Analgesic and Anti-Inflammatory Effects of Aucklandia lappa Root Extracts on Acetic Acid-Induced Writhing in Mice and Monosodium Iodoacetate-Induced Osteoarthritis in Rats
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
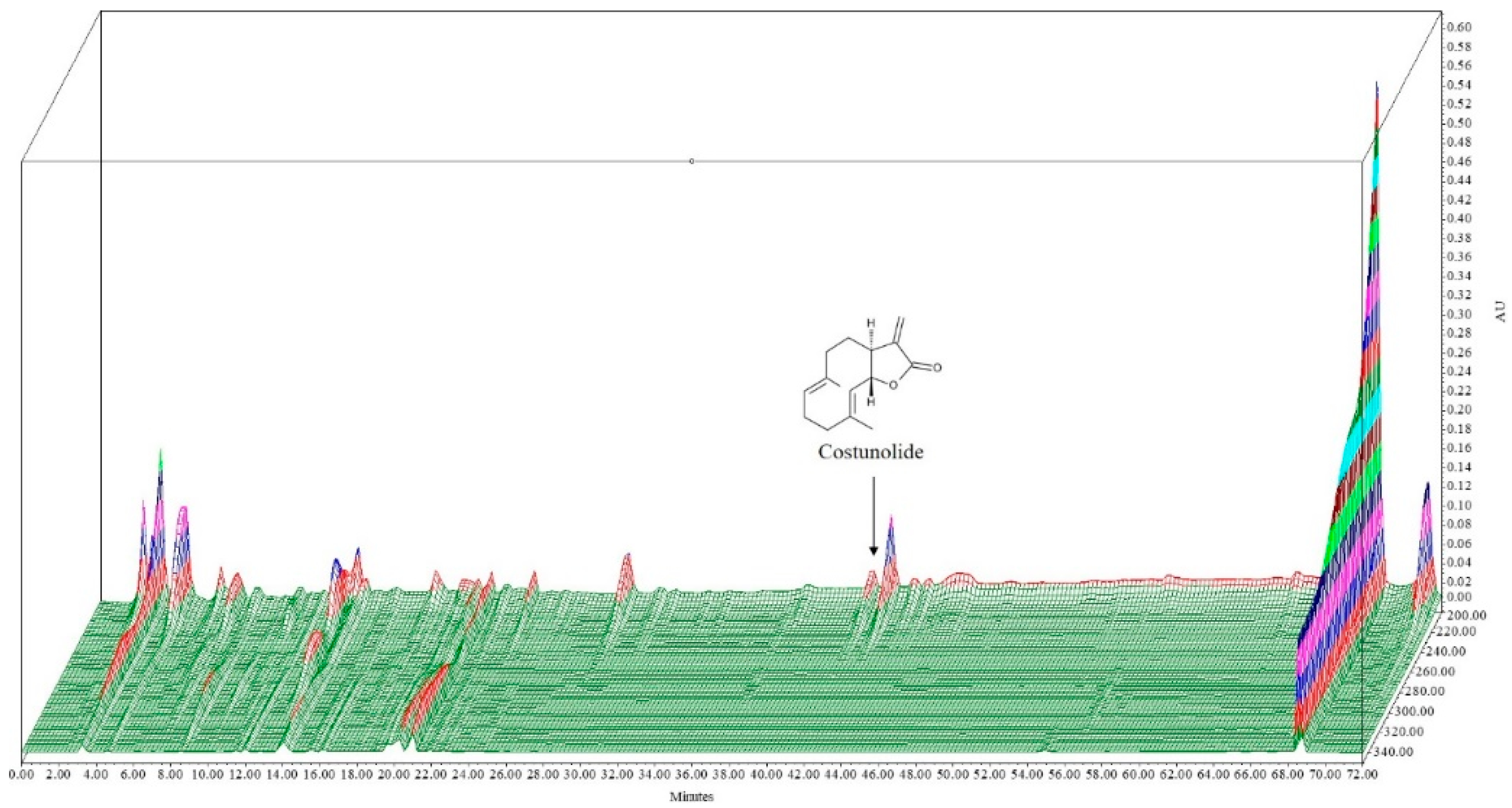
2.2. HPLC Analysis of A. lappa Extract
2.3. Animal Housing and Management
2.4. MIA Injection and Treatment
2.5. Weight Bearing Measurement
2.6. Serum Analysis
2.7. Micro-Computed Tomography Analysis
2.8. Acetic Acid Induced Writhing Responses
2.9. Cell Culture
2.10. NO and Cytotoxicity Measurement
2.11. Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR) Analysis
2.12. Protein Expression Analysis
2.13. Statistical Analysis
3. Results
3.1. HPLC Analysis
3.2. Weight Bearing Distribution of MIA Rats
3.3. Prevention of Knee Joint Damage in MIA Rats
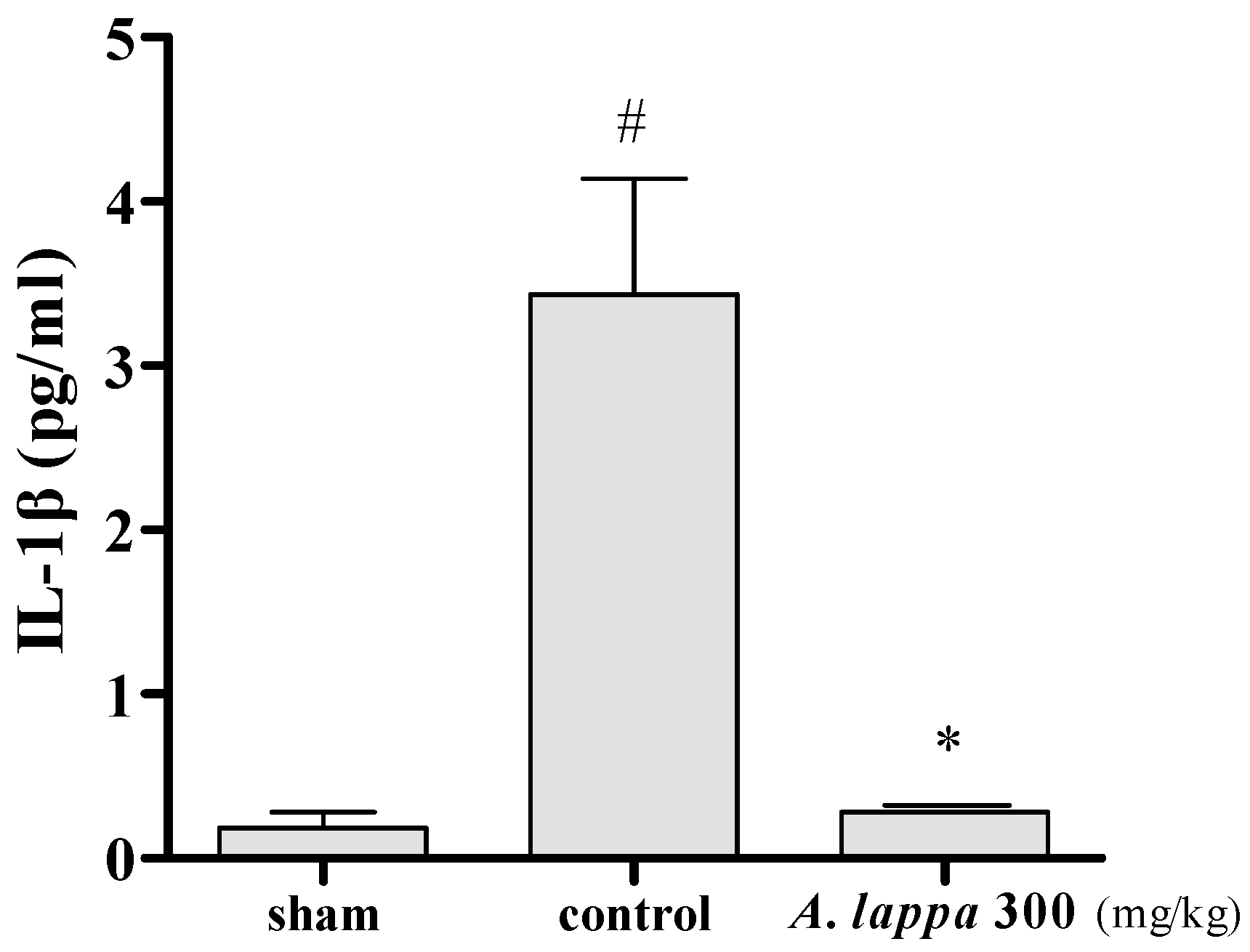
3.4. Inflammatory Cytokines in MIA Rats
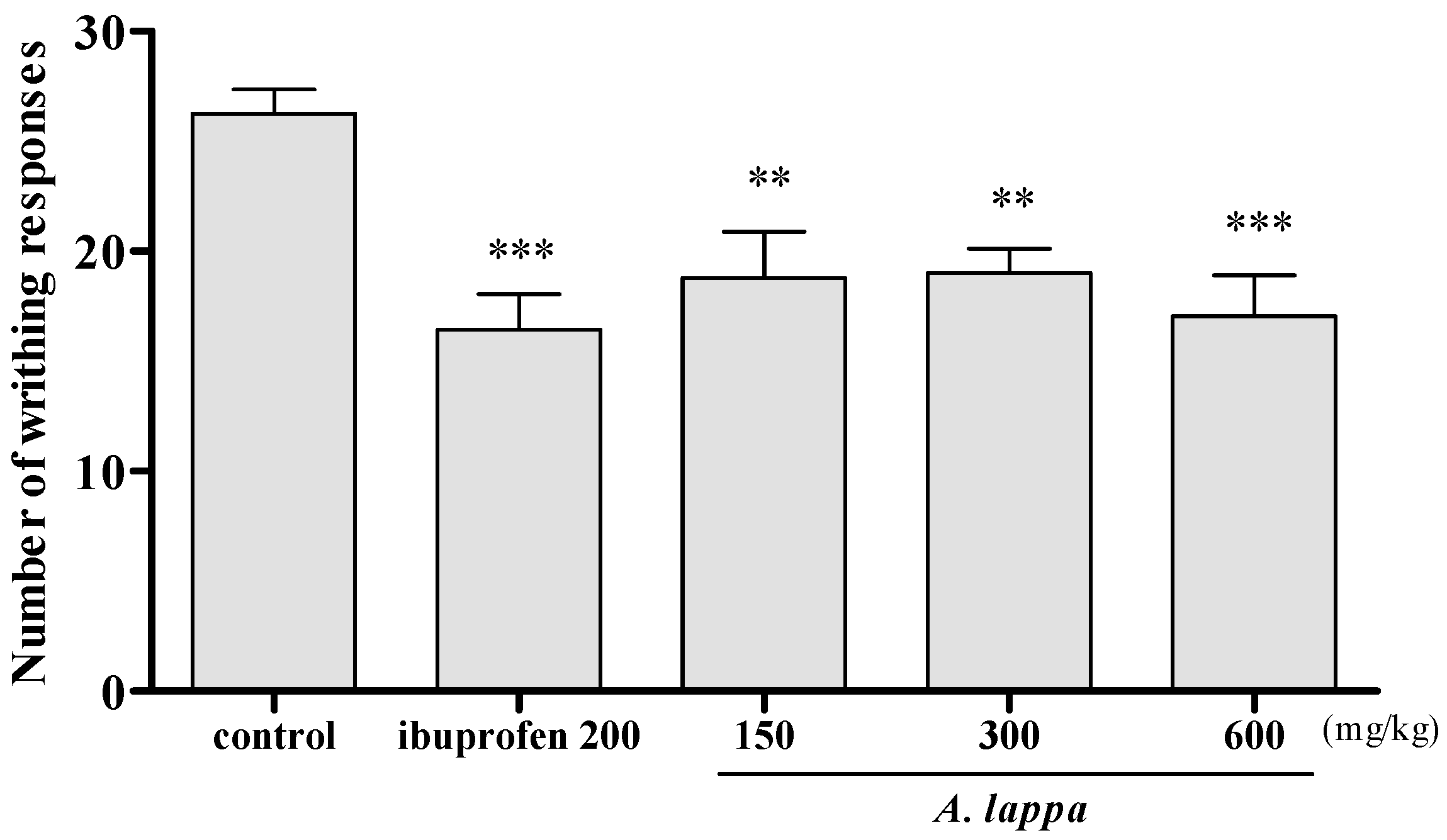
3.5. Acetic Acid Induced Writhing Responses
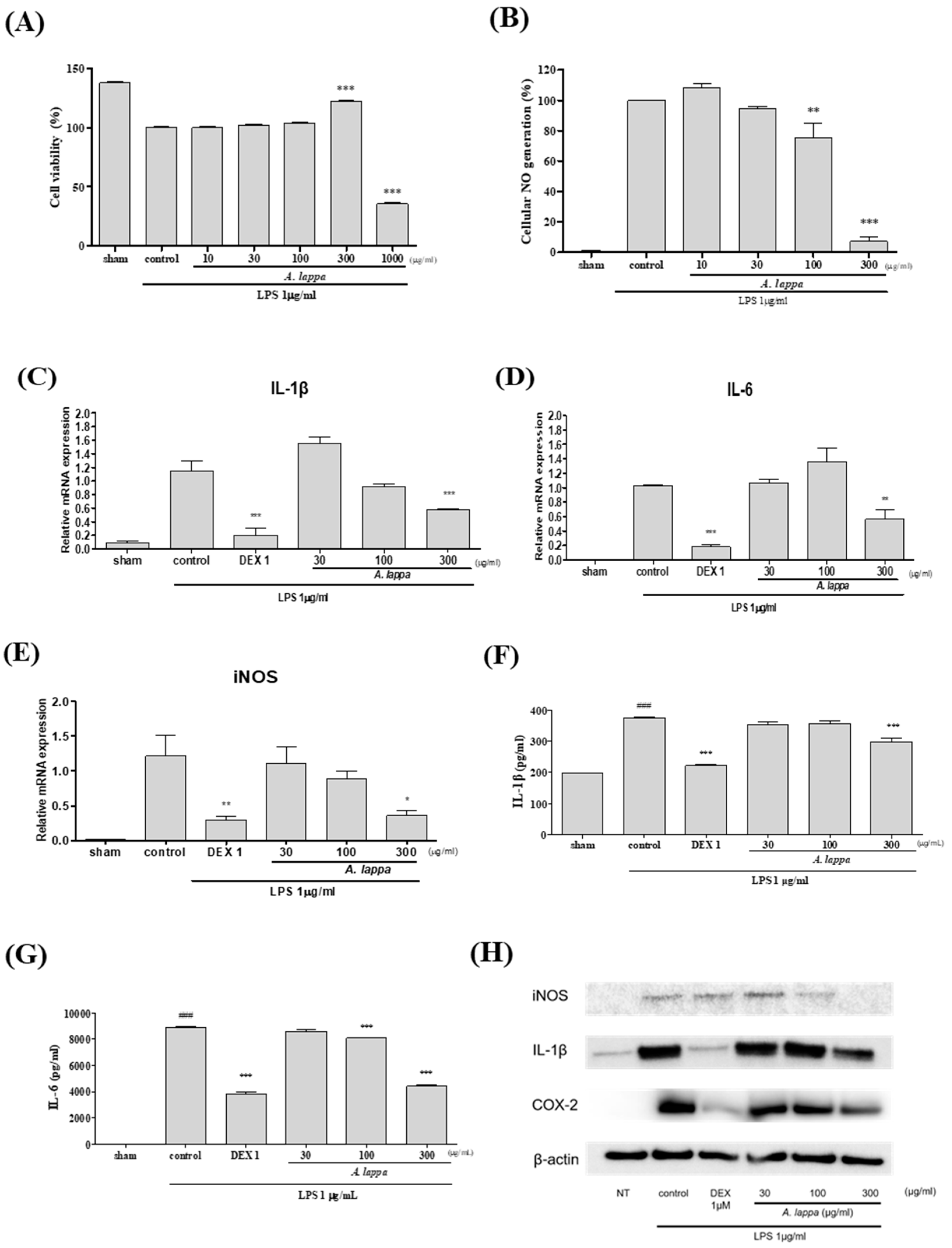
3.6. Effects of A. lappa on Inflammatory Response in LPS-Activated RAW264.7 Cells
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- O’Brien, M.; Philpott, H.T.; McDougall, J.J. Understanding osteoarthritis pain through animal models. Clin. Exp. Rheumatol. 2017, 35, S47–S52. [Google Scholar]
- Bijlsma, J.W.J.; Berenbaum, F.; Lafeber, F.P.J.G. Osteoarthritis: An update with relevance for clinical practice. Lancet 2011, 377, 2115–2126. [Google Scholar] [CrossRef]
- Burrage, P.S.; Mix, K.S.; Brinckerhoff, C.E. Matrix metalloproteinases: Role in arthritis. Front. Biosci. 2006, 11, 529–543. [Google Scholar] [CrossRef] [PubMed]
