Genetic Diversity of Stratiotes aloides L. (Hydrocharitaceae) Stands across Europe
Abstract
1. Introduction
2. Results
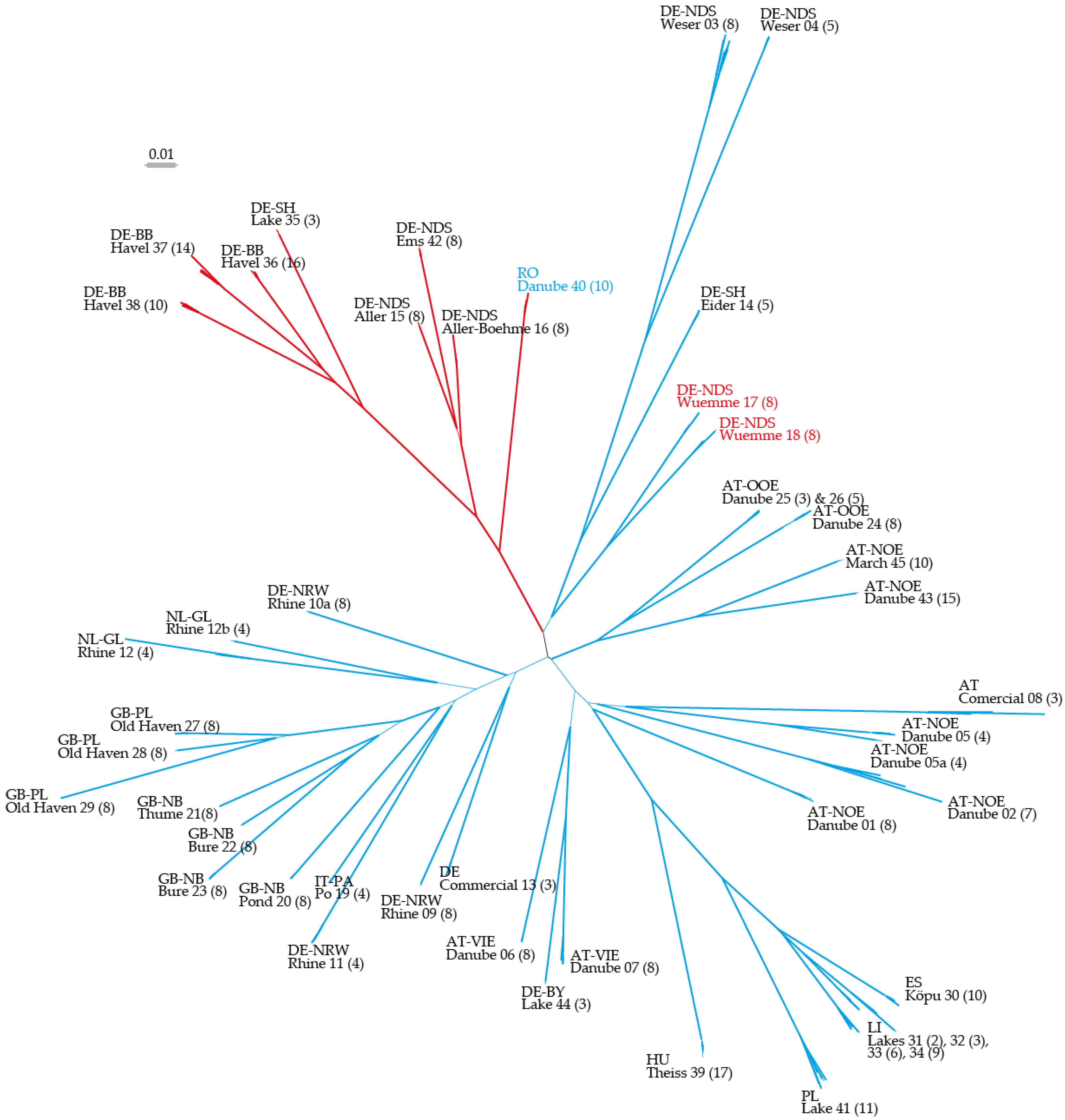
2.1. Neighbor Joining
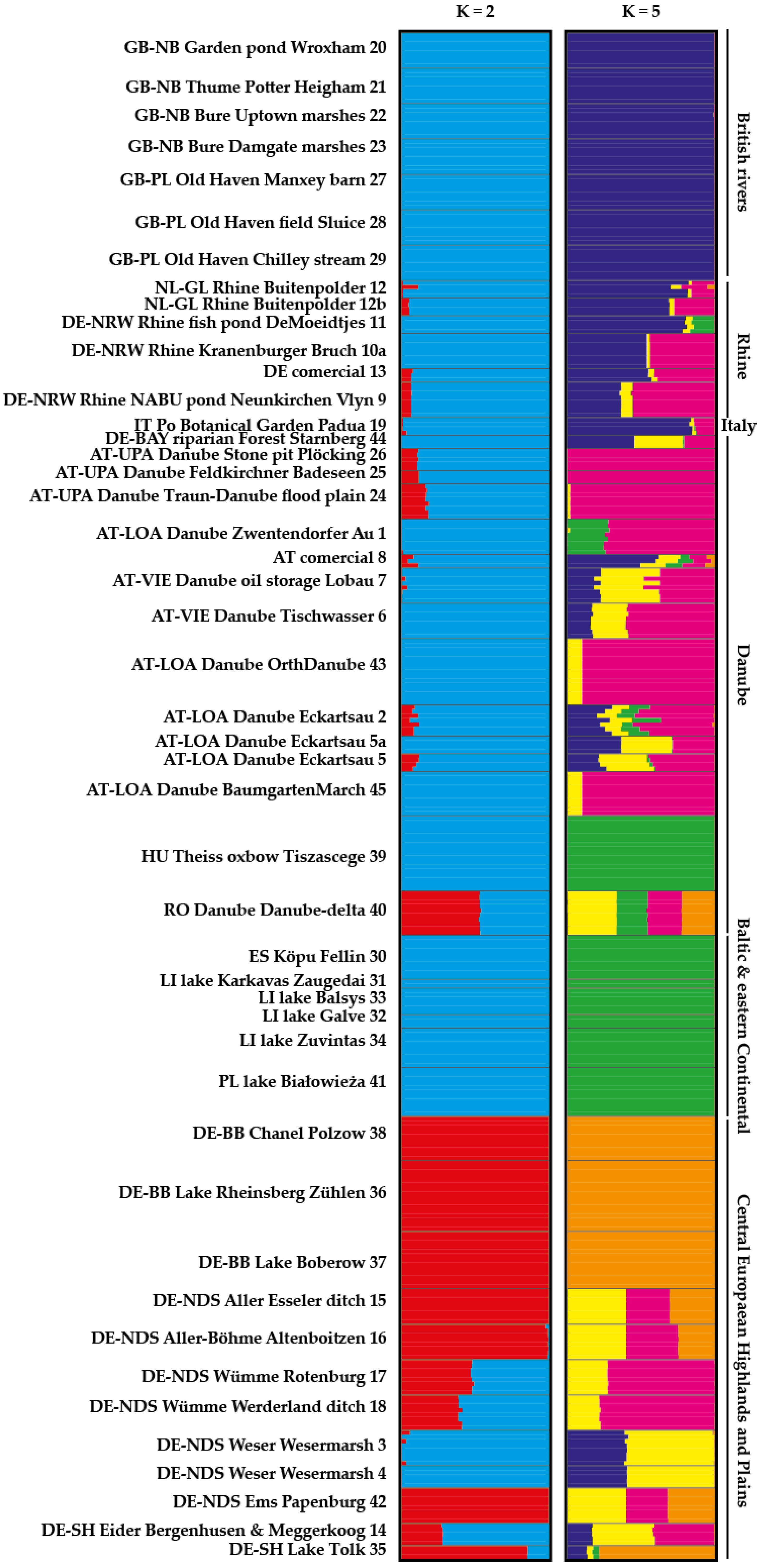
2.2. STRUCTURE
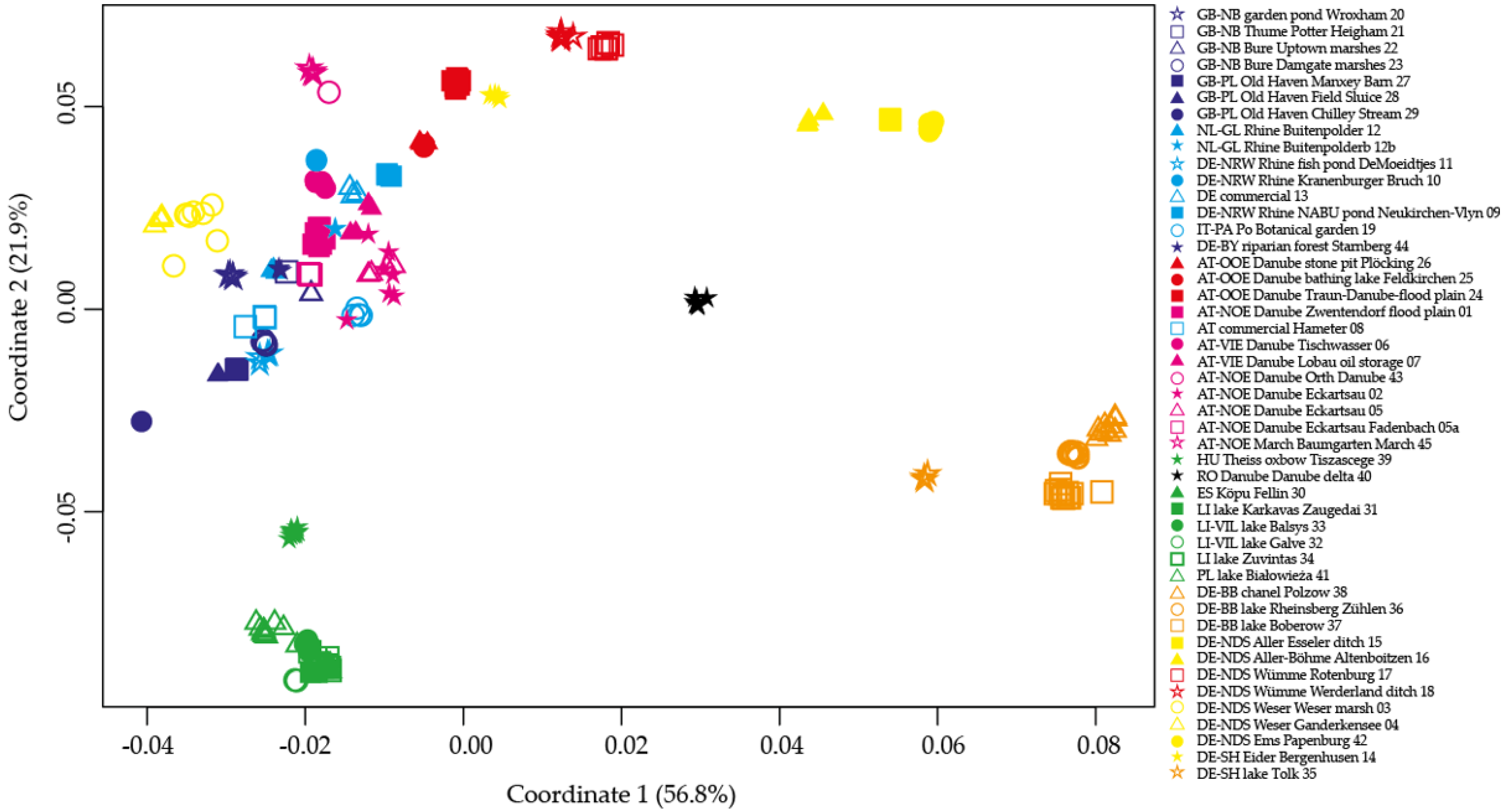
2.3. Principal Coordinate Analyses
2.4. AMOVA and Population Statistics
2.5. Mantel Tests
3. Discussion
4. Materials and Methods
4.1. Material
4.2. DNA Extraction
4.3. AFLP
4.4. Scoring and Phylogenetic Analysis
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Efremov, A.N.; Sviridenko, B.F.; Toma, C.; Mesterházy, A.; Murashko, Y.A. Ecology of Stratiotes aloides L. (Hydrochoritaceae) in Eurasia. Flora 2019, 253, 116–126. [Google Scholar] [CrossRef]
- Cook, C.; Urmi-König, K. A revision of the genus Stratiotes (Hydrocharitaceae). Aquat. Bot. 1983, 16, 213–249. [Google Scholar] [CrossRef]
- Küry, D. Krebsschere (Stratiotes aloides) in Naturschutzweihern der Schweiz. Bauhinia 2009, 21, 49–56. [Google Scholar]
- Rantala, M.J.; Ilmonen, J.; Koskimäki, J.; Suhonen, J.; Tynkkynen, K. The macrophyte, Stratiotes aloides, protects larvae of dragonfly Aeshna viridis against fish predation. Aquat. Ecol. 2004, 38, 77–82. [Google Scholar] [CrossRef]
- Suutari, E.; Salmela, J.; Paasivirta, L.; Rantala, M.J.; Tynkkynen, K.; Luojumäki, M.; Suhonen, J. Macroarthropod species richness and conservation priorities in Stratiotes aloides (L.) lakes. J. Insect Conserv. 2009, 13, 413–419. [Google Scholar] [CrossRef]
