Membrane Remodeling as a Key Player of the Hepatotoxicity Induced by Co-Exposure to Benzo[a]pyrene and Ethanol of Obese Zebrafish Larvae
Abstract
1. Introduction
2. Results
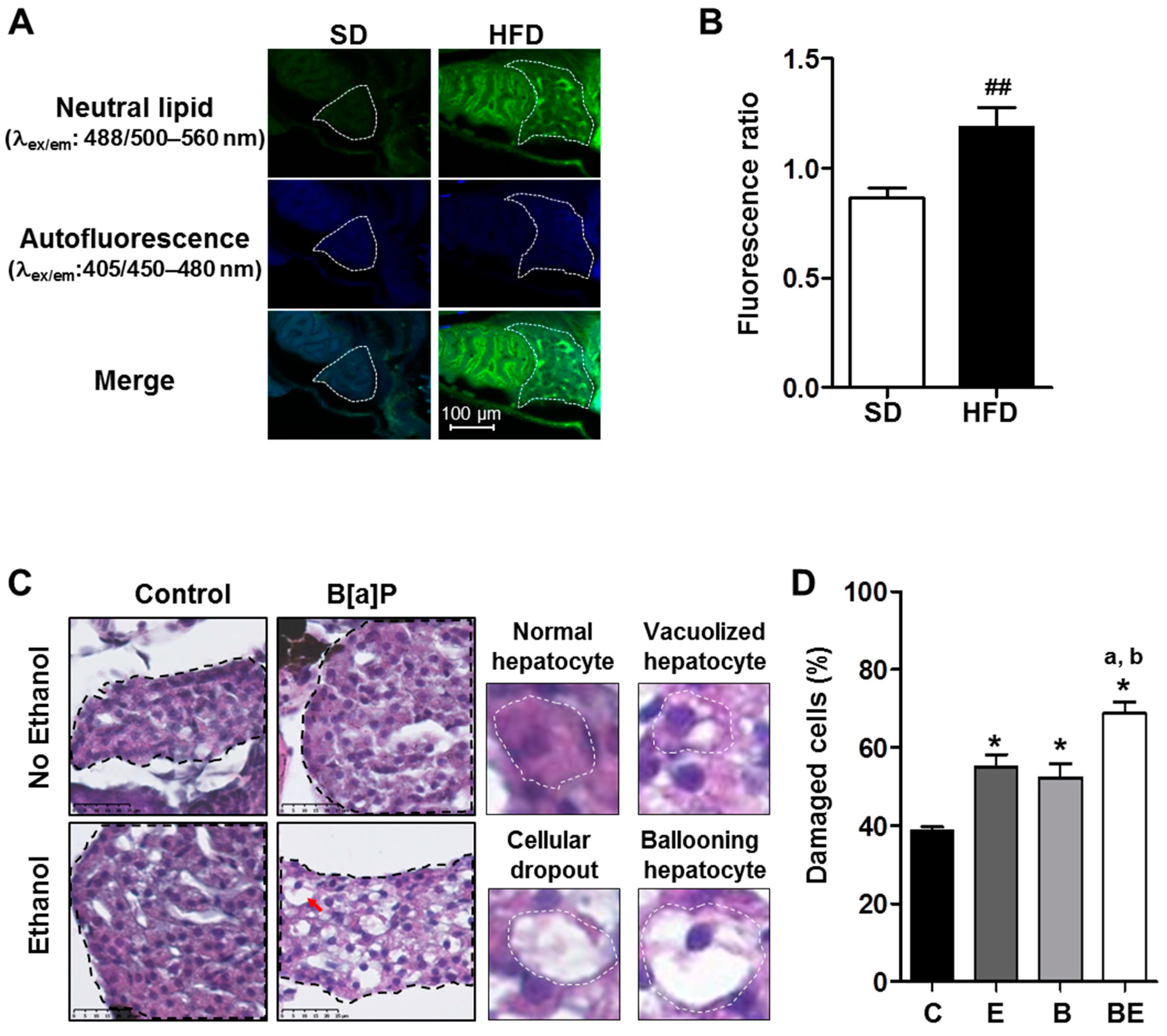
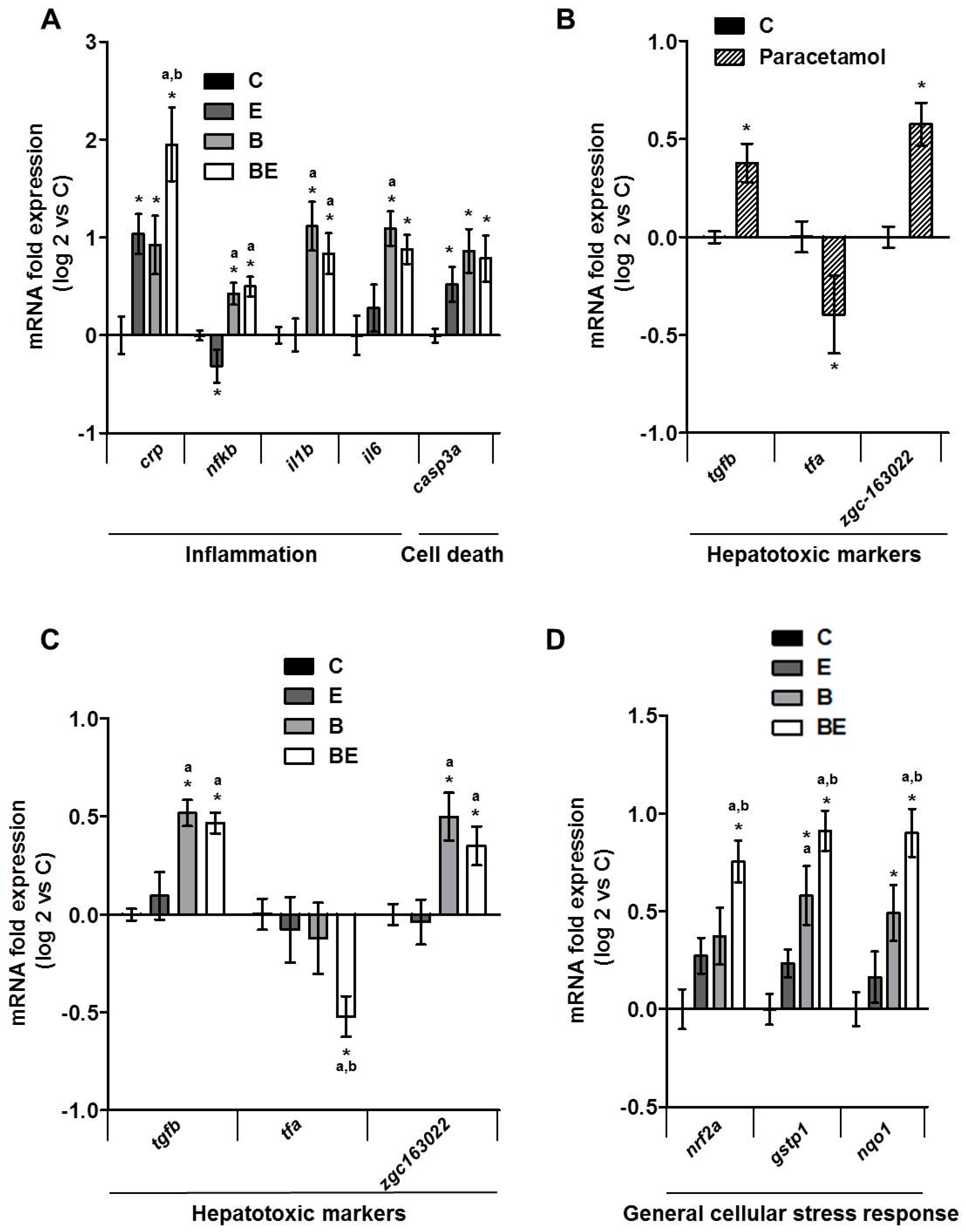
2.1. Progression of High-Fat Diet Induced Steatosis to a Steatohepatitis-Like State in Zebrafish Larvae upon Co-Exposure to B[a]P and Ethanol
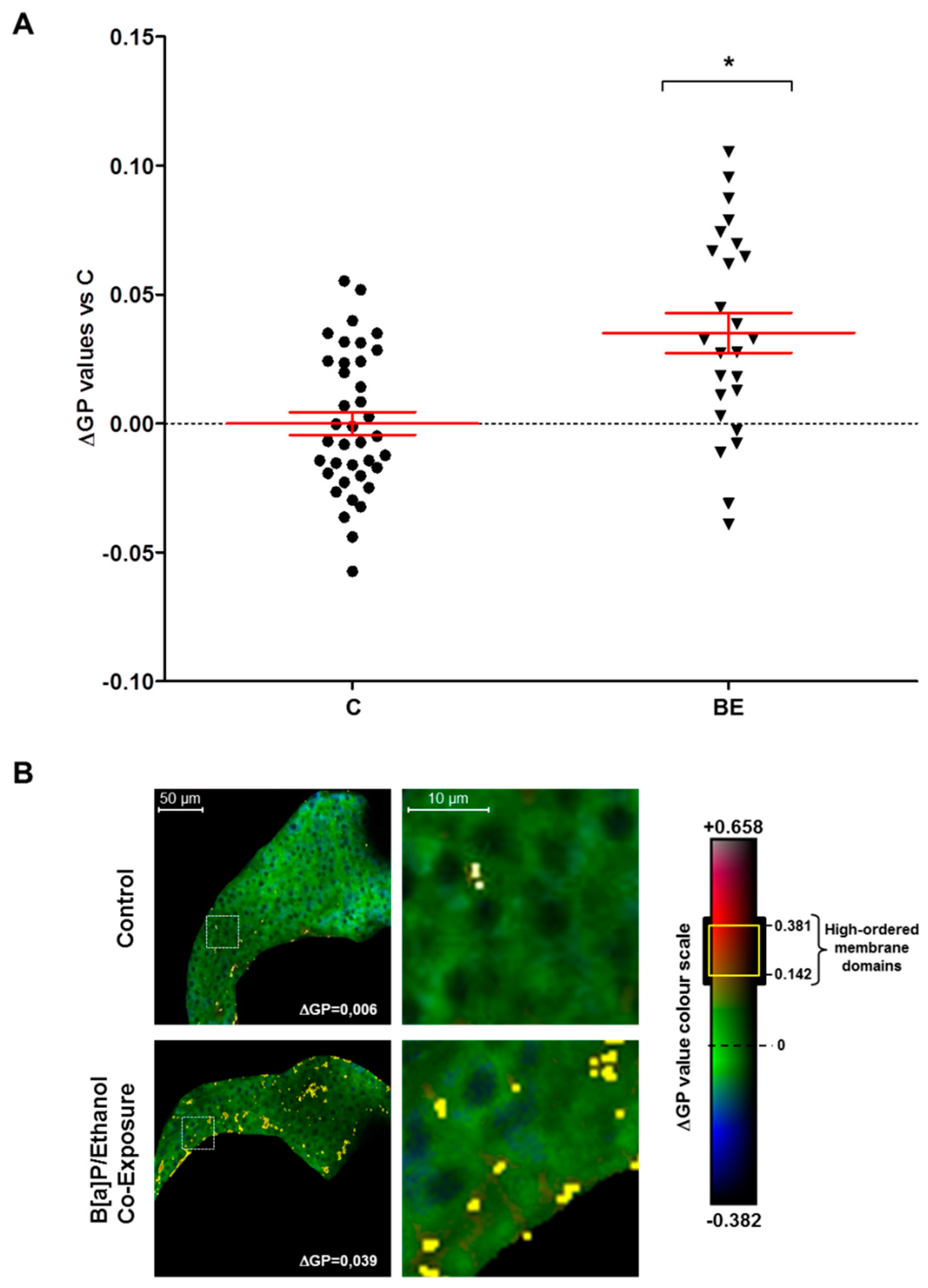
2.2. Involvement of Membrane Remodeling in the Hepatotoxicity Induced by B[a]P and Ethanol Co-Exposure in Zebrafish Larvae
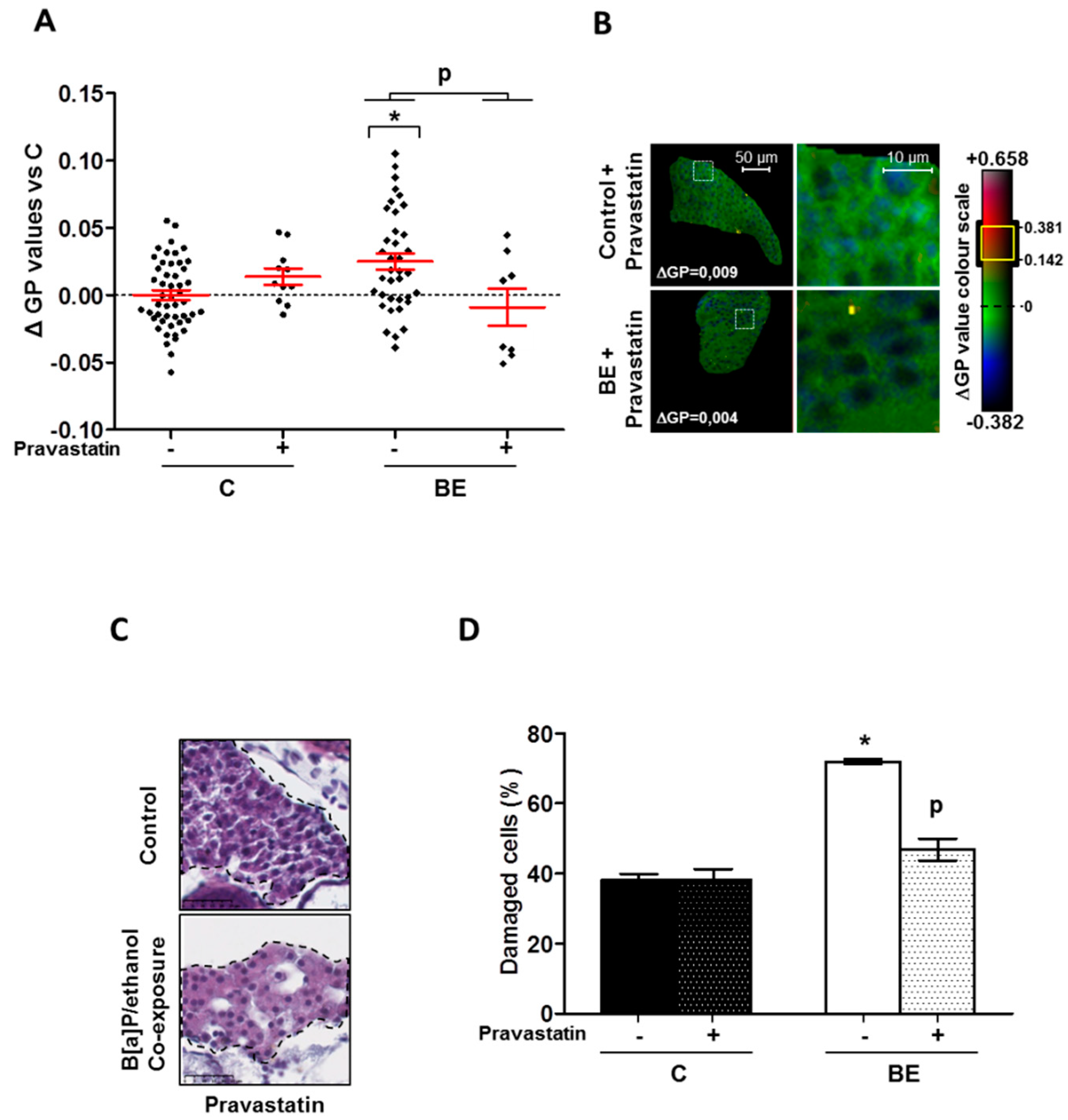
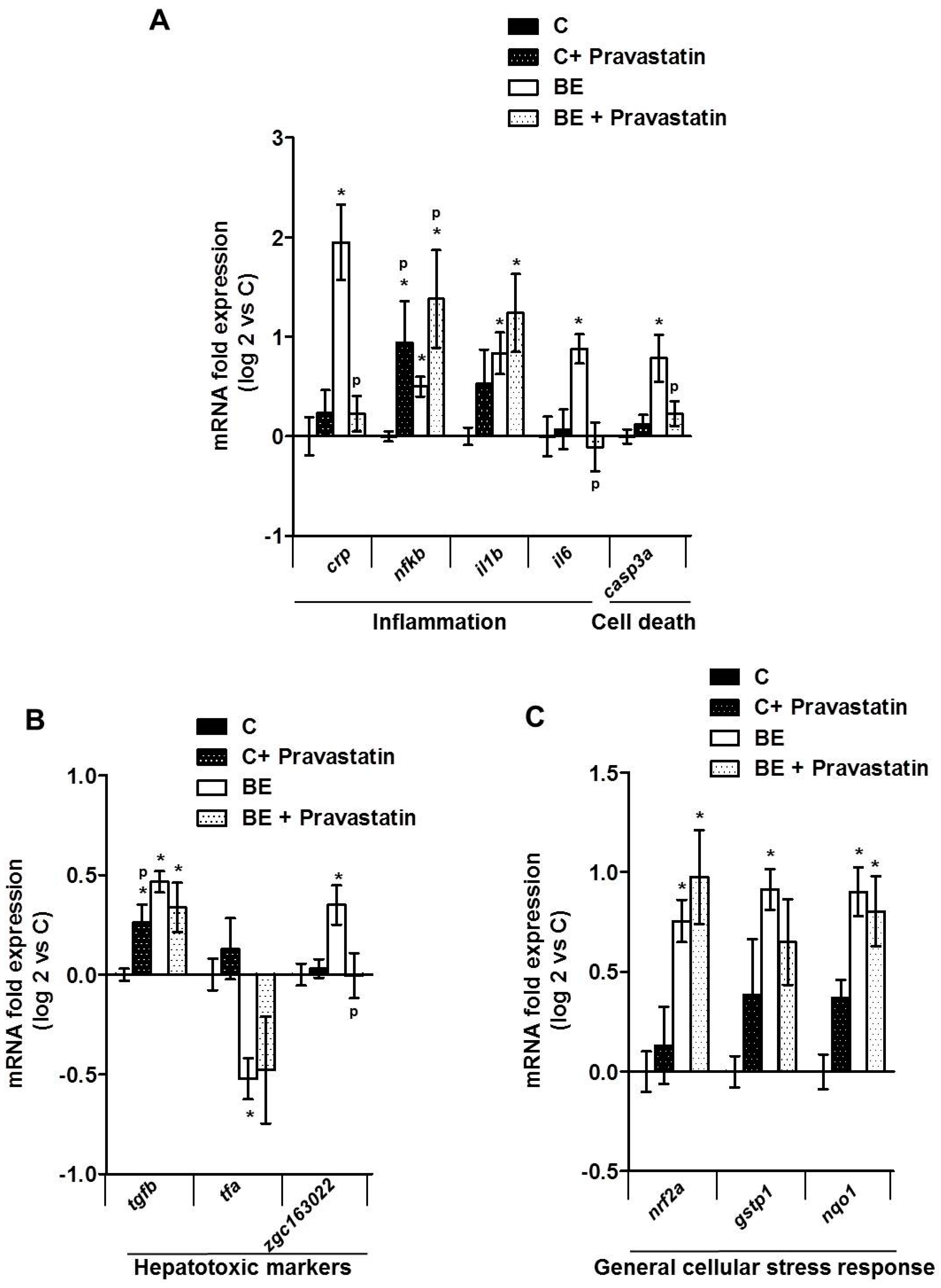
2.3. Role for Membrane Remodeling in the Protective Effect of Pravastatin against Co-Exposure-Induced Hepatotoxicity in Zebrafish Larvae
3. Discussion
4. Materials and Methods
4.1. Zebrafish Larvae Handling and Exposure
4.2. Neutral Lipid Staining with Nile Red
4.3. Histological Analysis of Liver Toxicity in Zebrafish Larvae
4.4. Analysis of Gene mRNA Expression
4.5. Membrane Order Determination by Fluorescence Staining
4.6. Statistical Analysis
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Jung, U.J.; Choi, M.-S. Obesity and its metabolic complications: The role of adipokines and the relationship between obesity, inflammation, insulin resistance, dyslipidemia and nonalcoholic fatty liver disease. Int. J. Mol. Sci. 2014, 15, 6184–6223. [Google Scholar] [CrossRef] [PubMed]
- Younossi, Z.M.; Koenig, A.B.; Abdelatif, D.; Fazel, Y.; Henry, L.; Wymer, M. Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology 2016, 64, 73–84. [Google Scholar] [CrossRef] [PubMed]
- Yasutake, K. Dietary habits and behaviors associated with nonalcoholic fatty liver disease. World J. Gastroenterol. 2014, 20, 1756. [Google Scholar] [CrossRef] [PubMed]
- Hardy, T.; Oakley, F.; Anstee, Q.M.; Day, C.P. Nonalcoholic fatty liver disease: Pathogenesis and disease spectrum. Ann. Rev. Pathol. 2016, 11, 451–496. [Google Scholar] [CrossRef] [PubMed]
- Diehl, A.M.; Day, C. Cause, pathogenesis, and treatment of nonalcoholic steatohepatitis. N. Engl. J. Med. 2017, 377, 2063–2072. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, S.H.; Hirsova, P.; Gores, G.J. Non-alcoholic steatohepatitis pathogenesis: Sublethal hepatocyte injury as a driver of liver inflammation. Gut 2018, 67, 963–972. [Google Scholar] [CrossRef] [PubMed]
- Cobbina, E.; Akhlaghi, F. Non-alcoholic fatty liver disease (NAFLD)—Pathogenesis, classification, and effect on drug metabolizing enzymes and transporters. Drug Metab. Rev. 2017, 49, 197–211. [Google Scholar] [CrossRef] [PubMed]
- Baršić, N.; Lerotić, I.; Smirčić-Duvnjak, L.; Tomašić, V.; Duvnjak, M. Overview and developments in noninvasive diagnosis of nonalcoholic fatty liver disease. World J. Gastroenterol. 2012, 18, 3945–3954. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Anderson, N.; Borlak, J. Molecular mechanisms and therapeutic targets in steatosis and steatohepatitis. Pharmacol. Rev. 2008, 60, 311–357. [Google Scholar] [CrossRef] [PubMed]
- Buzzetti, E.; Pinzani, M.; Tsochatzis, E.A. The multiple-hit pathogenesis of non-alcoholic fatty liver disease (NAFLD). Metab. Clin. Exp. 2016, 65, 1038–1048. [Google Scholar] [CrossRef] [PubMed]
- Patel, V.; Sanyal, A.J. Drug-Induced Steatohepatitis. Clin. Liver Dis. 2013, 17, 533–546. [Google Scholar] [CrossRef] [PubMed]
- Noureddin, M.; Rinella, M.E. Nonalcoholic fatty liver disease, diabetes, obesity, and hepatocellular carcinoma. Clin. Liver Dis. 2015, 19, 361–379. [Google Scholar] [CrossRef] [PubMed]
- Minato, T.; Tsutsumi, M.; Tsuchishima, M.; Hayashi, N.; Saito, T.; Matsue, Y.; Toshikuni, N.; Arisawa, T.; George, J. Binge alcohol consumption aggravates oxidative stress and promotes pathogenesis of NASH from obesity-induced simple steatosis. Mol. Med. 2014, 20, 490–502. [Google Scholar] [CrossRef] [PubMed]
