Biocontrol of Carrot Disease-Causing Pathogens Using Essential Oils
Abstract
1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. Fungal Material
4.2. Essential Oils Extraction
4.3. Evaluation of Essential Oils Efficiency
4.4. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Carvalho, F.P. Pesticides, environment, and food safety. Food Energy Secur. 2017, 6, 48–60. [Google Scholar] [CrossRef]
- Solaimalai, A.; Ramesh, R.T.; Baskar, M. Pesticides and environment. In Environmental Contamination and Bioreclamation; APH Publishing Corporation: New Delhi, India, 2004; pp. 345–382. ISBN 9788176485876. [Google Scholar]
- Rathore, H.S.; Nollet, L.M. (Eds.) Pesticides: Evaluation of Environmental Pollution; CRC Press: Boca Raton, FL, USA, 2012; p. 659. ISBN 9780367865191. [Google Scholar]
- Mahmood, I.; Imadi, S.R.; Shazadi, K.; Gul, A.; Hakeem, K.R. Effects of pesticides on environment. In Plant, Soil and Microbes; Springer: Cham, Switzerland, 2016; pp. 253–269. [Google Scholar]
- Rajmohan, K.S.; Chandrasekaran, R.; Varjani, S. A Review on occurrence of pesticides in environment and current technologies for their remediation and management. Indian J. Microbiol. 2020, 60, 125–138. [Google Scholar] [CrossRef]
- Antunes, M.D.C.; Cavaco, A.M. The use of essential oils for postharvest decay control. A review. Flavour Fragr. J. 2010, 25, 351–366. [Google Scholar] [CrossRef]
- Ricke, S.C.; Kundinger, M.M.; Miller, D.R.; Keeton, J.T. Alternatives to antibiotics: Chemical and physical antimicrobial interventions and foodborne pathogen response. Poult. Sci. 2005, 84, 667–675. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Farm to Fork Strategy: For a Fair, Healthy and Environmentally-Friendly Food System. DG SANTE/Unit ‘Food Information and Composition, Food Waste’. 2020. Available online: https://ec.europa.eu/food/sites/food/files/safety/docs/f2f_action-plan_2020_strategy-info_en.pdf (accessed on 4 March 2021).
