The Role of Nutraceutical Supplements, Monacolin K and Astaxanthin, and Diet in Blood Cholesterol Homeostasis in Patients with Myopathy
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Procedure
2.3. Ethics Statement
2.4. Statistical Analyses
3. Results
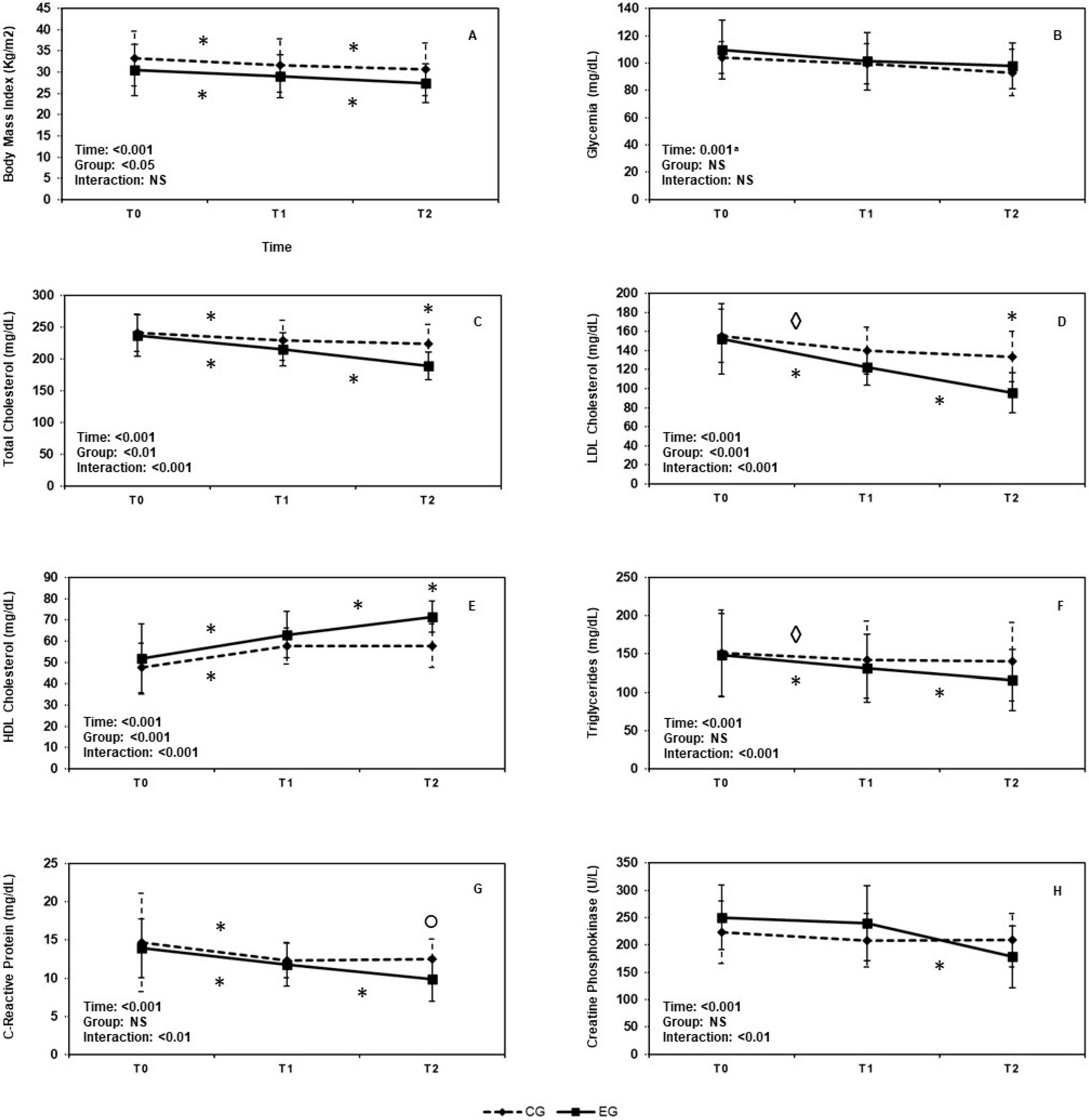
3.1. Mixed Factorial ANOVA
3.1.1. Body Mass Index
3.1.2. Glycemia
3.1.3. Serum Total Cholesterol Levels
3.1.4. LDL-Cholesterol Levels
3.1.5. HDL-Cholesterol Levels
3.1.6. Triglycerides Levels
3.1.7. C-Reactive Protein, CRP
3.1.8. Creatin Phosphokinase, CPK
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Franssen, R.; Monajemi, H.; Stroes, E.S.; Kastelein, J.J. Obesity and Dyslipidemia. Med. Clin. N. Am. 2011, 95, 893–902. [Google Scholar] [CrossRef] [PubMed]
- Găman, M.-A.; Cozma, M.-A.; Dobrică, E.-C.; Bacalbașa, N.; Bratu, O.G.; Diaconu, C.C. Dyslipidemia: A Trigger for Coronary Heart Disease in Romanian Patients with Diabetes. Metabolites 2020, 10, 195. [Google Scholar] [CrossRef] [PubMed]
- Pirro, M.; Mannarino, M.R.; Bianconi, V.; Simental-Mendía, L.E.; Bagaglia, F.; Mannarino, E.; Sahebkar, A. The effects of a nutraceutical combination on plasma lipids and glucose: A systematic review and meta-analysis of randomized controlled trials. Pharmacol. Res. 2016, 110, 76–88. [Google Scholar] [CrossRef] [PubMed]
- Cicero, A.F.; Fogacci, F.; Zambon, A. Red yeast rice for hypercholesterolemia: JACC focus seminar. J. Am. Coll. Cardiol. 2021, 77, 620–628. [Google Scholar] [CrossRef] [PubMed]
- Sciattella, P.; Maggioni, A.P.; Arcangeli, E.; Sidelnikov, E.; Kahangire, D.A.; Mennini, F.S. Healthcare Resource Utilization, Cardiovascular Event Rate and Use of Lipid-Lowering Therapies in Secondary Prevention of ASCVD in Hospitalized Patients in Italy. Adv. Ther. 2021, 39, 314–327. [Google Scholar] [CrossRef] [PubMed]
- Hirota, T.; Fujita, Y.; Ieiri, I. An updated review of pharmacokinetic drug interactions and pharmacogenetics of statins. Expert Opin. Drug Metab. Toxicol. 2020, 16, 809–822. [Google Scholar] [CrossRef] [PubMed]
- Thompson, P.D.; Panza, G.; Zaleski, A.; Taylor, B. Statin-associated side effects. J. Am. Coll. Cardiol. 2016, 67, 2395–2410. [Google Scholar] [CrossRef]
- Yebyo, H.G.; Aschmann, H.E.; Kaufmann, M.; Puhan, M.A. Comparative effectiveness and safety of statins as a class and of specific statins for primary prevention of cardiovascular disease: A systematic review, meta-analysis, and network meta-analysis of randomized trials with 94,283 participants. Am. Heart J. 2019, 210, 18–28. [Google Scholar] [CrossRef]
