Radial Oxygen Loss from Plant Roots—Methods
Abstract
:1. Introduction
2. Methods for Root Radial O2 Loss Determination
2.1. Qualitative Colorimetric Methods
2.1.1. Principle of the Method
2.1.2. Practical Set-Up
2.1.3. Generalities
2.2. Polarographic Cylindrical Oxygen Electrode
2.2.1. Principle of the Method
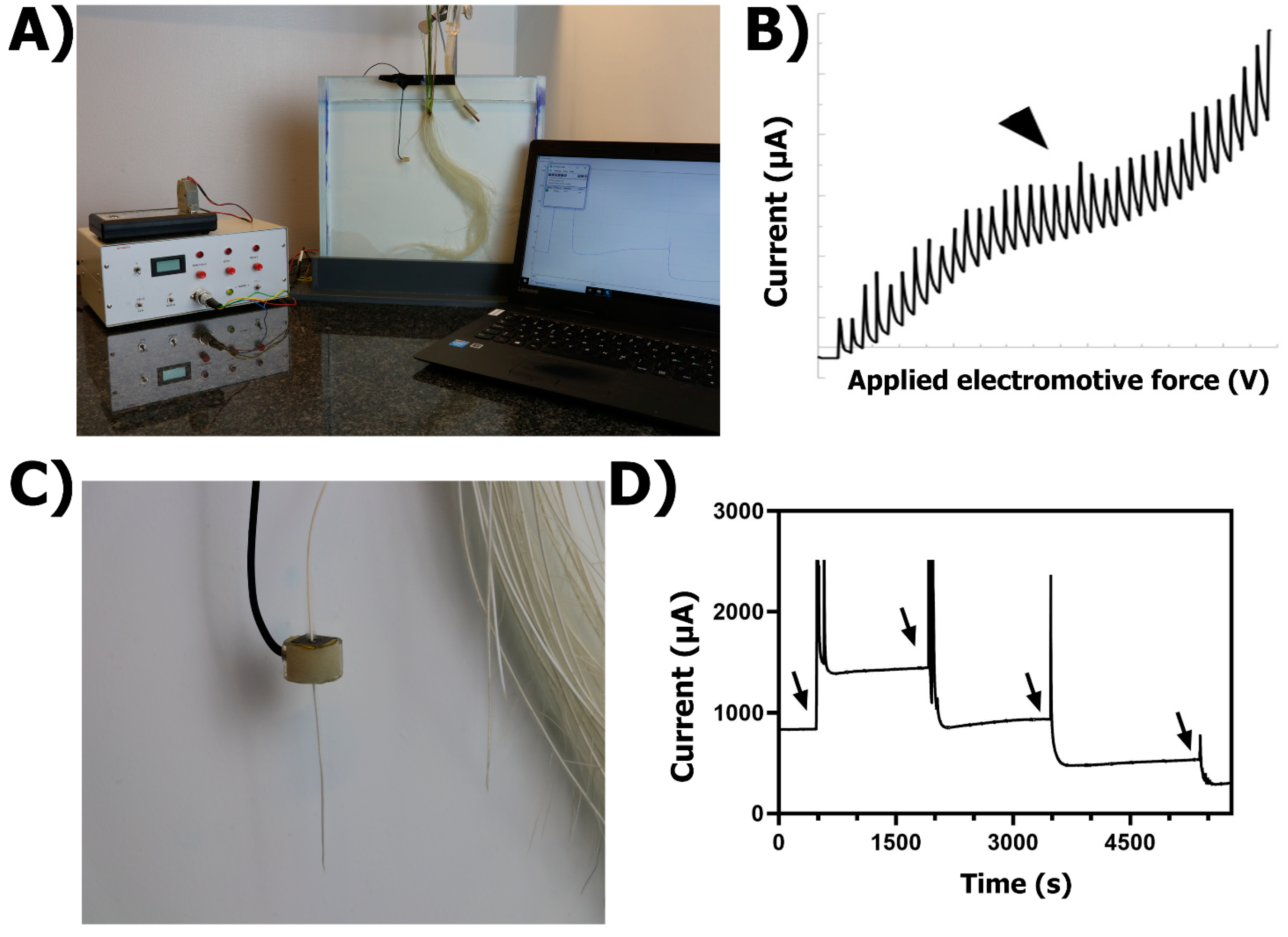
2.2.2. Practical Set-Up
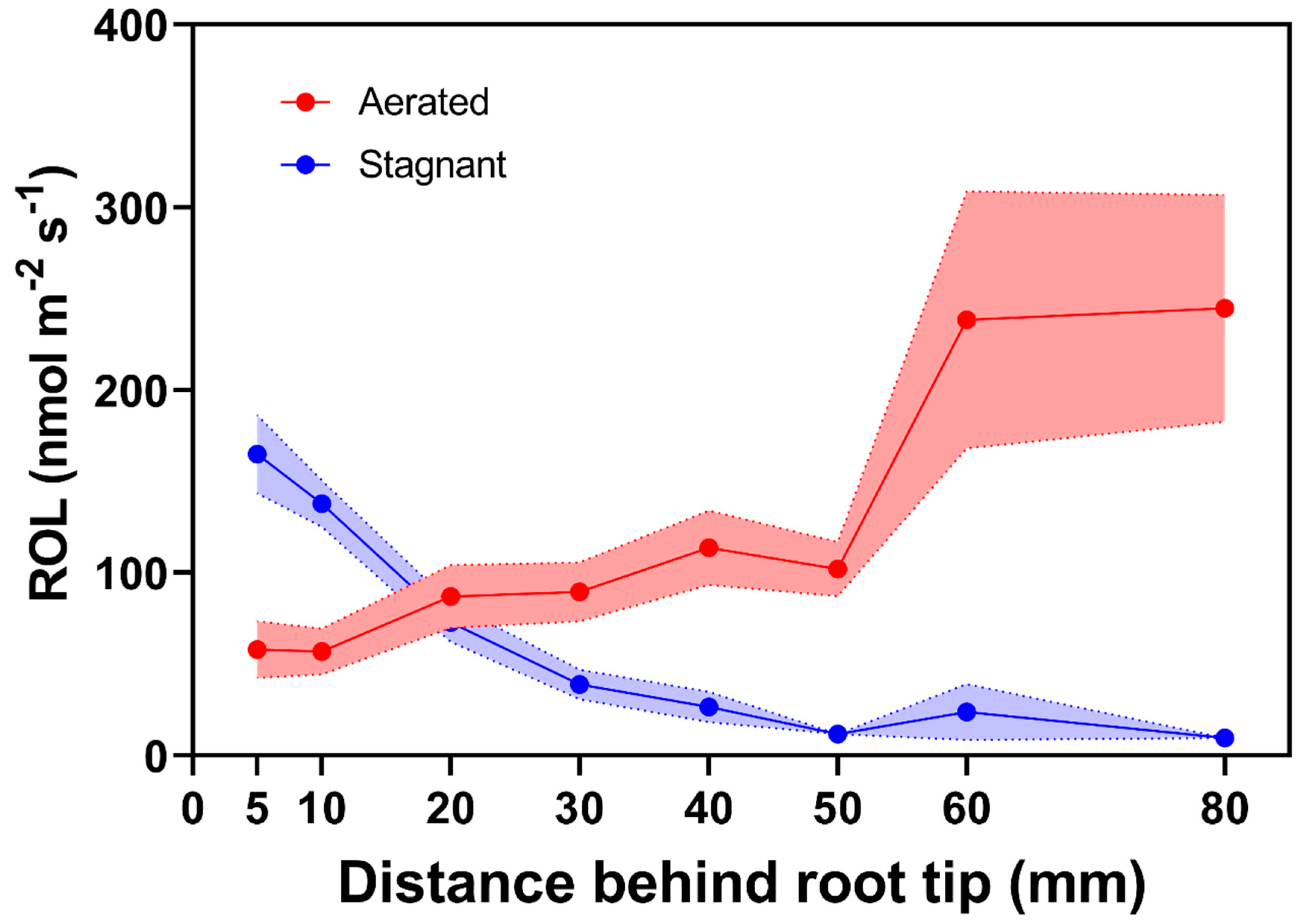
2.2.3. Generalities
2.3. Clark-Type O2 Microelectrodes and O2 Micro-Optodes
2.3.1. Principle of the Method: O2 Microelectrode
2.3.2. Principle of the Method: O2 Micro-Optodes
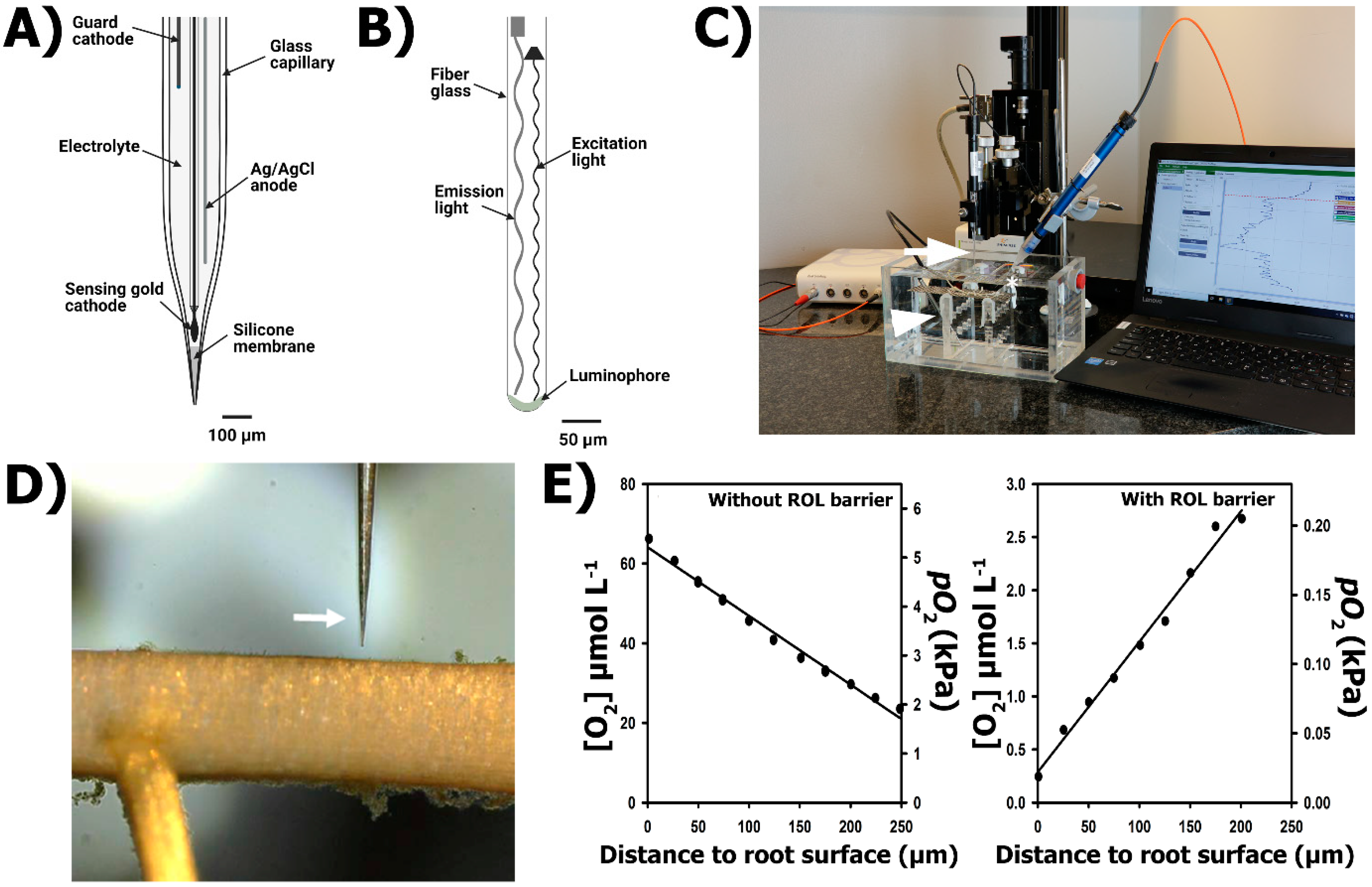
2.3.3. Practical Set-Up
2.3.4. Generalities
2.4. Planar Optodes
2.4.1. Principle of the Method
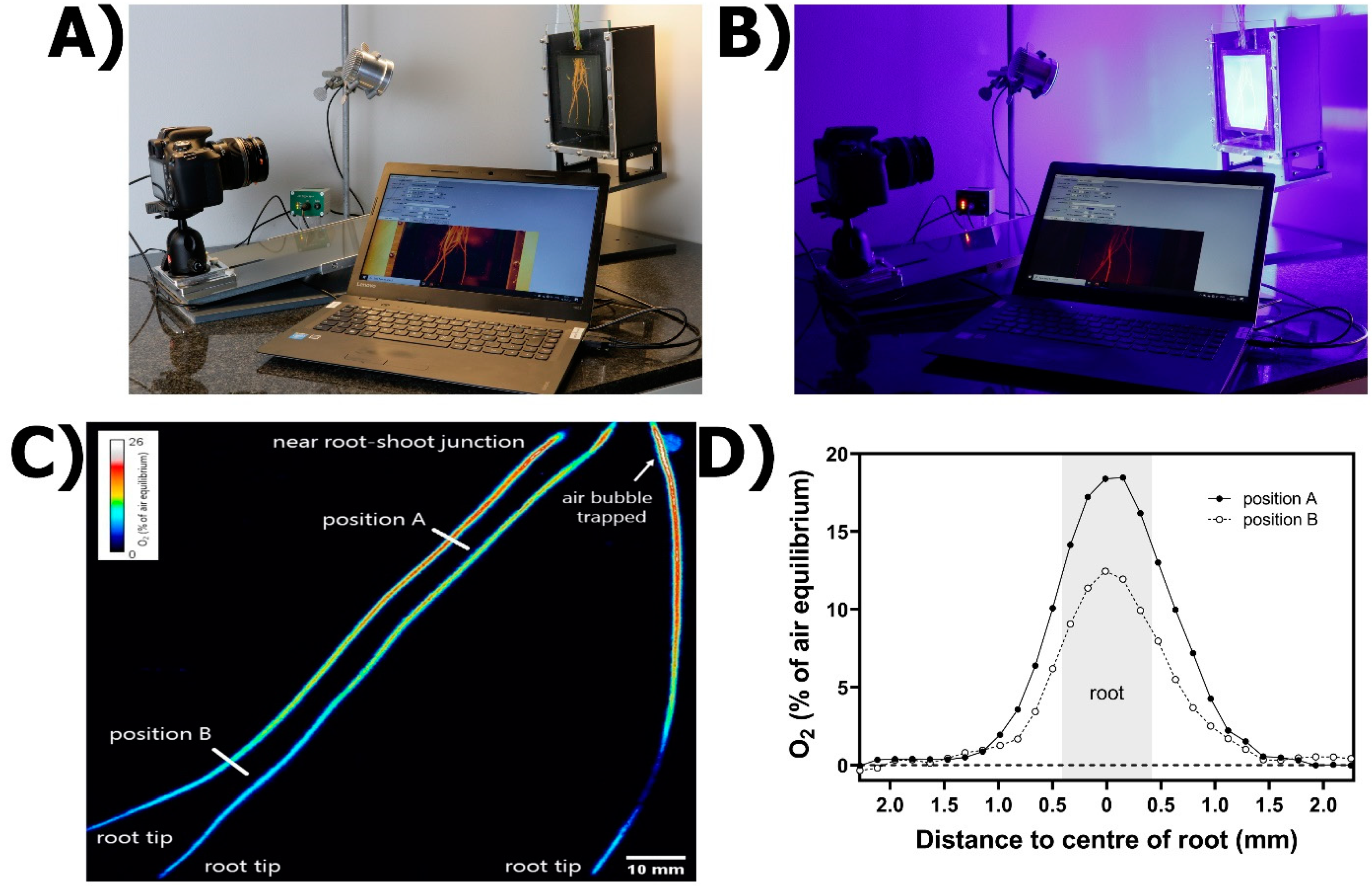
2.4.2. Practical Set-Up
2.4.3. Generalities
3. Conclusions and Further Perspectives
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Armstrong, W. Aeration in Higher Plants. Adv. Bot. Res. 1980, 7, 225–332. [Google Scholar] [CrossRef]
- Ponnamperuma, F. The Chemistry of Submerged Soils. Adv. Agron. 1972, 24, 29–96. [Google Scholar] [CrossRef]
- Setter, T.; Belford, B. Waterlogging: How it reduces plant growth and how plant overcome its effects. J. Agric. West. Aust. 1990, 31, 51–55. [Google Scholar]
- Colmer, T.D. Long-distance transport of gases in plants: A perspective on internal aeration and radial oxygen loss from roots. Plant Cell Environ. 2003, 26, 17–36. [Google Scholar] [CrossRef] [Green Version]
- Armstrong, W.; Healy, M.T.; Lythe, S. Oxygen diffusion in pea II. Oxygen concentrations in the primary pea root apex as affected by growth, the production of laterals and radial oxygen loss. New Phytol. 1983, 94, 549–559. [Google Scholar] [CrossRef]
- Armstrong, W.; Beckett, P. Measurement and modelling of oxygen release from roots of Phragmites australis. In Constructed Wetlands in Water Pollution Control; Elsevier: Amsterdam, The Netherlands, 1990; pp. 41–51. [Google Scholar]
- Jiménez, J.d.l.C.; Clode, P.L.; Signorelli, S.; Veneklaas, E.J.; Colmer, T.D.; Kotula, L. The barrier to radial oxygen loss impedes the apoplastic entry of Fe into the roots of Urochloa humidicola. J. Exp. Bot. 2021, 72, 3279–3293. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, W. Oxygen Diffusion from the Roots of Some British Bog Plants. Nat. Cell Biol. 1964, 204, 801–802. [Google Scholar] [CrossRef]
