Evaluation of Immune Dysregulation in an Austrian Patient Cohort Suffering from Myalgic Encephalomyelitis/Chronic Fatigue Syndrome
Abstract
:1. Introduction
2. Materials and Methods
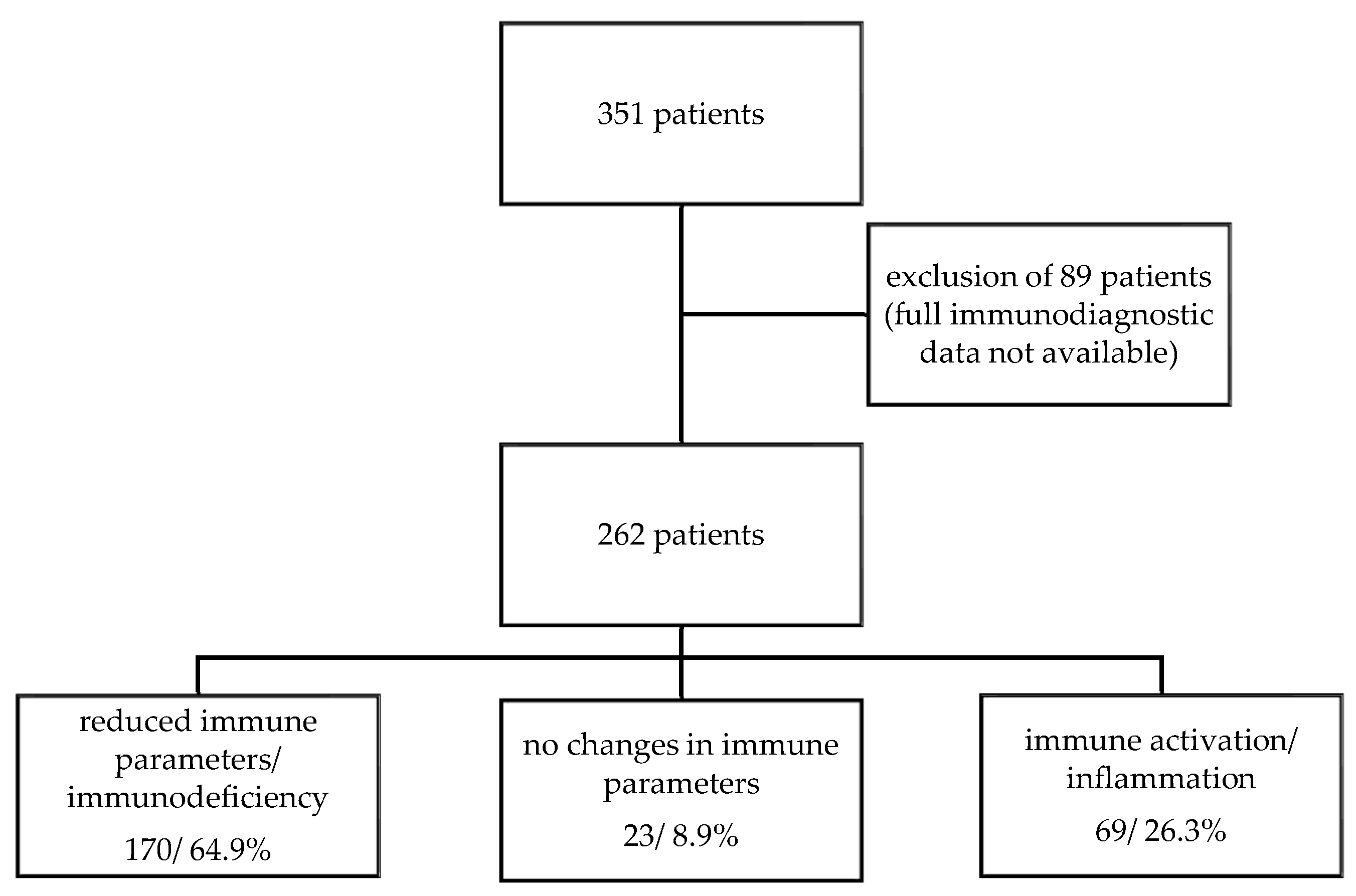
3. Results
3.1. Demographic Patient Data
3.2. Immune Parameters in ME/CFS Patients
