Seed Cryopreservation and Germination of Rhus glabra and the Critically Endangered Species Rhus michauxii
Abstract
1. Introduction
2. Results
2.1. R. glabra Seed Sterilization and Germination Experiments
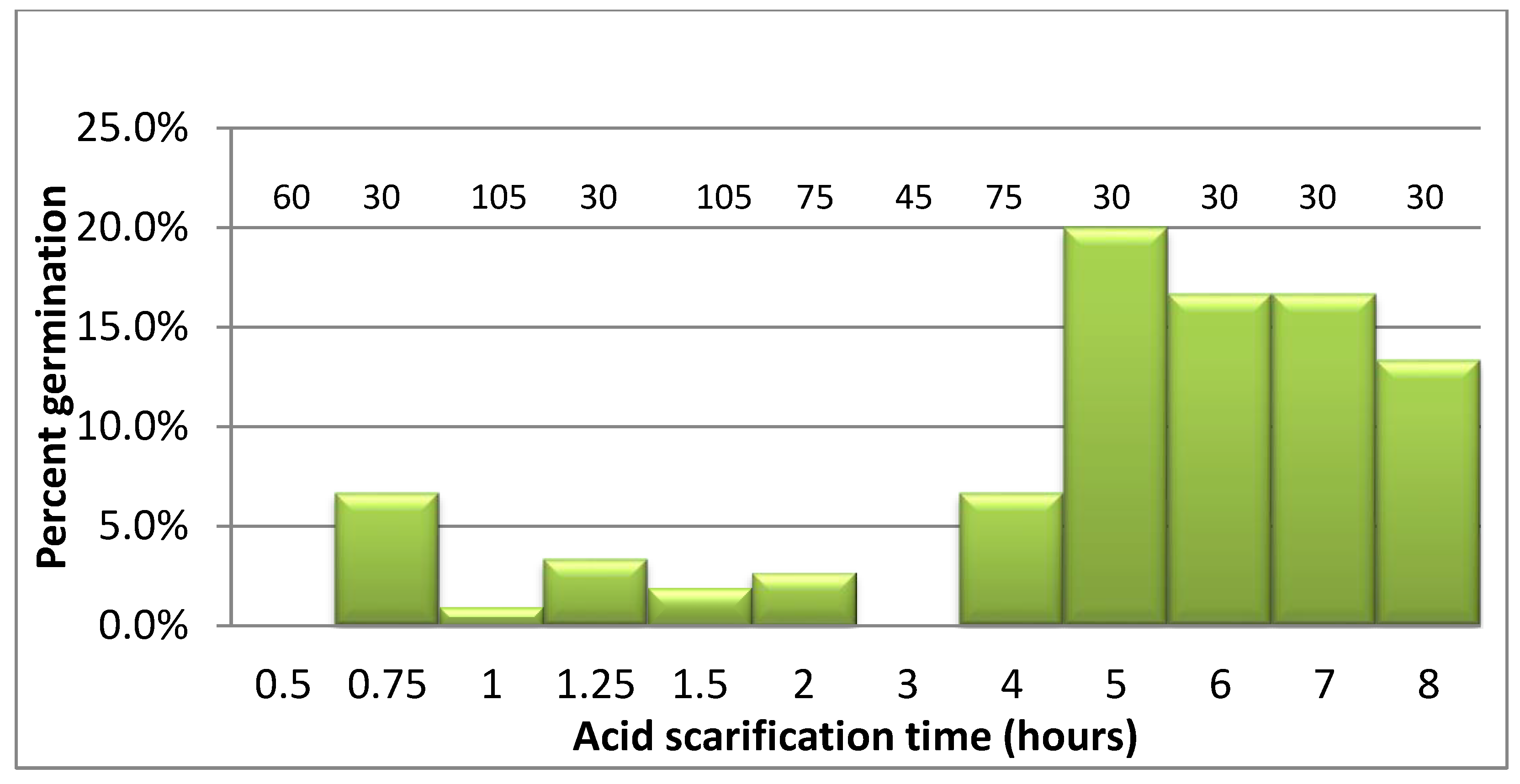
2.2. R. michauxii Seed Scarification, Germination, and Seedling Establishment in Potting Mix
2.3. R. glabra Shoot Culture Experiments
2.4. R. michauxii Bud Sterilization and Multiplication Experiments
2.5. R. michauxii Root Induction In Vitro and Acclimation to Soil
2.6. R. glabra and R. michauxii Seed Cryopreservation in Liquid Nitrogen
3. Discussion
4. Materials and Methods
4.1. Plant Materials, Experimental Design, and Evaluation
4.2. R. glabra Seed Sterilization and Germination Experiments
4.3. R. michauxii Seed Scarification, Germination, and Seedling Establishment in Potting Mix
4.4. R. glabra Shoot Culture Experiments
4.5. R. michauxii Bud Sterilization and Multiplication Experiments
4.6. R. michauxii Root Induction In Vitro and Acclimation to Soil
4.7. R. glabra and R. michauxii Seed Cryopreservation in Liquid Nitrogen
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- USFWS. Endangered and Threatened Wildlife and Plants; Determination of Endangered Status for Rhus michauxii (Michaux’s Sumac). Fed. Regist. 1989, 54, 39853–39857. [Google Scholar]
- Chafin, L.G. Field Guide to the Rare Plants of Georgia; University of Georgia Press: Athens, GA, USA, 2007. [Google Scholar]
- Emrick, V.; Jones, J. Influence of Competition on the Density of the Federally Endangered Michaux’s Sumac (Rhus michauxii) at Fort Pickett, Virginia. Southeast. Nat. 2008, 7, 61–68. [Google Scholar] [CrossRef]
- USFWS; Murdock, N.; Moore, J. Recovery Plan for Michaux’s Sumac (Rhus michauxii) Sargent; US Fish and Wildlife Service: Atlanta, GA, USA, 1993; p. 30.
- GADNR. Element Occurrence Records for Rhus michauxii; Georgia Department of Natural Resources, Wildlife Resources Division: Social Circle, GA, USA, 2020.
- NatureServe. Xerophyllum Asphodeloides|NatureServe Explorer 2.0. NatureServe Explorer: An Online Encyclopedia of Life [Web Application] 2020. Available online: https://explorer.natureserve.org/ (accessed on 20 October 2021).
- Sherman-Broyles, S.L.; Gibson, J.P.; Hamrick, J.L.; Bucher, M.A.; Gibson, M.J. Comparisons of Allozyme Diversity Among Rare and Widespread Rhus Species. Syst. Bot. 1992, 17, 551. [Google Scholar] [CrossRef]
- Burke, J.M.; Hamrick, J.L. Genetic Variation and Evidence of Hybridization in the Genus Rhus (Anacardiaceae). J. Hered. 2002, 93, 37–41. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fay, M.F. Conservation of rare and endangered plants using in vitro methods. Vitr. Cell. Dev. Biol. Anim. 1992, 28, 1–4. [Google Scholar] [CrossRef]
