Immunoglobulin Isotypes in Atlantic Salmon, Salmo Salar
Abstract
:1. Introduction
2. Immunoglobulin Heavy Chain Genes in Teleost Fish
3. Ig Expression in Teleost Fish
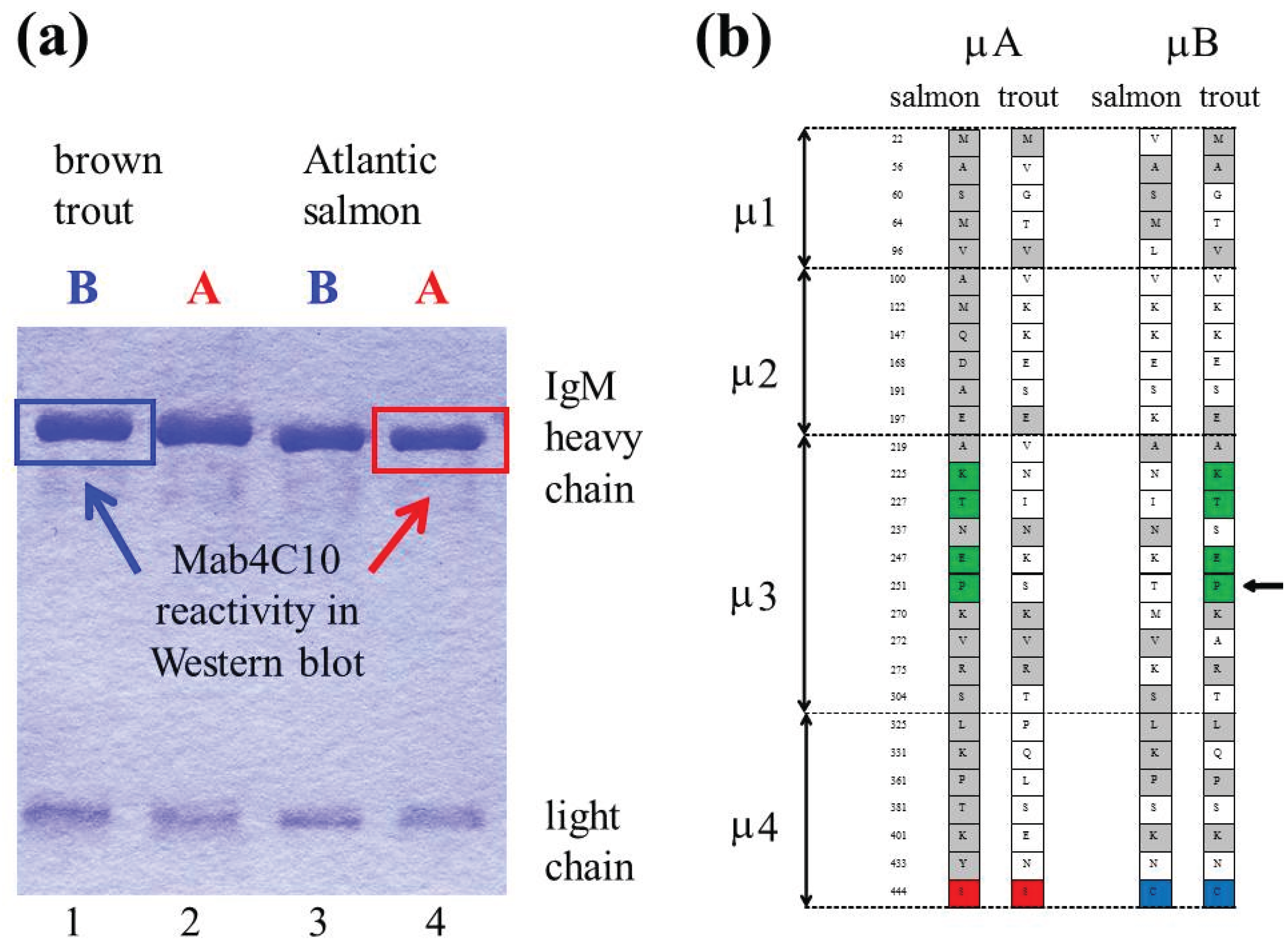
4. Ancestral Tetraploidy and Ig Sub-Variants in Salmonid Fish
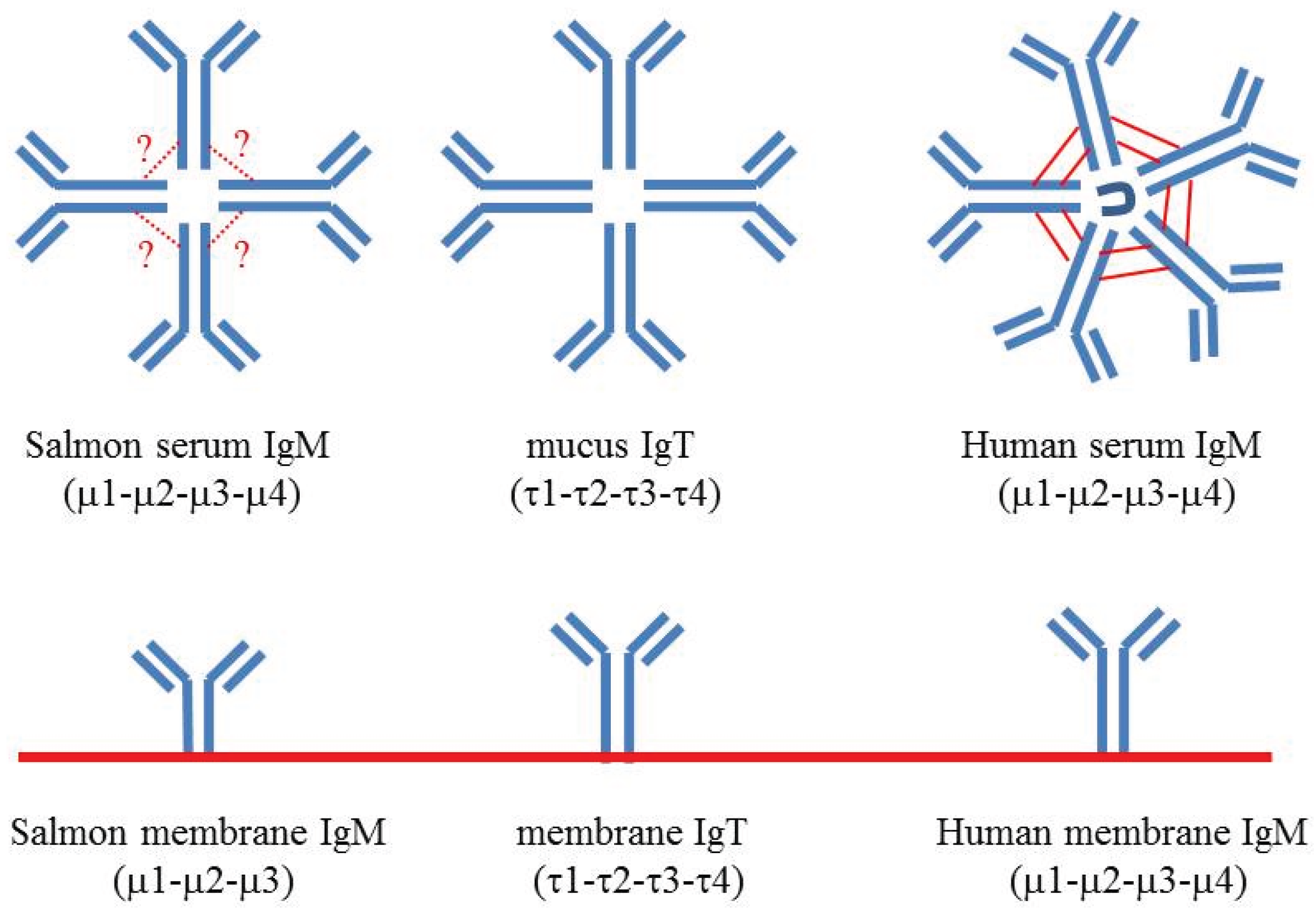
5. IgM Structure
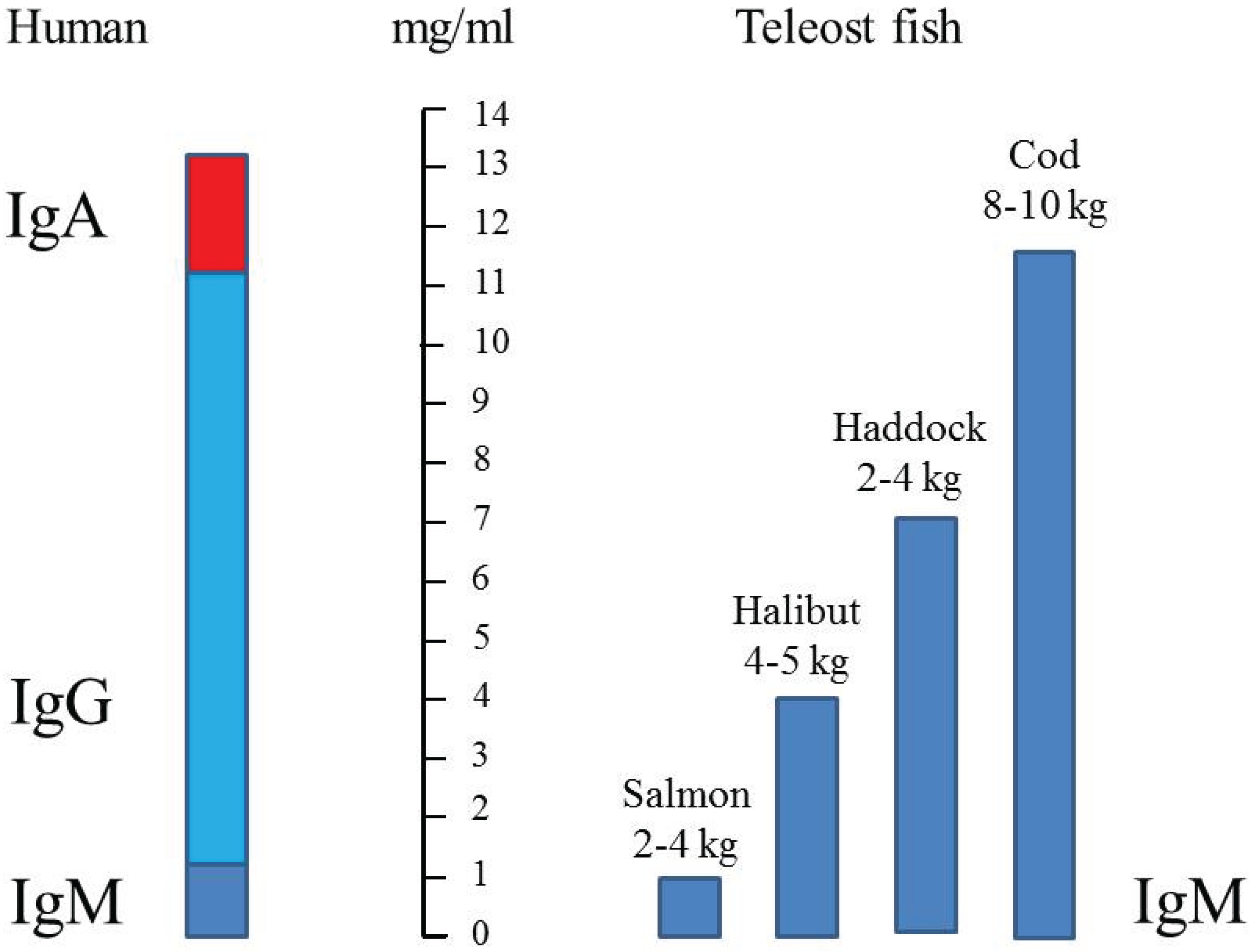
6. Immunoglobulin Concentration in Serum
| Species | Fish | Number of Fish | mg/mL Serum | Reference |
|---|---|---|---|---|
| Atlantic salmon | farm 2 to 8 kg | 10 | 0.8 to 1.3 | [31] |
| Atlantic salmon | farm 2.5 to 4 kg | 2–3 | ~1 | [40] |
| Brown trout | un-injected | 104 | 7.4 ± 0.3 | [47] |
| PBS-injected | 70 | 6.7 ± 0.2 | [47] | |
| antigen-stimulated | 399 | 7.5 to 16.8 | [47] | |
| Rainbow trout | stream | 58 | 3.3 ± 2.1 | [48] |
| farm | 25 | 2.2 ± 0.7 | [48] | |
| farm VHS/ERM | 40 | 4.2 ± 1.5 | [48] | |
| farm PKD | 10 | 10.9 ± 5.3 | [48] | |
| aquarium | 20 | 1.5 ± 0.4 | [48] | |
| Rainbow trout | 20 g (temp 19 °C) | 10 | 0.67 ± 0.66 | [49] |
| 125 g (temp 19 °C) | 15 | 9.1 ± 4.17 | [49] | |
| >1 kg (temp 19 °C) | 12 | 9.36 ± 2.78 | [49] | |
| >1 kg (temp 7 °C) | 12 | 3.51 ± 1.18 | [49] | |
| Masu salmon | larvae 88–235 dph | 100 + 50 (pooled) | <0.1 | [50] |
| 251–429 dph | 59 | 0.69 ± 0.037 | [50] | |
| adult males | 108 | 2.273 ± 118 | [50] | |
| adult females | 125 | 2.049 ± 97 | [50] | |
| Coho salmon | 50–200 g | 3 groups | 2.1 ± 0.1 | [51] |
7. Conclusions
Acknowledgements
Conflicts of Interest
References
- Hikima, J.-I.; Jung, T.-S.; Aoki, T. Immunoglobulin genes and their transcriptional control in teleosts. Dev. Comp. Immunol. 2011, 35, 924–936. [Google Scholar] [PubMed]
- Hansen, J.D.; Landis, E.D.; Phillips, R.B. Discovery of a unique Ig heavy-chain isotype (IgT) in rainbow trout: Implications for a distinctive B cell developmental pathway in teleost fish. Proc. Natl. Acad. Sci. USA 2005, 102, 6919–6924. [Google Scholar] [CrossRef] [PubMed]
- Danilova, N.; Bussmann, J.; Jekosch, K.; Steiner, L.A. The immunoglobulin heavy-chain locus in zebrafish: Identification and expression of a previously unknown isotype, immunoglobulin Z. Nat. Immunol. 2005, 6, 295–302. [Google Scholar] [CrossRef] [PubMed]
