Histamine Intolerance—A Kind of Pseudoallergic Reaction
Abstract
:1. Introduction
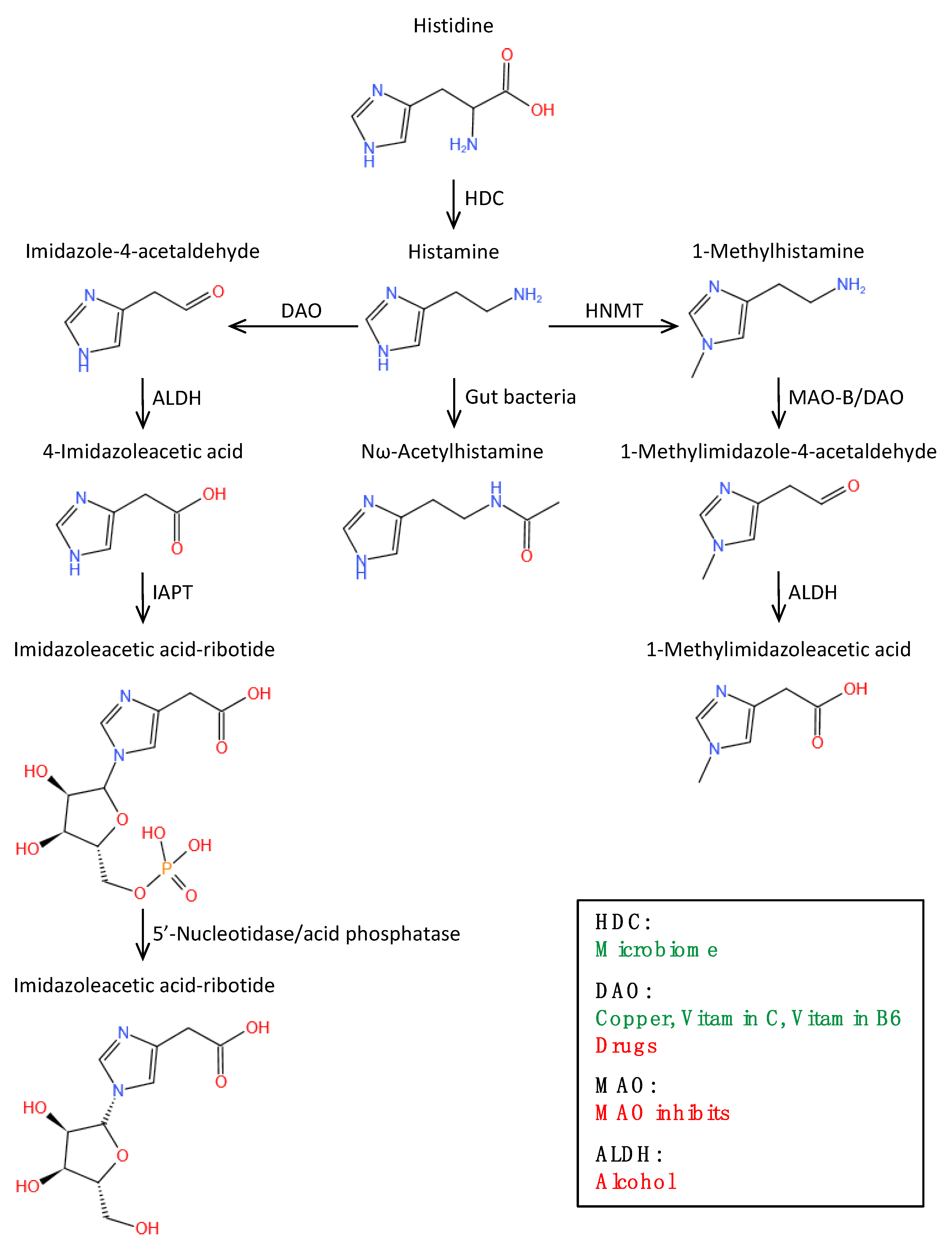
2. Histamine
2.1. Cellular Origin
2.2. Foods Rich in Histamine
2.3. Histamine Metabolic Pathway
3. Histamine Intolerance
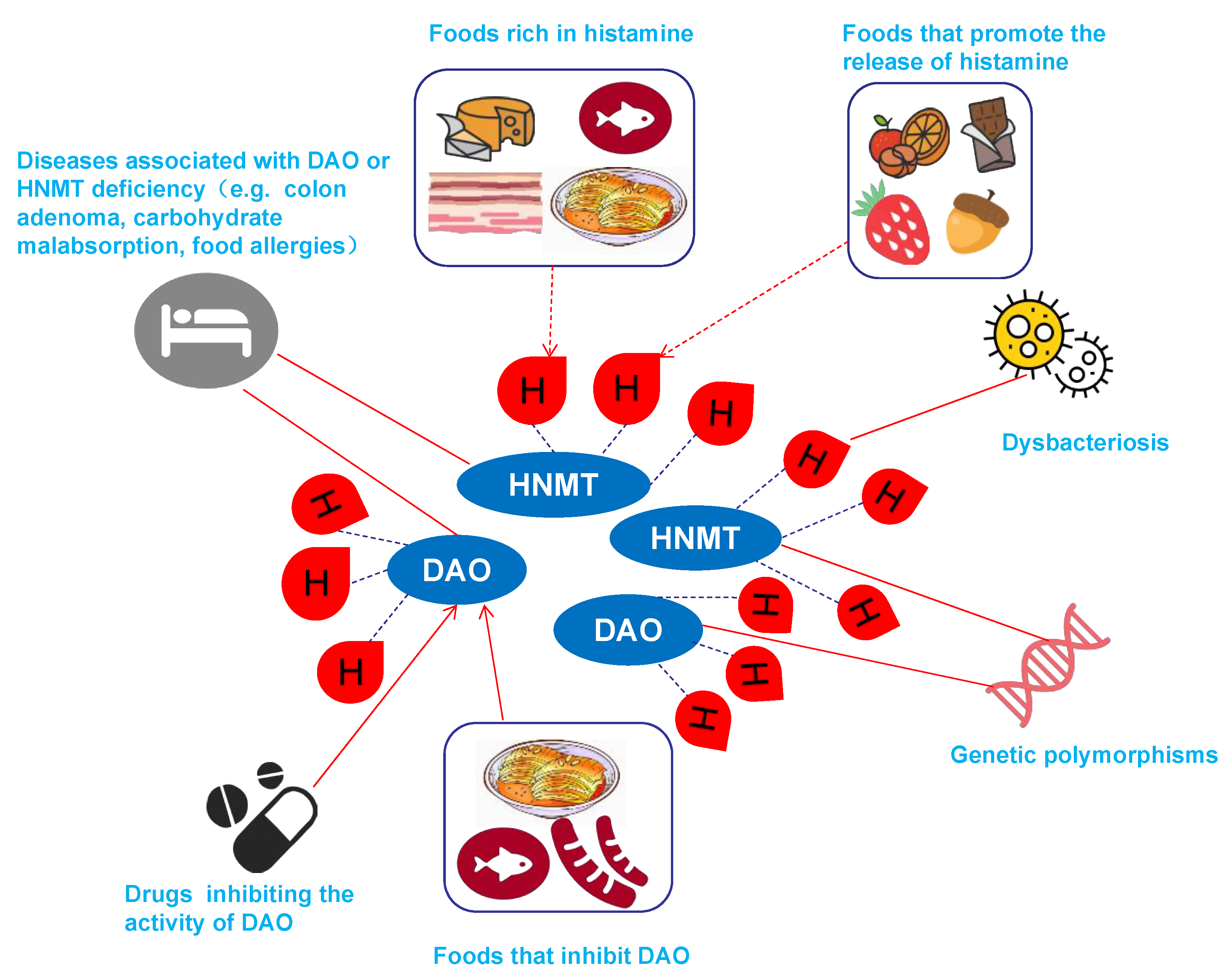
3.1. Causes of HIT
3.1.1. Genetically Induced HIT
3.1.2. HIT Associated with Dysbacteriosis
3.1.3. HIT Caused by Related Diseases
3.1.4. Drug-Induced HIT
3.1.5. Food-Induced HIT
3.2. Pathophysiological Mechanism of HIT
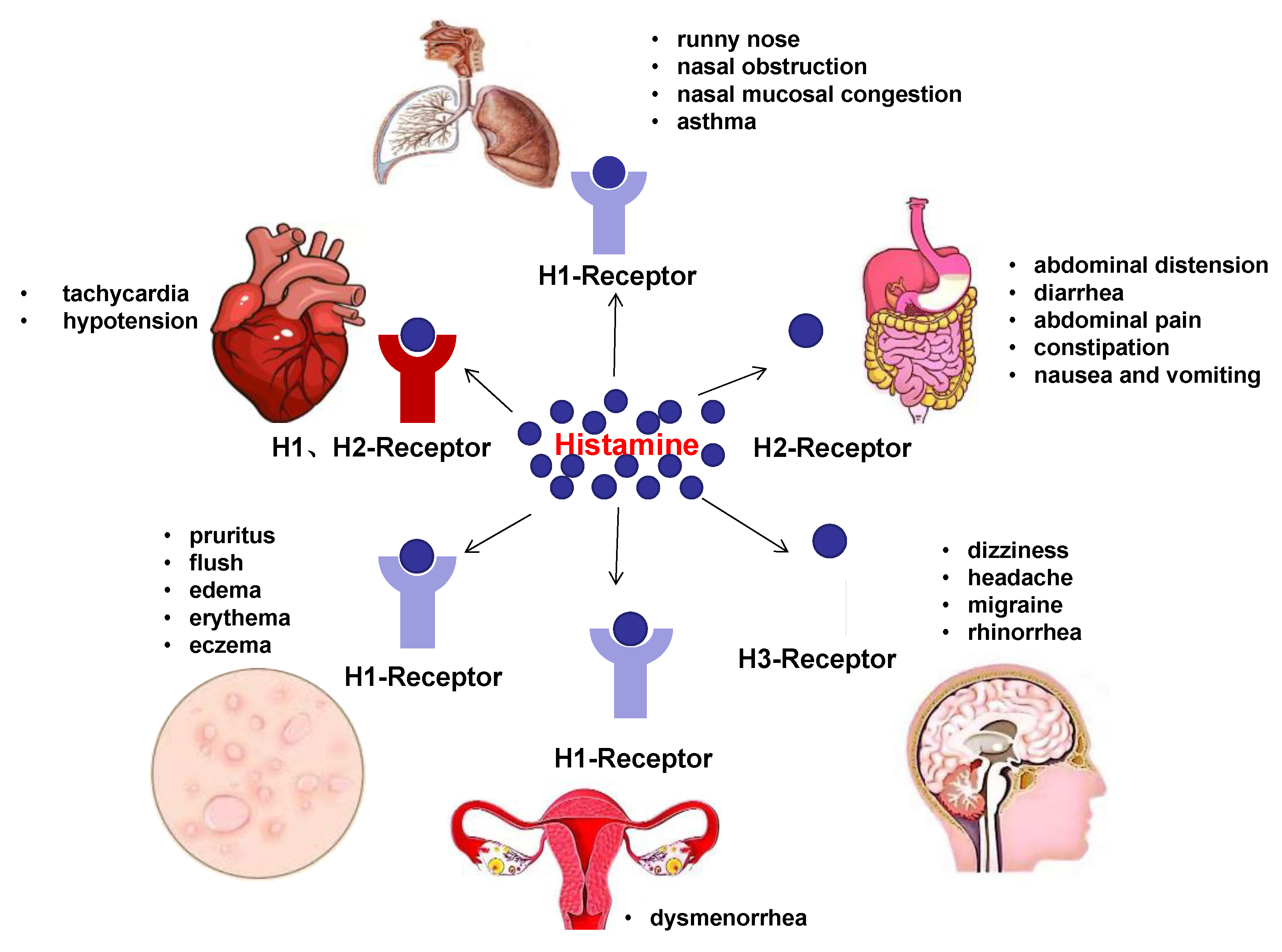
4. Clinical Manifestations of HIT
4.1. Skin
4.2. GI Tract
4.3. Respiratory System
4.4. Other Manifestations
4.5. Special Cases
4.5.1. Atopic Dermatitis (AD)
4.5.2. Chronic Urticaria (CU)
5. Differential Diagnostic Exclusion of Other Diseases
5.1. Food Allergy
5.2. Irritable Bowel Syndrome (IBS)
5.3. Other Food Intolerances and Celiac Disease (CD)
5.4. Helicobacter Pylori (HP) Infection
5.5. Eosinophilic Gastroenteritis (EGE)
5.6. Urticaria
5.7. Systemic Mastocytosis
6. Methods for Detecting HIT
6.1. History and Clinical Manifestations
6.2. DAO Concentration and Activity
6.3. Oral Histamine-Challenge Test
6.4. Histamine 50-Skin-Prick Test
6.5. Intestinal Biopsy
6.6. Diagnostic Therapy
6.7. Determination of Histamine and Its Metabolites in Urine
6.8. Determination of Histamine in Blood
6.9. SNPs of DAO Gene Assessment
6.10. To Rule Out Other Diseases with Similar Symptoms
7. Treatments of HIT
7.1. Antihistamine
7.2. Limit Histamine
7.3. Supplement of DAO
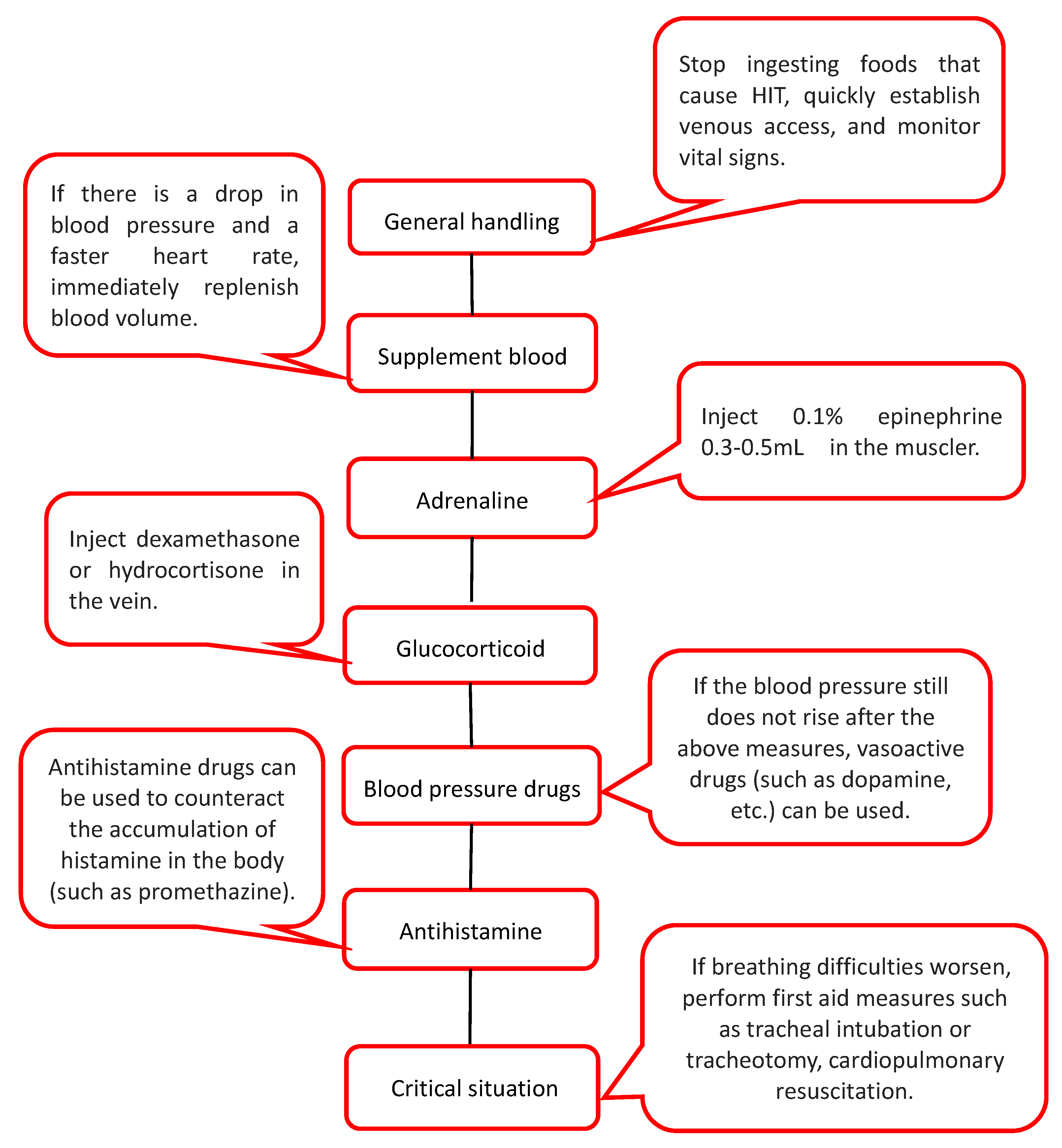
7.4. Treatment of Severe Urticaria Caused by HIT
7.5. Adjuvant Treatment of HIT
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Reese, I.; Ballmer-Weber, B.; Beyer, K.; Dölle-Bierke, S.; Kleine-Tebbe, J.; Klimek, L.; Lämmel, S.; Lepp, U.; Saloga, J.; Schäfer, C.; et al. Guideline on management of suspected adverse reactions to ingested histamine: Guideline of the German Society for Allergology and Clinical Immunology (DGAKI), the Society for Pediatric Allergology and Environmental Medicine (GPA), the Medical Association of German Allergologists (AeDA) as well as the Swiss Society for Allergology and Immunology (SGAI) and the Austrian Society for Allergology and Immunology (ÖGAI). Allergol. Sel. 2021, 5, 305–314. [Google Scholar] [CrossRef]
- Hrubisko, M.; Danis, R.; Huorka, M.; Wawruch, M. Histamine Intolerance-The More We Know the Less We Know. A Review. Nutrients 2021, 13, 2228. [Google Scholar] [CrossRef] [PubMed]
- Schnedl, W.J.; Lackner, S.; Enko, D.; Schenk, M.; Holasek, S.J.; Mangge, H. Evaluation of symptoms and symptom combinations in histamine intolerance. Intest. Res. 2019, 17, 427–433. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Komericki, P.; Klein, G.; Reider, N.; Hawranek, T.; Strimitzer, T.; Lang, R.; Kranzelbinder, B.; Aberer, W. Histamine intolerance: Lack of reproducibility of single symptoms by oral provocation with histamine: A randomised, double-blind, placebo-controlled cross-over study. Wien. Klin. Wochenschr. 2011, 123, 15–20. [Google Scholar] [CrossRef] [PubMed]
