Carbon-Carbon Double Bond and Resorcinol in Resveratrol and Its Analogues: What Is the Characteristic Structure in Quenching Singlet Oxygen?
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents and Sample Preparation
2.2. Density Functional Method
2.3. Qualitative Analysis by UHPLC-QTOF-MS2
2.4. Quantitative Analysis by UHPLC-QQQ-MS2
2.5. Statistical Analysis
3. Results and Discussion
3.1. Qualitative Comparison of the Activity of Double Bond and Resorcinol Ring Quenching 1O2
3.2. Quantitative Comparison of the Activity of the Catechol Ring, Carbon-Carbon Double Bond and Resorcinol Ring Quenching 1O2
3.3. Validation of the Mechanism by B3LYP Density Functional Method
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Defedericis, H.C.; Patrzyc, H.B.; Rajecki, M.J.; Budzinski, E.E.; Iijima, H.; Dawidzik, J.B.; Evans, M.S.; Greene, K.F.; Box, H.C. Singlet oxygen-induced DNA damage. Radiat. Res. 2006, 165, 445–451. [Google Scholar] [CrossRef] [PubMed]
- Liang, D.; Zhang, Y.N.; Wu, Z.Y.; Chen, Y.J.; Yang, X.; Sun, M.T.; Ni, R.Y.; Bian, J.S.; Huang, D.J. A near infrared singlet oxygen probe and its applications in in vivo imaging and measurement of singlet oxygen quenching activity of flavonoids. Sens. Actuator B Chem. 2018, 266, 645–654. [Google Scholar] [CrossRef]
