Combination of Stem Cells and Rehabilitation Therapies for Ischemic Stroke
Abstract
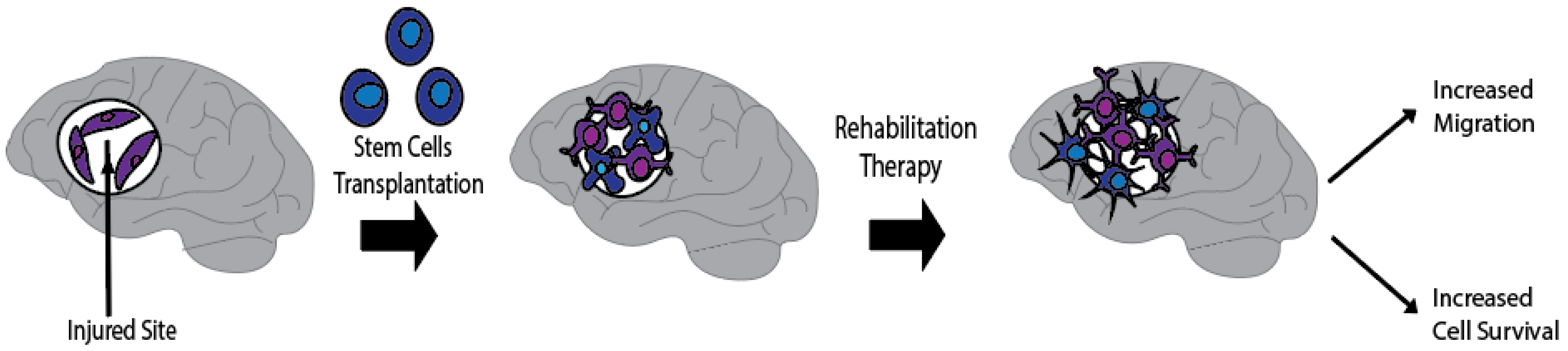
1. Introduction
1.1. Current Standing of Stem Cells as a Stand-Alone Therapeutic for Stroke
1.2. Exogenous Stem Cell Therapy for Stroke
1.2.1. Novel Preclinical Evidence
1.2.2. Various Exogenous Stem Cell Types Exhibit the Capacity to Recruit Endogenous Stem Cells
MSCs
NSCs
Other Cell Types
1.2.3. Stem Cell Source, Administration Route, Dose, and Timing for Stroke Treatment
1.2.4. Stems Cells and Stroke in the Aged Brain
1.2.5. Neuroprotective Effects of Exogenous Stem Cells
1.3. Endogenous Repair Mechanisms against Ischemic Stroke
1.3.1. Neurogenic Niches House Endogenous Cells
1.3.2. The Role of Neurogenesis and Angiogenesis in Stroke Recovery
1.3.3. White Matter Repair Mechanisms
1.3.4. Bystander Effect
1.4. Therapeutic Promise of Stem Cells
2. Remodeling of the Stroke Tissue Microenvironment within the Brain
2.1. Exogenous Stem Cell Remodeling
2.2. Endogenous Stem Cell Remodeling
2.3. Use of Biomaterials, Drugs, Photo- and Optogenetics
2.4. Novel Drug Therapies and Mechanism of Action
2.5. The Future of Therapeutic Approaches
3. Remodeling of Stroke Tissue Microenvironment outside the Brain
3.1. How Do Prominent Modalities of Stroke Rehabilitation Aid in Functional Recovery?
3.1.1. How Does Motor Rehabilitation Facilitate Functional Recovery?
3.1.2. How Does Functional Electrical Stimulation (FES) Aid in Functional Recovery?
3.1.3. How Does Functional Magnetic Stimulation Assist in Stroke Recovery?
3.1.4. How Does Biofeedback-Based Rehabilitation Training Aid Functional Recovery?
3.1.5. How Does Robot-Assisted Therapy Facilitate Functional Recovery?
3.1.6. How Does BCI Facilitate Functional Recovery?
3.1.7. How Does VR-Based Rehabilitation Improve Functional Recovery?
3.1.8. How Does Non-Motor Rehabilitation Assist in Functional Recovery?
3.2. How Do Stem Cells with Rehabilitation Offer an Additive or Synergistic Functional Recovery?
3.2.1. How Does Enriched Housing in Tandem with Stem Cells Enhance Functional Recovery?
3.2.2. How Does an EE with Stem Cells Work Together to Modulate the Microenvironment?
3.2.3. How Do Exercise and Stem Cells Elicit an Increased Therapeutic Effect When Used Together?
3.3. Combination Therapy Potential Benefits
4. Conclusions
Funding
Conflicts of Interest
Abbreviations
| Abbreviation | Meaning |
| tPA | tissue plasminogen activator |
| HSCs | hematopoietic stem cells |
| NSCs | neural stem cells |
| MSCs | mesenchymal stem cells |
| IPSCs | induced pluripotent stem cells |
| MAP2 | microtubule-associated protein 2 |
| ECM | extracellular matrix |
| GDNF | glial cell line-derived neurotrophic factor |
| EPCs | endothelial progenitor cells |
| USC-Exos | urine-derived stem cells |
| NPCs | neural progenitor cells |
| MMPs | matrix metalloproteinases |
| TBI | traumatic brain injury |
| SVZ | subventricular zone |
| IV | intravenous |
| OPCs | oligodendrocyte progenitor cells |
| BM-MSCs | bone marrow-derived mesenchymal stem cells |
| NVU | neurovascular unit |
| IGF-1 | insulin-like growth factor |
| VEGF | vascular endothelial growth factor |
| EGF | epidermal growth factor |
| bFGF | basic fibroblast growth factor |
| BMPs | bone morphogenic proteins |
| MCAO | middle cerebral artery occlusion |
| SDF-1 | stromal cell-derived factor-1 |
| CXC4 | chemokine receptor type 4 |
| IL | interleukin |
| PGE2 | prostaglandin E2 |
| FGF2 | fibroblast growth factor 2 |
| VEGFR2 | vascular endothelial growth factor receptor 2 |
| BDNF | brain-derived neurotrophic factor |
| PD | Parkinson’s disease |
| SGZ | subgranular zone |
| BBB | blood-brain barrier |
| SGZ | subgranular zone |
| PDGF | platelet-derived growth factor |
| OGD/R | oxygen glucose deprivation/re-oxygenation |
| IC | intracerebral |
| IA | intra-arterial |
| TNF-α | tumor necrosis factor-α |
| DAMPs | damage-associated molecular patterns |
| MCP-1 | monocyte chemoattractant protein-1 |
| TGF-β | transforming growth factor-β |
| NGF | nerve growth factor |
| PEI | polyethyleneimine |
| CCR4 | CC chemokine receptor 4 |
| ROS | reactive oxygen species |
| I/R | ischemia/reperfusion |
| CUECD2 | CUE domain-containing 2 |
| NPs | nanoparticles |
| IFN-γ | interferon-γ |
| GFAP | glial fibrillary acidic protein |
| EVs | extracellular vesicles |
| UC-MSCs | umbilical cord-mesenchymal stem cells |
| HIF-1α | hypoxia-inducible factor-1α |
| CTZ | coelenterazine |
| TSPO | translocator protein |
| EE | enriched environment |
| VR | virtual reality |
| BCI | brain-computer interfaces |
| FES | functional electrical stimulation |
| TMS | transcranial magnetic stimulation |
| NTF | neurofeedback therapy |
References
- Reeve, A.; Simcox, E.; Turnbull, D. Ageing and Parkinson’s disease: Why is advancing age the biggest risk factor? Ageing Res. Rev. 2014, 14, 19–30. [Google Scholar] [CrossRef]
- Wyss-Coray, T. Ageing, neurodegeneration and brain rejuvenation. Nature 2016, 539, 180–186. [Google Scholar] [CrossRef]
- Kuriakose, D.; Xiao, Z. Pathophysiology and Treatment of Stroke: Present Status and Future Perspectives. Int. J. Mol. Sci. 2020, 21, 7609. [Google Scholar] [CrossRef] [PubMed]
- Jauch, E.C.; Saver, J.L.; Adams, H.P., Jr.; Bruno, A.; Connors, J.J.; Demaerschalk, B.M.; Khatri, P.; McMullan, P.W., Jr.; Qureshi, A.I.; Rosenfield, K.; et al. Guidelines for the early management of patients with acute ischemic stroke: A guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2013, 44, 870–947. [Google Scholar] [CrossRef] [PubMed]
- Derex, L.; Cho, T.H. Mechanical thrombectomy in acute ischemic stroke. Rev. Neurol. 2017, 173, 106–113. [Google Scholar] [CrossRef]
- Blanc, R.; Escalard, S.; Baharvadhat, H.; Desilles, J.P.; Boisseau, W.; Fahed, R.; Redjem, H.; Ciccio, G.; Smajda, S.; Maier, B.; et al. Recent advances in devices for mechanical thrombectomy. Expert Rev. Med. Devices 2020, 17, 697–706. [Google Scholar] [CrossRef] [PubMed]
- Steffel, J.; Verhamme, P.; Potpara, T.S.; Albaladejo, P.; Antz, M.; Desteghe, L.; Haeusler, K.G.; Oldgren, J.; Reinecke, H.; Roldan-Schilling, V.; et al. The 2018 European Heart Rhythm Association Practical Guide on the use of non-vitamin K antagonist oral anticoagulants in patients with atrial fibrillation. Eur. Heart J. 2018, 39, 1330–1393. [Google Scholar] [CrossRef]
- Hugues, A.; Di Marco, J.; Ribault, S.; Ardaillon, H.; Janiaud, P.; Xue, Y.; Zhu, J.; Pires, J.; Khademi, H.; Rubio, L.; et al. Limited evidence of physical therapy on balance after stroke: A systematic review and meta-analysis. PLoS ONE 2019, 14, e0221700. [Google Scholar] [CrossRef]
- Borlongan, C.V. Concise Review: Stem Cell Therapy for Stroke Patients: Are We There Yet? Stem Cells Transl. Med. 2019, 8, 983–988. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Qu, J.; Xiang, C. The multi-functional roles of menstrual blood-derived stem cells in regenerative medicine. Stem Cell Res. Ther. 2019, 10, 1–10. [Google Scholar] [CrossRef]
- Huang, L.; Zhang, L. Neural stem cell therapies and hypoxic-ischemic brain injury. Prog Neurobiol. 2019, 173, 1–17. [Google Scholar] [CrossRef]
- Zhang, S.; Lachance, B.B.; Moiz, B.; Jia, X. Optimizing Stem Cell Therapy after Ischemic Brain Injury. J. Stroke 2020, 22, 286–305. [Google Scholar] [CrossRef] [PubMed]
- Napoli, E.; Borlongan, C.V. Recent Advances in Stem Cell-Based Therapeutics for Stroke. Transl. Stroke Res. 2016, 7, 452–457. [Google Scholar] [CrossRef]
- Banerjee, S.; Bentley, P.; Hamady, M.; Marley, S.; Davis, J.; Shlebak, A.; Nicholls, J.; Williamson, D.A.; Jensen, S.L.; Gordon, M.; et al. Intra-Arterial Immunoselected CD34+ Stem Cells for Acute Ischemic Stroke. Stem Cells Transl. Med. 2014, 3, 1322–1330. [Google Scholar] [CrossRef]
- Savitz, S.I.; Misra, V.; Kasam, M.; Juneja, H.; Cox, C.S., Jr.; Alderman, S.; Aisiku, I.; Kar, S.; Gee, A.; Grotta, J.C. Intravenous autologous bone marrow mononuclear cells for ischemic stroke. Ann. Neurol. 2011, 70, 59–69. [Google Scholar] [CrossRef] [PubMed]
- Chrostek, M.R.; Fellows, E.G.; Crane, A.T.; Grande, A.W.; Low, W.C. Efficacy of stem cell-based therapies for stroke. Brain Res. 2019, 1722, 146362. [Google Scholar] [CrossRef]
- Luo, L.; Albashari, A.A.; Wang, X.; Jin, L.; Zhang, Y.; Zheng, L.; Xia, J.; Xu, H.; Zhao, Y.; Xiao, J.; et al. Effects of Transplanted Heparin-Poloxamer Hydrogel Combining Dental Pulp Stem Cells and bFGF on Spinal Cord Injury Repair. Stem Cells Int. 2018, 2018, 2398521. [Google Scholar] [CrossRef] [PubMed]
- Ghuman, H.; Massensini, A.R.; Donnelly, J.; Kim, S.M.; Medberry, C.J.; Badylak, S.F.; Modo, M. ECM hydrogel for the treatment of stroke: Characterization of the host cell infiltrate. Biomaterials 2016, 91, 166–181. [Google Scholar] [CrossRef]
- Zhou, G.; Wang, Y.; Gao, S.; Fu, X.; Cao, Y.; Peng, Y.; Zhuang, J.; Hu, J.; Shao, A.; Wang, L. Potential Mechanisms and Perspectives in Ischemic Stroke Treatment Using Stem Cell Therapies. Front. Cell Dev. Biol. 2021, 9, 646927. [Google Scholar] [CrossRef]
- Sarmah, D.; Agrawal, V.; Rane, P.; Bhute, S.; Watanabe, M.; Kalia, K.; Ghosh, Z.; Dave, K.R.; Yavagal, D.R.; Bhattacharya, P. Mesenchymal Stem Cell Therapy in Ischemic Stroke: A Meta-analysis of Preclinical Studies. Clin. Pharm. Ther. 2018, 103, 990–998. [Google Scholar] [CrossRef]
- Esteban-Garcia, N.; Nombela, C.; Garrosa, J.; Rascón-Ramirez, F.J.; Barcia, J.A.; Sánchez-Sánchez-Rojas, L. Neurorestoration Approach by Biomaterials in Ischemic Stroke. Front. Neurosci. 2020, 14, 431. [Google Scholar] [CrossRef] [PubMed]
- Bang, O.Y.; Kim, E.H.; Cha, J.M.; Moon, G.J. Adult Stem Cell Therapy for Stroke: Challenges and Progress. J. Stroke 2016, 18, 256–266. [Google Scholar] [CrossRef]
- Volarevic, V.; Markovic, B.S.; Gazdic, M.; Volarevic, A.; Jovicic, N.; Arsenijevic, N.; Armstrong, L.; Djonov, V.; Lako, M.; Stojkovic, M. Ethical and Safety Issues of Stem Cell-Based Therapy. Inter. J. Med. Sci. 2018, 15, 36–45. [Google Scholar] [CrossRef]