- Armstrong, W. The relationship between oxidation-reduction potentials and oxygen-diffusion levels in some waterlogged organic soils. J. Soil Sci. 1967, 18, 27–34. [Google Scholar] [CrossRef]
- Armstrong, W. Radial Oxygen Losses from Intact Rice Roots as Affected by Distance from the Apex, Respiration and Waterlogging. Physiol. Plant. 1971, 25, 192–197. [Google Scholar] [CrossRef]
- Colmer, T.D.; Gibberd, M.R.; Wiengweera, A.; Tinh, T.K. The barrier to radial oxygen loss from roots of rice (Oryza sativa L.) is induced by growth in stagnant solution. J. Exp. Bot. 1998, 49, 1431–1436. [Google Scholar] [CrossRef]
- Colmer, T.D. Aerenchyma and an Inducible Barrier to Radial Oxygen Loss Facilitate Root Aeration in Upland, Paddy and Deep-water Rice (Oryza sativa L.). Ann. Bot. 2003, 91, 301–309. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Colmer, T.D.; Cox, M.C.H.; Voesenek, L. Root aeration in rice (Oryza sativa): Evaluation of oxygen, carbon dioxide, and ethylene as possible regulators of root acclimatizations. New Phytol. 2006, 170, 767–778. [Google Scholar] [CrossRef] [PubMed]
- Rubinigg, M.; Stulen, I.; Elzenga, J.T.M.; Colmer, T.D. Spatial patterns of radial oxygen loss and nitrate net flux along adventitious roots of rice raised in aerated or stagnant solution. Funct. Plant Biol. 2002, 29, 1475–1481. [Google Scholar] [CrossRef]
- Kotula, L.; Ranathunge, K.; Schreiber, L.; Steudle, E. Functional and chemical comparison of apoplastic barriers to radial oxygen loss in roots of rice (Oryza sativa L.) grown in aerated or deoxygenated solution. J. Exp. Bot. 2009, 60, 2155–2167. [Google Scholar] [CrossRef] [PubMed]
- Kulichikhin, K.; Yamauchi, T.; Watanabe, K.; Nakazono, M. Biochemical and molecular characterization of rice (oryza satival.) roots forming a barrier to radial oxygen loss. Plant Cell Environ. 2014, 37, 2406–2420. [Google Scholar]
- Mongon, J.; Konnerup, D.; Colmer, T.D.; Rerkasem, B. Responses of rice to Fe2+ in aerated and stagnant conditions: Growth, root porosity and radial oxygen loss barrier. Funct. Plant Biol. 2014, 41, 922–929. [Google Scholar] [CrossRef]
- Shiono, K.; Ogawa, S.; Yamazaki, S.; Isoda, H.; Fujimura, T.; Nakazono, M.; Colmer, T. Contrasting dynamics of radial O2-loss barrier induction and aerenchyma formation in rice roots of two lengths. Ann. Bot. 2010, 107, 89–99. [Google Scholar] [CrossRef]
- Wu, C.; Ye, Z.; Li, H.; Wu, S.; Deng, D.; Zhu, Y.; Wong, M. Do radial oxygen loss and external aeration affect iron plaque formation and arsenic accumulation and speciation in rice? J. Exp. Bot. 2012, 63, 2961–2970. [Google Scholar] [CrossRef] [Green Version]
