Aster glehni F. Schmidt Extract Modulates the Activities of HMG-CoA Reductase and Fatty Acid Synthase
Abstract
:1. Introduction
2. Results
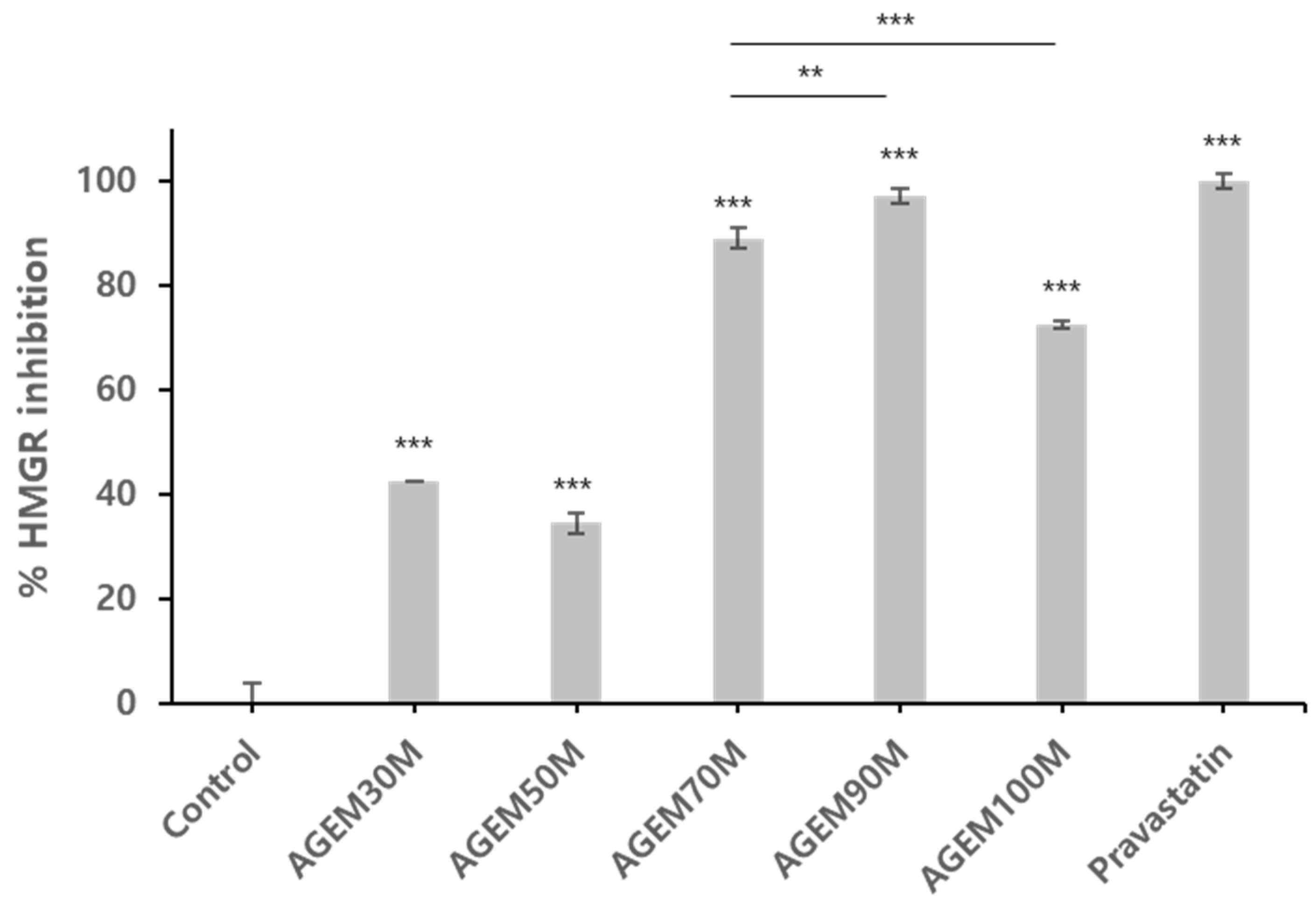
2.1. HMG-CoA Reductase Inhibition Assay
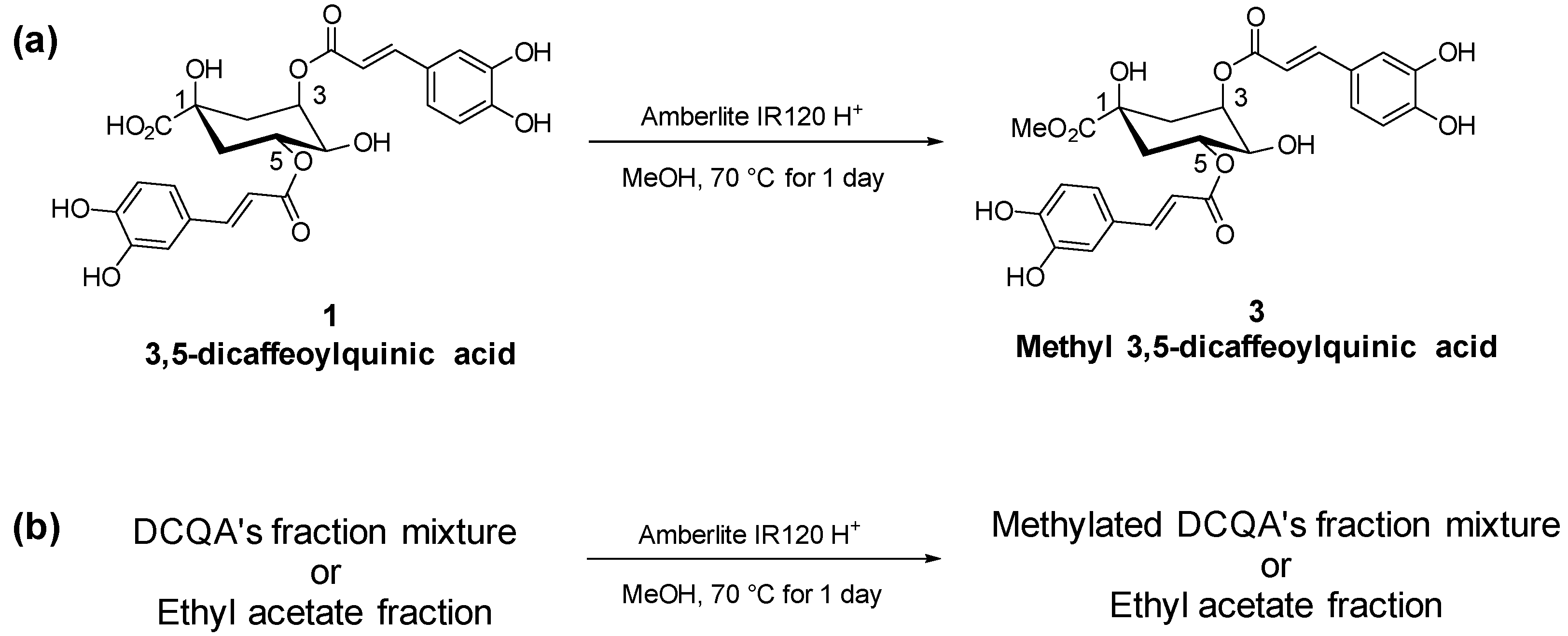
2.2. Esterification of AGE
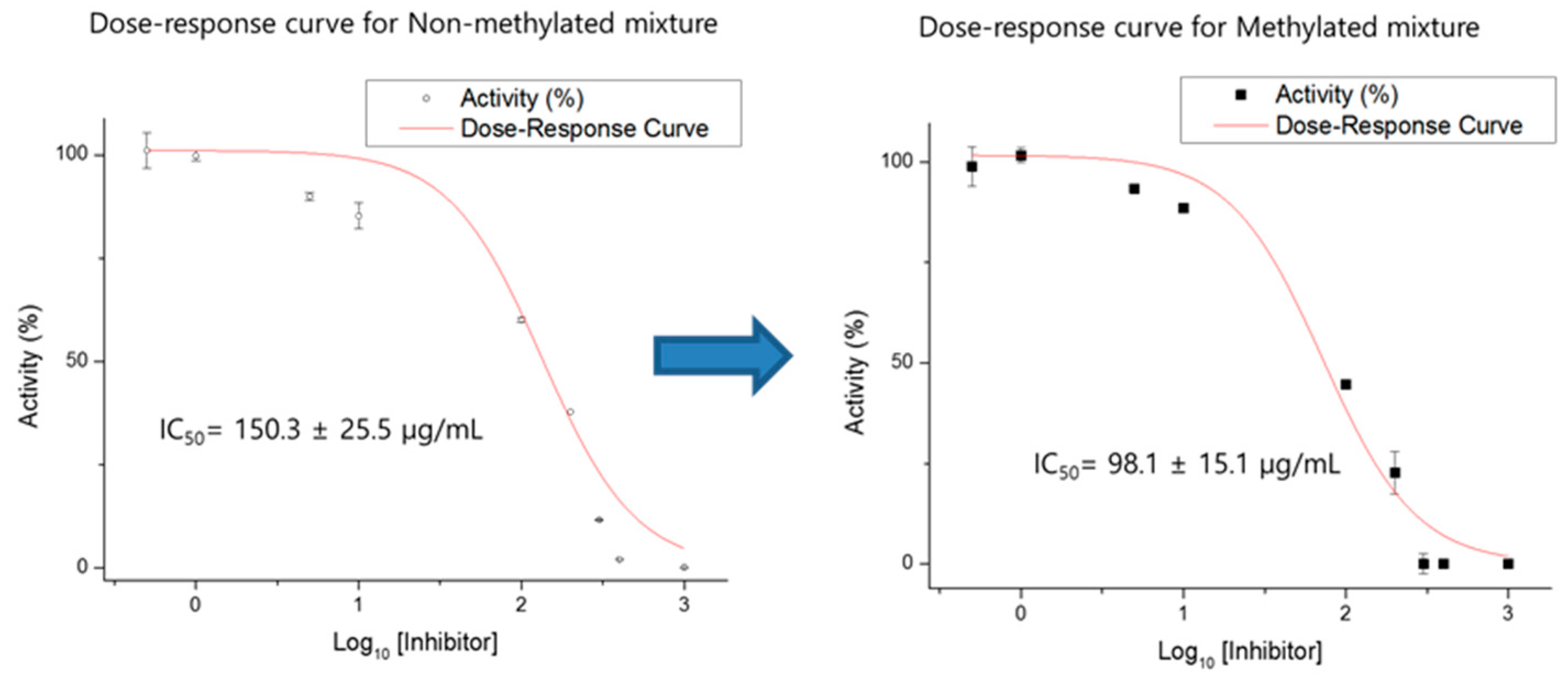
2.3. Concentration Dependent HMG-CoA Reductase Inhibition Assay
2.4. Concentration-Dependent Fatty Acid Synthase Inhibition Assay
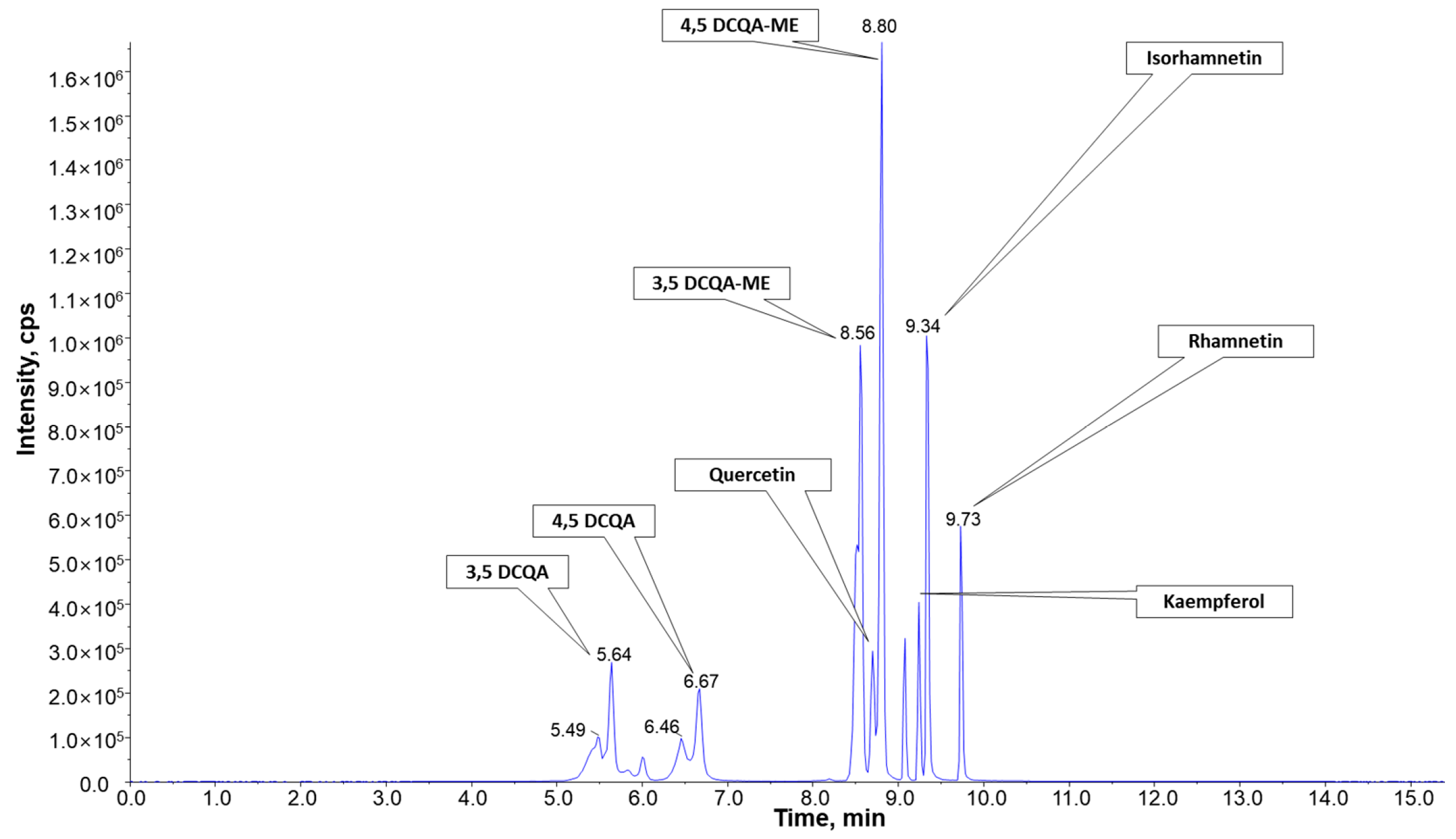
2.5. Qualitative Analysis of AGEM and Quantitative Analysis of AGEM70M
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Instruments
4.3. Plant Materials
4.4. HMG-CoA Reductase (HMGR) Inhibition Assay
4.5. Esterification of the Ethyl Acetate Fractions of AG (AGE)
4.6. Fractionation of the Methylated AGE (AGEM)
4.7. Fatty Acid Synthase (FAS) Inhibition Assay
4.8. Qualitative Analysis of Components in AGEM
4.9. Quantitative Analysis of DCQA and Isoflavonoids in AGEM
| Compound | m/z (Q1, Q3) | |
| DCQA | 517.197 | 163.027 |
| DCQA-ME | 531.177 | 163.000 |
| Rhamnetin | 317.003 | 273.900 |
| Isorhamnetin | 317.003 | 302.000 |
| Kaempferol | 286.977 | 153.000 |
| Quercetin | 302.950 | 229.000 |
| Quercetin-d3 | 306.058 | 232.000 |
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Ekor, M. The growing use of herbal medicines: Issues relating to adverse reactions and challenges in monitoring safety. Front. Pharmacol. 2014, 4, 177. [Google Scholar] [CrossRef] [Green Version]