- Hordvik, I. Identification of a novel immunoglobulin delta transcript and comparative analysis of the genes encoding IgD in Atlantic salmon and Atlantic halibut. Mol. Immunol. 2002, 39, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Ramirez-Gomez, F.; Greene, W.; Rego, K.; Hansen, J.D.; Costa, G.; Kataria, P.; Bromage, E.S. Discovery and characterization of secretory IgD in rainbow trout: Secretory IgD is produced through a novel splicing mechanism. J. Immunol. 2012, 188, 1341–1349. [Google Scholar] [CrossRef] [PubMed]
- Hordvik, I.; Thevarajan, J.; Samdal, I.; Bastani, N.; Krossøy, B. Molecular cloning and phylogenetic analysis of the Atlantic salmon immunoglobulin D gene. Scand. J. Immunol. 1999, 50, 202–210. [Google Scholar] [CrossRef] [PubMed]
- Tadiso, T.M.; Lie, K.K.; Hordvik, I. Molecular cloning of IgT from Atlantic salmon, and analysis of the relative expression of τ, μ and δ in different tissues. Vet. Immunol. Immunopathol. 2010, 139, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.-A.; Salinas, I.; Li, J.; Parra, D.; Bjork, S.; Xu, Z.; LaPatra, S.E.; Bartholomew, J.; Sunyer, J.O. IgT, a primitive immunoglobulin class specialized in mucosal immunity. Nat. Immunol. 2010, 11, 827–835. [Google Scholar] [CrossRef] [PubMed]
- Bengten, E.; Quiniou, S.M.; Stuge, T.B.; Katagiri, T.; Miller, N.W.; Clem, L.W.; Warr, G.W.; Wilson, M. The IgH locus of the channel catfish, Ictalurus punctatus, contains multiple constant region gene sequences: Different genes encode heavy chains of membrane and secreted IgD. J. Immunol. 2002, 169, 2488–2497. [Google Scholar] [CrossRef] [PubMed]
- Rombout, J.H.; Abelli, L.; Picchietti, S.; Scapigliati, G.; Kiron, V. Teleost intestinal immunology. Fish Shellfish Immunol. 2011, 31, 616–626. [Google Scholar] [CrossRef] [PubMed]
- Tadiso, T.M.; Sharma, A.; Hordvik, I. Analysis of polymeric immunoglobulin receptor-and CD300-like molecules from Atlantic salmon. Mol. Immunol. 2011, 49, 462–473. [Google Scholar] [CrossRef] [PubMed]