- Kalkman, V.J.; Boudot, J.-P.; Bernard, R.; Conze, K.-J.; De Knijf, G.; Dyatlova, E.; Ferreira, S.; Jović, M.; Ott, J.; Riservato, E.; et al. European Red List of Dragonflies; Publications Office of the European Union: Luxembourg, 2010. [Google Scholar]
- Sommerwerk, N.; Hein, T.; Schneider-Jakoby, M.; Baumgartner, C.; Ostojić, A.; Siber, R.; Bloesch, J.; Paunović, M.; Tockner, K. The Danube river basin. In Rivers of Europe; Tockner, K., Uehlinger, U., Robinson, C.T., Eds.; Elsevier: Amsterdam, The Netherlands, 2009; pp. 59–112. [Google Scholar]
- Schmidt-Mumm, U.; Janauer, G.A. Macrophyte assemblages in the aquatic-terrestrial transitional zone of oxbow lakes in the Danube floodplain (Austria). Folia Geobot. 2016, 51, 251–266. [Google Scholar] [CrossRef]
- Gerken, B. Auen, Verborgene Lebensadern der Natur, 1st ed.; Rombach GmbH + Co Verlagshaus KG: Freiburg im Breisgau, Germany, 1988; p. 131. [Google Scholar]
- Lange, G.; Lecher, K. Kleine Gewässer und landwirtschaftliche Vorfluter. In Gewässerregelung Gewässerpflege, 3rd ed.; Lange, G., Lecher, K., Eds.; Vieweg + Teubner Verlag: Wiesbaden, Germany, 1993; pp. 244–252. [Google Scholar]
- Smolders, A.J.P.; Lamers, L.P.M.; den Hartog, C.; Roelofs, J.G.M. Mechanisms involved in the decline of Stratiotes aloides L. in the Netherlands: Sulphate as a key variable. Hydrobiologia 2003, 506–509, 603–610. [Google Scholar] [CrossRef]