- Ejiri, M.; Sawazaki, Y.; Shiono, K. Some Accessions of Amazonian Wild Rice (Oryza glumaepatula) Constitutively Form a Barrier to Radial Oxygen Loss along Adventitious Roots under Aerated Conditions. Plants 2020, 9, 880. [Google Scholar] [CrossRef]
- Gibbs, J.; Turner, D.; Armstrong, W.; Darwent, M.; Greenway, H. Response to oxygen deficiency in primary maize roots. I. Development of oxygen deficiency in the stele reduces radial solute transport to the xylem. Funct. Plant Biol. 1998, 25, 745–758. [Google Scholar] [CrossRef]
- Colmer, T.D.; Greenway, H. Ion transport in seminal and adventitious roots of cereals during O2 deficiency. J. Exp. Bot. 2011, 62, 39–57. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Armstrong, W. Rhizosphere Oxidation in Rice and other Species: A Mathematical Model Based on the Oxygen Flux Component. Physiol. Plant. 1970, 23, 623–630. [Google Scholar] [CrossRef]
- Jiménez, J.d.l.C.; Kotula, L.; Veneklaas, E.J.; Colmer, T.D. Root-zone hypoxia reduces growth of the tropical forage grass Urochloa humidicola in high-nutrient but not low-nutrient conditions. Ann. Bot. 2019, 124, 1019–1032. [Google Scholar] [CrossRef]
- Armstrong, J.; Armstrong, W. Phragmites australis—A preliminary study of soil-oxidizing sites and internal gas transport pathways. New Phytol. 1988, 108, 373–382. [Google Scholar] [CrossRef]
- Kirk, G.J.D. Rice root properties for internal aeration and efficient nutrient acquisition in submerged soil. New Phytol. 2003, 159, 185–194. [Google Scholar] [CrossRef]
- Noorrohmah, S.; Takahashi, H.; Nakazono, M. Formation of a barrier to radial oxygen loss in L-type lateral roots of rice. Plant Root 2020, 14, 33–41. [Google Scholar] [CrossRef]
- Manzur, M.E.; Grimoldi, A.A.; Insausti, P.; Striker, G.G. Radial oxygen loss and physical barriers in relation to root tissue age in species with different types of aerenchyma. Funct. Plant Biol. 2015, 42, 9–17. [Google Scholar] [CrossRef]
- Kirk, G.J.; Boghi, A.; Affholder, M.; Keyes, S.D.; Heppell, J.; Roose, T. Soil carbon dioxide venting through rice roots. Plant Cell Environ. 2019, 42, 3197–3207. [Google Scholar] [CrossRef] [Green Version]