- Kortum, A.N.; Rodriguez-Nunez, I.; Yang, J.; Shim, J.; Runft, D.; O’Driscoll, M.L.; Haire, R.N.; Cannon, J.P.; Turner, P.M.; Litman, R.T.; et al. Differential expression and ligand binding indicate alternative functions for zebrafish polymeric immunoglobulin receptor (pIgR) and a family of pIgR-like (PIGRL) proteins. Immunogenetics 2014, 66, 267–279. [Google Scholar] [CrossRef] [PubMed]
- Warr, G.W.; Marchalonis, J.J. Lymphocyte surface immunoglobulin of the goldfish differs from its serum counterpart. Dev. Comp. Immunol. 1977, 1, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Wilson, M.R.; Marcuz, A.; van Ginkel, F.; Miller, N.W.; Clem, L.W.; Middleton, D.; Warr, G.W. The immunoglobulin M heavy chain constant region gene of the channel catfish, Ictalurus punctatus: An unusual mRNA splice pattern produces the membrane form of the molecule. Nucleic Acid Res. 1990, 18, 5227–5233. [Google Scholar] [CrossRef] [PubMed]
- Wilson, M.; Bengten, E.; Miller, N.W.; Clem, L.W.; Du Pasquier, L.; Warr, G.W. A novel chimeric Ig heavy chain from a teleost fish shares similarities to IgD. Proc. Natl. Acad. Sci. USA 1997, 94, 4593–4597. [Google Scholar] [CrossRef] [PubMed]
- Yasuike, M.; de Boer, J.; von Schalburg, K.R.; Cooper, G.A.; McKinnel, L.; Messmer, A.; So, S.; Davidson, W.S.; Koop, B.F. Evolution of duplicated IgH loci in Atlantic salmon, Salmo salar. BMC Genomics 2010. [Google Scholar] [CrossRef]
- Jackson, K.J.L.; Kidd, M.J.; Wang, Y.; Collins, A.M. The shape of the lymphocyte receptor repertoire: Lessons from the B cell receptor. Front. Immunol. 2013. [Google Scholar] [CrossRef]
- Castro, R.; Jouneau, L.; Pham, H.-P.; Bouchez, O.; Giudicelli, V.; Lefranc, P.; Quillet, E.; Benmansour, A.; Cazals, F.; Six, A.; et al. Teleost fish mount complex clonal IgM and IgT responses in spleen upon systemic viral infection. PLOS Pathog. 2013, 9, e1003098. [Google Scholar] [CrossRef] [PubMed]
- Yaniv, K.; Isogai, S.; Castranova, D.; Dye, L.; Hitomi, J.; Weinstein, B.M. Life imaging of lymphatic development in the zebrafish. Nat. Med. 2006, 12, 711–716. [Google Scholar] [CrossRef] [PubMed]