- Mach, F.; Ray, K.K.; Wiklund, O.; Corsini, A.; Catapano, A.L.; Bruckert, E.; De Backer, G.; Hegele, R.A.; Hovingh, G.K.; Jacobson, T.A.; et al. European Atherosclerosis Society Consensus Panel. Adverse effects of statin therapy: Perception vs. the evidence—Focus on glucose homeostasis, cognitive, renal and hepatic function, haemorrhagic stroke and cataract. Eur. Heart J. 2018, 39, 2526–2539. [Google Scholar] [CrossRef]
- Milajerdi, A.; Larijani, B.; Esmaillzadeh, A. Statins influence biomarkers of low grade inflammation in apparently healthy people or patients with chronic diseases: A systematic review and meta-analysis of randomized clinical trials. Cytokine 2019, 123, 154752. [Google Scholar] [CrossRef]
- Gaist, D.; Rodríguez, L.A.G.; Huerta, C.; Hallas, J.; Sindrup, S. Lipid-Lowering Drugs and Risk of Myopathy: A Population-Based Follow-Up Study. Epidemiology 2001, 12, 565–569. [Google Scholar] [CrossRef] [PubMed]
- Harper, C.R.; Jacobson, T. The broad spectrum of statin myopathy: From myalgia to rhabdomyolysis. Curr. Opin. Lipidol. 2007, 18, 401–408. [Google Scholar] [CrossRef] [PubMed]
- Taylor, B.A.; Thompson, P.D. Statin-Associated Muscle Disease: Advances in Diagnosis and Management. Neurotherapeutics 2018, 15, 1006–1017. [Google Scholar] [CrossRef]
- du Souich, P.; Roederer, G.; Dufour, R. Myotoxicity of statins: Mechanism of action. Pharmacol. Ther. 2017, 175, 1–16. [Google Scholar] [CrossRef]
- Koch, A.J.; Pereira, R.; Machado, M. The Creatine Kinase Response to Resistance Exercise. J. Musculoskelet Neuronal. Interact. 2014, 14, 68–77. [Google Scholar]
- Deichmann, R.; Lavie, C.; Andrews, S. Coenzyme q10 and statin-induced mitochondrial dysfunction. Ochsner J. 2010, 10, 16–21. [Google Scholar] [PubMed]
- Brown, A.J.; Ikonen, E.; Olkkonen, V.M. Cholesterol Precursors: More than Mere Markers of Biosynthesis. Curr. Opin. Lipidol. 2014, 25, 133–139. [Google Scholar] [CrossRef]
- Morikawa, S.; Murakami, T.; Yamazaki, H.; Izumi, A.; Saito, Y.; Hamakubo, T.; Kodama, T. Analysis of the Global RNA Expression Profiles of Skeletal Muscle Cells Treated with Statins. JAT 2005, 12, 121–131. [Google Scholar] [CrossRef]
