Improving the Activity of Trp-Rich Antimicrobial Peptides by Arg/Lys Substitutions and Changing the Length of Cationic Residues
Abstract
1. Introduction
2. Results
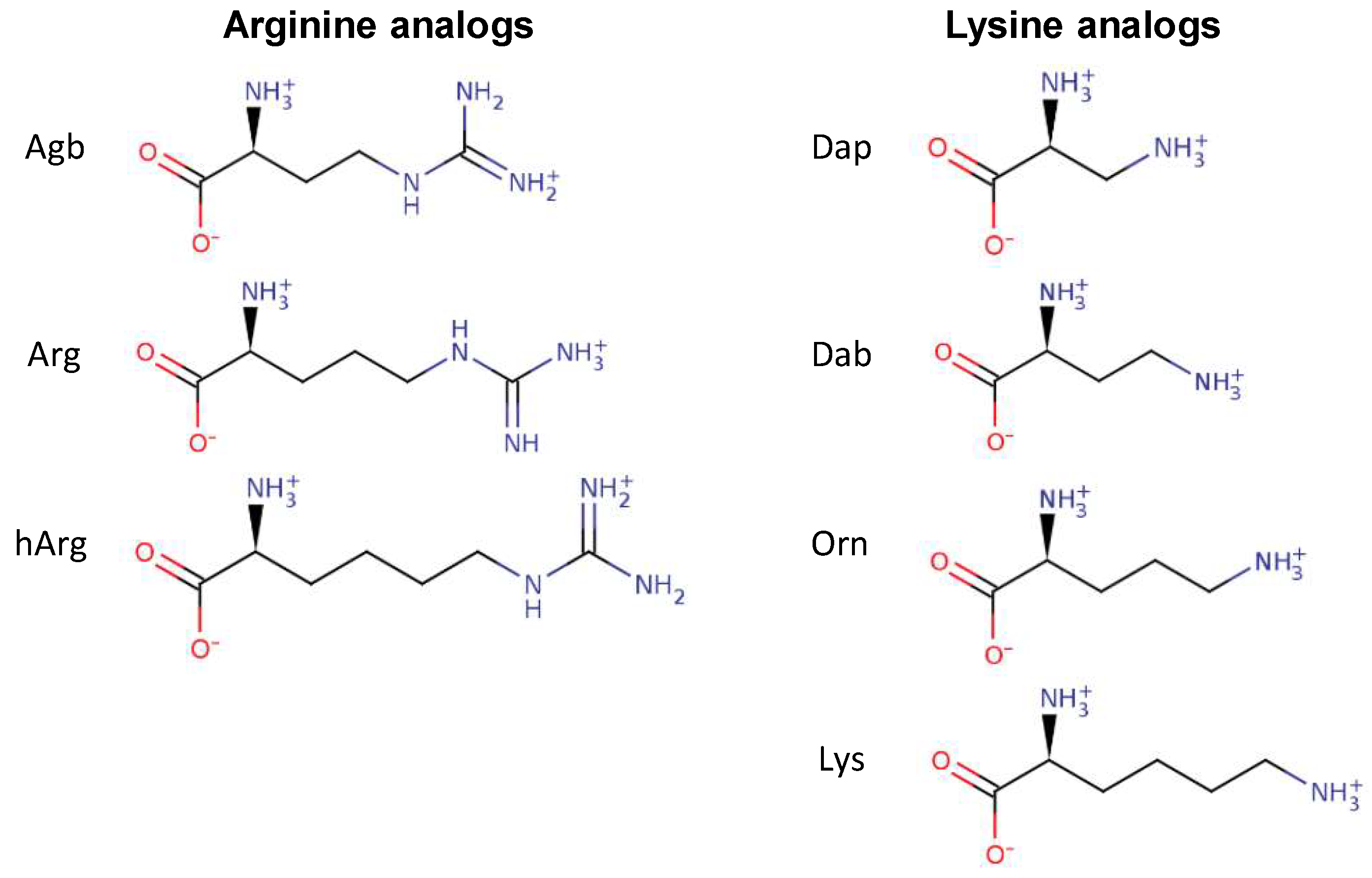
2.1. Peptide Design
2.2. Antibacterial Activity
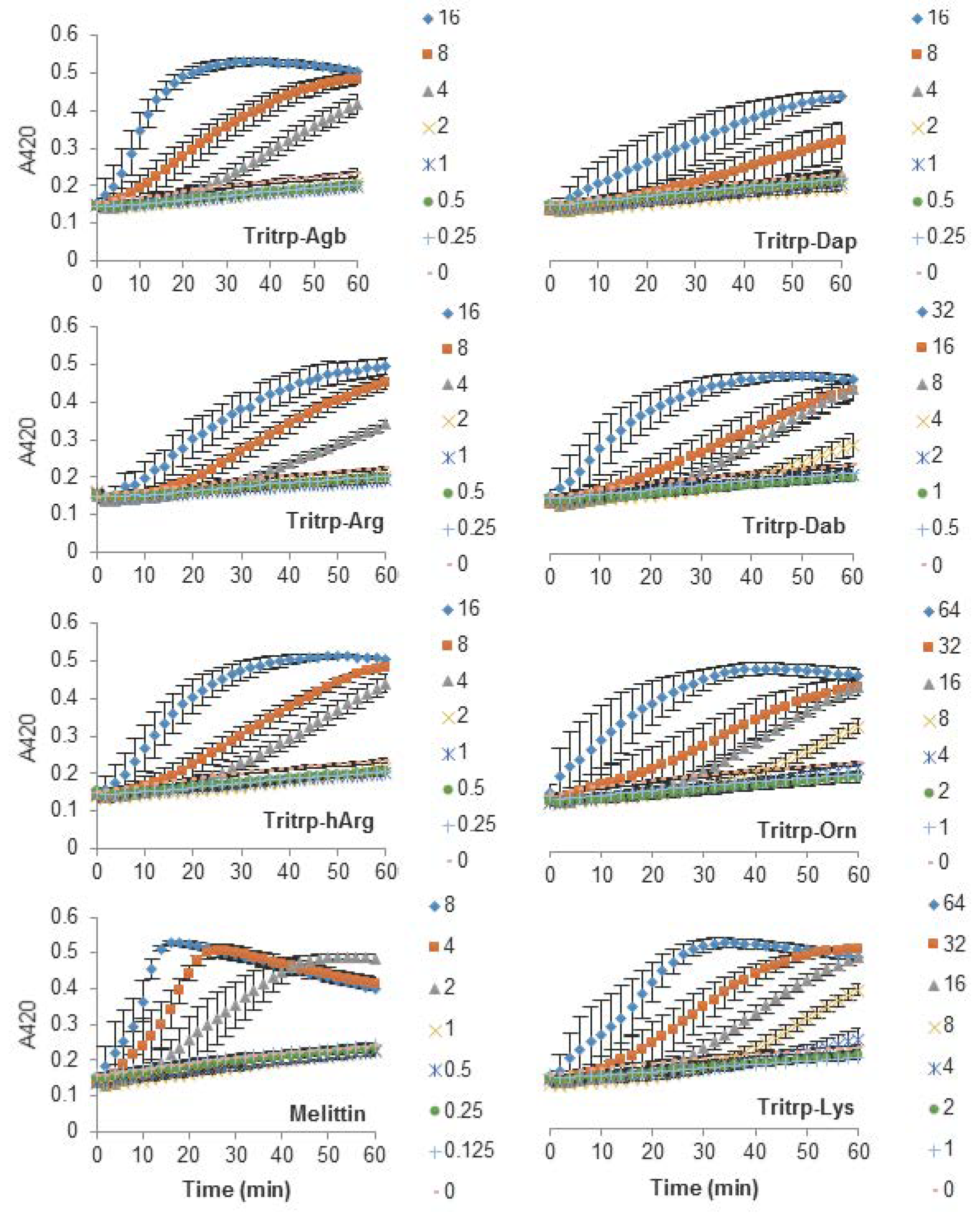
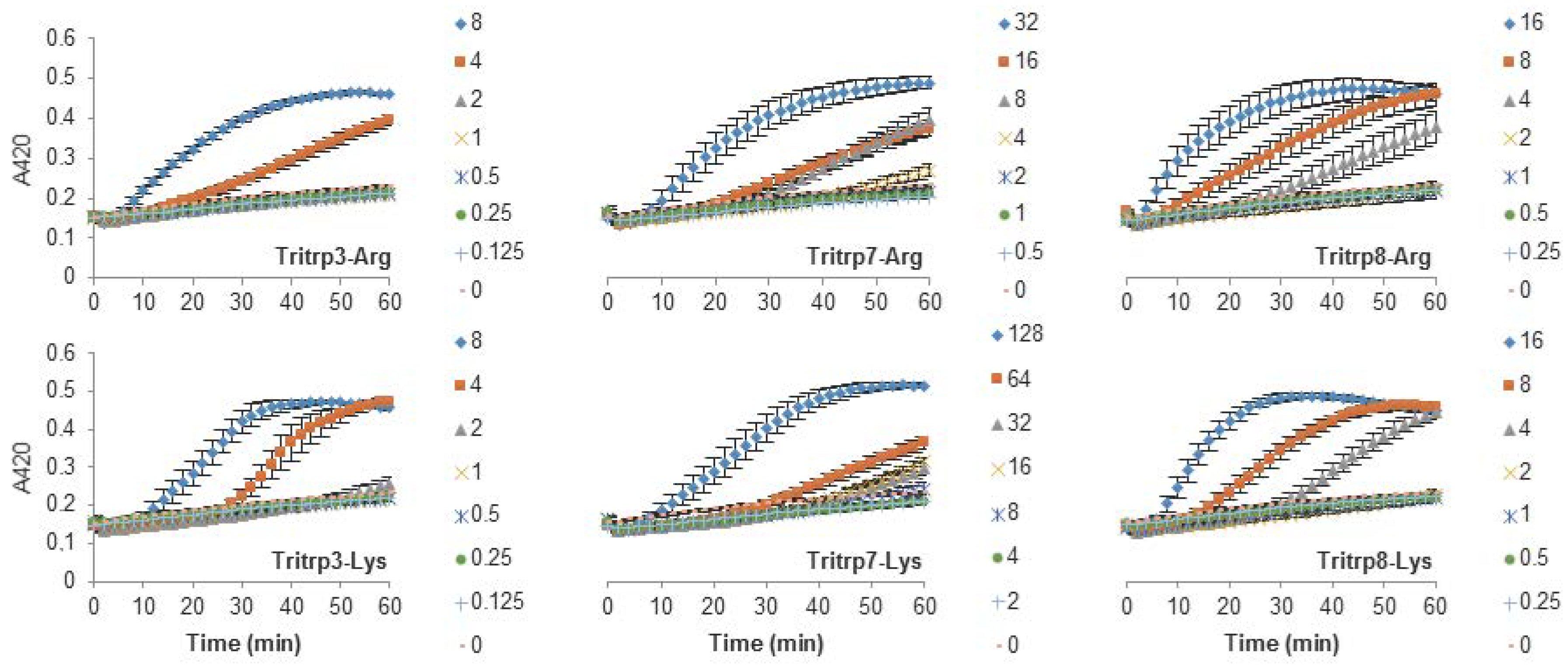
2.3. Escherichia coli Inner Membrane Permeabilization
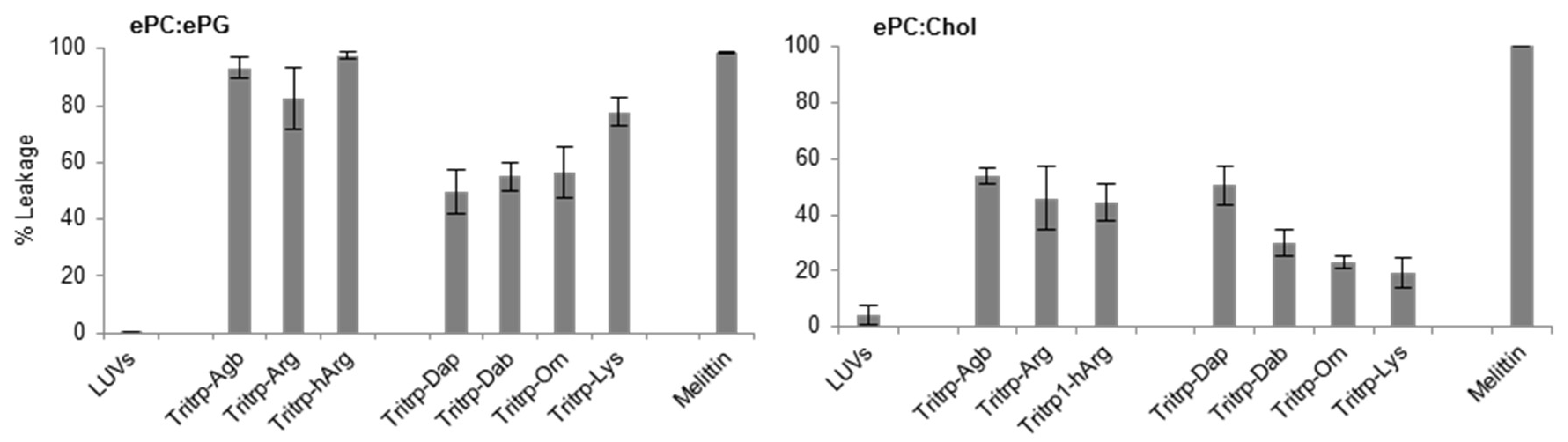
2.4. Large Unilamellar Vesicle Membrane Permeabilization
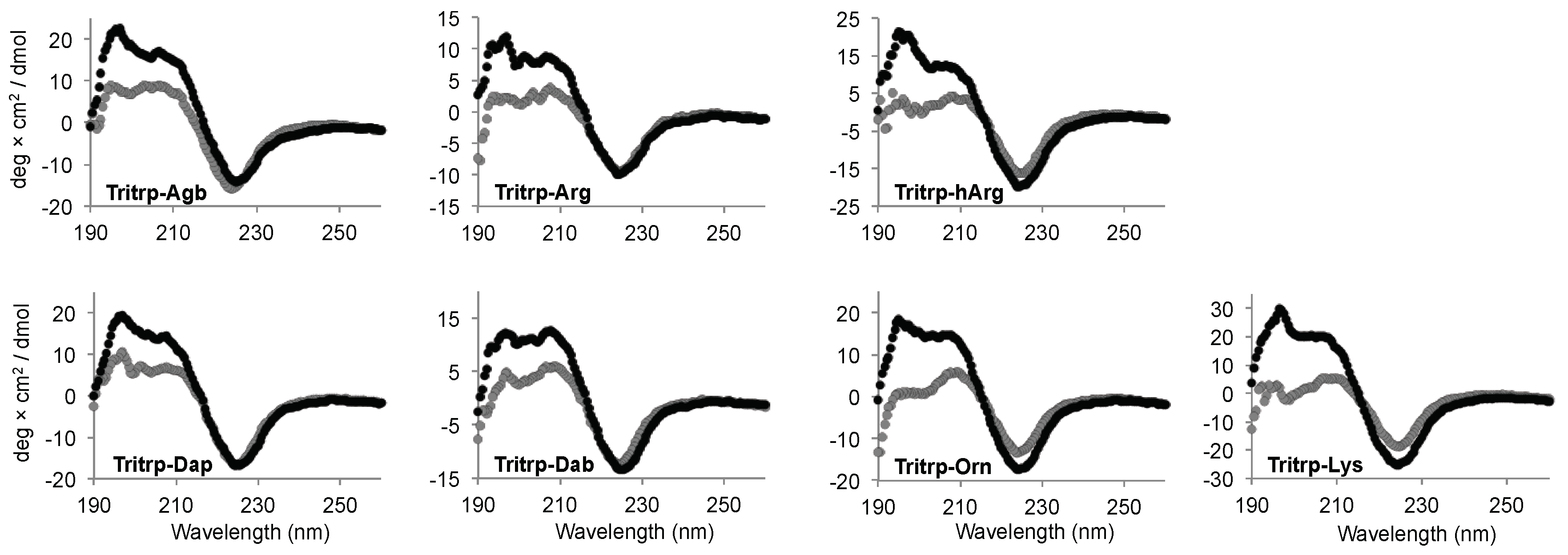
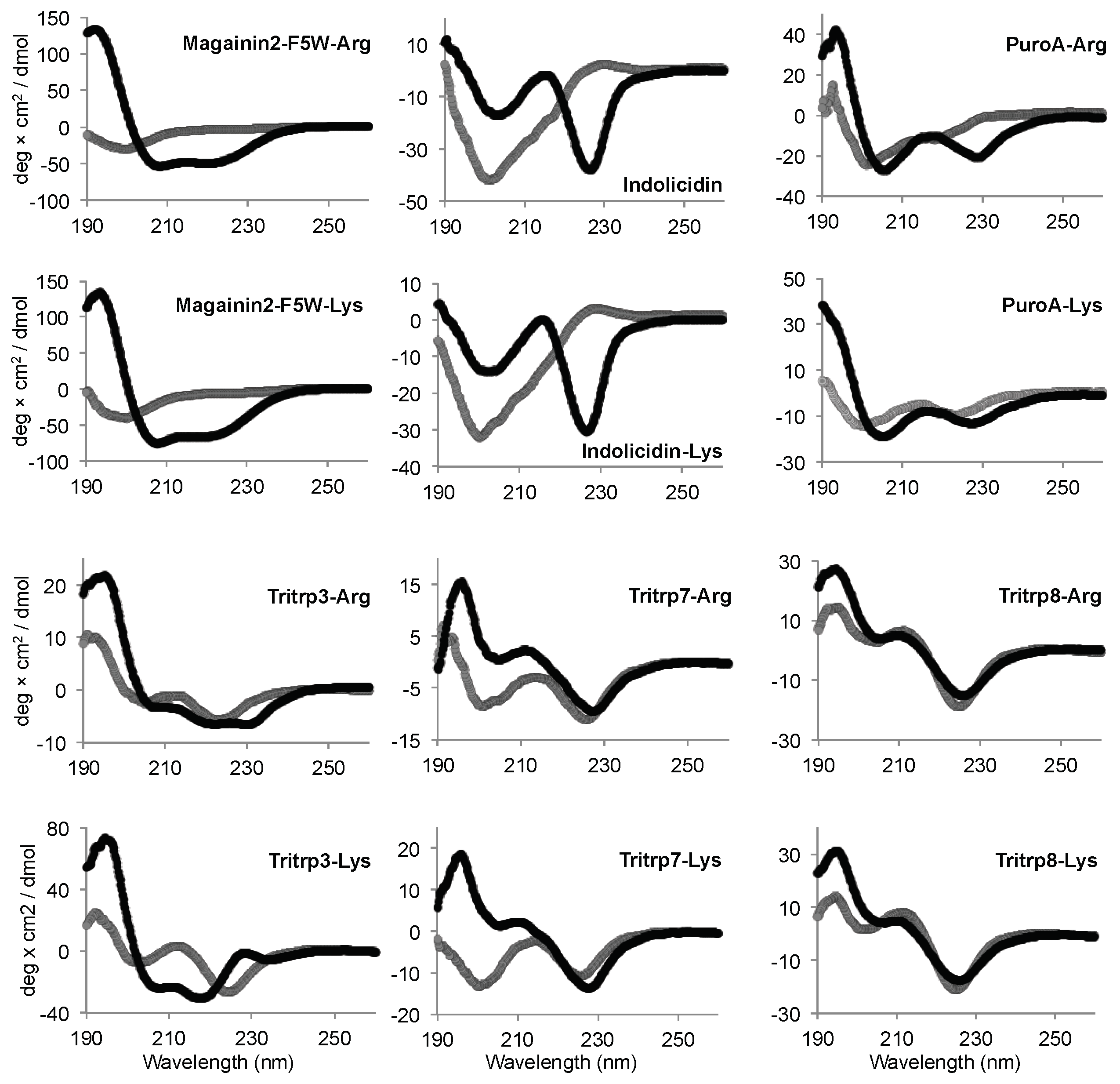
2.5. Secondary Structure Determination
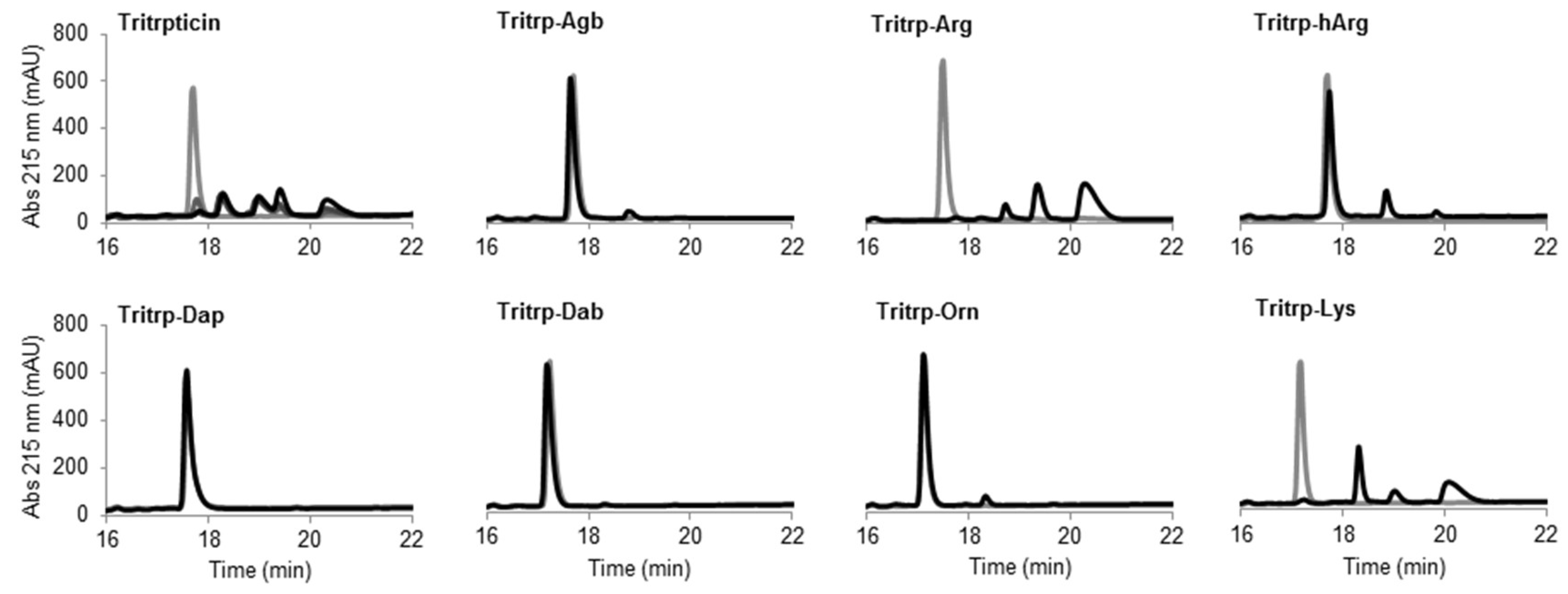
2.6. Trypsin Degradation
3. Discussion
4. Materials and Methods
4.1. Materials, Peptides, and Bacterial Strains
4.2. Antibacterial Activity
4.3. Escherichia coli ML35p Inner Membrane Permeabilization
4.4. Large Unilamellar Vesicles and Calcein Leakage
4.5. Far-UV Circular Dichroism
4.6. Trypsin Hydrolysis of Antimicrobial Peptides
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Epand, R.M.; Vogel, H.J. Diversity of antimicrobial peptides and their mechanisms of action. Biochim. Biophys. Acta 1999, 1462, 11–28. [Google Scholar] [CrossRef]
- Nguyen, L.T.; Haney, E.F.; Vogel, H.J. The expanding scope of antimicrobial peptide structures and their modes of action. Trends Biotechnol. 2011, 29, 464–472. [Google Scholar] [CrossRef] [PubMed]
- Jenssen, H.; Hamill, P.; Hancock, R.E.W. Peptide antimicrobial agents. Clin. Microbiol. Rev. 2006, 19, 491–511. [Google Scholar] [CrossRef] [PubMed]
- Zasloff, M. Antimicrobial peptides of multicellular organisms. Nature 2002, 415, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Steinstraesser, L.; Kraneburg, U.; Jacobsen, F.; Al-Benna, S. Host defense peptides and their antimicrobial-immunomodulatory duality. Immunobiology 2011, 216, 322–333. [Google Scholar] [CrossRef] [PubMed]
