Determination of ROS Scavenging, Antibacterial and Antifungal Potential of Methanolic Extract of Otostegia limbata (Benth.) Boiss.
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Extract
2.2. Antioxidant Assays
2.2.1. DPPH Radical Scavenging Activity Assay
2.2.2. Phosphomolybdate Assay
2.2.3. Reducing Power Assay
2.3. Antibacterial Assays
2.4. Antifungal Assay
3. Results
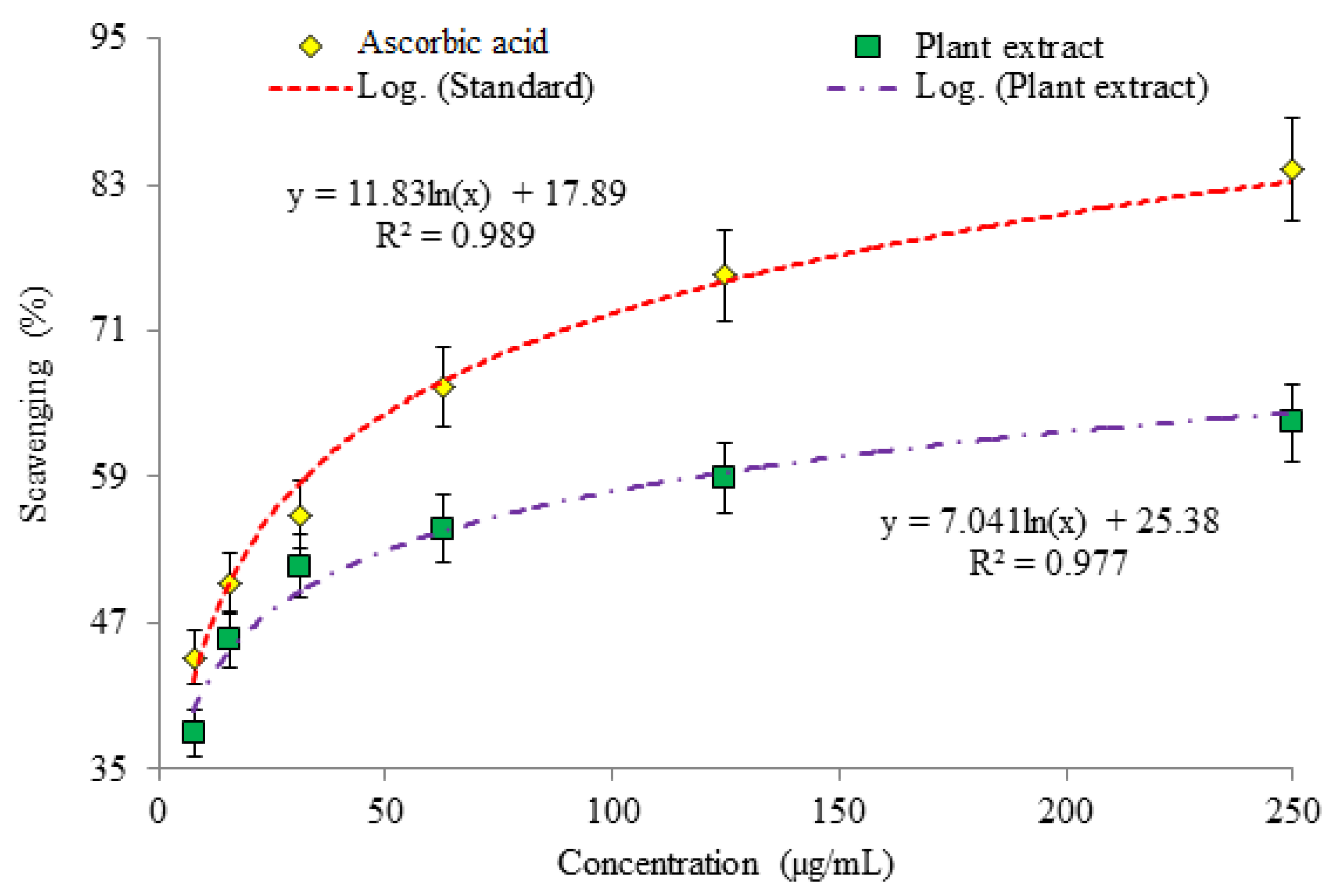
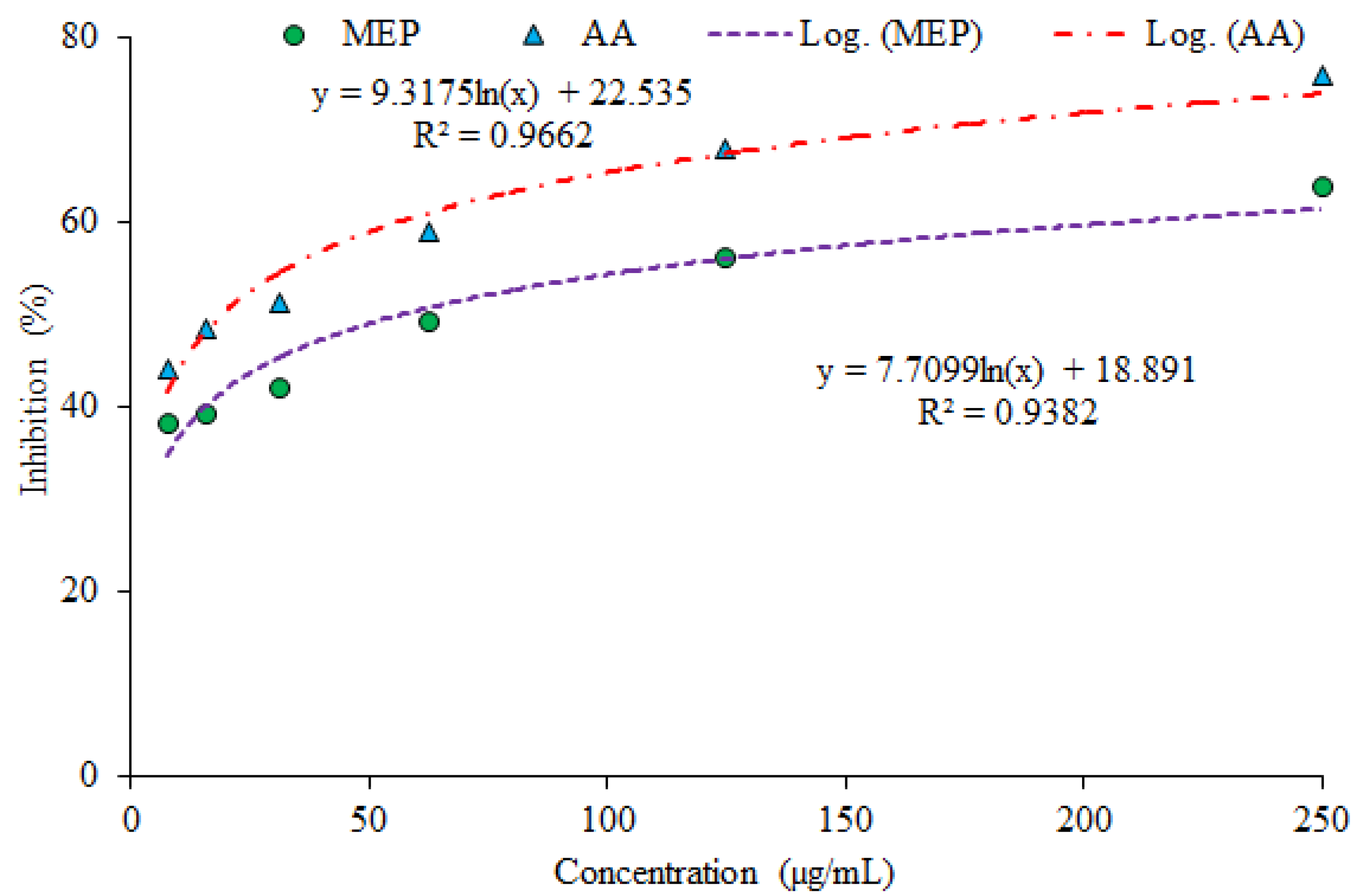
3.1. Evaluation of Antioxidant Potential
3.2. Antibacterial Potential
3.3. Antifungal Properties
4. Discussions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Poole, K. Overcoming antimicrobial resistance by targeting resistance mechanisms. J. Pharm. Pharmacol. 2001, 53, 283–294. [Google Scholar] [CrossRef] [PubMed]
- Rani, P.; Khullar, N. Antimicrobial evaluation of some medicinal plants for their anti-enteric potential against multi-drug resistantSalmonella typhi. Phytotherapy Res. 2004, 18, 670–673. [Google Scholar] [CrossRef] [PubMed]
- Essawi, T.; Srour, M. Screening of some Palestinian medicinal plants for antibacterial activity. J. Ethnopharmacol. 2000, 70, 343–349. [Google Scholar] [CrossRef]
- Menezes-Benavente, L.; Teixeira, F.K.; Kamei, C.L.A.; Margis-Pinheiro, M. Salt stress induces altered expression of genes encoding antioxidant enzymes in seedlings of a Brazilian indica rice (Oryza sativa L.). Plant Sci. 2004, 166, 323–331. [Google Scholar] [CrossRef]
- Yıldırım, A.; Mavi, A.; Kara, A.A. Determination of Antioxidant and Antimicrobial Activities of Rumex crispus L. Extracts. J. Agric. Food Chem. 2001, 49, 4083–4089. [Google Scholar] [CrossRef] [PubMed]
- Ksouri, R.; Megdiche, W.; Falleh, H.; Trabelsi, N.; Boulaaba, M.; Smaoui, A.; Abdelly, C. Influence of biological, environmental and technical factors on phenolic content and antioxidant activities of Tunisian halophytes. Comptes Rendus Biol. 2008, 331, 865–873. [Google Scholar] [CrossRef]
- Tepe, B.; Sokmen, M.; Akpulat, H.A.; Sokmen, A. Screening of the antioxidant potentials of six Salvia species from Turkey. Food Chem. 2006, 95, 200–204. [Google Scholar] [CrossRef]
