Ethnomedicinal Plants Traditionally Used for the Treatment of Jaundice (Icterus) in Himachal Pradesh in Western Himalaya—A Review
Abstract
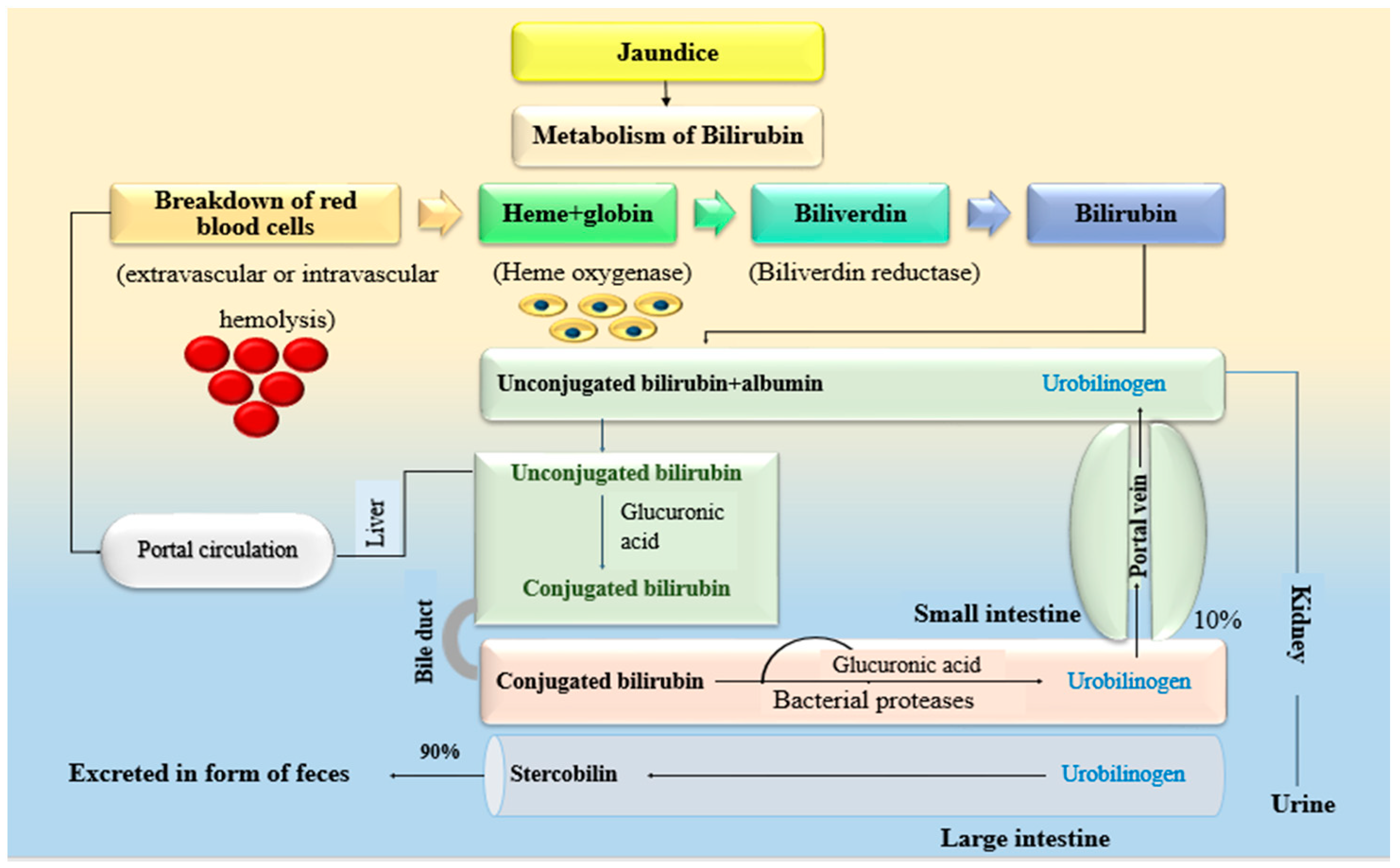
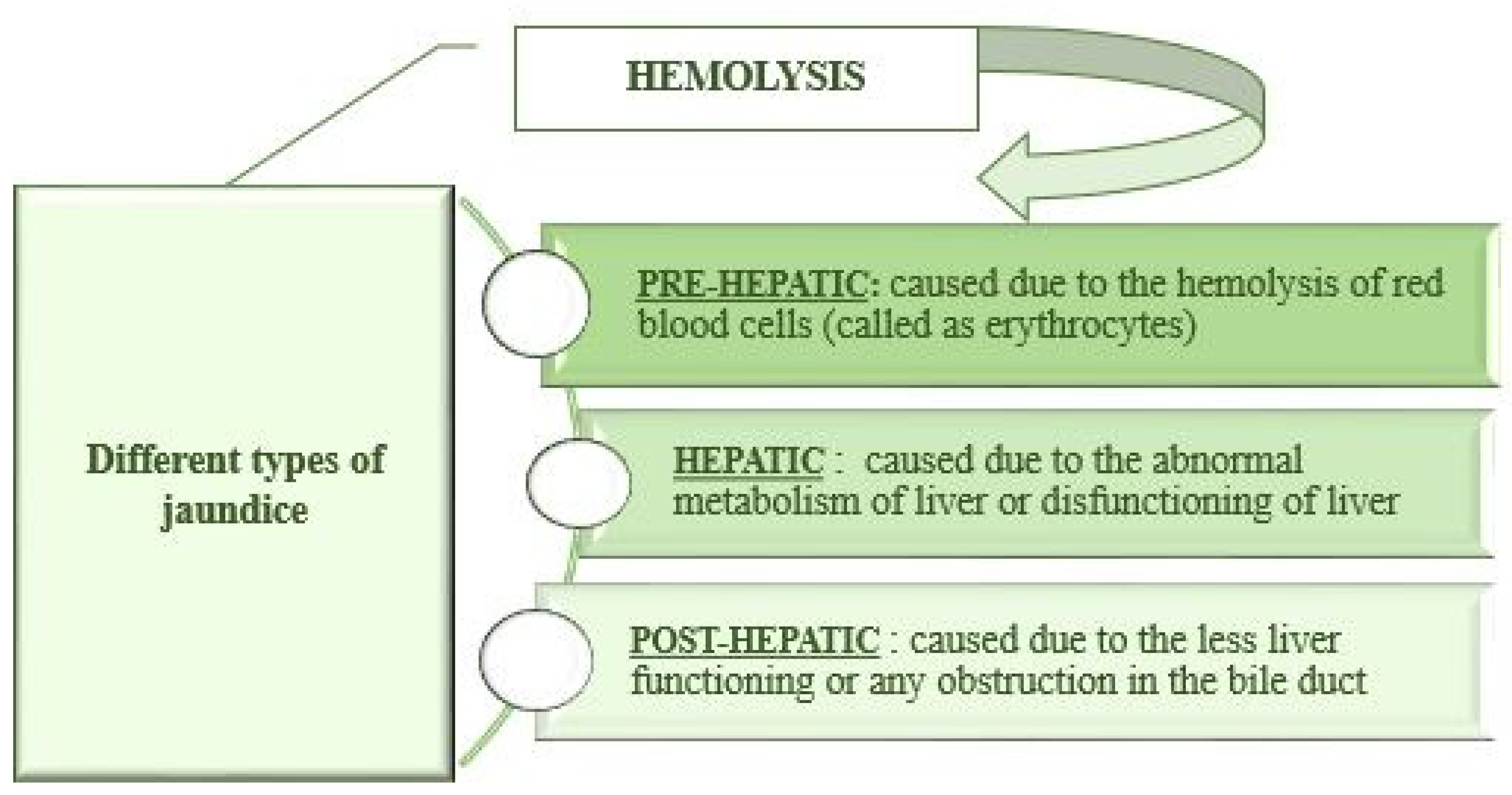
1. Introduction
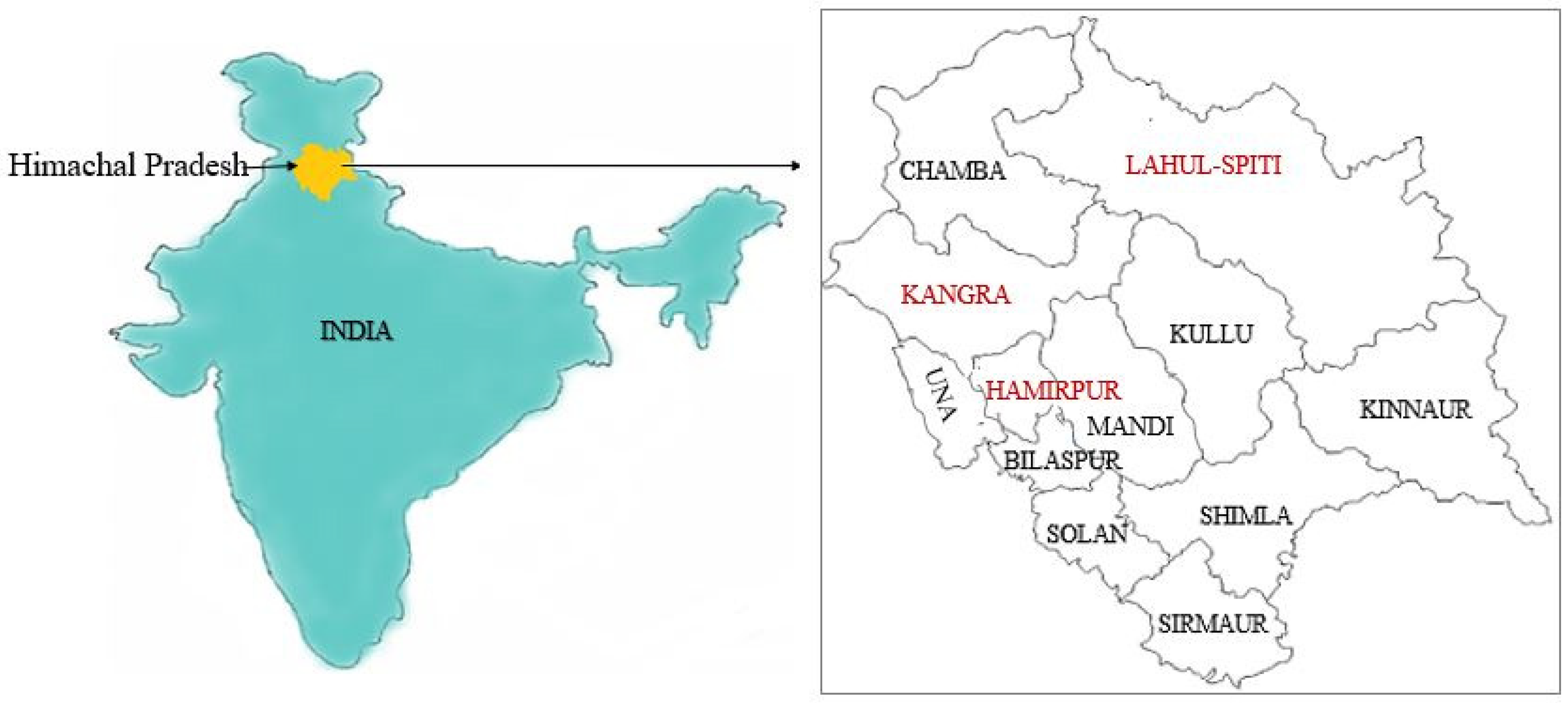
2. Materials and Methods
2.1. Ethnomedicinal Plants Used in the Treatment of Jaundice in Himachal Pradesh
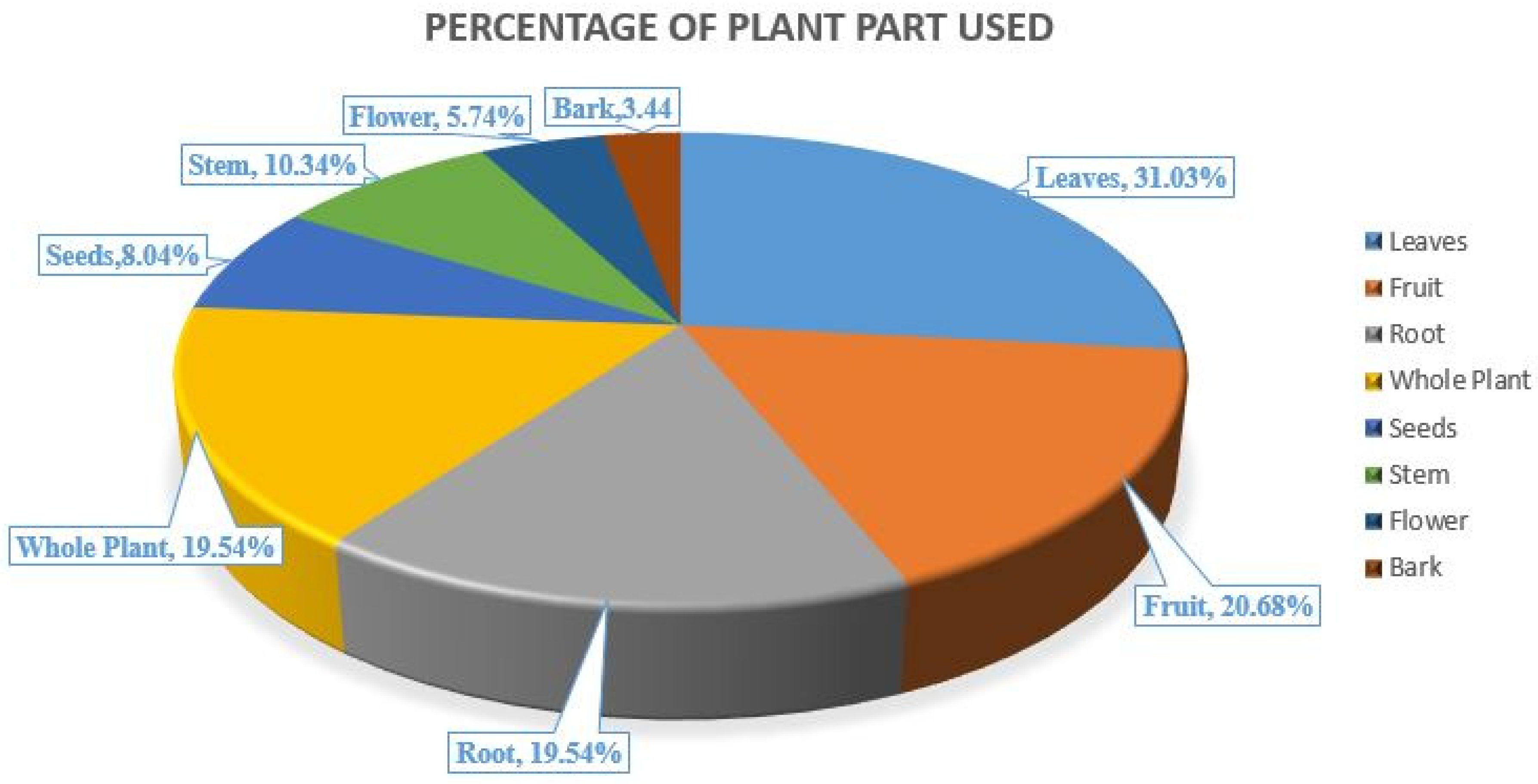
2.2. Mode of Plant Used for the Treatment of Jaundice in Himachal Pradesh
2.3. Phytochemical Constituents Present in the Ethnomedicinal Plants
2.4. Ethnopharmacological Evidence of Some Plant Species Used for the Treatment of Jaundice
3. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Thakur, K.; Bassi, K.; Sood, K. Textbook of Ethnobotany; S. Dinesh & Co.: Jalandhar, India, 2015; pp. 1–70. [Google Scholar]
- Aswal, B.S.; Mehrotra, B.N. Flora of Lahaul-Spiti; Bishen Singh Mahendra Pal Singh: Dehradun, India, 1994; pp. 10–15. [Google Scholar]
- Joshi, K.L. Geography of Himachal Pradesh; National Book Trust: New Delhi, India, 1987; pp. 220–222.
