Testosterone in Males as Enhanced by Onion (Allium Cepa L.)
Abstract
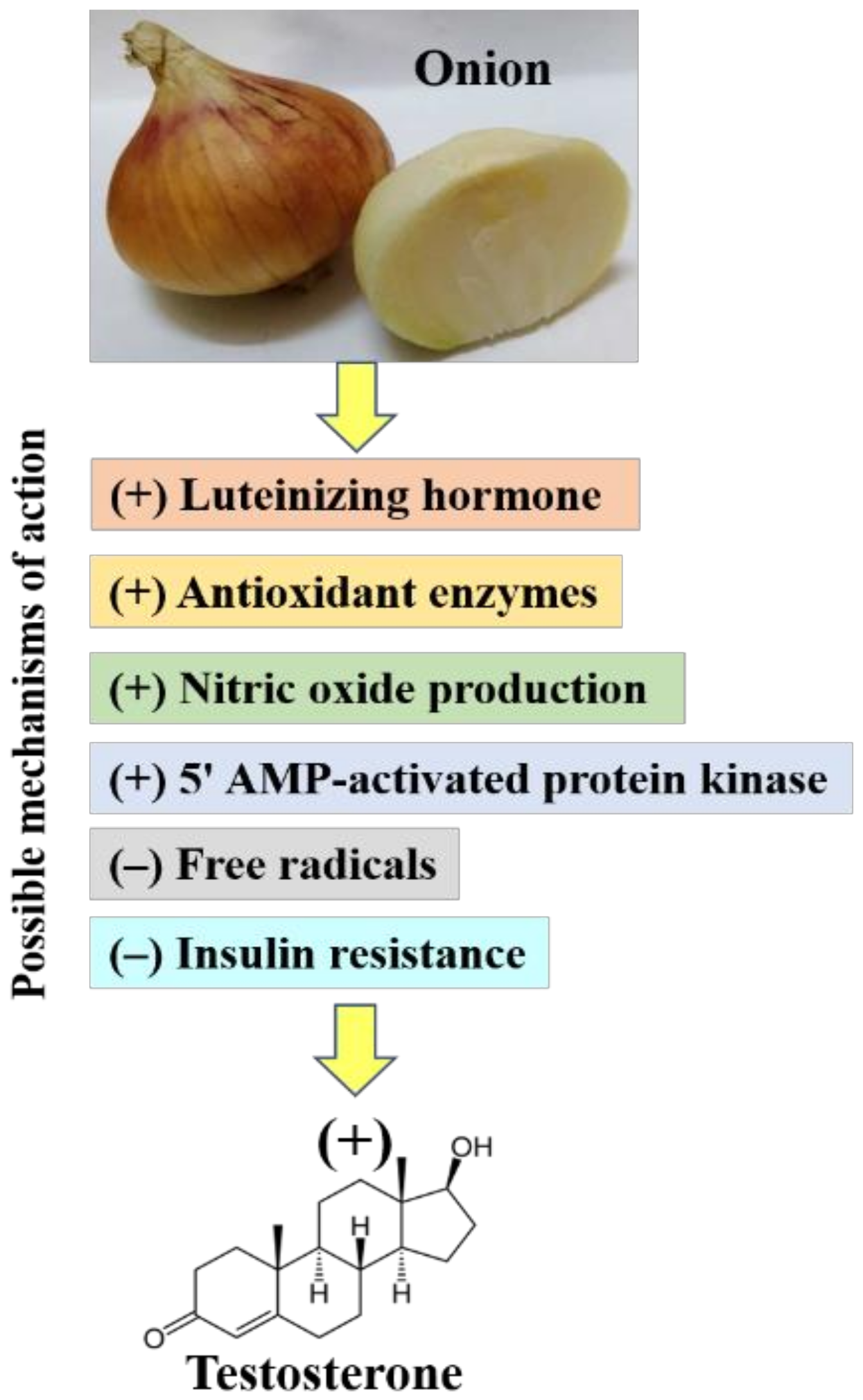
:1. Introduction
2. Effect of Onions on Testosterone
3. Mechanistic Studies
3.1. Effect of Onion on Luteinizing Hormone
3.2. Effect of Onion as a Potential Antioxidant on Testosterone
3.3. Effect of Onion as an Antihyperglycemic Affecter on Testosterone
3.4. Effect of Onion as a Nitric Oxide Stimulator on Testosterone
3.5. Effect of Onion on 5′ AMP-Activated Protein Kinase
4. Conclusions and Future Perspectives
Acknowledgments
Conflicts of Interest
References
- Kelly, D.M.; Jones, T.H. Testosterone: A metabolic hormone in health and disease. J. Endocrinol. 2013, 217, R25–R45. [Google Scholar] [CrossRef]
- Mooradian, A.D.; Morley, J.E.; Korenman, S.G. Biological actions of androgens. Endocr. Rev. 1987, 8, 1–28. [Google Scholar] [CrossRef] [PubMed]
- Karakas, M.; Schafer, S.; Appelbaum, S.; Ojeda, F.; Kuulasmaa, K.; Bruckmann, B.; Berisha, F.; Schulte-Steinberg, B.; Jousilahti, P.; Blankenberg, S.; et al. Testosterone levels and type 2 diabetes-no correlation with age, differential predictive value in men and women. Biomolecules 2018, 8, 76. [Google Scholar] [CrossRef] [PubMed]
- El-Migdadi, F.; Banihani, I.; Banihani, S.A. Clinico-hormonal correlation of oligospermic patients in the below sea level environment (Jordan Valley). Neuroendocrinol. Endocrinol. Lett. 2005, 26, 13–18. [Google Scholar]
- Sonmez, M.; Turk, G.; Yuce, A. The effect of ascorbic acid supplementation on sperm quality, lipid peroxidation and testosterone levels of male wistar rats. Theriogenology 2005, 63, 2063–2072. [Google Scholar] [CrossRef] [PubMed]
- Du Plessis, S.S.; Cabler, S.; McAlister, D.A.; Sabanegh, E.; Agarwal, A. The effect of obesity on sperm disorders and male infertility. Nat. Rev. Urol. 2010, 7, 153–161. [Google Scholar] [CrossRef] [PubMed]
- Lv, W.; Du, N.; Liu, Y.; Fan, X.; Wang, Y.; Jia, X.; Hou, X.; Wang, B. Low testosterone level and risk of Alzheimer’s disease in the elderly men: A systematic review and meta-analysis. Mol. Neurobiol. 2016, 53, 2679–2684. [Google Scholar] [CrossRef]
- Tuck, S.P.; Francis, R.M. Testosterone, bone and osteoporosis. Front. Horm. Res. 2009, 37, 123–132. [Google Scholar] [PubMed]
- McHenry, J.; Carrier, N.; Hull, E.; Kabbaj, M. Sex differences in anxiety and depression: Role of testosterone. Front. Neuroendocr. 2014, 35, 42–57. [Google Scholar] [CrossRef] [PubMed]
- Banihani, S.A. Effect of coenzyme q10 supplementation on testosterone. Biomolecules 2018, 8, 172. [Google Scholar] [CrossRef]
- Kloner, R.A.; Carson, C., 3rd.; Dobs, A.; Kopecky, S.; Mohler, E.R., 3rd. Testosterone and cardiovascular disease. J. Am. Coll. Cardiol. 2016, 67, 545–557. [Google Scholar] [CrossRef]
