Effects of Dipsacus asperoides and Phlomis umbrosa Extracts in a Rat Model of Osteoarthritis
Abstract
:1. Introduction
2. Results
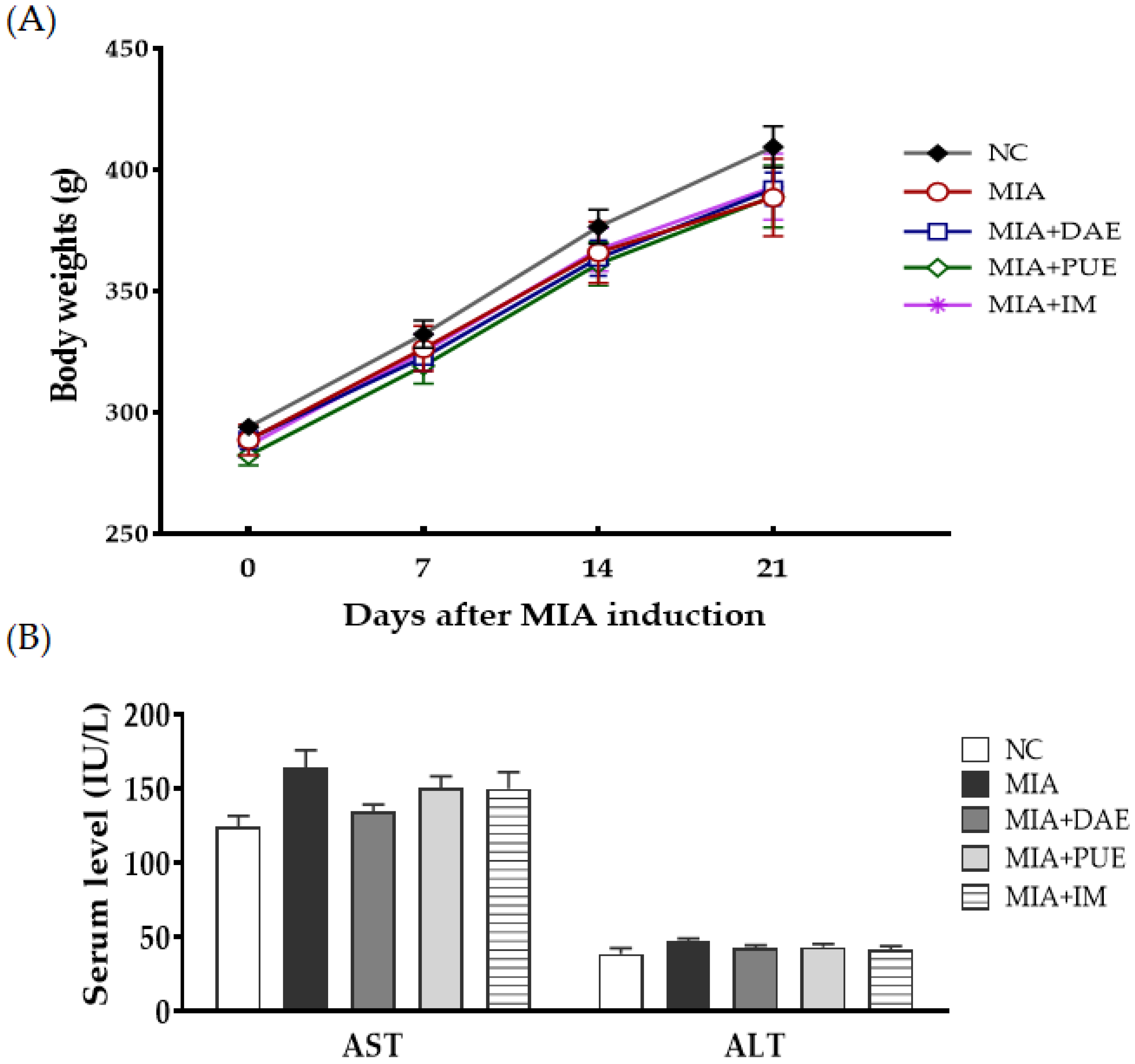
2.1. Effects of D. asperoides Extract (DAE) and P. umbrosa Extract (PUE) on Body Weight and Serum Markers of Toxicity
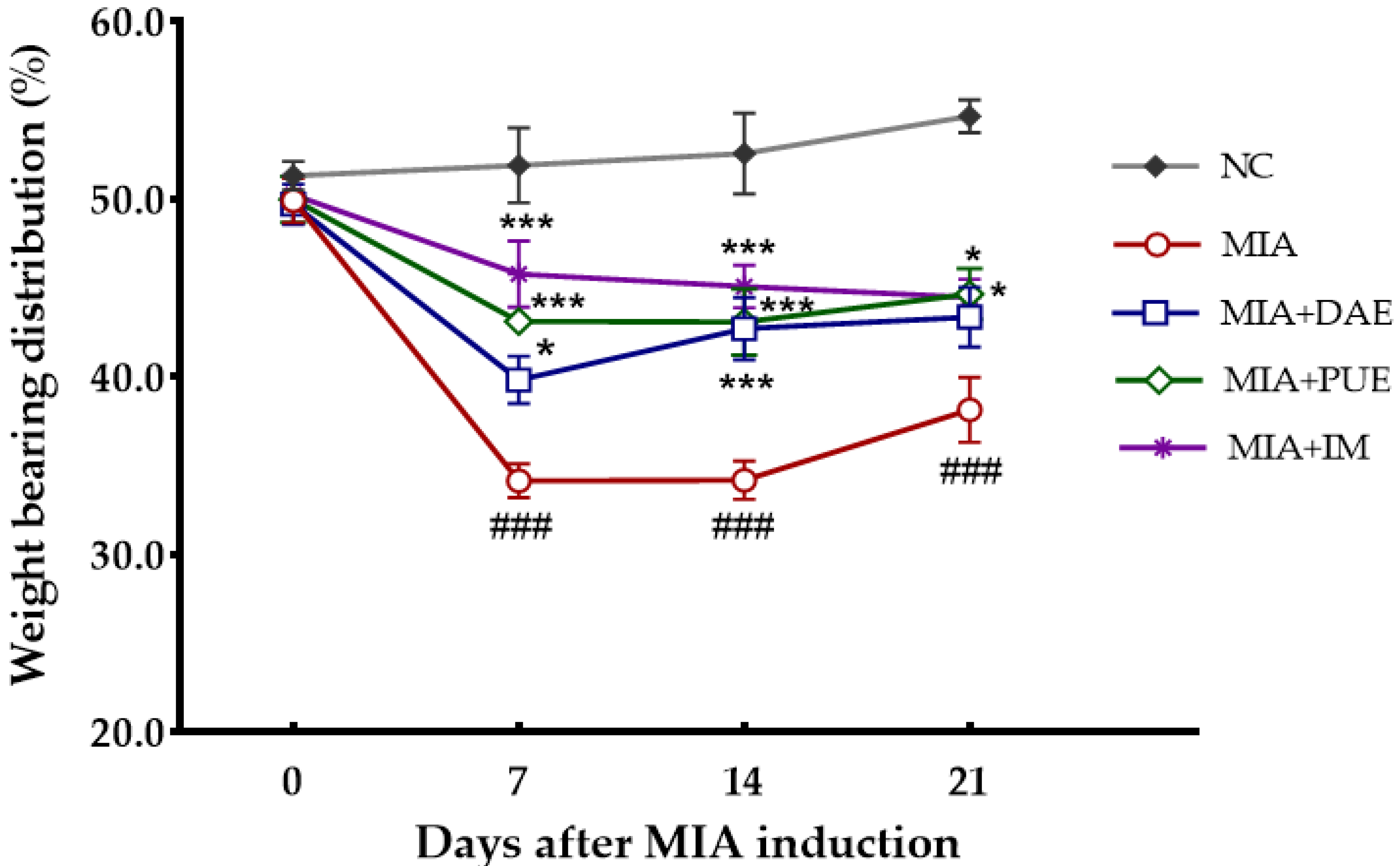
2.2. Effects of DAE and PUE on Hind Paw Weight-Bearing Distribution in MIA-Induced OA Rats
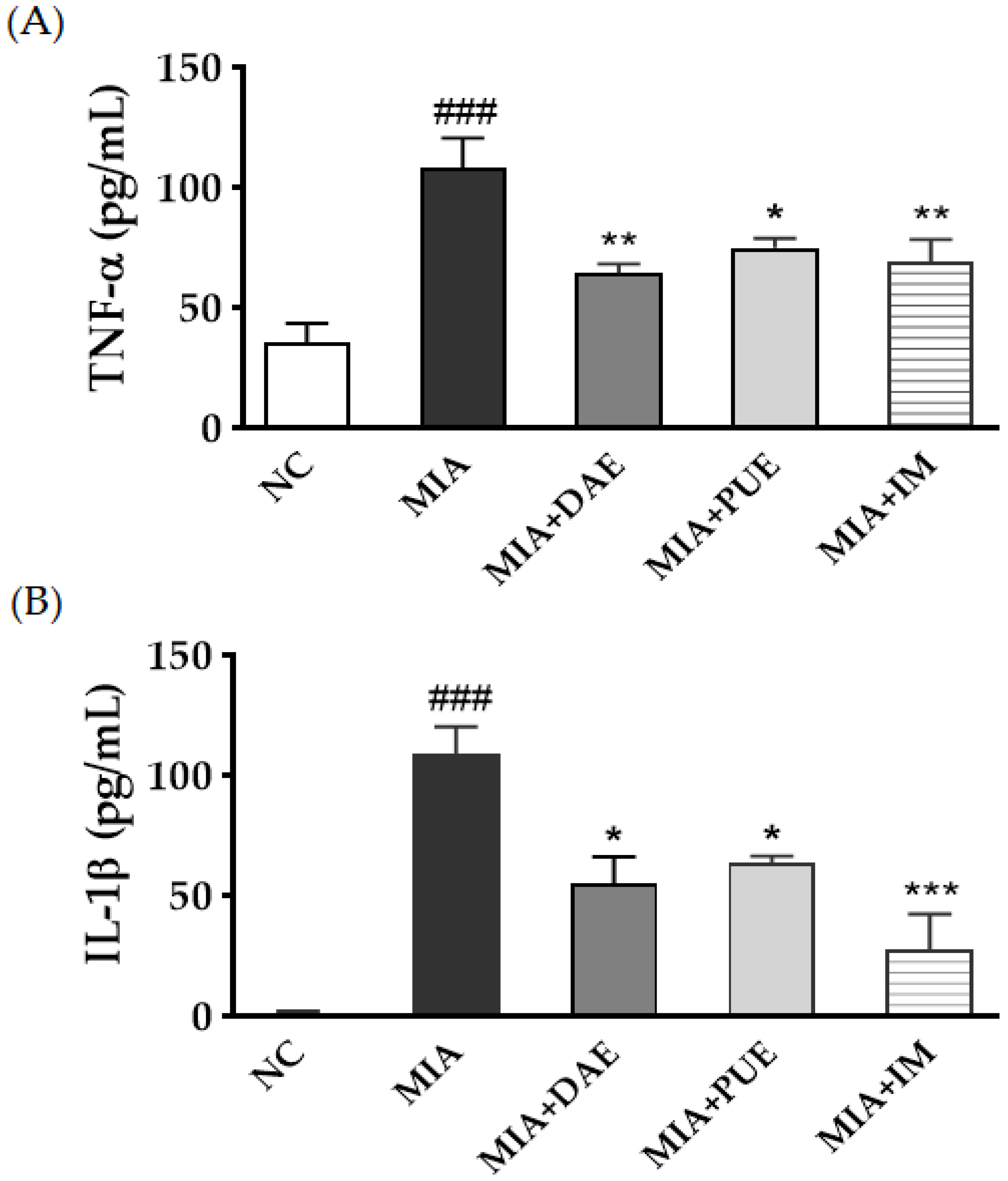
2.3. Effects of DAE and PUE on Sezrum Levels of Cytokines in MIA-Induced OA Rats
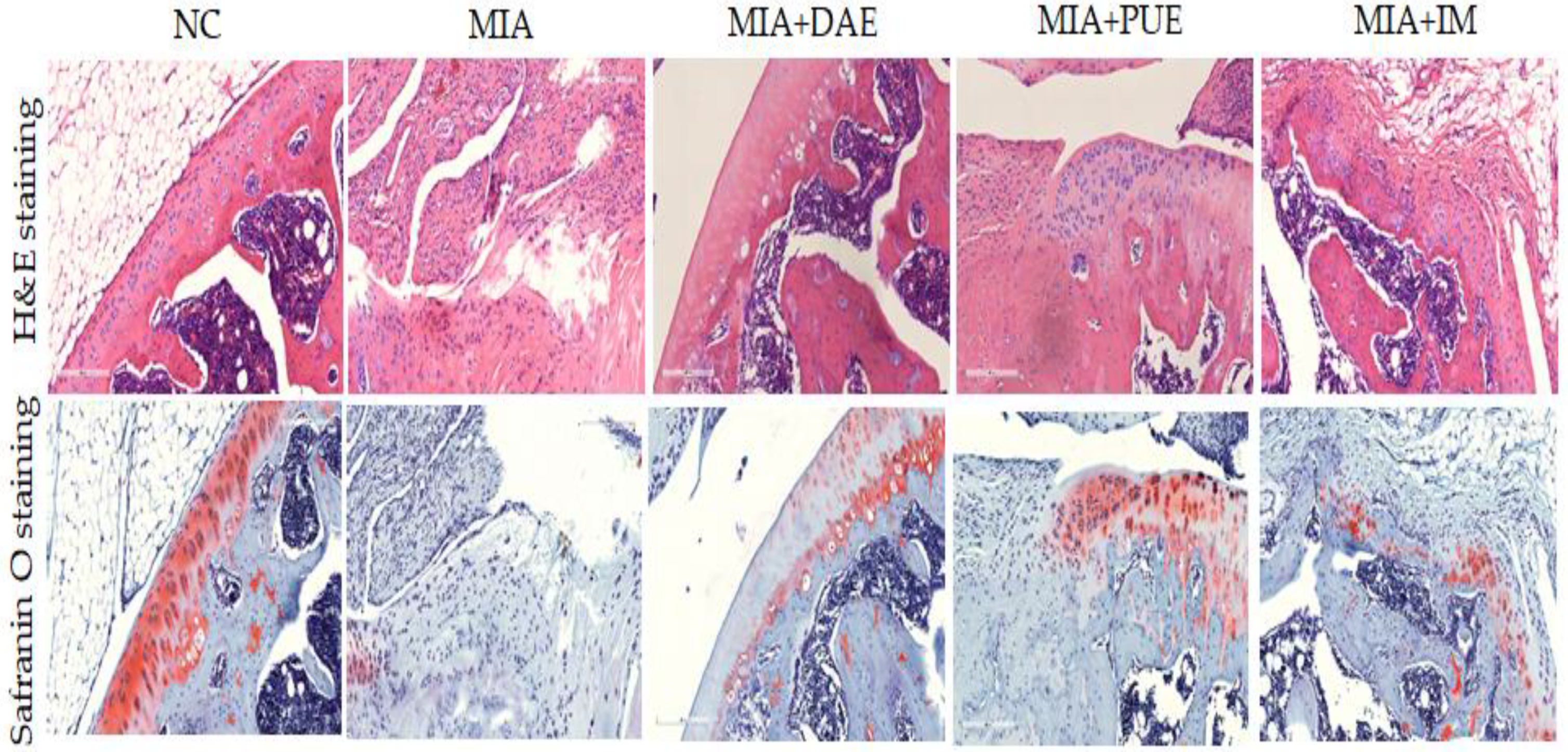
2.4. Effects of DAE and PUE on the Histopathological Features of Joint Tissues in MIA-Induced OA Rats
