High-Resolution Respirometry for Simultaneous Measurement of Oxygen and Hydrogen Peroxide Fluxes in Permeabilized Cells, Tissue Homogenate and Isolated Mitochondria
Abstract
:1. Introduction
| Abbreviation | Definition |
|---|---|
| CI-linked | The Complex I-linked substrate state is induced in mt-preparations by addition of NADH-generating substrates. |
| CII-linked | The Complex II-linked substrate state is induced in mt-preparations by addition of succinate and rotenone (Complex I inhibitor). |
| CI&II-linked | The Complex I&II-linked substrate state is induced in mt-preparations by addition of NADH-generating substrates (CI-linked) in combination with succinate (CII-linked). This physiological substrate combination is required for partial reconstitution of TCA cycle function and convergent electron-input into the Q-junction to compensate for metabolite depletion into the incubation medium. An additive effect of convergent CI&II-linked electron flow is observed in most types of mitochondria. |
| R | In the intact cell, ROUTINE respiration or ROUTINE activity in the physiological coupling state R is controlled by cellular energy demand, energy turnover and the degree of coupling to phosphorylation of ADP (intrinsic uncoupling and pathological dyscoupling; [16]). |
| L | LEAK respiration or LEAK oxygen flux compensating for proton leak, proton slip, cation cycling and electron leak, is a dissipative component of respiration which is not available for performing biochemical work and thus related to heat production. LEAK respiration is measured in state L, in the presence of reducing substrate(s), but absence of ADP (theoretically, absence of inorganic phosphate presents an alternative), or after enzymatic inhibition of the phosphorylation system. In this non-phosphorylating resting state, the electrochemical proton gradient is increased to a maximum, exerting feedback control by depressing oxygen flux to a level determined mainly by the proton leak and the H+/O2 ratio [17]. |