- Goldring, M.B.; Otero, M. Beyond individual Piaget and Vygotsky.pdf. Curr. Opin. Rheumatol. 2014, 23, 471–478. [Google Scholar] [CrossRef]
- Tantowi, N.A.C.A.; Lau, S.F.; Mohamed, S. Ficus deltoidea Prevented Bone Loss in Preclinical Osteoporosis/Osteoarthritis Model by Suppressing Inflammation. Calcif. Tissue Int. 2018, 103, 388–399. [Google Scholar] [CrossRef]
- Sharma, V.K.; Mamontov, E.; Tyagi, M. Effects of NSAIDs on the nanoscopic dynamics of lipid membrane. Biochim. Biophys. Acta (BBA) Biomembr. 2020, 1862, 183100. [Google Scholar] [CrossRef]
- Goudarzi, R.; Reid, A.; McDougall, J.J. Evaluation of the novel avocado/soybean unsaponifiable Arthrocen to alter joint pain and inflammation in a rat model of osteoarthritis. PLoS ONE 2018, 13, e0191906. [Google Scholar] [CrossRef]
- Kang, M.; Jung, I.; Hur, J.; Kim, S.H.; Lee, J.H.; Kang, J.-Y.; Jung, K.C.; Kim, K.S.; Yoo, M.C.; Park, D.-S.; et al. The analgesic and anti-inflammatory effect of WIN-34B, a new herbal formula for osteoarthritis composed of Lonicera japonica Thunb and Anemarrhena asphodeloides BUNGE in vivo. J. Ethnopharmacol. 2010, 131, 485–496. [Google Scholar] [CrossRef]
- Lee, M.; Ryu, D.G.; Kwon, K.B. Anti-inflammatory Effects of Saussurea Lappa Extracts in Murine Macrophages. J. Physiol. Pathol. Korean Med. 2011, 25, 275–279. [Google Scholar]
- Yang, H.J.; Kim, M.J.; Kang, S.; Moon, N.R.; Kim, D.S.; Lee, N.R.; Kim, K.S.; Park, S. Topical treatments of Saussurea costus root and Thuja orientalis L. synergistically alleviate atopic dermatitis-like skin lesions by inhibiting protease-activated receptor-2 and NF-κB signaling in HaCaT cells and Nc/Nga mice. J. Ethnopharmacol. 2017, 199, 97–105. [Google Scholar] [CrossRef]