- Wöhrl, S.; Hemmer, W.; Focke, M.; Rappersberger, K.; Jarisch, R. Histamine intolerance-like symptoms in healthy volunteers after oral provocation with liquid histamine. Allergy Asthma Proc. 2004, 25, 305–311. [Google Scholar] [PubMed]
- Talley, N.J. What Causes Functional Gastrointestinal Disorders? A Proposed Disease Model. Am. J. Gastroenterol. 2020, 115, 41–48. [Google Scholar] [CrossRef]
- Schnedl, W.J.; Enko, D. Histamine Intolerance Originates in the Gut. Nutrients 2021, 13, 1262. [Google Scholar] [CrossRef]
- Frossi, B.; Tripodo, C.; Guarnotta, C.; Carroccio, A.; De Carli, M.; De Carli, S.; Marino, M.; Calabrò, A.; Pucillo, C.E. Mast cells are associated with the onset and progression of celiac disease. J. Allergy Clin. Immunol. 2017, 139, 1266–1274. [Google Scholar] [CrossRef] [Green Version]
- Sunkara, T.; Rawla, P.; Yarlagadda, K.S.; Gaduputi, V. Eosinophilic gastroenteritis: Diagnosis and clinical perspectives. Clin. Exp. Gastroenterol. 2019, 12, 239–253. [Google Scholar] [CrossRef] [Green Version]
- Tuck, C.J.; Biesiekierski, J.R.; Schmid-Grendelmeier, P.; Pohl, D. Food Intolerances. Nutrients 2019, 11, 1684. [Google Scholar] [CrossRef] [Green Version]
- Comas-Basté, O.; Sánchez-Pérez, S.; Veciana-Nogués, M.; Latorre-Moratalla, M.; Vidal-Carou, M. Histamine Intolerance: The Current State of the Art. Biomolecules 2020, 10, 1181. [Google Scholar] [CrossRef] [PubMed]
- Petersen, J.; Raithel, M.; Schwelberger, H. Characterisation of functional polymorphisms of the human diamine oxidase gene. Inflamm. Res. Off. J. Eur. Histamine Res. Soc. 2005, 54, S58–S59. [Google Scholar] [CrossRef] [PubMed]
- Petersen, J.; Drasche, A.; Raithel, M.; Schwelberger, H. Analysis of genetic polymorphisms of enzymes involved in histamine metabolism. Inflamm. Res. Off. J. Eur. Histamine Res. Soc. 2003, 52, S69–S70. [Google Scholar] [CrossRef] [PubMed]
- Schwelberger, H.; Drasche, A.; Petersen, J.; Raithel, M. Genetic polymorphisms of histamine degrading enzymes: From small-scale screening to high-throughput routine testing. Inflamm. Res. Off. J. Eur. Histamine Res. Soc. 2003, 52, S71–S73. [Google Scholar] [CrossRef] [PubMed]
- Petersen, J.; Raithel, M.; Schwelberger, H. Histamine N-methyltransferase and diamine oxidase gene polymorphisms in patients with inflammatory and neoplastic intestinal diseases. Inflamm. Res. Off. J. Eur. Histamine Res. Soc. 2002, 51, S91–S92. [Google Scholar] [CrossRef]
- Meza-Velázquez, R.; López-Márquez, F.; Espinosa-Padilla, S.; Rivera-Guillen, M.; Gutíerrez-Díaz, N.; Pérez-Armendáriz, L.; Rosales-González, M. Association between two polymorphisms of histamine-metabolising enzymes and the severity of allergic rhinitis in a group of Mexican children. Allergol. Immunopathol. 2016, 44, 433–438. [Google Scholar] [CrossRef]