| P | OXPHOS capacity is the respiratory capacity of mitochondria in the ADP-activated state of oxidative phosphorylation, at saturating concentrations of ADP, inorganic phosphate, oxygen, and defined reduced substrates [17]. It thus differs from State 3 respiration which is respiration of isolated coupled mitochondria in the presence of high ADP and Pi concentrations [18]. ADP concentrations applied in State 3 are not necessarily saturating, whereas OXPHOS capacity is measured at saturating concentrations of ADP and Pi. |
| E | The mitochondrial electron transfer system (ETS) transfers electrons from externally supplied reduced substrates to oxygen. It consists of the membrane-bound ETS (mETS) with enzyme complexes located in the inner mt-membrane, mt-matrix dehydrogenases generating NADH, and the transport systems involved in metabolite exchange across the mt-membranes [19]. ETS capacity is max. O2 flux at optimum uncoupler concentration. |
2. Results and Discussion
2.1. Effect of Amplex Red on Respiration of Intact and Permeabilized HEK 293T Cells
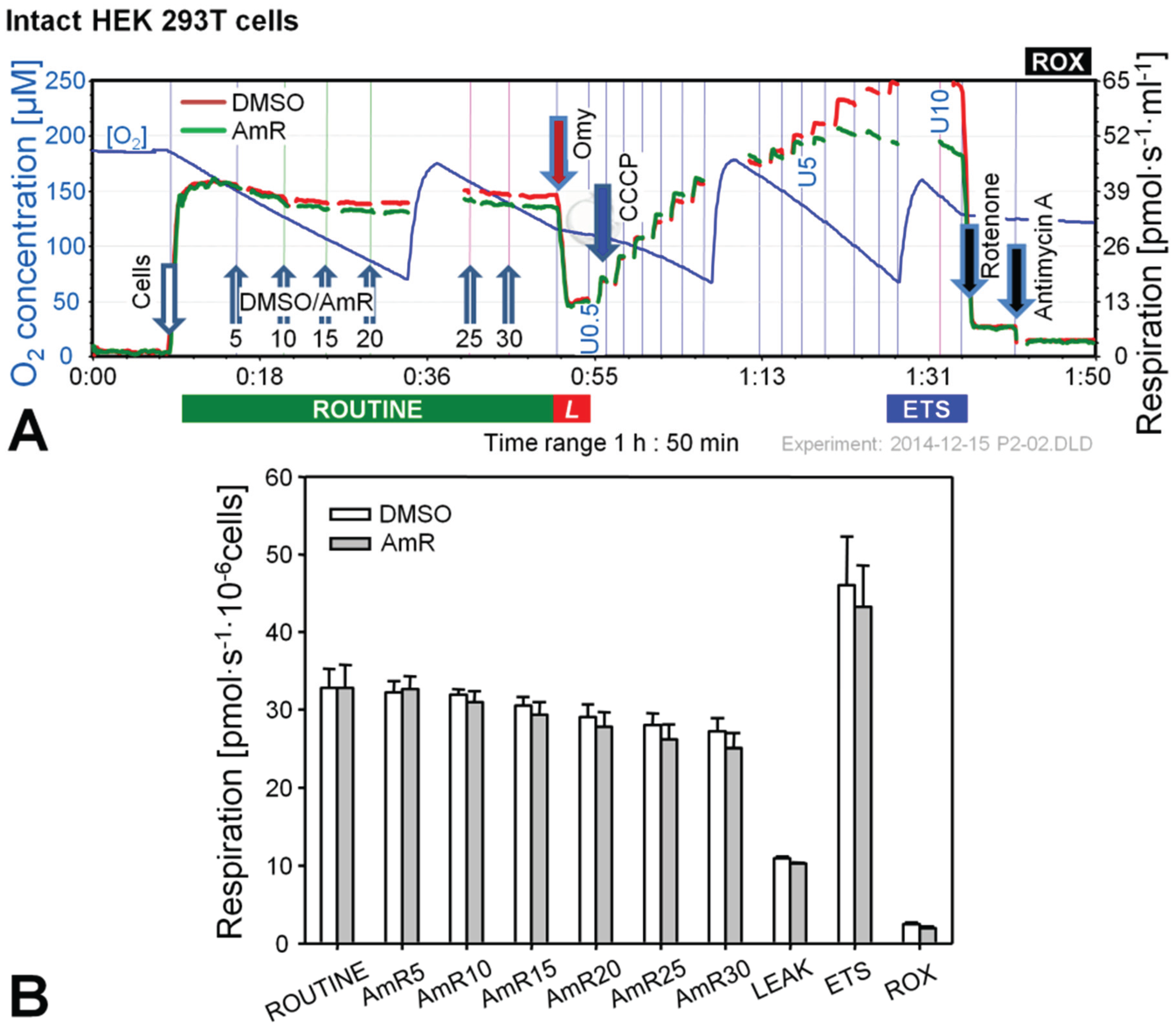
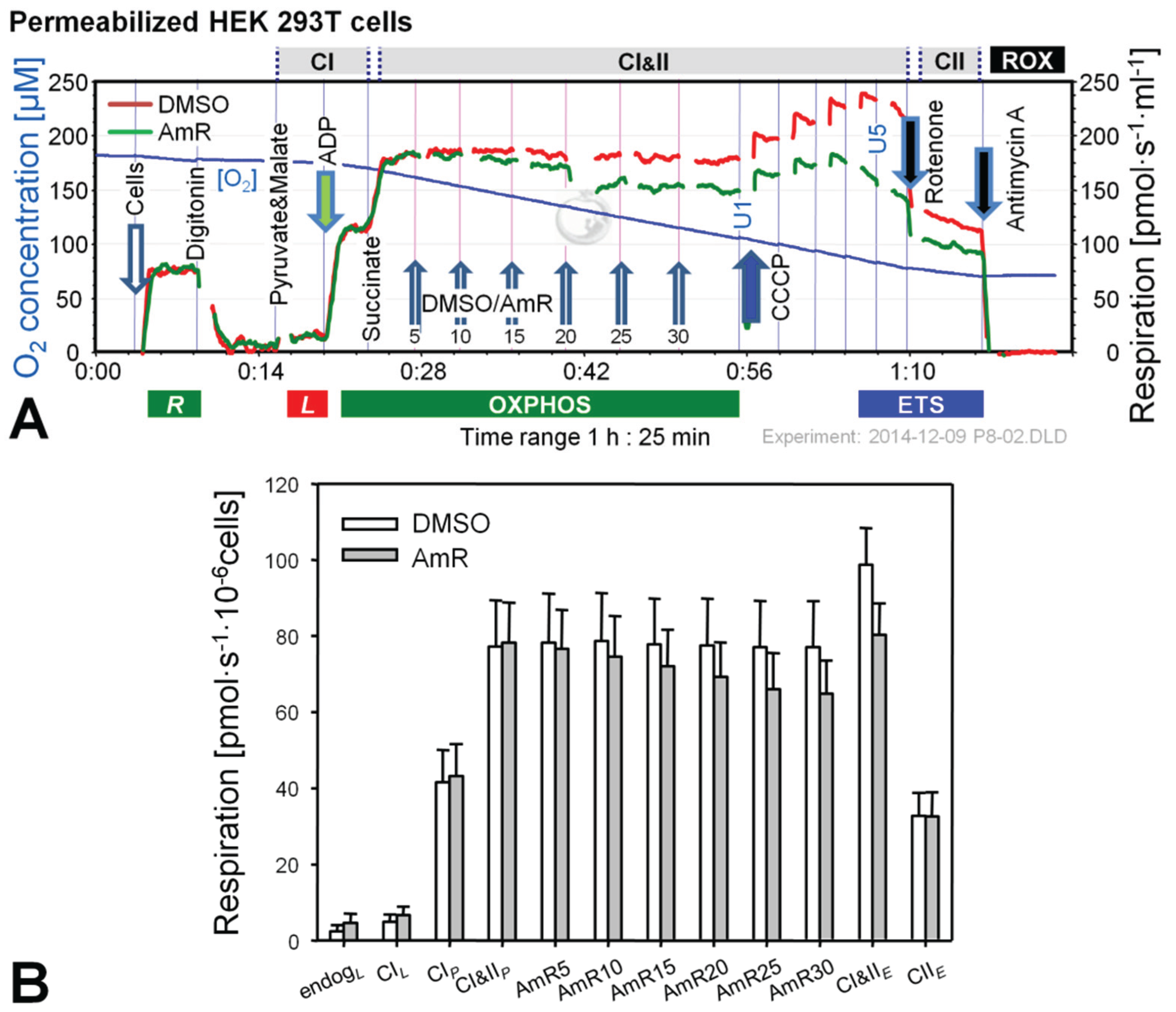
2.2. O2 and H2O2 Flow in Permeabilized HEK 293T Cells: Dependence on Substrate and Coupling State
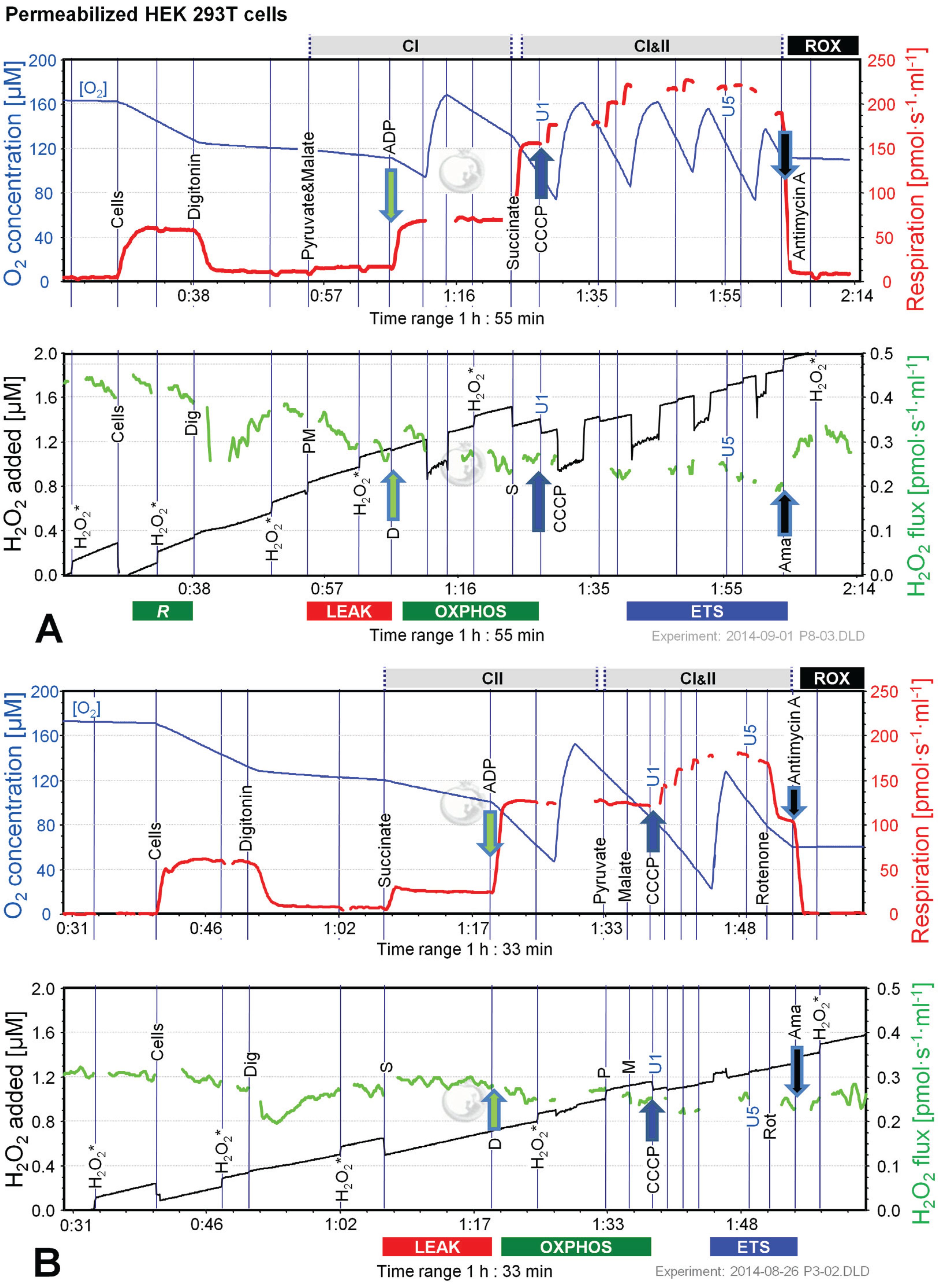

| Substrate | LEAK | OXPHOS | PMS OXPHOS | PMS ETS | ROX |
|---|---|---|---|---|---|
| Permeabilized HEK cells | |||||
| S | 0.85 ± 0.34 | 0.03 ± 0.08 | na | na | 7.2 ± 22.8 |
| S(Rot) | 0.81 ± 0.55 | 0.14 ± 0.07 | 0.16 ± 0.07 | 0.10 ± 0.7 | 7.9 ± 3.4 |
| Mouse brain homogenate | |||||
| PM | 1.2 ± 0.6 | 0.06 ± 0.13 | na | na | 60 ± 13 |
| S | 6.3 ± 0.2 | 0.05 ± 0.13 | 0.05 ± 0.09 | na | 26 ± 6 |
| S(Rot) | 1.5 ± 0.2 | 0.51 ± 0.08 | 1.34 ± 0.09 | 1.24 ± 0.06 | 33 ± 8 |