- Lo, B.; Parham, L. Ethical issues in stem cell research. Endocr. Rev. 2009, 30, 204–213. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Xu, K.; Nguyen, H.; Guedes, V.A.; Borlongan, C.V.; Acosta, S.A. Stem Cell-Induced Biobridges as Possible Tools to Aid Neuroreconstruction after CNS Injury. Front. Cell Dev. Biol. 2017, 5, 51. [Google Scholar] [CrossRef]
- Stonesifer, C.; Corey, S.; Ghanekar, S.; Diamandis, Z.; Acosta, S.A.; Borlongan, C.V. Stem cell therapy for abrogating stroke-induced neuroinflammation and relevant secondary cell death mechanisms. Prog Neurobiol. 2017, 158, 94–131. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.; Wei, Z.Z.; Jiang, M.Q.; Mohamad, O.; Yu, S.P. Stem cell transplantation therapy for multifaceted therapeutic benefits after stroke. Prog. Neurobiol. 2017, 157, 49–78. [Google Scholar] [CrossRef]
- Liu, Y.P.; Lang, B.T.; Baskaya, M.K.; Dempsey, R.J.; Vemuganti, R. The potential of neural stem cells to repair stroke-induced brain damage. Acta Neuropathol. 2009, 117, 469–480. [Google Scholar] [CrossRef]
- Incontri Abraham, D.; Gonzales, M.; Ibarra, A.; Borlongan, C.V. Stand alone or join forces? Stem cell therapy for stroke. Expert Opin. Biol. Ther. 2019, 19, 25–33. [Google Scholar] [CrossRef]
- Napoli, E.; Lippert, T.; Borlongan, C.V. Stem Cell Therapy: Repurposing Cell-Based Regenerative Medicine Beyond Cell Replacement. Adv. Exp. Med. Biol. 2018, 1079, 87–91. [Google Scholar]
- Nguyen, H.; Zarriello, S.; Coats, A.; Nelson, C.; Kingsbury, C.; Gorsky, A.; Rajani, M.; Neal, E.G.; Borlongan, C.V. Stem cell therapy for neurological disorders: A focus on aging. Neurobiol. Dis. 2019, 126, 85–104. [Google Scholar] [CrossRef]
- Haghighitalab, A.; Matin, M.M.; Amin, A.; Minaee, S.; Bidkhori, H.R.; Doeppner, T.R.; Bahrami, A.R. Investigating the effects of IDO1, PTGS2, and TGF-β1 overexpression on immunomodulatory properties of hTERT-MSCs and their extracellular vesicles. Sci. Rep. 2021, 11, 7825. [Google Scholar] [CrossRef]
- Dabrowska, S.; Andrzejewska, A.; Lukomska, B.; Janowski, M. Neuroinflammation as a target for treatment of stroke using mesenchymal stem cells and extracellular vesicles. J. Neuroinflammation 2019, 16, 178. [Google Scholar] [CrossRef]
- Cirillo, C.; Brihmat, N.; Castel-Lacanal, E.; Le Friec, A.; Barbieux-Guillot, M.; Raposo, N.; Pariente, J.; Viguier, A.; Simonetta-Moreau, M.; Albucher, J.-F.; et al. Post-stroke remodeling processes in animal models and humans. J. Cereb. Blood Flow Metab. 2020, 40, 3–22. [Google Scholar] [CrossRef]
- Regenhardt, R.W.; Takase, H.; Lo, E.H.; Lin, D.J. Translating concepts of neural repair after stroke: Structural and functional targets for recovery. Restor Neurol. Neurosci. 2020, 38, 67–92. [Google Scholar] [CrossRef]
- Liska, M.G.; Crowley, M.G.; Nguyen, H.; Borlongan, C.V. Biobridge concept in stem cell therapy for ischemic stroke. J. Neurosurg. Sci. 2017, 61, 173–179. [Google Scholar]
- Tuazon, J.P.; Castelli, V.; Lee, J.Y.; Desideri, G.B.; Stuppia, L.; Cimini, A.M.; Borlongan, C.V. Neural stem cells. Adv. Exp. Med. Biol. 2019, 1201, 79–91. [Google Scholar]
- Zhang, X.L.; Zhang, X.G.; Huang, Y.R.; Zheng, Y.Y.; Ying, P.J.; Zhang, X.J.; Lu, X.; Wang, Y.J.; Zheng, G.Q. Stem cell-based therapy for experimental ischemic stroke: A preclinical systematic review. Front. Cell Neurosci. 2021, 15, 628908. [Google Scholar] [CrossRef] [PubMed]
- Bliss, T.M.; Andres, R.H.; Steinberg, G.K. Optimizing the success of cell transplantation therapy for stroke. Neurobiol. Dis. 2010, 37, 275–283. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Zhang, B.; Chhatbar, P.Y.; Dong, Y.; Alawieh, A.; Lowe, F.; Hu, X.; Feng, W. Mesenchymal stem cell therapy in stroke: A systematic review of literature in pre-clinical and clinical research. Cell Transpl. 2018, 27, 1723–1730. [Google Scholar] [CrossRef] [PubMed]
- Borlongan, C.V.; Kaneko, Y.; Maki, M.; Yu, S.J.; Ali, M.; Allickson, J.G.; Sanberg, C.D.; Kuzmin-Nichols, N.; Sanberg, P.R. Menstrual blood cells display stem cell-like phenotypic markers and exert neuroprotection following transplantation in experimental stroke. Stem Cells Dev. 2010, 19, 439–452. [Google Scholar] [CrossRef]
- Hara, K.; Matsukawa, N.; Yasuhara, T.; Xu, L.; Yu, G.; Maki, M.; Kawase, T.; Hess, D.C.; Kim, S.U.; Borlongan, C.V. Transplantation of post-mitotic human neuroteratocarcinoma-overexpressing Nurr1 cells provides therapeutic benefits in experimental stroke: In vitro evidence of expedited neuronal differentiation and GDNF secretion. J. Neurosci. Res. 2007, 85, 1240–1251. [Google Scholar] [CrossRef] [PubMed]
- Tajiri, N.; Acosta, S.; Glover, L.E.; Bickford, P.C.; Jacotte Simancas, A.; Yasuhara, T.; Date, I.; Solomita, M.A.; Antonucci, I.; Stuppia, L.; et al. Intravenous grafts of amniotic fluid-derived stem cells induce endogenous cell proliferation and attenuate behavioral deficits in ischemic stroke rats. PLoS ONE 2012, 7, e43779. [Google Scholar] [CrossRef] [PubMed]
- Borlongan, C.V.; Evans, A.; Yu, G.; Hess, D.C. Limitations of intravenous human bone marrow CD133+ cell grafts in stroke rats. Brain Res. 2005, 1048, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Tajiri, N.; Duncan, K.; Antoine, A.; Pabon, M.; Acosta, S.A.; de la Pena, I.; Hernadez-Ontiveros, D.G.; Shinozuka, K.; Ishikawa, H.; Kaneko, Y.; et al. Stem cell-paved biobridge facilitates neural repair in traumatic brain injury. Front. Syst. Neurosci. 2014, 8, 116. [Google Scholar] [CrossRef]
- Bjorklund, G.R.; Anderson, T.R.; Stabenfeldt, S.E. Recent advances in stem cell therapies to address neuroinflammation, stem cell survival, and the need for rehabilitative therapies to treat traumatic train injuries. Int. J. Mol. Sci. 2021, 22, 1978. [Google Scholar] [CrossRef]
- Gójska-Grymajło, A.; Zieliński, M.; Wardowska, A.; Gąsecki, D.; Pikuła, M.; Karaszewski, B. CXCR7+ and CXCR4+ stem cells and neuron specific enolase in acute ischemic stroke patients. Neurochem. Int. 2018, 120, 134–139. [Google Scholar] [CrossRef]
- Jiao, Y.; Liu, Y.W.; Chen, W.G.; Liu, J. Neuroregeneration and functional recovery after stroke: Advancing neural stem cell therapy toward clinical application. Neural Regen. Res. 2021, 16, 80–92. [Google Scholar]
- Liang, H.; Zhao, H.; Gleichman, A.; Machnicki, M.; Telang, S.; Tang, S.; Rshtouni, M.; Ruddell, J.; Carmichael, S.T. Region-specific and activity-dependent regulation of SVZ neurogenesis and recovery after stroke. Proc. Natl. Acad. Sci. USA 2019, 116, 13621–13630. [Google Scholar] [CrossRef]
- Tajiri, N.; Kaneko, Y.; Shinozuka, K.; Ishikawa, H.; Yankee, E.; McGrogan, M.; Case, C.; Borlongan, C.V. Stem cell recruitment of newly formed host cells via a successful seduction? Filling the gap between neurogenic niche and injured brain site. PLoS ONE 2013, 8, e74857. [Google Scholar] [CrossRef]
- Enzmann, G.; Kargaran, S.; Engelhardt, B. Ischemia-reperfusion injury in stroke: Impact of the brain barriers and brain immune privilege on neutrophil function. Ther. Adv. Neurol. Disord. 2018, 11, 1756286418794184. [Google Scholar] [CrossRef] [PubMed]
- Crowley, M.G.; Tajiri, N. Exogenous stem cells pioneer a biobridge to the advantage of host brain cells following stroke: New insights for clinical applications. Brain Circ. 2017, 3, 130–134. [Google Scholar] [PubMed]
- Drago, D.; Cossetti, C.; Iraci, N.; Gaude, E.; Musco, G.; Bachi, A.; Pluchino, S. The stem cell secretome and its role in brain repair. Biochimie 2013, 95, 2271–2285. [Google Scholar] [CrossRef] [PubMed]
- Corey, S.; Bonsack, B.; Borlongan, C.V. Stem cell-based regenerative medicine for neurological disorders: A special tribute to Dr. Teng Ma. Brain Circ. 2019, 5, 97–100. [Google Scholar]
- Asgari Taei, A.; Nasoohi, S.; Hassanzadeh, G.; Kadivar, M.; Dargahi, L.; Farahmandfar, M. Enhancement of angiogenesis and neurogenesis by intracerebroventricular injection of secretome from human embryonic stem cell-derived mesenchymal stem cells in ischemic stroke model. Biomed. Pharm. 2021, 140, 111709. [Google Scholar] [CrossRef]
- Tobin, M.K.; Stephen, T.K.L.; Lopez, K.L.; Pergande, M.R.; Bartholomew, A.M.; Cologna, S.M.; Lazarov, O. Activated Mesenchymal Stem Cells Induce Recovery Following Stroke Via Regulation of Inflammation and Oligodendrogenesis. J. Am. Heart Assoc. 2020, 9, e013583. [Google Scholar] [CrossRef]
- Kiyose, R.; Sasaki, M.; Kataoka-Sasaki, Y.; Nakazaki, M.; Nagahama, H.; Magota, H.; Oka, S.; Ukai, R.; Takemura, M.; Yokoyama, T.; et al. Intravenous infusion of mesenchymal stem cells enhances therapeutic efficacy of reperfusion therapy in cerebral ischemia. World Neurosurg. 2021, 149, e160–e169. [Google Scholar] [CrossRef] [PubMed]
- Asgari Taei, A.; Dargahi, L.; Nasoohi, S.; Hassanzadeh, G.; Kadivar, M.; Farahmandfar, M. The conditioned medium of human embryonic stem cell-derived mesenchymal stem cells alleviates neurological deficits and improves synaptic recovery in experimental stroke. J. Cell Physiol. 2021, 236, 1967–1979. [Google Scholar] [CrossRef]
- Venkat, P.; Chen, J.; Chopp, M. Exosome-mediated amplification of endogenous brain repair mechanisms and brain and systemic organ interaction in modulating neurological outcome after stroke. J. Cereb. Blood Flow Metab. 2018, 38, 2165–2178. [Google Scholar] [CrossRef]
- Otero-Ortega, L.; Laso-García, F.; Gómez-de Frutos, M.D.; Rodríguez-Frutos, B.; Pascual-Guerra, J.; Fuentes, B.; Díez-Tejedor, E.; Gutiérrez-Fernández, M. White matter repair after extracellular vesicles administration in an experimental animal model of subcortical stroke. Sci. Rep. 2017, 7, 44433. [Google Scholar] [CrossRef]
- Ye, X.; Hu, J.; Cui, G. Therapy effects of bone marrow stromal cells on ischemic stroke. Oxid. Med. Cell Longev. 2016, 7682960. [Google Scholar] [CrossRef] [PubMed]
- Xin, H.; Li, Y.; Cui, Y.; Yang, J.J.; Zhang, Z.G.; Chopp, M. Systemic administration of exosomes released from mesenchymal stromal cells promote functional recovery and neurovascular plasticity after stroke in rats. J. Cereb. Blood Flow Metab. 2013, 33, 1711–1715. [Google Scholar] [CrossRef] [PubMed]
