Polyphasic OKJIP Chlorophyll a Fluorescence Transient in a Landrace and a Commercial Cultivar of Sweet Pepper (Capsicum annuum, L.) under Long-Term Salt Stress
Abstract
1. Introduction
2. Results and Discussion
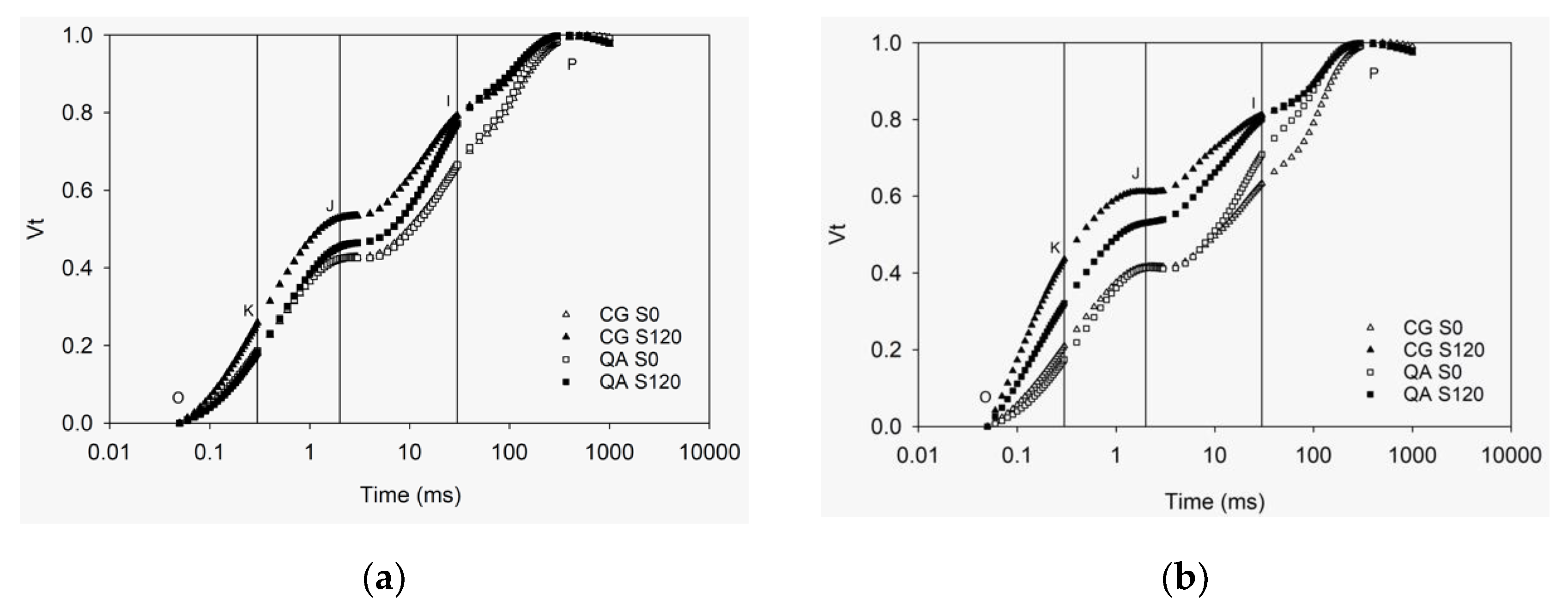
2.1. OKJIP Polyphasic Fluorescence Transients
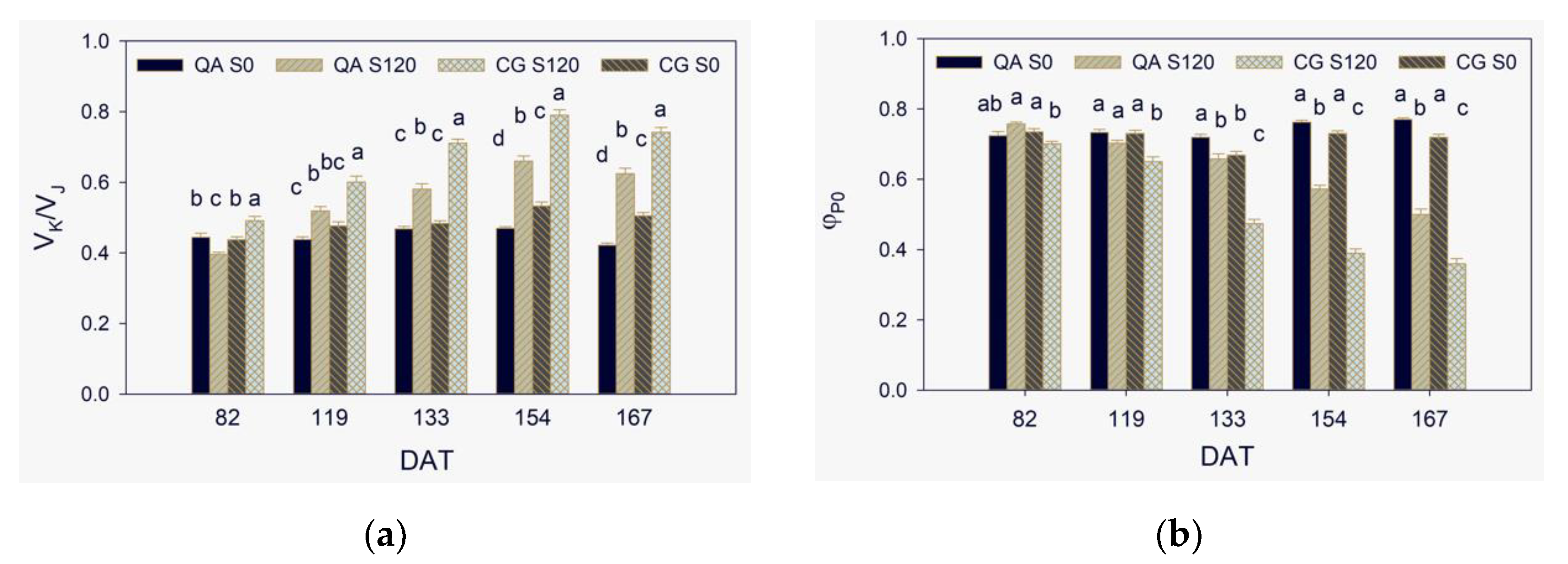
2.2. Donor Side of PSII
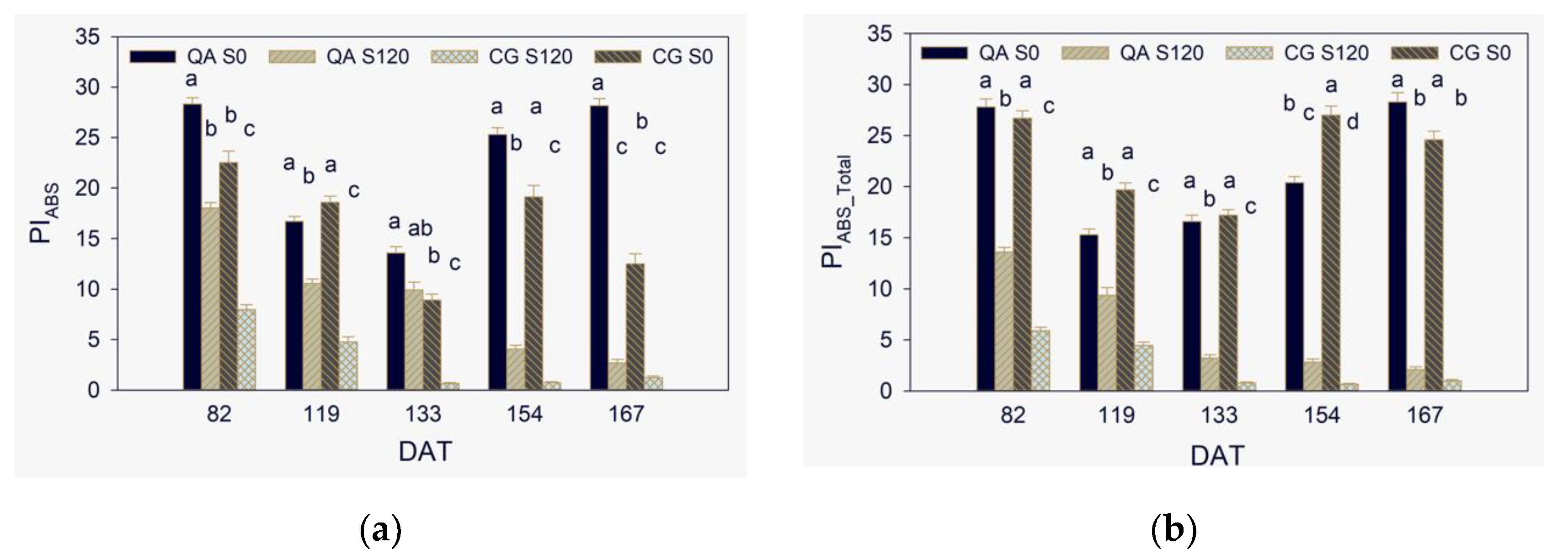
2.3. Maximum Quantum Yield, Efficiencies of Electron Transport, and Performance Indexes
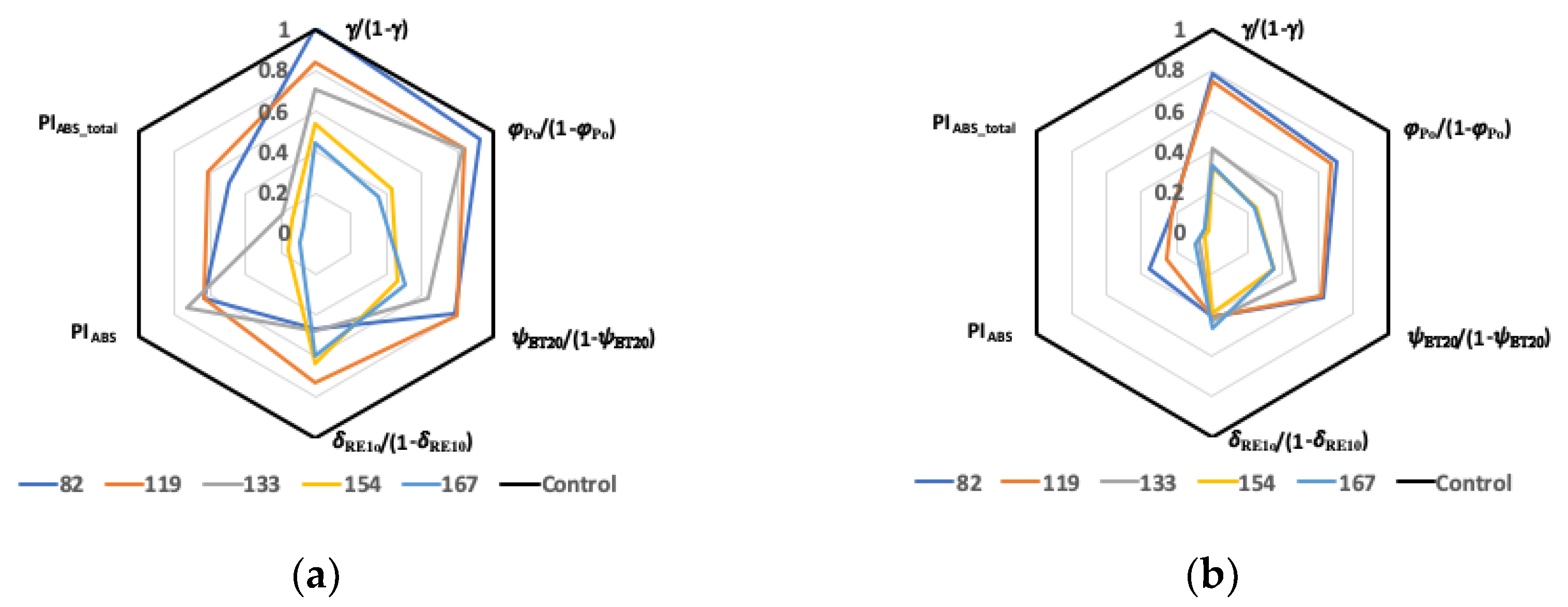
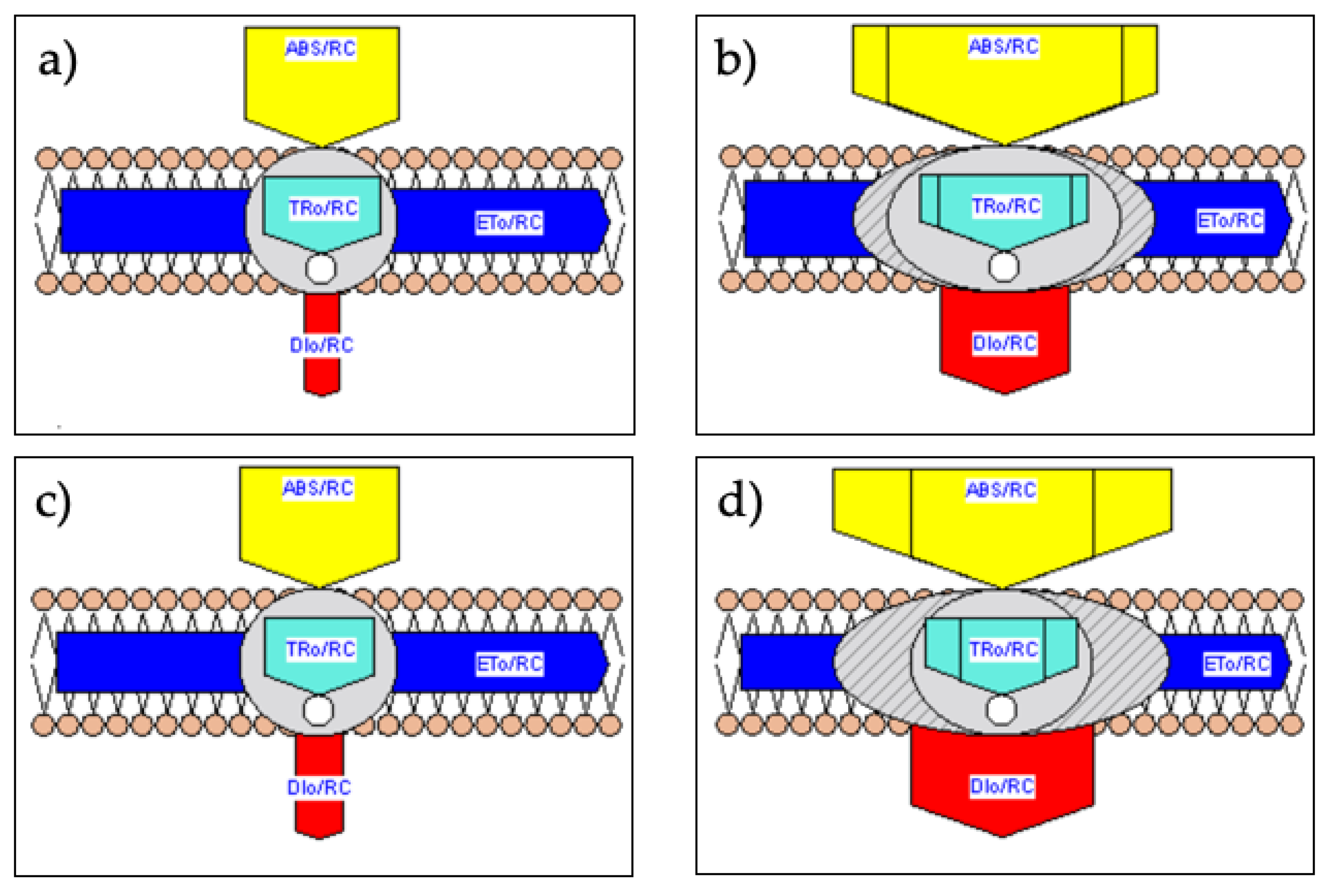
2.4. Membrane and Leaf Models of Energy Fluxes
2.5. Salt Stress Tolerance in Relation with Na+ and Cl− Leaf Content
3. Conclusions
4. Materials and Methods
4.1. Plant Material, Growth Conditions, and Salt Treatments
4.2. Fast Chlorophyll a Fluorescence Transient and JIP Test
4.3. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Duro, J.A.; Lauk, C.; Kastner, T.; Erb, K.-H.; Haberl, H. Global Inequalities in Food Consumption, Cropland Demand and Land-Use Efficiency: A Decomposition Analysis. Glob. Environ. Chang. 2020, 64, 102124. [Google Scholar] [CrossRef]
- Litalien, A.; Zeeb, B. Curing the Earth: A Review of Anthropogenic Soil Salinization and Plant-Based Strategies for Sustainable Mitigation. Sci. Total Environ. 2020, 698, 134235. [Google Scholar] [CrossRef]
- Zeven, A.C. Landraces: A Review of Definitions and Classifications. Euphytica 1998, 104, 127–139. [Google Scholar] [CrossRef]
- Fita, A.; Rodríguez-Burruezo, A.; Boscaiu, M.; Prohens, J.; Vicente, O. Breeding and Domesticating Crops Adapted to Drought and Salinity: A New Paradigm for Increasing Food Production. Front. Plant Sci. 2015, 6. [Google Scholar] [CrossRef]
- Razzaque, S.; Elias, S.M.; Haque, T.; Biswas, S.; Jewel, G.M.N.A.; Rahman, S.; Weng, X.; Ismail, A.M.; Walia, H.; Juenger, T.E.; et al. Gene Expression Analysis Associated with Salt Stress in a Reciprocally Crossed Rice Population. Sci. Rep. 2019, 9, 8249. [Google Scholar] [CrossRef]
- Munns, R.; Passioura, J.B.; Colmer, T.D.; Byrt, C.S. Osmotic Adjustment and Energy Limitations to Plant Growth in Saline Soil. New Phytol. 2020, 225, 1091–1096. [Google Scholar] [CrossRef]
- Chaves, M.M.; Flexas, J.; Pinheiro, C. Photosynthesis under Drought and Salt Stress: Regulation Mechanisms from Whole Plant to Cell. Ann. Bot. 2009, 103, 551–560. [Google Scholar] [CrossRef] [PubMed]
- Liang, W.; Ma, X.; Wan, P.; Liu, L. Plant Salt-Tolerance Mechanism: A Review. Biochem. Biophys. Res. Commun. 2018, 495, 286–291. [Google Scholar] [CrossRef]
- Van Zelm, E.; Zhang, Y.; Testerink, C. Salt Tolerance Mechanisms of Plants. Annu. Rev. Plant Biol. 2020, 71, 403–433. [Google Scholar] [CrossRef]
