In-depth Analysis of the Lid Subunits Assembly Mechanism in Mammals
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Culture and DNA Constructs
2.2. RNA Interference
2.3. Protein Extraction, Immunological Analysis, and Antibodies
2.4. Immunoprecipitation and Mass Spectrometry
2.5. In Vitro Transcription and Translation
3. Results
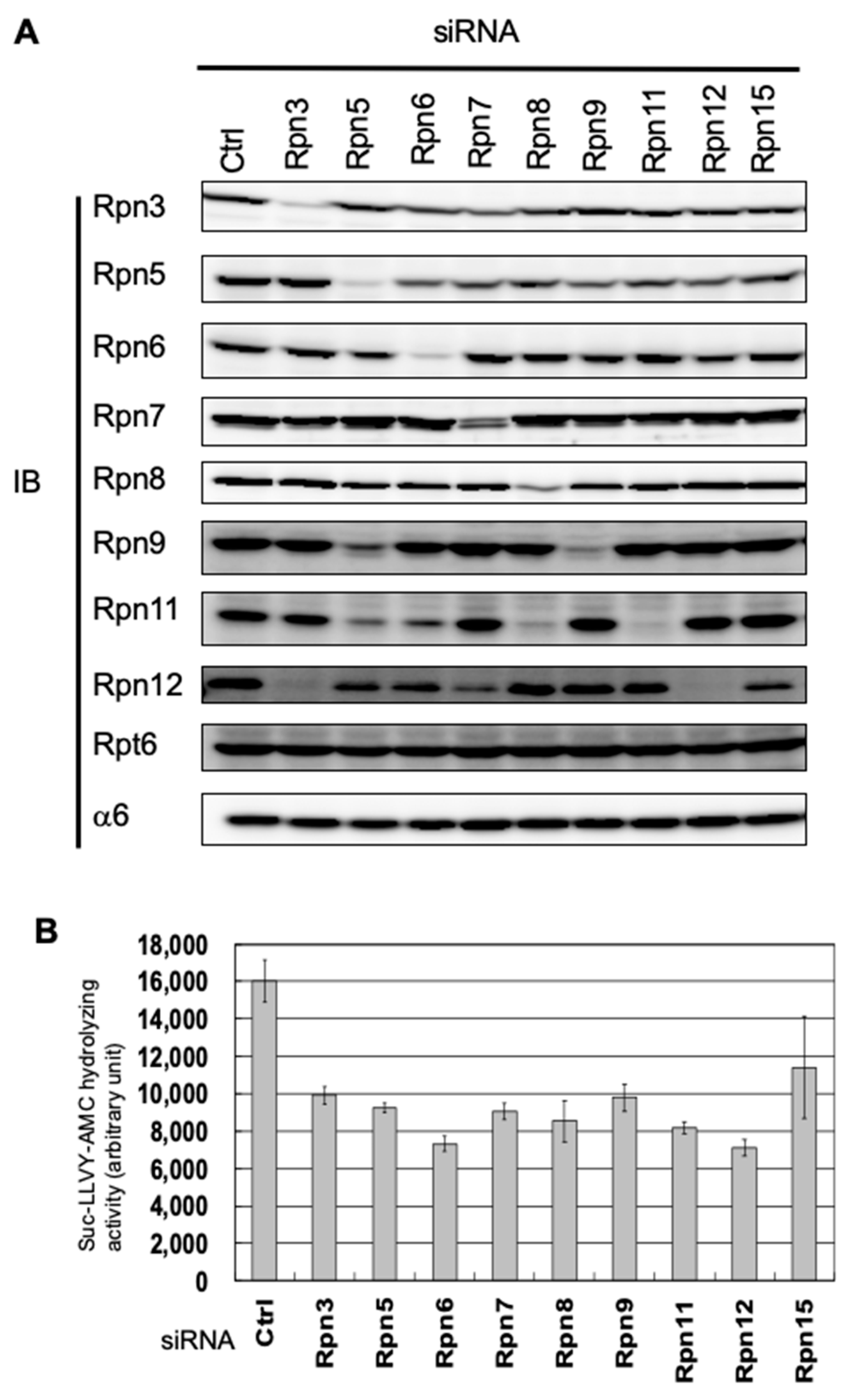
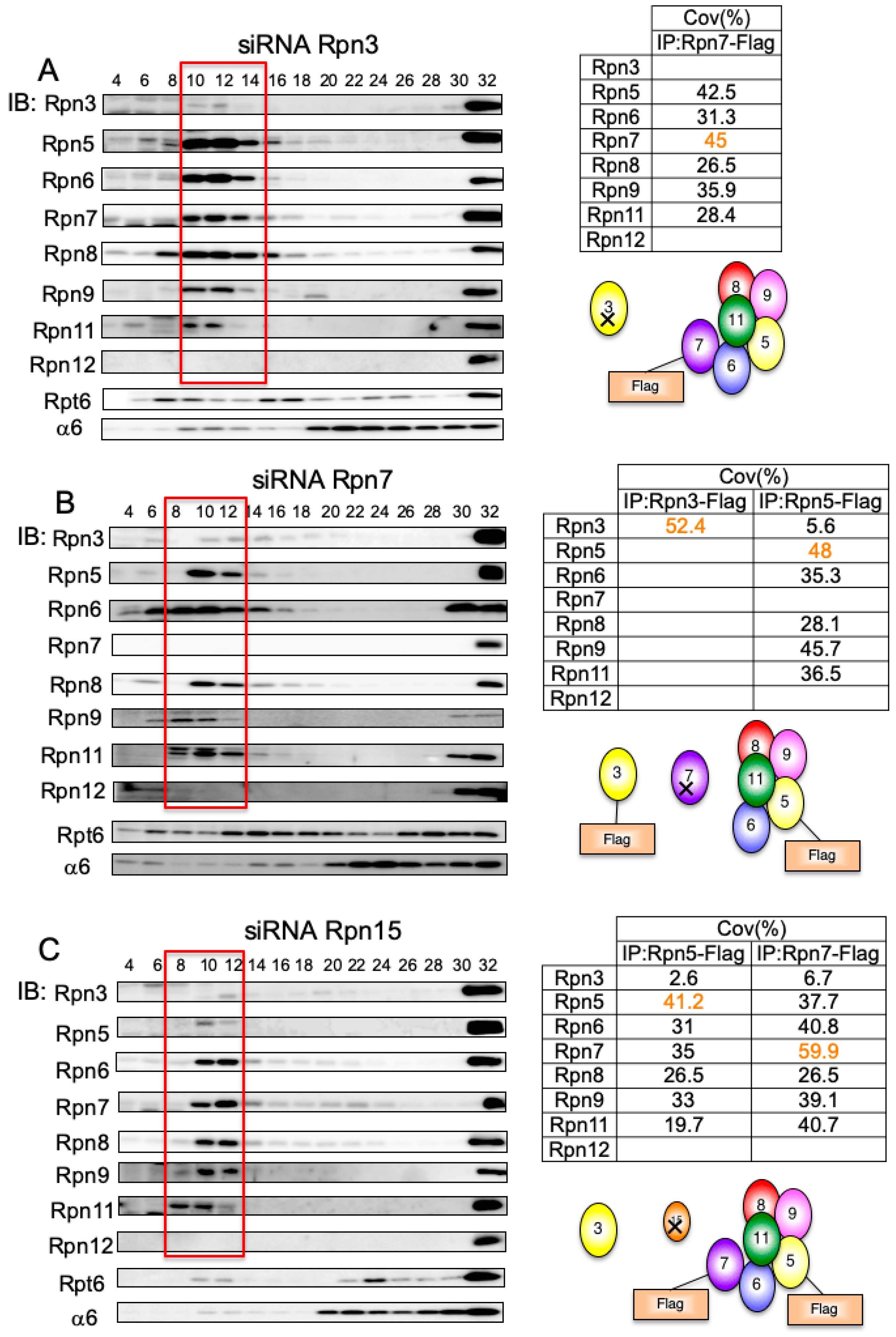
3.1. Interdependence Between the Lid Subunits for Protein Expression
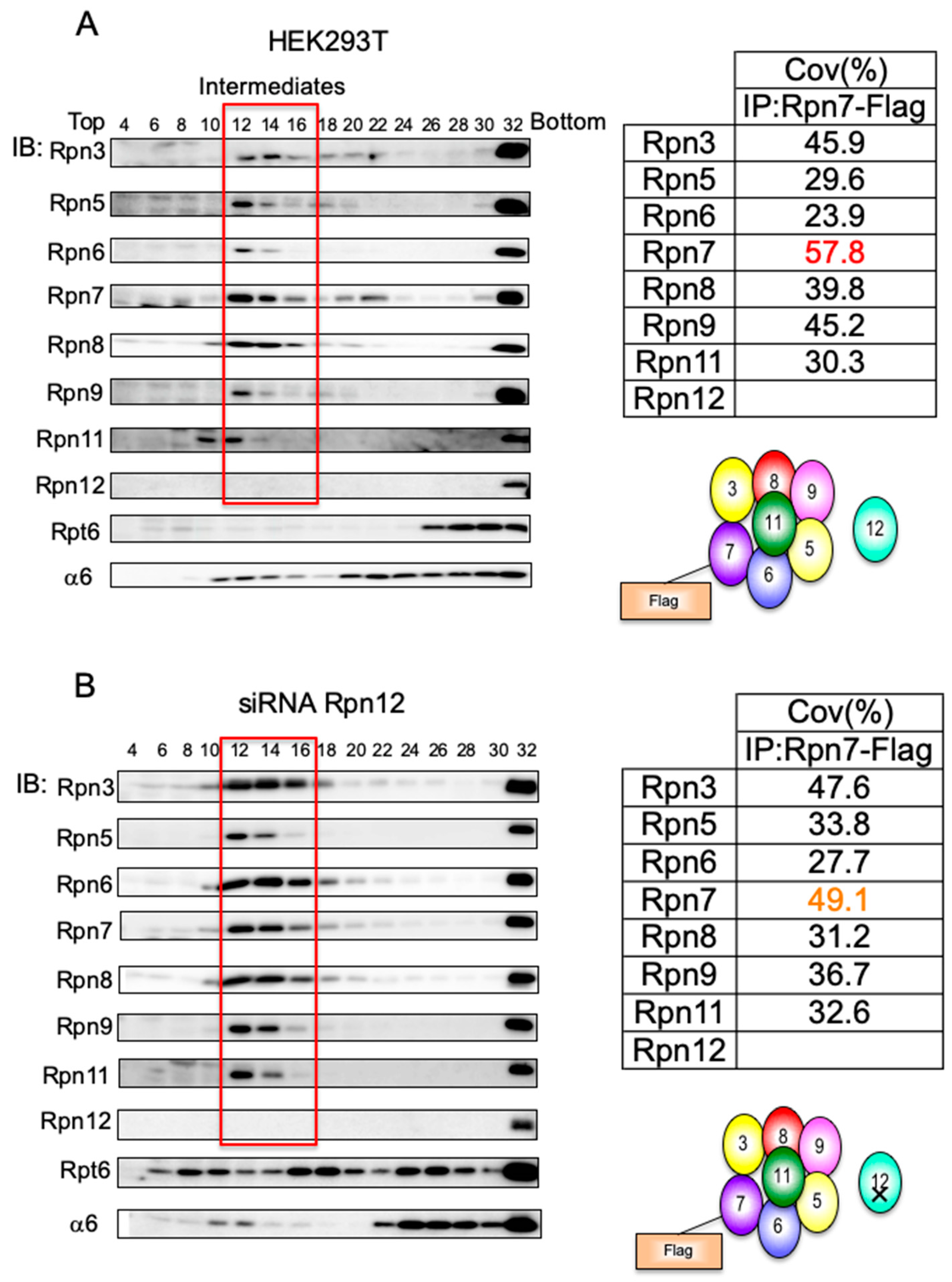
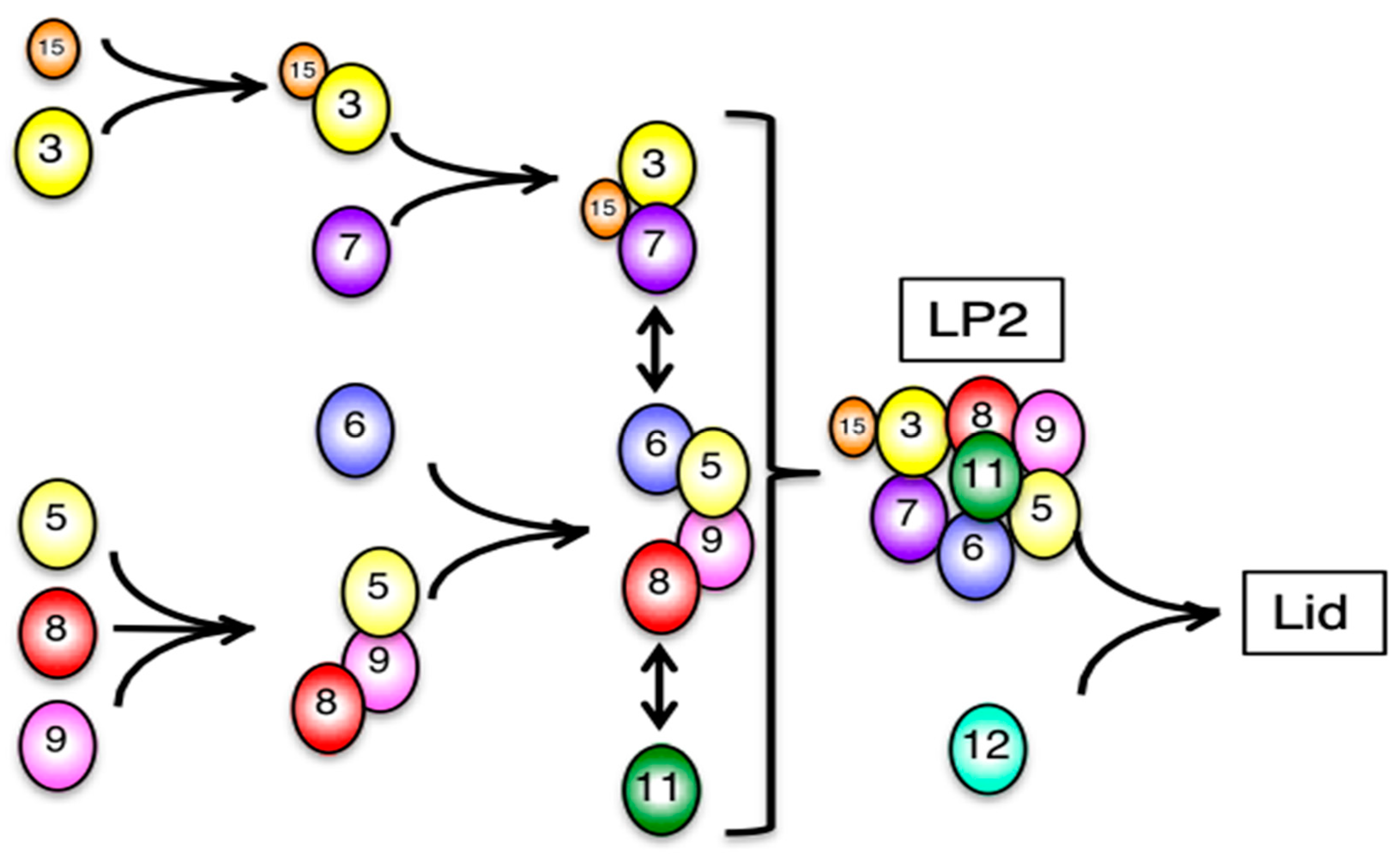
3.2. Mammalian Cells Express a Lid-like Complex Without Rpn12 (LP2)
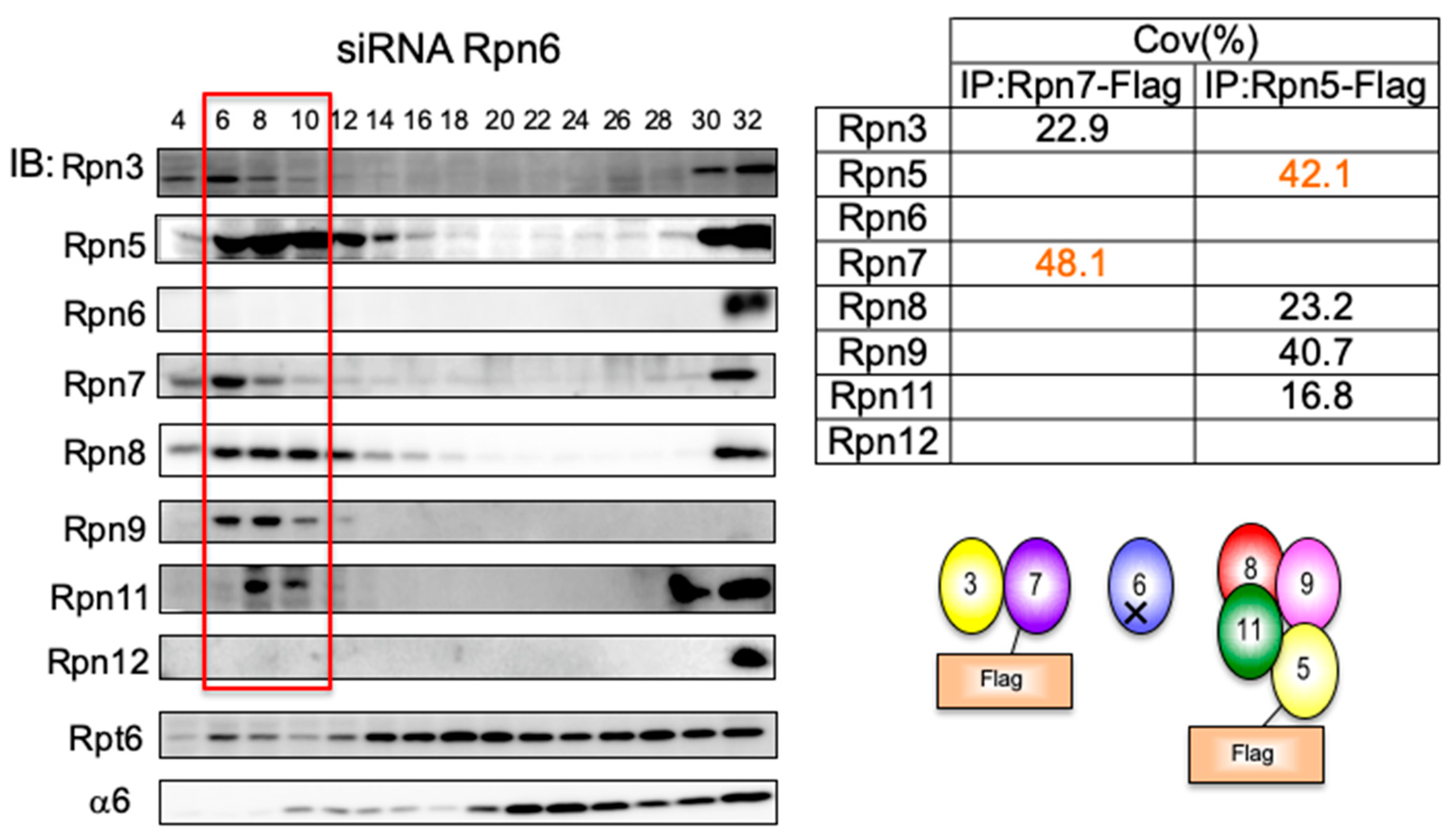
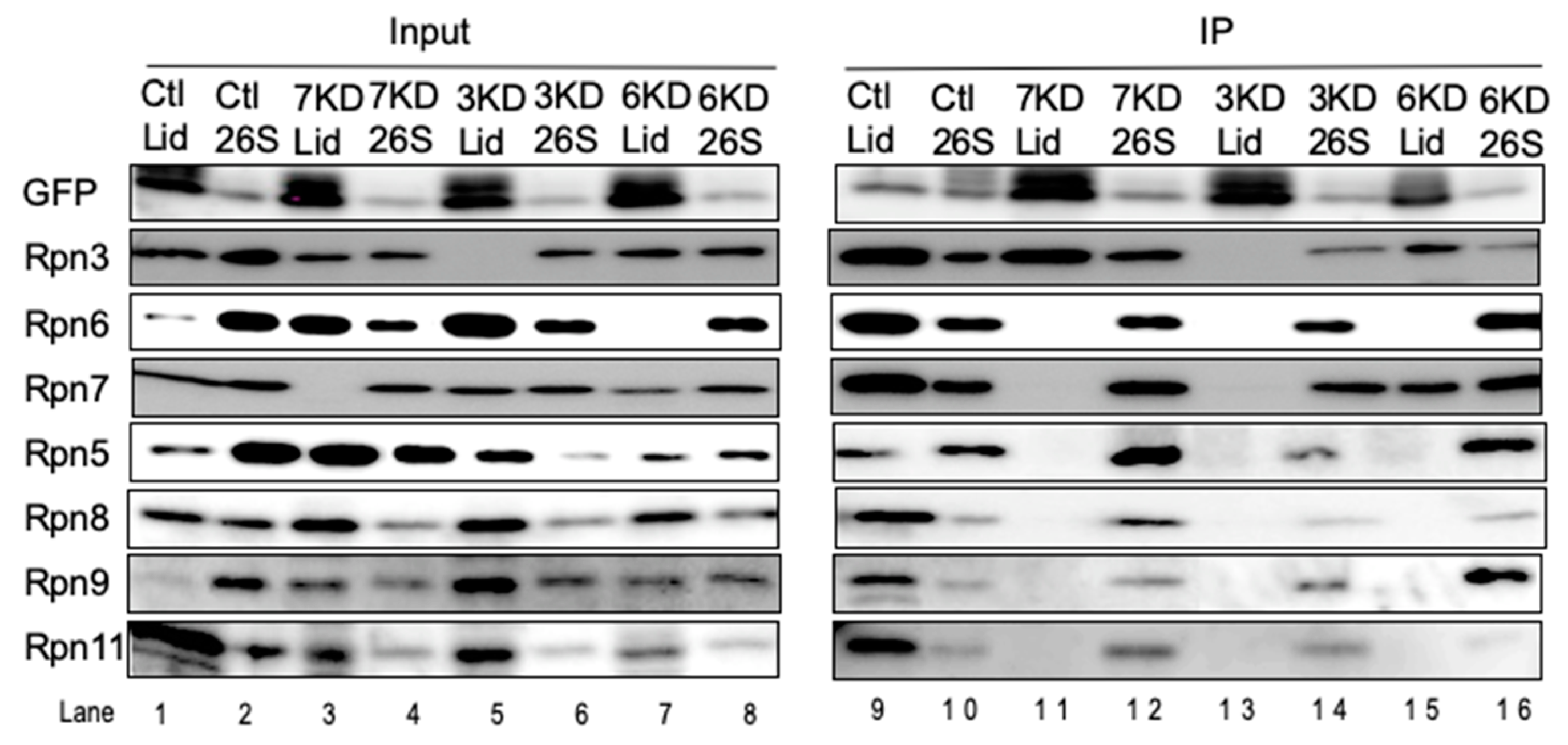
3.3. Rpn6 is Required for Interaction Between Rpn3-7-15 and Rpn5-8-9-11 and for Rpn11 Stability
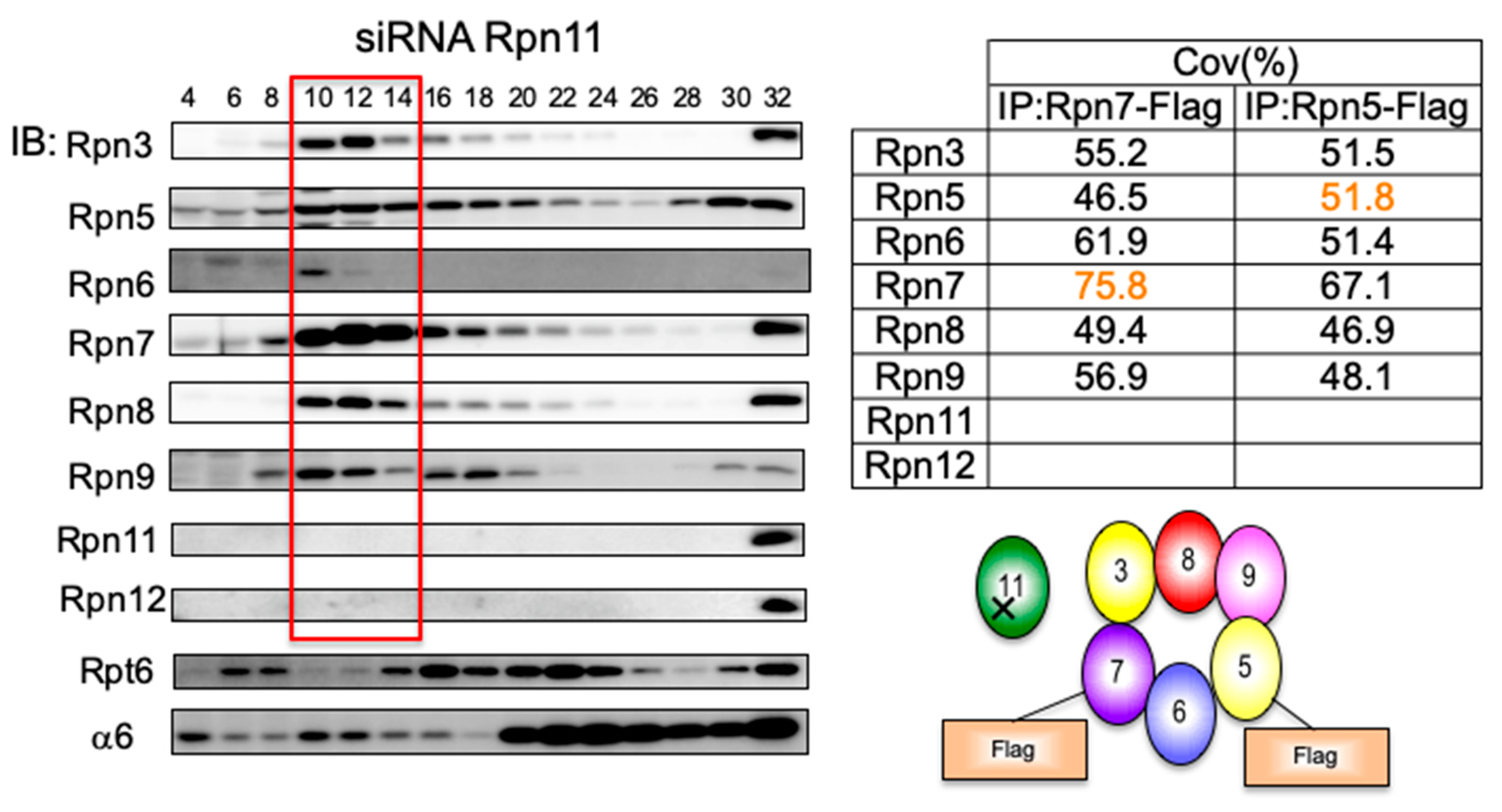
3.4. Loss of Rpn11 does not Affect the Assembly of Other Lid Subunits but Affects Lid–base Joining
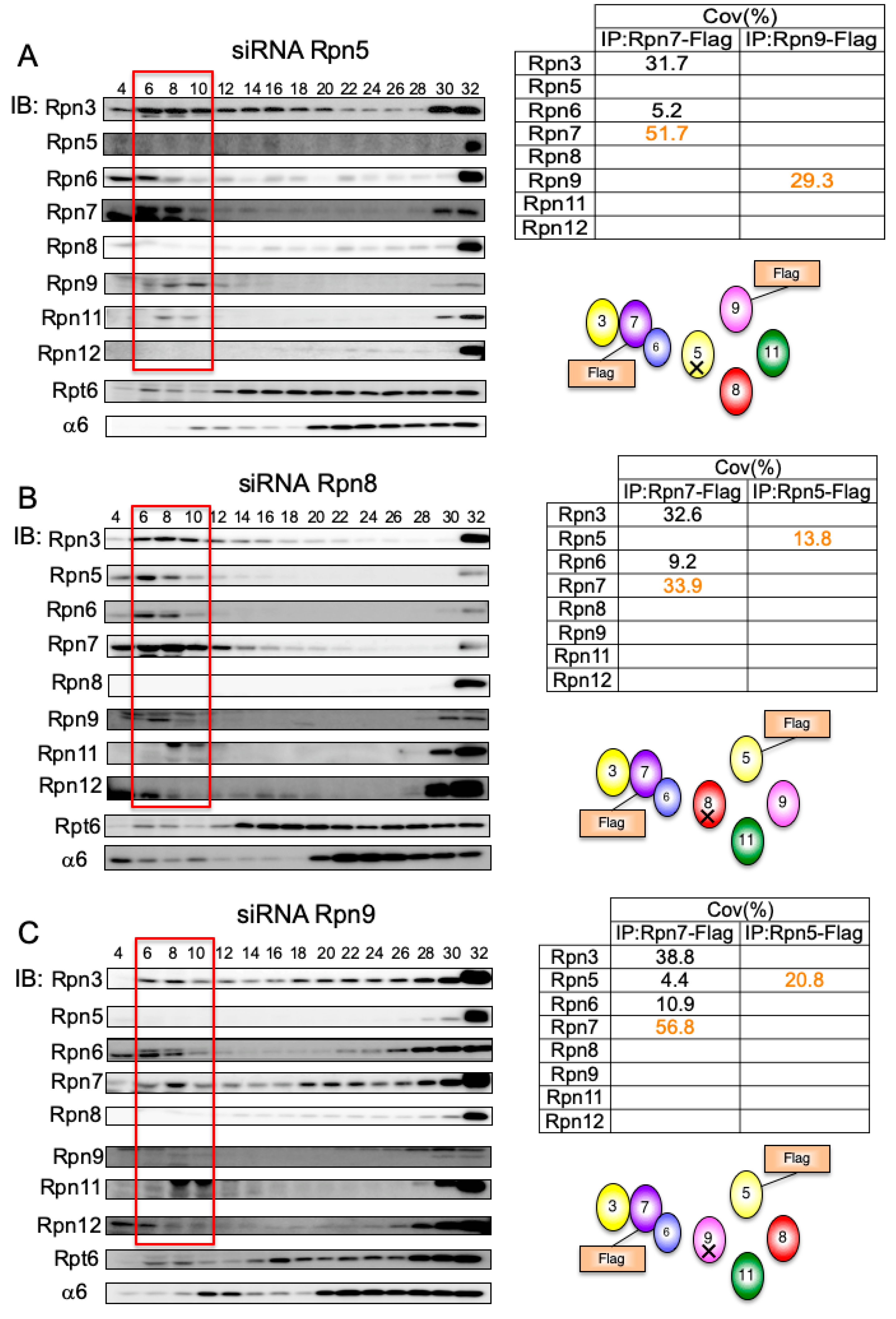
3.5. Interdependent Assembly of the Rpn5-8-9 Complex
3.6. Rpn7–Rpn6 Interaction Connects the Rpn3-7-15 Module with the Rpn5-6-8-9-11 Module
3.7. Rpn15 Mediates the Association of Rpn3 with Rpn7
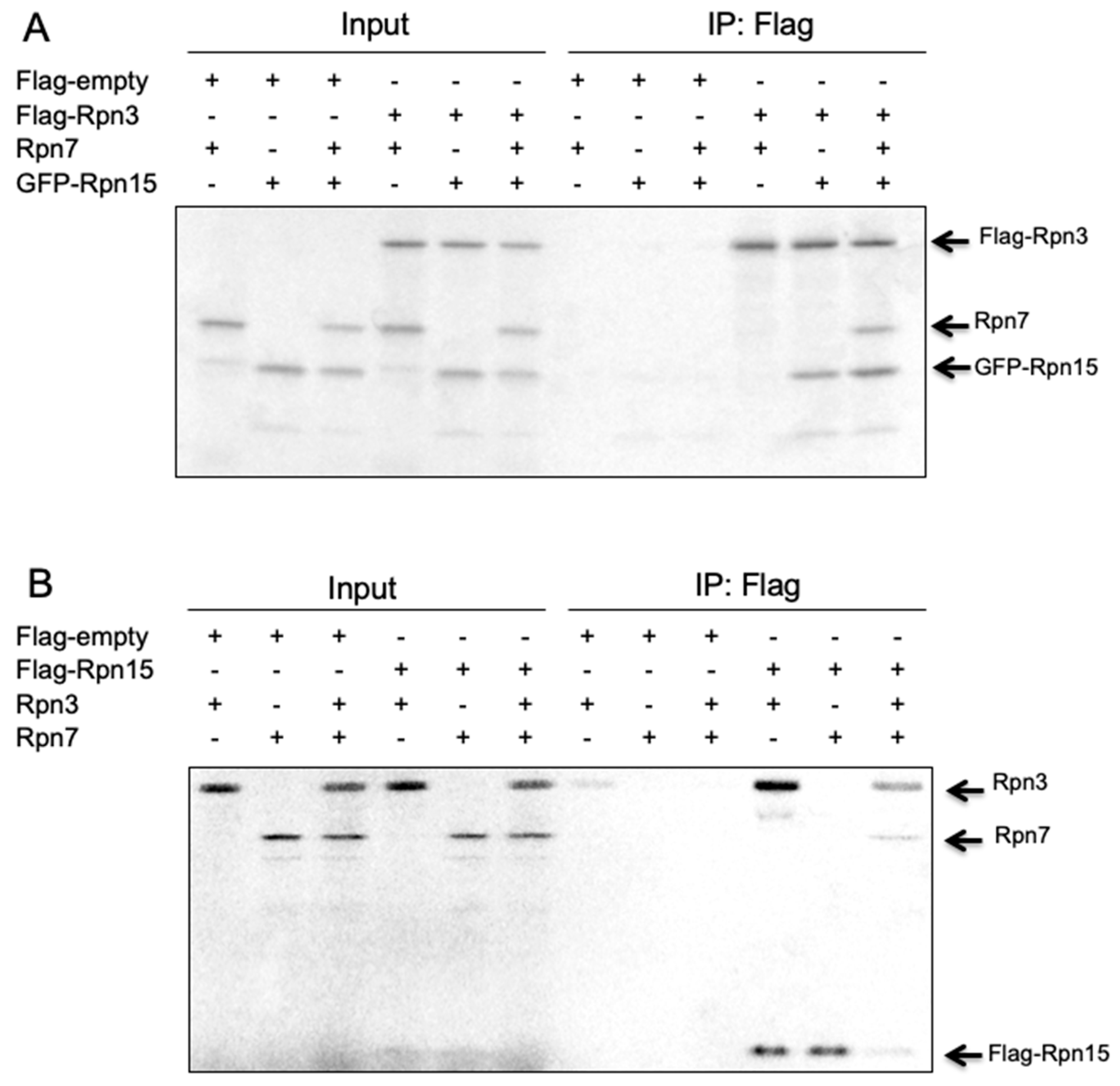
3.8. Rpn15 Directly Binds to Rpn3 and Promotes Rpn3–Rpn7 Association in Vitro
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Baumeister, W.; Walz, J.; Zuhl, F.; Seemuller, E. The proteasome: Paradigm of a self-compartmentalizing protease. Cell 1998, 92, 367–380. [Google Scholar] [CrossRef]
