Inclusion Complexes of β and HPβ-Cyclodextrin with α, β Amyrin and In Vitro Anti-Inflammatory Activity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of ICs
2.2.1. Physical Mixture (PM)
2.2.2. Kneading (KN)
2.3. Physicochemical Characterization
2.3.1. Fourier Transform Infrared (FT-IR)
2.3.2. Scanning Electronic Microscopy (SEM)
2.3.3. Powder X-Ray Diffraction (XRD)
2.3.4. Thermogravimetry (TG)
2.3.5. Differential Scanning Calorimetry (DSC)
2.4. In Vitro Anti-Inflammatory Study
Quantification of Nitric Oxide (NO−)
2.5. Cell Viability Assay
2.6. Statistical Analysis
3. Results
3.1. Physicochemical Characterization
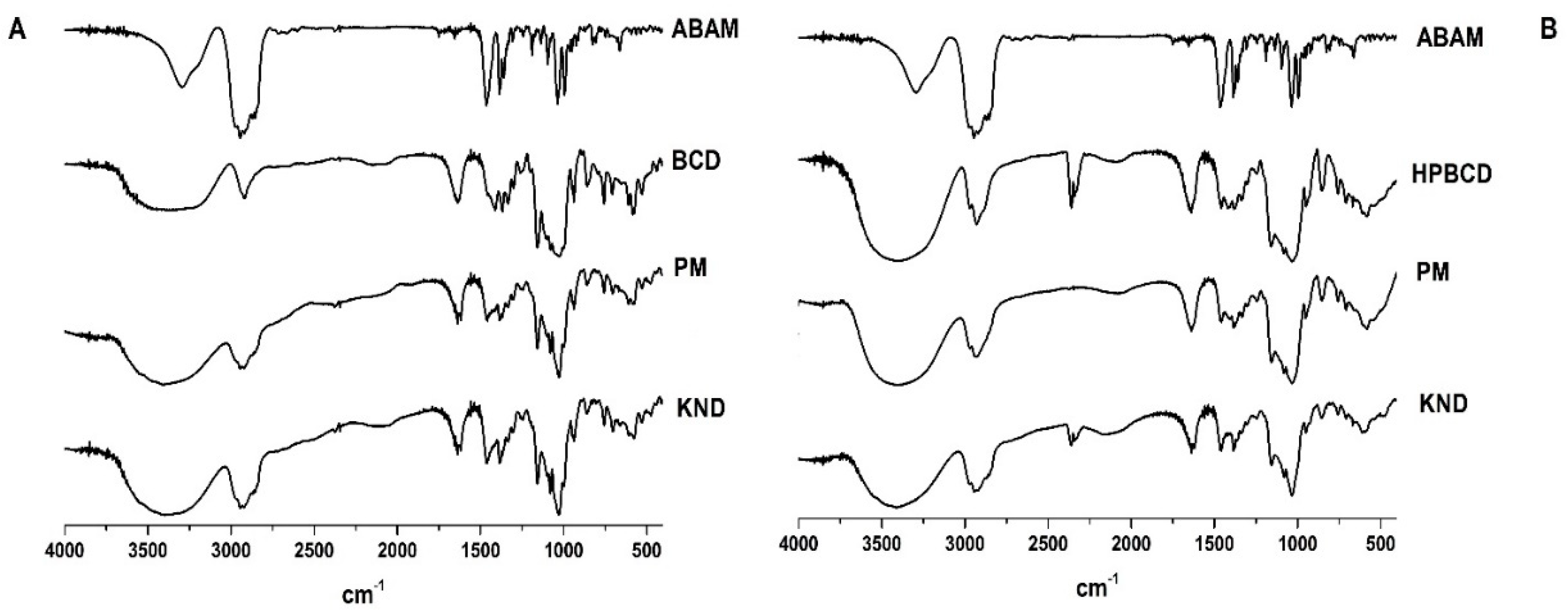
3.1.1. Fourier Transform Infrared (FT-IR)
3.1.2. Scanning Electronic Microscopy (SEM)
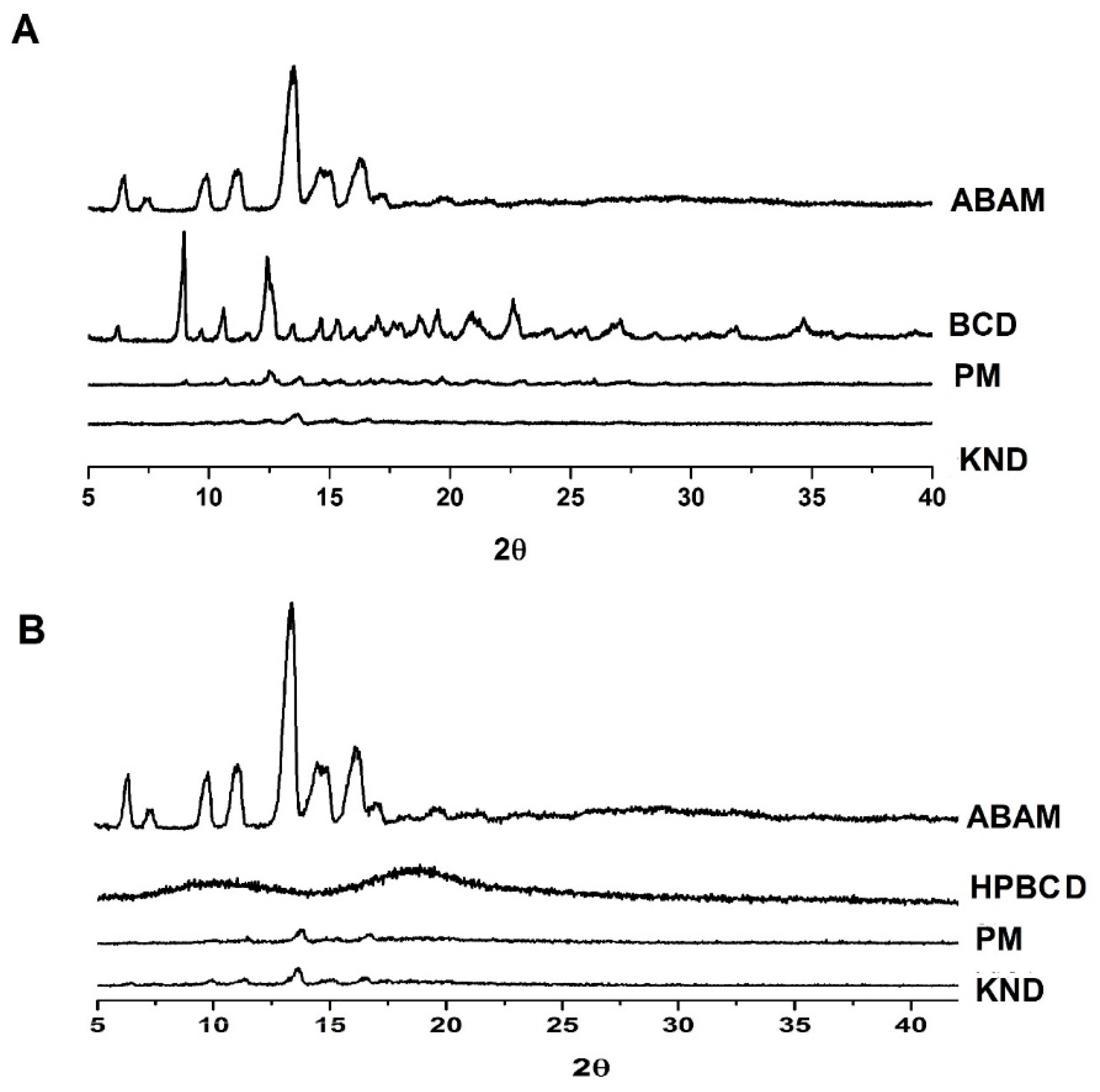
3.1.3. Powder X-ray Diffraction (XRD)
3.1.4. Thermogravimetry (TG)
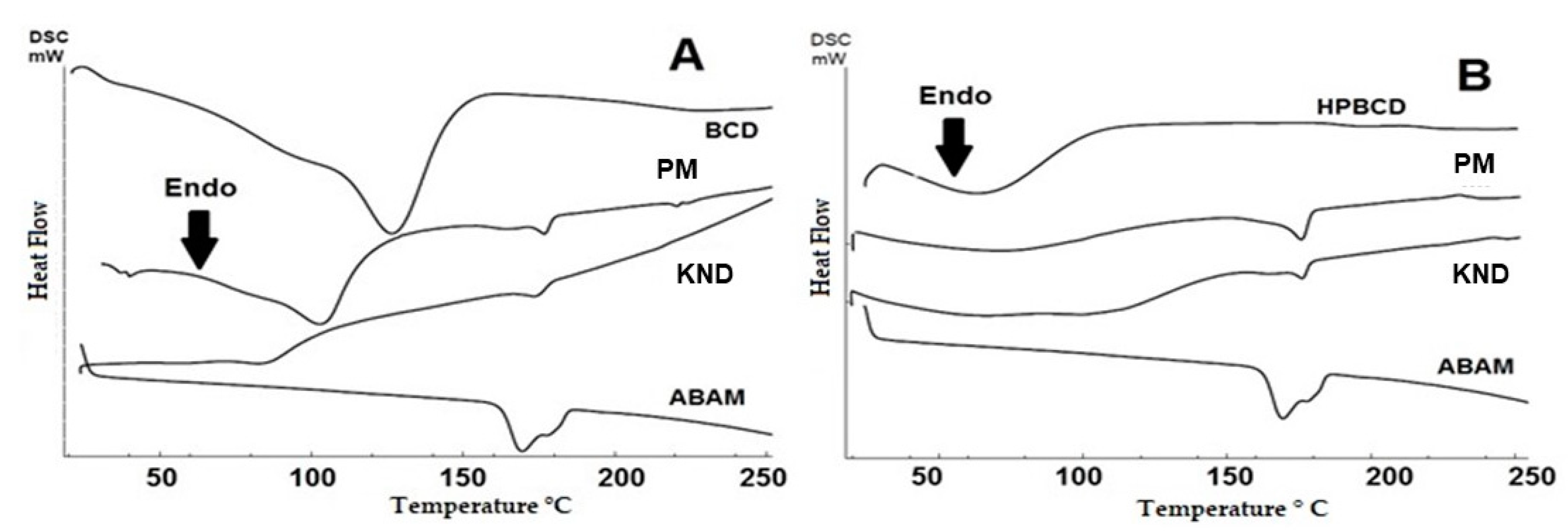
3.1.5. Differential Scanning Calorimetry (DSC)
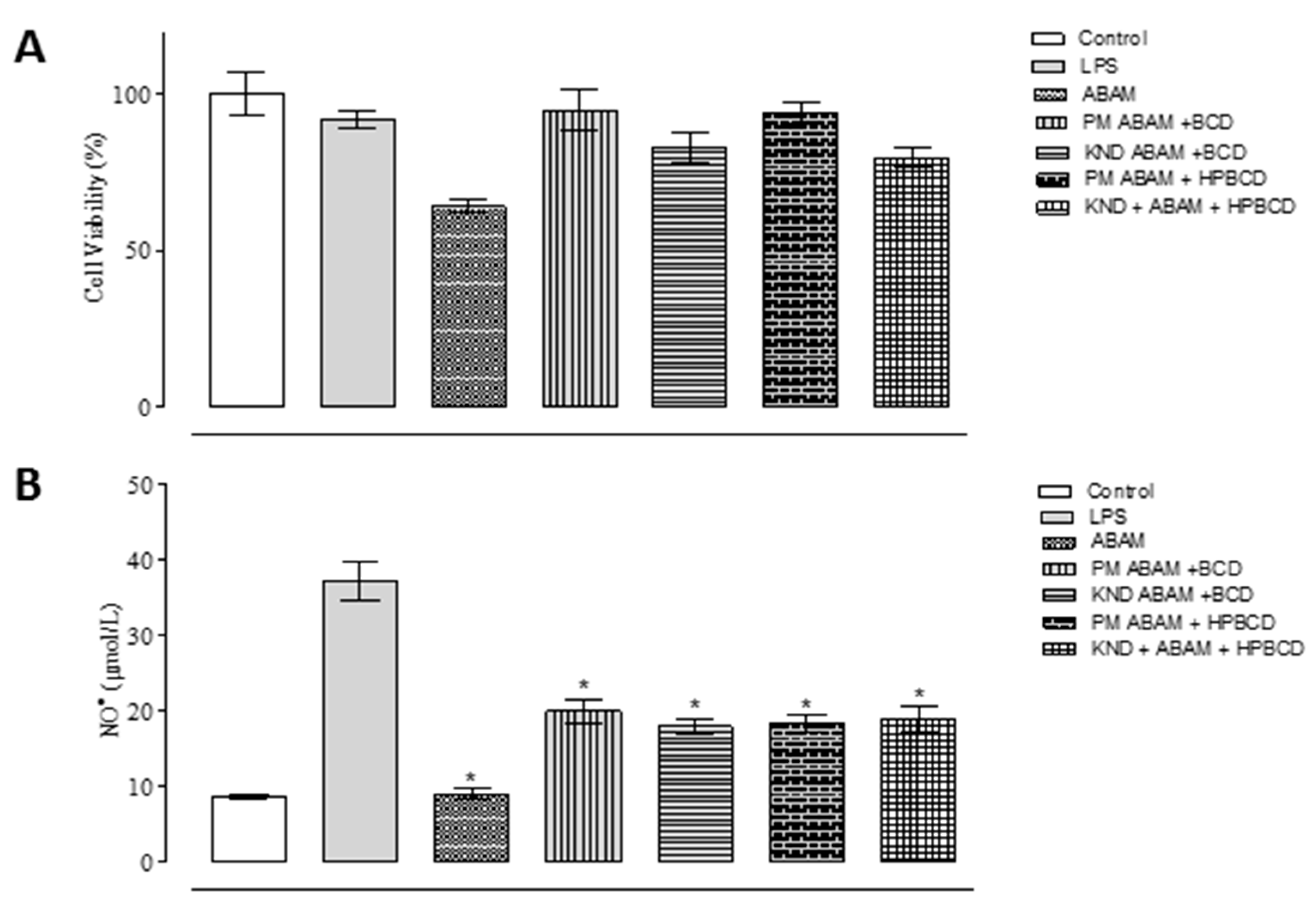
3.2. In Vitro Anti-inflammatory Study
3.3. Cell Viability Assay
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lorenzi, H. 1949- Árvores Brasileiras Manual de Identificacão e Cultivo de Plantas Arbóreas Nativas do Brasil; Instituto Plantarum de Estudos da Flora: Nova Odessa, Brazil, 2008; ISBN 8586714313. [Google Scholar]
- Siani, A.C.; Ramos, M.F.S.; Menezes-de-Lima, O., Jr.; Ribeiro-dos-Santos, R.; Fernadez-Ferreira, E.; Soares, R.O.A.; Rosas, E.C.; Susunaga, G.S.; Guimarães, A.C.; Zoghbi, M.G.B.; et al. Evaluation of anti-in ammatory-related activity of essential oils from the leaves and resin of species of Protium. J. Ethnopharmacol. 1999, 66, 57–69. [Google Scholar] [CrossRef]
- Lima, E.M.; Nascimento, A.M.; Lenz, D.; Scherer, R.; Meyrelles, S.S.; Boëchat, G.A.P.; Andrade, T.U.; Endringer, D.C. Triterpenes from the Protium heptaphyllum resin-chemical composition and cytotoxicity. Rev. Bras. Farmacogn. 2014, 24, 399–407. [Google Scholar] [CrossRef]
- Sheng, H.; Sun, H. Synthesis, biology and clinical significance of pentacyclic triterpenes: A multi-target approach to prevention and treatment of metabolic and vascular diseases. Nat. Prod. Rep. 2011, 28, 543. [Google Scholar] [CrossRef] [PubMed]