2.3. O2 and H2O2 Flow in Permeabilized HEK 293T Cells: Dependence on Cell Density
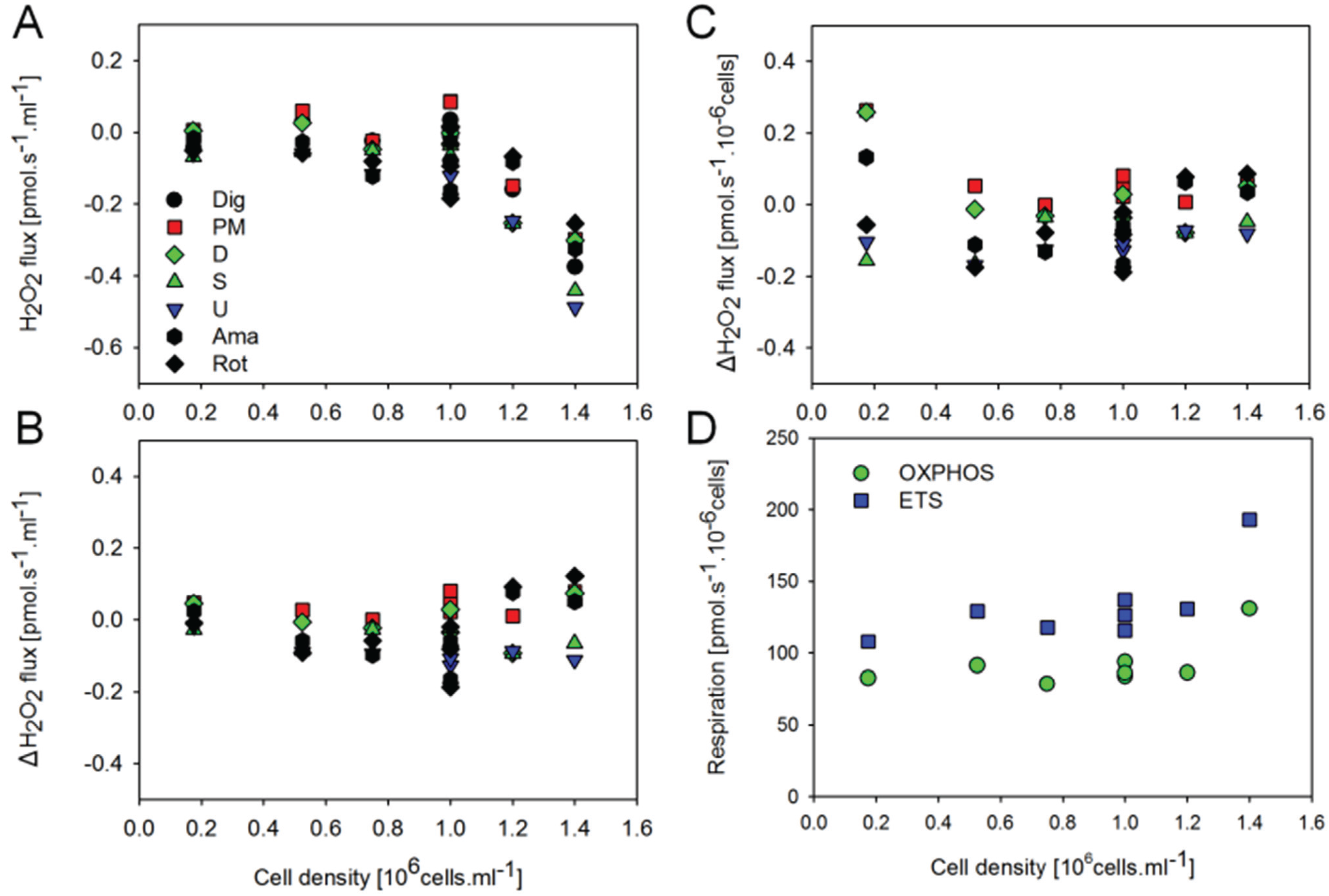
2.4. O2 and H2O2 Flux in Mouse Brain Homogenate: Dependence on Substrate and Coupling State

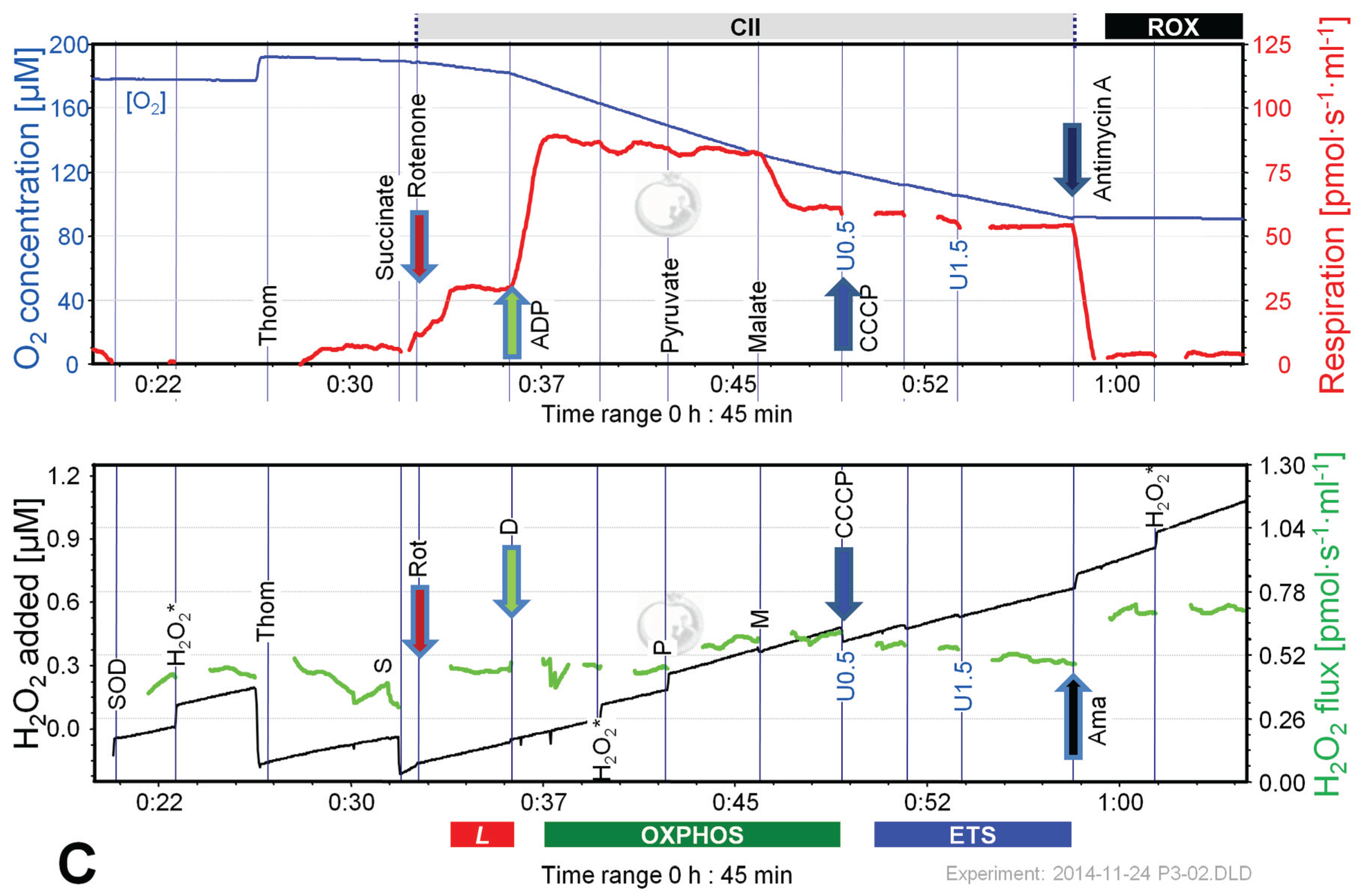
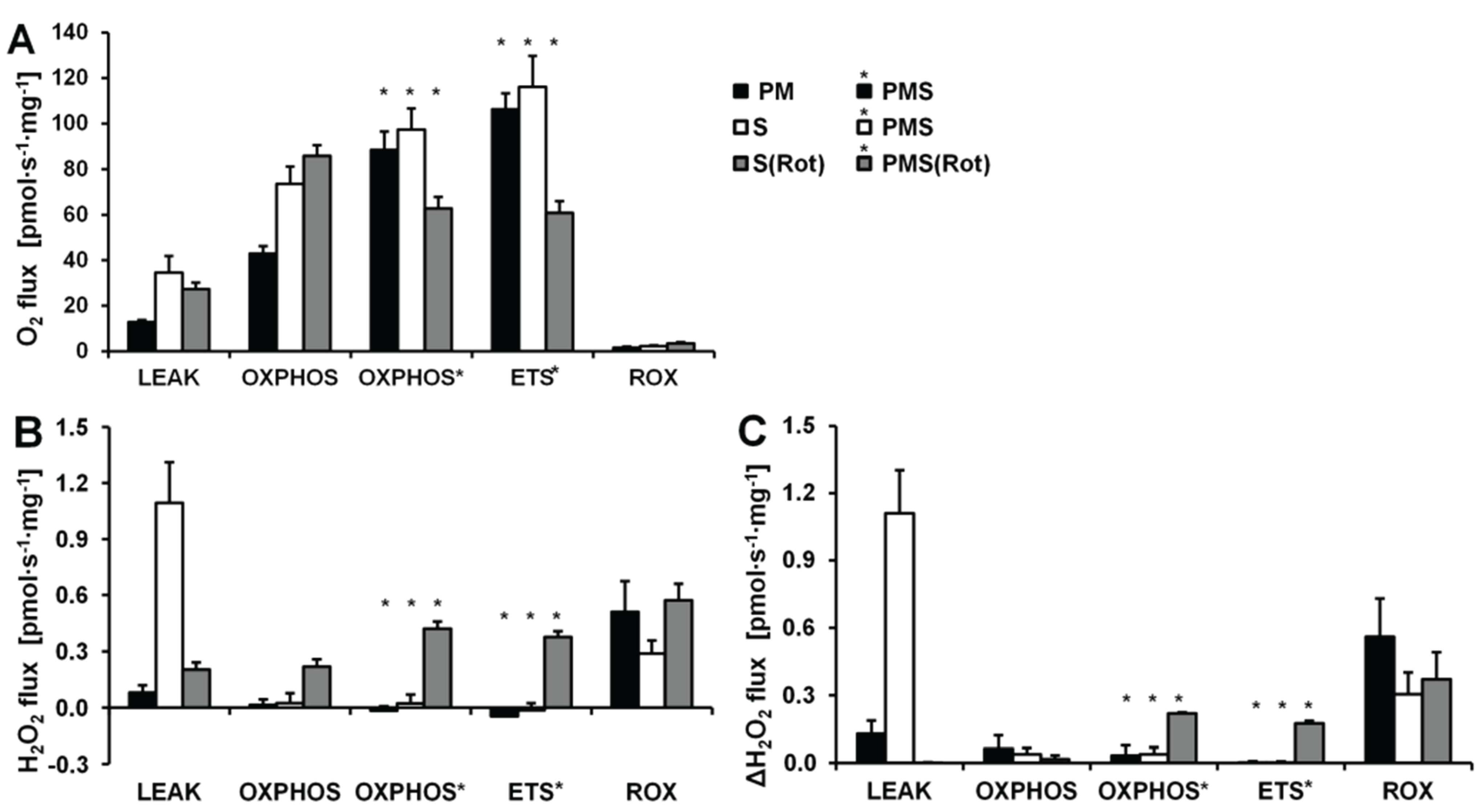
2.5. O2 and H2O2 Flux in Isolated Cardiac Mitochondria: Dependence on Substrate and Coupling State
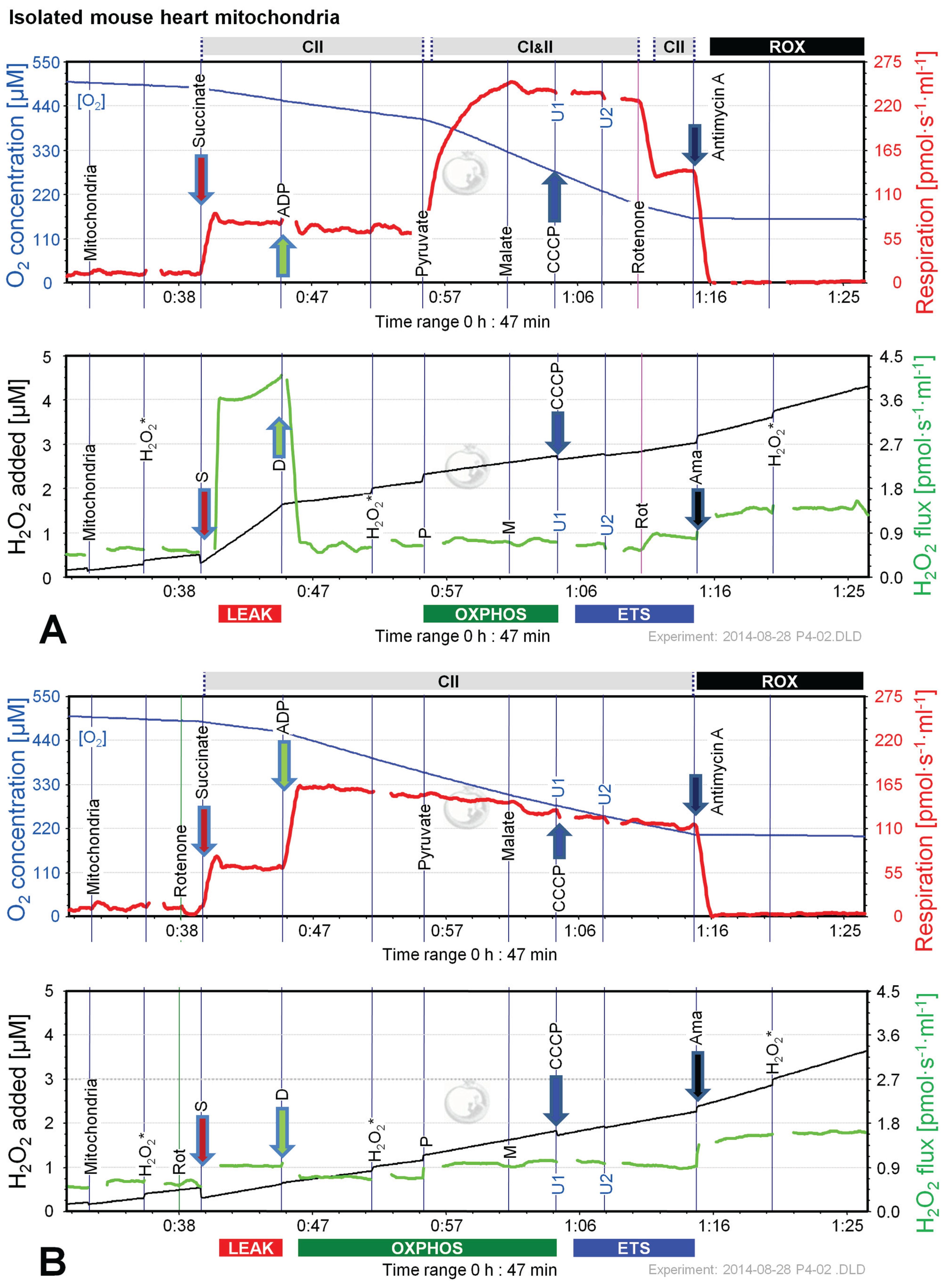
3. Experimental Section
3.1. Chemicals
3.2. High-Resolution Respirometry and O2k-Fluorometry
3.3. Experimental Procedure
3.4. Cell Culture
3.5. Preparation of Mouse Brain Homogenate
3.6. Isolation of Mouse Heart Mitochondria
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations (See also Table 1)
| Ama | Antimycin A | Rot | rotenone |
| AmR | Amplex UltraRed | ROX | residual oxygen consumption |
| Dig | digitonin | S | succinate |
| G | glutamate | SOD | superoxide dismutase |
| HRP | horse radish peroxidase | SUIT | substrate-uncoupler-inhibitor titration |
| M | malate | Thom | tissue homogenate |
| P | pyruvate | U | uncoupler |
References
- Sena, L.A.; Chandel, N.S. Physiological roles of mitochondrial reactive oxygen species. Mol. Cell 2012, 48, 158–167. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Szczepanek, K.; Hu, Y.; Thompson, J.; Lesnefsky, E.J. A deficiency of apoptosis inducing factor (AIF) in Harlequin mouse heart mitochondria paradoxically reduces ROS generation during ischemia-reperfusion. Front. Physiol. 2014. [Google Scholar] [CrossRef] [PubMed]
- Dai, D.F.; Chiao, Y.A.; Marcinek, D.J.; Szeto, H.H.; Rabinovitch, P.S. Mitochondrial oxidative stress in aging and healthspan. Longev. Healthspan. 2014. [Google Scholar] [CrossRef] [PubMed]
- Di Lisa, F.; Kaludercic, N.; Carpi, A.; Menabò, R.; Giorgio, M. Mitochondria and vascular pathology. Pharmacol. Rep. 2009, 61, 123–130. [Google Scholar]