- Martin, B.C.; Bougoure, J.; Ryan, M.; Bennett, W.; Colmer, T.; Joyce, N.K.; Olsen, Y.; Kendrick, G. Oxygen loss from seagrass roots coincides with colonisation of sulphide-oxidising cable bacteria and reduces sulphide stress. ISME J. 2019, 13, 707–719. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schmidt, H.; Eickhorst, T.; Tippkötter, R. Monitoring of root growth and redox conditions in paddy soil rhizotrons by redox electrodes and image analysis. Plant Soil 2011, 341, 221–232. [Google Scholar] [CrossRef]
- Maisch, M.; Lueder, U.; Kappler, A.; Schmidt, C. Iron Lung: How Rice Roots Induce Iron Redox Changes in the Rhizosphere and Create Niches for Microaerophilic Fe(II)-Oxidizing Bacteria. Environ. Sci. Technol. Lett. 2019, 6, 600–605. [Google Scholar] [CrossRef] [Green Version]
- Armstrong, J.; Armstrong, W. Rice: Sulfide-induced Barriers to Root Radial Oxygen Loss, Fe2+ and Water Uptake, and Lateral Root Emergence. Ann. Bot. 2005, 96, 625–638. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abiko, T.; Kotula, L.; Shiono, K.; Malik, A.I.; Colmer, T.D.; Nakazono, M. Enhanced formation of aerenchyma and induction of a barrier to radial oxygen loss in adventitious roots of Zea nicaraguensis contribute to its waterlogging tolerance as compared with maize (Zea mays ssp. mays). Plant Cell Environ. 2012, 35, 1618–1630. [Google Scholar] [CrossRef] [PubMed]
- Kotula, L.; Schreiber, L.; Colmer, T.; Nakazono, M. Anatomical and biochemical characterisation of a barrier to radial O2 loss in adventitious roots of two contrasting Hordeum marinum accessions. Funct. Plant Biol. 2017, 44, 845–857. [Google Scholar] [CrossRef]
- Shiono, K.; Yamauchi, T.; Yamazaki, S.; Mohanty, B.; Malik, A.I.; Nagamura, Y.; Nishizawa, N.K.; Tsutsumi, N.; Colmer, T.D.; Nakazono, M. Microarray analysis of laser-microdissected tissues indicates the biosynthesis of suberin in the outer part of roots during formation of a barrier to radial oxygen loss in rice (Oryza sativa). J. Exp. Bot. 2014, 65, 4795–4806. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Watanabe, K.; Takahashi, H.; Sato, S.; Nishiuchi, S.; Omori, F.; Malik, A.I.; Colmer, T.D.; Mano, Y.; Nakazono, M. A major locus involved in the formation of the radial oxygen loss barrier in adventitious roots of teosinte Zea nicaraguensis is located on the short-arm of chromosome. Plant Cell Environ. 2017, 40, 304–316. [Google Scholar] [CrossRef] [Green Version]
- Colmer, T.D.; Kotula, L.; Malik, A.I.; Takahashi, H.; Konnerup, D.; Nakazono, M.; Pedersen, O. Rice acclimation to soil flooding: Low concentrations of organic acids can trigger a barrier to radial oxygen loss in roots. Plant Cell Environ. 2019, 42, 2183–2197. [Google Scholar] [CrossRef]