- Kalaji, H.M.; Jajoo, A.; Oukarroum, A.; Brestic, M.; Zivcak, M.; Samborska, I.A.; Cetner, M.D.; Łukasik, I.; Goltsev, V.; Ladle, R.J.; et al. The Use of Chlorophyll Fluorescence Kinetics Analysis to Study the Performance of Photosynthetic Machinery in Plants. In Emerging Technologies and Management of Crop Stress Tolerance; Elsevier: Amsterdam, The Netherlands, 2014; pp. 347–384. ISBN 978-0-12-800875-1. [Google Scholar]
- Stirbet, A. On the Relation between the Kautsky Effect (Chlorophyll a Fluorescence Induction) and Photosystem II: Basics and Applications of the OJIP Fluorescence Transient. J. Photochem. Photobiol. B 2011, 104, 236–257. [Google Scholar] [CrossRef]
- Strasser, B.; Strasser, R. Measuring Fast Fluorescence Transients to Address Environmental Questions: The JIP-Test. Photosynth. Light Biosph. 1995, 5, 977–980. [Google Scholar]
- Guissé, B.; Srivastava, A.; Strasser, R.J. The Polyphasic Rise of the Chlorophyll A Fluorescence (O-K-J-I-P) in Heat-Stressed Leaves. Arch. Sci. 1995, 48, 147–160. [Google Scholar] [CrossRef]
- Strasser, B.J. Donor Side Capacity of Photosystem II Probed by Chlorophyll a Fluorescence Transients. Photosynth. Res. 1997, 52, 147–155. [Google Scholar] [CrossRef]
- Strasser, R.J.; Tsimilli-Michael, M.; Srivastava, A. Analysis of the Chlorophyll a Fluorescence Transient. In Chlorophyll a Fluorescence; Papageorgiou, G.C., Govindjee, Eds.; Advances in Photosynthesis and Respiration; Springer: Dordrecht, The Netherlands, 2004; Volume 19, pp. 321–362. ISBN 978-1-4020-3217-2. [Google Scholar]
- Schansker, G.; Tóth, S.Z.; Strasser, R.J. Methylviologen and Dibromothymoquinone Treatments of Pea Leaves Reveal the Role of Photosystem I in the Chl a Fluorescence Rise OJIP. Biochim. Biophys. Acta BBA Bioenerg. 2005, 1706, 250–261. [Google Scholar] [CrossRef]
- Kalaji, H.M.; Rastogi, A.; Zivcak, M.; Brestic, M.; Daszkowska-Golec, A.; Sitko, K.; Alsharafa, K.Y.; Lotfi, R.; Stypinski, P.; Samborska, I.A.; et al. Prompt Chlorophyll Fluorescence as a Tool for Crop Phenotyping: An Example of Barley Landraces Exposed to Various Abiotic Stress Factors. Photosynthetica 2018, 56, 953–961. [Google Scholar] [CrossRef]
- Faseela, P.; Sinisha, A.; Brestič, M.; Puthur, J. Chlorophyll a Fluorescence Parameters as Indicators of a Particular Abiotic Stress in Rice. Photosynthetica 2019, 57, 108–115. [Google Scholar]
- Brestic, M.; Zivcak, M.; Kalaji, H.M.; Carpentier, R.; Allakhverdiev, S.I. Photosystem II Thermostability in Situ: Environmentally Induced Acclimation and Genotype-Specific Reactions in Triticum Aestivum L. Plant Physiol. Biochem. 2012, 57, 93–105. [Google Scholar] [CrossRef]