- Lu, W.; Liu, J. Capturing Transient Endoperoxide in the Singlet Oxygen Oxidation of Guanine. Chemistry 2016, 22, 3127–3138. [Google Scholar] [CrossRef] [PubMed]
- Moriwaki, S.; Takahashi, Y.; Shimizu, H.; Inoue, M.; Sugiyama, Y.; Inoue, S. Decreased repair of singlet oxygen-induced DNA damage in xeroderma pigmentosum group A cells determined by plasmid host cell reactivation. J. Dermatol. Sci. 2012, 66, 242–244. [Google Scholar] [CrossRef] [PubMed]
- Runger, T.M.; Epe, B.; Moller, K. Repair of ultraviolet B and singlet oxygen-induced DNA damage in xeroderma pigmentosum cells. J. Investig. Dermatol. 1995, 104, 68–73. [Google Scholar] [CrossRef] [PubMed]
- Onyango, A.N. Endogenous Generation of Singlet Oxygen and Ozone in Human and Animal Tissues: Mechanisms, Biological Significance, and Influence of Dietary Components. Oxidative Med. Cell. Longev. 2016, 2016, 22. [Google Scholar] [CrossRef] [PubMed]
- Chiarelli-Neto, O.; Ferreira, A.S.; Martins, W.K.; Pavani, C.; Severino, D.; Faiao-Flores, F.; Maria-Engler, S.S.; Aliprandini, E.; Martinez, G.R.; Di Mascio, P.; et al. Melanin photosensitization and the effect of visible light on epithelial cells. PLoS ONE 2014, 9, e113266. [Google Scholar] [CrossRef] [PubMed]
