The Function of Flavonoids in the Diurnal Rhythm under Rapidly Changing UV Conditions—A Model Study on Okra
Abstract
1. Introduction
2. Results
2.1. Morphological Parameters
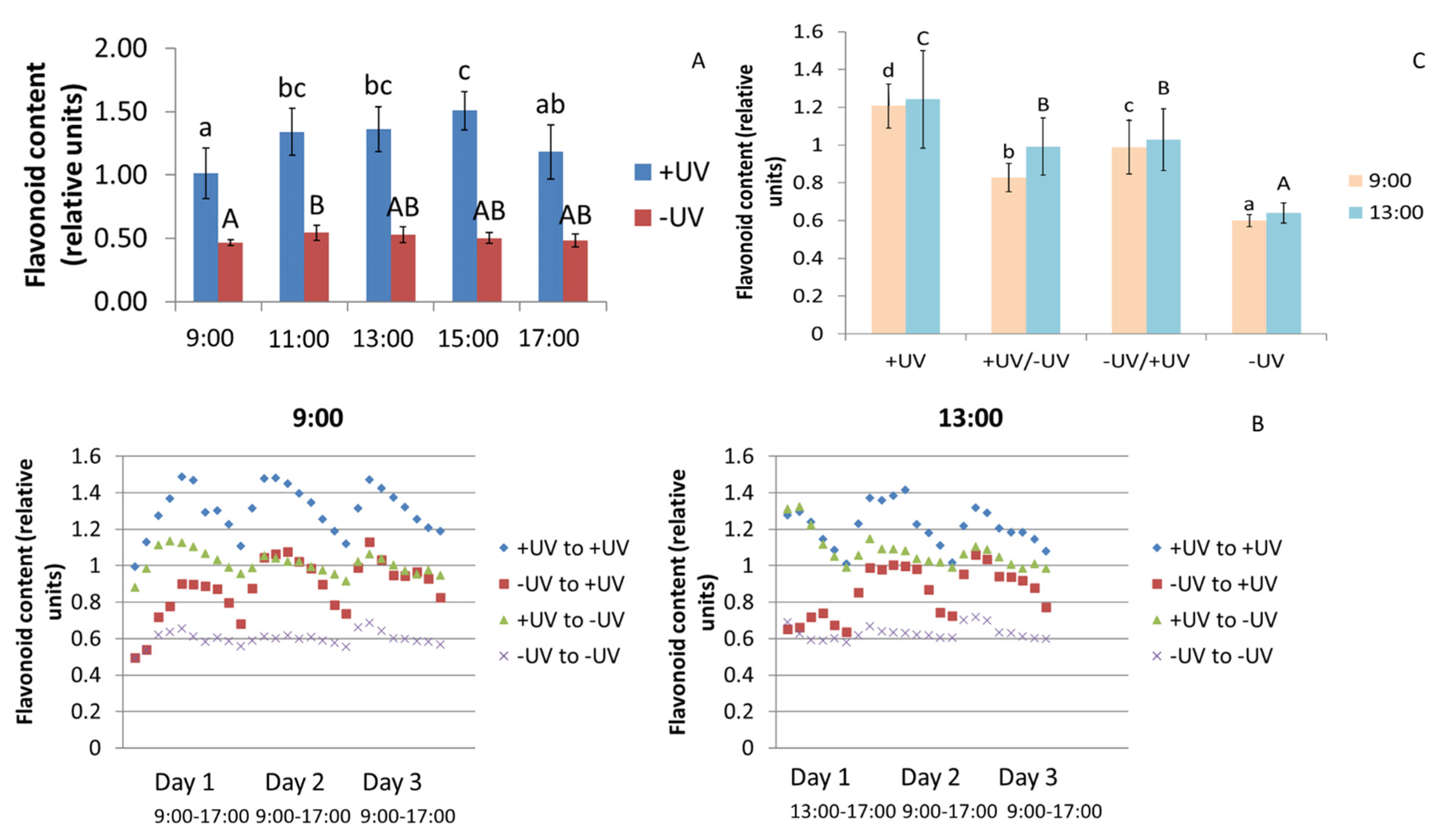
2.2. Flavonoids
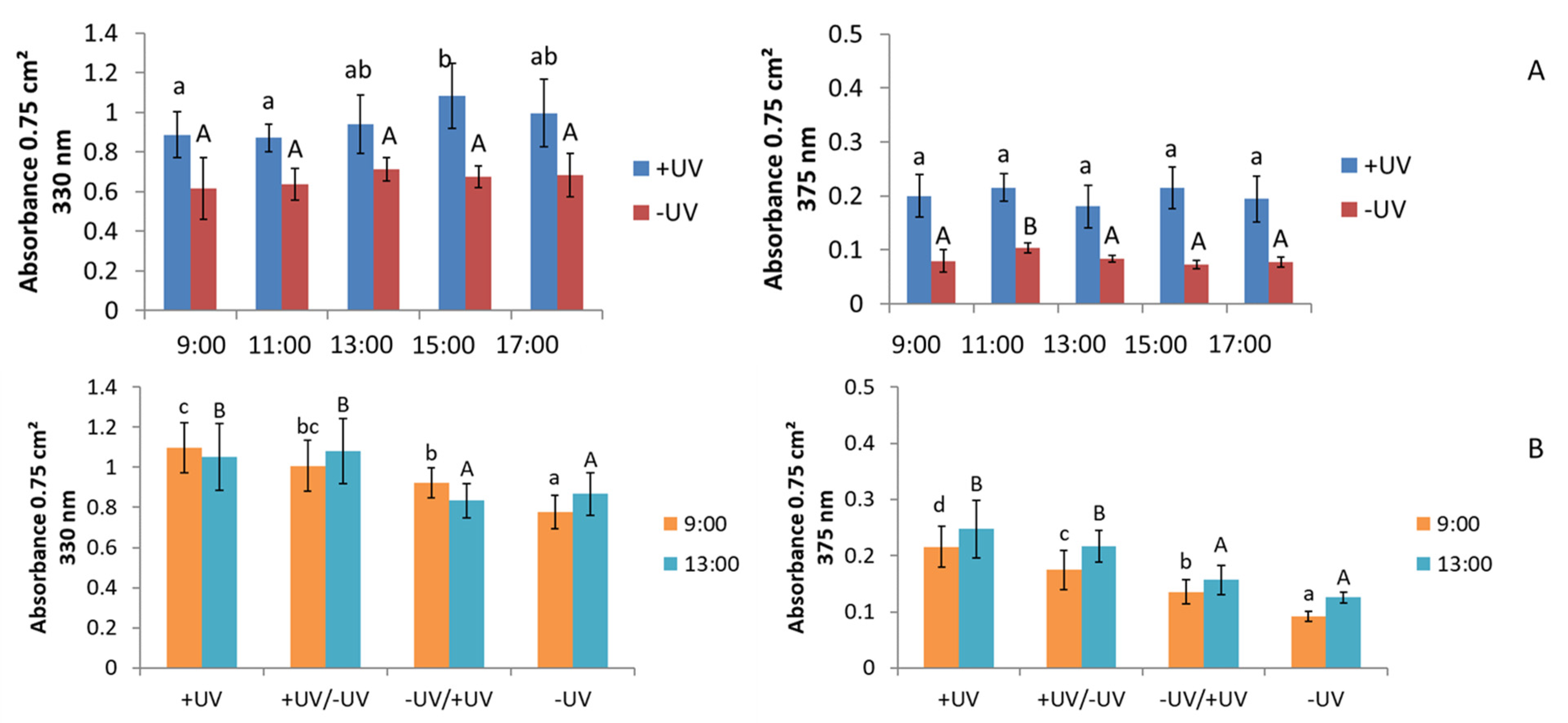
2.3. UV-Absorption
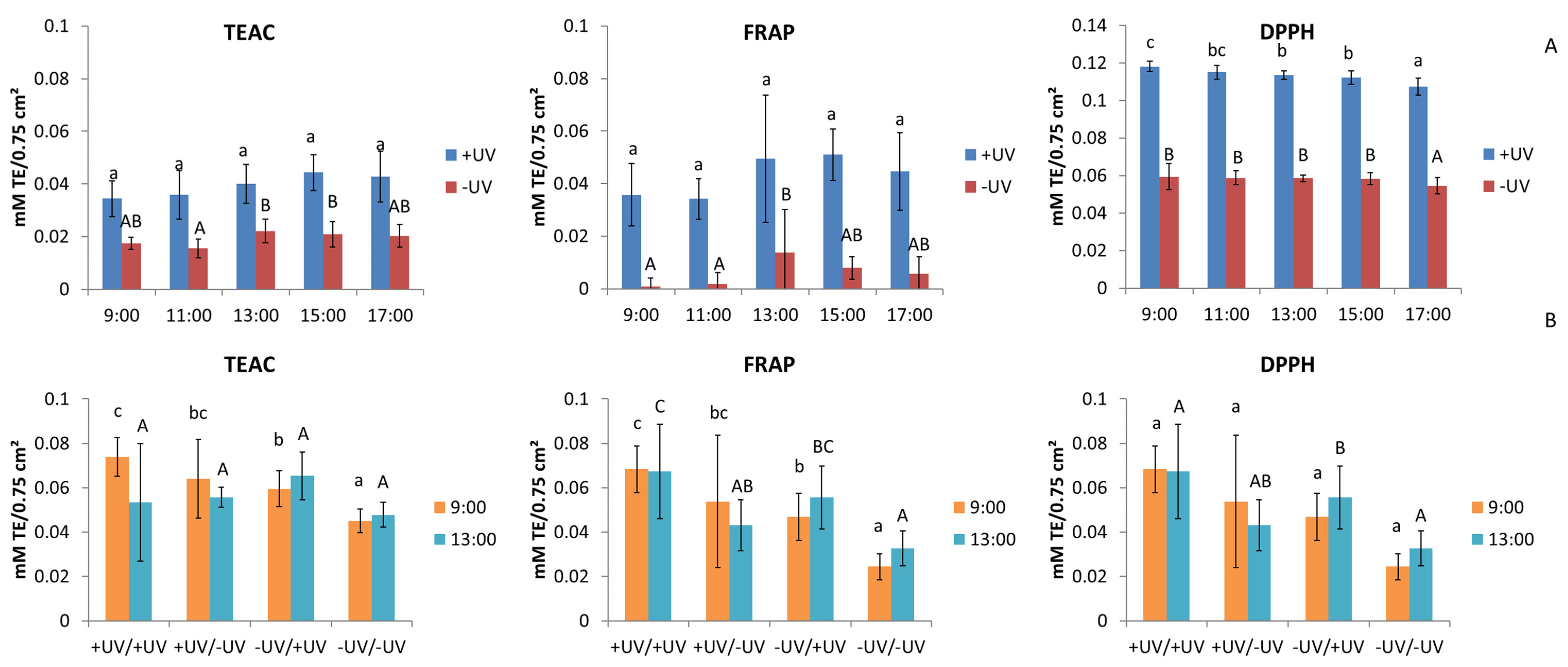
2.4. Antioxidant Activity
2.5. Relation of Flavonoids and Function in the Diurnal Rhythm
3. Discussion
3.1. Morphological Parameters
3.2. Flavonoids
3.3. UV-Absorption
3.4. Antioxidant Activity
3.5. Relation of Flavonoids and Function in the Diurnal Rhythm
4. Materials and Methods
4.1. Plant Experiments
4.1.1. Experiment 1: Diurnal Rhythm
4.1.2. Experiment 2: Changes in Diurnal Rhythm Due to Transfer to the Opposite UV Condition
4.2. Morphological Parameters
4.3. Flavonoid Content
4.4. Extraction and Chemical Analysis of Flavonoid Glycosides and Hydroxycinnamic Acid Derivatives
4.5. UV-Absorbance
4.6. Antioxidant Activity
4.6.1. The TEAC Assay (Trolox Euivalent Antioxidant Capacity) was Performed with the Following Method:
4.6.2. The FRAP Assay (Ferric Reducing Antioxidant Power) was Performed with the Following Method:
4.6.3. The DPPH Assay (2,2-Diphenyl-1-picrylhydrazyl Assay) Was Performed with the Following Method:
4.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yan, Y.; Stoddard, F.L.; Neugart, S.; Sadras, V.O.; Lindfors, A.; Morales, L.O.; Aphalo, P.J. Responses of flavonoid profile and associated gene expression to solar blue and UV radiation in two accessions of Vicia faba L. from contrasting UV environments. Photochem. Photobiol. Sci. 2019, 18, 434–447. [Google Scholar] [CrossRef]
- Neale, R.E.; Barnes, P.W.; Robson, T.M.; Neale, P.J.; Williamson, C.E.; Zepp, R.G.; Wilson, S.R.; Madronich, S.; Andrady, A.L.; HeikkilÃ, A.M.; et al. Environmental effects of stratospheric ozone depletion, UV radiation, and interactions with climate change: UNEP Environmental Effects Assessment Panel, Update 2020. Photochem. Photobiol. Sci. 2021, 20, 1–67. [Google Scholar] [CrossRef]
- Neugart, S.; Schreiner, M. UVB and UVA as eustressors in horticultural and agricultural crops. Sci. Hortic. 2018, 234, 370–381. [Google Scholar] [CrossRef]
- Majer, P.; Neugart, S.; Krumbein, A.; Schreiner, M.; Hideg, É. Singlet oxygen scavenging by leaf flavonoids contributes to sunlight acclimation in Tilia platyphyllos. Environ. Exp. Bot. 2014, 100, 1–9. [Google Scholar] [CrossRef]
- Fiol, M.; Weckmüller, A.; Neugart, S.; Schreiner, M.; Rohn, S.; Krumbein, A.; Kroh, L.W. Thermal-induced changes of kale’s antioxidant activity analyzed by HPLC–UV/Vis-online-TEAC detection. Food Chem. 2013, 138, 857–865. [Google Scholar] [CrossRef] [PubMed]
- Neugart, S.; Fiol, M.; Schreiner, M.; Rohn, S.; Zrenner, R.; Kroh, L.W.; Krumbein, A. Interaction of moderate UV-B exposure and temperature on the formation of structurally different flavonol glycosides and hydroxycinnamic acid derivatives in Kale (Brassica oleracea var. sabellica). J. Agric. Food Chem. 2014, 62, 4054–4062. [Google Scholar] [CrossRef]
