The Hormesis Concept: Strengths and Shortcomings
Abstract
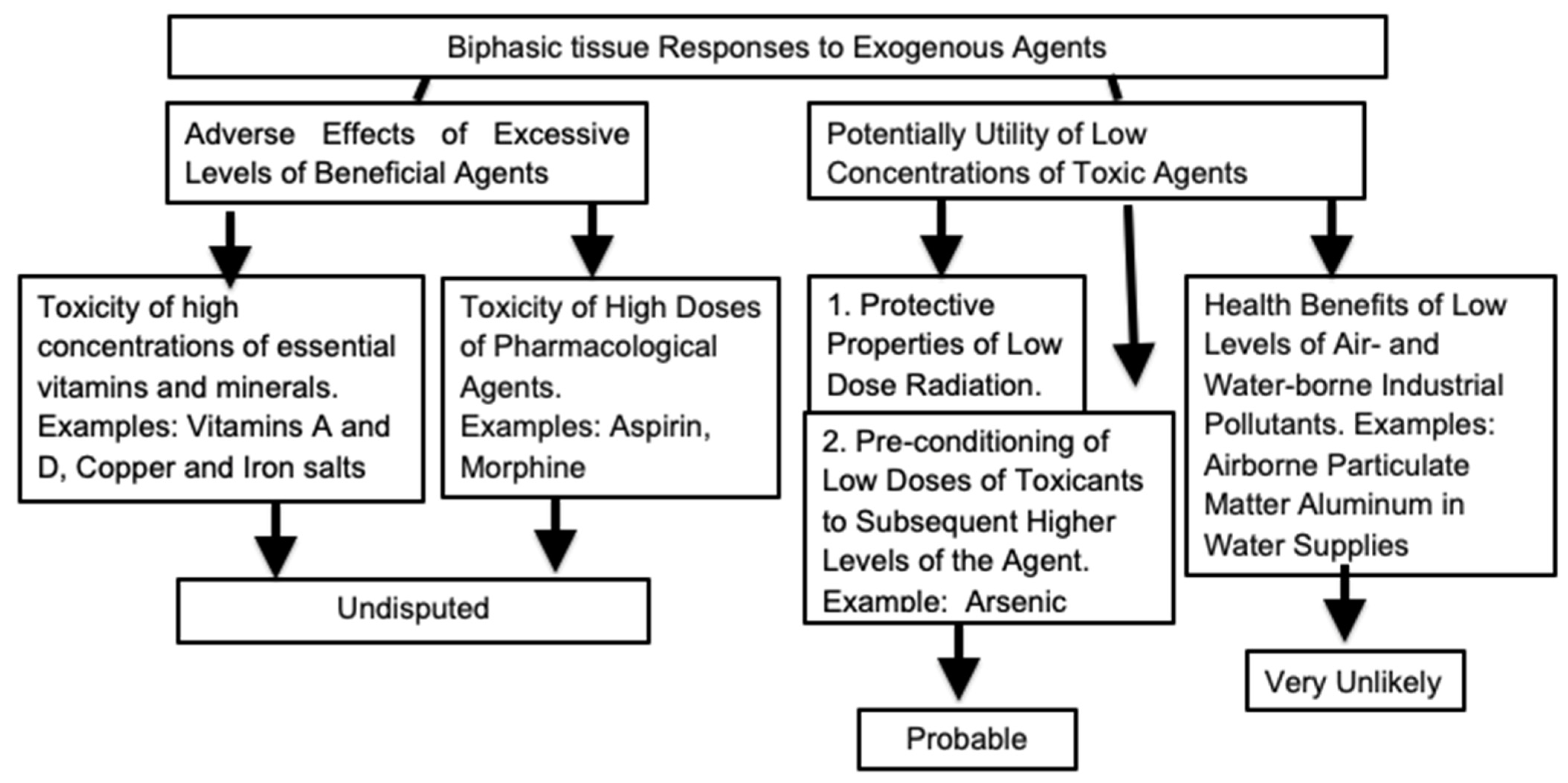
:1. Introduction
2. Problems with Evaluating the Current Literature
3. Intrinsic Events
4. Health Value of Moderate Exposure to Physical and Dietary Stressors
5. Dietary Components with Biphasic Effects on Wellbeing
6. Radiation and Hormesis
6.1. Epidemiological Reports
6.2. Clinical Reports
6.3. Findings from Isolated Systems and Animal Models
6.4. Closing Comment
7. Conclusions
Funding
Conflicts of Interest
References
- Schulz, H. Ueber hefegifte. Arch. Gesamte Physiol. Menschen Tiere 1888, 42, 517–541. [Google Scholar] [CrossRef]
- Overholser, B.R.; Foster, D.R. Opioid pharmacokinetic drug-drug interactions. Am. J. Manag. Care 2011, 17 (Suppl. S11), S276–S287. [Google Scholar]
- Schirrmacher, V. Less Can Be More: The Hormesis Theory of Stress Adaptation in the Global Biosphere and Its Implications. Biomedicines 2021, 9, 293. [Google Scholar] [CrossRef]
- Shrader-Frechette, K. Conceptual analysis and special-interest science: Toxicology and the case of Edward Calabrese. Synthese 2010, 177, 449–469. [Google Scholar] [CrossRef]
- Mushak, P.; Elliott, K.C. Structured Development and Promotion of a Research Field: Hormesis in Biology, Toxicology, and Environmental Regulatory Science. Kennedy Inst. Ethics J. 2015, 25, 335–367. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, E.J. Hormesis and homeopathy: A step forward. Homeopathy 2017, 106, 131–132. [Google Scholar] [CrossRef]
- Calabrese, E.J.; Giordano, J. Ultra low doses and biological amplification: Approaching Avogadro’s number. Pharmacol. Res. 2021, 170, 105738. [Google Scholar] [CrossRef]
- Jargin, S.V. Scientific Papers and Patents on Substances with Unproven Effects. Recent Pat. Drug Deliv. 2019, 13, 160–173. [Google Scholar] [CrossRef]
- Ullman, D. Exploring Possible Mechanisms of Hormesis and Homeopathy in the Light of Nanopharmacology and Ultra-High Dilutions. Dose Response 2021, 19, 15593258211022983. [Google Scholar] [CrossRef]
- Weltje, L.; vom Saal, F.S.; Oehlmann, J. Reproductive stimulation by low doses of xenoestrogens contrasts with the view of hormesis as an adaptive response. Hum. Exp. Toxicol. 2005, 24, 431–437. [Google Scholar] [CrossRef]
- Saal, F.S. Hormesis controversy. Environ. Sci. Technol. 2007, 41, 3. [Google Scholar] [CrossRef] [PubMed]
- Kendig, E.L.; Le, H.H.; Belcher, S.M. Defining hormesis: Evaluation of a complex concentration response phenomenon. Int. J. Toxicol. 2010, 29, 235–246. [Google Scholar] [CrossRef] [PubMed]
