The Pentose Phosphate Pathway in Yeasts–More Than a Poor Cousin of Glycolysis
Abstract
1. Introduction
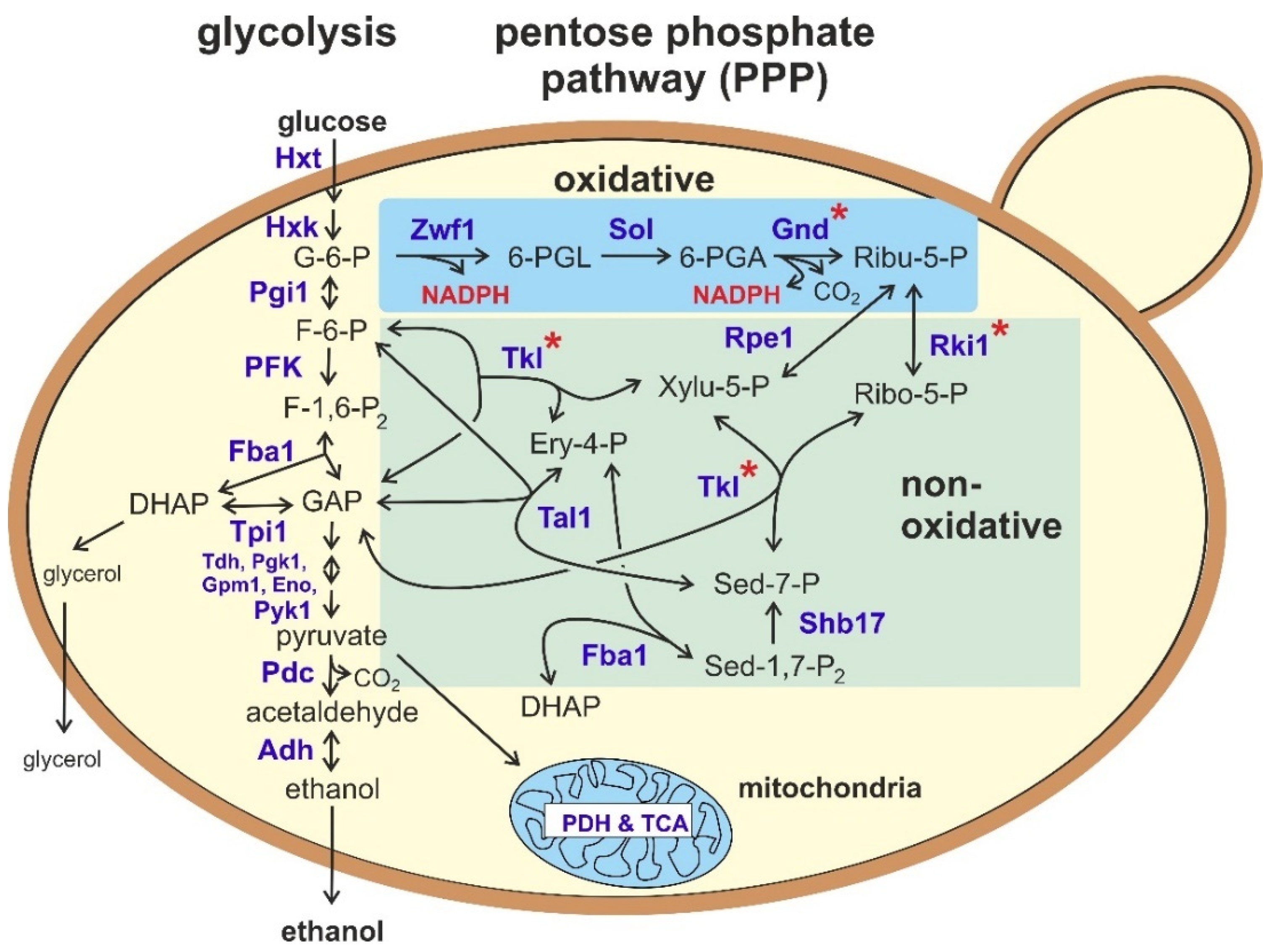
2. Overview on the Reactions of the Pentose Phosphate Pathway (PPP)
2.1. The Oxidative Part of the PPP
2.1.1. Glucose-6-phosphate Dehydrogenase
2.1.2. 6-Phosphoglucono Lactonase
2.1.3. 6-Phosphogluconate Dehydrogenase
2.2. The Non-Oxidative Part of the PPP
2.2.1. Ribulosephosphate Epimerase
2.2.2. Ribosephosphate Ketol Isomerase
2.2.3. Transketolase
2.2.4. Transaldolase
2.2.5. Sedoheptulose-1,7-bisphosphatase
3. Contribution of the PPP to Sugar Degradation
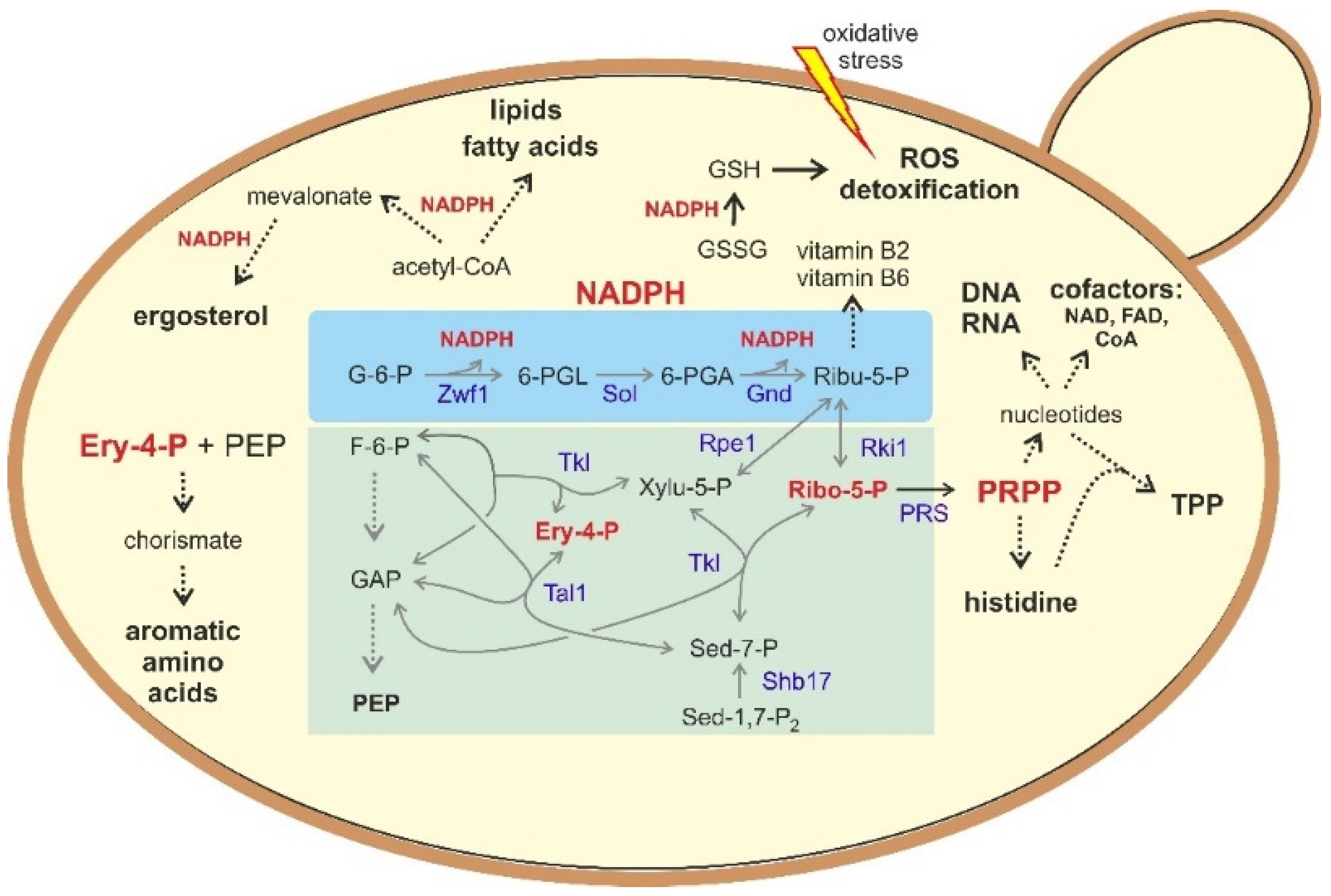
4. Biotechnological Implications
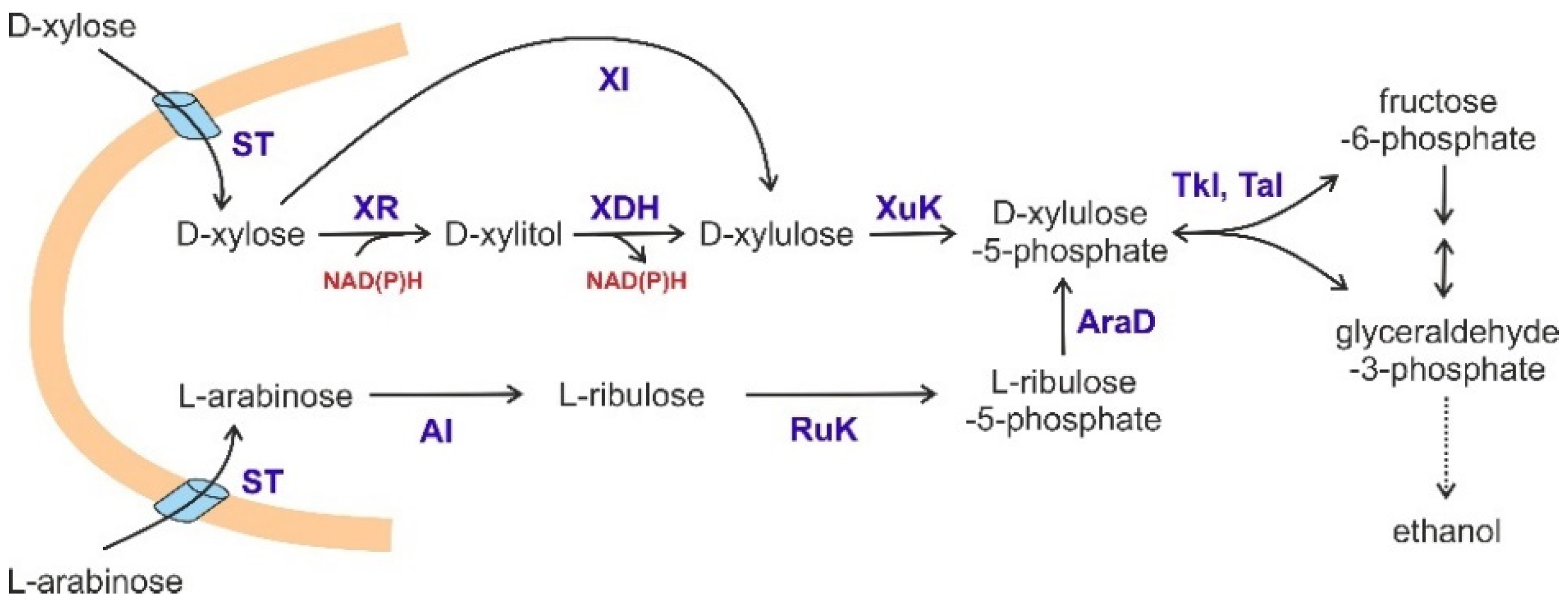
4.1. Fermentation of Pentoses by S. cerevisiae
4.2. The PPP and Synthetic Biology in the Production of Value-Added Products
4.3. Non-Conventional Yeasts
5. Yeasts as Workhorses to Study PPP-Related Diseases
6. Conclusions and Outlook
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Liu, Z.; Zhang, Y.; Nielsen, J. Synthetic Biology of Yeast. Biochemistry 2019, 58, 1511–1520. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, J. Yeast systems biology: Model organism and cell factory. Biotechnol. J. 2019, 14, e1800421. [Google Scholar] [CrossRef] [PubMed]
- Botstein, D.; Fink, G.R. Yeast: An Experimental Organism for 21st Century Biology. Genetics 2011, 189, 695–704. [Google Scholar] [CrossRef] [PubMed]
- Duina, A.; Miller, M.; Keeney, J.B. Budding Yeast for Budding Geneticists: A Primer on the Saccharomyces cerevisiae Model System. Genetics 2014, 197, 33–48. [Google Scholar] [CrossRef]
- Lacerda, M.P.; Oh, E.J.; Eckert, C. The Model System Saccharomyces cerevisiae versus Emerging Non-Model Yeasts for the Production of Biofuels. Life 2020, 10, 299. [Google Scholar] [CrossRef] [PubMed]
