Pomological Characteristics and Ploidy Levels of Japanese Plum (Prunus salicina Lindl.) Cultivars Preserved in Poland
Abstract
1. Introduction
2. Results and Discussion
2.1. Cold Hardiness
2.2. Susceptibility to Plum pox Virus (Sharka)
2.3. Tree Vigour
2.4. Flowering
2.5. Yield and Ripening Time of Fruit
2.6. Determination of Fruit Quality
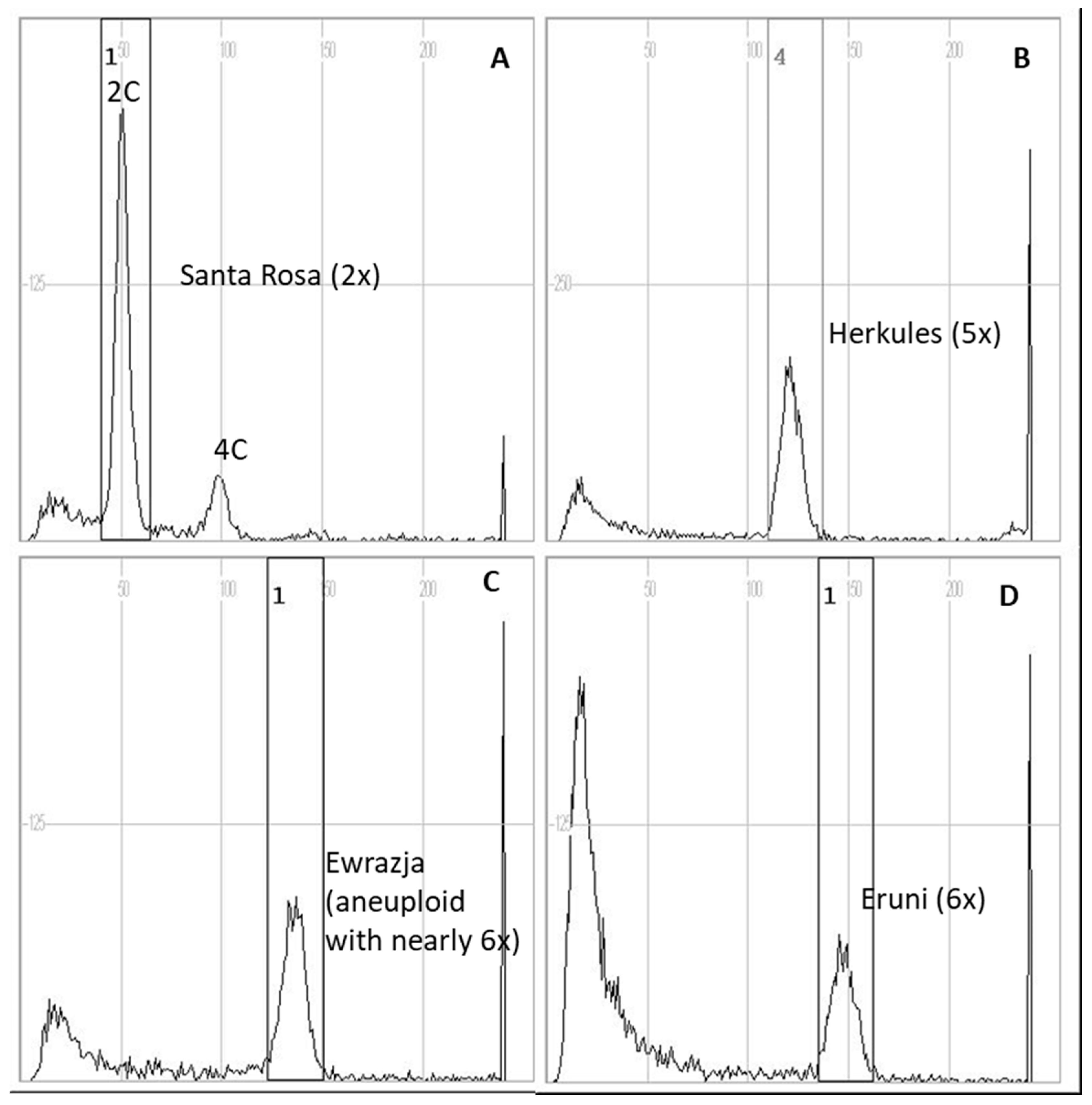
2.7. Ploidy Level
3. Materials and Methods
3.1. Location and Plant Material
3.2. Weather Conditions
3.3. Tree Growth and Productivity Assessment
- Frost damage to trees—assessed on a 1–9-point rating scale according to Perczak [33].
