Can Antiviral Activity of Licorice Help Fight COVID-19 Infection?
Abstract
1. Introduction
2. From Traditional to Medicinal Use of Licorice Extract
3. Antiviral Effects
| Virus Family | Virus | Study | Compound Tested | Effective Concentration | Effects | Ref | |
|---|---|---|---|---|---|---|---|
| Hepadnaviridae | Hepatitis A | In vitro PLC/PRF/5 cells | GR | 0.25–2 mg/mL | Reduction of antigen expression and virus infectivity | [27,28,29] | |
| Humans Patients with acute autoimmune hepatitis | SNMC | 200 mg/day i.v. x 4 weeks | Improvement of transaminases, prevention of disease progression | [30,31,32] | |||
| Hepatitis B | In vitro Rat hepatocytes | GR | 0.08 mg/mL | Suppression of transaminases | [33] | ||
| PLC/PRF/5 cells | GR | Not known | Suppression of surface antigen | [33] | |||
| PLC/PRF/5 cells | GR | 1–2.5 mg/mL | Suppression of surface antigen | [32] | |||
| GA | 0.5 mg/mL | ||||||
| In vivo Guinea pigs | GR | 3 or 67 mg/kg b.w. i.v | Suppression of surface antigen, reduction of transaminases | [34] | |||
| Humans Patients with chronic hepatitis and liver cirrhosis | SNMC | 120–160 mg/day i.v. 3 times/week for 4–36 months | Normalization of transaminases Reduction of viral load | [35,36] | |||
| Patients with chronic hepatitis and acute exacer-bation | SNMC | 200 mg/day i.v. × 5 days | Improvement of transaminases | [37] | |||
| Hepatitis C | Humans Patients with chronic hepatitis | SNMC | 80 mg/day i.v. × 4 weeks | Improvement of transaminases | [38] | ||
| Patients with chronic hepatitis | SNMC | 15 mg/day p.o. × 90 days | Improvement of liver function | [33] | |||
| Patients with chronic hepatitis | SNMC | 200 mg/day i.v. × 8 weeks | Improvement of transaminases and liver pathological features | [39] | |||
| Patients with chronic hepatitis and liver cirrhosis | SNMC | 120–160 mg/day i.v. 3 times/week for 4−36 months | Normalization of transaminases Reduced viral load | [40] | |||
| Retrospective study on patients with chronic hepatitis | SNMC | 200 mg/day i.v. × 8 weeks then 2–7 times a week × 2–16 years | Normalization of transaminases, reduced risk of hepatocellular carcinoma | [41] | |||
| Patients with chronic hepatitis | SNMC | 80 mg/day i.v. × 4 weeksor 200 mg/day i.v. × 8 weeks | Normalization of transaminases, reduced cirrhosis, reduced risk of hepatocellular carcinoma | [42] | |||
| Phase I/II study in patients with chronic hepatitis | SNMC | 200 mg i.v. 6 times a week × 4 weeks or 200 mg i.v. 6 times a week × 26 weeks | Improvement of biochemical and histological parameters, improvement quality | [43] | |||
| Retrospective study on patients with chronic hepatitis | SNMC | 200 mg/day i.v. 3 times a week × 4.3 years | Normalization of transaminases, reduced risk of hepatocellular carcinoma | [43] | |||
| Herpesviridae | Herpes simplex (HSV-1) | In vitro HEp2 cells | GR | 0.8–6.7 mg/mL | Inhibition of virus growth | [17] | |
| Vero cells | GR | 0.75 mg/mL | Inhibition of virus replication | [20] | |||
| Vero cells | Aqueous G. glabra extract | 2 mg/mL | Inhibition of virus entry | [21,22] | |||
| Aqueous G. glabra extract Alkaline extract | 0.3–3 mg/mL | Inhibition of virus growth | [22] | ||||
| Epstein-Barr | Raji cells | GR GA | 0.7–3 mg/mL | Inhibition of virus growth | [23] | ||
| Pseudorabies | Vero cells | GR | >0.3 mg/mL | Inhibition of virus growth | [19] | ||
| GA | >3 mg/mL | ||||||