- Abeli, T.; Rossi, G.; Smolders, A.J.P.; Orsenigo, S. Nitrogen pollution negatively affects Stratiotes aloides in Central-Eastern Europe. Implications for translocation actions. Aquat. Conserv. 2014, 24, 724–729. [Google Scholar] [CrossRef]
- Kunze, K. Erpobung von Managementmaßnahmen in Bremen zum Erhalt der Krebsschere; Abschlusstagung, Hochschule Bremen: Bremen, Germany, 2010. [Google Scholar]
- Orsenigo, S.; Gentili, R.; Smolders, A.J.P.; Efremov, A.; Rossi, G.; Ardenghi, N.M.G.; Citterio, S.; Abeli, T. Reintroduction of a dioecious aquatic macrophyte (Stratiotes aloides L.) regionally extinct in the wild. Interesting answers from genetics. Aquat. Conserv. 2017, 27, 10–23. [Google Scholar] [CrossRef]
- Zedler, J.B.; Kercher, S. Causes and consequences of invasive plants in Wetlands: Opportunities, opportunists, and outcomes. Crit. Rev. Plant Sci. 2004, 23, 431–452. [Google Scholar] [CrossRef]
- Bolpagni, R. Towards global dominance of invasive alien plants in freshwater ecosystems: The dawn of the Exocene? Hydrobiologia 2021. [Google Scholar] [CrossRef]
- Kleyheeg, E.; Fiedler, W.; Safi, K.; Waldenström, J.; Wikelski, M.; Liduine van Toor, M. A Comprehensive model for the quantitative estimation of seed dispersal by migratory Mallards. Front. Ecol. Evol. 2019, 7, 40. [Google Scholar] [CrossRef]
- Forbes, R.S. Assessing the status of Stratiotes aloides L. (water-soldier) in Co. Fermanagh, Northern Ireland (v.c. H33). Watsonia 2000, 23, 179–196. [Google Scholar]
