UV-Accelerated Photocatalytic Degradation of Pesticide over Magnetite and Cobalt Ferrite Decorated Graphene Oxide Composite
Abstract
1. Introduction
2. Results
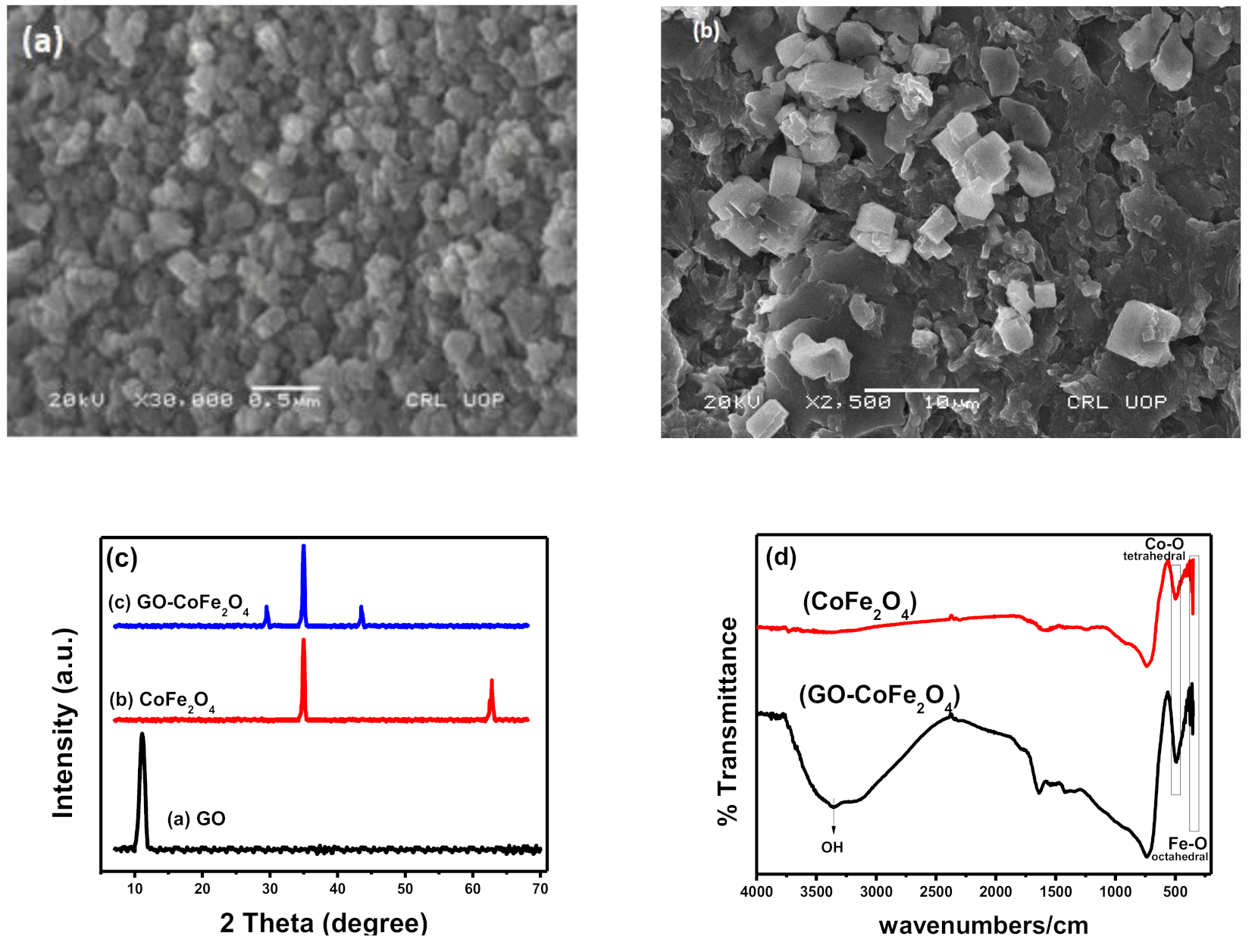
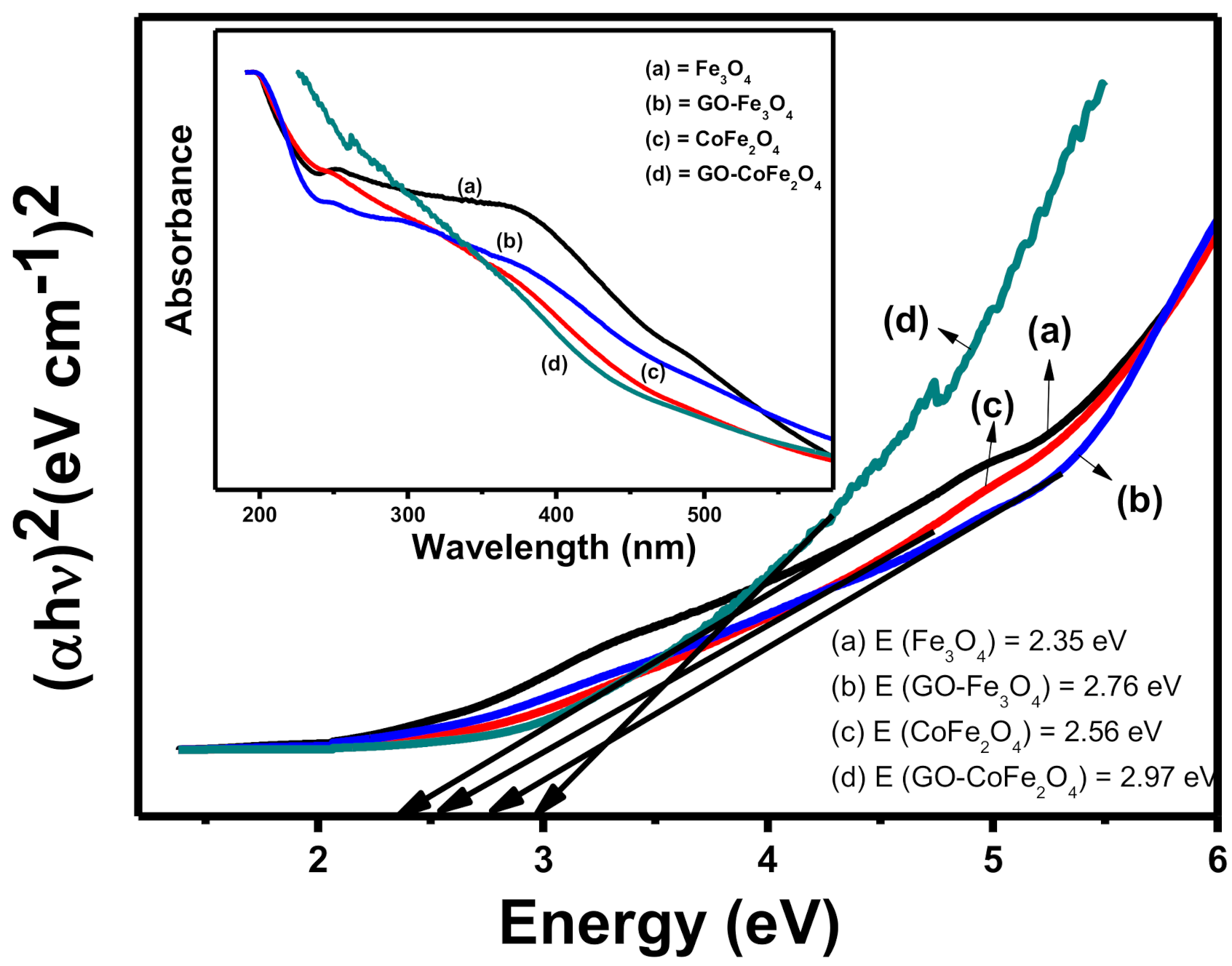
Characterization
3. Discussion
3.1. Degradation Potential of Catalysts against Acetamiprid’s Remediation
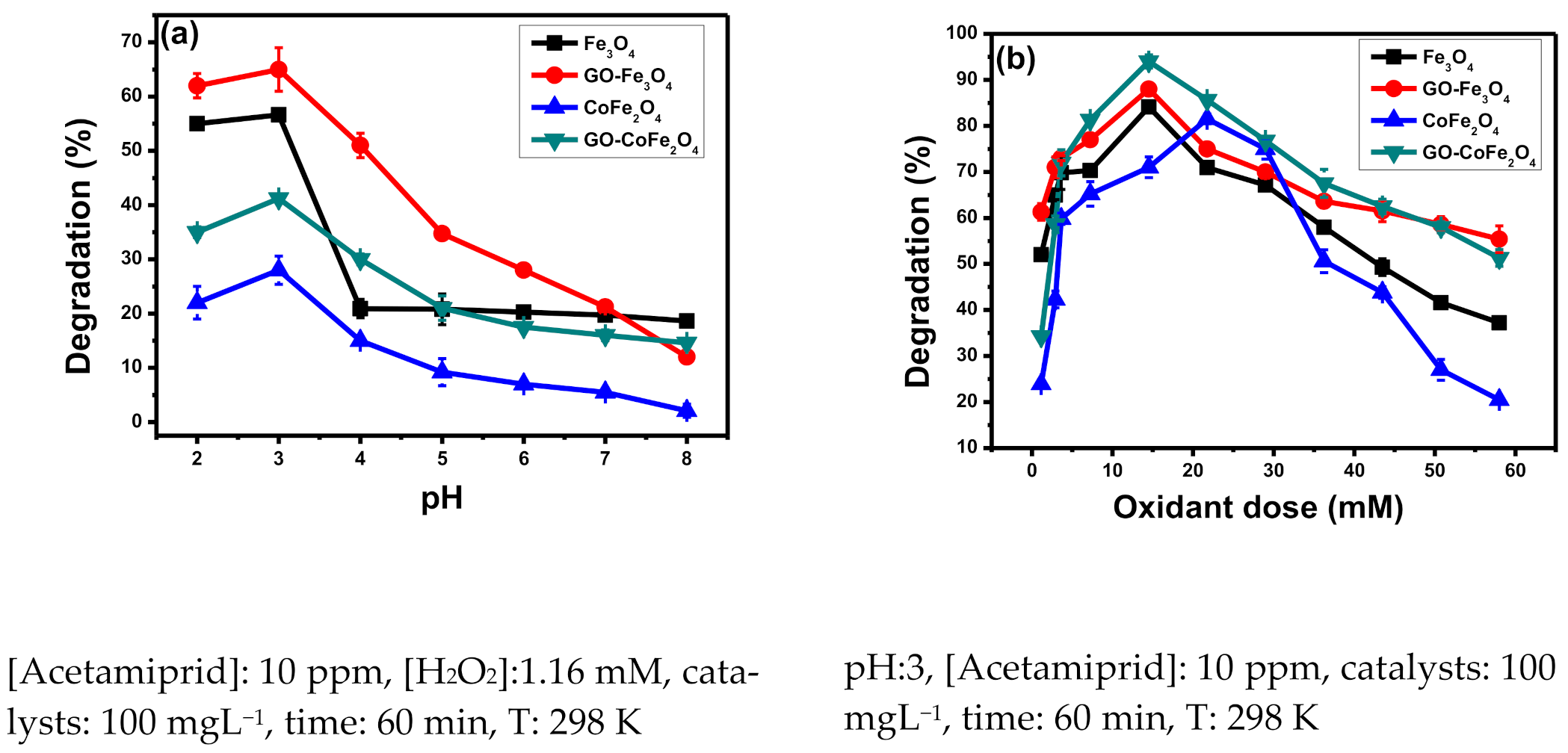
3.1.1. Effect of pH
3.1.2. Effect of Oxidant Dose
- (a)
- If oxidant in the photochemical reaction is added in a sufficient amount, it will absorb photons from the environment and help in promoting the rate of photolysis reaction occurring at the catalyst’s surface (Equation (5)). Consequently, more hydroxyl radicals would be available for pollutant degradation. Hydrogen peroxide not only enhances the Fenton’s reaction but also promotes the reusability of the catalyst (Equation (6)).