- Haugarvoll, E.; Thorsen, J.; Laane, M.; Huang, Q.; Koppang, E.O. Melanogenesis and evidence for melanosome transport to the plasma membrane in a CD83+ teleost leukocyte cell line. Pigment Cell Res. 2006, 19, 214–225. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, N.M.S.; Taverne-Thiele, J.J.; Barnes, A.C.; van Muiswinkel, W.B.; Ellis, A.E.; Rombout, J.H.W.M. The gill is a major organ for antibody secreting cell production following direct immersion of sea bass (Dicentrachus labrax, L.) in a Photobacterium damselae ssp. Piscicida bacterin: An ontogenetic study. Fish Shellfish Immunol. 2001, 11, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Haugarvoll, E.; Bjerkås, I.; Nowak, B.F.; Hordvik, I.; Koppang, E.O. Identification and characterization of a novel intraepithelial lymphoid tissue in the gills of Atlantic salmon. J. Anat. 2008, 213, 202–209. [Google Scholar] [CrossRef] [PubMed]
- Koppang, E.O.; Fischer, U.; Moore, L.; Tranulis, M.A.; Dijkstra, J.M.; Köllner, B.; Aune, L.; Jirillo, E.; Hordvik, I. Salmonid T cells assemble in the thymus, spleen and in novel interbranchial lymphoid tissue. J. Anat. 2010, 217, 728–739. [Google Scholar] [CrossRef] [PubMed]
- Aas, I.B.; Austbø, L.; König, M.; Syed, M.; Falk, K.; Hordvik, I.; Koppang, E.O. Transcriptional characterization of the T cell population within the salmonid interbranchial lymphoid tissue. J. Immunol. 2014, 193, 3463–3469. [Google Scholar] [CrossRef] [PubMed]
- Castro, R.; Bromage, E.; Abos, B.; Pignatelli, J.; Granja, A.G.; Luque, A.; Tafalla, C. CCR7 is mainly expressed in teleost gills where it defines an IgD + IgM- B lymphocyte subset. J. Immunol. 2014, 192, 1257–1266. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Parra, D.; Gomez, D.; Salinas, I.; Zhang, Y.-A.; von Gersdorff Jørgensen, L.; Heinecke, R.D.; Buchmann, K.; LaPatra, S.; Sunyer, J.O. Teleost skin, an ancient mucosal surface that elicits gut-like immune responses. Proc. Natl. Acad. Sci. USA 2013, 110, 13097–13102. [Google Scholar] [CrossRef] [PubMed]
- Glasauer, S.M.K.; Neuhauss, S.C.F. Whole-genome duplication in teleost fishes and its evolutionary consequences. Mol. Genet. Genomics 2014. [Google Scholar] [CrossRef]