| Varicella-zoster | Vero cells | Diammonium glycyrrhizinate | 0.025 mg/mL | Inhibition of virus replication | [24] | ||
| 0.005 mg/mL | |||||||
| GR in licorice powder extract | 0.01–1.25 mg/mL | Inhibition of virus growth and infectivity | [19] | ||||
| 0.125 mg/mL | Inhibition of virus growth | [24] | |||||
| Paramixoviridae | Newcastle disease | In vitro HEp2 cells | GR | 0.8–6.7 mg/mL | Inhibition of virus growth | [17] | |
| 0.01–0.3 mg/mL | Inhibition of virus growth, increase in IFN production | ||||||
| Human respiratory syncytial | HEp-2 cells A549 cells | Aqueous G. uralensis extract GA | 0.001–0.01 mg/mL | Inhibition of virus growth | [25] | ||
| Rhabdoviridae | Vesicular stomatitis | In vitro HEp2 cells | GA | 0.8–6.7 mg/mL | Inhibition of virus growth | [17] | |
| Flaviviridae | Japanese encephalitis | In vitro PS cells | GA | 1–2 mg/mL | Inhibition of virus growth and replication | [26] | |
| Vero cells | GR | 0.38 mg/mL | Inhibition of virus replication | ||||
| Dengue | In vitro Vero cells | GA | 0.01–0.1 mg/mL | Inhibition of virus replication | [44] | ||
| Vero cells | GA | 0.1–0.6 mg/mL | Reduction of infection | ||||
| West Nile | In vitro Vero cells | GR | 0.2 mg/mL | Inhibition of virus replication | [44] | ||
| Yellow fever | In vitro Vero cells | GR | 0.45 mg/mL | Inhibition of virus replication | [44] | ||
| Orthomyxoviridae | Influenza A | In vitro Hep2 cells | GR | 0.8–6.7 mg/mL | Inhibition of virus growth | [17,45,46] | |
| MDCK cells | GR | 0.25–1 mg/mL | Reduction of infectivity | [47] | |||
| A549 cells MDCK cells HFL-1 cells | GR | 0.4–0.8 mg/mL | Inhibition of virus replication | [48] | |||
| MDCK cells | GR conjugated with aromatic amino acids methyl ester | 0.2 mg/mL | Reduction of infectivity | [47] | |||
| GR conjugated with S-benzyl-cysteine | 0.2 mg/mL | ||||||
| In vivo Mice: C3H, ddY, CDF-1, C57BL, BALB/c, and athymic nude mice | GR | 20 mg/kg b.w i.v | Reduction of infectivity | [48] | |||
| 50 mg/kg b.w. i.p. | Increased IFN production | ||||||
| Influenza and upper respiratory tract infection | Human hospitalized patients | GR | 80 mg i.v infusion | Reduced hospitalization, body temperature lower 24 to 48 h after admission | [49,50] | ||
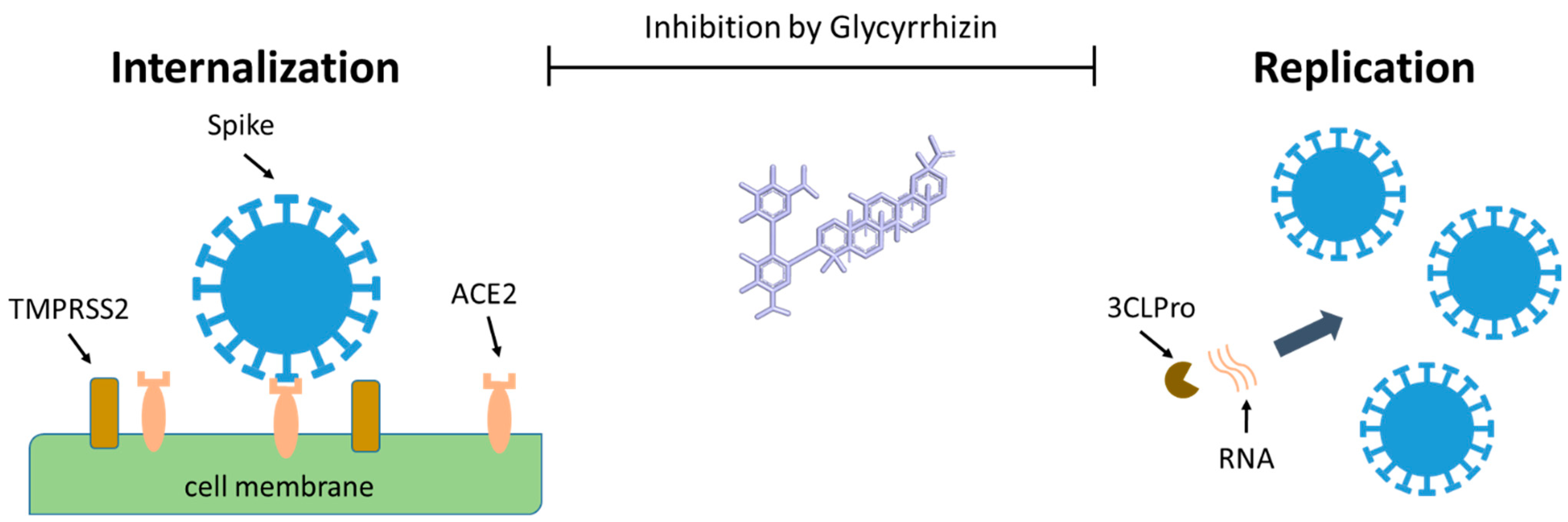
| Coronaviridae | SARS | In vitro Vero cells | GR | 0.3–4 mg/mL | Inhibition of virus replication, adsorption, and penetration | [6,7] | |
| Vero cells fRhK-4 cells | GR | 0.1 mg/mL>0.4 mg/mL | Inhibition of viral growth | ||||
| Avian infectious bronchitis | In vitro Vero cells | GR diammonium | 0.08–0.6 mg/mL | Inhibition viral growth | [7,51] | ||
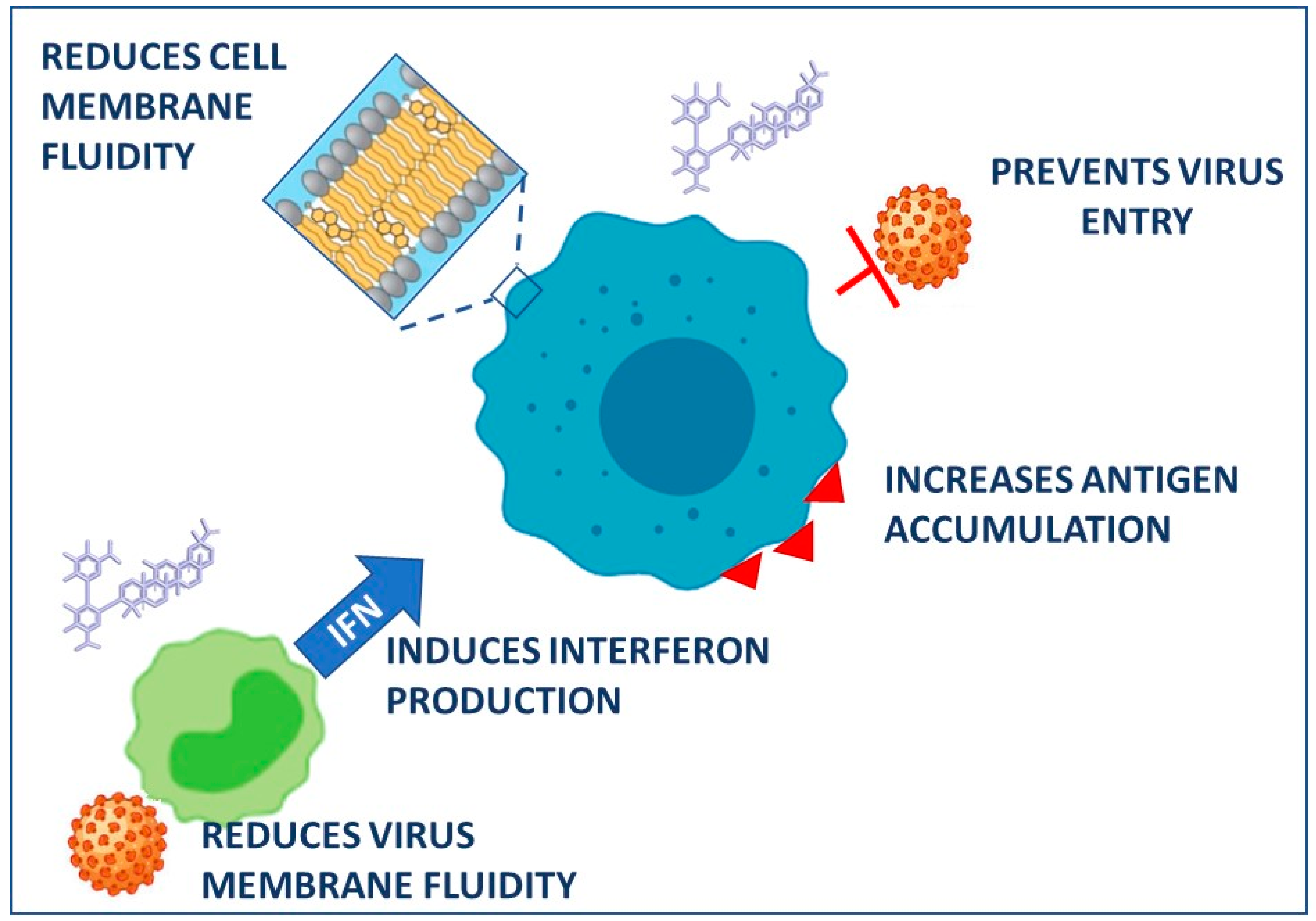
| Retroviridae | HIV | In vitro MT-2, MT-4 cells | GR | 0.1–1 mg/mL | Reduction of cellular and viral membrane fluidity; reduction of infectivity; inhibition of cell-to-cell | [45] | |
| GR | 0.01–1 mg/mL | Inhibition of virus growth | |||||
| GA | 0.01 mg/mL | ||||||
| MT-4 cells | Aqueous G. glabra extract | 0.3–0.5 mg/mL | Inhibition of virus growth | [52] | |||
| Alkaline extract | 0.2–0.05 mg/mL | Inhibition of virus growth | |||||
| MT-4 cells | GR | 0.4 mg/mL | Inhibition of virus growth | [22] | |||
| GA | >0.5 mg/mL | Inhibition of virus growth | |||||
| Reoviridae | Porcino rotavirus | In vivo colostrum-deprived piglets, 3 days of age | Methanol G. uralensis extract | 400 mg/mL p.o. 4 times/day | Cure of diarrhea, improvement of small intestinal lesions and fecal virus shedding, reduction of intestinalinflammation | [53] | |