- (b)
- On the other hand, hydroxyl radicals can efficiently react with hydrogen peroxide, so the excess of hydrogen peroxide will act as a hydroxyl radical scavenger (Equation (7)), which will lead to a decline in catalytic potential.
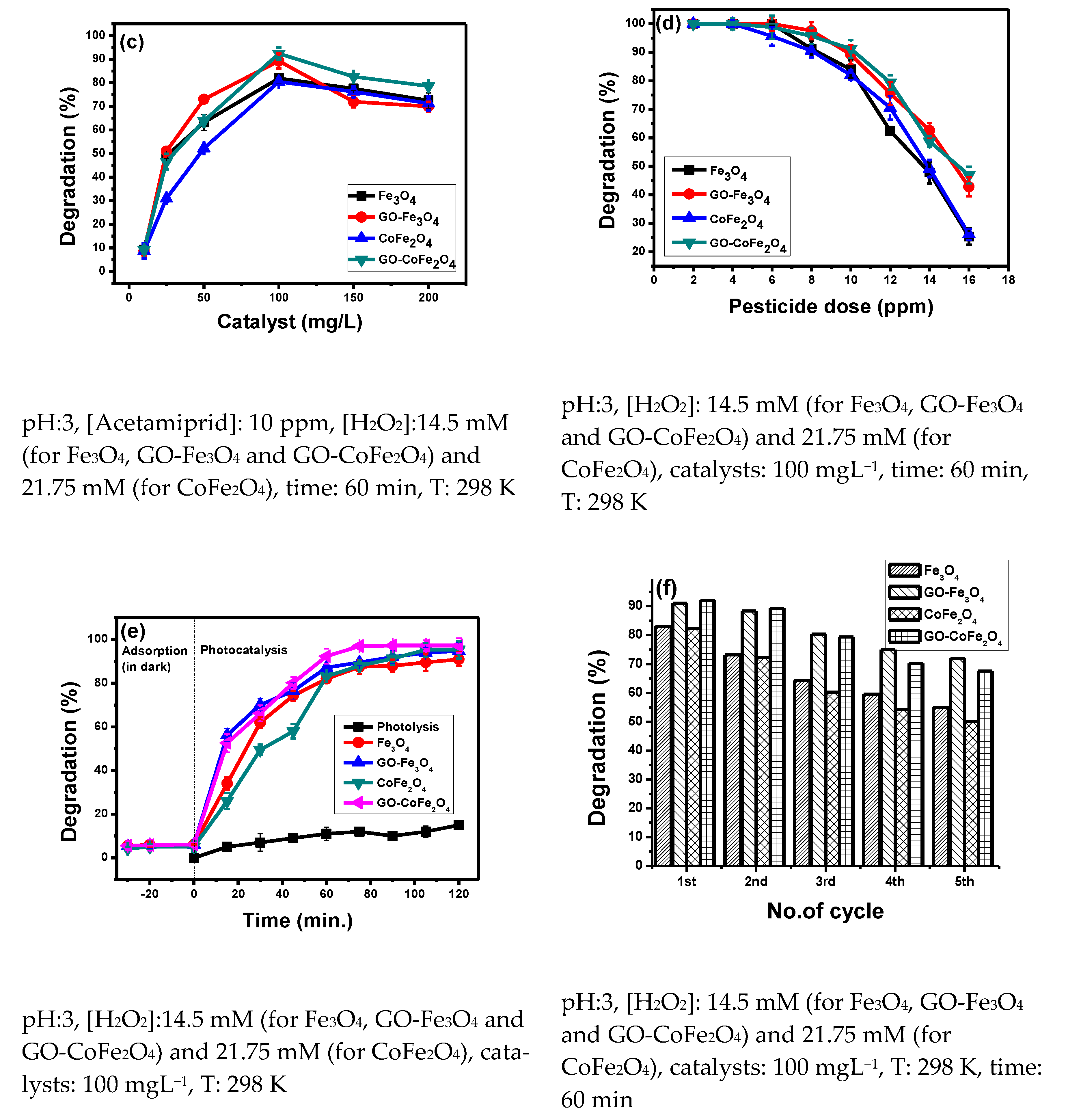
3.1.3. Effect of Catalyst Dose
3.1.4. Effect of Pesticide Load
3.1.5. Effect of Irradiation Time
3.1.6. Reusability and Stability of Catalysts
- As per EU directives, Fe leaching should not be higher than 2.0 ppm.