- Doeppner, T.R.; Herz, J.; Görgens, A.; Schlechter, J.; Ludwig, A.K.; Radtke, S.; de Miroschedji, K.; Horn, P.A.; Giebel, B.; Hermann, D.M. Extracellular vesicles improve post-stroke neuroregeneration and prevent postischemic immunosuppression. Stem Cells Transl. Med. 2015, 4, 1131–1143. [Google Scholar] [CrossRef] [PubMed]
- Shiota, Y.; Nagai, A.; Sheikh, A.M.; Mitaki, S.; Mishima, S.; Yano, S.; Haque, M.A.; Kobayashi, S.; Yamaguchi, S. Transplantation of a bone marrow mesenchymal stem cell line increases neuronal progenitor cell migration in a cerebral ischemia animal model. Sci. Rep. 2018, 8, 14951. [Google Scholar] [CrossRef] [PubMed]
- Go, V.; Sarikaya, D.; Zhou, Y.; Bowley, B.G.E.; Pessina, M.A.; Rosene, D.L.; Zhang, Z.G.; Chopp, M.; Finklestein, S.P.; Medalla, M.; et al. Extracellular vesicles derived from bone marrow mesenchymal stem cells enhance myelin maintenance after cortical injury in aged rhesus monkeys. Exp. Neurol. 2021, 337, 113540. [Google Scholar] [CrossRef]
- Ling, X.; Zhang, G.; Xia, Y.; Zhu, Q.; Zhang, J.; Li, Q.; Niu, X.; Hu, G.; Yang, Y.; Wang, Y.; et al. Exosomes from human urine-derived stem cells enhanced neurogenesis via miR-26a/HDAC6 axis after ischaemic stroke. J. Cell Mol. Med. 2020, 24, 640–654. [Google Scholar] [CrossRef]
- Kojima, T.; Hirota, Y.; Ema, M.; Takahashi, S.; Miyoshi, I.; Okano, H.; Sawamoto, K. Subventricular zone-derived neural progenitor cells migrate along a blood vessel scaffold toward the post-stroke striatum. Stem Cells 2010, 28, 545–554. [Google Scholar] [CrossRef]
- Walter, J.; Keiner, S.; Witte, O.W.; Redecker, C. Differential stroke-induced proliferative response of distinct precursor cell subpopulations in the young and aged dentate gyrus. Neuroscience 2010, 169, 1279–1286. [Google Scholar] [CrossRef]
- Mast, H.; Thompson, J.L.; Lee, S.H.; Mohr, J.P.; Sacco, R. Hypertension and diabetes mellitus as determinants of multiple lacunar infarcts. Stroke 1995, 26, 30–33. [Google Scholar] [CrossRef] [PubMed]
- Bakreen, A.; Juntunen, M.; Dunlop, Y.; Ugidos, I.F.; Malm, T.; Miettinen, S.; Jolkkonen, J. Additive Behavioral Improvement after Combined Cell Therapy and Rehabilitation Despite Long-Term Microglia Presence in Stroke Rats. Int. J. Mol. Sci. 2021, 22, 1512. [Google Scholar] [CrossRef]
- Becerra-Calixto, A.; Cardona-Gómez, G.P. The Role of Astrocytes in Neuroprotection after Brain Stroke: Potential in Cell Therapy. Front Mol. Neurosci. 2017, 10, 88. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Tang, H.; Zhu, J.; Zhang, J.H. Transplanting Mesenchymal Stem Cells for Treatment of Ischemic Stroke. Cell Transpl. 2018, 27, 1825–1834. [Google Scholar] [CrossRef]
- Xin, H.; Li, Y.; Chen, X.; Chopp, M. Bone marrow stromal cells induce BMP2/4 production in oxygen-glucose-deprived astrocytes, which promotes an astrocytic phenotype in adult subventricular progenitor cells. J. Neurosci. Res. 2006, 83, 1485–1493. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez-Fernandez, M.; Otero-Ortega, L.; Ramos-Cejudo, J.; Rodríguez-Frutos, B.; Fuentes, B.; Díez-Tejedor, E. Adipose tissue-derived mesenchymal stem cells as a strategy to improve recovery after stroke. Expert Opin. Biol. Ther. 2015, 15, 873–881. [Google Scholar] [CrossRef]
- Ryu, B.; Sekine, H.; Homma, J.; Kobayashi, T.; Kobayashi, E.; Kawamata, T.; Shimizu, T. Allogeneic adipose-derived mesenchymal stem cell sheet that produces neurological improvement with angiogenesis and neurogenesis in a rat stroke model. J. Neurosurg. 2019, 132, 442–455. [Google Scholar] [CrossRef] [PubMed]
- Petit, I.; Jin, D.; Rafii, S. The SDF-1-CXCR4 signaling pathway: A molecular hub modulating neo-angiogenesis. Trends Immunol. 2007, 28, 299–307. [Google Scholar] [CrossRef] [PubMed]
- Haque, N.; Fareez, I.M.; Fong, L.F.; Mandal, C.; Abu Kasim, N.H.; Kacharaju, K.R.; Soesilawati, P. Role of the CXCR4-SDF1-HMGB1 pathway in the directional migration of cells and regeneration of affected organs. World J. Stem Cells 2020, 12, 938–951. [Google Scholar] [CrossRef]
- Redondo-Castro, E.; Cunningham, C.; Miller, J.; Martuscelli, L.; Aoulad-Ali, S.; Rothwell, N.J.; Kielty, C.M.; Allan, S.M.; Pinteaux, E. Interleukin-1 primes human mesenchymal stem cells towards an anti-inflammatory and pro-trophic phenotype in vitro. Stem Cell Res. Ther. 2017, 8, 79. [Google Scholar] [CrossRef]
- Prasanna, S.J.; Gopalakrishnan, D.; Shankar, S.R.; Vasandan, A.B. Pro-inflammatory cytokines, IFNgamma and TNFalpha, influence immune properties of human bone marrow and Wharton jelly mesenchymal stem cells differentially. PLoS ONE 2010, 5, e9016. [Google Scholar] [CrossRef]
- Lu, Z.; Chen, Y.; Dunstan, C.; Roohani-Esfahani, S.; Zreiqat, H. Priming adipose stem cells with tumor necrosis factor-alpha preconditioning potentiates their exosome efficacy for bone regeneration. Tissue Eng. Part. 2017, 23, 1212–1220. [Google Scholar] [CrossRef]
- Barrachina, L.; Remacha, A.R.; Romero, A.; Vázquez, F.J.; Albareda, J.; Prades, M.; Gosálvez, J.; Roy, R.; Zaragoza, P.; Martín-Burriel, I.; et al. Priming equine bone marrow-derived mesenchymal stem cells with proinflammatory cytokines: Implications in immunomodulation-immunogenicity balance, cell viability, and differentiation potential. Stem Cells Dev. 2017, 26, 15–24. [Google Scholar] [CrossRef]
- Santos, J.M.; Camões, S.P.; Filipe, E.; Cipriano, M.; Barcia, R.N.; Filipe, M.; Teixeira, M.; Simões, S.; Gaspar, M.; Mosqueira, D.; et al. Three-dimensional spheroid cell culture of umbilical cord tissue-derived mesenchymal stromal cells leads to enhanced paracrine induction of wound healing. Stem Cell Res. Ther. 2015, 6, 90. [Google Scholar] [CrossRef] [PubMed]
- Bhang, S.H.; Lee, S.; Shin, J.Y.; Lee, T.J.; Kim, B.S. Transplantation of cord blood mesenchymal stem cells as spheroids enhances vascularization. Tissue Eng. Part A 2012, 18, 2138–2147. [Google Scholar] [CrossRef] [PubMed]
- Wakabayashi, K.; Nagai, A.; Sheikh, A.M.; Shiota, Y.; Narantuya, D.; Watanabe, T.; Masuda, J.; Kobayashi, S.; Kim, S.U.; Yamaguchi, S. Transplantation of human mesenchymal stem cells promotes functional improvement and increased expression of neurotrophic factors in a rat focal cerebral ischemia model. J. Neurosci. Res. 2010, 88, 1017–1025. [Google Scholar] [CrossRef] [PubMed]
- Schäbitz, W.R.; Sommer, C.; Zoder, W.; Kiessling, M.; Schwaninger, M.; Schwab, S. Intravenous brain-derived neurotrophic factor reduces infarct size and counterregulates Bax and Bcl-2 expression after temporary focal cerebral ischemia. Stroke 2000, 3, 2212–2217. [Google Scholar] [CrossRef]
- Schäbitz, W.R.; Steigleder, T.; Cooper-Kuhn, C.M.; Schwab, S.; Sommer, C.; Schneider, A.; Kuhn, H.G. Intravenous brain-derived neurotrophic factor enhances poststroke sensorimotor recovery and stimulates neurogenesis. Stroke 2007, 38, 2165–2172. [Google Scholar] [CrossRef]
- Carrero, R.; Cerrada, I.; Lledó, E.; Dopazo, J.; García-García, F.; Rubio, M.-P.; Trigueros, C.; Dorronsoro, A.; Ruiz-Sauri, A.; Montero, J.A.; et al. IL1β induces mesenchymal stem cells migration and leucocyte chemotaxis through NF-κB. Stem Cell Rev. Rep. 2012, 8, 905–916. [Google Scholar] [CrossRef]
- Fujita, J.; Mori, M.; Kawada, H.; Ieda, Y.; Tsuma, M.; Matsuzaki, Y.; Kawaguchi, H.; Yagi, T.; Yuasa, S.; Endo, J.; et al. Administration of granulocyte colony-stimulating factor after myocardial infarction enhances the recruitment of hematopoietic stem cell-derived myofibroblasts and contributes to cardiac repair. Stem Cells 2007, 25, 2750–2759. [Google Scholar] [CrossRef]
- Lee, J.S.; Hong, J.M.; Moon, G.J.; Lee, P.H.; Ahn, Y.H.; Bang, O.Y. A long-term follow-up study of intravenous autologous mesenchymal stem cell transplantation in patients with ischemic stroke. Stem Cells 2010, 28, 1099–1106. [Google Scholar] [CrossRef]
- Chung, J.W.; Chang, W.H.; Bang, O.Y.; Moon, G.J.; Kim, S.J.; Kim, S.K.; Lee, J.S.; Sohn, S.I.; Kim, Y.H. Efficacy and Safety of Intravenous Mesenchymal Stem Cells for Ischemic Stroke. Neurology 2021, 96, e1012–e1023. [Google Scholar]
- Bang, O.Y.; Lee, J.S.; Lee, P.H.; Lee, G. Autologous mesenchymal stem cell transplantation in stroke patients. Ann. Neurol. 2005, 57, 874–882. [Google Scholar] [CrossRef]
- Bhasin, A.; Srivastava, M.V.; Mohanty, S.; Bhatia, R.; Kumaran, S.S.; Bose, S. Stem cell therapy: A clinical trial of stroke. Clin. Neurol. Neurosurg. 2013, 115, 1003–1008. [Google Scholar] [CrossRef] [PubMed]
- Bohaciakova, D.; Hruska-Plochan, M.; Tsunemoto, R.; Gifford, W.D.; Driscoll, S.P.; Glenn, T.D.; Wu, S.; Marsala, S.; Navarro, M.; Tadokoro, T.; et al. Scalable Solution for Isolating Human Multipotent Clinical-grade Neural Stem Cells from ES precursors. Stem Cell Res. Ther. 2019, 10, 83. [Google Scholar] [CrossRef]
- Zuo, F.; Xiong, F.; Wang, X.; Li, X.; Wang, R.; Ge, W.; Bao, X. Intrastriatal transplantation of human neural stem cells restores the impaired subventricular zone in parkinsonian mice. Stem Cells 2017, 35, 1519–1531. [Google Scholar] [CrossRef]
- Napoli, E.; Borlongan, C.V. Cell therapy in Parkinson’s disease: Host brain repair machinery gets a boost from stem cell grafts. Stem Cells 2017, 35, 1443–1445. [Google Scholar] [CrossRef]
- Zhang, P.; Li, J.; Liu, Y.; Chen, X.; Lu, H.; Kang, Q.; Li, W.; Gao, M. Human embryonic neural stem cell transplantation increases subventricular zone cell proliferation and promotes peri-infarct angiogenesis after focal cerebral ischemia. Neuropathology 2011, 31, 384–391. [Google Scholar] [CrossRef]
- Park, D.H.; Eve, D.J.; Sanberg, P.R.; Musso, J., III; Bachstetter, A.D.; Wolfson, A.; Schlunk, A.; Baradez, M.O.; Sinden, J.D.; Gemma, C. Increased neuronal proliferation in the dentate gyrus of aged rats following neural stem cell implantation. Stem Cells Dev. 2010, 19, 175–180. [Google Scholar] [CrossRef]
- Jin, K.; Xie, L.; Mao, X.; Greenberg, M.B.; Moore, A.; Peng, B.; Greenberg, R.B.; Greenberg, D.A. Effect of human neural precursor cell transplantation on endogenous neurogenesis after focal cerebral ischemia in the rat. Brain Res. 2011, 1374, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.H.; Wang, J.X.; Lin, X.J.; Wang, L.Q.; Shao, B.; Jin, K.L.; Wang, Y.T.; Yang, G.Y. Neural stem cell protects aged rat brain from ischemia-reperfusion injury through neurogenesis and angiogenesis. J. Cereb. Blood Flow Met. 2014, 34, 1138–1147. [Google Scholar] [CrossRef] [PubMed]