| Arteriviridaein | Porcine reproductive and respiratory syndrome | In vitro MARC-145 cells | GR | 0.5–0.7 mg/mL | Reduction of virus penetration and proliferation | [54] | |
| Birnaviridae | Infectious bursal disease | In vivo chicken: 24-days old brown chicks | Glycyrrhizinate dipotassium | 20–80 mg/kg b.w. p.o. × 5 days | Increased immunological responses, increased IFN levels, inhibition of infection | [55] | |
4. Effects on COVID-19 Infection
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| GR | Glycyrrhizin |
| GA | Glycyrrhetinic acid |
| G. glabra | Glycyrrhiza glabra |
| G. uralensis | Glycyrrhiza uralensis |
| HIV | Human immunodeficiency virus |
| HIV-1 | Human immunodeficiency type-1 virus |
| SARS | Severe acute respiratory syndrome |
| SNMC | Stronger Neo-Minophagen C |
| HSV-1 | Herpes simplex virus type 1 |
| IFN | Interferon |
| ACE2 | Angiotensin-converting enzyme 2 |
| TLR4 | Toll-like receptor 4 |
| IL | Interleukin |
| TMPRSS2 | Transmembrane serine protease 2 |
| RBD | Receptor binding domain |
| 3CLpro | 3-chymotrypsin-like cysteine protease |
| SPR | Surface Plasmon Resonance |
| GRAS | Generally Recognized as Safe |
| FDA U.S. | Food and Drug Administration |
References
- Fiore, C.; Eisenhut, M.; Ragazzi, E.; Zanchin, G.; Armanini, D. A History of the Therapeutic Use of Liquorice in Europe. J. Ethnopharmacol. 2005, 99, 317–324. [Google Scholar] [CrossRef]
- Baltina, L.A.; Kondratenko, R.M.; Baltina, L.A.; Plyasunova, O.A.; Pokrovskii, A.G.; Tolstikov, G.A. Prospects for the Creation of New Antiviral Drugs Based on Glycyrrhizic Acid and Its Derivatives (a Review). Pharm. Chem. J. 2009, 43, 539–548. [Google Scholar] [CrossRef] [PubMed]
- Asl, M.N.; Hosseinzadeh, H. Review of Pharmacological Effects of Glycyrrhiza sp. and Its Bioactive Compounds. Phytother. Res. PTR 2008, 22, 709–724. [Google Scholar] [CrossRef]
- Pastorino, G.; Cornara, L.; Soares, S.; Rodrigues, F.; Oliveira, M.B.P.P. Liquorice (Glycyrrhiza Glabra): A Phytochemical and Pharmacological Review. Phytother. Res. 2018, 32, 2323–2339. [Google Scholar] [CrossRef] [PubMed]
- Fiore, C.; Dalbrenta, D.V.; Ragazzi, E.; Pellati, D.; Armanini, D. A Hypothesis on the Death of the Greek Philosopher Heraclitus. J. Endocrinol. Investig. 2008, 31, 742–743. [Google Scholar] [CrossRef] [PubMed]
- Cinatl, J.; Morgenstern, B.; Bauer, G.; Chandra, P.; Rabenau, H.; Doerr, H.W. Glycyrrhizin, an Active Component of Liquorice Roots, and Replication of SARS-Associated Coronavirus. Lancet 2003, 361, 2045–2046. [Google Scholar] [CrossRef]
- Chen, F.; Chan, K.H.; Jiang, Y.; Kao, R.Y.T.; Lu, H.T.; Fan, K.W.; Cheng, V.C.C.; Tsui, W.H.W.; Hung, I.F.N.; Lee, T.S.W.; et al. In Vitro Susceptibility of 10 Clinical Isolates of SARS Coronavirus to Selected Antiviral Compounds. J. Clin. Virol. 2004, 31, 69–75. [Google Scholar] [CrossRef] [PubMed]
- 21 CFR 184—Direct Food Substances Affirmed as Generally Recognized as Safe—Content Details—CFR-2011-Title21-Vol3-Part184. Available online: https://www.govinfo.gov/app/details/CFR-2011-title21-vol3/CFR-2011-title21-vol3-part184 (accessed on 18 March 2021).
- CFR—Code of Federal Regulations Title 21; Part 310; New Drugs. Available online: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?CFRPart=310 (accessed on 18 March 2021).