- Flint, O.P.; Masters, B.A.; Gregg, R.E.; Durham, S.K. HMG CoA Reductase Inhibitor-Induced Myotoxicity: Pravastatin and Lovastatin Inhibit the Geranylgeranylation of Low-Molecular-Weight Proteins in Neonatal Rat Muscle Cell Culture. Toxicol. Appl. Pharmacol. 1997, 145, 99–110. [Google Scholar] [CrossRef]
- Hirota, T.; Ieiri, I. Drug–drug interactions that interfere with statin metabolism. Expert Opin. Drug Metab. Toxicol. 2015, 11, 1435–1447. [Google Scholar] [CrossRef]
- Kellick, K.A.; Bottorff, M.; Toth, P.P. A clinician’s guide to statin drug-drug interactions. J. Clin. Lipidol. 2014, 8, S30–S46. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J.D.; Brinton, E.A.; Ito, M.K.; Jacobson, T.A. Understanding Statin Use in America and Gaps in Patient Education (USAGE): An internet-based survey of 10,138 current and former statin users. J. Clin. Lipidol. 2012, 6, 208–215. [Google Scholar] [CrossRef]
- Laufs, U.; Filipiak, K.J.; Gouni-Berthold, I.; Catapano, A.L.; Mandraffino, G.; Benlian, P. Practical aspects in the management of statin-associated muscle symptoms (SAMS). Atheroscler. Suppl. 2017, 26, 45–55. [Google Scholar] [CrossRef]
- Li, J.J.; Liu, H.H.; Wu, N.Q.; Yeo, K.K.; Tan, K.; Ako, J.; Krittayaphong, R.; Tan, R.S.; Aylward, P.E.; Baek, S.H.; et al. Statin intolerance: An updated, narrative review mainly focusing on muscle adverse effects. Expert Opin. Drug Metab. Toxicol. 2020, 16, 837–851. [Google Scholar] [CrossRef]
- Mazzanti, G.; Moro, P.A.; Raschi, E.; Da Cas, R.; Menniti-Ippolito, F. Adverse reactions to dietary supplements containing red yeast rice: Assessment of cases from the Italian surveillance system. Br. J. Clin. Pharmacol. 2017, 83, 894–908. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Z.; Cao, X.; Wen, Q.; Chen, Z.; Cheng, Z.; Huang, X.; Zhang, Y.; Long, C.; Zhang, Y.; Huang, Z. An overview of the bioactivity of monacolin K/lovastatin. Food Chem. Toxicol. 2019, 131, 110585. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.; Karl, M.; Santini, A. Red Yeast Rice. Foods 2017, 6, 19. [Google Scholar] [CrossRef] [PubMed]
- Mueller, P.S. Symptomatic Myopathy due to Red Yeast Rice. Ann. Intern. Med. 2006, 145, 474–475. [Google Scholar] [CrossRef] [PubMed]
- Opinion of the French Agency for Food, Environmental and Occupational Health & Safety (ANSES) on the Risks Associated with the Presence of ‘Red Yeast Rice’ in Food Supplements. Available online: https://www.anses.fr/en/system/files/NUT2012sa0228EN.pdf (accessed on 14 February 2014).