- Anchana, C.; Aphiwat, T.; Nuansri, R. Screening of antioxidant activity and antioxidant compounds of some edible plants of Thailand. Food Chem. 2005, 92, 491–497. [Google Scholar]
- Suhaj, M. Spice antioxidants isolation and their antiradical activity: A review. J. Food Compos. Anal. 2006, 19, 531–537. [Google Scholar] [CrossRef]
- Tadhani, M.; Patel, V.; Subhash, R. In vitro antioxidant activities of Stevia rebaudiana leaves and callus. J. Food Compos. Anal. 2007, 20, 323–329. [Google Scholar] [CrossRef]
- Anwar, N.; Salik, S.; Ahmad, D. Antibacterial activity of Otostegia limbata. Int. J. Agric. Biol. 2009, 11, 647–650. [Google Scholar]
- Ahmad, N.; Abbasi, B.H.; Fazal, H. Evaluation of antioxidant activity and its association with plant development in Silybum marianum L. Ind. Crop. Prod. 2013, 49, 164–168. [Google Scholar] [CrossRef]
- Kebede, T.; Gadisa, E.; Tufa, A. Antimicrobial activities evaluation and phytochemical screening of some selected medicinal plants: A possible alternative in the treatment of multidrug-resistant microbes. PLoS ONE 2021, 16, e0249253. [Google Scholar] [CrossRef]
- Umamaheswari, M.; Chatterjee, T.K. In vitro antioxidant activities of the fractions of Coccinia grandis l. leaf extract. Afr. J. Tradit. Complement. Altern. Med. 2008, 5, 61–73. [Google Scholar] [CrossRef]
- Jindal, H.M.K.; Mohamad, J. Antioxidant Activity of Ardisia crispa (Mata pelanduk). Sains Malays. 2012, 41, 539–545. [Google Scholar]
- Bibi, Y.; Nisa, S.; Chaudhary, F.M.; Zia, M. Antibacterial activity of some selected medicinal plants of Pakistan. BMC Complement. Altern. Med. 2011, 11, 52. [Google Scholar] [CrossRef] [PubMed]
- Bibi, Y.; Zia, M.; Nisa, S.; Habib, D.; Waheed, A.; Chaudhary, F.M. Regeneration of Centella asiatica plants from non-embryogenic cell lines and evaluation of antibacterial and antifungal properties of regenerated calli and plants. J. Biol. Eng. 2011, 5, 13. [Google Scholar] [CrossRef]
- Marxen, K.; Vanselow, K.H.; Lippemeier, S.; Hintze, R.; Ruser, A.; Hansen, U.P. Determination of DPPH radical oxidation caused by methanolic extracts of some microalgal species by linear regression analysis of spectrophotometric meas-urements. Sensors 2007, 7, 2080–2095. [Google Scholar] [CrossRef]