- Holanda Pinto, S.A.; Pinto, L.M.S.; Cunha, G.M.A.; Chaves, M.H.; Santos, F.A.; Rao, V.S. Anti-inflammatory effect of α, β-Amyrin, a pentacyclic triterpene from Protium heptaphyllum in rat model of acute periodontitis. Inflammopharmacology 2008, 16, 48–52. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, F.A.; Vieira-Júnior, G.M.; Chaves, M.H.; Almeida, F.R.C.; Florêncio, M.G.; Lima, R.C.P.; Silva, R.M.; Santos, F.A.; Rao, V.S.N. Gastroprotective and anti-inflammatory effects of resin from Protium heptaphyllum in mice and rats. Pharmacol. Res. 2004, 49, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Barros, F.W.A.; Bandeira, P.N.; Lima, D.J.B.; Meira, A.S.; de Farias, S.S.; Albuquerque, M.R.J.R.; dos Santos, H.S.; Lemos, T.L.G.; de Morais, M.O.; Costa-Lotufo, L.V.; et al. Amyrin esters induce cell death by apoptosis in HL-60 leukemia cells. Bioorg. Med. Chem. 2011, 19, 1268–1276. [Google Scholar] [CrossRef] [PubMed]
- Aragão, G.F.; Carneiro, L.M.V.; Junior, A.P.F.; Vieira, L.C.; Bandeira, P.N.; Lemos, T.L.G.; Viana, G.S.D.B. A possible mechanism for anxiolytic and antidepressant effects of alpha- and beta-amyrin from Protium heptaphyllum (Aubl.) March. Pharmacol. Biochem. Behav. 2006, 85, 827–834. [Google Scholar] [CrossRef]
- Bandeira, P.N.; Lemos, T.L.G.; Costa, S.M.O.; Santos, H.S. Obtenção de derivados da mistura triterpenoídica α- e β-amirina. Rev. Bras. Farmacogn. 2007, 17, 204–208. [Google Scholar] [CrossRef]
- Ching, J.; Lin, H.-S.; Tan, C.-H.; Koh, H.-L. Quantification of α- and β-amyrin in rat plasma by gas chromatography-mass spectrometry: Application to preclinical pharmacokinetic study. J. Mass Spectrom. 2011, 46, 457–464. [Google Scholar] [CrossRef]
- Silva, M.C.G.; Silva, S.C.B.L.; Santos, T.P.; Soares, P.R.L.; Andrade, A.L.C.; Cadena, M.R.S.; Cadena, P.G.; Silva, M.C.G.; Silva, S.C.B.L.; Santos, T.P.; et al. Avaliação do impacto causado pela disponibilidade de 17β-estradiol livre ou complexado à β-ciclodextrina no ambiente aquático sobre Oreochromis niloticus (tilápia). Arq. Bras. Med. Veterinária Zootec. 2018, 70, 222–230. [Google Scholar] [CrossRef]