- Joint FAO/WHO Expert Committee on Food Additives. Evaluation of Certain Food Additives: Sixty-Third Report of the Joint FAO/WHO Expert Committee on Food Additives; World Health Organization: Geneva, Switzerland, 2005; ISBN 9789241209281. [Google Scholar]
- Delbò, R.M. Assessment Report on Glycyrrhiza glabra L. and/or Glycyrrhiza Inflata Bat and/or Glycyrrhiza uralensis Fisch, Radix; European Medicines Agency: Amsterdam, The Netherlands, 2013; 40p. [Google Scholar]
- Van Rossum, T.G.J.; Vulto, A.G.; Hop, W.C.J.; Schalm, S.W. Pharmacokinetics of Intravenous Glycyrrhizin after Single and Multiple Doses in Patients with Chronic Hepatitis C Infection. Clin. Ther. 1999, 21, 2080–2090. [Google Scholar] [CrossRef]
- Krähenbühl, S.; Hasler, F.; Krapf, R. Analysis and Pharmacokinetics of Glycyrrhizic Acid and Glycyrrhetinic Acid in Humans and Experimental Animals. Steroids 1994, 59, 121–126. [Google Scholar] [CrossRef]
- De Groot, G.; Koops, R.; Hogendoorn, E.A.; Goewie, C.E.; Savelkoul, T.J.; van Vloten, P. Improvement of Selectivity and Sensitivity by Column Switching in the Determination of Glycyrrhizin and Glycyrrhetic Acid in Human Plasma by High-Performance Liquid Chromatography. J. Chromatogr. 1988, 456, 71–81. [Google Scholar] [CrossRef]
- Yamamura, Y.; Kawakami, J.; Santa, T.; Kotaki, H.; Uchino, K.; Sawada, Y.; Tanaka, N.; Iga, T. Pharmacokinetic Profile of Glycyrrhizin in Healthy Volunteers by a New High-Performance Liquid Chromatographic Method. J. Pharm. Sci. 1992, 81, 1042–1046. [Google Scholar] [CrossRef] [PubMed]
- Carlat, L.E.; Margraf, H.W.; Weathers, H.H.; Weichselbaum, T.E. Human Metabolism of Orally Ingested Glycyrrhetinic Acid and Monoammonium Glycyrrhizinate. Proc. Soc. Exp. Biol. Med. 1959, 102, 245–248. [Google Scholar] [CrossRef] [PubMed]
- Pompei, R.; Flore, O.; Marccialis, M.A.; Pani, A.; Loddo, B. Glycyrrhizic Acid Inhibits Virus Growth and Inactivates Virus Particles. Nature 1979, 281, 689–690. [Google Scholar] [CrossRef] [PubMed]
- Fiore, C.; Eisenhut, M.; Krausse, R.; Ragazzi, E.; Pellati, D.; Armanini, D.; Bielenberg, J. Antiviral Effects of Glycyrrhiza Species. Phytother. Res. 2008, 22, 141–148. [Google Scholar] [CrossRef]
- Sui, X.; Yin, J.; Ren, X. Antiviral Effect of Diammonium Glycyrrhizinate and Lithium Chloride on Cell Infection by Pseudorabies Herpesvirus. Antiviral Res. 2010, 85, 346–353. [Google Scholar] [CrossRef]
- Lampis, G.; Deidda, D.; Pinza, M.; Pompei, R. Enhancement of Anti-Herpetic Activity of Glycyrrhizic Acid by Physiological Proteins. Antivir. Chem. Chemother. 2001, 12, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Sabouri Ghannad, M.; Mohammadi, A.; Safiallahy, S.; Faradmal, J.; Azizi, M.; Ahmadvand, Z. The Effect of Aqueous Extract of Glycyrrhiza Glabra on Herpes Simplex Virus 1. Jundishapur J. Microbiol. 2014, 7. [Google Scholar] [CrossRef]
- Fukuchi, K.; Okudaira, N.; Adachi, K.; Odai-Ide, R.; Watanabe, S.; Ohno, H.; Yamamoto, M.; Kanamoto, T.; Terakubo, S.; Nakashima, H.; et al. Antiviral and Antitumor Activity of Licorice Root Extracts. Athens Greece 2016, 30, 777–785. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.-C.; Cherng, J.-M.; Hung, M.-S.; Baltina, L.A.; Baltina, L.; Kondratenko, R. Inhibitory Effects of Some Derivatives of Glycyrrhizic Acid against Epstein-Barr Virus Infection: Structure–Activity Relationships. Antiviral Res. 2008, 79, 6–11. [Google Scholar] [CrossRef] [PubMed]
- Shebl, R.I.; Amin, M.A.; Emad-Eldin, A.; Bin Dajem, S.M.; Mostafa, A.S.; Ibrahim, E.H.; Mohamed, A.F. Antiviral Activity of Liquorice Powder Extract against Varicella Zoster Virus Isolated from Egyptian Patients. Chang Gung Med. J. 2012, 35, 231–239. [Google Scholar] [CrossRef]
- Feng Yeh, C.; Wang, K.C.; Chiang, L.C.; Shieh, D.E.; Yen, M.H.; San Chang, J. Water Extract of Licorice Had Anti-Viral Activity against Human Respiratory Syncytial Virus in Human Respiratory Tract Cell Lines. J. Ethnopharmacol. 2013, 148, 466–473. [Google Scholar] [CrossRef]
- Badam, L. Ammonium Salt of Glycyrrhizic Acid as an Antiviral. Natl. Med. J. India 1997, 10, 98. [Google Scholar]
- Crance, J.M.; Biziagos, E.; Passagot, J.; van Cuyck-Gandré, H.; Deloince, R. Inhibition of Hepatitis A Virus Replication In Vitro by Antiviral Compounds. J. Med. Virol. 1990, 31, 155–160. [Google Scholar] [CrossRef]
