Further Findings Concerning Endothelial Damage in COVID-19 Patients
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Immunoassays
2.3. Statistical Analysis
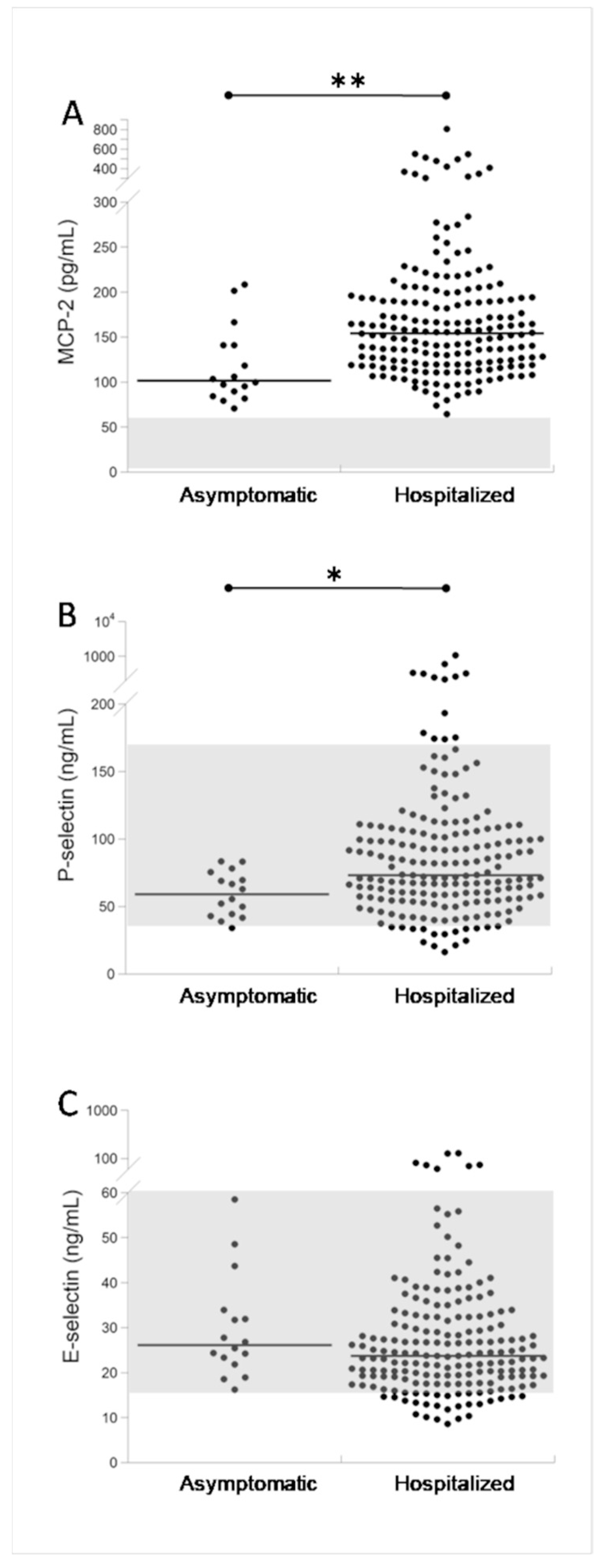
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Canzano, P.; Brambilla, M.; Porro, B.; Cosentino, N.; Tortorici, E.; Vicini, S.; Poggio, P.; Cascella, A.; Pengo, M.F.; Veglia, F.; et al. Platelet and Endothelial Activation as Potential Mechanisms Behind the Thrombotic Complications of COVID-19 Patients. JACC Basic Transl. Sci. 2021, 6, 202–218. [Google Scholar] [CrossRef]
- Jafarzadeh, A.; Chauhan, P.; Saha, B.; Jafarzadeh, S.; Nemati, M. Contribution of Monocytes and Macrophages to the Local Tissue Inflammation and Cytokine Storm in COVID-19: Lessons from SARS and MERS, and Potential Therapeutic Interventions. Life Sci. 2020, 257, 118102. [Google Scholar] [CrossRef]
- Poissy, J.; Goutay, J.; Caplan, M.; Parmentier, E.; Duburcq, T.; Lassalle, F.; Jeanpierre, E.; Rauch, A.; Labreuche, J.; Susen, S.; et al. Pulmonary Embolism in Patients with COVID-19: Awareness of an increased prevalence. Circulation 2020, 142, 184–186. [Google Scholar] [CrossRef]
- Comer, S.P.; Cullivan, S.; Szklanna, P.B.; Weiss, L.; Cullen, S.; Kelliher, S.; Smolenski, A.; Murphy, C.; Altaie, H.; Curran, J.; et al. COVID-19 Induces a Hyperactive Phenotype in Circulating Platelets. PLoS Biol. 2021, 19, e3001109. [Google Scholar] [CrossRef]
- Jamilloux, Y.; Henry, T.; Belot, A.; Viel, S.; Fauter, M.; El Jammal, T.; Walzer, T.; François, B.; Sève, P. Should We Stimulate or Suppress Immune Responses in COVID-19? Cytokine and anti-cytokine interventions. Autoimmun. Rev. 2020, 19, 102567. [Google Scholar] [CrossRef]
- Varga, Z.; Flammer, A.J.; Streiger, P.; Haberecker, M.; Andermatt, R.; Zinkernagel, A.S.; Mehra, M.R.; Schuepbach, R.A.; Ruschitzka, F.; Moch, H. Endothelial Cell Infection and Endothelitis in COVID-19. Lancet 2020, 395, 1417–1418. [Google Scholar] [CrossRef]
- E Fox, S.; Akmatbekov, A.; Harbert, J.L.; Li, G.; Brown, J.Q.; Heide, R.S.V. Pulmonary and Cardiac Pathology in African American Patients with COVID-19: An Autopsy Series from New Orleans. Lancet Respir. Med. 2020, 8, 681–686. [Google Scholar] [CrossRef]
- Ackermann, M.; Verleden, S.; Kuehnel, M.; Haverich, A.; Welte, T.; Laenger, F.; Vanstapel, A.; Werlein, C.; Stark, H.; Tzankov, A.; et al. Pulmonary Vascular Endothelialitis, Thrombosis, and Angiogenesis in Covid-19. N. Engl. J. Med. 2020, 383, 120–128. [Google Scholar] [CrossRef] [PubMed]