- Brewster, J. Onions and Other Vegetable Alliums; CABI: Wallingford, UK, 2008. [Google Scholar]
- US National Onion Association. History of Onion; US National Onion Association: Greeley, CO, USA, 2017. [Google Scholar]
- Tedesco, I.; Carbone, V.; Spagnuolo, C.; Minasi, P.; Russo, G.L. Identification and quantification of flavonoids from two southern Italian cultivars of Allium cepa L., tropea (red onion) and montoro (copper onion), and their capacity to protect human erythrocytes from oxidative stress. J. Agric. Food Chem. 2015, 63, 5229–5238. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.S.; Choi, H.; Loots du, T. Effects of dietary onion (Allium cepa L.) in a high-fat diet streptozotocin-induced diabetes rodent model. Ann. Nutr. Metab. 2008, 53, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Willcox, D.C.; Willcox, B.J.; Todoriki, H.; Suzuki, M. The Okinawan diet: Health implications of a low-calorie, nutrient-dense, antioxidant-rich dietary pattern low in glycemic load. J. Am. Coll. Nutr. 2009, 28, 500S–516S. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, Y.; Tanaka, K.; Hiramoto, S.; Yahata, N.; Inagawa, Y.; Nukui, K.; Hori, S. Alleviation of the aging males’ symptoms by the intake of onion-extracts containing concentrated cysteine sulfoxides for 4 weeks—Randomized, double-blind, placebo-controlled, parallel-group comparative study. Jpn. Pharmacol. Ther. 2017, 45, 595–608. [Google Scholar]
- Kumar, K.S.; Debjit, B.; Chiranjib, B.; Pankaj, T. Allium cepa: A traditional medicinal herb and its health benefits. J. Chem. Pharm. Res. 2010, 2, 283–291. [Google Scholar]
- Petering, R.C.; Brooks, N.A. Testosterone therapy: Review of clinical applications. Am. Fam. Physician 2017, 96, 441–449. [Google Scholar]
- Sharaf, A. Some hormone-like properties of Allium cepa L. (onion). Qual. Plant. Mater. Veg. 1967, 14, 267–275. [Google Scholar] [CrossRef]
- Khaki, A.; Fathiazad, F.; Nouri, M.; Khaki, A.A.; Khamenehi, H.J.; Hamadeh, M. Evaluation of androgenic activity of Allium cepa on spermatogenesis in the rat. Folia Morphol. 2009, 68, 45–51. [Google Scholar]
- Gholamhosini, B.; Khaki, A.; Ahmadi-Ashtiani, H.R.; Rezazadeh, S.; Rastegar, H.; Fathiazad, F.; Ghanbari, M. Treatment effects of onion on spermatogenesis in streptozotocin-induced diabetic rat. J. Med. Plants 2009, 8, 153–161. [Google Scholar]
- Khaki, A.; Farnam, A.; Ahmadi-Ashtiani, H.R.; Rezazadeh, S.; Rastgar, H.; Eftekharzadeh, S.; Aghamohamadi, R.; Abiri, N. Treatment effect of onion on sexual behavior after induces an antiepileptic drug (lamotrigine) in male rat. J. Med. Plants 2010, 9, 49–57. [Google Scholar]