- Yasui, S.; Fujiwara, K.; Tawada, A.; Fukuda, Y.; Nakano, M.; Yokosuka, O. Efficacy of Intravenous Glycyrrhizin in the Early Stage of Acute Onset Autoimmune Hepatitis. Dig. Dis. Sci. 2011, 56, 3638–3647. [Google Scholar] [CrossRef] [PubMed]
- Crance, J.M.; Lévêque, F.; Biziagos, E.; van Cuyck-Gandré, H.; Jouan, A.; Deloince, R. Studies on Mechanism of Action of Glycyrrhizin against Hepatitis A Virus Replication in Vitro. Antiviral Res. 1994, 23, 63–76. [Google Scholar] [CrossRef]
- Shiki, Y.; Shirai, K.; Saito, Y.; Yoshida, S.; Mori, Y.; Wakashin, M. Effect of Glycyrrhizin on Lysis of Hepatocyte Membranes Induced by Anti-Liver Cell Membrane Antibody. J. Gastroenterol. Hepatol. 1992, 7, 12–16. [Google Scholar] [CrossRef]
- Takahara, T.; Watanabe, A.; Shiraki, K. Effects of Glycyrrhizin on Hepatitis B Surface Antigen: A Biochemical and Morphological Study. J. Hepatol. 1994, 21, 601–609. [Google Scholar] [CrossRef]
- Sato, H.; Goto, W.; Yamamura, J.; Kurokawa, M.; Kageyama, S.; Takahara, T.; Watanabe, A.; Shiraki, K. Therapeutic Basis of Glycyrrhizin on Chronic Hepatitis B. Antiviral Res. 1996, 30, 171–177. [Google Scholar] [CrossRef]
- Wildhirt, E. Experience in Germany with Glycyrrhizinic Acid for the Treatment of Chronic Viral Hepatitis. In Viral Hepatitis and Liver Disease; Nishioka, K., Suzuki, H., Mishiro, S., Oda, T., Eds.; Springer: Tokyo, Japan, 1994; pp. 658–661. [Google Scholar]
- Lin, C.-C.; Wang, P.-H. Intravenous Glycyrrhizin Improved Serum Transaminases Rapidly in a Chronic Hepatitis B Patient with Acute Exacerbation. J. Formos. Med. Assoc. Taiwan Yi Zhi 2015, 114, 188–189. [Google Scholar] [CrossRef]
- Suzuki, H.; Fujisawa, K.; Ohta, Y.; Takino, T.; Hirayama, C.; Shimizu, N.; Aso, Y. The Therapeutic Effects of Stronger Neo Minophage. Igaku No Ayumi 1977, 102, 562–568. [Google Scholar]
- Suzuki, H.; Ohta, Y.; Takino, T.; Fujisawa, K.; Hirayama, C. Effects of Glycyrrhizin on Biochemical Tests in Patients with Chronic Hepatitis. Double Blind Trial. Asian Med. J. 1983, 26, 423–438. [Google Scholar]
- Su, X.S.; Chen, H.M.; Wang, L.H.; Jiang, C.F.; Liu, J.H.; Zhao, M.Q.; Ma, X.H.; Zhao, Y.C.; Han, D.W. Clinical and Laboratory Observation on the Effect of Glycyrrhizin in Acute and Chronic Viral Hepatitis. J. Tradit. Chin. Med. Chung Tsa Chih Ying Wen Pan 1984, 4, 127–132. [Google Scholar]
- Hino, K. Effects of Glycyrrhizin Therapy on Liver Histology in Chronic Aggressive Hepatitis. New Trends in Peptic Ulcer and Chronic Hepatitis. Excerpta Med. 1987, 295–303. [Google Scholar]
- Arase, Y.; Ikeda, K.; Murashima, N.; Chayama, K.; Tsubota, A.; Koida, I.; Suzuki, Y.; Saitoh, S.; Kobayashi, M.; Kumada, H. The Long Term Efficacy of Glycyrrhizin in Chronic Hepatitis C Patients. Cancer 1997, 79, 1494–1500. [Google Scholar] [CrossRef]
- Kumada, H. Long-Term Treatment of Chronic Hepatitis C with Glycyrrhizin (Stronger Neo-Minophagen C (SNMC)) for Preventing Liver Cirrhosis and Hepatocellular Carcinoma. Oncology 2002, 62 (Suppl. S1), 94–100. [Google Scholar] [CrossRef]
- Orlent, H.; Hansen, B.E.; Willems, M.; Brouwer, J.T.; Huber, R.; Kullak-Ublick, G.A.; Gerken, G.; Zeuzem, S.; Nevens, F.; Tielemans, W.C.M.; et al. Biochemical and Histological Effects of 26 Weeks of Glycyrrhizin Treatment in Chronic Hepatitis C: A Randomized Phase II Trial. J. Hepatol. 2006, 45, 539–546. [Google Scholar] [CrossRef]
- Ikeda, K.; Arase, Y.; Kobayashi, M.; Saitoh, S.; Someya, T.; Hosaka, T.; Sezaki, H.; Akuta, N.; Suzuki, Y.; Suzuki, F.; et al. A Long-Term Glycyrrhizin Injection Therapy Reduces Hepatocellular Carcinogenesis Rate in Patients with Interferon-Resistant Active Chronic Hepatitis C: A Cohort Study of 1249 Patients. Dig. Dis. Sci. 2006, 51, 603–609. [Google Scholar] [CrossRef] [PubMed]
- Van Rossum, T.G.; Vulto, A.G.; de Man, R.A.; Brouwer, J.T.; Schalm, S.W. Review Article: Glycyrrhizin as a Potential Treatment for Chronic Hepatitis C. Aliment. Pharmacol. Ther. 1998, 12, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Crance, J.M.; Scaramozzino, N.; Jouan, A.; Garin, D. Interferon, Ribavirin, 6-Azauridine and Glycyrrhizin: Antiviral Compounds Active against Pathogenic Flaviviruses. Antivir. Res. 2003, 58, 73–79. [Google Scholar] [CrossRef]
- Harada, S. The Broad Anti-Viral Agent Glycyrrhizin Directly Modulates the Fluidity of Plasma Membrane and HIV-1 Envelope. Biochem. J. 2005, 392, 191–199. [Google Scholar] [CrossRef]
