Status and Prospects of Botanical Biopesticides in Europe and Mediterranean Countries
Abstract
1. Introduction
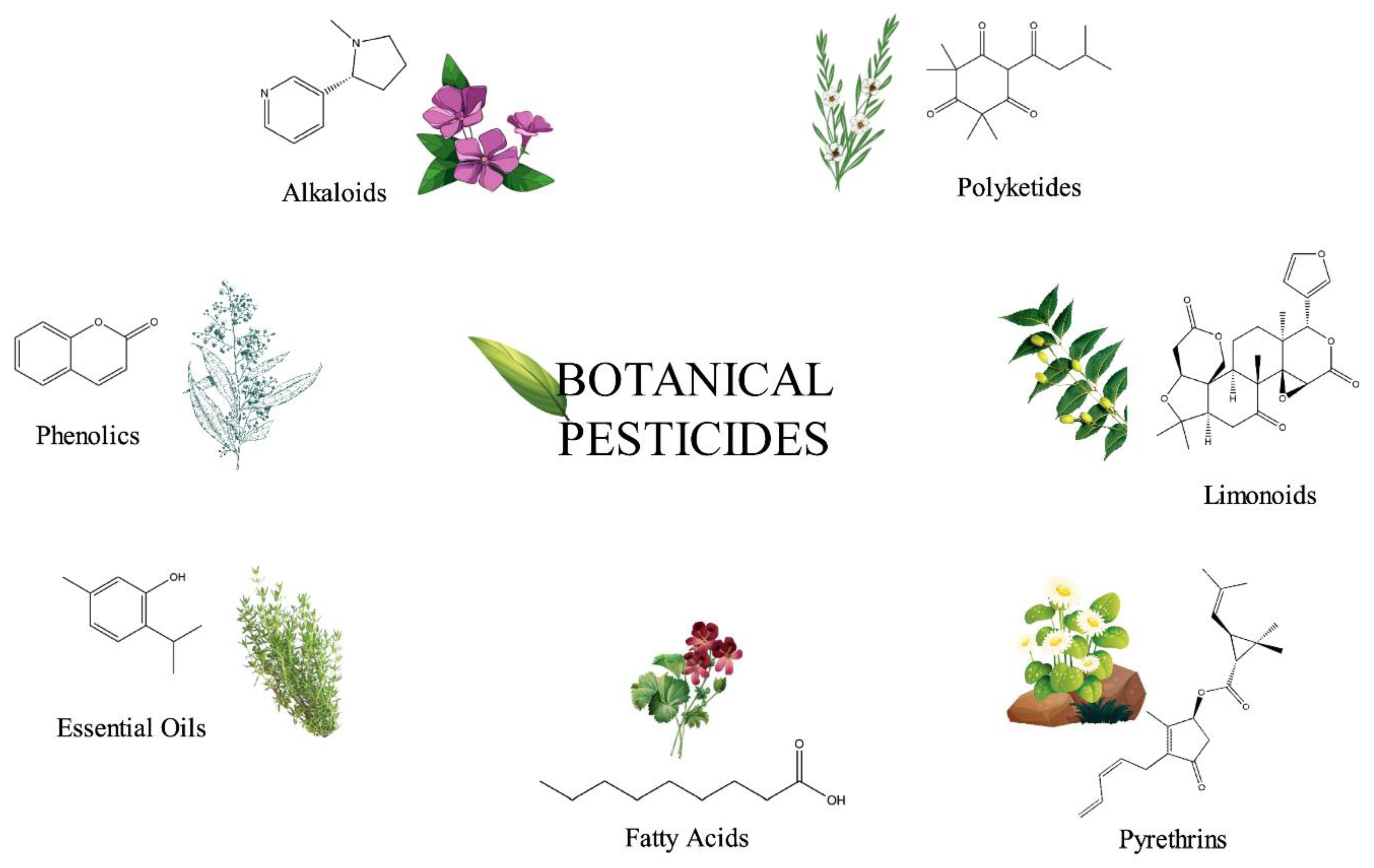
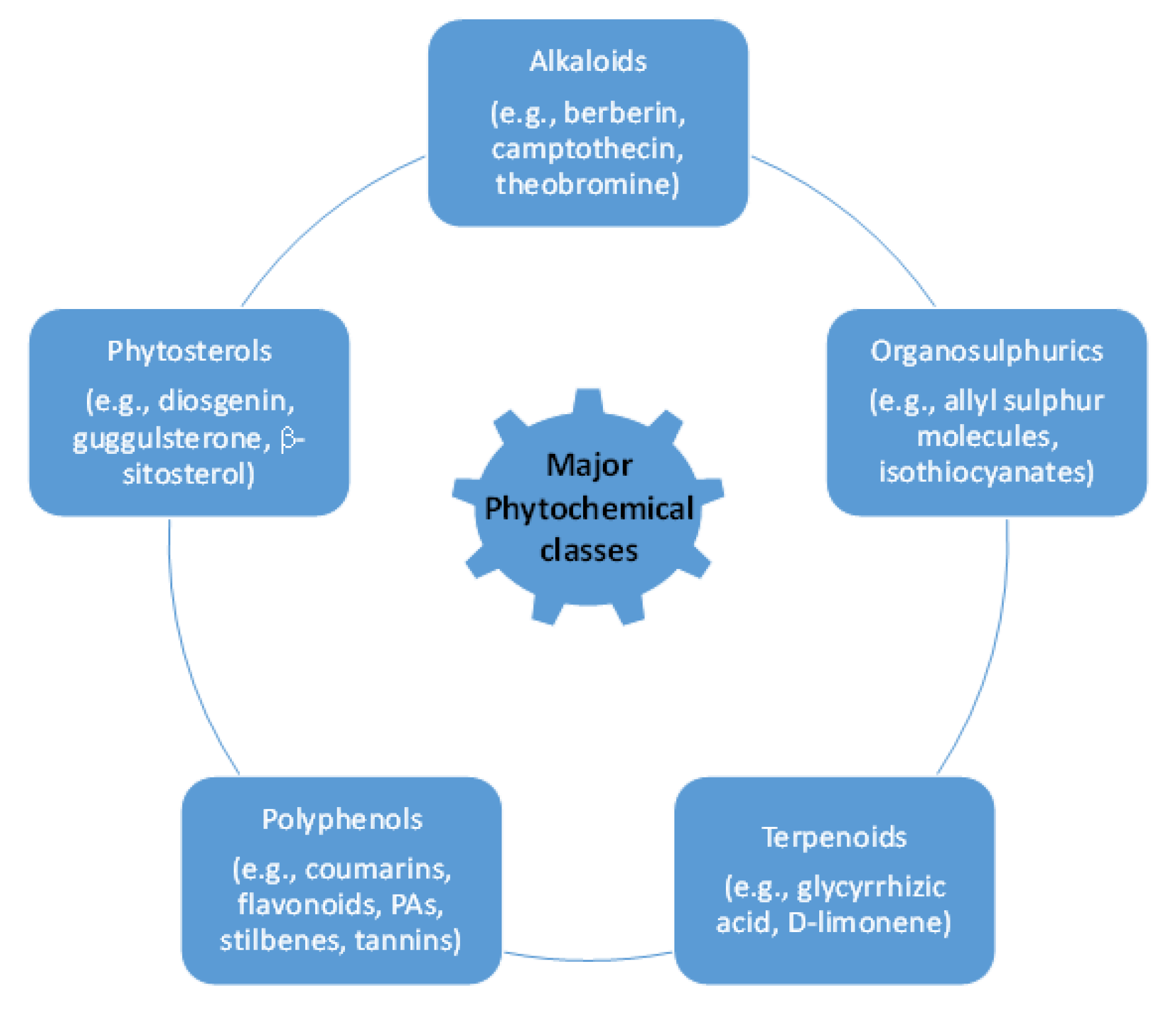
2. Bioactive Principles in Botanical Pesticides
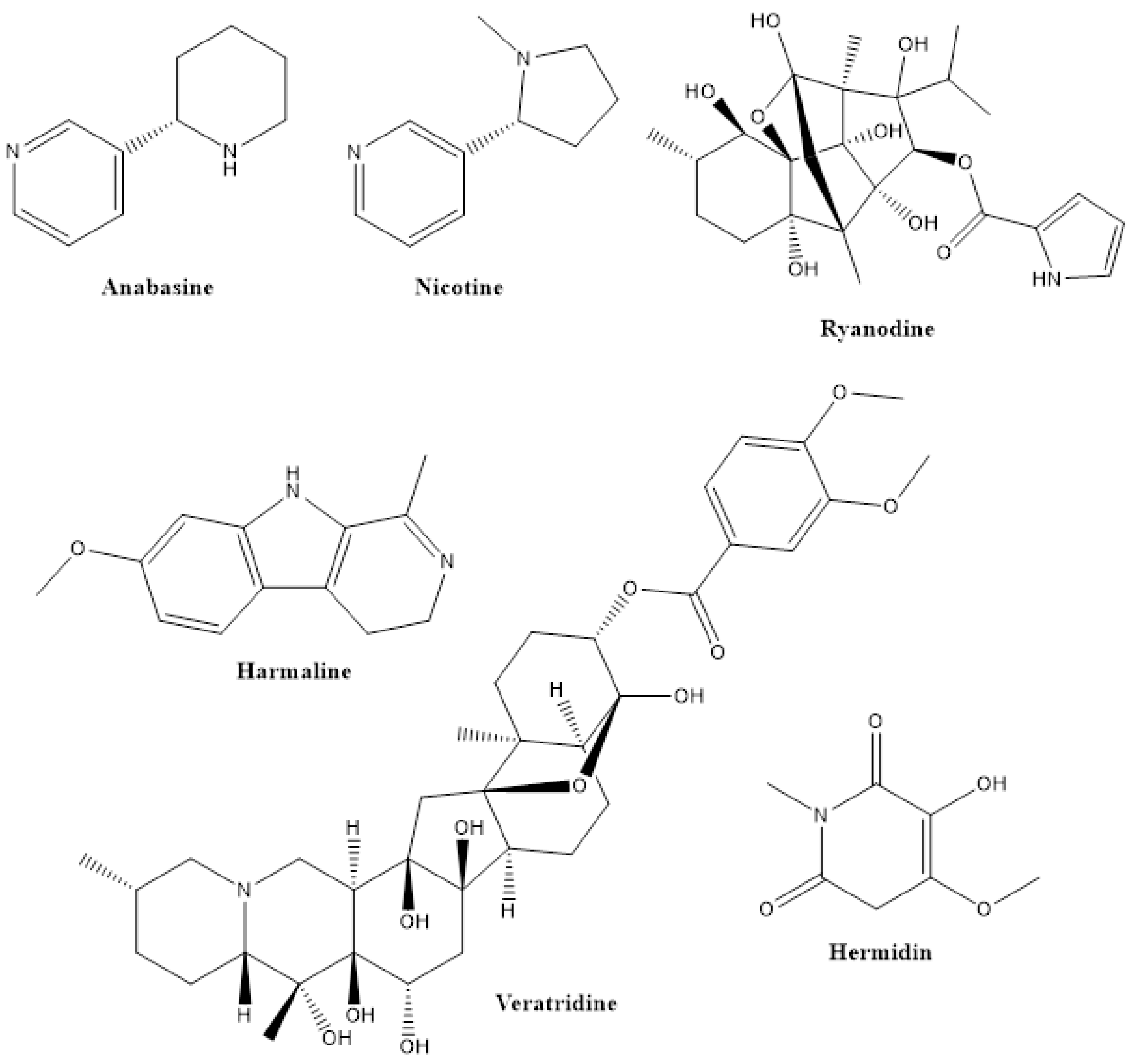
2.1. Alkaloids
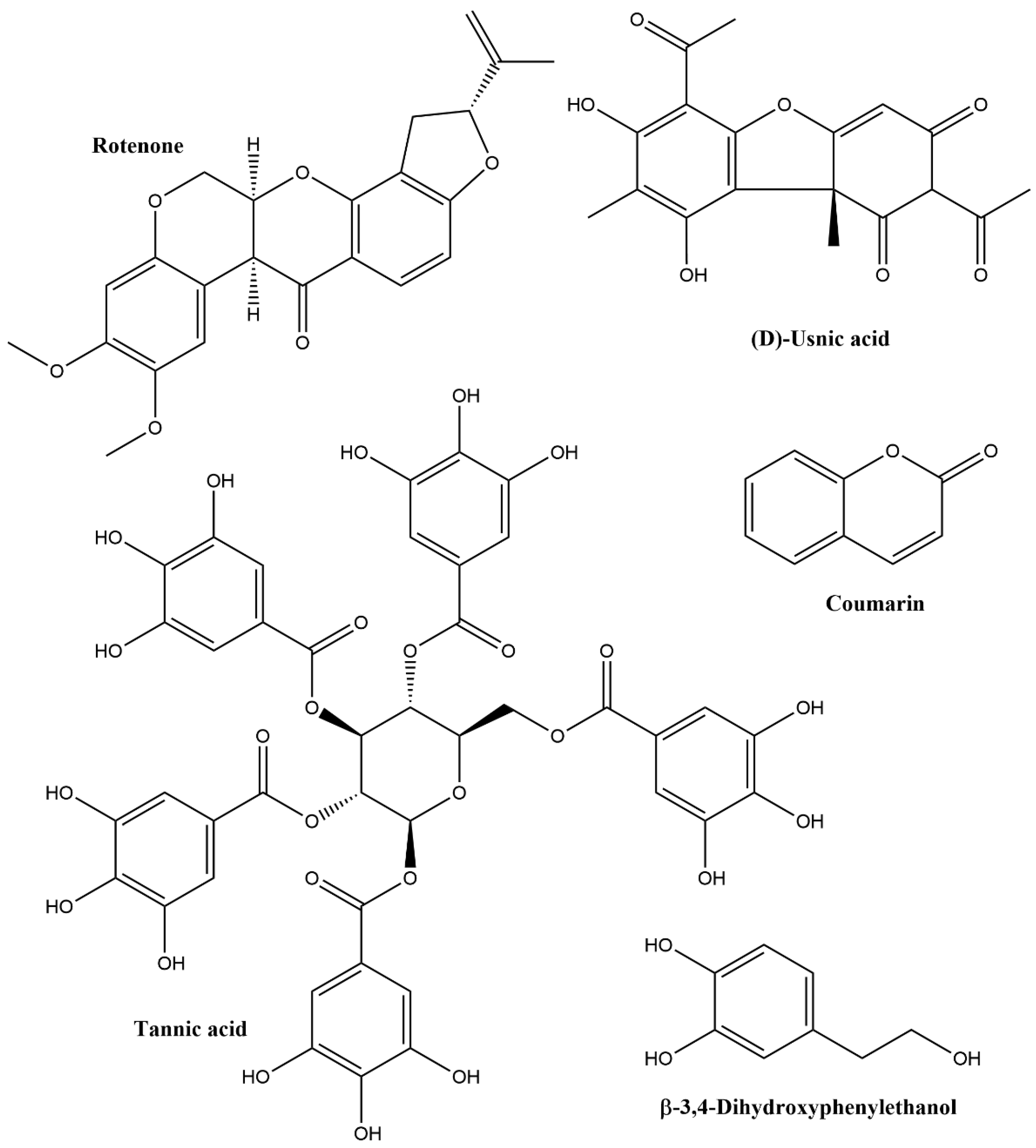
2.2. Phenolics
2.3. Essential Oils
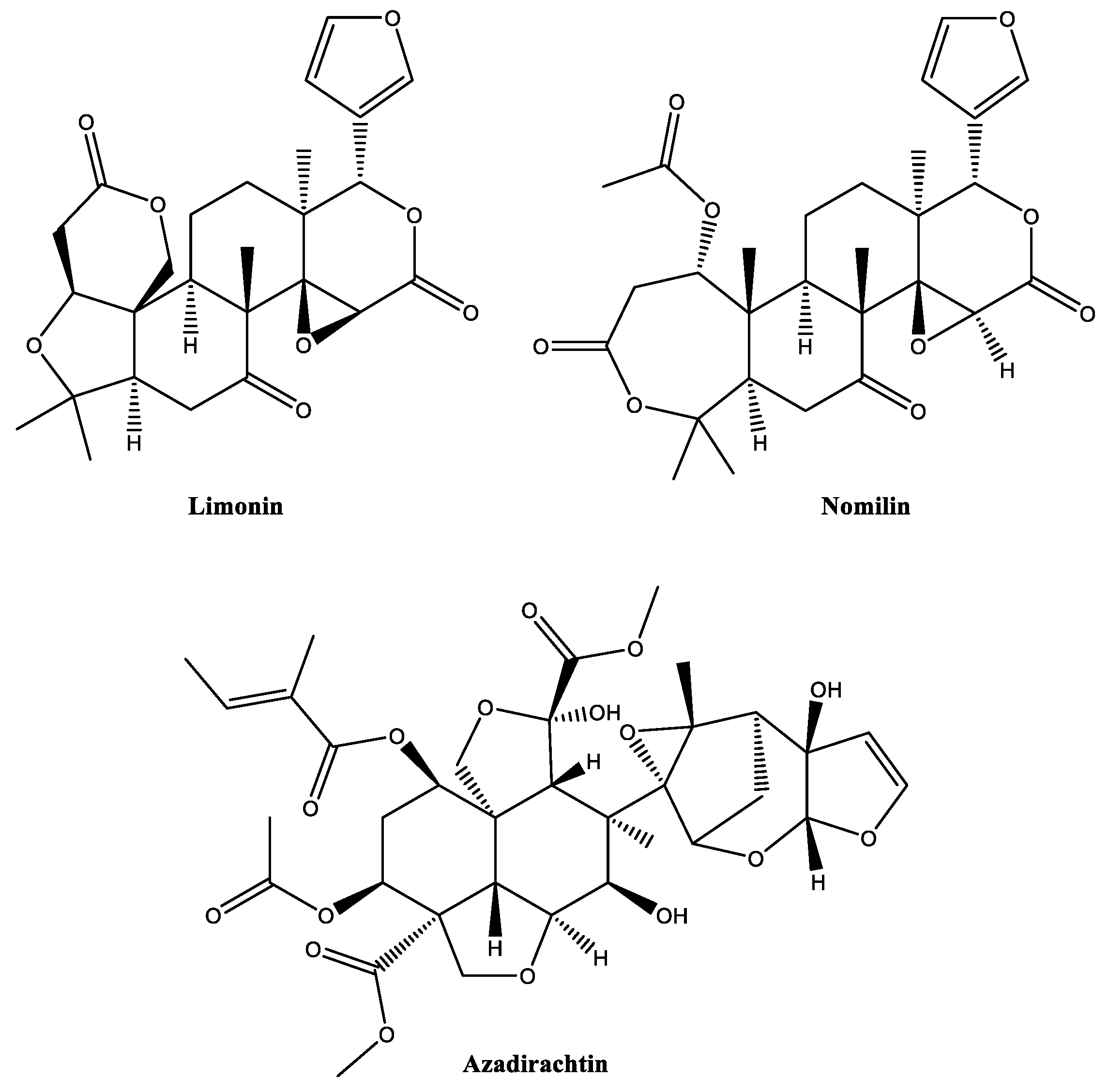
2.4. Limonoids
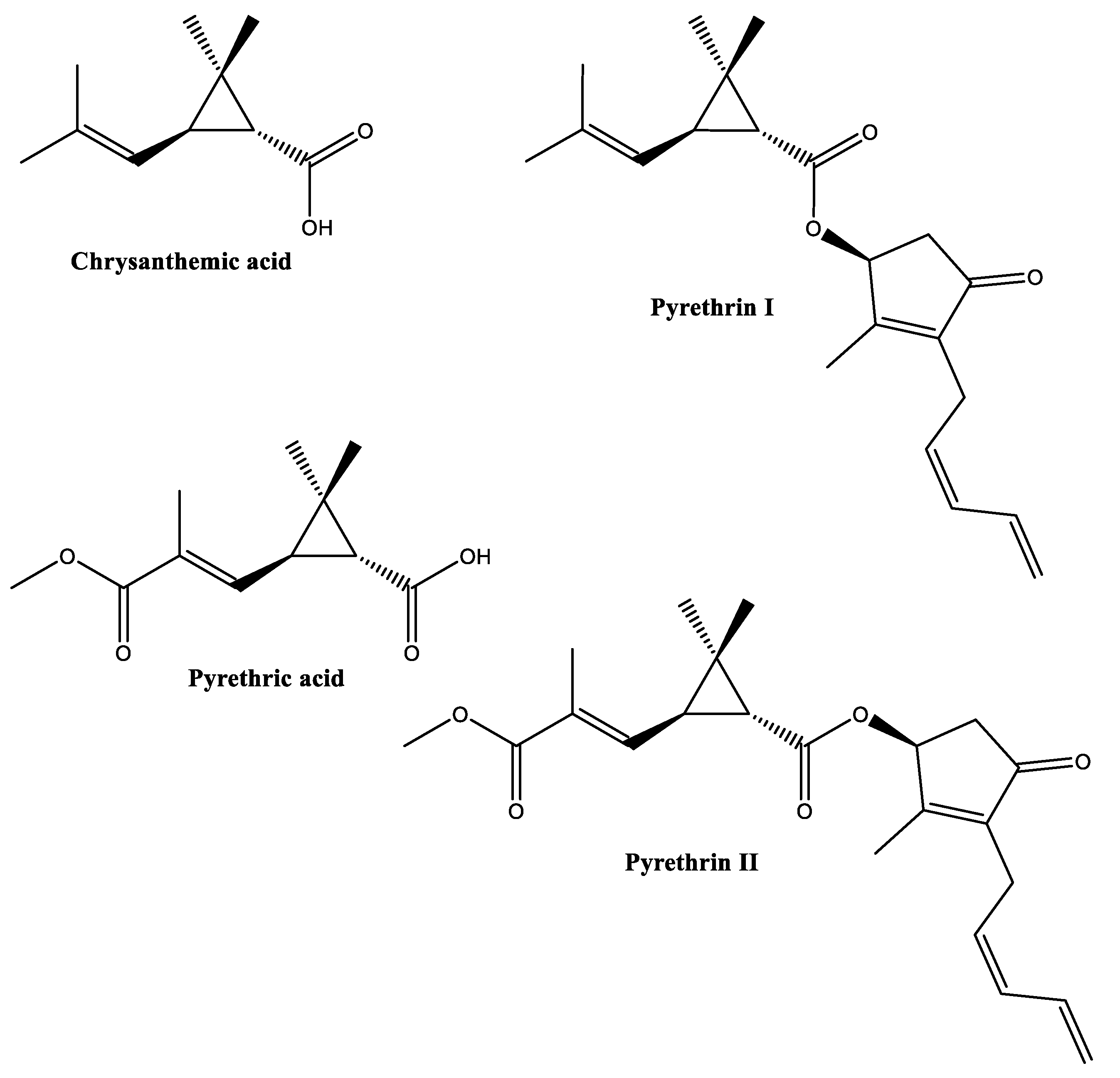
2.5. Pyrethrins
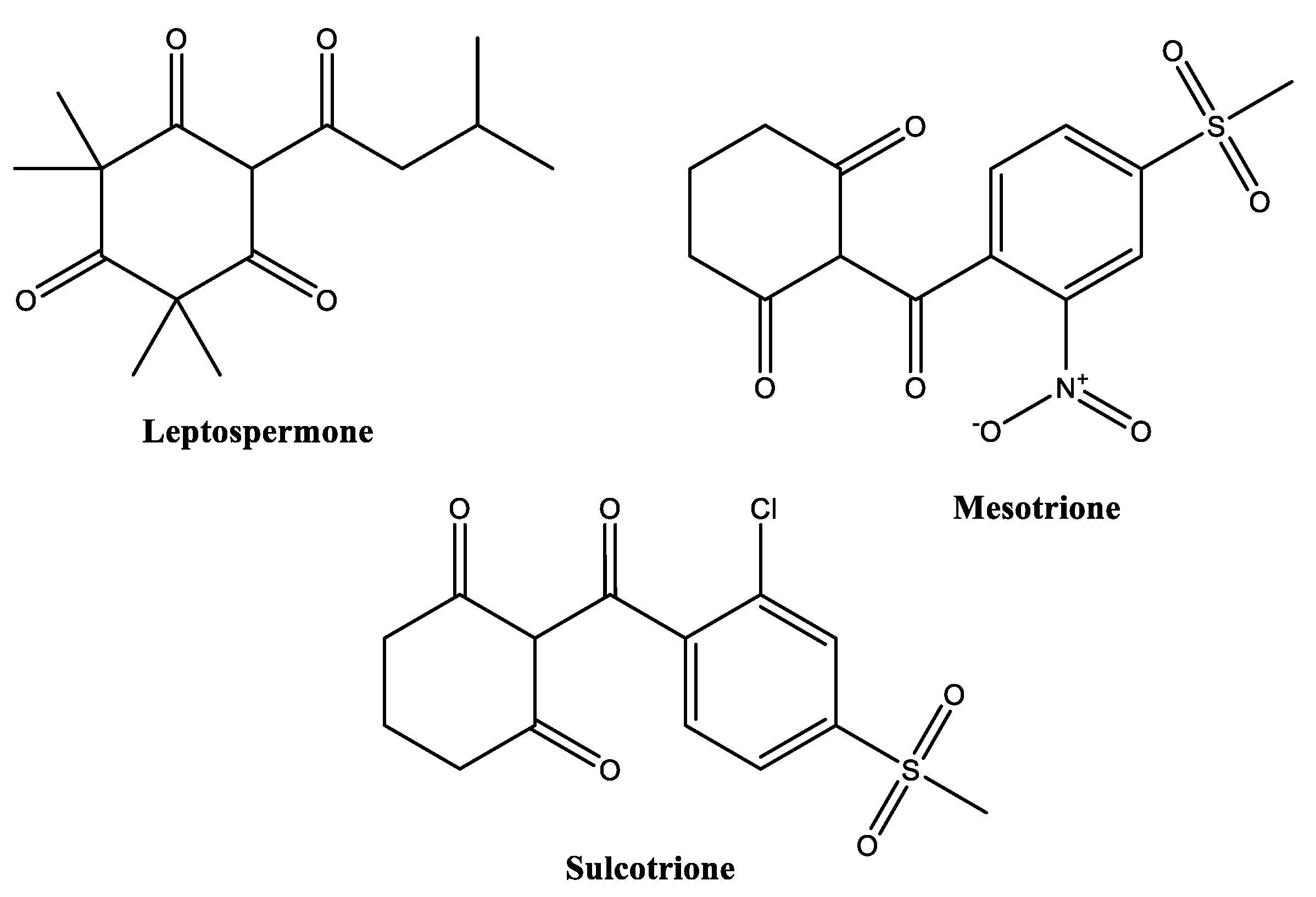
2.6. Polyketides
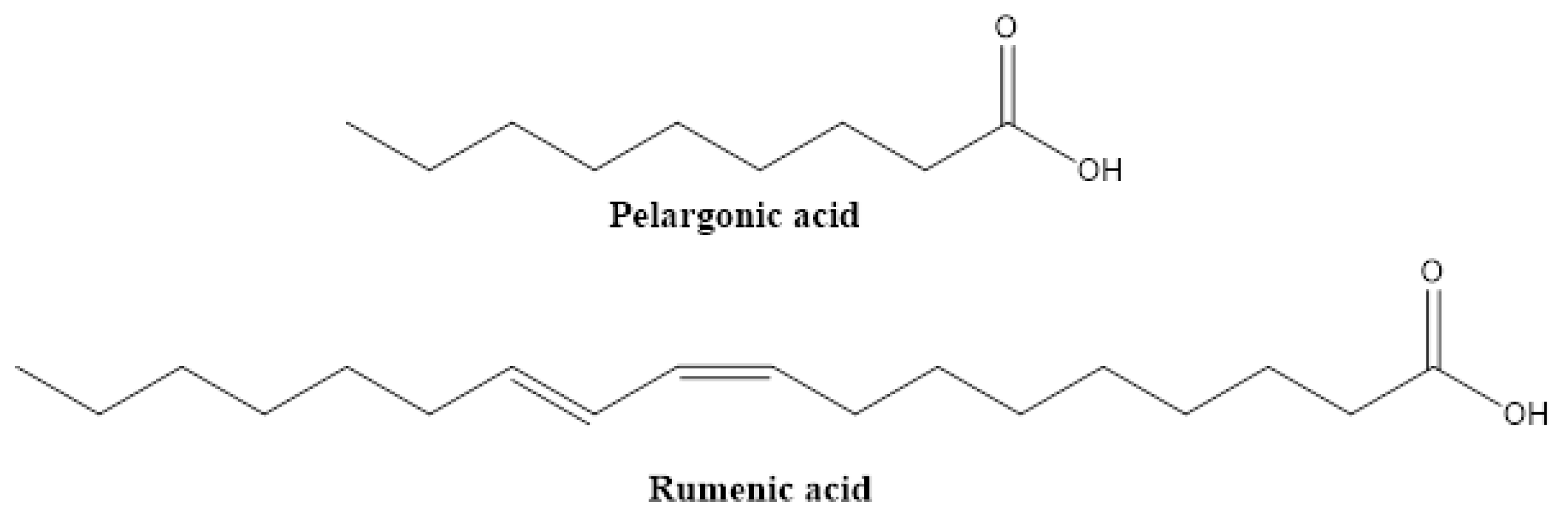
2.7. Fatty Acids
3. Botanical Insecticides
3.1. State of the Art
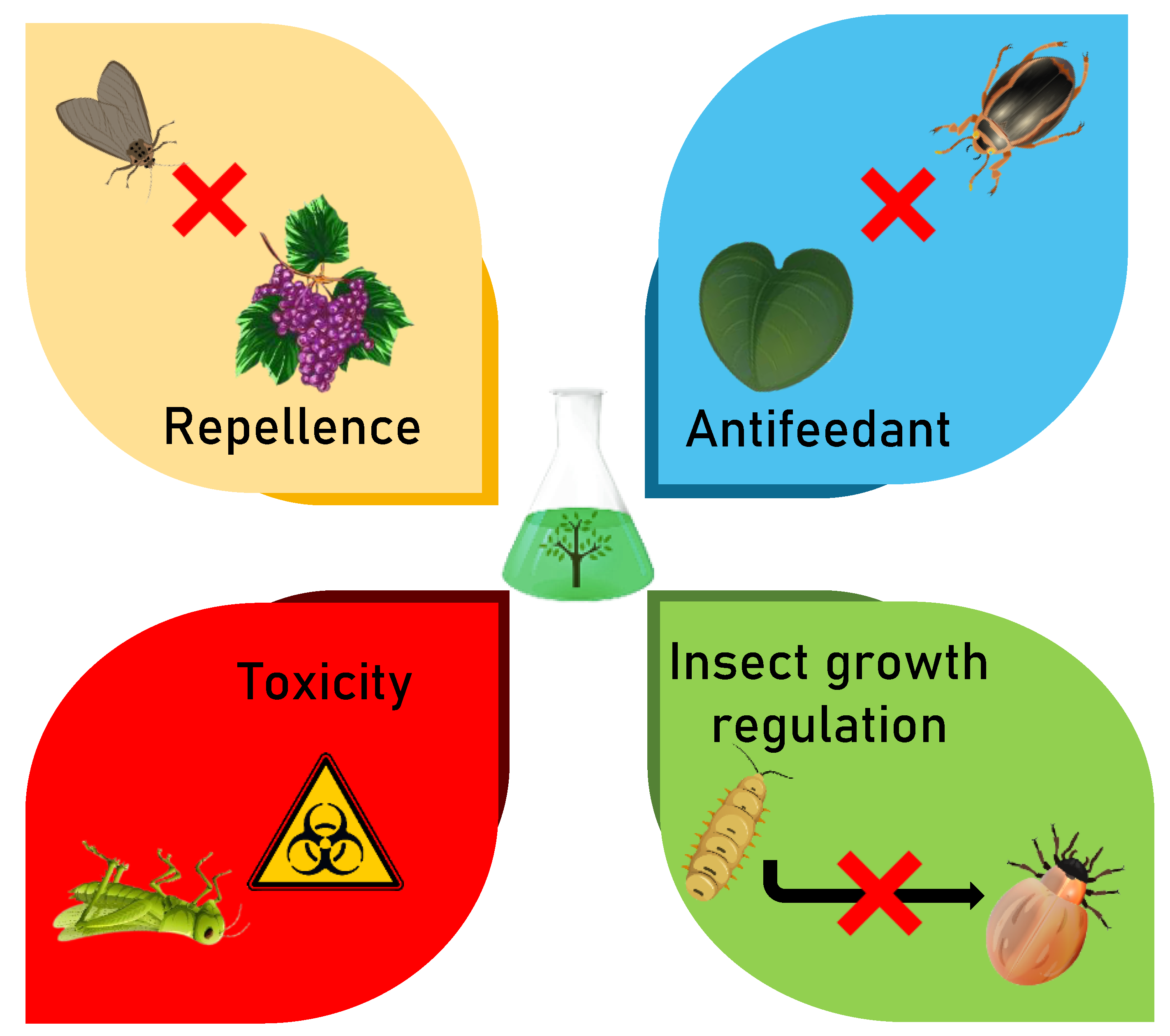
3.2. Modes of Action of Botanical Insecticides
3.2.1. Repellent Effects
3.2.2. Antifeedant Effects
3.2.3. Toxic Effects
3.2.4. Growth Regulation Effects: Larval Growth and Adult Reproduction
3.2.5. Metabolic Effects
4. Botanical Herbicides
4.1. State of the Art
4.2. Modes of Action of Botanical Herbicides
5. Botanical Fungicides
5.1. State of the Art
5.2. Modes of Action of Botanical Fungicides
6. Regulation of Biopesticides in the European Union
7. Current Challenges in the Development and Registration of Biopesticides
- Biopesticides and related products should be evaluated in a more biological and ecological context.
- A simplification of authorization should be enacted to enhance the further penetration of biopesticides into plant protection markets, shifting agriculture to more sustainable integrated pest management systems.
- There is a need for unification of legislation for low-risk biologically based plant protection products, separating their evaluation from conventional chemical pesticides, with a focus on food safety, human health and protection of the environment.
- Financial and in-depth scientific support through research programmes should be provided to facilitate SMEs to develop more biological control products and key pesticides producers to switch to more sustainable products.
- Support of networking approaches and effective links within farmers, SMEs and the industry will further stimulate the biopesticides market.
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Maggi, F.; la Cecilia, D.; Tang, F.H.M.; McBratney, A. The global environmental hazard of glyphosate use. Sci. Total Environ. 2020, 717, 137167. [Google Scholar] [CrossRef]
- Schrödl, W.; Krüger, S.; Konstantinova-Müller, T.; Shehata, A.A.; Rulff, R.; Krüger, M. Possible effects of glyphosate on Mucorales abundance in the rumen of dairy cows in Germany. Curr. Microbiol. 2014, 69, 817–823. [Google Scholar] [CrossRef]
- Krüger, M.; Basiouni, S.; Ederk, I.; Rodloff, A. Susceptibility of extended-spectrum ß-lactamase (ESBL)-producing Enterobacteriaaceae to Roundup. Ger. J. Microbiol. 2021, 1, 7–15. [Google Scholar] [CrossRef]
- Suratman, S.; Edwards, J.W.; Babina, K. Organophosphate pesticides exposure among farmworkers: Pathways and risk of adverse health effects. Rev. Environ. Health 2015, 30, 65–79. [Google Scholar] [CrossRef]
- Sola, P.; Mvumi, B.M.; Ogendo, J.O.; Mponda, O.; Kamanula, J.F.; Nyirenda, S.P.; Belmain, S.R.; Stevenson, P.C. Botanical pesticide production, trade and regulatory mechanisms in sub-Saharan Africa: Making a case for plant-based pesticidal products. Food Secur. 2014, 6, 369–384. [Google Scholar] [CrossRef]
- Isman, M.B. Bridging the gap: Moving botanical insecticides from the laboratory to the farm. Ind. Crops Prod. 2017, 110, 10–14. [Google Scholar] [CrossRef]
- Klein, R.A.; Dunkel, F.V. New pest management frontiers: Linking plant medicine to traditional knowledge. Am. Entomol. 2003, 49, 7–16. [Google Scholar] [CrossRef][Green Version]
- Isman, M.B. Botanical insecticides, deterrents, and repellents in modern agriculture and an increasingly regulated world. Annu. Rev. Entomol. 2006, 51, 45–66. [Google Scholar] [CrossRef]
- Belmain, S.; Stevenson, P. Ethnobotanicals in Ghana: Reviving and modernising age-old farmer practice. Pestic. Outlook 2001, 12, 233–238. [Google Scholar]
- Boeke, S.; Baumgart, I.; Van Loon, J.; Van Huis, A.; Dicke, M.; Kossou, D. Toxicity and repellence of African plants traditionally used for the protection of stored cowpea against Callosobruchus maculatus. J. Stored Prod. Res. 2004, 40, 423–438. [Google Scholar] [CrossRef]
- Wafukho, S.S.; Watiti, J.; Nang’ayo, F.; Masso, C.; Tarus, D. The building blocks for biofertiliser and biopesticides policymaking in Africa. Policy Brief 2013, 2. [Google Scholar]
- Regnault-Roger, C.; Philogène, B.J. Past and current prospects for the use of botanicals and plant allelochemicals in integrated pest management. Pharm. Biol. 2008, 46, 41–52. [Google Scholar] [CrossRef]
- Kaushik, B.D.; Kumar, D.; Shamim, M. Biofertilizers and Biopesticides in Sustainable Agriculture; Apple Academic Press and CRC Press: Boca Raton, FL, USA, 2019. [Google Scholar]
- Yadav, R.S.; Panwar, J.; Tarafdar, J.C.; Yadav, B.K.; Dave, S. Role of arbuscular mycorrhiza in dryland agriculture. Org. Agric. 2012, 119. [Google Scholar]
- Hammad, A.M.A.; Bashir, H.A.A.A.; Abdelbagi, A.O.; Ishag, A.E.S.A.; Ali, M.M.Y.; Bashir, M.O.; Hur, J.-H.; Laing, M.D. Efficacy of indigenous entomopathogenic fungi for the control of the tomato leafminer Tuta absoluta (Meyrick) in Sudan. Int. J. Trop. Insect Sci. 2021, 2021, 1–11. [Google Scholar] [CrossRef]
- Guijarro, B.; Larena, I.; Casals, C.; Teixidó, N.; Melgarejo, P.; De Cal, A. Compatibility interactions between the biocontrol agent Penicillium frequentans Pf909 and other existing strategies to brown rot control. Biol. Control 2019, 129, 45–54. [Google Scholar] [CrossRef]
- Lengai, G.M.; Muthomi, J.W.; Mbega, E.R. Phytochemical activity and role of botanical pesticides in pest management for sustainable agricultural crop production. Sci. Afr. 2020, 7, e00239. [Google Scholar] [CrossRef]
- Ahmad, W.; Singh, S.; Kumar, S. Phytochemical screening and antimicrobial study of Euphorbia hirta extracts. J. Med. Plants Stud. 2017, 5, 183–186. [Google Scholar]
- Banthorpe, D.V. Classification of terpenoids and general procedures for their characterization. In Methods in Plant Biochemistry; Charlwood, B.V., Banthorpe, D.V., Eds.; Academic Press: London, UK, 1991; Volume 7, pp. 1–41. [Google Scholar]
- Reverchon, E.; Senatore, F. Isolation of rosemary oil: Comparison between hydrodistillation and supercritical CO2 extraction. Flavour Fragr. J. 1992, 7, 227–230. [Google Scholar] [CrossRef]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—a review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef]
- Ippolito, A.; Nigro, F. Natural antimicrobials in postharvest storage of fresh fruits and vegetables. In Natural Antimicrobials for the Minimal Processing of Foods; Roller, S., Ed.; Woodhead Publishing: Cambridge, UK, 2003; pp. 176–200. [Google Scholar]
- Min, W.; Yi-Min, W.; Jin-Ming, G. Analysis of fatty acid and unsaponifiable matter from tartary buckwheat oil and buckwheat oil by GC/MS. In Advances in Buckwheat Research, Proceedings of the 9th International Symposium on Buckwheat, Prague, Czech Republic, 18–22 August 2004; Faberová, I., Dvořáček, V., Čepková, P., Hon, I., Holubec, V., Stehno, Z., Eds.; Research Institute of Crop Production Prague—Ruzynĕ: Prague—Suchdol, Czech Republic, 2004; pp. 723–729. [Google Scholar]