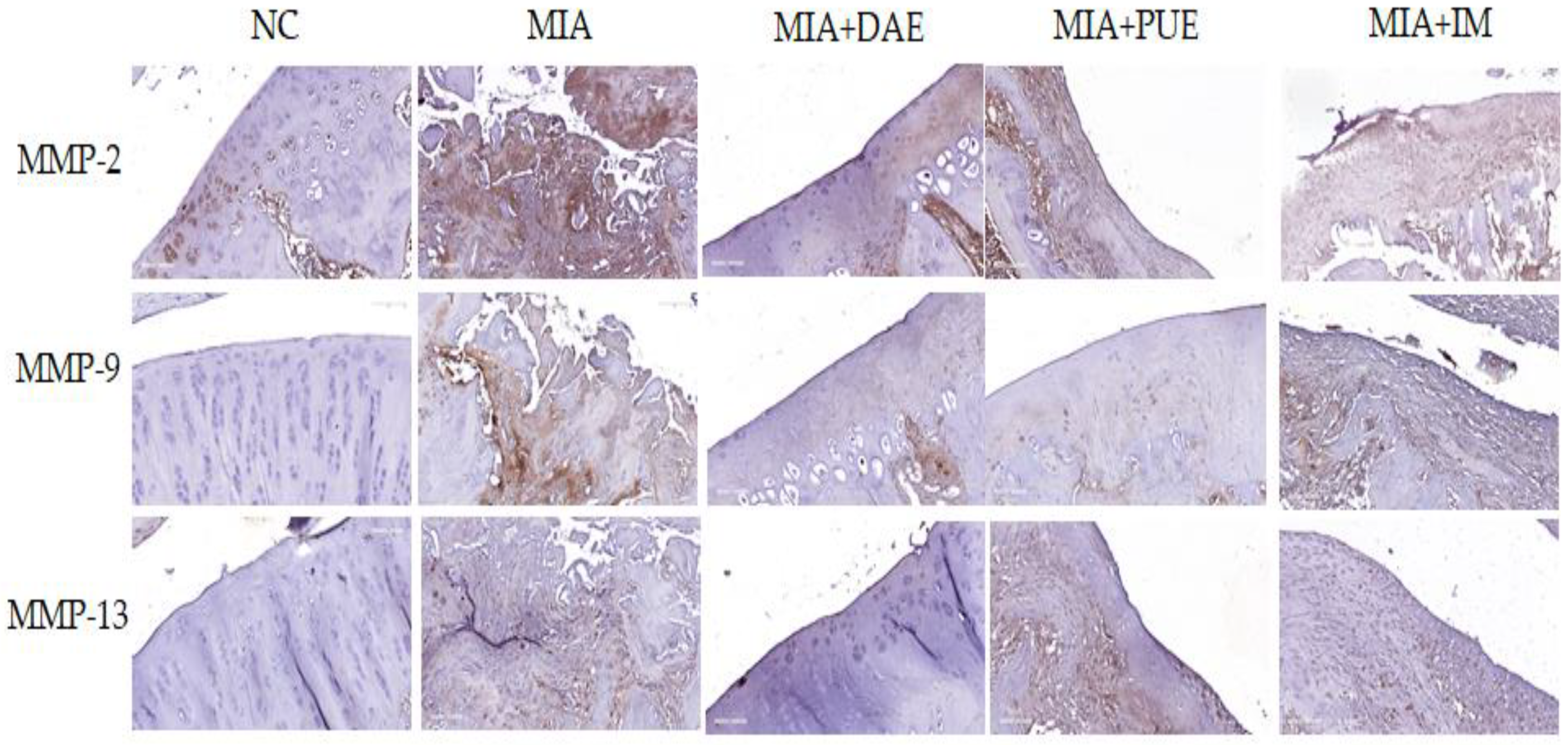
2.5. Effects of DAE and PUE on MMP-2, MMP-9, and MMP-13 Expression in Knee Joint Tissues in MIA-Induced OA Rats
3. Discussion
4. Materials and Methods
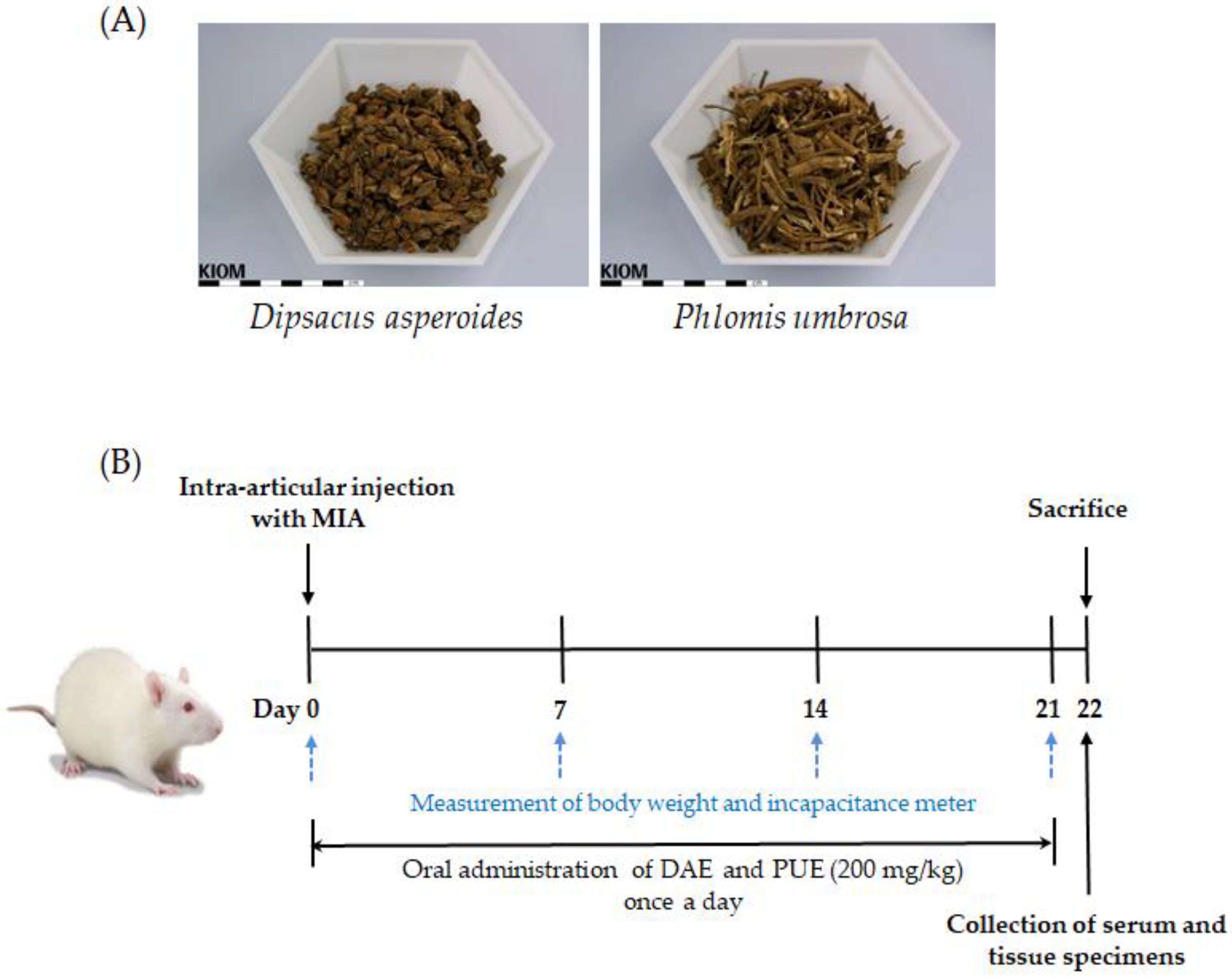
4.1. Plant Materials
4.2. Preparation of Herbal Extracts
4.3. Animals
4.4. Measurement of Hind Paw Weight-Bearing Distribution
4.5. Biochemical Blood Analysis
4.6. Histopathological Analysis
4.7. Immunohistochemical Analysis
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ALT | Alanine aminotransferase |
| AST | Aspartate aminotransferase |
| DAE | Dipsacus asperoides extract |
| H&E | Hematoxylin and eosin |
| IL-1β | Interleukin-1 beta |
| IM | Indomethacin |