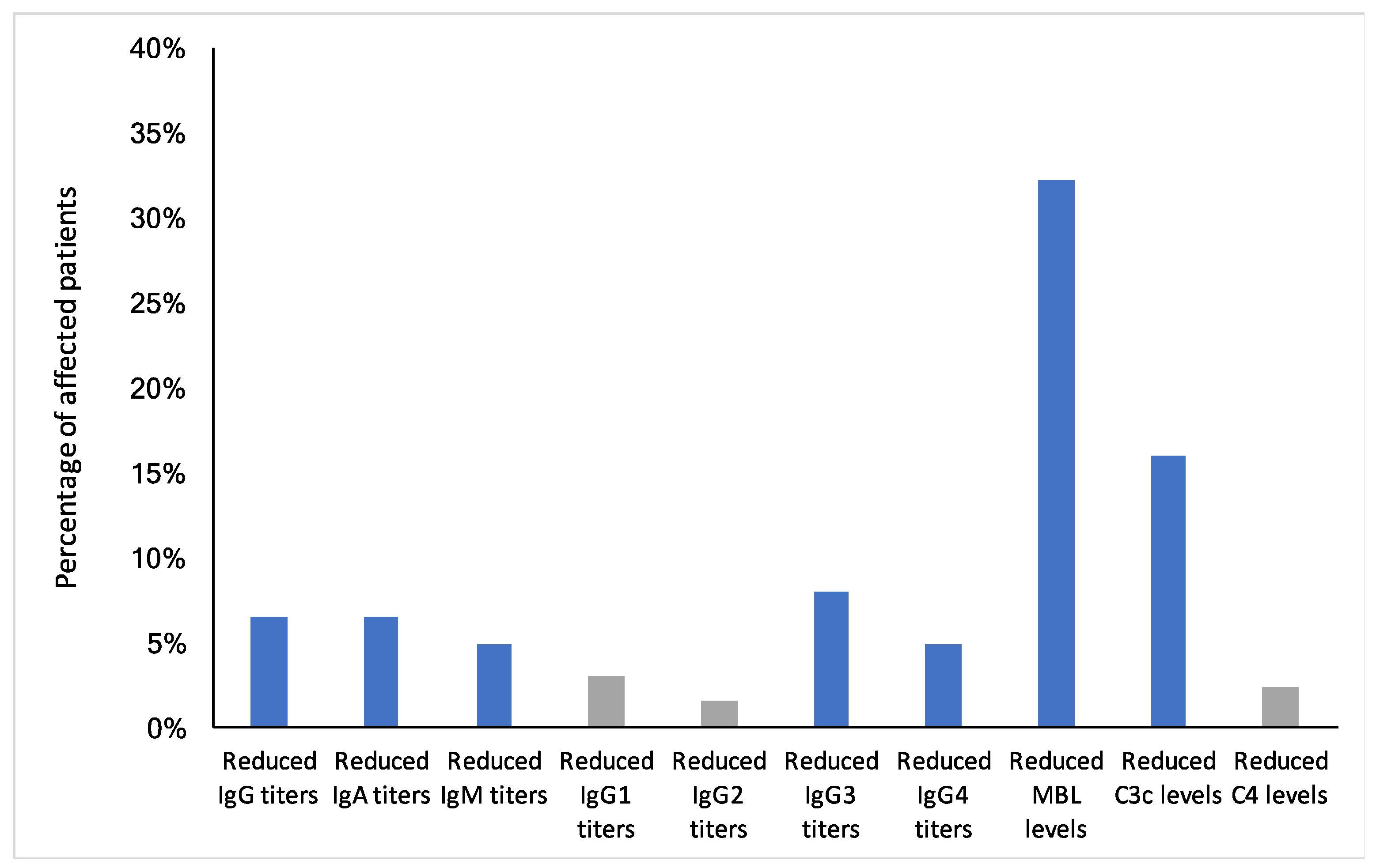
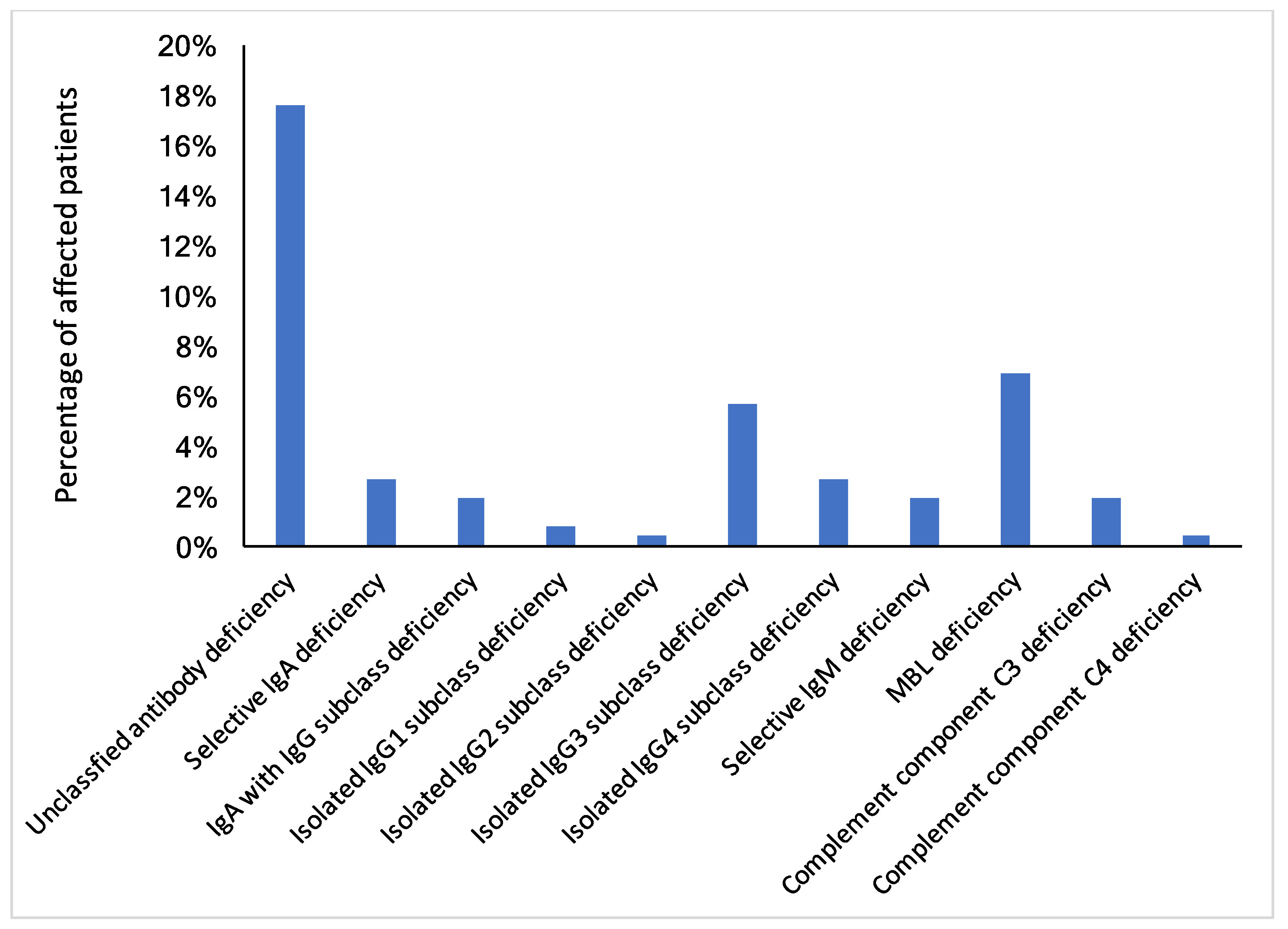
3.3. Reduced Humoral Immune Response in ME/CFS Patients
3.4. Combination of Reduced Cellular and Humoral Immune Response in ME/CFS Patients
3.5. Elevated Immune Parameters in ME/CFS Patients
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lim, E.-J.; Ahn, Y.-C.; Jang, E.-S.; Lee, S.-W.; Lee, S.-H.; Son, C.-G. Systematic review and meta-analysis of the prevalence of chronic fatigue syndrome/myalgic encephalomyelitis (CFS/ME). J. Transl. Med. 2020, 18, 100. [Google Scholar] [CrossRef] [PubMed]
- CFS Hilfe Austria. Available online: https://cfs-hilfe.at (accessed on 2 May 2021).