- Park, K.I.; Teng, Y.D.; Snyder, E.Y. The injured brain interacts reciprocally with neural stem cells supported by scaffolds to reconstitute lost tissue. Nat. Biotechnol. 2002, 20, 1111–1117. [Google Scholar] [CrossRef] [PubMed]
- Hassani, Z.; O’Reilly, J.; Pearse, Y.; Stroemer, P.; Tang, E.; Sinden, J.; Price, J.; Thuret, S. Human neural progenitor cell engraftment increases neurogenesis and microglial recruitment in the brain of rats with stroke. PLoS ONE 2012, 7, e50444. [Google Scholar] [CrossRef]
- Rumajogee, P.; Altamentova, S.; Li, L.; Li, J.; Wang, J.; Kuurstra, A.; Khazaei, M.; Beldick, S.; Menon, R.S.; van der Kooy, D.; et al. Exogenous neural precursor cell transplantation results in structural and functional recovery in a hypoxic-ischemic hemiplegic mouse model. Eneuro 2018, 5. [Google Scholar] [CrossRef]
- Kalladka, D.; Sinden, J.; Pollock, K.; Haig, C.; McLean, J.; Smith, W.; McConnachie, A.; Santosh, C.; Bath, P.M.; Dunn, L.; et al. Human neural stem cells in patients with chronic ischaemic stroke (PISCES): A phase 1, first-in-man study. Lancet 2016, 388, 787–796. [Google Scholar] [CrossRef]
- Liao, S.; Luo, C.; Cao, B.; Hu, H.; Wang, S.; Yue, H.; Chen, L.; Zhou, Z. Endothelial progenitor cells for ischemic stroke: Update on basic research and application. Stem Cells Int. 2017, 2017, 2193432. [Google Scholar] [CrossRef] [PubMed]
- Bayraktutan, U. Endothelial progenitor cells: Potential novel therapeutics for ischaemic stroke. Pharm. Res. 2019, 144, 181–191. [Google Scholar] [CrossRef]
- Iskander, A.; Knight, R.A.; Zhang, Z.G.; Ewing, J.R.; Shankar, A.; Varma, N.R.; Bagher-Ebadian, H.; Ali, M.M.; Arbab, A.S.; Janic, B. Intravenous administration of human umbilical cord blood-derived AC133+ endothelial progenitor cells in rat stroke model reduces infarct volume: Magnetic resonance imaging and histological findings. Stem Cells Transl. Med. 2012, 2, 703–714. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.F.; Ren, L.N.; Guo, G.; Cannella, L.A.; Chernaya, V.; Samuel, S.; Liu, S.X.; Wang, H.; Yang, X.F. Endothelial progenitor cells in ischemic stroke: An exploration from hypothesis to therapy. J. Hematol. Oncol. 2015, 8, 33. [Google Scholar] [CrossRef]
- Sanchez-Rojas, L.; Gómez-Pinedo, U.; Benito-Martin, M.S.; León-Espinosa, G.; Rascón-Ramirez, F.; Lendinez, C.; Martínez-Ramos, C.; Matías-Guiu, J.; Pradas, M.M.; Barcia, J.A. Biohybrids of scaffolding hyaluronic acid biomaterials plus adipose stem cells home local neural stem and endothelial cells: Implications for reconstruction of brain lesions after stroke. J. Biomed. Mater. Res. B Appl. Biomater. 2019, 107, 1598–1606. [Google Scholar] [CrossRef]
- De, D.; Karmakar, P.; Bhattacharya, D. Stem cell aging and regenerative medicine. Adv. Exp. Med. Biol. 2021, 1326, 11–37. [Google Scholar]
- Baker, N.; Boyette, L.B.; Tuan, R.S. Characterization of bone marrow-derived mesenchymal stem cells in aging. Bone 2015, 70, 37–47. [Google Scholar] [CrossRef]
- Deuse, T.; Hu, X.; Gravina, A.; Wang, D.; Tediashvili, G.; De, C.; Thayer, W.O.; Wahl, A.; Garcia, J.V.; Reichenspurner, H.; et al. Hypoimmunogenic derivatives of induced pluripotent stem cells evade immune rejection in fully immunocompetent allogeneic recipients. Nat. Biotechnol. 2019, 37, 252–258. [Google Scholar] [CrossRef]
- Gong, B.; Dong, Y.; He, C.; Jiang, W.; Shan, Y.; Zhou, B.Y.; Li, W. Intravenous Transplants of Human Adipose-Derived Stem Cell Protect the Rat Brain from Ischemia-Induced Damage. J. Stroke Cereb. Dis. 2019, 28, 595–603. [Google Scholar] [CrossRef] [PubMed]
- Chung, T.N.; Kim, J.H.; Choi, B.Y.; Jeong, J.Y.; Chung, S.P.; Kwon, S.W.; Suh, S.W. Effect of Adipose-Derived Mesenchymal Stem Cell Administration and Mild Hypothermia Induction on Delayed Neuronal Death After Transient Global Cerebral Ischemia. Crit. Care Med. 2017, 45, e508–e515. [Google Scholar] [CrossRef]
- Buga, A.M.; Bălşeanu, A.; Popa-Wagner, A.; Mogoantă, L. Strategies to improve post-stroke behavioral recovery in aged subjects. Rom. J. Morphol. Embryol. 2009, 50, 559–582. [Google Scholar] [PubMed]
- Popa-Wagner, A.; Buga, A.M.; Doeppner, T.R.; Hermann, D.M. Stem cell therapies in preclinical models of stroke associated with aging. Front. Cell Neurosci. 2014, 8, 347. [Google Scholar] [CrossRef] [PubMed]
- Capilla-Gonzalez, V.; Herranz-Pérez, V.; García-Verdugo, J.M. The aged brain: Genesis and fate of residual progenitor cells in the subventricular zone. Front. Cell Neurosci. 2015, 9, 365. [Google Scholar] [CrossRef] [PubMed]
- Popa-Wagner, A.; Filfan, M.; Uzoni, A.; Pourgolafshan, P.; Buga, A.M. Poststroke Cell Therapy of the Aged Brain. Neural Plast. 2015, 839638. [Google Scholar] [CrossRef] [PubMed]
- Balseanu, A.T.; Grigore, M.; Pinosanu, L.R.; Slevin, M.; Hermann, D.M.; Glavan, D.; Popa-Wagner, A. Electric Stimulation of Neurogenesis Improves Behavioral Recovery After Focal Ischemia in Aged Rats. Front. Neurosci. 2020, 14, 732. [Google Scholar] [CrossRef]
- Buga, A.M.; Vintilescu, R.; Balseanu, A.T.; Pop, O.T.; Streba, C.; Toescu, E.; Popa-Wagner, A. Repeated PTZ treatment at 25-day intervals leads to a highly efficient accumulation of doublecortin in the dorsal hippocampus of rats. PLoS ONE 2012, 7, e39302. [Google Scholar] [CrossRef]
- Korosi, A.; Naninck, E.F.; Oomen, C.A.; Schouten, M.; Krugers, H.; Fitzsimons, C.; Lucassen, P.J. Early-life stress mediated modulation of adult neurogenesis and behavior. Behav. Brain Res. 2012, 227, 400–409. [Google Scholar] [CrossRef]
- Tatarishvili, J.; Oki, K.; Monni, E.; Koch, P.; Memanishvili, T.; Buga, A.M.; Verma, V.; Popa-Wagner, A.; Brüstle, O.; Lindvall, O.; et al. Human induced pluripotent stem cells improve recovery in stroke-injured aged rats. Restor. Neurol. Neurosci. 2014, 32, 547–558. [Google Scholar] [CrossRef]
- Xu, W.; Zheng, J.; Gao, L.; Li, T.; Zhang, J.; Shao, A. Neuroprotective effects of stem cells in ischemic stroke. Stem Cells Int. 2017, 2017, 4653936. [Google Scholar] [CrossRef]
- Brown, J.; Park, Y.J.; Lee, J.Y.; Chase, T.N.; Koga, M.; Borlongan, C.V. Bone marrow-derived NCS-01 cells advance a novel cell-based therapy for stroke. Int. J. Mol. Sci. 2020, 21, 2845. [Google Scholar] [CrossRef]
- Mashkouri, S.; Crowley, M.G.; Liska, M.G.; Corey, S.; Borlongan, C.V. Utilizing pharmacotherapy and mesenchymal stem cell therapy to reduce inflammation following traumatic brain injury. Neural Regen Res. 2016, 11, 1379–1384. [Google Scholar] [CrossRef]
- Gurusamy, N.; Alsayari, A.; Rajasingh, S.; Rajasingh, J. Adult stem cells for regenerative therapy. Prog. Mol. Biol. Transl. Sci. 2018, 160, 1–22. [Google Scholar]
- Tuazon, J.P.; Castelli, V.; Borlongan, C.V. Drug-like delivery methods of stem cells as biologics for stroke. Expert Opin. Drug Deliv. 2019, 16, 823–833. [Google Scholar] [CrossRef]
- Hill, J.D.; Zuluaga-Ramirez, V.; Gajghate, S.; Winfield, M.; Sriram, U.; Rom, S.; Persidsky, Y. Activation of GPR55 induces neuroprotection of hippocampal neurogenesis and immune responses of neural stem cells following chronic, systemic inflammation. Brain Behav. Immun. 2019, 76, 165–181. [Google Scholar] [CrossRef] [PubMed]
- Acosta, S.A.; Tajiri, N.; Hoover, J.; Kaneko, Y.; Borlongan, C.V. Intravenous bone marrow stem cell grafts preferentially migrate to spleen and abrogate chronic inflammation in stroke. Stroke 2015, 46, 2616–2627. [Google Scholar] [CrossRef] [PubMed]
- Hansen, T.M.; Moss, A.J.; Brindle, N.P. Vascular endothelial growth factor and angiopoietins in neurovascular regeneration and protection following stroke. Curr. Neurovasc. Res. 2008, 5, 236–245. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Li, J.; Liu, Y.; Chen, X.; Kang, Q.; Zhao, J.; Li, W. Human neural stem cell transplantation attenuates apoptosis and improves neurological functions after cerebral ischemia in rats. Acta Anaesthesiol. Scand. 2009, 53, 1184–1191. [Google Scholar] [CrossRef]
- Zhang, R.; Zhang, Z.; Chopp, M. Function of neural stem cells in ischemic brain repair processes. J. Cereb. Blood Flow Metab. 2016, 36, 2034–2043. [Google Scholar] [CrossRef]
- Parent, J.M.; Vexler, Z.S.; Gong, C.; Derugin, N.; Ferriero, D.M. Rat forebrain neurogenesis and striatal neuron replacement after focal stroke. Ann. Neurol. 2002, 52, 802–813. [Google Scholar] [CrossRef] [PubMed]
- Ohab, J.J.; Fleming, S.; Blesch, A.; Carmichael, S.T. A neurovascular niche for neurogenesis after stroke. J. Neurosci. 2006, 26, 13007–13016. [Google Scholar] [CrossRef] [PubMed]
- Chintamen, S.; Imessadouene, F.; Kernie, S.G. Immune regulation of adult neurogenic niches in health and disease. Front. Cell Neurosci. 2021, 14, 571071. [Google Scholar] [CrossRef]
- Chen, J.; Venkat, P.; Zacharek, A.; Chopp, M. Neurorestorative therapy for stroke. Front. Hum. Neurosci. 2014, 8, 382. [Google Scholar] [CrossRef]
- Rahman, A.A.; Amruta, N.; Pinteaux, E.; Bix, G.J. Neurogenesis after stroke: A therapeutic perspective. Transl. Stroke Res. 2021, 12, 1–14. [Google Scholar] [CrossRef]
- Tuo, Q.Z.; Zhang, S.T.; Lei, P. Mechanisms of neuronal cell death in ischemic stroke and their therapeutic implications. In Medicinal Research Reviews; Wiley Online Library: Hoboken, NJ, USA, 2021; pp. 1–47. [Google Scholar]
- Arvidsson, A.; Collin, T.; Kirik, D.; Kokaia, Z.; Lindvall, O. Neuronal replacement from endogenous precursors in the adult brain after stroke. Nat. Med. 2002, 8, 963–970. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.L.; Chopp, M.; Gregg, S.R.; Toh, Y.; Roberts, C.; Letourneau, Y.; Buller, B.; Jia, L.; Davarani, S.P.N.; Zhang, Z.G. Patterns and dynamics of subventricular zone neuroblast migration in the ischemic striatum of the adult mouse. J. Cereb. Blood Flow Metab. 2009, 29, 1240–1250. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.L.; Chopp, M.; Roberts, C.; Liu, X.; Wei, M.; Nejad-Davarani, S.P.; Wang, X.; Zhang, Z.G. Stroke increases neural stem cells and angiogenesis in the neurogenic niche of the adult mouse. PLoS ONE 2014, 9, e113972. [Google Scholar] [CrossRef] [PubMed]