- Tahara, E.B.; Navarete, F.D.; Kowaltowski, A.J. Tissue-, substrate-, and site-specific characteristics of mitochondrial reactive oxygen species generation. Free Radic. Biol. Med. 2009, 46, 1283–1297. [Google Scholar] [CrossRef] [PubMed]
- Dröse, S.; Brandt, U. Molecular mechanisms of superoxide production by the mitochondrial respiratory chain. Adv. Exp. Med. Biol. 2012, 748, 145–169. [Google Scholar] [PubMed]
- Suski, J.M.; Lebiedzinska, M.; Bonora, M.; Pinton, P.; Duszynski, J.; Wieckowski, M.R. Relation between mitochondrial membrane potential and ROS formation. Methods Mol. Biol. 2012, 810, 183–205. [Google Scholar] [PubMed]
- Starkov, A.A. Measurement of mitochondrial ROS production. Methods Mol. Biol. 2010, 648, 245–255. [Google Scholar] [PubMed]
- Kowaltowski, A.J.; de Souza-Pinto, N.C.; Castilho, R.F.; Vercesi, A.E. Mitochondria and reactive oxygen species. Free Radic. Biol. Med. 2009, 47, 333–343. [Google Scholar] [CrossRef] [PubMed]
- Clarke, K.J.; Porter, R.K. Uncoupling protein 1 dependent reactive oxygen species production by thymus mitochondria. Int. J. Biochem. Cell Biol. 2013, 45, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Malinska, D.; Kudin, A.P.; Debska-Vielhaber, G.; Vielhaber, S.; Kunz, W.S. Quantification of superoxide production by mouse brain and skeletal muscle mitochondria. Methods Enzymol. 2009, 456, 419–437. [Google Scholar] [PubMed]
- Tretter, L.; Adam-Vizi, V. Uncoupling is without an effect on the production of reactive oxygen species by in situ synaptic mitochondria. J. Neurochem. 2007, 103, 1864–1871. [Google Scholar] [CrossRef] [PubMed]
- Tretter, L.; Adam-Vizi, V. High Ca2+ load promotes hydrogen peroxide generation via activation of α-glycerophosphate dehydrogenase in brain mitochondria. Free Radic. Biol. Med. 2012, 53, 2119–2130. [Google Scholar] [CrossRef] [PubMed]