- Abu-Rabia, A. Urinary diseases and ethnobotany among pastoral nomads in the Middle East. J. Ethnobiol. 2005, 1, 1–3. [Google Scholar]
- Sharma, J.; Gairola, S.; Gaur, R.D.; Painuli, R.M. The treatment of jaundice with medicinal plants in indigenous communities of the sub-Himalayan region of Uttarakhand, India. J. Ethnopharmacol. 2012, 143, 262–291. [Google Scholar] [CrossRef] [PubMed]
- Negi, P.S.; Subramani, S.P. Ethnobotanical study in the village Chhitkul of Sangla valley, district Kinnaur, Himachal Pradesh. J. Non-Timber For. Prod. 2002, 9, 113–120. [Google Scholar]
- Birhane, E.; Aynekulu, E.; Mekuria, W.; Endale, D. Management, use and ecology of medicinal plants in the degraded dry lands of Tigray, Northern Ethiopia. J. Med. Plants Res. 2011, 5, 308–319. [Google Scholar]
- Mesfin, K.; Tekle, G.; Tesfay, T. Ethnobotanical study of traditional medicinal plants used by indigenous people of Gemad district, Northern Ethiopia. J. Med. Plants. 2013, 1, 32–37. [Google Scholar]
- Suriyavathana, M.; Jeevitha, M.; Aranganathan, J. In vitro antioxidant profile of Justicia tranquebariensis. Int. J. Pharm. Sci. 2011, 4, 4259–4261. [Google Scholar]
- Yadav, R.S.; Agarwala, M. Phytochemical analysis of some medicinal plants. J. Phytol. 2011, 3, 10–14. [Google Scholar]
- Chauhan, N.S. Important medicinal and aromatic plants of Himachal Pradesh. Indian For. 2003, 129, 979–998. [Google Scholar]
- Verma, R.; Parkash, V.; Kumar, D. Ethno-medicinal uses of some plants of Kanag hill in Shimla, Himachal Pradesh, India. Int. J. Res. Ayurveda Pharm. 2012, 3, 319–322. [Google Scholar]
- Baqar, S.R. Text Book of Economic Botany; Ferozsons Ltd.: Rawalpindi, Pakistan, 2001; pp. 23–100. [Google Scholar]
- Abbasi, A.M.; Khan, M.A.; Ahmad, M.; Zafar, M.; Khan, H.; Muhammad, N.; Sultana, S. Medicinal plants used for the treatment of jaundice and hepatitis based on socio-economic documentation. Afr. J. Biotechnol. 2009, 8, 1644–1650. [Google Scholar]
- Janghel, V.; Patel, P.; Chandel, S.S. Plants used for the treatment of icterus (jaundice) in central India: A review. Ann. Hepatol. 2019, 18, 658–672. [Google Scholar] [CrossRef]
- Riva, M.A.; Riva, E.; Spicci, M.; Strazzabosco, M.; Giovannini, M.; Cesana, G. “The city of Hepar”: Rituals, gastronomy, and politics at the origins of the modern names for the liver. J. Hepatol. 2011, 55, 1132–1136. [Google Scholar] [CrossRef]
- Gao, B.; Bataller, R. Alcoholic liver disease: Pathogenesis and new therapeutic targets. Gastroenterology 2011, 141, 1572–1585. [Google Scholar] [CrossRef] [PubMed]
- Rahim, Z.B.; Rahman, M.M.; Saha, D.; Hosen, S.Z.; Paul, S.; Kader, S. Ethnomedicinal plants used against jaundice in Bangladesh and its economical prospects. Bull. Pharma. Res. 2012, 2, 91–105. [Google Scholar]
- Feldman, A.G.; Sokol, R.J. Neonatal cholestasis. Neoreviews 2013, 14, e63–e73. [Google Scholar] [CrossRef] [PubMed]
- Sarkhel, S. Ethnomedicinal uses of some plants in treatment of jaundice by tribal communities of Paschim Medinipur district, West Bengal, India. Open Access J. Med. Arom. Plants. 2015, 6, 43–48. [Google Scholar]
- Poduri, C.D. Jaundice: A brief historical perspective. Ann. Intern. Med. 2016, 13, 76–79. [Google Scholar] [CrossRef]
- Ebrahimimd, S.; Ashkani-Esfahani, S.; Poormahmudibs, A. Investigating the efficacy of Zizyphus jujuba on neonatal jaundice. Iran J. Pediatr. 2011, 21, 320–324. [Google Scholar]
- Kumar, S.; Paul, R. Ethnomedicinal plants used for jaundice in Kangra district (Himachal Pradesh). J. Plant Dev. Sci. 2009, 1, 35–39. [Google Scholar]
- Kumar, N.; Choyal, R. Traditional use of some plants of Hamirpur district of Himachal Pradesh for the treatment of jaundice, hepatitis and other liver disorders. Int. J. Theor. Appl. Sci. 2012, 4, 201–205. [Google Scholar]
- Bharadwaj, J.; Seth, M.K. Medicinal plant resources of Bilaspur, Hamirpur and Una districts of Himachal Pradesh: An ethnobotanical enumeration. J. Med. Plants Stud. 2017, 5, 99–110. [Google Scholar]
- Thakur, M.; Asrani, R.K.; Thakur, S.; Sharma, P.K.; Patil, R.D.; Lal, B.; Parkash, O. Observations on traditional usage of ethnomedicinal plants in humans and animals of Kangra and Chamba districts of Himachal Pradesh in north-western Himalaya, India. J. Ethnopharmacol. 2016, 191, 280–300. [Google Scholar] [CrossRef]
- Singh, J.; Singh, J.; Sharma, D. Traditional wisdom to treat the most common ailments in Chopal region of Shimla District, Himachal Pradesh, India. Plant Arch. 2018, 18, 2759–2769. [Google Scholar]
- Kala, C.P. Medicinal plants of the high-altitude cold desert in India: Diversity, distribution and traditional uses. Int. J. Biodivers. Sci. Ecosyst. Serv. Manag. 2006, 2, 43–56. [Google Scholar] [CrossRef]
- Kaur, M.; Singhal, V.K.; Singh, J. Use of some ethnomedicinal herbs by the natives of Solang Valley, Kullu District, Himachal Pradesh. Int. J. Pharm. Sci. 2017, 9, 222–227. [Google Scholar] [CrossRef]
- Sekar, K.C.; Srivastava, S.K. Traditional uses of plants in curing jaundice in the Pin Valley National Park, Himachal Pradesh. Indian J. Tradit. Know. 2005, 4, 314–316. [Google Scholar]
- Singh, K.N.; Lal, B. Ethnomedicines used against four common ailments by the tribal communities of Lahaul-Spiti in western Himalaya. J. Ethnopharmacol. 2008, 115, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Rani, S.; Rana, J.C.; Rana, P.K. Ethnomedicinal plants of Chamba district, Himachal Pradesh, India. J. Med. Plants Res. 2013, 7, 3147–3157. [Google Scholar]
- Kaur, I.; Sharma, S.; Lal, S. Ethnobotanical survey of medicinal plants used for different diseases in Mandi district, Himachal Pradesh. Int. J. Res. Pharm. Chem. 2011, 1, 1167–1171. [Google Scholar]
- Rani, S.; Rana, J.C.; Jeelani, S.M.; Gupta, R.C.; Kumari, S. Ethnobotanical notes on 30 medicinal polypetalous plants of district Kangra of Himachal Pradesh. J. Med. Plants Res. 2013, 7, 1362–1369. [Google Scholar]
- Thakur, S. Medicinal plants used by tribal inhabitants of Sirmour district, Himachal Pradesh. Indian J. Sci. Res. 2011, 2, 125–127. [Google Scholar]
- Verma, S.; Chauhan, N.S. Indigenous medicinal plants knowledge of Kunihar forest division, district Solan. J. Tradit. Know. 2007, 6, 494–497. [Google Scholar]
- Kumar, G.; Kumar, V.; Thakur, K. Some overlooked Ethno-medicinal plants of district Bilaspur, Himachal Pradesh (India). CPUH Res. J. 2018, 3, 116–123. [Google Scholar]
- Singh, K.J.; Thakur, A.K. Medicinal plants of the Shimla hills, Himachal Pradesh: A survey. Int. J. Herbal Med. 2014, 2, 118–127. [Google Scholar]
- Chand, R.; Kaur, R.; Kaur, A.; Kumar, V.; Nirmala, C.; Singh, A.N. Assessment of ethnomedicinal plant diversity of Una and Hamirpur district of Himachal Pradesh, India: An ethno-ecological approach. Ann. Plant Sci. 2016, 5, 1475–1490. [Google Scholar] [CrossRef]
- Rani, S.; Rana, J.C. Ethnobotanical uses of some plants of Bhattiyat block in district Chamba, Himachal Pradesh (Western Himalaya). Ethnobot. Res. Appl. 2014, 12, 407–414. [Google Scholar] [CrossRef]
- Boktapa, N.R.; Sharma, A.K. Wild medicinal plants used by local communities of Manali, Himachal Pradesh, India. Ethnobot. Leaflets. 2010, 4, 259–267. [Google Scholar]
- Rana, M.S.; Samant, S.S. Diversity, indigenous uses and conservation status of medicinal plants in Manali wildlife sanctuary, North western Himalaya. J. Tradit. Know. 2011, 10, 439–459. [Google Scholar]
- Rahman, A.M. Ethno-medicinal practices for the treatment of asthma, diuretic, jaundice, piles, rheumatism and vomiting at the village Abdullahpur under Akkelpur Upazilla of Joypurhat district, Bangladesh. J. Eng. Appl. Sci. 2014, 1, 4–8. [Google Scholar]
- Amiri, M.S.; Jabbarzadeh, P.; Akhondi, M. An ethnobotanical survey of medicinal plants used by indigenous people in Zangelanlo district, Northeast Iran. J. Med. Plants Res. 2012, 6, 749–753. [Google Scholar]
- Edeoga, H.O.; Okwu, D.E.; Mbaebie, B.O. Phytochemical constituents of some Nigerian medicinal plants. Afr. J. Biotechnol. 2005, 4, 685–688. [Google Scholar] [CrossRef]
- Njoku, V.O.; Obi, C.; Onyema, O.M. Phytochemical constituents of some selected medicinal plants. Afr. J. Biotechnol. 2011, 10, 15020–15024. [Google Scholar] [CrossRef]
- Tewari, D.; Mocan, A.; Parvanov, E.D.; Sah, A.N.; Nabavi, S.M.; Huminiecki, L.; Atanasov, A.G. Ethnopharmacological approaches for therapy of jaundice: Part II. Highly used plant species from Acanthaceae, Euphorbiaceae, Asteraceae, Combretaceae, and Fabaceae families. Front. Pharmacol. 2017, 8, 519. [Google Scholar] [CrossRef] [PubMed]
- Nascimento, G.G.; Locatelli, J.; Freitas, P.C.; Silva, G.L. Antibacterial activity of plant extracts and phytochemicals on antibiotic-resistant bacteria. Braz. J. Microbiol. 2000, 31, 247–256. [Google Scholar] [CrossRef]
- Bhat, V.S.; Nasavatl, D.D.; Mardikar, B.R. Adhatoda vasica-an ayurvedic medicinal plant. Drug Dev. Res. 1978, 15, 6–10. [Google Scholar]
- Pathak, R.R. Therapeutic Guide to Ayurvedic Medicine: A Hand Book of Ayurvedic Medicine; Shree Baidyanath Ayurved Bhawan Pvt. Ltd.: Patna, India, 1970; pp. 25–330. [Google Scholar]
- Irshad, A.H.; Pervaiz, A.H.; Abrar, Y.B.; Fahelboum, I.; Awen, B.Z. Antibacterial activity of Berberis lycium root extract. Trakia J. Sci. 2013, 1, 88–90. [Google Scholar]
- Pei-Gen, X.I.A.O.; Feng-Peng, W.A.N.G.; Feng, G.A.O.; Lu-Ping, Y.A.N.; Dong-Lin, C.H.E.N.; Yong, L.I. A pharmacophylogenetic study of Aconitum L. (Ranunculaceae) from China. J. Syst. Evol. 2005, 44, 1–46. [Google Scholar]
- Ranjan, V.; Vats, M.; Gupta, N.; Sardana, S. Antidiabetic potential of whole plant of Adiantum capillus veneris linn. in streptozotocin induced diabetic rats. Int. J. Curr. Pharm. Res. 2014, 6, 341–347. [Google Scholar]
- Ruhil, S.; Balhara, M.; Dhankhar, S.; Chhillar, A.K. Aegle marmelos (Linn.) Correa: A potential source of phytomedicines. J. Med. Plant Res. 2011, 5, 1497–1507. [Google Scholar]
- Mathela, C.S.; Padalia, R.C.; Joshi, R.K. Variability in fragrance constituents of Himalayan Tanacetum species: Commercial potential. J. Essent. Oil Bear. Plants 2008, 5, 503–513. [Google Scholar] [CrossRef]