- Jodynis-Liebert, J.; Kujawska, M. Biphasic Dose-Response Induced by Phytochemicals: Experimental Evidence. J. Clin. Med. 2020, 9, 718. [Google Scholar] [CrossRef] [PubMed]
- Devrim-Lanpir, A.; Hill, L.; Knechtle, B. How N-Acetylcysteine Supplementation Affects Redox Regulation, Especially at Mitohormesis and Sarcohormesis Level: Curr. Perspect. Antioxidants 2021, 10, 153. [Google Scholar] [CrossRef]
- Mattson, M.P. Hormesis defined. Ageing Res. Rev. 2008, 7, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Yang, T.; Sun, Z. Hormesis in Health and Chronic Diseases. Trends Endocrinol. Metab. 2019, 30, 944–958. [Google Scholar] [CrossRef]
- Jîtcă, G.; Ősz, B.E.; Tero-Vescan, A.; Miklos, A.P.; Rusz, C.M.; Bătrînu, M.G.; Vari, C.E. Positive Aspects of Oxidative Stress at Different Levels of the Human Body: A Review. Antioxidants 2022, 11, 572. [Google Scholar] [CrossRef]
- Berry, R., 3rd; López-Martínez, G. A dose of experimental hormesis: When mild stress protects and improves animal performance. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2020, 242, 110658. [Google Scholar] [CrossRef]
- Duregon, E.; Pomatto-Watson, L.C.D.D.; Bernier, M.; Price, N.L.; de Cabo, R. Intermittent fasting: From calories to time restriction. Geroscience 2021, 43, 1083–1092. [Google Scholar] [CrossRef]
- Patrick, R.P.; Johnson, T.L. Sauna use as a lifestyle practice to extend healthspan. Exp. Gerontol. 2021, 154, 111509. [Google Scholar] [CrossRef]
- Kolodziej, F.; O’Halloran, K.D. Re-Evaluating the Oxidative Phenotype: Can Endurance Exercise Save the Western World? Antioxidants 2021, 10, 609. [Google Scholar] [CrossRef] [PubMed]
- Pinches, I.J.L.; Pinches, Y.L.; Johnson, J.O.; Haddad, N.C.; Boueri, M.G.; Oke, L.M.; Haddad, G.E. Could “cellular exercise” be the missing ingredient in a healthy life? Diets, caloric restriction, and exercise-induced hormesis. Nutrition 2022, 99–100, 111629. [Google Scholar] [CrossRef] [PubMed]
- Powers, S.K.; Deminice, R.; Ozdemir, M.; Yoshihara, T.; Bomkamp, M.P.; Hyatt, H. Exercise-induced oxidative stress: Friend or foe? J. Sport Health Sci. 2020, 9, 415–425. [Google Scholar] [CrossRef] [PubMed]
- Martel, J.; Ojcius, D.M.; Ko, Y.F.; Ke, P.Y.; Wu, C.Y.; Peng, H.H.; Young, J.D. Hormetic Effects of Phytochemicals on Health and Longevity. Trends Endocrinol. Metab. 2019, 30, 335–346. [Google Scholar] [CrossRef]
- Epel, E.S. The geroscience agenda: Toxic stress, hormetic stress, and the rate of aging. Ageing Res Rev. 2020, 63, 101167. [Google Scholar] [CrossRef]