- Younossi, Z.M. Nonalcoholic fatty liver disease. Curr. Gastroenterol. Rep. 1999, 1, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Teli, M.R.; James, O.F.; Burt, A.D.; Bennett, M.K.; Day, C.P. The natural history of nonalcoholic fatty liver: A follow-up study. Hepatology 1995, 22, 1714–1719. [Google Scholar] [CrossRef] [PubMed]
- Heindel, J.J.; Blumberg, B.; Cave, M.; Machtinger, R.; Mantovani, A.; Mendez, M.A.; Nadal, A.; Palanza, P.; Panzica, G.; Sargis, R.; et al. Metabolism disrupting chemicals and metabolic disorders. Reprod. Toxicol. 2017, 68, 3–33. [Google Scholar] [CrossRef] [PubMed]
- Wahlang, B.; Beier, J.I.; Clair, H.B.; Bellis-Jones, H.J.; Falkner, K.C.; McClain, C.J.; Cave, M.C. Toxicant-associated steatohepatitis. Toxicol. Pathol. 2013, 41, 343–360. [Google Scholar] [CrossRef] [PubMed]
- Joshi-Barve, S.; Kirpich, I.; Cave, M.C.; Marsano, L.S.; McClain, C.J. Alcoholic, nonalcoholic, and toxicant-associated steatohepatitis: Mechanistic similarities and differences. Cell. Mol. Gastroenterol. Hepatol. 2015, 1, 356–367. [Google Scholar] [CrossRef] [PubMed]
- Foulds, C.E.; Treviño, L.S.; York, B.; Walker, C.L. Endocrine-disrupting chemicals and fatty liver disease. Nat. Rev. Endocrinol. 2017, 13, 445–457. [Google Scholar] [CrossRef] [PubMed]
- Åberg, F.; Helenius-Hietala, J.; Puukka, P.; Färkkilä, M.; Jula, A. Interaction between alcohol consumption and metabolic syndrome in predicting severe liver disease in the general population. Hepatology 2017. [Google Scholar] [CrossRef]
- Duly, A.M.P.; Alani, B.; Huang, E.Y.-W.; Yee, C.; Haber, P.S.; McLennan, S.V.; Seth, D. Effect of multiple binge alcohol on diet-induced liver injury in a mouse model of obesity. Nutr. Diabetes 2015, 5, e154. [Google Scholar] [CrossRef] [PubMed]
- Uno, S.; Nebert, D.W.; Makishima, M. Cytochrome P450 1A1 (CYP1A1) protects against nonalcoholic fatty liver disease caused by Western diet containing benzo[a]pyrene in mice. Food Chem. Toxicol. 2018, 113, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Robin, M.-A.; Demeilliers, C.; Sutton, A.; Paradis, V.; Maisonneuve, C.; Dubois, S.; Poirel, O.; Lettéron, P.; Pessayre, D.; Fromenty, B. Alcohol increases tumor necrosis factor alpha and decreases nuclear factor-kappab to activate hepatic apoptosis in genetically obese mice. Hepatology 2005, 42, 1280–1290. [Google Scholar] [CrossRef] [PubMed]
- Duval, C.; Teixeira-Clerc, F.; Leblanc, A.F.; Touch, S.; Emond, C.; Guerre-Millo, M.; Lotersztajn, S.; Barouki, R.; Aggerbeck, M.; Coumoul, X. Chronic exposure to low doses of dioxin promotes liver fibrosis development in the c57bl/6j diet-induced obesity mouse model. Environ. Health Perspect. 2017, 125, 428–436. [Google Scholar] [CrossRef] [PubMed]
- Massart, J.; Begriche, K.; Moreau, C.; Fromenty, B. Role of nonalcoholic fatty liver disease as risk factor for drug-induced hepatotoxicity. J. Clin. Transl. Res. 2017, 3, 212–232. [Google Scholar] [CrossRef] [PubMed]
- Bambino, K.; Zhang, C.; Austin, C.; Amarasiriwardena, C.; Arora, M.; Chu, J.; Sadler, K.C. Inorganic arsenic causes fatty liver and interacts with ethanol to cause alcoholic liver disease in zebrafish. Dis. Models Mech. 2018, 11. [Google Scholar] [CrossRef] [PubMed]
- Bucher, S.; Tête, A.; Podechard, N.; Liamin, M.; Le Guillou, D.; Chevanne, M.; Coulouarn, C.; Imran, M.; Gallais, I.; Fernier, M.; et al. Co-exposure to benzo[a]pyrene and ethanol induces a pathological progression of liver steatosis in vitro and in vivo. Sci. Rep. 2018, 8. [Google Scholar] [CrossRef] [PubMed]
- Ba, Q.; Li, J.; Huang, C.; Qiu, H.; Li, J.; Chu, R.; Zhang, W.; Xie, D.; Wu, Y.; Wang, H. Effects of benzo[a]pyrene exposure on human hepatocellular carcinoma cell angiogenesis, metastasis, and NF-κB signaling. Environ. Health Perspect. 2015, 123, 246–254. [Google Scholar] [CrossRef] [PubMed]
