Sweet Modifications Modulate Plant Development
Abstract
1. Plant Development and Sugars
1.1. Plant Development in a Nut Shell
1.2. Carbohydrate Classes: Free Saccharides and Glycoconjugates
1.3. Protein Glycosylation
1.3.1. N-Glycosylation
- Stepwise assembly of the Glc3Man9GlcNAc2-Dol-PP precursor build-up from Glc, mannose (Man), N-acetylglucosamine (GlcNAc) and a dolichol pyrophosphate (Dol-PP) lipid membrane anchor, in the cytosol and ER lumen [71];
- En bloc transfer of the N-glycan precursor to synthesized proteins in the ER lumen, through the oligosaccharyltransferase (OST) complex, which involves the actual transfer of the precursor to a protein, co-translationally, thus creating a glycoprotein [72];
- Processing of the N-glycan and channeling of correctly folded glycoproteins towards the cis-side of the Golgi apparatus whereas incorrectly folded glycoproteins, in case re-glycosylation did not yield a properly folded glycoprotein, are directed towards the ER-Associated Degradation (ERAD) pathway [65];
- Further processing, and modifications and maturation of the N-glycans occurs enzymatically, by the successive action of multiple GTs and GHs, while the glycoprotein is transported from the cis-Golgi to the cisternal trans-Golgi. Modifications include Man trimming as well as the addition of monosaccharides. Man trimming results in the removal of two to three Man residues from the high-Man N-glycans, creating oligo-Man N-glycans with five (Man5) or six (Man6) residues [65,73]. The addition of nucleotide-coupled monosaccharides like fucose (Fuc), xylose (Xyl) or GlcNAc to the pentavalent or hexavalent mannose structures gives rise to the typical plant complex and hybrid N-glycans [46,74,75]. However, the hybrid type is rather rare in plants [44];
- Glycoproteins continue their way along the secretory pathway and end up in the vacuole, plasma membrane or get secreted extracellularly with possible additional modifications, such as vacuolar glycan trimming, giving rise to typical truncated vacuolar (i.e., paucimannosidic) N-glycans [76,77,78,79].
1.3.2. O-Glycosylation
1.3.3. C-, P- and S-Glycosylation
1.4. Glycolipids
1.5. Nucleotide Sugars as Glycan Precursors
2. Developmental Consequences of Glycosylation: From Flowers to Germinating Seeds
2.1. Flowers Have a Sweet Tooth
2.1.1. Flower Reproductive Organs Rely on Glycans
2.1.2. Sugars: Aphrodisiacs for Plants?
2.2. Eradication of Sweet Cell Walls Mediates Fruit Ripening
2.2.1. N-Glycans Control Fruit Ripening
2.2.2. Glycoproteins, Polysaccharides and Fruit Ripening
2.3. The Sugar-Craving Cell Wall of Seeds
2.3.1. The Ever-Changing Cell Wall
2.3.2. Cellulose Biosynthesis-Related Problems Cause Cell Wall Disruptions
2.4. Glycosylation Decides over Seed Germination
2.4.1. (De)Glycosylation and Seed Germination
2.4.2. Glycan Maturation in the Golgi Apparatus Affects Seed Germination
2.4.3. O-GlcNAc Modification on Cytosolic Proteins Is Important for Seed Germination
2.4.4. Glycosylation of Endosperm Glycoproteins Influences Seed Morphology and Germination
2.4.5. Glycosylated Small Molecules and Their Role in Germination
3. Glycoproteins during Root and Leaf Development
3.1. The Deal with Glycosylation in Roots
3.1.1. N-Glycans: Influencers of Root Development
3.1.2. Impaired O-Glycosylation Affects the Root Tips and Root Hairs
3.1.3. Biotic Interactions at the Root Require Some Sweet Regulation
3.2. Phenotypical Disturbances in Leaves
3.2.1. Abnormal O-Glycosylation: Leaves in Distress
3.2.2. Specialized Leaf Tissues Are Annoyed by Absent Glycosylation
4. Functionality of Protein Glycosylation
5. Glycolipids and Plant Development
5.1. Roles of Glycoglycerolipids and Lipid Profile Alterations
5.2. Glycosylated Protein-Lipid Anchors: Glycophosphatidylinositols
5.3. Glycosphingolipids: Sugar Coating the ‘Sphinx’
6. Sugar Signaling as the Puppet Master of Plant Development
6.1. Sugar Signaling through the SnRK1/TOR Nexus
6.2. Developmental Regulation by Sugar Signaling
6.3. Interplay between Sugar Signaling and Glycoconjugates
7. Perspectives
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Liu, C.; Xi, W.; Shen, L.; Tan, C.; Yu, H. Regulation of floral patterning by flowering time genes. Dev. Cell 2009, 16, 711–722. [Google Scholar] [CrossRef] [PubMed]
- De Smet, I.; Vanneste, S.; Inze, D.; Beeckman, T. Lateral root initiation or the birth of a new meristem. Plant Mol. Biol. 2006, 60, 871–887. [Google Scholar] [CrossRef] [PubMed]
- Efroni, I.; Eshed, Y.; Lifschitz, E. Morphogenesis of simple and compound leaves: A critical review. Plant Cell 2010, 22, 1019–1032. [Google Scholar] [CrossRef] [PubMed]
- Eveland, A.L.; Jackson, D.P. Sugars, signalling, and plant development. J. Exp. Bot. 2012, 63, 3367–3377. [Google Scholar] [CrossRef]
- Van den Ende, W. Sugars take a central position in plant growth, development and, stress responses. A focus on apical dominance. Front. Plant Sci. 2014, 5, 313. [Google Scholar] [CrossRef]
- Narayanan, S.; Zoong-Lwe, Z.S.; Gandhi, N.; Welti, R.; Fallen, B.; Smith, J.R.; Rustgi, S. Comparative Lipidomic Analysis Reveals Heat Stress Responses of Two Soybean Genotypes Differing in Temperature Sensitivity. Plants 2020, 9, 457. [Google Scholar] [CrossRef] [PubMed]
- Strasser, R.; Seifert, G.J.; Doblin, M.S.; Johnson, K.L.; Ruprecht, C.; Pfrengle, F.; Bacic, A.; Estevez, J.M. Cracking the “Sugar Code”: A Snapshot of N- and O-Glycosylation Pathways and Functions in Plants Cells. Front. Plant Sci. 2021, 12. [Google Scholar] [CrossRef]
- Ciereszko, I. Regulatory roles of sugars in plant growth and development. Acta Soc. Bot. Pol. 2018, 87. [Google Scholar] [CrossRef]
- De Smet, I.; Lau, S.; Mayer, U.; Jurgens, G. Embryogenesis—The humble beginnings of plant life. Plant J. 2010, 61, 959–970. [Google Scholar] [CrossRef]
- Johnson, R.R. Embryogenesis. In Encyclopedia of Applied Plant Sciences; Elsevier: Amsterdam, The Netherlands, 2017; pp. 490–496. ISBN 9780123948083. [Google Scholar]
- Goldberg, R.B.; de Paiva, G.; Yadegari, R. Plant Embryogenesis: Zygote to Seed. Science 1994, 266, 605–614. [Google Scholar] [CrossRef]
- Kanei, M.; Horiguchi, G.; Tsukaya, H. Stable establishment of cotyledon identity during embryogenesis in Arabidopsis by ANGUSTIFOLIA3 and HANABA TARANU. Development 2012, 139, 2436–2446. [Google Scholar] [CrossRef] [PubMed]
- Manfre, A.J.; LaHatte, G.A.; Climer, C.R.; Marcotte, W.R. Seed dehydration and the establishment of desiccation tolerance during seed maturation is altered in the Arabidopsis thaliana mutant atem6-1. Plant Cell Physiol. 2009, 50, 243–253. [Google Scholar] [CrossRef] [PubMed]
- Tuan, P.A.; Kumar, R.; Rehal, P.K.; Toora, P.K.; Ayele, B.T. Molecular Mechanisms Underlying Abscisic Acid/Gibberellin Balance in the Control of Seed Dormancy and Germination in Cereals. Front. Plant Sci. 2018, 9, 668. [Google Scholar] [CrossRef] [PubMed]
- Balanza, V.; Roig-Villanova, I.; Di Marzo, M.; Masiero, S.; Colombo, L. Seed abscission and fruit dehiscence required for seed dispersal rely on similar genetic networks. Development 2016, 143, 3372–3381. [Google Scholar] [CrossRef] [PubMed]
- Spence, J.; Vercher, Y.; Gates, P.; Harris, N. ‘Pod shatter’ in Arabidopsis thaliana, Brassica napus and B. juncea. J. Microsc. 1996, 181, 195–203. [Google Scholar] [CrossRef]
- Nonogaki, M.; Nonogaki, H. Germination. In Encyclopedia of Applied Plant Sciences; Elsevier: Amsterdam, The Netherlands, 2017; pp. 509–512. ISBN 9780123948083. [Google Scholar]
- Leah, R.; Kigel, J.; Svendsen, I.; Mundy, J. Biochemical and Molecular Characterization of a Barley Seed β-Glucosidase. J. Biol. Chem. 1995, 270, 15789–15797. [Google Scholar] [CrossRef]
- Leubner-Metzger, G.; Meins, F. Sense transformation reveals a novel role for class I b-1,3-glucanase in tobacco seed germination. Plant J. 2000, 23, 215–221. [Google Scholar] [CrossRef]
- Schiltz, S.; Gaillard, I.; Pawlicki-Jullian, N.; Thiombiano, B.; Mesnard, F.; Gontier, E. A review: What is the spermosphere and how can it be studied? J. Appl. Microbiol. 2015, 119, 1467–1481. [Google Scholar] [CrossRef] [PubMed]
- Cosgrove, D.J. Relaxation in a high-stress environment: The molecular bases of extensible cell walls and cell enlargement. Plant Cell 1997, 9, 1031–1041. [Google Scholar] [CrossRef]
- Yue, J.-H.; Zhang, D.; Ren, L.; Shen, X.-H. Gibberellin and auxin signals control scape cell elongation and proliferation in Agapanthus praecox ssp. orientalis. J. Plant Biol. 2016, 59, 358–368. [Google Scholar] [CrossRef]
- Kutschera, U.; Köhler, K. Cell elongation, turgor and osmotic pressure in developing sunflower hypocotyls. J. Exp. Bot. 1994, 45, 591–595. [Google Scholar] [CrossRef]
- Seifert, G.J. On the Potential Function of Type II Arabinogalactan O-Glycosylation in Regulating the Fate of Plant Secretory Proteins. Front. Plant Sci. 2020, 11, 563735. [Google Scholar] [CrossRef] [PubMed]
- Deepak, S.; Shailasree, S.; Kini, R.K.; Muck, A.; Mithöfer, A.; Shetty, S.H. Hydroxyproline-rich Glycoproteins and Plant Defence. J. Phytopathol. 2010, 585–593. [Google Scholar] [CrossRef]
- Chaliha, C.; Rugen, M.D.; Field, R.A.; Kalita, E. Glycans as Modulators of Plant Defense Against Filamentous Pathogens. Front. Plant Sci. 2018, 9, 928. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Noël, G.M.A.; Tognetti, J.A. Sugar Signaling Under Abiotic Stress in Plants. In Plant Metabolites and Regulation Under Environmental Stress; Ahmad, P., Ahanger, M.A., Singh, V.P., Tripathi, D.K., Alam, P., Alyemeni, M.N., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 397–406. ISBN 9780128126899. [Google Scholar]
- Höfte, H.; Voxeur, A. Plant cell walls. Curr. Biol. 2017, 27, 865–870. [Google Scholar] [CrossRef]
- Cosgrove, D.J. Diffuse Growth of Plant Cell Walls. Plant Physiol. 2018, 176, 16–27. [Google Scholar] [CrossRef]
- Harris, P.J.; McQueen-Mason, S.; Darley, C.; Roberts, P.; Jones, L. Cell Growth. In Encyclopedia of Applied Plant Sciences; Elsevier: Amsterdam, The Netherlands, 2017; pp. 155–160. ISBN 9780123948083. [Google Scholar]
- Lashbrook, C.C.; Gonzalez-Bosch, C.; Bennett, A.B. Two divergent endo-beta-1,4-glucanase genes exhibit overlapping expression in ripening fruit and abscising flowers. Plant Cell 1994, 6, 1485–1493. [Google Scholar] [CrossRef][Green Version]
- Nicol, F. A plasma membrane-bound putative endo-1,4-beta -D-glucanase is required for normal wall assembly and cell elongation in Arabidopsis. EMBO J. 1998, 17, 5563–5576. [Google Scholar] [CrossRef]
- Minic, Z.; Jouanin, L. Plant glycoside hydrolases involved in cell wall polysaccharide degradation. Plant Physiol. Biochem. 2006, 44, 435–449. [Google Scholar] [CrossRef]
- Minic, Z. Physiological roles of plant glycoside hydrolases. Planta 2008, 227, 723–740. [Google Scholar] [CrossRef]
- Hong, S.-B.; Sexton, R.; Tucker, M.L. Analysis of Gene Promoters for Two Tomato Polygalacturonases Expressed in Abscission Zones and the Stigma. Plant Physiol. 2000, 123, 869–882. [Google Scholar] [CrossRef] [PubMed]
- Fulton, L.M.; Cobbett, C.S. Two alpha-L-arabinofuranosidase genes in Arabidopsis thaliana are differentially expressed during vegetative growth and flower development. J. Exp. Bot. 2003, 54, 2467–2477. [Google Scholar] [CrossRef]
- Xie, R.J.; Deng, L.; Jing, L.; He, S.L.; Ma, Y.T.; Yi, S.L.; Zheng, Y.Q.; Zheng, L. Recent advances in molecular events of fruit abscission. Biol. Plant. 2013, 57, 201–209. [Google Scholar] [CrossRef]
- Cosgrove, D.J. Plant cell wall extensibility: Connecting plant cell growth with cell wall structure, mechanics, and the action of wall-modifying enzymes. J. Exp. Bot. 2016, 67, 463–476. [Google Scholar] [CrossRef] [PubMed]
- Prestegard, J.H.; Liu, J.; Widmalm, G. Oligosaccharides and Polysaccharides. In Essentials of Glycobiology; Varki, A., Cummings, R.D., Esko, J.D., Stanley, P., Hart, G.W., Aebi, M., Darvill, A.G., Kinoshita, T., Packer, N.H., Prestegard, J.H., et al., Eds.; Cold Spring Harbor: New York, NY, USA, 2017; Volume 3. [Google Scholar]
- Streb, S.; Zeeman, S.C. Starch Metabolism in Arabidopsis. In The Arabidopsis Book; BioOne Complete: Washington, DC, USA, 2012. [Google Scholar]
- Rolland, F.; Baena-Gonzalez, E.; Sheen, J. Sugar sensing and signaling in plants: Conserved and Novel Mechanisms. Annu. Rev. Plant Biol. 2006, 57, 675–709. [Google Scholar] [CrossRef] [PubMed]
- Bolouri Moghaddam, M.R.; Van den Ende, W. Sweet immunity in the plant circadian regulatory network. J. Exp. Bot. 2013, 64, 1439–1449. [Google Scholar] [CrossRef]
- Janse van Rensburg, H.C.; Takács, Z.; Freynschlag, F.; Öner, E.T.; Jonak, C.; Van den Ende, W. Fructans Prime ROS Dynamics and Botrytis cinerea Resistance in Arabidopsis. Antioxidants 2020, 9, 805. [Google Scholar] [CrossRef]
- Lannoo, N.; Van Damme, E.J.M. Review/N-glycans: The making of a varied toolbox. Plant Sci. 2015, 239, 67–83. [Google Scholar] [CrossRef] [PubMed]
- Corfield, A. Eukaryotic protein glycosylation: A primer for histochemists and cell biologists. Histochem. Cell Biol. 2017, 147, 119–147. [Google Scholar] [CrossRef]
- Stanley, P.; Taniguchi, N.; Aebi, M. N-glycans. In Essentials of Glycobiology; Varki, A., Cummings, R.D., Esko, J.D., Stanley, P., Hart, G.W., Aebi, M., Darvill, A.G., Kinoshita, T., Packer, N.H., Prestegard, J.H., et al., Eds.; Cold Spring Harbor Laboratory Press: New York, NY, USA, 2017; Volume 3. [Google Scholar]
- Apweiler, R.; Hermjakob, H.; Sharon, N. On the frequency of protein glycosylation, as deduced from analysis of the SWISS-PROT database. Biochim. Biophys. Acta 1999, 1473, 4–8. [Google Scholar] [CrossRef]
- Ying, J.; Zhao, J.; Hou, Y.; Wang, Y.; Qiu, J.; Li, Z.; Tong, X.; Shi, Z.; Zhu, J.; Zhang, J. Mapping the N-linked glycosites of rice (Oryza sativa L.) germinating embryos. PLoS ONE 2017, 12, e0173853. [Google Scholar] [CrossRef]
- Nguema-Ona, E.; Vicre-Gibouin, M.; Gotte, M.; Plancot, B.; Lerouge, P.; Bardor, M.; Driouich, A. Cell wall O-glycoproteins and N-glycoproteins: Aspects of biosynthesis and function. Front. Plant Sci. 2014, 5, 499. [Google Scholar] [CrossRef]
- Gupta, R.; Leon, F.; Rauth, S.; Batra, S.K.; Ponnusamy, M.P. A Systematic Review on the Implications of O-linked Glycan Branching and Truncating Enzymes on Cancer Progression and Metastasis. Cells 2020, 9, 446. [Google Scholar] [CrossRef] [PubMed]
- Lombard, J. The multiple evolutionary origins of the eukaryotic N-glycosylation pathway. Biol. Direct 2016, 11, 36. [Google Scholar] [CrossRef] [PubMed]
- Chung, C.Y.; Majewska, N.I.; Wang, Q.; Paul, J.T.; Betenbaugh, M.J. SnapShot: N-Glycosylation Processing Pathways across Kingdoms. Cell 2017, 171, 258–258.e1. [Google Scholar] [CrossRef] [PubMed]
- Lannoo, N.; Van Damme, E.J.; Albenne, C.; Jamet, E. Plant Glycobiology-a diverse world of lectins, glycoproteins, glycolipids and glycans. Front. Plant Sci. 2014, 5, 604. [Google Scholar] [CrossRef]
- Mathieu-Rivet, E.; Mati-Baouche, N.; Walet-Balieu, M.L.; Lerouge, P.; Bardor, M. N- and O-Glycosylation Pathways in the Microalgae Polyphyletic Group. Front. Plant Sci. 2020, 11, 609993. [Google Scholar] [CrossRef]