- Lakshmi, P.T.V.; Rajalakshmi, P. Identification of phyto components and its biological activities of aloe vera through the gas chromatography-mass spectrometry. J. Pharm. Res. 2011, 2, 247–249. [Google Scholar]
- Brahmachari, G.; Gorai, D.; Roy, R. Argemone mexicana: Chemical and pharmacological aspects. Rev. Bras. Farmacogn. 2013, 23, 559–567. [Google Scholar] [CrossRef]
- Mushtaq, S.; Aga, M.A.; Qazi, P.H.; Ali, M.N.; Shah, A.M.; Lone, S.; Hussain, A.; Shah, Z.H. Isolation, characterization and HPLC quantification of compounds from Aquilegia fragrans Benth: Their in vitro antibacterial activities against bovine mastitis pathogens. J. Ethnopharmacol. 2016, 178, 9–12. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, D.; Kaushik, N. Phytochemical and pharmacological studies in genus Berberis. Phytochem Rev. 2012, 11, 523–542. [Google Scholar] [CrossRef]
- Joshi, H.; Saxena, G.K.; Singh, V.; Arya, E.; Singh, R.P. Phytochemical investigation, isolation and characterization of betulin from bark of Betula utilis. Res. J. Pharmacogn. Phytochem. 2013, 2, 145–151. [Google Scholar]
- Mishra, S.; Aeri, V.; Gaur, P.K.; Jachak, S.M. Phytochemical, therapeutic, and ethnopharmacological overview for a traditionally important herb: Boerhavia diffusa Linn. Biomed. Res. Int. 2014, 2014, 808302. [Google Scholar] [CrossRef]
- Karthikairaj, K.; Sevarkodiyone, S.P.; Pavaraj, M.; Balaji, S.; Senthikumar, P.; Kalaivani, A. Effect of organic amendments on the level of chemical constituents of redgram, Cajanus cajan infected with root-knot nematode, Meloidogyne javanica. Middle East J. Sci. Res. 2012, 12, 1068–1071. [Google Scholar]
- Al-Snafi, A.E. The chemical constituents and pharmacological effects of Capsella bursa-pastoris-a review. Int. J. Pharmacol. Toxicol. 2015, 5, 76–81. [Google Scholar]
- Sunil, P.; Sanjay, Y.; Vinod, S. Pharmacognostical investigation and standardization of capsicum annum L. roots. Int. J. Pharmacogn. 2012, 4, 21–24. [Google Scholar]
- Alorkpa, E.J.; Boadi, N.O.; Badu, M.; Saah, S.A. Phytochemical screening, antimicrobial and antioxidant properties of assorted Carica papaya leaves in Ghana. J. Med. Plant Res. 2016, 4, 193–198. [Google Scholar]
- Izhar, S.; Ahmed, D. Carissa opaca: A plant with great potential for future drugs for degenerative and infectious diseases. Chem. Select. 2016, 1, 3005–3011. [Google Scholar] [CrossRef]
- Sekar, T.; Arumugam, T.; Ayyanar, M.; Pillai, J. Phytochemical screening and antibacterial activity of leaf and callus extracts of Centella asiatica. Bangladesh J. Pharmacol. 2011, 6, 55–60. [Google Scholar] [CrossRef]
- Arora, M.; Singh, S.; Kaur, P. Pharmacognostic and phytochemical evaluation of selected seeds of ‘Cicer arietinum’Linn. seeds from Roopnagar, Punab. Int. J. Pharm. Sci. Invent. 2013, 2, 18–29. [Google Scholar]
- Prajapati, R.; Kalariya, M.; Umbarkar, R.; Parmar, S.; Sheth, N. Colocasia esculenta: A potent indigenous plant. Int. J. Nutr. Pharmacol. 2011, 1, 90–96. [Google Scholar] [CrossRef]
- Xiao, L.G.; Zhang, S.C.; Zhang, Y.; Liu, L.; Zhang, H.L.; Yu, Q.; An, L.K. Sesquiterpenoids from the aerial parts of Conyza japonica and their inhibitory activity against the nitric oxide production. Fitoterapia 2020, 142, 104473. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, W.; Zhang, J.; Chen, S.; Lin, X. Chemical constituents of Crepis flexuosa. Chin. J. Appl. Environ. Biol. 2011, 4, 509–511. [Google Scholar]
- Ahmad, A.; Tandon, S.; Xuan, T.D.; Nooreen, Z. A review on phytoconstituents and biological activities of Cuscuta species. Biomed. Pharmacother. 2017, 92, 772–795. [Google Scholar] [CrossRef]
- Das, J.; Chowdhury, A.; Biswas, K.; Karmakar, K.U.; Sharif, R.S.; Raihan, Z.S.; Muhit, A.M. Cytotoxicity and antifungal activities of ethanolic and chloroform extracts of Cucumis sativus Linn (Cucurbitaceae) leaves and stems. Res. J. Phytochem. 2012, 6, 25–30. [Google Scholar]
- Soni, P.; Siddiqui, A.A.; Dwivedi, J.; Soni, V. Pharmacological properties of Datura stramonium L. as a potential medicinal tree: An overview. Asian Pac. J. Trop. Biomed. 2012, 2, 1002–1008. [Google Scholar] [CrossRef]
- Variya, C.B.; Bakrania, K.; Patel, S.S. Emblica officinalis (Amla): A review for its phytochemistry, ethnomedicinal uses and medicinal potentials with respect to molecular mechanisms. Pharmacol. Res. 2016, 1, 180–200. [Google Scholar] [CrossRef]
- Li, E.T.; Liu, K.H.; Zang, M.H.; Zhang, X.L.; Jiang, H.Q.; Zhou, H.L.; Wu, Y. Chemical constituents from Euphorbia hirta. Biochem. Syst. Ecol. 2015, 62, 204–207. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Mizuguchi, R.; Yamada, Y. Chemical constituents of cultured cells of Euphorbia tirucalli and E. millii. Plant Cell Rep. 1981, 1, 29–30. [Google Scholar] [CrossRef] [PubMed]
- Al-Snafi, A.E. The pharmacology of Equisetum arvense-A review. IOSR J. Pharm. 2017, 7, 31–42. [Google Scholar] [CrossRef]
- Lalsare, S.; Verma, P.K.; Khatak, M.; Ranjan, S.; Rajurakar, S.; Gurav, S.S. Anti-inflammatory and antimicrobial activity of Flacourtia ramontchi leaves. Drug Dev. Res. 2011, 3, 308–313. [Google Scholar]
- Ismail, M.; Hussain, J.; Khan, A.U.; Khan, A.L.; Ali, L.; Khan, F.U.; Khan, Z.A.; Niaz, U.; Lee, I.J. Antibacterial, antifungal, cytotoxic, phytotoxic, insecticidal, and enzyme inhibitory activities of Geranium wallichianum. Evid. Based Complement. Altern. Med. 2012, 2012, 305906. [Google Scholar] [CrossRef] [PubMed]
- Shabir, M.; Agnihotri, P.; Husain, D.; Tiwari, J.K.; Husain, T. On the current status of the genus Gentiana L. (Gentianaceae) in India. Pleione 2017, 11, 16–24. [Google Scholar]
- Painuly, P.; Varma, N.; Tandon, J.S. Constituents of Geum elatum. J. Nat. Prod. 1984, 47, 181–189. [Google Scholar] [CrossRef]
- Al-Snafi, A.E. Pharmacological and therapeutic activities of Hedera helix-a review. IOSR J. Pharm. 2018, 8, 41–53. [Google Scholar]
- Kapoor, D.N.; Singh, G.; Kaur, T. A review on pharmacognostic, phytochemical and pharmacological data of various species of Hippophae (Sea buckthorn). Int. J. Green Pharm. 2017, 11, S62–S74. [Google Scholar]
- Shah, P.A.; Parmar, M.Y.; Thakkar, V.T.; Gandhi, T.R. Hepatoprotective activity of Hordeum vulgare Linn. seeds against ethanol-induced liver damage in rats. Int. J. Green Pharm. 2009, 3, 38–43. [Google Scholar]
- Singh, T.; Singh, O.; Singh, H. Adhatoda vasica Nees: Phytochemical and pharmacological profile. J. Nat. Prod. 2011, 1, 29–39. [Google Scholar] [CrossRef]
- Naidu, J.R.; Ismail, R.B.; Yeng, C.; Sasidharan, S.; Kumar, P. Chemical composition and antioxidant activity of the crude methanolic extracts of Mentha spicata. J. Phytol. 2012, 4, 13–18. [Google Scholar]
- Yang, Y.; Yang, X.; Xu, B.; Zeng, G.; Tan, J.; He, X.; Zhou, Y. Chemical constituents of Morus alba L. and their inhibitory effect on 3T3-L1 preadipocyte proliferation and differentiation. Fitoterapia 2014, 98, 222–227. [Google Scholar] [CrossRef] [PubMed]
- Mallhi, T.H.; Qadir, M.I.; Khan, Y.H. Determination of phytoconstituents of n-hexane extract of leaves of Morus nigra and evaluation of their effects on biochemical and histopathological parameters in paracetamol intoxicated mice liver. Braz. J. Pharm. Sci. 2018, 54, 1–9. [Google Scholar] [CrossRef]
- Dev, L.R.; Ranjeeta, P.; Anurag, M.; Rajiv, G. Pharmacognostic and phytochemical studies of bark of Oroxylum indicum. Rev. Bras. Farmacogn. 2010, 2, 297–303. [Google Scholar] [CrossRef]
- Sahidin, I.; Bahrun, A.; Taufik, M.; Mahatva Yodha, A.W.; Sabandar, C.W.; Imran, I.; Kadidae, L.O.; Diantini, A. Chemical constituents of Persicaria sagittata (L.) H. gross: Antioxidant activity and chemotaxonomy significance. Jundishapur J. Nat. Pharm. Prod. 2019, 15, 1–8. [Google Scholar] [CrossRef]
- Samali, A.; Florence, D.T.; Odeniran, O.A.; Cordelia, O.N. Evaluation of chemical constituents of Phyllanthus niruri. Afr. J. Pharm. Pharmacol. 2012, 6, 125–128. [Google Scholar] [CrossRef]
- Rathee, D.; Rathee, P.; Rathee, S.; Rathee, D. Phytochemical screening and antimicrobial activity of Picrorrhiza kurroa, an Indian traditional plant used to treat chronic diarrhoea. Arab. J. Chem. 2016, 9, 1307–1313. [Google Scholar] [CrossRef]
- Shiojima, K.; Masuda, K.; Ooishi, Y.; Suzuki, H.; Ageta, H. Composite constituents: New migrated gammacerane triterpenoids from roots of Picris hieracioides subsp. japonica. Tetrahedron Lett. 1989, 30, 6873–6874. [Google Scholar] [CrossRef]
- Bibi, Y.; Zia, M.; Qayyum, A. An overview of Pistacia integerrima a medicinal plant species: Ethnobotany, biological activities and phytochemistry. Pak. J. Pharm. Sci. 2015, 28, 1009–1013. [Google Scholar]
- Malik, M.A.; Srivastava, P.; Ahmad, S.B. Quantitative estimation of phytochemicals and antimicrobial activity of Podophyllum hexandrum. Int. J. Curr. Sci. 2018, 6, 1152–1155. [Google Scholar]
- Moorthy, K.; Punitha, T.; Vinodhini, R.; Thippan, B.; Sureshkumar, B.T.; Vijayalakshmi, P.; Thajuddin, N. Antimicrobial activity and qualitative phytochemical analysis of Punica granatum Linn. (Pericarp). J. Med. Plants Res. 2013, 7, 474–479. [Google Scholar]
- Poonam, V.; Raunak; Kumar, G.; Reddy, L.C.S.; Jain, R.; Sharma, S.; Prasad, A.K.; Parmar, V. Chemical constituents of the genus Prunus and their medicinal properties. Curr. Med. Chem. 2011, 18, 3758–3824. [Google Scholar] [CrossRef] [PubMed]
- Janjua, S.; Shahid, M. Phytochemical analysis and in vitro antibacterial activity of root peel extract of Raphanus sativus L. var niger. Adv. Med. Plant Res. 2013, 1, 1–7. [Google Scholar]
- Ribeiro, P.R.; de Castro, R.D.; Fernandez, L.G. Chemical constituents of the oilseed crop Ricinus communis and their pharmacological activities: A review. Ind Crops Prod. 2016, 91, 358–376. [Google Scholar] [CrossRef]
- Raj, J.; Basant, B.; Murugan, P.M.; da Silva, J.A.T.; Saurav, K.; Chaurasia, O.P.; Singh, S.B. Screening phytochemical constituents of 21 medicinal plants of trans-Himalayan region. Med. Aromat. Plant Sci. Biotechnol. 2010, 4, 90–93. [Google Scholar]
