Notochord Cells in Intervertebral Disc Development and Degeneration
Abstract
:1. Introduction
“Know from whence you came. If you know whence you came, there are absolutely no limitations to where you can go.”—James Baldwin
2. Functions and Structure of the Intervertebral Disc
3. Intervertebral Disc Development
3.1. Early Embryo and Node Development
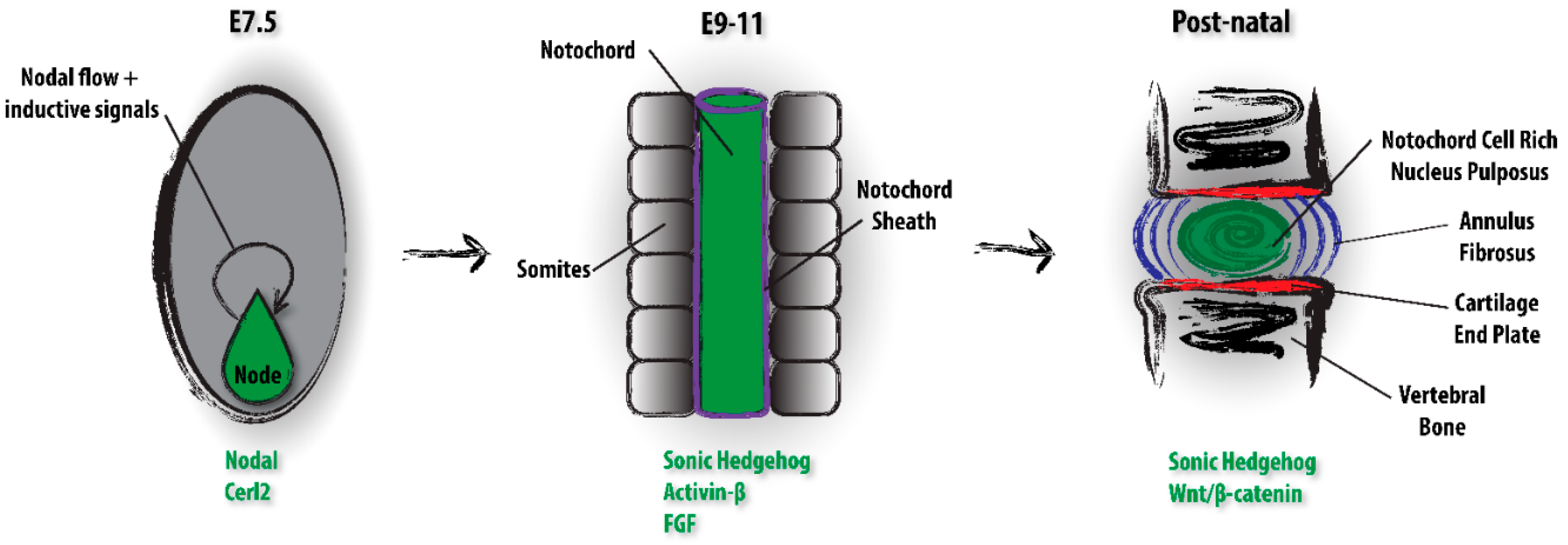
3.2. Notochord Development
3.3. Notochord Signalling
3.4. Role of the Mesenchyme in Intervertebral Disc Development
3.5. Notochord Cell Contribution to the Nucleus Pulposus
4. Intervertebral Disc Degeneration
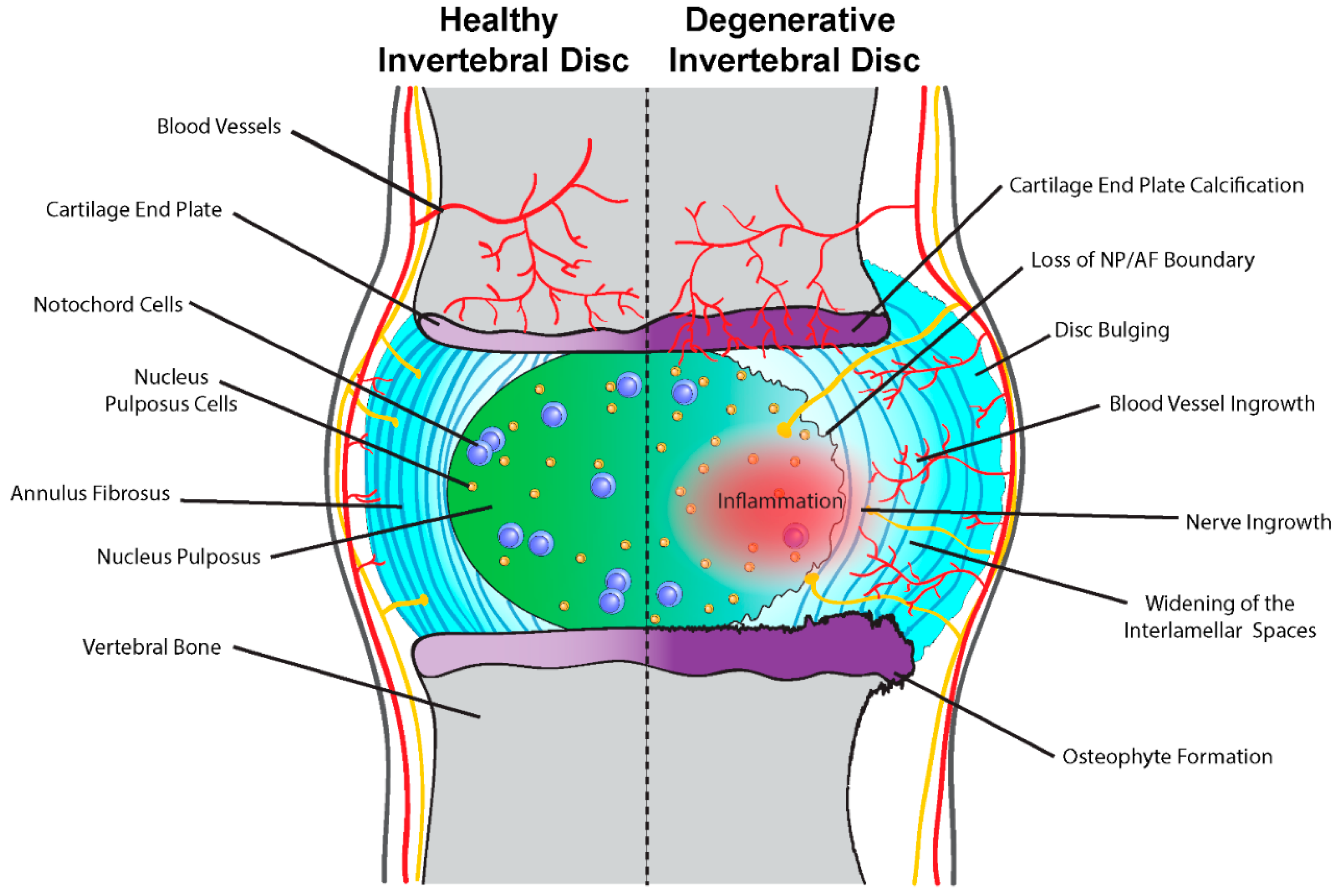
Notochord Cells and Intervertebral Disc Homeostasis
5. Small Animal Models to Study Intervertebral Disc Development and Degeneration
5.1. Notochord Cells in Animal Models
5.2. Notochord Associated Cre Mouse Strains
6. IVD Disease and Repair: A Mirror of Development?
7. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Balague, F.; Mannion, A.F.; Pellise, F.; Cedraschi, C. Non-specific low back pain. Lancet 2012, 379, 482–491. [Google Scholar] [CrossRef]
- Vos, T.; Flaxman, A.D.; Naghavi, M.; Lozano, R.; Michaud, C.; Ezzati, M.; Shibuya, K.; Salomon, J.A.; Abdalla, S.; Aboyans, V.; et al. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990–2010: A systematic analysis for the global burden of disease study 2010. Lancet 2012, 380, 2163–2196. [Google Scholar] [CrossRef]
- Manchikanti, L.; Singh, V.; Falco, F.J.; Benyamin, R.M.; Hirsch, J.A. Epidemiology of low back pain in adults. Neuromodulation 2014, 17 (Suppl. 2), 3–10. [Google Scholar] [CrossRef] [PubMed]
- Friedman, B.W.; O’Mahony, S.; Mulvey, L.; Davitt, M.; Choi, H.; Xia, S.; Esses, D.; Bijur, P.E.; Gallagher, E.J. One-week and 3-month outcomes after an emergency department visit for undifferentiated musculoskeletal low back pain. Ann. Emerg. Med. 2012, 59, 128–133. [Google Scholar] [CrossRef] [PubMed]
- Shiri, R.; Karppinen, J.; Leino-Arjas, P.; Solovieva, S.; Viikari-Juntura, E. The association between obesity and low back pain: A meta-analysis. Am. J. Epidemiol. 2010, 171, 135–154. [Google Scholar] [CrossRef] [PubMed]
- Andersson, G.B. Epidemiological features of chronic low-back pain. Lancet 1999, 354, 581–585. [Google Scholar] [CrossRef]
- Hoy, D.; Bain, C.; Williams, G.; March, L.; Brooks, P.; Blyth, F.; Woolf, A.; Vos, T.; Buchbinder, R. A systematic review of the global prevalence of low back pain. Arthritis Rheum. 2012, 64, 2028–2037. [Google Scholar] [CrossRef] [PubMed]
- Mayer, J.E.; Iatridis, J.C.; Chan, D.; Qureshi, S.A.; Gottesman, O.; Hecht, A.C. Genetic polymorphisms associated with intervertebral disc degeneration. Spine J. 2013, 13, 299–317. [Google Scholar] [CrossRef] [PubMed]
- Inoue, N.; Espinoza Orias, A.A. Biomechanics of intervertebral disk degeneration. Orthop. Clin. N. Am. 2011, 42, 487–499. [Google Scholar] [CrossRef] [PubMed]
- Moreland, L.W. Intra-articular hyaluronan (hyaluronic acid) and hylans for the treatment of osteoarthritis: Mechanisms of action. Arthritis Res. Ther. 2003, 5, 54–67. [Google Scholar] [CrossRef] [PubMed]
- Chandran, P.L.; Horkay, F. Aggrecan, an unusual polyelectrolyte: Review of solution behavior and physiological implications. Acta Biomater. 2012, 8, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Nap, R.J.; Szleifer, I. Structure and interactions of aggrecans: Statistical thermodynamic approach. Biophys. J. 2008, 95, 4570–4583. [Google Scholar] [CrossRef] [PubMed]
- Kraemer, J.; Kolditz, D.; Gowin, R. Water and electrolyte content of human intervertebral discs under variable load. Spine 1985, 10, 69–71. [Google Scholar] [CrossRef] [PubMed]
- Mwale, F. Collagen and other proteins of the nucleus pulposus, annulus fibrosus, and cartilage end plates. In The Intervertebral Disc; Springer: Berlin, Germany, 2014; pp. 79–92. [Google Scholar]
- Matsumura, Y.; Kasai, Y.; Obata, H.; Matsushima, S.; Inaba, T.; Uchida, A. Changes in water content of intervertebral discs and paravertebral muscles before and after bed rest. J. Orthop. Sci. 2009, 14, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Urban, J.P.G.; Roberts, S.; Ralphs, J.R. The nucleus of the intervertebral disc from development to degeneration. Am. Zool. 2000, 40, 53–61. [Google Scholar] [CrossRef]
- Setton, L.A.; Chen, J. Mechanobiology of the intervertebral disc and relevance to disc degeneration. J. Bone Joint Surg. Am. 2006, 88 (Suppl. 2), 52–57. [Google Scholar] [CrossRef] [PubMed]
- Adams, M.A.; Roughley, P.J. What is intervertebral disc degeneration, and what causes it? Spine 2006, 31, 2151–2161. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, H.; Yamada, Y.; Kimata, K. Roles of aggrecan, a large chondroitin sulfate proteoglycan, in cartilage structure and function. J. Biochem. 1998, 124, 687–693. [Google Scholar] [CrossRef] [PubMed]
- Cassidy, J.J.; Hiltner, A.; Baer, E. Hierarchical structure of the intervertebral disc. Connect. Tissue Res. 1989, 23, 75–88. [Google Scholar] [CrossRef] [PubMed]