- Allendorf, F.W.; Thorgaard, G.H. Tetraploidy and the evolution of salmonid fishes. In Evolutionary Genetics of Fishes; Turner, B.J., Ed.; Plenum Press: New York, NY, USA, 1984; pp. 1–55. [Google Scholar]
- Berthelot, C.; Brunet, F.; Chalopin, D.; Juanchich, A.; Bernard, M.; Noe, B.; Bento, P.; Silva, C.D.; Labadie, K.; Alberti, A.; et al. The rainbow trout genome provides novel insights into evolution after whole-genome duplication in vertebrates. Nat. Commun. 2014. [Google Scholar] [CrossRef]
- Hordvik, I. The impact of ancestral tetraploidy on antibody heterogeneity in salmonid fishes. Immunol. Rev. 1998, 166, 153–157. [Google Scholar] [CrossRef]
- Haavarstein, L.S.; Aasjord, P.M.; Ness, S.; Endresen, C. Purification and partial characterization of an IgM-like serum immunoglobulin from Atlantic salmon (Salmo salar). Dev. Comp. Immunol. 1988, 12, 773–785. [Google Scholar] [CrossRef] [PubMed]
- Hordvik, I.; Voie, A.M.; Glette, J.; Male, R.; Endresen, C. Cloning and sequence analysis of two isotypic IgM heavy chain genes from Atlantic salmon, Salmo salar L. Eur. J. Immunol. 1992, 22, 2957–2962. [Google Scholar] [CrossRef] [PubMed]
- Hordvik, I.; Berven, F.S.; Solem, S.T.; Hatten, F.; Endresen, C. Analysis of two IgM isotypes in Atlantic salmon and brown trout. Mol. Immunol. 2002, 39, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Kamil, A.; Falk, K.; Sharma, A.; Raae, A.; Berven, F.; Koppang, E.O.; Hordvik, I. A monoclonal antibody distinguishes between two IgM heavy chain isotypes in Atlantic salmon and brown trout: Protein characterization, 3D modeling and epitope mapping. Mol. Immunol. 2011, 48, 1859–1867. [Google Scholar] [CrossRef] [PubMed]
- Kamil, A.; Raae, A.; Fjelldal, P.G.; Koppang, E.O.; Kari, F.E.; Hordvik, I. Comparative analysis of IgM sub-variants in salmonid fish and identification of a residue in µ3 which is essential for MAb4C10 reactivity. Fish Shellfish Immunol. 2013, 34, 667–672. [Google Scholar] [CrossRef] [PubMed]