- Wolkerstorfer, A.; Kurz, H.; Bachhofner, N.; Szolar, O.H.J. Glycyrrhizin Inhibits Influenza A Virus Uptake into the Cell. Antivir. Res. 2009, 83, 171–178. [Google Scholar] [CrossRef] [PubMed]
- Baltina, L.A.; Zarubaev, V.V.; Baltina, L.A.; Orshanskaya, I.A.; Fairushina, A.I.; Kiselev, O.I.; Yunusov, M.S. Glycyrrhizic Acid Derivatives as Influenza A/H1N1 Virus Inhibitors. Bioorg. Med. Chem. Lett. 2015, 25, 1742–1746. [Google Scholar] [CrossRef]
- Abe, N.; Ebina, T.; Ishida, N. Interferon Induction by Glycyrrhizin and Glycyrrhetinic Acid in Mice. Microbiol. Immunol. 1982, 26, 535–539. [Google Scholar] [CrossRef] [PubMed]
- Yanagawa, Y.; Ogura, M.; Fujimoto, E.; Shono, S.; Okuda, E. Effects and Cost of Glycyrrhizin in the Treatment of Upper Respiratory Tract Infections in Members of the Japanese Maritime Self-Defense Force: Preliminary Report of a Prospective, Randomized, Double-Blind, Controlled, Parallel-Group, Alternate-Day Treatment Assignment Clinical Trial. Curr. Ther. Res. Clin. Exp. 2004, 65, 26–33. [Google Scholar] [CrossRef][Green Version]
- Mousa, H.A.-L. Prevention and Treatment of Influenza, Influenza-Like Illness, and Common Cold by Herbal, Complementary, and Natural Therapies. J. Evid. Based Complement. Altern. Med. 2017, 22, 166–174. [Google Scholar] [CrossRef]
- De Clercq, E. Potential Antivirals and Antiviral Strategies against SARS Coronavirus Infections. Expert Rev. Anti-Infect. Ther. 2006, 4, 291–302. [Google Scholar] [CrossRef]
- Baltina, L.A.; Stolyarova, O.V.; Baltina, L.A.; Kondratenko, R.M.; Plyasunova, O.A.; Pokrovskii, A.G. Synthesis and Antiviral Activity of 18α-Glycyrrhizic Acid and Its Esters. Pharm. Chem. J. 2010, 44, 299–302. [Google Scholar] [CrossRef]
- Alfajaro, M.M.; Kim, H.-J.; Park, J.-G.; Ryu, E.-H.; Kim, J.-Y.; Jeong, Y.-J.; Kim, D.-S.; Hosmillo, M.; Son, K.-Y.; Lee, J.-H.; et al. Anti-Rotaviral Effects of Glycyrrhiza Uralensis Extract in Piglets with Rotavirus Diarrhea. Virol. J. 2012, 9, 310. [Google Scholar] [CrossRef]
- Duan, E.; Wang, D.; Fang, L.; Ma, J.; Luo, J.; Chen, H.; Li, K.; Xiao, S. Suppression of Porcine Reproductive and Respiratory Syndrome Virus Proliferation by Glycyrrhizin. Antivir. Res. 2015, 120, 122–125. [Google Scholar] [CrossRef] [PubMed]
- Sun, N. Antiviral and Immunomodulatory Effects of Dipotassium Glycyrrhizinate in Chicks Artificially Infected with Infectious Bursal Disease Virus. Pak. Vet. J. 2019, 39, 43–48. [Google Scholar] [CrossRef]
- Yamamoto, S. Treatment of Hepatitis with the Antiallergic Drug, Stronger Neo-Minophagen C. Clin. Med. Pedatr. 1958, 13, 73–75. [Google Scholar]
- Bailly, C.; Vergoten, G. Glycyrrhizin: An Alternative Drug for the Treatment of COVID-19 Infection and the Associated Respiratory Syndrome? Pharmacol. Ther. 2020, 214, 107618. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, Y.; Matsuura, T.; Aoyagi, H.; Matsuda, M.; Hmwe, S.S.; Date, T.; Watanabe, N.; Watashi, K.; Suzuki, R.; Ichinose, S.; et al. Antiviral Activity of Glycyrrhizin against Hepatitis C Virus In Vitro. PLoS ONE 2013, 8, e68992. [Google Scholar] [CrossRef]
- Brendler, T.; Al-Harrasi, A.; Bauer, R.; Gafner, S.; Hardy, M.L.; Heinrich, M.; Hosseinzadeh, H.; Izzo, A.A.; Michaelis, M.; Nassiri-Asl, M.; et al. Botanical Drugs and Supplements Affecting the Immune Response in the Time of COVID-19: Implications for Research and Clinical Practice. Phytother. Res. 2020. [Google Scholar] [CrossRef]
- Wu, J.; Deng, W.; Li, S.; Yang, X. Advances in Research on ACE2 as a Receptor for 2019-NCoV. Cell. Mol. Life Sci. CMLS 2021, 78, 531–544. [Google Scholar] [CrossRef]
- Chen, H.; Du, Q. Potential Natural Compounds for Preventing SARS-CoV-2 (2019-NCoV) Infection. Preprints 2020. [Google Scholar] [CrossRef]
- Chen, X.; Wu, Y.; Chen, C.; Gu, Y.; Zhu, C.; Wang, S.; Chen, J.; Zhang, L.; Lv, L.; Zhang, G.; et al. Identifying Potential Anti-COVID-19 Pharmacological Components of Traditional Chinese Medicine Lianhuaqingwen Capsule Based on Human Exposure and ACE2 Biochromatography Screening. Acta Pharm. Sin. B 2021, 11, 222–236. [Google Scholar] [CrossRef]
- Mahdian, S.; Ebrahim-Habibi, A.; Zarrabi, M. Drug Repurposing Using Computational Methods to Identify Therapeutic Options for COVID-19. J. Diabetes Metab. Disord. 2020, 1–9. [Google Scholar] [CrossRef]