| KHP | Korean Herbal Pharmacopoeia |
| MIA | Monosodium iodoacetate |
| MMPs | Matrix metalloproteinases |
| OA | Osteoarthritis |
| PUE | Phlomis umbrosa extract |
| Safranin O | Safranin O-fast green |
| SCAR | Sequence-characterized amplified region |
| TNF-α | Tumor necrosis factor alpha |
References
- Mandl, L.A. Osteoarthritis year in review 2018: Clinical. Osteoarthr. Cartil. 2019, 27, 359–364. [Google Scholar] [CrossRef] [Green Version]
- Turkiewicz, A.; Petersson, I.F.; Björk, J.; Hawker, G.; Dahlberg, L.E.; Lohmander, L.S.; Englund, M. Current and future impact of osteoarthritis on health care: A population-based study with projections to year 2032. Osteoarthr. Cartil. 2014, 22, 1826–1832. [Google Scholar] [CrossRef] [Green Version]
- Chen, D.; Shen, J.; Zhao, W.; Wang, T.; Han, L.; Hamilton, J.L.; Im, H.J. Osteoarthritis: Toward a comprehensive understanding of pathological mechanism. Bone Res. 2017, 5, 16044. [Google Scholar] [CrossRef] [PubMed]
- Chun, J.M.; Lee, A.Y.; Kim, J.S.; Choi, G.; Kim, S.H. Protective Effects of Peucedanum japonicum Extract against Osteoarthritis in an Animal Model Using a Combined Systems Approach for Compound-Target Prediction. Nutrients 2018, 10, 754. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mével, E.; Merceron, C.; Vinatier, C.; Krisa, S.; Richard, T.; Masson, M.; Lesoeur, J.; Hivernaud, V.; Gauthier, O.; Abadie, J.; et al. Olive and