- Davis, M.E.; Brewster, M.E. Cyclodextrin-based pharmaceutics: Past, present and future. Nat. Rev. Drug Discov. 2004, 3, 1023–1035. [Google Scholar] [CrossRef]
- Serpe, L.; Franz-Montan, M.; dos Santos, C.P.; da Silva, C.B.; Nolasco, F.P.; Caldas, C.S.; Volpato, M.C.; de Paula, E.; Groppo, F.C. Anaesthetic efficacy of bupivacaine 2-hydroxypropyl-β-cyclodextrin for dental anaesthesia after inferior alveolar nerve block in rats. Br. J. Oral Maxillofac. Surg. 2014, 52, 452–457. [Google Scholar] [CrossRef] [PubMed]
- Dahan, A.; Miller, J.M.; Hoffman, A.; Amidon, G.E.; Amidon, G.L. The Solubility–Permeability Interplay in Using Cyclodextrins as Pharmaceutical Solubilizers: Mechanistic Modeling and Application to Progesterone. J. Pharm. Sci. 2010, 99, 2739–2749. [Google Scholar] [CrossRef] [PubMed]
- Santos, P.L.; Brito, R.G.; Quintans, J.S.S.; Araujo, A.A.S.; Menezes, I.R.A.; Brogden, N.K.; Quintans-Junior, L.J. Cyclodextrins as Complexation Agents to Improve the Anti-inflammatory Drugs Profile: A Systematic Review and Meta-Analysis. Curr. Pharm. Des. 2017, 23. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, L.B.; Martins, A.O.B.P.B.; Ribeiro-Filho, J.; Cesário, F.R.A.S.; e Castro, F.F.; de Albuquerque, T.R.; Fernandes, M.N.M.; da Silva, B.A.F.; Quintans Júnior, L.J.; de Sousa Araújo, A.A.; et al. Anti-inflammatory activity of the essential oil obtained from Ocimum basilicum complexed with β-cyclodextrin (β-CD) in mice. Food Chem. Toxicol. 2017, 109, 836–846. [Google Scholar] [CrossRef] [PubMed]
- Raza, A.; Sun, H.; Bano, S.; Zhao, Y.; Xu, X.; Tang, J. Preparation, characterization, and in vitro anti-inflammatory evaluation of novel water soluble kamebakaurin/hydroxypropyl-β-cyclodextrin inclusion complex. J. Mol. Struct. 2017, 1130, 319–326. [Google Scholar] [CrossRef]
- Pinheiro, J.; Tavares, E.; Silva, S.; Félix Silva, J.; Carvalho, Y.; Ferreira, M.; Araújo, A.; Barbosa, E.; Fernandes Pedrosa, M.; Soares, L.; et al. Inclusion Complexes of Copaiba (Copaifera multijuga Hayne) Oleoresin and Cyclodextrins: Physicochemical Characterization and Anti-Inflammatory Activity. Int. J. Mol. Sci. 2017, 18, 2388. [Google Scholar] [CrossRef] [PubMed]