- Klein, C.S.; Rodriguez-Concepcion, M. Carotenoids in carrot. In Pigments in Fruits and Vegetables; Chen, C., Ed.; Springer: New York, NY, USA, 2015; pp. 217–228. [Google Scholar] [CrossRef]
- Sharma, K.D.; Karki, S.; Thakur, N.S.; Attri, S. Chemical composition, functional properties and processing of carrot—A review. J. Food Sci. Technol. 2012, 49, 22–32. [Google Scholar] [CrossRef]
- Davis, R.M.; Nu, J. Integrated approaches for carrot pests and diseases management. In General Concepts in Integrated Pest and Disease Management; Ciancio, A., Mukerji, K.G., Eds.; Springer: Dordrecht, The Netherlands, 2007; pp. 149–188. [Google Scholar]
- Que, F.; Hou, X.L.; Wang, G.L.; Xu, Z.S.; Tan, G.F.; Li, T.; Xiong, A.S. Advances in research on the carrot, an important root vegetable in the Apiaceae family. Hortic. Res. 2019, 6, 69. [Google Scholar] [CrossRef]
- Le Clerc, V.; Briard, M. Carrot disease management. In Carrots and Related Apiaceae Crops, 2nd ed.; Geoffriau, E., Simon, P., Eds.; CABI Publishing: Wallingford, UK, 2020; Volume 33, pp. 115–129. [Google Scholar]
- Farrar, J.J.; Pryor, B.M.; Davis, R.M. Alternaria diseases of carrot. Plant Dis. 2004, 88, 776–784. [Google Scholar] [CrossRef] [PubMed]
- Escriva, L.; Oueslati, S.; Font, G.; Manyes, L. Alternaria mycotoxins in food and feed: An overview. J. Food Qual. 2017, 2017, 20. [Google Scholar] [CrossRef]
- Leyte-Lugo, M.; Richomme, P.; Poupard, P.; Peria-Rodriguez, L.M. Identification and Quantification of a Phytotoxic Metabolite from Alternaria dauci. Molecules 2020, 25, 4003. [Google Scholar] [CrossRef] [PubMed]