- Behnke, J.; Cohen, A.M.; LaRoche, J. N-linked glycosylation enzymes in the diatom Thalassiosira oceanica exhibit a diel cycle in transcript abundance and favor for NXT-type sites. Nat. Sci. Rep. 2021, 11, 3227. [Google Scholar] [CrossRef]
- Defaus, S.; Gupta, P.; Andreu, D.; Gutierrez-Gallego, R. Mammalian protein glycosylation--structure versus function. Analyst 2014, 139, 2944–2967. [Google Scholar] [CrossRef]
- Walski, T.; De Schutter, K.; Van Damme, E.J.M.; Smagghe, G. Diversity and functions of protein glycosylation in insects. Insect Biochem. Mol. Biol. 2017, 83, 21–34. [Google Scholar] [CrossRef]
- Kajiura, H.; Miyauchi, R.; Kakudo, A.; Ohashi, T.; Misaki, R.; Fujiyama, K. Bombyx mori β1,4-N-acetylgalactosaminyltransferase possesses relaxed donor substrate specificity in N-glycan synthesis. Nat. Sci. Rep. 2021, 11. [Google Scholar] [CrossRef]
- Deshpande, N.; Wilkins, M.R.; Packer, N.; Nevalainen, H. Protein glycosylation pathways in filamentous fungi. Glycobiology 2008, 18, 626–637. [Google Scholar] [CrossRef] [PubMed]
- Free, S.J. Fungal Cell Wall Organization and Biosynthesis. In Advances in Genetics; Elsevier: Amsterdam, The Netherlands, 2013; pp. 33–82. ISBN 0065-2660. [Google Scholar]
- Nothaft, H.; Szymanski, C.M. Protein glycosylation in bacteria: Sweeter than ever. Nat. Rev. Microbiol. 2010, 8, 765–778. [Google Scholar] [CrossRef] [PubMed]
- Lu, Q.; Li, S.; Shao, F. Sweet Talk: Protein Glycosylation in Bacterial Interaction With the Host. Trends Microbiol. 2015, 23, 630–641. [Google Scholar] [CrossRef]
- Schulze, S.; Pfeiffer, F.; Garcia, B.A.; Pohlschroder, M. Glycoproteomics of Haloferax volcanii reveals an extensive glycoproteome and concurrence of different N-glycosylation pathways. bioRxiv 2021. [Google Scholar] [CrossRef]
- Watanabe, Y.; Bowden, T.A.; Wilson, I.A.; Crispin, M. Exploitation of glycosylation in enveloped virus pathobiology. Biochim. Biophys. Acta. Gen. Subj. 2019, 1863, 1480–1497. [Google Scholar] [CrossRef]
- Strasser, R. Biological significance of complex N-glycans in plants and their impact on plant physiology. Front. Plant Sci. 2014, 5, 363. [Google Scholar] [CrossRef]
- Pereira, A.M.; Lopes, A.L.; Coimbra, S. Arabinogalactan Proteins as Interactors along the Crosstalk between the Pollen Tube and the Female Tissues. Front. Plant Sci. 2016, 7, 1895. [Google Scholar] [CrossRef]
- Nagashima, Y.; von Schaewen, A.; Koiwa, H. Function of N-glycosylation in plants. Plant Sci. 2018, 274, 70–79. [Google Scholar] [CrossRef]
- Matsui, T.; Takita, E.; Sato, T.; Kinjo, S.; Aizawa, M.; Sugiura, Y.; Hamabata, T.; Sawada, K.; Kato, K. N-glycosylation at noncanonical Asn-X-Cys sequences in plant cells. Glycobiology 2011, 21, 994–999. [Google Scholar] [CrossRef]
- Wang, J.; Wen, H.; Li, M.; Guo, T.; Chen, C.E. N-Glycoproteome Reveals That N-Glycosylation Plays Crucial Roles in Photosynthesis and Carbon Metabolism in Young Rice Leaves. J. Plant Biol. 2020, 63, 165–175. [Google Scholar] [CrossRef]
- Zabotina, O.A.; Zang, N.; Weerts, R. Polysaccharide Biosynthesis: Glycosyltransferases and Their Complexes. Front. Plant Sci. 2021, 12. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, F.; Aebi, M. Mechanisms and principles of N-linked protein glycosylation. Curr. Opin. Struct. Biol. 2011, 21, 576–582. [Google Scholar] [CrossRef]
- Harada, Y.; Li, H.; Li, H.; Lennarz, W.J. Oligosaccharyltransferase directly binds to ribosome at a location near the translocon-binding site. Proc. Natl. Acad. Sci. USA 2009, 106, 6945–6949. [Google Scholar] [CrossRef]
- Kajiura, H.; Koiwa, H.; Nakazawa, Y.; Okazawa, A.; Kobayashi, A.; Seki, T.; Fujiyama, K. Two Arabidopsis thaliana Golgi alpha-mannosidase I enzymes are responsible for plant N-glycan maturation. Glycobiology 2010, 20, 235–247. [Google Scholar] [CrossRef] [PubMed]
- Bardor, M. Analysis of the N-glycosylation of recombinant glycoproteins produced in transgenic plants. Trends Plant Sci. 1999, 4, 376–380. [Google Scholar] [CrossRef]
- Kaulfurst-Soboll, H.; Rips, S.; Koiwa, H.; Kajiura, H.; Fujiyama, K.; von Schaewen, A. Reduced immunogenicity of Arabidopsis hgl1 mutant N-glycans caused by altered accessibility of xylose and core fucose epitopes. J. Biol. Chem. 2011, 286, 22955–22964. [Google Scholar] [CrossRef]
- Gutternigg, M.; Kretschmer-Lubich, D.; Paschinger, K.; Rendic, D.; Hader, J.; Geier, P.; Ranftl, R.; Jantsch, V.; Lochnit, G.; Wilson, I.B. Biosynthesis of truncated N-linked oligosaccharides results from non-orthologous hexosaminidase-mediated mechanisms in nematodes, plants, and insects. J. Biol. Chem. 2007, 282, 27825–27840. [Google Scholar] [CrossRef]
- Liebminger, E.; Veit, C.; Pabst, M.; Batoux, M.; Zipfel, C.; Altmann, F.; Mach, L.; Strasser, R. Beta-N-acetylhexosaminidases HEXO1 and HEXO3 are responsible for the formation of paucimannosidic N-glycans in Arabidopsis thaliana. J. Biol. Chem. 2011, 286, 10793–10802. [Google Scholar] [CrossRef]
- Maeda, M.; Kimura, Y. Structural features of free N-glycans occurring in plants and functional features of de-N-glycosylation enzymes, ENGase, and PNGase: The presence of unusual plant complex type N-glycans. Front. Plant Sci. 2014, 5, 429. [Google Scholar] [CrossRef] [PubMed]
- Katsube, M.; Ebara, N.; Maeda, M.; Kimura, Y. Cytosolic Free N-Glycans Are Retro-Transported Into the Endoplasmic Reticulum in Plant Cells. Front. Plant Sci. 2020, 11, 610124. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.; Ferraz, R.; Dupree, P.; Showalter, A.M.; Coimbra, S. Three Decades of Advances in Arabinogalactan-Protein Biosynthesis. Front. Plant Sci. 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Ellis, M.; Egelund, J.; Schultz, C.J.; Bacic, A. Arabinogalactan-proteins: Key regulators at the cell surface? Plant Physiol. 2010, 153, 403–419. [Google Scholar] [CrossRef]
- Castilleux, R.; Plancot, B.; Gügi, B.; Attard, A.; Loutelier-Bourhis, C.; Lefranc, B.; Nguema-Ona, E.; Arkoun, M.; Yvin, J.-C.; Driouich, A.; et al. Extensin arabinosylation is involved in root response to elicitors and limits oomycete colonization. Ann. Bot. 2020, 125, 751–763. [Google Scholar] [CrossRef] [PubMed]
- Showalter, A.M.; Keppler, B.; Lichtenberg, J.; Gu, D.; Welch, L.R. A bioinformatics approach to the identification, classification, and analysis of hydroxyproline-rich glycoproteins. Plant Physiol. 2010, 153, 485–513. [Google Scholar] [CrossRef]
- Tian, E.; Ten Hagen, K.G. Recent insights into the biological roles of mucin-type O-glycosylation. Glycoconj. J. 2009, 26, 325–334. [Google Scholar] [CrossRef]
- Strasser, R. Plant protein glycosylation. Glycobiology 2016, 26, 926–939. [Google Scholar] [CrossRef]
- Xu, S.L.; Chalkley, R.J.; Maynard, J.C.; Wang, W.; Ni, W.; Jiang, X.; Shin, K.; Cheng, L.; Savage, D.; Huhmer, A.F.; et al. Proteomic analysis reveals O-GlcNAc modification on proteins with key regulatory functions in Arabidopsis. Proc. Natl. Acad. Sci. USA 2017, 114, E1536–E1543. [Google Scholar] [CrossRef]
- Holdener, B.C.; Haltiwanger, R.S. Protein O-fucosylation: Structure and function. Curr. Opin. Struct. Biol. 2019, 56, 78–86. [Google Scholar] [CrossRef]
- Joshi, H.J.; Narimatsu, Y.; Schjoldager, K.T.; Tytgat, H.L.P.; Aebi, M.; Clausen, H.; Halim, A. SnapShot: O-Glycosylation Pathways across Kingdoms. Cell 2018, 172, 632–632.e2. [Google Scholar] [CrossRef]
- Darula, Z.; Medzihradszky, K.F. Analysis of Mammalian O-Glycopeptides-We Have Made a Good Start, but There is a Long Way to Go. Mol. Cell. Proteom. 2018, 17, 2–17. [Google Scholar] [CrossRef] [PubMed]
- Olszewski, N.E.; West, C.M.; Sassi, S.O.; Hartweck, L.M. O-GlcNAc protein modification in plants: Evolution and function. Biochim. Biophys. Acta 2010, 1800, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Saito, F.; Suyama, A.; Oka, T.; Yoko-o, T.; Matsuoka, K.; Jigami, Y.; Shimma, Y.-I. Identification of Novel Peptidyl Serine α-Galactosyltransferase Gene Family in Plants. J. Biol. Chem. 2014, 289, 20405–20420. [Google Scholar] [CrossRef] [PubMed]
- Taylor, C.M.; Karunaratne, C.V.; Xie, N. Glycosides of hydroxyproline: Some recent, unusual discoveries. Glycobiology 2012, 22, 757–767. [Google Scholar] [CrossRef]
- Tryfona, T.; Liang, H.C.; Kotake, T.; Tsumuraya, Y.; Stephens, E.; Dupree, P. Structural characterization of Arabidopsis leaf arabinogalactan polysaccharides. Plant Physiol. 2012, 160, 653–666. [Google Scholar] [CrossRef] [PubMed]
- Hijazi, M.; Velasquez, S.M.; Jamet, E.; Estevez, J.M.; Albenne, C. An update on post-translational modifications of hydroxyproline-rich glycoproteins: Toward a model highlighting their contribution to plant cell wall architecture. Front. Plant Sci. 2014, 5, 395. [Google Scholar] [CrossRef]
- Showalter, A.M.; Basu, D. Extensin and Arabinogalactan-Protein Biosynthesis: Glycosyltransferases, Research Challenges, and Biosensors. Front. Plant Sci. 2016, 7, 814. [Google Scholar] [CrossRef]
- Larsen Petersen, B.; Ulvskov, P.; MacAlister, C.A. Plant Protein O-Arabinosylation. Front. Plant Sci. 2021, 12. [Google Scholar] [CrossRef]
- Hofsteenge, J.; Hess, D.; Furmanek, A.; Blommers, M.; Miroshnichenko, O. The Four Terminal Components of the Complement System AreC-Mannosylated on Multiple Tryptophan Residues. J. Biol. Chem. 1999, 274, 32786–32794. [Google Scholar] [CrossRef]
- Stepper, J.; Shastri, S.; Loo, T.S.; Preston, J.C.; Novak, P.; Man, P.; Moore, C.H.; Havlicek, V.; Patchett, M.L.; Norris, G.E. Cysteine S-glycosylation, a new post-translational modification found in glycopeptide bacteriocins. FEBS Lett. 2011, 585, 645–650. [Google Scholar] [CrossRef]
- Haynes, P.A. Phosphoglycosylation: A new structural class of glycosylation? Glycobiology 1998, 8, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Ren, Z.; Ji, X.; Jiao, Z.; Luo, Y.; Zhang, G.Q.; Tao, S.; Lei, Z.; Zhang, J.; Wang, Y.; Liu, Z.J.; et al. Functional analysis of a novel C-glycosyltransferase in the orchid Dendrobium catenatum. Hortic. Res. 2020, 7, 111. [Google Scholar] [CrossRef] [PubMed]
- Mumtaz, F.; Zubair, M.; Khan, F.; Niaz, K. Analysis of plants lipids. In Recent Advances in Natural Products Analysis; Elsevier: Amsterdam, The Netherlands, 2020; pp. 677–705. ISBN 9780128164556. [Google Scholar]
- Okazaki, Y.; Saito, K. Roles of lipids as signaling molecules and mitigators during stress response in plants. Plant J. 2014, 79, 584–596. [Google Scholar] [CrossRef] [PubMed]
- Hölzl, G.; Dörmann, P. Structure and function of glycoglycerolipids in plants and bacteria. Prog. Lipid Res. 2007, 46, 225–243. [Google Scholar] [CrossRef]
- Chester, M.A. IUPAC-IUB Joint Commission on Biochemical Nomenclature (JCBN) Nomenclature of glycolipids Recommendations 1997 Prepared for publication. Eur. J. Biochem. 1998, 257, 293–298. [Google Scholar] [PubMed]
- Kalisch, B.; Dörmann, P.; Holzl, G. DGDG and Glycolipids in Plants and Algae. In Lipids in Plant and Algae Development; Nakamura, Y., Li-Beisson, Y., Eds.; Springer International Publishing: Berlin/Heidelberg, Germany, 2016; Volume 86, pp. 51–83. ISBN 0306-0225. [Google Scholar]
- Schnaar, R.L.; Kinoshita, T. Glycosphingolipids. In Essentials of Glycobiology; Varki, A., Cummings, R.D., Esko, J.D., Stanley, P., Hart, G.W., Aebi, M., Darvill, A.G., Kinoshita, T., Packer, N.H., Prestegard, J.H., Eds.; Cold Spring Harbor Laboratory Press: New York, NY, USA, 2017; Volume 3. [Google Scholar]
- Caffalette, C.A.; Kuklewicz, J.; Spellmon, N.; Zimmer, J. Biosynthesis and Export of Bacterial Glycolipids. Annu. Rev. Biochem. 2020, 89, 741–768. [Google Scholar] [CrossRef]
- Lobasso, S.; Perez-Davo, A.; Vitale, R.; Sanchez, M.M.; Corcelli, A. Deciphering archaeal glycolipids of an extremely halophilic archaeon of the genus Halobellus by MALDI-TOF/MS. Chem. Phys. Lipids 2015, 186, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Nyström, L.; Schär, A.; Lampi, A.-M. Steryl glycosides and acylated steryl glycosides in plant foods reflect unique sterol patterns. Eur. J. Lipid Sci. Technol. 2012, 114, 656–669. [Google Scholar] [CrossRef]
- Kopitz, J. Lipid glycosylation: A primer for histochemists and cell biologists. Histochem Cell Biol 2017, 147, 175–198. [Google Scholar] [CrossRef]
- Bar-Peled, M.; O’Neill, M.A. Plant nucleotide sugar formation, interconversion, and salvage by sugar recycling. Annu. Rev. Plant Biol. 2011, 62, 127–155. [Google Scholar] [CrossRef]
- Geserick, C.; Tenhaken, R. UDP-sugar pyrophosphorylase is essential for arabinose and xylose recycling, and is required during vegetative and reproductive growth in Arabidopsis. Plant J. 2013, 74, 239–247. [Google Scholar] [CrossRef]
- Decker, D.; Kleczkowski, L.A. UDP-sugar producing pyrophosphorylases: Distinct and essential enzymes with overlapping substrate specificities, providing de novo precursors for glycosylation reactions. Front. Plant Sci. 2019, 9. [Google Scholar] [CrossRef]
- Granot, D.; David-Schwartz, R.; Kelly, G. Hexose kinases and their role in sugar-sensing and plant development. Front. Plant Sci. 2013, 4, 1–17. [Google Scholar] [CrossRef]
- Scialdone, A.; Howard, M. How plants manage food reserves at night: Quantitative models and open questions. Front. Plant Sci. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Seifert, G.J. Nucleotide sugar interconversions and cell wall biosynthesis: How to bring the inside to the outside. Curr. Opin. Plant Biol. 2004, 7, 277–284. [Google Scholar] [CrossRef]
- Niemann, M.C.E.; Bartrina, I.; Ashikov, A.; Weber, H.; Novák, O.; Spíchal, L.; Strnad, M.; Strasser, R.; Bakker, H.; Schmülling, T.; et al. Arabidopsis ROCK1 transports UDP-GlcNAc/UDP-GalNAc and regulates ER protein quality control and cytokinin activity. Proc. Natl. Acad. Sci. USA 2015, 112, 291–296. [Google Scholar] [CrossRef]
- Farid, A.; Pabst, M.; Schoberer, J.; Altmann, F.; Glössl, J.; Strasser, R. Arabidopsis thaliana alpha1,2-glucosyltransferase (ALG10) is required for efficient N-glycosylation and leaf growth. Plant J. 2011, 68, 314–325. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Henquet, M.; Chen, Z.; Zhang, H.; Zhang, Y.; Ren, X.; van der Krol, S.; Gonneau, M.; Bosch, D.; Gong, Z. LEW3, encoding a putative alpha-1,2-mannosyltransferase (ALG11) in N-linked glycoprotein, plays vital roles in cell-wall biosynthesis and the abiotic stress response in Arabidopsis thaliana. Plant J. 2009, 60, 983–999. [Google Scholar] [CrossRef]
- Dai, X.R.; Gao, X.Q.; Chen, G.H.; Tang, L.L.; Wang, H.; Zhang, X.S. ABNORMAL POLLEN TUBE GUIDANCE1, an Endoplasmic Reticulum-Localized Mannosyltransferase Homolog of GLYCOSYLPHOSPHATIDYLINOSITOL10 in Yeast and PHOSPHATIDYLINOSITOL GLYCAN ANCHOR BIOSYNTHESIS B in Human, Is Required for Arabidopsis Pollen Tube Micropylar Gu. Plant Physiol. 2014, 165, 1544–1556. [Google Scholar] [CrossRef] [PubMed]
- Lukowitz, W.; Nickle, T.C.; Meinke, D.W.; Last, R.L.; Conklin, P.L.; Somerville, C.R. Arabidopsis cyt1 mutants are deficient in a mannose-1-phosphate guanylyltransferase and point to a requirement of N-linked glycosylation for cellulose biosynthesis. Proc. Natl. Acad. Sci. USA 2001, 98, 2262–2267. [Google Scholar] [CrossRef] [PubMed]
- Jadid, N.; Mialoundama, A.S.; Heintz, D.; Ayoub, D.; Erhardt, M.; Mutterer, J.; Meyer, D.; Alioua, A.; Van Dorsselaer, A.; Rahier, A.; et al. DOLICHOL PHOSPHATE MANNOSE SYNTHASE1 mediates the biogenesis of isoprenyl-linked glycans and influences development, stress response, and ammonium hypersensitivity in Arabidopsis. Plant Cell 2011, 23, 1985–2005. [Google Scholar] [CrossRef] [PubMed]