- Smith, L.J.; Fazzalari, N.L. The elastic fibre network of the human lumbar anulus fibrosus: Architecture, mechanical function and potential role in the progression of intervertebral disc degeneration. Eur. Spine J. 2009, 18, 439–448. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Liu, C.; Guo, Q.; Yang, H.; Li, B. Regional variations in the cellular, biochemical, and biomechanical characteristics of rabbit annulus fibrosus. PLoS ONE 2014, 9, e91799. [Google Scholar] [CrossRef] [PubMed]
- Urban, J.P.; Smith, S.; Fairbank, J.C. Nutrition of the intervertebral disc. Spine 2004, 29, 2700–2709. [Google Scholar] [CrossRef] [PubMed]
- Davidson, B.P.; Tam, P.P. The node of the mouse embryo. Curr. Biol. 2000, 10, R617–R619. [Google Scholar] [CrossRef]
- Hirokawa, N.; Tanaka, Y.; Okada, Y.; Takeda, S. Nodal flow and the generation of left-right asymmetry. Cell 2006, 125, 33–45. [Google Scholar] [CrossRef] [PubMed]
- Brennan, J.; Norris, D.P.; Robertson, E.J. Nodal activity in the node governs left-right asymmetry. Genes Dev. 2002, 16, 2339–2344. [Google Scholar] [CrossRef] [PubMed]
- Marques, S.; Borges, A.C.; Silva, A.C.; Freitas, S.; Cordenonsi, M.; Belo, J.A. The activity of the Nodal antagonist Cerl-2 in the mouse node is required for correct L/R body axis. Genes Dev. 2004, 18, 2342–2347. [Google Scholar] [CrossRef] [PubMed]
- Babu, D.; Roy, S. Left-right asymmetry: Cilia stir up new surprises in the node. Open Biol. 2013, 3, 130052. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, Y.; Okada, Y.; Hirokawa, N. FGF-induced vesicular release of sonic hedgehog and retinoic acid in leftward nodal flow is critical for left-right determination. Nature 2005, 435, 172–177. [Google Scholar] [CrossRef] [PubMed]
- Nonaka, S.; Tanaka, Y.; Okada, Y.; Takeda, S.; Harada, A.; Kanai, Y.; Kido, M.; Hirokawa, N. Randomization of left-right asymmetry due to loss of nodal cilia generating leftward flow of extraembryonic fluid in mice lacking KIF3B motor protein. Cell 1998, 95, 829–837. [Google Scholar] [CrossRef]
- Adams, D.S.; Keller, R.; Koehl, M.A. The mechanics of notochord elongation, straightening and stiffening in the embryo of xenopus laevis. Development 1990, 110, 115–130. [Google Scholar] [PubMed]
- Stemple, D.L. Structure and function of the notochord: An essential organ for chordate development. Development 2005, 132, 2503–2512. [Google Scholar] [CrossRef] [PubMed]
- Choi, K.S.; Harfe, B.D. Hedgehog signaling is required for formation of the notochord sheath and patterning of nuclei pulposi within the intervertebral discs. Proc. Natl. Acad. Sci. USA 2011, 108, 9484–9489. [Google Scholar] [CrossRef] [PubMed]
- Gray, R.S.; Wilm, T.P.; Smith, J.; Bagnat, M.; Dale, R.M.; Topczewski, J.; Johnson, S.L.; Solnica-Krezel, L. Loss of col8a1a function during zebrafish embryogenesis results in congenital vertebral malformations. Dev. Biol. 2014, 386, 72–85. [Google Scholar] [CrossRef] [PubMed]
- Gotz, W.; Osmers, R.; Herken, R. Localisation of extracellular matrix components in the embryonic human notochord and axial mesenchyme. J. Anat. 1995, 186 Pt 1, 111–121. [Google Scholar] [PubMed]
- Corallo, D.; Schiavinato, A.; Trapani, V.; Moro, E.; Argenton, F.; Bonaldo, P. Emilin3 is required for notochord sheath integrity and interacts with Scube2 to regulate notochord-derived hedgehog signals. Development 2013, 140, 4594–4601. [Google Scholar] [CrossRef] [PubMed]
- Pagnon-Minot, A.; Malbouyres, M.; Haftek-Terreau, Z.; Kim, H.R.; Sasaki, T.; Thisse, C.; Thisse, B.; Ingham, P.W.; Ruggiero, F.; le Guellec, D. Collagen XV, a novel factor in zebrafish notochord differentiation and muscle development. Dev. Biol. 2008, 316, 21–35. [Google Scholar] [CrossRef] [PubMed]
- Mangos, S.; Lam, P.Y.; Zhao, A.; Liu, Y.; Mudumana, S.; Vasilyev, A.; Liu, A.; Drummond, I.A. The ADPKD genes pkd1a/b and pkd2 regulate extracellular matrix formation. Dis. Model. Mech. 2010, 3, 354–365. [Google Scholar] [CrossRef] [PubMed]
- Hunter, C.J.; Bianchi, S.; Cheng, P.; Muldrew, K. Osmoregulatory function of large vacuoles found in notochordal cells of the intervertebral disc running title: An osmoregulatory vacuole. Mol. Cell. Biomech. 2007, 4, 227–237. [Google Scholar] [PubMed]
- Waddington, C.; Perry, M.M. The ultrastructure of the developing urodele notochord. Proc. R. Soc. Lond. B Biol. Sci. 1962, 156, 459–482. [Google Scholar] [CrossRef]
- Bancroft, M.; Bellairs, R. The development of the notochord in the chick embryo, studied by scanning and transmission electron microscopy. J. Embryol. Exp. Morphol. 1976, 35, 383–401. [Google Scholar] [PubMed]
- Leeson, T.S.; Leeson, C.R. Observations on the histochemistry and fine structure of the notochord in rabbit embryos. J. Anat. 1958, 92, 278–285. [Google Scholar] [PubMed]
- Babic, M.S. Development of the notochord in normal and malformed human embryos and fetuses. Int. J. Dev. Biol. 1991, 35, 345–352. [Google Scholar] [PubMed]
- Ellis, K.; Bagwell, J.; Bagnat, M. Notochord vacuoles are lysosome-related organelles that function in axis and spine morphogenesis. J. Cell Biol. 2013, 200, 667–679. [Google Scholar] [CrossRef] [PubMed]