- Salmeron, K.E.; Maniskas, M.E.; Edwards, D.N.; Wong, R.; Rajkovic, I.; Trout, A.; Rahman, A.A.; Hamilton, S.; Fraser, J.F.; Pinteaux, E.; et al. Interleukin 1 alpha administration is neuroprotective and neuro-restorative following experimental ischemic stroke. J. Neuroinflamm. 2019, 16, 222. [Google Scholar] [CrossRef]
- Kiyota, T.; Ingraham, K.L.; Swan, R.J.; Jacobsen, M.T.; Andrews, S.J.; Ikezu, T. AAV serotype 2/1-mediated gene delivery of anti-inflammatory interleukin-10 enhances neurogenesis and cognitive function in APP+PS1 mice. Gene Ther. 2012, 19, 724–733. [Google Scholar] [CrossRef] [PubMed]
- Belenguer, G.; Duart-Abadia, P.; Jordán-Pla, A.; Domingo-Muelas, A.; Blasco-Chamarro, L.; Ferrón, S.R.; Morante-Redolat, J.M.; Fariñas, I. Adult neural stem cells are alerted by systemic inflammation through TNF-α receptor signaling. Cell Stem Cell 2021, 28, 285–299. [Google Scholar] [CrossRef]
- Christie, K.J.; Turnley, A.M. Regulation of endogenous neural stem/progenitor cells for neural repair-factors that promote neurogenesis and gliogenesis in the normal and damaged brain. Front. Cell Neurosci. 2013, 6, 70. [Google Scholar] [CrossRef] [PubMed]
- Rosenzweig, S.; Carmichael, S.T. The axon-glia unit in white matter stroke: Mechanisms of damage and recovery. Brain Res. 2015, 1623, 123–134. [Google Scholar] [CrossRef] [PubMed]
- Gregersen, R.; Christensen, T.; Lehrmann, E.; Diemer, N.H.; Finsen, B. Focal cerebral ischemia induces increased myelin basic protein and growth-associated protein-43 gene transcription in peri-infarct areas in the rat brain. Brain Res. 2001, 138, 384–392. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K.; Nogawa, S.; Suzuki, S.; Dembo, T.; Kosakai, A. Upregulation of oligodendrocyte progenitor cells associated with restoration of mature oligodendrocytes and myelination in peri-infarct area in the rat brain. Brain Res. 2003, 989, 172–179. [Google Scholar] [CrossRef]
- Cui, Y.; Jin, X.; Choi, D.J.; Choi, J.Y.; Kim, H.S.; Hwang, D.H.; Kim, B.G. Axonal degeneration in an in vitro model of ischemic white matter injury. Neurobiol. Dis. 2020, 134, 104672. [Google Scholar] [CrossRef]
- Raffaele, S.; Gelosa, P.; Bonfanti, E.; Lombardi, M.; Castiglioni, L.; Cimino, M.; Sironi, L.; Abbracchio, M.P.; Verderio, C.; Fumagalli, M. Microglial vesicles improve post-stroke recovery by preventing immune cell senescence and favoring oligodendrogenesis. Mol. Ther. 2021, 29, 1439–1458. [Google Scholar] [CrossRef]
- Ueno, Y.; Chopp, M.; Zhang, L.; Buller, B.; Liu, Z.; Lehman, N.L.; Liu, X.S.; Zhang, Y.; Roberts, C.; Zhang, Z.G. Axonal outgrowth and dendritic plasticity in the cortical peri-infarct area after experimental stroke. Stroke 2012, 43, 2221–2228. [Google Scholar] [CrossRef]
- Kawabori, M.; Shichinohe, H.; Kuroda, S.; Houkin, K. Clinical Trials of Stem Cell Therapy for Cerebral Ischemic Stroke. Int. J. Mol. Sci. 2020, 21, 7380. [Google Scholar] [CrossRef]
- Hicks, C.; Stevanato, L.; Stroemer, R.P.; Tang, E.; Richardson, S.; Sinden, J.D. In vivo and in vitro characterization of the angiogenic effect of CTX0E03 human neural stem cells. Cell Transpl. 2013, 22, 1541–1552. [Google Scholar] [CrossRef]
- Langhnoja, J.; Buch, L.; Pillai, P. Potential role of NGF, BDNF, and their receptors in oligodendrocytes differentiation from neural stem cell: An in vitro study. Cell Biol. Int. 2021, 45, 432–446. [Google Scholar] [CrossRef]
- Im, S.H.; Yu, J.H.; Park, E.S.; Lee, J.E.; Kim, H.O.; Park, K.I.; Kim, G.W.; Park, C.I.; Cho, S.R. Induction of striatal neurogenesis enhances functional recovery in an adult animal model of neonatal hypoxic-ischemic brain injury. Neuroscience 2010, 169, 259–268. [Google Scholar] [CrossRef]
- Ngo, M.T.; Harley, B.A.C. Progress in mimicking brain microenvironments to understand and treat neurological disorders. APL Bioeng. 2021, 5, 020902. [Google Scholar] [CrossRef]
- Schäfer, R.; Schwab, M.; Siegel, G.; von Ameln-Mayerhofer, A.; Buadze, M.; Lourhmati, A.; Wendel, H.-P.; Kluba, T.; Krueger, M.A.; Calaminus, C.; et al. Modulating endothelial adhesion and migration impacts stem cell therapies efficacy. EBioMedicine 2020, 60, 102987. [Google Scholar] [CrossRef]
- Cai, Y.; Liu, W.; Lian, L.; Xu, Y.; Bai, X.; Xu, S.; Zhang, J. Stroke treatment: Is exosome therapy superior to stem cell therapy? Biochimie 2020, 179, 190–204. [Google Scholar] [CrossRef]
- Wei, W.; Wu, D.; Duan, Y.; Elkin, K.B.; Chandra, A.; Guan, L.; Peng, C.; He, X.; Wu, C.; Ji, X.; et al. Neuroprotection by mesenchymal stem cell (MSC) administration is enhanced by local cooling infusion (LCI) in ischemia. Brain Res. 2019, 1724, 146406. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Xiao, X.; Xiao, H.; Hu, Z.; Tan, F. CUEDC2 ablation enhances the efficacy of mesenchymal stem cells in ameliorating cerebral ischemia/reperfusion insult. Aging 2021, 13, 4335–4356. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Zhang, S.; Liu, J.; Liu, F.; Du, F.; Li, M.; Chen, A.T.; Bao, Y.; Suh, H.W.; Avery, J.; et al. Targeted Drug Delivery to Stroke via Chemotactic Recruitment of Nanoparticles Coated with Membrane of Engineered Neural Stem Cells. Small 2019, 15, e1902011. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Yang, X.; Liu, T.; Shao, J.; Fu, N.; Yan, A.; Geng, K.; Xia, W. Adjudin-preconditioned neural stem cells enhance neuroprotection after ischemia reperfusion in mice. Stem Cell Res. Ther. 2017, 8, 248. [Google Scholar] [CrossRef]
- Zhang, J.J.; Zhu, J.J.; Hu, Y.B.; Xiang, G.H.; Deng, L.C.; Wu, F.Z.; Wei, X.J.; Wang, Y.H.; Sun, L.Y.; Lou, X.Q.; et al. Transplantation of bFGF-expressing neural stem cells promotes cell migration and functional recovery in rat brain after transient ischemic stroke. Oncotarget 2017, 8, 102067–102077. [Google Scholar] [CrossRef]
- Zhang, G.; Guo, X.; Chen, L.; Li, B.; Gu, B.; Wang, H.; Wu, G.; Kong, J.; Chen, W.; Yu, Y. Interferon-γ Promotes Neuronal Repair by Transplanted Neural Stem Cells in Ischemic Rats. Stem Cells Dev. 2018, 27, 355–366. [Google Scholar] [CrossRef]
- Zhang, G.; Chen, L.; Chen, W.; Li, B.; Yu, Y.; Lin, F.; Guo, X.; Wang, H.; Wu, G.; Gu, B.; et al. Neural Stem Cells Alleviate Inflammation via Neutralization of IFN-γ Negative Effect in Ischemic Stroke Model. J. Biomed. Nanotechnol. 2018, 14, 1178–1188. [Google Scholar] [CrossRef]
- Hosseini, S.M.; Ziaee, S.M.; Haider, K.H.; Karimi, A.; Tabeshmehr, P.; Abbasi, Z. Preconditioned neurons with NaB and nicorandil, a favorable source for stroke cell therapy. J. Cell Biochem 2018, 119, 10301–10313. [Google Scholar] [CrossRef] [PubMed]
- Spellicy, S.E.; Stice, S.L. Tissue and Stem Cell Sourced Extracellular Vesicle Communications with Microglia. Stem Cell Rev. Rep. 2021, 17, 357–368. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Liu, Z.; Tan, F.; Hu, Z.; Lu, M. Effects of the Insulted Neuronal Cells-Derived Extracellular Vesicles on the Survival of Umbilical Cord-Derived Mesenchymal Stem Cells following Cerebral Ischemia/Reperfusion Injury. Oxid. Med. Cell Longev. 2020, 2020, 9768713. [Google Scholar] [CrossRef] [PubMed]
- Chou, C.H.; Modo, M. Characterization of gene expression changes in human neural stem cells and endothelial cells modeling a neurovascular microenvironment. Brain Res. Bull. 2020, 158, 9–19. [Google Scholar] [CrossRef]
- Huang, L.; Wan, Y.; Dang, Z.; Yang, P.; Yang, Q.; Wu, S. Hypoxic preconditioning ameliorated neuronal injury after middle cerebral artery occlusion by promoting neurogenesis. Brain Behav. 2020, 10, e01804. [Google Scholar] [CrossRef] [PubMed]
- Hao, P.; Duan, H.; Hao, F.; Chen, L.; Sun, M.; Fan, K.S.; Sun, Y.E.; Williams, D.; Yang, Z.; Li, X. Neural repair by NT3-chitosan via enhancement of endogenous neurogenesis after adult focal aspiration brain injury. Biomaterials 2017, 140, 88–102. [Google Scholar] [CrossRef]
- Bernstock, J.D.; Peruzzotti-Jametti, L.; Leonardi, T.; Vicario, N.; Ye, D.; Lee, Y.-J.; Maric, D.; Johnson, K.R.; Mou, Y.; Van Den Bosch, A.; et al. SUMOylation promotes survival and integration of neural stem cell grafts in ischemic stroke. EBioMedicine 2019, 42, 214–224. [Google Scholar] [CrossRef]
- Jiang, X.C.; Xiang, J.J.; Wu, H.H.; Zhang, T.Y.; Zhang, D.P.; Xu, Q.H.; Huang, X.L.; Kong, X.L.; Sun, J.H.; Hu, Y.L.; et al. Neural Stem Cells Transfected with Reactive Oxygen Species-Responsive Polyplexes for Effective Treatment of Ischemic Stroke. Adv. Mater. 2019, 31, e1807591. [Google Scholar] [CrossRef] [PubMed]
- Song, S.; Amores, D.; Chen, C.; McConnell, K.; Oh, B.; Poon, A.; George, P.M. Controlling properties of human neural progenitor cells using 2D and 3D conductive polymer scaffolds. Sci. Rep. 2019, 9, 19565. [Google Scholar] [CrossRef]
- Petersen, M.A.; Ryu, J.K.; Chang, K.J.; Etxeberria, A.; Bardehle, S.; Mendiola, A.S.; Kamau-Devers, W.; Fancy, S.P.J.; Thor, A.; Bushong, E.A.; et al. Fibrinogen Activates BMP Signaling in Oligodendrocyte Progenitor Cells and Inhibits Remyelination after Vascular Damage. Neuron 2017, 96, 1003–1012. [Google Scholar] [CrossRef] [PubMed]
- Safakheil, M.; Safakheil, H. The Effect of Exosomes Derived from Bone Marrow Stem Cells in Combination with Rosuvastatin on Functional Recovery and Neuroprotection in Rats After Ischemic Stroke. J. Mol. Neurosci. 2020, 70, 724–737. [Google Scholar] [CrossRef]
- Chen, J.; Zhang, Z.G.; Li, Y.; Wang, Y.; Wang, L.; Jiang, H.; Zhang, C.; Lu, M.; Katakowski, M.; Feldkamp, C.S.; et al. Statins induce angiogenesis, neurogenesis, and synaptogenesis after stroke. Ann. Neurol. 2003, 53, 743–751. [Google Scholar] [CrossRef] [PubMed]
- Tuladhar, A.; Obermeyer, J.M.; Payne, S.L.; Siu, R.C.W.; Zand, S.; Morshead, C.M. Shoichet MS. Injectable hydrogel enables local and sustained co-delivery to the brain: Two clinically approved biomolecules, cyclosporine and erythropoietin, accelerate functional recovery in rat model of stroke. Biomaterials 2020, 235, 119794. [Google Scholar] [CrossRef] [PubMed]