- Plum pox virus (sharka) symptoms—on the basis of inspection performed at the end of June and in the middle of September. The state of trees was determined visually on a 0–3 point rating scale for symptoms on the leaves. The symptoms of the fruit were investigated, when the fruit consumption maturity was reached on a 0–3 point rating scale according to Iliev and Stoev [34].
- Tree vigor and canopy shape assessed on a 1–9 point rating scale.
- Flowering time—was recorded by recommendations of the international working group for pollination: start of flowering–10% open flowers; full bloom–80% of the flower buds on the tree had reached the open flower stage; and, end of flowering–90% of the petal fall [35].
3.4. Determination of Fruit Quality
3.5. Ploidy Level
3.6. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Blazek, J. A survey of the Genetic Resources used in Plum Breeding. Acta Hortic. 2007, 734, 31–45. [Google Scholar] [CrossRef]
- Topp, B.L.; Russell, D.M.; Neumüller, M.; Dalbo, M.A.; Liu, W. Plum. In Fruit Breeding; Chapter 15; Badenes, M.L., Byrne, D.H., Eds.; Springer: Berlin/Heidelberg, Germany, 2012; pp. 571–621. [Google Scholar]
- Okie, W.R.; Hancock, J.F. Plums. In Temperate Fruit Crop Breeding: Germplasm to Genomics; Hancock, J.F., Ed.; Springer: New York, NY, USA, 2008; pp. 337–357. [Google Scholar]
- Das, B.; Ahmed, N.; Singh, P. Prunus Diversity-Early and Present Development: A Review. Int. J. Biodivers. Conserv. 2011, 3, 721–734. [Google Scholar] [CrossRef]
- Ben Tamarzizt, H.; Walker, D.; Ben Mustapha, S.; Abdallah, D.; Baraket, G.; Salhi Hannachi, A.; Zehdi Azzouzi, S. DNA Variation and Polymorphism in Tunisian Plum Species (Prunus spp.): Contribution of Flow Cytometry and Molecular Markers. Genet. Mol. Res. 2015, 14, 18034–18046. [Google Scholar] [CrossRef]
- Prunus Crop Germplasm Committee. Prunus Vulnerability Statement. 2017; pp. 1–13. Available online: https://www.ars-grin.gov/npgs/cgc_reports/prunusvuln2017.pdf (accessed on 26 April 2021).