- Luzio, J.P.; Pryor, P.R.; Bright, N.A. Lysosomes: Fusion and function. Nat. Rev. Mol. Cell Biol. 2007, 8, 622–632. [Google Scholar] [CrossRef] [PubMed]
- Allen, R.D.; Naitoh, Y. Osmoregulation and contractile vacuoles of protozoa. Int. Rev. Cytol. 2002, 215, 351–394. [Google Scholar] [PubMed]
- McCann, M.R.; Patel, P.; Frimpong, A.; Xiao, Y.; Siqueira, W.L.; Seguin, C.A. Proteomic signature of the murine intervertebral disc. PLoS ONE 2015, 10, e0117807. [Google Scholar]
- Mavrogonatou, E.; Papadimitriou, K.; Urban, J.P.; Papadopoulos, V.; Kletsas, D. Deficiency in the α1 subunit of Na+/K+-ATPase enhances the anti-proliferative effect of high osmolality in nucleus pulposus intervertebral disc cells. J. Cell. Physiol. 2015, 230, 3037–3048. [Google Scholar] [CrossRef] [PubMed]
- Yamada, T.; Placzek, M.; Tanaka, H.; Dodd, J.; Jessell, T.M. Control of cell pattern in the developing nervous system: Polarizing activity of the floor plate and notochord. Cell 1991, 64, 635–647. [Google Scholar] [CrossRef]
- Fan, C.M.; Tessier-Lavigne, M. Patterning of mammalian somites by surface ectoderm and notochord: Evidence for sclerotome induction by a hedgehog homolog. Cell 1994, 79, 1175–1186. [Google Scholar] [CrossRef]
- Kim, S.K.; Hebrok, M.; Melton, D.A. Notochord to endoderm signaling is required for pancreas development. Development 1997, 124, 4243–4252. [Google Scholar] [PubMed]
- Meadows, S.M.; Fletcher, P.J.; Moran, C.; Xu, K.; Neufeld, G.; Chauvet, S.; Mann, F.; Krieg, P.A.; Cleaver, O. Integration of repulsive guidance cues generates avascular zones that shape mammalian blood vessels. Circ. Res. 2012, 110, 34–46. [Google Scholar] [CrossRef] [PubMed]
- Clarke, J.D.; Holder, N.; Soffe, S.R.; Storm-Mathisen, J. Neuroanatomical and functional analysis of neural tube formation in notochordless xenopus embryos; laterality of the ventral spinal cord is lost. Development 1991, 112, 499–516. [Google Scholar] [PubMed]
- Basler, K.; Edlund, T.; Jessell, T.M.; Yamada, T. Control of cell pattern in the neural tube: Regulation of cell differentiation by dorsalin-1, a novel TGF beta family member. Cell 1993, 73, 687–702. [Google Scholar] [CrossRef]
- Patten, I.; Placzek, M. Opponent activities of Shh and BMP signaling during floor plate induction in vivo. Curr. Biol. 2002, 12, 47–52. [Google Scholar] [CrossRef]
- Goulding, M.; Lumsden, A.; Paquette, A.J. Regulation of Pax-3 expression in the dermomyotome and its role in muscle development. Development 1994, 120, 957–971. [Google Scholar] [PubMed]
- Amacher, S.L.; Kimmel, C.B. Promoting notochord fate and repressing muscle development in zebrafish axial mesoderm. Development 1998, 125, 1397–1406. [Google Scholar] [PubMed]
- Brand-Saberi, B.; Ebensperger, C.; Wilting, J.; Balling, R.; Christ, B. The ventralizing effect of the notochord on somite differentiation in chick embryos. Anat. Embryol. 1993, 188, 239–245. [Google Scholar] [CrossRef] [PubMed]
- Cleaver, O.; Krieg, P.A. Notochord patterning of the endoderm. Dev. Biol. 2001, 234, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Hebrok, M.; Kim, S.K.; Melton, D.A. Notochord repression of endodermal sonic hedgehog permits pancreas development. Genes Dev. 1998, 12, 1705–1713. [Google Scholar] [CrossRef] [PubMed]
- Choi, K.S.; Cohn, M.J.; Harfe, B.D. Identification of nucleus pulposus precursor cells and notochordal remnants in the mouse: Implications for disk degeneration and chordoma formation. Dev. Dyn. 2008, 237, 3953–3958. [Google Scholar] [CrossRef] [PubMed]
- McCann, M.R.; Tamplin, O.J.; Rossant, J.; Seguin, C.A. Tracing notochord-derived cells using a Noto-cre mouse: Implications for intervertebral disc development. Dis. Model. Mech. 2012, 5, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Sivakamasundari, V.; Lufkin, T. Bridging the gap: Understanding embryonic intervertebral disc development. Cell Dev. Biol. 2012, 1, 103. [Google Scholar] [PubMed]
- Lawson, L.; Harfe, B.D. Notochord to nucleus pulposus transition. Curr. Osteoporos. Rep. 2015, 13, 336–341. [Google Scholar] [CrossRef] [PubMed]
- Aszodi, A.; Chan, D.; Hunziker, E.; Bateman, J.F.; Fassler, R. Collagen II is essential for the removal of the notochord and the formation of intervertebral discs. J. Cell Biol. 1998, 143, 1399–1412. [Google Scholar] [CrossRef] [PubMed]
- Forsberg, H.; Crozet, F.; Brown, N.A. Waves of mouse lunatic fringe expression, in four-hour cycles at two-hour intervals, precede somite boundary formation. Curr. Biol. 1998, 8, 1027–1030. [Google Scholar] [CrossRef]
- Brent, A.E.; Tabin, C.J. Developmental regulation of somite derivatives: Muscle, cartilage and tendon. Curr. Opin. Genet. Dev. 2002, 12, 548–557. [Google Scholar] [CrossRef]
- Christ, B.; Wilting, J. From somites to vertebral column. Ann. Anat. 1992, 174, 23–32. [Google Scholar] [CrossRef]
- Zhang, N.; Gridley, T. Defects in somite formation in lunatic fringe-deficient mice. Nature 1998, 394, 374–377. [Google Scholar] [PubMed]
- Barrantes, I.B.; Elia, A.J.; Wunsch, K.; Hrabe de Angelis, M.H.; Mak, T.W.; Rossant, J.; Conlon, R.A.; Gossler, A.; de la Pompa, J.L. Interaction between notch signalling and lunatic fringe during somite boundary formation in the mouse. Curr. Biol. 1999, 9, 470–480. [Google Scholar] [CrossRef]