- Lukošiūtė, S.; Šernaitė, L.; Morkeliūnė, A.; Rasiukevičiūtė, N.; Valiuškaitė, A. The effect of Lamiaceae plants essential oils on fungal plant pathogens in vitro. Agron. Res. 2020, 18, 2761–2769. [Google Scholar] [CrossRef]
- Šernaitė, L.; Rasiukevičiūtė, N.; Valiuškaitė, A. The extracts of cinnamon and clove as potential biofungicides against strawberry grey mould. Plants 2020, 9, 613. [Google Scholar] [CrossRef] [PubMed]
- Sil, A.; Pramanik, K.; Samantaray, P.; Firoz, M.; Yaday, V. Essential oils: A boon towards eco-friendly management of phytopathogenic fungi. J. Entomol. Zool. Stud 2020, 8, 1884–1891. [Google Scholar]
- Mugao, L.G.; Muturi, P.W.; Gichimu, B.M.; Njoroge, E.K. In Vitro Control of Phytophthora infestans and Alternaria solani Using Crude Extracts and Essential Oils from Selected Plants. Int. J. Agron. 2020, 2020, 8845692. [Google Scholar] [CrossRef]
- Pezzani, R.; Vitalini, S.; Iriti, M. Bioactivities of Origanum vulgare L.: An update. Phytochem. Rev. 2017, 16, 1253–1268. [Google Scholar] [CrossRef]
- Oniga, I.; Puscas, C.; Silaghi-Dumitrescu, R.; Olah, N.K.; Sevastre, B.; Marica, R.; Hanganu, D. Origanum vulgare ssp. vulgare: Chemical composition and biological studies. Molecules 2018, 23, 2077. [Google Scholar] [CrossRef]
- Esen, G.; Azaz, A.D.; Kurkcuoglu, M.; Baser, K.H.C.; Tinmaz, A. Essential oil and antimicrobial activity of wild and cultivated Origanum vulgare L. subsp. hirtum (Link) letswaart from the Marmara region, Turkey. Flavour Fragr. J. 2007, 22, 371–376. [Google Scholar] [CrossRef]