- Bayir, A.G.; Kiziltan, H.S.; Kocyigit, A. Plant family, carvacrol, and putative protection in gastric cancer. In Dietary Interventions in Gastrointestinal Diseases: Foods, Nutrients, and Dietary Supplements; Watson, R.R., Preedy, V.R., Eds.; Academic Press: London, UK, 2019; pp. 3–18. [Google Scholar]
- Schläger, S.; Dräger, B. Exploiting plant alkaloids. Curr. Opin. Biotechnol. 2016, 37, 155–164. [Google Scholar] [CrossRef]
- Bruneton, J. Pharmacognosie, Phytochimie, Plantes Médicinales, 4th ed.; Tec & Dac-Editions Médicinales Internationales: Paris, France, 1999. [Google Scholar]
- Taiz, L.; Zeiger, E. Plant Physiology, 4th ed.; Sinauer Associates, Inc.: Sunderland, MA, USA, 2006. [Google Scholar]
- Rattan, R.S. Mechanism of action of insecticidal secondary metabolites of plant origin. Crop Prot. 2010, 29, 913–920. [Google Scholar] [CrossRef]
- Ávila-Román, J.; Soliz-Rueda, J.R.; Bravo, F.I.; Aragonès, G.; Suárez, M.; Arola-Arnal, A.; Mulero, M.; Salvadó, M.-J.; Arola, L.; Torres-Fuentes, C.; et al. Phenolic compounds and biological rhythms: Who takes the lead? Trends Food Sci. Technol. 2021, 113, 77–85. [Google Scholar] [CrossRef]
- Manach, C.; Scalbert, A.; Morand, C.; Rémésy, C.; Jiménez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [CrossRef] [PubMed]
- Cutrim, C.S.; Cortez, M.A.S. A review on polyphenols: Classification, beneficial effects and their application in dairy products. Int. J. Dairy Technol. 2018, 71, 564–578. [Google Scholar] [CrossRef]
- Sharma, A.; Shahzad, B.; Rehman, A.; Bhardwaj, R.; Landi, M.; Zheng, B. Response of phenylpropanoid pathway and the role of polyphenols in plants under abiotic stress. Molecules 2019, 24, 2452. [Google Scholar] [CrossRef]
- Furiga, A.; Lonvaud-Funel, A.; Badet, C. In vitro study of antioxidant capacity and antibacterial activity on oral anaerobes of a grape seed extract. Food Chem. 2009, 113, 1037–1040. [Google Scholar] [CrossRef]
- Regnault-Roger, C.; Vincent, C.; Arnason, J.T. Essential oils in insect control: Low-risk products in a high-stakes world. Annu. Rev. Entomol. 2012, 57, 405–424. [Google Scholar] [CrossRef]
- Ikbal, C.; Pavela, R. Essential oils as active ingredients of botanical insecticides against aphids. J. Pest Sci. 2019, 92, 971–986. [Google Scholar] [CrossRef]
- Isman, M.B. Botanical insecticides in the twenty-first century—Fulfilling their promise? Annu. Rev. Entomol. 2020, 65, 233–249. [Google Scholar] [CrossRef]
- Isman, M.B.; Miresmailli, S.; Machial, C. Commercial opportunities for pesticides based on plant essential oils in agriculture, industry and consumer products. Phytochem. Rev. 2011, 10, 197–204. [Google Scholar] [CrossRef]
- Mitchell, M.J.; Smith, S.L.; Johnson, S.; Morgan, E.D. Effects of the neem tree compounds azadirachtin, salannin, nimbin, and 6-desacetylnimbin on ecdysone 20-monooxygenase activity. Arch. Insect Biochem. Physiol. 1997, 35, 199–209. [Google Scholar] [CrossRef]
- Saleem, S.; Muhammad, G.; Hussain, M.A.; Bukhari, S.N.A. A comprehensive review of phytochemical profile, bioactives for pharmaceuticals, and pharmacological attributes of Azadirachta indica. Phytother. Res. 2018, 32, 1241–1272. [Google Scholar] [CrossRef]
- Mordue, A.J. Present concepts of the mode of action of Azadirachtin from Neem. In Neem: Today and in the New Millennium; Koul, O., Wahab, S., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2004; pp. 229–242. [Google Scholar]
- Jeran, N.; Grdiša, M.; Varga, F.; Šatović, Z.; Liber, Z.; Dabić, D.; Biošić, M. Pyrethrin from Dalmatian pyrethrum (Tanacetum cinerariifolium (Trevir.) Sch. Bip. Biosynthesis, biological activity, methods of extraction and determination. Phytochem. Rev. 2021, 20, 875–905. [Google Scholar] [CrossRef]
- Casida, J.E. Pyrethrum flowers and pyrethroid insecticides. Environ. Health Perspect. 1980, 34, 189–202. [Google Scholar] [CrossRef]
- Grdiša, M.; Babić, S.; Periša, M.; Carović-Stanko, K.; Kolak, I.; Liber, Z.; Jug-Dujaković, M.; Satovic, Z. Chemical diversity of the natural populations of Dalmatian Pyrethrum (Tanacetum cinerariifolium (TREVIR.) SCH. BIP.) in Croatia. Chem. Biodivers. 2013, 10, 460–472. [Google Scholar] [CrossRef]
- Ambrožič-Dolinšek, J.; Kovač, M.; Žel, J.; Camloh, M. Pyrethrum (Tanacetum cinerariifolium) from the northern Adriatic as a potential source of natural insecticide. Ann. Ser. Hist. Nat. 2007, 17, 39–46. [Google Scholar]
- Clements, J.; Groves, R.L.; Cava, J.; Barry, C.C.; Chapman, S.; Olson, J.M. Conjugated linoleic acid as a novel insecticide targeting the agricultural pest Leptinotarsa decemlineata. PLoS ONE 2019, 14, e0220830. [Google Scholar] [CrossRef]
- Coleman, R.; Penner, D. Organic acid enhancement of pelargonic acid. Weed Technol. 2008, 22, 38–41. [Google Scholar] [CrossRef]
- Dayan, F.E.; Owens, D.K.; Watson, S.B.; Asolkar, R.N.; Boddy, L.G. Sarmentine, a natural herbicide from piper species with multiple herbicide mechanisms of action. Front. Plant Sci. 2015, 6, 222. [Google Scholar] [CrossRef]
- Muñoz, M.; Torres-Pagán, N.; Peiró, R.; Guijarro, R.; Sánchez-Moreiras, A.M.; Verdeguer, M. Phytotoxic effects of three natural compounds: Pelargonic acid, carvacrol, and cinnamic aldehyde, against problematic weeds in Mediterranean crops. Agronomy 2020, 10, 791. [Google Scholar] [CrossRef]
- Webber, C.L.; Shrefler, J.W. (220) Pelargonic acid weed control parameters. Hortscience 2006, 41, 1034. [Google Scholar] [CrossRef]
- Hikal, W.M.; Baeshen, R.S.; Said-Al Ahl, H.A.H. Botanical insecticide as simple extractives for pest control. Cogent Biol. 2017, 3, 1404274. [Google Scholar] [CrossRef]
- Nagegowda, D.A. Plant volatile terpenoid metabolism: Biosynthetic genes, transcriptional regulation and subcellular compartmentation. FEBS Lett. 2010, 584, 2965–2973. [Google Scholar] [CrossRef]
- Pavela, R. Antifeedant and larvicidal effects of some phenolic components of essential oils lasp lines of introduction against Spodoptera littoralis (Boisd.). J. Essent. Oil-Bear. Plants 2011, 14, 266–273. [Google Scholar] [CrossRef]
- Babaousmail, M.; Isman, M. Research on botanical insecticides in North Africa. Biopestic. Int. 2016, 12, 83–92. [Google Scholar]
- Stevenson, P.C.; Isman, M.B.; Belmain, S.R. Pesticidal plants in Africa: A global vision of new biological control products from local uses. Ind. Crops Prod. 2017, 110, 2–9. [Google Scholar] [CrossRef]
- Mulugeta, T.; Muhinyuza, J.B.; Gouws-Meyer, R.; Matsaunyane, L.; Andreasson, E.; Alexandersson, E. Botanicals and plant strengtheners for potato and tomato cultivation in Africa. J. Integr. Agric. 2020, 19, 406–427. [Google Scholar] [CrossRef]
- Isman, M.B.; Grieneisen, M.L. Botanical insecticide research: Many publications, limited useful data. Trends Plant Sci. 2014, 19, 140–145. [Google Scholar] [CrossRef]
- Velasques, J.; Cardoso, M.H.; Abrantes, G.; Frihling, B.E.; Franco, O.L.; Migliolo, L. The rescue of botanical insecticides: A bioinspiration for new niches and needs. Pestic. Biochem. Physiol. 2017, 143, 14–25. [Google Scholar] [CrossRef]
- Prakash, B.; Kumar, A.; Singh, P.P.; Das, S.; Dubey, N.K. Chapter 16—Prospects of plant products in the management of insect pests of food grains: Current status and future perspectives. In Natural Bioactive Compounds; Sinha, R.P., Häder, D.-P., Eds.; Academic Press: New York, NY, USA, 2021; pp. 317–335. [Google Scholar]