- Chiang, C.; Litingtung, Y.; Lee, E.; Young, K.E.; Corden, J.L.; Westphal, H.; Beachy, P.A. Cyclopia and defective axial patterning in mice lacking sonic hedgehog gene function. Nature 1996, 383, 407–413. [Google Scholar] [CrossRef] [PubMed]
- Choi, K.S.; Lee, C.; Harfe, B.D. Sonic hedgehog in the notochord is sufficient for patterning of the intervertebral discs. Mech. Dev. 2012, 129, 255–262. [Google Scholar] [CrossRef] [PubMed]
- Mundy, C.; Yasuda, T.; Kinumatsu, T.; Yamaguchi, Y.; Iwamoto, M.; Enomoto-Iwamoto, M.; Koyama, E.; Pacifici, M. Synovial joint formation requires local ext1 expression and heparan sulfate production in developing mouse embryo limbs and spine. Dev. Biol. 2011, 351, 70–81. [Google Scholar] [CrossRef] [PubMed]
- Bruggeman, B.J.; Maier, J.A.; Mohiuddin, Y.S.; Powers, R.; Lo, Y.; Guimaraes-Camboa, N.; Evans, S.M.; Harfe, B.D. Avian intervertebral disc arises from rostral sclerotome and lacks a nucleus pulposus: Implications for evolution of the vertebrate disc. Dev. Dyn. 2012, 241, 675–683. [Google Scholar] [CrossRef] [PubMed]
- Hunter, C.J.; Matyas, J.R.; Duncan, N.A. The notochordal cell in the nucleus pulposus: A review in the context of tissue engineering. Tissue Eng. 2003, 9, 667–677. [Google Scholar] [CrossRef] [PubMed]
- Roberts, S. Disc morphology in health and disease. Biochem. Soc. Trans. 2002, 30, 864–869. [Google Scholar] [CrossRef] [PubMed]
- Boos, N.; Weissbach, S.; Rohrbach, H.; Weiler, C.; Spratt, K.F.; Nerlich, A.G. Classification of age-related changes in lumbar intervertebral discs: 2002 Volvo Award in basic science. Spine 2002, 27, 2631–2644. [Google Scholar] [CrossRef] [PubMed]
- Aguiar, D.J.; Johnson, S.L.; Oegema, T.R. Notochordal cells interact with nucleus pulposus cells: Regulation of proteoglycan synthesis. Exp. Cell Res. 1999, 246, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Vujovic, S.; Henderson, S.; Presneau, N.; Odell, E.; Jacques, T.S.; Tirabosco, R.; Boshoff, C.; Flanagan, A.M. Brachyury, a crucial regulator of notochordal development, is a novel biomarker for chordomas. J. Pathol. 2006, 209, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Henriksson, H.; Thornemo, M.; Karlsson, C.; Hagg, O.; Junevik, K.; Lindahl, A.; Brisby, H. Identification of cell proliferation zones, progenitor cells and a potential stem cell niche in the intervertebral disc region: A study in four species. Spine 2009, 34, 2278–2287. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.W.; Lim, T.H.; Kim, J.G.; Jeong, S.T.; Masuda, K.; An, H.S. The origin of chondrocytes in the nucleus pulposus and histologic findings associated with the transition of a notochordal nucleus pulposus to a fibrocartilaginous nucleus pulposus in intact rabbit intervertebral discs. Spine 2003, 28, 982–990. [Google Scholar] [CrossRef] [PubMed]
- Trout, J.J.; Buckwalter, J.A.; Moore, K.C. Ultrastructure of the human intervertebral disc: II. Cells of the nucleus pulposus. Anat. Rec. 1982, 204, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Pazzaglia, U.E.; Salisbury, J.R.; Byers, P.D. Development and involution of the notochord in the human spine. J. R. Soc. Med. 1989, 82, 413–415. [Google Scholar] [PubMed]
- Liebscher, T.; Haefeli, M.; Wuertz, K.; Nerlich, A.G.; Boos, N. Age-related variation in cell density of human lumbar intervertebral disc. Spine 2010, 36, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Sakai, D.; Nishimura, K.; Tanaka, M.; Nakajima, D.; Grad, S.; Alini, M.; Kawada, H.; Ando, K.; Mochida, J. Migration of bone marrow-derived cells for endogenous repair in a new tail-looping disc degeneration model in the mouse: A pilot study. Spine J. 2015, 15, 1356–1365. [Google Scholar] [CrossRef] [PubMed]
- Colombier, P.; Camus, A.; Lescaudron, L.; Clouet, J.; Guicheux, J. Intervertebral disc regeneration: A great challenge for tissue engineers. Trends Biotechnol. 2014, 32, 433–435. [Google Scholar] [CrossRef] [PubMed]
- Mwale, F.; Roughley, P.; Antoniou, J. Distinction between the extracellular matrix of the nucleus pulposus and hyaline cartilage: A requisite for tissue engineering of intervertebral disc. Eur. Cell. Mater. 2004, 8, 58–63. [Google Scholar] [PubMed]
- Smith, L.J.; Nerurkar, N.L.; Choi, K.S.; Harfe, B.D.; Elliott, D.M. Degeneration and regeneration of the intervertebral disc: Lessons from development. Dis. Model. Mech. 2011, 4, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Freemont, A.J. The cellular pathobiology of the degenerate intervertebral disc and discogenic back pain. Rheumatology 2009, 48, 5–10. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Lv, F.J. Fibrosis in intervertebral disc degeneration: Knowledge and gaps. Austin J. Orthop. Rheumatol. 2014, 1, 3. [Google Scholar]
- Risbud, M.V.; Shapiro, I.M. Role of cytokines in intervertebral disc degeneration: Pain and disc content. Nat. Rev. Rheumatol. 2014, 10, 44–56. [Google Scholar] [CrossRef] [PubMed]