- Pedersen, O.; Nakayama, Y.; Yasue, H.; Kurokawa, Y.; Takahashi, H.; Floytrup, A.H.; Omori, F.; Mano, Y.; Colmer, T.D.; Nakazono, M. Lateral roots, in addition to adventitious roots, form a barrier to radial oxygen loss in Zea nicaraguensis and a chromosome segment introgression line in maize. New Phytol. 2021, 229, 94–105. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, W. The Oxidising Activity of Roots in Waterlogged Soils. Physiol. Plant. 1967, 20, 920–926. [Google Scholar] [CrossRef]
- Kludze, H.K.; DeLaune, R.D.; Patrick, W.H. A Colorimetric Method for Assaying Dissolved Oxygen Loss from Container-Grown Rice Roots. Agron. J. 1994, 86, 483–487. [Google Scholar] [CrossRef]
- Sorrell, B.K. Effect of external oxygen demand on radial oxygen loss by Juncus roots in titanium citrate solutions. Plant Cell Environ. 1999, 22, 1587–1593. [Google Scholar] [CrossRef]
- Matsui, T.; Tsuchiya, T. A Method to Estimate Practical Radial Oxygen Loss of Wetland Plant Roots. Plant Soil 2006, 279, 119–128. [Google Scholar] [CrossRef]
- Yamauchi, T.; Abe, F.; Tsutsumi, N.; Nakazono, M. Root Cortex Provides a Venue for Gas-Space Formation and Is Essential for Plant Adaptation to Waterlogging. Front. Plant Sci. 2019, 10, 259. [Google Scholar] [CrossRef]
- Armstrong, W.; Wright, E.J. Radial Oxygen Loss from Roots: The Theoretical Basis for the Manipulation of Flux Data Obtained by the Cylindrical Platinum Electrode Technique. Physiol. Plant. 1975, 35, 21–26. [Google Scholar] [CrossRef]
- Kotula, L.; Steudle, E. Measurements of oxygen permeability coefficients of rice (Oryza sativa L.) roots using a new perfusion technique. J. Exp. Bot. 2008, 60, 567–580. [Google Scholar] [CrossRef] [Green Version]
- Revsbech, N.P. An oxygen microelectrode with a guard cathode. Limnol. Oceanogr. 1989, 34, 474–478. [Google Scholar] [CrossRef]
- Clark, L.C., Jr. Monitor and control of blood and tissue oxygen tensions. Trans. Am. Soc. Artif. Intern. Organs 1956, 2, 41–48. [Google Scholar]
- Klimant, I.; Meyer, V.; Kühl, M. Fiber-optic oxygen microsensors, a new tool in aquatic biology. Limnol. Oceanogr. 1995, 40, 1159–1165. [Google Scholar] [CrossRef] [Green Version]
- Pedersen, O.; Revsbech, N.P.; Shabala, S. Microsensors in plant biology: In vivo visualization of inorganic analytes with high spatial and/or temporal resolution. J. Exp. Bot. 2020, 71, 3941–3954. [Google Scholar] [CrossRef] [PubMed]