- Wester, P.W.; Muller, J.J.A.; Slob, W.; Mohn, G.R.; Dortant, P.M.; Kroese, E.D. Carcinogenic activity of benzo[a]pyrene in a 2 year oral study in Wistar rats. Food Chem. Toxicol. 2012, 50, 927–935. [Google Scholar] [CrossRef] [PubMed]
- Hardonnière, K.; Huc, L.; Sergent, O.; Holme, J.A.; Lagadic-Gossmann, D. Environmental carcinogenesis and pH homeostasis: Not only a matter of dysregulated metabolism. Semin. Cancer Biol. 2017, 43, 49–65. [Google Scholar] [CrossRef] [PubMed]
- Tekpli, X.; Holme, J.A.; Sergent, O.; Lagadic-Gossmann, D. Role for membrane remodeling in cell death: Implication for health and disease. Toxicology 2013, 304, 141–157. [Google Scholar] [CrossRef] [PubMed]
- Tekpli, X.; Holme, J.A.; Sergent, O.; Lagadic-Gossmann, D. Importance of plasma membrane dynamics in chemical-induced carcinogenesis. Recent Pat. Anticancer Drug Discov. 2011, 6, 347–353. [Google Scholar] [CrossRef] [PubMed]
- Podechard, N.; Chevanne, M.; Fernier, M.; Tête, A.; Collin, A.; Cassio, D.; Kah, O.; Lagadic-Gossmann, D.; Sergent, O. Zebrafish larva as a reliable model for in vivo assessment of membrane remodeling involvement in the hepatotoxicity of chemical agents: Zebrafish larva for assessing membrane remodeling by hepatotoxicants. J. Appl. Toxicol. 2017, 37, 732–746. [Google Scholar] [CrossRef] [PubMed]
- Dolganiuc, A. Role of lipid rafts in liver health and disease. World J. Gastroenterol. 2011, 17, 2520–2535. [Google Scholar] [CrossRef] [PubMed]
- Inokuchi, S.; Tsukamoto, H.; Park, E.; Liu, Z.-X.; Brenner, D.A.; Seki, E. Toll-like receptor 4 mediates alcohol-induced steatohepatitis through bone marrow-derived and endogenous liver cells in mice. Alcohol. Clin. Exp. Res. 2011, 35, 1509–1518. [Google Scholar] [CrossRef] [PubMed]
- Tekpli, X.; Rissel, M.; Huc, L.; Catheline, D.; Sergent, O.; Rioux, V.; Legrand, P.; Holme, J.A.; Dimanche-Boitrel, M.-T.; Lagadic-Gossmann, D. Membrane remodeling, an early event in benzo[a]pyrene-induced apoptosis. Toxicol. Appl. Pharmacol. 2010, 243, 68–76. [Google Scholar] [CrossRef] [PubMed]
- Tekpli, X.; Huc, L.; Sergent, O.; Dendelé, B.; Dimanche-Boitrel, M.-T.; Holme, J.A.; Lagadic-Gossmann, D. NHE-1 relocation outside cholesterol-rich membrane microdomains is associated with its benzo[a]pyrene-related apoptotic function. Cell. Physiol. Biochem. 2012, 29, 657–666. [Google Scholar] [CrossRef] [PubMed]
- Nourissat, P.; Travert, M.; Chevanne, M.; Tekpli, X.; Rebillard, A.; Le Moigne-Müller, G.; Rissel, M.; Cillard, J.; Dimanche-Boitrel, M.-T.; Lagadic-Gossmann, D.; et al. Ethanol induces oxidative stress in primary rat hepatocytes through the early involvement of lipid raft clustering. Hepatology 2008, 47, 59–70. [Google Scholar] [CrossRef] [PubMed]
- Roh, Y.S.; Zhang, B.; Loomba, R.; Seki, E. TLR2 and TLR9 contribute to alcohol-mediated liver injury through induction of CXCL1 and neutrophil infiltration. Am. J. Physiol. Gastrointest. Liver Physiol. 2015, 309, G30–G41. [Google Scholar] [CrossRef] [PubMed]
- Collin, A.; Hardonnière, K.; Chevanne, M.; Vuillemin, J.; Podechard, N.; Burel, A.; Dimanche-Boitrel, M.-T.; Lagadic-Gossmann, D.; Sergent, O. Cooperative interaction of benzo[a]pyrene and ethanol on plasma membrane remodeling is responsible for enhanced oxidative stress and cell death in primary rat hepatocytes. Free Radic. Biol. Med. 2014, 72, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Alhasson, F.; Dattaroy, D.; Pourhoseini, S.; Seth, R.K.; Nagarkatti, M.; Nagarkatti, P.S.; Michelotti, G.A.; Diehl, A.M.; Kalyanaraman, B.; et al. NADPH Oxidase-derived peroxynitrite drives inflammation in mice and human nonalcoholic steatohepatitis via tlr4-lipid raft recruitment. Am. J. Pathol. 2015, 185, 1944–1957. [Google Scholar] [CrossRef] [PubMed]