- Jung, M.Y.; Choi, D.S. Electron spin resonance and luminescence spectroscopic observation and kinetic study of chemical and physical singlet oxygen quenching by resveratrol in methanol. J. Agric. Food Chem. 2010, 58, 11888–11895. [Google Scholar] [CrossRef]
- Akinwumi, B.C.; Bordun, K.M.; Anderson, H.D. Biological Activities of Stilbenoids. Int. J. Mol. Sci. 2018, 19, 792. [Google Scholar] [CrossRef]
- Rivière, C.; Pawlus, A.D.; Mérillon, J.M. Natural stilbenoids: Distribution in the plant kingdom and chemotaxonomic interest in Vitaceae. Nat. Prod. Rep. 2012, 29, 1317–1333. [Google Scholar]
- Ohguchi, K.; Tanaka, T.; Ito, T.; Iinuma, M.; Matsumoto, K.; Akao, Y.; Nozawa, Y. Inhibitory effects of resveratrol derivatives from dipterocarpaceae plants on tyrosinase activity. Biosci. Biotechnol. Biochem. 2003, 67, 1587–1589. [Google Scholar] [CrossRef] [PubMed]
- Murias, M.; Jäger, W.; Handler, N.; Erker, T.; Horvath, Z.; Szekeres, T.; Nohl, H.; Gille, L. Antioxidant, prooxidant and cytotoxic activity of hydroxylated resveratrol analogues: Structure–activity relationship. Biochem. Pharmacol. 2005, 69, 903–912. [Google Scholar] [CrossRef] [PubMed]
- Kotora, P.; Sersen, F.; Filo, J.; Loos, D.; Gregan, J.; Gregan, F. The Scavenging of DPPH, Galvinoxyl and ABTS Radicals by Imine Analogs of Resveratrol. Molecules 2016, 21, 127. [Google Scholar] [CrossRef] [PubMed]