- Harbaum-Piayda, B.; Walter, B.; Bengtsson, G.B.; Hubbermann, E.M.; Bilger, W.; Schwarz, K. Influence of pre-harvest UV-B irradiation and normal or controlled atmosphere storage on flavonoid and hydroxycinnamic acid contents of pak choi (Brassica campestris L. ssp chinensis var. communis). Postharvest Biol. Technol. 2010, 56, 202–208. [Google Scholar] [CrossRef]
- Olsson, L.C.; Veit, M.; Weissenbock, G.; Bornman, J.F. Differential flavonoid response to enhanced UV-B radiation in Brassica napus. Phytochemistry 1998, 49, 1021–1028. [Google Scholar] [CrossRef]
- Neugart, S.; Tobler, M.A.; Barnes, P.W. Different irradiances of UV and PAR in the same ratios alter the flavonoid profiles of Arabidopsis thaliana wild types and UV-signalling pathway mutants. Photochem. Photobiol. Sci. 2019, 18, 1685–1699. [Google Scholar] [CrossRef] [PubMed]
- Csepregi, K.; Neugart, S.; Schreiner, M.; Hideg, E. Comparative evaluation of total antioxidant capacities of plant polyphenols. Molecules 2016, 21, 208. [Google Scholar] [CrossRef]
- Agati, G.; Cerovic, Z.G.; Pinelli, P.; Tattini, M. Light-induced accumulation of ortho-dihydroxylated flavonoids as non-destructively monitored by chlorophyll fluorescence excitation techniques. Environ. Exp. Bot. 2011, 73, 3–9. [Google Scholar] [CrossRef]
- Zietz, M.; Weckmuller, A.; Schmidt, S.; Rohn, S.; Schreiner, M.; Krumbein, A.; Kroh, L.W. Genotypic and climatic influence on the antioxidant activity of flavonoids in kale (Brassica oleracea var. sabellica). J. Agric. Food Chem. 2010, 58, 2123–2130. [Google Scholar] [CrossRef] [PubMed]
- Moreira-Rodríguez, M.; Nair, V.; Benavides, J.; Cisneros-Zevallos, L.; Jacobo-Velázquez, D.A. UVA, UVB light doses and harvesting time differentially tailor glucosinolate and phenolic profiles in broccoli sprouts. Molecules 2017, 22, 1065. [Google Scholar] [CrossRef]
- Rechner, O.; Neugart, S.; Schreiner, M.; Wu, S.; Poehling, H.M. Different narrow-band light ranges alter plant secondary metabolism and plant defense response to aphids. J. Chem. Ecol. 2016, 42, 989–1003. [Google Scholar] [CrossRef]
- Bumke-Vogt, C.; Osterhoff, M.A.; Borchert, A.; Guzman-Perez, V.; Sarem, Z.; Birkenfeld, A.L.; Bähr, V.; Pfeiffer, A.F.H. The Flavones Apigenin and Luteolin Induce FOXO1 Translocation but Inhibit Gluconeogenic and Lipogenic Gene Expression in Human Cells. PLoS ONE 2014, 9, e104321. [Google Scholar] [CrossRef]
- Pan, M.H.; Lai, C.S.; Ho, C.T. Anti-inflammatory activity of natural dietary flavonoids. Food Funct. 2010, 1, 15–31. [Google Scholar] [CrossRef]
- Chen, A.Y.; Chen, Y.C. A review of the dietary flavonoid, kaempferol on human health and cancer chemoprevention. Food Chem. 2013, 138, 2099–2107. [Google Scholar] [CrossRef] [PubMed]
- Karakoltsidis, P.A.; Constantinides, S.M. Okra seeds: A new protein source. J. Agric. Food Chem. 1975, 23, 1204–1207. [Google Scholar] [CrossRef]
- Barnes, P.W.; Flint, S.D.; Slusser, J.R.; Gao, W.; Ryel, R.J. Diurnal changes in epidermal UV transmittance of plants in naturally high UV environments. Physiol. Plant. 2008, 133, 363–372. [Google Scholar] [CrossRef] [PubMed]