- Kolb, H.; Kempf, K.; Röhling, M.; Lenzen-Schulte, M.; Schloot, N.C.; Martin, S. Ketone bodies: From enemy to friend and guardian angel. BMC Med. 2021, 19, 313. [Google Scholar] [CrossRef]
- Bondy, S.C. Melatonin and Ageing in: Biochemistry and Cell Biology of Ageing: Part III Biomedical Science; Harris, R.J., Korolchuk, V.I., Eds.; Springer Nature: Basel, Switzerland, 2023; pp. 291–307. [Google Scholar]
- Ontario, M.L.; Siracusa, R.; Modafferi, S.; Scuto, M.; Sciuto, S.; Greco, V.; Bertuccio, M.P.; Trovato Salinaro, A.; Crea, R.; Calabrese, E.J.; et al. Potential prevention and treatment of neurodegenerative disorders by olive polyphenols and hidrox. Mech. Ageing Dev. 2022, 203, 111637. [Google Scholar] [CrossRef]
- Leger, K.A.; Charles, S.T.; Almeida, D.M. Let It Go: Lingering Negative Affect in Response to Daily Stressors Is Associated with Physical Health Years Later. Psychol. Sci. 2018, 29, 1283–1290. [Google Scholar] [CrossRef]
- Calabrese, E.J.; Mattson, M.P.; Calabrese, V. Resveratrol commonly displays hormesis: Occurrence and biomedical significance. Hum. Exp. Toxicol. 2010, 29, 980–1015. [Google Scholar] [CrossRef]
- Calabrese, E.J.; Dhawan, G.; Kapoor, R.; Mattson, M.P.; Rattan, S.I. Curcumin and hormesis with particular emphasis on neural cells. Food Chem. Toxicol. 2019, 129, 399–404. [Google Scholar] [CrossRef]
- Fang, E.F.; Waltz, T.B.; Kassahun, H.; Lu, Q.; Kerr, J.S.; Morevati, M.; Fivenson, E.M.; Wollman, B.N.; Marosi, K.; Wilson, M.A.; et al. Tomatidine enhances lifespan and healthspan in C. elegans through mitophagy induction via the SKN-1/Nrf2 pathway. Sci. Rep. 2017, 7, 46208. [Google Scholar] [CrossRef] [PubMed]
- Varela-López, A.; Romero-Márquez, J.M.; Navarro-Hortal, M.D.; Ramirez-Tortosa, C.L.; Battino, M.; Forbes-Hernández, T.Y.; Quiles, J.L. Dietary antioxidants and lifespan: Relevance of environmental conditions, diet, and genotype of experimental models. Exp. Gerontol. 2023, 178, 112221. [Google Scholar] [CrossRef] [PubMed]
- Alì, S.; Davinelli, S.; Accardi, G.; Aiello, A.; Caruso, C.; Duro, G.; Ligotti, M.E.; Pojero, F.; Scapagnini, G.; Candore, G. Healthy ageing and Mediterranean diet: A focus on hormetic phytochemicals. Mech. Ageing Dev. 2021, 200, 111592. [Google Scholar] [CrossRef]
- Sauer, S.; Plauth, A. Health-beneficial nutraceuticals-myth or reality? Appl. Microbiol. Biotechnol. 2017, 101, 951–961. [Google Scholar] [CrossRef]