- Kennedy, M.J.; Loehle, J.A.; Griffin, A.R.; Doll, M.A.; Kearns, G.L.; Sullivan, J.E.; Hein, D.W. Association of the histamine N-methyltransferase C314T (Thr105Ile) polymorphism with atopic dermatitis in Caucasian children. Pharmacotherapy 2008, 28, 1495–1501. [Google Scholar] [CrossRef] [Green Version]
- Deindl, P.; Peri-Jerkan, S.; Deichmann, K.; Niggemann, B.; Lau, S.; Sommerfeld, C.; Sengler, C.; Müller, S.; Wahn, U.; Nickel, R.; et al. No association of histamine- N-methyltransferase polymorphism with asthma or bronchial hyperresponsiveness in two German pediatric populations. Pediatr. Allergy Immunol. 2005, 16, 40–42. [Google Scholar] [CrossRef]
- Szczepankiewicz, A.; Bręborowicz, A.; Sobkowiak, P.; Popiel, A. Polymorphisms of two histamine-metabolizing enzymes genes and childhood allergic asthma: A case control study. Clin. Mol. Allergy 2010, 8, 14. [Google Scholar] [CrossRef] [Green Version]
- Anvari, S.; Vyhlidal, C.A.; Dai, H.; Jones, B.L. Genetic Variation along the Histamine Pathway in Children with Allergic versus Nonallergic Asthma. Am. J. Respir. Cell Mol. Biol. 2015, 53, 802–809. [Google Scholar] [CrossRef] [Green Version]
- Schink, M.; Konturek, P.C.; Tietz, E.; Dieterich, W.; Pinzer, T.C.; Wirtz, S.; Neurath, M.F.; Zopf, Y. Microbial patterns in patients with histamine intolerance. J. Physiol. Pharmacol. 2018, 69, 579–593. [Google Scholar] [CrossRef]
- Frei, R.; Ferstl, R.; Konieczna, P.; Ziegler, M.; Simon, T.; Rugeles, T.M.; Mailand, S.; Watanabe, T.; Lauener, R.; Akdis, C.A.; et al. Histamine receptor 2 modifies dendritic cell responses to microbial ligands. J. Allergy Clin. Immunol. 2013, 132, 194–204. [Google Scholar] [CrossRef] [PubMed]
- Sattler, J.; Lorenz, W. Intestinal diamine oxidases and enteral-induced histaminosis: Studies on three prognostic variables in an epidemiological model. J. Neural. Transm. Suppl. 1990, 32, 291–314. [Google Scholar] [PubMed]
- Maintz, L.; Novak, N. Histamine and histamine intolerance. Am. J. Clin. Nutr. 2007, 85, 1185–1196. [Google Scholar] [CrossRef]
- Rosell-Camps, A.; Zibetti, S.; Pérez-Esteban, G.; Vila-Vidal, M.; Ferrés-Ramis, L.; García-Teresa-García, E. Histamine intolerance as a cause of chronic digestive complaints in pediatric patients. Rev. Esp. Enferm. Dig. 2013, 105, 201–206. [Google Scholar] [CrossRef]
- Amon, U.; Bangha, E.; Küster, T.; Menne, A.; Vollrath, I.B.; Gibbs, B.F. Enteral histaminosis: Clinical implications. Inflamm. Res. Off. J. Eur. Histamine Res. Soc. 1999, 48, 291–295. [Google Scholar] [CrossRef]
- Bodmer, S.; Imark, C.; Kneubühl, M. Biogenic amines in foods: Histamine and food processing. Inflamm. Res. Off. J. Eur. Histamine Res. Soc. 1999, 48, 296–300. [Google Scholar] [CrossRef]
- Muñoz-Esparza, N.C.; Latorre-Moratalla, M.L.; Comas-Basté, O.; Toro-Funes, N.; Veciana-Nogués, M.T.; Vidal-Carou, M.C. Polyamines in Food. Front. Nutr. 2019, 6, 108. [Google Scholar] [CrossRef]
- Muñoz-Esparza, N.C.; Costa-Catala, J.; Comas-Basté, O.; Toro-Funes, N.; Latorre-Moratalla, M.L.; Veciana-Nogués, M.T.; Vidal-Carou, M.C. Occurrence of Polyamines in Foods and the Influence of Cooking Processes. Foods 2021, 10, 1752. [Google Scholar] [CrossRef]