- Smolders, A.J.P.; den Hartog, C.; Roelofs, J.G.M. Observations on fruiting and seed-set of Stratiotes aloides L. in The Netherlands. Aquat. Bot. 1995, 51, 259–268. [Google Scholar] [CrossRef]
- Silvertown, J. The evolutionary maintenance of sexual reproduction: Evidence from the ecological distribution of asexual reproduction in clonal plants. Int. J. Plant Sci. 2008, 169, 157–168. [Google Scholar] [CrossRef]
- Lambertini, C.; Gustafsson, M.H.G.; Frydenberg, J.; Speranza, M.; Brix, H. Genetic diversity patterns in Phragmites australis at the population, regional and continental scales. Aquat. Bot. 2008, 88, 160–170. [Google Scholar] [CrossRef]
- Loeschcke, V.; Tomiuk, J.; Jain, S.K. Introductory remarks: Genetics and conservation biology. In Conservation Genetics; Loeschcke, V., Tomiuk, J., Eds.; Birkhäuser Verlag: Basel, Switzerland, 1994; EXS; Volume 68, pp. 3–8. [Google Scholar]
- Uehlinger, U.; Arndt, H.; Wantzen, K.M.; Leuven, R.S.E.W. The Rhine river basin. In Rivers of Europe; Tockner, K., Uehlinger, U., Robinson, C.T., Eds.; Elsevier: Amsterdam, The Netherlands, 2009; pp. 59–112. [Google Scholar]
- Tolf, C.; Bengtsson, D.; Rodrigues, D.; Latorre-Margalef, N.; Wille, M.; Figueiredo, M.E.; Jankowska-Hjortaas, M.; Germundsson, A.; Duby, P.-Y.; Lebarbenchon, C.; et al. Birds and viruses at a crossroads—Surveillance of influenza A virus in Portuguese waterfowl. PLoS ONE 2012, 7, e49002. [Google Scholar] [CrossRef] [PubMed]
- Kranichschutz Deutschland. Available online: https://www.kraniche.de/en/crane-migration.html (accessed on 18 April 2021).