- Dougoud, J.; Toepfer, S.; Bateman, M.; Jenner, W.H. Efficacy of homemade botanical insecticides based on traditional knowledge. A review. Agron. Sustain. Dev. 2019, 39, 37. [Google Scholar] [CrossRef]
- Patil, R.S.; Nandihalli, B.S. Efficacy of promising botanicals a red spider mite on brinjal. Karnataka J. Agric. Sci. 2009, 22, 690–692. [Google Scholar]
- Degri, M.M.; Sharah, H.S. Field evaluation of two aqueous plant extracts on water melon Citrullus lanatus (Thumb) insect pets in northern Guinea Savannah of Nigeria. Int. Lett. Nat. Sci. 2014, 9, 59–67. [Google Scholar] [CrossRef]
- Koul, O. Antifeedant phytochemicals in insect management (so close yet so far). In Ecofriendly Pest Management for Food Security; Omkar, O., Ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2016; pp. 525–544. [Google Scholar]
- Pavela, R. History, presence and perspective of using plant extracts as commercial botanical insecticides and farm products for protection against insects—A review. Plant Prot. Sci. 2016, 52, 229–241. [Google Scholar]
- Cao, J.Q.; Guo, S.S.; Wang, Y.; Pang, X.; Geng, Z.F.; Du, S.S. Toxicity and repellency of essential oil from Evodia lenticellata Huang fruits and its major monoterpenes against three stored-product insects. Ecotoxicol. Environ. Saf. 2018, 160, 342–348. [Google Scholar] [CrossRef]
- Hernández-Lambraño, R.; Caballero-Gallardo, K.; Olivero-Verbel, J. Toxicity and antifeedant activity of essential oils from three aromatic plants grown in Colombia against Euprosterna elaeasa and Acharia fusca (Lepidoptera: Limacodidae). Asian Pac. J. Trop. Biomed. 2014, 4, 695–700. [Google Scholar] [CrossRef]
- Khan, H.A.A. Toxicity, repellent and oviposition deterrent effects of select essential oils against the house fly Musca domestica. J. Asia-Pac. Entomol. 2021, 24, 15–20. [Google Scholar] [CrossRef]
- Peixoto, M.G.; Bacci, L.; Fitzgerald Blank, A.; Araújo, A.P.A.; Alves, P.B.; Silva, J.H.S.; Santos, A.A.; Oliveira, A.P.; da Costa, A.S.; Arrigoni-Blank, M.d.F. Toxicity and repellency of essential oils of Lippia alba chemotypes and their major monoterpenes against stored grain insects. Ind. Crops Prod. 2015, 71, 31–36. [Google Scholar] [CrossRef]
- Soonwera, M.; Wongnet, O.; Sittichok, S. Ovicidal effect of essential oils from Zingiberaceae plants and Eucalyptus globulus on eggs of head lice, Pediculus humanus capitis De Geer. Phytomedicine 2018, 47, 93–104. [Google Scholar] [CrossRef]
- Tandon, S.; Ak, M. Insecticidal and growth inhibitory activity of essential oils of Boenninghausenia albiflora and Teucrium quadrifarium against Spilarctia obliqua. Biochem. Syst. Ecol. 2018, 81, 70–73. [Google Scholar] [CrossRef]
- Priestley, C.M.; Williamson, E.M.; Wafford, K.A.; Sattelle, D.B. Thymol, a constituent of thyme essential oil, is a positive allosteric modulator of human GABA(A) receptors and a homo-oligomeric GABA receptor from Drosophila melanogaster. Br. J. Pharmacol. 2003, 140, 1363–1372. [Google Scholar] [CrossRef]
- Bentley, M.D.; Leonard, D.E.; Reynolds, E.K.; Leach, S.; Beck, A.B.; Murakoshi, I. Lupine alkaloids as larval feeding deterrents for spruce budworm, Choristoneura fumiferana (Lepidoptera: Tortricidae). Ann. Entomol. Soc. Am. 1984, 77, 398–400. [Google Scholar] [CrossRef]
- Brem, B.; Seger, C.; Pacher, T.; Hofer, O.; Vajrodaya, S.; Greger, H. Feeding deterrence and contact toxicity of Stemona alkaloids-a source of potent natural insecticides. J. Agric. Food Chem. 2002, 50, 6383–6388. [Google Scholar] [CrossRef] [PubMed]
- Rharrabe, K.; Jbilou, R.; Bouayad, N.; Ajaha, A.; Aarab, A. Harmaline ingestion effect on development, metabolites and midgut of the red flour beetle, Tribolium castaneum. J. Asia-Pac. Entomol. 2020, 23, 29–35. [Google Scholar] [CrossRef]
- Dhaliwal, G.; Arora, R. Role of phytochemicals in integrated pest management. In Phytochemical Biopesticides; Koul, O., Dhaliwa, G.S., Eds.; Harwood Academic Publishers: Amsterdam, The Netherlands, 2001; pp. 97–117. [Google Scholar]
- Silver, K.S.; Du, Y.; Nomura, Y.; Oliveira, E.E.; Salgado, V.L.; Zhorov, B.S.; Dong, K. Voltage-gated sodium channels as insecticide targets. Adv. Insect Physiol. 2014, 46, 389–433. [Google Scholar]
- Tandon, S.; Mittal, A.K.; Pant, A.K. Insect growth regulatory activity of Vitex trifolia and Vitex agnus-castus essential oils against Spilosoma obliqua. Fitoterapia 2008, 79, 283–286. [Google Scholar] [CrossRef]
- Abdelkhalek, A.; Salem, M.Z.M.; Kordy, A.M.; Salem, A.Z.M.; Behiry, S.I. Antiviral, antifungal, and insecticidal activities of Eucalyptus bark extract: HPLC analysis of polyphenolic compounds. Microb. Pathog. 2020, 147, 104383. [Google Scholar] [CrossRef]
- Geng, Z.F.; Liu, Z.L.; Wang, C.F.; Liu, Q.Z.; Shen, S.M.; Liu, Z.M.; Du, S.S.; Deng, Z.W. Feeding deterrents against two grain storage insects from Euphorbia fischeriana. Molecules 2011, 16, 466–476. [Google Scholar] [CrossRef]
- Hashim, M.S.; Devi, K.S. Insecticidal action of the polyphenolic rich fractions from the stem bark of Streblus asper on Dysdercus cingulatus. Fitoterapia 2003, 74, 670–676. [Google Scholar] [CrossRef]
- Regnault-Roger, C.; Ribodeau, M.; Hamraoui, A.; Bareau, I.; Blanchard, P.; Gil-Munoz, M.-I.; Barberan, F.T. Polyphenolic compounds of Mediterranean Lamiaceae and investigation of orientational effects on Acanthoscelides obtectus (Say). J. Stored Prod. Res. 2004, 40, 395–408. [Google Scholar] [CrossRef]
- Regnault-Roger, C.; Philogène, B.J.; Vincent, C. Biopesticides d’origine Végétale; Editions Tec & Doc: Paris, France, 2002. [Google Scholar]
- Beckage, N.E. Insect growth regulators. In Biological and Biotechnological Control of Insect Pests; Beckage, N.E., Rechcigl, J., Rechcigl, N., Eds.; Lewis Publishers: Boca Raton, FL, USA, 1998; pp. 123–137. [Google Scholar]
- Dawkar, V.V.; Barage, S.H.; Barbole, R.S.; Fatangare, A.; Grimalt, S.; Haldar, S.; Heckel, D.G.; Gupta, V.S.; Thulasiram, H.V.; Svatoš, A.; et al. Azadirachtin-A from Azadirachta indica impacts multiple biological targets in cotton bollworm Helicoverpa armigera. ACS Omega 2019, 4, 9531–9541. [Google Scholar] [CrossRef]
- Zhou, Y.; Qin, D.Q.; Zhang, P.W.; Chen, X.T.; Liu, B.J.; Cheng, D.M.; Zhang, Z.X. Integrated LC–MS and GC–MS-based untargeted metabolomics studies of the effect of azadirachtin on Bactrocera dorsalis larvae. Sci. Rep. 2020, 10, 2306. [Google Scholar] [CrossRef]
- Dively, G.P.; Patton, T.; Barranco, L.; Kulhanek, K. Comparative efficacy of common active ingredients in organic insecticides against difficult to control insect pests. Insects 2020, 11, 614. [Google Scholar] [CrossRef]
- Soderlund, D.M.; Bloomquist, J.R. Neurotoxic actions of pyrethroid insecticides. Annu. Rev. Entomol. 1989, 34, 77–96. [Google Scholar] [CrossRef]
- Liu, F.; Wang, Q.; Xu, P.; Andreazza, F.; Valbon, W.R.; Bandason, E.; Chen, M.; Yan, R.; Feng, B.; Smith, L.B. A dual-target molecular mechanism of pyrethrum repellency against mosquitoes. Nature Commun. 2021, 12, 1–9. [Google Scholar] [CrossRef]
- Hilje, L.; Mora, G. Chapter 15 Promissory botanical repellents/deterrents for managing two key tropical insect pests, the whitefly Bemisia tabaci and the mahogany shootborer Hypsipyla grandella. In Advances in Phytomedicine; Rai, M., Carpinella, M.C., Eds.; Elsevier: Amsterdam, The Netherlands, 2006; Volume 3, pp. 379–403. [Google Scholar]