- Nybakken, L.; Aubert, S.; Bilger, W. Epidermal UV-screening of arctic and alpine plants along a latitudinal gradient in Europe. Polar Biol. 2004, 27, 391–398. [Google Scholar] [CrossRef]
- Barnes, P.W.; Flint, S.D.; Tobler, M.A.; Ryel, R.J. Diurnal adjustment in ultraviolet sunscreen protection is widespread among higher plants. Oecologia 2016, 181, 55–63. [Google Scholar] [CrossRef]
- Arapitsas, P. Identification and quantification of polyphenolic compounds from okra seeds and skins. Food Chem. 2008, 110, 1041–1045. [Google Scholar] [CrossRef] [PubMed]
- Neugart, S.; Tobler, M.A.; Barnes, P.W. Rapid adjustment in epidermal UV sunscreen: Comparison of optical measurement techniques and response to changing solar UV radiation conditions. Physiol. Plant. 2021, 173, 725–735. [Google Scholar] [CrossRef]
- Agati, G.; Brunetti, C.; Di Ferdinando, M.; Ferrini, F.; Pollastri, S.; Tattini, M. Functional roles of flavonoids in photoprotection—New evidence, lessons from the past. Plant Physiol. Biochem. 2013, 72, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Caldwell, M.M.; Flint, S.D.; Searles, P.S. Spectral balance and UV-B sensitivity of soybean: A field experiment. Plant Cell Environ. 1994, 17, 267–276. [Google Scholar] [CrossRef]
- Jansen, M.A.K. Ultraviolet-B radiation effects on plants: Induction of morphogenic responses. Physiol. Plant. 2002, 116, 423–429. [Google Scholar] [CrossRef]
- Kolb, C.A.; Kopecky, J.; Riederer, M.; Pfuendel, E.E. UV screening by phenolics in berries of grapevine (Vitis vinifera). Funct. Plant Biol. 2003, 30, 1177–1186. [Google Scholar] [CrossRef]
- Wang, Q.-W.; Robson, T.M.; Pieristè, M.; Oguro, M.; Oguchi, R.; Murai, Y.; Kurokawa, H. Testing trait plasticity over the range of spectral composition of sunlight in forb species differing in shade tolerance. J. Ecol. 2020, 108, 1923–1940. [Google Scholar] [CrossRef]
- Siipola, S.M.; Kotilainen, T.; Sipari, N.; Morales, L.O.; Lindfors, A.V.; Robson, T.M.; Aphalo, P.J. Epidermal UV-A absorbance and whole-leaf flavonoid composition in pea respond more to solar blue light than to solar UV radiation. Plant Cell Environ. 2015, 38, 941–952. [Google Scholar] [CrossRef]
- Barnes, P.W.; Flint, S.D.; Ryel, R.J.; Tobler, M.A.; Barkley, A.E.; Wargent, J.J. Rediscovering leaf optical properties: New insights into plant acclimation to solar UV radiation. Plant Physiol. Biochem. 2015, 93, 94–100. [Google Scholar] [CrossRef]
- Barnes, P.W.; Tobler, M.A.; Keefover-Ring, K.; Flint, S.D.; Barkley, A.E.; Ryel, R.J.; Lindroth, R.L. Rapid modulation of ultra-violet shielding in plants is influenced by solar ultraviolet radiation and linked to alterations in flavonoids. Plant Cell Envron. 2016, 39, 222–230. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, J.H.; Gitz, D.C., 3rd; Liu-Gitz, L.; Xu, C.; Gao, W.; Slusser, J. Coupling short-term changes in ambient UV-B levels with induction of UV-screening compounds. Photochem. Photobiol. 2007, 83, 863–870. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Lu, M.-F.; Liao, H.-B.; Li, Y.-X.; Han, W.; Yuan, K. Content determination of the flavonoids in the different parts and different species of Abelmoschus esculentus L. by reversed phase-high performance liquid chromatograph and colorimetric method. Pharm. Mag. 2014, 10, 278–284. [Google Scholar] [CrossRef]