- Tretter, L.; Takacs, K.; Kövér, K.; Adam-Vizi, V. Stimulation of H2O2 generation by calcium in brain mitochondria respiring on alpha-glycerophosphate. J. Neurosci. Res. 2007, 85, 3471–3479. [Google Scholar] [CrossRef] [PubMed]
- Krumschnabel, G.; Fontana-Ayoub, M.; Sumbalova, Z.; Heidler, J.; Gauper, K.; Fasching, M.; Gnaiger, E. Simultaneous high-resolution measurement of mitochondrial respiration and hydrogen peroxide production. Methods Mol. Biol. 2015, 1264, 245–261. [Google Scholar] [PubMed]
- Gnaiger, E. Polarographic Oxygen Sensors, the Oxygraph and High-Resolution Respirometry to Assess Mitochondrial Function. In Mitochondrial Dysfunction in Drug-Induced Toxicity; Dykens, J.A., Will, Y., Eds.; John Wiley: Hoboken, NJ, USA, 2008; pp. 327–352. [Google Scholar]
- Gnaiger, E. Capacity of oxidative phosphorylation in human skeletal muscle. New perspectives of mitochondrial physiology. Int. J. Biochem. Cell Biol. 2009, 41, 1837–1845. [Google Scholar] [CrossRef] [PubMed]
- Chance, B.; Williams, G.R. Respiratory enzymes in oxidative phosphorylation: III. The steady state. J. Biol. Chem. 1955, 217, 409–427. [Google Scholar] [PubMed]
- Gnaiger, E. Mitochondrial Pathways and Respiratory Control. An Introduction to OXPHOS Analysis, 4th ed.; Mitochondr Physiol Network 19.12; OROBOROS MiPNet Publications: Innsbruck, Austria, 2014; p. 80. [Google Scholar]
- Kovács, K.; Erdélyi, K.; Hegedűs, C.; Lakatos, P.; Regdon, Z.; Bai, P.; Haskó, G.; Szabó, E.; Virág, L. Poly(ADP-ribosyl)ation is a survival mechanism in cigarette smoke-induced and hydrogen peroxide-mediated cell death. Free Radic. Biol. Med. 2012, 53, 1680–1688. [Google Scholar] [CrossRef] [PubMed]
- Marchissio, M.J.; Francés, D.E.; Carnovale, C.E.; Marinelli, R.A. Mitochondrial aquaporin-8 knockdown in human hepatoma HepG2 cells causes ROS-induced mitochondrial depolarization and loss of viability. Toxicol. Appl. Pharmacol. 2012, 264, 246–254. [Google Scholar] [CrossRef] [PubMed]
