Role of microRNAs in Alcohol-Induced Multi-Organ Injury
Abstract
:1. Introduction to microRNA
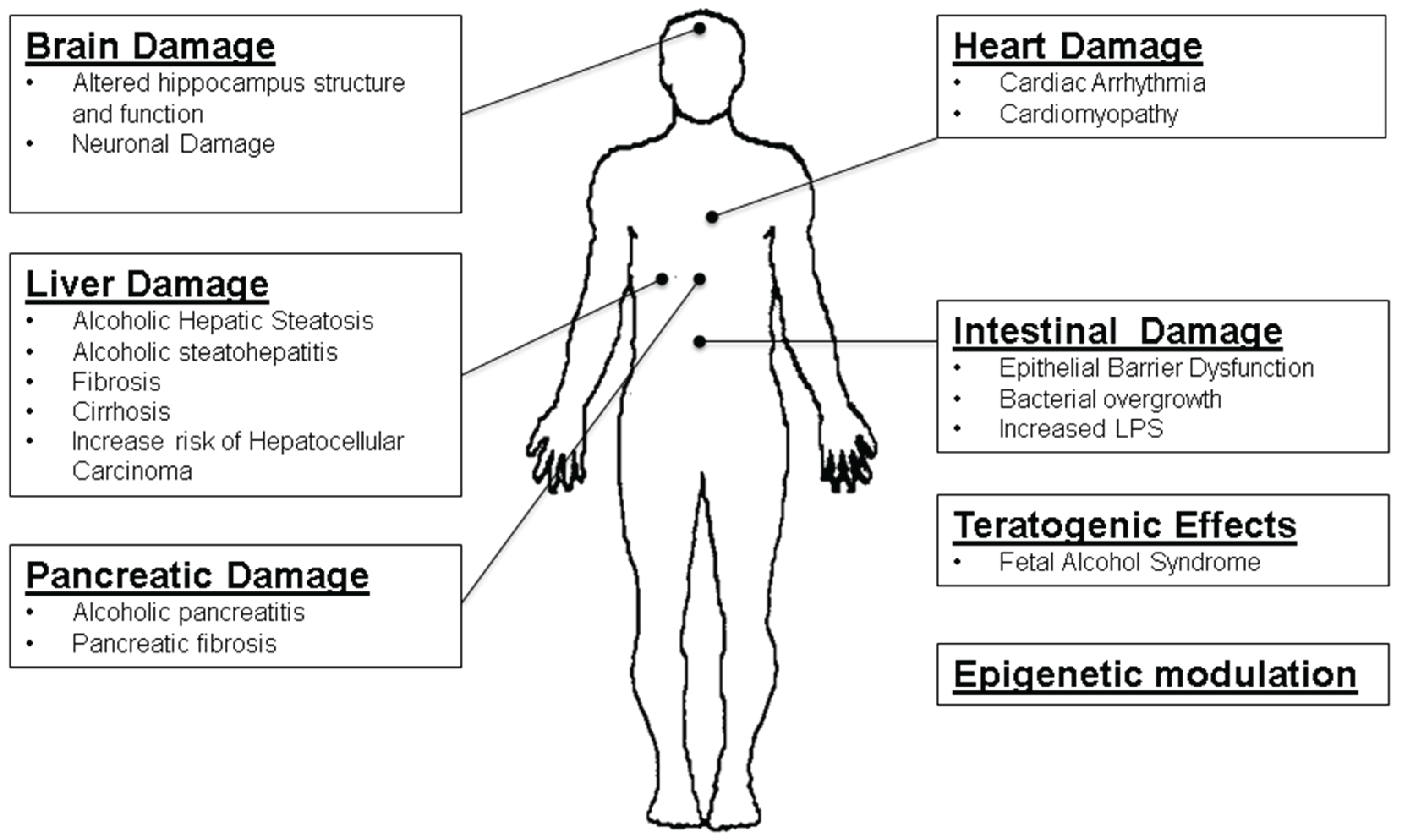
2. Alcohol Metabolism and Pancreatitis
Altered microRNAs in Alcoholic Pancreatitis
3. Circulating microRNAs in ALD
3.1. Ethanol Alters Monocyte Function via Circulating microRNAs
3.2. Ethanol Abuse and Bone Marrow Depression
3.3. Pitfalls of Circulating microRNA Analysis
4. Liver microRNAs in Alcoholic Liver Disease
4.1. Liver miR-34a in ALD
4.2. Role of miR-21 in ALD
5. Gut-Liver Axis in the Pathogenesis of ALD
5.1. Altered Intestinal microRNAs in ALD
5.2. microRNAs and Intestinal Tight Junctions in ALD
6. Brain Injury and microRNAs with Alcohol Consumption
6.1. Binge Drinking Alters Hippocampal microRNA
6.2. Neuronal Damage and microRNA
6.3. Long Noncoding RNA and Ethanol
6.4. Teratogenic Effect of Ethanol
6.5. Maternal Ethanol Consumption
6.6. Paternal Ethanol Consumption and Epigenetics
7. Alcoholic Cardiomyopathy
Role of microRNAs in Alcohol-Induced Muscle Injury
8. Conclusions
| Affected Organ | Upregulated microRNA | Targets | References |
|---|---|---|---|
| Pancreas | miR-21 | Connective tissue growth factor | [18,19] |
| miR-199a-3p | [18] | ||
| miR-211 | [18] | ||
| Liver | miR-21 | Btg2, IL-6, STAT3 | [24,36,45,46,47] |
| miR-33 | VLDL | [37,47] | |
| miR-34a | Caspase-2, SIRT1 | [24] | |
| miR-132 | p65 subunit of NFkB | [34] | |
| miR-137 | [24] | ||
| miR-155 | TNF-α, MCL1,SIRT1 | [25,32,35] | |
| miR-185 | [84] | ||
| miR-199a-3p | [84] | ||
| miR-214 | [84] | ||
| miR-217 | SIRT1, Lipin-1 | [39] | |
| miR-320 | [23] | ||
| miR-409-5p | [24] | ||
| miR-486 | [23] | ||
| miR-490 | [84] | ||
| miR-509-3p | [24] | ||
| miR-705 | [23] | ||
| miR-882 | [24] | ||
| miR-1224 | [23] | ||
| Intestine | miR-122 | TNF-α | [71,72] |
| miR-155 | FASLG, DR5, TNF-α, Regb, Src homology domain 2-containing inosital phosphatase | [36,67,68,69] | |
| miR-212 | ZO-1 | [70] | |
| Circulation | miR-122 | TLR4, NADPH Oxidase | [82] |
| miR-155 | [36,50,78,82] | ||
| miR-185 | [84] | ||
| miR-199a-3p | [84] | ||
| miR-214 | [84] | ||
| miR-490 | [84] | ||
| Brain | Let 7 Family members | [109] | |
| miR-9 | [120] | ||
| miR-10a, miR-10b | TBX5, Homeobox | [97,99,120,121,122,123] | |
| miR-21 | |||
| miR-26b | CB1R | [130] | |
| miR-29b | SP1, RAX, Phospho-PKR | [91] | |
| miR-30a-3p | [120] | ||
| miR-34a | [110] | ||
| miR-145 | [120] | ||
| miR-146a | [106] | ||
| miR-132 | [69] | ||
| miR-152 | [120] | ||
| miR-155 | TNF-α | [69] | |
| miR-194 | [106] | ||
| miR-203 | [106] | ||
| miR-302b | Bcl2, Cyclin D2 | [110] | |
| miR-369 | [106] | ||
| miR-497 | Bcl2 | [110] | |
| Heart | miR-138 | [149] | |
| miR-485-5p | [149] | ||
| miR-506 | [149] | ||
| miR-512-5p | [149] | ||
| miR-548-3p | [149] | ||
| miR-4262 | [149] |
| Affected Organ | Downregulated microRNA | Targets | References |
|---|---|---|---|
| Pancreas | miR-148a | [18] | |
| miR-802 | [18] | ||
| Liver | Let-7a | [24] | |
| Let-7b | [24] | ||
| Let-7g | [24] | ||
| miR-27b | [23] | ||
| miR-122 | [24] | ||
| miR-127 | [24] | ||
| miR-27b | [23] | ||
| miR-181a,b | [24] | ||
| miR-182 | [23] | ||
| miR-183 | [23] | ||
| miR-192 | [24] | ||
| miR-199a-3p | Endothelin-1, HIF1-α | [23,25,32] | |
| miR-200a | [23] | ||
| miR-214 | [23] | ||
| miR-322 | [23] | ||
| miR-871 | [24] | ||
| Brain | miR-9 | [94,123,124,125,126] | |
| miR-21 | [79,80,81] | ||
| miR-26a | BDNF, SIRT1 | [97] | |
| miR-29a | [91] | ||
| miR-29b | [91] | ||
| miR-133 | [91] | ||
| miR-153 | [79,80,81] | ||
| miR-335 | [126] | ||
| miR-495 | BDNF, SIRT1 | [97] | |
| Heart | miR-30a | Beclin 1 | [148] |
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ALD | alcoholic liver disease; |
| ALT | alanine amino transferase; |
| BCL2 | B-cell lymphoma 2; |
| MCL1 | myeloid cell lymphoma 1; |
| BDNR | brain-derived neurotropic factor; |
| CB1R | cannabinoid receptor 1; |
| CCL2 | chemokine (c-c motif) ligand 2; |
| CYP2E1 | cytochrome p450-2E1 oxidase; |
| DR5 | death receptor 5; |
| DGCR8 | DiGeorge syndrome critical region 8; |
| RBC | red blood cells; |
| ROS | reactive oxygen species; |
| FAEE | fatty acid ethyl ester; |
| FASLG | FAS ligand G; |
| FoxM1 | forkhead box protein M1; |
| LCFA | long chain fatty acids; |
| IL | interleukin; |
| lncRNA | long non-coding RNA; |
| LPS | lipopolysaccharide; |
| MALAT1 | metastasis associated lung adenocarcinoma transcript 1; |
| PGC-1α | peroxisome proliferator-activated receptor 1 alpha; |
| RAR | retinoic acid receptor; |
| STAT3 | signal transducer and activator of transcription 3; |
| SCFA | short chain fatty acids; |
| SOCS1 | suppressor of cytokine signaling1; |
| SIRT | Sirtuins; |
| SREBP | sterol regulatory element-binding protein; |
| TLR4 | toll-like receptor 4; |
| TGF-β | transforming growth factor-β; |
| TNF-α | tumor necrosis factor-α; |
| TBX5 | T-box transcription factor 5; |
| UTR | un-translated region; |
| VLDL | very low density lipoproteins; |
| ZO-1 | zona occludens. |
References
- Bartel, D.P. MicroRNAs: Target recognition and regulatory functions. Cell 2009, 136, 215–233. [Google Scholar] [CrossRef] [PubMed]
- Ruby, J.G.; Jan, C.H.; Bartel, D.P. Intronic microRNA precursors that bypass drosha processing. Nature 2007, 448, 83–86. [Google Scholar] [CrossRef] [PubMed]
- Bohnsack, M.T.; Czaplinski, K.; Gorlich, D. Exportin 5 is a RanGTP-dependent dsRNA-binding protein that mediates nuclear export of pre-miRNAs. RNA 2004, 10, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Natarajan, S.K.; Rasineni, K.; Ganesan, M.; Feng, D.; McVicker, B.L.; McNiven, M.A.; Osna, N.A.; Mott, J.L.; Casey, C.A.; Kharbanda, K.K. Structure, function and metabolism of hepatic and adipose tissue lipid droplets: Implications in alcoholic liver disease. Curr. Mol. Pharmacol. 2015, in press. [Google Scholar]
- Miranda, R.C.; Pietrzykowski, A.Z.; Tang, Y.; Sathyan, P.; Mayfield, D.; Keshavarzian, A.; Sampson, W.; Hereld, D. Micrornas: Master regulators of ethanol abuse and toxicity? Alcohol. Clin. Exp. Res. 2010, 34, 575–587. [Google Scholar] [CrossRef] [PubMed]
- Osna, N.A.; Donohue, T.M., Jr. Cyp2e1-catalyzed alcohol metabolism: Role of oxidant generation in interferon signaling, antigen presentation and autophagy. Subcell. Biochem. 2013, 67, 177–197. [Google Scholar] [PubMed]