- Fan, L.; Fang, C.; Dubé, C.; Tremblay, N.; Khanizadeh, S.; Canada, A. A non-destructive method to predict polyphenol content in strawberry. J. Food Agric. Environ. 2011, 9, 59–62. [Google Scholar]
- Scordino, M.; Sabatino, L.; Lazzaro, F.; Borzì, M.; Gargano, M.; Traulo, P.; Gagliano, G. Blood Orange Anthocyanins in Fruit Beverages: How the Commercial Shelf Life Reflects the Quality Parameter. Beverages 2015, 1, 82–94. [Google Scholar] [CrossRef]
- dos Nascimento, L.B.; Brunetti, C.; Agati, G.; Lo Iacono, C.; Detti, C.; Giordani, E.; Ferrini, F.; Gori, A. Short-Term Pre-Harvest UV-B Supplement Enhances the Polyphenol Content and Antioxidant Capacity of Ocimum basilicum Leaves during Storage. Plants 2020, 9, 797. [Google Scholar] [CrossRef] [PubMed]
- Agati, G.; Cerovic, Z.G.; Marta, A.D.; Di Stefano, V.; Pinelli, P.; Traversi, M.L.; Orlandini, S. Optically-assessed preformed flavonoids and susceptibility of grapevine to Plasmopara viticola under different light regimes. Funct. Plant Biol. 2008, 35, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Liu, X.; Ma, Y.; Zhang, R.; Cao, Q.; Zhu, Y.; Cao, W.; Tian, Y. A Comparative Assessment of Measures of Leaf Nitrogen in Rice Using Two Leaf-Clip Meters. Sensors 2019, 20, 175. [Google Scholar] [CrossRef] [PubMed]
- Lefebvre, T.; Millery-Vigues, A.; Gallet, C. Does leaf optical absorbance reflect the polyphenol content of alpine plants along an elevational gradient? Alp Bot. 2016, 126, 177–185. [Google Scholar] [CrossRef]
- Kolb, C.A.; Kaser, M.A.; Kopecky, J.; Zotz, G.; Riederer, M.; Pfundel, E.E. Effects of natural intensities of visible and ultraviolet radiation on epidermal ultraviolet screening and photosynthesis in grape leaves. Plant Physiol. 2001, 127, 863–875. [Google Scholar] [CrossRef]
- Barnes, P.W.; Kersting, A.R.; Flint, S.D.; Beyschlag, W.; Ryel, R.J. Adjustments in epidermal UV-transmittance of leaves in sun-shade transitions. Physiol. Plant. 2013, 149, 200–213. [Google Scholar] [CrossRef]
- Ellenberger, J.; Siefen, N.; Krefting, P.; Schulze Lutum, J.-B.; Pfarr, D.; Remmel, M.; Schröder, L.; Röhlen-Schmittgen, S. Effect of UV Radiation and Salt Stress on the Accumulation of Economically Relevant Secondary Metabolites in Bell Pepper Plants. Agronomy 2020, 10, 142. [Google Scholar] [CrossRef]