- Howe, K.; Clark, M.D.; Torroja, C.F.; Torrance, J.; Berthelot, C.; Muffato, M.; Collins, J.E.; Humphray, S.; McLaren, K.; Matthews, L.; et al. The zebrafish reference genome sequence and its relationship to the human genome. Nature 2013, 496, 498–503. [Google Scholar] [CrossRef] [PubMed]
- Chu, J.; Sadler, K.C. New school in liver development: Lessons from zebrafish. Hepatology 2009, 50, 1656–1663. [Google Scholar] [CrossRef] [PubMed]
- Schlegel, A. Studying non-alcoholic fatty liver disease with zebrafish: A confluence of optics, genetics, and physiology. Cell. Mol. Life Sci. 2012, 69, 3953–3961. [Google Scholar] [CrossRef] [PubMed]
- Forn-Cuní, G.; Varela, M.; Pereiro, P.; Novoa, B.; Figueras, A. Conserved gene regulation during acute inflammation between zebrafish and mammals. Sci. Rep. 2017, 7, 41905. [Google Scholar] [CrossRef] [PubMed]
- Goessling, W.; Sadler, K.C. Zebrafish: An Important Tool for Liver Disease Research. Gastroenterology 2015, 149, 1361–1377. [Google Scholar] [CrossRef] [PubMed]
- Passeri, M.J.; Cinaroglu, A.; Gao, C.; Sadler, K.C. Hepatic steatosis in response to acute alcohol exposure in zebrafish requires sterol regulatory element binding protein activation. Hepatology 2009, 49, 443–452. [Google Scholar] [CrossRef] [PubMed]
- Alderton, W.; Berghmans, S.; Butler, P.; Chassaing, H.; Fleming, A.; Golder, Z.; Richards, F.; Gardner, I. Accumulation and metabolism of drugs and CYP probe substrates in zebrafish larvae. Xenobiotica 2010, 40, 547–557. [Google Scholar] [CrossRef] [PubMed]
- Bugiak, B.; Weber, L.P. Hepatic and vascular mRNA expression in adult zebrafish (Danio rerio) following exposure to benzo-a-pyrene and 2,3,7,8-tetrachlorodibenzo-p-dioxin. Aquat. Toxicol. 2009, 95, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Verstraelen, S.; Peers, B.; Maho, W.; Hollanders, K.; Remy, S.; Berckmans, P.; Covaci, A.; Witters, H. Phenotypic and biomarker evaluation of zebrafish larvae as an alternative model to predict mammalian hepatotoxicity. J. Appl. Toxicol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Driessen, M.; Kienhuis, A.S.; Pennings, J.L.A.; Pronk, T.E.; Brandhof, E.-J.; Roodbergen, M.; Spaink, H.P.; Water, B.; Ven, L.T.M. Exploring the zebrafish embryo as an alternative model for the evaluation of liver toxicity by histopathology and expression profiling. Arch. Toxicol. 2013, 87, 807–823. [Google Scholar] [CrossRef] [PubMed]
- Driessen, M.; Kienhuis, A.S.; Vitins, A.P.; Pennings, J.L.A.; Pronk, T.E.; Brandhof, E.-J.; Roodbergen, M.; Water, B.; Ven, L.T.M. Gene expression markers in the zebrafish embryo reflect a hepatotoxic response in animal models and humans. Toxicol. Lett. 2014, 230, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Driessen, M.; Vitins, A.P.; Pennings, J.L.A.; Kienhuis, A.S.; Water, B.; Ven, L.T.M. A transcriptomics-based hepatotoxicity comparison between the zebrafish embryo and established human and rodent in vitro and in vivo models using cyclosporine A, amiodarone and acetaminophen. Toxicol. Lett. 2015, 232, 403–412. [Google Scholar] [CrossRef] [PubMed]
- Tsedensodnom, O.; Vacaru, A.M.; Howarth, D.L.; Yin, C.; Sadler, K.C. Ethanol metabolism and oxidative stress are required for unfolded protein response activation and steatosis in zebrafish with alcoholic liver disease. Dis. Models Mech. 2013, 6, 1213–1226. [Google Scholar] [CrossRef] [PubMed]
- Dai, W.; Wang, K.; Zheng, X.; Chen, X.; Zhang, W.; Zhang, Y.; Hou, J.; Liu, L. High fat plus high cholesterol diet lead to hepatic steatosis in zebrafish larvae: A novel model for screening anti-hepatic steatosis drugs. Nutr. Metab. 2015, 12, 42. [Google Scholar] [CrossRef] [PubMed]