- Kelly-Reif, K.; Sandler, D.P.; Shore, D.; Schubauer-Berigan, M.K.; Troester, M.A.; Nylander-French, L.; Richardson, D.B. Radon and cancer mortality among underground uranium miners in the Příbram region of the Czech Republic. Am. J. Ind. Med. 2020, 63, 859–867. [Google Scholar] [CrossRef] [PubMed]
- Kurkela, O.; Nevalainen, J.; Pätsi, S.M.; Kojo, K.; Holmgren, O.; Auvinen, A. Lung cancer incidence attributable to residential radon exposure in Finland. Radiat. Environ. Biophys. 2023, 62, 35–49. [Google Scholar] [CrossRef] [PubMed]
- Scott, B.R. Residential radon appears to prevent lung cancer. Dose Response 2011, 9, 444–464. [Google Scholar] [CrossRef]
- Thompson, R.E. Epidemiological Evidence for Possible Radiation Hormesis from Radon Exposure: A Case-Control Study Conducted in Worcester, MA. Dose Response 2010, 9, 59–75. [Google Scholar] [CrossRef]
- Lehrer, S.; Green, S.; Rosenzweig, K.E. Reduced Ovarian Cancer Incidence in Women Exposed to Low Dose Ionizing Background Radiation or Radiation to the Ovaries after Treatment for Breast Cancer or Rectosigmoid Cancer. Asian Pac. J. Cancer Prev. 2016, 17, 2979–2982. [Google Scholar]
- Feinendegen, L.E. Evidence for beneficial low level radiation effects and radiation hormesis. Br. J. Radiol. 2005, 78, 3–7. [Google Scholar] [CrossRef]
- John, E.M.; Phipps, A.I.; Knight, J.A.; Milne, R.L.; Dite, G.S.; Hopper, J.L.; Andrulis, I.L.; Southey, M.; Giles, G.G.; West, D.W.; et al. Medical radiation exposure and breast cancer risk: Findings from the Breast Cancer Family Registry. Int. J. Cancer 2007, 21, 386–394. [Google Scholar] [CrossRef]
- David, E.; Bitan, R.; Atlas, S.; Wolfson, M.; Fraifeld, V.E. Correlative links between natural radiation and life expectancy in the US population. Biogerontology 2022, 23, 425–430. [Google Scholar] [CrossRef]
- Doss, M. Are We Approaching the End of the Linear No-Threshold Era? J. Nucl. Med. 2018, 59, 1786–1793. [Google Scholar] [CrossRef]
- Vaiserman, A.; Koliada, A.; Zabuga, O.; Socol, Y. Health Impacts of Low-Dose Ionizing Radiation: Current Scientific Debates and Regulatory Issues. Dose Response 2018, 16, 1559325818796331. [Google Scholar] [CrossRef]
- Kojima, S.; Thukimoto, M.; Cuttler, J.M.; Inoguchi, K.; Ootaki, T.; Shimura, N.; Koga, H.; Murata, A. Recovery From Rheumatoid Arthritis Following 15 Months of Therapy With Low Doses of Ionizing Radiation: A Case Report. Dose Response 2018, 16, 1559325818784719. [Google Scholar] [CrossRef]