- Patra, P.; Das, M.; Kundu, P.; Ghosh, A. Recent advances in systems and synthetic biology approaches for developing novel cell-factories in non-conventional yeasts. Biotechnol. Adv. 2021, 47, 107695. [Google Scholar] [CrossRef]
- Wagner, J.M.; Alper, H.S. Synthetic biology and molecular genetics in non-conventional yeasts: Current tools and future advances. Fungal Genet. Biol. 2016, 89, 126–136. [Google Scholar] [CrossRef]
- Rodicio, R.; Heinisch, J.J. Yeast on the milky way: Genetics, physiology and biotechnology of Kluyveromyces lactis. Yeast 2013, 30, 165–177. [Google Scholar] [CrossRef]
- Ernst, J.F.; Tielker, D. Responses to hypoxia in fungal pathogens. Cell. Microbiol. 2009, 11, 183–190. [Google Scholar] [CrossRef]
- Kumamoto, C.; Gresnigt, M.S.; Hube, B. The gut, the bad and the harmless: Candida albicans as a commensal and opportunistic pathogen in the intestine. Curr. Opin. Microbiol. 2020, 56, 7–15. [Google Scholar] [CrossRef]
- Stincone, A.; Prigione, A.; Cramer, T.; Wamelink, M.M.C.; Campbell, K.; Cheung, E.; Olin-Sandoval, V.; Greuning, N.-M.; Krueger, A.; Alam, M.T.; et al. The return of metabolism: Biochemistry and physiology of the pentose phosphate pathway. Biol. Rev. 2015, 90, 927–963. [Google Scholar] [CrossRef]
- Dashko, S.; Zhou, N.; Compagno, C.; Piškur, J. Why, when, and how did yeast evolve alcoholic fermentation? FEMS Yeast Res. 2014, 14, 826–832. [Google Scholar] [CrossRef] [PubMed]
- Flores, C.-L.; Rodríguez, C.; Petit, T.; Gancedo, C. Carbohydrate and energy-yielding metabolism in non-conventional yeasts. FEMS Microbiol. Rev. 2000, 24, 507–529. [Google Scholar] [CrossRef] [PubMed]