- Oukarroum, A.; Madidi, S.E.; Schansker, G.; Strasser, R.J. Probing the Responses of Barley Cultivars (Hordeum vulgare L.) by Chlorophyll a Fluorescence OLKJIP under Drought Stress and Re-Watering. Environ. Exp. Bot. 2007, 60, 438–446. [Google Scholar] [CrossRef]
- Živčák, M.; Brestič, M.; Olšovská, K.; Slamka, P. Performance Index as a Sensitive Indicator of Water Stress in Triticum aestivum L. Plant Soil Environ. 2008, 54, 133–139. [Google Scholar] [CrossRef]
- Zhao, J.; Lang, Y.; Zhang, S.; Zhao, Q.; Zhang, C.; Xia, J. Photosynthetic Characteristics and Chlorophyll a Fluorescence Transient in Lonicera Japonica under Drought Stress. Acta Physiol. Plant. 2019, 41, 124. [Google Scholar] [CrossRef]
- Bano, H.; Athar, H.; Zafar, Z.U.; Kalaji, H.M.; Ashraf, M. Linking Changes in Chlorophyll a Fluorescence with Drought Stress Susceptibility in Mung Bean [Vigna radiata (L.) Wilczek]. Physiol. Plant. 2021. [Google Scholar] [CrossRef] [PubMed]
- Kalaji, H.M.; Govindjee; Bosa, K.; Kościelniak, J.; Żuk-Gołaszewska, K. Effects of Salt Stress on Photosystem II Efficiency and CO2 Assimilation of Two Syrian Barley Landraces. Environ. Exp. Bot. 2011, 73, 64–72. [Google Scholar] [CrossRef]
- Rastogi, A.; Kovar, M.; He, X.; Zivcak, M.; Kataria, S.; Kalaji, H.M.; Skalicky, M.; Ibrahimova, U.F.; Hussain, S.; Mbarki, S.; et al. Special Issue in Honour of Prof. Reto J. Strasser—JIP-Test as a Tool to Identify Salinity Tolerance in Sweet Sorghum Genotypes. Photosynthetica 2020, 58, 518–528. [Google Scholar] [CrossRef]
- Kalaji, H.M.; Račková, L.; Paganová, V.; Swoczyna, T.; Rusinowski, S.; Sitko, K. Can Chlorophyll-a Fluorescence Parameters Be Used as Bio-Indicators to Distinguish between Drought and Salinity Stress in Tilia Cordata Mill? Environ. Exp. Bot. 2018, 152, 149–157. [Google Scholar] [CrossRef]
- Umar, M.; Uddin, Z.; Siddiqui, Z.S. Responses of Photosynthetic Apparatus in Sunflower Cultivars to Combined Drought and Salt Stress. Photosynthetica 2019, 57, 627–639. [Google Scholar] [CrossRef]
- Bussotti, F.; Desotgiu, R.; Cascio, C.; Pollastrini, M.; Gravano, E.; Gerosa, G.; Marzuoli, R.; Nali, C.; Lorenzini, G.; Salvatori, E.; et al. Ozone Stress in Woody Plants Assessed with Chlorophyll a Fluorescence. A Critical Reassessment of Existing Data. Environ. Exp. Bot. 2011, 73, 19–30. [Google Scholar] [CrossRef]
- Maliba, B.G.; Inbaraj, P.M.; Berner, J.M. The Use of OJIP Fluorescence Transients to Monitor the Effect of Elevated Ozone on Biomass of Canola Plants. Water. Air. Soil Pollut. 2019, 230, 75. [Google Scholar] [CrossRef]
