Sas-4 Colocalizes with the Ciliary Rootlets of the Drosophila Sensory Organs
Abstract
:1. Introduction
2. Materials and Methods
2.1. Drosophila Strains
2.2. Antibodies
2.3. Immunofluorescence Staining
2.4. Transmission Electron Microscopy
2.5. Image Acquisition
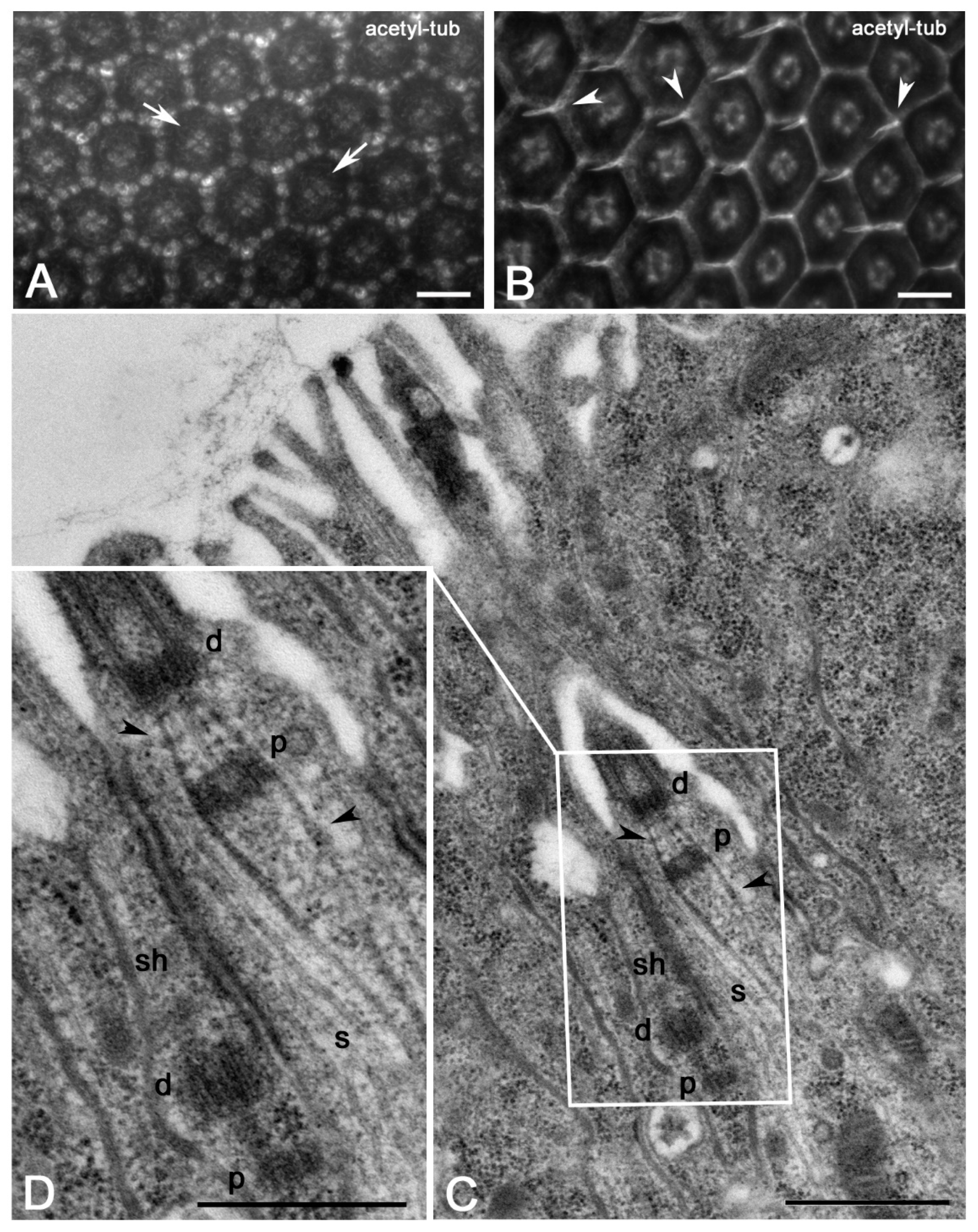
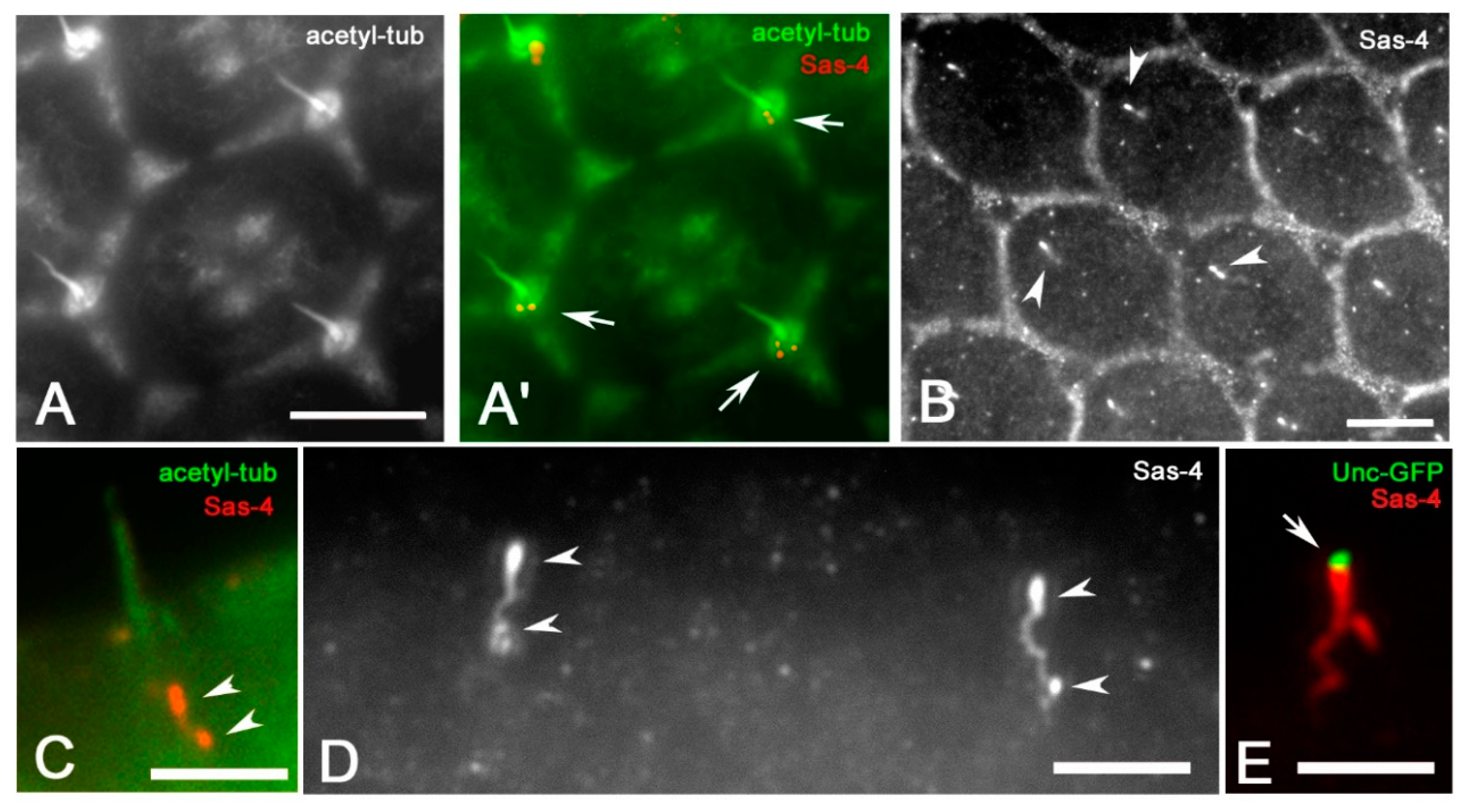
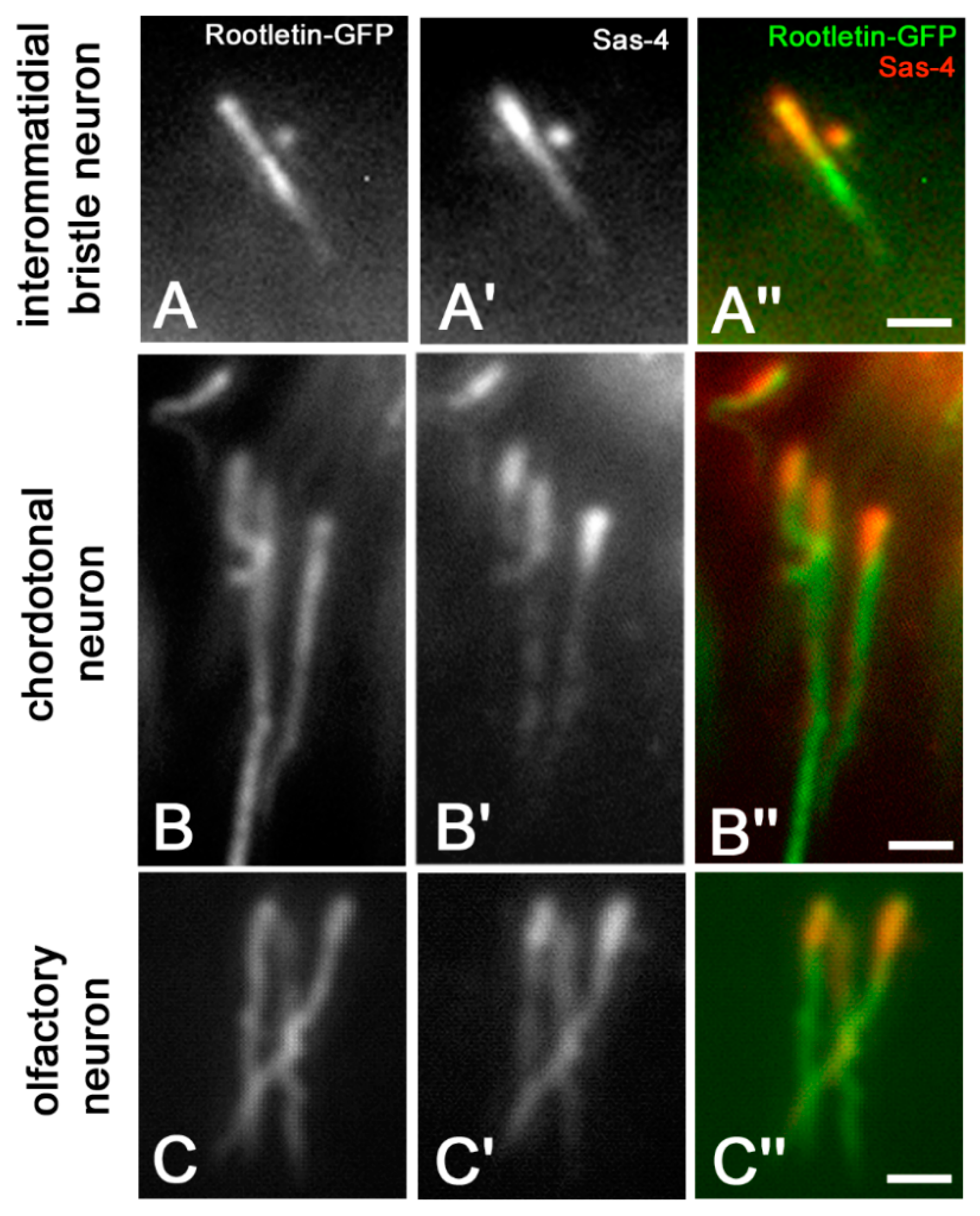
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jana, S.C.; Mendonça, S.; Machado, P.; Werner, S.; Rocha, J.; Pereira, A.; Maiato, H.; Bettencourt-Dias, M. Differential regulation of transition zone and centriole proteins contributes to ciliary base diversity. Nat. Cell Biol. 2018, 20, 928–941. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Keil, T.A. Sensory cilia in arthropods. Arthropod. Struct. Dev. 2012, 41, 515–534. [Google Scholar] [CrossRef] [PubMed]
- Riparbelli, M.G.; Callaini, G.; Megraw, T.L. Assembly and persistence of primary cilia in dividing Drosophila spermatocytes. Dev. Cell 2012, 23, 425–432. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Januschke, J.; Llamazares, S.; Reina, J.; Gonzalez, C. Drosophila neuroblasts retain the daughter centrosome. Nat. Commun. 2011, 2, 243. [Google Scholar] [CrossRef] [PubMed]
- Gottardo, M.; Callaini, G.; Riparbelli, M.G. Does Unc–GFP uncover ciliary structures in the rhabdomeric eye of Drosophila? J. Cell Sci. 2016, 129, 2726–2731. [Google Scholar] [CrossRef] [Green Version]
- Gottardo, M.; Pollarolo, G.; Llamazares, S.; Reina, J.; Riparbelli, M.G.; Callaini, G.; Gonzalez, C. Loss of Centrobin enables daughter centrioles to form sensory cilia in Drosophila. Curr. Biol. 2015, 25, 2319–2324. [Google Scholar] [CrossRef] [Green Version]