- Werner, J.; Saghir, M.; Warshaw, A.L.; Lewandrowski, K.B.; Laposata, M.; Iozzo, R.V.; Carter, E.A.; Schatz, R.J.; Fernandez-Del Castillo, C. Alcoholic pancreatitis in rats: Injury from nonoxidative metabolites of ethanol. Am. J. Physiol. Gastrointest. Liver Physiol. 2002, 283, G65–G73. [Google Scholar] [CrossRef] [PubMed]
- Criddle, D.N. The role of fat and alcohol in acute pancreatitis: A dangerous liaison. Pancreatology 2015, 15, S6–S12. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Booth, D.M.; Cane, M.C.; Chvanov, M.; Javed, M.A.; Elliott, V.L.; Armstrong, J.A.; Dingsdale, H.; Cash, N.; Li, Y.; et al. Fatty acid ethyl ester synthase inhibition ameliorates ethanol-induced Ca2+-dependent mitochondrial dysfunction and acute pancreatitis. Gut 2014, 63, 1313–1324. [Google Scholar] [CrossRef] [PubMed]
- Laposata, M. Fatty acid ethyl esters: Short-term and long-term serum markers of ethanol intake. Clin. Chem. 1997, 43, 1527–1534. [Google Scholar] [PubMed]
- Wurst, F.M.; Alexson, S.; Wolfersdorf, M.; Bechtel, G.; Forster, S.; Alling, C.; Aradottir, S.; Jachau, K.; Huber, P.; Allen, J.P.; et al. Concentration of fatty acid ethyl esters in hair of alcoholics: Comparison to other biological state markers and self reported-ethanol intake. Alcohol. Alcohol. 2004, 39, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Elamin, E.; Masclee, A.; Juuti-Uusitalo, K.; van Ijzendoorn, S.; Troost, F.; Pieters, H.J.; Dekker, J.; Jonkers, D. Fatty acid ethyl esters induce intestinal epithelial barrier dysfunction via a reactive oxygen species-dependent mechanism in a three-dimensional cell culture model. PLoS ONE 2013, 8, e58561. [Google Scholar] [CrossRef] [PubMed]
- Nichitailo, M.E.; Kravchenko, D.A.; Medvetskii, E.B.; Shpon’ka, I.S.; Savitskaia, I.M. Inhibition of pancreatic stellate cell activation by lisinopril for prevention fibrogenesis in experimental chronic alcoholic pancreatitis. Klin. khirurhiia 2012, 7, 52–56. (In Russian) [Google Scholar]
- Cyriac, J.; Mahadevan, P.; Augustine, P.; Ramesh, H.; Koshy, A. Stellate cell activation in tropical calcific pancreatitis compared to alcoholic pancreatitis, adenocarcinoma of pancreas and normal pancreas. JOP 2012, 13, 376–386. [Google Scholar] [PubMed]
- Apte, M.V.; Wilson, J.S. Stellate cell activation in alcoholic pancreatitis. Pancreas 2003, 27, 316–320. [Google Scholar] [CrossRef] [PubMed]
- Vonlaufen, A.; Wilson, J.S.; Pirola, R.C.; Apte, M.V. Role of alcohol metabolism in chronic pancreatitis. Alcohol. Res. Health 2007, 30, 48–54. [Google Scholar] [PubMed]
- Suda, K.; Shiotsu, H.; Nakamura, T.; Akai, J.; Nakamura, T. Pancreatic fibrosis in patients with chronic alcohol abuse: Correlation with alcoholic pancreatitis. Am. J. Gastroenterol. 1994, 89, 2060–2062. [Google Scholar] [PubMed]
- Charrier, A.; Chen, R.; Chen, L.; Kemper, S.; Hattori, T.; Takigawa, M.; Brigstock, D.R. Connective tissue growth factor (CCN2) and microRNA-21 are components of a positive feedback loop in pancreatic stellate cells (PSC) during chronic pancreatitis and are exported in PSC-derived exosomes. J. Cell Commun. Signal. 2014, 8, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Charrier, A.L.; Brigstock, D.R. Connective tissue growth factor production by activated pancreatic stellate cells in mouse alcoholic chronic pancreatitis. Lab. Investig. 2010, 90, 1179–1188. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Charrier, A.; Zhou, Y.; Chen, R.; Yu, B.; Agarwal, K.; Tsukamoto, H.; Lee, L.J.; Paulaitis, M.E.; Brigstock, D.R. Epigenetic regulation of connective tissue growth factor by microRNA-214 delivery in exosomes from mouse or human hepatic stellate cells. Hepatology 2014, 59, 1118–1129. [Google Scholar] [CrossRef] [PubMed]
- Arroyo, J.D.; Chevillet, J.R.; Kroh, E.M.; Ruf, I.K.; Pritchard, C.C.; Gibson, D.F.; Mitchell, P.S.; Bennett, C.F.; Pogosova-Agadjanyan, E.L.; Stirewalt, D.L.; et al. Argonaute2 complexes carry a population of circulating microRNAs independent of vesicles in human plasma. Proc. Natl. Acad. Sci. USA 2011, 108, 5003–5008. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Zhang, S.; Weber, J.; Baxter, D.; Galas, D.J. Export of microRNAs and microRNA-protective protein by mammalian cells. Nucleic Acids Res. 2010, 38, 7248–7259. [Google Scholar] [CrossRef] [PubMed]
- Turchinovich, A.; Weiz, L.; Langheinz, A.; Burwinkel, B. Characterization of extracellular circulating microrna. Nucleic Acids Res. 2011, 39, 7223–7233. [Google Scholar] [CrossRef] [PubMed]
- Turchinovich, A.; Burwinkel, B. Distinct AGO1 and AGO2 associated miRNA profiles in human cells and blood plasma. RNA Biol. 2012, 9, 1066–1075. [Google Scholar] [CrossRef] [PubMed]
- Turchinovich, A.; Weiz, L.; Burwinkel, B. Isolation of circulating microRNA associated with RNA-binding protein. Methods Mol. Biol. 2013, 1024, 97–107. [Google Scholar] [PubMed]
- Valadi, H.; Ekstrom, K.; Bossios, A.; Sjostrand, M.; Lee, J.J.; Lotvall, J.O. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat. Cell Biol. 2007, 9, 654–659. [Google Scholar] [CrossRef] [PubMed]
- Bala, S.; Petrasek, J.; Mundkur, S.; Catalano, D.; Levin, I.; Ward, J.; Alao, H.; Kodys, K.; Szabo, G. Circulating microRNAs in exosomes indicate hepatocyte injury and inflammation in alcoholic, drug-induced, and inflammatory liver diseases. Hepatology 2012, 56, 1946–1957. [Google Scholar] [CrossRef] [PubMed]
- Pirola, C.J.; Fernandez Gianotti, T.; Castano, G.O.; Mallardi, P.; San Martino, J.; Mora Gonzalez Lopez Ledesma, M.; Flichman, D.; Mirshahi, F.; Sanyal, A.J.; Sookoian, S. Circulating microRNA signature in non-alcoholic fatty liver disease: From serum non-coding rnas to liver histology and disease pathogenesis. Gut 2015, 64, 800–812. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.P.; Jin, X.; Xiang, Z.; Chen, S.H.; Li, Y.M. Circulating microRNAs as potential biomarkers for alcoholic steatohepatitis. Liver Int. 2013, 33, 1257–1265. [Google Scholar] [CrossRef] [PubMed]
- Pritchard, C.C.; Kroh, E.; Wood, B.; Arroyo, J.D.; Dougherty, K.J.; Miyaji, M.M.; Tait, J.F.; Tewari, M. Blood cell origin of circulating microRNAs: A cautionary note for cancer biomarker studies. Cancer Prev. Res. 2012, 5, 492–497. [Google Scholar] [CrossRef] [PubMed]
- Natarajan, S.K.; Smith, M.A.; Wehrkamp, C.J.; Mohr, A.M.; Mott, J.L. MicroRNA function in human diseases. Med. Epigenetics 2013, 1, 106–115. [Google Scholar] [CrossRef]