- Latouche, G.; Bellow, S.; Poutaraud, A.; Meyer, S.; Cerovic, Z.G. Influence of constitutive phenolic compounds on the response of grapevine (Vitis vinifera L.) leaves to infection by Plasmopara viticola. Planta 2013, 237, 351–361. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Calzada, T.; Qian, M.; Strid, Å.; Neugart, S.; Schreiner, M.; Torres-Pacheco, I.; Guevara-González, R.G. Effect of UV-B radiation on morphology, phenolic compound production, gene expression, and subsequent drought stress responses in chili pepper (Capsicum annuum L.). Plant Physiol. Biochem. 2019, 134, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Fgaier, S.; de Almeida Lopes, M.M.; de Oliveira Silva, E.; Aarrouf, J.; Urban, L. Xenon lamps used for fruit surface sterilization can increase the content of total flavonols in leaves of Lactuca sativa L. without any negative effect on net pho-tosynthesis. PLoS ONE 2019, 14, e0223787. [Google Scholar] [CrossRef] [PubMed]
- Pollastrini, M.; Di Stefano, V.; Ferretti, M.; Agati, G.; Grifoni, D.; Zipoli, G.; Orlandini, S.; Bussotti, F. Influence of different light intensity regimes on leaf features of Vitis vinifera L. in ultraviolet radiation filtered condition. Environ. Exp. Bot. 2011, 73, 108–115. [Google Scholar] [CrossRef]
- Barthod, S.; Cerovic, Z.; Epron, D. Can dual chlorophyll fluorescence excitation be used to assess the variation in the content of UV-absorbing phenolic compounds in leaves of temperate tree species along a light gradient? J. Exp. Bot. 2007, 58, 1753–1760. [Google Scholar] [CrossRef]
- Hartikainen, S.M.; Pieristè, M.; Lassila, J.; Robson, T.M. Seasonal Patterns in Spectral Irradiance and Leaf UV-A Absorbance Under Forest Canopies. Front. Plant Sci. 2020, 10, 1762. [Google Scholar] [CrossRef]
- Morales, L.O.; Tegelberg, R.; Brosche, M.; Lindfors, A.; Siipola, S.; Aphalo, P.J. Temporal variation in epidermal flavonoids due to altered solar UV radiation is moderated by the leaf position in Betula pendula. Physiol. Plant. 2011, 143, 261–270. [Google Scholar] [CrossRef]
- Gabriel, J.L.; Zarco-Tejada, P.J.; López-Herrera, P.J.; Pérez-Martín, E.; Alonso-Ayuso, M.; Quemada, M. Airborne and ground level sensors for monitoring nitrogen status in a maize crop. Biosyst. Eng. 2017, 160, 124–133. [Google Scholar] [CrossRef]
- Dalla Marta, A.; Di Stefano, V.; Cerovic, Z.G.; Agati, G.; Orlandini, S. Solar radiation affects grapevine susceptibility to Plasmopara Viticola. Sci. Agric. 2008, 65, 65–70. [Google Scholar] [CrossRef][Green Version]
- Quemada, M.; Gabriel, J.; Zarco-Tejada, P. Airborne Hyperspectral Images and Ground-Level Optical Sensors As Assessment Tools for Maize Nitrogen Fertilization. Remote Sens. 2014, 6, 2940–2962. [Google Scholar] [CrossRef]
- Li, J.W.; Zhang, J.X.; Zhao, Z.; Lei, X.D.; Xu, X.L.; Lu, X.X.; Weng, D.L.; Gao, Y.; Cao, L.K. Use of fluorescence-based sensors to determine the nitrogen status of paddy rice. J. Agric. Sci. 2013, 151, 862–871. [Google Scholar] [CrossRef]
- Hideg, E.; Jansen, M.A.K.; Strid, A. UV-B exposure, ROS, and stress: Inseparable companions or loosely linked associates? Trends Plant Sci. 2013, 18, 107–115. [Google Scholar] [CrossRef]
- Burchard, P.; Bilger, W.; Weissenbock, G. Contribution of hydroxycinnamates and flavonoids to epidermal shielding of UV-A and UV-B radiation in developing rye primary leaves as assessed by ultraviolet-induced chlorophyll fluorescence measure-ments. Plant Cell Environ. 2000, 23, 1373–1380. [Google Scholar] [CrossRef]