- Russo, R.; Gallelli, L.; Cannataro, R.; Perri, M.; Calignano, A.; Citraro, R.; Russo, E.; Gareri, P.; Corsonello, A.; De Sarro, G. When Nutraceuticals Reinforce Drugs Side Effects: A Case Report. Curr. Drug Saf. 2016, 11, 264–266. [Google Scholar] [CrossRef]
- Landi, F.; Martone, A.M.; Salini, S.; Zazzara, B.; Calvani, R.; Marzetti, E.; Nesci, A.; Di Giorgio, A.; Giupponi, B.; Santoro, L.; et al. Effects of a New Combination of Medical Food on Endothelial Function and Lipid Profile in Dyslipidemic Subjects: A Pilot Randomized Trial. BioMed Res. Int. 2019, 2019, 1970878. [Google Scholar] [CrossRef]
- Fogacci, F.; Banach, M.; Mikhailidis, D.P.; Bruckert, E.; Toth, P.P.; Watts, G.F.; Reiner, Ž.; Mancini, J.; Rizzo, M.; Mitchenko, O.; et al. Lipid and Blood Pressure Meta-analysis Collaboration. Safety of red yeast rice supplementation: A systematic review and meta-analysis of randomized controlled trials. Pharmacol. Res. 2019, 143, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Cicero, A.F.G.; Morbini, M.; Parini, A.; Urso, R.; Rosticci, M.; Grandi, E.; Borghi, C. Effect of red yeast rice combined with antioxidants on lipid pattern, hs-CRP level, and endothelial function in moderately hypercholesterolemic subjects. Ther. Clin. Risk Manag. 2016, 12, 281–286. [Google Scholar] [CrossRef] [PubMed]
- Galletti, F.; Fazio, V.; Gentile, M.; Schillaci, G.; Pucci, G.; Battista, F.; Mercurio, V.; Bosso, G.; Bonaduce, D.; Brambilla, N.; et al. Efficacy of a nutraceutical combination on lipid metabolism in patients with metabolic syndrome: A multicenter, double blind, randomized, placebo controlled trial. Lipids Health Dis. 2019, 18, 66. [Google Scholar] [CrossRef] [PubMed]
- Zhuge, F.; Ni, Y.; Wan, C.; Liu, F.; Fu, Z. Anti-diabetic effects of astaxanthin on an STZ-induced diabetic model in rats. Endocr. J. 2021, 68, 451–459. [Google Scholar] [CrossRef] [PubMed]
- Mashhadi, N.S.; Zakerkish, M.; Mohammadiasl, J.; Zarei, M.; Mohammadshahi, M.; Haghighizadeh, M.H. Astaxanthin improves glucose metabolism and reduces blood pressure in patients with type 2 diabetes mellitus. Asia Pac. J. Clin. Nutr. 2018, 27, 341–346. [Google Scholar]
- Ni, Y.; Nagashimada, M.; Zhuge, F.; Zhan, L.; Nagata, N.; Tsutsui, A.; Shuichi, K.; Ota, T. Astaxanthin prevents and reverses diet-induced insulin resistance and steatohepatitis in mice: A comparison with vitamin E. Sci. Rep. 2015, 5, 17192. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.; Kim, J.-Y.; Jun, H.-J.; Kim, S.-J.; Lee, J.-H.; Hoang, M.H.; Hwang, K.-Y.; Um, S.-J.; Chang, H.I.; Lee, S.-J. The natural carotenoid astaxanthin, a PPAR-α agonist and PPAR-γ antagonist, reduces hepatic lipid accumulation by rewiring the transcriptome in lipid-loaded hepatocytes. Mol. Nutr. Food Res. 2012, 56, 878–888. [Google Scholar] [CrossRef] [PubMed]
- DiNicolantonio, J.J.; McCarty, M.; Okeefe, J. Astaxanthin plus berberine: A nutraceutical strategy for replicating the benefits of a metformin/fibrate regimen in metabolic syndrome. Open Heart 2019, 6, e000977. [Google Scholar] [CrossRef] [PubMed]
- Visioli, F.; Artaria, C. Astaxanthin in cardiovascular health and disease: Mechanisms of action, therapeutic merits, and knowledge gaps. Food Funct. 2016, 8, 39–63. [Google Scholar] [CrossRef] [PubMed]