- Freemont, A.J.; Peacock, T.E.; Goupille, P.; Hoyland, J.A.; O’Brien, J.; Jayson, M.I. Nerve ingrowth into diseased intervertebral disc in chronic back pain. Lancet 1997, 350, 178–181. [Google Scholar] [CrossRef]
- Pearce, R.H.; Grimmer, B.J.; Adams, M.E. Degeneration and the chemical composition of the human lumbar intervertebral disc. J. Orthop. Res. 1987, 5, 198–205. [Google Scholar] [CrossRef] [PubMed]
- Sobajima, S.; Kim, J.S.; Gilbertson, L.G.; Kang, J.D. Gene therapy for degenerative disc disease. Gene Ther. 2004, 11, 390–401. [Google Scholar] [CrossRef] [PubMed]
- Roughley, P.J. Biology of intervertebral disc aging and degeneration: Involvement of the extracellular matrix. Spine 2004, 29, 2691–2699. [Google Scholar] [CrossRef] [PubMed]
- Boxberger, J.I.; Sen, S.; Yerramalli, C.S.; Elliott, D.M. Nucleus pulposus glycosaminoglycan content is correlated with axial mechanics in rat lumbar motion segments. J. Orthop. Res. 2006, 24, 1906–1915. [Google Scholar] [CrossRef] [PubMed]
- Buckwalter, J.A. Aging and degeneration of the human intervertebral disc. Spine 1995, 20, 1307–1314. [Google Scholar] [PubMed]
- Barbir, A.; Godburn, K.E.; Michalek, A.J.; Lai, A.; Monsey, R.D.; Iatridis, J.C. Effects of torsion on intervertebral disc gene expression and biomechanics, using a rat tail model. Spine 2011, 36, 607–614. [Google Scholar] [CrossRef] [PubMed]
- O’Connell, G.D.; Guerin, H.L.; Elliott, D.M. Theoretical and uniaxial experimental evaluation of human annulus fibrosus degeneration. J. Biomech. Eng. 2009, 131, 111007. [Google Scholar] [CrossRef] [PubMed]
- Natarajan, R.N.; Williams, J.R.; Andersson, G.B. Modeling changes in intervertebral disc mechanics with degeneration. J. Bone Joint Surg. Am. 2006, 88 (Suppl. 2), 36–40. [Google Scholar] [CrossRef] [PubMed]
- Haefeli, M.; Kalberer, F.; Saegesser, D.; Nerlich, A.G.; Boos, N.; Paesold, G. The course of macroscopic degeneration in the human lumbar intervertebral disc. Spine 2006, 31, 1522–1531. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, E.; Nakamae, T.; Olmarker, K. Pain behavior changes following disc puncture relate to nucleus pulposus rather than to the disc injury per se: An experimental study in rats. Open Orthop. J. 2011, 5, 72–77. [Google Scholar] [CrossRef] [PubMed]
- Risbud, M.V.; Schaer, T.P.; Shapiro, I.M. Toward an understanding of the role of notochordal cells in the adult intervertebral disc: From discord to accord. Dev. Dyn. 2011, 239, 2141–2148. [Google Scholar] [CrossRef] [PubMed]
- Korecki, C.L.; MacLean, J.J.; Iatridis, J.C. Dynamic compression effects on intervertebral disc mechanics and biology. Spine 2008, 33, 1403–1409. [Google Scholar] [CrossRef] [PubMed]
- Gantenbein, B.; Calandriello, E.; Wuertz-Kozak, K.; Benneker, L.M.; Keel, M.J.; Chan, S.C. Activation of intervertebral disc cells by co-culture with notochordal cells, conditioned medium and hypoxia. BMC Musculoskelet. Disord. 2014, 15, 422. [Google Scholar] [CrossRef] [PubMed]
- Boyd, L.M.; Chen, J.; Kraus, V.B.; Setton, L.A. Conditioned medium differentially regulates matrix protein gene expression in cells of the intervertebral disc. Spine 2004, 29, 2217–2222. [Google Scholar] [CrossRef] [PubMed]
- Erwin, W.M.; Ashman, K.; O’Donnel, P.; Inman, R.D. Nucleus pulposus notochord cells secrete connective tissue growth factor and up-regulate proteoglycan expression by intervertebral disc chondrocytes. Arthritis Rheum. 2006, 54, 3859–3867. [Google Scholar] [CrossRef] [PubMed]
- Bedore, J.; Sha, W.; McCann, M.R.; Liu, S.; Leask, A.; Seguin, C.A. Impaired intervertebral disc development and premature disc degeneration in mice with notochord-specific deletion of CCN2. Arthritis Rheum. 2013, 65, 2634–2644. [Google Scholar] [PubMed]
- Bedore, J.; Leask, A.; Seguin, C.A. Targeting the extracellular matrix: Matricellular proteins regulate cell-extracellular matrix communication within distinct niches of the intervertebral disc. Matrix Biol. 2014, 37, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Erwin, W.M.; Islam, D.; Inman, R.D.; Fehlings, M.G.; Tsui, F.W. Notochordal cells protect nucleus pulposus cells from degradation and apoptosis: Implications for the mechanisms of intervertebral disc degeneration. Arthritis Res. Ther. 2011, 13, R215. [Google Scholar] [CrossRef] [PubMed]
- Cornejo, M.C.; Cho, S.K.; Giannarelli, C.; Iatridis, J.C.; Purmessur, D. Soluble factors from the notochordal-rich intervertebral disc inhibit endothelial cell invasion and vessel formation in the presence and absence of pro-inflammatory cytokines. Osteoarthritis Cartilage 2015, 23, 487–496. [Google Scholar] [CrossRef] [PubMed]
- Davies, J.A.; Cook, G.M.; Stern, C.D.; Keynes, R.J. Isolation from chick somites of a glycoprotein fraction that causes collapse of dorsal root ganglion growth cones. Neuron 1990, 4, 11–20. [Google Scholar] [CrossRef]
- Anderson, C.N.; Ohta, K.; Quick, M.M.; Fleming, A.; Keynes, R.; Tannahill, D. Molecular analysis of axon repulsion by the notochord. Development 2003, 130, 1123–1133. [Google Scholar] [CrossRef] [PubMed]