- Bilger, W.; Johnsen, T.; Schreiber, U. UV-excited chlorophyll fluorescence as a tool for the assessment of UV-protection by the epidermis of plants. J. Exp. Bot. 2001, 52, 2007–2014. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.Q.; Yao, Y. Photosynthetic responses to solar UV-A and UV-B radiation in low- and high-altitude populations of Hippophae rhamnoides. Photosynthetica 2008, 46, 307–311. [Google Scholar] [CrossRef]
- Krause, G.H.; Gallé, A.; Gademann, R.; Winter, K. Capacity of protection against ultraviolet radiation in sun and shade leaves of tropical forest plants. Funct. Plant Biol. 2003, 30, 533–542. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Shahzad, B.; Rehman, A.; Bhardwaj, R.; Landi, M.; Zheng, B. Response of Phenylpropanoid Pathway and the Role of Polyphenols in Plants under Abiotic Stress. Molecules 2019, 24, 2452. [Google Scholar] [CrossRef] [PubMed]
- Geng, S.; Liu, Y.; Ma, H.; Chen, C. Extraction and Antioxidant Activity of Phenolic Compounds from Okra Flowers. Trop. J. Pharm. Res. 2015, 14, 807. [Google Scholar] [CrossRef]
- Majer, P.; Hideg, E. Existing antioxidant levels are more important in acclimation to supplemental UV-B irradiation than inducible ones: Studies with high light pretreated tobacco leaves. Emir. J. Food Agric. 2012, 24, 598–606. [Google Scholar] [CrossRef]
- Stelzner, J.; Roemhild, R.; Garibay-Hernández, A.; Harbaum-Piayda, B.; Mock, H.-P.; Bilger, W. Hydroxycinnamic acids in sunflower leaves serve as UV-A screening pigments. Photochem. Photobiol. Sci. 2019, 18, 1649–1659. [Google Scholar] [CrossRef] [PubMed]
- Neugart, S.; Rohn, S.; Schreiner, M. Identification of complex, naturally occurring flavonoid glycosides in Vicia faba and Pisum sativum leaves by HPLC-DAD-ESI-MSn and the genotypic effect on their flavonoid profile. Food Res. Int. 2015, 76, 114–121. [Google Scholar] [CrossRef]
- Neugart, S.; Baldermann, S.; Ngwene, B.; Wesonga, J.; Schreiner, M. Indigenous leafy vegetables of Eastern Africa—A source of extraordinary secondary plant metabolites. Food Res. Int. 2017, 100, 411–422. [Google Scholar] [CrossRef] [PubMed]
| Experiment 1 | Experiment 2 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 13:00 | 9:00 | 13:00 | ||||||||
| +UV | -UV | +UV | +UV/-UV | -UV/+UV | -UV | +UV | +UV/-UV | -UV/+UV | -UV | |
| Plant height (cm) | 23.14 ± 3.86 a | 27.07 ± 1.29 b | 20.40 ± 1.14 | 20.73 ± 8.82 | 20.08 ± 1.84 | 21.29 ± 1.08 | 19.46 ± 2.42 | 19.47 ± 2.53 | 20.93 ± 1.82 | 21.38 ± 1.27 |
| Internode length (cm) | 4.53 ± 1.34 a | 5.88 ± 0.65 b | 2.45 ± 0.38 A | 3.0 ± 0.76 AB | 2.71 ± 0.68 AB | 3.21 ± 1.05 B | 2.41 ± 0.35 | 2.49 ± 0.25 | 2.60 ± 0.29 | 3.02 ± 0.62 |
| Leaf area (cm2) | 61.76 ± 9.91 | 63.50 ± 10.25 | 36.25 ± 6.36 | 38.10 ± 6.70 | 33.98 ± 5.40 | 45.25 ± 5.57 | 37.83 ± 8.73 | 37.28 ± 6.27 | 43.40 ± 6.61 | 44.57 ± 8.48 |