- Willeit, P.; Zampetaki, A.; Dudek, K.; Kaudewitz, D.; King, A.; Kirkby, N.S.; Crosby-Nwaobi, R.; Prokopi, M.; Drozdov, I.; Langley, S.R.; et al. Circulating microRNAs as novel biomarkers for platelet activation. Circ. Res. 2013, 112, 595–600. [Google Scholar] [CrossRef] [PubMed]
- Curtis, A.M.; FitzGerald, G.A. Platelet microRNAs: Novy Mir* or mired in human complexity? Circ. Res. 2013, 112, 576–578. [Google Scholar] [CrossRef] [PubMed]
- Mukamal, K.J.; Massaro, J.M.; Ault, K.A.; Mittleman, M.A.; Sutherland, P.A.; Lipinska, I.; Levy, D.; D’Agostino, R.B.; Tofler, G.H. Alcohol consumption and platelet activation and aggregation among women and men: The framingham offspring study. Alcohol. Clin. Exp. Res. 2005, 29, 1906–1912. [Google Scholar] [CrossRef] [PubMed]
- McMullen, M.R.; Cocuzzi, E.; Hatzoglou, M.; Nagy, L.E. Chronic ethanol exposure increases the binding of hur to the tnfalpha 3'-untranslated region in macrophages. J. Biol. Chem. 2003, 278, 38333–38341. [Google Scholar] [CrossRef] [PubMed]
- Degre, D.; Lemmers, A.; Gustot, T.; Ouziel, R.; Trepo, E.; Demetter, P.; Verset, L.; Quertinmont, E.; Vercruysse, V.; Le Moine, O.; et al. Hepatic expression of CCL2 in alcoholic liver disease is associated with disease severity and neutrophil infiltrates. Clin. Exp. Immunol. 2012, 169, 302–310. [Google Scholar] [CrossRef] [PubMed]
- Lemmers, A.; Moreno, C.; Gustot, T.; Marechal, R.; Degre, D.; Demetter, P.; de Nadai, P.; Geerts, A.; Quertinmont, E.; Vercruysse, V.; et al. The interleukin-17 pathway is involved in human alcoholic liver disease. Hepatology 2009, 49, 646–657. [Google Scholar] [CrossRef] [PubMed]
- Szabo, G.; Satishchandran, A. MicroRNAs in alcoholic liver disease. Semin. Liver Dis. 2015, 35, 36–42. [Google Scholar] [PubMed]
- Momen-Heravi, F.; Bala, S.; Kodys, K.; Szabo, G. Exosomes derived from alcohol-treated hepatocytes horizontally transfer liver specific miRNA-122 and sensitize monocytes to LPS. Sci. Rep. 2015. [Google Scholar] [CrossRef] [PubMed]
- Gimsing, P.; Melgaard, B.; Andersen, K.; Vilstrup, H.; Hippe, E. Vitamin B-12 and folate function in chronic alcoholic men with peripheral neuropathy and encephalopathy. J. Nutr. 1989, 119, 416–424. [Google Scholar] [PubMed]
- Gonzalez-Reimers, E.; Alvisa-Negrin, J.; Santolaria-Fernandez, F.; Candelaria Martin-Gonzalez, M.; Hernandez-Betancor, I.; Fernandez-Rodriguez, C.M.; Vina-Rodriguez, J.; Gonzalez-Diaz, A. Vitamin D and nutritional status are related to bone fractures in alcoholics. Alcohol. Alcohol. 2011, 46, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Larramona, G.; Lucendo, A.J.; Gonzalez-Delgado, L. Alcoholic liver disease and changes in bone mineral density. Rev. Esp. Enferm. Dig. 2013, 105, 609–621. [Google Scholar] [CrossRef] [PubMed]
- Malik, P.; Gasser, R.W.; Kemmler, G.; Moncayo, R.; Finkenstedt, G.; Kurz, M.; Fleischhacker, W.W. Low bone mineral density and impaired bone metabolism in young alcoholic patients without liver cirrhosis: A cross-sectional study. Alcohol. Clin. Exp. Res. 2009, 33, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Giuliani, N.; Girasole, G.; Vescovi, P.P.; Passeri, G.; Pedrazzoni, M. Ethanol and acetaldehyde inhibit the formation of early osteoblast progenitors in murine and human bone marrow cultures. Alcohol. Clin. Exp. Res. 1999, 23, 381–385. [Google Scholar] [CrossRef] [PubMed]
- Johnson, T.L.; Gaddini, G.; Branscum, A.J.; Olson, D.A.; Caroline-Westerlind, K.; Turner, R.T.; Iwaniec, U.T. Effects of chronic heavy alcohol consumption and endurance exercise on cancellous and cortical bone microarchitecture in adult male rats. Alcohol. Clin. Exp. Res. 2014, 38, 1365–1372. [Google Scholar] [CrossRef] [PubMed]
- Pai, M.; Zacharoulis, D.; Milicevic, M.N.; Helmy, S.; Jiao, L.R.; Levicar, N.; Tait, P.; Scott, M.; Marley, S.B.; Jestice, K.; et al. Autologous infusion of expanded mobilized adult bone marrow-derived CD34+ cells into patients with alcoholic liver cirrhosis. Am. J. Gastroenterol. 2008, 103, 1952–1958. [Google Scholar] [CrossRef] [PubMed]
- Saito, T.; Okumoto, K.; Haga, H.; Nishise, Y.; Ishii, R.; Sato, C.; Watanabe, H.; Okada, A.; Ikeda, M.; Togashi, H.; et al. Potential therapeutic application of intravenous autologous bone marrow infusion in patients with alcoholic liver cirrhosis. Stem Cells Dev. 2011, 20, 1503–1510. [Google Scholar] [CrossRef] [PubMed]
- Spahr, L.; Chalandon, Y.; Terraz, S.; Kindler, V.; Rubbia-Brandt, L.; Frossard, J.L.; Breguet, R.; Lanthier, N.; Farina, A.; Passweg, J.; et al. Autologous bone marrow mononuclear cell transplantation in patients with decompensated alcoholic liver disease: A randomized controlled trial. PLoS ONE 2013, 8, e53719. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Reimers, E.; Quintero-Platt, G.; Rodriguez-Rodriguez, E.; Martinez-Riera, A.; Alvisa-Negrin, J.; Santolaria-Fernandez, F. Bone changes in alcoholic liver disease. World J. Hepatol. 2015, 7, 1258–1264. [Google Scholar] [CrossRef] [PubMed]
- Tang, P.; Xiong, Q.; Ge, W.; Zhang, L. The role of microRNAs in osteoclasts and osteoporosis. RNA Biol. 2014, 11, 1355–1363. [Google Scholar] [CrossRef] [PubMed]
- Weilner, S.; Skalicky, S.; Salzer, B.; Keider, V.; Wagner, M.; Hildner, F.; Gabriel, C.; Dovjak, P.; Pietschmann, P.; Grillari-Voglauer, R.; et al. Differentially circulating miRNAs after recent osteoporotic fractures can influence osteogenic differentiation. Bone 2015, 79, 43–51. [Google Scholar] [CrossRef] [PubMed]
- McDaniel, K.; Herrera, L.; Zhou, T.; Francis, H.; Han, Y.; Levine, P.; Lin, E.; Glaser, S.; Alpini, G.; Meng, F. The functional role of microRNAs in alcoholic liver injury. J. Cell. Mol. Med. 2014, 18, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Roderburg, C.; Luedde, T. Circulating microRNAs as markers of liver inflammation, fibrosis and cancer. J. Hepatol. 2014, 61, 1434–1437. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, P.S.; Parkin, R.K.; Kroh, E.M.; Fritz, B.R.; Wyman, S.K.; Pogosova-Agadjanyan, E.L.; Peterson, A.; Noteboom, J.; O’Briant, K.C.; Allen, A.; et al. Circulating microRNAs as stable blood-based markers for cancer detection. Proc. Natl. Acad. Sci. USA 2008, 105, 10513–10518. [Google Scholar] [CrossRef] [PubMed]