- Capelli, B.; Talbott, S.; Ding, L. Astaxanthin sources: Suitability for human health and nutrition. Funct. Foods Health Dis. 2019, 9, 430. [Google Scholar] [CrossRef]
- Galasso, C.; Orefice, I.; Pellone, P.; Cirino, P.; Miele, R.; Ianora, A.; Brunet, C.; Sansone, C. On the Neuroprotective Role of Astaxanthin: New Perspectives? Mar. Drugs 2018, 16, 247. [Google Scholar] [CrossRef] [PubMed]
- La Torre, M.E.; Villano, I.; Monda, M.; Messina, A.; Cibelli, G.; Valenzano, A.; Pisanelli, D.; Panaro, M.A.; Tartaglia, N.; Ambrosi, A.; et al. Role of Vitamin E and the Orexin System in Neuroprotection. Brain Sci. 2021, 11, 1098. [Google Scholar] [CrossRef] [PubMed]
- Beutner, S.; Bloedorn, B.; Frixel, S.; Blanco, I.H.; Hoffmann, T.; Martin, H.-D.; Mayer, B.; Noack, P.; Ruck, C.; Schmidt, M.; et al. Quantitative Assessment of Antioxidant Properties of Natural Colorants and Phytochemicals: Carotenoids, Flavonoids, Phenols and Indigoids. The Role of β-Carotene in Antioxidant Functions: Antioxidant Properties of Natural Colorants and Phytochemicals. J. Sci. Food Agric. 2001, 81, 559–568. [Google Scholar] [CrossRef]
- Capelli, B.; Bagchi, D.; Cysewski, G.R. Synthetic astaxanthin is significantly inferior to algal-based astaxanthin as an antioxidant and may not be suitable as a human nutraceutical supplement. Nutrafoods 2013, 12, 145–152. [Google Scholar] [CrossRef]
- Fassett, R.G.; Coombes, J.S. Astaxanthin, oxidative stress, inflammation and cardiovascular disease. Futur. Cardiol. 2009, 5, 333–342. [Google Scholar] [CrossRef]
- Kurashige, M.; Okimasu, E.; Inoue, M.; Utsumi, K. Inhibition of oxidative injury of biological membranes by astaxanthin. Physiol. Chem. Phys. Med NMR 1990, 22, 27–38. [Google Scholar] [PubMed]
- Jia, Y.; Wu, C.; Kim, J.; Kim, B.; Lee, S.-J. Astaxanthin reduces hepatic lipid accumulations in high-fat-fed C57BL/6J mice via activation of peroxisome proliferator-activated receptor (PPAR) alpha and inhibition of PPAR gamma and Akt. J. Nutr. Biochem. 2015, 28, 9–18. [Google Scholar] [CrossRef]
- Lardi, C.R.; Garofalo, E.; Chieffi, S.; Gamboz, N.; La Marra, M.; Iavarone, A. Daily exposure to digital displays may affect the clock-drawing test: From psychometrics to serendipity. Neurol. Sci. 2020, 41, 3683–3690. [Google Scholar] [CrossRef]
- La Marra, M.; Villano, I.; Ilardi, C.R.; Carosella, M.; Staiano, M.; Iavarone, A.; Chieffi, S.; Messina, G.; Polito, R.; Porro, C.; et al. Executive Functions in Overweight and Obese Treatment-Seeking Patients: Cross-Sectional Data and Longitudinal Perspectives. Brain Sci. 2022, 12, 777. [Google Scholar] [CrossRef]
- Barrios, V.; Escobar, C.; Cicero, A.F.G.; Burke, D.; Fasching, P.; Banach, M.; Bruckert, E. A nutraceutical approach (Armolipid Plus) to reduce total and LDL cholesterol in individuals with mild to moderate dyslipidemia: Review of the clinical evidence. Atheroscler. Suppl. 2017, 24, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Ruscica, M.; Gomaraschi, M.; Mombelli, G.; Macchi, C.; Bosisio, R.; Pazzucconi, F.; Pavanello, C.; Calabresi, L.; Arnoldi, A.; Sirtori, C.R.; et al. Nutraceutical approach to moderate cardiometabolic risk: Results of a randomized, double-blind and crossover study with Armolipid Plus. J. Clin. Lipidol. 2014, 8, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Crispino, M.; Trinchese, G.; Penna, E.; Cimmino, F.; Catapano, A.; Villano, I.; Perrone-Capano, C.; Mollica, M.P. Interplay between Peripheral and Central Inflammation in Obesity-Promoted Disorders: The Impact on Synaptic Mitochondrial Functions. Int. J. Mol. Sci. 2020, 21, 5964. [Google Scholar] [CrossRef] [PubMed]