- Kłyś, M.; Malejky, N.; Nowak-Chmura, M. The repellent effect of plants and their active substances against the beetle storage pests. J. Stored Prod. Res. 2017, 74, 66–77. [Google Scholar] [CrossRef]
- Czerniewicz, P.; Chrzanowski, G.; Sprawka, I.; Sytykiewicz, H. Aphicidal activity of selected Asteraceae essential oils and their effect on enzyme activities of the green peach aphid, Myzus persicae (Sulzer). Pestic. Biochem. Physiol. 2018, 145, 84–92. [Google Scholar] [CrossRef]
- Pascual-Villalobos, M.J.; Cantó-Tejero, M.; Vallejo, R.; Guirao, P.; Rodríguez-Rojo, S.; Cocero, M.J. Use of nanoemulsions of plant essential oils as aphid repellents. Ind. Crops Prod. 2017, 110, 45–57. [Google Scholar] [CrossRef]
- Lucia, A.; Guzmán, E. Emulsions containing essential oils, their components or volatile semiochemicals as promising tools for insect pest and pathogen management. Adv. Colloid Interface Sci. 2021, 287, 102330. [Google Scholar] [CrossRef]
- Isman, M.B. Botanical insecticides and antifeedants: New sources and perspectives. Pestic. Res. J. 1994, 6, 11–19. [Google Scholar]
- Zapata, N.; Budia, F.; Viñuela, E.; Medina, P. Antifeedant and growth inhibitory effects of extracts and drimanes of Drimys winteri stem bark against Spodoptera littoralis (Lep., Noctuidae). Ind. Crops Products 2009, 30, 119–125. [Google Scholar] [CrossRef]
- Koul, O. Phytochemicals and insect control: An antifeedant approach. Crit. Rev. Plant Sci. 2008, 27, 1–24. [Google Scholar] [CrossRef]
- Mierziak, J.; Kostyn, K.; Kulma, A. Flavonoids as important molecules of plant interactions with the environment. Molecules 2014, 19, 16240–16265. [Google Scholar] [CrossRef]
- Zhang, A.; Liu, Z.; Lei, F.; Fu, J.; Zhang, X.; Ma, W.; Zhang, L. Antifeedant and oviposition-deterring activity of total ginsenosides against Pieris rapae. Saudi J. Biol. Sci. 2017, 24, 1751–1753. [Google Scholar] [CrossRef]
- Singh, B.; Kaur, A. Control of insect pests in crop plants and stored food grains using plant saponins: A review. LWT 2018, 87, 93–101. [Google Scholar] [CrossRef]
- Akhtar, A.; Abbasi, N.A.; Hussain, A.; Bakhsh, A. Preserving quality of loquat fruit during storage by modified atmosphere packaging. Pak. J. Agric. Sci. 2012, 49, 419–423. [Google Scholar]
- Céspedes, C.L.; Torres, P.; Marın, J.C.; Arciniegas, A.; de Vivar, A.R.; Pérez-Castorena, A.L.; Aranda, E. Insect growth inhibition by tocotrienols and hydroquinones from Roldana barba-johannis. Phytochemistry 2004, 65, 1963–1975. [Google Scholar] [CrossRef]
- Rhoades, D.F. Evolution of plant defense against herbivores. In Herbivores: Their Interaction with Secondary Plant Metabolites; Rosenthal, G.A., Janzen, D.H., Eds.; Academic Press: New York, NY, USA, 1979; pp. 1–55. [Google Scholar]
- Céspedes, C.L.; Lina-Garcia, L.; Kubo, I.; Salazar, J.R.; Ariza-Castolo, A.; Alarcon, J.; Aqueveque, P.; Werner, E.; Seigler, D.S. Calceolaria integrifolia s.l. complex, reduces feeding and growth of Acanthoscelides obtectus, and Epilachna varivestis. A new source of bioactive compounds against dry bean pests. Ind. Crops Prod. 2016, 89, 257–267. [Google Scholar] [CrossRef]
- Chakraborty, R.; Thakur, T.S.; Chauhan, R.; Manzar, Z.; Das, G.; Mitra, A. Exploring the neural mechanisms of aversion to bitter gourd phytochemicals in insects using Drosophila. J. Asia-Pac. Entomol. 2021, 24, 749–758. [Google Scholar] [CrossRef]
- Kabir, K.E.; Choudhary, M.I.; Ahmed, S.; Tariq, R.M. Growth-disrupting, larvicidal and neurobehavioral toxicity effects of seed extract of Seseli diffusum against Aedes aegypti (L.) (Diptera: Culicidae). Ecotoxicol. Environ. Saf. 2013, 90, 52–60. [Google Scholar] [CrossRef]
- Rani, P.U. Fumigant and contact toxic potential of essential oils from plant extracts against stored product pests. J. Biopestic. 2012, 5, 120. [Google Scholar]
- López, M.D.; Pascual-Villalobos, M.J. Mode of inhibition of acetylcholinesterase by monoterpenoids and implications for pest control. Ind. Crops Prod. 2010, 31, 284–288. [Google Scholar] [CrossRef]
- Nenaah, G.E. Toxic and antifeedant activities of potato glycoalkaloids against Trogoderma granarium (Coleoptera: Dermestidae). J. Stored Prod. Res. 2011, 47, 185–190. [Google Scholar] [CrossRef]
- Kubo, I.; Kinst-Hori, I.; Chaudhuri, S.K.; Kubo, Y.; Sánchez, Y.; Ogura, T. Flavonols from Heterotheca inuloides: Tyrosinase inhibitory activity and structural criteria. Bioorg. Med. Chem. 2000, 8, 1749–1755. [Google Scholar] [CrossRef]
- Ma, T.; Yan, H.; Shi, X.; Liu, B.; Ma, Z.; Zhang, X. Comprehensive evaluation of effective constituents in total alkaloids from Sophora alopecuroides L. and their joint action against aphids by laboratory toxicity and field efficacy. Ind. Crops Prod. 2018, 111, 149–157. [Google Scholar] [CrossRef]
- Céspedes, C.L.; Salazar, J.R.; Martínez, M.; Aranda, E. Insect growth regulatory effects of some extracts and sterols from Myrtillocactus geometrizans (Cactaceae) against Spodoptera frugiperda and Tenebrio molitor. Phytochemistry 2005, 66, 2481–2493. [Google Scholar] [CrossRef]
- Varma, J.; Dubey, N. Prospectives of botanical and microbial products as pesticides of tomorrow. Curr. Sci. 1999, 76, 172–179. [Google Scholar]
- Miresmailli, S.; Isman, M.B. Botanical insecticides inspired by plant-herbivore chemical interactions. Trends Plant Sci. 2014, 19, 29–35. [Google Scholar] [CrossRef]
- Singh, K.D.; Mobolade, A.J.; Bharali, R.; Sahoo, D.; Rajashekar, Y. Main plant volatiles as stored grain pest management approach: A review. J. Agric. Food Res. 2021, 4, 100127. [Google Scholar] [CrossRef]
- Céspedes, C.L.; Sampietro, D.; Seigler, D.S.; Rai, M. Natural Antioxidants and Biocides from Wild Medicinal Plants; CAB International: Wallingford, UK, 2013. [Google Scholar]
- Muñoz, E.; Lamilla, C.; Marin, J.C.; Alarcon, J.; Cespedes, C.L. Antifeedant, insect growth regulatory and insecticidal effects of Calceolaria talcana (Calceolariaceae) on Drosophila melanogaster and Spodoptera frugiperda. Ind. Crops Prod. 2013, 42, 137–144. [Google Scholar] [CrossRef]
- Mulla, M.S.; Su, T. Activity and biological effects of neem products against arthropods of medical and veterinary importance. J. Am. Mosq. Control Assoc. 1999, 15, 133–152. [Google Scholar]
- Muema, J.M.; Bargul, J.L.; Mutunga, J.M.; Obonyo, M.A.; Asudi, G.O.; Njeru, S.N. Neurotoxic Zanthoxylum chalybeum root constituents invoke mosquito larval growth retardation through ecdysteroidogenic CYP450s transcriptional perturbations. Pestic. Biochem. Physiol. 2021, 178, 104912. [Google Scholar] [CrossRef]
- Quiroz-Carreño, S.; Pastene-Navarrete, E.; Espinoza-Pinochet, C.; Muñoz-Núñez, E.; Devotto-Moreno, L.; Céspedes-Acuña, C.L.; Alarcón-Enos, J. Assessment of insecticidal activity of benzylisoquinoline alkaloids from Chilean Rhamnaceae plants against fruit-fly Drosophila melanogaster and the lepidopteran crop pest Cydia pomonella. Molecules 2020, 25, 5094. [Google Scholar] [CrossRef]
- Hasheminia, S.M.; Sendi, J.J.; Jahromi, K.T.; Moharramipour, S. The effects of Artemisia annua L. and Achillea millefolium L. crude leaf extracts on the toxicity, development, feeding efficiency and chemical activities of small cabbage Pieris rapae L. (Lepidoptera: Pieridae). Pestic. Biochem. Physiol. 2011, 99, 244–249. [Google Scholar] [CrossRef]