- da Silva Junior, W.F.; de Oliveira Pinheiro, J.G.; de França Alm Moreira, C.D.L.; Rudiger, A.L.; Barbosa, E.G.; Lima, E.S.; da Veiga Junior, V.F.; da Silva Junior, A.A.; Aragão, C.F.S.; de Lima, Á.A.N. Thermal behavior and termal degradation kinetic parameters of triterpene α, β amyrin. J. Therm. Anal. Calorim. 2017, 192, 788–802. [Google Scholar]
- National Center for Biotechnology Information. PubChem Database. alpha-Amyrin, CID=73170. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/alpha-Amyrin (accessed on 3 June 2019).
- National Center for Biotechnology Information. PubChem Database. beta-Amyrin, CID=73145. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/beta-Amyrin (accessed on 3 June 2019).
- Galvão, J.G.; Silva, V.F.; Ferreira, S.G.; França, F.R.M.; Santos, D.A.; Freitas, L.S.; Alves, P.B.; Araújo, A.A.S.; Cavalcanti, S.C.H.; Nunes, R.S. β-cyclodextrin inclusion complexes containing Citrus sinensis (L.) Osbeck essential oil: An alternative to control Aedes aegypti larvae. Thermochim. Acta 2015, 608, 14–19. [Google Scholar] [CrossRef]
- Verma, N.; Tripathi, S.K.; Sahu, D.; Das, H.R.; Das, R.H. Evaluation of inhibitory activities of plant extracts on production of LPS-stimulated pro-inflammatory mediators in J774 murine macrophages. Mol. Cell. Biochem. 2010, 336, 127–135. [Google Scholar] [CrossRef]
- Lopez-Zamora, S.; Alkhlel, A.; de Lasa, H. Monitoring the progress of catalytic cracking for model compounds in the mid-infrared (MIR) 3200–2800 cm−1 range. Chem. Eng. Sci. 2018, 192, 788–802. [Google Scholar] [CrossRef]
- Abarca, R.L.; Rodríguez, F.J.; Guarda, A.; Galotto, M.J.; Bruna, J.E. Characterization of beta-cyclodextrin inclusion complexes containing an essential oil component. Food Chem. 2016, 196, 968–975. [Google Scholar] [CrossRef] [PubMed]
- Passos, J.J.; De Sousa, F.B.; Mundim, I.M.; Bonfim, R.R.; Melo, R.; Viana, A.F.; Stolz, E.D.; Borsoi, M.; Rates, S.M.K.; Sinisterra, R.D. In vivo evaluation of the highly soluble oral β-cyclodextrin–Sertraline supramolecular complexes. Int. J. Pharm. 2012, 436, 478–485. [Google Scholar] [CrossRef] [PubMed]