- Askun, T.; Tumen, G.; Satil, F.; Kilic, T. Effects of some Lamiaceae species methanol extracts on potential mycotoxin producer fungi. Pharm. Biol. 2008, 46, 688–694. [Google Scholar] [CrossRef][Green Version]
- Sarikurkcu, C.; Zengin, G.; Oskay, M.; Uysal, S.; Ceylan, R.; Aktumsek, A. Composition, antioxidant, antimicrobial and enzyme inhibition activities of two Origanum vulgare subspecies (subsp. vulgare and subsp. hirtum) essential oils. Ind. Crop. Prod. 2015, 70, 178–184. [Google Scholar] [CrossRef]
- Baranauskienė, R.; Venskutonis, P.R.; Dambrauskienė, E.; Viškelis, P. Harvesting time influences the yield and oil composition of Origanum vulgare L. ssp. vulgare and ssp. hirtum. Ind. Crop. Prod. 2013, 49, 43–51. [Google Scholar] [CrossRef]
- Sanit, S. Antifungal activity of selected medicinal plants against Alternaria species: The pathogen of dirty panicle disease in rice. J. Med. Plants Res. 2016, 10, 195–201. [Google Scholar] [CrossRef][Green Version]
- Plotto, A.; Roberts, D.D.; Roberts, R.G. Evaluation of plant essential oils as natural postharvest disease control of tomato (Lycopersicon esculentum). In Proceedings of the XXVI International Horticultural Congress: Issues and Advances in Postharvest Horticulture, Toronto, ON, Canada, 11–17 August 2002; pp. 737–745. [Google Scholar] [CrossRef]
- Scariot, F.J.; Foresti, L.; Delamare, A.P.L.; Echeverrigaray, A.S. Activity of monoterpenoids on the in vitro growth of two Colletotrichum species and the mode of action on C. acutatum. Pestic. Biochem. Physiol. 2020, 170, 104698. [Google Scholar] [CrossRef] [PubMed]
- Ghalem, B.R.; Talia, B.; Omar, H. Antifungal Activities of Five Commercial Extracts against Alternaria alternate. J. Biotechnol. Res. 2020, 6, 98–103. [Google Scholar] [CrossRef]