- Enko, D.; Meinitzer, A.; Mangge, H.; Kriegshäuser, G.; Halwachs-Baumann, G.; Reininghaus, E.Z.; Bengesser, S.A.; Schnedl, W.J. Concomitant Prevalence of Low Serum Diamine Oxidase Activity and Carbohydrate Malabsorption. Can. J. Gastroenterol. Hepatol. 2016, 2016, 4893501. [Google Scholar] [CrossRef] [Green Version]
- Raithel, M.; Ulrich, P.; Keymling, J.; Hahn, E.G. Analysis and topographical distribution of gut diamine oxidase activity in patients with food allergy. Ann. N. Y. Acad. Sci. 1998, 859, 258–261. [Google Scholar] [CrossRef] [PubMed]
- Kuefner, M.A.; Schwelberger, H.G.; Hahn, E.G.; Raithel, M. Decreased histamine catabolism in the colonic mucosa of patients with colonic adenoma. Dig. Dis. Sci. 2008, 53, 436–442. [Google Scholar] [CrossRef] [PubMed]
- Schnedl, W.J.; Mangge, H.; Schenk, M.; Enko, D. Non-responsive celiac disease may coincide with additional food intolerance/malabsorption, including histamine intolerance. Med. Hypotheses 2021, 146, 110404. [Google Scholar] [CrossRef] [PubMed]
- Beltrán-Ortiz, C.; Peralta, T.; Ramos, V.; Durán, M.; Behrens, C.; Maureira, D.; Guzmán, M.A.; Bastias, C.; Ferrer, P. Standardization of a colorimetric technique for determination of enzymatic activity of diamine oxidase (DAO) and its application in patients with clinical diagnosis of histamine intolerance. World Allergy Organ. J. 2020, 13, 100457. [Google Scholar] [CrossRef]
- Manzotti, G.; Breda, D.; Di Gioacchino, M.; Burastero, S.E. Serum diamine oxidase activity in patients with histamine intolerance. Int. J. Immunopathol. Pharm. 2016, 29, 105–111. [Google Scholar] [CrossRef] [Green Version]
- Kofler, L.; Ulmer, H.; Kofler, H. Histamine 50-skin-prick test: A tool to diagnose histamine intolerance. ISRN Allergy 2011, 2011, 353045. [Google Scholar] [CrossRef] [Green Version]
- Pereira, A.R.; Araújo, A.N.; Montenegro, M.C.B.S.M.; Amorim, C.M.P.G. A simpler potentiometric method for histamine assessment in blood sera. Anal. Bioanal. Chem. 2020, 412, 3629–3637. [Google Scholar] [CrossRef]
- Comas-Basté, O.; Latorre-Moratalla, M.L.; Bernacchia, R.; Veciana-Nogués, M.T.; Vidal-Carou, M.C. New approach for the diagnosis of histamine intolerance based on the determination of histamine and methylhistamine in urine. J Pharm. Biomed. Anal. 2017, 145, 379–385. [Google Scholar] [CrossRef]
- Steinbrecher, I.; Hemmer, W.; Jarisch, R. Adverse reaction to the azo dye Pigment Red 170 in a tattoo. J. Dtsch. Dermatol. Ges. 2004, 2, 1007–1008. [Google Scholar]
- Kovacova-Hanuskova, E.; Buday, T.; Gavliakova, S.; Plevkova, J. Histamine, histamine intoxication and intolerance. Allergol. Immunopathol. 2015, 43, 498–506. [Google Scholar] [CrossRef]
- Mušič, E.; Korošec, P.; Šilar, M.; Adamič, K.; Košnik, M.; Rijavec, M. Serum diamine oxidase activity as a diagnostic test for histamine intolerance. Wien. Klin. Wochenschr. 2013, 125, 239–243. [Google Scholar] [CrossRef] [PubMed]