- Lim, H.-S.; Soo-Jin, J.; Kim, O.-S.; Shin, H.-K.; Jeong, S.-J. Alantolactone from Saussurea lappa Exerts Antiinflammatory Effects by Inhibiting Chemokine Production and STAT1 Phosphorylation in TNF-α and IFN-γ-induced in HaCaT cells. Phytotherapy Res. 2015, 29, 1088–1096. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Yan, L.H.; Feng, W.H.; Ma, G.X.; Peng, Y.; Wang, Z.M.; Xiao, P.G. Research Progress on Active Ingredients and Pharmacologic Properties of Saussurea lappa. Curr. Opin. Complement. Altern. Med. 2014, 1, 1–7. [Google Scholar] [CrossRef]
- Lin, X.; Peng, Z.; Su, C. Potential Anti-Cancer Activities and Mechanisms of Costunolide and Dehydrocostuslactone. Int. J. Mol. Sci. 2015, 16, 10888–10906. [Google Scholar] [CrossRef] [PubMed]
- Shah, N. Herbal folk medicines in Northern India. J. Ethnopharmacol. 1982, 6, 293–301. [Google Scholar] [CrossRef]
- Chik, W.; Zhu, L.; Fan, L.; Yi, T.; Zhu, G.; Gou, X.; Tang, Y.; Xu, J.; Yeung, W.; Zhao, Z.; et al. Saussurea involucrata: A review of the botany, phytochemistry and ethnopharmacology of a rare traditional herbal medicine. J. Ethnopharmacol. 2015, 172, 44–160. [Google Scholar] [CrossRef]
- Matsuda, H.; Toguchida, I.; Ninomiya, K.; Kageura, T.; Morikawa, T.; Yoshikawa, M. Effects of sesquiterpenes and amino acid–sesquiterpene conjugates from the roots of Saussurea lappa on inducible nitric oxide synthase and heat shock protein in lipopolysaccharide-activated macrophages. Bioorganic Med. Chem. 2003, 11, 709–715. [Google Scholar] [CrossRef]
- Matsuda, H.; Kagerura, T.; Toguchida, I.; Ueda, H.; Morikawa, T.; Yoshikawa, M. Inhibitory effects of sesquiterpenes from bay leaf on nitric oxide production in lipopolysaccharide-activated macrophages: Structure requirement and role of heat shock protein induction. Life Sci. 2000, 66, 2151–2157. [Google Scholar] [CrossRef]