- Yan, C.; Yang, Q.; Shen, H.-M.; Spitsbergen, J.M.; Gong, Z. Chronically high level of tgfb1a induction causes both hepatocellular carcinoma and cholangiocarcinoma via a dominant Erk pathway in zebrafish. Oncotarget 2017, 8, 77096–77109. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Garcia, L.; Schlegel, A. Lxr-driven enterocyte lipid droplet formation delays transport of ingested lipids. J. Lipid Res. 2014, 55, 1944–1958. [Google Scholar] [CrossRef] [PubMed]
- Amali, A.A.; Rekha, R.D.; Lin, C.J.-F.; Wang, W.-L.; Gong, H.-Y.; Her, G.-M.; Wu, J.-L. Thioacetamide induced liver damage in zebrafish embryo as a disease model for steatohepatitis. J. Biomed. Sci. 2006, 13, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Hugo, S.E.; Schlegel, A. A genetic screen for zebrafish mutants with hepatic steatosis identifies a locus required for larval growth. J. Anat. 2017, 230, 407–413. [Google Scholar] [CrossRef] [PubMed]
- IARD (International Alliance for Responsible Drinking). Drinking Guidelines: General Population. Available online: http://www.iard.org/policy-tables/drinking-guidelines-general-population (accessed on 6 April 2018).
- Walker, D.I.; Pennell, K.D.; Uppal, K.; Xia, X.; Hopke, P.K.; Utell, M.J.; Phipps, R.P.; Sime, P.J.; Rohrbeck, P.; Mallon, C.T.M.; et al. Pilot Metabolome-Wide Association Study of Benzo(a)pyrene in Serum From Military Personnel. J. Occup. Environ. Med. 2016, 58, 445. [Google Scholar] [CrossRef] [PubMed]
- Magee, N.; Zou, A.; Zhang, Y. Pathogenesis of Nonalcoholic Steatohepatitis: Interactions between Liver Parenchymal and Nonparenchymal Cells. BioMed Res. Int. 2016, 2016, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Issa, D.; Patel, V.; Sanyal, A.J. Future therapy for non-alcoholic fatty liver disease. Liver Int. 2018, 38, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Sumida, Y.; Yoneda, M. Current and future pharmacological therapies for NAFLD/NASH. J. Gastroenterol. 2018, 53, 362–376. [Google Scholar] [CrossRef] [PubMed]
- Shen, G.; Kong, A.-N. Nrf2 plays an important role in coordinated regulation of Phase II drug metabolism enzymes and Phase III drug transporters. Biopharm. Drug Dispos. 2009, 30, 345–355. [Google Scholar] [CrossRef] [PubMed]
- Chambel, S.S.; Santos-Gonçalves, A.; Duarte, T.L. The Dual Role of Nrf2 in Nonalcoholic Fatty Liver Disease: Regulation of Antioxidant Defenses and Hepatic Lipid Metabolism. Biomed. Res. Int. 2015, 2015, 597134. [Google Scholar] [CrossRef] [PubMed]
- Owen, D.M.; Rentero, C.; Magenau, A.; Abu-Siniyeh, A.; Gaus, K. Quantitative imaging of membrane lipid order in cells and organisms. Nat. Protoc. 2012, 7, 24–35. [Google Scholar] [CrossRef] [PubMed]
- Sezgin, E.; Sadowski, T.; Simons, K. Measuring Lipid Packing of Model and Cellular Membranes with Environment Sensitive Probes. Langmuir 2014, 30, 8160–8166. [Google Scholar] [CrossRef] [PubMed]
- Aron, M.; Browning, R.; Carugo, D.; Sezgin, E.; Bernardino de la Serna, J.; Eggeling, C.; Stride, E. Spectral imaging toolbox: Segmentation, hyperstack reconstruction, and batch processing of spectral images for the determination of cell and model membrane lipid order. BMC Bioinform. 2017, 18. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.-M.; Li, X.; Xiong, J.; Abais, J.M.; Xia, M.; Boini, K.M.; Zhang, Y.; Li, P.-L. Attenuation by statins of membrane raft-redox signaling in coronary arterial endothelium. J. Pharmacol. Exp. Ther. 2013, 345, 170–179. [Google Scholar] [CrossRef] [PubMed]
- Hardonnière, K.; Fernier, M.; Gallais, I.; Mograbi, B.; Podechard, N.; Le Ferrec, E.; Grova, N.; Appenzeller, B.; Burel, A.; Chevanne, M.; et al. Role for the ATPase inhibitory factor 1 in the environmental carcinogen-induced Warburg phenotype. Sci. Rep. 2017, 7, 195. [Google Scholar] [CrossRef] [PubMed]