- Kamil, A.; Fjelldal, P.G.; Hansen, T.; Raae, A.; Koppang, E.O.; Hordvik, I. Vaccination of Atlantic salmon leads to long-lasting higher levels of serum immunoglobulin and possible skewed ratios of two distinct IgM isotypes. Adv. Biosci. Biotechnol. 2013, 4, 85–90. [Google Scholar] [CrossRef]
- Hedfors, I.A.; Bakke, H.; Skjødt, K.; Grimholt, U. Antibodies recognizing both IgM isotypes in Atlantic salmon. Fish Shellfish Immunol. 2012, 33, 1199–1206. [Google Scholar] [CrossRef] [PubMed]
- Thuvander, A.; Fossum, C.; Lorenzen, N. Monoclonal-antibodies to salmonid immunoglobulin: Characterization and applicability in immunoassays. Dev. Comp. Immunol. 1990, 14, 415–423. [Google Scholar] [CrossRef] [PubMed]
- Acton, R.T.; Weinheimer, P.F.; Hall, S.J.; Niedermeier, W.; Shelton, E.; Bennett, J.C. Tetrameric immune macroglobulins in three orders of bony fishes. Proc. Natl. Acad. Sci. USA 1971, 68, 107–111. [Google Scholar] [CrossRef] [PubMed]
- Magnadottir, B. Comparison of immunoglobulin (IgM) from four fish species. Icel. Agric. Sci. 1998, 12, 47–59. [Google Scholar]
- Klimovich, V.B.; Samoilovich, M.P.; Klimovich, B.V. Problem of J-chain of immunoglobulins. J. Evol. Biochem. Physiol. 2008, 44, 151–166. [Google Scholar] [CrossRef]
- Czajkowsky, D.M.; Zhifeng, S. The human IgM pentamer is a mushroom-shaped molecule with flexural bias. Proc. Natl. Acad. Sci. USA 2009, 106, 14960–14965. [Google Scholar] [CrossRef] [PubMed]
- Getahun, A.; Lundqvist, M.; Middleton, D.; Warr, G.; Pilström, L. Influence of the μ-chain C-terminal sequence on polymerization of immunoglobulin M. Immunology 1999, 97, 408–413. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Bromage, E.S.; Kaattari, S.L. The strength of B cell interaction with antigen determines the degree of IgM polymerization. J. Immunol. 2010, 184, 844–850. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Bromage, E.; Kaattari, I.; Kaattari, S. Transduction of binding affinity by B lymphocytes: A new dimension in immunological regulation. Dev. Comp. Immunol. 2011, 35, 982–990. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Kaattari, I.M.; Ma, C.; Kaattari, S. The teleost humoral immune response. Fish Shellfish Immunol. 2013, 35, 1719–1728. [Google Scholar] [CrossRef] [PubMed]