- Khaki, A.; Farnam, A.; Badie, A.D.; Nikniaz, H. Treatment effects of onion (Allium cepa) and ginger (Zingiber officinale) on sexual behavior of rat after inducing an antiepileptic drug (lamotrigine). Balk. Med. J. 2012, 29, 236–242. [Google Scholar] [CrossRef] [PubMed]
- Akhigbe, R.E.; Ige, S.F. The role of Allium cepa on aluminum-induced reproductive dysfunction in experimental male rat models. J. Hum. Reprod. Sci. 2012, 5, 200–205. [Google Scholar]
- Ghalehkandi, J.G.; Asghari, A.; Beheshti, R.; Valilu, M.; Yeghaneh, A. Effect of onion (Allium cepa. Linn) aqueous extract on serum concentration of lh, fsh and testosterone compared with zinc sulfate supplementation in the rats. J. Anim. Vet. Adv. 2012, 11, 3346–3349. [Google Scholar]
- Ige, S.F.; Akhigbe, R.E. Common onion (Allium cepa) extract reverses cadmium-induced organ toxicity and dyslipidaemia via redox alteration in rats. Pathophysiology 2013, 20, 269–274. [Google Scholar] [CrossRef]
- Allouh, M.Z.; Daradka, H.M.; Barbarawi, M.M.; Mustafa, A.G. Fresh onion juice enhanced copulatory behavior in male rats with and without paroxetine-induced sexual dysfunction. Exp. Boil. Med. 2014, 239, 177–182. [Google Scholar] [CrossRef]
- Shahverdi, S.; Khaki, A.; Jafari, B. Investigating the antioxidant effect of Allium cepa after exposure to escherichia coli on biochemical factors, the blood antioxidants, and testis tissue in rats. Crescent J. Med. Boil. Sci. 2017, 4, 32–38. [Google Scholar]
- Khaki, A.; Khaki, A.A.; Rajabzadeh, A. The effects of Permethrin and antioxidant properties of Allium cepa (onion) on testicles parameters of male rats. Toxin Rev. 2017, 36, 1–6. [Google Scholar] [CrossRef]
- Shokoohi, M.; Madarek, E.O.S.; Khaki, A.; Shoorei, H.; Khaki, A.A.; Soltani, M.; Ainehchi, N. Investigating the effects of onion juice on male fertility factors and pregnancy rate after testicular torsion/detorsion by intrauterine insemination method. Int. J. Womens Health Reprod. Sci. 2018, 6, 499–505. [Google Scholar] [CrossRef]
- Khaki, A.; Khaki, A.A.; Hajhosseini, L.; Golzar, F.S.; Ainehchi, N. The anti-oxidant effects of ginger and cinnamon on spermatogenesis dys-function of diabetes rats. Afr. J. Tradit. Complement. Altern. Med. 2014, 11, 1–8. [Google Scholar] [CrossRef]
- Banihani, S.A. Ginger and testosterone. Biomolecules 2018, 8, 119. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, F.; Ye, L.; Zirkin, B.; Chen, H. Steroidogenesis in Leydig cells: Effects of aging and environmental factors. Reproduction 2017, 154, R111–R122. [Google Scholar] [CrossRef]
- Griswold, M.D. The central role of Sertoli cells in spermatogenesis. Semin. Cell Dev. Biol. 1998, 9, 411–416. [Google Scholar] [CrossRef]