- Amin, A.R.; Attur, M.; Patel, R.N.; Thakker, G.D.; Marshall, P.J.; Rediske, J.J.; Stuchin, S.A.; Patel, I.R.; Abramson, S.B. Superinduction of cyclooxygenase-2 activity in human osteoarthritis-affected cartilage. Influence of nitric oxide. J. Clin. Investig. 1997, 99, 1231–1237. [Google Scholar] [CrossRef]
- Kim, K.S.; Choi, H.M.; Yang, H.-I.; Yoo, M.C. WIN-34B May Have Analgesic and Anti-Inflammatory Effects by Reducing the Production of Pro-Inflammatory Mediators in Cells via Inhibition of IκB Signaling Pathways. Biomol. Ther. 2012, 20, 50–56. [Google Scholar] [CrossRef]
- Bognar, E.; Sarszegi, Z.; Szabo, A.; Debreceni, B.; Kalman, N.; Tucsek, Z.; Sumegi, B.; Gallyas, J.F. Antioxidant and Anti-Inflammatory Effects in RAW264.7 Macrophages of Malvidin, a Major Red Wine Polyphenol. PLoS ONE 2013, 8, e65355. [Google Scholar] [CrossRef]
- Güler, N.; Kurkcu, M.; Duygu, G.; Çam, B. Sodium iodoacetate induced osteoarthrosis model in rabbit temporomandibular joint: CT and histological study (Part I). Int. J. Oral Maxillofac. Surg. 2011, 40, 1289–1295. [Google Scholar] [CrossRef] [PubMed]
- Van Der Kraan, P.M.; Vitters, E.L.; Van De Putte, L.B.; Berg, W.B.V.D. Development of osteoarthritic lesions in mice by “metabolic” and "mechanical" alterations in the knee joints. Am. J. Pathol. 1989, 135, 1001–1014. [Google Scholar] [PubMed]
- Alaaeddine, N.; di Battista, J.A.; Pelletier, J.P.; Kiansa, K.; Cloutier, J.M.; Martel-Pelletier, J. Inhibition of tumor necrosis factor α-induced prostaglandin E2 production by the antiinflammatory cytokines interleukin-4, interleukin-10, and interleukin-13 in osteoarthritic synovial fibroblasts: Distinct targeting in the signaling pathways. Arthritis Rheum. 1999, 42, 710–718. [Google Scholar] [CrossRef]