- Medarević, D.; Kachrimanis, K.; Djurić, Z.; Ibrić, S. Influence of hydrophilic polymers on the complexation of carbamazepine with hydroxypropyl-β-cyclodextrin. Eur. J. Pharm. Sci. 2015, 78, 273–285. [Google Scholar] [CrossRef]
- Marques, H.M.C. A review on cyclodextrin encapsulation of essential oils and volatiles. Flavour Fragr. J. 2010, 25, 313–326. [Google Scholar] [CrossRef]
- Li, J.; Zhang, M.; Chao, J.; Shuang, S. Preparation and characterization of the inclusion complex of Baicalin (BG) with β-CD and HP-β-CD in solution: An antioxidant ability study. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2009, 73, 752–756. [Google Scholar] [CrossRef]
- Bulani, V.D.; Kothavade, P.S.; Kundaikar, H.S.; Gawali, N.B.; Chowdhury, A.A.; Degani, M.S.; Juvekar, A.R. Inclusion complex of ellagic acid with β-cyclodextrin: Characterization and in vitro anti-inflammatory evaluation. J. Mol. Struct. 2016, 1105, 308–315. [Google Scholar] [CrossRef]
- de Melo, P.N.; Barbosa, E.G.; Garnero, C.; de Caland, L.B.; Fernandes-Pedrosa, M.F.; Longhi, M.R.; da Silva-Júnior, A.A. Interaction pathways of specific co-solvents with hydroxypropyl-β-cyclodextrin inclusion complexes with benznidazole in liquid and solid phase. J. Mol. Liq. 2016, 223, 350–359. [Google Scholar] [CrossRef]
- Ding, L.; He, J.; Huang, L.; Lu, R. Studies on a novel modified β-cyclodextrin inclusion complex. J. Mol. Struct. 2010, 979, 122–127. [Google Scholar] [CrossRef]
- Sharma, V.K.; Mazumdar, B. Feasibility and characterization of gummy exudate of Cochlospermum religiosum as pharmaceutical excipient. Ind. Crops Prod. 2013, 50, 776–786. [Google Scholar] [CrossRef]
- Cappello, B.; Maio, C.; Iervolino, M.; Miro, A. Combined effect of hydroxypropyl methylcellulose and hydroxypropyl-β-cyclodextrin on physicochemical and dissolution properties of celecoxib. J. Incl. Phenom. Macrocycl. Chem. 2007, 59, 237–244. [Google Scholar] [CrossRef]