- Chen, J.V.; Kao, L.R.; Jana, S.C.; Sivan-Loukianova, E.; Mendonça, S.; Cabrera, O.A.; Singh, P.; Cabernard, C.; Eberl, D.F.; Bettencourt-Dias, M.; et al. Rootletin organizes the ciliary rootlet to achieve neuron sensory function in Drosophila. J. Cell Biol. 2015, 211, 435–453. [Google Scholar] [CrossRef] [Green Version]
- Yang, J.; Liu, X.; Yue, G.; Adamian, M.; Bulgakov, O.; Li, T. Rootletin, a novel coiled-coil protein, is a structural component of the ciliary rootlet. J. Cell Biol. 2002, 159, 431–440. [Google Scholar] [CrossRef] [Green Version]
- Mohan, S.; Timbers, T.A.; Kennedy, J.; Blacque, O.E.; Leroux, M.R. Striated rootlet and nonfilamentous forms of rootletin maintain ciliary function. Curr. Biol. 2013, 23, 2016–2022. [Google Scholar] [CrossRef] [Green Version]
- Joukov, V.; De Nicolo, A. The Centrosome and the Primary Cilium: The Yin and Yang of a Hybrid Organelle. Cells 2019, 8, 701. [Google Scholar] [CrossRef] [Green Version]
- Yang, J.; Gao, J.; Adamian, M.; Wen, X.H.; Pawlyk, B.; Zhang, L.; Sanderson, M.J.; Zuo, J.; Makino, C.L.; Li, T. The ciliary rootlet maintains long-term stability of sensory cilia. Mol. Cell Biol. 2005, 25, 4129–4137. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Styczynska-Soczka, K.; Jarman, A.P. The Drosophila homologue of Rootletin is required for mechanosensory function and ciliary rootlet formation in chordotonal sensory neurons. Cilia 2015. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ko, D.; Kim, J.; Rhee, K.; Choi, H.J. Identification of a Structurally Dynamic Domain for Oligomer Formation in Rootletin. J. Mol. Biol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Vlijm, R.; Li, X.; Panic, M.; Rüthnick, D.; Hata, S.; Herrmannsdörfer, F.; Kuner, T.; Heilemann, M.; Engelhardt, J.; Hell, S.W.; et al. STED nanoscopy of the centrosome linker reveals a CEP68-organized, periodic rootletin network anchored to a C-Nap1 ring at centrioles. Proc. Natl. Acad. Sci. USA 2018, 115, 2246–2253. [Google Scholar] [CrossRef] [Green Version]