- Valente, J.D.S. The Pharmacology of Pain Associated With the Monoiodoacetate Model of Osteoarthritis. Front. Pharmacol. 2019, 10, 974. [Google Scholar] [CrossRef]
- Fernandes, J.C.; Martel-Pelletier, J.; Pelletier, J.-P. The role of cytokines in osteoarthritis pathophysiology. Biorheology 2002, 39, 237–246. [Google Scholar]
- Hart, P.H.; Vitti, G.F.; Burgess, D.R.; Whitty, G.A.; Piccoli, D.S.; Hamilton, J.A. Potential antiinflammatory effects of interleukin 4: Suppression of human monocyte tumor necrosis factor alpha, interleukin 1, and prostaglandin E2. Proc. Natl. Acad. Sci. USA 1989, 86, 3803–3807. [Google Scholar] [CrossRef]
- Hart, P.H.; Ahern, M.J.; Smith, M.D.; Finlay-Jones, J.J. Comparison of the suppressive effects of interleukin-10 and interleukin-4 on synovial fluid macrophages and blood monocytes from patients with inflammatory arthritis. Immunology 1995, 84, 536–542. [Google Scholar]
- Woo, Y.J.; Bin Joo, Y.; Jung, Y.O.; Ju, J.H.; La Cho, M.; Oh, H.J.; Jhun, J.Y.; Park, M.K.; Park, J.S.; Kang, C.M.; et al. Grape seed proanthocyanidin extract ameliorates monosodium iodoacetate-induced osteoarthritis. Exp. Mol. Med. 2011, 43, 561–570. [Google Scholar] [CrossRef]
- Xu, C.; Oyajobi, B.; Frazer, A.; Kozaci, L.D.; Russell, R.G.G.; Hollander, A.P. Effects of growth factors and interleukin-1 alpha on proteoglycan and type II collagen turnover in bovine nasal and articular chondrocyte pellet cultures. Endocrinology 1996, 137, 3557–3565. [Google Scholar] [CrossRef][Green Version]