- Caillau, M.; Quick, W.P. New insights into plant transaldolase. Plant J. 2005, 43, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Kern, A.; Tilley, E.; Hunter, I.S.; Legiša, M.; Glieder, A. Engineering primary metabolic pathways of industrial micro-organisms. J. Biotechnol. 2007, 129, 6–29. [Google Scholar] [CrossRef]
- Kruger, N.J.; von Schaewen, A. The oxidative pentose phosphate pathway: Structure and organisation. Curr. Opin. Plant Biol. 2003, 6, 236–246. [Google Scholar] [CrossRef]
- Papagianni, M. Recent advances in engineering the central carbon metabolism of industrially important bacteria. Microb. Cell Factories 2012, 11, 50. [Google Scholar] [CrossRef]
- Piranavan, P.; Bhamra, M.; Perl, A. Metabolic Targets for Treatment of Autoimmune Diseases. Immunometabolism 2020, 2, e200012. [Google Scholar] [CrossRef] [PubMed]
- Rajas, F.; Gautier-Stein, A.; Mithieux, G. Glucose-6 Phosphate, a Central Hub for Liver Carbohydrate Metabolism. Metabolites 2019, 9, 282. [Google Scholar] [CrossRef]
- Heinisch, J. Construction and physiological characterization of mutants disrupted in the phosphofructokinase genes of Saccharomyces cerevisiae. Curr. Genet. 1986, 11, 227–234. [Google Scholar] [CrossRef]
- Alfarouk, K.O.; Ahmed, S.B.M.; Elliott, R.L.; Benoit, A.; Alqahtani, S.S.; Ibrahim, M.E.; Bashir, A.H.H.; AlHoufie, S.T.S.; Elhassan, G.O.; Wales, C.C.; et al. The Pentose Phosphate Pathway Dynamics in Cancer and Its Dependency on Intracellular pH. Metabolites 2020, 10, 285. [Google Scholar] [CrossRef]
- Manganelli, G.; Masullo, U.; Passarelli, S.; Filosa, S. Glucose-6-Phosphate Dehydrogenase Deficiency: Disadvantages and Possible Benefits. Cardiovasc. Hematol. Disord. Drug Targets 2013, 13, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Soderberg, T. Biosynthesis of ribose-5-phosphate and erythrose-4-phosphate in archaea: A phylogenetic analysis of archaeal genomes. Archaea 2005, 1, 347–352. [Google Scholar] [CrossRef] [PubMed]