- Yu, S.; Zhu, Y.; Xu, J.; Yao, G.; Zhang, P.; Wang, M.; Zhao, Y.; Lin, G.; Chen, H.; Chen, L.; et al. Glycyrrhizic Acid Exerts Inhibitory Activity against the Spike Protein of SARS-CoV-2. Phytomedicine 2020, 153364. [Google Scholar] [CrossRef]
- Zígolo, M.A.; Goytia, M.R.; Poma, H.R.; Rajal, V.B.; Irazusta, V.P. Virtual Screening of Plant-Derived Compounds against SARS-CoV-2 Viral Proteins Using Computational Tools. Sci. Total Environ. 2021, 781, 146400. [Google Scholar] [CrossRef]
- Carino, A.; Moraca, F.; Fiorillo, B.; Marchianò, S.; Sepe, V.; Biagioli, M.; Finamore, C.; Bozza, S.; Francisci, D.; Distrutti, E.; et al. Hijacking SARS-CoV-2/ACE2 Receptor Interaction by Natural and Semi-Synthetic Steroidal Agents Acting on Functional Pockets on the Receptor Binding Domain. Front. Chem. 2020, 8. [Google Scholar] [CrossRef]
- Das, S.; Sarmah, S.; Lyndem, S.; Roy, A.S. An Investigation into the Identification of Potential Inhibitors of SARS-CoV-2 Main Protease Using Molecular Docking Study. J. Biomol. Struct. Dyn. 2020, 1–11. [Google Scholar] [CrossRef]
- Diniz, L.R.L.; Perez-Castillo, Y.; Elshabrawy, H.A.; Filho, C.d.S.M.B.; de Sousa, D.P. Bioactive Terpenes and Their Derivatives as Potential SARS-CoV-2 Proteases Inhibitors from Molecular Modeling Studies. Biomolecules 2021, 11. [Google Scholar] [CrossRef]
- Koulgi, S.; Jani, V.; Uppuladinne, M.; Sonavane, U.; Nath, A.K.; Darbari, H.; Joshi, R. Drug Repurposing Studies Targeting SARS-CoV-2: An Ensemble Docking Approach on Drug Target 3C-like Protease (3CLpro). J. Biomol. Struct. Dyn. 2020, 1–21. [Google Scholar] [CrossRef]
- Quan Pham, M.; Vu, K.B.; Pham, T.N.H.; Huong, L.T.T.; Hoang Tran, L.; Thanh Tung, N.; Vu, V.V.; Hai Nguyen, T.; Tung Ngo, S. Rapid Prediction of Possible Inhibitors for SARS-CoV-2 Main Protease Using Docking and FPL Simulations. RSC Adv. 2020, 10, 31991–31996. [Google Scholar] [CrossRef]
- Rai, H.; Barik, A.; Singh, Y.P.; Suresh, A.; Singh, L.; Singh, G.; Nayak, U.Y.; Dubey, V.K.; Modi, G. Molecular Docking, Binding Mode Analysis, Molecular Dynamics, and Prediction of ADMET/Toxicity Properties of Selective Potential Antiviral Agents against SARS-CoV-2 Main Protease: An Effort toward Drug Repurposing to Combat COVID-19. Mol. Divers. 2021. [Google Scholar] [CrossRef]
- Sampangi-Ramaiah, M.H.; Vishwakarma, R.; Shaanker, R.U. Molecular Docking Analysis of Selected Natural Products from Plants for Inhibition of SARS-CoV-2 Main Protease. Curr. Sci. 2020, 118, 6. [Google Scholar]
- Shawky, E.; Nada, A.A.; Ibrahim, R.S. Potential Role of Medicinal Plants and Their Constituents in the Mitigation of SARS-CoV-2: Identifying Related Therapeutic Targets Using Network Pharmacology and Molecular Docking Analyses. RSC Adv. 2020, 10, 27961–27983. [Google Scholar] [CrossRef]
- Srivastava, V.; Yadav, A.; Sarkar, P. Molecular Docking and ADMET Study of Bioactive Compounds of Glycyrrhiza Glabra against Main Protease of SARS-CoV2. Mater. Today Proc. 2020. [Google Scholar] [CrossRef]
- Günther, S.; Reinke, P.Y.A.; Fernández-García, Y.; Lieske, J.; Lane, T.J.; Ginn, H.M.; Koua, F.H.M.; Ehrt, C.; Ewert, W.; Oberthuer, D.; et al. X-Ray Screening Identifies Active Site and Allosteric Inhibitors of SARS-CoV-2 Main Protease. Science 2021. [Google Scholar] [CrossRef]
- De Oliveira, O.V.; Rocha, G.B.; Paluch, A.S.; Costa, L.T. Repurposing Approved Drugs as Inhibitors of SARS-CoV-2 S-Protein from Molecular Modeling and Virtual Screening. J. Biomol. Struct. Dyn. 2020, 1–10. [Google Scholar] [CrossRef]
- Jin, Z.; Du, X.; Xu, Y.; Deng, Y.; Liu, M.; Zhao, Y.; Zhang, B.; Li, X.; Zhang, L.; Peng, C.; et al. Structure of M pro from SARS-CoV-2 and Discovery of Its Inhibitors. Nature 2020, 582, 289–293. [Google Scholar] [CrossRef]
- Van de Sand, L.; Bormann, M.; Alt, M.; Schipper, L.; Heilingloh, C.S.; Todt, D.; Dittmer, U.; Elsner, C.; Witzke, O.; Krawczyk, A. Glycyrrhizin Effectively Neutralizes SARS-CoV-2 in Vitro by Inhibiting the Viral Main Protease. BioRxiv 2020. [Google Scholar] [CrossRef]
- Boozer, C.; Kim, G.; Cong, S.; Guan, H.; Londergan, T. Looking towards Label-Free Biomolecular Interaction Analysis in a High-Throughput Format: A Review of New Surface Plasmon Resonance Technologies. Curr. Opin. Biotechnol. 2006, 17, 400–405. [Google Scholar] [CrossRef]