- Krumschnabel, G.; Eigentler, A.; Fasching, M.; Gnaiger, E. Use of safranin for the assessment of mitochondrial membrane potential by high-resolution respirometry and fluorometry. Methods Enzymol. 2014, 542, 163–181. [Google Scholar] [PubMed]
- Scaduto, R.C., Jr.; Grotyohann, L.W. Measurement of mitochondrial membrane potential using fluorescent rhodamine derivatives. Biophys. J. 1999, 76, 469–477. [Google Scholar] [CrossRef]
- Krumschnabel, G.; Nydlova, E.; Vrbova, M.; OROBOROS INSTRUMENTS, Innsbruck, Austria. Unpublished work. 2015.
- Wojtala, A.; Bonora, M.; Malinska, D.; Pinton, P.; Duszynski, J.; Wieckowski, M.R. Methods to monitor ROS production by fluorescence microscopy and fluorometry. Methods Enzymol. 2014, 542, 243–262. [Google Scholar] [PubMed]
- Kwak, H.B.; Thalacker-Mercer, A.; Anderson, E.J.; Lin, C.T.; Kane, D.A.; Lee, N.S.; Cortright, R.N.; Bamman, M.M.; Neufer, P.D. Simvastatin impairs ADP-stimulated respiration and increases mitochondrial oxidative stress in primary human skeletal myotubes. Free Radic. Biol. Med. 2012, 52, 198–207. [Google Scholar] [CrossRef] [PubMed]
- Cantu, D.; Fulton, R.E.; Drechsel, D.A.; Patel, M. Mitochondrial aconitase knockdown attenuates paraquat-induced dopaminergic cell death via decreased cellular metabolism and release of iron and H2O2. J. Neurochem. 2011, 118, 79–92. [Google Scholar] [CrossRef] [PubMed]
- Zhao, B.; Summers, F.A.; Mason, R.P. Photooxidation of Amplex Red to resorufin: Implications of exposing the Amplex Red assay to light. Free Radic. Biol. Med. 2012, 53, 1080–1087. [Google Scholar] [CrossRef] [PubMed]
- Suhane, S.; Kanzaki, H.; Arumugaswami, V.; Murali, R.; Ramanujan, V.K. Mitochondrial NDUFS3 regulates the ROS-mediated onset of metabolic switch in transformed cells. Biol. Open. 2013, 2, 295–305. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Tak, E.; Lee, J.; Rashid, M.A.; Murphy, M.P.; Ha, J.; Kim, S.S. Mitochondrial H2O2 generated from electron transport chain complex I stimulates muscle differentiation. Cell Res. 2011, 21, 817–834. [Google Scholar] [CrossRef] [PubMed]