3.1.7. Radical Scavenging Test
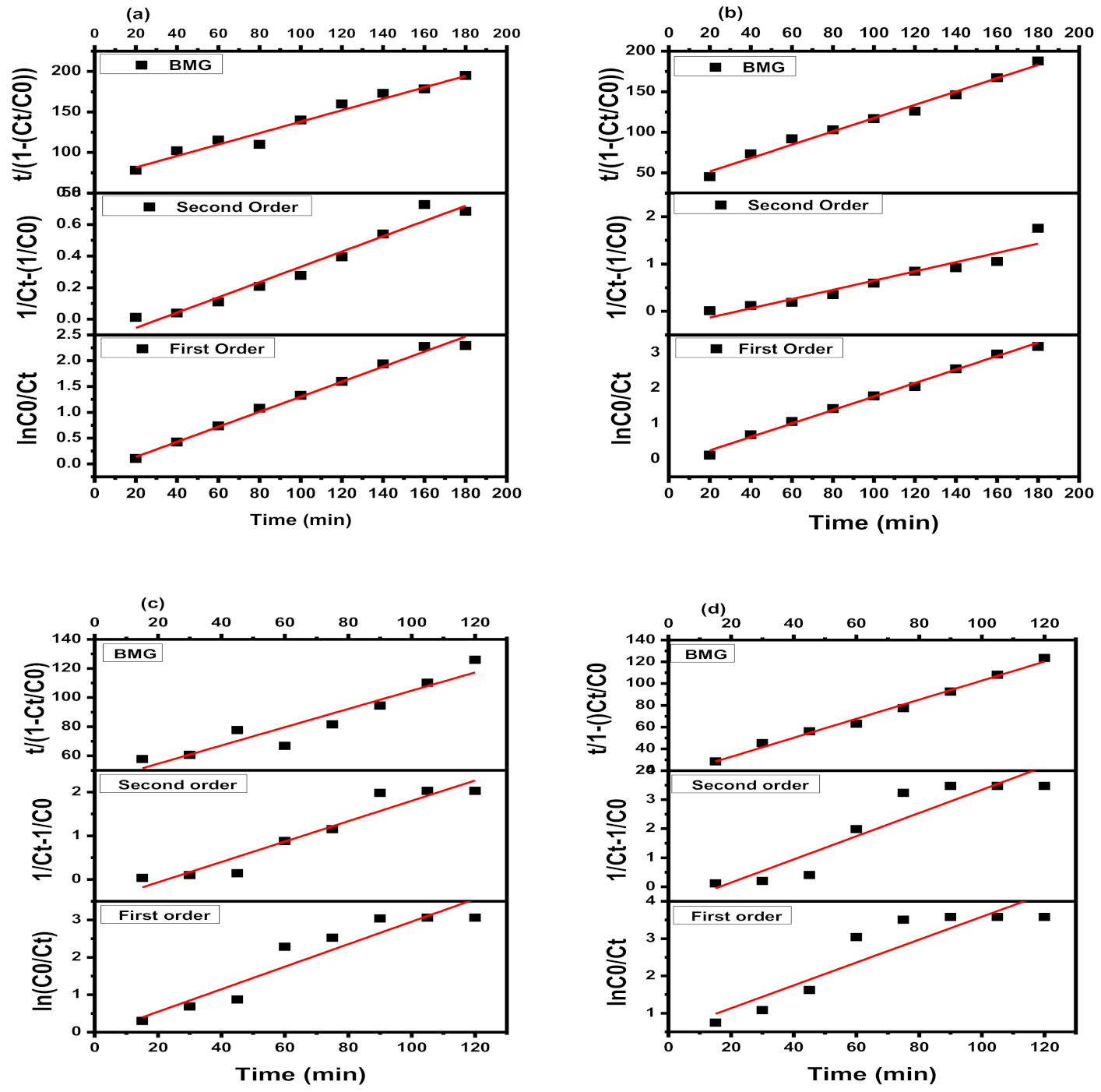
3.2. Kinetics of Photo-Degradation Reaction
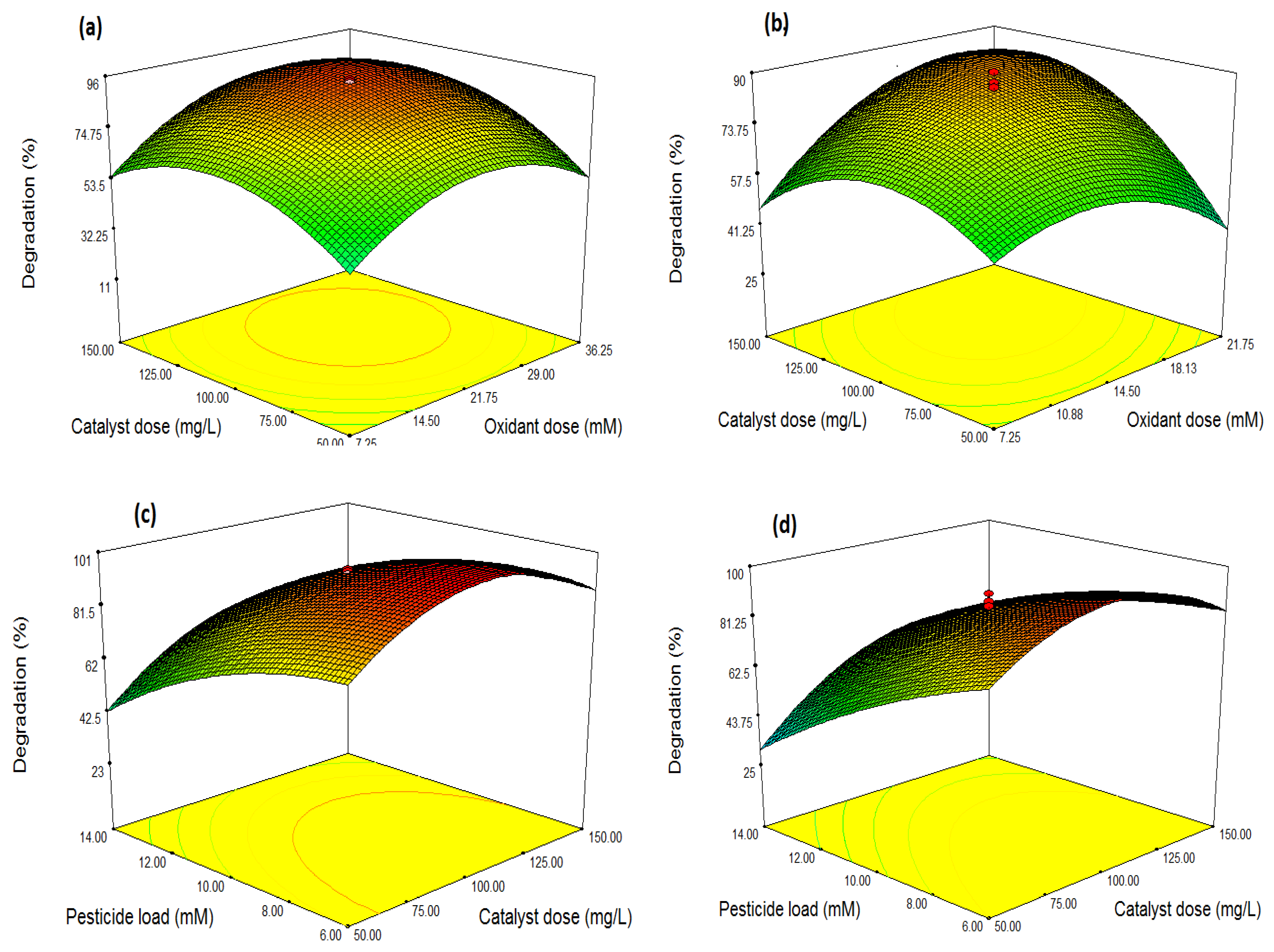
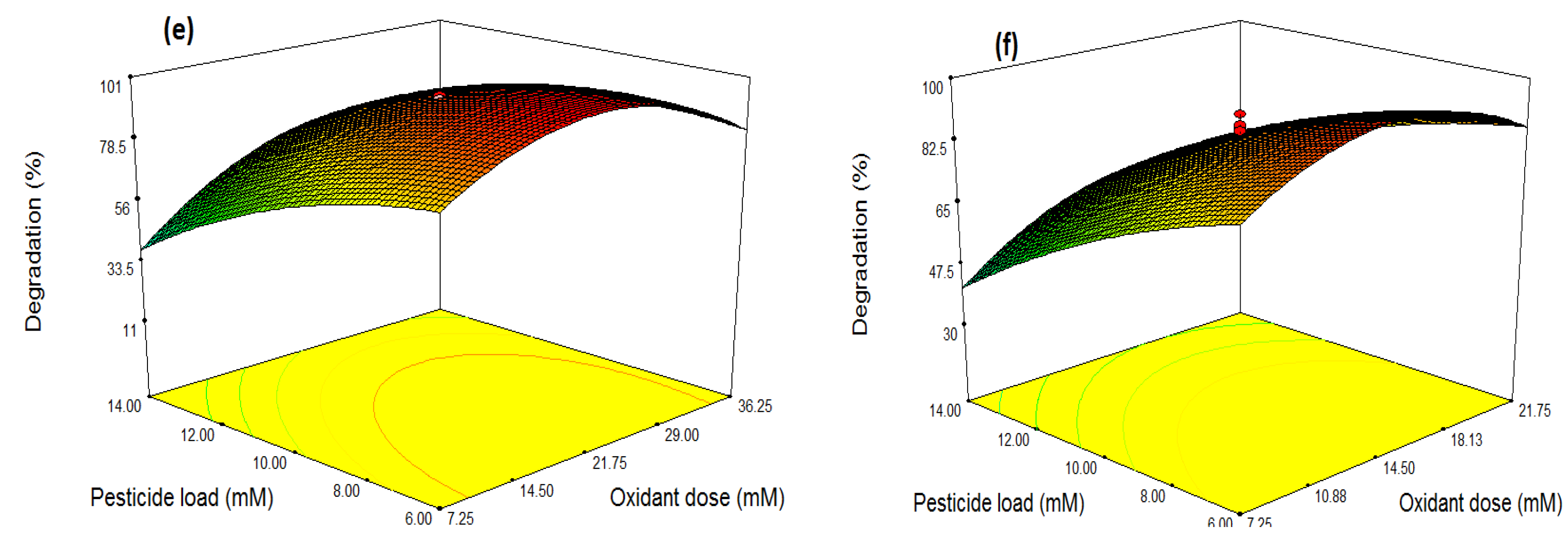
3.3. Optimization through Response Surface Methodology (RSM)
Optimization and Combined Effects of Independent Variables
4. Materials and Methods
4.1. Materials
4.2. Methods
4.2.1. Preparation of Graphene Oxide and Catalysts
4.2.2. Characterization of Catalysts
4.2.3. Degradation Experiment
4.2.4. Kinetic Study of Degradation Reaction
4.2.5. Optimization Using Response Surface Methodology (RSM)
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Loewy, R.M.; Monza, L.B.; Kirs, V.E.; Savini, M.C. Pesticide distribution in an agricultural environment in Argentina. J. Environ. Sci. Health Part B 2011, 46, 662–670. [Google Scholar]
- Žabar, R.D.; Dolenc, T.; Jerman, M.; Franko, P.; Trebše. Photolytic and photocatalytic degradation of 6-chloronicotinic acid. Chemosphere 2011, 85, 861–868. [Google Scholar] [PubMed]
- Huang, Z.H.; Li, Y.; Wei, J.; Xiong, J.Y. Distribution and ecological risk of neonicotinoid insecticides in sediment in South China: Impact of regional characteristics and chemical properties. Sci. Total Environ. 2020, 714, 136878. [Google Scholar] [PubMed]
- Dujaković, N.S.; Grujić, M.; Radišić, T.; Vasiljević, M.L. Determination of pesticides in surface and ground waters by liquid chromatography–electrospray–tandem mass spectrometry. Anal. Chim. Acta 2010, 678, 63–72. [Google Scholar]
- Tabasum, A.M.; Zahid, H.N.; Bhatti, M.A. Fe3O4-GO composite as efficient heterogeneous photo-Fenton’s catalyst to degrade pesticides. Mater. Res. Express 2019, 6, 015608. [Google Scholar]
- Zahid, M.N.; Nadeem, N.; Tahir, M.I.; Majeed, S.A.R.; Naqvi, T.H. Hybrid Nanomaterials for Water Purification. In Multifunctional Hybrid Nanomaterials for Sustainable Agri-Food and Ecosystems; Elsevier: Amsterdam, The Netherlands, 2020; pp. 155–188. [Google Scholar]
- Matsuda, K.S.D.; Buckingham, D.; Kleier, J.J.; Rauh, M.; Grauso, D.B.S. Neonicotinoids: Insecticides acting on insect nicotinic acetylcholine receptors. Trends Pharmacol. Sci. 2001, 22, 573–580. [Google Scholar]
- Saleh, I.A.N.; Zouari, M.A.; Ghouti, M.A. Removal of pesticides from water and wastewater: Chemical, physical and biological treatment approaches. Environ. Technol. Innov. 2020, 19, 101026. [Google Scholar]
- Imamura, T.Y.; Yanagawa, K.; Nishikawa, N.; Matsumoto, T.; Sakamoto, T. Two cases of acute poisoning with acetamiprid in humans. Clin. Toxicol. 2010, 48, 851–853. [Google Scholar]
- Ma, X.; Li, H.; Xiong, J.; Mehler, W.T.; You, J. Developmental toxicity of a neonicotinoid insecticide, acetamiprid to zebrafish embryos. J. Agric. Food Chem. 2019, 67, 2429–2436. [Google Scholar]
- Saher, R.; Hanif, M.; Mansha, A.; Javed, H.; Zahid, M.; Nadeem, N.; Mustafa, G.; Shaheen, A.; Riaz, O. Sunlight-driven photocatalytic degradation of rhodamine B dye by Ag/FeWO4/gC3N4 composites. Int. J. Environ. Sci. Technol. 2020. [CrossRef]