| Fresh matter (g/leaf) | 1.27 ± 0.33 | 1.40 ± 0.17 | 0.94 ± 0.17 AB | 0.99 ± 0.18 AB | 0.86 ± 0.13 A | 1.09 ± 0.14 B | 0.95 ± 0.23 | 0.93 ± 0.14 | 1.05 ± 0.16 | 1.09 ± 0.20 |
| Dry matter (g/leaf) | 0.22 ± 0.05 | 0.27 ± 0.03 | 0.14 ± 0.02 AB | 0.15 ± 0.02 AB | 0.13 ± 0.02 A | 0.17 ± 0.02 B | 0.16 ± 0.03 | 0.15 ± 0.03 | 0.17 ± 0.03 | 0.18 ± 0.04 |
| Dry matter (mg/0.75 cm2) | 2.18 ± 0.35 | 2.26 ± 0.24 | 2.26 ± 0.42 | 2.22 ± 0.28 | 2.33 ± 0.24 | 2.09 ± 0.34 | 2.47 ± 0.34 | 2.21 ± 0.40 | 2.25 ± 0.22 | 2.36 ± 0.23 |
| Experiment 1 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 9:00 | 11:00 | 13:00 | 15:00 | 17:00 | ||||||
| +UV | -UV | +UV | -UV | +UV | -UV | +UV | -UV | +UV | -UV | |
| HFer-glc | 4.78 ± 1.95 bc | 2.48 ± 1.42 B | 6.45 ± 1.50 c | 6.25 ± 0.60 C | 2.45 ± 1.75 a | 0.75 ± 0.58 A | 3.54 ± 2.33 ab | 2.14 ± 1.26 B | 6.36 ± 1.24 c | 5.47 ± 0.97 C |
| I-3-glc | 0.03 ± 0.03 a | 0.00 ± 0.01 A | 0.09 ± 0.02 c | 0.02 ± 0.01 B | 0.01 ± 0.01 a | 0.00 ± 0.00 A | 0.01 ± 0.01 a | 0.00 ± 0.00 A | 0.06 ± 0.02 b | 0.01 ± 0.01 A |
| K-3-glc | 0.15 ± 0.07 a | 0.07 ± 0.13 A | 0.28 ± 0.07 b | 0.08 ± 0.03 A | 0.07 ± 0.05 a | 0.01 ± 0.01 A | 0.11 ± 0.08 a | 0.02 ± 0.01 A | 0.28 ± 0.12 b | 0.08 ± 0.04 A |
| K-3-glc-xyl | 0.38 ± 0.22 b | 0.09 ± 0.07 A | 0.74 ± 0.12 c | 0.26 ± 0.09 B | 0.08 ± 0.06 a | 0.02 ± 0.01 A | 0.18 ± 0.14 a | 0.04 ± 0.03 A | 0.54 ± 0.14 b | 0.18 ± 0.09 B |
| Q-3-glc | 0.28 ± 0.18 ab | 0.01 ± 0.01 A | 0.63 ± 0.14 c | 0.02 ± 0.01 B | 0.08 ± 0.07 a | 0.00 ± 0.00 A | 0.19 ± 0.15 a | 0.00 ± 0.00 A | 0.48 ± 0.23 bc | 0.02 ± 0.02 A |
| Q-3-glc-xyl | 3.90 ± 2.67 ab | 0.15 ± 0.14 A | 8.56 ± 3.22 c | 0.44 ± 0.20 B | 0.97 ± 1.16 a | 0.03 ± 0.02 A | 2.04 ± 1.85 a | 0.08 ± 0.06 A | 6.02 ± 2.50 bc | 0.35 ± 0.22 B |
| Sin-glc | 0.57 ± 0.32 a | 0.11 ± 0.02 A | 1.10 ± 0.49 b | 0.18 ± 0.02 A | 0.43 ± 0.18 a | 0.13 ± 0.03 A | 0.35 ± 0.09 a | 0.19 ± 0.18 A | 0.55 ± 0.36 a | 0.15 ± 0.03 A |
| Experiment 2 | ||||||||
|---|---|---|---|---|---|---|---|---|
| 9:00 | 13:00 | |||||||
| +UV | +UV/-UV | -UV/+UV | -UV | +UV | +UV/-UV | -UV/+UV | -UV | |
| HFer-glc | 6.44 ± 0.71 | 6.56 ± 0.24 | 6.69 ± 0.40 | 6.31 ± 0.75 | 6.42 ± 0.86 | 6.18 ± 0.44 | 6.66 ± 0.73 | 6.16 ± 0.31 |
| I-3-glc | 0.05 ± 0.02 b | 0.03 ± 0.01 a | 0.03 ± 0.01 a | 0.07 ± 0.04 b | 0.08 ± 0.04 B | 0.01 ± 0.01 A | 0.05 ± 0.03 B | 0.01 ± 0.01 A |
| K-3-glc | 0.13 ± 0.07 | 0.12 ± 0.05 | 0.14 ± 0.09 | 0.13 ± 0.07 | 0.16 ± 0.04 B | 0.07 ± 0.02 A | 0.14 ± 0.04 B | 0.05 ± 0.02 A |
| K-3-glc-xyl | 0.49 ± 0.23 | 0.46 ± 0.14 | 0.48 ± 0.14 | 0.42 ± 0.23 | 0.59 ± 0.20 B | 0.34 ± 0.08 A | 0.58 ± 0.15 B | 0.29 ± 0.05 A |
| Q-3-glc | 0.23 ± 0.19 a | 0.27 ± 0.09 ab | 0.25 ± 0.11 ab | 0.43 ± 0.24 b | 0.35 ± 0.12 C | 0.15 ± 0.09 B | 0.28 ± 0.13 C | 0.02 ± 0.01 A |
| Q-3-glc-xyl | 6.61 ± 2.05 | 4.70 ± 1.28 | 5.28 ± 1.89 | 7.15 ± 3.28 | 7.61 ± 1.92 C | 2.92 ± 0.91 B | 6.23 ± 1.98 C | 0.55 ± 0.16 A |
| Sin-glc | 1.59 ± 0.08 c | 1.37 ± 0.11 ab | 1.45 ± 0.17 bc | 1.18 ± 0.27 a | 1.48 ± 0.16 B | 1.16 ± 0.25 B | 1.35 ± 0.27 B | 0.13 ± 0.40 A |