- Hershko, A.; Ciechanover, A. The ubiquitin system. Annu. Rev. Biochem. 1998, 67, 425–479. [Google Scholar] [CrossRef] [PubMed]
- Glickman, M.H.; Ciechanover, A. The ubiquitin-proteasome proteolytic pathway: Destruction for the sake of construction. Physiol. Rev. 2002, 82, 373–428. [Google Scholar] [CrossRef] [PubMed]
- Kohler, A.; Cascio, P.; Leggett, D.S.; Woo, K.M.; Goldberg, A.L.; Finley, D. The axial channel of the proteasome core particle is gated by the Rpt2 ATPase and controls both substrate entry and product release. Mol. Cell 2001, 7, 1143–1152. [Google Scholar] [CrossRef]
- Saeki, Y.; Sone, T.; Toh-e, A.; Yokosawa, H. Identification of ubiquitin-like protein-binding subunits of the 26S proteasome. Biochem. Biophys. Res. Commun. 2002, 296, 813–819. [Google Scholar] [CrossRef]
- Deveraux, Q.; Ustrell, V.; Pickart, C.; Rechsteiner, M. A 26 S protease subunit that binds ubiquitin conjugates. J. Biol. Chem. 1994, 269, 7059–7061. [Google Scholar]
- Husnjak, K.; Elsasser, S.; Zhang, N.; Chen, X.; Randles, L.; Shi, Y.; Hofmann, K.; Walters, K.J.; Finley, D.; Dikic, I. Proteasome subunit Rpn13 is a novel ubiquitin receptor. Nature 2008, 453, 481–488. [Google Scholar] [CrossRef]
- Rosenzweig, R.; Bronner, V.; Zhang, D.; Fushman, D.; Glickman, M.H. Rpn1 and Rpn2 coordinate ubiquitin processing factors at proteasome. J. Biol. Chem. 2012, 287, 14659–14671. [Google Scholar] [CrossRef]