- Rashrash, M.; Schommer, J.C.; Brown, L.M. Prevalence and predictors of herbal medicine use among adults in the United States. J. Patient Exp. 2017, 4, 108–113. [Google Scholar] [CrossRef] [PubMed]
- WHO. WHO Global Report on Traditional and Complementary Medicine 2019; World Health Organization: Geneva, Switzerland, 2019. [Google Scholar]
- Thomford, N.E.; Senthebane, D.A.; Rowe, A.; Munro, D.; Seele, P.; Maroyi, A.; Dzobo, K. Natural products for drug discovery in the 21st century: Innovations for novel drug discovery. Int. J. Mol. Sci. 2018, 19, 1578. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs from 1981 to 2014. J. Nat. Prod. 2016, 79, 629–661. [Google Scholar] [CrossRef] [Green Version]
- Son, M.J.; Jin, C.; Lee, Y.S.; Lee, J.Y.; Kim, H.J. Characterization of caffeoylglucoside derivatives and hypouricemic activity of the ethyl acetate fraction from Aster glehni. Bull. Korean Chem. Soc. 2015, 36, 503–512. [Google Scholar] [CrossRef]
- Lee, Y.J.; Jang, Y.N.; Han, Y.M.; Kim, H.M.; Jeong, J.M.; Son, M.J.; Jin, C.B.; Kim, H.J.; Seo, H.S. Caffeoylquinic acid-rich extract of Aster glehni F. Schmidt ameliorates nonalcoholic fatty liver through the regulation of PPARδ and adiponectin in ApoE KO Mice. PPAR Res. 2017, 2017, 3912567. [Google Scholar] [CrossRef] [Green Version]
- Lee, H.-M.; Yang, G.; Ahn, T.-G.; Kim, M.-D.; Nugroho, A.; Park, H.-J.; Lee, K.-T.; Park, W.; An, H.-J. Antiadipogenic effects of Aster glehni extract: In vivo and in vitro effects. Evid. Based Complement. Altern. Med. 2013, 2013, 859624. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nugroho, A.; Kim, M.