- Sousa, R.M.O.F.; Rosa, J.S.; Oliveira, L.; Cunha, A.C.; Fernandes-Ferreira, M. Activities of Apiaceae essential oils and volatile compounds on hatchability, development, reproduction and nutrition of Pseudaletia unipuncta (Lepidoptera: Noctuidae). Ind. Crops Prod. 2015, 63, 226–237. [Google Scholar] [CrossRef]
- Chowański, S.; Chudzińska, E.; Lelario, F.; Ventrella, E.; Marciniak, P.; Miądowicz-Kobielska, M.; Spochacz, M.; Szymczak, M.; Scrano, L.; Bufo, S.A.; et al. Insecticidal properties of Solanum nigrum and Armoracia rusticana extracts on reproduction and development of Drosophila melanogaster. Ecotoxicol. Environ. Saf. 2018, 162, 454–463. [Google Scholar] [CrossRef]
- Ibanez, S.; Gallet, C.; Després, L. Plant insecticidal toxins in ecological networks. Toxins 2012, 4, 228–243. [Google Scholar] [CrossRef]
- Abdellaoui, K.; Hazzoug, M.; Boussadia, O.; Miladi, M.; Omri, G.; Acheuk, F.; Ben Halima-Kamel, M.; Brahem, M. Physiological and biochemical effects of Olea europaea leaf extracts from four phenological growth stages on the oogenesis of female locust Locusta migratoria. Physiol. Entomol. 2018, 43, 129–139. [Google Scholar] [CrossRef]
- Acheuk, F.; Cusson, M.; Doumandji-Mitiche, B. Effects of a methanolic extract of the plant Haplophyllum tuberculatum and of teflubenzuron on female reproduction in the migratory locust, Locusta migratoria (Orthoptera: Oedipodinae). J. Insect Physiol. 2012, 58, 335–341. [Google Scholar] [CrossRef]
- Bowers, W.S. Insect hormones and antihormones in plants. In Herbivores: Their Interactions with Secondary Plant Metabolites; Academic Press: New York, NY, USA, 2012; pp. 431–456. [Google Scholar]
- Lee, S.-H.; Ha, K.B.; Park, D.H.; Fang, Y.; Kim, J.H.; Park, M.G.; Woo, R.M.; Kim, W.J.; Park, I.-K.; Choi, J.Y.; et al. Plant-derived compounds regulate formation of the insect juvenile hormone receptor complex. Pestic. Biochem. Physiol. 2018, 150, 27–32. [Google Scholar] [CrossRef]
- Chaieb, K.; Hajlaoui, H.; Zmantar, T.; Kahla-Nakbi, A.B.; Rouabhia, M.; Mahdouani, K.; Bakhrouf, A. The chemical composition and biological activity of clove essential oil, Eugenia caryophyllata (Syzigium aromaticum L. Myrtaceae): A short review. Phytother. Res. 2007, 21, 501–506. [Google Scholar] [CrossRef]
- Jirovetz, L.; Buchbauer, G.; Stoilova, I.; Stoyanova, A.; Krastanov, A.; Schmidt, E. Chemical composition and antioxidant properties of clove leaf essential oil. J. Agric. Food Chem. 2006, 54, 6303–6307. [Google Scholar] [CrossRef] [PubMed]
- Pinto, E.; Vale-Silva, L.; Cavaleiro, C.; Salgueiro, L. Antifungal activity of the clove essential oil from Syzygium aromaticum on Candida, Aspergillus and dermatophyte species. J. Med. Microbiol. 2009, 58, 1454–1462. [Google Scholar] [CrossRef] [PubMed]
- Abbas, H.K.; Bader, Q.A.; Hussein, A.H.; Shaheed, D.Q. Preparation and in vitro evaluation of clove oil microemulsion. Int. J. Pharm. Res. 2020, 12, 422–430. [Google Scholar]
- Almarie, A. Bioherbicidal potential of Eucalyptus and clove oil and their combinations on four weedy species. Iraqi J. Sci. 2021, 62, 1494–1502. [Google Scholar] [CrossRef]
- Ahuja, N.; Singh, H.P.; Batish, D.R.; Kohli, R.K. Eugenol-inhibited root growth in Avena fatua involves ROS-mediated oxidative damage. Pestic. Biochem. Physiol. 2015, 118, 64–70. [Google Scholar] [CrossRef]
- Bainard, L.D.; Isman, M.B.; Upadhyaya, M.K. Phytotoxicity of clove oil and its primary constituent eugenol and the role of leaf epicuticular wax in the susceptibility to these essential oils. Weed Sci. 2006, 54, 833–837. [Google Scholar] [CrossRef]
- Boyd, N.S.; Brennan, E.B. Burning nettle, common purslane, and rye response to a clove oil herbicide. Weed Technol. 2006, 20, 646–650. [Google Scholar] [CrossRef]
- Akhila, A. Essential Oil-Bearing Grasses: The Genus Cymbopogon; CRC Press: Boca Raton, FL, USA, 2009. [Google Scholar]
- Ootani, M.A.; dos Reis, M.R.; Cangussu, A.S.R.; Capone, A.; Fidelis, R.R.; Oliveira, W.; Barros, H.B.; Portella, A.C.F.; de Souza Aguiar, R.; dos Santos, W.F. Phytotoxic effects of essential oils in controlling weed species Digitaria horizontalis and Cenchrus echinatus. Biocatal. Agric. Biotechnol. 2017, 12, 59–65. [Google Scholar] [CrossRef]
- Lins, L.; Dal Maso, S.; Foncoux, B.; Kamili, A.; Laurin, Y.; Genva, M.; Jijakli, M.H.; De Clerck, C.; Fauconnier, M.L.; Deleu, M. Insights into the relationships between herbicide activities, molecular structure and membrane interaction of cinnamon and citronella essential oils components. Int. J. Mol. Sci. 2019, 20, 4007. [Google Scholar] [CrossRef]
- Hensel, A.S.; Engelshowe, T.J.; Lechtenberg, R.; Petereit, M.; Krüger, F. Arzneipflanzen Entdecken. 2. Erweiterte Auflage; Eigenverlag, 280 Seiten mit Farbabbildungen: Münster, Deutschland, 2010. [Google Scholar]
- Yanakiev, S. Effects of cinnamon (Cinnamomum spp.) in dentistry: A review. Molecules 2020, 25, 4184. [Google Scholar] [CrossRef]
- Prasanna, B.; Anand, A.V. Cinnamon species: In vivo anti-oxidant activity of ethanolic extracts of Cinnamon zeylanicum and Cinnamon cassicae Barks. Pharmacogn. J. 2019, 11, 245–247. [Google Scholar] [CrossRef]
- Vangalapati, M.; Satya, N.S.; Prakash, D.S.; Avanigadda, S. A review on pharmacological activities and clinical effects of Cinnamon species. Res. J. Pharm. Biol. Chem. Sci. 2012, 3, 653–663. [Google Scholar]
- Campiglia, E.; Mancinelli, R.; Cavalieri, A.; Caporali, F. Use of essential oils of cinnamon, lavender and peppermint for weed control. Ital. J. Agron. 2007, 2, 171–178. [Google Scholar] [CrossRef]
- Tworkoski, T. Herbicide effects of essential oils. Weed Sci. 2002, 50, 425–431. [Google Scholar] [CrossRef]
- Zimmermann, G. Übersichtsarbeit-Wirkung und Anwendungsmöglichkeiten ätherischer Öle im Pflanzenschutz: Eine Übersicht. J. Kult. 2012, 64, 1. [Google Scholar]
- Chotsaeng, N.; Laosinwattana, C.; Charoenying, P. Inhibitory effects of a variety of aldehydes on Amaranthus tricolor L. and Echinochloa crus-galli (L.) Beauv. Molecules 2018, 23, 471. [Google Scholar] [CrossRef]
- Coppen, J.J. Production, trade and markets for eucalyptus oils. In Eucalyptus; CRC Press: Boca-Raton, FL, USA, 2002; pp. 379–397. [Google Scholar]
- Batish, D.R.; Singh, H.P.; Kohli, R.K.; Kaur, S. Eucalyptus essential oil as a natural pesticide. Forest Ecol. Manage. 2008, 256, 2166–2174. [Google Scholar] [CrossRef]
- Verdeguer, M.; Sánchez-Moreiras, A.M.; Araniti, F. Phytotoxic effects and mechanism of action of essential oils and terpenoids. Plants 2020, 9, 1571. [Google Scholar] [CrossRef]
- Grosso, C.; Figueiredo, A.C.; Burillo, J.; Mainar, A.M.; Urieta, J.S.; Barroso, J.G.; Coelho, J.A.; Palavra, A.M. Composition and antioxidant activity of Thymus vulgaris volatiles: Comparison between supercritical fluid extraction and hydrodistillation. J. Sep. Sci. 2010, 33, 2211–2218. [Google Scholar] [CrossRef]
- Soltys, D.; Krasuska, U.; Bogatek, R.; Gniazdowska, A. Allelochemicals as bioherbicides—Present and perspectives. In Herbicides —Current Research and Case Studies in Use; Price, A., Ed.; IntechOpen: London, UK, 2013. [Google Scholar]
- Grosso, C.; Coelho, J.A.; Urieta, J.S.; Palavra, A.M.F.; Barroso, J.G. Herbicidal activity of volatiles from coriander, winter savory, cotton lavender, and thyme isolated by hydrodistillation and supercritical fluid extraction. J. Agr. Food Chem. 2010, 58, 11007–11013. [Google Scholar] [CrossRef]
- Ramezani, S.; Saharkhiz, M.J.; Ramezani, F.; Fotokian, M.H. Use of essential oils as bioherbicides. J. Essent. Oil-Bear. Plants 2008, 11, 319–327. [Google Scholar] [CrossRef]