- Yeung, A.T.Y.; Gellatly, S.L.; Hancock, R.E.W. Multifunctional cationic host defence peptides and their clinical applications. Cell. Mol. Life Sci. 2011, 68, 2161–2176. [Google Scholar] [CrossRef] [PubMed]
- Hancock, R.E.W.; Haney, E.F.; Gill, E.E. The immunology of host defence peptides: beyond antimicrobial activity. Nat. Rev. Immunol. 2016, 16, 321–334. [Google Scholar] [CrossRef] [PubMed]
- Nicolas, P. Multifunctional host defense peptides: Intracellular-targeting antimicrobial peptides. FEBS J. 2009, 276, 6483–6496. [Google Scholar] [CrossRef] [PubMed]
- Lohner, K.; Sevcsik, E.; Pabst, G. Liposome-based biomembrane mimetic systems: Implications for lipid-peptide interactions. In Advances in Planar Lipid Bilayers and Liposomes, 1st ed.; Liu, A.L., Ed.; Elsevier Inc.: Waltham, MA, USA, 2008; Volume 6, pp. 103–132. [Google Scholar]
- Yeaman, M.R.; Yount, N.Y. Mechanisms of antimicrobial peptide action and resistance. Pharmacol. Rev. 2003, 55, 27–55. [Google Scholar] [CrossRef] [PubMed]
- Deslouches, B.; Hasek, M.L.; Craigo, J.K.; Steckbeck, J.D.; Montelaro, R.C. Comparative functional properties of engineered cationic antimicrobial peptides consisting exclusively of tryptophan and either lysine or arginine. J. Med. Microbiol. 2016, 65, 554–565. [Google Scholar] [CrossRef] [PubMed]
- Chan, D.I.; Prenner, E.J.; Vogel, H.J. Tryptophan- and arginine-rich antimicrobial peptides: structures and mechanisms of action. Biochim. Biophys. Acta 2006, 1758, 1184–1202. [Google Scholar] [CrossRef] [PubMed]
- Karstad, R.; Isaksen, G.; Wynendaele, E.; Guttormsen, Y.; De Spiegeleer, B.; Brandsdal, B.; Svendsen, J.S.; Svenson, J. Targeting the S1 and S3 subsite of trypsin with unnatural cationic amino acids generates antimicrobial peptides with potential for oral administration. J. Med. Chem. 2012, 55, 6294–6305. [Google Scholar] [CrossRef] [PubMed]
- Werle, M.; Bernkop-Schnurch, A. Strategies to improve plasma half life time of peptide and protein drugs. Amino Acids 2006, 30, 351–367. [Google Scholar] [CrossRef] [PubMed]