- dos Passos Menezes, P.; Serafini, M.R.; de Carvalho, Y.M.B.G.; Soares Santana, D.V.; Lima, B.S.; Quintans-Júnior, L.J.; Marreto, R.N.; de Aquino, T.M.; Sabino, A.R.; Scotti, L.; et al. Kinetic and physical-chemical study of the inclusion complex of β-cyclodextrin containing carvacrol. J. Mol. Struct. 2016, 1125, 323–330. [Google Scholar] [CrossRef]
- Shih, M.-F.; Chen, L.-Y.; Tsai, P.-J.; Cherng, J.-Y. In Vitro and in Vivo Therapeutics of β-Thujaplicin on Lps-Induced Inflammation in Macrophages and Septic Shock in Mice. Int. J. Immunopathol. Pharmacol. 2012, 25, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Mura, P. Analytical techniques for characterization of cyclodextrin complexes in the solid state: A review. J. Pharm. Biomed. Anal. 2015, 113, 226–238. [Google Scholar] [CrossRef] [PubMed]
- Frömming, K.-H.; Szejtli, J. Preparation and Characterization of Cyclodextrin Complexes; Springer: Dordrecht, The Netherlands, 1994; pp. 83–104. [Google Scholar]
- Yang, M.; He, S.; Fan, Y.; Wang, Y.; Ge, Z.; Shan, L.; Gong, W.; Huang, X.; Tong, Y.; Gao, C. Microenvironmental pH-modified solid dispersions to enhance the dissolution and bioavailability of poorly water-soluble weakly basic GT0918, a developing anti-prostate cancer drug: Preparation, characterization and evaluation in vivo. Int. J. Pharm. 2014, 475, 97–109. [Google Scholar] [CrossRef] [PubMed]
- Quintans, J.S.S.; Pereira, E.W.M.; Carvalho, Y.M.B.G.; Menezes, P.P.; Serafini, M.R.; Batista, M.V.A.; Moreira, C.D.L.F.A.; Lima, Á.A.N.; Branco, A.; Almeida, J.R.G.S.; et al. Host–guest inclusion complexation of β-cyclodextrin and hecogenin acetate to enhance anti-hyperalgesic effect in an animal model of musculoskeletal pain. Process. Biochem. 2017, 59, 123–131. [Google Scholar] [CrossRef]
- Fernandes, C.M.; Veiga, F.J.B. Effect of the hydrophobic nature of triacetyl-beta-cyclodextrin on the complexation with nicardipine hydrochloride: Physicochemical and dissolution properties of the kneaded and spray-dried complexes. Chem. Pharm. Bull. 2002, 50, 1597–1602. [Google Scholar] [CrossRef] [PubMed]