- Nadeem, N.; Zahid, M.; Tabasum, A.; Mansha, A.; Jilani, I.A.; Bhatti, H.A.; Bhatti, N. Degradation of reactive dye using heterogeneous photo-Fenton catalysts: ZnFe2O4 and GO-ZnFe2O4 composite. Mater. Res. Express 2020, 7, 015519. [Google Scholar]
- Zhang, Z.; Hossain, F.M.; Takahashi, T. Self-assembled hematite (α-Fe2O3) nanotube arrays for photoelectrocatalytic degradation of azo dye under simulated solar light irradiation. Appl. Catal. B Environ. 2010, 95, 423–429. [Google Scholar]
- Bandara, J.; Klehm, U.; Kiwi, J. Raschig rings-Fe2O3 composite photocatalyst activate in the degradation of 4-chlorophenol and Orange II under daylight irradiation. Appl. Catal. B Environ. 2007, 76, 73–81. [Google Scholar]
- Zahid, M.; Nadeem, N.; Hanif, M.A.; Bhatti, I.A.; Bhatti, H.N.; Mustafa, G. Metal Ferrites and Their Graphene-Based Nanocomposites: Synthesis, Characterization and Applications in Wastewater Treatment. In Magnetic Nanostructures; Springer: Cham, Switzerland, 2019; pp. 181–212. [Google Scholar]
- Pignatello, J.J.; Oliveros, E.; MacKay, A. Advanced oxidation processes for organic contaminant destruction based on the Fenton reaction and related chemistry. Crit. Rev. Environ. Sci. Technol. 2006, 36, 1–84. [Google Scholar]
- Zhou, T.; Li, Y.; Ji, J.; Wong, F.S.; Lu, X. Oxidation of 4-chlorophenol in a heterogeneous zero valent iron/H2O2 Fenton-like system: Kinetic, pathway and effect factors. Sep. Purif. Technol. 2008, 62, 551–558. [Google Scholar]
- Murase, T.; Irie, H.; Hashimoto, K. Visible light sensitive photocatalysts, nitrogen-doped Ta2O5 powders. J. Phys. Chem. B 2004, 108, 15803–15807. [Google Scholar]
- Pradhan, G.K.; Sahu, N.; Parida, K. Fabrication of S, N co-doped α-Fe2 O3 nanostructures: Effect of doping, OH radical formation, surface area,[110] plane and particle size on the photocatalytic activity. Rsc Adv. 2013, 3, 7912–7920. [Google Scholar]
- Gumy, D.; Giraldo, S.; Rengifo, J.; Pulgarin, C. Effect of suspended TiO2 physicochemical characteristics on benzene derivatives photocatalytic degradation. Appl. Catal. B Environ. 2008, 78, 19–29. [Google Scholar]
- Choi, Y.W.; Lee, H.; Song, Y.; Sohn, D. Colloidal stability of iron oxide nanoparticles with multivalent polymer surfactants. J. Colloid Interface Sci. 2015, 443, 8–12. [Google Scholar]
- Zhu, P.; Nair, A.S.; Shengjie, P.; Shengyuan, Y.; Ramakrishna, S. Facile fabrication of TiO2–graphene composite with enhanced photovoltaic and photocatalytic properties by electrospinning. ACS Appl. Mater. Interfaces 2012, 4, 581–585. [Google Scholar]
- Liu, C.; Teng, Y.; Liu, R.; Luo, S.; Tang, Y.; Chen, L.; Cai, Q. Fabrication of graphene films on TiO2 nanotube arrays for photocatalytic application. Carbon 2011, 49, 5312–5320. [Google Scholar] [CrossRef]
- Abd, E.; Zahid, K.A.M. Aquananotechnology: Applications of Nanomaterials for Water Purification; Elsevier: Amsterdam, The Netherlands, 2020. [Google Scholar]
- Jauhar, S.; Kaur, J.; Goyal, A.; Singhal, S. Tuning the properties of cobalt ferrite: A road towards diverse applications. RSC Adv. 2016, 6, 97694–97719. [Google Scholar] [CrossRef]