- Hirano, Y.; Hendil, K.B.; Yashiroda, H.; Iemura, S.; Nagane, R.; Hioki, Y.; Natsume, T.; Tanaka, K.; Murata, S. A heterodimeric complex that promotes the assembly of mammalian 20S proteasomes. Nature 2005, 437, 1381–1385. [Google Scholar] [CrossRef]
- Hirano, Y.; Hayashi, H.; Iemura, S.; Hendil, K.B.; Niwa, S.; Kishimoto, T.; Kasahara, M.; Natsume, T.; Tanaka, K.; Murata, S. Cooperation of multiple chaperones required for the assembly of mammalian 20S proteasomes. Mol. Cell 2006, 24, 977–984. [Google Scholar] [CrossRef]
- Murata, S. Multiple chaperone-assisted formation of mammalian 20S proteasomes. IUBMB Life 2006, 58, 344–348. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Kusmierczyk, A.R.; Wong, P.; Emili, A.; Hochstrasser, M. beta-Subunit appendages promote 20S proteasome assembly by overcoming an Ump1-dependent checkpoint. EMBO J. 2007, 26, 2339–2349. [Google Scholar] [CrossRef] [PubMed]
- Le Tallec, B.; Barrault, M.B.; Courbeyrette, R.; Guerois, R.; Marsolier-Kergoat, M.C.; Peyroche, A. 20S proteasome assembly is orchestrated by two distinct pairs of chaperones in yeast and in mammals. Mol. Cell 2007, 27, 660–674. [Google Scholar] [CrossRef] [PubMed]
- Yashiroda, H.; Mizushima, T.; Okamoto, K.; Kameyama, T.; Hayashi, H.; Kishimoto, T.; Niwa, S.; Kasahara, M.; Kurimoto, E.; Sakata, E.; et al. Crystal structure of a chaperone complex that contributes to the assembly of yeast 20S proteasomes. Nat. Struct. Mol. Biol. 2008, 15, 228–236. [Google Scholar] [CrossRef] [PubMed]