- Bagheri, M.; Hancock, R.E.W. High-performance liquid chromatogray and mass spectrometry-based design of proteolytically stable antimicrobial peptides. In Antimicrobial Peptides: Methods and Protocols, 1st ed.; Hansen, P.R., Ed.; Springer: New York, NY, USA, 2017; Volume 1548, pp. 61–71. [Google Scholar]
- Gentilucci, L.; De Marco, R.; Cerisoli, L. Chemical modifications designed to improve peptide stability: Incorporation of non-natural amino acids, pseudo-peptide bonds, and cyclization. Curr. Pharm. Des. 2010, 16, 3185–3203. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.T.; Chau, J.K.; Perry, N.A.; de Boer, L.; Zaat, S.A.; Vogel, H.J. Serum stabilities of short tryptophan- and arginine-rich antimicrobial peptide analogs. PLoS ONE 2010, 5, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Bottger, R.; Hoffmann, R.; Knappe, D. Differential stability of therapeutic peptides with different proteolytic cleavage sites in blood, plasma and serum. PLoS ONE 2017, 12, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Berthold, N.; Czihal, P.; Fritsche, S.; Sauer, U.; Schiffer, G.; Knappe, D.; Alber, G.; Hoffmann, R. Novel apidaecin 1b analogs with superior serum stabilities for treatment of infections by Gram-negative pathogens. Antimicrob. Agents Chemother. 2013, 57, 402–409. [Google Scholar] [CrossRef] [PubMed]
- Henklein, P.; Bruckdorfer, T. Shorter arginine homologues to stabilize peptides towards tryptic digestion. Chem. Today 2008, 26, 12–15. [Google Scholar]
- Bagheri, M.; Arasteh, S.; Haney, E.F.; Hancock, R.E.W. Tryptic stability of synthetic bactenecin derivatives is determined by the side chain length of cationic residues and the peptide conformation. J. Med. Chem. 2016, 59, 3079–3086. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, N.W.; Wong, G.C.L. Antimicrobial peptides and induced membrane curvature: Geometry, coordination chemistry, and molecular engineering. Curr. Opin. Solid State Mater. Sci. 2013, 17, 151–163. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Vorobyov, I.; Allen, T.W. The different interactions of lysine and arginine side chains with lipid membranes. J. Phys. Chem. B 2013, 117, 11906–11920. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.T.; de Boer, L.; Zaat, S.A.; Vogel, H.J. Investigating the cationic side chains of the antimicrobial peptide tritrpticin: Hydrogen bonding properties govern its membrane-disruptive activities. Biochim. Biophys. Acta 2011, 1808, 2297–2303. [Google Scholar] [CrossRef] [PubMed]
- Schibli, D.J.; Nguyen, L.T.; Kernaghan, S.D.; Rekdal, Ø.; Vogel, H.J. Structure–function analysis of tritrpticin analogs: potential relationships between antimicrobial activities, model membrane interactions, and their micelle-bound NMR structures. Biophys. J. 2006, 91, 4413–4426. [Google Scholar] [CrossRef] [PubMed]
- Gesell, J.; Zasloff, M.; Opella, S.J. Two-dimensional 1H-NMR experiments show that the 23-residue magainin antibiotic peptide is an α-helix in dodecylphosphocholine micelles, sodium dodecylsulfate micelles, and trifluoroethanol/water solution. J. Biomol. NMR 1997, 9, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Matsuzaki, K.; Murase, O.; Fujii, N.; Miyajima, K. An antimicrobial peptide, magainin2, induced rapid flip-flop of phospholipids coupled with pore formation and peptide translocation. Biochemistry 1996, 35, 11361–11368. [Google Scholar] [CrossRef] [PubMed]
- Rozek, A.; Friedrich, C.L.; Hancock, R.E. Structure of the bovine antimicrobial peptide indolicidin bound to dodecylphosphocholine and sodium dodecyl sulfate micelles. Biochemistry 2000, 39, 15765–15774. [Google Scholar] [CrossRef] [PubMed]
- Subbalakshmi, C.; Sitaram, N. Mechanism of antimicrobial action of indolicidin. FEMS Microbiol. Lett. 1998, 160, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Jing, W.; Demcoe, A.R.; Vogel, H.J. Conformation of a bactericidal domain of Puroindoline a: structure and mechanism of action of a 13-residue antimicrobial peptide. J. Bacteriol. 2003, 185, 4938–4947. [Google Scholar] [CrossRef] [PubMed]