- Cagan, R.L.; Ready, D.F. The emergence of order in the Drosophila pupal retina. Dev. Biol. 1989, 136, 346–362. [Google Scholar] [CrossRef]
- Lai, E.C.; Orgogozo, V. A hidden program in Drosophila peripheral neurogenesis revealed: Fundamental principles underlying sensory organ diversity. Dev. Biol. 2004, 269, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Fu, J.; Glover, D. How the newborn centriole becomes a mother. Cell Cycle 2016, 15, 1521–1522. [Google Scholar] [CrossRef] [Green Version]
- Zheng, X.; Gooi, L.M.; Wason, A.; Gabriel, E.; Mehrjardi, N.Z.; Yang, Q.; Zhang, X.; Debec, A.; Basiri, M.L.; Avidor-Reiss, T.; et al. Conserved TCP domain of Sas-4/CPAP is essential for pericentriolar material tethering during centrosome biogenesis. Proc. Natl. Acad. Sci. USA 2014, 111, E354–E363. [Google Scholar] [CrossRef] [Green Version]
- Ramani, A.; Mariappan, A.; Gottardo, M.; Mandad, S.; Urlaub, H.; Avidor-Reiss, T.; Riparbelli, M.; Callaini, G.; Debec, A.; Feederle, R.; et al. Plk1/Polo Phosphorylates Sas-4 at the Onset of Mitosis for an Efficient Recruitment of Pericentriolar Material to Centrosomes. Cell Rep. 2018, 25, 3618–3630. [Google Scholar] [CrossRef] [Green Version]
- Gabriel, E.; Wason, A.; Ramani, A.; Gooi, L.M.; Keller, P.; Pozniakovsky, A.; Poser, I.; Noack, F.; Telugu, N.S.; Calegari, F.; et al. CPAP promotes timely cilium disassembly to maintain neural progenitor pool. EMBO J. 2016, 35, 803–819. [Google Scholar] [CrossRef]