- Wantke, F.; Götz, M.; Jarisch, R. Histamine-free diet: Treatment of choice for histamine-induced food intolerance and supporting treatment for chronic headaches. Clin. Exp. Allergy 1993, 23, 982–985. [Google Scholar] [CrossRef] [PubMed]
- Son, J.H.; Chung, B.Y.; Kim, H.O.; Park, C.W. A Histamine-Free Diet Is Helpful for Treatment of Adult Patients with Chronic Spontaneous Urticaria. Ann. Dermatol. 2018, 30, 164–172. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- King, W.; McCargar, L.; Joneja, J.M.; Barr, S.I. Benefits of a Histamine-Reducing Diet for Some Patients with Chronic Urticaria and Angioedema. Can. J. Diet. Pract. Res. 2000, 61, 24–26. [Google Scholar]
- Böhn, L.; Störsrud, S.; Törnblom, H.; Bengtsson, U.; Simrén, M. Self-reported food-related gastrointestinal symptoms in IBS are common and associated with more severe symptoms and reduced quality of life. Am. J. Gastroenterol. 2013, 108, 634–641. [Google Scholar] [CrossRef] [Green Version]
- Maintz, L.; Benfadal, S.; Allam, J.-P.; Hagemann, T.; Fimmers, R.; Novak, N. Evidence for a reduced histamine degradation capacity in a subgroup of patients with atopic eczema. J. Allergy Clin. Immunol. 2006, 117, 1106–1112. [Google Scholar] [CrossRef]
- Wagner, N.; Dirk, D.; Peveling-Oberhag, A.; Reese, I.; Rady-Pizarro, U.; Mitzel, H.; Staubach, P. A Popular myth-low-histamine diet improves chronic spontaneous urticaria-fact or fiction? J. Eur. Acad. Dermatol. Venereol. 2017, 31, 650–655. [Google Scholar] [CrossRef] [Green Version]
- Joneja, J.M.V.; Carmona-Silva, C. Outcome of a Histamine-restricted Diet Based on Chart Audit. J. Nutr. Environ. Med. 2001, 11, 249–262. [Google Scholar] [CrossRef]
- Schnedl, W.J.; Schenk, M.; Lackner, S.; Enko, D.; Mangge, H.; Forster, F. Diamine oxidase supplementation improves symptoms in patients with histamine intolerance. Food Sci. Biotechnol. 2019, 28, 1779–1784. [Google Scholar] [CrossRef] [Green Version]
- Dressler, C.; Rosumeck, S.; Werner, R.N.; Magerl, M.; Metz, M.; Maurer, M.; Nast, A.; Zuberbier, T. Executive summary of the methods report for ‘The EAACI/GA LEN/EDF/WAO Guideline for the Definition, Classification, Diagnosis and Management of Urticaria. The 2017 Revision and Update’. Allergy 2018, 73, 1145–1146. [Google Scholar] [CrossRef]
- Riley, J.F. HISTAMINE AND SIR HENRY DALE. Br. Med. J. 1965, 1, 1488–1490. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smolinska, S.; Jutel, M.; Crameri, R.; O’Mahony, L. Histamine and gut mucosal immune regulation. Allergy 2014, 69, 273–281. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Panula, P.; Chazot, P.L.; Cowart, M.; Gutzmer, R.; Leurs, R.; Liu, W.L.S.; Stark, H.; Thurmond, R.L.; Haas, H.L. International Union of Basic and Clinical Pharmacology. XCVIII. Histamine Receptors. Pharmacol. Rev. 2015, 67, 601–655. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Z.; Kurashima, Y. Two Sides of the Coin: Mast Cells as a Key Regulator of Allergy and Acute/Chronic Inflammation. Cells 2021, 10, 1615. [Google Scholar] [CrossRef] [PubMed]