- Gnaiger, E.; Kuznetsov, A.V.; Schneeberger, S.; Seiler, R.; Brandacher, G.; Steurer, W.; Margreiter, R. Mitochondria in the Cold. In Life in the Cold; Heldmaier, G., Klingenspor, M., Eds.; Springer: Heiderlberg, Germany, 2000; pp. 431–442. [Google Scholar]
- Borutaite, V.; Toleikis, A.; Brown, G.C. In the eye of the storm: mitochondrial damage during heart and brain ischaemia. FEBS J. 2013, 280, 4999–5014. [Google Scholar] [CrossRef] [PubMed]
- Fassone, E.; Rahman, S. Complex I deficiency: Clinical features, biochemistry and molecular genetics. J. Med. Genet. 2012, 49, 578–590. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, D.L.; Brookes, P.S. Oxygen sensitivity of mitochondrial reactive oxygen species generation depends on metabolic conditions. J. Biol. Chem. 2009, 284, 16236–16245. [Google Scholar] [CrossRef] [PubMed]
- Pham, T.; Loiselle, D.; Power, A.; Hickey, A.J. Mitochondrial inefficiencies and anoxic ATP hydrolysis capacities in diabetic rat heart. Am. J. Physiol. Cell Physiol. 2014, 307, C499–C507. [Google Scholar] [CrossRef] [PubMed]
- Henry, O.; Jolicoeur, M.; Kamen, A. Unraveling the metabolism of HEK-293 cells using lactate isotopomer analysis. Bioprocess Biosyst. Eng. 2011, 34, 263–267. [Google Scholar] [CrossRef] [PubMed]
- Sauer, L.A.; Dauchy, R.T.; Nagel, W.O.; Morris, H.P. Mitochondrial malic enzymes. Mitochondrial NAD(P)+-dependent malic enzyme activity and malate-dependent pyruvate formation are progression-linked in Morris hepatomas. J. Biol. Chem. 1980, 255, 3844–3848. [Google Scholar] [PubMed]
- Gnaiger, E. P/E from Mouse to Man. Available online: http://wiki.oroboros.at/index.php/OXPHOS_control_ratio#P.2FE_from_mouse_to_man (accessed on 5 June 2015).