- Kalaji, H.M.; Oukarroum, A.; Alexandrov, V.; Kouzmanova, M.; Brestic, M.; Zivcak, M.; Samborska, I.A.; Cetner, M.D.; Allakhverdiev, S.I.; Goltsev, V. Identification of Nutrient Deficiency in Maize and Tomato Plants by in Vivo Chlorophyll a Fluorescence Measurements. Plant Physiol. Biochem. 2014, 81, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Swoczyna, T.; Lata, B.; Stasiak, A.; Stefaniak, J.; Latocha, P. JIP-Test in Assessing Sensitivity to Nitrogen Deficiency in Two Cultivars of Actinidia Arguta (Siebold et Zucc.) Planch. Ex Miq. Photosynthetica 2019, 57, 646–658. [Google Scholar] [CrossRef]
- Bussotti, F.; Desotgiu, R.; Pollastrini, M.; Cascio, C. The JIP Test: A Tool to Screen the Capacity of Plant Adaptation to Climate Change. Scand. J. For. Res. 2010, 25, 43–50. [Google Scholar] [CrossRef]
- Fratianni, F.; d’Acierno, A.; Cozzolino, A.; Spigno, P.; Riccardi, R.; Raimo, F.; Pane, C.; Zaccardelli, M.; Tranchida Lombardo, V.; Tucci, M.; et al. Biochemical Characterization of Traditional Varieties of Sweet Pepper (Capsicum annuum L.) of the Campania Region, Southern Italy. Antioxidants 2020, 9, 556. [Google Scholar] [CrossRef]
- Rameshwaran, P.; Tepe, A.; Yazar, A.; Ragab, R. Effects of Drip-Irrigation Regimes with Saline Water on Pepper Productivity and Soil Salinity under Greenhouse Conditions. Sci. Hortic. 2016, 199, 114–123. [Google Scholar] [CrossRef]
- Kaya, C.; Ashraf, M.; Alyemeni, M.N.; Ahmad, P. The Role of Endogenous Nitric Oxide in Salicylic Acid-Induced up-Regulation of Ascorbate-Glutathione Cycle Involved in Salinity Tolerance of Pepper (Capsicum annuum L.) Plants. Plant Physiol. Biochem. 2020, 147, 10–20. [Google Scholar]
- Sonneveld, C.; Van der Burg, A. Sodium Chloride Salinity in Fruit Vegetable Crops in Soilless Culture. NJAS Wagening. J. Life Sci. 1991, 39, 115–122. [Google Scholar]
- Chartzoulakis, K.; Klapaki, G. Response of Two Greenhouse Pepper Hybrids to NaCl Salinity during Different Growth Stages. Sci. Hortic. 2000, 86, 247–260. [Google Scholar] [CrossRef]
- Aktas, H.; Abak, K.; Cakmak, I. Genotypic Variation in the Response of Pepper to Salinity. Sci. Hortic. 2006, 110, 260–266. [Google Scholar] [CrossRef]
- Ruggiero, A.; Landi, S.; Punzo, P.; Possenti, M.; Van Oosten, M.J.; Costa, A.; Morelli, G.; Maggio, A.; Grillo, S.; Batelli, G. Salinity and ABA Seed Responses in Pepper: Expression and Interaction of ABA Core Signaling Components. Front. Plant Sci. 2019, 10, 304. [Google Scholar] [CrossRef]
- Giorio, P.; Cirillo, V.; Caramante, M.; Oliva, M.; Guida, G.; Venezia, A.; Grillo, S.; Maggio, A.; Albrizio, R. Physiological Basis of Salt Stress Tolerance in a Landrace and a Commercial Variety of Sweet Pepper (Capsicum annuum L.). Plants 2020, 9, 795. [Google Scholar] [CrossRef] [PubMed]