- Pickl, A.; Schönheit, P. The oxidative pentose phosphate pathway in the haloarchaeon Haloferax volcanii involves a novel type of glucose-6-phosphate dehydrogenase—The archaeal Zwischenferment. FEBS Lett. 2015, 589, 1105–1111. [Google Scholar] [CrossRef]
- Clasquin, M.F.; Melamud, E.; Singer, A.; Gooding, J.R.; Xu, X.; Dong, A.; Cui, H.; Campagna, S.R.; Savchenko, A.; Yakunin, A.F.; et al. Riboneogenesis in Yeast. Cell 2011, 145, 969–980. [Google Scholar] [CrossRef] [PubMed]
- Heinisch, J.J.; Knuesting, J.; Scheibe, R. Investigation of Heterologously Expressed Glucose-6-Phosphate Dehydrogenase Genes in a Yeast zwf1 Deletion. Microorganisms 2020, 8, 546. [Google Scholar] [CrossRef]
- Jacoby, J.J.; Heinisch, J.J. Analysis of a transketolase gene from Kluyveromyces lactis reveals that the yeast enzymes are more related to transketolases of prokaryotic origins than to those of higher eukaryotes. Curr. Genet. 1997, 31, 15–21. [Google Scholar] [CrossRef]
- Juhnke, H.; Krems, B.; Kötter, P.; Entian, K.-D. Mutants that show increased sensitivity to hydrogen peroxide reveal an important role for the pentose phosphate pathway in protection of yeast against oxidative stress. Mol. Genet. Genom. 1996, 252, 456–464. [Google Scholar] [CrossRef] [PubMed]
- Nogae, I.; Johnston, M. Isolation and characterization of the ZWF1 gene of Saccharomyces cerevisiae, encoding glucose-6-phosphate dehydrogenase. Gene 1990, 96, 161–169. [Google Scholar] [CrossRef]
- Thomas, D.; Cherest, H.; Surdin-Kerjan, Y. Identification of the structural gene for glucose-6-phosphate dehydrogenase in yeast. Inactivation leads to a nutritional requirement for organic sulfur. EMBO J. 1991, 10, 547–553. [Google Scholar] [CrossRef]