- Haney, E.F.; Petersen, A.P.; Lau, C.K.; Jing, W.; Storey, D.G.; Vogel, H.J. Mechanism of action of puroindoline derived tryptophan-rich antimicrobial peptides. Biochim. Biophys. Acta 2013, 1828, 1802–1813. [Google Scholar] [CrossRef] [PubMed]
- Arias, M.; Vogel, H.J. Fluorescence and absorbance spectroscopy methods to study membrane perturbations by antimicrobial host defense peptides. In Antimicrobial Peptides: Methods and Protocols, Methods in Molecular Biology, 1st ed.; Hansen, P.R., Ed.; Springer Science + Business Media: New York, NY, USA, 2017; Volume 1548, pp. 141–157. [Google Scholar]
- Andrushchenko, V.V.; Vogel, H.J.; Prenner, E.J. Solvent-dependent structure of two tryptophan-rich antimicrobial peptides and their analogs studied by FTIR and CD spectroscopy. Biochim. Biophys. Acta 2006, 1758, 1596–1608. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Falla, T.J.; Karunaratne, D.N.; Hancock, R.E.W. Mode of action of the antimicrobial peptide indolicidin. J. Biol. Chem. 1996, 271, 19298–19303. [Google Scholar] [CrossRef] [PubMed]
- Ludtke, S.J.; He, K.; Heller, W.T.; Harroun, T.A.; Yang, L.; Huang, H.W. Membrane pores induced by magainin. Biochemistry 1996, 35, 13723–13728. [Google Scholar] [CrossRef] [PubMed]
- Selsted, M.E.; Novotny, M.J.; Morris, W.L.; Tang, Y.; Smith, W.; Cullor, J.S. Indolicidin, a novel bactericidal tridecapeptide amide from neutrophils. J. Biol. Chem. 1992, 267, 4292–4295. [Google Scholar] [PubMed]
- Staubitz, P.; Peschel, A.; Nieuwenhuizen, W.F.; Otto, M.; Jung, G.; Jack, R.W.; Gotz, F. Structure-function relationships in the tryptophan-rich, antimicrobial peptide indolicidin. J. Pept. Sci. 2001, 564, 552–564. [Google Scholar] [CrossRef] [PubMed]