- Podwyszyńska, M.; Sitarek, M.; Marasek-Ciołakowska, A.; Kowalska, U. Nuclear DNA Content and Phenotypic Traits of the Prunus Rootstocks from Poland’s Gene Resources. Zemdirb. Agric. 2020, 107, 71–78. [Google Scholar] [CrossRef]
- Grzyb, Z.S.; Rozpara, E. Intensywny Sad Śliwowy; Hortpress: Warszawa, Poland, 2007; pp. 5–214. [Google Scholar]
- Grzyb, Z.S.; Rozpara, E. Klasyfikacja Niektórych Odmian Śliwy Pod Względem Wielkości Owoców i Terminu Dojrzewania. Zesz. Probl. Postępów Nauk Rol. 2007, 517, 331–337. [Google Scholar]
- Hartmann, W.; Neumüller, M. Plum Breeding Plantation Tree Crops: Temperate Species; Jain, S.M., Priyadaeshan, P.M., Eds.; Springer: Berlin/Heidelberg, Germany, 2009; pp. 161–231. [Google Scholar]
- Grzyb, Z.S.; Rozpara, E. Plum Production in Poland. Acta Hortic. 1998, 478, 19–24. [Google Scholar] [CrossRef]
- Malinowski, T.; Rozpara, E.; Grzyb, Z.S. Evaluation of the Susceptibility of Several Plum (Prunus domestica L.) Cultivars to Plum Pox Virus (PPV) Infection in the Field. Sharka-Like Symptoms Observed on ‘Jojo’ Fruit are not Related to PPV. J. Hortic. Res. 2013, 21, 61–65. [Google Scholar] [CrossRef]
- Milatović, D.; Durović, D.; Dorđević, B. Evaluation of Japanese Plum Cultivars in Serbia. Acta Hortic. 2013, 981, 173–176. [Google Scholar] [CrossRef]
- Milatović, D.; Durović, D.; Zec, G.; Radović, A. Evaluation of Some Diploid Plum Cultivars in the Region of Belgrade. Acta Hortic. 2019, 1260, 153–158. [Google Scholar] [CrossRef]
- Butac, M.; Militaru, E.; Chitu, E.; Plopa, C.; Sumedrea, M.; Sumedrea, D. Differences and Similarities between some European and Japanese Plum Cultivars. Acta Hortic. 2019, 1260, 129–136. [Google Scholar] [CrossRef]
- Isaczkin, A.W.; Worobiew, B.N. Sortowoj Katalog Płodowych Kultur Rossii. Śliwa. AST Actrel. Moskwa 2003, 427, 293–369. [Google Scholar]
- Pawliuk, W.W. Pomologia. Śliwa, Wiśnia, Czereśnia. Kijew 2004, 4, 7–104. [Google Scholar]
- Brooks, R.M.; Olmo, H.P. Register of New Fruit and Nut Varieties 1920–1950; University of California Press: Berkely, CA, USA; Cambridge University Press: Cambridge, UK, 1952; pp. 7–50. [Google Scholar]
- Szabo, Z.; Nyeki, J. Japanese Plums in Hungary. Acta Hortic. 2002, 577, 65–72. [Google Scholar] [CrossRef]
- Pirkhezri, M.; Fatahi Mogagam, M.R.; Edabi, E.; Hassani, D.; Abdoosi, V. Morphological Study of Some New Japanese Plum (Prunus Salicina Lindl.) Cultivars Grown in Iran. Int. J. Biosci. 2014, 5, 180–187. [Google Scholar]
- Karklins, J.; Skrivele, M.; Kaufmane, E.; Ikase, L. Plumju Skirnes. Latvijas Pomologija; Mintis: Ithaca, NY, USA, 2007; pp. 3–204. [Google Scholar]
- Vangdal, E.; Flatland, S.; Nordbo, R. Fruit Quality Changes during Marketing of New Plum Cultivars (Prunus Domestica L.). Hortic. Sci. 2007, 34, 91–95. [Google Scholar] [CrossRef]
- Kemp, H.; Wustenberghs, H. Screening of Plums and Prunes for Fresh Consumption, 1988–1997. Acta Hortic. 1998, 478, 207–216. [Google Scholar] [CrossRef]
- Mayfield, D.; Chen, Z.J.; Pires, J.C. Epigenetic Regulation of Flowering Time in Polyploids. Curr. Opin. Plant Biol. 2011, 14, 174–178. [Google Scholar] [CrossRef]
- Xue, H.; Zhang, B.; Tian, J.R.; Chen, M.M.; Zhang, Y.Y.; Zhang, Z.H.; Ma, Y. Comparison of the Morphology, Growth and Development of Diploid and Autotetraploid ‘Hanfu’ Apple Trees. Sci. Hortic. 2017, 225, 277–285. [Google Scholar] [CrossRef]
- Gantait, S.; Mandal, N.; Bhattacharyya, S.; Das, P.K. Induction and Identification of Tetraploids Using in vitro Colchicine Treatment of Gerbera jamesonii Bolus Cv. Sciella. Plant Cell Tissue Organ Cult. 2011, 106, 485–493. [Google Scholar] [CrossRef]
- Liu, S.; Chen, S.; Chen, Y.; Guan, Z.; Yin, D.; Chen, F. In vitro Induced Tetraploid of Dendranthema Nankingense (Nakai) Tzvel. Shows an Improved Level of Abiotic Stress Tolerance. Sci. Hortic. 2011, 127, 411–419. [Google Scholar] [CrossRef]
- Ramsey, J.; Schemske, D.W. Pathways, Mechanisms, and Rates of Polyploid Formation in Flowering Plants. Annu. Rev. Ecol. Syst. 1998, 29, 467–501. [Google Scholar] [CrossRef]
- Wang, Y.; Du, H.M.; Zhang, J.; Chen, T.; Chen, Q.; Tang, H.R.; Wang, X.R. Ploidy Level of Chinese Cherry (Cerasus Pseudocerasus Lindl.) and Comparative Study on Karyotypes with Four Cerasus Species. Sci. Hortic. 2018, 232, 46–51. [Google Scholar] [CrossRef]
- Neumüller, M. Fundamental and Applied Aspects of Plum (Prunus domestica) Breeding. Fruit Veg. Cereal Sci. Biotechnol. 2011, 139–156. Available online: www.globalsciencebooks.info (accessed on 26 April 2021).