- Suttorp, N.; Möckel, M.; Siegmund, B.; Dietel, M. Chronisches Fatigue Syndrome, in Harrisons Innere Medizin; ABW Wissenschaftsverlagsgesellschaft: Berlin, Germany, 2016; p. 464. [Google Scholar]
- Pertl, D.; Stürzlinger, H. Chronic Fatigue Syndrom—Chronisches Erschöpfungssyndrom. In Kurzrecherche: Im Auftrag des Bundesministeriums für Gesundheit; Gesundheit Österreich GmbH: Vienna, Austria, 2015. [Google Scholar]
- Bested, C.A.; Marshall, L.M. Review of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: An evidence-based approach to diagnosis and management by clinicians. Rev. Environ. Health 2015, 30, 223–249. [Google Scholar] [CrossRef] [PubMed]
- Hausotter, W. Medical certification of chronic fatigue syndrome. Versicherungsmedizin 2015, 67, 13–18. [Google Scholar]
- Sandler, X.C.; Lloyd, A.R. Chronic fatigue syndrome: Progress and possibilities. Med. J. Aust. 2020, 212, 428–433. [Google Scholar] [CrossRef]
- Wood, E.; Hall, K.H.; Tate, W. Role of mitochondria, oxidative stress and the response to antioxidants in myalgic encephalomyelitis/chronic fatigue syndrome: A possible approach to SARS-CoV-2 ‘long-haulers’? Chronic Dis. Transl. Med. 2021, 7, 14–26. [Google Scholar] [PubMed]
- Sotzny, F.; Blanco, J.; Capelli, E.; Castro-Marrero, J.; Steiner, S.; Murovska, M.; Scheibenbogen, C. Myalgic Encephalomyelitis/Chronic Fatigue Syndrome—Evidence for an autoimmune disease. Autoimmun. Rev. 2018, 17, 601–609. [Google Scholar] [CrossRef]
- Underhill, R.A. Myalgic encephalomyelitis, chronic fatigue syndrome: An infectious disease. Med. Hypotheses 2015, 85, 765–773. [Google Scholar] [CrossRef]
- Rasa, S.; Nora-Krukle, Z.; Henning, N.; Eliassen, E.; Shikova, E.; Harrer, T.; Scheibenbogen, C.; Murovska, M.; Prusty, B.K. Chronic viral infections in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS). J. Transl. Med. 2018, 16, 268. [Google Scholar] [CrossRef] [Green Version]
- Blomberg, J.; Rizwan, M.; Böhlin-Wiener, A.; Elfaitouri, A.; Julin, P.; Zachrisson, O.; Rosén, A.; Gottfries, C.-G. Antibodies to Human Herpesviruses in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome Patients. Front. Immunol. 2019, 10, 1946. [Google Scholar] [CrossRef] [Green Version]
- Arnett, S.; Alleva, L.; Korossy-Horwood, R.; Clark, I. Chronic fatigue syndrome—A neuroimmunological model. Med. Hypotheses 2011, 77, 77–83. [Google Scholar] [CrossRef]
- Morris, G.; Maes, M.; Berk, M.; Puri, B.K. Myalgic encephalomyelitis or chronic fatigue syndrome: How could the illness develop? Metab. Brain Dis. 2019, 34, 385–415. [Google Scholar] [CrossRef] [Green Version]
- Skowera, A.; Cleare, A.; Blair, D.; Bevis, L.; Wessely, S.C.; Peakman, M. High levels of type 2 cytokine-producing cells in chronic fatigue syndrome. Clin. Exp. Immunol. 2004, 135, 294–302. [Google Scholar] [CrossRef]