- Marabita, F.; de Candia, P.; Torri, A.; Tegner, J.; Abrignani, S.; Rossi, R.L. Normalization of circulating microRNA expression data obtained by quantitative real-time RT-PCR. Brief. Bioinform. 2015. [Google Scholar] [CrossRef] [PubMed]
- Benz, F.; Roderburg, C.; Vargas Cardenas, D.; Vucur, M.; Gautheron, J.; Koch, A.; Zimmermann, H.; Janssen, J.; Nieuwenhuijsen, L.; Luedde, M.; et al. U6 is unsuitable for normalization of serum miRNA levels in patients with sepsis or liver fibrosis. Exp. Mol. Med. 2013. [Google Scholar] [CrossRef] [PubMed]
- McDonald, J.S.; Milosevic, D.; Reddi, H.V.; Grebe, S.K.; Algeciras-Schimnich, A. Analysis of circulating microRNA: Preanalytical and analytical challenges. Clin. Chem. 2011, 57, 833–840. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.K.; Yeo, J.; Kim, B.; Ha, M.; Kim, V.N. Short structured rnas with low GC content are selectively lost during extraction from a small number of cells. Mol. Cell 2012, 46, 893–895. [Google Scholar] [CrossRef] [PubMed]
- Lal, A.; Thomas, M.P.; Altschuler, G.; Navarro, F.; O’Day, E.; Li, X.L.; Concepcion, C.; Han, Y.C.; Thiery, J.; Rajani, D.K.; et al. Capture of microRNA-bound mrnas identifies the tumor suppressor miR-34a as a regulator of growth factor signaling. PLoS Genet. 2011, 7, e1002363. [Google Scholar] [CrossRef] [PubMed]
- Bala, S.; Szabo, G. MicroRNA signature in alcoholic liver disease. Int. J. Hepatol. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Dolganiuc, A.; Petrasek, J.; Kodys, K.; Catalano, D.; Mandrekar, P.; Velayudham, A.; Szabo, G. MicroRNA expression profile in lieber-decarli diet-induced alcoholic and methionine choline deficient diet-induced nonalcoholic steatohepatitis models in mice. Alcohol. Clin. Exp. Res. 2009, 33, 1704–1710. [Google Scholar] [CrossRef] [PubMed]
- Meng, F.; Glaser, S.S.; Francis, H.; Yang, F.; Han, Y.; Stokes, A.; Staloch, D.; McCarra, J.; Liu, J.; Venter, J.; et al. Epigenetic regulation of miR-34a expression in alcoholic liver injury. Am. J. Pathol. 2012, 181, 804–817. [Google Scholar] [CrossRef] [PubMed]
- Yeligar, S.; Tsukamoto, H.; Kalra, V.K. Ethanol-induced expression of ET-1 and ET-BR in liver sinusoidal endothelial cells and human endothelial cells involves hypoxia-inducible factor-1alpha and microRNA-199. J. Immunol. 2009, 183, 5232–5243. [Google Scholar] [CrossRef] [PubMed]
- Nishiyama, Y.; Goda, N.; Kanai, M.; Niwa, D.; Osanai, K.; Yamamoto, Y.; Senoo-Matsuda, N.; Johnson, R.S.; Miura, S.; Kabe, Y.; et al. HIF-1alpha induction suppresses excessive lipid accumulation in alcoholic fatty liver in mice. J. Hepatol. 2012, 56, 441–447. [Google Scholar] [CrossRef] [PubMed]
- Nath, B.; Szabo, G. Hypoxia and hypoxia inducible factors: Diverse roles in liver diseases. Hepatology 2012, 55, 622–633. [Google Scholar] [CrossRef] [PubMed]
- Paredes-Turrubiarte, G.; Gonzalez-Chavez, A.; Perez-Tamayo, R.; Salazar-Vazquez, B.Y.; Hernandez, V.S.; Garibay-Nieto, N.; Fragoso, J.M.; Escobedo, G. Severity of non-alcoholic fatty liver disease is associated with high systemic levels of tumor necrosis factor alpha and low serum interleukin 10 in morbidly obese patients. Clin. Exp. Med. 2015. [Google Scholar] [CrossRef] [PubMed]
- Barve, S.; Kirpich, I.A.; McClain, C.J. Tumor necrosis factor alpha-induced receptor 1 signaling in alcoholic liver disease: A gut reaction? Hepatology 2015, 61, 754–756. [Google Scholar] [CrossRef] [PubMed]
- McClain, C.J.; Barve, S.; Barve, S.; Deaciuc, I.; Hill, D.B. Tumor necrosis factor and alcoholic liver disease. Alcohol. Clin. Exp. Res. 1998, 22, 248S–252S. [Google Scholar] [CrossRef] [PubMed]
- Tili, E.; Michaille, J.J.; Cimino, A.; Costinean, S.; Dumitru, C.D.; Adair, B.; Fabbri, M.; Alder, H.; Liu, C.G.; Calin, G.A.; et al. Modulation of miR-155 and miR-125b levels following lipopolysaccharide/TNF-alpha stimulation and their possible roles in regulating the response to endotoxin shock. J. Immunol. 2007, 179, 5082–5089. [Google Scholar] [CrossRef] [PubMed]
- Bala, S.; Marcos, M.; Kodys, K.; Csak, T.; Catalano, D.; Mandrekar, P.; Szabo, G. Up-regulation of microRNA-155 in macrophages contributes to increased tumor necrosis factor α (TNFα) production via increased mRNA half-life in alcoholic liver disease. J. Biol. Chem. 2011, 286, 1436–1444. [Google Scholar] [CrossRef] [PubMed]
- Strum, J.C.; Johnson, J.H.; Ward, J.; Xie, H.; Feild, J.; Hester, A.; Alford, A.; Waters, K.M. MicroRNA 132 regulates nutritional stress-induced chemokine production through repression of SirT1. Mol. Endocrinol. 2009, 23, 1876–1884. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Han, C.; Zhang, J.; Song, K.; Wang, Y.; Wu, T. Deletion of miR155 prevents fas-induced liver injury through up-regulation of MCL-1. Am. J. Pathol. 2015, 185, 1033–1044. [Google Scholar] [CrossRef] [PubMed]
- Lippai, D.; Bala, S.; Catalano, D.; Kodys, K.; Szabo, G. Micro-Rna-155 deficiency prevents alcohol-induced serum endotoxin increase and small bowel inflammation in mice. Alcohol. Clin. Exp. Res. 2014, 38, 2217–2224. [Google Scholar] [CrossRef] [PubMed]
- Yin, H.; Hu, M.; Zhang, R.; Shen, Z.; Flatow, L.; You, M. microRNA-217 promotes ethanol-induced fat accumulation in hepatocytes by down-regulating sirt1. J. Biol. Chem. 2012, 287, 9817–9826. [Google Scholar] [CrossRef] [PubMed]
- You, M.; Jogasuria, A.; Taylor, C.; Wu, J. Sirtuin 1 signaling and alcoholic fatty liver disease. Hepatobiliary Surg. Nutr. 2015, 4, 88–100. [Google Scholar] [PubMed]
- Yin, H.; Liang, X.; Jogasuria, A.; Davidson, N.O.; You, M. miR-217 regulates ethanol-induced hepatic inflammation by disrupting sirtuin 1-lipin-1 signaling. Am. J. Pathol. 2015, 185, 1286–1296. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.; Wang, F.; Li, X.; Rogers, C.Q.; Liang, X.; Finck, B.N.; Mitra, M.S.; Zhang, R.; Mitchell, D.A.; You, M. Regulation of hepatic lipin-1 by ethanol: Role of AMP-activated protein kinase/sterol regulatory element-binding protein 1 signaling in mice. Hepatology 2012, 55, 437–446. [Google Scholar] [CrossRef] [PubMed]
- Stow, J.L.; Manderson, A.P.; Murray, R.Z. Snareing immunity: The role of snares in the immune system. Nat. Rev. Immunol. 2006, 6, 919–929. [Google Scholar] [CrossRef] [PubMed]
- Allen, R.M.; Marquart, T.J.; Jesse, J.J.; Baldan, A. Control of very low-density lipoprotein secretion by N-ethylmaleimide-sensitive factor and miR-33. Circ. Res. 2014, 115, 10–22. [Google Scholar] [CrossRef] [PubMed]