- Naila, A.; Flint, S.; Fletcher, G.; Bremer, P.; Meerdink, G. Control of biogenic amines in food-existing and emerging approaches. J. Food Sci. 2010, 75, R139–R150. [Google Scholar] [CrossRef] [Green Version]
- Shulpekova, Y.O.; Nechaev, V.M.; Popova, I.R.; Deeva, T.A.; Kopylov, A.T.; Malsagova, K.A.; Kaysheva, A.L.; Ivashkin, V.T. Food Intolerance: The Role of Histamine. Nutrients 2021, 13, 3207. [Google Scholar] [CrossRef]
- Grosman, N. Histamine: Biology and medical aspects. Oncology 2004, 359. [Google Scholar]
- Jarisch, R. Histamin-Intoleranz. Histamin und Seekrankheit; Springer: Berlin/Heidelberg, Germany, 2004. [Google Scholar]
- Beutling, D.M. Biogenic amines in nutrition. Arch. Fuer Lebensm. 1996, 47, 97–102. [Google Scholar]
- Gagic, M.; Jamroz, E.; Krizkova, S.; Milosavljevic, V.; Kopel, P.; Adam, V. Current Trends in Detection of Histamine in Food and Beverages. J. Agric. Food Chem. 2019, 67, 773–783. [Google Scholar] [CrossRef]
- Izquierdo-Pulido, M.; Hernández-Jover, T.; Mariné-Font, A.; Vidal-Carou, M.C. Biogenic Amines in European Beers. J. Agric. Food Chem. 2008, 44, 3159–3163. [Google Scholar] [CrossRef]
- Sánchez-Pérez, S.; Comas-Basté, O.; Rabell-González, J.; Veciana-Nogués, M.T.; Latorre-Moratalla, M.L.; Vidal-Carou, M.C. Biogenic Amines in Plant-Origin Foods: Are They Frequently Underestimated in Low-Histamine Diets? Foods 2018, 7, 205. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barnes, W.G.; Hough, L.B. Membrane-bound histamine N-methyltransferase in mouse brain: Possible role in the synaptic inactivation of neuronal histamine. J. Neurochem. 2002, 82, 1262–1271. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moriguchi, T.; Takai, J. Histamine and histidine decarboxylase: Immunomodulatory functions and regulatory mechanisms. Genes Cells 2020, 25, 443–449. [Google Scholar] [CrossRef] [PubMed]
- Leitner, R.; Zoernpfenning, E.; Missbichler, A. Evaluation of the inhibitory effect of various drugs / active ingredients on the activity of human diamine oxidase in vitro. Clin. Transl. Allergy 2014, 4, 23. [Google Scholar] [CrossRef] [Green Version]
- Colombo, F.; Cattaneo, P.; Confalonieri, E.; Bernardi, C. Histamine food poisonings: A systematic review and meta-analysis. Crit. Rev. Food Sci. Nutr. 2018, 58, 1131–1151. [Google Scholar] [CrossRef] [Green Version]
- Yu, Y.; Wang, P.; Bian, L.; Hong, S. Rare Death Via Histamine Poisoning Following Crab Consumption: A Case Report. J. Forensic Sci. 2018, 63, 980–982. [Google Scholar] [CrossRef]
- Linares, D.M.; Martín, M.C.; Ladero, V.; Alvarez, M.A.; Fernández, M. Biogenic amines in dairy products. Crit. Rev. Food Sci. Nutr. 2011, 51, 691–703. [Google Scholar] [CrossRef]
- Doeun, D.; Davaatseren, M.; Chung, M.-S. Biogenic amines in foods. Food Sci. Biotechnol. 2017, 26, 1463–1474. [Google Scholar] [CrossRef]