- Lin, M.T.; Beal, M.F. Mitochondrial dysfunction and oxidative stress in neurodegenerative diseases. Nature 2006, 443, 787–795. [Google Scholar] [CrossRef] [PubMed]
- Banihani, S.A. Role of uric acid in semen. Biomolecules 2018, 8, 65. [Google Scholar] [CrossRef]
- Mohammad, N.S.; Arafa, M.H.; Atteia, H.H. Coenzyme q10 and fish oil synergistically alleviate aluminum chloride-induced suppression of testicular steroidogenesis and antioxidant defense. Free Radic. Res. 2015, 49, 1319–1334. [Google Scholar] [CrossRef]
- Jaiswal, N.; Rizvi, S.I. Onion extract (Allium cepa L.), quercetin and catechin up-regulate paraoxonase 1 activity with concomitant protection against low-density lipoprotein oxidation in male wistar rats subjected to oxidative stress. J. Sci. Food Agric. 2014, 94, 2752–2757. [Google Scholar] [CrossRef] [PubMed]
- Mlcek, J.; Jurikova, T.; Skrovankova, S.; Sochor, J. Quercetin and its anti-allergic immune response. Molecules 2016, 21, 623. [Google Scholar] [CrossRef]
- Eitah, H.E.; Maklad, Y.A.; Abdelkader, N.F.; Gamal El Din, A.A.; Badawi, M.A.; Kenawy, S.A. Modulating impacts of quercetin/sitagliptin combination on streptozotocin-induced diabetes mellitus in rats. Toxicol. Appl. Pharm. 2019, 36, 30–40. [Google Scholar] [CrossRef]
- Ruiz-Miyazawa, K.W.; Staurengo-Ferrari, L.; Mizokami, S.S.; Domiciano, T.P.; Vicentini, F.; Camilios-Neto, D.; Pavanelli, W.R.; Pinge-Filho, P.; Amaral, F.A.; Teixeira, M.M.; et al. Quercetin inhibits gout arthritis in mice: Induction of an opioid-dependent regulation of inflammasome. Inflammopharmacology 2017, 25, 555–570. [Google Scholar] [CrossRef] [PubMed]
- Brito, A.F.; Ribeiro, M.; Abrantes, A.M.; Pires, A.S.; Teixo, R.J.; Tralhao, J.G.; Botelho, M.F. Quercetin in cancer treatment, alone or in combination with conventional therapeutics? Curr. Med. Chem. 2015, 22, 3025–3039. [Google Scholar] [CrossRef] [PubMed]
- Khanna, D.; Sethi, G.; Ahn, K.S.; Pandey, M.K.; Kunnumakkara, A.B.; Sung, B.; Aggarwal, A.; Aggarwal, B.B. Natural products as a gold mine for arthritis treatment. Curr. Opin. Pharm. 2007, 7, 344–351. [Google Scholar] [CrossRef] [PubMed]
- Law, Y.Y.; Chiu, H.F.; Lee, H.H.; Shen, Y.C.; Venkatakrishnan, K.; Wang, C.K. Consumption of onion juice modulates oxidative stress and attenuates the risk of bone disorders in middle-aged and post-menopausal healthy subjects. Food Funct. 2016, 7, 902–912. [Google Scholar] [CrossRef] [PubMed]
- Ren, F.; Reilly, K.; Kerry, J.P.; Gaffney, M.; Hossain, M.; Rai, D.K. Higher antioxidant activity, total flavonols, and specific quercetin glucosides in two different onion (Allium cepa L.) varieties grown under organic production: Results from a 6-year field study. J. Agric. Food Chem. 2017, 65, 5122–5132. [Google Scholar] [CrossRef] [PubMed]