- Mitsunobu, F.; Yamaoka, K.; Hanamoto, K.; Kojima, S.; Hosaki, Y.; Ashida, K.; Sugita, K.; Tanizaki, Y. Elevation of antioxidant enzymes in the clinical effects of radon and thermal therapy for bronchial asthma. J. Radiat. Res. 2003, 44, 95–99. [Google Scholar] [CrossRef] [PubMed]
- Park, B.S.; Hong, G.U.; Ro, J.Y. Foxp3(+)-Treg cells enhanced by repeated low-dose gamma-irradiation attenuate ovalbumin-induced allergic asthma in mice. Radiat. Res. 2013, 179, 570–583. [Google Scholar] [CrossRef] [PubMed]
- Le Reun, E.; Foray, N. Low-Dose Radiation Therapy (LDRT) against Cancer and Inflammatory or Degenerative Diseases: Three Parallel Stories with a Common Molecular Mechanism Involving the Nucleoshuttling of the ATM Protein? Cancers 2023, 15, 1482. [Google Scholar] [CrossRef] [PubMed]
- Dunlap, N.E.; van Berkel, V.; Cai, L. COVID-19 and low-dose radiation therapy. Radiat. Med. Prot. 2021, 2, 139–145. [Google Scholar] [CrossRef]
- Liu, S.Z. On radiation hormesis expressed in the immune system. Crit. Rev. Toxicol. 2003, 33, 431–441. [Google Scholar] [CrossRef]
- Lehrer, S.; Rosenzweig, K.E. Lung cancer hormesis in high impact states where nuclear testing occurred. Clin. Lung Cancer 2015, 16, 152–155. [Google Scholar] [CrossRef] [PubMed]
- Averbeck, D.; Rodriguez-Lafrasse, C. Role of Mitochondria in Radiation Responses: Epigenetic, Metabolic, and Signaling Impacts. Int. J. Mol. Sci. 2021, 22, 11047. [Google Scholar] [CrossRef] [PubMed]
- Fisher, D.R.; Weller, R.E. Carcinogenesis from inhaled (239)PuO(2) in beagles: Evidence for radiation homeostasis at low doses? Health Phys. 2010, 99, 357–362. [Google Scholar] [CrossRef] [PubMed]
- Song, K.H.; Jung, S.Y.; Kho, S.H.; Hwang, S.G.; Ha, H.; Nam, S.Y.; Song, J.Y. Effects of low-dose irradiation on mice with Escherichia coli-induced sepsis. Toxicol. Appl. Pharmacol. 2017, 333, 17–25. [Google Scholar] [CrossRef]
- Jargin, S.V. Radiation Safety and Hormesis. Front. Public Health 2020, 8, 278. [Google Scholar] [CrossRef]
- Lowe, D.; Roy, L.; Tabocchini, M.A.; Rühm, W.; Wakeford, R.; Woloschak, G.E.; Laurier, D. Radiation dose rate effects: What is new and what is needed? Radiat. Environ. Biophys. 2022, 61, 507–543. [Google Scholar] [CrossRef]
- Sutou, S. A message to Fukushima: Nothing to fear but fear itself. Genes Environ. 2016, 38, 12. [Google Scholar] [CrossRef]
- Mossman, K.L. Deconstructing radiation hormesis. Health Phys. 2001, 80, 263–269. [Google Scholar] [CrossRef]
- Cuttler, J.M. The LNT Issue Is About Politics and Economics, Not Safety. Dose Response 2020, 18, 1559325820949066. [Google Scholar] [CrossRef]