- Kusmierczyk, A.R.; Kunjappu, M.J.; Funakoshi, M.; Hochstrasser, M. A multimeric assembly factor controls the formation of alternative 20S proteasomes. Nat. Struct. Mol. Biol. 2008, 15, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Sahara, K.; Hirayama, S.; Zhao, X.; Watanabe, A.; Hamazaki, J.; Yashiroda, H.; Murata, S. PAC1-PAC2 proteasome assembly chaperone retains the core alpha4-alpha7 assembly intermediates in the cytoplasm. Genes Cells 2018, 23, 839–848. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, T.; Hamazaki, J.; Iemura, S.; Sasaki, K.; Furuyama, K.; Natsume, T.; Tanaka, K.; Murata, S. Assembly pathway of the Mammalian proteasome base subcomplex is mediated by multiple specific chaperones. Cell 2009, 137, 914–925. [Google Scholar] [CrossRef]
- Funakoshi, M.; Tomko, R.J., Jr.; Kobayashi, H.; Hochstrasser, M. Multiple assembly chaperones govern biogenesis of the proteasome regulatory particle base. Cell 2009, 137, 887–899. [Google Scholar] [CrossRef]
- Hirano, Y.; Kaneko, T.; Okamoto, K.; Bai, M.; Yashiroda, H.; Furuyama, K.; Kato, K.; Tanaka, K.; Murata, S. Dissecting beta-ring assembly pathway of the mammalian 20S proteasome. EMBO J. 2008, 27, 2204–2213. [Google Scholar] [CrossRef]
- Isono, E.; Nishihara, K.; Saeki, Y.; Yashiroda, H.; Kamata, N.; Ge, L.; Ueda, T.; Kikuchi, Y.; Tanaka, K.; Nakano, A.; et al. The assembly pathway of the 19S regulatory particle of the yeast 26S proteasome. Mol. Biol Cell 2007, 18, 569–580. [Google Scholar] [CrossRef]