- Cavaliere, G.; Trinchese, G.; Bergamo, P.; De Filippo, C.; Mattace Raso, G.; Gifuni, G.; Putti, R.; Moni, B.H.; Canani, R.B.; Meli, R.; et al. Polyunsaturated fatty acids attenuate diet induced obesity and insulin resistance, modulating mitochondrial respiratory uncoupling in rat skeletal muscle. PLoS ONE 2016, 11, e0149033. [Google Scholar] [CrossRef]
- Di Maio, G.; Monda, V.; Messina, A.; Polito, R.; Monda, M.; Tartaglia, N.; Ambrosio, A.; Pisanelli, D.; Asmundo, A.; Nunno, N.D.; et al. Physical activity and modification of lifestyle induce benefits on the health status. Acta Med. Medit. 2020, 36, 1913. [Google Scholar]
- Villano, I.; La Marra, M.; Messina, A.; Di Maio, G.; Moscatelli, F.; Chieffi, S.; Monda, M.; Messina, G.; Monda, V. Effects of vegetarian and vegan nutrition on body composition in competitive futsal athletes. Prog. Nutr. 2021, 23, e2021126. [Google Scholar] [CrossRef]
- Iwabu, M.; Yamauchi, T.; Okada-Iwabu, M.; Sato, K.; Nakagawa, T.; Funata, M.; Yamaguchi, M.; Namiki, S.; Nakayama, R.; Tabata, M.; et al. Adiponectin and AdipoR1 regulate PGC-1α and mitochondria by Ca2+ and AMPK/SIRT1. Nature 2010, 464, 1313–1319. [Google Scholar] [CrossRef]
- Grahame Hardie, D. AMP-activated protein kinase: A key regulator of energy balance with many roles in human disease. J. Intern. Med. 2014, 276, 543–559. [Google Scholar] [CrossRef] [PubMed]
- Monda, V.; Villano, I.; Messina, A.; Valenzano, A.; Salerno, M.; Signorelli, S.S.; Monda, M.; Russo, V.; Sessa, F.; Messina, G. Aerobic exercise and Orexin A: Role of sympathetic activity and redox system. J. Biol. Regul. Homeost. Agents 2019, 33, 587–592. [Google Scholar] [PubMed]
- Herzig, S.; Shaw, R.J. AMPK: Guardian of metabolism and mitochondrial homeostasis. Nat. Rev. Mol. Cell Biol. 2018, 19, 121–135. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.J.; Kim, M.; Park, H.-S.; Kim, H.S.; Jeon, M.J.; Oh, K.S.; Koh, E.H.; Won, J.C.; Kim, M.-S.; Oh, G.T.; et al. AMPK activation increases fatty acid oxidation in skeletal muscle by activating PPARα and PGC-1. Biochem. Biophys. Res. Commun. 2006, 340, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Lundåsen, T.; Hunt, M.C.; Nilsson, L.-M.; Sanyal, S.; Angelin, B.; Alexson, S.E.; Rudling, M. PPARα is a key regulator of hepatic FGF21. Biochem. Biophys. Res. Commun. 2007, 360, 437–440. [Google Scholar] [CrossRef] [PubMed]
- Di Maio, G.; Alessio, N.; Demirsoy, I.; Peluso, G.; Perrotta, S.; Monda, M.; Di Bernardo, G. Evaluation of Browning Agents on the White Adipogenesis of Bone Marrow Mesenchymal Stromal Cells: A Contribution to Fighting Obesity. Cells 2021, 10, 403. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Marcos, P.J.; Auwerx, J. Regulation of PGC-1α, a nodal regulator of mitochondrial biogenesis. Am. J. Clin. Nutr. 2011, 93, 884S–890S. [Google Scholar] [CrossRef] [PubMed]
- Irrcher, I.; Ljubicic, V.; Kirwan, A.F.; Hood, D.A. AMP-Activated Protein Kinase-Regulated Activation of the PGC-1α Promoter in Skeletal Muscle Cells. PLoS ONE 2008, 3, e3614. [Google Scholar] [CrossRef]