- Baltaci, B.B.; Uygur, R.; Caglar, V.; Aktas, C.; Aydin, M.; Ozen, O.A. Protective effects of quercetin against arsenic-induced testicular damage in rats. Andrologia 2016, 48, 1202–1213. [Google Scholar] [CrossRef] [PubMed]
- Abd-Ellah, M.F.; Aly, H.A.; Mokhlis, H.A.; Abdel-Aziz, A.H. Quercetin attenuates di-(2-ethylhexyl) phthalate-induced testicular toxicity in adult rats. Hum. Exp. Toxicol. 2016, 35, 232–243. [Google Scholar] [CrossRef] [PubMed]
- Abdel Aziz, R.L.; Abdel-Wahab, A.; Abo El-Ela, F.I.; Hassan, N.E.Y.; El-Nahass, E.S.; Ibrahim, M.A.; Khalil, A.A.Y. Dose- dependent ameliorative effects of quercetin and l-carnitine against atrazine- induced reproductive toxicity in adult male albino rats. Biomed. Pharm. 2018, 102, 855–864. [Google Scholar] [CrossRef]
- Osawe, S.O.; Farombi, E.O. Quercetin and rutin ameliorates sulphasalazine-induced spermiotoxicity, alterations in reproductive hormones and steroidogenic enzyme imbalance in rats. Andrologia 2018, 50, e12981. [Google Scholar] [CrossRef]
- Ujah, G.A.; Nna, V.U.; Agah, M.I.; Omue, L.O.; Leku, C.B.; Osim, E.E. Effect of quercetin on cadmium chloride-induced impairments in sexual behaviour and steroidogenesis in male wistar rats. Andrologia 2018, 50, e12866. [Google Scholar] [CrossRef]
- Sharma, P.; Aslam Khan, I.; Singh, R. Curcumin and quercetin ameliorated cypermethrin and deltamethrin-induced reproductive system impairment in male wistar rats by upregulating the activity of pituitary-gonadal hormones and steroidogenic enzymes. Int. J. Fertil. Steril. 2018, 12, 72–80. [Google Scholar]
- Murota, K.; Mitsukuni, Y.; Ichikawa, M.; Tsushida, T.; Miyamoto, S.; Terao, J. Quercetin-4′-glucoside is more potent than quercetin-3-glucoside in protection of rat intestinal mucosa homogenates against iron ion-induced lipid peroxidation. J. Agric. Food Chem. 2004, 52, 1907–1912. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.S.; Yang, M.; Goo, T.H.; Jo, C.; Ahn, D.U.; Park, J.H.; Lee, O.H.; Kang, S.N. Radical scavenging-linked antioxidant activities of commonly used herbs and spices in Korea. Int. J. Food Sci. Nutr. 2012, 63, 603–609. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.L.; Zhu, D.Y.; Thakur, K.; Wang, C.H.; Wang, H.; Ren, Y.F.; Zhang, J.G.; Wei, Z.J. Antioxidant and antibacterial evaluation of polysaccharides sequentially extracted from onion (Allium cepa L.). Int. J. Biol. Macromol. 2018, 111, 92–101. [Google Scholar] [CrossRef]