- Santos, A.A.; Hancox, T.J.J.; Picanço, M.C.; Delaporte, K.; Hogendoorn, K. Potential distribution of Leptospermum species (Myrtaceae) in Australia for bioactive honey production purposes. N. Z. J. Crop Hortic. Sci. 2021, 1–12. [Google Scholar] [CrossRef]
- Thompson, J. Revision of the genus Leptospermum (Myrtaceae). Telopea 1989, 3, 301–449. [Google Scholar] [CrossRef]
- Bean, A. The genus Leptospermum Forst et Forst. F. (Myrtaceae) in northern Australia and Malesia. Austrobaileya 1992, 3, 643–659. [Google Scholar]
- Brophy, J.J.; Goldsack, R.J.; Punruckvong, A.; Bean, A.R.; Forster, P.I.; Lepschi, B.J.; Doran, J.C.; Rozefelds, A.C. Leaf essential oils of the genus Leptospermum (Myrtaceae) in eastern Australia. Part 7 Leptospermum petersonii, L. liversidgei and allies. Flavour Fragr. J. 2000, 15, 342–351. [Google Scholar] [CrossRef]
- Dawson, M. A history of Leptospermum scoparium in cultivation: Discoveries from the wild. N. Z. Gard. J. 2009, 12, 21–25. [Google Scholar]
- Dayan, F.E.; Howell, J.L.; Marais, J.P.; Ferreira, D.; Koivunen, M. Manuka oil, a natural herbicide with preemergence activity. Weed Sci. 2011, 59, 464–469. [Google Scholar] [CrossRef]
- Harris, S.A. Ιnvestigating Tank Mix Partners to Improve the Efficacy of Manuka Oil as a Herbicide for Organic Vegetable Production. Master’s Thesis, The University of Guelph, Guelph, ON, Canada, 2017. [Google Scholar]
- Killeen, D.P.; Klink, J.W.; Smallfield, B.M.; Gordon, K.C.; Perry, N.B. Herbicidal β-triketones are compartmentalized in leaves of Leptospermum species: Localization by Raman microscopy and rapid screening. N. Phytol. 2015, 205, 339–349. [Google Scholar] [CrossRef] [PubMed]
- Dayan, E.; Censor, N.; Buch, E.R.; Sandrini, M.; Cohen, L.G. Noninvasive brain stimulation: From physiology to network dynamics and back. Nat. Neurosci. 2013, 16, 838–844. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.; Sharma, R.A. Secondary Metabolites of Medicinal Plants: Ethnopharmacological Properties, Biological Activity and Production Strategies; Wiley-VCH Verlag GmbH & Co. KGaA.: Weinheim, Germany, 2020; Volume 3. [Google Scholar]
- Jnanesha, A.A.K.S. Agrotechnology for Ocimum Species in India. Agrobios Newsl. 2019, XVII, 67–68. [Google Scholar]
- Maurya, S.; Sangwan, N.S. Profiling of essential oil constituents in Ocimum species. Proc. Natl. Acad. Sci. India Sect. B Biol. Sci. 2020, 90, 577–583. [Google Scholar] [CrossRef]
- Grayer, R.J.; Kite, G.C.; Goldstone, F.J.; Bryan, S.E.; Paton, A.; Putievsky, E. Infraspecific taxonomy and essential oil chemotypes in sweet basil, Ocimum basilicum. Phytochemistry 1996, 43, 1033–1039. [Google Scholar] [CrossRef]
- Mekky, M.S.; Hassanien, A.M.A.; Kamel, E.M.; Ismail, A.E.A. Allelopathic effect of Ocimum basilicum L. extracts on weeds and some crops and its possible use as new crude bio-herbicide. Ann. Agric. Sci. 2019, 64, 211–221. [Google Scholar] [CrossRef]
- Telci, I.; Bayram, E.; Yılmaz, G.; Avcı, B. Variability in essential oil composition of Turkish basils (Ocimum basilicum L.). Biochem. Syst. Ecol. 2006, 34, 489–497. [Google Scholar] [CrossRef]
- Wogiatzi, E.; Gougoulias, N.; Papachatzis, A.; Vagelas, I.; Chouliaras, N. Greek oregano essential oils production, phytotoxicity and antifungal activity. Biotechnol. Biotechnol. Equip. 2009, 23, 1150–1152. [Google Scholar] [CrossRef]
- Ibáñez, M.D.; Blázquez, M.A. Phytotoxic effects of commercial Eucalyptus citriodora, Lavandula angustifolia, and Pinus sylvestris essential oils on weeds, crops, and invasive species. Molecules 2019, 24, 2847. [Google Scholar] [CrossRef]
- Islam, A.K.M.M.; Kato-Noguchi, H. Phytotoxic activity of Ocimum tenuiflorum extracts on germination and seedling growth of different plant species. Sci. World J. 2014, 2014, 676242. [Google Scholar] [CrossRef]
- De Mastro, G.; El Mahdi, J.; Ruta, C. Bioherbicidal potential of the essential oils from Mediterranean Lamiaceae for weed control in organic farming. Plants 2021, 10, 818. [Google Scholar] [CrossRef]
- Figuérédo, G.; Cabassu, P.; Chalchat, J.-C.; Pasquier, B. Studies of Mediterranean oregano populations. VIII—Chemical composition of essential oils of oreganos of various origins. Flavour Fragr. J. 2006, 21, 134–139. [Google Scholar] [CrossRef]
- Grana, E.; Diaz-Tielas, C.; Sanchez-Moreiras, A.M.; Reigosa, M.J. Mode of action of monoterpenes in plant-plant interactions. Curr. Bioact. Compd. 2012, 8, 80–89. [Google Scholar] [CrossRef]
- Oliva, M.d.l.M.; Carezzano, M.E.; Giuliano, M.; Daghero, J.; Zygadlo, J.; Bogino, P.; Giordano, W.; Demo, M. Antimicrobial activity of essential oils of Thymus vulgaris and Origanum vulgare on phytopathogenic strains isolated from soybean. Plant Biol. J. 2015, 17, 758–765. [Google Scholar] [CrossRef]
- Argyropoulos, E.; Eleftherohorinos, I.; Vokou, D. In vitro evaluation of essential oils from Mediterranean aromatic plants of the Lamiaceae for weed control in tomato and cotton crops. Allelopath. J. 2008, 22, 69–78. [Google Scholar]
- Ibáñez, M.D.; Blázquez, M.A. Herbicidal value of essential oils from oregano-like flavour species. Food Agric. Immunol. 2017, 28, 1168–1180. [Google Scholar] [CrossRef]
- Kordali, S.; Cakir, A.; Ozer, H.; Cakmakci, R.; Kesdek, M.; Mete, E. Antifungal, phytotoxic and insecticidal properties of essential oil isolated from Turkish Origanum acutidens and its three components, carvacrol, thymol and p-cymene. Bioresour. Technol. 2008, 99, 8788–8795. [Google Scholar] [CrossRef] [PubMed]
- Uludag, A.; Uremis, I.; Arslan, M.; Gozcu, D. Allelopathy studies in weed science in Turkey—A review. Z. Pflanzenkrankh. Pflanzenpathol. Pflanzenschutz, Sonderh. 2006, 20, 419. [Google Scholar]
- Atak, M.; Mavİ, K.; Üremİș, İ. Allelopathic effects of oregano and rosemary essential oils on germination and seedling growth of durum wheat. In Proceedings of the VII International Scientific Agriculture Symposium,”Agrosym 2016”, Jahorina, Bosnia and Herzegovina, 6–9 October 2016; pp. 1178–1183. [Google Scholar]
- Hanana, M.; Mansour, M.B.; Algabr, M.; Amri, I.; Gargouri, S.; Romane, A.; Jamoussi, B.; Hamrouni, L. Potential use of essential oils from four Tunisian species of Lamiaceae: Biological alternative for fungal and weed control. Rec. Nat. Prod. 2017, 11, 258–269. [Google Scholar]
- Frantz, J.; Pitchay, D.; Tay, D.; Ehrenberger, J.; Gray, J.; Heckathorn, S.; Leisner, S. Nutrient ranges in Pelargonium ×hortorum and Pelargonium species. HortScience 2006, 41, 980. [Google Scholar] [CrossRef]
- Saraswathi, J.; Venkatesh, K.; Baburao, N.; Hilal, M.H.; Rani, A.R. Phytopharmacological importance of Pelargonium species. J. Med. Plants Res. 2011, 5, 2587–2598. [Google Scholar]
- Singh, H.; Batish, D.; Kohli, R. Allelopathic effect of two volatile monoterpenes against bill goat weed (Ageratum conyzoides L.). Crop Prot. 2002, 21, 347–350. [Google Scholar] [CrossRef]
- Butler, G.; Rasmussen, M.D.; Lin, M.F.; Santos, M.A.S.; Sakthikumar, S.; Munro, C.A.; Rheinbay, E.; Grabherr, M.; Forche, A.; Reedy, J.L.; et al. Evolution of pathogenicity and sexual reproduction in eight Candida genomes. Nature 2009, 459, 657–662. [Google Scholar] [CrossRef]
- Kolodziej, H. Traditionally-used Pelargonium species: Chemistry and biological activity of umckaloabo extracts and their constituents. Curr. Top. Phytochem. 2000, 3, 77–93. [Google Scholar]