- Saliola, M.; Scappucci, G.; De Maria, I.; Lodi, T.; Mancini, P.; Falcone, C. Deletion of the Glucose-6-Phosphate Dehydrogenase Gene KlZWF1 Affects both Fermentative and Respiratory Metabolism in Kluyveromyces lactis. Eukaryot. Cell 2006, 6, 19–27. [Google Scholar] [CrossRef]
- Saliola, M.; Tramonti, A.; Lanini, C.; Cialfi, S.; De Biase, D.; Falcone, C. Intracellular NADPH Levels Affect the Oligomeric State of the Glucose 6-Phosphate Dehydrogenase. Eukaryot. Cell 2012, 11, 1503–1511. [Google Scholar] [CrossRef]
- Blank, L.M.; Kuepfer, L.; Sauer, U. Large-scale 13C-flux analysis reveals mechanistic principles of metabolic network robustness to null mutations in yeast. Genome Biol. 2005, 6, R49. [Google Scholar] [CrossRef]
- Stanford, D.R.; Whitney, M.L.; Hurto, R.L.; Eisaman, D.M.; Shen, W.-C.; Hopper, A.K. Division of Labor Among the Yeast Sol Proteins Implicated in tRNA Nuclear Export and Carbohydrate Metabolism. Genetics 2004, 168, 117–127. [Google Scholar] [CrossRef]
- He, W.; Wang, Y.; Liu, W.; Zhou, C.-Z. Crystal structure of Saccharomyces cerevisiae 6-phosphogluconate dehydrogenase Gnd1. BMC Struct. Biol. 2007, 7, 38. [Google Scholar] [CrossRef] [PubMed]
- Lobo, Z.; Maitra, P.K. Pentose phosphate pathway mutants of yeast. Mol. Genet. Genom. 1982, 185, 367–368. [Google Scholar] [CrossRef] [PubMed]
- Miosga, T.; Schaaff-Gerstenschläger, I.; Franken, E.; Zimmermann, F.K. Lysine144 is essential for the catalytic activity of Saccharomyces cerevisiae transaldolase. Yeast 1993, 9, 1241–1249. [Google Scholar] [CrossRef] [PubMed]
- Kondo, H.; Nakamura, Y.; Dong, Y.-X.; Nikawa, J.-I.; Sueda, S. Pyridoxine biosynthesis in yeast: Participation of ribose 5-phosphate ketol-isomerase. Biochem. J. 2004, 379, 65–70. [Google Scholar] [CrossRef]