- Guenther, S.; Loebel, M.; Mooslechner, A.A.; Knops, M.; Hanitsch, L.G.; Grabowski, P.; Wittke, K.; Meisel, C.; Unterwalder, N.; Volk, H.-D.; et al. Frequent IgG subclass and mannose binding lectin deficiency in patients with chronic fatigue syndrome. Hum. Immunol. 2015, 76, 729–735. [Google Scholar] [CrossRef] [PubMed]
- Wirth, K.; Scheibenbogen, C. A Unifying Hypothesis of the Pathophysiology of Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS): Recognitions from the finding of autoantibodies against ss2-adrenergic receptors. Autoimmun. Rev. 2020, 19, 102527. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. IOM 2015 Diagnostic Criteria. 2021. Available online: https://www.cdc.gov/me-cfs/healthcare-providers/diagnosis/iom-2015-diagnostic-criteria.html (accessed on 27 April 2021).
- European Society for Immunodeficiencies (ESID). 2021. Available online: https://esid.org/Working-Parties/Registry-Working-Party/Diagnosis-criteria (accessed on 27 April 2021).
- Centers for Disease Control and Prevention. Epstein-Barr Virus and Infectious Mononucleosis. 2020. Available online: https://www.cdc.gov/epstein-barr/laboratory-testing.html (accessed on 28 September 2020).
- Fourcade, G.; Germi, R.; Guerber, F.; Lupo, J.; Baccard, M.; Seigneurin, A.; Semenova, T.; Morand, P.; Epaulard, O. Evolution of EBV seroprevalence and primary infection age in a French hospital and a city laboratory network, 2000–2016. PLoS ONE 2017, 12, e0175574. [Google Scholar] [CrossRef] [PubMed]
- Pottgießer, T.; Bauer, G. Epstein-Barr-Virus-Infektionen. Dtsch. Z. Sportmed. 2014, 64, 337–341. [Google Scholar]
- De Paschale, M.; Clerici, P. Serological diagnosis of Epstein-Barr virus infection: Problems and solutions. World J. Virol. 2012, 1, 31–43. [Google Scholar] [CrossRef]
- Yancey, J.R.; Thomas, S.M. Chronic fatigue syndrome: Diagnosis and treatment. Am. Fam. Physician 2012, 86, 741–746. [Google Scholar] [PubMed]
- Wakefield, D.; Lloyd, A.; Brockman, A. Immunoglobulin subclass abnormalities in patients with chronic fatigue syndrome. Pediatr. Infect. Dis. J. 1990, 9 (Suppl. S8), S50–S53. [Google Scholar] [CrossRef]
- Peterson, P.K.; Shepard, J.; Macres, M.; Schenck, C.; Crosson, J.; Rechtman, D.; Lurie, N. A controlled trial of intravenous immunoglobulin G in chronic fatigue syndrome. Am. J. Med. 1990, 89, 554–560. [Google Scholar] [CrossRef]
- Ballow, M. Primary Immunodeficiency Diseases. In Goldman’s Cecil Medicine, 24th ed.; Goldman, L., Schafer, A.I., Eds.; Elsevier Saunders: Philadelphia, PA, USA, 2012; pp. 1615–1622. [Google Scholar]
- Hammarstrom, L.; Vorechovsky, I.; Webster, D. Selective IgA deficiency (SIgAD) and common variable immunodeficiency (CVID). Clin. Exp. Immunol. 2000, 120, 225–231. [Google Scholar] [CrossRef]