- Lambert, R.J.W.; Skandamis, P.N.; Coote, P.J.; Nychas, G.J. A study of the minimum inhibitory concentration and mode of action of oregano essential oil, thymol and carvacrol. J. Appl. Microbiol. 2001, 91, 453–462. [Google Scholar] [CrossRef]
- Abbaszadeh, S.; Sharifzadeh, A.; Shokri, H.; Khosravi, A.R.; Abbaszadeh, A. Antifungal efficacy of thymol, carvacrol, eugenol and menthol as alternative agents to control the growth of food-relevant fungi. J. Mycol. Med. 2014, 24, e51–e56. [Google Scholar] [CrossRef] [PubMed]
- Simmons, E.G. Alternaria: An Identification Manual; ASM Press: Washington, DC, USA, 2007; 775p, ISBN 978-9070351687. [Google Scholar]
- AOAC. Volatile oil in spices. In Official Methods of Analysis, 15th ed.; Helrich, K., Ed.; Association of Official Analytical Chemists: Washington, DC, USA, 1990; Volume 1. [Google Scholar]
- Morkeliūnė, A.; Rasiukevičiūtė, N.; Valiuškaitė, A. Pathogenicity of Colletotrichum acutatum to different strawberry cultivars and anthracnose control with essential oils. Zemdirb. Agric. 2021, 10, 173–180. [Google Scholar] [CrossRef]
- Marei, G.I.K.; Rasoul, M.A.A.; Abdelgaleil, S.A. Comparative antifungal activities and biochemical effects of monoterpenes on plant pathogenic fungi. Pestic. Biochem. Physiol. 2012, 103, 56–61. [Google Scholar] [CrossRef]
- Feng, W.; Zheng, X. Essential oils to control Alternaria alternata in vitro and in vivo. Food Control 2007, 18, 1126–1130. [Google Scholar] [CrossRef]
- Raudonius, S. Mokslinių Tyrimų Planavimas Ir Analizė: Mokomoji Knyga; Akademija, IDP Solutions: Kaunas, Lithuania, 2008; 139p, ISBN 9789955865186. [Google Scholar]