- Lindner, H.; Kessler, S.A.; Muller, L.M.; Shimosato-Asano, H.; Boisson-Dernier, A.; Grossniklaus, U. TURAN and EVAN mediate pollen tube reception in Arabidopsis Synergids through protein glycosylation. PLoS Biol. 2015, 13, e1002139. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.H.; Shen, H.L.; Hsu, P.J.; Hwang, S.G.; Cheng, W.H. N-acetylglucosamine-1-P uridylyltransferase 1 and 2 are required for gametogenesis and embryo development in Arabidopsis thaliana. Plant Cell Physiol. 2014, 55, 1977–1993. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lerouxel, O.; Mouille, G.; Andeme-Onzighi, C.; Bruyant, M.P.; Seveno, M.; Loutelier-Bourhis, C.; Driouich, A.; Hofte, H.; Lerouge, P. Mutants in DEFECTIVE GLYCOSYLATION, an Arabidopsis homolog of an oligosaccharyltransferase complex subunit, show protein underglycosylation and defects in cell differentiation and growth. Plant J. 2005, 42, 455–468. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.A.; von Besser, K.; Zhou, Q.; Smith, E.; Aux, G.; Patton, D.; Levin, J.Z.; Preuss, D. Arabidopsis hapless mutations define essential gametophytic functions. Genetics 2004, 168, 971–982. [Google Scholar] [CrossRef]
- Farid, A.; Malinovsky, F.G.; Veit, C.; Schoberer, J.; Zipfel, C.; Strasser, R. Specialized roles of the conserved subunit OST3/6 of the oligosaccharyltransferase complex in innate immunity and tolerance to abiotic stresses. Plant Physiol. 2013, 162, 24–38. [Google Scholar] [CrossRef] [PubMed]
- Koiwa, H.; Li, F.; McCully, M.G.; Mendoza, I.; Koizumi, N.; Manabe, Y.; Nakagawa, Y.; Zhu, J.; Rus, A.; Pardo, J.M.; et al. The STT3a subunit isoform of the Arabidopsis oligosaccharyltransferase controls adaptive responses to salt/osmotic stress. Plant Cell 2003, 15, 2273–2284. [Google Scholar] [CrossRef] [PubMed]
- Frank, J.; Kaulfurst-Soboll, H.; Rips, S.; Koiwa, H.; von Schaewen, A. Comparative analyses of Arabidopsis complex glycan1 mutants and genetic interaction with staurosporin and temperature sensitive3a. Plant Physiol. 2008, 148, 1354–1367. [Google Scholar] [CrossRef][Green Version]
- Boisson, M. Arabidopsis glucosidase I mutants reveal a critical role of N-glycan trimming in seed development. EMBO J. 2001, 20, 1010–1019. [Google Scholar] [CrossRef]
- Gillmor, C.S.; Poindexter, P.; Lorieau, J.; Palcic, M.M.; Somerville, C. Alpha-glucosidase I is required for cellulose biosynthesis and morphogenesis in Arabidopsis. J. Cell Biol. 2002, 156, 1003–1013. [Google Scholar] [CrossRef]
- Peng, L.; Hocart, C.H.; Redmond, J.W.; Williamson, R.E. Fractionation of carbohydrates in Arabidopsis root cell walls shows that three radial swelling loci are specifically involved in cellulose production. Planta 2000, 211, 406–414. [Google Scholar] [CrossRef] [PubMed]
- Burn, J.E.; Hurley, U.A.; Birch, R.J.; Arioli, T.; Cork, A.; Williamson, R.E. The cellulose-deficient Arabidopsis mutant rsw3 is defective in a gene encoding a putative glucosidase II, an enzyme processing N-glycans during ER quality control. Plant J. 2002, 32, 949–960. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Xu, Y.; Li, Z.; Zhang, S.; Lim, J.-M.; Lee, K.O.; Li, C.; Qian, Q.; Jiang, D.A.; Qi, Y. OsMOGS is required forN-glycan formation and auxin-mediated root development in rice (Oryza sativaL.). Plant J. 2014, 78, 632–645. [Google Scholar] [CrossRef]
- Hong, Z.; Jin, H.; Tzfira, T.; Li, J. Multiple mechanism-mediated retention of a defective brassinosteroid receptor in the endoplasmic reticulum of Arabidopsis. Plant Cell 2008, 20, 3418–3429. [Google Scholar] [CrossRef]
- Berger, S.; Menudier, A.; Julien, R.; Karamanos, Y. Endo-N-acetyl-β-D-glucosaminidase and peptide-N4-(N-acetyl-glucosaminyl) asparagine amidase activities during germination of Raphanus sativus. Phytochemistry 1995, 39, 481–487. [Google Scholar] [CrossRef]
- Vuylsteker, C.; Cuvellier, G.; Berger, S.; Faugeron, C.; Karamanos, Y. Evidence of two enzymes performing the de-N glycosylation of proteins in barley: Expression during germination, localization within the grain and set-up during grain formatio. J. Exp. Bot. 2000, 51, 839–845. [Google Scholar] [CrossRef] [PubMed]
- Jagadeesh, B.H.; Prabha, T.N.; Srinivasan, K. Activities of β-hexosaminidase and α-mannosidase during development and ripening of bell capsicum (Capsicum annuum var. variata). Plant Sci. 2004, 167, 1263–1271. [Google Scholar] [CrossRef]
- Ghosh, S.; Meli, V.S.; Kumar, A.; Thakur, A.; Chakraborty, N.; Chakraborty, S.; Datta, A. The N-glycan processing enzymes alpha-mannosidase and beta-D-N-acetylhexosaminidase are involved in ripening-associated softening in the non-climacteric fruits of capsicum. J. Exp. Bot. 2011, 62, 571–582. [Google Scholar] [CrossRef]
- Pedersen, C.T.; Loke, I.; Lorentzen, A.; Wolf, S.; Kamble, M.; Kristensen, S.K.; Munch, D.; Radutoiu, S.; Spillner, E.; Roepstorff, P.; et al. N-glycan maturation mutants in Lotus japonicus for basic and applied glycoprotein research. Plant J. 2017, 91, 394–407. [Google Scholar] [CrossRef]
- Frank, M.; Kaulfürst-Soboll, H.; Fischer, K.; Von Schaewen, A. Complex-Type N-Glycans Influence the Root Hair Landscape of Arabidopsis Seedlings by Altering the Auxin Output. Front. Plant Sci. 2021, 12. [Google Scholar] [CrossRef]
- Fanata, W.I.; Lee, K.H.; Son, B.H.; Yoo, J.Y.; Harmoko, R.; Ko, K.S.; Ramasamy, N.K.; Kim, K.H.; Oh, D.B.; Jung, H.S.; et al. N-glycan maturation is crucial for cytokinin-mediated development and cellulose synthesis in Oryza sativa. Plant J. 2013, 73, 966–979. [Google Scholar] [CrossRef]
- Kaulfürst-Soboll, H.; Mertens-Beer, M.; Brehler, R.; Albert, M.; Von Schaewen, A. Complex N-Glycans Are Important for Normal Fruit Ripening and Seed Development in Tomato. Front. Plant Sci. 2021, 12. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.S.; Frank, J.; Kang, C.H.; Kajiura, H.; Vikram, M.; Ueda, A.; Kim, S.; Bahk, J.D.; Triplett, B.; Fujiyama, K.; et al. Salt tolerance of Arabidopsis thaliana requires maturation of N-glycosylated proteins in the Golgi apparatus. Proc. Natl. Acad. Sci. USA 2008, 105, 5933–5938. [Google Scholar] [CrossRef]
- Liebminger, E.; Hüttner, S.; Vavra, U.; Fischl, R.; Schoberer, J.; Grass, J.; Blaukopf, C.; Seifert, G.J.; Altmann, F.; Mach, L.; et al. Class I α-Mannosidases Are Required for N-Glycan Processing and Root Development in Arabidopsis thaliana. Plant Cell 2009, 21, 3850–3867. [Google Scholar] [CrossRef] [PubMed]
- Bencúr, P.; Steinkellner, H.; Svoboda, B.; Mucha, J.; Strasser, R.; Kolarich, D.; Hann, S.; Köllensperger, G.; Glössl, J.; Altmann, F.; et al. Arabidopsis thaliana β1,2-xylosyltransferase: An unusual glycosyltransferase with the potential to act at multiple stages of the plant N-glycosylation pathway. Biochem. J. 2005, 388, 515–525. [Google Scholar] [CrossRef] [PubMed]
- Takano, S.; Matsuda, S.; Funabiki, A.; Furukawa, J.; Yamauchi, T.; Tokuji, Y.; Nakazono, M.; Shinohara, Y.; Takamure, I.; Kato, K. The rice RCN11 gene encodes beta1,2-xylosyltransferase and is required for plant responses to abiotic stresses and phytohormones. Plant Sci. 2015, 236, 75–88. [Google Scholar] [CrossRef] [PubMed]
- Vu, K.V.; Jeong, C.Y.; Nguyen, T.T.; Dinh, T.T.H.; Lee, H.; Hong, S.W. Deficiency of AtGFAT1 activity impairs growth, pollen germination and tolerance to tunicamycin in Arabidopsis. J. Exp. Bot. 2019, 70, 1775–1787. [Google Scholar] [CrossRef]
- Hanover, J.A. Glycan-dependent signaling: O-linked N-acetylglucosamine. FASEB J. 2001, 15, 1865–1876. [Google Scholar] [CrossRef]
- Smith, D.K.; Jones, D.M.; Lau, J.B.R.; Cruz, E.R.; Brown, E.; Harper, J.F.; Wallace, I.S. A Putative Protein O-Fucosyltransferase Facilitates Pollen Tube Penetration through the Stigma-Style Interface. Plant Physiol. 2018, 176, 2804–2818. [Google Scholar] [CrossRef]
- Hartweck, L.M.; Scott, C.L.; Olszewski, N.E. Two O-linked N-acetylglucosamine transferase genes of Arabidopsis thaliana L. Heynh. have overlapping functions necessary for gamete and seed development. Genetics 2002, 161, 1279–1291. [Google Scholar] [CrossRef]
- Zentella, R.; Sui, N.; Barnhill, B.; Hsieh, W.-P.; Hu, J.; Shabanowitz, J.; Boyce, M.; Olszewski, N.E.; Zhou, P.; Hunt, D.F.; et al. The Arabidopsis O-fucosyltransferase SPINDLY activates nuclear growth repressor DELLA. Nat. Chem. Biol. 2017, 13, 479–485. [Google Scholar] [CrossRef]
- Silverstone, A.L.; Tseng, T.-S.; Swain, S.M.; Dill, A.; Jeong, S.Y.; Olszewski, N.E.; Sun, T.-P. Functional Analysis of SPINDLY in Gibberellin Signaling in Arabidopsis. Plant Physiol. 2007, 143, 987–1000. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; He, Y.; Su, C.; Zentella, R.; Sun, T.-P.; Wang, L. Nuclear Localized O-Fucosyltransferase SPY Facilitates PRR5 Proteolysis to Fine-Tune the Pace of Arabidopsis Circadian Clock. Mol. Plant 2020, 13, 446–458. [Google Scholar] [CrossRef] [PubMed]
- Ke, S.; Liu, S.; Luan, X.; Xie, X.M.; Hsieh, T.F.; Zhang, X.Q. Mutation in a putative glycosyltransferase-like gene causes programmed cell death and early leaf senescence in rice. Rice 2019, 12, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Tang, Y.; Ruan, N.; Dang, Z.; Huang, Y.; Miao, W.; Xu, Z.; Li, F. The Rice BZ1 Locus Is Required for Glycosylation of Arabinogalactan Proteins and Galactolipid and Plays a Role in both Mechanical Strength and Leaf Color. Rice 2020, 13. [Google Scholar] [CrossRef]
- Asif, M.H.; Trivedi, P.K.; Misra, P.; Nath, P. Prolyl-4-hydroxylase (AtP4H1) mediates and mimics low oxygen response in Arabidopsis thaliana. Funct. Integr. Genomics 2009, 9, 525–535. [Google Scholar] [CrossRef] [PubMed]
- Fragkostefanakis, S.; Sedeek, K.E.M.; Raad, M.; Zaki, M.S.; Kalaitzis, P. Virus induced gene silencing of three putative prolyl 4-hydroxylases enhances plant growth in tomato (Solanum lycopersicum). Plant Mol. Biol. 2014, 85, 459–471. [Google Scholar] [CrossRef]
- Velasquez, S.M.; Ricardi, M.M.; Dorosz, J.G.; Fernandez, P.V.; Nadra, A.D.; Pol-Fachin, L.; Egelund, J.; Gille, S.; Harholt, J.; Ciancia, M.; et al. O-Glycosylated Cell Wall Proteins Are Essential in Root Hair Growth. Science 2011, 332, 1401–1403. [Google Scholar] [CrossRef]
- Velasquez, S.M.; Iusem, N.D.; Estevez, J.M. Root hair sweet growth. Plant Signal. Behav. 2011, 6, 1600–1602. [Google Scholar] [CrossRef][Green Version]
- Velasquez, S.M.; Ricardi, M.M.; Poulsen, C.P.; Oikawa, A.; Dilokpimol, A.; Halim, A.; Mangano, S.; Juarez, S.P.D.; Marzol, E.; Salter, J.D.S.; et al. Complex Regulation of Prolyl-4-HydroxylasesImpacts Root Hair Expansion. Mol. Plant 2014, 8, 734–746. [Google Scholar] [CrossRef]
- Ogawa-Ohnishi, M.; Matsushita, W.; Matsubayashi, Y. Identification of three hydroxyproline O-arabinosyltransferases in Arabidopsis thaliana. Nat. Chem. Biol. 2013, 9, 726–730. [Google Scholar] [CrossRef]
- Gille, S.; Hansel, U.; Ziemann, M.; Pauly, M. Identification of plant cell wall mutants by means of a forward chemical genetic approach using hydrolases. Proc. Natl. Acad. Sci. USA 2009, 106, 14699–14704. [Google Scholar] [CrossRef]
- Møller, S.R.; Yi, X.; Velásquez, S.M.; Gille, S.; Hansen, P.L.M.; Poulsen, C.P.; Olsen, C.E.; Rejzek, M.; Parsons, H.; Zhang, Y.; et al. Identification and evolution of a plant cell wall specific glycoprotein glycosyl transferase, ExAD. Sci. Rep. 2017, 7, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Gille, S.; Sharma, V.; Baidoo, E.E.K.; Keasling, J.D.; Scheller, H.V.; Pauly, M. Arabinosylation of a Yariv-Precipitable Cell Wall Polymer Impacts Plant Growth as Exemplified by the Arabidopsis Glycosyltransferase Mutant ray1. Mol. Plant 2013, 6, 1369–1372. [Google Scholar] [CrossRef] [PubMed]
- Ogawa-Ohnishi, M.; Matsubayashi, Y. Identification of three potent hydroxyprolineO-galactosyltransferases in Arabidopsis. Plant J. 2015, 81, 736–746. [Google Scholar] [CrossRef] [PubMed]
- Basu, D.; Tian, L.; Wang, W.; Bobbs, S.; Herock, H.; Travers, A.; Showalter, A.M. A small multigene hydroxyproline-O-galactosyltransferase family functions in arabinogalactan-protein glycosylation, growth and development in Arabidopsis. BMC Plant Biol. 2015, 15, 295. [Google Scholar] [CrossRef]
- Basu, D.; Wang, W.; Ma, S.; Debrosse, T.; Poirier, E.; Emch, K.; Soukup, E.; Tian, L.; Showalter, A.M. Two Hydroxyproline Galactosyltransferases, GALT5 and GALT2, Function in Arabinogalactan-Protein Glycosylation, Growth and Development in Arabidopsis. PLoS ONE 2015, 10, e0125624. [Google Scholar] [CrossRef]
- Zhang, Y.; Held, M.A.; Kaur, D.; Showalter, A.M. CRISPR-Cas9 multiplex genome editing of the hydroxyproline-O-galactosyltransferase gene family alters arabinogalactan-protein glycosylation and function in Arabidopsis. BMC Plant Biol. 2021, 21, 16. [Google Scholar] [CrossRef]
- Sauquet, H.; von Balthazar, M.; Magallon, S.; Doyle, J.A.; Endress, P.K.; Bailes, E.J.; Barroso de Morais, E.; Bull-Herenu, K.; Carrive, L.; Chartier, M.; et al. The ancestral flower of angiosperms and its early diversification. Nat. Commun. 2017, 8, 16047. [Google Scholar] [CrossRef]
- Endress, P.K. Origins of flower morphology. J. Exp. Zool. 2001, 291, 105–115. [Google Scholar] [CrossRef]
- Wilson, Z.A.; Zhang, D.B. From Arabidopsis to rice: Pathways in pollen development. J. Exp. Bot. 2009, 60, 1479–1492. [Google Scholar] [CrossRef] [PubMed]
- Tan, H.; Liang, W.; Hu, J.; Zhang, D. MTR1 encodes a secretory fasciclin glycoprotein required for male reproductive development in rice. Dev. Cell 2012, 22, 1127–1137. [Google Scholar] [CrossRef]
- Fitchette-Laine, A.-C.; Gomord, V.; Cabanes, M.; Michalski, J.-C.; Macary, M.; Foucher, B.; Cavelier, B.; Hawes, C.; Lerouge, P.; Faye, L. N-glycans harboring the Lewis a epitope are expressed at the surface of plant cells. Plant J. 1997, 12, 1411–1417. [Google Scholar] [CrossRef] [PubMed]
- Joly, C.; Léonard, R.; Maftah, A.; Riou-Khamlichi, C. α4-Fucosyltransferase is regulated during flower development: Increases in activity are targeted to pollen maturation and pollen tube elongation. J. Exp. Bot. 2002, 53, 1429–1436. [Google Scholar] [CrossRef]
- Lara-Mondragón, C.M.; MacAlister, C.A. Arabinogalactan glycoprotein dynamics during the progamic phase in the tomato pistil. Plant Reprod. 2021. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Stoddard, F.L.; Baldwin, T.C. Developmental Regulation of Mannan, Arabinogalactan-Protein, and Pectic Epitopes in Pistils of Vicia faba (Faba Bean). Int. J. Plant Sci. 2006, 167, 919–932. [Google Scholar] [CrossRef]
- Losada, J.M.; Herrero, M. Glycoprotein composition along the pistil of Malus x domestica and the modulation of pollen tube growth. BMC Plant Biol. 2014, 14, 1. [Google Scholar] [CrossRef]
- Heslopha-Harrison, J. Pollen Germination and Pollen-Tube Growth. In International Review of Cytology Volume 107; Elsevier: Amsterdam, The Netherlands, 1987; pp. 1–78. ISBN 9780123645074. [Google Scholar]
- Wu, H.-M.; Wong, E.; Ogdahl, J.; Cheung, A.Y. A pollen tube growth-promoting arabinogalactan protein from Nicotiana alata is similar to the tobacco TTS protein. Plant J. 2000, 22, 165–176. [Google Scholar] [CrossRef] [PubMed]