- Demmig-Adams, B.; Adams, W.W. Photoprotection in an Ecological Context: The Remarkable Complexity of Thermal Energy Dissipation. New Phytol. 2006, 172, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Strasser, R.J. The Fo and the OJIP fluorescence rise in higher plants and algae. In Regulation of Chloroplast Biogenesis; Springer: Berlin/Heidelberg, Germany, 1992; pp. 423–426. [Google Scholar]
- Strasserf, R.J.; Srivastava, A. Govindjee Polyphasic Chlorophyll a Fluorescence Transient in Plants and Cyanobacteria. Photochem. Photobiol. 1995, 61, 32–42. [Google Scholar] [CrossRef]
- Duysens, L. Mechanism of the Two Photochemical Reactions in Algae as Studied by Means of Fluorescence. In Studies on Microalgae and Photosynthetic Bacteria; Special Issue of Plant and Cell Physiology; Japanese Society of Plant Physiologists, University of Tokyo: Tokyo, Japan, 1963; pp. 353–372. [Google Scholar]
- Goltsev, V.N.; Kalaji, H.M.; Paunov, M.; Bąba, W.; Horaczek, T.; Mojski, J.; Kociel, H.; Allakhverdiev, S.I. Variable Chlorophyll Fluorescence and Its Use for Assessing Physiological Condition of Plant Photosynthetic Apparatus. Russ. J. Plant Physiol. 2016, 63, 869–893. [Google Scholar] [CrossRef]
- Oukarroum, A.; Schansker, G.; Strasser, R.J. Drought Stress Effects on Photosystem I Content and Photosystem II Thermotolerance Analyzed Using Chl a Fluorescence Kinetics in Barley Varieties Differing in Their Drought Tolerance. Physiol. Plant. 2009, 137, 188–199. [Google Scholar] [CrossRef]
- Srivastava, A.; Guisse, B.; Greppin, H.; Strasser, R.J. Regulation of Antenna Structure and Electron Transport in Photosystem II of Pisum Sativum under Elevated Temperature Probed by the Fast Polyphasic Chlorophyll a Fluorescence Transient: OKJIP. Biochim. Biophys. Acta (BBA) Bioenerg. 1997, 1320, 95–106. [Google Scholar] [CrossRef]
- Mehta, P.; Jajoo, A.; Mathur, S.; Bharti, S. Chlorophyll a Fluorescence Study Revealing Effects of High Salt Stress on Photosystem II in Wheat Leaves. Plant Physiol. Biochem. 2010, 48, 16–20. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.W.; Xu, C.; Lv, C.F.; Wu, M.; Cai, X.J.; Liu, Z.T.; Song, X.M.; Chen, G.X.; Lv, C.G. Chlorophyll a Fluorescence Analysis of High-Yield Rice (Oryza sativa L.) LYPJ during Leaf Senescence. Photosynthetica 2016, 54, 422–429. [Google Scholar] [CrossRef]
- Henmi, T.; Miyao, M.; Yamamoto, Y. Release and Reactive-Oxygen-Mediated Damage of the Oxygen-Evolving Complex Subunits of PSII during Photoinhibition. Plant Cell Physiol. 2004, 45, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Tahjib-UI-Arif, M.; Sohag, A.A.M.; Afrin, S.; Bashar, K.K.; Afrin, T.; Mahamud, A.; Polash, M.A.S.; Hossain, M.; Sohel, M.; Taher, A. Differential Response of Sugar Beet to Long-Term Mild to Severe Salinity in a Soil–Pot Culture. Agriculture 2019, 9, 223. [Google Scholar] [CrossRef]