- Sugimoto, K.; Takei, Y. Pathogenesis of alcoholic liver disease: Pathogenesis of alcoholic liver disease. Hepatol. Res. 2017, 47, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Souza, T.; Jennen, D.; van Delft, J.; van Herwijnen, M.; Kyrtoupolos, S.; Kleinjans, J. New insights into BaP-induced toxicity: Role of major metabolites in transcriptomics and contribution to hepatocarcinogenesis. Arch. Toxicol. 2016, 90, 1449–1458. [Google Scholar] [CrossRef] [PubMed]
- Hardonnière, K.; Saunier, E.; Lemarié, A.; Fernier, M.; Gallais, I.; Héliès-Toussaint, C.; Mograbi, B.; Antonio, S.; Bénit, P.; Rustin, P.; et al. The environmental carcinogen benzo[a]pyrene induces a Warburg-like metabolic reprogramming dependent on NHE1 and associated with cell survival. Sci. Rep. 2016, 6, 30776. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Zhuang, Z.-J.; Bian, D.-X.; Ma, X.-J.; Xun, Y.-H.; Yang, W.-J.; Luo, Y.; Liu, Y.-L.; Jia, L.; Wang, Y.; et al. Toll-like receptor-4 signalling in the progression of non-alcoholic fatty liver disease induced by high-fat and high-fructose diet in mice. Clin. Exp. Pharmacol. Physiol. 2014, 41, 482–488. [Google Scholar] [CrossRef] [PubMed]
- Sutter, A.G.; Palanisamy, A.P.; Lench, J.H.; Esckilsen, S.; Geng, T.; Lewin, D.N.B.; Cowart, L.A.; Chavin, K.D. Dietary Saturated Fat Promotes Development of Hepatic Inflammation Through Toll-Like Receptor 4 in Mice. J. Cell. Biochem. 2016, 117, 1613–1621. [Google Scholar] [CrossRef] [PubMed]
- Gianfrancesco, M.A.; Paquot, N.; Piette, J.; Legrand-Poels, S. Lipid bilayer stress in obesity-linked inflammatory and metabolic disorders. Biochem. Pharmacol. 2018, in press. [Google Scholar] [CrossRef]
- Roh, Y.S.; Seki, E. Toll-like receptors in alcoholic liver disease, non-alcoholic steatohepatitis and carcinogenesis. J. Gastroenterol. Hepatol. 2013, 28, 38–42. [Google Scholar] [CrossRef] [PubMed]
- Decaens, C.; Rodriguez, P.; Bouchaud, C.; Cassio, D. Establishment of hepatic cell polarity in the rat hepatoma-human fibroblast hybrid WIF-B9. A biphasic phenomenon going from a simple epithelial polarized phenotype to an hepatic polarized one. J. Cell Sci. 1996, 109, 1623–1635. [Google Scholar] [PubMed]
- Biagini, C.; Bender, V.; Borde, F.; Boissel, E.; Bonnet, M.-C.; Masson, M.-T.; Cassio, D.; Chevalier, S. Cytochrome P450 expression-induction profile and chemically mediated alterations of the WIF-B9 cell line. Biol. Cell 2006, 98, 23–32. [Google Scholar] [CrossRef] [PubMed]
- McVicker, B.L.; Tuma, D.J.; Kubik, J.L.; Tuma, P.L.; Casey, C.A. Ethanol-induced apoptosis in polarized hepatic cells possibly through regulation of the Fas pathway. Alcohol. Clin. Exp. Res. 2006, 30, 1906–1915. [Google Scholar] [CrossRef] [PubMed]
- Ozeki, J.; Uno, S.; Ogura, M.; Choi, M.; Maeda, T.; Sakurai, K.; Matsuo, S.; Amano, S.; Nebert, D.W.; Makishima, M. Aryl hydrocarbon receptor ligand 2,3,7,8-tetrachlorodibenzo-p-dioxin enhances liver damage in bile duct-ligated mice. Toxicology 2011, 280, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Lieber, C.S. Alcoholic fatty liver: Its pathogenesis and mechanism of progression to inflammation and fibrosis. Alcohol 2004, 34, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wu, K.C.; Lu, Y.-F.; Ekuase, E.; Klaassen, C.D. NRF2 Protection against Liver Injury Produced by Various Hepatotoxicants. Oxid. Med. Cell. Longev. 2013, 2013, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Wu, K.C.; Cui, J.Y.; Klaassen, C.D. Effect of Graded Nrf2 Activation on Phase-I and -II Drug Metabolizing Enzymes and Transporters in Mouse Liver. PLoS ONE 2012, 7, e39006. [Google Scholar] [CrossRef] [PubMed]