- Sattler, J.; Lorenz, W.; Kubo, K.; Schmal, A.; Sauer, S.; Lüben, L. Food-induced histaminosis under diamine oxidase (DAO) blockade in pigs: Further evidence of the key role of elevated plasma histamine levels as demonstrated by successful prophylaxis with antihistamines. Agents Actions 1989, 27, 212–214. [Google Scholar] [CrossRef]
- Akdis, C.A.; Simons, F.E.R. Histamine receptors are hot in immunopharmacology. Eur. J. Pharmacol. 2006, 533, 69–76. [Google Scholar] [CrossRef]
- Parsons, M.E.; Ganellin, C.R. Histamine and its receptors. Br. J. Pharmacol. 2006, 147, S127–S135. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maintz, L.; Yu, C.F.; Rodríguez, E.; Baurecht, H.; Bieber, T.; Illig, T.; Weidinger, S.; Novak, N. Association of single nucleotide polymorphisms in the diamine oxidase gene with diamine oxidase serum activities. Allergy 2011, 66, 893–902. [Google Scholar] [CrossRef] [PubMed]
- García-Martín, E.; García-Menaya, J.; Sánchez, B.; Martínez, C.; Rosendo, R.; Agúndez, J.A.G. Polymorphisms of histamine-metabolizing enzymes and clinical manifestations of asthma and allergic rhinitis. Clin. Exp. Allergy 2007, 37, 1175–1182. [Google Scholar] [CrossRef] [PubMed]
- Ayuso, P.; García-Martín, E.; Martínez, C.; Agúndez, J.A.G. Genetic variability of human diamine oxidase: Occurrence of three nonsynonymous polymorphisms and study of their effect on serum enzyme activity. Pharm. Genom. 2007, 17, 687–693. [Google Scholar] [CrossRef] [PubMed]
- Messlinger, K. Migraine: Where and how does the pain originate? Exp. Brain Res. 2009, 196, 179–193. [Google Scholar] [CrossRef]
- Ionescu, G.; Kiehl, R. Monoamine and diamine oxidase activities in atopic eczema. Allergy 1988, 43, 318–319. [Google Scholar] [CrossRef]
- Ring, J. Plasma histamine concentrations in atopic eczema. Clin. Allergy 1983, 13, 545–552. [Google Scholar] [CrossRef]
- Guida, B.; De Martino, C.D.; De Martino, S.D.; Tritto, G.; Patella, V.; Trio, R.; D’Agostino, C.; Pecoraro, P.; D’Agostino, L. Histamine plasma levels and elimination diet in chronic idiopathic urticaria. Eur. J. Clin. Nutr. 2000, 54, 155–158. [Google Scholar] [CrossRef] [Green Version]
- Yacoub, M.-R.; Ramirez, G.A.; Berti, A.; Mercurio, G.; Breda, D.; Saporiti, N.; Burastero, S.; Dagna, L.; Colombo, G. Diamine Oxidase Supplementation in Chronic Spontaneous Urticaria: A Randomized, Double-Blind Placebo-Controlled Study. Int. Arch. Allergy Immunol. 2018, 176, 268–271. [Google Scholar] [CrossRef]
- Siebenhaar, F.; Melde, A.; Magerl, M.; Zuberbier, T.; Church, M.K.; Maurer, M. Histamine intolerance in patients with chronic spontaneous urticaria. J. Eur. Acad. Dermatol. Venereol. 2016, 30, 1774–1777. [Google Scholar] [CrossRef]
- Harer, K.N.; Eswaran, S.L. Irritable Bowel Syndrome: Food as a Friend or Foe? Gastroenterol. Clin. N. Am. 2021, 50, 183–199. [Google Scholar] [CrossRef] [PubMed]
- Bai, T.; Wang, W.F.; Zhang, L.; Wang, H.; Qian, W.; Song, J.; Hou, X.H. Positive endoscopic and ultrasonographic findings in patients with symptom-diagnosed functional gastrointestinal disorder: Data from a Chinese cross-sectional study. J. Dig. Dis. 2018, 19, 759–765. [Google Scholar] [CrossRef] [PubMed]