- Sarasan, V.; Cripps, R.; Ramsay, M.M.; Atherton, C.; McMichen, M.; Prendergast, G.; Rowntree, J.K. Conservation In Vitro of Threatened Plants—Progress in the Past Decade. Vitr. Cell. Dev. Biol. Plant 2006, 42, 206–214. [Google Scholar] [CrossRef]
- Reed, B.M.; Sarasan, V.; Kane, M.; Bunn, E.; Pence, V.C. Biodiversity Conservation and Conservation Biotechnology Tools. Vitr. Cell. Dev. Biol. Plant 2011, 47, 1–4. [Google Scholar] [CrossRef]
- Engelmann, F. Cryopreservation of Tropical Plant Germplasm. In Current Research Progress and Application; Japan International Research Center for Agriculture Sciences and International Plant Genetic Resources Institte: Rome, Italy, 2000; pp. 8–20. [Google Scholar]
- Pritchard, H.W. Cryopreservation of Desiccation-Tolerant Seeds. In Cryopreservation and Freeze-Drying Protocols, 2nd ed.; Day, J.G., Stacey, G.N., Eds.; Humana Press: Totowa, NJ, USA, 2007; pp. 185–201. [Google Scholar] [CrossRef]
- Engelmann, F. Use of Biotechnologies for the Conservation of Plant Biodiversity. Vitr. Cell. Dev. Biol. Plant 2011, 47, 5–16. [Google Scholar] [CrossRef]
- Pence, V.C.; Ballesteros, D.; Walters, C.; Reed, B.M.; Philpott, M.; Dixon, K.W.; Pritchard, H.W.; Culley, T.M.; Vanhove, A.-C. Cryobiotechnologies: Tools for Expanding Long-Term Ex Situ Conservation to All Plant Species. Biol. Conserv. 2020, 250, 108736. [Google Scholar] [CrossRef]
- Rayne, S.; Mazza, G. Biological Activities of Extracts from Sumac (Rhus spp.): A Review. Nat. Preced. 2007. [Google Scholar] [CrossRef]
- Djakpo, O.; Yao, W. Rhus Chinensis and Galla Chinensis-Folklore to Modern Evidence: Review. Phytother. Res. 2010, 24, 1739–1747. [Google Scholar] [CrossRef]
- Choi, W.; Jung, H.; Kim, K.; Lee, S.; Yoon, S.; Park, J.; Kim, S.; Cheon, S.; Eo, W.; Lee, S. Rhus Verniciflua Stokes against Advanced Cancer: A Perspective from the Korean Integrative Cancer Center. J. Biomed. Biotechnol. 2012, 2012, 874276. [Google Scholar] [CrossRef]
- Shabbir, A. Rhus Coriaria Linn, a Plant of Medicinal, Nutritional and Industrial Importance: A Review. J. Anim. Plant Sci. 2012, 22, 505–512. [Google Scholar]
- Martinetti, P.J. Method of Treating Respiratory Disease in Animals. U.S. Patent 4,654,214, 31 March 1987. [Google Scholar]
- Warren, L.E. Rhus michauxii—A Non-Poisonous Plant. Am. J. Pharm. 1910, 82, 499. [Google Scholar]
- Li, X.; Baskin, J.M.; Baskin, C.C. Anatomy of Two Mechanisms of Breaking Physical Dormancy by Experimental Treatments in Seeds of Two North American Rhus species (Anacardiaceae). Am. J. Bot. 1999, 86, 1505–1511. [Google Scholar] [CrossRef] [PubMed]
- Baskin, C.C.; Baskin, J.M. Seeds: Ecology, Biogeography, and, Evolution of Dormancy and Germination; Elsevier: Amsterdam, The Netherlands, 1998. [Google Scholar]