- Philp, A.M.; Davis, E.T.; Jones, S.W. Developing anti-inflammatory therapeutics for patients with osteoarthritis. Rheumatology 2016, 56, kew278. [Google Scholar] [CrossRef]
- Guzman, R.E.; Evans, M.G.; Bove, S.; Morenko, B.; Kilgore, K. Mono-Iodoacetate-Induced Histologic Changes in Subchondral Bone and Articular Cartilage of Rat Femorotibial Joints: An Animal Model of Osteoarthritis. Toxicol. Pathol. 2003, 31, 619–624. [Google Scholar] [CrossRef] [PubMed]
- Hayer, S.; Bauer, G.; Willburger, M.; Sinn, K.; Alasti, F.; Plasenzotti, R.; Shvets, T.; Niederreiter, B.; Aschauer, C.; Steiner, G.; et al. Cartilage damage and bone erosion are more prominent determinants of functional impairment in longstanding experimental arthritis than synovial inflammation. Dis. Model. Mech. 2016, 9, 1329–1338. [Google Scholar] [CrossRef] [PubMed]
- An, S.-S.; Heo, D.-S. Effects of Kyejiinsam-tang in MIA-Induced Osteoarthritis Rats. J. Korean Med. 2013, 34, 69–85. [Google Scholar] [CrossRef]
- Mohan, G.; Perilli, E.; Kuliwaba, J.; Humphries, J.M.; Parkinson, I.H.; Fazzalari, N.L. Application of in vivo micro-computed tomography in the temporal characterisation of subchondral bone architecture in a rat model of low-dose monosodium iodoacetate-induced osteoarthritis. Arthritis Res. Ther. 2011, 13, R210. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.E.; Lee, J.Y.; Shim, K.-S.; Lee, S.; Min, K.; Bae, J.-H.; Kim, H.-J.; Park, K.; Song, H.-R. Attenuation of inflammation and cartilage degradation by sulfasalazine-containing hyaluronic acid on osteoarthritis rat model. Int. J. Biol. Macromol. 2018, 114, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Pauly, H.M.; Larson, B.E.; Coatney, G.A.; Button, K.D.; DeCamp, C.E.; Fajardo, R.S.; Haut, R.C.; Donahue, T.L.H. Assessment of cortical and trabecular bone changes in two models of post-traumatic osteoarthritis. J. Orthop. Res. 2015, 33, 1835–1845. [Google Scholar] [CrossRef] [PubMed]