- Gelzo, M.; Cacciapuoti, S.; Pinchera, B.; De Rosa, A.; Cernera, G.; Scialò, F.; Mormile, M.; Fabbrocini, G.; Parrella, R.; Gentile, I.; et al. Prognostic Role of Neutrophil to Lymphocyte Ratio in COVID-19 Patients: Still Valid in Patients That Had Started Therapy? Front. Public Heal. 2021, 9, 664108. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Chen, L.; Dang, W.Q.; Cao, M.F.; Xiao, J.F.; Lv, S.Q.; Jiang, W.J.; Yao, X.H.; Lu, H.M.; Miao, J.Y.; et al. CCL8 secreted by Tumor-Associated Macrophages Promotes Invasion and Stemness of Glioblastoma Cells Via ERK1/2 Signaling. Lab. Invest. 2020, 100, 619–629. [Google Scholar] [CrossRef]
- Asano, K.; Takahashi, N.; Ushiki, M.; Monya, M.; Aihara, F.; Kuboki, E.; Moriyama, S.; Iida, M.; Kitamura, H.; Qiu, C.-H.; et al. Intestinal CD169+ Macrophages Initiate Mucosal Inflammation by Secreting CCL8 that Recruits Inflammatory Monocytes. Nat. Commun. 2015, 6, 7802. [Google Scholar] [CrossRef]
- Cheung, C.Y.; Poon, L.L.; Ng, I.H.; Luk, W.; Sia, S.F.; Wu, M.H.; Chan, K.H.; Yuen, K.Y.; Gordon, S.; Guan, Y.; et al. Cyto-kine Responses in Severe Acute Respiratory Syndrome Coronavirus-Infected Macrophages In Vitro: Possible Relevance To patho-Genesis. J. Virol. 2005, 79, 7819–7826. [Google Scholar] [CrossRef]
- Chevrier, S.; Zurbuchen, Y.; Cervia, C.; Adamo, S.; Raeber, M.E.; de Souza, N.; Sivapatham, S.; Jacobs, A.; Bachli, E.; Rudiger, A.; et al. A Distinct Innate Immune Signature Marks Progression from Mild to Severe COVID-19. Cell Rep. Med. 2020, 2, 100166. [Google Scholar] [CrossRef] [PubMed]
- Barthel, S.R.; Gavino, J.D.; Descheny, L.; Dimitroff, C.J. Targeting Selectins and Selectin Ligands in Inflammation and Cancer. Expert Opin. Ther. Targets 2007, 11, 1473–1491. [Google Scholar] [CrossRef]
- Guo, L.; Sun, G.; Wang, G.; Ning, W.; Zhao, K. Soluble P-selectin Promotes Acute Myocardial Infarction Onset But Not Severity. Mol. Med. Rep. 2014, 11, 2027–2033. [Google Scholar] [CrossRef]
- Panicker, S.R.; Mehta-D’souza, P.; Zhang, N.; Klopocki, A.G.; Shao, B.; McEver, R.P. Circulating Soluble P-selectin Must Di-merize to Promote Inflammation and Coagulation in Mice. Blood 2017, 130, 181–191. [Google Scholar] [CrossRef]
- Grobler, C.; Maphumulo, S.; Grobbelaar, L.; Bredenkamp, J.; Laubscher, G.; Lourens, P.; Steenkamp, J.; Kell, D.; Pretorius, E. Covid-19: The Rollercoaster of Fibrin(Ogen), D-Dimer, Von Willebrand Factor, P-Selectin and Their Interactions with Endothelial Cells, Platelets and Erythrocytes. Int. J. Mol. Sci. 2020, 21, 5168. [Google Scholar] [CrossRef]
- Binder, F.P.C.; Ernst, B. E- and P-selectin: Differences, Similarities and Implications for the Design of P-selectin Antagonists. Chim. Int. J. Chem. 2011, 65, 210–213. [Google Scholar] [CrossRef] [PubMed]
- Karsli, E.; Sabirli, R.; Altintas, E.; Canacik, O.; Sabirli, G.T.; Kaymaz, B.; Özgür, K.; Koseler, A. Soluble P-selectin as a Potential Diagnostic and Prognostic Biomarker for COVID-19 Disease: A Case-Control Study. Life Sci. 2021, 277, 119634. [Google Scholar] [CrossRef] [PubMed]