- Guerra, M.E.; Rodrigo, J. Japanese Plum Pollination: A Review. Sci. Hortic. 2017, 197, 674–686. [Google Scholar] [CrossRef]
- Hjalmarsson, I. Plum Cultivars in Sweden:m History and Conservation for Future Use. Proc. Latv. Acad. Sci. Sect. B 2019, 73, 207–213. [Google Scholar] [CrossRef]
- Perczak, J. Metodyka Badania Wartości Gospodarczej Odmian (WGO), Odrębności, Wyrównania I Trwałości (OWT) Roślin Uprawnych. Śliwa COBORU Słupia Wielka 2006, I, 1–33. [Google Scholar]
- Iliev, P.; Stoev, A. Reaction of Some Plum Cultivars to Sharka Virus (Plum pox Virus, PPP-M Strain). Acta Hortic. 2002, 577, 73–77. [Google Scholar] [CrossRef]
- Wertheim, S.J. Methods for Cross Pollination and Flowering Assessment and their Interpretation. Acta Hortic. 1996, 423, 237–242. [Google Scholar] [CrossRef]
| Cultivar. | Frost Damage to the Trees * | Dead Trees | Symptoms of Plum pox Virus | |
|---|---|---|---|---|
| On the Leaves ** | On the Fruits *** | |||
| Angeleno | 9 | 0 | 0 | 0 |
| Barkhatnaya | 9 | 3 | 3 | 3 |
| Black Amber | 7 | 1 | 0 | 0 |
| Black Diamond | 9 | 1 | 0 | 0 |
| Blue Gigant | 7 | 0 | 0 | 0 |
| Chuk | 7 | 0 | 0 | 0 |
| Desertnaya Rannaya | 9 | 1 | 0 | 0 |
| Dofi Sandra | 7 | 1 | 3 | 0 |
| Early Golden | 7 | 2 | 2 | 2 |
| Ewierch Rannyj | 9 | 1 | 0 | 0 |
| Friar | 7 | 1 | 0 | 0 |
| Formosa | 7 | 0 | 0 | 0 |
| Gauota | 7 | 1 | 0 | 0 |
| Gek | 9 | 0 | 0 | 0 |
| General | 9 | 0 | 0 | 0 |
| Herkules | 9 | 1 | 1 | 1 |
| Inese | 9 | 0 | 0 | 0 |
| Kometa | 9 | 2 | 0 | 0 |
| Kometa Late | 9 | 0 | 0 | 0 |
| Maschenka | 9 | 1 | 0 | 0 |
| Naidyona | 9 | 0 | 0 | 0 |
| Obilnaya | 7 | 0 | 0 | 0 |
| Oishi Wase | 9 | 1 | 0 | 0 |
| Ozark Premier | 9 | 1 | 0 | 0 |
| Puteshestvennitsa | 9 | 0 | 0 | 0 |
| Santa Rosa | 9 | 0 | 0 | 0 |
| Shater | 7 | 0 | 0 | 0 |
| Shiro | 9 | 0 | 0 | 0 |
| Skoroplodnaya | 9 | 1 | 1 | 0 |
| Slivovidnaya | 9 | 1 | 0 | 0 |
| Superior | 7 | 2 | 0 | 0 |
| Tatyana | 9 | 3 | 3 | 3 |
| Tsernushka | 9 | 0 | 0 | 0 |
| Tsernushka Rannaya | 9 | 0 | 0 | 0 |
| Vanier | 9 | 1 | 0 | 0 |
| Yevraziya | 9 | 0 | 0 | 0 |
| Cultivar | Growth Vigour * | Crown Habit ** | Date of Flowering | Duration of Flowering (days) | Beginning of Fruiting (Year after Planting) | Productivity *** | Time of Harvest | ||
|---|---|---|---|---|---|---|---|---|---|
| Start | Full | End | |||||||