- Henriksen, G.H.; Raman, D.R.; Walker, L.P.; Spanswick, R.M. Measurement of Net Fluxes of Ammonium and Nitrate at the Surface of Barley Roots Using Ion-Selective Microelectrodes. Plant Physiol. 1992, 99, 734–747. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peralta Ogorek, L.L.; Pellegrini, E.; Pedersen, O. Novel functions of the root barrier to radial oxygen loss—Radial diffusion resistance to H2 and water vapour. New Phytol. 2021, 231, 1365–1376. [Google Scholar] [CrossRef]
- Armstrong, W.; Cousins, D.; Turner, D.; Beckett, P.M. Oxygen Distribution in Wetland Plant Roots and Permeability Barriers to Gas-exchange with the Rhizosphere: A Microelectrode and Modelling Study with Phragmites australis. Ann. Bot. 2000, 86, 687–703. [Google Scholar] [CrossRef] [Green Version]
- Armstrong, W.; Beckett, P.M.; Colmer, T.; Setter, T.L.; Greenway, H. Tolerance of roots to low oxygen: ‘Anoxic’ cores, the phytoglobin-nitric oxide cycle, and energy or oxygen sensing. J. Plant Physiol. 2019, 239, 92–108. [Google Scholar] [CrossRef]
- Soukup, A.; Votrubová, O.; Čížková, H. Development of anatomical structure of roots of Phragmites australis. New Phytol. 2002, 153, 277–287. [Google Scholar] [CrossRef]
- Lassen, C.; Revsbech, N.P.; Pedersen, O. Macrophyte development and resuspension regulate the photosynthesis and production of benthic microalgae. Hydrobiologia 1997, 350, 1–11. [Google Scholar] [CrossRef]
- Gundersen, J.K.; Ramsing, N.B.; Glud, R.N. Predicting the signal of O2 microsensors from physical dimensions, temperature, salinity, and O2 concentration. Limnol. Oceanogr. 1998, 43, 1932–1937. [Google Scholar] [CrossRef]
- Larsen, M.; Borisov, S.M.; Grunwald, B.; Klimant, I.; Glud, R.N. A simple and inexpensive high resolution color ratiometric planar optode imaging approach: Application to oxygen and pH sensing. Limnol. Oceanogr. Methods 2011, 9, 348–360. [Google Scholar] [CrossRef]
- Glud, R.; Ramsing, N.; Gundersen, J.; Klimant, I. Planar optrodes:a new tool for fine scale measurements of two-dimensional O2 distribution in benthic communities. Mar. Ecol. Prog. Ser. 1996, 140, 217–226. [Google Scholar] [CrossRef]
- Santner, J.; Larsen, M.; Kreuzeder, A.; Glud, R.N. Two decades of chemical imaging of solutes in sediments and soils—A review. Anal. Chim. Acta 2015, 878, 9–42. [Google Scholar] [CrossRef] [Green Version]