- Björkman, O.; Demmig, B. Photon Yield of O2 Evolution and Chlorophyll Fluorescence Characteristics at 77 K among Vascular Plants of Diverse Origins. Planta 1987, 170, 489–504. [Google Scholar] [CrossRef] [PubMed]
- Malnoë, A. Photoinhibition or Photoprotection of Photosynthesis? Update on the (Newly Termed) Sustained Quenching Component QH. Environ. Exp. Bot. 2018, 154, 123–133. [Google Scholar] [CrossRef]
- Demmig-Adams, B.; Cohu, C.M.; Muller, O.; Adams, W.W. Modulation of Photosynthetic Energy Conversion Efficiency in Nature: From Seconds to Seasons. Photosynth. Res. 2012, 113, 75–88. [Google Scholar] [CrossRef]
- Maxwell, K.; Johnson, G.N. Chlorophyll Fluorescence—A Practical Guide. J. Exp. Bot. 2000, 51, 659–668. [Google Scholar] [CrossRef] [PubMed]
- Digrado, A.; Bachy, A.; Mozaffar, A.; Schoon, N.; Bussotti, F.; Amelynck, C.; Dalcq, A.; Fauconnier, M.; Aubinet, M.; Heinesch, B. Long-term Measurements of Chlorophyll a Fluorescence Using the JIP-test Show That Combined Abiotic Stresses Influence the Photosynthetic Performance of the Perennial Ryegrass (Lolium perenne) in a Managed Temperate Grassland. Physiol. Plant. 2017, 161, 355–371. [Google Scholar] [CrossRef]
- Strasser, R.J.; Srivastava, A.; Tsimilli-Michael, M. The Fluorescence Transient as a Tool to Characterize and Screen Photosynthetic Samples. In Probing Photosynthesis: Mechanisms, Regulation and Adaptation; Yunus, M., Pathre, U., Mohanty, P., Eds.; Taylor and Francis: London, UK, 2000; pp. 445–483. [Google Scholar]
- Tsimilli-Michael, M.; Strasser, R.J. In vivo assessment of stress impact on plant’s vitality: Applications in detecting and evaluating the beneficial role of mycorrhization on host plants. In Mycorrhiza; Springer: Berlin/Heidelberg, Germany, 2008; pp. 679–703. [Google Scholar]
- Wang, Z.X.; Chen, L.; Ai, J.; Qin, H.Y.; Liu, Y.X.; Xu, P.L.; Jiao, Z.Q.; Zhao, Y.; Zhang, Q.T. Photosynthesis and Activity of Photosystem II in Response to Drought Stress in Amur Grape (Vitis Amurensis Rupr.). Photosynthetica 2012, 50, 189–196. [Google Scholar] [CrossRef]
- Joliot, P.; Joliot, A. Cyclic Electron Flow in C3 Plants. Biochim. Biophys. Acta BBA Bioenerg. 2006, 1757, 362–368. [Google Scholar] [CrossRef] [PubMed]
- Redondo-Gómez, S.; Wharmby, C.; Castillo, J.M.; Mateos-Naranjo, E.; Luque, C.J.; de Cires, A.; Luque, T.; Davy, A.J.; Enrique Figueroa, M. Growth and Photosynthetic Responses to Salinity in an Extreme Halophyte, Sarcocornia Fruticosa. Physiol. Plant. 2006, 128, 116–124. [Google Scholar] [CrossRef]
- Zivcak, M.; Brestic, M.; Balatova, Z.; Drevenakova, P.; Olsovska, K.; Kalaji, H.M.; Yang, X.; Allakhverdiev, S.I. Photosynthetic Electron Transport and Specific Photoprotective Responses in Wheat Leaves under Drought Stress. Photosynth. Res. 2013, 117, 529–546. [Google Scholar] [CrossRef]