- Periasamy, R.; Kothainayaki, S.; Sivakumar, K. Preparation, physicochemical analysis and molecular modeling investigation of 2,2′-Bipyridine: β-Cyclodextrin inclusion complex in solution and solid state. J. Mol. Struct. 2015, 1100, 59–69. [Google Scholar] [CrossRef]
- Zhu, J.; Bailly, A.; Zwiewka, M.; Sovero, V.; di Donato, M.; Ge, P.; Oehri, J.; Aryal, B.; Hao, P.; Linnert, M.; et al. TWISTED DWARF1 mediates the action of auxin transport inhibitors on actin cytoskeleton dynamics. Plant Cell 2016, 28, 930–948. [Google Scholar] [CrossRef]
- Rajendiran, N.; Thulasidhasan, J.; Saravanan, J. Inclusion complexation of isoprenaline and methyl dopa with α- and β-cyclodextrin nanocavities: Spectral and theoretical study. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2014, 122, 411–421. [Google Scholar] [CrossRef]
- Spamer, E.; Müller, D.G.; Wessels, P.L.; Venter, J.P. Characterization of the complexes of furosemide with 2-hydroxypropyl-beta-cyclodextrin and sulfobutyl ether-7-beta-cyclodextrin. Eur. J. Pharm. Sci. 2002, 16, 247–253. [Google Scholar] [CrossRef]
- Tang, P.; Tang, B.; Wang, Q.; Xu, K.; Xiong, X.; Li, H. Effect of hydroxypropyl-β-cyclodextrin on the bounding of salazosulfapyridine to human serum albumin. Int. J. Biol. Macromol. 2016, 92, 105–115. [Google Scholar] [CrossRef] [PubMed]
- van Ekeren, P.J.; van Genderen, A.C.G.; van den Berg, G.J.K. Redetermination of the thermodynamic properties of the solid–solid transition of adamantane by adiabatic calorimetry to investigate the suitability as a reference material for low-temperature DSC-calibration. Thermochim. Acta 2006, 446, 33–35. [Google Scholar] [CrossRef]
- de Araujo, D.R.; Tsuneda, S.S.; Cereda, C.M.S.; Del, G.F.; Carvalho, F.; Preté, P.S.C.; Fernandes, S.A.; Yokaichiya, F.; Franco, M.K.K.D.; Mazzaro, I.; et al. Development and pharmacological evaluation of ropivacaine-2-hydroxypropyl-β-cyclodextrin inclusion complex. Eur. J. Pharm. Sci. 2008, 33, 60–71. [Google Scholar] [CrossRef] [PubMed]