- Venter, C.; Bezuidenhout, J.; Laubscher, G.; Lourens, P.; Steenkamp, J.; Kell, D.; Pretorius, E. Erythrocyte, Platelet, Serum Ferritin, and P-Selectin Pathophysiology Implicated In Severe Hypercoagulation and Vascular Complications in COVID-19. Int. J. Mol. Sci. 2020, 21, 8234. [Google Scholar] [CrossRef]
- Roldán, V.; Marín, F.; Lip, G.Y.H.; Blann, A.D. Soluble E-selectin in Cardiovascular Disease and Its Risk Factors. Thromb. Haemost. 2003, 90, 1007–1020. [Google Scholar] [CrossRef] [PubMed]
- Smadja, D.M.; Guerin, C.L.; Chocron, R.; Yatim, N.; Boussier, J.; Gendron, N.; Khider, L.; Hadjadj, J.; Goudot, G.; Debuc, B.; et al. Angiopoietin-2 as a Marker of Endothelial Activation Is a Good Predictor Factor for Intensive Care Unit Admission of COVID-19 Patients. Angiogenesis 2020, 23, 611–620. [Google Scholar] [CrossRef] [PubMed]
- Blann, A.D.; Taberner, D.A. A Reliable Marker of Endothelial Cell Dysfunction: Does It Exist? Br. J. Haematol. 1995, 90, 244–248. [Google Scholar] [CrossRef]
- Scavone, C.; Mascolo, A.; Rafaniello, C.; Sportiello, L.; Trama, U.; Zoccoli, A.; Bernardi, F.F.; Racagni, G.; Berrino, L.; Castaldo, G.; et al. Therapeutic Strategies to Fight COVID-19: Which Is the Status Artis? Br. J. Pharmacol. 2021. [Google Scholar] [CrossRef]
- Zollo, M.; Ferrucci, V.; Izzo, B.; Quarantelli, F.; Di Domenico, C.; Comegna, M.; Paolillo, C.; Amato, F.; Siciliano, R.; Castaldo, G.; et al. SARS-CoV_Subgenomic N (sgN) Transcripts in Oro-nasopharingeal Swabs Correlate With the Highest Viral Load, as evaluated by five different methods. Diagnostics 2021, 11, 288. [Google Scholar] [CrossRef]
- Cacciapuoti, S.; De Rosa, A.; Gelzo, M.; Megna, M.; Raia, M.; Pinchera, B.; Pontarelli, A.; Scotto, R.; Scala, E.; Scarano, F.; et al. Immunocytometric Analysis of COVID Patients: A Contribution to Personalized Therapy? Life Sci. 2020, 261, 118355. [Google Scholar] [CrossRef]
- von Cube, M.; Grodd, M.; Wolkewitz, M.; Hazard, D.; Wengenmayer, T.; Canet, E.; Lambert, J. Harmonizing Heterogeneous Endpoints in Coronavirus Disease 2019 Trials Without Loss of Information. Crit. Care Med. 2021, 49, e11–e19. [Google Scholar] [CrossRef]
- WHO Working Group on the Clinical Characterization and Management of COVID-19 Infection. A minimal Common Out-come Measure Set for COVID-19 Clinical Research. Lancet Infect. Dis. 2020, 20, e192–e197. [Google Scholar] [CrossRef]
- Ponthieux, A.; Herbeth, B.; Droesch, S.; Lambert, D.; Visvikis, S. Age- and Sex-related Reference Values for Serum Adhesion Molecule Concentrations in Healthy Individuals: Intercellular Adhesion Molecule-1 and E-, P-, and L-Selectin. Clin. Chem. 2003, 49, 1544–1546. [Google Scholar] [CrossRef][Green Version]
- Elmoselhi, H.; Mansell, H.; Soliman, M.; Shoker, A. Circulating Chemokine Ligand Levels Before and After Successful Kidney Transplantation. J. Inflamm. 2016, 13, 1–9. [Google Scholar] [CrossRef]