- Bennike, O.; Hoek, W. Late Glacial and early Holocene records of Stratiotes aloides L. from northwestern Europe. Rev. Palaeobot. Palynol. 1999, 107, 259–263. [Google Scholar] [CrossRef]
- Guo, J.-L.; Yu, Y.-H.; Zhang, J.-W.; Li, Z.-M.; Zhang, Y.-H.; Volis, S. Conservation strategy for aquatic plants: Endangered Ottelia acuminata (Hydrocharitaceae) as a case study. Biodivers. Conserv. 2019, 28, 1533–1548. [Google Scholar] [CrossRef]
- Schratt-Ehrendorfer, L. Geobotanisch-ökologische Untersuchungen zum Indikatorwert von Wasserpflanzen und ihren Gesellschaften in Donaualtwässern bei Wien. Stapfia 1999, 64, 23–162. [Google Scholar]
- Suhonen, J.; Suutari, E.; Kaunisto, K.M.; Krams, I. Patch area of marophyte Stratiotes aloides as a critical resource for declining dragonfly Aeshna viridis. J. Insect Conserv. 2013, 17, 393–398. [Google Scholar] [CrossRef]
- Beintema, A.J.; van der Winden, J.; Baarspul, T.; de Krijger, J.P.; van Oers, K.; Keller, M. Black terns Chlidonias niger and their dietary problems in Dutch wetlands. Ardea 2010, 98, 365–372. [Google Scholar] [CrossRef]
- Trockner, K.; Tonolla, D.; Uehlinger, U.; Siber, R.; Robinson, C.T.; Peter, F.D. Introduction to European rivers. In Rivers of Europe; Tockner, K., Uehlinger, U., Robinson, C.T., Eds.; Elsevier: Amsterdam, The Netherlands, 2009; pp. 59–112. [Google Scholar]
- Vos, P.; Hogers, R.; Bleeker, M.; Reijans, M.; Van de Lee, T.; Hornes, M.; Frijters, A.; Pot, J.; Peleman, J.; Kuiper, M.; et al. AFLP: A new technique for DNA fingerprinting. Nucleic Acids Res. 1995, 23, 4407–4414. [Google Scholar] [CrossRef] [PubMed]
- Safer, S.; Tremetsberger, K.; Guo, Y.-P.; Kohl, G.; Samuel, M.R.; Stuessy, T.F.; Stuppner, H. Phylogenetic relationships in the genus Leontopodium (Asteraceae: Gnaphalieae) based on AFLP data. Bot. J. Linn. Soc. 2011, 165, 364–377. [Google Scholar] [CrossRef] [PubMed]
- Swofford, D.L. PAUP*: Phylogenetic Analysis Using Parsimony (and Other Methods) Version 4; Sinauer Associates: Sunderland, MA, USA, 2003. [Google Scholar]
- Huson, D.H.; Bryant, D. Application of phylogenetic networks in evolutionary studies. Mol. Biol. Evol. 2006, 23, 254–267. [Google Scholar] [CrossRef] [PubMed]
- Goslee, S.C.; Urban, D.L. The ecodist package for dissimilarity-based analysis of ecological data. J. Stat. Softw. 2007, 22, 1–19. [Google Scholar] [CrossRef]
- Ligges, U.; Mächler, M. Scatterplot3d—An R package for visualizing multivariate data. J. Stat. Softw. 2003, 8, 1–20. [Google Scholar] [CrossRef]
- Excoffier, L.; Lischer, H.E.L. Arlequin suite ver 3.5: A new series of programs to perform population genetics analyses under Linux and Windows. Mol. Ecol. Resour. 2010, 10, 564–567. [Google Scholar] [CrossRef]
- Pritchard, J.K.; Stephens, M.; Donelly, P. Inference of population structure using multilocus genotype data. Gentetics 2000, 155, 945–959. [Google Scholar]
- Falush, D.; Stephens, M.; Pritchard, J.K. Inference of population structure using multilocus genotype data: Linked loci and correlated allele frequencies. Genetics 2003, 164, 1567–1587. [Google Scholar] [PubMed]
- Falush, D.; Stephens, M.; Pritchard, J.K. Inference of population structure using multilocus genotype data: Dominant markers and null alleles. Mol. Ecol. Notes 2007, 7, 574–578. [Google Scholar] [CrossRef] [PubMed]