- Tolofari, S.K.; Richardson, S.M.; Freemont, A.J.; Hoyland, J.A. Expression of semaphorin 3a and its receptors in the human intervertebral disc: Potential role in regulating neural ingrowth in the degenerate intervertebral disc. Arthritis Res. Ther. 2010, 12, R1. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Jing, L.; Chen, J. Changes in the molecular phenotype of nucleus pulposus cells with intervertebral disc aging. PLoS ONE 2012, 7, e52020. [Google Scholar] [CrossRef] [PubMed]
- Mathews, M.B. Comparative biochemistry of chondroitin sulphate-proteins of cartilage and notochord. Biochem. J. 1971, 125, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Bernhardt, R.R.; Schachner, M. Chondroitin sulfates affect the formation of the segmental motor nerves in zebrafish embryos. Dev. Biol. 2000, 221, 206–219. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Masuda, T.; Fukamauchi, F.; Takeda, Y.; Fujisawa, H.; Watanabe, K.; Okado, N.; Shiga, T. Developmental regulation of notochord-derived repulsion for dorsal root ganglion axons. Mol. Cell. Neurosci. 2004, 25, 217–227. [Google Scholar] [CrossRef] [PubMed]
- Purmessur, D.; Cornejo, M.C.; Cho, S.K.; Roughley, P.J.; Linhardt, R.J.; Hecht, A.C.; Iatridis, J.C. Intact glycosaminoglycans from intervertebral disc-derived notochordal cell-conditioned media inhibit neurite growth while maintaining neuronal cell viability. Spine J. 2015, 15, 1060–1069. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.Q.; Karasugi, T.; Cheung, K.M.; Chiba, K.; Ho, D.W.; Miyake, A.; Kao, P.Y.; Sze, K.L.; Yee, A.; Takahashi, A.; et al. Lumbar disc degeneration is linked to a carbohydrate sulfotransferase 3 variant. J. Clin. Investig. 2013, 123, 4909–4917. [Google Scholar] [CrossRef] [PubMed]
- Thiele, H.; Sakano, M.; Kitagawa, H.; Sugahara, K.; Rajab, A.; Hohne, W.; Ritter, H.; Leschik, G.; Nurnberg, P.; Mundlos, S. Loss of chondroitin 6-O-sulfotransferase-1 function results in severe human chondrodysplasia with progressive spinal involvement. Proc. Natl. Acad. Sci. USA 2004, 101, 10155–10160. [Google Scholar] [CrossRef] [PubMed]
- Hermanns, P.; Unger, S.; Rossi, A.; Perez-Aytes, A.; Cortina, H.; Bonafe, L.; Boccone, L.; Setzu, V.; Dutoit, M.; Sangiorgi, L.; et al. Congenital joint dislocations caused by carbohydrate sulfotransferase 3 deficiency in recessive larsen syndrome and humero-spinal dysostosis. Am. J. Hum. Genet. 2008, 82, 1368–1374. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.; Sakamoto, K.; Zhang, H.; Ito, Z.; Imagama, S.; Kishida, S.; Natori, T.; Sawada, M.; Matsuyama, Y.; Kadomatsu, K. Transforming growth factor-beta1 upregulates keratan sulfate and chondroitin sulfate biosynthesis in microglias after brain injury. Brain Res. 2009, 1263, 10–22. [Google Scholar] [CrossRef] [PubMed]
- Johnson, W.E.; Caterson, B.; Eisenstein, S.M.; Hynds, D.L.; Snow, D.M.; Roberts, S. Human intervertebral disc aggrecan inhibits nerve growth in vitro. Arthritis Rheum. 2002, 46, 2658–2664. [Google Scholar] [CrossRef] [PubMed]
- Olmarker, K.; Rydevik, B.; Nordborg, C. Autologous nucleus pulposus induces neurophysiologic and histologic changes in porcine cauda equina nerve roots. Spine 1993, 18, 1425–1432. [Google Scholar] [CrossRef] [PubMed]
- Alini, M.; Eisenstein, S.M.; Ito, K.; Little, C.; Kettler, A.A.; Masuda, K.; Melrose, J.; Ralphs, J.; Stokes, I.; Wilke, H.J. Are animal models useful for studying human disc disorders/degeneration? Eur. Spine J. 2008, 17, 2–19. [Google Scholar] [CrossRef] [PubMed]
- Hunter, C.J.; Matyas, J.R.; Duncan, N.A. The three-dimensional architecture of the notochordal nucleus pulposus: Novel observations on cell structures in the canine intervertebral disc. J. Anat. 2003, 202, 279–291. [Google Scholar] [CrossRef] [PubMed]
- Hunter, C.J.; Matyas, J.R.; Duncan, N.A. The functional significance of cell clusters in the notochordal nucleus pulposus: Survival and signaling in the canine intervertebral disc. Spine 2004, 29, 1099–1104. [Google Scholar] [CrossRef] [PubMed]
- Beachley, M.C.; Graham, F.H., Jr. Hypochondroplastic dwarfism (enchondral chondrodystrophy) in a dog. J. Am. Vet. Med. Assoc. 1973, 163, 283–284. [Google Scholar] [PubMed]
- Hansen, H.J. Some animal bone and joint diseases of comparative interest. Calcif. Tissue Res. 1968, 2, 23. [Google Scholar] [CrossRef]
- Smolders, L.A.; Bergknut, N.; Grinwis, G.C.; Hagman, R.; Lagerstedt, A.S.; Hazewinkel, H.A.; Tryfonidou, M.A.; Meij, B.P. Intervertebral disc degeneration in the dog. Part 2: Chondrodystrophic and non-chondrodystrophic breeds. Vet. J. 2013, 195, 292–299. [Google Scholar] [CrossRef] [PubMed]
- Olby, N.; Harris, T.; Burr, J.; Munana, K.; Sharp, N.; Keene, B. Recovery of pelvic limb function in dogs following acute intervertebral disc herniations. J. Neurotrauma 2004, 21, 49–59. [Google Scholar] [CrossRef] [PubMed]
- Cappello, R.; Bird, J.L.; Pfeiffer, D.; Bayliss, M.T.; Dudhia, J. Notochordal cell produce and assemble extracellular matrix in a distinct manner, which may be responsible for the maintenance of healthy nucleus pulposus. Spine 2006, 31, 873–882. [Google Scholar] [CrossRef] [PubMed]