- Van Damme, J.; Proost, P.; Lenaerts, J.P.; Opdenakker, G. Structural and Functional Identification of Two Human, Tumor-Derived Monocyte Chemotactic Proteins (MCP-2 and MCP-3) Belonging to the Chemokine Family. J. Exp. Med. 1992, 176, 59–65. [Google Scholar] [CrossRef]
- Lu, L.; Zhang, H.; Dauphars, D.J.; He, Y.-W. A Potential Role of Interleukin 10 in COVID-19 Pathogenesis. Trends Immunol. 2021, 42, 3–5. [Google Scholar] [CrossRef]
- Zhao, Y.; Quin, L.; Zhang, P.; Li, K.; Liang, L.; Sun, J.; Xu, B.; Dai, Y.; Li, X.; Zhang, C.; et al. Longitudinal COVID-19 Profil-ing Associates IL-1RA and IL-10 with Disease Severity and RANTES with Mild Disease. JCI Insight 2020, 5, e139834. [Google Scholar] [CrossRef]
- Han, H.; Ma, Q.; Li, C.; Liu, R.; Zhao, L.; Wang, W.; Zhang, P.; Liu, X.; Gao, G.; Liu, F.; et al. Profiling Serum Cytokines in COVID-19 Patients Reveals IL-6 and IL-10 are Disease Severity Predictors. Emerg. Microbes Infect. 2020, 9, 1123–1130. [Google Scholar] [CrossRef]
- Goshua, G.; Pine, A.B.; Meizlish, M.L.; Chang, C.-H.; Zhang, H.; Bahel, P.; Baluha, A.; Bar, N.; Bona, R.D.; Burns, A.J.; et al. Endotheliopathy in COVID-19-Associated Coagulopathy: Evidence from a Single-Centre, Cross-sectional Study. Lancet Haematol. 2020, 7, e575–e582. [Google Scholar] [CrossRef]
- Neri, T.; Nieri, D.; Celi, A. P-selectin Blockade in COVID-19-Related ARDS. Am. J. Physiol. Cell. Mol. Physiol. 2020, 318, L1237–L1238. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Xiang, D.; Gao, F.; Yao, H.; Ye, Q.; Wang, Y. The inhibition of P-selectin Reduced Severe Acute Lung Injury in Immun-ocompromised Mice. Oxid. Med. Cell Longev. 2020, 23, 8430465. [Google Scholar]
- Littler, A.J.; Buckley, C.D.; Wordsworth, P.; Collins, I.; Martinson, J.; Simmons, D.L. A distinct Profile of Six Soluble Adhesion Molecules (ICAM-1, ICAM-3, VCAM-1, E-selectin, L-selectin and P-selectin) in rheumatoid arthritis. Br. J. Rheumatol. 1997, 36, 164–169. [Google Scholar] [CrossRef] [PubMed]
| WHO 3 | WHO 4 | WHO 5–7 | Kruskal–Wallis | |
|---|---|---|---|---|
| N | 64 | 78 | 46 | - |
| Females, N (%) | 48 (75) | 27 (35) a | 11 (24) a | - |
| Age | 35.5 (29.0–52.3) | 55.5 (41.0–67.0) a | 57.5 (48.0–75.8) a | <0.0001 |
| MCP-2 (pg/mL) | 133 (112–167) | 158 (123–206) | 172 (141–201) a | 0.001 |
| P-selectin (ng/mL) | 82.3 (54.3–109) | 72.7 (58.3–100) | 64.5 (49.2–108) | n.s. |
| E-selectin (ng/mL) | 23.6 (17.5–30.5) | 22.3 (19.0–32.4) | 25.4 (18.7–32.8) | n.s. |
| Platelets (MN/mmc) | 231 (201–301) | 251 (206–361) | 224 (155–257) | n.s. |
| Smokers, N (%) | 11 (17) | 13 (17) | 3 (7) b | - |
| CAD, N (%) | 10 (16) | 5 (7) | 6 (13) | - |
| Diabetes, N (%) | 9 (14) | 8 (10) | 12 (27) c | - |
| CKD, N (%) | none | 5 (7) a | 9 (20) a | - |
| WHO 3 (n = 18) | WHO 4 (n = 24) | WHO 5–7 (n = 10) | |