- Syx, D.; Tran, P.B.; Miller, R.E.; Malfait, A.-M. Peripheral Mechanisms Contributing to Osteoarthritis Pain. Curr. Rheumatol. Rep. 2018, 20, 1–11. [Google Scholar] [CrossRef]
- Ren, K.; Dubner, R. Inflammatory Models of Pain and Hyperalgesia. ILAR J. 1999, 40, 111–118. [Google Scholar] [CrossRef]
- Deraedt, R.; Jouquey, S.; Delevallée, F.; Flahaut, M. Release of prostaglandins E and F in an algogenic reaction and its inhibition. Eur. J. Pharmacol. 1980, 61, 17–24. [Google Scholar] [CrossRef]
- Sugita, R.; Kuwabara, H.; Kubota, K.; Sugimoto, K.; Kiho, T.; Tengeiji, A.; Kawakami, K.; Shimada, K. Simultaneous Inhibition of PGE2 and PGI2 Signals Is Necessary to Suppress Hyperalgesia in Rat Inflammatory Pain Models. Mediat. Inflamm. 2016, 2016, 9847840. [Google Scholar] [CrossRef]
- Zhao, F.; Xu, H.; He, E.-Q.; Jiang, Y.-T.; Liu, K. Inhibitory effects of sesquiterpenes fromSaussurea lappaon the overproduction of nitric oxide and TNF-α release in LPS-activated macrophages. J. Asian Nat. Prod. Res. 2008, 10, 1045–1053. [Google Scholar] [CrossRef] [PubMed]
- Parente, L. Pros and cons of selective inhibition of cyclooxygenase-2 versus dual lipoxygenase/cyclooxygenase inhibition: Is two better than one? J. Rheumatol. 2001, 28, 2375–2382. [Google Scholar] [PubMed]
- Daheshia, M.; Yao, J.Q. The Interleukin 1β Pathway in the Pathogenesis of Osteoarthritis. J. Rheumatol. 2008, 35, 2306–2312. [Google Scholar] [CrossRef] [PubMed]


| IL-6 | F | 5′-ACCAGAGGAAATTTTCAATAGG-3′ |