- Noyszewski, A.K.; Liu, Y.C.; Tamura, K.; Smith, A.G. Polymorphism and structure of style-specific arabinogalactan proteins as determinants of pollen tube growth in Nicotiana. BMC Evol. Biol. 2017, 17, 186. [Google Scholar] [CrossRef]
- Alves, C.M.L.; Noyszewski, A.K.; Smith, A.G. Structure and function of class III pistil-specific extensin-like protein in interspecific reproductive barriers. BMC Plant Biol. 2019, 19, 118. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, M.; Tantikanjana, T.; Nishio, T.; Nasrallah, M.E.; Nasrallah, J.B. Site-specific N-glycosylation of the S-locus receptor kinase and its role in the self-incompatibility response of the brassicaceae. Plant Cell 2014, 26, 4749–4762. [Google Scholar] [CrossRef] [PubMed]
- Mohanty, S.; Chaudhary, B.P.; Zoetewey, D. Structural Insight into the Mechanism of N-Linked Glycosylation by Oligosaccharyltransferase. Biomolecules 2020, 10, 624. [Google Scholar] [CrossRef] [PubMed]
- Boisson-Dernier, A.; Roy, S.; Kritsas, K.; Grobei, M.A.; Jaciubek, M.; Schroeder, J.I.; Grossniklaus, U. Disruption of the pollen-expressed FERONIA homologs ANXUR1 and ANXUR2 triggers pollen tube discharge. Development 2009, 136, 3279–3288. [Google Scholar] [CrossRef] [PubMed]
- Ezura, H.; Hiwasa-Tanase, K. Fruit Development. In Plant Developmental Biology; Springer International Publishing: Berlin/Heidelberg, Germany, 2010; Volume 1, pp. 301–318. ISBN 978-3-642-02300-2978-3-642-02301-9. [Google Scholar]
- Palma, J.M.; Corpas, F.J.; Freschi, L.; Valpuesta, V. Editorial: Fruit Ripening: From Present Knowledge to Future Development. Front. Plant Sci. 2019, 10, 545. [Google Scholar] [CrossRef] [PubMed]
- Levi, A.; Davis, A.; Hernandez, A.; Wechter, P.; Thimmapuram, J.; Trebitsh, T.; Tadmor, Y.; Katzir, N.; Portnoy, V.; King, S. Genes expressed during the development and ripening of watermelon fruit. Plant Cell Rep. 2006, 25, 1233–1245. [Google Scholar] [CrossRef] [PubMed]
- Klee, H.J.; Giovannoni, J.J. Genetics and control of tomato fruit ripening and quality attributes. Annu. Rev. Genet. 2011, 45, 41–59. [Google Scholar] [CrossRef]
- Seymour, G.B.; Granell, A. Fruit development and ripening. J. Exp. Bot. 2014, 65, 4489–4490. [Google Scholar] [CrossRef]
- Bouzayen, M.; Latché, A.; Nath, P.; Pech, J.C. Mechanism of Fruit Ripening. In Plant Developmental Biology-Biotechnological Perspectives; Springer: Berlin/Heidelberg, Germany, 2010; Volume 1, ISBN 978-3-642-02300-2. [Google Scholar]
- Brummell, D.A.; Harpster, M.H.; Dunsmuir, P. Differential expression of expansin gene family members during growth and ripening of tomato fruit. Plant Mol. Biol. 1999, 39, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Goulao, L.; Oliveira, C. Cell wall modifications during fruit ripening: When a fruit is not the fruit. Trends Food Sci. Technol. 2008, 19, 4–25. [Google Scholar] [CrossRef]
- Zhang, X.; Tang, H.; Du, H.; Liu, Z.; Bao, Z.; Shi, Q. Comparative N-glycoproteome analysis provides novel insights into the regulation mechanism in tomato (solanum lycopersicum L.) During fruit ripening process. Plant Sci. 2020, 293, 110413. [Google Scholar] [CrossRef]
- Faugeron, C.; Mollet, J.-C.; Karamanos, Y.; Morvan, H. Activities of de-N-glycosylation are ubiquitously found in tomato plant. Acta Physiol. Plant. 2006, 28, 557–565. [Google Scholar] [CrossRef]
- Chang, T.; Kuo, M.-C.; Khoo, K.-H.; Inoue, S.; Inoue, Y. Developmentally Regulated Expression of a Peptide:N-Glycanase during Germination of Rice Seeds (Oryza sativa) and Its Purification and Characterization. J. Biol. Chem. 2000, 275, 129–134. [Google Scholar] [CrossRef]
- Nakamura, K.; Inoue, M.; Yoshiie, T.; Hosoi, K.; Kimura, Y. Changes in structural features of free N-glycan and endoglycosidase activity during tomato fruit ripening. Biosci. Biotechnol. Biochem. 2008, 72, 2936–2945. [Google Scholar] [CrossRef]
- Kimura, Y.; Takeoka, Y.; Inoue, M.; Maeda, M.; Fujiyama, K. Double-knockout of putative endo-beta-N-acetylglucosaminidase (ENGase) genes in Arabidopsis thaliana: Loss of ENGase activity induced accumulation of high-mannose type free N-glycans bearing N,N’-acetylchitobiosyl unit. Biosci. Biotechnol. Biochem. 2011, 75, 1019–1021. [Google Scholar] [CrossRef]
- Yokouchi, D.; Ono, N.; Nakamura, K.; Maeda, M.; Kimura, Y. Purification and characterization of beta-xylosidase that is active for plant complex type N-glycans from tomato (Solanum lycopersicum): Removal of core alpha1-3 mannosyl residue is prerequisite for hydrolysis of beta1-2 xylosyl residue. Glycoconj. J. 2013, 30, 463–472. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wu, J.; Liu, L.; Li, J. The Crucial Role of Demannosylating Asparagine-Linked Glycans in ERADicating Misfolded Glycoproteins in the Endoplasmic Reticulum. Front. Plant Sci. 2021, 11. [Google Scholar] [CrossRef]
- Priem, B.; Gitti, R.; Bush, C.A.; Gross, K.C. Structure of Ten Free N-Glycans in Ripening Tomato Fruit (Arabinose Is a Constituent of a Plant N-Glycan). Plant Physiol. 1993, 102, 445–458. [Google Scholar] [CrossRef] [PubMed]
- Yunovitz, H.; Gross, K.C. Delay of tomato fruit ripening by an oligosaccharide n-glycan—Interactions with IAA, galactose and lectins. Physiol. Plant. 1994, 90, 152–156. [Google Scholar] [CrossRef]
- Handa, A.K.; Singh, N.K.; Biggs, M.S. Effect of tunicamycin on in vitro ripening of tomato pericarp tissue. Physiol. Plant. 1985, 63, 417–424. [Google Scholar] [CrossRef]
- Yunovitz, H.; Gross, K.C. Effect of tunicamycin on metabolism of unconjugated n-glycans in relation to regulation of tomato fruit ripening. Phytochemistry 1994, 37, 663–668. [Google Scholar] [CrossRef]
- Nakamura, K.; Inoue, M.; Hosoi, K.; Kimura, Y. Predominant occurrence of truncated complex type N-glycans among glycoproteins in mature red tomato. Biosci. Biotechnol. Biochem. 2009, 73, 221–223. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Irfan, M.; Ghosh, S.; Meli, V.S.; Kumar, A.; Kumar, V.; Chakraborty, N.; Chakraborty, S.; Datta, A. Fruit Ripening Regulation of alpha-Mannosidase Expression by the MADS Box Transcription Factor RIPENING INHIBITOR and Ethylene. Front. Plant Sci. 2016, 7, 10. [Google Scholar] [CrossRef] [PubMed]
- Bose, S.K.; He, Y.; Howlader, P.; Wang, W.; Yin, H. The N-glycan processing enzymes beta-D-N-acetylhexosaminidase are involved in ripening-associated softening in strawberry fruit. J. Food Sci. Technol. 2021, 58, 621–631. [Google Scholar] [CrossRef] [PubMed]
- Alvisi, N.; Van Noort, K.; Dwiani, S.; Geschiere, N.; Sukarta, O.; Varossieau, K.; Nguyen, D.-L.; Strasser, R.; Hokke, C.H.; Schots, A.; et al. β-Hexosaminidases Along the Secretory Pathway of Nicotiana benthamiana Have Distinct Specificities Toward Engineered Helminth N-Glycans on Recombinant Glycoproteins. Front. Plant Sci. 2021, 12. [Google Scholar] [CrossRef] [PubMed]
- Meli, V.S.; Ghosh, S.; Prabha, T.N.; Chakraborty, N.; Chakraborty, S.; Datta, A. Enhancement of fruit shelf life by suppressing N-glycan processing enzymes. Proc. Natl. Acad. Sci. USA 2010, 107, 2413–2418. [Google Scholar] [CrossRef]
- Mendez-Yanez, A.; Beltran, D.; Campano-Romero, C.; Molinett, S.; Herrera, R.; Moya-Leon, M.A.; Morales-Quintana, L. Glycosylation is important for FcXTH1 activity as judged by its structural and biochemical characterization. Plant Physiol. Biochem. 2017, 119, 200–210. [Google Scholar] [CrossRef]
- Schröder, J.; Atkinson, R.G.; Langenkämper, G.; Redgwell, R.J. Biochemical and molecular characterisation of xyloglucan endotransglycosylase from ripe kiwifruit. Planta 1998, 204, 242–251. [Google Scholar] [CrossRef]
- Strohmeier, M.; Hrmova, M.; Fischer, M.; Harvey, A.J.; Fincher, G.B.; Pleiss, J. Molecular modeling of family GH16 glycoside hydrolases: Potential roles for xyloglucan transglucosylases/hydrolases in cell wall modification in the poaceae. Protein Sci. 2004, 13. [Google Scholar] [CrossRef]
- Eklöf, J.M.; Brumer, H. The XTH gene family: An update on enzyme structure, function, and phylogeny in xyloglucan remodeling. Plant Physiol. 2010, 153, 456–466. [Google Scholar] [CrossRef]
- Fry, S.C.; Smith, R.C.; Renwick, K.F.; Martin, D.J.; Hodge, S.K.; Matthews, K.J. Xyloglucan endotransglycosylase, a new wall-loosening enzyme activity from plants. Biochem. J. 1992, 282, 821–828. [Google Scholar] [CrossRef]
- Miedes, E.; Lorences, E.P. Xyloglucan endotransglucosylase/hydrolases (XTHs) during tomato fruit growth and ripening. J. Plant Physiol. 2009, 166, 489–498. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Ban, Q.; Li, H.; Hou, Y.; Jin, M.; Han, S.; Rao, J. DkXTH8, a novel xyloglucan endotransglucosylase/hydrolase in persimmon, alters cell wall structure and promotes leaf senescence and fruit postharvest softening. Nat. Sci. Rep. 2016, 6, 39155. [Google Scholar] [CrossRef]
- Albersheim, P.; Anderson, A.J. Proteins from Plant Cell Walls Inhibit Polygalacturonases Secreted by Plant Pathogens. Proc. Natl. Acad. Sci. USA 1971, 68, 1815–1819. [Google Scholar] [CrossRef]
- Shastri, N.V.; Dias, S.; Deo, A. Polygalacturonase Inhibiting Proteins (PGIPs) of Plants. J. Biochem. Biotechnol. 2002, 11, 11–20. [Google Scholar] [CrossRef]
- Protsenko, M.A.; Bulantseva, E.A.; Korableva, N.P. Polygalacturonase-inhibiting proteins in plant fleshy fruits during their ripening and infections. Russ. J. Plant Physiol. 2010, 57, 356–362. [Google Scholar] [CrossRef]
- Kalunke, R.M.; Tundo, S.; Benedetti, M.; Cervone, F.; De Lorenzo, G.; D’Ovidio, R. An update on polygalacturonase-inhibiting protein (PGIP), a leucine-rich repeat protein that protects crop plants against pathogens. Front. Plant Sci. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Akagi, A.; Engelberth, J.; Stotz, H.U. Interaction between polygalacturonase-inhibiting protein and jasmonic acid during defense activation in tomato against Botrytis cinerea. Eur. J. Plant Pathol. 2010, 128, 423–428. [Google Scholar] [CrossRef]
- Kajava, A.V. Structural diversity of leucine-rich repeat proteins. J. Mol. Biol. 1998, 277, 519–527. [Google Scholar] [CrossRef]
- Lim, J.-M.; Aoki, K.; Angel, P.; Garrison, D.; King, D.; Tiemeyer, M.; Bergmann, C.; Wells, L. Mapping Glycans onto Specific N-Linked Glycosylation Sites ofPyrus communisPGIP Redefines the Interface for EPG−PGIP Interactions. J. Proteome Res. 2009, 8, 673–680. [Google Scholar] [CrossRef]
- Fragkostefanakis, S.; Dandachi, F.; Kalaitzis, P. Expression of arabinogalactan proteins during tomato fruit ripening and in response to mechanical wounding, hypoxia and anoxia. Plant Physiol. Biochem. 2012, 52, 112–118. [Google Scholar] [CrossRef]
- Leszczuk, A.; Cybulska, J.; Skrzypek, T.; Zdunek, A. Properties of Arabinogalactan Proteins (AGPs) in Apple (Malus × Domestica) Fruit at Different Stages of Ripening. Biology 2020, 9, 225. [Google Scholar] [CrossRef]
- Leszczuk, A.; Kalaitzis, P.; Blazakis, K.N.; Zdunek, A. The role of arabinogalactan proteins (AGPs) in fruit ripening-a review. Hortic. Res. 2020, 7, 176. [Google Scholar] [CrossRef]
- Leszczuk, A.; Zajac, A.; Kurzyna-Szklarek, M.; Cybulska, J.; Zdunek, A. Investigations of changes in the arabinogalactan proteins (AGPs) structure, size and composition during the fruit ripening process. Nat. Sci. Rep. 2020, 10, 20621. [Google Scholar] [CrossRef]
- Wang, D.; Yeats, T.H.; Uluisik, S.; Rose, J.K.C.; Seymour, G.B. Fruit Softening: Revisiting the Role of Pectin. Trends Plant Sci. 2018, 23, 302–310. [Google Scholar] [CrossRef] [PubMed]
- Brummell, D.A.; Dal Cin, V.; Crisosto, C.H.; Labavitch, J.M. Cell wall metabolism during maturation, ripening and senescence of peach fruit. J. Exp. Bot. 2004, 55, 2029–2039. [Google Scholar] [CrossRef] [PubMed]
- Konozy, E.H.; Causse, M.; Faurobert, M. Cell wall glycosidase activities and protein content variations during fruit development and ripening in three texture contrasted tomato cultivars. Saudi J. Biol. Sci. 2012, 19, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Jiang, F.; Lopez, A.; Jeon, S.; de Freitas, S.T.; Yu, Q.; Wu, Z.; Labavitch, J.M.; Tian, S.; Powell, A.L.T.; Mitcham, E. Disassembly of the fruit cell wall by the ripening-associated polygalacturonase and expansin influences tomato cracking. Hortic. Res. 2019, 6, 17. [Google Scholar] [CrossRef] [PubMed]
- Bidhendi, A.J.; Geitmann, A. Relating the mechanics of the primary plant cell wall to morphogenesis. J. Exp. Bot. 2016, 67, 449–461. [Google Scholar] [CrossRef]
- Nuhse, T.S. Cell wall integrity signaling and innate immunity in plants. Front. Plant Sci. 2012, 3, 280. [Google Scholar] [CrossRef]
- Keegstra, K. Plant cell walls. Plant Physiol. 2010, 154, 483–486. [Google Scholar] [CrossRef]
- Harholt, J.; Suttangkakul, A.; Vibe Scheller, H. Biosynthesis of pectin. Plant Physiol. 2010, 153, 384–395. [Google Scholar] [CrossRef] [PubMed]
- Staehelin, L.A.; Moore, I. The Plant Golgi Apparatus: Structure, Functional Organization and Trafficking Mechanisms. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1995, 46, 261–288. [Google Scholar] [CrossRef]
- Zamil, M.S.; Geitmann, A. The middle lamella-more than a glue. Phys. Biol. 2017, 14, 15004. [Google Scholar] [CrossRef] [PubMed]
- Zhong, R.; Ye, Z.H. Secondary cell walls: Biosynthesis, patterned deposition and transcriptional regulation. Plant Cell Physiol. 2015, 56, 195–214. [Google Scholar] [CrossRef]
- Gillmor, C.S.; Lukowitz, W.; Brininstool, G.; Sedbrook, J.C.; Hamann, T.; Poindexter, P.; Somerville, C. Glycosylphosphatidylinositol-anchored proteins are required for cell wall synthesis and morphogenesis in Arabidopsis. Plant Cell 2005, 17, 1128–1140. [Google Scholar] [CrossRef] [PubMed]
- Walker, A.K.; Soo, K.Y.; Levina, V.; Talbo, G.H.; Atkin, J.D. N-linked glycosylation modulates dimerization of protein disulfide isomerase family A member 2 (PDIA2). FEBS J. 2013, 280, 233–243. [Google Scholar] [CrossRef]
- Beihammer, G.; Maresch, D.; Altmann, F.; Van Damme, E.J.M.; Strasser, R. Lewis A Glycans Are Present on Proteins Involved in Cell Wall Biosynthesis and Appear Evolutionarily Conserved Among Natural Arabidopsis thaliana Accessions. Front. Plant Sci. 2021, 12. [Google Scholar] [CrossRef]
- Basu, D.; Tian, L.; Debrosse, T.; Poirier, E.; Emch, K.; Herock, H.; Travers, A.; Showalter, A.M. Glycosylation of a Fasciclin-Like Arabinogalactan-Protein (SOS5) Mediates Root Growth and Seed Mucilage Adherence via a Cell Wall Receptor-Like Kinase (FEI1/FEI2) Pathway in Arabidopsis. PLoS ONE 2016, 11, e0145092. [Google Scholar] [CrossRef]
- Horiuchi, R.; Hirotsu, N.; Miyanishi, N. Comparative analysis of N-glycans in the ungerminated and germinated stages of Oryza sativa. Carbohydr. Res. 2015, 418, 1–8. [Google Scholar] [CrossRef]
- Priem, B.; Morvan, H.; Hafez, A.M.A.; Morvan, C. Influence of a plant glycan of the oligomannoside type on the growth of flax plantlets. C. R. Acad. Sci Paris 1990, 311, 411–416. [Google Scholar]
- Jin, H.; Yan, Z.; Nam, K.H.; Li, J. Allele-specific suppression of a defective brassinosteroid receptor reveals a physiological role of UGGT in ER quality control. Mol. Cell 2007, 26, 821–830. [Google Scholar] [CrossRef]
- Römisch, K. Endoplasmic Reticulum-Associated Degradation. Annu. Rev. Cell Dev. Biol. 2005, 21, 435–456. [Google Scholar] [CrossRef]