- Loschen, G.; Flohe, L.; Chance, B. Respiratory chain linked H2O2 production in pigeon heart mitochondria. FEBS Lett. 1971, 18, 261–264. [Google Scholar] [CrossRef]
- Sumbalova, Z.; Vancova, O.; Krumschnabel, G.; Gnaiger, E. Optimization of malate concentration for high-resolution respirometry: Mitochondria from rat liver and brain. Mitochondr. Physiol. Network 2014, 19.13, 37. [Google Scholar]
- Bleier, L.; Dröse, S. Superoxide generation by complex III: From mechanistic rationales to functional consequences. Biochim. Biophys. Acta 2013, 1827, 1320–1331. [Google Scholar] [CrossRef] [PubMed]
- Murphy, M.P. How mitochondria produce reactive oxygen species. Biochem. J. 2009, 417, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Picard, M.; Taivassalo, T.; Gouspillou, G.; Hepple, R.T. Mitochondria: Isolation, structure and function. J. Physiol. 2011, 589, 4413–4421. [Google Scholar] [CrossRef] [PubMed]
- Picard, M.; Taivassalo, T.; Ritchie, D.; Wright, K.J.; Thomas, M.M.; Romestaing, C.; Hepple, R.T. Mitochondrial structure and function are disrupted by standard isolation methods. PLoS ONE 2011, 6, e18317. [Google Scholar] [CrossRef] [PubMed]
- Eigentler, A.; Fontana-Ayoub, M.; Gnaiger, E. O2k-Fluorometry: HRR and H2O2 production in mouse cardiac tissue homogenate. Mitochondr. Physiol. Network 2013, 18, 1–6. [Google Scholar]
- Gross, V.S.; Greenberg, H.K.; Baranov, S.V.; Carlson, G.M.; Stavrovskaya, I.G.; Lazarev, A.V.; Kristal, B.S. Isolation of functional mitochondria from rat kidney and skeletal muscle without manual homogenization. Anal. Biochem. 2011, 418, 213–223. [Google Scholar] [CrossRef] [PubMed]
- Tahara, E.B.; Barros, M.H.; Oliveira, G.A.; Netto, L.E.; Kowaltowski, A.J. Dihydrolipoyl dehydrogenase as a source of reactive oxygen species inhibited by caloric restriction and involved in Saccharomyces cerevisiae aging. FASEB J. 2007, 21, 274–283. [Google Scholar] [CrossRef] [PubMed]
- Seaver, L.C.; Imlay, J.A. Are respiratory enzymes the primary sources of intracellular hydrogen peroxide? J. Biol. Chem. 2004, 279, 48742–48750. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.R.; Zweier, J.L. Cardiac mitochondria and reactive oxygen species generation. Circ. Res. 2014, 114, 524–537. [Google Scholar] [CrossRef] [PubMed]
- Pesta, D.; Gnaiger, E. High-resolution respirometry: OXPHOS protocols for human cells and permeabilized fibers from small biopsies of human muscle. Methods Mol. Biol. 2012, 810, 25–58. [Google Scholar] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Makrecka-Kuka, M.; Krumschnabel, G.; Gnaiger, E. High-Resolution Respirometry for Simultaneous Measurement of Oxygen and Hydrogen Peroxide Fluxes in Permeabilized Cells, Tissue Homogenate and Isolated Mitochondria. Biomolecules 2015, 5, 1319-1338. https://doi.org/10.3390/biom5031319
Makrecka-Kuka M, Krumschnabel G, Gnaiger E. High-Resolution Respirometry for Simultaneous Measurement of Oxygen and Hydrogen Peroxide Fluxes in Permeabilized Cells, Tissue Homogenate and Isolated Mitochondria. Biomolecules. 2015; 5(3):1319-1338. https://doi.org/10.3390/biom5031319
Chicago/Turabian StyleMakrecka-Kuka, Marina, Gerhard Krumschnabel, and Erich Gnaiger. 2015. "High-Resolution Respirometry for Simultaneous Measurement of Oxygen and Hydrogen Peroxide Fluxes in Permeabilized Cells, Tissue Homogenate and Isolated Mitochondria" Biomolecules 5, no. 3: 1319-1338. https://doi.org/10.3390/biom5031319
APA StyleMakrecka-Kuka, M., Krumschnabel, G., & Gnaiger, E. (2015). High-Resolution Respirometry for Simultaneous Measurement of Oxygen and Hydrogen Peroxide Fluxes in Permeabilized Cells, Tissue Homogenate and Isolated Mitochondria. Biomolecules, 5(3), 1319-1338. https://doi.org/10.3390/biom5031319