| R | 5′-TGATGCACTTGCAGAAAACA-3′ | |
| COX-2 | F | 5′-AACCGCATTGCCTCTGAAT-3′ |
| R | 5′-CATGTTCCAGGAGGATGGAG-3′ | |
| TNF-α | F | 5′-ATGGGCTTTCCGAATTCAC-3′ |
| R | 5′-GAGGCAACCTGACCACTCTC-3′ | |
| IL-1β | F | 5′-CCTAAAGTATGGGCTGGACTGT-3′ |
| R | 5′-GACTAAGGAGTCCCCTGGAGAT-3′ | |
| iNOS | F | 5′-CCCTTCCGAAGTTTCTGGCAGCAGC-3′ |
| R | 5′-GGCTGTCAGAGCCTCGTGGCTTTGG-3′ | |
| GAPDH | F | 5′-TGGCCTCCAAGGAGTAAGAAAC-3′ |
| R | 5′-CAGCAACTGAGGGCCTCTCT-3′ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jo, H.-G.; Lee, G.-Y.; Baek, C.Y.; Song, H.S.; Lee, D. Analgesic and Anti-Inflammatory Effects of Aucklandia lappa Root Extracts on Acetic Acid-Induced Writhing in Mice and Monosodium Iodoacetate-Induced Osteoarthritis in Rats. Plants 2021, 10, 42. https://doi.org/10.3390/plants10010042
Jo H-G, Lee G-Y, Baek CY, Song HS, Lee D. Analgesic and Anti-Inflammatory Effects of Aucklandia lappa Root Extracts on Acetic Acid-Induced Writhing in Mice and Monosodium Iodoacetate-Induced Osteoarthritis in Rats. Plants. 2021; 10(1):42. https://doi.org/10.3390/plants10010042
Chicago/Turabian StyleJo, Hee-Geun, Geon-Yeong Lee, Chae Yun Baek, Ho Sueb Song, and Donghun Lee. 2021. "Analgesic and Anti-Inflammatory Effects of Aucklandia lappa Root Extracts on Acetic Acid-Induced Writhing in Mice and Monosodium Iodoacetate-Induced Osteoarthritis in Rats" Plants 10, no. 1: 42. https://doi.org/10.3390/plants10010042
APA StyleJo, H.-G., Lee, G.-Y., Baek, C. Y., Song, H. S., & Lee, D. (2021). Analgesic and Anti-Inflammatory Effects of Aucklandia lappa Root Extracts on Acetic Acid-Induced Writhing in Mice and Monosodium Iodoacetate-Induced Osteoarthritis in Rats. Plants, 10(1), 42. https://doi.org/10.3390/plants10010042