- Clouse, S.D.; Langford, M.; McMorris, T.C. A Brassinosteroid-Insensitive Mutant in Arabidopsis thaliana Exhibits Multiple Defects in Growth and Development. Plant Physiol. 1996, 111, 671–678. [Google Scholar] [CrossRef]
- Steber, C.M.; McCourt, P. A Role for Brassinosteroids in Germination in Arabidopsis. Plant Physiol. 2001, 125, 763–769. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, T.; Cano-Delgado, A.; Hideharu, S.; Hiranuma, S.; Fujioka, S.; Yoshida, S.; Chory, J. Binding of brassinosteroids to the extracellular domain of plant receptor kinase BRI1. Nat. Lett. 2005, 43, 167–171. [Google Scholar] [CrossRef]
- Noguchi, T.; Fujioka, S.; Choe, S.; Takatsuto, S.; Yoshida, S.; Yuan, H.; Feldmann, K.A.; Tax, F.E. Brassinosteroid-Insensitive Dwarf Mutants of Arabidopsis Accumulate Brassinosteroids. Plant Physiol. 1999, 121, 743–752. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.W.; Shin, J.H.; Lee, W.K.; Begum, H.; Min, C.H.; Jang, M.H.; Oh, H.B.; Yang, M.S.; Kim, S.R. Inactivation of the beta (1, 2)-xylosyltransferase and the alpha (1, 3)-fucosyltransferase gene in rice (Oryza sativa) by multiplex CRISPR/Cas9 strategy. Plant Cell Rep. 2021. [Google Scholar] [CrossRef] [PubMed]
- Izhaki, A.; Swain, S.M.; Tseng, T.-S.; Borochov, A.; Olszewski, N.E.; Weiss, D. The role of SPY and its TPR domain in the regulation of gibberellin action throughout the life cycle of Petunia hybrida plants. Plant J. 2001, 28, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Swain, S.M.; Tseng, T.-S.; Olszewski, N.E. Altered Expression of SPINDLY Affects Gibberellin Response and Plant Development. Plant Physiol. 2001, 126, 1174–1185. [Google Scholar] [CrossRef]
- Filardo, F.F.; Swain, S.M. SPYing on GA Signaling and Plant Development. J. Plant Growth Regul. 2003, 22, 163–175. [Google Scholar] [CrossRef]
- Singh, D.P.; Jermakow, A.M.; Swain, S.M. Gibberellins are required for seed development and pollen tube growth in Arabidopsis. Plant Cell 2002, 14, 3133–3147. [Google Scholar] [CrossRef]
- Ripoll, J.J.; Roeder, A.H.; Ditta, G.S.; Yanofsky, M.F. A novel role for the floral homeotic gene APETALA2 during Arabidopsis fruit development. Development 2011, 138, 5167–5176. [Google Scholar] [CrossRef] [PubMed]
- Okamuro, J.K.; Szeto, W.; Lotys-Prass, C.; Jofuku, K.D. Photo and hormonal control of meristem identity in the Arabidopsis flower mutants apetala2 and apetala1. Plant Cell 1997, 9, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Hartweck, L.M.; Genger, R.K.; Grey, W.M.; Olszewski, N.E. SECRET AGENT and SPINDLY have overlapping roles in the development of Arabidopsis thaliana L. Heyn. J. Exp. Bot. 2006, 57, 865–875. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bi, Y.; Deng, Z.; Ni, W.; Shrestha, R.; Savage, D.; Hartwig, T.; Patil, S.; Hong, S.H.; Zhang, Z.; Oses-Prieto, J.A.; et al. Arabidopsis ACINUS is O-glycosylated and regulates transcription and alternative splicing of regulators of reproductive transitions. Nat. Commun. 2021, 12, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Yan, D.; Duermeyer, L.; Leoveanu, C.; Nambara, E. The Functions of the Endosperm During Seed Germination. Plant Cell Physiol. 2014, 55, 1521–1533. [Google Scholar] [CrossRef] [PubMed]
- Müntz, K. Deposition of storage proteins. Plant Mol. Biol. 1998, 38, 77–98. [Google Scholar] [CrossRef]
- Tosi, P.; Parker, M.; Gritsch, C.S.; Carzaniga, R.; Martin, B.; Shewry, P.R. Trafficking of storage proteins in developing grain of wheat. J. Exp. Bot. 2009, 60, 979–991. [Google Scholar] [CrossRef]
- Sturm, A.; Van Kuik, J.A.; Vliegenthart, J.F.; Chrispeels, M.J. Structure, position, and biosynthesis of the high mannose and the complex oligosaccharide side chains of the bean storage protein phaseolin. J. Biol. Chem. 1987, 262, 13392–13403. [Google Scholar] [CrossRef]
- Arcalis, E.; Stadlmann, J.; Marcel, S.; Drakakaki, G.; Winter, V.; Rodriguez, J.; Fischer, R.; Altmann, F.; Stoger, E. The changing fate of a secretory glycoprotein in developing maize endosperm. Plant Physiol. 2010, 153, 693–702. [Google Scholar] [CrossRef]
- Ohta, M.; Takaiwa, F. OsHrd3 is necessary for maintaining the quality of endoplasmic reticulum-derived protein bodies in rice endosperm. J. Exp. Bot. 2015, 66, 4585–4593. [Google Scholar] [CrossRef][Green Version]
- Schoebel, S.; Mi, W.; Stein, A.; Ovchinnikov, S.; Pavlovicz, R.; DiMaio, F.; Baker, D.; Chambers, M.G.; Su, H.; Li, D.; et al. Cryo-EM structure of the protein-conducting ERAD channel Hrd1 in complex with Hrd3. Nat. Lett. 2017, 548, 352–355. [Google Scholar] [CrossRef]
- Qian, D.; Chen, G.; Tian, L.; Qu, L.Q. OsDER1 Is an ER-Associated Protein Degradation Factor That Responds to ER Stress. Plant Physiol. 2018, 178, 402–412. [Google Scholar] [CrossRef] [PubMed]
- Santos, C.N.; Alves, M.; Oliveira, A.; Ferreira, R.B. beta-N-Acetylhexosaminidase involvement in alpha-conglutin mobilization in Lupinus albus. J. Plant Physiol. 2013, 170, 1047–1056. [Google Scholar] [CrossRef]
- Hodge, A.; Alexander, I.J.; Gooday, G.W. Chitinolytic activities of Eucalyptus pilularis and Pinus sylvestris root systems challenged with mycorrhizal and pathogenic fungi. New Phytol. 1995, 131, 255–261. [Google Scholar] [CrossRef]
- Müntz, K.; Belozersky, M.A.; Dunaevsky, Y.E.; Schlereth, A.; Tiedemann, J. Stored proteinases and the initiation of storage protein mobilization in seeds during germination and seedling growth. J. Exp. Bot. 2001, 52, 1741–1752. [Google Scholar] [CrossRef] [PubMed]
- Silva-Sanchez, C.; Chen, S.; Li, J.; Chourey, P.S. A comparative glycoproteome study of developing endosperm in the hexose-deficient miniature1 (mn1) seed mutant and its wild type Mn1 in maize. Front. Plant Sci. 2014, 5, 63. [Google Scholar] [CrossRef]
- Ruan, Y.L.; Jin, Y.; Yang, Y.J.; Li, G.J.; Boyer, J.S. Sugar input, metabolism, and signaling mediated by invertase: Roles in development, yield potential, and response to drought and heat. Mol. Plant 2010, 3, 942–955. [Google Scholar] [CrossRef] [PubMed]
- Chourey, P.S.; Li, Q.B.; Cevallos-Cevallos, J. Pleiotropy and its dissection through a metabolic gene Miniature1 (Mn1) that encodes a cell wall invertase in developing seeds of maize. Plant Sci. 2012, 184, 45–53. [Google Scholar] [CrossRef]
- Kang, B.H.; Xiong, Y.; Williams, D.S.; Pozueta-Romero, D.; Chourey, P.S. Miniature1-encoded cell wall invertase is essential for assembly and function of wall-in-growth in the maize endosperm transfer cell. Plant Physiol. 2009, 151, 1366–1376. [Google Scholar] [CrossRef]
- Bowles, D.; Isayenkova, J.; Lim, E.K.; Poppenberger, B. Glycosyltransferases: Managers of small molecules. Curr. Opin. Plant Biol. 2005, 8, 254–263. [Google Scholar] [CrossRef] [PubMed]
- Louveau, T.; Osbourn, A. The Sweet Side of Plant-Specialized Metabolism. Cold Spring Harb. Perspect. Biol. 2019, 11. [Google Scholar] [CrossRef]
- Li, W.; Zhang, F.; Chang, Y.; Zhao, T.; Schranz, M.E.; Wang, G. Nicotinate O-Glucosylation Is an Evolutionarily Metabolic Trait Important for Seed Germination under Stress Conditions in Arabidopsis thaliana. Plant Cell 2015, 27, 1907–1924. [Google Scholar] [CrossRef]
- Wang, T.; Li, P.; Mu, T.; Dong, G.; Zheng, C.; Jin, S.; Chen, T.; Hou, B.; Li, Y. Overexpression of UGT74E2, an Arabidopsis IBA Glycosyltransferase, Enhances Seed Germination and Modulates Stress Tolerance via ABA Signaling in Rice. Int. J. Mol. Sci. 2020, 21, 7239. [Google Scholar] [CrossRef]
- Behr, M.; Neutelings, G.; El Jaziri, M.; Baucher, M. You Want it Sweeter: How Glycosylation Affects Plant Response to Oxidative Stress. Front. Plant Sci. 2020, 11, 571399. [Google Scholar] [CrossRef]
- Pandey, V.; Niranjan, A.; Atri, N.; Chandrashekhar, K.; Mishra, M.K.; Trivedi, P.K.; Misra, P. WsSGTL1 gene from Withania somnifera, modulates glycosylation profile, antioxidant system and confers biotic and salt stress tolerance in transgenic tobacco. Planta 2014, 239, 1217–1231. [Google Scholar] [CrossRef] [PubMed]
- Vissenberg, K.; Claeijs, N.; Balcerowicz, D.; Schoenaers, S. Hormonal regulation of root hair growth and responses to the environment in Arabidopsis. J. Exp. Bot. 2020, 71, 2412–2427. [Google Scholar] [CrossRef] [PubMed]
- Qin, C.; Li, Y.; Gan, J.; Wang, W.; Zhang, H.; Liu, Y.; Wu, P. OsDGL1, a Homolog of an Oligosaccharyltransferase Complex Subunit, is Involved in N-Glycosylation and Root Development in Rice. Plant Cell Physiol. 2013, 54, 129–137. [Google Scholar] [CrossRef]
- Veit, C.; König, J.; Altmann, F.; Strasser, R. Processing of the Terminal Alpha-1,2-Linked Mannose Residues From Oligomannosidic N-Glycans Is Critical for Proper Root Growth. Front. Plant Sci. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Niu, G.; Zhang, H.; Sun, Y.; Sun, S.; Yu, F.; Lu, S.; Yang, Y.; Li, J.; Hong, Z. Trimming of N-Glycans by the Golgi-Localized α-1,2-Mannosidases, MNS1 and MNS2, Is Crucial for Maintaining RSW2 Protein Abundance during Salt Stress in Arabidopsis. Mol. Plant 2018, 11, 678–690. [Google Scholar] [CrossRef]
- Endler, A.; Kesten, C.; Schneider, R.; Zhang, Y.; Ivakov, A.; Froehlich, A.; Funke, N.; Persson, S. A Mechanism for Sustained Cellulose Synthesisduring Salt Stress. Cell 2015, 162, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Nagashima, Y.; Ma, Z.; Liu, X.; Qian, X.; Zhang, X.; Von Schaewen, A.; Koiwa, H. Multiple Quality Control Mechanisms in the ER and TGN Determine Subcellular Dynamics and Salt-Stress Tolerance Function of KORRIGAN1. Plant Cell 2020, 32, 470–485. [Google Scholar] [CrossRef] [PubMed]
- Bao, Y.; Howell, S.H. The Unfolded Protein Response Supports Plant Development and Defense as well as Responses to Abiotic Stress. Front. Plant Sci. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Depaepe, T.; Hendrix, S.; Janse van Rensburg, H.C.; Van den Ende, W.; Cuypers, A.; Van Der Straeten, D. At the Crossroads of Survival and Death: The Reactive Oxygen Species–Ethylene–Sugar Triad and the Unfolded Protein Response. Trends Plant Sci. 2021. [Google Scholar] [CrossRef]
- Von Schaewen, A.; Rips, S.; Jeong, I.S.; Koiwa, H. Arabidopsis thalianaKORRIGAN1 protein: N-glycan modification, localization, and function in cellulose biosynthesis and osmotic stress responses. Plant Signal. Behav. 2015, 10, e1024397. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rips, S.; Bentley, N.; Jeong, I.S.; Welch, J.L.; Von Schaewen, A.; Koiwa, H. Multiple N-Glycans Cooperate in the Subcellular Targeting and Functioning of Arabidopsis KORRIGAN1. Plant Cell 2014, 26, 3792–3808. [Google Scholar] [CrossRef]
- Mócsai, R.; Göritzer, K.; Stenitzer, D.; Maresch, D.; Strasser, R.; Altmann, F. Prolyl Hydroxylase Paralogs in Nicotiana benthamiana Show High Similarity With Regard to Substrate Specificity. Front. Plant Sci. 2021, 12. [Google Scholar] [CrossRef] [PubMed]
- Velasquez, M.; Salter, J.S.; Dorosz, J.G.; Petersen, B.L.; Estevez, J.M. Recent Advances on the Posttranslational Modifications of EXTs and Their Roles in Plant Cell Walls. Front. Plant Sci. 2012, 3. [Google Scholar] [CrossRef] [PubMed]
- Parniske, M. Arbuscular mycorrhiza: The mother ofplant root endosymbioses. Nat. Rev. Microbiol. 2008, 6, 763–775. [Google Scholar] [CrossRef] [PubMed]
- Concha, C.; Doerner, P. The impact of the rhizobia–legume symbiosis on host root system architecture. J. Exp. Bot. 2020, 71, 3902–3921. [Google Scholar] [CrossRef]
- Bonfante, P.; Genre, A. Mechanisms underlying beneficial plant–fungus interactions in mycorrhizal symbiosis. Nat. Commun. 2010, 1, 1–11. [Google Scholar] [CrossRef]
- Ferguson, B.J.; Mens, C.; Hastwell, A.H.; Zhang, M.; Su, H.; Jones, C.H.; Chu, X.; Gresshoff, P.M. Legume nodulation: The host controls the party. Plant. Cell Environ. 2019, 42, 41–51. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, B.J.; Li, D.; Hastwell, A.H.; Reid, D.E.; Li, Y.; Jackson, S.A.; Gresshoff, P.M. The soybean (Glycine max) nodulation-suppressive CLE peptide, GmRIC1, functions interspecifically in common white bean (Phaseolus vulgaris), but not in a supernodulating line mutated in the receptor PvNARK. Plant Biotechnol. J. 2014, 12, 1085–1097. [Google Scholar] [CrossRef] [PubMed]
- Mortier, V.; Den Herder, G.; Whitford, R.; Van De Velde, W.; Rombauts, S.; D’Haeseleer, K.; Holsters, M.; Goormachtig, S. CLE Peptides Control Medicago truncatula Nodulation Locally and Systemically. Plant Physiol. 2010, 153, 222–237. [Google Scholar] [CrossRef] [PubMed]
- Hastwell, A.H.; Corcilius, L.; Williams, J.T.; Gresshoff, P.M.; Payne, R.J.; Ferguson, B.J. Triarabinosylation is required for nodulation-suppressive CLE peptides to systemically inhibit nodulation inPisum sativum. Plant. Cell Environ. 2019, 42, 188–197. [Google Scholar] [CrossRef] [PubMed]
- Imin, N.; Patel, N.; Corcilius, L.; Payne, R.J.; Djordjevic, M.A. CLE peptide tri-arabinosylation and peptide domain sequence composition are essential for SUNN-dependent autoregulation of nodulation in Medicago truncatula. New Phytol. 2018, 218, 73–80. [Google Scholar] [CrossRef]
- Karlo, M.; Boschiero, C.; Landerslev, K.G.; Blanco, G.S.; Wen, J.; Mysore, K.S.; Dai, X.; Zhao, P.X.; De Bang, T.C. The CLE53–SUNN genetic pathway negatively regulates arbuscular mycorrhiza root colonization in Medicago truncatula. J. Exp. Bot. 2020, 71, 4972–4984. [Google Scholar] [CrossRef]
- Cannesan, M.A.; Durand, C.; Burel, C.; Gangneux, C.; Lerouge, P.; Ishii, T.; Laval, K.; Follet-Gueye, M.-L.; Driouich, A.; Vicré-Gibouin, M. Effect of Arabinogalactan Proteins from the Root Caps of Pea and Brassica napus on Aphanomyces euteiches Zoospore Chemotaxis and Germination. Plant Physiol. 2012, 159, 1658–1670. [Google Scholar] [CrossRef] [PubMed]
- Xie, D.; Ma, L.; Šamaj, J.; Xu, C. Immunohistochemical analysis of cell wall hydroxyproline-rich glycoproteins in the roots of resistant and susceptible wax gourd cultivars in response to Fusarium oxysporum f. sp. Benincasae infection and fusaric acid treatment. Plant Cell Rep. 2011, 30, 1555–1569. [Google Scholar] [CrossRef]
- Xie, F.; Williams, A.; Edwards, A.; Downie, J.A. A plant arabinogalactan-like glycoprotein promotes a novel type of polar surface attachment by Rhizobium leguminosarum. Mol. Plant Microbe Interact. 2012, 25, 250–258. [Google Scholar] [CrossRef]
- Lin, B.; Qing, X.; Liao, J.; Zhuo, K. Role of Protein Glycosylation in Host-Pathogen Interaction. Cells 2020, 9, 1022. [Google Scholar] [CrossRef] [PubMed]
- Tang, D.; Wang, G.; Zhou, J.-M. Receptor Kinases in Plant-Pathogen Interactions: More Than Pattern Recognition. Plant Cell 2017, 29, 618–637. [Google Scholar] [CrossRef] [PubMed]
- Svara, A.; Tarkowski, Ł.P.; Janse van Rensburg, H.C.; Deleye, E.; Vaerten, J.; De Storme, N.; Keulemans, W.; Van den Ende, W. Sweet Immunity: The Effect of Exogenous Fructans on the Susceptibility of Apple (Malus × domestica Borkh.) to Venturia inaequalis. Int. J. Mol. Sci. 2020, 21, 5885. [Google Scholar] [CrossRef] [PubMed]
- Ghorashi, A.C.; Kohler, J.J. Not All Quiet on the Sugar Front: Glycan Combatants in Host–Pathogen Interactions. Biochemistry 2020, 59, 3061–3063. [Google Scholar] [CrossRef]