- Verma, A.; Kumar, B.; Alam, P.; Singh, V.; Gupta, S.K. Rubia cordifolia—A review on pharmaconosy and phytochemistry. Int. J. Pharm. Sci. Res. 2016, 7, 2720–2731. [Google Scholar]
- Singh, A.; Lal, R.U.; Mukhtar, H.; Singh, P.S.; Shah, G.; Dhawan, R.K. Phytochemical profile of sugarcane and its potential health aspects. Phcog. Rev. 2015, 9, 45–53. [Google Scholar] [CrossRef]
- Kumar, P.S.; Patel, J.S.; Saraf, M.N. Mechanism of vasorelaxant activity of a fraction of root extract of Sesamum indicum Linn. Indian J. Exp. Biol. 2008, 46, 457–464. [Google Scholar]
- Chauhan, R.; Ruby, K.M.; Shori, A.; Dwivedi, J. Solanum nigrum with dynamic therapeutic role: A review. Int. J. Pharm. Sci. Res. 2012, 15, 65–71. [Google Scholar]
- Ahmed, M.M.; Andleeb, S.; Saqib, F.; Hussain, M.; Khatun, M.N.; Ahmad, B.; Rahman, H. Diuretic and serum electrolyte regulation potential of aqueous methanolic extract of Solanum surattense fruit validates its folkloric use in dysuria. BMC Complement. Altern. Med. 2016, 16, 2–8. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Khanzada, S.K.; Shaikh, W.; Sofia, S.; Kazi, T.G.; Usmanghani, K.; Kabir, A.; Sheerazi, T.H. Chemical constituents of Tamarindus indica L. medicinal plant in Sindh. Pak. J. Bot. 2008, 40, 2553–2559. [Google Scholar]
- Kumar, N.; Khurana, S.M. Phytochemistry and medicinal potential of the Terminalia bellirica Roxb. (Bahera). J. Nat. Prod. Res. 2018, 9, 97–107. [Google Scholar]
- Upadhyay, A.; Agrahari, P.; Singh, D.K. A review on the pharmacological aspects of Terminalia chebula. Int. J. Pharmacol. 2014, 10, 289–298. [Google Scholar] [CrossRef]
- Sharma, N.; Kumar, V.; Chopra, M.P.; Sourirajan, A.; Dev, K. Thalictrum foliolosum: A lesser unexplored medicinal herb from the himalayan region as a source of valuable phytocompounds. J. Ethnopharmacol. 2020, 255, 112736. [Google Scholar] [CrossRef] [PubMed]
- Shervani, Z.A.; Mishra, P.K. Phytochemical study of Tinospora cordifolia grown on three different soil conditions. Res. J. Life Sci. Bioinform. Pharm. Chem. Sci. 2017, 5, 810–815. [Google Scholar]
- Vigh, S.; Cziaky, Z.; Sinka, L.T.; Pribac, C.; Moş, L.; Turcuş, V.; Remenyik, J.; Mathe, E. Analysis of phytoconstituent profile of fenugreek–Trigonella foenuem-graecum L.-seed extracts. Studia Univ. Babes-Bolyai Chem. 2017, 62, 145–166. [Google Scholar] [CrossRef]
- Akbar, S. Vernonia anthelmintica Willd. (Asteraceae/Compositae). In Handbook of 200 Medicinal Plants; Akbar, S., Ed.; Springer: Cham, Switzerland, 2020; pp. 1895–1899. [Google Scholar]
- Chandra, D.; Kohli, G.; Prasad, K.; Bisht, G.; Punetha, V.D.; Khetwal, K.S.; Devrani, M.K.; Pandey, H.K. Phytochemical and ethnomedicinal uses of family Violaceae. Curr. Res. Chem. 2015, 7, 44–52. [Google Scholar] [CrossRef]
- Chandra, D.; Kohli, G.; Prasad, K.; Bisht, G.; Punetha, V.D.; Pandey, H.K. Chemical composition of the essential oil of Viola serpens from Bageshwar (Shama), Uttarakhad, India. J. Med. Plant Res. 2017, 11, 513–517. [Google Scholar]
- Das, P.K.; Goswami, S.; Chinniah, A.; Panda, N.; Banerjee, S.; Sahu, N.P.; Achari, B. Woodfordia fruticosa: Traditional uses and recent findings. J. Ethnopharmacol. 2007, 110, 189–199. [Google Scholar] [CrossRef]
- Dhalaria, R.; Verma, R.; Kumar, D.; Puri, S.; Tapwal, A.; Kumar, V.; Nepovimova, E.; Kuca, K. Bioactive compounds of edible fruits with their anti-aging properties: A comprehensive review to prolong human life. Antioxidants 2020, 9, 1123. [Google Scholar] [CrossRef] [PubMed]
- Shantabi, L.; Jagetia, G.C.; Vabeiryureilai, M.; Lalrinzuali, K. Phytochemical screening of certain medicinal plants of Mizoram, India and their folklore use. J. Biodivers. Biopros. Dev. 2014, 1, 1–9. [Google Scholar]
- Bodakhe, S.H.; Ram, A. Hepatoprotective properties of Bauhinia variegata bark extract. Yakugaku zasshi. Farumashia 2007, 127, 1503–1507. [Google Scholar]
- Dehar, N.A.; Walia, R.A.; Verma, R.B.; Pandey, P.I. Hepatoprotective activity of Berberis aristata root extract against chemical induced acute hepatotoxicity in rats. Asian J. Pharm. Clin. Res. 2013, 6, 53–56. [Google Scholar]
- Bera, T.K.; Chatterjee, K.; De, D.; Ali, K.M.; Jana, K.; Maiti, S.; Ghosh, D. Hepatoprotective activity of livshis, a polyherbal formulation in CCl4-Induced hepatotoxic male Wistar rats: A toxicity screening approach. Biosciences 2011, 3, 103–110. [Google Scholar] [CrossRef]
- Setty, S.R.; Quereshi, A.A.; Swamy, A.V.; Patil, T.; Prakash, T.; Prabhu, K.; Gouda, A.V. Hepatoprotective activity of Calotropis procera flowers against paracetamol-induced hepatic injury in rats. Fitoterapia 2007, 78, 451–455. [Google Scholar] [CrossRef]
- Aghel, N.; Rashidi, I.; Mombeini, A. Hepatoprotective activity of Capparis spinosa root bark against CCl4 induced hepatic damage in mice. Iran J. Pharm. Res. 2007, 6, 285–290. [Google Scholar]
- Adeneye, A.A.; Olagunju, J.A.; Banjo, A.A.; Abdul, S.F.; Sanusi, O.A.; Sanni, O.O.; Osarodion, B.A.; Shonoiki, O.E. The aqueous seed extract of Carica papaya Linn. prevents carbon tetrachloride induced hepatotoxicity in rats. Int. J. Appl. Res. Nat. Prod. 2009, 2, 19–32. [Google Scholar]
- Sahreen, S.; Khan, M.R.; Khan, R.A. Hepatoprotective effects of methanol extract of Carissa opaca leaves on CCl4-induced damage in rat. BMC Comp. Alt. Med. 2011, 11, 48. [Google Scholar] [CrossRef]
- Tuse, T.A.; Harle, U.N.; Bore, V.V. Hepatoprotective activity of Colocasia antiquorum against experimentally induced liver injury in rats. Malays. J. Pharma. Sci. 2009, 7, 99–112. [Google Scholar]
- Dubey, S.; Mehta, S. Hepatoprotective activity of Euphorbia hirta Linn. Plant against carbon tetrachloride-induced hepatic injury in rats. Food Bio. Med. Sci. 2014, 1, 108–111. [Google Scholar]
- Jyothi, T.M.; Shankariah, M.M.; Prabhu, K.; Lakshminarasu, S.; Srinivasa, G.M.; Ramachandra, S.S. Hepatoprotective and antioxidant activity of Euphorbia tirucalli. Iran. J. Pharma. Ther. 2008, 7, 25–30. [Google Scholar]
- Adil, A.; Khan, S.S.; Naeem, S.; Ikram, R.; Jaffer, N. Evaluation of acute and chronic hepatoprotective activity of Hedera helix L. Aqueous leaf extract on carbon tetrachloride-induced hepatotoxicity in rat model. Pak. J. Phytopathol. 2017, 34, 1–15. [Google Scholar]
- Afzal, U.; Gulfraz, M.; Hussain, S.; Malik, F.; Maqsood, S.; Shah, I.; Mahmood, S. Hepatoprotective effects of Justicia adhatoda L. against carbon tetrachloride (CCl4) induced liver injury in Swiss albino mice. Afr. J. Pharm. Pharmacogn. 2013, 7, 8–14. [Google Scholar] [CrossRef]
- Hogade, M.G.; Patil, K.S.; Wadkar, G.H.; Mathapati, S.S.; Dhumal, P.B. Hepatoprotective activity of Morus alba (Linn.) leaves extract against carbon tetrachloride induced hepatotoxicity in rats. Afr. J. Pharma. Pharmacogn. 2010, 4, 731–734. [Google Scholar]
- Mallhi, T.H.; Qadir, M.I.; Khan, Y.H.; Ali, M. Hepatoprotective activity of aqueous methanolic extract of Morus nigra against paracetamol-induced hepatotoxicity in mice. Bangladesh J. Pharmacol. 2014, 9, 60–66. [Google Scholar] [CrossRef]
- Tenpe, C.R.; Upaganlawar, A.; Burle, S.; Yeole, Y.G. In vitro antioxidant and preliminary hepatoprotective activity of Oroxylum indicum Vent leaf extracts. Pharmacologyonline 2009, 1, 35–43. [Google Scholar]
- Harish, R.; Shivanandappa, T. Antioxidant activity and hepatoprotective potential of Phyllanthus niruri. Food Chem. 2006, 95, 180–185. [Google Scholar] [CrossRef]
- Ganie, S.A.; Zargar, B.A.; Masood, A.; Zargar, M.A. Hepatoprotective and antioxidant activity of rhizome of Podophyllum hexandrum against carbon tetra chloride induced hepatotoxicity in rats. Biomedicine 2013, 26, 209–221. [Google Scholar] [CrossRef]
- Soni, M.; Tanwar, Y.S. Hepatoprotective activity of Prunus domestica fruit extract against paracetamol-induced liver damage in Albino rats. J. Crit. Rev. 2020, 7, 4729–4734. [Google Scholar]
- Kumar, M.; Dandapat, S.; Sinha, M.P. Hepatoprotective activity of Punica granatum leaf extract against carbon tetrachloride induced hepatotoxicity in rats. Balneo 2018, 9, 24–27. [Google Scholar] [CrossRef]
- Visen, P.S.; Shukla, B.; Patnaik, G.K.; Tripathi, S.C.; Kulshreshtha, D.K.; Srimal, R.C.; Dhawan, B.N. Hepatoprotective activity of Ricinus communis leaves. Int. J. Pharmacogn. 1992, 30, 241–250. [Google Scholar] [CrossRef]
- Elhag, R.A.M.; El Badwi, S.M.A.; Bakhiet, A.O.; Galal, M. Hepatoprotective activity of Solanum nigrum extracts on chemically induced liver damage in rats. J. Vet. Med. Anim. Health 2011, 3, 45–50. [Google Scholar]
- Meena, S.Z.; Rahman, M.A.; Bagga, P.; Mujahid, M. Hepatoprotective activity of Tamarindus indica Linn stem bark ethanolic extract against hepatic damage induced by co-administration of antitubercular drugs isoniazid and rifampicin in Sprague Dawley rats. J. Basic Clin. Physiol. Pharmacol. 2018, 30, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Marslin, G.; Prakash, J. Hepatoprotective Activity of Thalictrum foliolosum (Ranunculaceae) Root Ethanolic extract. Int. J. Life Sci. Pharma. Res. 2020, 10, 8–11. [Google Scholar]
- Kanter, M.; Coskun, O.; Budancamanak, M. Hepatoprotective effects of Nigella sativa L and Urtica dioica L on lipid peroxidation, antioxidant enzyme systems and liver enzymes in carbon tetrachloride-treated rats. World J. Gastroenterol. 2005, 11, 6684–6688. [Google Scholar] [CrossRef]
- Kumar, H.; Bhardwaj, K.; Dhanjal, D.S.; Nepovimova, E.; Șen, F.; Regassa, H.; Singh, R.; Verma, R.; Kumar, V.; Kumar, D.; et al. Fruit extract mediated green synthesis of metallic nanoparticles: A new avenue in pomology applications. Int. J. Mol. Sci. 2020, 21, 8458. [Google Scholar] [CrossRef]
- Kumar, H.; Bhardwaj, K.; Kuca, K.; Kalia, A.; Nepovimova, E.; Verma, R.; Kumar, D. Flower-Based green synthesis of metallic nanoparticles: Applications beyond fragrance. Nanomaterials 2020, 10, 766. [Google Scholar] [CrossRef]
| Botanical Name | Local Name | Family | Region | Data Source |
|---|---|---|---|---|
| Justicia adhatoda L. | Basuti, Arusha | Acanthaceae | Kangra, Hamirpur | [23,24] |
| Adiantum capillus-veneris L. | Dooman tuli | Adiantaceae | Hamirpur | [24] |