| Angeleno | 7 | 7 | 12.04 | 14.04 | 22.04 | 8.5 | 2 | 4.2 | b. X |
| Barkhatnaya | 3 | 7 | 05.04 | 07.04 | 15.04 | 9.3 | 1 | 9 | b. VIII |
| Black Amber | 5 | 3 | 12.04 | 14.04 | 23.04 | 11.7 | 1 | 4.6 | b. IX |
| Black Diamond | 5 | 7 | 11.04 | 13.04 | 20.04 | 12.3 | 2 | 3.8 | II dec. VIII |
| Blue Gigant | 3 | 3 | 12.04 | 13.04 | 23.04 | 11.0 | 3 | 3.5 | II dec. VIII |
| Chuk | 5 | 7 | 04.04 | 06.04 | 12.04 | 10.0 | 2 | 8.5 | e. VII |
| Desertnaya Rannaya | 7 | 7 | 06.04 | 08.04 | 19.04 | 10.7 | 1 | 5 | e. VII |
| Dofi Sandra | 7 | 5 | 06.04 | 08.04 | 19.04 | 10.0 | 2 | 8 | e. VII |
| Early Golden | 5 | 7 | 11.04 | 13.04 | 23.04 | 10.8 | 1 | 8 | b. VIII |
| Ewierch Rannyj | 7 | 7 | 10.04 | 12.04 | 19.04 | 9.0 | 2 | 7 | b. VII |
| Friar | 5 | 7 | 12.04 | 14.04 | 23.04 | 11.7 | 2 | 4 | e. IX |
| Formosa | 7 | 7 | 08.04 | 11.04 | 19.04 | 9.0 | 2 | 2.3 | b. VIII |
| Gauota | 5 | 3 | 11.04 | 13.04 | 21.04 | 10.0 | 2 | 5 | b. VII |
| Gek | 5 | 7 | 13.04 | 15.04 | 25.04 | 9.7 | 1 | 7.5 | e. VII |
| General | 7 | 7 | 06.04 | 09.04 | 21.04 | 14.3 | 1 | 7 | e. VII |
| Herkules | 9 | 3 | 12.04 | 14.04 | 23.04 | 8.7 | 1 | 3.3 | II dec. VIII |
| Inese | 7 | 7 | 11.04 | 13.04 | 19.04 | 9.3 | 1 | 9 | b. VIII |
| Kometa | 5 | 7 | 11.04 | 13.04 | 21.04 | 10.3 | 1 | 7.5 | e. VII |
| Kometa Late | 5 | 7 | 10.04 | 12.04 | 24.04 | 13.0 | 1 | 7.5 | b. VIII |
| Maschenka | 7 | 7 | 10.04 | 12.04 | 22.04 | 11.,3 | 1 | 7 | II dec. VII |
| Naidyona | 5 | 5 | 10.04 | 12.04 | 19.04 | 8.0 | 2 | 7 | e. VII |
| Obilnaya | 5 | 7 | 08.04 | 10.04 | 21.04 | 10.7 | 1 | 5 | e. VII |
| Oishi Wase | 5 | 7 | 08.04 | 10.04 | 16.04 | 10.0 | 1 | 4.3 | b. VII |
| Ozark Premier | 5 | 7 | 08.04 | 10.04 | 16.04 | 9.0 | 1 | 6 | b. VIII |
| Puteshestvennitsa | 7 | 7 | 10.04 | 12.04 | 21.04 | 11.3 | 1 | 6.5 | II dec. VII |
| Santa Rosa | 7 | 7 | 09.04 | 11.04 | 22.04 | 11.7 | 1 | 6 | II dec. VII |
| Shater | 3 | 7 | 08.04 | 10.04 | 21.04 | 11.3 | 1 | 9 | II dec. VII |
| Shiro | 7 | 7 | 11.04 | 13.04 | 25.04 | 12.7 | 1 | 6 | b. VIII |
| Skoroplodnaya | 5 | 5 | 11.04 | 13.04 | 21.04 | 8.0 | 2 | 5 | II dec. VII |
| Slivovidnaya | 7 | 7 | 11.04 | 13.04 | 22.04 | 11.3 | 2 | 6 | e. VII |
| Superior | 5 | 7 | 11.04 | 13.04 | 19.04 | 9.3 | 1 | 6 | II dec. VIII |
| Tatyana | 3 | 7 | 04.04 | 07.04 | 16.04 | 12.0 | 1 | 9 | b. VIII |