- Akinyemi, A.J.; Adedara, I.A.; Thome, G.R.; Morsch, V.M.; Rovani, M.T.; Mujica, L.K.S.; Duarte, T.; Duarte, M.; Oboh, G.; Schetinger, M.R.C. Dietary supplementation of ginger and turmeric improves reproductive function in hypertensive male rats. Toxicol. Rep. 2015, 2, 1357–1366. [Google Scholar] [CrossRef] [PubMed]
- Banihani, S.A. Radish (Raphanus sativus) and diabetes. Nutrients 2017, 9, 1014. [Google Scholar] [CrossRef] [PubMed]
- Maiese, K. New insights for oxidative stress and diabetes mellitus. Oxid. Med. Cell. Longev. 2015, 2015, 875961. [Google Scholar] [CrossRef] [PubMed]
- Thakur, P.; Kumar, A.; Kumar, A. Targeting oxidative stress through antioxidants in diabetes mellitus. J. Drug Target. 2018, 26, 766–776. [Google Scholar] [CrossRef] [PubMed]
- Denu, R.A.; Hematti, P. Effects of oxidative stress on mesenchymal stem cell biology. Oxid. Med. Cell. Longev. 2016, 2016, 2989076. [Google Scholar] [CrossRef]
- Taj-Eldin, I.M.; Ahmed, E.M.; Elwahab, H.M.A. Preliminary study of the clinical hypoglycemic effects of Allium cepa (red onion) in type 1 and type 2 diabetic patients. Environ. Health Insights 2010, 4, 71–77. [Google Scholar]
- Jung, J.Y.; Lim, Y.; Moon, M.S.; Kim, J.Y.; Kwon, O. Onion peel extracts ameliorate hyperglycemia and insulin resistance in high fat diet/streptozotocin-induced diabetic rats. Nutr. Metab. 2011, 8, 18. [Google Scholar] [CrossRef]
- Pradeep, S.R.; Srinivasan, K. Amelioration of oxidative stress by dietary fenugreek (Trigonella foenum-graecum L.) seeds is potentiated by onion (Allium cepa L.) in streptozotocin-induced diabetic rats. Appl. Physiol. Nutr. Metab. 2017, 42, 816–828. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.M.; Gu, C.H.; Tao, L.; Wu, X.L. Effect of surgery and efferent duct ligation on testicular blood flow and testicular steroidogenesis in the rat. J. Reprod. Fertil. 1985, 73, 191–196. [Google Scholar] [CrossRef] [PubMed]
- Clavijo, R.I.; Carrasquillo, R.; Ramasamy, R. Varicoceles: Prevalence and pathogenesis in adult men. Fertil. Steril. 2017, 108, 364–369. [Google Scholar] [CrossRef] [PubMed]
- Grman, M.; Misak, A.; Cacanyiova, S.; Kristek, F.; Tomaskova, Z.; Bertova, A.; Ondrias, K. The aqueous garlic, onion and leek extracts release nitric oxide from S-nitrosoglutathione and prolong relaxation of aortic rings. Gen. Physiol. Biophys. 2011, 30, 396–402. [Google Scholar] [CrossRef] [PubMed]
- Merrill, G.F.; Kurth, E.J.; Hardie, D.G.; Winder, W.W. Aica riboside increases amp-activated protein kinase, fatty acid oxidation, and glucose uptake in rat muscle. Am. J. Physiol. 1997, 273, E1107–E1112. [Google Scholar] [CrossRef] [PubMed]