- Isono, E.; Saeki, Y.; Yokosawa, H.; Toh-e, A. Rpn7 Is required for the structural integrity of the 26 S proteasome of Saccharomyces cerevisiae. J. Biol. Chem. 2004, 279, 27168–27176. [Google Scholar] [CrossRef] [PubMed]
- Isono, E.; Saito, N.; Kamata, N.; Saeki, Y.; Toh, E.A. Functional analysis of Rpn6p, a lid component of the 26 S proteasome, using temperature-sensitive rpn6 mutants of the yeast Saccharomyces cerevisiae. J. Biol. Chem. 2005, 280, 6537–6547. [Google Scholar] [CrossRef] [PubMed]
- Sharon, M.; Taverner, T.; Ambroggio, X.I.; Deshaies, R.J.; Robinson, C.V. Structural organization of the 19S proteasome lid: Insights from MS of intact complexes. PLoS Biol. 2006, 4, e267. [Google Scholar] [CrossRef] [PubMed]
- Tomko, R.J., Jr.; Hochstrasser, M. Incorporation of the Rpn12 subunit couples completion of proteasome regulatory particle lid assembly to lid-base joining. Mol. Cell 2011, 44, 907–917. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Luan, B.; Wu, J.; Shi, Y. An atomic structure of the human 26S proteasome. Nat. Struct. Mol. Biol. 2016, 23, 778–785. [Google Scholar] [CrossRef] [PubMed]
- Lander, G.C.; Estrin, E.; Matyskiela, M.E.; Bashore, C.; Nogales, E.; Martin, A. Complete subunit architecture of the proteasome regulatory particle. Nature 2012, 482, 186–191. [Google Scholar] [CrossRef]