| Tsernushka | 7 | 7 | 06.04 | 08.04 | 19.04 | 11.6 | 1 | 8 | b. VII |
| Tsernushka Rannaya | 7 | 7 | 05.04 | 07.04 | 14.04 | 9.7 | 2 | 7.9 | b. VII |
| Vanier | 5 | 5 | 11.04 | 13.04 | 21.04 | 9.5 | 1 | 9 | II dec. VIII |
| Yevraziya | 7 | 7 | 13.04 | 15.04 | 25.04 | 9.3 | 3 | 7 | e. VII |
| Cultivar | Mean Fruit Weight [g] | Shape * | Skin Colour ** | Flesh Colour *** | Soluble Solids [%] | Stone Separating from Flesh **** |
|---|---|---|---|---|---|---|
| Angeleno | 57.3 de | 1 | 9 | 2/6 | 14.3 c–i | 1 |
| Barkhatnaya | 27.7 l–p | 2 | 9 | 2 | 14.0 d–j | 7 |
| Black Amber | 76.7 ab | 2 | 11 | 2 | 13.9 d–k | 1 |
| Black Diamond | 72.1 bc | 1 | 9 | 6 | 10.6 n–o | 1 |
| Blue Gigant | 87.7 a | 5 | 11 | 6 | 11.7 m–n | 1 |
| Chuk | 21.0 p | 2 | 8 | 2 | 17.8 a | 1 |
| Desertnaya Rannaya | 42.0 f–j | 1 | 8 | 2/6 | 12.9 h–l | 1 |
| Dofi Sandra | 39.0 f–l | 4 | 10 | 2 | 14.8 c–g | 1 |
| Early Golden | 41.3 f–k | 2 | 4 | 2 | 12.4 j–m | 3 |
| Ewierch Rannyj | 29.8 j–p | 2 | 9 | 2/6 | 15.5 b–e | 1 |
| Friar | 64.1 cd | 1 | 11 | 2 | 13.2 g–m | 1 |
| Formosa | 51.4 ef | 5 | 6 | 2 | 13.5 f–m | 1 |
| Gauota | 46.6 efg | 2 | 6 | 2 | 12.4 j–m | 5 |
| Gek | 28.3 k–p | 4 | 4 | 2 | 13.6 f–l | 5 |
| General | 51.8 ef | 5 | 8 | 2/6 | 13.0 g–m | 1 |
| Herkules | 78.8 ab | 2 | 6 | 2 | 12,6 i–m | 1 |
| Inese | 21.3 p | 4 | 6 | 2 | 15.1 c–f | 1 |
| Kometa | 35.2 g–o | 2 | 6 | 2 | 11.8 l–n | 1 |
| Kometa Late | 36.6 g–m | 5 | 8 | 2/6 | 12.1 k–n | 3 |
| Maschenka | 24.0 n–p | 2 | 6 | 2/6 | 13.5 f–m | 1 |
| Naidyona | 36.1 g–n | 2 | 6 | 2 | 13.9 d–k | 1 |
| Obilnaya | 43.4 f–i | 2 | 9 | 2/6 | 13.6 f–l | 7 |
| Oishi Wase | 49.7 ef | 5 | 6 | 2 | 16.0 bc | 1 |
| Ozark Premier | 73.2 bc | 2 | 8 | 2 | 15.7 bcd | 5 |
| Puteshestvennitsa | 27.9 l–p | 4 | 8 | 2/6 | 17.0 ab | 1 |
| Santa Rosa | 44.1 fgh | 5 | 9 | 6 | 15.2 c–f | 1 |
| Shater | 28.0 l–p | 4 | 10 | 2 | 9.7 o | 5 |
| Shiro | 35.2 g–o | 5 | 4 | 2 | 13.0 g–m | 1 |
| Skoroplodnaya | 26.7 l–p | 2 | 5/6 | 2 | 9.6 o | 5 |
| Slivovidnaya | 23.2 n–p | 2 | 8 | 2 | 13.7 e–k | 9 |
| Superior | 75.8 bc | 5 | 8 | 2 | 15.7 bcd | 1 |
| Tatyana | 32.2 h–p | 1 | 6 | 2 | 14.7 c–h | 3 |
| Tsernushka | 22.5 o–p | 2 | 8 | 2 | 15.9 bc | 1 |
| Tsernushka Rannaya | 25.9 l–p | 2 | 8 | 2 | 15.1 c–f | 1 |
| Vanier | 51.9 ef | 2 | 3 | 2 | 14.0 d–j | 1 |