- Kohandel, Z.; Farkhondeh, T.; Aschner, M.; Pourbagher-Shahri, A.M.; Samarghandian, S. Anti-inflammatory action of astaxanthin and its use in the treatment of various diseases. Biomed. Pharmacother. 2021, 145, 112179. [Google Scholar] [CrossRef]
- Yoshida, H.; Yanai, H.; Ito, K.; Tomono, Y.; Koikeda, T.; Tsukahara, H.; Tada, N. Administration of natural astaxanthin increases serum HDL-cholesterol and adiponectin in subjects with mild hyperlipidemia. Atherosclerosis 2010, 209, 520–523. [Google Scholar] [CrossRef]
- Yamauchi, T.; Kamon, J.; Minokoshi, Y.; Ito, Y.; Waki, H.; Uchida, S.; Yamashita, S.; Noda, M.; Kita, S.; Ueki, K.; et al. Adiponectin stimulates glucose utilization and fatty-acid oxidation by activating AMP-activated protein kinase. Nat. Med. 2002, 8, 1288–1295. [Google Scholar] [CrossRef]
- Frühbeck, G.; Catalán, V.; Rodríguez, A.; Ramírez, B.; Becerril, S.; Salvador, J.; Portincasa, P.; Colina, I.; Gómez-Ambrosi, J. Involvement of the leptin-adiponectin axis in inflammation and oxidative stress in the metabolic syndrome. Sci. Rep. 2017, 7, 6619. [Google Scholar] [CrossRef]
- Messina, A.; Monda, M.; Valenzano, A.; Messina, G.; Villano, I.; Moscatelli, F.; Cibelli, G.; Marsala, G.; Polito, R.; Ruberto, M.; et al. Functional Changes Induced by Orexin A and Adiponectin on the Sympathetic/Parasympathetic Balance. Front. Physiol. 2018, 9, 259. [Google Scholar] [CrossRef]
- Messina, A.; Monda, V.; Avola, R.; Moscatelli, F.; Avalenzano, A.A.V.; Villano, I.; Ruberto, M.; Monda, E.; La Marra, M.; Tafuri, D.; et al. Role of the orexin system on arousal, attention, feeding behaviour and sleep disorders. Acta Med. Medit. 2017, 4, 645. [Google Scholar] [CrossRef]
- Polito, R.; Francavilla, V.C.; Ambrosi, A.; Tartaglia, N.; Tafuri, D.; Monda, M.; Messina, A.; Sessa, F.; Di Maio, G.; Ametta, A.; et al. The Orexin-A Serum Levels Are Strongly Modulated by Physical Activity Intervention in Diabetes Mellitus Patients. J. Hum. Sport Exerc. 2020, 15, S244–S251. [Google Scholar]
- Tsubakio-Yamamoto, K.; Matsuura, F.; Koseki, M.; Oku, H.; Sandoval, J.C.; Inagaki, M.; Nakatani, K.; Nakaoka, H.; Kawase, R.; Yuasa-Kawase, M.; et al. Adiponectin prevents atherosclerosis by increasing cholesterol efflux from macrophages. Biochem. Biophys. Res. Commun. 2008, 375, 390–394. [Google Scholar] [CrossRef] [PubMed]
- Marazzi, G.; Cacciotti, L.; Pelliccia, F.; Iaia, L.; Volterrani, M.; Caminiti, G.; Sposato, B.; Massaro, R.; Grieco, F.; Rosano, G. Long-term effects of nutraceuticals (berberine, red yeast rice, policosanol) in elderly hypercholesterolemic patients. Adv. Ther. 2011, 28, 1105–1113. [Google Scholar] [CrossRef] [PubMed]
- Park, J.S.; Chyun, J.H.; Kim, Y.K.; Line, L.L.; Chew, B.P. Astaxanthin decreased oxidative stress and inflammation and enhanced immune response in humans. Nutr. Metab. 2010, 7, 18. [Google Scholar] [CrossRef]
- La Marra, M.; Caviglia, G.; Perrella, R. Using Smartphones When Eating Increases Caloric Intake in Young People: An Overview of the Literature. Front. Psychol. 2020, 11, 587886. [Google Scholar] [CrossRef]
- Monda, V.; la Marra, M.; Perrella, R.; Caviglia, G.; Iavarone, A.; Chieffi, S.; Messina, G.; Carotenuto, M.; Monda, M.; Messina, A. Obesity and brain illness: From cognitive and psychological evidences to obesity paradox. Diabetes. Metab. Syndr. Obes. Targets Ther. 2017, 10, 473–479. [Google Scholar] [CrossRef]