- Schwelberger, H.G. Histamine intolerance: A metabolic disease? Inflamm. Res. Off. J. Eur. Histamine Res. Soc. 2010, 59, S219–S221. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Pérez, S.; Comas-Basté, O.; Veciana-Nogués, M.T.; Latorre-Moratalla, M.L.; Vidal-Carou, M.C. Low-Histamine Diets: Is the Exclusion of Foods Justified by Their Histamine Content? Nutrients 2021, 13, 1395. [Google Scholar] [CrossRef]
- San Mauro Martin, I.; Brachero, S.; Garicano Vilar, E. Histamine intolerance and dietary management: A complete review. Allergol. Immunopathol. 2016, 44, 475–483. [Google Scholar] [CrossRef]

| Food Categories | Histamine | Recommended Upper Limit for Histamine | ||
|---|---|---|---|---|
| mg/kg | mg/L | mg/kg | mg/L | |
| Fish (frozen/smoked or salted/canned) | 200 | |||
| Mackerel | 1–20/1–1788/ND–210 | |||
| Herring | 1–4/5–121/1–479 | |||
| Sardine | ND/14–150/3–2000 | |||
| Tuna | ND/ND/1–402 | |||
| Cheese | No recommendation | |||
| Gouda | 10–900 | |||
| Camembert | 0–1000 | |||
| Cheddar | 0–2100 | |||
| Emmental | 5–2500 | |||
| Swiss | 4–2500 | |||
| Parmesan | 10–581 | |||
| Meat | No recommendation | |||
| Fermented sausage | ND–650 | |||
| Salami | 1–654 | |||
| Fermented ham | 38–271 | |||
| Vegetables | ||||
| Sauerkraut | 0–229 | 10 | ||
| Spinach | 9–70 | |||
| Eggplant | 4–101 | |||
| Tomato | ND–17 | |||
| Ketchup | ND–22 | |||
| Avocado | ND–23 | |||
| Red wine vinegar | 4 | |||
| Alcohol | ||||
| White wine | ND–10 | 2 | ||
| Red wine | ND–30 | 2 | ||
| Top-fermented beer | ND–14 | |||
| Bottom-fermented beer | ND–17 | |||
| Champagne | 670 | |||
| Foods Rich in Histamine | Fish, sauerkraut, smoked meat products and cheeses |
| Foods that Promote the Release of Histamine | Citrus fruits, papaya, strawberries, egg whites, chocolate, nuts, fish, pork, cheese, fermented sausage, green peppers, wheat germ and bean sprouts |
| Foods that Competitively Inhibit DAO | Fish, fermented sausage, and sauerkraut |
| Anamnesis |
|
|
|
| Diagnostic Therapy (4–8 weeks) |
|
|
|
|
| Additional Tests |
|
|
|
|
|
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, Y.; Zhang, X.; Jin, H.; Chen, L.; Ji, J.; Zhang, Z. Histamine Intolerance—A Kind of Pseudoallergic Reaction. Biomolecules 2022, 12, 454. https://doi.org/10.3390/biom12030454
Zhao Y, Zhang X, Jin H, Chen L, Ji J, Zhang Z. Histamine Intolerance—A Kind of Pseudoallergic Reaction. Biomolecules. 2022; 12(3):454. https://doi.org/10.3390/biom12030454
Chicago/Turabian StyleZhao, Ying, Xiaoyan Zhang, Hengxi Jin, Lu Chen, Jiang Ji, and Zhongwei Zhang. 2022. "Histamine Intolerance—A Kind of Pseudoallergic Reaction" Biomolecules 12, no. 3: 454. https://doi.org/10.3390/biom12030454
APA StyleZhao, Y., Zhang, X., Jin, H., Chen, L., Ji, J., & Zhang, Z. (2022). Histamine Intolerance—A Kind of Pseudoallergic Reaction. Biomolecules, 12(3), 454. https://doi.org/10.3390/biom12030454