- Hubisz, M.J.; Falush, D.; Stephens, M.; Pritchard, J.K. Inferring weak population structure with the assistance of sample group information. Mol. Ecol. Resour. 2009, 9, 1322–1332. [Google Scholar] [CrossRef]
- Raj, A.; Stephens, M.; Pritchard, J.K. fastSTRUCTURE: Variational inference of population structure in large SNP data sets. Genetics 2014, 197, 573–589. [Google Scholar] [CrossRef]
- Evanno, G.; Regnaut, S.; Goudet, J. Detecting the number of clusters of individuals using the software STRUCTURE: A simulation study. Mol. Ecol. 2005, 14, 2611–2620. [Google Scholar] [CrossRef]
- Earl, D.A.; Von Holdt, B.M. STRUCTURE HARVESTER: A website and program for visualizing STRUCTURE output and implementing the Evanno method. Conserv. Genet. Resour. 2012, 4, 359–361. [Google Scholar] [CrossRef]
- Jakobsson, M.; Rosenberg, N.A. CLUMPP: A cluster matching and permutation program for dealing with label switching and multimodality in analysis of population structure. Bioinformatics 2007, 23, 1801–1806. [Google Scholar] [CrossRef]
- Rosenberg, N.A. DISTRUCT: A program for the graphical display of population structure. Mol. Ecol. Notes 2004, 4, 137–138. [Google Scholar] [CrossRef]
- Peakall, R.; Smouse, P.E. GenAlEx 6.5: Genetic analysis in Excel. Population genetic software for teaching and research—An update. Bioinformatics 2012, 28, 2537–2539. [Google Scholar] [CrossRef] [PubMed]
- Mantel, N. The detection of disease clustering and a generalized regression approach. Cancer Res. 1967, 27, 209–220. [Google Scholar] [PubMed]
- Ersts, P.J. Geographic Distance Matrix Generator (Version 1.2.3); American Museum of Natural History, Center for Biodiversity and Conservation: New York, NY, USA; Available online: http://biodiversityinformatics.amnh.org/open_source/gdmg (accessed on 12 January 2021).
- Nei, M. Analysis of gene diversity in subdivided populations. Proc. Natl. Acad. Sci. USA 1973, 70, 3321–3323. [Google Scholar] [CrossRef]
- Lewontin, R.C. The apportionment of human diversity. In Evolutionary Biology; Dobzhansky, T., Hecht, M.K., Steere, W.C., Eds.; Springer: New York, NY, USA, 1972; pp. 381–398. [Google Scholar]
| Pop nr | River System 1 | Country | Region | Location | Nr. Indivs. | Year | Collector | HBV Acc. Nr | Coordinates |
|---|---|---|---|---|---|---|---|---|---|
| 8 | commercial | Austria | commercial | Stauden Hameter | 3 | 2012 | (Hameister S) | N 48°17′03.03″ E 16°02′19.84″ | |
| 1 | Danube | Austria | Lower Austria | Floodplain Zwentendorf, Obere Placken | 8 | 2012 | Bernhardt K-G | 56059 57014 | N 48°22′14.00″ E 15°47′47.00″ |
| 2 | Danube | Austria | Lower Austria | Eckartsau Fadenbach | 7 | 2012 | Hermann | N 48°08′03.96″ E 16°45′45.03″ | |
| 5 | Danube | Austria | Lower Austria | Eckartsau Fadenbach | 4 | 2012 | Bernhardt K-G | N 48°08′10.50″ E 16°46′52.80″ | |
| 5a | Danube | Austria | Lower Austria | Eckartsau Fadenbach | 4 | 2012 | Hameister S | M 48°08′10.50″ E 16°46′52.80″ | |
| 6 | Danube | Austria | Vienna | Tischwasser | 8 | 2012 | Hameister S | N 48°11′34.49″ E 16°28′54.84″ | |
| 7 | Danube | Austria | Vienna | Oilstrage Lobau | 8 | 2012 | Hameister S | N 48°10′48.55″ E 16°29′47.30″ | |