- Hart, G.W. Minireview Series on the Thirtieth Anniversary of Research on O-GlcNAcylation of Nuclear and Cytoplasmic Proteins: Nutrient Regulation of Cellular Metabolism and Physiology by O-GlcNAcylation. J. Biol. Chem. 2014, 289, 34422–34423. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Sardar, H.S.; McGovern, K.R.; Zhang, Y.; Showalter, A.M. A lysine-rich arabinogalactan protein in Arabidopsis is essential for plant growth and development, including cell division and expansion. Plant J. 2007, 49, 629–640. [Google Scholar] [CrossRef] [PubMed]
- Wagner, G.J. New Approaches for Studying and Exploiting an Old Protuberance, the Plant Trichome. Ann. Bot. 2004, 93, 3–11. [Google Scholar] [CrossRef]
- Kaur, J.; Kariyat, R. Role of Trichomes in Plant Stress Biology. In Evolutionary Ecology of Plant-Herbivore Interaction; Núñez-Farfán, J., Luis Valverde, P., Eds.; Springer International Publishing: Berlin/Heidelberg, Germany, 2020; pp. 15–35. [Google Scholar]
- Mustafa, A.; Ensikat, H.-J.; Weigend, M. Ontogeny and the process of biomineralization in the trichomes of Loasaceae. Am. J. Bot. 2017, 104, 367–378. [Google Scholar] [CrossRef]
- Hauser, M.-T. Molecular basis of natural variation and environmental control of trichome patterning. Front. Plant Sci. 2014, 5. [Google Scholar] [CrossRef]
- Mithöfer, A.; Boland, W. Plant Defense Against Herbivores: Chemical Aspects. Annu. Rev. Plant Biol. 2012, 63, 431–450. [Google Scholar] [CrossRef]
- Tominaga-Wada, R.; Ishida, T.; Wada, T. New Insights into the Mechanism ofDevelopment of Arabidopsis RootHairs and Trichomes. Int. Rev. Cell Mol. Biol. 2011, 286, 67–106. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Held, M.A.; Showalter, A.M. Elucidating the roles of three β-glucuronosyltransferases (GLCATs) acting on arabinogalactan-proteins using a CRISPR-Cas9 multiplexing approach in Arabidopsis. BMC Plant Biol. 2020, 20, 221. [Google Scholar] [CrossRef] [PubMed]
- Lamport, D.T.A.; Várnai, P. Periplasmic arabinogalactan glycoproteins act as a calcium capacitor that regulates plant growth and development. New Phytol. 2012, 197, 58–64. [Google Scholar] [CrossRef]
- Lopez-Hernandez, F.; Tryfona, T.; Rizza, A.; Yu, X.L.; Harris, M.O.B.; Webb, A.A.R.; Kotake, T.; Dupree, P. Calcium Binding by Arabinogalactan Polysaccharides Is Important for Normal Plant Development. Plant Cell 2020, 32, 3346–3369. [Google Scholar] [CrossRef] [PubMed]
- Zentella, R.; Hu, J.; Hsieh, W.-P.; Matsumoto, P.A.; Dawdy, A.; Barnhill, B.; Oldenhof, H.; Hartweck, L.M.; Maitra, S.; Thomas, S.G.; et al. O-GlcNAcylation of master growth repressor DELLA by SECRET AGENT modulates multiple signaling pathways inArabidopsis. Genes Dev. 2016, 30, 164–176. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Liu, Q.; Yao, T.; Fu, X. Shedding light on integrative GA signaling. Curr. Opin. Plant Biol. 2014, 21, 89–95. [Google Scholar] [CrossRef]
- Feng, S.; Martinez, C.; Gusmaroli, G.; Wang, Y.; Zhou, J.; Wang, F.; Chen, L.; Yu, L.; Iglesias-Pedraz, J.M.; Kircher, S.; et al. Coordinated regulation of Arabidopsis thaliana development by light and gibberellins. Nature 2008, 451, 475–479. [Google Scholar] [CrossRef]
- De Lucas, M.; Davière, J.M.; Rodríguez-Falcón, M.; Pontin, M.; Iglesias-Pedraz, J.M.; Lorrain, S.; Fankhauser, C.; Blázquez, M.A.; Titarenko, E.; Prat, S. A molecular framework for light and gibberellin control of cell elongation. Nature 2008, 451, 480–484. [Google Scholar] [CrossRef]
- Liebminger, E.; Grass, J.; Altmann, F.; Mach, L.; Strasser, R. Characterizing the link between glycosylation state and enzymatic activity of the endo-β1,4-glucanase KORRIGAN1 from Arabidopsis thaliana. J. Biol. Chem. 2013, 288, 22270–22280. [Google Scholar] [CrossRef]
- Häweker, H.; Rips, S.; Koiwa, H.; Salomon, S.; Saijo, Y.; Chinchilla, D.; Robatzek, S.; Von Schaewen, A. Pattern recognition receptors require N-glycosylation to mediate plant immunity. J. Biol. Chem. 2010, 285, 4629–4636. [Google Scholar] [CrossRef]
- Esko, J.D.; Bertozzi, C.; Schnaar, R.L. Chemical Tools for Inhibiting Glycosylation. In Essentials of Glycobiology; Varki, A., Cummings, R.D., Esko, J.D., Stanley, P., Hart, G.W., Aebi, M., Darvill, A.G., Kinoshita, T., Packer, N.H., Prestegard, J.H., et al., Eds.; Cold Spring Harbor: New York, NY, USA, 2017. [Google Scholar]
- Hölzl, G.; Dörmann, P. Chloroplast Lipids and Their Biosynthesis. Annu. Rev. Plant Biol. 2019, 70, 51–81. [Google Scholar] [CrossRef] [PubMed]
- Shimojima, M.; Ohta, H. Critical regulation of galactolipid synthesis controls membrane differentiation and remodeling in distinct plant organs and following environmental changes. Prog. Lipid Res. 2011, 50, 258–266. [Google Scholar] [CrossRef] [PubMed]
- Sebastiana, M.; Duarte, B.; Monteiro, F.; Malho, R.; Cacador, I.; Matos, A.R. The leaf lipid composition of ectomycorrhizal oak plants shows a drought-tolerance signature. Plant Physiol. Biochem. 2019, 144, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Boudière, L.; Michaud, M.; Petroutsos, D.; Rébeillé, F.; Falconet, D.; Bastien, O.; Roy, S.; Finazzi, G.; Rolland, N.; Jouhet, J.; et al. Glycerolipids in photosynthesis: Composition, synthesis and trafficking. Biochim. Biophys. Acta Bioenerg. 2014, 1837, 470–480. [Google Scholar] [CrossRef]
- Rocha, J.; Nitenberg, M.; Girard-Egrot, A.; Jouhet, J.; Maréchal, E.; Block, M.A.; Breton, C. Do Galactolipid Synthases Play a Key Role in the Biogenesis of Chloroplast Membranes of Higher Plants? Front. Plant Sci. 2018, 9. [Google Scholar] [CrossRef]
- Li, H.-M.; Yu, C.-W. Chloroplast Galactolipids: The Link Between Photosynthesis, Chloroplast Shape, Jasmonates, Phosphate Starvation and Freezing Tolerance. Plant Cell Physiol. 2018, 59, 1128–1134. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, K. Role of membrane glycerolipids in photosynthesis, thylakoid biogenesis and chloroplast development. J. Plant Res. 2016, 129, 565–580. [Google Scholar] [CrossRef] [PubMed]
- Standfuss, J.; Terwisscha Van Scheltinga, A.C.; Lamborghini, M.; Kühlbrandt, W. Mechanisms of photoprotection and nonphotochemical quenching in pea light-harvesting complex at 2.5 Å resolution. EMBO J. 2005, 24, 919–928. [Google Scholar] [CrossRef]
- Jarvis, P.; Dörmann, P.; Peto, C.A.; Lutes, J.; Benning, C.; Chory, J. Galactolipid deficiency and abnormal chloroplast development in the Arabidopsis MGD synthase 1 mutant. Proc. Natl. Acad. Sci. USA 2000, 97, 8175–8179. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Yan, H.; Wang, K.; Kuang, T.; Zhang, J.; Gui, L.; An, X.; Chang, W. Crystal structure of spinach major light-harvesting complex at 2.72 Å resolution. Nat. Artic. 2004, 428, 287–292. [Google Scholar] [CrossRef]
- Chetal, S.; Wagle, D.S.; Nainawatee, H.S. Alterations in glycolipids of wheat and barley leaves under water stress. Phytochemistry 1982, 21, 51–53. [Google Scholar] [CrossRef]
- Torres-Franklin, M.-L.; Gigon, A.; De Melo, D.F.; Zuily-Fodil, Y.; Pham-Thi, A.-T. Drought stress and rehydration affect the balance between MGDG and DGDG synthesis in cowpea leaves. Physiol. Plant. 2007, 131, 201–210. [Google Scholar] [CrossRef]
- Guerfel, M.; Baccouri, O.; Boujnah, D.; Zarrouk, M. Changes in lipid composition, water relations and gas exchange in leaves of two young ‘Chemlali’ and ‘Chetoui’ olive trees in response to water stress. Plant Soil 2008, 311, 121–129. [Google Scholar] [CrossRef]
- Chen, J.; Burke, J.J.; Xin, Z.; Xu, C.; Velten, J. Characterization of the Arabidopsis thermosensitive mutant atts02 reveals an important role for galactolipids in thermotolerance. Plant. Cell Environ. 2006, 29, 1437–1448. [Google Scholar] [CrossRef]
- Shiva, S.; Samarakoon, T.; Lowe, K.A.; Roach, C.; Vu, H.S.; Colter, M.; Porras, H.; Hwang, C.; Roth, M.R.; Tamura, P.; et al. Leaf Lipid Alterations in Response to Heat Stress of Arabidopsis thaliana. Plants 2020, 9, 845. [Google Scholar] [CrossRef] [PubMed]
- Mueller, S.P.; Unger, M.; Guender, L.; Fekete, A.; Mueller, M.J. Phospholipid: Diacylglycerol Acyltransferase-Mediated Triacylglyerol Synthesis Augments Basal Thermotolerance. Plant Physiol. 2017, 175, 486–497. [Google Scholar] [CrossRef]
- Süss, K.-H.; Yordanov, I.T. Biosynthetic Cause of in Vivo Acquired Thermotolerance of Photosynthetic Light Reactions and Metabolic Responses of Chloroplasts to Heat Stress. Plant Physiol. 1986, 81, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, S.; Tamura, P.J.; Roth, M.R.; Prasad, P.V.V.; Welti, R. Wheat leaf lipids during heat stress: I. High day and night temperatures result in major lipid alterations. Plant. Cell Environ. 2016, 39, 787–803. [Google Scholar] [CrossRef]
- Mueller, S.P.; Krause, D.M.; Mueller, M.J.; Fekete, A. Accumulation of extra-chloroplastic triacylglycerols inArabidopsisseedlings during heat acclimation. J. Exp. Bot. 2015, 66, 4517–4526. [Google Scholar] [CrossRef]
- Moellering, E.R.; Muthan, B.; Benning, C. Freezing Tolerance in Plants Requires Lipid Remodeling at the Outer Chloroplast Membrane. Science 2010, 330, 226–228. [Google Scholar] [CrossRef]
- Barnes, A.C.; Benning, C.; Roston, R.L. Chloroplast Membrane Remodeling during Freezing Stress Is Accompanied by Cytoplasmic Acidification Activating SENSITIVE TO FREEZING2. Plant Physiol. 2016, 171, 2140–2149. [Google Scholar] [CrossRef]
- Roston, R.L.; Wang, K.; Kuhn, L.A.; Benning, C. Structural Determinants Allowing Transferase Activity in SENSITIVE TO FREEZING 2, Classified as a Family I Glycosyl Hydrolase. J. Biol. Chem. 2014, 289, 26089–26106. [Google Scholar] [CrossRef]
- Fourrier, N.; Bédard, J.; Lopez-Juez, E.; Barbrook, A.; Bowyer, J.; Jarvis, P.; Warren, G.; Thorlby, G. A role forSENSITIVE TO FREEZING2in protecting chloroplasts against freeze-induced damage in Arabidopsis. Plant J. 2008, 55, 734–745. [Google Scholar] [CrossRef] [PubMed]
- Warren, G.; McKown, R.; Marin, A.; Teutonico, R. Isolation of Mutations Affecting the Development of Freezing Tolerance in Arabidopsis thaliana (L.) Heynh. Plant Physiol. 1996, 111, 1011–1019. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, Y. Phosphate starvation and membrane lipid remodeling in seed plants. Prog. Lipid Res. 2013, 52, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Gaude, N.; Bréhélin, C.; Tischendorf, G.; Kessler, F.; Dörmann, P. Nitrogen deficiency in Arabidopsis affects galactolipid composition and gene expression and results in accumulation of fatty acid phytyl esters. Plant J. 2007, 49, 729–739. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, M.A.J.; Hart, G.W.; Kinoshita, T. Glycosylphosphatidylinositol Anchors. In Essentials of Glycobiology; Varki, A., Cummings, D., Esko, J.D., Stanley, P., Hart, G.W., Aebi, M., Darvill, A.G., Kinoshita, T., Packer, N.H., Prestegard, J.H., et al., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2017; pp. 476–478. ISBN 9780123786319. [Google Scholar]
- Kinoshita, T.; Fujita, M. Thematic Review Series: Glycosylphosphatidylinositol (GPI) Anchors: Biochemistry and Cell Biology Biosynthesis of GPI-anchored proteins: Special emphasis on GPI lipid remodeling. J. Lipid Res. 2016, 57, 6–24. [Google Scholar] [CrossRef]
- Zhou, K. Glycosylphosphatidylinositol-Anchored Proteins in Arabidopsis and One of Their Common Roles in Signaling Transduction. Front. Plant Sci. 2019, 10. [Google Scholar] [CrossRef]
- Yeats, T.H.; Bacic, A.; Johnson, K.L. Plant glycosylphosphatidylinositol anchored proteins at the plasma membrane-cell wall nexus. J. Integr. Plant Biol. 2018, 60, 649–669. [Google Scholar] [CrossRef]
- Beihammer, G.; Maresch, D.; Altmann, F.; Strasser, R. Glycosylphosphatidylinositol-Anchor Synthesis in Plants: A Glycobiology Perspective. Front. Plant Sci. 2020, 11. [Google Scholar] [CrossRef]
- Oxley, D.; Bacic, A. Structure of the glycosylphosphatidylinositol anchor of an arabinogalactan protein from Pyrus communis suspension-cultured cells. Proc. Natl. Acad. Sci. USA 1999, 96, 14246–14251. [Google Scholar] [CrossRef]
- Cheung, A.Y.; Li, C.; Zou, Y.J.; Wu, H.M. Glycosylphosphatidylinositol Anchoring: Control through Modification. Plant Physiol. 2014, 166, 748–750. [Google Scholar] [CrossRef]
- Lalanne, E.; Honys, D.; Johnson, A.; Borner, G.H.; Lilley, K.S.; Dupree, P.; Grossniklaus, U.; Twell, D. SETH1 and SETH2, two components of the glycosylphosphatidylinositol anchor biosynthetic pathway, are required for pollen germination and tube growth in Arabidopsis. Plant Cell 2004, 16, 229–240. [Google Scholar] [CrossRef] [PubMed]
- Desnoyer, N.; Palanivelu, R. Bridging the GAPs in plant reproduction: A comparison of plant and animal GPI-anchored proteins. Plant Reprod. 2020, 33, 129–142. [Google Scholar] [CrossRef]
- Poisson, G.; Chauve, C.; Chen, X.; Bergeron, A. FragAnchor: A Large-Scale Predictor of Glycosylphosphatidylinositol Anchors in Eukaryote Protein Sequences by Qualitative Scoring. Genom. Proteom. Bioinform. 2007, 5, 121–130. [Google Scholar] [CrossRef]
- Zhou, K. The alternative splicing of SKU5-Similar3 in Arabidopsis. Plant Signal. Behav. 2019, 14, e1651182. [Google Scholar] [CrossRef] [PubMed]
- Grojean, J.; Downes, B. Riboswitches as hormone receptors: Hypothetical cytokinin-binding riboswitches in Arabidopsis thaliana. Biol. Direct 2010, 5, 60. [Google Scholar] [CrossRef]
- Millar, A.H.; Heazlewood, J.L.; Giglione, C.; Holdsworth, M.J.; Bachmair, A.; Schulze, W.X. The Scope, Functions, and Dynamics of Posttranslational Protein Modifications. Annu. Rev. Plant Biol. 2019, 70, 119–151. [Google Scholar] [CrossRef] [PubMed]
- Ytterberg, A.J.; Jensen, O.N. Modification-specific proteomics in plant biology. J. Proteom. 2010, 73, 2249–2266. [Google Scholar] [CrossRef]
- Borner, G.H.H.; Sherrier, D.J.; Stevens, T.J.; Arkin, I.T.; Dupree, P. Prediction of Glycosylphosphatidylinositol-Anchored Proteins in Arabidopsis. A Genomic Analysis: Table I. Plant Physiol. 2002, 129, 486–499. [Google Scholar] [CrossRef]
- Leipelt, M.; Warnecke, D.; Zähringer, U.; Ott, C.; Müller, F.; Hube, B.; Heinz, E. Glucosylceramide Synthases, a Gene Family Responsible for the Biosynthesis of Glucosphingolipids in Animals, Plants, and Fungi. J. Biol. Chem. 2001, 276, 33621–33629. [Google Scholar] [CrossRef]
- Cacas, J.-L.; Buré, C.; Grosjean, K.; Gerbeau-Pissot, P.; Lherminier, J.; Rombouts, Y.; Maes, E.; Bossard, C.; Gronnier, J.; Furt, F.; et al. Revisiting Plant Plasma Membrane Lipids in Tobacco: A Focus on Sphingolipids. Plant Physiol. 2016, 170, 367–384. [Google Scholar] [CrossRef]
- Mamode Cassim, A.; Grison, M.; Ito, Y.; Simon-Plas, F.; Mongrand, S.; Boutté, Y. Sphingolipids in plants: A guidebook on their function in membrane architecture, cellular processes, and environmental or developmental responses. FEBS Lett. 2020, 594, 3719–3738. [Google Scholar] [CrossRef] [PubMed]
- Mortimer, J.C.; Scheller, H.V. Synthesis and Function of Complex Sphingolipid Glycosylation. Trends Plant Sci. 2020, 25, 522–524. [Google Scholar] [CrossRef] [PubMed]
- Warnecke, D.; Heinz, E. Recently discovered functions of glucosylceramides in plants and fungi. Cell. Mol. Life Sci. 2003, 60, 919–941. [Google Scholar] [CrossRef]
- Mamode Cassim, A.; Gouguet, P.; Gronnier, J.; Laurent, N.; Germain, V.; Grison, M.; Boutté, Y.; Gerbeau-Pissot, P.; Simon-Plas, F.; Mongrand, S. Plant lipids: Key players of plasma membrane organization and function. Prog. Lipid Res. 2019, 73, 1–27. [Google Scholar] [CrossRef]