- Copple, I.M.; Dinkova-Kostova, A.T.; Kensler, T.W.; Liby, K.T.; Wigley, W.C. NRF2 as an Emerging Therapeutic Target. Oxid. Med. Cell. Longev. 2017, 2017, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Henriksbo, B.D.; Schertzer, J.D. Is immunity a mechanism contributing to statin-induced diabetes? Adipocyte 2015, 4, 232–238. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Zhao, L.; Yang, P.; Zhou, W.; Li, B.; Moorhead, J.F.; Varghese, Z.; Ruan, X.Z.; Chen, Y. Inflammatory Stress Sensitizes the Liver to Atorvastatin-Induced Injury in ApoE-/- Mice. PLoS ONE 2016, 11, e0159512. [Google Scholar] [CrossRef] [PubMed]
- Pastori, D.; Polimeni, L.; Baratta, F.; Pani, A.; Del Ben, M.; Angelico, F. The efficacy and safety of statins for the treatment of non-alcoholic fatty liver disease. Dig. Liver Dis. 2015, 47, 4–11. [Google Scholar] [CrossRef] [PubMed]
- Park, H.-S.; Jang, J.E.; Ko, M.S.; Woo, S.H.; Kim, B.J.; Kim, H.S.; Park, H.S.; Park, I.-S.; Koh, E.H.; Lee, K.-U. Statins Increase Mitochondrial and Peroxisomal Fatty Acid Oxidation in the Liver and Prevent Non-Alcoholic Steatohepatitis in Mice. Diabetes Metab. J. 2016, 40, 376. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.-Y.; Jang, H.-J.; Yang, Y.R.; Park, K.-I.; Seo, J.; Shin, I.-W.; Jeon, T.-I.; Ahn, S.; Suh, P.-G.; Osborne, T.F.; et al. SREBP-2/PNPLA8 axis improves non-alcoholic fatty liver disease through activation of autophagy. Sci. Rep. 2016, 6, 35732. [Google Scholar] [CrossRef]
- Wang, W.; Zhao, C.; Zhou, J.; Zhen, Z.; Wang, Y.; Shen, C. Simvastatin Ameliorates Liver Fibrosis via Mediating Nitric Oxide Synthase in Rats with Non-Alcoholic Steatohepatitis-Related Liver Fibrosis. PLoS ONE 2013, 8, e76538. [Google Scholar] [CrossRef] [PubMed]
- Hyogo, H.; Yamagishi, S.; Maeda, S.; Kimura, Y.; Ishitobi, T.; Chayama, K. Atorvastatin improves disease activity of nonalcoholic steatohepatitis partly through its tumour necrosis factor-α-lowering property. Dig. Liver Dis. 2012, 44, 492–496. [Google Scholar] [CrossRef] [PubMed]
- Samy, W.; Hassanian, M.A. Paraoxonase-1 activity, malondialdehyde and glutathione peroxidase in non-alcoholic fatty liver disease and the effect of atorvastatin. Arab J. Gastroenterol. 2011, 12, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Orime, K.; Shirakawa, J.; Togashi, Y.; Tajima, K.; Inoue, H.; Nagashima, Y.; Terauchi, Y. Lipid-lowering agents inhibit hepatic steatosis in a non-alcoholic steatohepatitis-derived hepatocellular carcinoma mouse model. Eur. J. Pharmacol. 2016, 772, 22–32. [Google Scholar] [CrossRef] [PubMed]
- Tziomalos, K.; Athyros, V.G.; Paschos, P.; Karagiannis, A. Nonalcoholic fatty liver disease and statins. Metab. Clin. Exp. 2015, 64, 1215–1223. [Google Scholar] [CrossRef] [PubMed]
- Okada, Y.; Yamaguchi, K.; Nakajima, T.; Nishikawa, T.; Jo, M.; Mitsumoto, Y.; Kimura, H.; Nishimura, T.; Tochiki, N.; Yasui, K.; et al. Rosuvastatin ameliorates high-fat and high-cholesterol diet-induced nonalcoholic steatohepatitis in rats. Liver Int. 2013, 33, 301–311. [Google Scholar] [CrossRef] [PubMed]
- Musso, G.; Cassader, M.; Gambino, R. Cholesterol-lowering therapy for the treatment of nonalcoholic fatty liver disease: An update. Curr. Opin. Lipidol. 2011, 22, 489–496. [Google Scholar] [CrossRef] [PubMed]
- Ji, G.; Zhao, X.; Leng, L.; Liu, P.; Jiang, Z. Comparison of Dietary Control and Atorvastatin on High Fat Diet Induced Hepatic Steatosis and Hyperlipidemia in Rats. Lipids Health Dis. 2011, 10, 23. [Google Scholar] [CrossRef] [PubMed]
- Otis, J.P.; Farber, S.A. High-fat Feeding Paradigm for Larval Zebrafish: Feeding, Live Imaging, and Quantification of Food Intake. J. Vis. Exp. 2016, 116. [Google Scholar] [CrossRef] [PubMed]
- Marza, E.; Barthe, C.; André, M.; Villeneuve, L.; Hélou, C.; Babin, P.J. Developmental expression and nutritional regulation of a zebrafish gene homologous to mammalian microsomal triglyceride transfer protein large subunit: Regulation of mtp Expression in Zebrafish. Dev. Dyn. 2005, 232, 506–518. [Google Scholar] [CrossRef] [PubMed]