- Sharma, A.; Aher, A.; Dynes, N.J.; Frey, D.; Katrukha, E.A.; Jaussi, R.; Grigoriev, I.; Croisier, M.; Kammerer, R.A.; Akhmanova, A.; et al. Centriolar CPAP/SAS-4 Imparts Slow Processive Microtubule Growth. Dev. Cell. 2016, 37, 362–376. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Avidor-Reiss, T.; Gopalakrishnan, J. Building a centriole. Curr. Opin. Cell Biol. 2013, 25, 72–77. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hatzopoulos, G.N.; Erat, M.C.; Cutts, E.; Rogala, K.B.; Slater, L.M.; Stansfeld, P.J.; Vakonakis, I. Structural analysis of the G-box domain of the microcephaly protein CPAP suggests a role in centriole architecture. Structure 2013, 21, 2069–2077. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gartenmann, L.; Wainman, A.; Qurashi, M.; Kaufmann, R.; Schubert, S.; Raff, J.W.; Dobbie, I.M. A combined 3D-SIM/SMLM approach allows centriole proteins to be localized with a precision of ~4–5 nm. Curr. Biol. 2017, 27, R1054–R1055. [Google Scholar] [CrossRef] [Green Version]
- Varadarajan, R.; Rusan, N.M. Bridging centrioles and PCM in proper space and time. Essays Biochem. 2018, 62, 793–801. [Google Scholar]
- Baker, J.D.; Adhikarakunnathu, S.; Kernan, M.J. Mechanosensory-defective, male-sterile unc mutants identify a novel basal body protein required for ciliogenesis in Drosophila. Development 2004, 131, 3411–3422. [Google Scholar] [CrossRef] [Green Version]
- Zheng, X.; Ramani, A.; Soni, K.; Gottardo, M.; Zheng, S.; Ming Gooi, L.; Li, W.; Feng, S.; Mariappan, A.; Wason, A.; et al. Molecular basis for CPAP-tubulin interaction in controlling centriolar and ciliary length. Nat. Commun. 2016. [Google Scholar] [CrossRef] [Green Version]
- Basto, R.; Lau, J.; Vinogradova, T.; Gardiol, A.; Woods, C.G.; Khodjakov, A.; Raff, J.W. Flies without centrioles. Cell 2006, 125, 1375–1386. [Google Scholar] [CrossRef] [Green Version]
- Lewis, E. A new standard food medium. Dros. Inf. Service 1960, 34, 117–118. [Google Scholar]