- Luttgeharm, K.D.; Kimberlin, A.N.; Cahoon, E.B. Plant Sphingolipid Metabolism and Function. In Lipids in Plant and Algae Development; Nakamura, Y., Li-Beisson, Y., Eds.; Springer International Publishing: Berlin/Heidelberg, Germany, 2016; Volume 86, pp. 249–286. ISBN 0306-0225. [Google Scholar]
- Pata, M.O.; Hannun, Y.A.; Ng, C.K.Y. Plant sphingolipids: Decoding the enigma of the Sphinx. New Phytol. 2010, 185, 611–630. [Google Scholar] [CrossRef]
- Huby, E.; Napier, J.A.; Baillieul, F.; Michaelson, L.V.; Dhondt-Cordelier, S. Sphingolipids: Towards an integrated view of metabolism during the plant stress response. New Phytol. 2020, 225, 659–670. [Google Scholar] [CrossRef] [PubMed]
- Zäuner, S.; Ternes, P.; Warnecke, D. Biosynthesis of Sphingolipids in Plants (and Some of Their Functions). In Sphingolipids as Signaling and Regulatory Molecules; Chalfant, C., Del Poeta, M., Eds.; Springer: New York, NY, USA, 2010. [Google Scholar]
- Markham, J.E.; Li, J.; Cahoon, E.B.; Jaworski, J.G. Separation and Identification of Major Plant Sphingolipid Classes from Leaves. J. Biol. Chem. 2006, 281, 22684–22694. [Google Scholar] [CrossRef]
- Daleke, D.L. Regulation of transbilayer plasma membrane phospholipid asymmetry. J. Lipid Res. 2003, 44, 233–242. [Google Scholar] [CrossRef] [PubMed]
- Buré, C.; Cacas, J.-L.; Mongrand, S.; Schmitter, J.-M. Characterization of glycosyl inositol phosphoryl ceramides from plants and fungi by mass spectrometry. Anal. Bioanal. Chem. 2014, 406, 995–1010. [Google Scholar] [CrossRef]
- Borner, G.H.H.; Sherrier, D.J.; Weimar, T.; Michaelson, L.V.; Hawkins, N.D.; Macaskill, A.; Napier, J.A.; Beale, M.H.; Lilley, K.S.; Dupree, P. Analysis of Detergent-Resistant Membranes in Arabidopsis. Evidence for Plasma Membrane Lipid Rafts. Plant Physiol. 2005, 137, 104–116. [Google Scholar] [CrossRef] [PubMed]
- Tapken, W.; Murphy, A.S. Membrane nanodomains in plants: Capturing form, function, and movement. J. Exp. Bot. 2015, 66, 1573–1586. [Google Scholar] [CrossRef]
- Iswanto, A.B.B.; Kim, J.-Y. Lipid Raft, Regulator of Plasmodesmal Callose Homeostasis. Plants 2017, 6, 15. [Google Scholar] [CrossRef]
- Gronnier, J.; Germain, V.; Gouguet, P.; Cacas, J.-L.; Mongrand, S. GIPC: Glycosyl Inositol Phospho Ceramides, the major sphingolipids on earth. Plant Signal. Behav. 2016, 11, e1152438. [Google Scholar] [CrossRef] [PubMed]
- Cacas, J.-L.; Buré, C.; Furt, F.; Maalouf, J.-P.; Badoc, A.; Cluzet, S.; Schmitter, J.-M.; Antajan, E.; Mongrand, S. Biochemical survey of the polar head of plant glycosylinositolphosphoceramides unravels broad diversity. Phytochemistry 2013, 96, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, T.; Fang, L.; Rennie, E.A.; Sechet, J.; Yan, J.; Jing, B.; Moore, W.; Cahoon, E.B.; Scheller, H.V.; Kawai-Yamada, M.; et al. GLUCOSAMINE INOSITOLPHOSPHORYLCERAMIDE TRANSFERASE1 (GINT1) Is a GlcNAc-Containing Glycosylinositol Phosphorylceramide Glycosyltransferase. Plant Physiol. 2018, 177, 938–952. [Google Scholar] [CrossRef]
- Lynch, D.V.; Dunn, T.M. An introduction to plant sphingolipids and a review of recent advances in understanding their metabolism and function. New Phytol. 2004, 161, 677–702. [Google Scholar] [CrossRef]
- Michaelson, L.V.; Napier, J.A.; Molino, D.; Faure, J.D. Plant sphingolipids: Their importance in cellular organization and adaption. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2016, 1861, 1329–1335. [Google Scholar] [CrossRef]
- Melser, S.; Batailler, B.; Peypelut, M.; Poujol, C.; Bellec, Y.; Wattelet-Boyer, V.; Maneta-Peyret, L.; Faure, J.-D.; Moreau, P. Glucosylceramide Biosynthesis is Involved in Golgi Morphology and Protein Secretion in Plant Cells. Traffic 2010, 11, 479–490. [Google Scholar] [CrossRef]
- Melser, S.; Molino, D.; Batailler, B.; Peypelut, M.; Laloi, M.; Wattelet-Boyer, V.; Bellec, Y.; Faure, J.-D.; Moreau, P. Links between lipid homeostasis, organelle morphodynamics and protein trafficking in eukaryotic and plant secretory pathways. Plant Cell Rep. 2011, 30, 177–193. [Google Scholar] [CrossRef]
- Uemura, M.; Steponkus, P.L. A Contrast of the Plasma Membrane Lipid Composition of Oat and Rye Leaves in Relation to Freezing Tolerance. Plant Physiol. 1994, 104, 479–496. [Google Scholar] [CrossRef]
- Xie, L.-J.; Chen, Q.-F.; Chen, M.-X.; Yu, L.-J.; Huang, L.; Chen, L.; Wang, F.-Z.; Xia, F.-N.; Zhu, T.-R.; Wu, J.-X.; et al. Unsaturation of Very-Long-Chain Ceramides Protects Plant from Hypoxia-Induced Damages by Modulating Ethylene Signaling in Arabidopsis. PLoS Genet. 2015, 11, e1005143. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.-J.; Yu, L.-J.; Chen, Q.-F.; Wang, F.-Z.; Huang, L.; Xia, F.-N.; Zhu, T.-R.; Wu, J.-X.; Yin, J.; Liao, B.; et al. Arabidopsis acyl-CoA-binding protein ACBP 3 participates in plant response to hypoxia by modulating very-long-chain fatty acid metabolism. Plant J. 2015, 81, 53–67. [Google Scholar] [CrossRef] [PubMed]
- Voxeur, A.; Fry, S.C. Glycosylinositol phosphorylceramides fromRosacell cultures are boron-bridged in the plasma membrane and form complexes with rhamnogalacturonan II. Plant J. 2014, 79, 139–149. [Google Scholar] [CrossRef]
- Ji, H.; Pardo, J.M.; Batelli, G.; Van Oosten, M.J.; Bressan, R.A.; Li, X. The Salt Overly Sensitive (SOS) Pathway: Established and Emerging Roles. Mol. Plant 2013, 6, 275–286. [Google Scholar] [CrossRef]
- Jiang, Z.; Zhou, X.; Tao, M.; Yuan, F.; Liu, L.; Wu, F.; Wu, X.; Xiang, Y.; Niu, Y.; Liu, F.; et al. Plant cell-surface GIPC sphingolipids sense salt to trigger Ca2+ influx. Nat. Artic. 2019, 572, 341–346. [Google Scholar] [CrossRef]
- Lenarčič, T.; Albert, I.; Böhm, H.; Hodnik, V.; Pirc, K.; Zavec, A.B.; Podobnik, M.; Pahovnik, D.; Žagar, E.; Pruitt, R.; et al. Eudicot plant-specific sphingolipids determine host selectivity of microbial NLP cytolysins. Science 2017, 358, 1431–1434. [Google Scholar] [CrossRef]
- Baena-González, E.; Hanson, J. Shaping plant development through the SnRK1–TOR metabolic regulators. Curr. Opin. Plant Biol. 2017, 35, 152–157. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Sheen, J. Dynamic and diverse sugar signaling. Curr. Opin. Plant Biol. 2016, 33, 116–125. [Google Scholar] [CrossRef] [PubMed]
- Bledsoe, S.W.; Henry, C.; Griffiths, C.A.; Paul, M.J.; Feil, R.; Lunn, J.E.; Stitt, M.; Lagrimini, L.M. The role of Tre6P and SnRK1 in maize early kernel development and events leading to stress-induced kernel abortion. BMC Plant Biol. 2017, 17, 74. [Google Scholar] [CrossRef]
- Lastdrager, J.; Hanson, J.; Smeekens, S. Sugar signals and the control of plant growth and development. J. Exp. Bot. 2014, 65, 799–807. [Google Scholar] [CrossRef]
- Ponnu, J.; Schlereth, A.; Zacharaki, V.; Działo, M.A.; Abel, C.; Feil, R.; Schmid, M.; Wahl, V. The trehalose 6-phosphate pathway impacts vegetative phase change in Arabidopsis thaliana. Plant J. 2020, 104, 768–780. [Google Scholar] [CrossRef] [PubMed]
- Wingler, A.; Delatte, T.L.; O’Hara, L.E.; Primavesi, L.F.; Jhurreea, D.; Paul, M.J.; Schluepmann, H. Trehalose 6-Phosphate Is Required for the Onset of Leaf Senescence Associated with High Carbon Availability. Plant Physiol. 2012, 158, 1241–1251. [Google Scholar] [CrossRef] [PubMed]
- Baena-González, E.; Rolland, F.; Thevelein, J.M.; Sheen, J. A central integrator of transcription networks in plant stress and energy signalling. Nat. Lett. 2007, 448, 938–942. [Google Scholar] [CrossRef]
- Broeckx, T.; Hulsmans, S.; Rolland, F. The plant energy sensor: Evolutionary conservation and divergence of SnRK1 structure, regulation, and function. J. Exp. Bot. 2016, 67, 6215–6252. [Google Scholar] [CrossRef] [PubMed]
- Nunes, C.; Primavesi, L.F.; Patel, M.K.; Martinez-Barajas, E.; Powers, S.J.; Sagar, R.; Fevereiro, P.S.; Davis, B.G.; Paul, M.J. Inhibition of SnRK1 by metabolites: Tissue-dependent effects and cooperative inhibition by glucose 1-phosphate in combination with trehalose 6-phosphate. Plant Physiol. Biochem. 2013, 63, 89–98. [Google Scholar] [CrossRef]
- Toroser, D.; Plaut, Z.; Huber, S.C. Regulation of a Plant SNF1-Related Protein Kinase by Glucose-6-Phosphate. Plant Physiol. 2000, 123, 403–412. [Google Scholar] [CrossRef]
- Zhang, Y.; Primavesi, L.F.; Jhurreea, D.; Andralojc, P.J.; Mitchell, R.A.C.; Powers, S.J.; Schluepmann, H.; Delatte, T.; Wingler, A.; Paul, M.J. Inhibition of SNF1-Related Protein Kinase1 Activity and Regulation of Metabolic Pathways by Trehalose-6-Phosphate. Plant Physiol. 2009, 149, 1860–1871. [Google Scholar] [CrossRef]
- Ryabova, L.A.; Robaglia, C.; Meyer, C. Target of Rapamycin kinase: Central regulatory hub for plant growth and metabolism. J. Exp. Bot. 2019, 70, 2211–2216. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.; McCormack, M.; Li, L.; Hall, Q.; Xiang, C.; Sheen, J. Glucose–TOR signalling reprograms the transcriptome and activates meristems. Nat. Artic. 2013, 496, 181–186. [Google Scholar] [CrossRef]
- Radchuk, R.; Emery, R.J.N.; Weier, D.; Vigeolas, H.; Geigenberger, P.; Lunn, J.E.; Feil, R.; Weschke, W.; Weber, H. Sucrose non-fermenting kinase 1 (SnRK1) coordinates metabolic and hormonal signals during pea cotyledon growth and differentiation. Plant J. 2009, 61, 324–338. [Google Scholar] [CrossRef] [PubMed]
- Radchuk, R.; Radchuk, V.; Weschke, W.; Borisjuk, L.; Weber, H. Repressing the Expression of the SUCROSE NONFERMENTING-1-RELATED PROTEIN KINASE Gene in Pea Embryo Causes Pleiotropic Defects of Maturation Similar to an Abscisic Acid-Insensitive Phenotype. Plant Physiol. 2006, 140, 263–278. [Google Scholar] [CrossRef]
- Tsai, A.Y.L.; Gazzarrini, S. Trehalose-6-phosphate and SnRK1 kinases in plant development and signaling: The emerging picture. Front. Plant Sci. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Belda-Palazón, B.; Adamo, M.; Valerio, C.; Ferreira, L.J.; Confraria, A.; Reis-Barata, D.; Rodrigues, A.; Meyer, C.; Rodriguez, P.L.; Baena-González, E. A dual function of SnRK2 kinases in the regulation of SnRK1 and plant growth. Nat. Plants 2020, 6, 1345–1353. [Google Scholar] [CrossRef]
- Chan, A.; Carianopol, C.; Tsai, A.Y.-L.; Varatharajah, K.; Chiu, R.S.; Gazzarrini, S. SnRK1 phosphorylation of FUSCA3 positively regulates embryogenesis, seed yield, and plant growth at high temperature in Arabidopsis. J. Exp. Bot. 2017, 68, 4219–4231. [Google Scholar] [CrossRef] [PubMed]
- Meitzel, T.; Radchuk, R.; McAdam, E.L.; Thormählen, I.; Feil, R.; Munz, E.; Hilo, A.; Geigenberger, P.; Ross, J.J.; Lunn, J.E.; et al. Trehalose 6-phosphate promotes seed filling by activating auxin biosynthesis. New Phytol. 2021, 229, 1553–1565. [Google Scholar] [CrossRef]
- Menand, B.; Desnos, T.; Nussaume, L.; Berger, F.; Bouchez, D.; Meyer, C.; Robaglia, C. Expression and disruption of the Arabidopsis TOR (target of rapamycin) gene. Proc. Natl. Acad. Sci. USA 2002, 99, 6422–6427. [Google Scholar] [CrossRef]
- Ahn, C.S.; Ahn, H.-K.; Pai, H.-S. Overexpression of the PP2A regulatory subunit Tap46 leads to enhanced plant growth through stimulation of the TOR signalling pathway. J. Exp. Bot. 2015, 66, 827–840. [Google Scholar] [CrossRef]
- Deprost, D.; Yao, L.; Sormani, R.; Moreau, M.; Leterreux, G.; Nicolaï, M.; Bedu, M.; Robaglia, C.; Meyer, C. The Arabidopsis TOR kinase links plant growth, yield, stress resistance and mRNA translation. EMBO Rep. 2007, 8, 864–870. [Google Scholar] [CrossRef]
- Roldan, M.; Gomez-Mena, C.; Ruiz-Garcia, L.; Salinas, J.; Martinez-Zapater, J.M. Sucrose availability on the aerial part of the plant promotes morphogenesis and flowering of Arabidopsis in the dark. Plant J. 1999, 20, 581–590. [Google Scholar] [CrossRef]
- Dobrenel, T.; Caldana, C.; Hanson, J.; Robaglia, C.; Vincentz, M.; Veit, B.; Meyer, C. TOR Signaling and Nutrient Sensing. Annu. Rev. Plant Biol. 2016, 67, 261–285. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Song, Y.; Wang, K.; Dong, P.; Zhang, X.; Li, F.; Li, Z.; Ren, M. TOR-inhibitor insensitive-1 (TRIN1) regulates cotyledons greening in Arabidopsis. Front. Plant Sci. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Pfeiffer, A.; Janocha, D.; Dong, Y.; Medzihradszky, A.; Schöne, S.; Daum, G.; Suzaki, T.; Forner, J.; Langenecker, T.; Rempel, E.; et al. Integration of light and metabolic signals for stem cell activation at the shoot apical meristem. Elife 2016, 5, e17023. [Google Scholar] [CrossRef]
- Carvalho, R.F.; Szakonyi, D.; Simpson, C.G.; Barbosa, I.C.R.; Brown, J.W.S.; Baena-González, E.; Duque, P. The Arabidopsis SR45 Splicing Factor, a Negative Regulator of Sugar Signaling, Modulates SNF1-Related Protein Kinase 1 Stability. Plant Cell 2016, 28, 1910–1925. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Xu, M.; Koo, Y.; He, J.; Poethig, R.S. Sugar promotes vegetative phase change in Arabidopsis thaliana by repressing the expression of MIR156A and MIR156C. Elife 2013, 2, e00260. [Google Scholar] [CrossRef]
- Yu, S.; Cao, L.; Zhou, C.-M.; Zhang, T.-Q.; Lian, H.; Sun, Y.; Wu, J.; Huang, J.; Wang, G.; Wang, J.-W. Sugar is an endogenous cue for juvenile-to-adult phase transition in plants. Elife 2013, 2, e00269. [Google Scholar] [CrossRef] [PubMed]
- Mutanwad, K.V.; Neumayer, N.; Freitag, C.; Zangl, I.; Lucyshyn, D. O-glycosylation of SPL transcription factors regulates plant developmental transitions downstream of miR156. bioRxiv 2019. [Google Scholar] [CrossRef]
- Mason, M.G.; Ross, J.J.; Babst, B.A.; Wienclaw, B.N.; Beveridge, C.A. Sugar demand, not auxin, is the initial regulator of apical dominance. Proc. Natl. Acad. Sci. USA 2014, 111, 6092–6097. [Google Scholar] [CrossRef]
- Heyer, A.G.; Raap, M.; Schroeer, B.; Marty, B.; Willmitzer, L. Cell wall invertase expression at the apical meristem alters floral, architectural, and reproductive traits in Arabidopsis thaliana. Plant J. 2004, 39, 161–169. [Google Scholar] [CrossRef]
- Schluepmann, H.; Pellny, T.; Van Dijken, A.; Smeekens, S.; Paul, M. Trehalose 6-phosphate is indispensable for carbohydrate utilization and growth in Arabidopsis thaliana. Proc. Natl. Acad. Sci. USA 2003, 100, 6849–6854. [Google Scholar] [CrossRef] [PubMed]
- Van Dijken, A.J.H.; Schluepmann, H.; Smeekens, S.C.M. Arabidopsis Trehalose-6-Phosphate Synthase 1 Is Essential for Normal Vegetative Growth and Transition to Flowering. Plant Physiol. 2004, 135, 969–977. [Google Scholar] [CrossRef] [PubMed]
- Fichtner, F.; Barbier, F.F.; Annunziata, M.G.; Feil, R.; Olas, J.J.; Mueller-Roeber, B.; Stitt, M.; Beveridge, C.A.; Lunn, J.E. Regulation of shoot branching in arabidopsis by trehalose 6-phosphate. New Phytol. 2021, 229, 2135–2151. [Google Scholar] [CrossRef]
- Anderson, G.H.; Veit, B.; Hanson, M.R. The Arabidopsis AtRaptor genes are essential for post-embryonic plant growth. BMC Biol. 2005, 3, 12. [Google Scholar] [CrossRef] [PubMed]
- Moreau, M.; Azzopardi, M.; Clément, G.; Dobrenel, T.; Marchive, C.; Renne, C.; Martin-Magniette, M.-L.; Taconnat, L.; Renou, J.-P.; Robaglia, C.; et al. Mutations in the Arabidopsis Homolog of LST8/GβL, a Partner of the Target of Rapamycin Kinase, Impair Plant Growth, Flowering, and Metabolic Adaptation to Long Days. Plant Cell 2012, 24, 463–481. [Google Scholar] [CrossRef]