- Kharbanda, K.K.; Todero, S.L.; Ward, B.W.; Cannella, J.J., 3rd; Tuma, D.J. Betaine administration corrects ethanol-induced defective VLDL secretion. Mol. Cell. Biochem. 2009, 327, 75–78. [Google Scholar] [CrossRef] [PubMed]
- Liang, X.; Hu, M.; Rogers, C.Q.; Shen, Z.; You, M. Role of sirt1-foxo1 signaling in dietary saturated fat-dependent upregulation of liver adiponectin receptor 2 in ethanol-administered mice. Antioxid. Redox Signal. 2011, 15, 425–435. [Google Scholar] [CrossRef] [PubMed]
- Caito, S.; Rajendrasozhan, S.; Cook, S.; Chung, S.; Yao, H.; Friedman, A.E.; Brookes, P.S.; Rahman, I. SIRT1 is a redox-sensitive deacetylase that is post-translationally modified by oxidants and carbonyl stress. FASEB J. 2010, 24, 3145–3159. [Google Scholar] [CrossRef] [PubMed]
- Song, G.; Sharma, A.D.; Roll, G.R.; Ng, R.; Lee, A.Y.; Blelloch, R.H.; Frandsen, N.M.; Willenbring, H. MicroRNAs control hepatocyte proliferation during liver regeneration. Hepatology 2010, 51, 1735–1743. [Google Scholar] [CrossRef] [PubMed]
- Dippold, R.P.; Vadigepalli, R.; Gonye, G.E.; Hoek, J.B. Chronic ethanol feeding enhances miR-21 induction during liver regeneration while inhibiting proliferation in rats. Am. J. Physiol. Gastrointest. Liver Physiol. 2012, 303, G733–G743. [Google Scholar] [CrossRef] [PubMed]
- Francis, H.; McDaniel, K.; Han, Y.; Liu, X.; Kennedy, L.; Yang, F.; McCarra, J.; Zhou, T.; Glaser, S.; Venter, J.; et al. Regulation of the extrinsic apoptotic pathway by microRNA-21 in alcoholic liver injury. J. Biol. Chem. 2014, 289, 27526–27539. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.K.; Blansit, K.; Kiet, T.; Sherman, A.; Wong, G.; Earle, C.; Bourguignon, L.Y. The inhibition of miR-21 promotes apoptosis and chemosensitivity in ovarian cancer. Gynecol. Oncol. 2014, 132, 739–744. [Google Scholar] [CrossRef] [PubMed]
- Medina, P.P.; Nolde, M.; Slack, F.J. Oncomir addiction in an in vivo model of microRNA-21-induced pre-B-cell lymphoma. Nature 2010, 467, 86–90. [Google Scholar] [CrossRef] [PubMed]
- Spolarics, Z.; Spitzer, J.J.; Wang, J.F.; Xie, J.; Kolls, J.; Greenberg, S. Alcohol administration attenuates LPS-induced expression of inducible nitric oxide synthase in kupffer and hepatic endothelial cells. Biochem. Biophys. Res. Commun. 1993, 197, 606–611. [Google Scholar] [CrossRef] [PubMed]
- Ghoshal, S.; Witta, J.; Zhong, J.; de Villiers, W.; Eckhardt, E. Chylomicrons promote intestinal absorption of lipopolysaccharides. J. Lipid Res. 2009, 50, 90–97. [Google Scholar] [CrossRef] [PubMed]
- Hauton, D.; Evans, R.D. Fatty acid and triacylglycerol utilisation by perfused rat spleen: Differential metabolic fate and the effect of endotoxin. Cell. Physiol. Biochem. 2002, 12, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Freudenberg, M.; Galanos, C. Metabolic fate of endotoxin in rat. Adv. Exp. Med. Biol. 1990, 256, 499–509. [Google Scholar] [PubMed]
- Bala, S.; Marcos, M.; Gattu, A.; Catalano, D.; Szabo, G. Acute binge drinking increases serum endotoxin and bacterial DNA levels in healthy individuals. PLoS ONE 2014, 9, e96864. [Google Scholar] [CrossRef] [PubMed]
- Bull-Otterson, L.; Feng, W.; Kirpich, I.; Wang, Y.; Qin, X.; Liu, Y.; Gobejishvili, L.; Joshi-Barve, S.; Ayvaz, T.; Petrosino, J.; et al. Metagenomic analyses of alcohol induced pathogenic alterations in the intestinal microbiome and the effect of lactobacillus rhamnosus GG treatment. PLoS ONE 2013, 8, e53028. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Torralba, M.; Tan, J.; Embree, M.; Zengler, K.; Starkel, P.; van Pijkeren, J.P.; DePew, J.; Loomba, R.; Ho, S.B.; et al. Supplementation of saturated long-chain fatty acids maintains intestinal eubiosis and reduces ethanol-induced liver injury in mice. Gastroenterology 2015, 148, 203–214. [Google Scholar] [CrossRef] [PubMed]
- Xie, G.; Zhong, W.; Zheng, X.; Li, Q.; Qiu, Y.; Li, H.; Chen, H.; Zhou, Z.; Jia, W. Chronic ethanol consumption alters mammalian gastrointestinal content metabolites. J. Proteome Res. 2013, 12, 3297–3306. [Google Scholar] [CrossRef] [PubMed]
- Venkatraman, A.; Ramakrishna, B.S.; Pulimood, A.B. Butyrate hastens restoration of barrier function after thermal and detergent injury to rat distal colon in vitro. Scand. J. Gastroenterol. 1999, 34, 1087–1092. [Google Scholar] [CrossRef] [PubMed]
- Donohoe, D.R.; Garge, N.; Zhang, X.; Sun, W.; O’Connell, T.M.; Bunger, M.K.; Bultman, S.J. The microbiome and butyrate regulate energy metabolism and autophagy in the mammalian colon. Cell Metab. 2011, 13, 517–526. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.H.; Kang, S.G.; Park, J.H.; Yanagisawa, M.; Kim, C.H. Short-chain fatty acids activate GPR41 and GPR43 on intestinal epithelial cells to promote inflammatory responses in mice. Gastroenterology 2013, 145, 396–406. [Google Scholar] [CrossRef] [PubMed]
- Van Ampting, M.T.; Loonen, L.M.; Schonewille, A.J.; Konings, I.; Vink, C.; Iovanna, J.; Chamaillard, M.; Dekker, J.; van der Meer, R.; Wells, J.M.; et al. Intestinally secreted C-type lectin Reg3b attenuates salmonellosis but not listeriosis in mice. Infect. Immun. 2012, 80, 1115–1120. [Google Scholar] [CrossRef] [PubMed]
- Faraoni, I.; Antonetti, F.R.; Cardone, J.; Bonmassar, E. miR-155 gene: A typical multifunctional microRNA. Biochim. Biophys. Acta 2009, 1792, 497–505. [Google Scholar] [CrossRef] [PubMed]
- Pathak, S.; Grillo, A.R.; Scarpa, M.; Brun, P.; D’Inca, R.; Nai, L.; Banerjee, A.; Cavallo, D.; Barzon, L.; Palu, G.; et al. miR-155 modulates the inflammatory phenotype of intestinal myofibroblasts by targeting SOCS1 in ulcerative colitis. Exp. Mol. Med. 2015. [Google Scholar] [CrossRef] [PubMed]
- Bala, S.; Petrasek, J.; Csak, T.; Catalano, D.; Kodys, K.; Mundkur, S.; Szabo, G. MicroRNA-155 regulates inflammation in alcoholic liver disease via targeting SOCS1 and SHIP1. J. Immunol. 2012, 188, 54.15. [Google Scholar]