- Loebel, M.; Grabowski, P.; Heidecke, H.; Bauer, S.; Hanitsch, L.G.; Wittke, K.; Meisel, C.; Reinke, P.; Volk, H.-D.; Fluge, Ø.; et al. Antibodies to beta adrenergic and muscarinic cholinergic receptors in patients with Chronic Fatigue Syndrome. Brain Behav. Immun. 2016, 52, 32–39. [Google Scholar] [CrossRef] [Green Version]
- Cakalaroski, K.; Ivanovski, N.; Popov, Z.; Dohcev, S.; Kolevski, P.; Weil, B.; Lang, P. Mono- and oligoclonal immunoglobulin anomalies in kidney transplant patients. Ann. Urol. 2000, 34, 340–344. [Google Scholar]
- Vilibic-Cavlek, T.; Ljubin-Sternak, S.; Kos, L.; Mlinaric-Galinovic, G. The role of IgG avidity determination in diagnosis of Epstein-Barr virus infection in immunocompetent and immunocompromised patients. Acta Microbiol. Immunol. Hung. 2011, 58, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Lerner, A.M.; Beqaj, S.H.; Deeter, R.G.; Fitzgerald, J.T. IgM serum antibodies to Epstein-Barr virus are uniquely present in a subset of patients with the chronic fatigue syndrome. Vivo 2004, 18, 101–106. [Google Scholar]
- Zhang, H.; Li, P.; Wu, D.; Xu, D.; Hou, Y.; Wang, Q.; Li, M.; Li, Y.; Zeng, X.; Zhang, F.; et al. Serum IgG subclasses in autoimmune diseases. Medicine 2015, 94, e387. [Google Scholar] [CrossRef] [PubMed]
- Scheibenbogen, C.; Loebel, M.; Freitag, H.; Krueger, A.; Bauer, S.; Antelmann, M.; Doehner, W.; Scherbakov, N.; Heidecke, H.; Reinke, P.; et al. Immunoadsorption to remove ss2 adrenergic receptor antibodies in Chronic Fatigue Syndrome CFS/ME. PLoS ONE 2018, 13, e0193672. [Google Scholar]
- Sorensen, B.; Streib, J.E.; Strand, M.; Make, B.; Giclas, P.C.; Fleshner, M.; Jones, J.F. Complement activation in a model of chronic fatigue syndrome. J. Allergy Clin. Immunol. 2003, 112, 397–403. [Google Scholar] [CrossRef] [PubMed]
- Torres-Harding, S.; Sorenson, M.; Jason, L.A.; Maher, K.; Fletcher, M.A. Evidence for T-helper 2 shift and association with illness parameters in chronic fatigue syndrome (CFS). Bull. IACFS ME 2008, 16, 19–33. [Google Scholar]
- Brenu, E.W.; Huth, T.K.; Hardcastle, S.L.; Fuller, K.; Kaur, M.; Johnston, S.; Ramos, S.B.; Staines, D.R.; Marshall-Gradisnik, S.M. Role of adaptive and innate immune cells in chronic fatigue syndrome/myalgic encephalomyelitis. Int. Immunol. 2014, 26, 233–242. [Google Scholar] [CrossRef]
- Cosmi, L.; Maggi, L.; Santarlasci, V.; Liotta, F.; Annunziato, F. T helper cells plasticity in inflammation. Cytom. A 2014, 85, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Tirelli, U.; Marotta, G.; Improta, S.; Pinto, A. Immunological Abnormalities in Patients with Chronic Fatigue Syndrome. Scand. J. Immunol. 1994, 40, 601–608. [Google Scholar] [CrossRef]
| Parameter | Absolute Frequency | Mean | Range |