grape seed extract prevents post-traumatic osteoarthritis damages and exhibits in vitro anti IL-1β activities before and after oral consumption. Sci. Rep. 2016, 6, 33527. [Google Scholar] [CrossRef] [Green Version]
- Nagoya Protocol. Available online: https://en.wikipedia.org/wiki/Convention_on_Biological_Diversity (accessed on 17 May 2021).
- Lee, A.R.; Chun, J.M.; Lee, A.Y.; Kim, H.S.; Gu, G.J.; Kwon, B.I. Reduced allergic lung inflammation by root extracts from two species of Peucedanum through inhibition of Th2 cell activation. J. Ethnopharmacol. 2017, 196, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Yea, S.J.; Kim, B.Y.; Kim, C.; Yi, M.Y. A framework for the targeted selection of herbs with similar efficacy by exploiting drug repositioning technique and curated biomedical knowledge. J. Ethnopharmacol. 2017, 208, 117–128. [Google Scholar] [CrossRef]
- Park, I.; Yang, S.; Kim, W.J.; Noh, P.; Lee, H.O.; Moon, B.C. Authentication of Herbal Medicines Dipsacus asper and Phlomoides umbrosa Using DNA Barcodes, Chloroplast Genome, and Sequence Characterized Amplified Region (SCAR) Marker. Molecules 2018, 23, 1748. [Google Scholar] [CrossRef] [Green Version]
- Korea Institute of Oriental Medicine. Defining Dictionary for Medicinal Herbs [Korean, ‘Hanyak Giwon Sajeon’]. Available online: https://oasis.kiom.re.kr/herblib/hminfo/hbmcod/hbmcodList.do (accessed on 17 May 2021).