| +UV | 330 | 370 | TEAC | FRAP | DPPH | HFer-glc | I-3-glc | K-3-glc | K-3-glc-xyl | Q-3-glc | Q-3-glc-xyl | Sin-glc | FLAV |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 330 | 1.00 | 0.66 | 0.71 | 0.73 | −0.36 | −0.30 | −0.19 | 0.01 | −0.27 | −0.07 | −0.24 | −0.30 | 0.19 |
| 370 | 1.00 | 0.63 | 0.54 | −0.08 | −0.02 | 0.30 | 0.31 | 0.23 | 0.38 | 0.31 | 0.18 | 0.10 | |
| TEAC | 1.00 | 0.75 | −0.34 | −0.13 | 0.07 | 0.10 | −0.07 | 0.11 | 0.06 | 0.00 | 0.26 | ||
| FRAP | 1.00 | −0.34 | −0.29 | −0.14 | −0.11 | −0.27 | −0.10 | −0.14 | −0.20 | 0.23 | |||
| DPPH | 1.00 | −0.07 | −0.02 | −0.08 | 0.04 | −0.02 | −0.02 | 0.19 | −0.21 | ||||
| HFer-glc | 1.00 | 0.64 | 0.67 | 0.74 | 0.68 | 0.66 | 0.28 | −0.15 | |||||
| I-3-glc | 1.00 | 0.84 | 0.93 | 0.92 | 0.91 | 0.57 | 0.00 | ||||||
| K-3-glc | 1.00 | 0.87 | 0.91 | 0.78 | 0.37 | −0.11 | |||||||
| K-3-glc-xyl | 1.00 | 0.90 | 0.90 | 0.59 | −0.17 | ||||||||
| Q-3-glc | 1.00 | 0.90 | 0.41 | −0.10 | |||||||||
| Q-3-glc-xyl | 1.00 | 0.61 | −0.04 | ||||||||||
| Sin-glc | 1.00 | −0.04 | |||||||||||
| FLAV | 1.00 |
| -UV | 330 | 370 | TEAC | FRAP | DPPH | HFer-glc | I-3-glc | K-3-glc | K-3-glc-xyl | Q-3-glc | Q-3-glc-xyl | Sin-glc | FLAV |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 330 | 1.00 | 0.51 | 0.51 | 0.46 | −0.11 | −0.07 | 0.01 | −0.08 | −0.08 | −0.01 | −0.07 | 0.01 | 0.24 |
| 370 | 1.00 | −0.16 | 0.05 | 0.12 | 0.38 | 0.39 | 0.09 | 0.41 | 0.31 | 0.34 | 0.06 | 0.38 | |
| TEAC | 1.00 | 0.63 | −0.16 | −0.26 | −0.17 | −0.12 | −0.20 | −0.22 | −0.14 | 0.05 | 0.12 | ||
| FRAP | 1.00 | −0.01 | −0.24 | −0.21 | −0.17 | −0.17 | −0.14 | −0.16 | −0.04 | 0.43 | |||
| DPPH | 1.00 | −0.28 | −0.05 | −0.21 | −0.16 | −0.28 | −0.23 | −0.16 | 0.16 | ||||
| HFer-glc | 1.00 | 0.54 | 0.44 | 0.91 | 0.78 | 0.87 | 0.10 | 0.10 | |||||
| I-3-glc | 1.00 | 0.20 | 0.51 | 0.36 | 0.49 | 0.12 | 0.23 | ||||||
| K-3-glc | 1.00 | 0.49 | 0.39 | 0.50 | 0.03 | −0.03 | |||||||
| K-3-glc-xyl | 1.00 | 0.78 | 0.97 | 0.07 | 0.17 | ||||||||
| Q-3-glc | 1.00 | 0.79 | 0.08 | 0.16 | |||||||||
| Q-3-glc-xyl | 1.00 | 0.05 | 0.18 | ||||||||||
| Sin-glc | 1.00 | −0.12 | |||||||||||
| FLAV | 1.00 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Neugart, S.; Tobler, M.A.; Barnes, P.W. The Function of Flavonoids in the Diurnal Rhythm under Rapidly Changing UV Conditions—A Model Study on Okra. Plants 2021, 10, 2268. https://doi.org/10.3390/plants10112268
Neugart S, Tobler MA, Barnes PW. The Function of Flavonoids in the Diurnal Rhythm under Rapidly Changing UV Conditions—A Model Study on Okra. Plants. 2021; 10(11):2268. https://doi.org/10.3390/plants10112268
Chicago/Turabian StyleNeugart, Susanne, Mark A. Tobler, and Paul W. Barnes. 2021. "The Function of Flavonoids in the Diurnal Rhythm under Rapidly Changing UV Conditions—A Model Study on Okra" Plants 10, no. 11: 2268. https://doi.org/10.3390/plants10112268
APA StyleNeugart, S., Tobler, M. A., & Barnes, P. W. (2021). The Function of Flavonoids in the Diurnal Rhythm under Rapidly Changing UV Conditions—A Model Study on Okra. Plants, 10(11), 2268. https://doi.org/10.3390/plants10112268