- South, P.F.; Cavanagh, A.P.; Lopez-Calcagno, P.E.; Raines, C.A.; Ort, D.R. Optimizing Photorespiration for Improved Crop Productivity. J. Integr. Plant Biol. 2018, 60, 1217–1230. [Google Scholar] [CrossRef]
- Ziotti, A.B.; Silva, B.P.; Neto, M.C.L. Photorespiration Is Crucial for Salinity Acclimation in Castor Bean. Environ. Exp. Bot. 2019, 167, 103845. [Google Scholar] [CrossRef]
- Hendrickson, L.; Förster, B.; Furbank, R.T.; Chow, W.S. Processes Contributing to Photoprotection of Grapevine Leaves Illuminated at Low Temperature. Physiol. Plant. 2004, 121, 272–281. [Google Scholar] [CrossRef]
- Brestic, M.; Zivcak, M. PSII fluorescence techniques for measurement of drought and high temperature stress signal in crop plants: Protocols and applications. In Molecular Stress Physiology of Plants; Springer: Berlin/Heidelberg, Germany, 2013; pp. 87–131. [Google Scholar]
- Jajoo, A. Changes in photosystem II in response to salt stress. In Ecophysiology and Responses of Plants under Salt Stress; Springer: Berlin/Heidelberg, Germany, 2013; pp. 149–168. [Google Scholar]
- Geilfus, C.-M. Chloride: From Nutrient to Toxicant. Plant Cell Physiol. 2018, 59, 877–886. [Google Scholar] [CrossRef]
- Rajendran, K.; Tester, M.; Roy, S.J. Quantifying the Three Main Components of Salinity Tolerance in Cereals. Plant Cell Environ. 2009, 32, 237–249. [Google Scholar] [CrossRef]
- Isayenkov, S.V.; Maathuis, F.J.M. Plant Salinity Stress: Many Unanswered Questions Remain. Front. Plant Sci. 2019, 10, 80. [Google Scholar] [CrossRef] [PubMed]
- Tavakkoli, E.; Rengasamy, P.; McDonald, G.K. High Concentrations of Na+ and Cl– Ions in Soil Solution Have Simultaneous Detrimental Effects on Growth of Faba Bean under Salinity Stress. J. Exp. Bot. 2010, 61, 4449–4459. [Google Scholar] [CrossRef]
- Carillo, P.; Woodrow, P.; Raimondi, G.; El-Nakhel, C.; Pannico, A.; Kyriacou, M.C.; Colla, G.; Mori, M.; Giordano, M.; De Pascale, S.; et al. Omeprazole Promotes Chloride Exclusion and Induces Salt Tolerance in Greenhouse Basil. Agronomy 2019, 9, 355. [Google Scholar] [CrossRef]
- Zuo, Z.; Ye, F.; Wang, Z.; Li, S.; Li, H.; Guo, J.; Mao, H.; Zhu, X.; Li, X. Salt Acclimation Induced Salt Tolerance in Wild-Type and Chlorophyl b-Deficient Mutant Wheat. Plant Soil Environ. 2021, 67, 26–32. [Google Scholar] [CrossRef]
- Hoagland, D.R.; Arnon, D.I. The Water-Culture Method for Growing Plants without Soil. Circ. Calif. Agric. Exp. Stn. 1950, 347, 32. [Google Scholar]
- Addinsoft, A. XLSTAT Statistical and Data Analysis Solution; Long Island, NY, USA, 2019. Available online: https://www.xlstat.com/en/news/2020 (accessed on 5 November 2020).