- Ingram, G.A.; Alexander, J.B. Immunoglobulin of the brown trout, Salmo trutta and its concentration in the serum of antigen-stimulated and non-stimulated fish. J. Fish Biol. 1979, 14, 249–260. [Google Scholar] [CrossRef]
- Olesen, N.J.; Jørgensen, P.E. Quantification of serum immunoglobulin in rainbow trout Salmo gairdneri under various environmental conditions. Dis. Aquat. Org. 1986, 1, 183–189. [Google Scholar] [CrossRef]
- Sanchez, C.; Babin, M.; Tomillo, J.; Ubeira, F.M.; Dominguez, J. Quantification of low-levels of rainbow-trout immunoglobulin by enzyme-immunoassay using 2 monoclonal-antibodies. Vet. Immunol. Immunopathol. 1993, 36, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Fuda, H.; Soyano, K.; Yamazaki, F.; Hara, A. Serum immunoglobulin M (IgM) during early development of masu salmon (Oncorhynchus masou). Comp. Biochem. Physiol. A. Comp. Physiol. 1991, 99, 637–643. [Google Scholar] [CrossRef] [PubMed]
- Voss, E.W.; Groberg, W.J.; Fryer, J.L. Metabolism of coho salmon Ig: Catabolic rate of coho salmon tetrameric Ig in serum. Mol. Immunol. 1980, 17, 445–452. [Google Scholar] [CrossRef] [PubMed]
- Koppang, E.O.; Bjerkås, I.; Haugarvoll, E.; Chan, E.K.L.; Szabo, N.J.; Ono, N.; Akikusa, B.; Jirillo, E.; Poppe, T.T.; Sveier, H.; et al. Vaccination-induced systemic autoimmunity in farmed Atlantic salmon. J. Immunol. 2008, 181, 4807–4814. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hordvik, I. Immunoglobulin Isotypes in Atlantic Salmon, Salmo Salar. Biomolecules 2015, 5, 166-177. https://doi.org/10.3390/biom5010166
Hordvik I. Immunoglobulin Isotypes in Atlantic Salmon, Salmo Salar. Biomolecules. 2015; 5(1):166-177. https://doi.org/10.3390/biom5010166
Chicago/Turabian StyleHordvik, Ivar. 2015. "Immunoglobulin Isotypes in Atlantic Salmon, Salmo Salar" Biomolecules 5, no. 1: 166-177. https://doi.org/10.3390/biom5010166
APA StyleHordvik, I. (2015). Immunoglobulin Isotypes in Atlantic Salmon, Salmo Salar. Biomolecules, 5(1), 166-177. https://doi.org/10.3390/biom5010166