| Pistacia integerrima J. L. Stewart ex Brandis | Kakar Singhi | Anacardiaceae | Hamirpur | [24] |
| Carissa opaca Stapf. ex Haines. | Karaunda, Garnu | Apocynaceae | Kangra, Mandi, and Una | [25] |
| Calotropis procera (Aiton) Dryand. | Aak | Apocynaceae | Kangra and Chamba | [26] |
| Colocasia antiquorum Schott | Ghandiale, Arbi | Araceae | Kangra | [23] |
| Colocasia esculanata (L.) Schott | Arbi kuchawari | Araceae | Hamirpur | [24] |
| Hedera helix L. | Kannauri (Bail) | Araliaceae | Shimla | [27] |
| Ajania tibetica (Hook.f. and Thomson) Tzvelev | Tibetan Tansy | Asteraceae | Lahul and Spiti | [28] |
| Crepis flexuosa (Ledeb.) Benth. ex C.B. Clarke | Homa-silli | Asteraceae | Lahul and Spiti | [28] |
| Conyza japonica (Thunb.) Less. ex Less. | Gaadi | Asteraceae | Kullu (Solang Valley) | [29] |
| Picris hieracioides subsp. japonica (Thunb.) Hand-Mazz. | Cherakpa | Asteraceae | Lahul and Spiti | [30] |
| Scorzonera divaricata Turcz. | Himalayan viper grass | Asteraceae | Lahul and Spiti | [31] |
| Taraxacum officinale (L.) Weber ex F.H. Wigg. | Dudhli, Dulal | Asteraceae | Kangra, Hamirpur | [23,24] |
| Youngia tenuifolia (Willd.) Babc. and Stebbins | Seertik | Asteraceae | Lahul and Spiti | [28] |
| Vernonia anthelmintica (L.) Willd. | Kaliziri | Asteraceae | Shimla | [27] |
| Berberis aristata DC. | Kashmal, Chunchari | Berberidaceae | Kangra, Mandi, Shimla, Chamba | [23,27,32,33] |
| Berberis ceratophylla G. Don | Kashmal | Berberidaceae | Kangra | [34] |
| Berberis lycium Royle | Kashmal, Dauhaldi | Berberidaceae | Kangra, Hamirpur, Shimla, Chamba, and Sirmour | [23,24,27,32,35] |
| Berberis chitria Buch. -Ham. ex. Lindl. | Kashmal | Berberidaceae | Shimla | [27] |
| Betula utilis D. Don | Bhojpatra | Betulaceae | Kangra and Chamba | [23,32] |
| Capsella bursa-pastoris (L.) Medik. | Jangli sarson | Brassicaceae | Kullu (Solang Valley) | [29] |
| Raphanus sativus L. | Muli | Brassicaceae | Kangra, Hamirpur | [23,24] |
| Oroxylum indicum (L.) Kurz | Tatpalnga | Bignoniaceae | Kangra | [23] |
| Cassia fistula L. | Kyar, Alsi ki tat | Caesalpinaceae | Kangra | [34] |
| Tamarindus indica L. | Imli | Caesalpinaceae | Hamirpur | [24] |
| Capparis spinosa L. | Kabra findus rose | Capparaceae | Lahul and Spiti | [31] |
| Carica papaya L. | Pump, Papita | Caricaceae | Kangra | [23] |
| Terminalia bellirica (Gaertn.) Roxb. | Behada | Combretaceae | Solan, Kangra | [23,36] |
| Terminalia chebula Retz. | Harad | Combretaceae | Kangra, Solan (Kunihar forest divison), and Shimla | [23,27,36] |
| Cuscuta reflexa Roxb. | Akash bel | Convalvulaceae | Hamirpur, Bilaspur, Solan (Kunihar forest divison) | [24,27,37] |
| Cucumis sativus L. | Kheera | Cucurbitaceae | Hamirpur | [24] |
| Elaeagnus rhamnoides (L.) A. Nelson | Sea-buck thorn | Elaegnaceae | Lahul and Spiti | [31] |
| Hippophae tibetana Schltdl. | Star bn | Elaegnaceae | Lahul and Spiti | [31] |
| Emblica officinalis Gaertn. | Amla, Amlika | Euphorbiaceae | Kangra, Shimla, Bilaspur, Chamba, Hamirpur, Sirmour, Solan, Una | [23,24,25,32,37,38] |
| Euphorbia hirta L. | Doodhli | Euphorbiaceae | Hamirpur | [24] |
| Euphorbia tirucalli L. | Tohar | Euphorbiaceae | Una, Hamirpur | [39] |
| Mallotus philippinensis Mull.Arg. | Kamla tree, Kumkum | Euphorbiaceae | Kunihar forest division, Solan | [36] |
| Phyllanthus fraternus G.L. Webster | Chota amla, Bhoomi ambla | Euphorbiaceae | Kangra | [23] |
| Ricinus communis L. | Erand | Euphorbiaceae | Kangra | [23] |
| Equisetum arvense L. | Girthan | Equisetaceae | Kangra | [23] |
| Bauhinia variegata L. | Karale, Kachnar | Fabaceae | Kangra | [23] |
| Cajanus cajan (L.) Millsp. | Arhar, Pigeonpea | Fabaceae | Chamba | [40] |
| Cicer microphyllum Benth. | Chana, Cowpea | Fabaceae | Lahul and Spiti | [28] |
| Trigonella emodi Benth. | Methi, Fenugreek | Fabaceae | Lahul and Spiti | [31] |
| Flacourtia ramontchi L. | Governor’s plum, Bilangra | Flacourtiaceae | Bilaspur, Chamba, Kangra, Hamirpur, Sirmour (Nahan), Solan, Una | [25] |
| Gentiana kurrroo Royle | Kanauri (Bail) | Gentianaceae | Shimla | [27] |
| Gentiana tubiflora (G. Don) Griseb. | Tikta anupo Mensa | Gentianaceae | Lahul and Spiti | [30] |
| Gentiana leucomelaena Maxim. | Buksuk shipo | Gentianaceae | Lahul and Spiti | [30] |
| Gentianopsis detonsa (Rottb.) Ma | Chateek | Gentianaceae | Lahul and Spiti | [30,31] |
| Gentianopsis paludosa (Hook.f.) Ma | Gyatheek | Gentianaceae | Lahul and Spiti | [30,31] |
| Gentianella moorcroftiana (Wall. ex Griseb.) | Airy shaw | Gentianaceae | Lahul and Spiti | [31] |
| Geranium nepalense Sw. | Tirahi | Geraniaceae | Manali | [26] |
| Mentha spicata L. | Pudina | Lamiaceae | Kangra | [23] |
| Aloe vera (L.) Burm.f. | Kware, Ghritkumar, Gavrapatha | Liliaceae | Kangra, Shimla | [23,38] |
| Asparagus adscendens Roxb. | Sanspan | Liliaceae | Kangra | [23] |
| Woodfordia fruticosa (L.) Kurz | Dhoaien, Dhai | Lythraceae | Kangra, Hamirpur | [23,24] |
| Tinospora cordifolia (Willd.) Miers | Giloe, Giloen, Guljae | Menispermaceae | Kangra, Chamba | [23,32] |
| Morus alba L. | Chitta toot | Moraceae | Hamirpur, Bilaspur | [24,37] |
| Morus nigra L. | Kala toot | Moraceae | Hamirpur | [24] |
| Leucas cephalotes (Roth) Spreng. | Mal bhedu | Lamiaceae | Kangra | [23] |
| Boerhavia diffusa L. | Punarnava | Nyctaginaceae | Una and Hamirpur | [39] |
| Argemone mexicana L. | Kantili, Pili Kantili, Bharbhand | Papaveraceae | Kangra, Hamirpur, and Sirmour | [23,24,35] |
| Sesamum indicum L. | Til | Pedaliaceae | Hamirpur | [24] |
| Polygonum tortuosum D. Don | Agel davaj | Polygonaceae | Lahul and Spiti | [30] |
| Persicaria amplexicaulis (D. Don) Ronse Decr. | Amli/kutrya | Polygonaceae | Chamba and Kangra | [26] |
| Hordeum vulgare L. | Jou, Joui | Poaceae | Hamirpur, Bilaspur | [24,37] |
| Saccharum officinarum L. | Ganna, Kamandi | Poaceae | Kangra, Bilaspur, and Hamirpur | [23,24,37] |
| Podophyllum hexandrum Royle | Bankakdi | Podophyllaceae | Manali | [41] |
| Punica granatum L. | Daran | Punicaceae | Hamirpur | [24] |
| Aquilegia fragrans Benth. | Zadul | Rannunculaceae | Kangra | [34] |
| Aconitum rotundifolium Kar. and Kir. | Atish, Patish | Rannunculaceae | Lahul and Spiti | [31] |
| Thalictrum foliolosum DC. | Pili jari, Chabra | Rannunculaceae | Shimla | [27] |
| Geum elatum Wall. ex G. Don | Gyampar mendok, Turu silva Mensa | Rosaceae | Lahul and Spiti | [30] |
| Prunus domestica L. | alubhukhara, Palam | Rosaceae | Hamirpur | [24] |
| Rosa webbiana Wall. ex Royle | Seba, Webb’s rose | Rosaceae | Lahul and Spiti | [31] |
| Rubia manjith Roxb. ex Fleming | Jamithi, Manjit | Rubiaceae | Manali | [42] |
| Aegle marmelos (L.) Correa | Bil, Bil patri | Rutaceae | Hamirpur, Bilaspur | [24,37] |
| Saxifraga flagellaris Willd. | Spider plant | Saxifragaceae | Lahul and Spiti | [28] |
| Picrorhiza kurroa Royl ex. Benth. | Karru, Kutki | Scrophulariaceae | Hamirpur, Manali, Chamba | [24,32,41] |
| Capsicum annum L. | Mircha, Pippali | Solanaceae | Kangra | [23] |
| Datura stramonium L. | Dhatura | Solanaceae | Manali | [42] |
| Solanum nigrum L. | Choote tamatter, Makoi | Solanaceae | Kangra, Hamirpur | [23,24] |
| Solanum surattense Burm. f. | Kantkari | Solanaceae | Hamirpur | [24] |
| Centella asiatica (L.) Urb. | Brahmi, Minki | Umblellifereae | Kangra | [23] |
| Urtica dioica L. | Bichu butti | Urticaceae | Manali | [42] |
| Viola serpens Wall. ex Ging. | Bhanaksha | Violaceae | Hamirpur | [24] |
| Botanical Name | Plant Part Used | Mode of Use | Reference |
|---|---|---|---|
| Aconitum rotundifolium | Whole plant | Plant juice is taken orally along with an equal volume of water for five to seven days to cure jaundice. | [31] |
| Adiantum capillus | Leaves | A decoction of fresh leaves is taken two times for seven days to cure jaundice. | [24] |
| Aegle marmelos | Leaves and fruit | A decoction of leaves and unripe fruit is used to treat jaundice. | [24] |
| Ajania tibetica | Leaves and flower | Leaves and flowers are used to cure jaundice. | [28] |
| Aloe vera | Fleshy leaves | The pulp of the leaves is directly consumed by the patient for two weeks to cure jaundice. | [23] |
| Argemone mexicana | Whole plant | Yellow sap of the plant is used to treat jaundice. | [24] |
| Asparagus adscendens | Roots | A decoction of roots (10–15 mL) is given for eight to ten days to cure jaundice. | [23] |
| Aquilegia fragrans | Seeds | A decoction of seeds is used to treat jaundice. | [34] |
| Bauhinia variegata | Leaves | Leaves juice is taken for seven days for the treatment of jaundice. | [23] |
| Berberis aristata | New leaves (twigs) and roots | New leaves are directly consumed, and a decoction of ground roots (100 mL) is taken to cure jaundice. | [23] |
| Berberis ceratophylla | Roots | Fresh roots are cut into small pieces and further shade-dried to make pills. These pills are consumed with “Kujja-Mishri” with water to cure jaundice. | [34] |
| Berberis chitria | Roots | A decoction of roots is used to treat jaundice. | [27] |
| Berberis lycium | Roots | A decoction of roots (80–100 mL) is given to cure jaundice. | [23] |
| Betula utilis | Papery bark | A decoction of the bark is given to the patient for ten to twelve days to cure jaundice. | [23] |
| Boerhavia diffusa | Whole plant | The whole plant is used to cure jaundice. | [39] |
| Cajanus cajan | Leaves | Leaf juice or leaf decoction is given with sugar (regularly in the morning) for about one month to cure jaundice. | [40] |
| Calotropis procera | Flowers | Flowers and betel leaf are taken with honey to treat jaundice. | [26] |
| Capparis spinosa | Shoot | Stem powder is taken with water at least for five to six days. | [31] |
| Capsella bursa-pastoris | Stem | The stem is used for the treatment of jaundice. | [29] |
| Capsicum annuum | Leaves | Boiled leaves are used as a vegetable (saag) and given for two to three days to cure jaundice. | [23] |
| Carica papaya | Raw fruit | Boiled vegetable of raw fruit is given to the patient to cure jaundice. | [23] |
| Carissa opaca | Roots | Roots are used for the treatment of jaundice. | [25] |