- Gopalakrishnan, J.; Mennella, V.; Blachon, S.; Zhai, B.; Smith, A.H.; Megraw, T.L.; Nicastro, D.; Gygi, S.P.; Agard, D.A.; Avidor-Reiss, T. Sas-4 provides a scaffold for cytoplasmic complexes and tethers them in a centrosome. Nat. Commun. 2011, 2, 359. [Google Scholar] [CrossRef] [Green Version]
- Reina, J.; Gottardo, M.; Riparbelli, M.G.; Llamazares, S.; Callaini, G.; Gonzalez, C. Centrobin is essential for C-tubule assembly and flagellum development in Drosophila melanogaster spermatogenesis. J. Cell Biol. 2018, 201, 801032. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues-Martins, A.; Bettencourt-Dias, M.; Riparbelli, M.; Ferreira, C.; Ferreira, I.; Callaini, G.; Glover, D.M. DSAS-6 organizes a tube-like centriole precursor, and its absence suggests modularity in centriole assembly. Curr. Biol. 2007, 17, 1465–1472. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carthew, R.W. Pattern formation in the Drosophila eye. Curr. Opin. Genet. Dev. 2007, 17, 309–313. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cagan, R.L. Principles of Drosophila eye differentiation. Curr. Top. Dev. Biol. 2009, 89, 115–135. [Google Scholar] [PubMed] [Green Version]
- Perry, M.M. Further studies on the development of the of Drosophila melanogaster. II. The interommatidial bristles. J. Morphol. 1968, 124, 249–262. [Google Scholar] [CrossRef] [PubMed]
- Hsu, W.B.; Hung, L.Y.; Tang, C.J.; Su, C.L.; Chang, Y.; Tang, T.K. Functional characterization of the microtubule-binding and -destabilizing domains of CPAP and d-SAS-4. Exp. Cell Res. 2008, 314, 2591–2602. [Google Scholar] [CrossRef]
- Tang, C.J.; Fu, R.H.; Wu, K.S.; Hsu, W.B.; Tang, T.K. CPAP is a cell-cycle regulated protein that controls centriole length. Nat. Cell Biol. 2009, 11, 825–831. [Google Scholar] [CrossRef]