-H.; Choi, J.; Choi, J.S.; Jung, W.T.; Lee, K.-T.; Park, H.-J. Phytochemical studies of the phenolic substances in Aster glehni extract and its sedative and anticonvulsant activity. Arch. Pharmacal Res. 2012, 35, 423–430. [Google Scholar] [CrossRef]
- Seo, S.; Lee, K.-G.; Shin, J.-S.; Chung, E.K.; Lee, J.Y.; Kim, H.J.; Lee, K.-T. 6′-O-Caffeoyldihydrosyringin isolated from Aster glehni suppresses lipopolysaccharide-induced iNOS, COX-2, TNF-α, IL-1β and IL-6 expression via NF-κB and AP-1 inactivation in RAW 264.7 macrophages. Bioorganic Med. Chem. Lett. 2016, 26, 4592–4598. [Google Scholar] [CrossRef]
- Roth, G.A.; Forouzanfar, M.H.; Moran, A.E.; Barber, R.; Nguyen, G.; Feigin, V.L.; Naghavi, M.; Mensah, G.A.; Murray, C.J.L. Demographic and epidemiologic drivers of global cardiovascular mortality. N. Engl. J. Med. 2015, 372, 1333–1341. [Google Scholar] [CrossRef] [Green Version]
- Siri-Tarino, P.W.; Sun, Q.; Hu, F.B.; Krauss, R.M. Saturated fatty acids and risk of coronary heart disease: Modulation by replacement nutrients. Curr. Atheroscler. Rep. 2010, 12, 384–390. [Google Scholar] [CrossRef] [Green Version]
- Yeganeh, B.; Wiechec, E.; Ande, S.R.; Sharma, P.; Moghadam, A.R.; Post, M.; Freed, D.H.; Hashemi, M.; Shojaei, S.; Zeki, A.A.; et al. Targeting the mevalonate cascade as a new therapeutic approach in heart disease, cancer and pulmonary disease. Pharmacol. Ther. 2014, 143, 87–110. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nagarajan, S.R.; Butler, L.M.; Hoy, A.J. The diversity and breadth of cancer cell fatty acid metabolism. Cancer Metab. 