- Wang, G.; Ma, Y.; Tong, Y.; Dong, X. Development of manganese ferrite/graphene oxide nanocomposites for magnetorheological fluid with enhanced sedimentation stability. J. Ind. Eng. Chem. 2017, 48, 142–150. [Google Scholar] [CrossRef]
- Ambikeswari, N.; Manivannan, S. Superior magnetodielectric properties of room temperature synthesized superparamagnetic cobalt ferrite–graphene oxide composite. J. Alloy. Compd. 2018, 763, 711–718. [Google Scholar] [CrossRef]
- Rani, M.; Shanker, U. Efficient photocatalytic degradation of Bisphenol A by metal ferrites nanoparticles under sunlight. Environ. Technol. Innov. 2020, 19, 100792. [Google Scholar] [CrossRef]
- Kamakshi, T.; Sundari, G.S.; Erothu, H.; Rao, T. Synthesis and characterization of graphene based iron oxide (Fe3O4) nanocomposites. Rasayan J. Chem. 2018, 11, 1113–1119. [Google Scholar] [CrossRef]
- Cong, Y.; Chen, M.; Xu, T.; Zhang, Y.; Wang, Q. Tantalum and aluminum co-doped iron oxide as a robust photocatalyst for water oxidation. Appl. Catal. B Environ. 2014, 147, 733–740. [Google Scholar] [CrossRef]
- Shao, P.; Yu, S.; Duan, X.; Yang, L.; Shi, H.; Ding, L.; Tian, J.; Yang, L.; Luo, X.; Wang, S. Potential Difference Driving Electron Transfer via Defective Carbon Nanotube towards Selective Oxidation of Organic Micro-Pollutants. Environ. Sci. Technol. 2020, 54, 8472–8474. [Google Scholar] [CrossRef]
- Shao, P.; Tian, F.; Yang, X.; Duan, S.; Gao, W.; Shi, X.; Luo, F.; Cui, S.; Luo; Wang, J. Identification and regulation of active sites on nanodiamonds: Establishing a highly efficient catalytic system for oxidation of organic contaminants. Adv. Funct. Mater. 2018, 28, 1705295. [Google Scholar]
- Shao, P.; Duan, J.; Xu, J.; Tian, W.; Shi, S.; Gao, M.; Xu, F.; Cui, S.; Luo; Wang, X. Heterogeneous activation of peroxymonosulfate by amorphous boron for degradation of bisphenol S. J. Hazard. Mater. 2017, 322, 532–539. [Google Scholar] [CrossRef]
- Sharma, G.; Kumar, S.; Sharma, M.; Naushad, T.; Ahamad, S.I.; Saeedi, G.M.; Senani, N.S.; Kadhi, F.J.; Stadler, A. Facile fabrication of Zr2Ni1Cu7 trimetallic nano-alloy and its composite with Si3N4 for visible light assisted photodegradation of methylene blue. J. Mol. Liq. 2018, 272, 170–179. [Google Scholar] [CrossRef]
- Di, L.; Yang, T.; Xian, X.; Liu, X.; Chen, H. Photocatalytic and photo-Fenton catalytic degradation activities of Z-scheme Ag2S/BiFeO3 heterojunction composites under visible-light irradiation. Nanomaterials 2019, 9, 399. [Google Scholar] [CrossRef] [PubMed]
- Shiraz, A.D.; Takdastan, S.M.; Borghei, A. Photo-Fenton like degradation of catechol using persulfate activated by UV and ferrous ions: Influencing operational parameters and feasibility studies. J. Mol. Liq. 2018, 249, 463–469. [Google Scholar] [CrossRef]
- Behnajady, M.; Modirshahla, N.; Daneshvar, M.; Rabbani, N. Photocatalytic degradation of an azo dye in a tubular continuous-flow photoreactor with immobilized TiO2 on glass plates. Chem. Eng. J. 2007, 127, 167–176. [Google Scholar] [CrossRef]