- Holst, G.; Kohls, O.; Klimant, I.; König, B.; Kühl, M.; Richter, T. A modular luminescence lifetime imaging system for mapping oxygen distribution in biological samples. Sens. Actuators B Chem. 1998, 51, 163–170. [Google Scholar] [CrossRef]
- Liebsch, G.; Klimant, I.; Frank, B.; Holst, G.; Wolfbeis, O.S. Luminescence Lifetime Imaging of Oxygen, pH, and Carbon Dioxide Distribution Using Optical Sensors. Appl. Spectrosc. 2000, 54, 548–559. [Google Scholar] [CrossRef]
- Glud, R.N.; Tengberg, A.; Kühl, M.; Hall, P.O.J.; Klimant, I. An in situ instrument for planar O2 optode measurements at benthic interfaces. Limnol. Oceanogr. 2001, 46, 2073–2080. [Google Scholar] [CrossRef] [Green Version]
- Fischer, J.P.; Wenzhöfer, F. A novel planar optode setup for concurrent oxygen and light field imaging: Application to a benthic phototrophic community. Limnol. Oceanogr. Methods 2010, 8, 254–268. [Google Scholar] [CrossRef] [Green Version]
- Larsen, M.; Santner, J.; Oburger, E.; Wenzel, W.W.; Glud, R.N. O2 dynamics in the rhizosphere of young rice plants (Oryza sativa L.) as studied by planar optodes. Plant Soil 2015, 390, 279–292. [Google Scholar] [CrossRef] [Green Version]
- Frederiksen, M.S.; Glud, R.N. Oxygen dynamics in the rhizosphere of Zostera marina: A two-dimensional planar optode study. Limnol. Oceanogr. 2006, 51, 1072–1083. [Google Scholar] [CrossRef] [Green Version]
- Jovanovic, Z.; Larsen, M.; Kristensen, E.; Glud, R.; Pedersen, M. Rhizosphere O2 dynamics in young Zostera marina and Ruppia maritima. Mar. Ecol. Prog. Ser. 2015, 518, 95–105. [Google Scholar] [CrossRef] [Green Version]
- Jensen, S.I.; Kühl, M.; Glud, R.N.; Jørgensen, L.B.; Priemé, A. Oxic microzones and radial oxygen loss from roots of Zostera marina. Mar. Ecol. Prog. Ser. 2005, 293, 49–58. [Google Scholar] [CrossRef]
- Glud, R.N. Oxygen dynamics of marine sediments. Mar. Biol. Res. 2008, 4, 243–289. [Google Scholar] [CrossRef]
- Quaranta, M.; Borisov, S.; Klimant, I. Indicators for optical oxygen sensors. Bioanal. Rev. 2012, 4, 115–157. [Google Scholar] [CrossRef] [Green Version]
- Koren, K.; Brodersen, K.E.; Jakobsen, S.L.; Kühl, M. Optical Sensor Nanoparticles in Artificial Sediments–A New Tool To Visualize O2 Dynamics around the Rhizome and Roots of Seagrasses. Environ. Sci. Technol. 2015, 49, 2286–2292. [Google Scholar] [CrossRef]
- Williams, P.N.; Santner, J.; Larsen, M.; Lehto, N.J.; Oburger, E.; Wenzel, W.; Glud, R.N.; Davison, W.; Zhang, H. Localized Flux Maxima of Arsenic, Lead, and Iron around Root Apices in Flooded Lowland Rice. Environ. Sci. Technol. 2014, 48, 8498–8506. [Google Scholar] [CrossRef] [PubMed]