- Wenk, M.R.; Seelig, J. Magainin 2 amide interaction with lipid membranes: Calorimetric detection of peptide binding and pore formation. Biochemistry 1998, 37, 3909–3916. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.T.; Shin, S.Y.; Kim, J.I. Interaction mode of a symmetric Trp-rich undeca peptide PST11-RK with lipid bilayers. FEBS Lett. 2007, 581, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Tokunaga, Y.; Niidome, T.; Hatakeyama, T.; Aoyagi, H. Antibacterial activity of bactenecin 5 fragments and their interaction with phospholipid membranes. J. Pept. Sci. 2001, 7, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Rajasekaran, G.; Kamalakannan, R.; Shin, S.Y. Enhancement of the anti-inflammatory activity of temporin-1Tl-derived antimicrobial peptides by tryptophan, arginine and lysine substitutions. J. Pept. Sci. 2015, 21, 779–785. [Google Scholar] [CrossRef] [PubMed]
- Muhle, S.A.; Tam, J.P. Design of Gram-negative selective antimicrobial peptides. Biochemistry 2001, 40, 5777–5785. [Google Scholar] [CrossRef] [PubMed]
- Rice, A.; Wereszczynski, J. Probing the disparate effects of arginine and lysine residues on antimicrobial peptide/bilayer association. Biochim. Biophys. Acta 2017, 1859, 1941–1950. [Google Scholar] [CrossRef] [PubMed]
- Chin, W.; Zhong, G.; Pu, Q.; Yang, C.; Lou, W.; De Sessions, P.F.; Periaswamy, B.; Lee, A.; Liang, Z.C.; Ding, X.; Gao, S.; et al. A macromolecular approach to eradicate multidrug resistant bacterial infections while mitigating drug resistance onset. Nat. Commun. 2018, 9, 917. [Google Scholar] [CrossRef] [PubMed]
- Kohn, E.M.; Shirley, D.J.; Arotsky, L.; Picciano, A.M.; Ridgway, Z.; Urban, M.W.; Carone, B.R.; Caputo, G.A. Role of cationic side chains in the antimicrobial activity of C18G. Molecules 2018, 23, 329. [Google Scholar] [CrossRef] [PubMed]
- Palermo, E.F.; Vemparala, S.; Kuroda, K. Cationic spacer arm design strategy for control of antimicrobial activity and conformation of amphiphilic methacrylate random copolymers. Biomacromolecules 2012, 13, 1632–1641. [Google Scholar] [CrossRef] [PubMed]