- Schweitzer, A.; Aufderheide, A.; Rudack, T.; Beck, F.; Pfeifer, G.; Plitzko, J.M.; Sakata, E.; Schulten, K.; Forster, F.; Baumeister, W. Structure of the human 26S proteasome at a resolution of 3.9 A. Proc. Nat. Acad. Sci. USA 2016, 113, 7816–7821. [Google Scholar] [CrossRef]
- Murata, S.; Udono, H.; Tanahashi, N.; Hamada, N.; Watanabe, K.; Adachi, K.; Yamano, T.; Yui, K.; Kobayashi, N.; Kasahara, M.; et al. Immunoproteasome assembly and antigen presentation in mice lacking both PA28alpha and PA28beta. EMBO J. 2001, 20, 5898–5907. [Google Scholar] [CrossRef]
- Fukunaga, K.; Kudo, T.; Toh-e, A.; Tanaka, K.; Saeki, Y. Dissection of the assembly pathway of the proteasome lid in Saccharomyces cerevisiae. Biochem. Biophys. Res. Commun. 2010, 396, 1048–1053. [Google Scholar] [CrossRef]
- Budenholzer, L.; Cheng, C.L.; Li, Y.; Hochstrasser, M. Proteasome Structure and Assembly. J. Mol. Biol. 2017, 429, 3500–3524. [Google Scholar] [CrossRef]
- Wei, S.J.; Williams, J.G.; Dang, H.; Darden, T.A.; Betz, B.L.; Humble, M.M.; Chang, F.M.; Trempus, C.S.; Johnson, K.; Cannon, R.E.; et al. Identification of a specific motif of the DSS1 protein required for proteasome interaction and p53 protein degradation. J. Mol. Biol. 2008, 383, 693–712. [Google Scholar] [CrossRef] [PubMed]
- Tomko, R.J., Jr.; Hochstrasser, M. The intrinsically disordered Sem1 protein functions as a molecular tether during proteasome lid biogenesis. Mol. Cell 2014, 53, 433–443. [Google Scholar] [CrossRef] [PubMed]