- Klauser, M.; Forterre, F.; Doherr, M.; Zurbriggen, A.; Spreng, D.; Forterre, S. Evaluation of apoptotic cell death in normal and chondrodystrophic canine intervertebral discs. Vet. Sci. Dev. 2012, 2, e6. [Google Scholar] [CrossRef]
- Rosenthal, N.; Brown, S. The mouse ascending: Perspectives for human-disease models. Nat. Cell Biol. 2007, 9, 993–999. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, D.; Xu, T. The expanding role of mouse genetics for understanding human biology and disease. Dis. Model. Mech. 2008, 1, 56–66. [Google Scholar] [CrossRef] [PubMed]
- Hunter, C.J.; Matyas, J.R.; Duncan, N.A. Cytomorphology of notochordal and chondrocytic cells from the nucleus pulposus: A species comparison. J. Anat. 2004, 205, 357–362. [Google Scholar] [CrossRef] [PubMed]
- Nagy, A. Cre recombinase: The universal reagent for genome tailoring. Genesis 2000, 26, 99–109. [Google Scholar] [CrossRef]
- Perantoni, A.O.; Timofeeva, O.; Naillat, F.; Richman, C.; Pajni-Underwood, S.; Wilson, C.; Vainio, S.; Dove, L.F.; Lewandoski, M. Inactivation of FGF8 in early mesoderm reveals an essential role in kidney development. Development 2005, 132, 3859–3871. [Google Scholar] [CrossRef] [PubMed]
- Lange, A.; Gegg, M.; Burtscher, I.; Bengel, D.; Kremmer, E.; Lickert, H. Fltp(T2AiCre): A new knock-in mouse line for conditional gene targeting in distinct mono- and multiciliated tissues. Differentiation 2012, 83, S105–S113. [Google Scholar] [CrossRef] [PubMed]
- Ovchinnikov, D.A.; Deng, J.M.; Ogunrinu, G.; Behringer, R.R. Col2a1-directed expression of Cre recombinase in differentiating chondrocytes in transgenic mice. Genesis 2000, 26, 145–146. [Google Scholar] [CrossRef]
- Schipani, E.; Ryan, H.E.; Didrickson, S.; Kobayashi, T.; Knight, M.; Johnson, R.S. Hypoxia in cartilage: HIF-1alpha is essential for chondrocyte growth arrest and survival. Genes Dev. 2001, 15, 2865–2876. [Google Scholar] [PubMed]
- Sakai, K.; Hiripi, L.; Glumoff, V.; Brandau, O.; Eerola, R.; Vuorio, E.; Bosze, Z.; Fassler, R.; Aszodi, A. Stage-and tissue-specific expression of a Col2a1-Cre fusion gene in transgenic mice. Matrix Biol. 2001, 19, 761–767. [Google Scholar] [CrossRef]
- Terpstra, L.; Prud’homme, J.; Arabian, A.; Takeda, S.; Karsenty, G.; Dedhar, S.; St-Arnaud, R. Reduced chondrocyte proliferation and chondrodysplasia in mice lacking the integrin-linked kinase in chondrocytes. J. Cell Biol. 2003, 162, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Harfe, B.D.; Scherz, P.J.; Nissim, S.; Tian, H.; McMahon, A.P.; Tabin, C.J. Evidence for an expansion-based temporal shh gradient in specifying vertebrate digit identities. Cell 2004, 118, 517–528. [Google Scholar] [CrossRef] [PubMed]
- Semba, K.; Araki, K.; Li, Z.; Matsumoto, K.; Suzuki, M.; Nakagata, N.; Takagi, K.; Takeya, M.; Yoshinobu, K.; Araki, M.; et al. A novel murine gene, sickle tail, linked to the danforth’s short tail locus, is required for normal development of the intervertebral disc. Genetics 2006, 172, 445–456. [Google Scholar] [CrossRef] [PubMed]
- Abe, K.; Araki, K.; Tanigawa, M.; Semba, K.; Ando, T.; Sato, M.; Sakai, D.; Hiyama, A.; Mochida, J.; Yamamura, K. A Cre knock-in mouse line on the sickle tail locus induces recombination in the notochord and intervertebral disks. Genesis 2012, 50, 758–765. [Google Scholar] [CrossRef] [PubMed]
- Ang, S.L.; Wierda, A.; Wong, D.; Stevens, K.A.; Cascio, S.; Rossant, J.; Zaret, K.S. The formation and maintenance of the definitive endoderm lineage in the mouse: Involvement of HNF3/forkhead proteins. Development 1993, 119, 1301–1315. [Google Scholar] [PubMed]
- Ang, S.L.; Rossant, J. HNF-3 beta is essential for node and notochord formation in mouse development. Cell 1994, 78, 561–574. [Google Scholar] [CrossRef]
- Park, E.J.; Sun, X.; Nichol, P.; Saijoh, Y.; Martin, J.F.; Moon, A.M. System for tamoxifen-inducible expression of Cre-recombinase from the Foxa2 locus in mice. Dev. Dyn. 2008, 237, 447–453. [Google Scholar] [CrossRef] [PubMed]
- Uetzmann, L.; Burtscher, I.; Lickert, H. A mouse line expressing Foxa2-driven Cre recombinase in node, notochord, floorplate, and endoderm. Genesis 2008, 46, 515–522. [Google Scholar] [CrossRef] [PubMed]
- Merceron, C.; Mangiavini, L.; Robling, A.; Wilson, T.L.; Giaccia, A.J.; Shapiro, I.M.; Schipani, E.; Risbud, M.V. Loss of HIF-1alpha in the notochord results in cell death and complete disappearance of the nucleus pulposus. PLoS ONE 2014, 9, e110768. [Google Scholar] [CrossRef] [PubMed]
- Beckers, A.; Alten, L.; Viebahn, C.; Andre, P.; Gossler, A. The mouse homeobox gene noto regulates node morphogenesis, notochordal ciliogenesis, and left right patterning. Proc. Natl. Acad. Sci. USA 2007, 104, 15765–15770. [Google Scholar] [CrossRef] [PubMed]
- Darras, S.; Nishida, H. The BMP signaling pathway is required together with the FGF pathway for notochord induction in the ascidian embryo. Development 2001, 128, 2629–2638. [Google Scholar] [PubMed]