| Cassia fistula | Seeds | A decoction of seeds is consumed empty stomach for a week, daily in the morning. | [34] |
| Centella asiatica | Whole plant (entire herb) | The dried herb is crushed with kali mirch, and its paste (5–10 g is taken for seven days to cure jaundice. | [23] |
| Cicer microphyllum | Seeds | Seeds are used for the treatment of jaundice. | [28] |
| Colocacia antiquorum | Corm | Corm (cooked or pealed) is kept in open places overnight. In the morning, chopped pieces are given with honey to the patient for five days. | [23] |
| Colocacia esculenta | Bulb | The dried bulb in the powder form is used for the treatment of jaundice. | [24] |
| Conyza japonica | Leaves | Leaf paste is used to cure jaundice. | [29] |
| Cucumis sativus | Fruit | Fresh fruit is cut into small pieces and taken thrice a day for three weeks to cure jaundice. | [24] |
| Cuscuta reflexa | Whole plant | A decoction of the whole plant is used to treat jaundice. | [24] |
| Crepis flexuosa | Whole plant | Whole plant juice is mixed with water in equal proportion and taken once a day to cure jaundice. | [31] |
| Datura stramonium | Leaves and fruit | Fruits and leaves are used to cure jaundice. | [42] |
| Emblica officinalis | Roots | A decoction of roots is recommended for two weeks to cure jaundice. | [24] |
| Elaeagnus rhamnoides | Fruit | Fruit juice is used to cure jaundice. | [31] |
| Euphorbia hirta | Stem and leaves | Stem and leaf extract is used to cure jaundice. | [24] |
| Euphorbia tirucalli | Leaves | A decoction of leaves is used for the treatment of jaundice. | [39] |
| Equisetum arvense | Young branch | Young branches are dipped in the water overnight, and juice is (mix a small quantity of Kujja-Mishri and two and a half kali mirch seeds) taken daily empty stomach at least for seven days to cure jaundice. | [23] |
| Flacourtia ramontchi | Bark, fruits, and roots | Bark, fruits, and roots are used to treat jaundice. | [25] |
| Gentiana kurrroo | Roots | The root powder is used for treating jaundice. | [27] |
| Gentiana tubiflora | Whole plant | The whole plant is ground with lazi (salted curd) to form a paste and given for forty to forty-five days to cure jaundice. | [30] |
| Gentiana leucomelaena Gentianopsis detonsa Gentianopsis paludosa | Whole plant | Plants are crushed with a small proportion of petals of Polemonium caerulem, and this mixture is given with curd or cow milk empty stomach for fifteen to twenty-two days to cure jaundice. | [30] |
| Gentianella moorcroftiana | Aerial plant part | Juice of fresh extracted aerial plant part is taken empty stomach to cure jaundice. | [31] |
| Gernaium nepalenses | Roots | Root powder (2 g) is administered thrice a day to cure jaundice. | [42] |
| Geum elatum Wallich | Leaves | Leaves extract, mixed with cow milk or curd, is given for fifteen to twenty-two days to cure jaundice. | [30] |
| Hedera helix | Leaves | Crushed leaves’ juice is used to cure jaundice. | [27] |
| Hippophae tibetana | Fruit | A decoction of the fruit is taken to cure jaundice. | [31] |
| Hordeum vulgare | Seeds | Dried seed powder is mixed with a sugar solution to cure jaundice. | [37] |
| Justicia adhatoda | Roots | A decoction of its roots is given to the patient for one month to cure jaundice. | [24] |
| Leucas cephalotes | Entire herb | Juice of the entire herb (10–15 mL) is given to the patient for eight to ten days. | [23,36] |
| Mallotus philippinensis | Seeds | Seed powder is given for the treatment of jaundice. | |
| Mentha spicata | Leaves | Fresh leaf juice is taken with Kujja-Mishri and given twice a day for two or more weeks. | [23] |
| Morus alba | Fruit | Fruit juice is used for treating jaundice. | [24] |
| Morus nigra | Fruit | Fresh fruit juice is given to the patients twice a day for two weeks. | [24] |
| Oroxylum indicum | Bark of the stem | Crushed bark is soaked in water overnight and given with a small amount of kapoor to cure jaundice. A decoction of the bark is also used for treating jaundice. | [23] |
| Persicaria amplexicaulis | Whole plant | A decoction of the whole plant is given orally to treat jaundice. | [26] |
| Picris hieracioides | Whole plant | Plant extract with salted curd is given to the patient for twenty to thirty days. | [30] |
| Pistacia integerrima | Fruit | Fresh fruit juice is given daily for seven days to curing jaundice. | [24] |
| Phyllanthus fraternus | Whole plant, roots | A decoction of the entire herb and juice of fresh roots is given for seven days to cure jaundice. | [23] |
| Picrorhiza kurroa | Rhizome | Rhizome powder is used to cure jaundice. | [24] |
| Polygonum tortuosum | Whole plant | A paste of the whole plant is mixed with curd (prepared from goat’s milk) and given an empty stomach for fifteen to twenty-two days. | [30] |
| Podophyllum hexandrum | Flower and leaves | The juice of flowers and leaves is mixed with butter and taken orally to cure jaundice. | [41] |
| Prunus domestica | Fruit | The fruit extract is used for the treatment of jaundice. | [24] |
| Punica granatum | Fruit and seeds | Seeds and fruit powder is taken with water and sugar solution to cure jaundice. | [24] |
| Raphanus sativus | Root and fleshy part | A decoction of roots and juice of fleshy part is given to cure jaundice. | [24] |
| Ricinus communis | Leaves | Leaf juice is given with cow’s milk early in the morning for seven days to treat jaundice. | [23] |
| Rosa webbiana | Fruit | Fruit powder is mixed with little quantity of water and taken daily to cure jaundice. | [31] |
| Rubia manjith | Roots and stem | Roots and stem paste is given to cure jaundice. | [42] |
| Saccharum officinarum | Stem | Stem juice is used to cure jaundice. | [24] |
| Saxifraga flagellaris | Leaves and stem | Leaves and stems are used to cure jaundice. | [28] |
| Scorzonera divaricata | Leaves and shoot | A decoction of leaves and shoots is taken orally to cure jaundice. | [31] |
| Sesamum indicum | Leaves | Powder made from fresh leaves is used to cure jaundice. | [24] |
| Solanum nigrum | Leaves | Tablets are made from crushed leaves and taken with imli (tamarind) or curd for treating jaundice. | [23] |
| Solanum surattense | Fruit | The fruit is directly consumed for the treatment of jaundice. | [24] |
| Tamarindus indica | Fruit and root | A decoction of its roots is used to treat jaundice. The fruit is also used to cure jaundice. | [24] |
| Taraxacum officinale | Root and leaves and whole herb | The entire herb in the crushed form (10 gm) is given to the patient for ten days to curing jaundice. | [23] |
| Terminalia bellirica | Leaves | A decoction of leaf powder is taken to cure jaundice. | [36] |
| Terminalia chebula | Fruit rind | Fruit powder is mixed with rock salt and taken with warm water for eight to ten days to cure jaundice. | [23] |
| Thalictrum foliolosum | Roots | A decoction of roots is used to treat jaundice. | [27] |
| Tinospora cordifolia | Fresh stem | The dried stem of the giloe is crushed with punarnava mool, and its juice is taken for seven to ten days. | [23] |
| Trigonella emodi | Leaves and flower | Leaves and flower powder is taken with water twice a day for seven to ten days. | [31] |
| Urtica dioica | Whole plant | The whole plant is used to treat jaundice. | [42] |
| Vernonia anthelmintica | Seeds and leaves | A decoction of seeds and leaves is given to cure jaundice. | [27] |
| Viola serpens | Whole plant | A decoction of dried plant is taken with sugar for more than fifteen days. | [24] |
| Woodfordia fruticosa | Flowers | Flower extract is used to cure jaundice. | [24] |
| Youngia tenuifolia | Leaves | Leaves are used to treat jaundice. | [28] |
| Plant Name | Phytochemical Constituent | Data Source |
|---|---|---|
| Aconitum rotundifolium | Diterpenoid alkaloids (isoatisine, atisine chloride) | [52] |
| Adiantum capillus-veneris | Flavonoids, phenolic acids (sulfate esters of hydroxycinnamic acid), alkaloids, terpenoids (triterpenes), steroids, tannins, saponins | [53] |
| Aegle marmelos | Phenolic compounds (coumarins, such as marmelosin, marmesin, imperatorin, scopoletin, and esculetin), alkaloids (aeglin, aegelenine, skimmianine), tannins | [54] |
| Ajania tibetica | Terpenoids (bornyl acetate 60.7%, β-caryophyllene 9.1%, β-eudesmol 5.3%, methyl thymol 4.3% and borneol 2.2%) | [55] |
| Aloe vera | Fatty acids (such as n-hexadecanoic acid 20.47%, oleic acid 14.53%, tetradecanoic acid 1.04%, 1,2-benzenedicarboxylic acid, diisooctyl ester 13.60%, squalene 6.60%, butyl octyl ester 2.30%) | [56] |
| Argemone mexicana | Alkaloids (argemexicaine A, argemexicaine B, protopine, columbamin, muramine, cryptopine, isocorydine), carbohydrates (arabinose, lactose), steroids (β-sitosterol, stigma-4-en-3,6-dione), terpenoids (β-amyrin, trans-phytol), flavonoids (eriodictyol, luteolin, quercetin, rutin), tannins, phenolic acids (vanillic acid) | [57] |
| Asparagus adscendens | Saponins and steroids (stigmasterol glycosides) | [26] |
| Aquilegia fragrans | Steroids (β-sitosterol), 2,4-dihydroxyphenylacetic acid methyl ester, aquilegiolide, glochidiono lactone-A, and alkaloids (magnoflorine) | [58] |
| Bauhinia variegata | Terpenoids, flavonoids, tannins, saponins, steroids, and cardiac glycosides | [26] |
| Berberis aristata | Phenolic acids (e-caffeic acid, chlorogenic acid), flavonoids (quercetin, rutin), and alkaloids (berberine, berbamine, palmatine, columbamine, jatrorrhizine, oxyacanthine) | [59] |
| Berberis lycium | Alkaloids (berberine, berbamine, chenabine, karakoramine, palmatine, baluchistanamine, gilgitine, jhelumine, punjabine, sindamine) | [26] |
| Betula utilis | Terpenoids (betulin, betulinic acid, oleanolic acid, acetyl-oleanolic acid, lupeol, lupenone, methyl betulonate, methyl betulate, karachic acid), steroids (sitosterol), flavonoids (leucocyanidin, polymeric leucoanthocyanidins) | [60] |
| Boerhavia diffusa | Flavonoids (rotenoids, quercetin, kaempferol, borhaavone), lignans, steroids, phenolic glycosides, phenolic compounds (trans-caftaric acid, xanthones), fatty acids, and hydrocarbons | [61] |