- Garcia, A.; Rhoden, S.A.; Bernardi-Wenzel, J.; Orlandelli, R.C.; Azevedo, J.L.; Pamphile, J.A. Antimicrobial Activity of Crude Extracts of Endophytic Fungi Isolated from Medicinal Plant Sapindus saponaria L. J. Appl. Pharm. Sci. 2012, 2, 35–40. [Google Scholar] [CrossRef][Green Version]
- Clarke, G.; Ting, K.N.; Wiart, C.; Fry, J. High Correlation of 2,2-diphenyl-1-picrylhydrazyl (DPPH) Radical Scavenging, Ferric Reducing Activity Potential and Total Phenolics Content Indicates Redundancy in Use of All Three Assays to Screen for Antioxidant Activity of Extracts of Plants from the Malaysian Rainforest. Antioxidants 2013, 2, 1–10. [Google Scholar] [CrossRef]
- Khatoon, M.; Islam, E.; Islam, R.; Rahman, A.A.; Alam, A.K.; Khondkar, P.; Rashid, M.; Parvin, S. Estimation of total phenol and in vitro antioxidant activity of Albizia procera leaves. BMC Res. Notes 2013, 6, 121. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, I.C.F.R.; Baptista, P.; Boas, M.V.; Barros, L. Free radical scavenging capacity and reducing power of wild edible mushrooms from northeast Portugal. Food Chem. 2007, 100, 1511–1516. [Google Scholar] [CrossRef]
- Katalinic, V.; Milos, M.; Kulisic, T.; Jukic, M. Screening of 70 medicinal plant extracts for antioxidant capacity and total phenols. Food Chem. 2006, 94, 550–557. [Google Scholar] [CrossRef]
- Ahmed, D.; Arshad, M.A.; Asghar, M.N.; Aujla, M.I. Antioxidant and free radical scavenging potential of Otostegia limbata. Asian J. Chem. 2010, 22, 4524. [Google Scholar]
- Mun, S.-H.; Joung, D.-K.; Kim, Y.-S.; Kang, O.-H.; Kim, S.-B.; Seo, Y.-S.; Kim, Y.-C.; Lee, D.-S.; Shin, D.-W.; Kweon, K.-T.; et al. Synergistic antibacterial effect of curcumin against methicillin-resistant Staphylococcus aureus. Phytomedicine 2013, 20, 714–718. [Google Scholar] [CrossRef]
- Chovanová, R.; Mikulášová, M.; Vaverková, S. In VitroAntibacterial and Antibiotic Resistance Modifying Effect of Bioactive Plant Extracts on Methicillin-ResistantStaphylococcus epidermidis. Int. J. Microbiol. 2013, 2013, 1–7. [Google Scholar] [CrossRef]