- Lippai, D.; Bala, S.; Csak, T.; Kurt-Jones, E.A.; Szabo, G. Chronic alcohol-induced microRNA-155 contributes to neuroinflammation in a TLR4-dependent manner in mice. PLoS ONE 2013, 8, e70945. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Banan, A.; Forsyth, C.B.; Fields, J.Z.; Lau, C.K.; Zhang, L.J.; Keshavarzian, A. Effect of alcohol on miR-212 expression in intestinal epithelial cells and its potential role in alcoholic liver disease. Alcohol. Clin. Exp. Res. 2008, 32, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Ye, D.; Guo, S.; Al-Sadi, R.; Ma, T.Y. MicroRNA regulation of intestinal epithelial tight junction permeability. Gastroenterology 2011, 141, 1323–1333. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Zhao, C.; Dong, Y.; Zhang, M.; Wang, Y.; Li, F.; Li, X.; McClain, C.; Yang, S.; Feng, W. Inhibition of miR122a by lactobacillus rhamnosus GG culture supernatant increases intestinal occludin expression and protects mice from alcoholic liver disease. Toxicol. Lett. 2015, 234, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Natarajan, S.K.; Ramamoorthy, P.; Thomas, S.; Basivireddy, J.; Kang, G.; Ramachandran, A.; Pulimood, A.B.; Balasubramanian, K.A. Intestinal mucosal alterations in rats with carbon tetrachloride-induced cirrhosis: Changes in glycosylation and luminal bacteria. Hepatology 2006, 43, 837–846. [Google Scholar] [CrossRef] [PubMed]
- Ramachandran, A.; Balasubramanian, K.A. Intestinal dysfunction in liver cirrhosis: Its role in spontaneous bacterial peritonitis. J. Gastroenterol. Hepatol. 2001, 16, 607–612. [Google Scholar] [CrossRef] [PubMed]
- Ramachandran, A.; Prabhu, R.; Thomas, S.; Reddy, J.B.; Pulimood, A.; Balasubramanian, K.A. Intestinal mucosal alterations in experimental cirrhosis in the rat: Role of oxygen free radicals. Hepatology 2002, 35, 622–629. [Google Scholar] [CrossRef] [PubMed]
- Tsai, P.C.; Bake, S.; Balaraman, S.; Rawlings, J.; Holgate, R.R.; Dubois, D.; Miranda, R.C. MiR-153 targets the nuclear factor-1 family and protects against teratogenic effects of ethanol exposure in fetal neural stem cells. Biol. Open 2014, 3, 741–758. [Google Scholar] [CrossRef] [PubMed]
- Sathyan, P.; Golden, H.B.; Miranda, R.C. Competing interactions between micro-RNAs determine neural progenitor survival and proliferation after ethanol exposure: Evidence from an ex vivo model of the fetal cerebral cortical neuroepithelium. J. Neurosci. 2007, 27, 8546–8557. [Google Scholar] [CrossRef] [PubMed]
- Miranda, R.C. MicroRNAs and fetal brain development: Implications for ethanol teratology during the second trimester period of neurogenesis. Front. Genet. 2012. [Google Scholar] [CrossRef] [PubMed]
- Qi, Y.; Zhang, M.; Li, H.; Frank, J.A.; Dai, L.; Liu, H.; Chen, G. MicroRNA-29b regulates ethanol-induced neuronal apoptosis in the developing cerebellum through SP1/RAX/PKR cascade. J. Biol. Chem. 2014, 289, 10201–10210. [Google Scholar] [CrossRef] [PubMed]
- Peters, G.A.; Seachrist, D.D.; Keri, R.A.; Sen, G.C. The double-stranded Rna-binding protein, pact, is required for postnatal anterior pituitary proliferation. Proc. Natl. Acad. Sci. USA 2009, 106, 10696–10701. [Google Scholar] [CrossRef] [PubMed]
- Pietrzykowski, A.Z.; Friesen, R.M.; Martin, G.E.; Puig, S.I.; Nowak, C.L.; Wynne, P.M.; Siegelmann, H.T.; Treistman, S.N. Posttranscriptional regulation of BK channel splice variant stability by miR-9 underlies neuroadaptation to alcohol. Neuron 2008, 59, 274–287. [Google Scholar] [CrossRef] [PubMed]
- Martin, G.E.; Hendrickson, L.M.; Penta, K.L.; Friesen, R.M.; Pietrzykowski, A.Z.; Tapper, A.R.; Treistman, S.N. Identification of a BK channel auxiliary protein controlling molecular and behavioral tolerance to alcohol. Proc. Natl. Acad. Sci. USA 2008, 105, 17543–17548. [Google Scholar] [CrossRef] [PubMed]
- Kann, L.; Kinchen, S.; Shanklin, S.L.; Flint, K.H.; Kawkins, J.; Harris, W.A.; Lowry, R.; Olsen, E.O.; McManus, T.; Chyen, D.; et al. Youth risk behavior surveillance—United States, 2013. MMWR Surveill. Summ. 2014, 63, S1–S168. [Google Scholar]
- White, A.M.; Ghia, A.J.; Levin, E.D.; Swartzwelder, H.S. Binge pattern ethanol exposure in adolescent and adult rats: Differential impact on subsequent responsiveness to ethanol. Alcohol. Clin. Exp. Res. 2000, 24, 1251–1256. [Google Scholar] [CrossRef] [PubMed]
- Prins, S.A.; Przybycien-Szymanska, M.M.; Rao, Y.S.; Pak, T.R. Long-term effects of peripubertal binge etoh exposure on hippocampal microRNA expression in the rat. PLoS ONE 2014, 9, e83166. [Google Scholar] [CrossRef] [PubMed]
- Przybycien-Szymanska, M.M.; Mott, N.N.; Paul, C.R.; Gillespie, R.A.; Pak, T.R. Binge-pattern alcohol exposure during puberty induces long-term changes in HPA axis reactivity. PLoS ONE 2011, 6, e18350. [Google Scholar] [CrossRef] [PubMed]
- Pappalardo-Carter, D.L.; Balaraman, S.; Sathyan, P.; Carter, E.S.; Chen, W.J.; Miranda, R.C. Suppression and epigenetic regulation of miR-9 contributes to ethanol teratology: Evidence from zebrafish and murine fetal neural stem cell models. Alcohol. Clin. Exp. Res. 2013, 37, 1657–1667. [Google Scholar] [CrossRef] [PubMed]
- Napoli, J.L. Effects of ethanol on physiological retinoic acid levels. IUBMB Life 2011, 63, 701–706. [Google Scholar] [CrossRef] [PubMed]
- Nimmagadda, V.K.; Bever, C.T.; Vattikunta, N.R.; Talat, S.; Ahmad, V.; Nagalla, N.K.; Trisler, D.; Judge, S.I.; Royal, W., 3rd; Chandrasekaran, K.; et al. Overexpression of SIRT1 protein in neurons protects against experimental autoimmune encephalomyelitis through activation of multiple SIRT1 targets. J. Immunol. 2013, 190, 4595–4607. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Qiu, Y.; Gao, X.; Yuan, X.B.; Zhai, Q. Overexpression of Sirt1 in mouse forebrain impairs lipid/glucose metabolism and motor function. PLoS ONE 2011, 6, e21759. [Google Scholar] [CrossRef] [PubMed]
- Neumann, J.T.; Thompson, J.W.; Raval, A.P.; Cohan, C.H.; Koronowski, K.B.; Perez-Pinzon, M.A. Increased BDNF protein expression after ischemic or PKC epsilon preconditioning promotes electrophysiologic changes that lead to neuroprotection. J. Cereb. Blood Flow Metab. 2015, 35, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Harper, C.G.; Kril, J.J. Neuropathology of alcoholism. Alcohol. Alcohol. 1990, 25, 207–216. [Google Scholar] [PubMed]
- Kril, J.J. The contribution of alcohol, thiamine deficiency and cirrhosis of the liver to cerebral cortical damage in alcoholics. Metab. Brain Dis. 1995, 10, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Lewohl, J.M.; Nunez, Y.O.; Dodd, P.R.; Tiwari, G.R.; Harris, R.A.; Mayfield, R.D. Up-regulation of microRNAs in brain of human alcoholics. Alcohol. Clin. Exp. Res. 2011, 35, 1928–1937. [Google Scholar] [CrossRef] [PubMed]