- Ould-Brahim, F.; Sarma, S.N.; Syal, C.; Lu, K.J.; Seegobin, M.; Carter, A.; Jeffers, M.S.; Doré, C.; Stanford, W.L.; Corbett, D.; et al. Metformin Preconditioning of Human Induced Pluripotent Stem Cell-Derived Neural Stem Cells Promotes Their Engraftment and Improves Post-Stroke Regeneration and Recovery. Stem Cells Dev. 2018, 27, 1085–1096. [Google Scholar] [CrossRef] [PubMed]
- Peña, I.D.; Borlongan, C.V. Translating G-CSF as an Adjunct Therapy to Stem Cell Transplantation for Stroke. Transl. Stroke Res. 2015, 6, 421–429. [Google Scholar] [CrossRef] [PubMed]
- Choi, C.; Kim, H.M.; Shon, J.; Park, J.; Kim, H.-T.; Kang, S.H.; Oh, S.-H.; Kim, N.K.; Kim, O.J. The combination of mannitol and temozolomide increases the effectiveness of stem cell treatment in a Chronic stroke model. Cytotherapy 2018, 20, 820–829. [Google Scholar] [CrossRef]
- Gonzales-Portillo, G.S.; Sanberg, P.R.; Franzblau, M.; Gonzales-Portillo, C.; Diamandis, T.; Staples, M.; Sanberg, C.D.; Borlongan, C.V. Mannitol-enhanced delivery of stem cells and their growth factors across the blood-brain barrier. Cell Transpl. 2014, 23, 531–539. [Google Scholar] [CrossRef]
- Tajiri, N.; Lee, J.Y.; Acosta, S.; Sanberg, P.R.; Borlongan, C.V. Breaking the Blood-Brain Barrier with Mannitol to Aid Stem Cell Therapeutics in the Chronic Stroke Brain. Cell Transpl. 2016, 25, 1453–1460. [Google Scholar] [CrossRef]
- Choi, C.; Kim, H.M.; Shon, J.; Park, J.; Kim, H.-T.; Oh, S.-H.; Kim, N.K.; Kim, O.J. Additional increased effects of mannitol-temozolomide combined treatment on blood-brain barrier permeability. Biochem. Biophys. Res. Commun. 2018, 497, 769–775. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.P.; Tung, J.K.; Wei, Z.Z.; Chen, D.; Berglund, K.; Zhong, W.; Zhang, J.Y.; Gu, X.; Song, M.; Gross, R.E.; et al. Optochemogenetic Stimulation of Transplanted iPS-NPCs Enhances Neuronal Repair and Functional Recovery after Ischemic Stroke. J. Neurosci. 2019, 39, 6571–6594. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, E.P.; Malysz-Cymborska, I.; Golubczyk, D.; Kalkowski, L.; Kwiatkowska, J.; Reis, R.L.; Oliveira, J.M.; Walczak, P. Advances in bioinks and in vivo imaging of biomaterials for CNS applications. Acta Biomater. 2019, 95, 60–72. [Google Scholar] [CrossRef] [PubMed]
- Gopalakrishnan, A.; Shankarappa, S.A.; Rajanikant, G.K. Hydrogel Scaffolds: Towards Restitution of Ischemic Stroke-Injured Brain. Transl. Stroke Res. 2019, 10, 1–18. [Google Scholar] [CrossRef]
- Piejko, M.; Jablonska, A.; Walczak, P.; Janowski, M. Proteolytic Rafts for Improving Intraparenchymal Migration of Minimally Invasively Administered Hydrogel-Embedded Stem Cells. Int. J. Mol. Sci. 2019, 20, 3830. [Google Scholar] [CrossRef]
- Fernández-García, L.; Pérez-Rigueiro, J.; Martinez-Murillo, R.; Panetsos, F.; Ramos, M.; Guinea, G.V.; González-Nieto, D. Cortical Reshaping and Functional Recovery Induced by Silk Fibroin Hydrogels-Encapsulated Stem Cells Implanted in Stroke Animals. Front. Cell Neurosci. 2018, 12, 296. [Google Scholar] [CrossRef] [PubMed]
- Ballios, B.G.; Cooke, M.J.; Donaldson, L.; Coles, B.L.; Morshead, C.M.; van der Kooy, D.; Shoichet, M.S. A Hyaluronan-Based Injectable Hydrogel Improves the Survival and Integration of Stem Cell Progeny following Transplantation. Stem Cell Rep. 2015, 4, 1031–1045. [Google Scholar] [CrossRef]
- Zhuo, F.; Liu, X.; Gao, Q.; Wang, Y.; Hu, K.; Cai, Q. Injectable hyaluronan-methylcellulose composite hydrogel crosslinked by polyethylene glycol for central nervous system tissue engineering. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 81, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Kang, P.; Kumar, S.; Schaffer, D. Novel biomaterials to study neural stem cell mechanobiology and improve cell-replacement therapies. Curr. Opin. Biomed. Eng. 2017, 4, 13–20. [Google Scholar] [CrossRef]
- Marquardt, L.M.; Heilshorn, S.C. Design of Injectable Materials to Improve Stem Cell Transplantation. Curr. Stem Cell Rep. 2016, 2, 207–220. [Google Scholar] [CrossRef] [PubMed]
- Unal, D.B.; Caliari, S.R.; Lampe, K.J. Engineering biomaterial microenvironments to promote myelination in the central nervous system. Brain Res. Bull. 2019, 152, 159–174. [Google Scholar] [CrossRef] [PubMed]
- Tsintou, M.; Dalamagkas, K.; Moore, T.L.; Rathi, Y.; Kubicki, M.; Rosene, D.L.; Makris, N. The use of hydrogel-delivered extracellular vesicles in recovery of motor function in stroke: A testable experimental hypothesis for clinical translation including behavioral and neuroimaging assessment approaches. Neural. Regen Res. 2021, 16, 605–613. [Google Scholar]
- Nordström, T.; Andersson, L.C.; Åkerman, K.E.O. Regulation of intracellular pH by electrogenic Na+/HCO3- co-transporters in embryonic neural stem cell-derived radial glia-like cells. Biochim. Biophys. Acta Biomembr. 2019, 1861, 1037–1048. [Google Scholar] [CrossRef]
- Hongjin, W.; Han, C.; Baoxiang, J.; Shiqi, Y.; Xiaoyu, X. Reconstituting neurovascular unit based on the close relations between neural stem cells and endothelial cells: An effective method to explore neurogenesis and angiogenesis. Rev. Neurosci. 2020, 31, 143–159. [Google Scholar] [CrossRef]
- Veenman, L. Raloxifene as Treatment for Various Types of Brain Injuries and Neurodegenerative Diseases: A Good Start. Int. J. Mol. Sci. 2020, 21, 7586. [Google Scholar] [CrossRef] [PubMed]
- Dimitrova-Shumkovska, J.; Krstanoski, L.; Veenman, L. Diagnostic and Therapeutic Potential of TSPO Studies Regarding Neurodegenerative Diseases, Psychiatric Disorders, Alcohol Use Disorders, Traumatic Brain Injury, and Stroke: An Update. Cells 2020, 9, 870. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulos, V.; Lecanu, L.; Brown, R.C.; Han, Z.; Yao, Z.X. Peripheral-type benzodiazepine receptor in neurosteroid biosynthesis, neuropathology and neurological disorders. Neuroscience 2006, 138, 749–756. [Google Scholar] [CrossRef]
- Chen, W.H.; Yeh, H.L.; Tsao, C.W.; Lien, L.M.; Chiwaya, A.; Alizargar, J.; Bai, C.H. Plasma Translocator Protein Levels and Outcomes of Acute Ischemic Stroke: A Pilot Study. Dis. Markers. 2018, 2018, 9831079. [Google Scholar] [CrossRef]
- Khan, M.M.; Wakade, C.; de Sevilla, L.; Brann, D.W. Selective estrogen receptor modulators (SERMs) enhance neurogenesis and spine density following focal cerebral ischemia. J. Steroid Biochem. Mol. Biol. 2015, 146, 38–47. [Google Scholar] [CrossRef]
- Culmsee, C.; Zhu, X.; Yu, Q.S.; Chan, S.L.; Camandola, S.; Guo, Z.; Greig, N.H.; Mattson, M.P. A synthetic inhibitor of p53 protects neurons against death induced by ischemic and excitotoxic insults, and amyloid beta-peptide. J. Neurochem. 2001, 77, 220–228. [Google Scholar] [CrossRef] [PubMed]
- Huo, C.C.; Zheng, Y.; Lu, W.W.; Zhang, T.Y.; Wang, D.F.; Xu, D.S.; Li, Z.Y. Prospects for intelligent rehabilitation techniques to treat motor dysfunction. Neural Regen Res. 2021, 16, 264–269. [Google Scholar] [PubMed]
- Hara, Y. Brain plasticity and rehabilitation in stroke patients. J. Nippon Med. Sch. 2015, 82, 4–13. [Google Scholar] [CrossRef]
- Mu, J.; Bakreen, A.; Juntunen, M.; Korhonen, P.; Oinonen, E.; Cui, L.; Myllyniemi, M.; Zhao, S.; Miettinen, S.; Jolkkonen, J. Combined Adipose Tissue-Derived Mesenchymal Stem Cell Therapy and Rehabilitation in Experimental Stroke. Front. Neurol. 2019, 10, 235. [Google Scholar] [CrossRef]
- Komleva Iu, K.; Salmina, A.B.; Prokopenko, S.V.; Shestakova, L.A.; Petrova, M.M.; Malinovskaia, N.A.; Lopatina, O.L. Changes in structural and functional plasticity of the brain induced by environmental enrichment. Ann. Russ. Acad. Med. Sci. 2013, 68, 39–48. [Google Scholar] [CrossRef]
- Kempermann, G. Environmental enrichment, new neurons and the neurobiology of individuality. Nat. Rev. Neurosci. 2019, 20, 235–245. [Google Scholar] [CrossRef]
- Komitova, M.; Mattsson, B.; Johansson, B.B.; Eriksson, P.S. Enriched environment increases neural stem/progenitor cell proliferation and neurogenesis in the subventricular zone of stroke-lesioned adult rats. Stroke 2005, 36, 1278–1282. [Google Scholar] [CrossRef]
- Caliaperumal, J.; Colbourne, F. Rehabilitation improves behavioral recovery and lessens cell death without affecting iron, ferritin, transferrin, or inflammation after intracerebral hemorrhage in rats. Neurorehabil. Neural Repair 2014, 28, 395–404. [Google Scholar] [CrossRef] [PubMed]
- Zhan, Y.; Li, M.Z.; Yang, L.; Feng, X.F.; Lei, J.F.; Zhang, N.; Zhao, Y.Y.; Zhao, H. The three-phase enriched environment paradigm promotes neurovascular restorative and prevents learning impairment after ischemic stroke in rats. Neurobiol. Dis. 2020, 146, 105091. [Google Scholar] [CrossRef] [PubMed]
- Di Pino, G.; Pellegrino, G.; Assenza, G.; Capone, F.; Ferreri, F.; Formica, D.; Ranieri, F.; Tombini, M.; Ziemann, U.; Rothwell, J.C.; et al. Modulation of brain plasticity in stroke: A novel model for neurorehabilitation. Nat. Rev. Neurol. 2014, 10, 597–608. [Google Scholar] [CrossRef] [PubMed]
- Borich, M.R.; Brown, K.E.; Boyd, L.A. Motor skill learning is associated with diffusion characteristics of white matter in individuals with chronic stroke. J. Neurol. Phys. Ther. 2014, 38, 151–160. [Google Scholar] [CrossRef] [PubMed]
- Resquín, F.; Cuesta Gómez, A.; Gonzalez-Vargas, J.; Brunetti, F.; Torricelli, D.; Molina Rueda, F.; Cano de la Cuerda, R.; Miangolarra, J.C.; Pons, J.L. Hybrid robotic systems for upper limb rehabilitation after stroke: A review. Med. Eng. Phys. 2016, 38, 1279–1288. [Google Scholar] [CrossRef] [PubMed]
- Bang, D.H.; Shin, W.S.; Choi, H.S. Effects of modified constraint-induced movement therapy with trunk restraint in early stroke patients: A single-blinded, randomized, controlled, pilot trial. NeuroRehabilitation 2018, 42, 29–35. [Google Scholar] [CrossRef]
- Huang, S.; Yu, X.; Lu, Y.; Qiao, J.; Wang, H.; Jiang, L.M.; Wu, X.; Niu, W. Body weight support-Tai Chi footwork for balance of stroke survivors with fear of falling: A pilot randomized controlled trial. Complement. Ther. Clin. Pr. 2019, 37, 140–147. [Google Scholar] [CrossRef]
- Takeuchi, N.; Izumi, S.-I. Rehabilitation with Poststroke Motor Recovery: A Review with a Focus on Neural Plasticity. Stroke Res. Treat. 2013, 2013, 128641. [Google Scholar] [CrossRef] [PubMed]