- Bigos, M.; Wasiela, M.; Kalemba, D.; Sienkiewicz, M. Antimicrobial activity of geranium oil against clinical strains of Staphylococcus aureus. Molecules 2012, 17, 10276–10291. [Google Scholar] [CrossRef]
- Southwell, I.A.; Stiff, I.A. Chemical composition of an Australian geranium oil. J. Essent. Oil Res. 1995, 7, 545–547. [Google Scholar] [CrossRef]
- Alvarez, F.; Arena, M.; Auteri, D.; Borroto, J.; Brancato, A.; Carrasco Cabrera, L.; Castoldi, A.F.; Chiusolo, A.; Colagiorgi, A. Peer review of the pesticide risk assessment of the active substance pelargonic acid (nonanoic acid). EFSA J. 2021, 19, e06813. [Google Scholar]
- Capatina, F.; Suciu, E.; Benedec, D. Phytochemical analysis and antioxidant activity of some Thymus species from Romania. Rom. J. Pharm. Pract. 2021, 14, 27–33. [Google Scholar] [CrossRef]
- Tohidi, B.; Rahimmalek, M.; Arzani, A. Variation in phytochemical, morphological, and ploidy levels of Iranian Thymus species. Chem. Biodivers. 2021, 18, e2000911. [Google Scholar] [CrossRef]
- Wang, J.; Dong, H.; Zhang, H.; Dai, S.; Jiang, J.; Zhao, Z. Isopropyl cresols: Synthesis and herbicidal activity. ChemistrySelect 2020, 5, 1294–1299. [Google Scholar] [CrossRef]
- Zhou, S.; Han, C.; Zhang, C.; Kuchkarova, N.; Wei, C.; Zhang, C.; Shao, H. Allelopathic, phytotoxic, and insecticidal effects of Thymus proximus Serg. essential oil and its major constituents. Front. Plant Sci. 2021, 12, 689875. [Google Scholar] [CrossRef]
- Ali, I.B.E.H.; Chaouachi, M.; Bahri, R.; Chaieb, I.; Boussaïd, M.; Harzallah-Skhiri, F. Chemical composition and antioxidant, antibacterial, allelopathic and insecticidal activities of essential oil of Thymus algeriensis Boiss. et Reut. Ind. Crops Prod. 2015, 77, 631–639. [Google Scholar]
- Ibrahim, H.; Salama, A.M.; Abou El-Leel, O.F. Analysis of genetic diversity of Lavandula species using taxonomic, essential oil and molecular genetic markers. Middle East J. Appl. Sci. 2017, 7, 141–154. [Google Scholar]
- Sancaktaroğlu, S.; Kumlay, A.; Yıldırım, B. Micropropogation studies on Lavandula species. In Proceedings of the 4th International Conference on Advances in Natural & Applied Science Agriculture, Ağrı, Turkey, 19–22 June 2019. [Google Scholar]
- Zuzarte, M.; Dinis, A.; Canhoto, J.; Salgueiro, L. Leaf trichomes of Portuguese Lavandula species: A comparative morphological study. Microsc. Microanal. 2009, 15, 37–38. [Google Scholar] [CrossRef]
- Kim, N.-S.; Lee, D.-S. Comparison of different extraction methods for the analysis of fragrances from Lavandula species by gas chromatography–mass spectrometry. J. Chromatogr. A 2002, 982, 31–47. [Google Scholar] [CrossRef]
- Singh, H.; Batish, D.; Kaur, S.; Ramezani, H.; Kohli, R. Comparative phytotoxicity of four monoterpenes against Cassia occidentalis. Ann. Appl. Biol. 2002, 141, 111–116. [Google Scholar] [CrossRef]
- Karaman, S.; Toker, Ö.S.; Yüksel, F.; Çam, M.; Kayacier, A.; Dogan, M. Physicochemical, bioactive, and sensory properties of persimmon-based ice cream: Technique for order preference by similarity to ideal solution to determine optimum concentration. J. Dairy Sci. 2014, 97, 97–110. [Google Scholar] [CrossRef]
- Di Pasqua, R.; Hoskins, N.; Betts, G.; Mauriello, G. Changes in membrane fatty acids composition of microbial cells induced by addiction of thymol, carvacrol, limonene, cinnamaldehyde, and eugenol in the growing media. J. Agric. Food Chem. 2006, 54, 2745–2749. [Google Scholar] [CrossRef]
- dos Santos Silva, C.T.; Wanderley-Teixeira, V.; da Cunha, F.M.; de Oliveira, J.V.; de Andrade Dutra, K.; Navarro, D.M.d.A.F.; Teixeira, Á.A.C. Biochemical parameters of Spodoptera frugiperda (JE Smith) treated with citronella oil (Cymbopogon winterianus Jowitt ex Bor) and its influence on reproduction. Acta Histochem. 2016, 118, 347–352. [Google Scholar] [CrossRef]
- Fagodia, S.; Batish, D.; Singh, H.P. Comparitive phytotoxicity of three monoterpenes against Cassia occidentalis. Int. J. Adv. Technol. Eng. Sci. 2017, 5, 79–84. [Google Scholar]
- Vaid, S. Phytotoxicity of citronellol against Amaranthus viridis L. Int. J. Eng. Appl. Sci. 2015, 2, 257763. [Google Scholar]
- Gilles, M.; Zhao, J.; An, M.; Agboola, S. Chemical composition and antimicrobial properties of essential oils of three Australian Eucalyptus species. Food Chem. 2010, 119, 731–737. [Google Scholar] [CrossRef]
- Rodenak-Kladniew, B.; Castro, A.; Stärkel, P.; Galle, M.; Crespo, R. 1, 8-Cineole promotes G0/G1 cell cycle arrest and oxidative stress-induced senescence in HepG2 cells and sensitizes cells to anti-senescence drugs. Life Sci. 2020, 243, 117271. [Google Scholar] [CrossRef]
- Werrie, P.-Y.; Durenne, B.; Delaplace, P.; Fauconnier, M.-L. Phytotoxicity of essential oils: Opportunities and constraints for the development of biopesticides. A review. Foods 2020, 9, 1291. [Google Scholar] [CrossRef]
- Sampaio, L.A.; Pina, L.T.S.; Serafini, M.R.; dos Santos Tavares, D.; Guimarães, A.G. Antitumor effects of carvacrol and thymol: A systematic review. Front. Pharmacol. 2021, 12, 702487. [Google Scholar] [CrossRef]
- Ciriminna, R.; Fidalgo, A.; Ilharco, L.M.; Pagliaro, M. Herbicides based on pelargonic acid: Herbicides of the bioeconomy. Biofuel Bioprod. Biorefin. 2019, 13, 1476–1482. [Google Scholar] [CrossRef]
- Foyer, C.H.; Lelandais, M.; Kunert, K.J. Photooxidative stress in plants. Physiol. Plant 1994, 92, 696–717. [Google Scholar] [CrossRef]
- Travlos, I.; Rapti, E.; Gazoulis, I.; Kanatas, P.; Tataridas, A.; Kakabouki, I.; Papastylianou, P. The herbicidal potential of different pelargonic acid products and essential oils against several important weed species. Agronomy 2020, 10, 1687. [Google Scholar] [CrossRef]
- Yoon, M.-Y.; Cha, B.; Kim, J.-C. Recent trends in studies on botanical fungicides in agriculture. Plant Pathol. J. 2013, 29, 1–9. [Google Scholar] [CrossRef]
- Lattanzio, V.; Cardinali, A.; Palmieri, S. The role of phenolics in the postharvest physiology of fruits and vegetables: Browning reaction and fungal diseases. Ital. J. Food Sci. 1994, 1, 3–22. [Google Scholar]
- Cowan, M.M. Plant products as antimicrobial agents. Clin. Microbiol. Rev. 1999, 12, 564–582. [Google Scholar] [CrossRef]
- Mihai, A.L.; Popa, M.E. In vitro activity of natural antimicrobial compounds against Aspergillus strains. Agric. Agric. Sci. Procedia 2015, 6, 585–592. [Google Scholar] [CrossRef]
- Mendoza, L.; Wilkens, M.; Urzúa, A. Antimicrobial study of the resinous exudates and of diterpenoids and flavonoids isolated from some Chilean Pseudognaphalium (Asteraceae). J. Ethnopharmacol. 1997, 58, 85–88. [Google Scholar] [CrossRef]
- Garcia, D.; Ramos, A.J.; Sanchis, V.; Marín, S. Effect of Equisetum arvense and Stevia rebaudiana extracts on growth and mycotoxin production by Aspergillus flavus and Fusarium verticillioides in maize seeds as affected by water activity. Int. J. Food Microbiol. 2012, 153, 21–27. [Google Scholar] [CrossRef]
- Zhou, C.-X.; Liu, J.-Y.; Ye, W.-C.; Liu, C.-H.; Tan, R.-X. Neoverataline A and B, two antifungal alkaloids with a novel carbon skeleton from Veratrum taliense. Tetrahedron 2003, 59, 5743–5747. [Google Scholar] [CrossRef]
- Houghton, P.J.; Woldemariam, T.Z.; Khan, A.I.; Burke, A.; Mahmood, N. Antiviral activity of natural and semi-synthetic chromone alkaloids. Antiviral Res. 1994, 25, 235–244. [Google Scholar] [CrossRef]