- Malama, A.A.; Mironova, S.N.; Filimonova, T.V.; Moiseev, Y.V.; Gumargalieva, K.Z. The effect of temperature on growth of mycelial fungi colonies. Microbiology 1987, 54, 994–997. [Google Scholar]
- Šernaitė, L.; Rasiukevičiūtė, N.; Valiuškaitė, A. Application of plant extracts to control postharvest gray mold and susceptibility of apple fruits to B. cinerea from different plant hosts. Foods 2020, 9, 1430. [Google Scholar] [CrossRef] [PubMed]
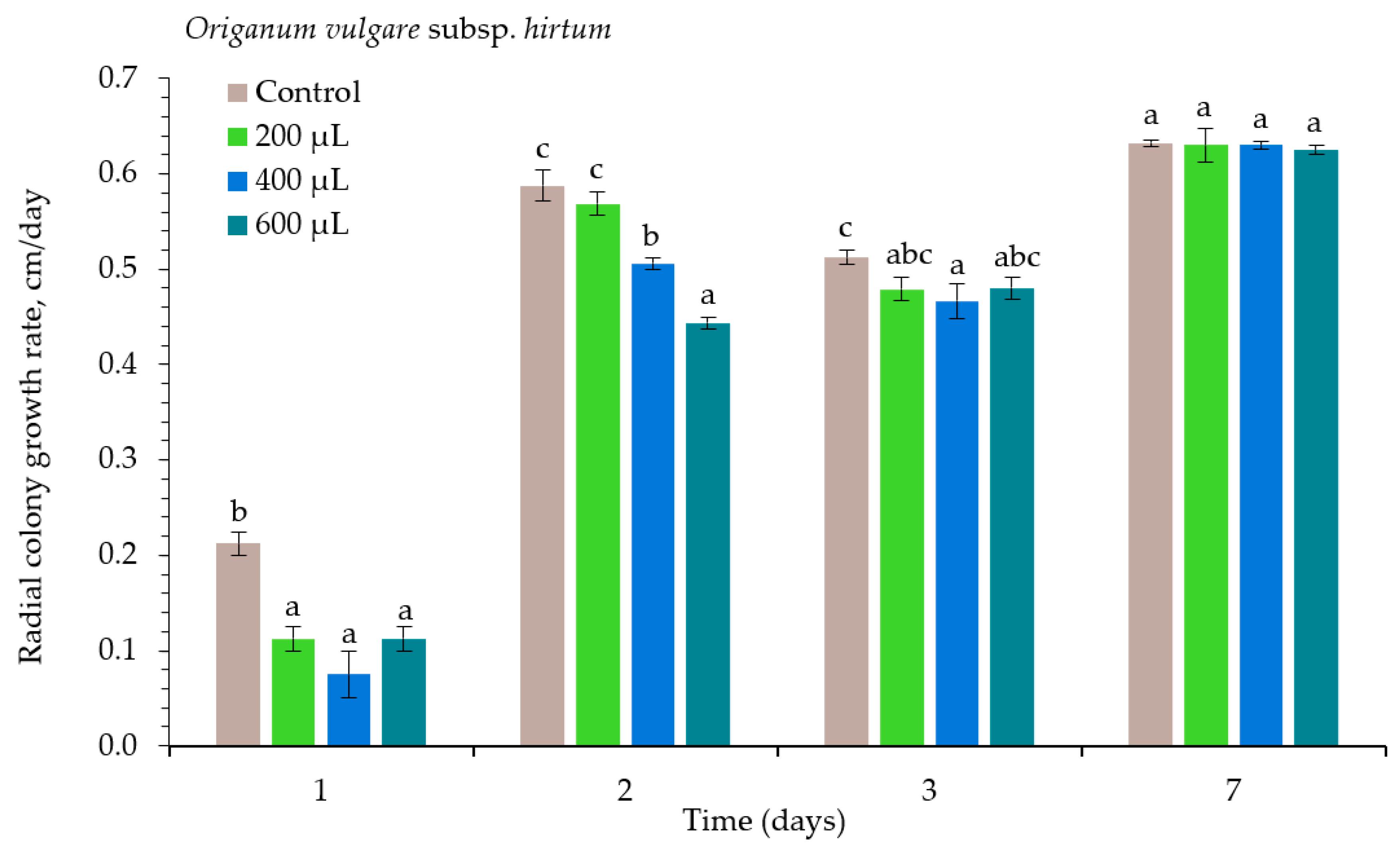
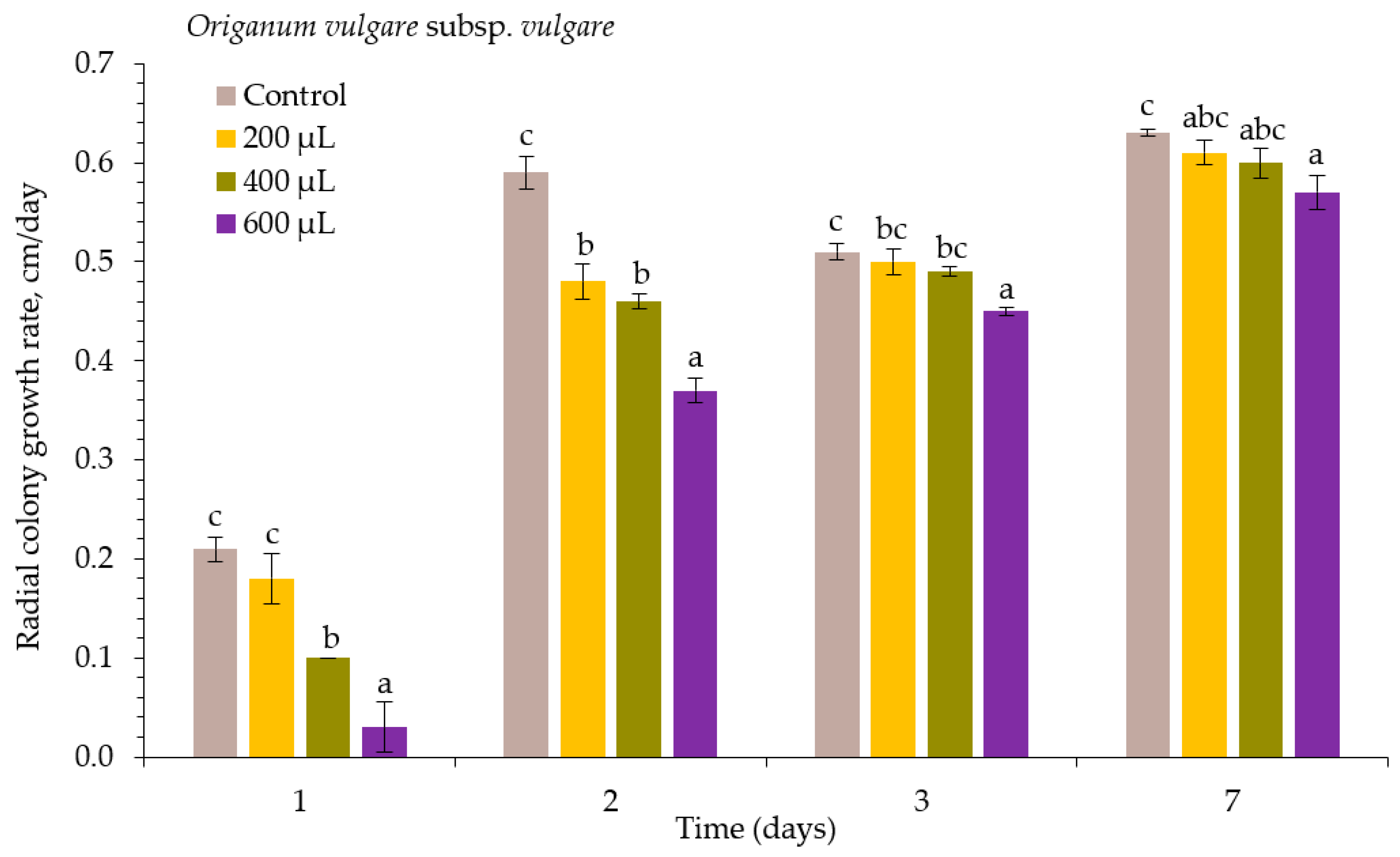
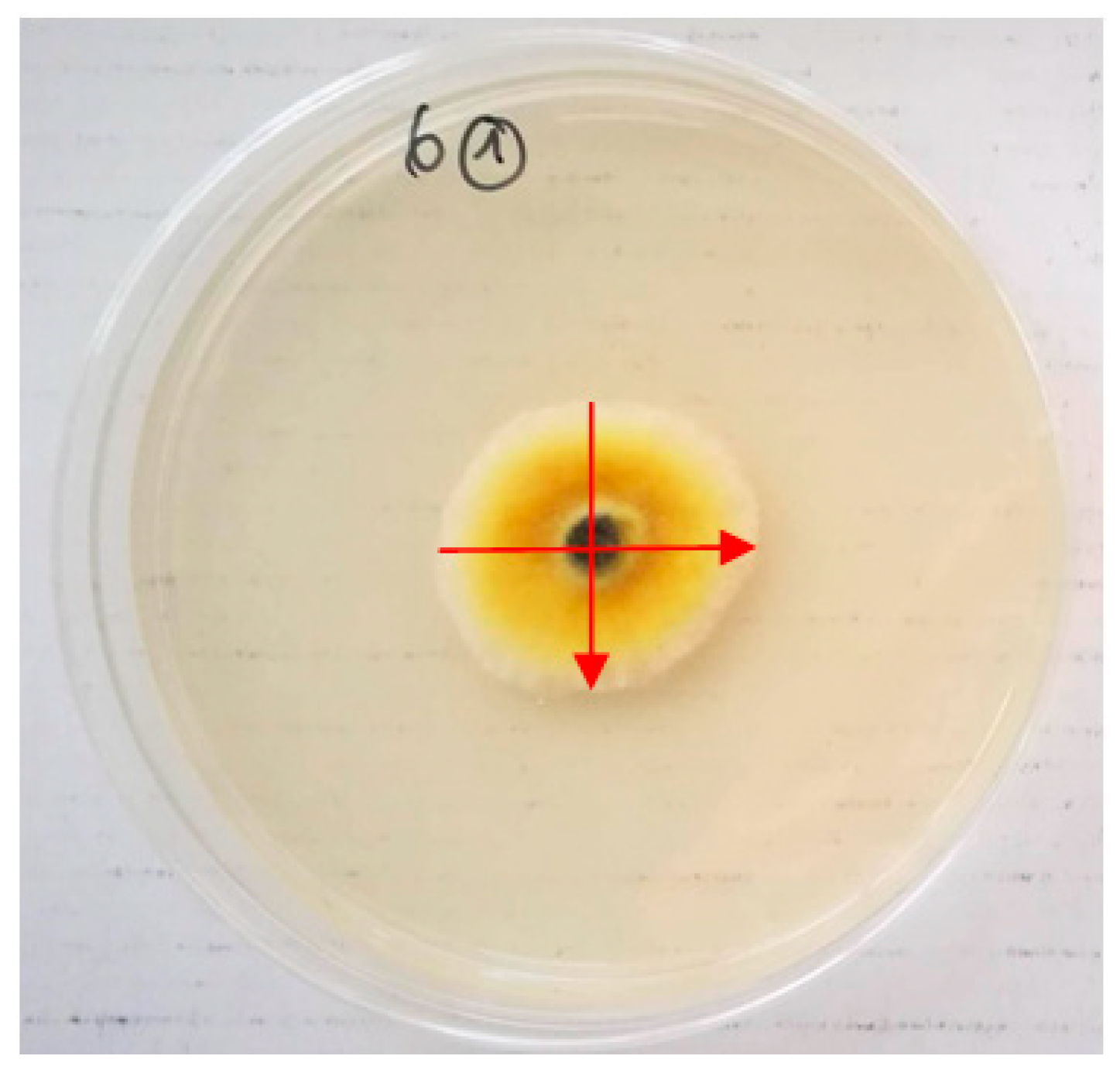
| Essential Oil | Concentration μL L−1 | Inhibition of Alternaria spp., % | ||
|---|---|---|---|---|
| 1 DAI | 3 DAI | 7 DAI | ||
| Origanum vulgare subsp. vulgare | 200 | 14.29 | 1.96 | 3.17 |
| 400 | 52.38 | 3.92 | 4.76 | |
| 600 | 85.71 | 11.76 | 9.52 | |
| Origanum vulgare subsp. hirtum | 200 | 47.06 | 6.50 | 0.34 |
| 400 | 64.71 | 8.94 | 0.34 | |
| 600 | 47.06 | 6.34 | 1.13 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chrapačienė, S.; Rasiukevičiūtė, N.; Valiuškaitė, A. Biocontrol of Carrot Disease-Causing Pathogens Using Essential Oils. Plants 2021, 10, 2231. https://doi.org/10.3390/plants10112231
Chrapačienė S, Rasiukevičiūtė N, Valiuškaitė A. Biocontrol of Carrot Disease-Causing Pathogens Using Essential Oils. Plants. 2021; 10(11):2231. https://doi.org/10.3390/plants10112231
Chicago/Turabian StyleChrapačienė, Simona, Neringa Rasiukevičiūtė, and Alma Valiuškaitė. 2021. "Biocontrol of Carrot Disease-Causing Pathogens Using Essential Oils" Plants 10, no. 11: 2231. https://doi.org/10.3390/plants10112231
APA StyleChrapačienė, S., Rasiukevičiūtė, N., & Valiuškaitė, A. (2021). Biocontrol of Carrot Disease-Causing Pathogens Using Essential Oils. Plants, 10(11), 2231. https://doi.org/10.3390/plants10112231