- De Filippis, B.; Ammazzalorso, A.; Fantacuzzi, M.; Giampietro, L.; Maccallini, C.; Amoroso, R. Anticancer Activity of Stilbene-Based Derivatives. Chem. Med. Chem. 2017, 12, 558–570. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Jiang, L.; Wu, B.; Pan, Y.; Sun, C. Pallidol, a resveratrol dimer from red wine, is a selective singlet oxygen quencher. Biochem. Biophys. Res. Commun. 2009, 379, 283–287. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Lu, Y.; Jiang, L.; Wu, B.; Zhang, F.; Pan, Y. Preparative isolation and purification of antioxidative stilbene oligomers from Vitis chunganeniss using high-speed counter-current chromatography in stepwise elution mode. J. Sep. Sci. 2009, 32, 2339–2345. [Google Scholar] [CrossRef]
- He, S.; Jiang, L.Y.; Wu, B.; Zhou, J.; Pan, Y.J. Two Novel Antioxidative Stilbene Tetramers from Parthenocissus laetevirens. Helv. Chim. Acta 2009, 92, 1260–1267. [Google Scholar] [CrossRef]
- He, S.; Jiang, L.Y.; Wu, B.; Li, C.; Li, Y.J. Chunganenol: An Unusual Antioxidative Resveratrol Hexamer from Vitis chunganensis. J. Org. Chem. 2009, 74, 7966–7969. [Google Scholar] [CrossRef]