- Gallard, H.; Laat, J. Kinetics of oxidation of chlorobenzenes and phenyl-ureas by Fe (II)/H2O2 and Fe (III)/H2O2. Evidence of reduction and oxidation reactions of intermediates by Fe (II) or Fe (III). Chemosphere 2001, 42, 405–413. [Google Scholar]
- Guedes, A.M.; Madeira, R.A.; Boaventura, C.A.; Costa, L.M. Fenton oxidation of cork cooking wastewater—overall kinetic analysis. Water Res. 2003, 37, 3061–3069. [Google Scholar] [CrossRef]
- Behnajady, M.; Modirshahla, F.; Ghanbary, N. A kinetic model for the decolorization of CI Acid Yellow 23 by Fenton process. J. Hazard. Mater. 2007, 148, 98–102. [Google Scholar] [CrossRef]
- Chun, J.; Lee, S.H.; Lee, S.W.; Hong, J.; Lee, C.; Lee, J.; Lee, H. Magnetite/mesocellular carbon foam as a magnetically recoverable fenton catalyst for removal of phenol and arsenic. Chemosphere 2012, 89, 1230–1237. [Google Scholar] [CrossRef]
- Selvam, K.; Muruganandham, M.; Swaminathan, M. Enhanced heterogeneous ferrioxalate photo-fenton degradation of reactive orange 4 by solar light. Sol. Energy Mater. Sol. Cells 2005, 89, 61–74. [Google Scholar] [CrossRef]
- Tabasum, A.; Bhatti, N.; Nadeem, M.; Zahid, Z.A.; Rehan, T.; Hussain, A.; Jilani, I.A. Degradation of acetamiprid using graphene-oxide-based metal (Mn and Ni) ferrites as Fenton-like photocatalysts. Water Sci. Technol. 2020, 81, 178–189. [Google Scholar] [CrossRef]
- Yao, Y.; Cai, F.; Lu, F.; Wei, X.; Wang, S.; Wang, Y. Magnetic recoverable MnFe2O4 and MnFe2O4-graphene hybrid as heterogeneous catalysts of peroxymonosulfate activation for efficient degradation of aqueous organic pollutants. J. Hazard. Mater. 2014, 270, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Luna, M.D.G.; Briones, C.C.; Su, M.C.; Lu, R.M. Kinetics of acetaminophen degradation by Fenton oxidation in a fluidized-bed reactor. Chemosphere 2013, 90, 1444–1448. [Google Scholar] [CrossRef] [PubMed]

| Run | Fe3O4 | Fe3O4-GO | CoFe2O4 | CoFe2O4-GO | ||||
|---|---|---|---|---|---|---|---|---|
| Degradation (%) | Iron Leaching (ppm) | Degradation (%) | Iron Leaching (ppm) | Degradation (%) | Iron Leaching (ppm) | Degradation (%) | Iron Leaching (ppm) | |
| 1st | 83 | 1.28 | 91 | 0.62 | 82.3 | 1.13 | 96.43 | 0.72 |
| 2nd | 73.2 | 1.22 | 88.3 | 0.60 | 72.3 | 1.02 | 89.2 | 0.68 |
| 3rd | 64.3 | 1.02 | 80.3 | 0.53 | 70.3 | 0.94 | 79.3 | 0.62 |
| 4th | 59.6 | 0.94 | 75 | 0.50 | 54.2 | 0.88 | 70.2 | 0.57 |
| 5th | 55 | 0.79 | 72 | 0.44 | 50.1 | 0.82 | 67.6 | 0.46 |
| Catalysts | First-Order | Second-Order | BMG | ||||
|---|---|---|---|---|---|---|---|
| R2 | K1(min−1) | R2 | K2(L µmol−1min−1) | R2 | m | b | |
| Fe3O4 | 0.8971 | 0.0178 | 0.9891 | 0.0096 | 0.9767 | 24.396 | 0.8653 |
| GO-Fe3O4 | 0.8474 | 0.0193 | 0.9679 | 0.0193 | 0.9971 | 9.7356 | 0.9546 |
| CoFe2O4 | 0.8946 | 0.0233 | 0.9207 | 0.0233 | 0.9008 | 41.982 | 0.6278 |
| GO-CoFe2O4 | 0.8397 | 0.0306 | 0.8688 | 0.0400 | 0.9911 | 15.267 | 0.8745 |
| Source | Sum of Square | df | Mean Square | F Value | p-Value (Prob>F) | |
|---|---|---|---|---|---|---|
| Model | 1572.94 | 9 | 1747.22 | 2527.16 | <0.0001 | Significant |
| A-Oxidant Dose | 327.41 | 1 | 327.41 | 473.56 | <0.0001 | |