2021, 9, 2. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.-Y.; Li, H.; Tang, J.-J.; Wang, J.; Luo, J.; Liu, B.; Wang, J.-K.; Shi, X.-J.; Cui, H.-W.; Tang, J.; et al. Discovery of a potent HMG-CoA reductase degrader that eliminates statin-induced reductase accumulation and lowers cholesterol. Nat. Commun. 2018, 9, 5138. [Google Scholar] [CrossRef] [Green Version]
- De Silva, G.S.; Desai, K.; Darwech, M.; Naim, U.; Jin, X.; Adak, S.; Harroun, N.; Sanchez, L.A.; Semenkovich, C.F.; Zayed, M.A. Circulating serum fatty acid synthase is elevated in patients with diabetes and carotid artery stenosis and is LDL-associated. Atherosclerosis 2019, 287, 38–45. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheng, G.; Palanisamy, A.P.; Evans, Z.P.; Sutter, A.G.; Jin, L.; Singh, I.; May, H.; Schmidt, M.G.; Chavin, K.D. Cerulenin blockade of fatty acid synthase reverses hepatic steatosis in ob/ob mice. PLoS ONE 2013, 8, e75980. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; Hudson, R.; Sintim, H.O. Inhibitors of fatty acid synthesis in prokaryotes and eukaryotes as anti-infective, anticancer and anti-obesity drugs. Future Med. Chem. 2012, 4, 1113–1151. [Google Scholar] [CrossRef] [PubMed]
- Softic, S.; Cohen, D.E.; Kahn, C.R. Role of dietary fructose and hepatic de novo lipogenesis in fatty liver disease. Dig. Dis. Sci. 2016, 61, 1282–1293. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hong, S.; Joo, T.; Jhoo, J.-W. Antioxidant and anti-inflammatory activities of 3,5-dicaffeoylquinic acid isolated from Ligularia fischeri leaves. Food Sci. Biotechnol. 2015, 24, 257–263. [Google Scholar] [CrossRef]