| 24 | Danube | Austria | Upper Austria | Traun-Danube-floodplain | 8 | 2013 | Hameister S Hudler A | N 48°15′16.90″ E 14°23′18.20″ | |
| 25 | Danube | Austria | Upper Austria | Bathing lake Feldkirchen | 3 | 2013 | Hameister S Hudler A | N 48°19′41.20″ E 14°03′45.90″ | |
| 26 | Danube | Austria | Upper Austria | Stone-pit Plöcking | 5 | 2013 | Hameister S Hudler A | N 48°26′35.00″ E 14°00′14.20″ | |
| 43 | Danube | Austria | Lower Austria | Orth an der Donau/Steinafurt | 15 | 2015 | Lapin K | N 48°08′31.90″ E 16°41′03.70″ | |
| 45 | Danube | Austria | Lower Austria | Baumgarten ad March, Maritz South | 10 | 2018 | Gregor L | N 48°18′50.00″ E 16°53′12.00″ | |
| 13 | commercial | Germany | commercial | Holzum | 1 | 2013 | (Hameister S) | N 51°46′36.13″ E 06°24′12.77″ | |
| 13 | commercial | Germany | commercial | Stauden Förster | 2 | 2013 | (Hameister S) | N 52°25′10.68″ E 13°01′11.81″ | |
| 9 | Rhine | Germany | Nordrhein-Westfalen | NABU pond Neukirchen Vlyn | 8 | 2013 | Hameister S | N 51°26′48.60″ E 06°32′35.20″ | |
| 10 | Rhine | Germany | Nordrhein-Westfalen | Kranenburger Bruch | 8 | 2013 | Hameister S | N 51°47′14.20″ E 06°01′37.50″ | |
| 11 | Rhine | Germany | Nordrhein-Westfalen | Fishpond “De Moeidtjes” | 4 | 2013 | Hameister S | N 51°51′04.00″ E 06°10′14.60″ | |
| 12 | Rhine | Netherlands | Gelderland | Buitenpolder (Rhine back water) | 8 | 2013 | Hameister S | N 51°54′03.30″ E 06°03′39.90″ | |
| 12b | Rhine | Netherlands | Gelderland | Buitenpolder (Rhine back water) | 8 | 2013 | Hameister S | N 51°54′03.50″ E 06°03′44.60″ | |
| 14 | CHP | Germany | Schleswig-Holstein | Eider-Bergenhusen | 1 | 2013 | Rasran L | N 54°22′06.41″ E 09°20′55.39″ | |
| 14 | CHP | Germany | Schleswig-Holstein | Eider-Bergenhusen NABU | 2 | 2013 | Rasran L | N 54°22′27.17″ E 09°19′24.49″ | |
| 14 | CHP | Germany | Schleswig-Holstein | Eider-Meggerkoog | 2 | 2013 | Rasran L | N 54°21′55.82″ E 09°22′47.45″ | |
| 35 | CHP | Germany | Schleswig-Holstein | Tolk-lake | 3 | 2014 | Rasran L | N 54°34′37.37″ E 09°37′37.37″ | |
| 3 | CHP | Germany | Niedersachsen | Weser marsh Bremen | 8 | 2012 | Bernhardt K-G | N 53°08′38.60″ E 08°39′24.60″ | |
| 4 | CHP | Germany | Niedersachsen | Ganderkensee-Werderland | 5 | 2012 | Hanke K | N 53°02′03.24″ E 08°32′33.52″ | |
| 15 | CHP | Germany | Niedersachsen | Aller, Esseler ditch | 8 | 2013 | Turner F | N 52°42′12.26″ E 09°37′30.91″ | |
| 16 | CHP | Germany | Niedersachsen | Aller (Böhme), Altenboitzen | 8 | 2013 | Turner F | N 52°48′49.25″ E 09°32′14.69″ | |
| 17 | CHP | Germany | Niedersachsen | Wümme, Rotenburg | 8 | 2013 | Turner F | N 53°05′49.86″ E 09°21′20.31″ | |
| 18 | CHP | Germany | Niedersachsen | Wümme, Werderland ditch | 8 | 2013 | Turner F | 57455 57456 | N 53°08′49.41″ E 08°38′25.49″ |
| 36 | CHP | Germany | Brandenburg | Havel, Rheinsberg-Zühlen lake | 16 | 2014 | Grimm Oldorff S | N 53°03′55.44″ E 12°48′54.84″ | |
| 37 | CHP | Germany | Brandenburg | Havel, Boberow lake | 13 | 2014 | Grimm Oldorff S | N 53°10′57.11″ E 13°01′12.76″ | |
| 38 | CHP | Germany | Brandenburg | Havel, Chanel Polzow | 10 | 2014 | Grimm Oldorff S | N 53°07′03.17″ E 13°01′07.05″ | |