| B-Catalyst Dose | 1492.72 | 1 | 1492.72 | 2159.05 | <0.0001 | |
| C-Pesticide load | 2508.02 | 1 | 2508.02 | 3627.57 | <0.0001 | |
| AB | 632.72 | 1 | 632.72 | 915.16 | <0.0001 | |
| AC | 1211.65 | 1 | 1211.65 | 1752.52 | <0.0001 | |
| BC | 388.01 | 1 | 388.01 | 561.21 | <0.0001 | |
| A2 | 7396.43 | 1 | 7396.43 | 10698.12 | <0.0001 | |
| B2 | 2418.69 | 1 | 2418.69 | 3498.36 | <0.0001 | |
| C2 | 489.09 | 1 | 489.09 | 707.41 | <0.0001 | |
| Residual | 6.91 | 10 | .069 | |||
| Lack of Fit | 4.95 | 5 | .99 | 2.51 | 0.1674 | Non-significant |
| Pure Error | 1.97 | 5 | .39 | |||
| Cor Total | 15,731.86 | 19 | ||||
| Std. Dev. | 0.83 | R-squared | 0.99996 | |||
| Mean | 65.48 | Adj. R-squared | 0.9992 | |||
| C.V.% | 1.27 | Pred. R-squared | 0.9971 | |||
| PRESS | 45.46 | Adeq. precision | 148.021 | |||
| Source | Sum of Square | df | Mean Square | F Value | p-Value (Prob>F) | |
|---|---|---|---|---|---|---|
| Model | 12,924.55 | 9 | 1436.06 | 20.37 | <0.0001 | Significant |
| A-Oxidant Dose | 392.60 | 1 | 392.60 | 5.57 | 0.0399 | |
| B-Catalyst Dose | 1060.05 | 1 | 1060.05 | 15.04 | 0.0031 | |
| C-Pesticide load | 4225.51 | 1 | 4225.51 | 59.95 | <0.0001 | |
| AB | 1164.76 | 1 | 1164.76 | 16.52 | 0.0023 | |
| AC | 81.47 | 1 | 81.47 | 1.16 | 0.3076 | |
| BC | 354.71 | 1 | 354.71 | 5.03 | 0.0487 | |
| A2 | 2482.81 | 1 | 2482.81 | 35.22 | 0.0001 | |
| B2 | 3651.90 | 1 | 3651.90 | 51.81 | <0.0001 | |
| C2 | 296.29 | 1 | 296.29 | 4.20 | 0.0675 | |
| Residual | 704.86 | 10 | 70.49 | |||
| Lack of Fit | 497.67 | 5 | 99.53 | 2.40 | 0.1791 | Non-significant |
| Pure Error | 207.19 | 5 | 4144 | |||
| Cor Total | 13,629.40 | 19 | ||||
| Std. Dev. | 8.40 | R-squared | 0.9483 | |||
| Mean | 61.56 | Adj. R-squared | 0.9017 | |||
| C.V.% | 13.64 | Pred. R-squared | 0.6815 | |||
| PRESS | 4340.44 | Adeq. precision | 14.640 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tabasum, A.; Alghuthaymi, M.; Qazi, U.Y.; Shahid, I.; Abbas, Q.; Javaid, R.; Nadeem, N.; Zahid, M. UV-Accelerated Photocatalytic Degradation of Pesticide over Magnetite and Cobalt Ferrite Decorated Graphene Oxide Composite. Plants 2021, 10, 6. https://doi.org/10.3390/plants10010006
Tabasum A, Alghuthaymi M, Qazi UY, Shahid I, Abbas Q, Javaid R, Nadeem N, Zahid M. UV-Accelerated Photocatalytic Degradation of Pesticide over Magnetite and Cobalt Ferrite Decorated Graphene Oxide Composite. Plants. 2021; 10(1):6. https://doi.org/10.3390/plants10010006
Chicago/Turabian StyleTabasum, Asma, Mousa Alghuthaymi, Umair Yaqub Qazi, Imran Shahid, Qamar Abbas, Rahat Javaid, Nimra Nadeem, and Muhammad Zahid. 2021. "UV-Accelerated Photocatalytic Degradation of Pesticide over Magnetite and Cobalt Ferrite Decorated Graphene Oxide Composite" Plants 10, no. 1: 6. https://doi.org/10.3390/plants10010006
APA StyleTabasum, A., Alghuthaymi, M., Qazi, U. Y., Shahid, I., Abbas, Q., Javaid, R., Nadeem, N., & Zahid, M. (2021). UV-Accelerated Photocatalytic Degradation of Pesticide over Magnetite and Cobalt Ferrite Decorated Graphene Oxide Composite. Plants, 10(1), 6. https://doi.org/10.3390/plants10010006