- Gong, L.L.; Yang, S.; Liu, H.; Zhang, W.; Ren, L.L.; Han, F.F.; Lv, Y.L.; Wan, Z.R.; Liu, L.H. Anti-nociceptive and anti-inflammatory potentials of Akebia saponin D. Eur. J. Pharmacol. 2019, 845, 85–90. [Google Scholar] [CrossRef]
- WHO. Medicinal Plants in the Republic of Korea: Information on 150 Commonly Used Medicinal Plants; WHO Regional Office for the Western Pacific: Manila, Philippines, 1998. [Google Scholar]
- Lee, D.; Kim, Y.S.; Song, J.; Kim, H.S.; Lee, H.J.; Guo, H.; Kim, H. Effects of Phlomis umbrosa Root on Longitudinal Bone Growth Rate in Adolescent Female Rats. Molecules 2016, 21, 461. [Google Scholar] [CrossRef] [Green Version]
- Shang, X.; Wang, J.; Li, M.; Miao, X.; Pan, H.; Yang, Y.; Wang, Y. Antinociceptive and anti-inflammatory activities of Phlomis umbrosa Turcz extract. Fitoterapia 2011, 82, 716–721. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.E.; Lee, H.; Kim, M.H.; Yang, W.M. Osteogenic effects of Phlomis umbrosa via up-regulation of Runx2 in osteoporosis. Biomed Rep. 2019, 10, 17–22. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chun, J.M.; Lee, A.Y.; Nam, J.Y.; Lee, M.Y.; Choe, M.S.; Lim, K.S.; Kim, C.; Kim, J.S. Protective effects of Phlomis umbrosa extract on a monosodium iodoacetate-induced osteoarthritis model and prediction of molecular mechanisms using transcriptomics. Phytomedicine 2021, 81, 153429. [Google Scholar] [CrossRef] [PubMed]
- Chun, J.M.; Lee, A.Y.; Nam, J.Y.; Lim, K.S.; Choe, M.S.; Lee, M.Y.; Kim, C.; Kim, J.S. Effects of Dipsacus asperoides Extract on Monosodium Iodoacetate-Induced Osteoarthritis in Rats Based on Gene Expression Profiling. Front. Pharmacol. 2021, 12, 615157. [Google Scholar] [CrossRef] [PubMed]
- McGill, M.R. The past and present of serum aminotransferases and the future of liver injury biomarkers. Excli J. 2016, 15, 817–828. [Google Scholar] [CrossRef]
- Atsamo, A.D.; Nguelefack, T.B.; Datté, J.Y.; Kamanyi, A. Acute and subchronic oral toxicity assessment of the aqueous extract from the stem bark of Erythrina senegalensis DC (Fabaceae) in rodents. J. Ethnopharmacol. 2011, 134, 697–702. [Google Scholar] [CrossRef]
- Singh, A.; Bhat, T.K.; Sharma, O.P. Clinical Biochemistry of Hepatotoxicity. J. Clin. Toxicol. 2011. [Google Scholar] [CrossRef] [Green Version]
- Sung, Y.Y.; Kim, D.S.; Kim, H.K. Viola mandshurica ethanolic extract prevents high-fat-diet-induced obesity in mice by activating AMP-activated protein kinase. Environ. Toxicol. Pharmacol. 2014, 38, 41–50. [Google Scholar] [CrossRef]
- He, Q.; Su, G.; Liu, K.; Zhang, F.; Jiang, Y.; Gao, J.; Liu, L.; Jiang, Z.; Jin, M.; Xie, H. Sex-specific reference intervals of hematologic and biochemical analytes in Sprague-Dawley rats using the nonparametric rank percentile method. PLoS ONE 2017, 12, e0189837. [Google Scholar] [CrossRef] [Green Version]
- Matsuzawa, T.; Nomura, M.; Unno, T. Clinical pathology reference ranges of laboratory animals. Working Group II, Nonclinical Safety Evaluation Subcommittee of the Japan Pharmaceutical Manufacturers Association. J. Vet. Med. Sci. 1993, 55, 351–362. [Google Scholar] [CrossRef] [Green Version]
- Troeberg, L.; Nagase, H. Proteases involved in cartilage matrix degradation in osteoarthritis. Biochim. Biophys. Acta 2012, 1824, 133–145. [Google Scholar] [CrossRef] [Green Version]
- Shin, N.R.; Lee, A.Y.; Park, G.; Ko, J.W.; Kim, J.C.; Shin, I.S.; Kim, J.S. Therapeutic Effect of Dipsacus asperoides C. Y. Cheng et T. M. Ai in Ovalbumin-Induced Murine Model of Asthma. Int. J. Mol. Sci. 2019, 20, 1855. [Google Scholar] [CrossRef] [Green Version]