- Farmer, R.E.; Farmer, R.E.; Lockley, G.C.; Cunningham, M. Germination Patterns of the Sumacs, Rhus Glabra and Rhus Coppalina: Effects of Scarification Time, Temperature and Genotype. Seed Sci. Technol. Neth. 1982, 10, 223–231. [Google Scholar]
- Li, X.; Baskin, J.M.; Baskin, C.C. Seed Morphology and Physical Dormancy of Several North American Rhus Species (Anacardiaceae). Seed Sci. Res. 1999, 9, 247–258. [Google Scholar] [CrossRef]
- Hartmann, H.T.; Kester, D.E.; Davies, F.T., Jr.; Geneve, R.I. Hartman and Kester’s Plant Propagation: Principles and Practices, 8th ed.; Prentice-Hall: Upper Saddle River, NJ, USA, 2011. [Google Scholar]
- Dumroese, R.K.; Luna, T.; Landis, T.D. Nursery Manual for Native Plants: A Guide for Tribal Nurseries—Volume 1: Nursery Management. In Agriculture Handbook; U.S. Department of Agriculture Forestry Service: Washington, DC, USA, 2009; Volume 302, p. 730. [Google Scholar]
- Brinkman, K.A.; Rhus, L. Seeds of Woody Plants in the United States; Schopmeyer, C.S., Ed.; U.S. Department of Agriculture Forestry Service: Washington, DC, USA, 1974.
- Yi, T.; Miller, A.J.; Wen, J. Phylogenetic and Biogeographic Diversification of Rhus (Anacardiaceae) in the Northern Hemisphere. Mol. Phylogenet. Evol. 2004, 33, 861–879. [Google Scholar] [CrossRef]
- Wilkinson, C.A.; DeMarco, H.A.; Jones, L.J. Report to the United States Department of the Army: Viability, Germination and Propagation of Rhus Michauxii at Fort Pickett; Virginia Tech Southern Piedmont Agricultural Research Station: Blacksburg, VA, USA, 1996. [Google Scholar]
- Bolin, J.F.; Jones, M.E.; Musselman, L.J. Germination of the Federally Endangered Michaux’s Sumac (Rhus michauxii). Nativ. Plants J. 2011, 12, 119–122. [Google Scholar] [CrossRef]
- Welfare, R.B.; Proctor, J.D.; Jones, M.E.; Bolin, J.F. The Germination Ecology of the Federally Endangered Michaux’s Sumac (Rhus michauxii, Anacardiaceae) Including Endozoochory. In Proceedings of the 73rd Annual meeting of the Association of Southeastern Biologists, University of Georgia, Athens, GA, USA, 4–7 April 2012; p. 205. [Google Scholar]
- Braham, R.; Murray, C.; Boyer, M. Mitigating Impacts to Michaux’s Sumac (Rhus michauxii Sarg.): A Case Study of Transplanting an Endangered Shrub. Castanea 2006, 71, 265–271. [Google Scholar] [CrossRef]
- Emrick, V.I.; Fields, M.; Fitzpatrick, J. Removal, Propagation, and Transplantation of Michaux’s Sumac (Rhus michauxii) Colonies from the Infantry Platoon Battle Course, Fort Pickett–Maneuver Training Center, Virginia; Conservation Management Institute: Blacksburg, VA, USA, 2018; Available online: https://vtechworks.lib.vt.edu/bitstream/handle/10919/95906/Final%20Report_Removal%2C%20Propagation%2C%20and%20Transplant%20of%20Michaux%27s%20Sumac.pdf (accessed on 20 October 2021).