- Gasteiger, E.; Hoogland, C.; Gattiker, A.; Duvaud, S.; Wilkins, M.R.; Appel, R.D.; Bairoch, A. Protein identification and analysis tools on the ExPASy server. In The Proteomics Protocols Handbook, 1st ed.; Walker, J.M., Ed.; Humana Press: Totowa, NJ, USA, 2005; Volume 1, pp. 571–607. [Google Scholar]
- Wiegand, I.; Hilpert, K.; Hancock, R.E.W. Agar and broth dilution methods to determine the minimal inhibitory concentration (MIC) of antimicrobial substances. Nat. Protoc. 2008, 3, 163–175. [Google Scholar] [CrossRef] [PubMed]
- Epand, R.F.; Pollard, J.E.; Wright, J.O.; Savage, P.B.; Epand, R.M. Depolarization, bacterial membrane composition, and the antimicrobial action of ceragenins. Antimicrob. Agents Chemother. 2010, 54, 3708–3713. [Google Scholar] [CrossRef] [PubMed]
- Arias, M.; Jensen, K.V.; Nguyen, L.T.; Storey, D.G.; Vogel, H.J. Hydroxy-tryptophan containing derivatives of tritrpticin: Modification of antimicrobial activity and membrane interactions. Biochim. Biophys. Acta 2014, 1848, 277–288. [Google Scholar] [CrossRef] [PubMed]
- Ames, B.N.; Neufeld, E.F.; Ginsberg, V. Assay of inorganic phosphate, total phosphate and phosphatases. Methods Enzymol. 1966, 8, 115–118. [Google Scholar]

| Peptide | Sequence | Net Charge a | 3D Structure b | Mechanism of Action | Ref. |
|---|---|---|---|---|---|
| Magainin2 | GIGKFLHSAKKFGKAFVGEIMNS | +3 | α-helix (I2–N22) | Membranolytic | [26,27] |
| Indolicidin | ILPWKWPWWPWRR-NH2 | +4 | β-turns (K5, W8) | Intracellular target | [28,29] |
| PuroA | FPVTWKWWKWWKG-NH2 | +5 | β-turn (T4–W7) helical turn (W8–W10) | Membranolytic | [30,31] |
| Tritrp1 c | VRRFPWWWPFLRR-NH2 | +5 | β-turn (P5–W8) helical turn (W9–R12) | Membranolytic | [25] |
| Tritrp3 | VRRFAWWWAFLRR-NH2 | +5 | α-helix (F4–L11) | Membranolytic | [25] |
| Tritrp7 | VRRFAWWWPFLRR-NH2 | +5 | α-helix-like (F4–L11) | Membranolytic | [25] |
| Tritrp8 | VRRFPWWWAFLRR-NH2 | +5 | β-turn (P5–W8) helical turn (W9–R12) | Membranolytic | [25] |
| Peptide | Sequence | MIC (µM) |
|---|---|---|
| Magainin2-F5W–Arg | GIGRFLHSARRFGRAFVGEIMNS | 8 |
| Magainin2-F5W–Lys | GIGKFLHSAKKFGKAFVGEIMNS | 16 |