- Kong, Q.; Ren, X.; Jiang, L.; Pan, Y.; Sun, C. Scirpusin A, a hydroxystilbene dimer from Xinjiang wine grape, acts as an effective singlet oxygen quencher and DNA damage protector. J. Sci. Food Agric. 2010, 90, 823–828. [Google Scholar] [CrossRef]
- Jiang, L.Y.; He, S.; Jiang, K.Z.; Jiang, C.R.; Pan, Y.J. Resveratrol and its oligomers from wine grapes are selective 1O2 quenchers: Mechanistic implication by high-performance liquid chromatography-electrospray ionization-tandem mass spectrometry and theoretical calculation. J. Agric. Food Chem. 2010, 58, 9020–9027. [Google Scholar] [CrossRef]
- Celaje, J.A.; Zhang, D.; Guerrero, A.M.; Selke, M. Chemistry of trans-Resveratrol with Singlet Oxygen: [2+2] Addition, [4+2] Addition, and Formation of the Phytoalexin Moracin M. Org. Lett. 2011, 13, 4846–4849. [Google Scholar] [CrossRef] [PubMed]
- Yin, X.; Yu, J.; Kong, Q.; Ren, X. Mechanism of isomers and analogues of resveratrol dimers selectively quenching singlet oxygen by UHPLC-ESI-MS2. Food Chem. 2017, 237, 1101–1111. [Google Scholar] [CrossRef] [PubMed]
- Choo, Q.Y.; Yeo, S.C.M.; Yeo, P.C.; Tanaka, Y.; Lin, H.S. Pterostilbene surpassed resveratrol for anti-inflammatory application: Potency consideration and pharmacokinetics perspective. J. Funct. Foods 2014, 11, 352–362. [Google Scholar] [CrossRef]