| Yevraziya | 30.8 i–p | 2 | 10 | 2 | 15.2 c–f | 5 |
| No. | Cultivar | Ploidy Level | No. | Cultivar | Ploidy Level |
|---|---|---|---|---|---|
| 1 | Angeleno | 2× | 19 | Kometa Late | 2× |
| 2 | Barkhatnaya * | - | 20 | Maschenka | 2× |
| 3 | Black Amber | 2× | 21 | Naidyona | 2× |
| 4 | Black Diamond | 2× | 22 | Obilnaya | 2× |
| 5 | Blue Gigant | 2× | 23 | Oishi Wase | 2× |
| 6 | Chuk | 2× | 24 | Ozark Premier | 2× |
| 7 | Desertnaya Rannaya | 2× | 25 | Puteshestvennitsa | 2× |
| 8 | Dofi Sandra | 2× | 26 | Santa Rosa | 2× |
| 9 | Early Golden | 2× | 27 | Shiro | 2× |
| 10 | Ewierch Rannyj | 2× | 28 | Shater * | - |
| 11 | Friar | 2× | 29 | Skoroplodnaya | 2× |
| 12 | Formosa | 2× | 30 | Slivovidnaya | 2× |
| 13 | Gauota | 2× | 31 | Superior | 2× |
| 14 | Gek | 2× | 32 | Tatyana * | - |
| 15 | General | 2× | 33 | Tsernushka | 2× |
| 16 | Herkules | 5× | 34 | Tsernushka Rannaya | 2× |
| 17 | Inese | 2× | 35 | Vanier | 2× |
| 18 | Kometa | 2× | 36 | Yevraziya | 6× aneuploid |
| No. | Cultivar | Reported Parentage | Country |
|---|---|---|---|
| 1 | Angeleno | Selection within a population of seedlings resulted from open pollination | USA |
| 2 | Barkhatnaya | unknown | Ukraine |
| 3 | Black Amber | Friar × Queen Rosa | USA |
| 4 | Black Diamond | Angeleno × open pollination | England |
| 5 | Blue Gigant | No date | No date |
| 6 | Chuk | P. salicina × P.cerasifera (Skoroplodnaya × Otlichnitsa) | Russia |
| 7 | Desertnaya Rannaya | Wickson × P.cerasifera Tavricheskaya | Ukraine |
| 8 | Dofi Sandra | Black Gold × Burmosa | Italy |
| 9 | Early Golden | A changce seedling of Burbank or Shiro | Canada |
| 10 | Ewierch Rannyj | No date | Ukraine |
| 11 | Friar | Gaviota × Nubiana | USA |
| 12 | Formosa | unknown | USA |
| 13 | Gauota | unknown | Russia |
| 14 | Gek | Skoroplodnaya × P.cerasifera Otlichnitsa | Russia |
| 15 | General | Obilnaya × open pollination | Ukraine |
| 16 | Herkules | Ontario × Formosa | Sweden |
| 17 | Inese | Seedling of breeding number PU-16807 | Latvia |
| 18 | Kometa | P. salicina × P.cerasifera (Skoroplodnaya × Pionerka) | Russia |
| 19 | Kometa Late | Kubanska Kometa × open pollination | Russia |
| 20 | Maschenka | No date | Ukraine |
| 21 | Naidyona | P. salicina × P.cerasifera (Skoroplodnaya × Desertnaya) | Belarus |