- Shirgurkar, M.V.; John, C.K.; Nadgauda, R.S. Factors affecting in vitro microrhizome production in turmeric. Plant Cell Tissue Organ Cult. 2001, 64, 5–11. [Google Scholar] [CrossRef]
- Nayak, S.; Naik, P.K. Factors Effecting in Vitro Microrhizome Formation and Growth in Curcuma Longa, L. and Improved Field Performance of Micropropagated Plants. 2006. Available online: http://scienceasia.org/2006.32.n1/v32_031_037.pdf (accessed on 20 October 2021).
- Pence, V.C. Cryopreservation of Seeds of Ohio Native Plants and Related Species. Seed Sci. Technol. 1991, 19, 235–251. [Google Scholar]
- Engelmann, F. Cryopreservation of Embryos: An Overview. In Plant Embryo Culture: Methods and Protocols; Thorpe, T.A., Yeung, E.C., Eds.; Humana Press: Totowa, NJ, USA, 2011; Volume 710, pp. 155–184. [Google Scholar] [CrossRef]
- Salomão, A.N. Tropical Seed Species’ Responses to Liquid Nitrogen Exposure. Braz. J. Plant Physiol. 2002, 14, 133–138. [Google Scholar] [CrossRef]
- Khanna, S.; Jenkins, H.; Bucalo, K.; Determann, R.O.; Cruse-Sanders, J.M.; Pullman, G.S. Effects of Seed Cryopreservation, Stratification and Scarification on Germination for Five Rare Species of Pitcher Plants. Cryoletters 2014, 35, 29–39. [Google Scholar]
- Hardin, J.W.; Phillips, L.L. Hybridization in Eastern North American Rhus (Anacardiaceae). ASB Bull. 1985, 32, 99–106. [Google Scholar]
- Murashige, T.; Skoog, F. A Revised Medium for Rapid Growth and Bio Assays with Tobacco Tissue Cultures. Physiol. Plant. 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Lloyd, G.; McCown, B. Commercially-Feasible Micropropagation of Mountain Laurel, Kalmia Latifolia, by Use of Shoot-Tip Culture. Combined Proceedings, International Plant Propagators’ Society 1980, 30, 421–427. [Google Scholar]
- Rowe, B.; Blazich., F.A. Anacardiaceae–Sumac family Rhus L. sumac. In Woody Plant Seed Manual-Agric. Handbook No. 727. Washington, DC. U.S. Department of Agriculture, Forest Service. 1223 p; Bonner, F.T., Karrfalt, P., Eds.; United States Department of Agriculture: Washington, DC, USA, 2008; pp. 954–960. [Google Scholar]
- Ma, X.; Bucalo, K.; Determann, R.O.; Cruse-Sanders, J.M.; Pullman, G.S. Somatic Embryogenesis, Plant Regeneration, and Cryopreservation for Torreya Taxifolia, a Highly Endangered Coniferous Species. Vitr. Cell. Dev. Biol. Plant 2012, 48, 324–334. [Google Scholar] [CrossRef]
- Pullman, G.S.; Zeng, X.; Copeland-Kamp, B.; Crockett, J.; Lucrezi, J.; May, S.W.; Bucalo, K. Conifer Somatic Embryogenesis: Improvements by Supplementation of Medium with Oxidation–Reduction Agents. Tree Physiol. 2015, 35, 209–224. [Google Scholar] [CrossRef] [PubMed]







| Seed Source and Origin | No. of Seeds Collected | Seeds Sinking (%) | No. (%) of Germinants after 1 Month | No. (%) of Contaminated Seeds after 1 Month | Seedlings Planted in Potting Soil | Seedlings Surviving after 1 Month |
|---|---|---|---|---|---|---|
| Source 4—LBRWMA, females near the male plants | 515 | 10.3 | 22/53 (39.6) | 14/53 (26.4) | 16 | 11 |
| Source 5—LBRWMA, females away from the male plants | 243 | 0.4 | 0/1 (0) | 0/1 (0) | 0 | 0 |
| Source 6—Zoo Atlanta rooftop garden | 2600 | 0.3 | 1/8 (12.5) | 1/8 (12.5) | 1 | 1 |
| Seed Cryopreservation |
|---|