| 42 | CHP | Germany | Niedersachsen | Ems, Channel Papenburg | 8 | 2015 | Tremetsberger K | 64641 | N 53°04′27.15″ E 07°27′03.61″ |
| 44 | Danube | Germany | Bavaria | Isar, Riparian forest Starnberg | 3 | 2016 | Bernhardt K-G | 67455 67456 | N 48°01′37.10″ E 11°23′32.60″ |
| 19 | Italian | Italy | Po, Botanical Garden Padua | 4 | 2013 | Bernhard K-G, Hameister S | N 45°23′55.94″ E 11°52′50.69″ | ||
| 20 | British | Great Britain | Norfolk | Garden pond, Norfolk Broads Wroxham | 8 | 2013 | Leaney B | N 52°42′21.02″ E 01°24′04.56″ | |
| 21 | British | Great Britain | Norfolk | Thume, Norfolk Broads, Potter Heigham | 8 | 2013 | Leaney B | N 52°42′14.34″ E 01°34′31.31″ | |
| 22 | British | Great Britain | Norfolk | Bure, Norfolk Broads, Uptown Marshes | 8 | 2013 | Leaney B | N 52°39′46.43″ E 01°32′31.98″ | |
| 23 | British | Great Britain | Norfolk | Bure, Norfolk Broads, Damgate Marshes | 8 | 2013 | Leaney B | N 52°37′54.15″ E 01°33′34.24″ | |
| 27 | British | Great Britain | East Sussex | Old haven, Pevensey Level, Manxey Barn | 8 | 2013 | Birch J | N 50°49′16.21″ E 00°21′3.45″ | |
| 28 | British | Great Britain | East Sussex | Old haven, Pevensey Level, Field Sluice | 8 | 2013 | Birch J | N 50°49′16.21″ E 00°21′03.45″ | |
| 29 | British | Great Britain | East Sussex | Old haven, Pevensey Level, Chilley Stream | 8 | 2013 | Birch J | N 50°49′16.21″ E 00°21′03.45″ | |
| 30 | BEC | Estonia | Vijandi | Köpu, Fellin | 10 | 2014 | Vellak K | N 58°20′09.00″ E 25°20′08.00″ | |
| 31 | BEC | Lithuania | Utena | Karkavas lake, Zaugedai | 2 | 2014 | Bernhardt K-G | 62094 | N 55°06′22.30″ E 25°40′08.78″ |
| 32 | BEC | Lithuania | Vilnius | Galve lake | 3 | 2014 | Bernhardt K-G | N 54°39′00.20″ E 24°55′49.90″ | |
| 33 | BEC | Lithuania | Vilnius | Balsys lake | 6 | 2014 | Bernhardt K-G | N 54°47′01.50″ E 25°20′00.90″ | |
| 34 | BEC | Lithuania | Alytus | Zuvintas lake | 9 | 2014 | Bernhardt K-G | 62091 62092 62093 | N 54°27′26.40″ E 23°38′18.40″ |
| 39 | Danube | Hungary | BH | Theiss oxbow, Tiszascege. | 17 | 2014 | Hameister S Oschatz | N 47°40′45.20″ E 20°59′01.90″ | |
| 40 | Danube | Romania | Tulcea | Danube-delta; E Tulcea. NE Murighiol. | 10 | 2015 | Bernhardt K-G | 64193 | N 45°08′38.10″ E 29°19′30.70″ |
| 41 | BEC | Poland | Podlachien | Białowieża; Palace Park | 11 | 2015 | Wernisch MM | 64088 | N 52°42′05.32″ E 23°50′42.42″ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Turner, B.; Hameister, S.; Hudler, A.; Bernhardt, K.-G. Genetic Diversity of Stratiotes aloides L. (Hydrocharitaceae) Stands across Europe. Plants 2021, 10, 863. https://doi.org/10.3390/plants10050863
Turner B, Hameister S, Hudler A, Bernhardt K-G. Genetic Diversity of Stratiotes aloides L. (Hydrocharitaceae) Stands across Europe. Plants. 2021; 10(5):863. https://doi.org/10.3390/plants10050863
Chicago/Turabian StyleTurner, Barbara, Steffen Hameister, Andreas Hudler, and Karl-Georg Bernhardt. 2021. "Genetic Diversity of Stratiotes aloides L. (Hydrocharitaceae) Stands across Europe" Plants 10, no. 5: 863. https://doi.org/10.3390/plants10050863
APA StyleTurner, B., Hameister, S., Hudler, A., & Bernhardt, K.-G. (2021). Genetic Diversity of Stratiotes aloides L. (Hydrocharitaceae) Stands across Europe. Plants, 10(5), 863. https://doi.org/10.3390/plants10050863