- Taibi, N.; Dupont, J.; Bouguermouh, Z.; Froment, P.; Rame, C.; Anane, A.; Amirat, Z.; Khammar, F. Expression of adenosine 5′-monophosphate-activated protein kinase (AMPK) in ovine testis (ovis aries): In vivo regulation by nutritional state. Anim. Reprod. Sci. 2017, 178, 9–22. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.G.; Kim, J.R.; Choi, H.C. Quercetin-induced AMP-activated protein kinase activation attenuates vasoconstriction through LKB1-AMPK signaling pathway. J. Med. Food 2018, 21, 146–153. [Google Scholar] [CrossRef]
- Rivera Rivera, A.; Castillo-Pichardo, L.; Gerena, Y.; Dharmawardhane, S. Anti-breast cancer potential of quercetin via the Akt/AMPK/mammalian target of rapamycin (mTOR) signaling cascade. PLoS ONE 2016, 11, e0157251. [Google Scholar] [CrossRef]
| (Onion or Onion Derivative) | Dose (Mode of Treatment) | Duration | Population | Effect on Testosterone | Reference |
|---|---|---|---|---|---|
| Onion juice | 2, 4, and 6 mL day−1 (Orally) | 10 days | Male albino rats | (+) | [20] |
| Onion juice | 0.5 and 1 g rat−1 day−1 (Orally) | 20 days | Wistar male rats | (+) | [21] |
| Onion | 15 mg kg−1 day−1 (Orally) | 4 weeks | Streptozotocin-induced diabetic | (+) | [22] |
| Onion juice | 3 mL day−1 rat−1 (Orally) | 4 weeks | Lamotrigine-induced male rats | (+) | [23,24] |
| Aqueous extract of onion | 1 mL of the extract/100 g of body weight (Orally) | 8 weeks | Male rats with aluminum-induced reproductive toxicity | (±) | [25] |
| Aqueous extract of onion | 1 and 2 mL rat−1 (Orally) | 4 weeks | Rats | (±) | [26] |
| Aqueous extract of onion | 1 mL/100g of body weight (Orally) | 4 weeks | Male rats with cadmium-induced organ toxicity | (±) | [27] |
| Onion juice | 1 and 2 mL day−1 rat−1 (Orally) | 20 days | Adult male abino rats | (+) | [28] |
| Onion juice | 3 mL rat−1 (Orally) | 60 days | Eschericcia coli infected male rats | (+) | [29] |
| Onion juice | 3 mL rat−1 (Orally) | 60 days | Permethrin-induced male rats | (+) | [30] |
| Onion juice | 40 mg kg−1 (Orally) | 14 days | Testicular torsion/detorsion rat models | (+) | [31] |
| Onion extracts containing concentrated cysteine sulfoxides | 30 mg day−1 (Orally) | 14 days | Healthy men | (+) | [17] |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Banihani, S.A. Testosterone in Males as Enhanced by Onion (Allium Cepa L.). Biomolecules 2019, 9, 75. https://doi.org/10.3390/biom9020075
Banihani SA. Testosterone in Males as Enhanced by Onion (Allium Cepa L.). Biomolecules. 2019; 9(2):75. https://doi.org/10.3390/biom9020075
Chicago/Turabian StyleBanihani, Saleem Ali. 2019. "Testosterone in Males as Enhanced by Onion (Allium Cepa L.)" Biomolecules 9, no. 2: 75. https://doi.org/10.3390/biom9020075
APA StyleBanihani, S. A. (2019). Testosterone in Males as Enhanced by Onion (Allium Cepa L.). Biomolecules, 9(2), 75. https://doi.org/10.3390/biom9020075