- Hirayama, O.; Nakamura, K.; Hamada, S.; Kobayasi, Y. Singlet oxygen quenching ability of naturally occurring carotenoids. Lipids 1994, 29, 149–150. [Google Scholar] [CrossRef] [PubMed]
- Thomas, H.; Thomas, H.; Glomb, M.A. Lycopene Inhibits the Isomerization of β-Carotene during Quenching of Singlet Oxygen and Free Radicals. J. Agric. Food Chem. 2015, 63, 3279–3287. [Google Scholar]
- Yun, S.C.; Jung, M.Y. Kinetic study on the singlet oxygen quenching activity of nordihydroguaiaretic acid (NDGA) using methylene blue sensitized photooxidation of α-terpinene. Food Sci. Biotechnol. 2016, 25, 1333–1336. [Google Scholar]
- Liu, C.; Si, H.; Han, P.; Tang, M. Density functional theory study on structure and stability of BeBn+clusters. Rapid Commun. Mass Spectrom. 2017, 31, 1437–1444. [Google Scholar] [CrossRef] [PubMed]
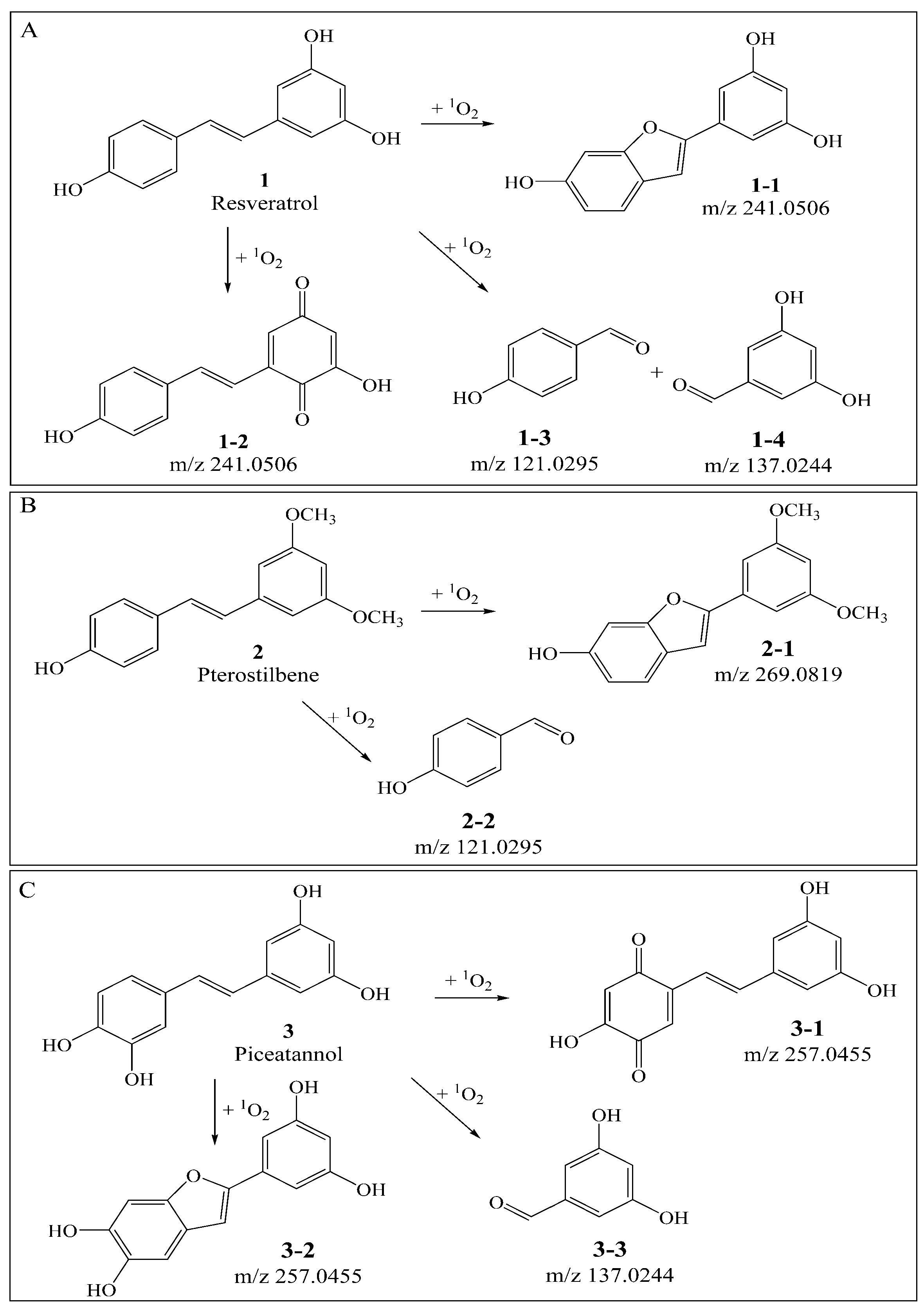
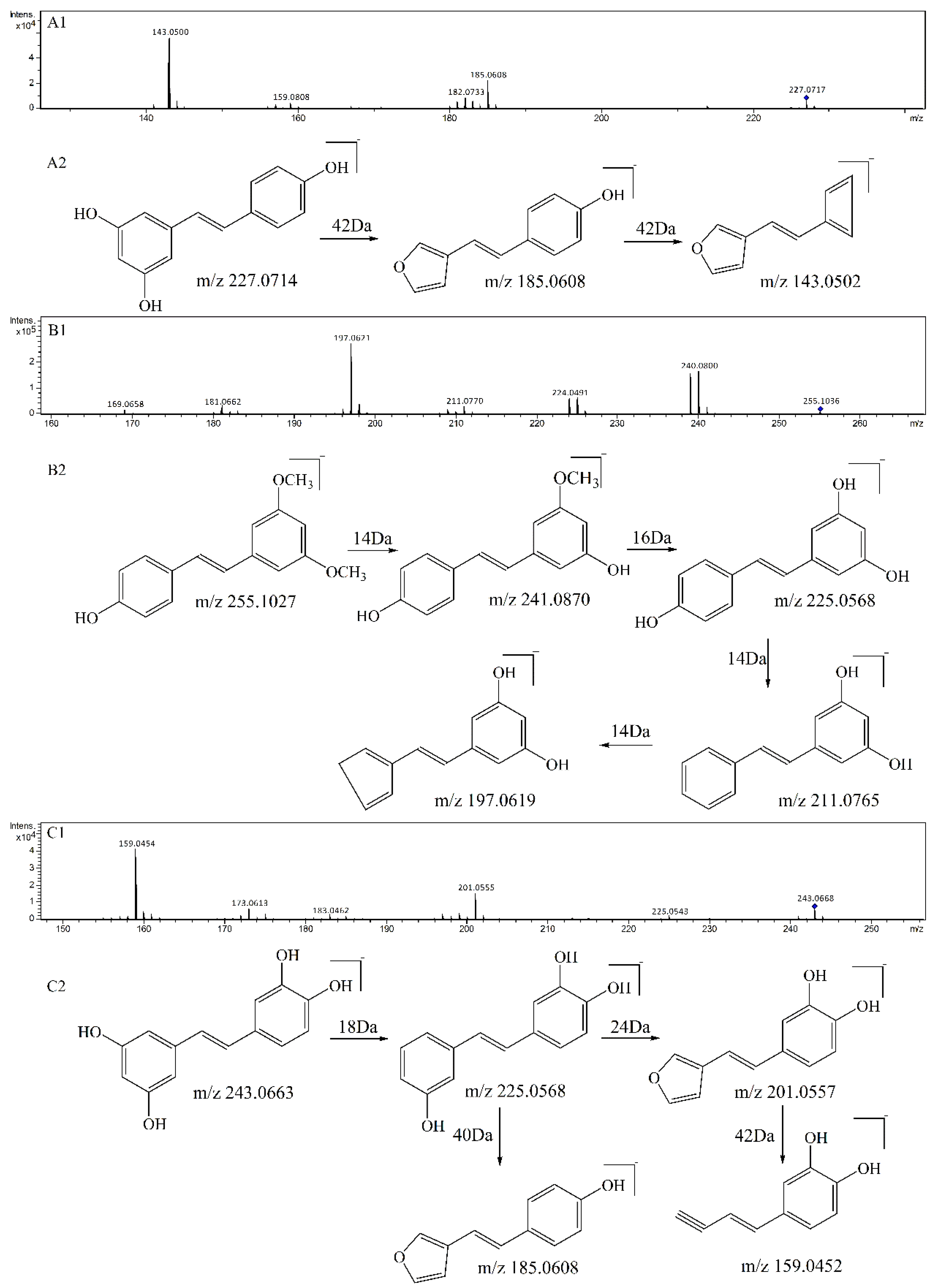
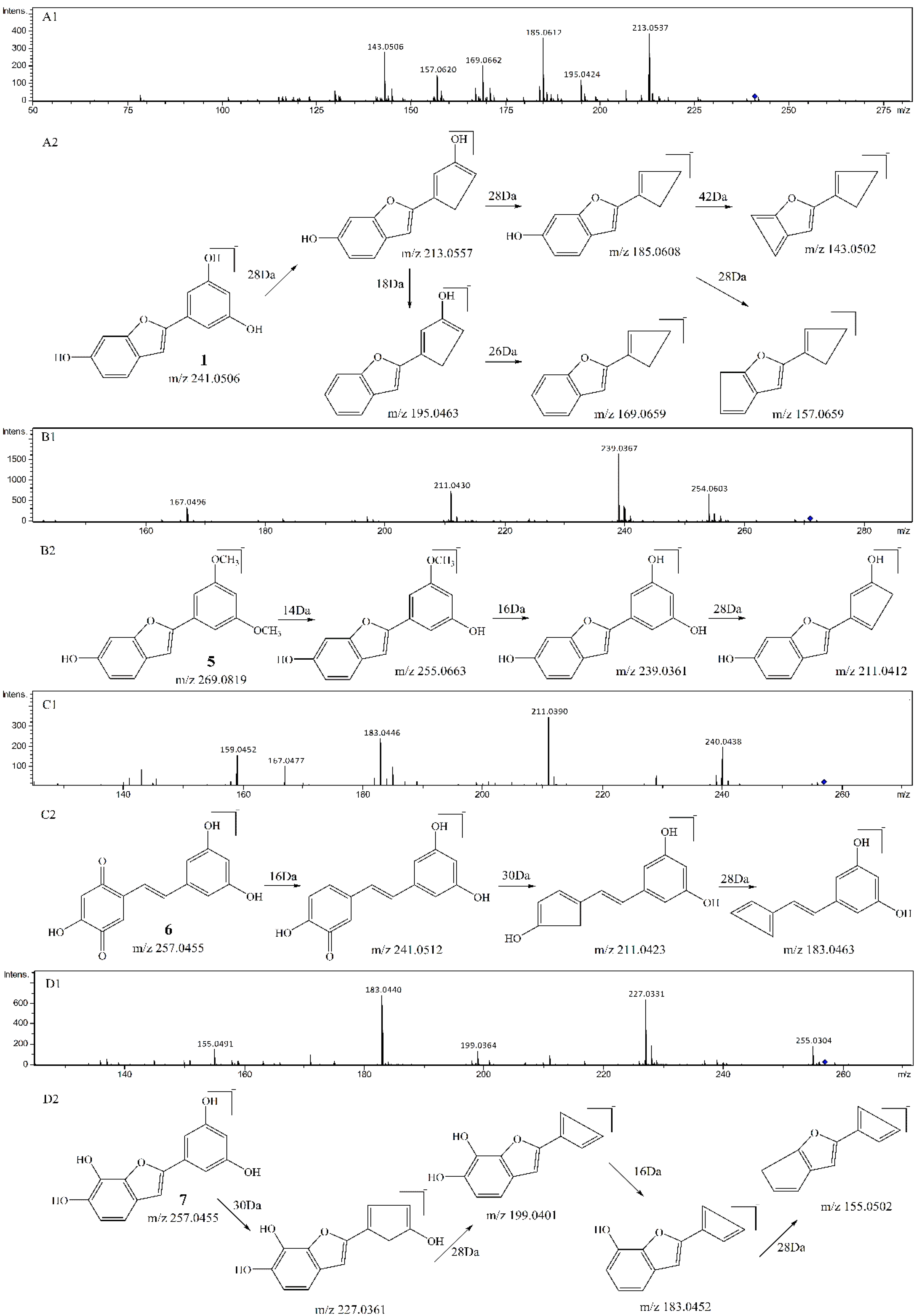
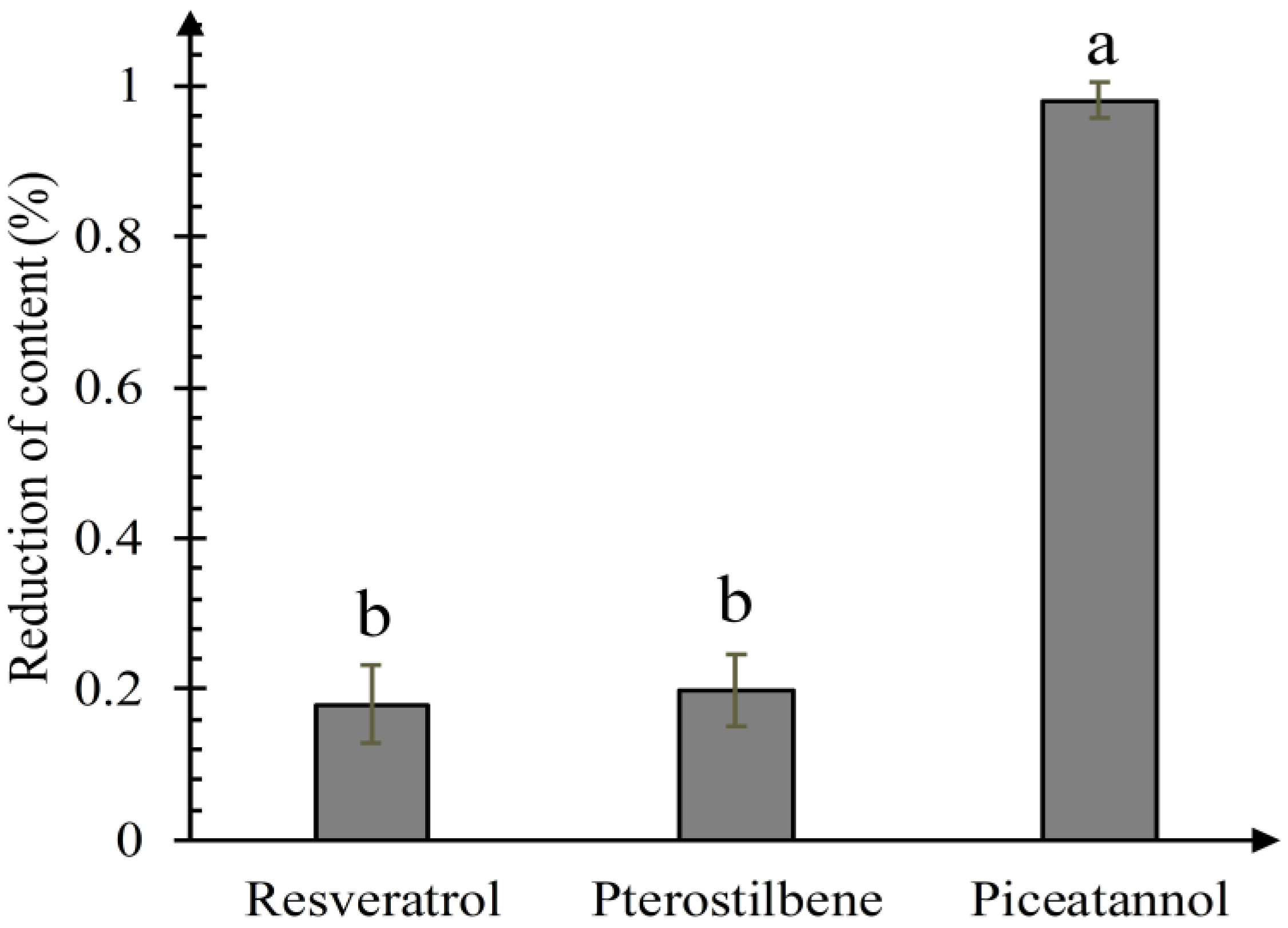
| Compounds | Precursorion | Production | Fragmentor (V) | Collision Energy (eV) |
|---|---|---|---|---|
| resveratrol | m/z 227 | m/z 185 | 75 | 15 |
| pterostilbene | m/z 255 | m/z 239.8 | 75 | 15 |
| piceatannol | m/z 243 | m/z 200.6 | 90 | 8 |
| Compound 1-1 | Compound 1-2 |
|---|---|