- Tingaud-Sequeira, A.; Ouadah, N.; Babin, P.J. Zebrafish obesogenic test: A tool for screening molecules that target adiposity. J. Lipid Res. 2011, 52, 1765–1772. [Google Scholar] [CrossRef] [PubMed]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: An open-source platform for biological-image analysis. Nat. Methods 2012, 9, 676–682. [Google Scholar] [CrossRef] [PubMed]
- de Lara Rodrigues, E.; Fanta, E. Liver histopathology of the fish Brachydanio rerio hamilton-buchman after acute exposure to sublethal levels of the organophosphate dimethoate 500. Rev. Bras. Zool. 1998, 15, 441–450. [Google Scholar]
- Caldwell, S.; Ikura, Y.; Dias, D.; Isomoto, K.; Yabu, A.; Moskaluk, C.; Pramoonjago, P.; Simmons, W.; Scruggs, H.; Rosenbaum, N.; et al. Hepatocellular ballooning in NASH. J. Hepatol. 2010, 53, 719–723. [Google Scholar] [CrossRef] [PubMed]
| Gene | Official Full Name | Accession Number | Forward Primer | Reverse Primer |
|---|---|---|---|---|
| actb2 | Actin, beta 2 | NM_181601.4 | 5′-TTCTCTTAAGTCGACAACCCCC-3′ | 5′-TACCAACCATGACACCCTGAT-3′ |
| 18s | - | NR_145818.1 | 5′-TTACCCCAGGCTCGGAAAAC-3′ | 5′-CGGGAAGGTCTTTGAACCCA-3′ |
| gapdh | Glyceraldehyde-3-phosphate dehydrogenase | NM_001115114.1 | 5′-GAGGCTTCTCACAAACGAGGA-3′ | 5′-TGGCCACGATCTCCACTTTC-3′ |
| crp | C-reactive protein | NM_001045860.1 | 5′-CATTAGAGGCTACCGAAGGTTT-3′ | 5’-GACTCAGGGGTTTTTCAGGATA-3′ |
| nfkb3 (rela) | Nuclear factor kappa B | NM_001001839.2 | 5′-CAACGACACCACGAAAACG-3′ | 5′-CGTCAGGAATCTTGAATGGGT-3′ |
| il1b | Interleukin 1β | NM_212844.2 | 5′-GAACAGAATGAAGCACATCAAACC-3′ | 5′-ACGGCACTGAATCCACCAC-3′ |
| il6 | Interleukun6 | NM_001261449.1 | 5’-TCAACTTCTCCAGCGTGATG-3′ | 5’-TCTTTCCCTCTTTTCCTCCTG-3′ |
| casp3a | Caspase 3a | NM_131877.3 | 5’-TCGGTTCTCGCTGTTGAAGG-3′ | 5′-GTCTCCGTATCCGCATGTCC-3′ |
| tgfb1a | Transforming growth factor β 1a | NM_182873.1 | 5′-GGAAGGCAACACAAGGTGGA-3′ | 5′-GGCTTACTTATCAATCCCGACT-3′ |
| tfa | Transferrin a | NM_001291499.1 | 5’-GAAAATCCCAGAGTCAGCCA-3’ | 5′-TTCATCTCCAACAGCCTTCC-3′ |
| zgc163022 | Ferric chelate reductase 1 | NM_001089557.2 | 5’-CCCAGAGGCTGCTGTTTATT-3’ | 5′-GCCGTGATTAGGCATCATAGAG-3′ |
| nrf2a | Nuclear factor (eruthroid-derived 2)-like2 | NM_182889.1 | 5′-TCGGGTTTGTCCCTAGATG-3′ | 5′-AGGTTTGGAGTGTCCGCTA-3′ |
| gstp1 | Glutathione S-transferase pi | NM_131734.3 | 5′-ACACACTCACATACTTCGCA-3′ | 5′-GTCGCCCTTCATCCACTCTT-3′ |
| nqo1 | NADPH dehydrogenase, quinone 1 | NM_001204272.1 | 5′-TCTGACAAAGAAAGGCTACAAAGTC-3′ | 5′-ATACACAAAGTGCTCGGGATT-3′ |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Imran, M.; Sergent, O.; Tête, A.; Gallais, I.; Chevanne, M.; Lagadic-Gossmann, D.; Podechard, N. Membrane Remodeling as a Key Player of the Hepatotoxicity Induced by Co-Exposure to Benzo[a]pyrene and Ethanol of Obese Zebrafish Larvae. Biomolecules 2018, 8, 26. https://doi.org/10.3390/biom8020026
Imran M, Sergent O, Tête A, Gallais I, Chevanne M, Lagadic-Gossmann D, Podechard N. Membrane Remodeling as a Key Player of the Hepatotoxicity Induced by Co-Exposure to Benzo[a]pyrene and Ethanol of Obese Zebrafish Larvae. Biomolecules. 2018; 8(2):26. https://doi.org/10.3390/biom8020026
Chicago/Turabian StyleImran, Muhammad, Odile Sergent, Arnaud Tête, Isabelle Gallais, Martine Chevanne, Dominique Lagadic-Gossmann, and Normand Podechard. 2018. "Membrane Remodeling as a Key Player of the Hepatotoxicity Induced by Co-Exposure to Benzo[a]pyrene and Ethanol of Obese Zebrafish Larvae" Biomolecules 8, no. 2: 26. https://doi.org/10.3390/biom8020026
APA StyleImran, M., Sergent, O., Tête, A., Gallais, I., Chevanne, M., Lagadic-Gossmann, D., & Podechard, N. (2018). Membrane Remodeling as a Key Player of the Hepatotoxicity Induced by Co-Exposure to Benzo[a]pyrene and Ethanol of Obese Zebrafish Larvae. Biomolecules, 8(2), 26. https://doi.org/10.3390/biom8020026