- Kamonwannasit, S.; Nantapong, N.; Kumkrai, P.; Luecha, P.; Kupittayanant, S.; Chudapongse, N. Antibacterial activity of Aquilaria crassna leaf extract against Staphylococcus epidermidis by disruption of cell wall. Ann. Clin. Microbiol. Antimicrob. 2013, 12, 20. [Google Scholar] [CrossRef]
- Chowdhury, F.; Pal, S.; Sharmin, T.; Rashid, R.B.; Sikder, A.; Kabir, S.; Rahman, M.S.; Rashid, M.A. Bioactivities of artocarpus chaplasha Roxb. and Bougainvillea spectabillis Willd. Bang. Pharm. J. 2013, 16, 63–68. [Google Scholar] [CrossRef]
- Abubakar, M.S. Practical Manual of Pharmacognosy and Ethnomedicine; Usmanu Danfodiyo University Press: Sokoto, Nigeria, 2009; pp. 15–44. [Google Scholar]
- Mosafa, E.; Yahyaabadi, S.; Doudi, M. In-Vitro Antibacterial Properties of Sage (Salvia officinalis) Ethanol Extract against Multidrug Resistant Staphylococcus aureus, Escherichia coli, Pseudomonas aeruginosa and Klebsiella pneumoniae. Zahedan J. Res. Med. Sci. 2014, 16, 42–46. [Google Scholar]


| Concentration (µg/mL) | Absorbance (nm) | |
|---|---|---|
| AA | MEP | |
| 250 | 0.153 f ± 0.044 | 0.357 f ± 0.001 |
| 125 | 0.239 e ± 0.036 | 0.402 e ± 0.005 |
| 62.5 | 0.329 d ± 0.00 | 0.443 d ± 0.046 |
| 31.25 | 0.432 c ± 0.071 | 0.473 c ± 0.088 |
| 15.625 | 0.487 b ± 0.090 | 0.532 b ± 0.091 |
| 7.81 | 0.547 a ± 0.028 | 0.608 a ± 0.037 |
| Sample | IC 50 Value (µg/mL) | ||
|---|---|---|---|
| DPPH | PMA | RPA | |
| MEP | 33.01 | 27.40 | 56.54 |
| AA | 15.09 | 10.79 | 19.06 |
| Bacterial Strains | Zone of Inhibition (mm) | ||
|---|---|---|---|
| Cefotaxime | MEP | DMSO | |
| Klebsiella pneumonia | 17.40 ± 0.40 | 15.07 ± 0.30 | 0.00 ± 0.00 |
| Staphylococcus epidermidis | 17.83 ± 0.15 | 17.47 ± 0.45 | 0.00 ± 0.00 |
| Escherichia coli | 15.10 ± 0.17 | 12.17 ± 0.15 | 0.00 ± 0.00 |
| Staphylococcus aureus | 19.07 ± 0.40 | 18.80 ± 0.70 | 0.00 ± 0.00 |
| Pseudomonas spp. | 17.68 ± 0.73 | 14.73 ± 0.87 | 0.00 ± 0.00 |
| Analysis of Variance | |||||
|---|---|---|---|---|---|
| Sum of Squares | df | Mean Square | F | Sig. | |
| Between Groups | 571.255 | 4 | 142.814 | 9.512 | 0.002 |