- Bohn, S.; Sakata, E.; Beck, F.; Pathare, G.R.; Schnitger, J.; Nagy, I.; Baumeister, W.; Forster, F. Localization of the regulatory particle subunit Sem1 in the 26S proteasome. Biochem. Biophys. Res. Commun. 2013, 435, 250–254. [Google Scholar] [CrossRef] [PubMed]
- Vilchez, D.; Morantte, I.; Liu, Z.; Douglas, P.M.; Merkwirth, C.; Rodrigues, A.P.; Manning, G.; Dillin, A. RPN-6 determines C. elegans longevity under proteotoxic stress conditions. Nature 2012, 489, 263–268. [Google Scholar] [CrossRef] [PubMed]
- Vilchez, D.; Boyer, L.; Morantte, I.; Lutz, M.; Merkwirth, C.; Joyce, D.; Spencer, B.; Page, L.; Masliah, E.; Berggren, W.T.; et al. Increased proteasome activity in human embryonic stem cells is regulated by PSMD11. Nature 2012, 489, 304–308. [Google Scholar] [CrossRef] [PubMed]
- Tsolou, A.; Nelson, G.; Trachana, V.; Chondrogianni, N.; Saretzki, G.; von Zglinicki, T.; Gonos, E.S. The 19S proteasome subunit Rpn7 stabilizes DNA damage foci upon genotoxic insult. IUBMB life 2012, 64, 432–442. [Google Scholar] [CrossRef] [PubMed]
- Butler, L.R.; Densham, R.M.; Jia, J.; Garvin, A.J.; Stone, H.R.; Shah, V.; Weekes, D.; Festy, F.; Beesley, J.; Morris, J.R. The proteasomal de-ubiquitinating enzyme POH1 promotes the double-strand DNA break response. EMBO J. 2012, 31, 3918–3934. [Google Scholar] [CrossRef]
| Name | Sequence | Supplier |
|---|---|---|
| Human Rpn3 | 5′-UGUCCUGACAGCUUGAGUCAGAAGG-3′ | Invitrogen |
| Human Rpn5 | 5′-UUCCAUAGUCCUCAACAAGUGUGGA-3′ | Invitrogen |