| 22 | Obilnaya | P. salicina × P.cerasifera (Berbank × Tavricheskaya) | Ukraine |
| 23 | Oishi Wase | No date | No date |
| 24 | Ozark Premier | Burbank × Methly | USA |
| 25 | Puteshestvennitsa | P. salicina × P.cerasifera (seedling of cultivar Desertnaya) | Russia |
| 26 | Santa Rosa | P. salicina × P.simonii × P.americana | USA |
| 27 | Shater | Fibing × open pollination | Russia |
| 28 | Shiro | A chance seedling | USA |
| 29 | Skoroplodnaya | P. salicina × P.ussuriensis Ussuriyskaya Krasnaya × Klaymeks | Russia |
| 30 | Slivovidnaya | Obilnaya x open pollination | Ukraine |
| 31 | Superior | P. salicina Burbank × (P. americana × P. simonii) Kaga | USA |
| 32 | Tatyana | No date | Ukraine |
| 33 | Tsernushka | No date | Ukraine |
| 34 | Tsernushka Rannaya | No date | Ukraine |
| 35 | Vanier | Burbank × Wickson | Canada |
| 36 | Yevraziya | Lakrescent × open pollination | Russia |
| Year | Temperature [ °C] | Precipitation [mm] | ||
|---|---|---|---|---|
| Minimum | Maximum | Mean | Total | |
| 2015 | −11.4 | 37.9 | 9.9 | 384.0 |
| 2016 | −17.9 | 34.2 | 9.3 | 503.8 |
| 2017 | −20.9 | 37.4 | 9.0 | 564.0 |
| 2018 | −20.6 | 35.5 | 9.7 | 364.2 |
| 2019 | −11.5 | 39.0 | 10.6 | 359.7 |
| 2020 | −10.1 | 35.9 | 9.3 | 499.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Głowacka, A.; Sitarek, M.; Rozpara, E.; Podwyszyńska, M. Pomological Characteristics and Ploidy Levels of Japanese Plum (Prunus salicina Lindl.) Cultivars Preserved in Poland. Plants 2021, 10, 884. https://doi.org/10.3390/plants10050884
Głowacka A, Sitarek M, Rozpara E, Podwyszyńska M. Pomological Characteristics and Ploidy Levels of Japanese Plum (Prunus salicina Lindl.) Cultivars Preserved in Poland. Plants. 2021; 10(5):884. https://doi.org/10.3390/plants10050884
Chicago/Turabian StyleGłowacka, Agnieszka, Mirosław Sitarek, Elżbieta Rozpara, and Małgorzata Podwyszyńska. 2021. "Pomological Characteristics and Ploidy Levels of Japanese Plum (Prunus salicina Lindl.) Cultivars Preserved in Poland" Plants 10, no. 5: 884. https://doi.org/10.3390/plants10050884
APA StyleGłowacka, A., Sitarek, M., Rozpara, E., & Podwyszyńska, M. (2021). Pomological Characteristics and Ploidy Levels of Japanese Plum (Prunus salicina Lindl.) Cultivars Preserved in Poland. Plants, 10(5), 884. https://doi.org/10.3390/plants10050884