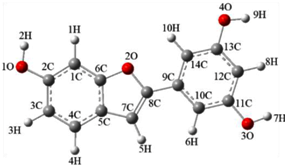 | 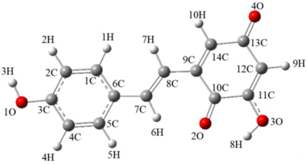 |
| E(b3lyp/6-311g(d,p)) | |
| −840.6133 Hartree/Particle | −840.5894 Hartree/Particle |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kong, Q.; Ren, X.; Qi, J.; Yu, J.; Lu, J.; Wang, S. Carbon-Carbon Double Bond and Resorcinol in Resveratrol and Its Analogues: What Is the Characteristic Structure in Quenching Singlet Oxygen? Biomolecules 2019, 9, 268. https://doi.org/10.3390/biom9070268
Kong Q, Ren X, Qi J, Yu J, Lu J, Wang S. Carbon-Carbon Double Bond and Resorcinol in Resveratrol and Its Analogues: What Is the Characteristic Structure in Quenching Singlet Oxygen? Biomolecules. 2019; 9(7):268. https://doi.org/10.3390/biom9070268
Chicago/Turabian StyleKong, Qingjun, Xueyan Ren, Jianrui Qi, Jia Yu, Jun Lu, and Shuo Wang. 2019. "Carbon-Carbon Double Bond and Resorcinol in Resveratrol and Its Analogues: What Is the Characteristic Structure in Quenching Singlet Oxygen?" Biomolecules 9, no. 7: 268. https://doi.org/10.3390/biom9070268
APA StyleKong, Q., Ren, X., Qi, J., Yu, J., Lu, J., & Wang, S. (2019). Carbon-Carbon Double Bond and Resorcinol in Resveratrol and Its Analogues: What Is the Characteristic Structure in Quenching Singlet Oxygen? Biomolecules, 9(7), 268. https://doi.org/10.3390/biom9070268