| Cajanus cajan | Carbohydrates 26.425 ± 0.32, proteins 12.83 ± 0.285, lipids and phenols | [62] |
| Calotropis procera | Alkaloids (calotropin, calotoxin, uskerin), flavonoids, tannins, saponins, cardiac glycosides, volatile oil, and steroids | [26] |
| Capparis spinosa | Flavonoids (flavonol, quercetin-7-O-β-d-glucopyranoside-β-l-rhamnopyranoside, quercetin-3-rutinoside, rutin), fatty oil, carbohydrates (pentosans), and saponin | [31] |
| Capsella bursa-pastoris | Fatty acids (dodecanoic acid 5.66 ± 1.17, tetradecanoic acid 29.63 ± 5.79, pentadecanoic acid 18.05 ± 3.06, hexadecanoic acid 284.48 ± 41.06, heptadecanoic acid 7.11 ± 1.60, octadecanoic acid 53.20 ± 0.68, eicosanoic acid 2.52 ± 0.33), steroids (phytosterol, cholesterol, campesterol, stigmasterol, β-sitosterol), amino acids (glycine, histidine), and flavonoids (tricin, kaempferol, quercetin) | [63] |
| Capsicum annum | Alkaloids (capsaicin 0.5%–0.9%), glycosides, carbohydrates, steroids, terpenoids (triterpenes), and carotenoids (capsanthin, capsorubin 4–16%) | [64] |
| Carica papaya | Flavonoids, saponins, tannins, glycosides, and steroids | [65] |
| Carissa opaca | Cardiac glycosides (digitoxigenin-3-O-β-d-digitalopyranoside), phenolic compounds, lignans, terpenoids (17-hydroxy-11-oxo-nor-β-amyrone, urs-12-ene-3β, 22β-diol-17-carboxylic acid), steroids (stigmasterol, campesterol, β-sitosterol), flavonoids (rutin, quercetin), essential oils (hydroxyacetophenone 89.5%, benzyl salicylate 6.0%, benzyl benzoate 4.6%, (E,E)-α-farnesene 3.5%), protein (1.3%), and carbohydrates (17.39%) | [66] |
| Cassia fistula | Terpenoids (lupeol), steroids (β-sitosterol), fatty acid alcohols (hexacosanol), flavonoids (kaempferol, leucopelargonidin, rhamnetin-3-O-gentiobioside), phenolic acids (rhein, 3-formyl-1-hydroxy-8- methoxy anthaquinone), and alkaloids | [26] |
| Centella asiatica | Alkaloids, glycosides, terpenoids, steroids, flavonoids, tannins, and reducing sugars | [67] |
| Cicer microphyllum | Steroids (phytosterols), flavonoids, phenolic compounds, tannins, carbohydrates, proteins, and amino acids | [68] |
| Colocasia esculenta | Flavonoids (flavones, apigenin, luteolin, anthocyanins), carbohydrate (starch 0.23–0.52%), and lipids 0.017–0.025% | [69] |
| Conyza japonica | Terpenoids (sesquiterpenoids, conyterpenols A−D, strictic acids), flavonoids, and their glycosides | [70] |
| Crepis flexuosa | Phenolic acids (p-hydroxybenzoic acid, ethyl p-hydroxybenzoate, esculetin) terpenoids (taraxast-20(30)-ene-3β, 21α-diol, ursolic acid, oleanolic acid), flavonoids (apigenin, luteolin, luteolin-7-O-β-D-glucoside), fatty acids (octacosanoic acid, 2′,3′-dihydroxypropyl pentacosanoate), and steroids (daucosterol) | [71] |
| Cuscuta reflexa | Flavonoids (aromandendrin), glycosides, carotenoids (lutein- 10–22%, lycopene), alkaloids, steroids (campesterol, stigmasterol, stigmast-5-en-3-O-β-D-glucopyranoside), lignin (sesamin), and terpenoids (lupeol, maragenin) | [72] |
| Cucumis sativus | Steroids, glycosides, flavonoids, alkaloids, saponins, and tannins (except gums and reducing sugars) | [73] |
| Datura stramonium | Alkaloids (atropine, hyoscyamine, scopolamine), glycosides, saponins, and tannins | [74] |
| Elaeagnus rhamnoides | Alkaloids (carboline), terpenoids (ursolic acid, uvaol, amyrin), flavonoids (quercetin, myricetin, isorhamnetin, glucosides, rutin), carotenoids, fatty oil, and steroids (sitosterol, citrostandienol) | [31] |
| Emblica officinalis | Phenolic acids (propnyl 3,4,5-trihydroxybezonate, 2,3,7,8-tetrahydroxy chromeno [5,4,3-cde]chromene-5,10-dione, chlorogenic acid, ellagic acid), flavonoids (rutin, quercetin), tannins, amino acids, fixed oils | [75] |
| Euphorbia hirta | Terpenoids (diterpenes, triterpenes), phenolic acids (coumarins), and lignans | [76] |
| Euphorbia tirucalli | Fatty acids (palmitic acid, linoleic acid), steroids (sitosterol, stigmasterol, campesterol), flavonoids (anthocyanin, cyanidin glycoside) | [77] |
| Equisetum arvense | Flavonoids 0.6–0.9% (such as apigenin-5-O-glucoside, genkwanin-5-O-glucoside, kaempferol-3,7-di-O-glucoside, kaempferol-3-O-(6′-O-malonylglucoside)-7-O-glucoside, kaempferol-3-O-sophoroside, luteolin-5-O-glucoside, quercetin-3-O-glucoside), terpenoids (cis-geranyl acetone 13.74%, thymol 12.09%, trans-phytol 10.06%, triterpenes), alkaloids, carbohydrates, proteins, amino acids, steroids (phytosterols), saponins, and tannins | [78] |
| Flacourtia ramontchi | Saponins, steroids, sugar, lignans, and terpenoids (triterpenes) | [79] |
| Geranium nepalenses | Steroids (β-sitosterol, β-sitosterol-glactoside, stigmasterol) and terpenoids (ursolic acid) | [80] |
| Gentiana kurroo | Terpenoids (iridoids, triterpenoids), flavonoids, alkaloids | [81] |
| Gentianopsis paludosa | Flavonoids (luteolin), xanthones (gentiacaulein, phenolics-1-hydroxy-3,7,8-trimethoxyxanthone), terpenoids (ursolic acid), dicarboxylic acid–succinic acid | [31] |
| Gentianopsis detonsa | Flavonoids (luteolin), dicarboxylic acid–succinic acid, xanthones (gentiacaulein, 1-hydroxy-3,7,8-trimethoxyxanthone), terpenoids (ursolic acid) | [31] |
| Geum elatum | Fatty acid alcohols (hentriacontanol, hentriacontanone), sterols (β-sitosterol), phenolic acids (tetra-O-methyl ellagic acid, ellagic acid), and flavonoids (isoquercetrin) | [82] |
| Hedera helix | Phenolic acids (gallic acid 131.25 ± 1.54), flavonoids (quercetin 18.61 ± 0.37), alkaloids (emetine), amino acids, and saponins (hederacoside C, α-hederin, hederagenin) | [83] |
| Hippophae tibetana | Flavonoids (isorhamnetin, quercetin, kaempferol, rhamnetin, quercetin -3-O- rutinoside, quercetin-3-O-galactoside), fatty acids (2-hydroxydecanoic acid, nona-7-enoic acid, undec-9-en-7-ynoic acid, 13-phenyl tridecanoic acid, 5,9,21-nonacosatrienoic acid, 1,3-dicapryloyl-2- linoleoylglycerol, oleic, linoleic, linolenic acids and fats 3.5–4.8%) | [84] |
| Hordeum vulgare | Flavonoids, phenolic acids, terpenoids, glycosides, and saponins | [85] |
| Justicia adhatoda | Alkaloids (vasicine, vasicol, vasicinone, peganine, adhatonine, vasicinol, vasicinolone), flavonoids (kaempferol, quercetin), and steroids (β-sitosterol) | [26,86] |
| Leucas cephalotes | Phenolic acids (gallic acid, protocatechuic acid, chlorogenic acid, caffeic acid, and ferulic acid) | [26] |
| Mentha spicata | Fatty acids (methyl esters, methyl acetate 2–11%), terpenoids (menthol 33–60%, menthone 15–32%, isomenthone 2–8%, 1,8 cineole eucalyptol 5–13%, menthofuaran 1–10%, limonene 1–7%), flavonoids, alkaloids, and sugars | [87] |
| Morus alba | Phenolic acids (coumarins, benzofurans), flavonoids (chalcones, flavones, flavane derivative, (2S)-4′-hydroxy-7-methoxy-8-prenylflavan) | [88] |
| Morus nigra | Phenolic acids, alkaloids, terpenoids (oleanolic acid), and flavonoids (quercetin, luteolin, apigenin, kuwanon, kaempferol) | [89] |
| Oroxylum indicum | Steroids, tannins, alkaloids, glycosides, and flavonoids | [90] |
| Persicaria amplexicaulis | Flavonoids (quercetin), steroids (β-sitosterol), phenolic compounds (methyl-4-hydroxy cinnamate, gallic acid, protocatechuic acid, methyl gallate, vanicoside A, vanicoside B), terpenoids (arborinone), 25-hydroxycholest-5-en3β-yl acetate | [91] |
| Phyllanthus niruri | Alkaloids, terpenoids (triterpenes), phenols, flavonoids (quercetin, kaempferol), lignin glycoside, tannins, and fatty acids | [92] |
| Picrorrhiza kurroa | Terpenoids (iridoid glycoside, triterpenoids, kutkin, picroside I 3.66 ± 0.11%, kutkoside 4.44 ± 0.02%), steroids, tannins, and saponins | [93] |
| Picris hieracioides | Terpenoids (gammacer-16-ene derivatives, gammacer-16-en-3β-yl acetate) | [94] |
| Pistacia integerrima | Phenolic acids, carotenoids, terpenoids (monoterpens 91%, triterpenes), flavonoids (catechins), saponins, tannins, and steroids | [95] |
| Podophyllum hexandrum | Terpenoids, steroids, flavonoids, saponins, tannins, glycosides, and amino acids | [96] |
| Punica granatum | Flavonoids, saponins, tannins, phenolic acids, glycosides, steroids (phytosterols), terpenoids, carbohydrates, and proteins | [97] |
| Prunus domestica | Flavonoids, phenylpropanoid esters, phenolic acids (caffeoylquinic acids), steroids, and terpenoids | [98] |
| Raphanus sativus | Tannins (phlobatannins), saponins, flavonoids, phenolic acids (anthraquinones), steroids (phytosterol), alkaloids, terpenoids, cardiac glycosides, glucosinolates, isothiocyanates, protein 28.57%, carbohydrates 39.82% and fats 27.76% | [99] |
| Ricinus communis | Alkaloids, steroids, glycosides, flavonoids (quercetin, vitexin, rutin, kaempferol, epicatechin), terpenoids, phenolic acids (gentisic acid, ellagic acid, gallic acid, coumarins), and essential oils 37% | [100] |
| Rosa webbiana | Flavonoids, alkaloids, tannins, and saponins | [101] |
| Rubia manjith | Phenolic acids (quinones like glycosides, including rubiadin,1-hydroxy,2-methoxy anthraquinone, 3-dimethoxy 2 carboxy anthraquinone, munjistin, purpurin, pseudopurpurin, mollugin, furomollugin), fatty acids (rubiprasin A,B,C), ruiearbonls, and terpenoids (aborane, triterpenes) | [102] |
| Saccharum officinarum | Steroids (phytosterols), terpenoids, flavonoids, -O- and -C-glycosides, phenolic acids, fatty acid alcohols (policosanoles 2.5–80%, octacosanol 50–80% of the total policosanoles) | [103] |
| Sesamum indicum | Phenolic acids (anthraquinone) and tannins | [104] |
| Solanum nigrum | Flavonoids (catechin, epicatechin, rutin), phenolic acids (caffeic acid, gallic acid, protocatechuic acid), fatty acids (linoleic acid 67.9%), carbohydrates (polysaccharides), and proteins content 17% | [105] |
| Solanum surattense | Alkaloids, tannins, saponins, phenolic acids (phenolic methyl caffeate, caffeic acid, coumarins like imperatorin, scopoletin and esculetin), steroids (β-sitosterol), tri-terpenoids, and other major constituents like solasonine, solamargine, solasurine, torvoside K and L, khasianine, aculeatiside A, and solamargine | [106] |