- Yadav, S.; Pandey, A.; Shukla, A.; Talwelkar, S.S.; Kumar, A.; Pant, A.B.; Parmar, D. MiR-497 and miR-302B regulate ethanol-induced neuronal cell death through BCL2 protein and cyclin D2. J. Biol. Chem. 2011, 286, 37347–37357. [Google Scholar] [CrossRef] [PubMed]
- Nunez, Y.O.; Mayfield, R.D. Understanding alcoholism through microRNA signatures in brains of human alcoholics. Front. Genet. 2012, 3. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, V.; Ellis, J.D.; Shen, Z.; Song, D.Y.; Pan, Q.; Watt, A.T.; Freier, S.M.; Bennett, C.F.; Sharma, A.; Bubulya, P.A.; et al. The nuclear-retained noncoding RNA malat1 regulates alternative splicing by modulating sr splicing factor phosphorylation. Mol. Cell 2010, 39, 925–938. [Google Scholar] [CrossRef] [PubMed]
- Volders, P.J.; Verheggen, K.; Menschaert, G.; Vandepoele, K.; Martens, L.; Vandesompele, J.; Mestdagh, P. An update on lncipedia: A database for annotated human lncRNA sequences. Nucleic Acids Res. 2015, 43, D174–D180. [Google Scholar] [CrossRef] [PubMed]
- Clark, B.S.; Blackshaw, S. Long non-coding RNA-dependent transcriptional regulation in neuronal development and disease. Front. Genet. 2014. [Google Scholar] [CrossRef] [PubMed]
- Kryger, R.; Fan, L.; Wilce, P.A.; Jaquet, V. Malat-1, a non protein-coding RNA is upregulated in the cerebellum, hippocampus and brain stem of human alcoholics. Alcohol 2012, 46, 629–634. [Google Scholar] [CrossRef] [PubMed]
- Koshimizu, T.A.; Fujiwara, Y.; Sakai, N.; Shibata, K.; Tsuchiya, H. Oxytocin stimulates expression of a noncoding RNA tumor marker in a human neuroblastoma cell line. Life Sci. 2010, 86, 455–460. [Google Scholar] [CrossRef] [PubMed]
- Marchesi, C.; Chiodera, P.; Brusamonti, E.; Volpi, R.; Coiro, V. Abnormal plasma oxytocin and beta-endorphin levels in alcoholics after short and long term abstinence. Prog. Neuropsychopharmacol. Biol. Psychiatry 1997, 21, 797–807. [Google Scholar] [CrossRef]
- Farris, S.P.; Mayfield, R.D. RNA-Seq reveals novel transcriptional reorganization in human alcoholic brain. Int. Rev. Neurobiol. 2014, 116, 275–300. [Google Scholar] [PubMed]
- Young, J.K.; Giesbrecht, H.E.; Eskin, M.N.; Aliani, M.; Suh, M. Nutrition implications for fetal alcohol spectrum disorder. Adv. Nutr. 2014, 5, 675–692. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.L.; Zhang, Z.; Li, Q.; Yang, R.; Pei, X.; Xu, Y.; Wang, J.; Zhou, S.F.; Li, Y. Ethanol exposure induces differential microrna and target gene expression and teratogenic effects which can be suppressed by folic acid supplementation. Hum. Reprod. 2009, 24, 562–579. [Google Scholar] [CrossRef] [PubMed]
- Weiss, F.U.; Marques, I.J.; Woltering, J.M.; Vlecken, D.H.; Aghdassi, A.; Partecke, L.I.; Heidecke, C.D.; Lerch, M.M.; Bagowski, C.P. Retinoic acid receptor antagonists inhibit miR-10A expression and block metastatic behavior of pancreatic cancer. Gastroenterology 2009, 137, 2136–2145. [Google Scholar] [CrossRef] [PubMed]
- Mansfield, J.H.; Harfe, B.D.; Nissen, R.; Obenauer, J.; Srineel, J.; Chaudhuri, A.; Farzan-Kashani, R.; Zuker, M.; Pasquinelli, A.E.; Ruvkun, G.; et al. Microrna-responsive “sensor” transgenes uncover hox-like and other developmentally regulated patterns of vertebrate microrna expression. Nat. Genet. 2004, 36, 1079–1083. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Yang, X.Y.; Zhao, J.Y.; Yu, L.W.; Zhang, P.; Duan, W.Y.; Chong, M.; Gui, Y.H. MiR-10a and miR-10b target the 3'-untranslated region of TBX5 to repress its expression. Pediatr. Cardiol. 2014, 35, 1072–1079. [Google Scholar] [CrossRef] [PubMed]
- Serrano, M.; Han, M.; Brinez, P.; Linask, K.K. Fetal alcohol syndrome: Cardiac birth defects in mice and prevention with folate. Am. J. Obstet. Gynecol. 2010, 203, 75.e7–75.e15. [Google Scholar] [CrossRef] [PubMed]
- Grummer, M.A.; Langhough, R.E.; Zachman, R.D. Maternal ethanol ingestion effects on fetal rat brain vitamin a as a model for fetal alcohol syndrome. Alcohol. Clin. Exp. Res. 1993, 17, 592–597. [Google Scholar] [CrossRef] [PubMed]
- Chokroborty-Hoque, A.; Alberry, B.; Singh, S.M. Exploring the complexity of intellectual disability in fetal alcohol spectrum disorders. Front. Pediatr. 2014. [Google Scholar] [CrossRef] [PubMed]
- Kleiber, M.L.; Wright, E.; Singh, S.M. Maternal voluntary drinking in c57bl/6j mice: Advancing a model for fetal alcohol spectrum disorders. Behav. Brain Res. 2011, 223, 376–387. [Google Scholar] [CrossRef] [PubMed]
- Mantha, K.; Laufer, B.I.; Singh, S.M. Molecular changes during neurodevelopment following second-trimester binge ethanol exposure in a mouse model of fetal alcohol spectrum disorder: From immediate effects to long-term adaptation. Dev. Neurosci. 2014, 36, 29–43. [Google Scholar] [CrossRef] [PubMed]
- Burke, M.W.; Ptito, M.; Ervin, F.R.; Palmour, R.M. Hippocampal neuron populations are reduced in vervet monkeys with fetal alcohol exposure. Dev. Psychobiol. 2015, 57, 470–485. [Google Scholar] [CrossRef] [PubMed]
- Stringer, R.L.; Laufer, B.I.; Kleiber, M.L.; Singh, S.M. Reduced expression of brain cannabinoid receptor 1 (CNR1) is coupled with an increased complementary micro-RNA (miR-26b) in a mouse model of fetal alcohol spectrum disorders. Clin. Epigenetics 2013, 5, 1868–1870. [Google Scholar] [CrossRef] [PubMed]
- Blazquez, C.; Chiarlone, A.; Bellocchio, L.; Resel, E.; Pruunsild, P.; Garcia-Rincon, D.; Sendtner, M.; Timmusk, T.; Lutz, B.; Galve-Roperh, I.; et al. The cb cannabinoid receptor signals striatal neuroprotection via a PI3K/AKT/MTORC1/BDNF pathway. Cell Death Differ. 2015. [Google Scholar] [CrossRef] [PubMed]
- Ceccarini, J.; Hompes, T.; Verhaeghen, A.; Casteels, C.; Peuskens, H.; Bormans, G.; Claes, S.; van Laere, K. Changes in cerebral CB1 receptor availability after acute and chronic alcohol abuse and monitored abstinence. J. Neurosci. 2014, 34, 2822–2831. [Google Scholar] [CrossRef] [PubMed]
- Liang, F.; Diao, L.; Liu, J.; Jiang, N.; Zhang, J.; Wang, H.; Zhou, W.; Huang, G.; Ma, D. Paternal ethanol exposure and behavioral abnormities in offspring: Associated alterations in imprinted gene methylation. Neuropharmacology 2014, 81, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Curley, J.P.; Mashoodh, R.; Champagne, F.A. Epigenetics and the origins of paternal effects. Horm. Behav. 2011, 59, 306–314. [Google Scholar] [CrossRef] [PubMed]