| (1) Select seeds sinking in distilled water, dry seeds; (2) Determine if seed moisture content is below 14% and further dry, if needed; (3) Place seeds in cryogenic storage vials; (4) Rapidly immerse vials in liquid nitrogen (LN); (5) Remove vials from LN and re-warm in a 37 °C water bath for 1–2 min; (6) Germinate seeds in vitro. |
| Seed germination in vitro (production of seedlings for planting or shoots for micropropagation) |
| (1) Scarify non-cryopreserved or cryopreserved seeds in concentrated sulfuric acid for 6 h; (2) Rinse seeds in sterile distilled water three times; (3) Place on the ½ MS medium without PGRs in sterile containers; (4) Incubate in light at room temperature; (5) Remove germinants for planting or micropropagation. |
| Germinant/seedling establishment in the potting mix |
| (1) Select germinating seeds when the radicle becomes visible; (2) Carefully remove from agar, rinse in tap water, and plant in the potting mix; (3) Cover with humidity domes for about 2 weeks. Water every other day. Transfer to larger pots after about 3 months. |
| Species | Plant Part and Source | Collection Site | Collection Time | Pollination | Notes |
|---|---|---|---|---|---|
| R. glabra | seeds | SSCN | Oct 2010 | Natural | Wild plants |
| R. michauxii | buds | ABG | Oct, Nov 2010 | Buds and shoot tips from cuttings of a female plant from the natural population at Covington, GA | |
| R. michauxii | Seeds 1 | NCBG | 2005 | Natural | Seeds were stored in the NCBG Seed Banking Program at −18 °C |
| R. michauxii | Seeds 2 | LBRWMA | Dec 2012 | Natural | From female cuttings planted near natural male plants at the LBRWMA |
| R. michauxii | Seeds 3 | SSCN | Oct 2013 | Hand-pollinated | From female cuttings mixed with male cuttings planted in the SSCN |
| R. michauxii | Seeds 4 | LBRWMA | Sept 2018 | Natural | Seeds collected from cuttings of females planted near natural male plants at the LBRWMA |
| R. michauxii | Seeds 5 | LBRWMA | Sept 2018 | Natural | Seeds collected from females away from male plants at the LBRWMA |
| R. michauxii | Seeds 6 | Atlanta Zoo | Sept 2018 | Natural | Seeds collected from female cuttings planted in 2015 with male cuttings |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pullman, G.S.; Bucalo, K.; Determann, R.O.; Cruse-Sanders, J.M. Seed Cryopreservation and Germination of Rhus glabra and the Critically Endangered Species Rhus michauxii. Plants 2021, 10, 2277. https://doi.org/10.3390/plants10112277
Pullman GS, Bucalo K, Determann RO, Cruse-Sanders JM. Seed Cryopreservation and Germination of Rhus glabra and the Critically Endangered Species Rhus michauxii. Plants. 2021; 10(11):2277. https://doi.org/10.3390/plants10112277
Chicago/Turabian StylePullman, Gerald S., Kylie Bucalo, Ron O. Determann, and Jennifer M. Cruse-Sanders. 2021. "Seed Cryopreservation and Germination of Rhus glabra and the Critically Endangered Species Rhus michauxii" Plants 10, no. 11: 2277. https://doi.org/10.3390/plants10112277
APA StylePullman, G. S., Bucalo, K., Determann, R. O., & Cruse-Sanders, J. M. (2021). Seed Cryopreservation and Germination of Rhus glabra and the Critically Endangered Species Rhus michauxii. Plants, 10(11), 2277. https://doi.org/10.3390/plants10112277