| Within Groups | 150.133 | 10 | 15.013 | ||
| Total | 721.388 | 14 | |||
| Fungal Strains | Linear Growth (mm) | Percentage Inhibition | |||
|---|---|---|---|---|---|
| MEP | Fluconazole | DMSO | MEP | Fluconazole | |
| Alternaria spp. | 22.3 ± 1.5 | 14.16 ± 1.0 | 99 ± 1.73 | 78.45 | 86.71 |
| Aspergillus terreus | 32.0 ± 1.7 | 22.67 ± 1.5 | 100 ± 0.0 | 68.0 | 77.33 |
| Analysis of Variance | ||||||
|---|---|---|---|---|---|---|
| Sum of Squares | df | Mean Square | F | Sig. | ||
| MEP | Between groups | 163.804 | 1 | 163.804 | 324.364 | 0.000 |
| Within groups | 2.020 | 4 | 0.505 | |||
| Total | 165.824 | 5 | ||||
| Fluconazole | Between groups | 84.600 | 1 | 84.600 | 82.987 | 0.001 |
| Within groups | 4.078 | 4 | 1.019 | |||
| Total | 88.678 | 5 | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sadaf, H.M.; Bibi, Y.; Ishaque, M.; Nisa, S.; Qayyum, A.; Safdar, N.; Shah, Z.H.; Alsamadany, H.; Chung, G. Determination of ROS Scavenging, Antibacterial and Antifungal Potential of Methanolic Extract of Otostegia limbata (Benth.) Boiss. Plants 2021, 10, 2360. https://doi.org/10.3390/plants10112360
Sadaf HM, Bibi Y, Ishaque M, Nisa S, Qayyum A, Safdar N, Shah ZH, Alsamadany H, Chung G. Determination of ROS Scavenging, Antibacterial and Antifungal Potential of Methanolic Extract of Otostegia limbata (Benth.) Boiss. Plants. 2021; 10(11):2360. https://doi.org/10.3390/plants10112360
Chicago/Turabian StyleSadaf, Huma Mehreen, Yamin Bibi, Muhammad Ishaque, Sobia Nisa, Abdul Qayyum, Naila Safdar, Zahid Hussain Shah, Hameed Alsamadany, and Gyuhwa Chung. 2021. "Determination of ROS Scavenging, Antibacterial and Antifungal Potential of Methanolic Extract of Otostegia limbata (Benth.) Boiss." Plants 10, no. 11: 2360. https://doi.org/10.3390/plants10112360
APA StyleSadaf, H. M., Bibi, Y., Ishaque, M., Nisa, S., Qayyum, A., Safdar, N., Shah, Z. H., Alsamadany, H., & Chung, G. (2021). Determination of ROS Scavenging, Antibacterial and Antifungal Potential of Methanolic Extract of Otostegia limbata (Benth.) Boiss. Plants, 10(11), 2360. https://doi.org/10.3390/plants10112360