- Bayona, N.A.; Bitensky, J.; Salter, K.; Teasell, R. The Role of Task-Specific Training in Rehabilitation Therapies. Top Stroke Rehabil. 2005, 12, 58–65. [Google Scholar] [CrossRef]
- Lai, M.I.; Pan, L.L.; Tsai, M.W.; Shih, Y.F.; Wei, S.H.; Chou, L.W. Investigating the Effects of Peripheral Electrical Stimulation on Corticomuscular Functional Connectivity Stroke Survivors. Top. Stroke Rehabil. 2016, 23, 154–162. [Google Scholar] [CrossRef]
- Christiansen, L.; Perez, M.A. Targeted-Plasticity in the Corticospinal Tract After Human Spinal Cord Injury. Neurotherapeutics 2018, 15, 618–627. [Google Scholar] [CrossRef]
- Biasiucci, A.; Leeb, R.; Iturrate, I.; Perdikis, S.; Al-Khodairy, A.; Corbet, T.; Schnider, A.; Schmidlin, T.; Zhang, H.; Bassolino, M.; et al. Brain-actuated functional electrical stimulation elicits lasting arm motor recovery after stroke. Nat. Commun. 2018, 9, 2421. [Google Scholar] [CrossRef] [PubMed]
- Salhab, G.; Sarraj, A.R.; Saleh, S. Mirror therapy combined with functional electrical stimulation for rehabilitation of stroke survivors’ ankle dorsiflexion. Annu. Int. Conf. IEEE Eng. Med Biol. Soc. 2016, 4699–4702. [Google Scholar]
- Quandt, F.; Hummel, F.C. The influence of functional electrical stimulation on hand motor recovery in stroke patients: A review. Exp. Transl. Stroke Med. 2014, 6, 9. [Google Scholar] [CrossRef] [PubMed]
- Staudt, M.D.; Herring, E.Z.; Gao, K.; Miller, J.P.; Sweet, J.A. Evolution in the Treatment of Psychiatric Disorders: From Psychosurgery to Psychopharmacology to Neuromodulation. Front. Neurosci. 2019, 13, 108. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Chen, Z. Neuromodulation for Pain Management. Adv. Exp. Med. Biol. 2019, 1101, 207–223. [Google Scholar]
- Dodd, K.C.; Nair, V.A.; Prabhakaran, V. Role of the contralesional vs. ipsilesional hemisphere in stroke recovery. Front. Hum. Neurosci. 2017, 11, 469. [Google Scholar] [CrossRef] [PubMed]
- Yamada, N.; Kakuda, W.; Senoo, A.; Kondo, T.; Mitani, S.; Shimizu, M.; Abo, M. Functional cortical reorganization after low-frequency repetitive transcranial magnetic stimulation plus intensive occupational therapy for upper limb hemiparesis: Evaluation by functional magnetic resonance imaging in poststroke patients. Int. J. Stroke 2013, 8, 422–429. [Google Scholar] [CrossRef]
- Tamburella, F.; Moreno, J.C.; Herrera Valenzuela, D.S.; Pisotta, I.; Iosa, M.; Cincotti, F.; Mattia, D.; Pons, J.L.; Molinari, M. Influences of the biofeedback content on robotic post-stroke gait rehabilitation: Electromyographic vs joint torque biofeedback. J. Neuroeng. Rehabil. 2019, 16, 95. [Google Scholar] [CrossRef]
- Lehrer, N.; Chen, Y.; Duff, M.; Wolf, S.L.; Rikakis, T. Exploring the bases for a mixed reality stroke rehabilitation system, Part II: Design of interactive feedback for upper limb rehabilitation. J. Neuroeng. Rehabil. 2011, 8, 54. [Google Scholar] [CrossRef]
- Afzal, M.R.; Byun, H.-Y.; Oh, M.-K.; Yoon, J. Effects of kinesthetic haptic feedback on standing stability of young healthy subjects and stroke patients. J. Neuroeng. Rehabil. 2015, 12, 27. [Google Scholar] [CrossRef]
- Afzal, M.R.; Oh, M.-K.; Choi, H.Y.; Yoon, J. A novel balance training system using multimodal biofeedback. Biomed. Eng. Online 2016, 15, 42. [Google Scholar] [CrossRef][Green Version]
- Wang, T.; Mantini, D.; Gillebert, C.R. The potential of real-time fMRI neurofeedback for stroke rehabilitation: A systematic review. Cortex 2018, 107, 148–165. [Google Scholar] [CrossRef]
- Mihara, M. S3-1. Therapeutic intervention for neurological patients with neurofeedback technique. Clin. Neurophysiol. 2018, 129, e23. [Google Scholar] [CrossRef]
- Hasegawa, N.; Takeda, K.; Mancini, M.; King, L.A.; Horak, F.B.; Asaka, T. Differential effects of visual versus auditory biofeedback training for voluntary postural sway. PLoS ONE 2020, 15, e0244583. [Google Scholar] [CrossRef]
- Renton, T.; Tibbles, A.; Topolovec-Vranic, J. Neurofeedback as a form of cognitive rehabilitation therapy following stroke: A systematic review. PLoS ONE 2017, 12, e0177290. [Google Scholar] [CrossRef]
- Mehrholz, J.; Pohl, M. Electromechanical-assisted gait training after stroke: A systematic review comparing end-effector and exoskeleton devices. J. Rehabil. Med. 2012, 44, 193–199. [Google Scholar] [CrossRef]
- Veerbeek, J.M.; Langbroek-Amersfoort, A.C.; van Wegen, E.E.; Meskers, C.G.; Kwakkel, G. Effects of Robot-Assisted Therapy for the Upper Limb After Stroke. Neurorehabil. Neural Repair 2017, 31, 107–121. [Google Scholar] [CrossRef]
- Beom, J.; Koh, S.; Nam, H.S.; Kim, W.; Kim, Y.; Seo, H.G.; Oh, B.M.; Chung, S.G.; Kim, S. Robotic Mirror Therapy System for Functional Recovery of Hemiplegic Arms. J. Vis. Exp. 2016, 114. [Google Scholar] [CrossRef]
- Huang, X.; Naghdy, F.; Naghdy, G.; Du, H.; Todd, C. Robot-assisted post-stroke motion rehabilitation in upper extremities: A survey. Int. J. Disabil Hum. Dev. 2017, 16, 233–247. [Google Scholar] [CrossRef]
- Morone, G.; Cocchi, I.; Paolucci, S.; Iosa, M. Robot-assisted therapy for arm recovery for stroke patients: State of the art and clinical implication. Expert Rev. Med. Devices 2020, 17, 223–233. [Google Scholar] [CrossRef]
- Sale, P.; Infarinato, F.; Del Percio, C.; Lizio, R.; Babiloni, C.; Foti, C.; Franceschini, M. Electroencephalographic markers of robot-aided therapy in stroke patients for the evaluation of upper limb rehabilitation. Int. J. Rehabil. Res. 2015, 38, 294–305. [Google Scholar] [CrossRef]
- Major, Z.Z.; Vaida, C.; Major, K.A.; Tucan, P.; Simori, G.; Banica, A.; Brusturean, E.; Burz, A.; Craciunas, R.; Ulinici, I.; et al. The Impact of Robotic Rehabilitation on the Motor System in Neurological Diseases. A Multimodal Neurophysiological Approach. Int. J. Environ. Res. Public Health 2020, 17, 6557. [Google Scholar] [CrossRef]
- Cervera, M.A.; Soekadar, S.R.; Ushiba, J.; Millán, J.D.R.; Liu, M.; Birbaumer, N.; Garipelli, G. Brain-computer interfaces for post-stroke motor rehabilitation: A meta-analysis. Ann. Clin. Transl. Neurol. 2018, 5, 651–663. [Google Scholar] [CrossRef]
- Abiri, R.; Borhani, S.; Sellers, E.W.; Jiang, Y.; Zhao, X. A comprehensive review of EEG-based brain-computer interface paradigms. J. Neural Eng. 2019, 16, 011001. [Google Scholar] [CrossRef]
- Buch, E.; Weber, C.; Cohen, L.G.; Braun, C.; Dimyan, M.A.; Ard, T.; Mellinger, J.; Caria, A.; Soekadar, S.; Fourkas, A.; et al. Think to move: A neuromagnetic brain-computer interface (BCI) system for chronic stroke. Stroke 2008, 39, 910–917. [Google Scholar] [CrossRef]
- Khan, M.A.; Das, R.; Iversen, H.K.; Puthusserypady, S. Review on motor imagery based BCI systems for upper limb post-stroke neurorehabilitation: From designing to application. Comput. Biol. Med. 2020, 123, 103843. [Google Scholar] [CrossRef]
- Mane, R.; Chouhan, T.; Guan, C. BCI for stroke rehabilitation: Motor and beyond. J. Neural Eng. 2020, 17, 041001. [Google Scholar] [CrossRef]
- Laver, K.E.; George, S.; Thomas, S.; Deutsch, J.E.; Crotty, M. Virtual reality for stroke rehabilitation. Cochrane Database Syst. Rev. 2015, 2015, Cd008349. [Google Scholar] [CrossRef]
- Maier, M.; Rubio Ballester, B.; Duff, A.; Duarte Oller, E.; Verschure, P. Effect of Specific Over Nonspecific VR-Based Rehabilitation on Poststroke Motor Recovery: A Systematic Meta-analysis. Neurorehabil. Neural Repair 2019, 33, 112–129. [Google Scholar] [CrossRef]
- Cheung, K.L.; Tunik, E.; Adamovich, S.V.; Boyd, L.A. Neuroplasticity and virtual reality. In Virtual Reality for Physical and Motor Rehabilitation; Springer: New York, NY, USA, 2014; pp. 5–24. [Google Scholar]
- Ferreira Dos Santos, L.; Christ, O.; Mate, K.; Schmidt, H.; Krüger, J.; Dohle, C. Movement visualisation in virtual reality rehabilitation of the lower limb: A systematic review. Biomed. Eng. Online 2016, 15 (Suppl. S3), 144. [Google Scholar] [CrossRef]
- Modroño, C.; Bermúdez, S.; Cameirão, M.; Pereira, F.; Paulino, T.; Marcano, F.; Hernández-Martín, E.; Plata-Bello, J.; Palenzuela, N.; Núñez-Pádron, D.; et al. Is it necessary to show virtual limbs in action observation neurorehabilitation systems? J. Rehab Ass Tech. Eng. 2019, 6, 2055668319859140. [Google Scholar] [CrossRef]
- Cano Porras, D.; Sharon, H.; Inzelberg, R.; Ziv-Ner, Y.; Zeilig, G.; Plotnik, M. Advanced virtual reality-based rehabilitation of balance and gait in clinical practice. Ther. Adv. Chronic Dis. 2019, 10, 2040622319868379. [Google Scholar] [CrossRef]
- Song, R.; Grabowska, W.; Park, M.; Osypiuk, K.; Vergara-Diaz, G.P.; Bonato, P.; Hausdorff, J.M.; Fox, M.; Sudarsky, L.R.; Macklin, E.; et al. The impact of Tai Chi and Qigong mind-body exercises on motor and non-motor function and quality of life in Parkinson’s disease: A systematic review and meta-analysis. Parkinsonism Relat. Disord. 2017, 41, 3–13. [Google Scholar] [CrossRef]
- Petry-Schmelzer, J.N.; Krause, M.; Dembek, T.A.; Horn, A.; Evans, J.; Ashkan, K.; Rizos, A.; Silverdale, M.; Schumacher, W.; Sack, C.; et al. Non-motor outcomes depend on location of neurostimulation in Parkinson’s disease. Brain 2019, 142, 3592–3604. [Google Scholar] [CrossRef]
- Dafsari, H.S.; Silverdale, M.; Strack, M.; Rizos, A.; Ashkan, K.; Mahlstedt, P.; Sachse, L.; Steffen, J.; Dembek, T.A.; Visser-Vandewalle, V.; et al. Nonmotor symptoms evolution during 24 months of bilateral subthalamic stimulation in Parkinson’s disease. Mov. Disord. 2018, 33, 421–430. [Google Scholar] [CrossRef]
- Beyaert, C.; Vasa, R.; Frykberg, G.E. Gait post-stroke: Pathophysiology and rehabilitation strategies. Neurophysiol. Clin. 2015, 45, 335–355. [Google Scholar] [CrossRef]
- Bolognini, N.; Russo, C.; Edwards, D.J. The sensory side of post-stroke motor rehabilitation. Restor. Neurol. Neurosci. 2016, 34, 571–586. [Google Scholar] [CrossRef] [PubMed]
- Stein, C.; Fritsch, C.G.; Robinson, C.; Sbruzzi, G.; Plentz, R.D. Effects of Electrical Stimulation in Spastic Muscles After Stroke: Systematic Review and Meta-Analysis of Randomized Controlled Trials. Stroke 2015, 46, 2197–2205. [Google Scholar] [CrossRef]