- Liu, Z.G.; Zhang, R.; Li, C.; Ma, X.; Liu, L.; Wang, J.P.; Mei, Q.B. The osteoprotective effect of Radix Dipsaci extract in ovariectomized rats. J. Ethnopharmacol. 2009, 123, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Bove, S.E.; Calcaterra, S.L.; Brooker, R.M.; Huber, C.M.; Guzman, R.E.; Juneau, P.L.; Schrier, D.J.; Kilgore, K.S. Weight bearing as a measure of disease progression and efficacy of anti-inflammatory compounds in a model of monosodium iodoacetate-induced osteoarthritis. Osteoarthr. Cartil. 2003, 11, 821–830. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.R.; Yoo, J.J.; Kim, H.A. Therapeutics in Osteoarthritis Based on an Understanding of Its Molecular Pathogenesis. Int. J. Mol. Sci. 2018, 19, 674. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mehana, E.E.; Khafaga, A.F.; El-Blehi, S.S. The role of matrix metalloproteinases in osteoarthritis pathogenesis: An updated review. Life Sci. 2019, 234, 116786. [Google Scholar] [CrossRef] [PubMed]
- Lipari, L.; Gerbino, A. Expression of gelatinases (MMP-2, MMP-9) in human articular cartilage. Int. J. Immunopathol. Pharmacol. 2013, 26, 817–823. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shiomi, T.; Lemaître, V.; D’Armiento, J.; Okada, Y. Matrix metalloproteinases, a disintegrin and metalloproteinases, and a disintegrin and metalloproteinases with thrombospondin motifs in non-neoplastic diseases. Pathol. Int. 2010, 60, 477–496. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Sampson, E.R.; Jin, H.; Li, J.; Ke, Q.H.; Im, H.J.; Chen, D. MMP13 is a critical target gene during the progression of osteoarthritis. Arthritis Res. Ther. 2013, 15, R5. [Google Scholar] [CrossRef] [Green Version]
- Kim, S.J.; Jin, S.W.; Lee, G.H.; Kim, Y.A.; Jeong, H.G. Evaluation of Estrogenic Activity of Extract from the Herbal Mixture Cynanchum wilfordii Hemsley, Phlomis umbrosa Turczaninow, and Angelica gigas Nakai. Toxicol. Res. 2017, 33, 71–77. [Google Scholar] [CrossRef] [Green Version]
- Lee, D.; Lee, S.H.; Song, J.; Jee, H.J.; Cha, S.H.; Chang, G.T. Effects of Astragalus Extract Mixture HT042 on Height Growth in Children with Mild Short Stature: A Multicenter Randomized Controlled Trial. Phytother Res. 2018, 32, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Lee, D.; Min, B.; Bae, J.S.; Chang, G.T.; Kim, H. Safety evaluation of Astragalus extract mixture HT042 and its constituent herbs in Sprague-Dawley rats. Phytomedicine 2017, 32, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Combe, R.; Bramwell, S.; Field, M.J. The monosodium iodoacetate model of osteoarthritis: A model of chronic nociceptive pain in rats? Neurosci. Lett. 2004, 370, 236–240. [Google Scholar] [CrossRef] [PubMed]
- McDougall, J.J.; Watkins, L.; Li, Z. Vasoactive intestinal peptide (VIP) is a modulator of joint pain in a rat model of osteoarthritis. Pain 2006, 123, 98–105. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.S.; Son, Y.; Bae, M.J.; Lee, M.; Lee, C.G.; Jo, W.S.; Kim, S.D.; Yang, K. Administration of granulocyte colony-stimulating factor with radiotherapy promotes tumor growth by stimulating vascularization in tumor-bearing mice. Oncol. Rep. 2015, 34, 147–154. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chun, J.M.; Lee, A.Y.; Moon, B.C.; Choi, G.; Kim, J.-S. Effects of Dipsacus asperoides and Phlomis umbrosa Extracts in a Rat Model of Osteoarthritis. Plants 2021, 10, 2030. https://doi.org/10.3390/plants10102030
Chun JM, Lee AY, Moon BC, Choi G, Kim J-S. Effects of Dipsacus asperoides and Phlomis umbrosa Extracts in a Rat Model of Osteoarthritis. Plants. 2021; 10(10):2030. https://doi.org/10.3390/plants10102030
Chicago/Turabian StyleChun, Jin Mi, A Yeong Lee, Byeong Cheol Moon, Goya Choi, and Joong-Sun Kim. 2021. "Effects of Dipsacus asperoides and Phlomis umbrosa Extracts in a Rat Model of Osteoarthritis" Plants 10, no. 10: 2030. https://doi.org/10.3390/plants10102030
APA StyleChun, J. M., Lee, A. Y., Moon, B. C., Choi, G., & Kim, J.-S. (2021). Effects of Dipsacus asperoides and Phlomis umbrosa Extracts in a Rat Model of Osteoarthritis. Plants, 10(10), 2030. https://doi.org/10.3390/plants10102030