- Rouhi-Boroujeni, H.; Rouhi-Boroujeni, H.; Heidarian, E.; Mohammadizadeh, F.; Rafieian-Kopaei, M. Herbs with anti-lipid effects and their interactions with statins as a chemical anti- hyperlipidemia group drugs: A systematic review. ARYA Atheroscler. 2015, 11, 244–251. [Google Scholar]
- Rouhi-Boroujeni, H.; Rouhi-Boroujeni, H.; Gharipour, M.; Mohammadizadeh, F.; Ahmadi, S.; Rafieian-Kopaei, M. Systematic review on safety and drug interaction of herbal therapy in hyperlipidemia: A guide for internist. Acta Biomed. 2015, 86, 130–136. [Google Scholar] [PubMed]
- Rouhi-Boroujeni, H.; Heidarian, E.; Rouhi-Boroujeni, H.; Deris, F.; Rafieian-Kopaei, M. Medicinal plants with multiple effects on cardiovascular diseases: A systematic review. Curr. Pharm. Des 2017, 23, 999–1015. [Google Scholar] [CrossRef] [PubMed]
- Rafieian-Kopaei, M.; Shahinfard, N.; Rouhi-Boroujeni, H.; Gharipour, M.; Darvishzadeh-Boroujeni, P. Effects of Ferulago angulata extract on serum lipids and lipid peroxidation. Evid. Based Complement. Alternat. Med. 2014, 2014, 680856. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Efferth, T.; Koch, E. Complex interactions between phytochemicals. The multi-target therapeutic concept of phytotherapy. Curr. Drug Targets 2011, 12, 122–132. [Google Scholar] [CrossRef] [PubMed]