- Scott, B.R. Epidemiologic Studies Cannot Reveal the True Shape of the Dose-Response Relationship for Radon-Induced Lung Cancer. Dose Response 2019, 17, 1559325819828617. [Google Scholar] [CrossRef]
- Kino, K. The prospective mathematical idea satisfying both radiation hormesis under low radiation doses and linear non-threshold theory under high radiation doses. Genes Environ. 2020, 42, 4. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, A.; Bauer, M.A.; Kroemer, G.; Madeo, F.; Carmona-Gutierrez, D. When less is more: Hormesis against stress and disease. Microb. Cell 2014, 1, 150–153. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, E.J.; Kozumbo, W.J. The hormetic dose-response mechanism: Nrf2 activation. Pharmacol. Res. 2021, 167, 105526. [Google Scholar] [CrossRef] [PubMed]
- Beal, S.A.; Osterberg, E.C.; Zdanowicz, C.M.; Fisher, D.A. Ice Core Perspective on Mercury Pollution during the Past 600 Years. Environ. Sci. Technol. 2015, 49, 7641–7647. [Google Scholar] [CrossRef]
- Calabrese, E.J. Hormesis: Path and Progression to Significance. Int. J. Mol. Sci. 2018, 19, 2871. [Google Scholar] [CrossRef]
- Sikakana, P.; Roberts, R.A. A decade of toxicological trends: What the papers say. Toxicol. Res. 2020, 9, 676–682. [Google Scholar] [CrossRef]
- Lave, L.B. Hormesis: Policy implications. J. Appl. Toxicol. 2000, 20, 141–145. [Google Scholar] [CrossRef]
- Thayer, K.A.; Melnick, R.; Burns, K.; Davis, D.; Huff, J. Fundamental flaws of hormesis for public health decisions. Environ. Health Perspect. 2005, 113, 1271–1276. [Google Scholar] [CrossRef]
- Axelrod, D.; Burns, K.; Davis, D.; von Larebeke, N. “Hormesis”—An inappropriate extrapolation from the specific to the universal. Int. J. Occup. Environ. Health 2004, 10, 335–339. [Google Scholar] [CrossRef]
- Hoffmann, G.; Stempsey, W. The hormesis concept and risk assessment: Are there unique ethical and policy considerations? Hum. Exp. Toxicol. 2008, 27, 613–620. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bondy, S.C. The Hormesis Concept: Strengths and Shortcomings. Biomolecules 2023, 13, 1512. https://doi.org/10.3390/biom13101512
Bondy SC. The Hormesis Concept: Strengths and Shortcomings. Biomolecules. 2023; 13(10):1512. https://doi.org/10.3390/biom13101512
Chicago/Turabian StyleBondy, Stephen C. 2023. "The Hormesis Concept: Strengths and Shortcomings" Biomolecules 13, no. 10: 1512. https://doi.org/10.3390/biom13101512
APA StyleBondy, S. C. (2023). The Hormesis Concept: Strengths and Shortcomings. Biomolecules, 13(10), 1512. https://doi.org/10.3390/biom13101512