- Baber, M.; Koren, G. Investigating the fetal and postnatal effects of paternal alcohol exposure in mouse offspring: A review. J. Popul. Ther. Clin. Pharmacol. 2015, 22, e57–e58. [Google Scholar] [PubMed]
- Finegersh, A.; Rompala, G.R.; Martin, D.I.; Homanics, G.E. Drinking beyond a lifetime: New and emerging insights into paternal alcohol exposure on subsequent generations. Alcohol 2015, 49, 461–470. [Google Scholar] [CrossRef] [PubMed]
- Bielawski, D.M.; Zaher, F.M.; Svinarich, D.M.; Abel, E.L. Paternal alcohol exposure affects sperm cytosine methyltransferase messenger RNA levels. Alcohol. Clin. Exp. Res. 2002, 26, 347–351. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.M.; Pang, R.T.; Chiu, P.C.; Wong, B.P.; Lao, K.; Lee, K.F.; Yeung, W.S. Sperm-borne microRNA-34c is required for the first cleavage division in mouse. Proc. Natl. Acad. Sci. USA 2012, 109, 490–494. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Galindo, C.; Hermsen, M.; Garcia-Pedrero, J.M.; Fresno, M.F.; Suarez, C.; Rodrigo, J.P. P27 and BCL2 expression predicts response to chemotherapy in head and neck squamous cell carcinomas. Oral Oncol. 2014, 50, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Rodgers, A.B.; Morgan, C.P.; Bronson, S.L.; Revello, S.; Bale, T.L. Paternal stress exposure alters sperm microRNA content and reprograms offspring HPA stress axis regulation. J. Neurosci. 2013, 33, 9003–9012. [Google Scholar] [CrossRef] [PubMed]
- McPherson, N.O.; Fullston, T.; Aitken, R.J.; Lane, M. Paternal obesity, interventions, and mechanistic pathways to impaired health in offspring. Ann. Nutr. Metab. 2014, 64, 231–238. [Google Scholar] [CrossRef] [PubMed]
- McPherson, N.O.; Owens, J.A.; Fullston, T.; Lane, M. Preconception diet or exercise intervention in obese fathers normalizes sperm microRNA profile and metabolic syndrome in female offspring. Am. J. Physiol. Endocrinol. Metab. 2015, 308, E805–E821. [Google Scholar] [CrossRef] [PubMed]
- Djousse, L.; Gaziano, J.M. Alcohol consumption and heart failure: A systematic review. Curr. Atheroscler Rep. 2008, 10, 117–120. [Google Scholar] [CrossRef] [PubMed]
- Guzzo-Merello, G.; Segovia, J.; Dominguez, F.; Cobo-Marcos, M.; Gomez-Bueno, M.; Avellana, P.; Millan, I.; Alonso-Pulpon, L.; Garcia-Pavia, P. Natural history and prognostic factors in alcoholic cardiomyopathy. JACC Heart Fail. 2015, 3, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Xiao, F.; Yuan, W.; Li, X.; Wang, G.; Jiang, T.; Wang, W.; Zhang, J.; Li, P.; Qi, L.; Chen, Y. A case of heart failure due to alcoholic cardiomyopathy combined with acute pulmonary embolism. J. Thorac. Dis. 2014, 6, E176–E180. [Google Scholar] [PubMed]
- Guzzo-Merello, G.; Cobo-Marcos, M.; Gallego-Delgado, M.; Garcia-Pavia, P. Alcoholic cardiomyopathy. World J. Cardiol. 2014, 6, 771–781. [Google Scholar] [CrossRef] [PubMed]
- Piano, M.R.; Phillips, S.A. Alcoholic cardiomyopathy: Pathophysiologic insights. Cardiovasc Toxicol. 2014, 14, 291–308. [Google Scholar] [CrossRef] [PubMed]
- Lai, Y.J.; Hung, C.L.; Hong, R.C.; Tseng, Y.M.; Lin, C.I.; Ko, Y.S.; Tsai, C.H.; Yeh, H.I. Slow conduction and gap junction remodeling in murine ventricle after chronic alcohol ingestion. J. Biomed. Sci. 2011. [Google Scholar] [CrossRef] [PubMed]
- Laszlo, R.; Eick, C.; Schwiebert, M.; Schreiner, B.; Weig, H.J.; Weretka, S.; Bosch, R.F.; Schreieck, J. Alcohol-induced electrical remodeling: Effects of sustained short-term ethanol infusion on ion currents in rabbit atrium. Alcohol. Clin. Exp. Res. 2009, 33, 1697–1703. [Google Scholar] [CrossRef] [PubMed]
- Boon, R.A.; Iekushi, K.; Lechner, S.; Seeger, T.; Fischer, A.; Heydt, S.; Kaluza, D.; Treguer, K.; Carmona, G.; Bonauer, A.; et al. MicroRNA-34a regulates cardiac ageing and function. Nature 2013, 495, 107–110. [Google Scholar] [CrossRef] [PubMed]
- Guo, R.; Hu, N.; Kandadi, M.R.; Ren, J. Facilitated ethanol metabolism promotes cardiomyocyte contractile dysfunction through autophagy in murine hearts. Autophagy 2012, 8, 593–608. [Google Scholar] [CrossRef] [PubMed]
- Jing, L.; Jin, C.; Lu, Y.; Huo, P.; Zhou, L.; Wang, Y.; Tian, Y. Investigation of microRNA expression profiles associated with human alcoholic cardiomyopathy. Cardiology 2015, 130, 223–233. [Google Scholar] [CrossRef] [PubMed]
- Hunter, R.J.; Neagoe, C.; Jarvelainen, H.A.; Martin, C.R.; Lindros, K.O.; Linke, W.A.; Preedy, V.R. Alcohol affects the skeletal muscle proteins, titin and nebulin in male and female rats. J. Nutr. 2003, 133, 1154–1157. [Google Scholar] [PubMed]
- Urbano-Marquez, A.; Estruch, R.; Fernandez-Sola, J.; Nicolas, J.M.; Pare, J.C.; Rubin, E. The greater risk of alcoholic cardiomyopathy and myopathy in women compared with men. JAMA 1995, 274, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Andersen, H.; Borre, M.; Jakobsen, J.; Andersen, P.H.; Vilstrup, H. Decreased muscle strength in patients with alcoholic liver cirrhosis in relation to nutritional status, alcohol abstinence, liver function, and neuropathy. Hepatology 1998, 27, 1200–1206. [Google Scholar] [CrossRef] [PubMed]
- Aagaard, N.K.; Andersen, H.; Vilstrup, H.; Clausen, T.; Jakobsen, J.; Dorup, I. Magnesium supplementation and muscle function in patients with alcoholic liver disease: A randomized, placebo-controlled trial. Scand. J. Gastroenterol. 2005, 40, 972–979. [Google Scholar] [CrossRef] [PubMed]
- Ignacio, C.; Hicks, S.D.; Burke, P.; Lewis, L.; Szombathyne-Meszaros, Z.; Middleton, F.A. Alterations in serum microRNA in humans with alcohol use disorders impact cell proliferation and cell death pathways and predict structural and functional changes in brain. BMC Neurosci. 2015. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Natarajan, S.K.; Pachunka, J.M.; Mott, J.L. Role of microRNAs in Alcohol-Induced Multi-Organ Injury. Biomolecules 2015, 5, 3309-3338. https://doi.org/10.3390/biom5043309
Natarajan SK, Pachunka JM, Mott JL. Role of microRNAs in Alcohol-Induced Multi-Organ Injury. Biomolecules. 2015; 5(4):3309-3338. https://doi.org/10.3390/biom5043309
Chicago/Turabian StyleNatarajan, Sathish Kumar, Joseph M. Pachunka, and Justin L. Mott. 2015. "Role of microRNAs in Alcohol-Induced Multi-Organ Injury" Biomolecules 5, no. 4: 3309-3338. https://doi.org/10.3390/biom5043309
APA StyleNatarajan, S. K., Pachunka, J. M., & Mott, J. L. (2015). Role of microRNAs in Alcohol-Induced Multi-Organ Injury. Biomolecules, 5(4), 3309-3338. https://doi.org/10.3390/biom5043309