- Rathee, P.; Chaudhary, H.; Rathee, S.; Rathee, D.; Kumar, V.; Kohli, K. Mechanism of action of flavonoids as anti-inflammatory agents: A review. Inflamm. Allergy Drug Targets 2009, 8, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Pascua-Maestro, R.; Corraliza-Gomez, M.; Diez-Hermano, S.; Perez-Segurado, C.; Ganfornina, M.D.; Sanchez, D. The MTT-formazan assay: Complementary technical approaches and in vivo validation in Drosophila larvae. Acta Histochem. 2018, 120, 179–186. [Google Scholar] [CrossRef] [PubMed]

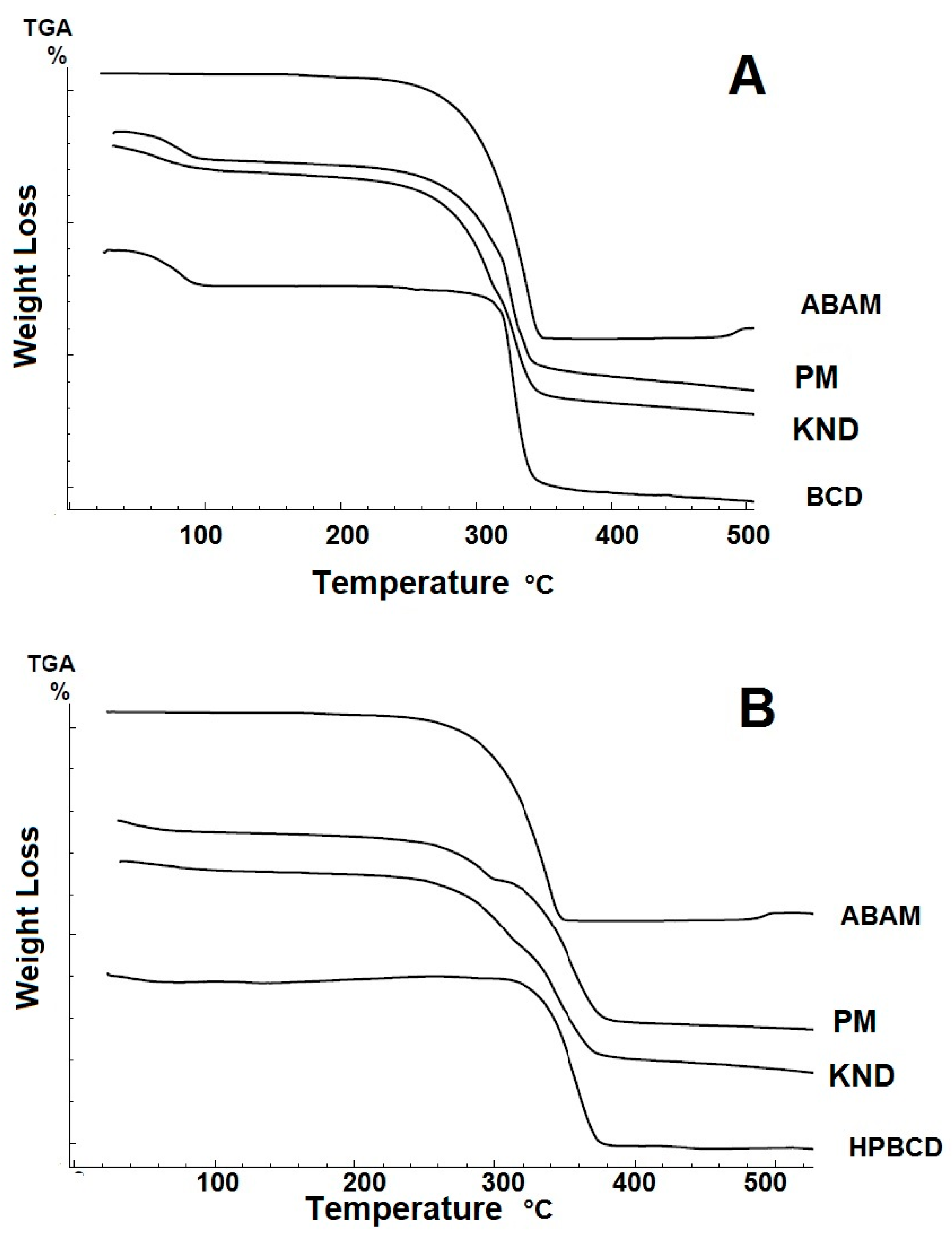
| Samples | ∆m1 (%) |
|---|---|
| 25–200 °C | |
| ABAM | 0.02 |
| βCD | 13.60 |
| PM (βCD) | 10.45 |
| KN (βCD) | 9.65 |
| HPβCD | 3.93 |
| PM (HPβCD) | 5.5 |
| KN (HPβCD) | 3.81 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
da Silva Júnior, W.F.; Bezerra de Menezes, D.L.; de Oliveira, L.C.; Koester, L.S.; Oliveira de Almeida, P.D.; Lima, E.S.; de Azevedo, E.P.; da Veiga Júnior, V.F.; Neves de Lima, Á.A. Inclusion Complexes of β and HPβ-Cyclodextrin with α, β Amyrin and In Vitro Anti-Inflammatory Activity. Biomolecules 2019, 9, 241. https://doi.org/10.3390/biom9060241
da Silva Júnior WF, Bezerra de Menezes DL, de Oliveira LC, Koester LS, Oliveira de Almeida PD, Lima ES, de Azevedo EP, da Veiga Júnior VF, Neves de Lima ÁA. Inclusion Complexes of β and HPβ-Cyclodextrin with α, β Amyrin and In Vitro Anti-Inflammatory Activity. Biomolecules. 2019; 9(6):241. https://doi.org/10.3390/biom9060241
Chicago/Turabian Styleda Silva Júnior, Walter Ferreira, Danielle Lima Bezerra de Menezes, Luana Carvalho de Oliveira, Letícia Scherer Koester, Patrícia Danielle Oliveira de Almeida, Emerson Silva Lima, Eduardo Pereira de Azevedo, Valdir Florêncio da Veiga Júnior, and Ádley Antonini Neves de Lima. 2019. "Inclusion Complexes of β and HPβ-Cyclodextrin with α, β Amyrin and In Vitro Anti-Inflammatory Activity" Biomolecules 9, no. 6: 241. https://doi.org/10.3390/biom9060241
APA Styleda Silva Júnior, W. F., Bezerra de Menezes, D. L., de Oliveira, L. C., Koester, L. S., Oliveira de Almeida, P. D., Lima, E. S., de Azevedo, E. P., da Veiga Júnior, V. F., & Neves de Lima, Á. A. (2019). Inclusion Complexes of β and HPβ-Cyclodextrin with α, β Amyrin and In Vitro Anti-Inflammatory Activity. Biomolecules, 9(6), 241. https://doi.org/10.3390/biom9060241