- Zieger, S.E.; Mosshammer, M.; Kühl, M.; Koren, K. Hyperspectral Luminescence Imaging in Combination with Signal Deconvolution Enables Reliable Multi-Indicator-Based Chemical Sensing. ACS Sens. 2021, 6, 183–191. [Google Scholar] [CrossRef] [PubMed]

| Technique | Indication | Advantages | Disadvantages |
|---|---|---|---|
| Methylene blue staining | Qualitative | Cheapest and quickest method; rapid screening approach for a large number of plants | Qualitative nature; unknown detection limit |
| Polarographic cylindrical O2 electrodes | Quantitative | Accurate quantification of very small amounts of radial fluxes of O2 | Use of custom-built equipment is necessary; frequent calibration is needed; time-consuming method |
| Clark-type O2 microelectrodes | Quantitative | Commercially available; rapid and accurate O2 quantification; linear response to O2; resolution to tissue and cell level; appropriate for penetration into tissues | Relatively short lifetime (especially in <10 µm sensors); expensive equipment and frequent calibration needed; signal can be affected by chemical compounds and electrical noise |
| O2 micro-optodes | Quantitative | Commercially available; rapid and precise O2 quantification; resolution to tissue level; long lifetime | Possible interferences with inorganic substances; low signal to noise ratio at high O2 concentrations; not suitable for penetration into tissue |
| Planar optodes | Qualitative and Quantitative | Quantitative and qualitative 2D determination of O2 | Custom-built equipment and technical knowledge needed; non-linear response to O2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiménez, J.d.l.C.; Pellegrini, E.; Pedersen, O.; Nakazono, M. Radial Oxygen Loss from Plant Roots—Methods. Plants 2021, 10, 2322. https://doi.org/10.3390/plants10112322
Jiménez JdlC, Pellegrini E, Pedersen O, Nakazono M. Radial Oxygen Loss from Plant Roots—Methods. Plants. 2021; 10(11):2322. https://doi.org/10.3390/plants10112322
Chicago/Turabian StyleJiménez, Juan de la Cruz, Elisa Pellegrini, Ole Pedersen, and Mikio Nakazono. 2021. "Radial Oxygen Loss from Plant Roots—Methods" Plants 10, no. 11: 2322. https://doi.org/10.3390/plants10112322
APA StyleJiménez, J. d. l. C., Pellegrini, E., Pedersen, O., & Nakazono, M. (2021). Radial Oxygen Loss from Plant Roots—Methods. Plants, 10(11), 2322. https://doi.org/10.3390/plants10112322