- Feng, S.; Luan, X.; Wang, Y.; Wang, H.; Zhang, Z.; Wang, Y.; Tian, Z.; Liu, M.; Xiao, Y.; Zhao, Y.; et al. Eltrombopag Is a Potential Target for Drug Intervention in SARS-CoV-2 Spike Protein. Infect. Genet. Evol. 2020, 85, 104419. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Mcpartlon, M.; Li, J. Improved Protein Structure Prediction by Deep Learning Irrespective of Co-Evolution Information. BioRxiv 2020. [Google Scholar] [CrossRef]
- Murck, H. Symptomatic Protective Action of Glycyrrhizin (Licorice) in COVID-19 Infection? Front. Immunol. 2020, 11. [Google Scholar] [CrossRef]
- Janes, J.; Young, M.E.; Chen, E.; Rogers, N.H.; Burgstaller-Muehlbacher, S.; Hughes, L.D.; Love, M.S.; Hull, M.V.; Kuhen, K.L.; Woods, A.K.; et al. The ReFRAME Library as a Comprehensive Drug Repurposing Library and Its Application to the Treatment of Cryptosporidiosis. Proc. Natl. Acad. Sci. USA 2018, 115, 10750–10755. [Google Scholar] [CrossRef]
- Bakowski, M.A.; Beutler, N.; Chen, E.; Nguyen, T.-T.H.; Kirkpatrick, M.G.; Parren, M.; Yang, L.; Ricketts, J.; Gupta, A.K.; Hull, M.V.; et al. Oral Drug Repositioning Candidates and Synergistic Remdesivir Combinations for the Prophylaxis and Treatment of COVID-19. BioRxiv 2020. [Google Scholar] [CrossRef]
- Riva, L.; Yuan, S.; Yin, X.; Martin-Sancho, L.; Matsunaga, N.; Pache, L.; Burgstaller-Muehlbacher, S.; De Jesus, P.D.; Teriete, P.; Hull, M.V.; et al. Discovery of SARS-CoV-2 Antiviral Drugs through Large-Scale Compound Repurposing. Nature 2020, 1–11. [Google Scholar] [CrossRef]
- Chrzanowski, J.; Chrzanowska, A.; Graboń, W. Glycyrrhizin: An Old Weapon against a Novel Coronavirus. Phytother. Res. 2020, 35. [Google Scholar] [CrossRef] [PubMed]
- Safa, O.; Hassani-Azad, M.; Farashahinejad, M.; Davoodian, P.; Dadvand, H.; Hassanipour, S.; Fathalipour, M. Effects of Licorice on Clinical Symptoms and Laboratory Signs in Moderately Ill Patients with Pneumonia from COVID-19: A Structured Summary of a Study Protocol for a Randomized Controlled Trial. Trials 2020, 21, 790. [Google Scholar] [CrossRef] [PubMed]
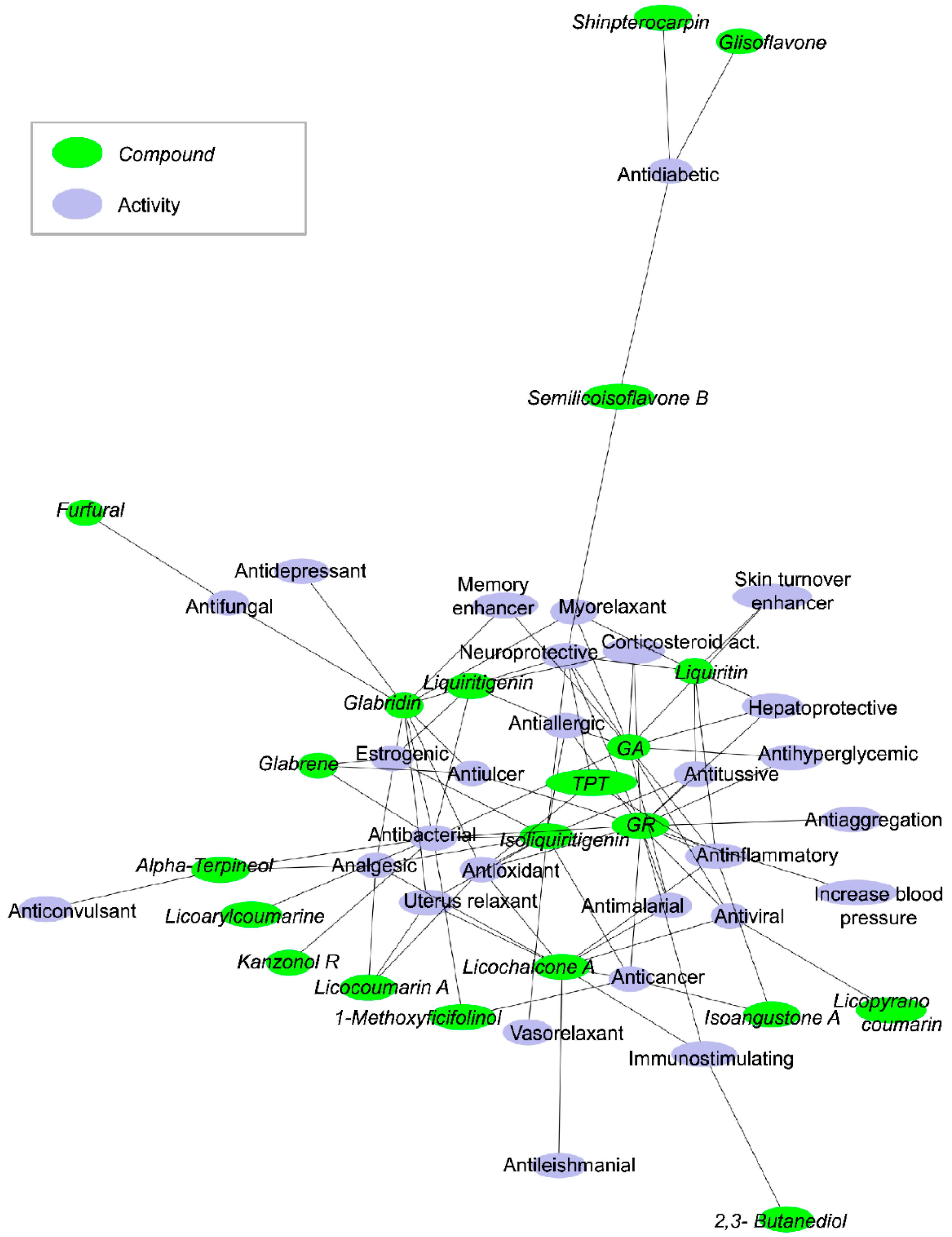
| Glycyrrhizin (Glycyrrhizic acid) CAS: 1405-86-3 | 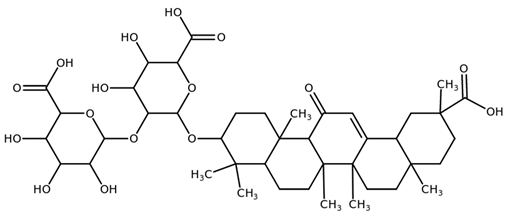 |
| Glycyrrhetinic acid (Enoxolone) CAS: 471-53-4 | 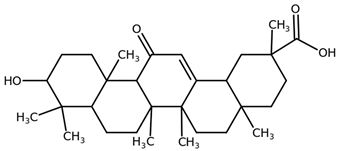 |
| Isoliquiritigenin CAS: 961-29-5 |  |
| Licochalcone A CAS: 58749-22-7 |  |
| Liquiritigenin CAS: 578-86-9 | 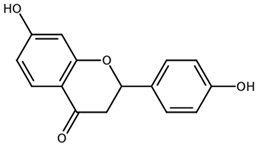 |
| Liquiritin CAS: 551-15-5 |  |
| Licoflavone B CAS: 91433-17-9 | 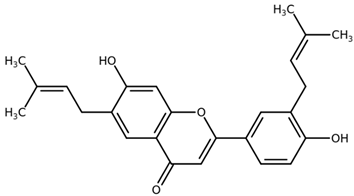 |
| Glisoflavone CAS: 125709-32-2 |  |
| Isoangustone A CAS: 129280-34-8 |  |
| Semilicoisoflavone B CAS: 129280-33-7 | 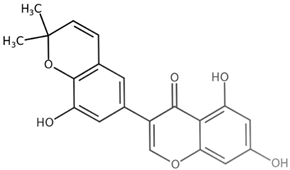 |
| Shinpterocarpin CAS: 157414-04-5 |  |
| 1-Methoxyficifolinol CHEBI: 69096 |  |
| Licoriphenone CAS: 129280-36-0 | 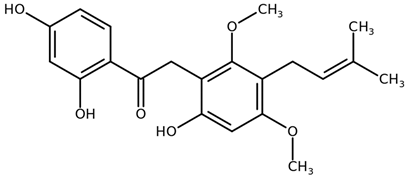 |
| Glabridin CAS: 59870-68-7 |  |
| Glabrene CAS: 60008-03-9 |  |
| Kanzonol R CAS: 156250-73-6 |  |
| Licopyranocoumarin CAS: 117038-80-9 | 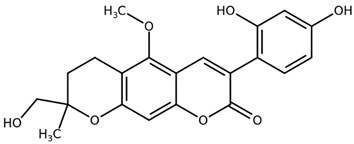 |
| Licoarylcoumarin CAS: 125709-31-1 | 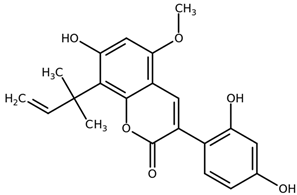 |
| Licocoumarin A |  |
| Tetramethyl pyrazine-2,3,5,6-tetracarboxylate CAS: 35042-21-8 | 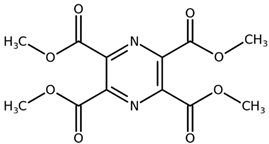 |
| Alpha-terpineol CAS: 98-55-5 | 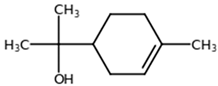 |
| Furfural CAS: 98-01-1 |  |
| 2,3-Butanediol CAS: 513-85-9 | 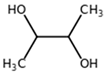 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Diomede, L.; Beeg, M.; Gamba, A.; Fumagalli, O.; Gobbi, M.; Salmona, M. Can Antiviral Activity of Licorice Help Fight COVID-19 Infection? Biomolecules 2021, 11, 855. https://doi.org/10.3390/biom11060855
Diomede L, Beeg M, Gamba A, Fumagalli O, Gobbi M, Salmona M. Can Antiviral Activity of Licorice Help Fight COVID-19 Infection? Biomolecules. 2021; 11(6):855. https://doi.org/10.3390/biom11060855
Chicago/Turabian StyleDiomede, Luisa, Marten Beeg, Alessio Gamba, Oscar Fumagalli, Marco Gobbi, and Mario Salmona. 2021. "Can Antiviral Activity of Licorice Help Fight COVID-19 Infection?" Biomolecules 11, no. 6: 855. https://doi.org/10.3390/biom11060855
APA StyleDiomede, L., Beeg, M., Gamba, A., Fumagalli, O., Gobbi, M., & Salmona, M. (2021). Can Antiviral Activity of Licorice Help Fight COVID-19 Infection? Biomolecules, 11(6), 855. https://doi.org/10.3390/biom11060855