| Raw data obtained from fluorescence induction curve | |
| Ft | Fluorescence intensity at any time (t) since start of actinic illumination |
| Fo = F20μs or 50μs | Fluorescence intensity when all PSII RCs are open. The used measure depends on instrumental reliability |
| F300μs | Fluorescence intensity at the K step (300 μs) of the induction curve |
| FJ ≡ F2ms | Fluorescence intensity at the J step (2 ms) of the induction curve |
| FI ≡ F30ms | Fluorescence intensity at the I step (30 ms) of the induction curve |
| FP ≡ FM | Fluorescence intensity when all PSII RCs are closed due to saturating illumination |
| Technical fluorescence parameters calculated from the raw data | |
| Vv = Ft − Fo | Variable Chl fluorescence |
| FV = FM − Fo | Maximum variable Chl fluorescence |
| Vt = (Ft − Fo)/(FM − Fo) | Relative variable Chl fluorescence |
| Mo ≈ (∆V/∆t)o = 4 ms−1 (F300μs − F50μs)/(FM − F50μs) | Approximate value of the initial slope of Vt curve |
| Energy fluxes | |
| ABS = TR + DI | Rate of light energy (photons) absorbed by PSII antenna |
| TR | Rate of excitation energy (excitons) trapped by the PSII RCs (causing reduction of the primary PSII acceptor, QA) |
| TRo | Maximum TR (initial, at time t = 0) |
| DI | Rate of energy dissipation in the PSIIs, in processes other than trapping |
| ET2o | Electron transport flux from primary (QA) to secondary (QB) PSII acceptor |
| RE1o | Electron transport flux from QB until PSI acceptors (initial, at time t = 0) |
| Quantum yields and efficiencies/probabilities | |
| φPo ≡ TRo / ABS = 1 − Fo/FM | Maximum quantum yield of primary PSII photochemistry (initial, at time t = 0) |
| φET2o ≡ ET2o / ABS = 1 − FJ/FM = φPo(1 − VJ) | Quantum yield of the electron transport flux from QA to QB (initial, at time t = 0) |
| φRE1o ≡ RE1o / ABS = 1 − FI/FM = φPo(1 − VI) | Quantum yield of the electron transport flux until the PSI electron acceptors (initial, at time t = 0) |
| ψET2o ≡ ET2o / TRo = 1 − VJ | Efficiency/probability with which a PSII trapped electron is transferred from QA to QB (initial, at time t = 0) |
| ψRE1o ≡ RE1o / TRo = 1 – VI | Efficiency/probability with which a PSII trapped electron is transferred until PSI acceptors (initial, at time t = 0) |
| δRE1o ≡ RE1o / ET2o = (1 – VI)/(1 – VJ) | Efficiency/probability with which an electron from QB is transferred until PSI acceptors (initial, at time t = 0) |
| Specific energy fluxes (per active PSII reaction center) | |
| ABS /RC = (Mo/VJ) (1/φPo) | Light energy (photons) absorption flux per PSII reaction center (i.e., apparent antenna size of an active PSII) |
| γRC2 ≡ ChlRC/ChlTotal | Probability that a PSII Chl functions as RC |
| RC/ABS = φPoVJ/Mo = γRC2/(1 −γRC2) | Number of QA-reducing RCs (i.e., active) per PSII antenna Chl |
| TRo/RC = Mo/VJ | Maximum trapped exciton flux per PSII (initial, at time t = 0) |
| ET2o /RC = (Mo/VJ) (1 − VJ) | Electron transport flux from QA to QB per PSII RC (initial, at time t = 0) |
| RE1o /RC = (Mo/VJ) (1 − VI) | Electron transport flux until PSI acceptors per PSII RC (initial, at time t = 0) |
| Phenomenological energy fluxes/activities (per excited cross section CS) | |
| ABS /CSo = Fo or ABS /CSM = FM | Light energy (photons) absorption flux per cross section (or also, apparent PSII antenna size) |
| RC/CS = (RC/ABS) (ABS /CS) | The number of active PSII RCs per cross section |
| TRo/CS = (TRo/ABS) (ABS /CS) | Maximum trapped exciton flux per cross section |
| ET2o/CS = (ET2o / ABS) (ABS /CS) | Electron transport flux from QA to QB per cross section |
| RE1o/CS = (RE1o / ABS) (ABS /CS) | Electron transport flux until PSI acceptors per cross section |
| Performance indexes on absorption basis | |
| PIABS = [γRC2/(1 −γRC2)] [φPo/ (1 − φPo)] [ψET2o /(1 −ψET2o)] | Performance index for energy conservation from photons absorbed by PSII antenna, to the reduction of QB |
| PIABS_Total = PIABS [δRE1o /(1 −δRE1o)] | Performance index for energy conservation from photons absorbed by PSII antenna, until the reduction of PSI acceptors |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giorio, P.; Sellami, M.H. Polyphasic OKJIP Chlorophyll a Fluorescence Transient in a Landrace and a Commercial Cultivar of Sweet Pepper (Capsicum annuum, L.) under Long-Term Salt Stress. Plants 2021, 10, 887. https://doi.org/10.3390/plants10050887
Giorio P, Sellami MH. Polyphasic OKJIP Chlorophyll a Fluorescence Transient in a Landrace and a Commercial Cultivar of Sweet Pepper (Capsicum annuum, L.) under Long-Term Salt Stress. Plants. 2021; 10(5):887. https://doi.org/10.3390/plants10050887
Chicago/Turabian StyleGiorio, Pasquale, and Mohamed Houssemeddine Sellami. 2021. "Polyphasic OKJIP Chlorophyll a Fluorescence Transient in a Landrace and a Commercial Cultivar of Sweet Pepper (Capsicum annuum, L.) under Long-Term Salt Stress" Plants 10, no. 5: 887. https://doi.org/10.3390/plants10050887
APA StyleGiorio, P., & Sellami, M. H. (2021). Polyphasic OKJIP Chlorophyll a Fluorescence Transient in a Landrace and a Commercial Cultivar of Sweet Pepper (Capsicum annuum, L.) under Long-Term Salt Stress. Plants, 10(5), 887. https://doi.org/10.3390/plants10050887