|---|---|---|---|
| Sex (male/female) | 83/179 | ||
| Age (years) | 41 | 18–79 | |
| Age groups (18–40 years/41–80 years) | 131/131 | ||
| Fulfilling of all IOM-criteria for the diagnosis ME/CFS | 207 | ||
| Duration of disease until immunodiagnostics (years) | 9.4 | 1–39 | |
| Patients with recurrent infections | 158 | ||
| Patients diagnosed with acute EBV infection at onset of fatigue | 45 | ||
| positive EBV antibodies | 194 |
| Humoral Parameter | Cellular Parameter | No. of Patients (95%CI) (n = 262) |
|---|---|---|
| CD4+ T-cell lymphopenia | 5 (2–12) | |
| reduced CD8-CD57+ NK cell counts | 6 (2–13) | |
| Reduction of at least one of total IgG, IgG1, IgG2, IgG3, IgA or IgM levels in combination with… | reduced CD3+CD16+CD56+ NKT cell counts | 0 (0–4) |
| reduced CD3-CD16+CD56+ NK cell counts | 8 (3–16) | |
| CD4+ T-cell lymphopenia | 4 (1–10) | |
| CD8+ T-cell lymphopenia | 3 (1–9) | |
| Unclassified antibody deficiency in combination with… | reduced CD8-CD57+ NK cell counts | 4 (1–10) |
| reduced CD3+CD16+CD56+ NKT cell counts | 0 (0–4) | |
| reduced CD3-CD16+CD56+ NK cell counts | 6 (2–13) | |
| CD4+ T-cell lymphopenia | 4 (1–10) |
| Parameter | No. of Patients (95%CI) (n = 262) |
|---|---|
| CD4+ T-cells | 12 (6–21) |
| CD8+ T-cells | 4 (1–10) |
| CD8-CD57+ NK cell counts | 23 (15–34) |
| CD3+CD16+CD56+ NKT cell counts | 7 (3–14) |
| CD3-CD16+CD56+ NK cell counts | 4 (1–10) |
| IgG | 4 (1–10) |
| IgA | 4 (1–10) |
| IgM | 8 (3–16) |
| IgG1 | 5 (2–12) |
| IgG2 | 13 (7–22) |
| IgG3 | 3 (1–9) |
| IgG4 | 5 (2–12) |
| C3 | 1 (0–6) |
| C4 | 3 (1–9) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lutz, L.; Rohrhofer, J.; Zehetmayer, S.; Stingl, M.; Untersmayr, E. Evaluation of Immune Dysregulation in an Austrian Patient Cohort Suffering from Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. Biomolecules 2021, 11, 1359. https://doi.org/10.3390/biom11091359
Lutz L, Rohrhofer J, Zehetmayer S, Stingl M, Untersmayr E. Evaluation of Immune Dysregulation in an Austrian Patient Cohort Suffering from Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. Biomolecules. 2021; 11(9):1359. https://doi.org/10.3390/biom11091359
Chicago/Turabian StyleLutz, Lena, Johanna Rohrhofer, Sonja Zehetmayer, Michael Stingl, and Eva Untersmayr. 2021. "Evaluation of Immune Dysregulation in an Austrian Patient Cohort Suffering from Myalgic Encephalomyelitis/Chronic Fatigue Syndrome" Biomolecules 11, no. 9: 1359. https://doi.org/10.3390/biom11091359
APA StyleLutz, L., Rohrhofer, J., Zehetmayer, S., Stingl, M., & Untersmayr, E. (2021). Evaluation of Immune Dysregulation in an Austrian Patient Cohort Suffering from Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. Biomolecules, 11(9), 1359. https://doi.org/10.3390/biom11091359