- Riparbelli, M.G.; Persico, V.; Callaini, G. A transient microtubule-based structure uncovers a new intrinsic asymmetry between the mother centrioles in the early Drosophila spermatocytes. Cytoskeleton 2018, 75, 472–480. [Google Scholar] [CrossRef]
- Hartenstein, V.; Posakony, J.W. Development of adult sensilla on the wing and notum of Drosophila melanogaster. Development 1989, 107, 389–405. [Google Scholar]
- Schweisguth, F. Asymmetric cell division in the Drosophila bristle lineage: From the polarization of sensory organ precursor cells to Notch-mediated binary fate decision. Wiley Interdiscip. Rev. Dev. Biol. 2015, 4, 299–309. [Google Scholar] [CrossRef]
- Cagan, R.L.; Ready, D.F. Notch is required for successive cell decisions in the developing Drosophila retina. Genes Dev. 1989, 3, 1099–1112. [Google Scholar] [CrossRef] [PubMed] [Green Version]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Persico, V.; Callaini, G.; Riparbelli, M.G. Sas-4 Colocalizes with the Ciliary Rootlets of the Drosophila Sensory Organs. J. Dev. Biol. 2021, 9, 1. https://doi.org/10.3390/jdb9010001
Persico V, Callaini G, Riparbelli MG. Sas-4 Colocalizes with the Ciliary Rootlets of the Drosophila Sensory Organs. Journal of Developmental Biology. 2021; 9(1):1. https://doi.org/10.3390/jdb9010001
Chicago/Turabian StylePersico, Veronica, Giuliano Callaini, and Maria Giovanna Riparbelli. 2021. "Sas-4 Colocalizes with the Ciliary Rootlets of the Drosophila Sensory Organs" Journal of Developmental Biology 9, no. 1: 1. https://doi.org/10.3390/jdb9010001
APA StylePersico, V., Callaini, G., & Riparbelli, M. G. (2021). Sas-4 Colocalizes with the Ciliary Rootlets of the Drosophila Sensory Organs. Journal of Developmental Biology, 9(1), 1. https://doi.org/10.3390/jdb9010001