|---|---|---|---|
| MCP-2 (pg/mL) | |||
| Basal | 142 (111–204) | 169 (122–263) | 151 (125–175) |
| After 1 week | 132 (101–160) | 124 (96–158) | 105 (76.7–128) |
| p-value a | 0.035 | <0.0001 | 0.017 |
| P-selectin (ng/mL) | |||
| Basal | 93.8 (66.7–124) | 98.9 (74.7–135) | 109 (74.6–192) |
| After 1 week | 130 (95.1–171) | 102 (95.4–127) | 72.5 (57.4–165) |
| p-value a | 0.003 | n.s. | n.s. |
| E-selectin (ng/mL) | |||
| Basal | 28.9 (22.6–40.7) | 23.8 (19.0–38.4) | 26.3 (20.0–30.0) |
| After 1 week | 26.2 (21.9–36.5) | 24.1 (17.5–39.7) | 20.9 (13.8–25.1) |
| p-value a | n.s. | n.s. | n.s. |
| Platelets (MN/mmc) | |||
| Basal | 248 (193–292) | 263 (207–395) | 168 (119–259) |
| After 1 week | 339 (251–446) | 319 (206–370) | 229 (155–273) |
| p-value a | 0.003 | n.s. | n.s. |
| MCP-2 (pg/mL) | P-Selectin (ng/mL) | E-Selectin (ng/mL) | ||||
|---|---|---|---|---|---|---|
| rs | p-Value | rs | p-Value | rs | p-Value | |
| MCP-2 (pg/mL) | - | - | 0.183 | 0.012 | 0.271 | <0.0001 |
| P-selectin (ng/mL) | 0.183 | 0.012 | - | - | 0.145 | 0.047 |
| E-selectin (ng/mL) | 0.271 | <0.001 | 0.145 | 0.047 | - | - |
| IL-6 (pg/mL) | 0.065 | 0.377 | 0.278 | <0.0001 | 0.340 | <0.0001 |
| IL-17A (pg/mL) | −0.099 | 0.330 | −0.077 | 0.452 | −0.051 | 0.616 |
| IL-10 (pg/mL) | 0.203 | 0.006 | 0.463 | <0.0001 | 0.347 | <0.0001 |
| Neutrophils (N/mmc) | 0.004 | 0.954 | −0.012 | 0.867 | 0.046 | 0.527 |
| Monocytes (N/mmc) | 0.113 | 0.121 | 0.082 | 0.262 | 0.099 | 0.178 |
| Platelets (N/mmc) | −0.039 | 0.691 | 0.340 | <0.0001 | −0.157 | 0.108 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gelzo, M.; Cacciapuoti, S.; Pinchera, B.; De Rosa, A.; Cernera, G.; Scialò, F.; Comegna, M.; Mormile, M.; Fabbrocini, G.; Parrella, R.; et al. Further Findings Concerning Endothelial Damage in COVID-19 Patients. Biomolecules 2021, 11, 1368. https://doi.org/10.3390/biom11091368
Gelzo M, Cacciapuoti S, Pinchera B, De Rosa A, Cernera G, Scialò F, Comegna M, Mormile M, Fabbrocini G, Parrella R, et al. Further Findings Concerning Endothelial Damage in COVID-19 Patients. Biomolecules. 2021; 11(9):1368. https://doi.org/10.3390/biom11091368
Chicago/Turabian StyleGelzo, Monica, Sara Cacciapuoti, Biagio Pinchera, Annunziata De Rosa, Gustavo Cernera, Filippo Scialò, Marika Comegna, Mauro Mormile, Gabriella Fabbrocini, Roberto Parrella, and et al. 2021. "Further Findings Concerning Endothelial Damage in COVID-19 Patients" Biomolecules 11, no. 9: 1368. https://doi.org/10.3390/biom11091368
APA StyleGelzo, M., Cacciapuoti, S., Pinchera, B., De Rosa, A., Cernera, G., Scialò, F., Comegna, M., Mormile, M., Fabbrocini, G., Parrella, R., Corso, G., Gentile, I., & Castaldo, G. (2021). Further Findings Concerning Endothelial Damage in COVID-19 Patients. Biomolecules, 11(9), 1368. https://doi.org/10.3390/biom11091368