- El-Readi, M.Z.; Al-Abd, A.M.; Althubiti, M.A.; Almaimani, R.A.; Al-Amoodi, H.S.; Ashour, M.L.; Wink, M.; Eid, S.Y. Multiple molecular mechanisms to overcome multidrug resistance in cancer by natural secondary metabolites. Front. Pharmacol. 2021, 12, 658513. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Liu, Y.; Zhang, J.-T. A new mechanism of drug resistance in breast cancer cells: Fatty acid synthase overexpression-mediated palmitate overproduction. Mol. Cancer Ther. 2008, 7, 263–270. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, D.; Xu, J.; Yu, G.; Zhang, B.; Wang, H.; Wang, C.; Ru, G.; Sun, A.; Shen, L.; Wei, Q. Expression status of fatty acid synthase (FAS) but not HER2 is correlated with the differentiation grade and prognosis of esophageal carcinoma. Hepatogastroenterology 2013, 60, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Buckley, D.; Duke, G.; Heuer, T.S.; O’Farrell, M.; Wagman, A.S.; McCulloch, W.; Kemble, G. Fatty acid synthase–Modern tumor cell biology insights into a classical oncology target. Pharmacol. Ther. 2017, 177, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Ademosun, A.O.; Oboh, G.; Passamonti, S.; Tramer, F.; Ziberna, L.; Boligon, A.A.; Athayde, M.L. Phenolics from grapefruit peels inhibit HMG-CoA reductase and angiotensin-I converting enzyme and show antioxidative properties in endothelial EA.Hy 926 cells. Food Sci. Hum. Wellness 2015, 4, 80–85. [Google Scholar] [CrossRef] [Green Version]
- Leopoldini, M.; Malaj, N.; Toscano, M.; Sindona, G.; Russo, N. On the inhibitor effects of bergamot juice flavonoids binding to the 3-hydroxy-3-methylglutaryl-CoA reductase (HMGR) enzyme. J. Agric. Food Chem. 2010, 58, 10768–10773. [Google Scholar] [CrossRef]