- Hulot, J.F.; Hiller, N. Exploring the Benefits of Biocontrol for Sustainable Agriculture—A Literature Review on Biocontrol in Light of the European Green Deal; Institute for European Environmental Policy: Brussels, Belgium, 2021. [Google Scholar]
- Sundh, I.; Eilenberg, J. Why has the authorization of microbial biological control agents been slower in the EU than in comparable jurisdictions? Pest Manag. Sci. 2021, 77, 2170–2178. [Google Scholar] [CrossRef] [PubMed]
- Scheepmaker, J.W.A.; Busschers, M.; Sundh, I.; Eilenberg, J.; Butt, T.M. Sense and nonsense of the secondary metabolites data requirements in the EU for beneficial microbial control agents. Biol. Control 2019, 136, 104005. [Google Scholar] [CrossRef]
- Ward, M. The regulatory landscape for biological control agents. EPPO Bull. 2016, 46, 249–253. [Google Scholar] [CrossRef]
- Marchand, P.A. Basic and low-risk substances under European Union pesticide regulations: A new choice for biorational portfolios of small and medium-sized enterprises. J. Plant Prot. Res. 2017, 57, 433–440. [Google Scholar] [CrossRef][Green Version]
- COM_(2020)_208_final. Evaluation of Regulation (EC) No 1107/2009 on the Placing of Plant Protection Products on the Market and of Regulation (EC) No 396/2005 on Maximum Residue Levels of Pesticides; Report from the Commission to the European Parliament and the Council; The European Commission: Brussels, Belgium, 2020. [Google Scholar]
- Matyjaszczyk, E. Products containing microorganisms as a tool in integrated pest management and the rules of their market placement in the European Union. Pest Manag. Sci. 2015, 71, 1201–1206. [Google Scholar] [CrossRef]
- Rakshit, A.; Meena, V.S.; Abhilash, P.C.; Sarma, B.K.; Singh, H.B.; Fraceto, L.; Parihar, M.; Kumar Singh, A. Biopesticides: Advances in Bio-Inoculants; Woodhead Publishing: Cambridge, UK, 2021; Volume 2. [Google Scholar]
- Balog, A.; Hartel, T.; Loxdale, H.D.; Wilson, K. Differences in the progress of the biopesticide revolution between the EU and other major crop-growing regions. Pest Manag. Sci. 2017, 73, 2203–2208. [Google Scholar] [CrossRef] [PubMed]
- Kumar, J.; Ramlal, A.; Mallick, D.; Mishra, V. An overview of some biopesticides and their importance in plant protection for commercial acceptance. Plants 2021, 10, 1185. [Google Scholar] [CrossRef] [PubMed]
- Hassan, E.; Gökçe, A. Production and consumption of biopesticides. In Advances in Plant Biopesticides; Springer: New Delhi, India, 2014; pp. 361–379. [Google Scholar]
- Bailey, K.L.; Boyetchko, S.M.; Längle, T. Social and economic drivers shaping the future of biological control: A Canadian perspective on the factors affecting the development and use of microbial biopesticides. Biol. Control 2010, 52, 221–229. [Google Scholar] [CrossRef]
- Glare, T.R.; Gwynn, R.L.; Morán-Diez, M.E. Development of biopesticides and future opportunities. In Microbial-Based Biopesticides. Methods in Molecular Biology; Glare, T., Moran-Diez, M., Eds.; Humana Press: New York, NY, USA, 2016; Volume 1477, pp. 211–221. [Google Scholar]
- Bourguignon, D. EU Policy and Legislation on Pesticides—Plant Protection Products and Biocides; PE 599.428; European Parliamentary Research Service: Brussels, Belgium, 2017. [Google Scholar]
- Buckwell, A.; de Wachter, E.; Nadeu, E.; Williams, A. Crop Protection & the EU Food System. Where Are They Going? RISE Foundation: Brussels, Belgium, 2020. [Google Scholar]
- Arora, N.K.; Verma, M.; Prakash, J.; Mishra, J. Regulation of biopesticides: Global concerns and policies. In Bioformulations: For Sustainable Agriculture; Arora, N., Mehnaz, S., Balestrini, R., Eds.; Springer: New Delhi, India, 2016. [Google Scholar]
- Gupta, S.; Dikshit, A.K. Biopesticides: An ecofriendly approach for pest control. J. Biopestic. 2010, 3, 186–188. [Google Scholar]
- Damalas, C.A.; Koutroubas, S.D. Current status and recent developments in biopesticide use. Agriculture 2018, 8, 13. [Google Scholar] [CrossRef]
- OECD. Report of the 8th Biopesticides Expert Group Seminar on Niche Uses of Highly Specific Biocontrol Products. OECD Environment, Health and Safety Publications Series on Pesticides No. 95: Environment Directorate Organisation for Economic Cooperation and Development; OECD: Paris, France, 2018. [Google Scholar]
- Wandahwa, P.; Van Ranst, E.; Van Damme, P. Pyrethrum (Chrysanthemum cinerariaefolium Vis.) cultivation in West Kenya: Origin, ecological conditions and management. Ind. Crops Prod. 1996, 5, 307–322. [Google Scholar] [CrossRef]

| Insecticide | Main Mode of Action | Remarks |
|---|---|---|
| Azadirachtin | Stops larval moulting by inhibiting ecdysteroids synthesis (moulting hormones). Acts as repellent and antifeedant. Causes sterility in adult females. | Broad spectrum insecticide targeting aphids, whiteflies, fungus gnats and two-spotted mits. |
| Nicotinoids | Mimic acetylcholine neurotransmitter. | Banned by the EU in 2018, due to its harmful effect on honeybees. |
| Pyrethrins | Disruption of sodium and potassium ion exchange in insect nerve fibres, leading to immediate paralysis. | Synergized by piperonyl butoxide (PBO). |
| Ryanodine | Acts as stomach poison, with ryanodine receptors influencing the secretion of Ca2+. | Synergized by PBO. |
| Rayania | Inhibits cellular respiration (mitochondrial poison). | Extremely toxic to fish and insects. EU announced in 2008 a phase-out of rotenone (EC 2008/317). |
| Rotenone | Neurotoxic, causing paralysis and death. | Synergized by PBO or N-octyl bicycloheptene dicarboximide (MGK-264). |
| Sabadilla | Repellent, anti-feedant, Na+ channel agonist, neurotoxic. | Broad spectrum insecticide, mild activities, highly toxic to bees. |
| Compound | Remarks |
|---|---|
| Flavonoids | Bind to adhesions |
| Phenol | Substrate deprivation |
| Phenolic acids | Membrane disruption |
| Quinones | Bind to adhesions link to cell wall, enzyme inactivation |
| Tannins | Bind to proteins, bind to adhesions, membrane disruption, enzyme inhibition, substrate deprivation and metal ions complexation |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Acheuk, F.; Basiouni, S.; Shehata, A.A.; Dick, K.; Hajri, H.; Lasram, S.; Yilmaz, M.; Emekci, M.; Tsiamis, G.; Spona-Friedl, M.; et al. Status and Prospects of Botanical Biopesticides in Europe and Mediterranean Countries. Biomolecules 2022, 12, 311. https://doi.org/10.3390/biom12020311
Acheuk F, Basiouni S, Shehata AA, Dick K, Hajri H, Lasram S, Yilmaz M, Emekci M, Tsiamis G, Spona-Friedl M, et al. Status and Prospects of Botanical Biopesticides in Europe and Mediterranean Countries. Biomolecules. 2022; 12(2):311. https://doi.org/10.3390/biom12020311
Chicago/Turabian StyleAcheuk, Fatma, Shereen Basiouni, Awad A. Shehata, Katie Dick, Haifa Hajri, Salma Lasram, Mete Yilmaz, Mevlüt Emekci, George Tsiamis, Marina Spona-Friedl, and et al. 2022. "Status and Prospects of Botanical Biopesticides in Europe and Mediterranean Countries" Biomolecules 12, no. 2: 311. https://doi.org/10.3390/biom12020311
APA StyleAcheuk, F., Basiouni, S., Shehata, A. A., Dick, K., Hajri, H., Lasram, S., Yilmaz, M., Emekci, M., Tsiamis, G., Spona-Friedl, M., May-Simera, H., Eisenreich, W., & Ntougias, S. (2022). Status and Prospects of Botanical Biopesticides in Europe and Mediterranean Countries. Biomolecules, 12(2), 311. https://doi.org/10.3390/biom12020311