| Indolicidin | ILPWKWPWWPWRR-NH2 | 8 |
| Indolicidin–Lys | ILPWKWPWWPWKK-NH2 | 16 |
| PuroA–Arg | FPVTWRWWRWWRG-NH2 | 8–16 |
| PuroA–Lys | FPVTWKWWKWWKG-NH2 | 8 |
| Tritrp3–Arg | V R R FAWWWAFL R R -NH2 | 2 |
| Tritrp3–Lys | V K K FAWWWAFL K K -NH2 | 2 |
| Tritrp7–Arg | V R R FAWWWPFL R R -NH2 | 8 |
| Tritrp7–Lys | V K K FAWWWPFL K K -NH2 | 32 |
| Tritrp8–Arg | V R R FPWWWAFL R R -NH2 | 2–4 |
| Tritrp8–Lys | V K K FPWWWAFL K K -NH2 | 4 |
| Tritrp–Agb | V (Agb) (Agb) FPWWWPFL (Agb) (Agb) -NH2 | 4 |
| Tritrp–Arg | V R R FPWWWPFL R R -NH2 | 2–4 |
| Tritrp–hArg | V (hArg)(hArg)FPWWWPFL(hArg)(hArg) -NH2 | 4 |
| Tritrp–Dap | V (Dap) (Dap) FPWWWPFL (Dap) (Dap) -NH2 | 4 |
| Tritrp–Dab | V (Dab) (Dab) FPWWWPFL (Dab) (Dab) -NH2 | 4–8 |
| Tritrp–Orn | V (Orn) (Orn) FPWWWPFL (Orn) (Orn) -NH2 | 16 |
| Tritrp–Lys | V K K FPWWWPFL K K -NH2 | 16 |
| Melittin | GIGAVLKVLTTGLPALISWIKRKRQQ-NH2 | 2 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arias, M.; Piga, K.B.; Hyndman, M.E.; Vogel, H.J. Improving the Activity of Trp-Rich Antimicrobial Peptides by Arg/Lys Substitutions and Changing the Length of Cationic Residues. Biomolecules 2018, 8, 19. https://doi.org/10.3390/biom8020019
Arias M, Piga KB, Hyndman ME, Vogel HJ. Improving the Activity of Trp-Rich Antimicrobial Peptides by Arg/Lys Substitutions and Changing the Length of Cationic Residues. Biomolecules. 2018; 8(2):19. https://doi.org/10.3390/biom8020019
Chicago/Turabian StyleArias, Mauricio, Kathlyn B. Piga, M. Eric Hyndman, and Hans J. Vogel. 2018. "Improving the Activity of Trp-Rich Antimicrobial Peptides by Arg/Lys Substitutions and Changing the Length of Cationic Residues" Biomolecules 8, no. 2: 19. https://doi.org/10.3390/biom8020019
APA StyleArias, M., Piga, K. B., Hyndman, M. E., & Vogel, H. J. (2018). Improving the Activity of Trp-Rich Antimicrobial Peptides by Arg/Lys Substitutions and Changing the Length of Cationic Residues. Biomolecules, 8(2), 19. https://doi.org/10.3390/biom8020019