| Human Rpn6 | 5′-UAGUAAGUUAUCAUACAACUUGGCC-3′ | Invitrogen |
| Human Rpn7 | 5′-UAGUAAGGAGCCAUGUUGUUAUCGC-3′ | Invitrogen |
| Human Rpn8 | 5′-UGUGGUACCAGCCAACUAUUCUUUC-3′ | Invitrogen |
| Human Rpn9 | 5′-AAUUGUUUCCUUUGUAACCUGUAGG-3′ | Invitrogen |
| Human Rpn11 | 5′-AUACCAACCAACAACCAUCUCCGGC-3′ | Invitrogen |
| Human Rpn12 | 5′-UAUGUCACGGGCCAGAAUUAGCUGC-3′ | Greiner bio-one |
| Human Rpn15 | 5′-UAACAGACCUAAGUCUACCGGCUGC-3′ | Greiner bio-one |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bai, M.; Zhao, X.; Sahara, K.; Ohte, Y.; Hirano, Y.; Kaneko, T.; Yashiroda, H.; Murata, S. In-depth Analysis of the Lid Subunits Assembly Mechanism in Mammals. Biomolecules 2019, 9, 213. https://doi.org/10.3390/biom9060213
Bai M, Zhao X, Sahara K, Ohte Y, Hirano Y, Kaneko T, Yashiroda H, Murata S. In-depth Analysis of the Lid Subunits Assembly Mechanism in Mammals. Biomolecules. 2019; 9(6):213. https://doi.org/10.3390/biom9060213
Chicago/Turabian StyleBai, Minghui, Xian Zhao, Kazutaka Sahara, Yuki Ohte, Yuko Hirano, Takeumi Kaneko, Hideki Yashiroda, and Shigeo Murata. 2019. "In-depth Analysis of the Lid Subunits Assembly Mechanism in Mammals" Biomolecules 9, no. 6: 213. https://doi.org/10.3390/biom9060213
APA StyleBai, M., Zhao, X., Sahara, K., Ohte, Y., Hirano, Y., Kaneko, T., Yashiroda, H., & Murata, S. (2019). In-depth Analysis of the Lid Subunits Assembly Mechanism in Mammals. Biomolecules, 9(6), 213. https://doi.org/10.3390/biom9060213