- Calder, P.C.; Ahluwalia, N.; Brouns, F.; Buetler, T.; Clement, K.; Cunningham, K.; Esposito, K.; Jönsson, L.S.; Kolb, H.; Lansink, M.; et al. Dietary factors and low-grade inflammation in relation to overweight and obesity. Br. J. Nutr. 2011, 106 (Suppl. S3), S1–S78. [Google Scholar] [CrossRef]
- Galmés, S.; Cifre, M.; Palou, A.; Oliver, P.; Serra, F. A Genetic Score of Predisposition to Low-Grade Inflammation Associated with Obesity May Contribute to Discern Population at Risk for Metabolic Syndrome. Nutrients 2019, 11, 298. [Google Scholar] [CrossRef]
- Villano, I.; La Marra, M.; Di Maio, G.; Monda, V.; Chieffi, S.; Guatteo, E.; Messina, G.; Moscatelli, F.; Monda, M.; Messina, A. Physiological Role of Orexinergic System for Health. Int. J. Environ. Res. Public Health 2022, 19, 8353. [Google Scholar] [CrossRef]
| T0 | T1 | T2 | ||||
|---|---|---|---|---|---|---|
| CG | EG | CG | EG | CG | EG | |
| BMI, mean (SD) | 33.26 (6.45) | 30.56 (6.03) | 31.58 (6.32) | 29.03 (5.1) | 30.63 (6.21) | 27.38 (4.53) |
| Glycemia mg/dL, mean (SD) | 104.18 (11.59) | 109.75 (21.58) | 99.45 (14.57) | 101.25 (21.26) | 93 (17.04) | 97,95 (16.74) |
| TC mg/dL, mean (SD) | 241.50 (29.8) | 237.05 (32.92) | 229.50 (32,01) | 215.60 (26.18) | 223.7 (30.76) | 189.15 (21.66) |
| LDL-C mg/dL, mean (SD) | 155.40 (27.98) | 152.30 (36.86) | 139.85 (24.61) | 122.15 (18.48) | 133.7 (26.49) | 95.65 (21.37) |
| HDL-C mg/dL, mean (SD) | 47.45 (11.56) | 51.70 (16.57) | 57.70 (8.43) | 63.10 (10.81) | 57.80 (10.31) | 71.50 (7.36) |
| TG mg/dL, mean (SD) | 151.65 (56.21) | 148.95 (54.38) | 142.45 (50.49) | 131,60 (44.33) | 140.35 (51.3) | 115.95 (39.98) |
| CRP mg/L, mean (SD) | 14.66 (6.47) | 13.91 (3.86) | 12.35 (2.33) | 11.74 (2.82) | 12.47 (2.66) | 9.86 (2.89) |
| CPK U/L, mean (SD) | 223.49 (56.9) | 250.31 (59.05) | 208.37 (48.83) | 239.47 (68.6) | 208.97 (48.63) | 178.32 (56.62) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Villano, I.; La Marra, M.; Allocca, S.; Ilardi, C.R.; Polito, R.; Porro, C.; Chieffi, S.; Messina, G.; Monda, V.; Di Maio, G.; et al. The Role of Nutraceutical Supplements, Monacolin K and Astaxanthin, and Diet in Blood Cholesterol Homeostasis in Patients with Myopathy. Biomolecules 2022, 12, 1118. https://doi.org/10.3390/biom12081118
Villano I, La Marra M, Allocca S, Ilardi CR, Polito R, Porro C, Chieffi S, Messina G, Monda V, Di Maio G, et al. The Role of Nutraceutical Supplements, Monacolin K and Astaxanthin, and Diet in Blood Cholesterol Homeostasis in Patients with Myopathy. Biomolecules. 2022; 12(8):1118. https://doi.org/10.3390/biom12081118
Chicago/Turabian StyleVillano, Ines, Marco La Marra, Salvatore Allocca, Ciro Rosario Ilardi, Rita Polito, Chiara Porro, Sergio Chieffi, Giovanni Messina, Vincenzo Monda, Girolamo Di Maio, and et al. 2022. "The Role of Nutraceutical Supplements, Monacolin K and Astaxanthin, and Diet in Blood Cholesterol Homeostasis in Patients with Myopathy" Biomolecules 12, no. 8: 1118. https://doi.org/10.3390/biom12081118
APA StyleVillano, I., La Marra, M., Allocca, S., Ilardi, C. R., Polito, R., Porro, C., Chieffi, S., Messina, G., Monda, V., Di Maio, G., & Messina, A. (2022). The Role of Nutraceutical Supplements, Monacolin K and Astaxanthin, and Diet in Blood Cholesterol Homeostasis in Patients with Myopathy. Biomolecules, 12(8), 1118. https://doi.org/10.3390/biom12081118