- Brunkard, J.O.; Xu, M.; Scarpin, M.R.; Chatterjee, S.; Shemyakina, E.A.; Goodman, H.M.; Zambryski, P. TOR dynamically regulates plant cell–cell transport. Proc. Natl. Acad. Sci. USA 2020, 117, 5049–5058. [Google Scholar] [CrossRef]
- Kelly, G.; David-Schwartz, R.; Sade, N.; Moshelion, M.; Levi, A.; Alchanatis, V.; Granot, D. The Pitfalls of Transgenic Selection and New Roles of AtHXK1: A High Level of AtHXK1 Expression Uncouples Hexokinase1-Dependent Sugar Signaling from Exogenous Sugar. Plant Physiol. 2012, 159, 47–51. [Google Scholar] [CrossRef] [PubMed]
- Barbier, F.F.; Cao, D.; Fichtner, F.; Weiste, C.; Perez-Garcia, M.-D.; Caradeuc, M.; Le Gourrierec, J.; Sakr, S.; Beveridge, C.A. HEXOKINASE1 signalling promotes shoot branching and interacts with cytokinin and strigolactone pathways. New Phytol. 2021. [Google Scholar] [CrossRef]
- Williams, S.P.; Rangarajan, P.; Donahue, J.L.; Hess, J.E.; Gillaspy, G.E. Regulation of Sucrose non-Fermenting Related Kinase 1 genes in Arabidopsis thaliana. Front. Plant Sci. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.-H.; Hong, J.-W.; Kim, E.-C.; Yoo, S.-D. Regulatory Functions of SnRK1 in Stress-Responsive Gene Expression and in Plant Growth and Development. Plant Physiol. 2012, 158, 1955–1964. [Google Scholar] [CrossRef] [PubMed]
- Ren, M.; Qiu, S.; Venglat, P.; Xiang, D.; Feng, L.; Selvaraj, G.; Datla, R. Target of Rapamycin Regulates Development and Ribosomal RNA Expression through Kinase Domain in Arabidopsis. Plant Physiol. 2011, 155, 1367–1382. [Google Scholar] [CrossRef] [PubMed]
- Avila-Ospina, L.; Moison, M.; Yoshimoto, K.; Masclaux-Daubresse, C. Autophagy, plant senescence, and nutrient recycling. J. Exp. Bot. 2014, 65, 3799–3811. [Google Scholar] [CrossRef] [PubMed]
- Janse van Rensburg, H.C.; Van den Ende, W.; Signorelli, S. Autophagy in Plants: Both a Puppet and a Puppet Master of Sugars. Front. Plant Sci. 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Bassham, D.C. TOR Is a Negative Regulator of Autophagy in Arabidopsis thaliana. PLoS ONE 2010, 5, e11883. [Google Scholar] [CrossRef] [PubMed]
- Soto-Burgos, J.; Bassham, D.C. SnRK1 activates autophagy via the TOR signaling pathway in Arabidopsis thaliana. PLoS ONE 2017, 12, e0182591. [Google Scholar] [CrossRef]
- Liu, J.-X.; Howell, S.H. Endoplasmic Reticulum Protein Quality Control and Its Relationship to Environmental Stress Responses in Plants. Plant Cell 2010, 22, 2930–2942. [Google Scholar] [CrossRef]
- Da-Silva, W.S.; Rezende, G.L.; Galina, A. Subcellular distribution and kinetic properties of cytosolic and non-cytosolic hexokinases in maize seedling roots: Implications for hexose phosphorylation. J. Exp. Bot. 2001, 52, 1191–1201. [Google Scholar] [CrossRef]
- Galina, A.; Da Silva, W.S. Hexokinase activity alters sugar-nucleotide formation in maize root homogenates. Phytochemistry 2000, 53, 29–37. [Google Scholar] [CrossRef]
- Herbers, K.; Meuwly, P.; Frommer, W.B.; Metraux, J.P.; Sonnewald, U. Systemic Acquired Resistance Mediated by the Ectopic Expression of Invertase: Possible Hexose Sensing in the Secretory Pathway. Plant Cell 1996, 8, 793–803. [Google Scholar] [CrossRef] [PubMed]
- van der Harg, J.M.; van Heest, J.C.; Bangel, F.N.; Patiwael, S.; van Weering, J.R.T.; Scheper, W. The UPR reduces glucose metabolism via IRE1 signaling. Biochim. Biophys. Acta Mol. Cell Res. 2017, 1864, 655–665. [Google Scholar] [CrossRef]
- Fujiki, R.; Hashiba, W.; Sekine, H.; Yokoyama, A.; Chikanishi, T.; Ito, S.; Imai, Y.; Kim, J.; He, H.H.; Igarashi, K.; et al. GlcNAcylation of histone H2B facilitates its monoubiquitination. Nat. Lett. 2011, 480, 557–560. [Google Scholar] [CrossRef]
- Wu, D.; Cai, Y.; Jin, J. Potential coordination role between O-GlcNAcylation and epigenetics. Protein Cell 2017, 8, 713–723. [Google Scholar] [CrossRef]
- Zhang, X.; Zheng, X.; Ke, S.; Zhu, H.; Liu, F.; Zhang, Z.; Peng, X.; Guo, L.; Zeng, R.; Hou, P.; et al. ER-localized adenine nucleotide transporter ER-ANT1: An integrator of energy and stress signaling in rice. Plant Mol. Biol. 2016, 92, 701–715. [Google Scholar] [CrossRef] [PubMed]
- Bullen, J.W.; Balsbaugh, J.L.; Chanda, D.; Shabanowitz, J.; Hunt, D.F.; Neumann, D.; Hart, G.W. Cross-talk between Two Essential Nutrient-sensitive Enzymes. J. Biol. Chem. 2014, 289, 10592–10606. [Google Scholar] [CrossRef] [PubMed]
- Reyes, F.; Marchant, L.; Norambuena, L.; Nilo, R.; Silva, H.; Orellana, A. AtUTr1, a UDP-glucose/UDP-galactose Transporter from Arabidopsis thaliana, Is Located in the Endoplasmic Reticulum and Up-regulated by the Unfolded Protein Response*. J. Biol. Chem. 2006, 281, 9145–9151. [Google Scholar] [CrossRef] [PubMed]
- Handford, M.; Rodríguez-Furlán, C.; Marchant, L.; Segura, M.; Gómez, D.; Alvarez-Buylla, E.; Xiong, G.-Y.; Pauly, M.; Orellana, A. Arabidopsis thaliana AtUTr7 Encodes a Golgi-Localized UDP–Glucose/UDP–Galactose Transporter that Affects Lateral Root Emergence. Mol. Plant 2012, 5, 1263–1280. [Google Scholar] [CrossRef]
- Janse van Rensburg, H.C.; Van den Ende, W. UDP-Glucose: A Potential Signaling Molecule in Plants? Front. Plant Sci. 2018, 8. [Google Scholar] [CrossRef] [PubMed]
- Park, J.-I.; Ishimizu, T.; Suwabe, K.; Sudo, K.; Masuko, H.; Hakozaki, H.; Nou, I.-S.; Suzuki, G.; Watanabe, M. UDP-Glucose Pyrophosphorylase is Rate Limiting in Vegetative and Reproductive Phases in Arabidopsis thaliana. Plant Cell Physiol. 2010, 51, 981–996. [Google Scholar] [CrossRef]
- Chivasa, S.; Tomé, D.F.A.; Slabas, A.R. UDP-Glucose Pyrophosphorylase Is a Novel Plant Cell Death Regulator. J. Proteome Res. 2013, 12, 1743–1753. [Google Scholar] [CrossRef]
- Li, Y.; Van den Ende, W.; Rolland, F. Sucrose Induction of Anthocyanin Biosynthesis Is Mediated by DELLA. Mol. Plant 2014, 7, 570–572. [Google Scholar] [CrossRef]
- Paweł Tarkowski, Ł.; Van De Poel, B.; Höfte, M.; Van den Ende, W. Molecular Sciences Sweet Immunity: Inulin Boosts Resistance of Lettuce (Lactuca sativa) against Grey Mold (Botrytis cinerea) in an Ethylene-Dependent Manner. Int. J. Mol. Sci. 2019, 20, 52. [Google Scholar] [CrossRef]
- Sagervanshi, A.; Naeem, A.; Geilfus, C.-M.; Kaiser, H.; Mühling, K.H. One-time abscisic acid priming induces long-term salinity resistance in Vicia faba: Changes in key transcripts, metabolites, and ionic relations. Physiol. Plant. 2020, 172, 146–161. [Google Scholar] [CrossRef]
- Jia, X.; Zeng, H.; Bose, S.K.; Wang, W.; Yin, H. Chitosan oligosaccharide induces resistance to Pst DC3000 in Arabidopsis via a non-canonical N-glycosylation regulation pattern. Carbohydr. Polym. 2020, 250, 116939. [Google Scholar] [CrossRef] [PubMed]
- Mohnike, L.; Rekhter, D.; Huang, W.; Feussner, K.; Tian, H.; Herrfurth, C.; Zhang, Y.; Feussner, I. The glycosyltransferase UGT76B1 is critical for plant immunity as it governs the homeostasis of N-hydroxy-pipecolic acid. bioRxiv 2020. [Google Scholar] [CrossRef]
- Jiao, Q.S.; Niu, G.T.; Wang, F.F.; Dong, J.Y.; Chen, T.S.; Zhou, C.F.; Hong, Z. N-glycosylation regulates photosynthetic efficiency of Arabidopsis thaliana. Photosynthetica 2020, 58, 72–79. [Google Scholar] [CrossRef]
- Moradi, A.; El-Shetehy, M.; Gamir, J.; Austerlitz, T.; Dahlin, P.; Wieczorek, K.; Künzler, M.; Mauch, F. Expression of a fungal lectin in Arabidopsis enhances plant growth and resistance towards microbial pathogens and plant-parasitic nematode. bioRxiv 2021. [Google Scholar] [CrossRef]
- Zhang, L.; Paasch, B.C.; Chen, J.; Day, B.; He, S.Y. An important role ofl-fucose biosynthesis and protein fucosylation genes in Arabidopsis immunity. New Phytol. 2019, 222, 981–994. [Google Scholar] [CrossRef]
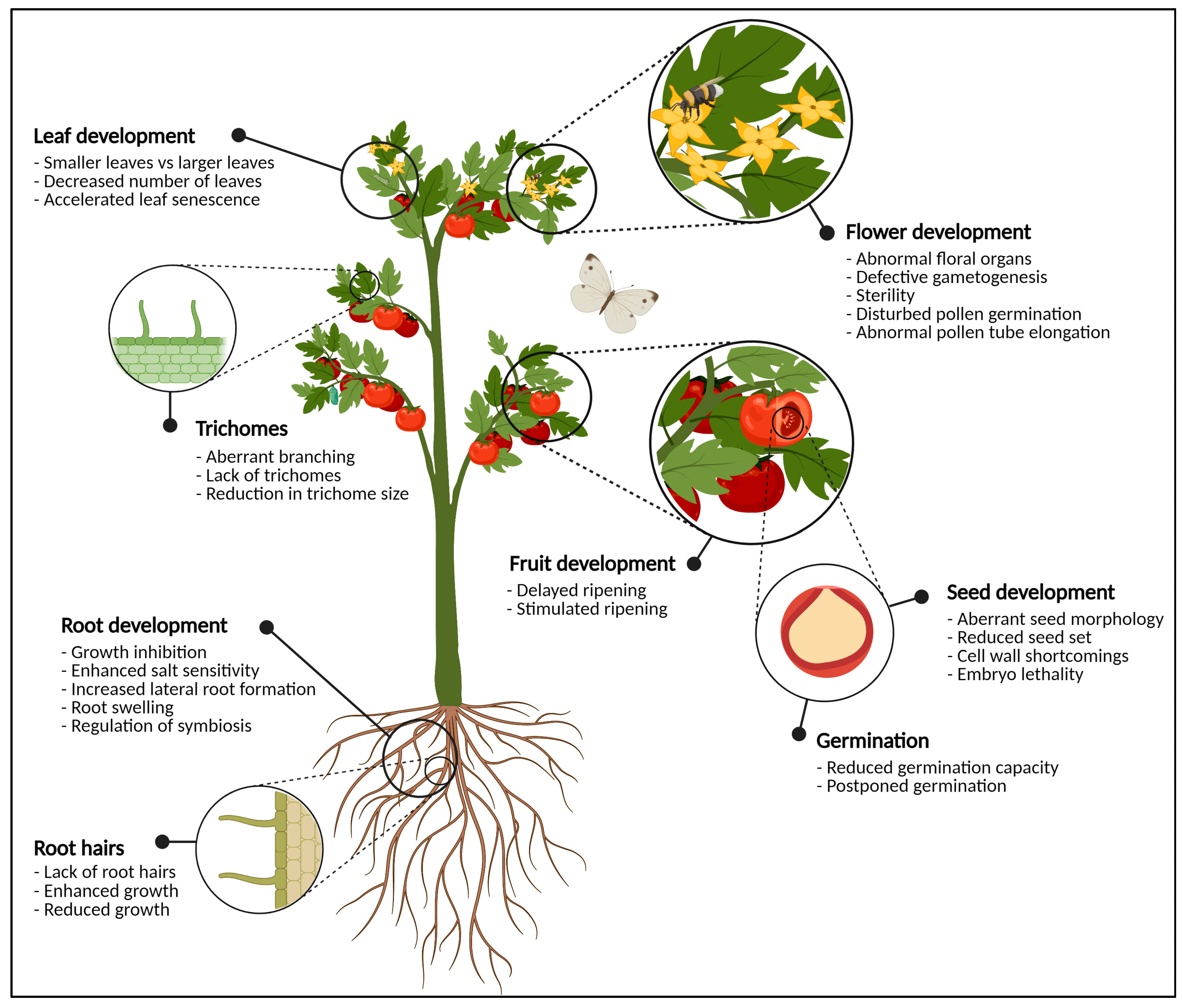
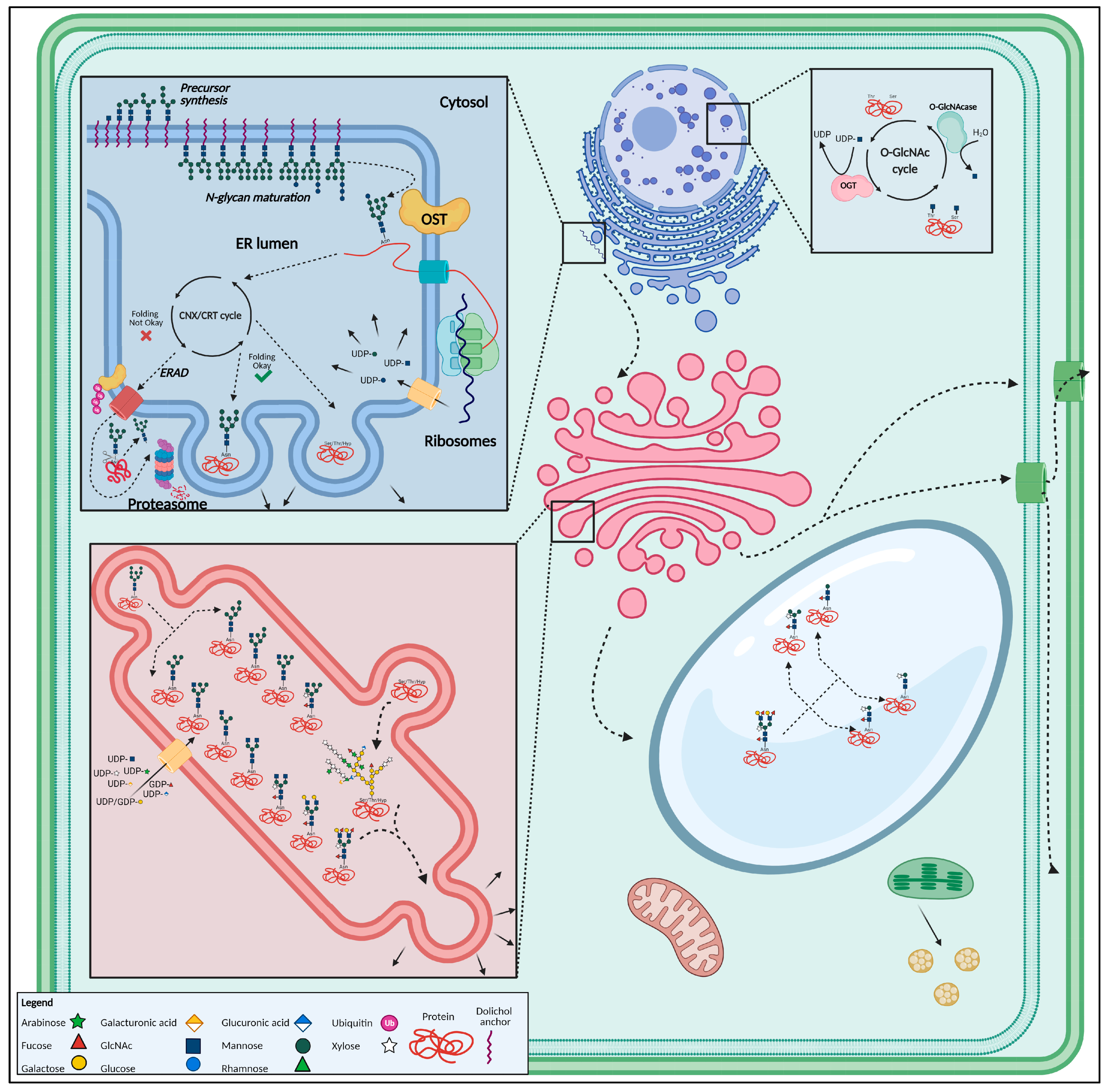
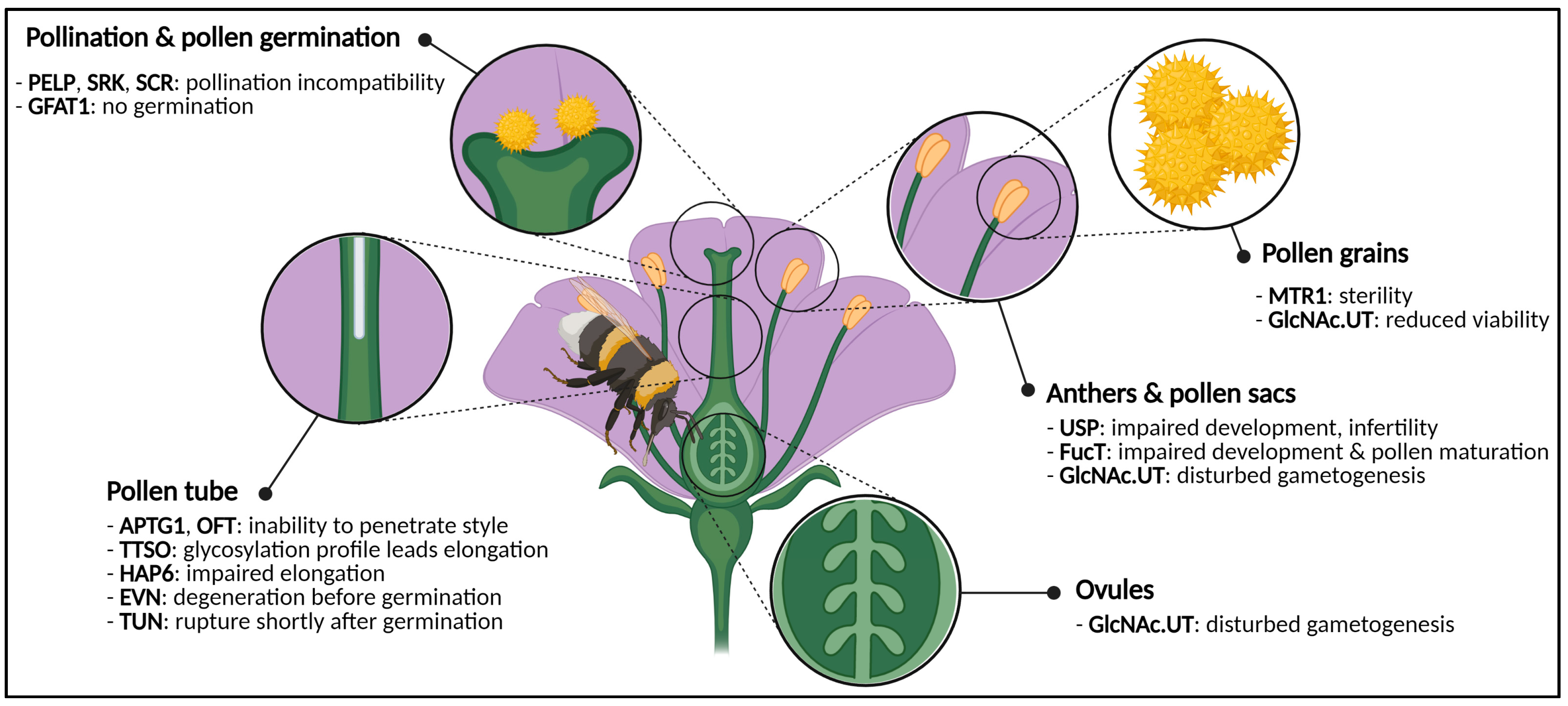
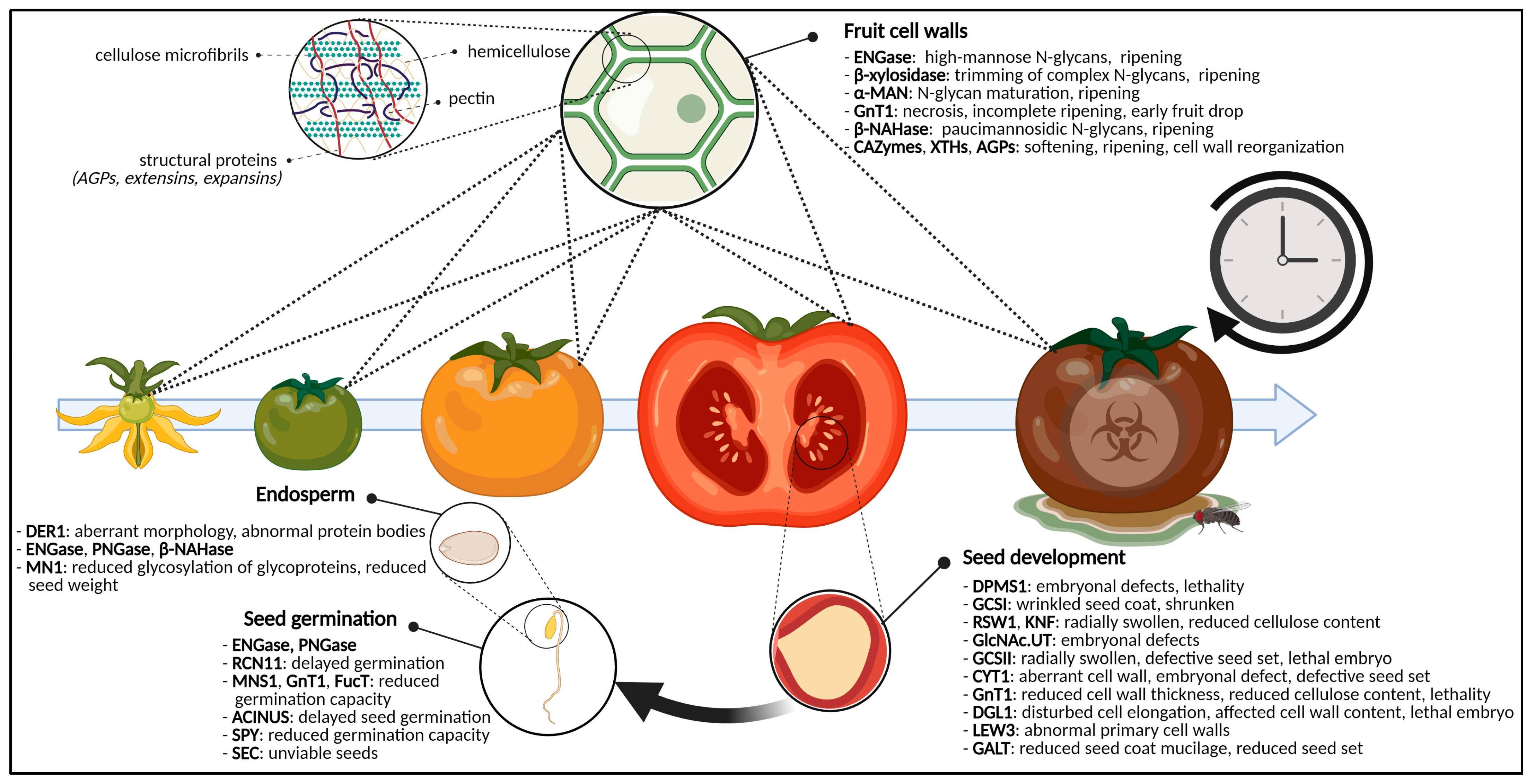
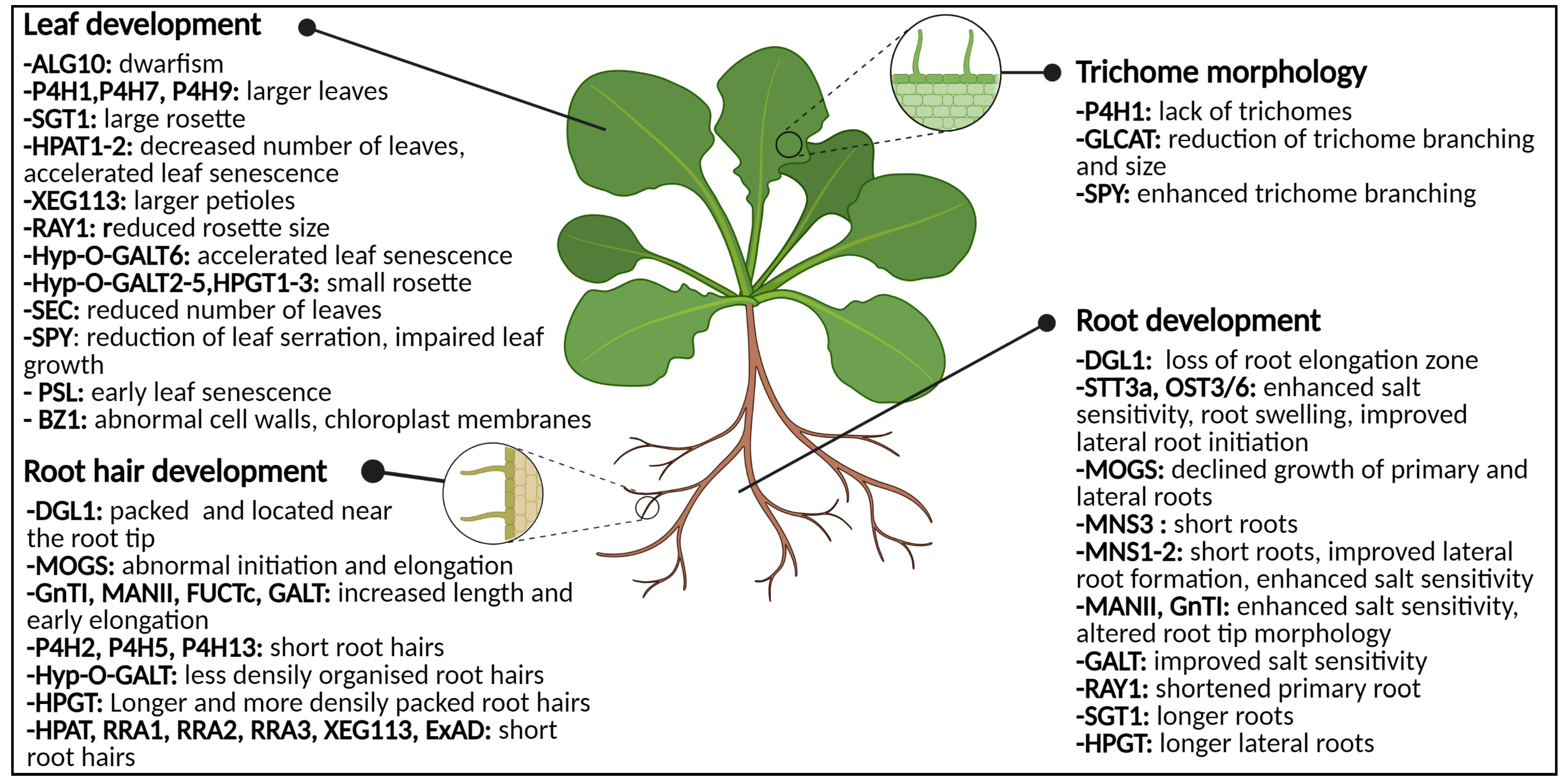
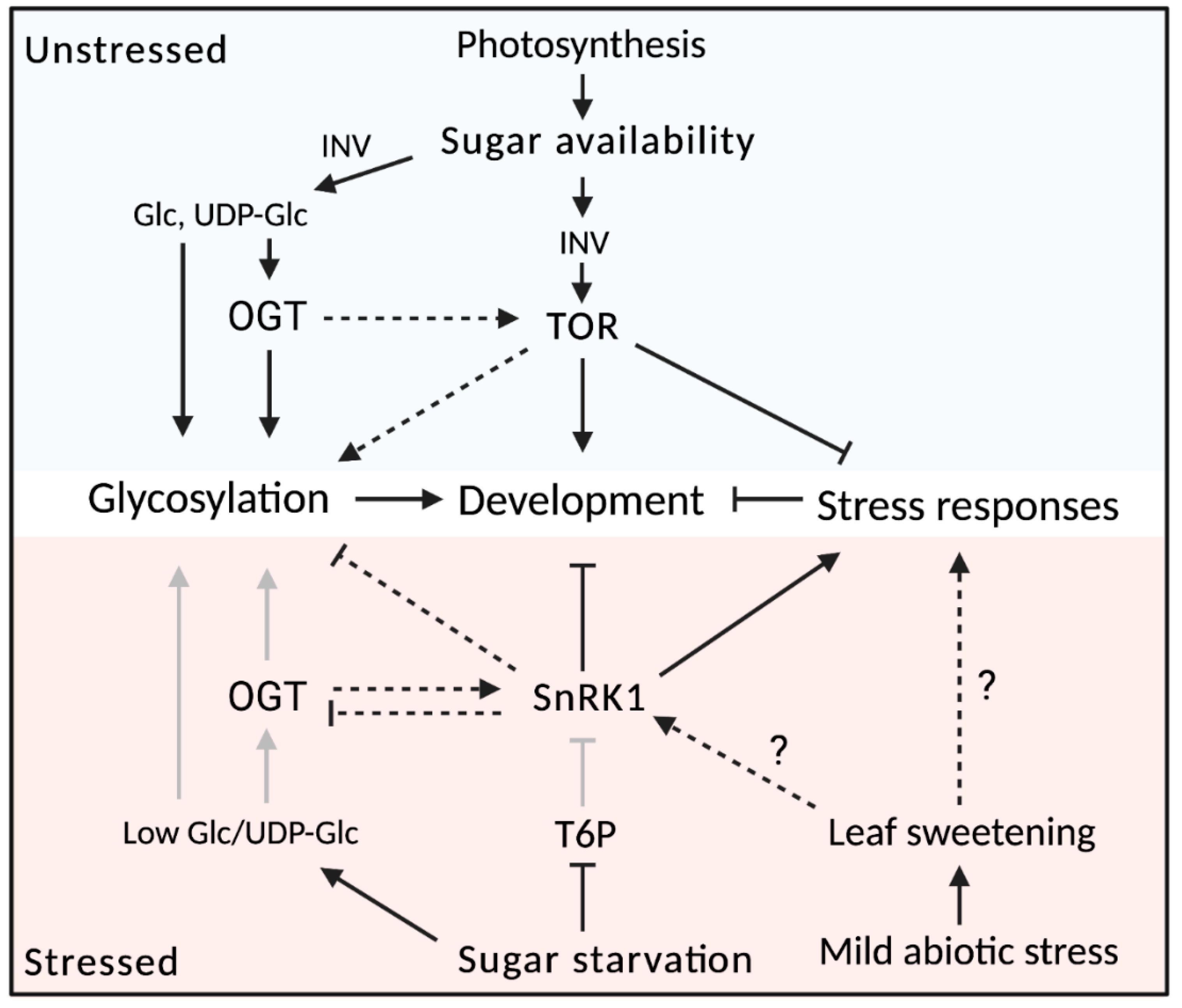
| Enzyme | Activity | Role during Glycosylation | Mutant | Phenotype | Sources |
|---|---|---|---|---|---|
| ALG10 | α-1,2-glucosyltransferase | N-glycan precursor synthesis | alg10 | Dwarfism | [118] |
| ALG11 | α-1,2-mannosyltransferase | lew3 | Aberrant distribution of glycans, reduced cellulose synthesis, abnormal cell walls, reduced fertility | [119] | |
| APTG1 | mannosyltransferase | aptg1 | Pollen tube is unable to penetrate the style; embryo lethality | [120] | |
| CYT1 | GTP:α-D-mannose-1-phosphate guanylyltransferase | cyt1 | Disrupted embryonal development | [121] | |
| DPMS1 | dolichol phosphate mannose synthase 1 | dpms1 | Embryo lethality, wrinkled seed coats | [122] | |
| EVN | dolichol kinase | evn | Pollen degeneration before pollen germination | [123] | |
| GlcNA.UT | GlcNAc-phosphate UDP transferase | glcna.ut1/2 | Aberrant gametogenesis, reduced fertilization, shorter siliques | [124] | |
| DGL1 | Subunit of OST complex | En bloc transfer | dgl1 | Altered cell wall composition, embryo lethality, reduced growth of hypocotyls and roots, dense packed root hairs | [125] |
| HAP6 | hap6 | Shorter pollen tube | [126] | ||
| OST3/6 | ost3/6 | Enhanced salt sensitivity resulting in decreased root growth and root swelling | [127,128,129] | ||
| STT3a | stt3a | ||||
| TUN | UDP-glycosyltransferase | tun | Pollen defects, premature pollen rupture | [123] | |
| GCSI | α-glucosidase I | N-glycan processing in the ER | gcs1 | Embryo lethality, wrinkled seed coats | [130,131] |
| GCSII | α-glucosidase II | gcs2, rsw3 | Embryo lethality, radially swollen roots, modified seed set | [132,133] | |
| MOGS | mannosyloligosaccharide glucosidase | mogs | Reduced growth of primary and lateral roots, abnormal root hair initiation and elongation | [134] | |
| UGGT | UDP-glucose:glycoprotein glucosyltransferase | ERAD | - | No mutant | [135] |
| PNGase | peptide-N4-(N-acetyl-β-D-glucosaminyl) asparagine amidase | - | No mutant | [78,136,137] | |
| ENGase | endo-N-acetyl-β-D-glucosaminidase | De-N-glycosylation | - | No mutant | [78,136,137] |
| α-MAN | α-mannosidase | - | No mutant | [138,139] | |
| β-NAHase | β-D-N-acetylhexosaminidase | - | No mutant | [138,139] | |
| FucT | α-1,3-fucosyltransferase | N-glycan maturation in the Golgi apparatus | fuct | Reduced seed set | [140] |
| FUCTc | α-1,4-fucosyltransferase | fuctc | Increased root hair length, early root hair elongation | [141] | |
| GALT | β-1,3-galactosyl-transferase | galt | Increased root hair length, early root hair elongation | [141] | |
| GnT1 | N-acetylglucosaminyl transferase I | gnt1 | Cell wall shortcomings: reduced thickness and cellulose content; lethality | [140,142] | |
| Necrosis on fruits, incomplete fruit ripening, early fruit drop | [143] | ||||
| Increased root hair length, early root hair elongation, salt sensitivity resulting in altered root tip morphology | [141,144] | ||||
| MANII | α-mannosidase II | manII | Increased root hair length, early root hair elongation, salt sensitivity resulting in altered root tip morphology | [141] | |
| MNS1 | mannosidase I | mns1 | Reduced germination capacity | [140] | |
| MNS2 | mns1, mns2, mns3 | Shorter roots | [145] | ||
| MNS3 | |||||
| XylT | β-1,2-xylosyltransferase | rcn11 | Postponed seed germination | [140,146,147] | |
| GFAT1 | Glutamine:fructose-6-phosphate amidotransferase 1 | Sugar delivery | gfat1 | Aberrant pollen cell wall | [148] |
| USP | UDP-sugar pyrophosphorylase | usp | Infertile pollen, improper pollen sac development | [112] | |
| ROCK1 | UDP-GlcNAc transferase, UDP-GalNAc transferase | rock1 | Cytokinin shortage-like phenotype, increased organ formation rate, increased UPR | [117] | |
| GlcNAc.UT | UDP-GlcNAc transferase | glcna.ut1, glcna.ut2 | Severe developmental shortcomings. Disturbed gametogenesis of male and female gametes. Embryonal problems, shorter siliques with fewer and shrunken seeds in double mutant | [149] | |
| OFT | O-fucosyltransferase | O-glycan modification | oft | Impaired pollen tube penetration ability | [150] |
| SEC | O-GlcNAc transferase | sec | Nonviable seeds, decreased number of leaves | [90,151] | |
| SPY | O-fucosyltransferase | spy | Germination in presence of germination inhibitors | [152] | |
| Enhanced trichome branching, reduced leaf serration size and number of leaf serrations | [153,154] | ||||
| PSL | β-1,6-GlcNAc-transferase | psl | Early leaf senescence, enhanced ethylene production | [155] | |
| BZ1 | UDP-Gal/Glu epimerase | bz1 | Abnormal cell walls, abnormal chloroplast membranes | [156] | |
| P4H1 | Prolyl-4-hydroxylase | Hydroxyproline production | p4h1 | Lack of trichomes, large leaves | [157,158] |
| P4H2 | p4h2 | Short root hairs | [94,159,160] | ||
| P4H5 | p4h5 | ||||
| P4H13 | p4h13 | ||||
| P4H7 | p4h7 | Large leaves | [158] | ||
| P4H9 | p4h9 | ||||
| HPAT1 | Hydroxyproline O-β-arabinosyltransferase | O-arabinosylation of extensins | hpat1 | Decreased number of leaves, accelerated leaf senescence, short root hairs | [161,162] |
| HPAT2 | hpat2 | ||||
| HPAT3 | hpat3 | ||||
| RRA1 | β-1,2-arabinosyltransferase | rra1 | Short root hairs | [95] | |
| RRA2 | rra2 | ||||
| RRA3 | rra3 | ||||
| XEG113 | β-1,2-arabinosyltransferase | xeg113 | Larger petioles, short root hairs | [95,163] | |
| ExAD | α-1,3-arabinosyltransferase | Short root hairs | [164] | ||
| SGT1 | Serine O-α-galactosyltransferase | O-galactosylation of extensins | sgt1 | Larger leaf rosette, longer roots | [91] |
| RAY1 | β-arabinofuranosyltransferase | O-arabinosylation of arabinogalactan proteins | ray1 | Short primary root, reduced leaf rosette size | [165] |
| GALT2 | Hydroxyproline-O-β-galactosyltransferase | O-galactosylation of arabinogalactan proteins | galt2 | Reduced seed coat mucilage, reduced seed set; pleiotropic effects on roots, leaves and root hairs in different single or multiple mutants | [162,166,167,168,169] |
| GALT3 | galt3 | ||||
| GALT4 | galt4 | ||||
| GALT5 | galt5 | ||||
| GALT6 | galt6 | ||||
| HPGT1 | hpgt1 | ||||
| HPGT2 | hpgt2 | ||||
| HPGT3 | hpgt3 | ||||
| GLCAT14a | β-1,6-glucuronosyltransferase | O-glucuronidation of arabinogalactan proteins | glcat14a | Reduced trichome size, branching in double (glcat14a/glcat14b) and triple mutant | [169] |
| GLCAT14b | glcat14b | ||||
| GLCAT14c | glcat14c |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Coninck, T.; Gistelinck, K.; Janse van Rensburg, H.C.; Van den Ende, W.; Van Damme, E.J.M. Sweet Modifications Modulate Plant Development. Biomolecules 2021, 11, 756. https://doi.org/10.3390/biom11050756
De Coninck T, Gistelinck K, Janse van Rensburg HC, Van den Ende W, Van Damme EJM. Sweet Modifications Modulate Plant Development. Biomolecules. 2021; 11(5):756. https://doi.org/10.3390/biom11050756
Chicago/Turabian StyleDe Coninck, Tibo, Koen Gistelinck, Henry C. Janse van Rensburg, Wim Van den Ende, and Els J. M. Van Damme. 2021. "Sweet Modifications Modulate Plant Development" Biomolecules 11, no. 5: 756. https://doi.org/10.3390/biom11050756
APA StyleDe Coninck, T., Gistelinck, K., Janse van Rensburg, H. C., Van den Ende, W., & Van Damme, E. J. M. (2021). Sweet Modifications Modulate Plant Development. Biomolecules, 11(5), 756. https://doi.org/10.3390/biom11050756