- Sobajima, S.; Shimer, A.L.; Chadderdon, R.C.; Kompel, J.F.; Kim, J.S.; Gilbertson, L.G.; Kang, J.D. Quantitative analysis of gene expression in a rabbit model of intervertebral disc degeneration by real-time polymerase chain reaction. Spine J. 2005, 5, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Takae, R.; Matsunaga, S.; Origuchi, N.; Yamamoto, T.; Morimoto, N.; Suzuki, S.; Sakou, T. Immunolocalization of bone morphogenetic protein and its receptors in degeneration of intervertebral disc. Spine 1999, 24, 1397–1401. [Google Scholar] [PubMed]
- Kim, H.; Lee, J.U.; Moon, S.H.; Kim, H.C.; Kwon, U.H.; Seol, N.H.; Kim, H.J.; Park, J.O.; Chun, H.J.; Kwon, I.K.; et al. Zonal responsiveness of the human intervertebral disc to bone morphogenetic protein-2. Spine 2009, 34, 1834–1838. [Google Scholar] [CrossRef] [PubMed]
- Than, K.D.; Rahman, S.U.; Vanaman, M.J.; Wang, A.C.; Lin, C.Y.; Zhang, H.; la Marca, F.; Park, P. Bone morphogenetic proteins and degenerative disk disease. Neurosurgery 2012, 70, 996–1002. [Google Scholar] [CrossRef] [PubMed]
- Wei, A.; Brisby, H.; Chung, S.A.; Diwan, A.D. Bone morphogenetic protein-7 protects human intervertebral disc cells in vitro from apoptosis. Spine J. 2008, 8, 466–474. [Google Scholar] [CrossRef] [PubMed]
- Carragee, E.J.; Hurwitz, E.L.; Weiner, B.K. A critical review of recombinant human bone morphogenetic protein-2 trials in spinal surgery: Emerging safety concerns and lessons learned. Spine J. 2011, 11, 471–491. [Google Scholar] [CrossRef] [PubMed]
- Smucker, J.D.; Rhee, J.M.; Singh, K.; Yoon, S.T.; Heller, J.G. Increased swelling complications associated with off-label usage of rhBMP-2 in the anterior cervical spine. Spine 2006, 31, 2813–2819. [Google Scholar] [CrossRef] [PubMed]
- Ukita, K.; Hirahara, S.; Oshima, N.; Imuta, Y.; Yoshimoto, A.; Jang, C.W.; Oginuma, M.; Saga, Y.; Behringer, R.R.; Kondoh, H.; et al. Wnt signaling maintains the notochord fate for progenitor cells and supports the posterior extension of the notochord. Mech. Dev. 2009, 126, 791–803. [Google Scholar] [CrossRef] [PubMed]
- Kondo, N.; Yuasa, T.; Shimono, K.; Tung, W.; Okabe, T.; Yasuhara, R.; Pacifici, M.; Zhang, Y.; Iwamoto, M.; Enomoto-Iwamoto, M. Intervertebral disc development is regulated by Wnt/β-catenin signaling. Spine 2011, 36, E513–E518. [Google Scholar] [CrossRef] [PubMed]
- Winkler, T.; Mahoney, E.J.; Sinner, D.; Wylie, C.C.; Dahia, C.L. Wnt signaling activates Shh signaling in early postnatal intervertebral discs, and re-activates Shh signaling in old discs in the mouse. PLoS ONE 2014, 9, e98444. [Google Scholar] [CrossRef] [PubMed]
- Dahia, C.L.; Mahoney, E.; Wylie, C. Shh signaling from the nucleus pulposus is required for the postnatal growth and differentiation of the mouse intervertebral disc. PLoS ONE 2012, 7, e35944. [Google Scholar] [CrossRef] [PubMed]
- Smolders, L.A.; Meij, B.P.; Riemers, F.M.; Licht, R.; Wubbolts, R.; Heuvel, D.; Grinwis, G.C.; Vernooij, H.C.; Hazewinkel, H.A.; Penning, L.C.; et al. Canonical Wnt signaling in the notochordal cell is upregulated in early intervertebral disk degeneration. J. Orthop. Res. 2012, 30, 950–957. [Google Scholar] [CrossRef] [PubMed]
- Showell, C.; Binder, O.; Conlon, F.L. T-box genes in early embryogenesis. Dev. Dyn. 2004, 229, 201–218. [Google Scholar] [CrossRef] [PubMed]
- Pillay, N.; Plagnol, V.; Tarpey, P.S.; Lobo, S.B.; Presneau, N.; Szuhai, K.; Halai, D.; Berisha, F.; Cannon, S.R.; Mead, S.; et al. A common single-nucleotide variant in T is strongly associated with chordoma. Nat. Genet. 2012, 44, 1185–1187. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.R.; Ng, D.; Alcorta, D.A.; Liebsch, N.J.; Sheridan, E.; Li, S.; Goldstein, A.M.; Parry, D.M.; Kelley, M.J. T (brachyury) gene duplication confers major susceptibility to familial chordoma. Nat. Genet. 2009, 41, 1176–1178. [Google Scholar] [CrossRef] [PubMed]
- McMaster, M.L.; Goldstein, A.M.; Bromley, C.M.; Ishibe, N.; Parry, D.M. Chordoma: Incidence and survival patterns in the united states, 1973–1995. Cancer Causes Control 2001, 12, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Chambers, K.J.; Lin, D.T.; Meier, J.; Remenschneider, A.; Herr, M.; Gray, S.T. Incidence and survival patterns of cranial chordoma in the united states. Laryngoscope 2014, 124, 1097–1102. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, T.; Suzuki, S.; Ishiiwa, H.; Ueda, Y. Intraosseous benign notochordal cell tumours: Overlooked precursors of classic chordomas? Histopathology 2004, 44, 597–602. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
McCann, M.R.; Séguin, C.A. Notochord Cells in Intervertebral Disc Development and Degeneration. J. Dev. Biol. 2016, 4, 3. https://doi.org/10.3390/jdb4010003
McCann MR, Séguin CA. Notochord Cells in Intervertebral Disc Development and Degeneration. Journal of Developmental Biology. 2016; 4(1):3. https://doi.org/10.3390/jdb4010003
Chicago/Turabian StyleMcCann, Matthew R., and Cheryle A. Séguin. 2016. "Notochord Cells in Intervertebral Disc Development and Degeneration" Journal of Developmental Biology 4, no. 1: 3. https://doi.org/10.3390/jdb4010003
APA StyleMcCann, M. R., & Séguin, C. A. (2016). Notochord Cells in Intervertebral Disc Development and Degeneration. Journal of Developmental Biology, 4(1), 3. https://doi.org/10.3390/jdb4010003