- Ibrahim, A.; Shafie, N.H.; Mohd Esa, N.; Shafie, S.R.; Bahari, H.; Abdullah, M.A. Mikania micrantha extract inhibits HMG-CoA reductase and ACAT2 and ameliorates hypercholesterolemia and lipid peroxidation in high cholesterol-fed rats. Nutrients 2020, 12, 3077. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.-S.; Lei, J.-P.; Wei, G.-Q.; Chen, H.; Ma, C.-Y.; Jiang, H.-Z. Natural fatty acid synthase inhibitors as potent therapeutic agents for cancers: A review. Pharm. Biol. 2016, 54, 1919–1925. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, H.; Kim, H.J.; Chae, H.; Yoon, N.E.; Jung, B.H. Aster glehni F. Schmidt Extract Modulates the Activities of HMG-CoA Reductase and Fatty Acid Synthase. Plants 2021, 10, 2287. https://doi.org/10.3390/plants10112287
Lee H, Kim HJ, Chae H, Yoon NE, Jung BH. Aster glehni F. Schmidt Extract Modulates the Activities of HMG-CoA Reductase and Fatty Acid Synthase. Plants. 2021; 10(11):2287. https://doi.org/10.3390/plants10112287
Chicago/Turabian StyleLee, Hyunbeom, Hyoung Ja Kim, Hyungi Chae, Na Eun Yoon, and Byung Hwa Jung. 2021. "Aster glehni F. Schmidt Extract Modulates the Activities of HMG-CoA Reductase and Fatty Acid Synthase" Plants 10, no. 11: 2287. https://doi.org/10.3390/plants10112287
APA StyleLee, H., Kim, H. J., Chae, H., Yoon, N. E., & Jung, B. H. (2021). Aster glehni F. Schmidt Extract Modulates the Activities of HMG-CoA Reductase and Fatty Acid Synthase. Plants, 10(11), 2287. https://doi.org/10.3390/plants10112287