| Taraxacum officinale | Free amino acids, terpenoids (germacranolide, taraxacin, taraxacerin, a diester of taraxanthin, lactupicrin, triterpenes), carbohydrates (glucans, mannan), phenolic acids (scopoletin, esculetin), steroids (phytosterols, taraxasterol, homotaraxasterol), eudesmanolic-tetrahydroridentin B, eudesmanolide-d-glucopyranoside, and proteins | [31] |
| Tamarindus indica | Fatty acids (n-heptadecanoate 13.00%, n-octadecanoic 6.1%, methyl-n-pentacosanoic 4.45%, nonanoic acid 1.92%, nonadecanoic acid 9.2%, 10-octadecenoicacid 7.8%, heptadecanoate 3.3%, n-pentacosenoic acid 2.54%, hexacoseoic acid 0.7%) and proteins 7.5–6.6% | [107] |
| Terminalia bellirica | Fatty acids (stearic acid 14.93%, myristic acid 17.70%, palmitic acid 21.6%, oleic acid 45.67%), proteins, carbohydrates, steroids (β-sitosterol), tannins (chebulanic acid, galloyl glucose), phenolic acids (gallic, ellagic acid, ethyl gallate), alkaloids, flavonoids, saponins, and terpenoids | [108] |
| Terminalia chebula | Tannins (chebulic acid, chebulagic acid, corilagin), phenolic acids (gallic acid, ellagic acid), steroids (β-sitosterol), terpenoids (triterpenes), and flavonoids (flavonol glycosides) | [109] |
| Thalictrum foliolosum | Alkaloids (berberine, jatrorrhizine, palmatine, thalrugosidine, thalrugosaminine, thalisopine thaligosine, thalirugidine, trhalirugine, 8-oxyberberine, berlambine, noroxyhydrastinine, N, O, O-trimethylsparsiflorine, thalicarpine, thalidasine, thalfoliolosumines A, and thalfoliolosumines B) | [110] |
| Tinospora cordifolia | Phenolic acids, flavonoids, glycosides, saponins, and alkaloids | [111] |
| Trigonella emodi Ben | Flavonoids (quercetin, luteolin, vitexin, orientin, isoorientin, vicenin-1, vicenin-2, naringenin, kaempferol, 7,4′-dimethoxyflavanone), protein, and carbohydrates | [112] |
| Urtica dioica | Alkaloids (betaine, choline), amino acids, carbohydrates, protein polymer (neutral and acidic), carotenoids (carotenes), and saponins | [31] |
| Vernonia anthelmintica | Fatty acids (vernolic acid) and terpenoids (vernodalin, vernodalol) | [113] |
| Viola serpens | Tannins, amino acids, reducing sugars, flavonoids (rutin),organic ester (methyl salicylate), glycosides (quercitrin), alkaloids (violin), terpenoids (monoterpens, sesquiterpenes), saponin, bis (2-ethylhexyl) maleate 15.62%, 2,4,4,6-tetramethyl-2-heptene 11.52%, hexen-3-ol 6.56% and cis verbeno l 4.77% | [114,115] |
| Woodfordia fruticosa | Phenolic acids, tannins (hydrolyzable tannins, such as woodfordins A, B, C), flavonoids (quercetin glycosides, naringenin 7-glucoside, kaempferol 3-O-glucoside), fatty acid alcohols (octacosanol), steroids (β-sitosterol, hecogenin), and terpenoids (lupeol, betulin, ursolic acid, oleanolic acid) | [116] |
| Plant Species | Plant Part Used (Extract Taken/Total Amount or Dose Required) | Test Dose/ Experimental Model | Constituents Responsible for (May/May Not Be Present) | Data Source |
|---|---|---|---|---|
| Aegle marmelos | Pulp/seeds (aq. extract/NA) | CCl4-induced hepatotoxicity/albino Wistar rat | NA | [15] |
| Powdered fruit pulp | Gentamicin-induced liver injury/oral/Wistar albino rats | NA | ||
| Leaves/powder | Ethanol-induced liver toxicity/orally/male albino Wistar rat | NA | ||
| Aloe vera | Aerial part (aq. extract/500 mg/kg bw) | CCl4 (1 mL/kg)/albino Wistar rat | NA | [5] |
| Argemone mexicana | Stem (aq. extract/250 and 150 mg/kg bw) | CCl4-induced hepatotoxicity (2 mL/kg bw)/albino Wistar rat | NA | [5] |
| Bauhinia variegata | Stem/bark extract/100 and 200 mg/kg) | CCl4 (1 mL/kg)/Sprague-Dawley rats | NA | [119] |
| Berberis aristata | Root extract/NA) | CCl4 (1 mL/kg)/albino rats | Berberine | [120] |
| Berberis chitria | Bark, stem extract/80 mg Livshis sample (450 mg) | CCl4 (1 mL/kg)/male Wistar albino rats | NA | [121] |
| Boerhavia diffusa | Root (aq. extract/2 mL/kg and 150 mg/kg bw) | Thioacetamide (100 mg/kg bw)/albino Wistar rat | Ursolic acids | [5] |
| Cajanus cajan | Leaves (methanolic extract/100 mg/kg bw) | Acetaminophen and D-galactosamine-induced hepatotoxicity/albino Wistar rat | Alkaloids and flavonoids | [15] |
| Calotropis procera | Flowers (hydro-ethanolic extract/200 mg/kg and 400 mg/kg) | Paracetamol (2 g/kg)/Wistar rats | Quercetin-3-rutinoside and other flavonoids | [122] |
| Capparis spinosa | Root bark (ethanolic extract/100, 200, and 400 mg/kg) | CCl4 (0.2 mg/kg)/mice | NA | [123] |
| Carica papaya | Seeds (aq. extract/(400 mg/kg) | CCl4 (1.5 mL/kg)/male Wistar rat | NA | [124] |
| Carissa opaca | Leaves (methanolic extract/(200 mg/kg bw) | CCl4 (0.5 mL/kg)/Sprague-Dawley rats | Isoquercetin, hyperoside, vitexin, myricetin, and kaempferol | [125] |
| Cassia fistula | Fruit pulp (aq. extract 2000 mg/kg bw) | CCl4 (1 mL/kg bw)/albino Wistar rat | Lupenol | [5] |
| Centella asiatica | Whole plant (aq. extract/(0.7 g/kg bw)) | CCl4 (0.7 mL/kg bw)/albino Wistar rat | NA | [5] |
| Cuscuta reflexa | Whole plant (hydro- alcoholic extract/400 mg/kg bw) | Paracetamol (200 mg/kg bw)/albino Wistar rat | Phenolic compounds | [5] |
| Colocasia antiquorum | Corms (petroleum ether extract/NA) | Paracetamol-induced hepatotoxicity (100 mg/kg bw)/albino mice | Anthocyanins | [126] |
| Euphorbia hirta | Whole plant (aq. extract/100–300 mg/kg bw) | CCl4-induced hepatotoxicity (NA)/adult male Wistar rat | Flavonoids | [127] |
| Euphorbia tirucalli | Arial parts (aq. extract/125–200 mg/kg bw) | CCI4 intoxicated (NA)/albino Wistar rat | Flavonoids | [128] |
| Hedera helix | Leaf (aq. extract/150 mg/kg bw) | CCl4-induced hepatotoxicity (5 mL/kg)/ | NA | [129] |
| Hordeum vulgare | Seeds (methanolic extract/300–500 mg/kg) | ethanol-induced liver damage (3.76 g/kg/day)/Wistar albino rat | Phenolic compounds | [85] |
| Justicia adhatoda | Leaves and flowers (methanolic extract/200 mg/kg bw) | CCl4-induced hepatotoxicity (1 mL/kg of body bw)/Swiss albino mice | NA | [130] |
| Flacourtia ramontchi | Leaf (aq. extract/250 and 500 mg/kg bw) | CCl4 (1.5 mL/kg bw)/albino Wistar rat | Phenolic compounds | [5] |
| Leucas cephalotes | Whole plant (methanolic extract/400 mg/kg bw) | CCl4-induced liver toxicity (NA)/male mice | Flavonoid | [15] |
| Morus alba | Leaves (petroleum ether chloroform alcoholic and water extract 200–500 mg/kg) | CCl4-induced hepatotoxicity (1 mL/kg)/Swiss albino mice | Alkaloids, carbohydrates, flavonoids, tannins, steroid | [131] |
| Morus nigra | Leaves (aq. methanolic extract/200 and 500 mg/kg) | Paracetamol-induced hepatotoxicity (NA)/mice | Quercetin, luteolin, and isorhamnetin | [132] |
| Oroxylum indicum | Leaves (ethanol, water, chloroform, and petroleum ether/300 mg/kg) | CCl4 hepatoprotective activity (0.7 mL/kg)/adult albino rats | Flavonoids and phenolics | [133] |
| Phyllanthus emblica | Fruit (ethanol extract/75 mg/kg/day bw) | Ethanol induced (4 g/kg/day bw)/albino Wistar rat | Phyllanthin and hypophyllanthin | [5] |
| Phyllanthus niruri | Leaves (aq. extract/100 mg/kg bw) | CCl4-induced hepatotoxicity (1 mL/kg body weight))/male adult rats | Phenolic compounds, Phyllanthin, and hypophyllanthin | [134] |
| Podophyllum hexandrum | (Hexane extract/50 mg/kg) | CCl4-induced hepatotoxicity (NA)/male albino rats | Polyphenols | [135] |
| Prunus domestica | Fruit (methanolic and ethanolic extract/20–100 mg/kg) | Paracetamol-induced hepatotoxicity (2 g/kg)/albino rats | NA | [136] |
| Punica granatum | Leaf (aq. extract/NA) | CCl4-induced hepatotoxicity (NA)/albino rats | Flavonoids | [137] |
| Raphanus sativus | Leaves (methanolic extract/300 mg/kg bw) | CCl4-induced cytotoxicity/male rats | NA | [15] |
| Ricinus communis | Leaves (ethanolic extract/800 mg/kg) | Paracetamol-induced cholestasis (2 g/kg)/adult Druckrey rats | Ricinine and n-demethyl-ricinine | [138] |
| Solanum nigrum | Whole plant (water or methanolic extract 500 mg/kg) | CCl4-induced hepatotoxicity (0.2 mg/kg)/Wistar albino rat | NA | [139] |
| Tamarindus Indica | Stem bark (ethanolic extract/NA) | Hepatic damage induced (100 to 200 mg/kg bw)/female SD rats | NA | [140] |
| Terminalia chebula | Fruit (aq. extract/NA) | t-BHP-induced hepatotoxicity/(NA)/mice | Phenolic compounds | [47] |
| Thalictrum foliolosum | Roots (ethanolic extract (200 to 1000 mg/kg bw)) | Paracetamol-induced hepatotoxicity (2 g/kg bw)/Wistar rat and male albino mice | NA | [141] |
| Tinospora cordifolia | Root, stem (petroleum ether/ethanol and aq. extract/400 mg/kg bw) | CCl4-induced | Flavonoids, alkaloids, and phenolics | [15] |
| liver toxicity/albino Wistar rat | ||||
| Urtica dioica | Seeds (polar extract/NA) | CCl4-induced | Phenolics | [142] |
| hepatotoxicity (150 to 200 g/kg)/male Wistar albino rat | ||||
| Woodfordia fruticosa | Flower (petroleum ether, chloroform, ethanolic/250 mg/kg bw) | CCl4 (1%)/albino Wistar rat | NA | [5] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Raghuvanshi, D.; Dhalaria, R.; Sharma, A.; Kumar, D.; Kumar, H.; Valis, M.; Kuča, K.; Verma, R.; Puri, S. Ethnomedicinal Plants Traditionally Used for the Treatment of Jaundice (Icterus) in Himachal Pradesh in Western Himalaya—A Review. Plants 2021, 10, 232. https://doi.org/10.3390/plants10020232
Raghuvanshi D, Dhalaria R, Sharma A, Kumar D, Kumar H, Valis M, Kuča K, Verma R, Puri S. Ethnomedicinal Plants Traditionally Used for the Treatment of Jaundice (Icterus) in Himachal Pradesh in Western Himalaya—A Review. Plants. 2021; 10(2):232. https://doi.org/10.3390/plants10020232
Chicago/Turabian StyleRaghuvanshi, Disha, Rajni Dhalaria, Anjali Sharma, Dinesh Kumar, Harsh Kumar, Martin Valis, Kamil Kuča, Rachna Verma, and Sunil Puri. 2021. "Ethnomedicinal Plants Traditionally Used for the Treatment of Jaundice (Icterus) in Himachal Pradesh in Western Himalaya—A Review" Plants 10, no. 2: 232. https://doi.org/10.3390/plants10020232
APA StyleRaghuvanshi, D., Dhalaria, R., Sharma, A., Kumar, D., Kumar, H., Valis, M., Kuča, K., Verma, R., & Puri, S. (2021). Ethnomedicinal Plants Traditionally Used for the Treatment of Jaundice (Icterus) in Himachal Pradesh in Western Himalaya—A Review. Plants, 10(2), 232. https://doi.org/10.3390/plants10020232