- Khan, F.; Chevidikunnan, F. Theta burst stimulation a new paradigm of non-invasive brain stimulation for post-stroke upper limb motor rehabilitation. Turk. J. Phys. Med. Rehabil. 2017, 63, 193–196. [Google Scholar] [CrossRef]
- Coleman, E.R.; Moudgal, R.; Lang, K.; Hyacinth, H.I.; Awosika, O.O.; Kissela, B.M.; Feng, W. Early Rehabilitation After Stroke: A Narrative Review. Curr. Atheroscler. Rep. 2017, 19, 59. [Google Scholar] [CrossRef]
- Döbrössy, M.D.; Dunnett, S.B. Optimising plasticity: Environmental and training associated factors in transplant-mediated brain repair. Rev. Neurosci. 2005, 16, 1–21. [Google Scholar] [CrossRef]
- Aichner, F.; Adelwöhrer, C.; Haring, H.P. Rehabilitation approaches to stroke. In Stroke-Vascular Diseases; Springer: Vienna, Austria, 2002; pp. 59–73. [Google Scholar]
- Ward, N.S. Restoring brain function after stroke—Bridging the gap between animals and humans. Nat. Rev. Neurol. 2017, 13, 244–255. [Google Scholar] [CrossRef] [PubMed]
- Hicks, A.U.; Hewlett, K.; Windle, V.; Chernenko, G.; Ploughman, M.; Jolkkonen, J.; Weiss, S.; Corbett, D. Enriched environment enhances transplanted subventricular zone stem cell migration and functional recovery after stroke. Neuroscience 2007, 146, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.H.; Seo, J.H.; Lee, J.Y.; Lee, M.Y.; Cho, S.R. Induction of Neurorestoration From Endogenous Stem Cells. Cell Transpl. 2016, 25, 863–882. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Hu, X.; Zhang, L.; Li, L.; Zheng, H.; Li, M.; Zhang, Q. Physical exercise regulates neural stem cells proliferation and migration via SDF-1α/CXCR4 pathway in rats after ischemic stroke. Neurosci. Lett. 2014, 578, 203–208. [Google Scholar] [CrossRef]
- Komitova, M.; Zhao, L.R.; Gidö, G.; Johansson, B.B.; Eriksson, P. Postischemic exercise attenuates whereas enriched environment has certain enhancing effects on lesion-induced subventricular zone activation in the adult rat. Eur. J. Neurosci. 2005, 21, 2397–2405. [Google Scholar] [CrossRef]
- Hicks, A.U.; MacLellan, C.L.; Chernenko, G.A.; Corbett, D. Long-term assessment of enriched housing and subventricular zone derived cell transplantation after focal ischemia in rats. Brain Res. 2008, 1231, 103–112. [Google Scholar] [CrossRef]
- Tan, X.D.; Liu, B.; Jiang, Y.; Yu, H.J.; Li, C.Q. Gadd45b mediates environmental enrichment-induced neurogenesis in the SVZ of rats following ischemia stroke via BDNF. Neurosci. Lett. 2021, 745, 135616. [Google Scholar] [CrossRef]
- Park, H.; Poo, M. Neurotrophin regulation of neural circuit development and function. Nat. Rev. Neurosci. 2013, 14, 7–23. [Google Scholar] [CrossRef]
- Sasaki, Y.; Sasaki, M.; Kataoka-Sasaki, Y.; Nakazaki, M.; Nagahama, H.; Suzuki, J.; Tateyama, D.; Oka, S.; Namioka, T.; Namioka, A.; et al. Synergic Effects of Rehabilitation and Intravenous Infusion of Mesenchymal Stem Cells After Stroke in Rats. Phys. Ther. 2016, 96, 1791–1798. [Google Scholar] [CrossRef]
- Zha, K.; Yang, Y.; Tian, G.; Sun, Z.; Yang, Z.; Li, X.; Sui, X.; Liu, S.; Zhao, J.; Guo, Q. Nerve growth factor (NGF) and NGF receptors in mesenchymal stem/stromal cells: Impact on potential therapies. Stem Cells Transl. Med. 2021, 10, 1008–1020. [Google Scholar] [CrossRef]
- Gonçalves, L.V.; Herlinger, A.L.; Ferreira, T.A.A.; Coitinho, J.B.; Pires, R.G.W.; Martins-Silva, C. Environmental enrichment cognitive neuroprotection in an experimental model of cerebral ischemia: Biochemical and molecular aspects. Behav. Brain Res. 2018, 348, 171–183. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, D.; Qi, H.; Yuan, Y.; Liu, H.; Yao, S.; Yuan, S.; Zhang, J. Enriched environment promotes post-stroke neurogenesis through NF-κB-mediated secretion of IL-17A from astrocytes. Brain Res. 2018, 1687, 20–31. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.-X.; Yuan, M.-Z.; Cheng, L.; Lin, L.-Z.; Du, H.-W.; Chen, R.-H.; Liu, N. Treadmill exercise enhances therapeutic potency of transplanted bone mesenchymal stem cells in cerebral ischemic rats via anti-apoptotic effects. BMC Neurosci. 2015, 16, 56. [Google Scholar] [CrossRef]
- Curtis, M.A.; Eriksson, P.S.; Faull, R.L. Progenitor cells and adult neurogenesis in neurodegenerative diseases and injuries of the basal ganglia. Clin. Exp. Pharm. Physiol. 2007, 34, 528–532. [Google Scholar] [CrossRef]
- Lee, S.H.; Kim, Y.H.; Kim, Y.J.; Yoon, B.W. Enforced physical training promotes neurogenesis in the subgranular zone after focal cerebral ischemia. J. Neurol. Sci. 2008, 269, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Morishita, S.; Hokamura, K.; Yoshikawa, A.; Agata, N.; Tsutsui, Y.; Umemura, K.; Kumada, T. Different exercises can modulate the differentiation/maturation of neural stem/progenitor cells after photochemically induced focal cerebral infarction. Brain Behav. 2020, 10, e01535. [Google Scholar] [CrossRef]
- Akhoundzadeh, K.; Vakili, A.; Sameni, H.R.; Vafaei, A.A.; Rashidy-Pour, A.; Safari, M.; Mohammadkhani, R. Effects of the combined treatment of bone marrow stromal cells with mild exercise and thyroid hormone on brain damage and apoptosis in a mouse focal cerebral ischemia model. Metab. Brain Dis. 2017, 32, 1267–1277. [Google Scholar] [CrossRef] [PubMed]
- Scutelnic, A.; Heldner, M.R. Vascular Events, Vascular Disease and Vascular Risk Factors-Strongly Intertwined with COVID-19. Curr. Treat. Options Neurol. 2020, 22, 40. [Google Scholar] [CrossRef]
| Type of Intervention (Exercise/Stem Cells/Exercise + Stem Cells) | Title, Author, Year | Stem Cell Variety | Significant Findings |
|---|---|---|---|
| Exercise | Enforced physical training promotes neurogenesis in the subgranular zone after focal cerebral ischemia [277] | NSCs | Enforced physical training promotes neurogenesis in the SGZ after focal cerebral ischemia [277] |
| Exercise | Physical exercise regulates neural stem cells proliferation and migration via SDF-1α/CXCR4 pathway in rats after ischemic stroke [266] | NSCs | Exercise improved functional recovery by increasing NSC proliferation, migration from the SVZ and differentiation in the damaged striatum of MCAO occluded rats [266] |
| Exercise | Postischemic exercise attenuates whereas enriched environment has certain enhancing effects on lesion-induced subventricular zone activation in the adult rat [267] | NSCs | Exercise modulated the stroke induced increase in neural stem cell proliferation in the SVZ early after cortical infarction [267] |
| Exercise | Different exercises can modulate the differentiation/maturation of neural stem/progenitor cells after photochemically induced focal cerebral infarction [278] | NSCs | Exercise improved neuronal maturation and increased generation of endogenous NSCs [278] |
| Stem cell transplantation | Stem cell-paved biobridge facilitates neural repair in traumatic brain injury [45] | MSC | MSCs aided endogenous NSCs to the area of infarction, improved behavioral outcomes [45] |
| Stem cell exosome transplantation | Enhancement of angiogenesis and neurogenesis by intracerebroventricular injection of secretome from human embryonic stem cell-derived mesenchymal stem cells in ischemic stroke model [55] | MSC | MSCs transplantation suppresses inflammation, reduces cell death, promotes angiogenesis, and stimulates neurogenesis [55] |
| Stem cell transplantation | Activated Mesenchymal Stem Cells Induce Recovery Following Stroke Via Regulation of Inflammation and Oligodendrogenesis [56] | MSCs | MSCs lower overall inflammation, ameliorate potentially toxic environments, and increase neurotrophic factor release, enabling both endogenous NSC survival and function [56] |
| Exercise and stem cell transplantation | Treadmill exercise enhances therapeutic potency of transplanted bone mesenchymal stem cells in cerebral ischemic rats via anti-apoptotic effects [275] | MSCs | Treadmill exercise enhances the therapeutic potency of MSCs by improving neurological function and possibly inhibiting the apoptosis of neuron cells and transplanted MSCs [275] |
| Exercise and stem cell transplantation | Synergic Effects of Rehabilitation and Intravenous Infusion of Mesenchymal Stem Cells After Stroke in Rats [262] | MSCs | Both combined therapy and MSC infusion reduced lesion volume, induced synaptogenesis, and elicited functional improvement compared with the groups without MSC infusion, but the effect was greater in the combined therapy group [262] |
| Exercise and stem cell transplantation | Effects of the combined treatment of bone marrow stromal cells with mild exercise and thyroid hormone on brain damage and apoptosis in a mouse focal cerebral ischemia model [279] | MSCs | Decrease in infarct volume and decrease in apoptosis [279] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Berlet, R.; Anthony, S.; Brooks, B.; Wang, Z.-J.; Sadanandan, N.; Shear, A.; Cozene, B.; Gonzales-Portillo, B.; Parsons, B.; Salazar, F.E.; et al. Combination of Stem Cells and Rehabilitation Therapies for Ischemic Stroke. Biomolecules 2021, 11, 1316. https://doi.org/10.3390/biom11091316
Berlet R, Anthony S, Brooks B, Wang Z-J, Sadanandan N, Shear A, Cozene B, Gonzales-Portillo B, Parsons B, Salazar FE, et al. Combination of Stem Cells and Rehabilitation Therapies for Ischemic Stroke. Biomolecules. 2021; 11(9):1316. https://doi.org/10.3390/biom11091316
Chicago/Turabian StyleBerlet, Reed, Stefan Anthony, Beverly Brooks, Zhen-Jie Wang, Nadia Sadanandan, Alex Shear, Blaise Cozene, Bella Gonzales-Portillo, Blake Parsons, Felipe Esparza Salazar, and et al. 2021. "Combination of Stem Cells and Rehabilitation Therapies for Ischemic Stroke" Biomolecules 11, no. 9: 1316. https://doi.org/10.3390/biom11091316
APA StyleBerlet, R., Anthony, S., Brooks, B., Wang, Z.-J., Sadanandan, N., Shear, A., Cozene, B., Gonzales-Portillo, B., Parsons, B., Salazar, F. E., Lezama Toledo, A. R., Monroy, G. R., Gonzales-Portillo, J. V., & Borlongan, C. V. (2021). Combination of Stem Cells and Rehabilitation Therapies for Ischemic Stroke. Biomolecules, 11(9), 1316. https://doi.org/10.3390/biom11091316