- Horecker, B.L. The Pentose Phosphate Pathway. J. Biol. Chem. 2002, 277, 47965–47971. [Google Scholar] [CrossRef]
- Lindqvist, Y.; Schneider, G.; Ermler, U.; Sundstrom, M. Three-dimensional structure of transketolase, a thiamine diphosphate dependent enzyme, at 2.5 A resolution. EMBO J. 1992, 11, 2373–2379. [Google Scholar] [CrossRef]
- Schaaff, I.; Hohmann, S.; Zimmermann, F.K. Molecular analysis of the structural gene for yeast transaldolase. Eur. J. Biochem. 1990, 188, 597–603. [Google Scholar] [CrossRef]
- Schaaff-Gerstenschlager, I.; Mannhaupt, G.; Vetter, I.; Zimmermann, F.K.; Feldmann, H. TKL2, a second transketolase gene of Saccharomyces cerevisiae. Cloning, sequence and deletion analysis of the gene. Eur. J. Biochem. 1993, 217, 487–492. [Google Scholar] [CrossRef]
- Schaaff-Gerstenschläger, I.; Zimmermann, F.K. Pentose-phosphate pathway in Saccharomyces cerevisiae: Analysis of deletion mutants for transketolase, transaldolase, and glucose 6-phosphate dehydrogenase. Curr. Genet. 1993, 24, 373–376. [Google Scholar] [CrossRef] [PubMed]
- Jacoby, J.; Hollenberg, C.P.; Heinisch, J.J. Transaldolase mutants in the yeast Kluyveromyces lactis provide evidence that glucose can be metabolized through the pentose phosphate pathway. Mol. Microbiol. 1993, 10, 867–876. [Google Scholar] [CrossRef] [PubMed]
- Nkhoma, E.T.; Poole, C.; Vannappagari, V.; Hall, S.A.; Beutler, E. The global prevalence of glucose-6-phosphate dehydrogenase deficiency: A systematic review and meta-analysis. Blood Cells Mol. Dis. 2009, 42, 267–278. [Google Scholar] [CrossRef] [PubMed]
- Richardson, S.R.; O’Malley, G.F. Glucose-6-Phosphate Dehydrogenase Deficiency; StatPearls: Treasure Island, FL, USA, 2021. [Google Scholar]
- Perli, T.; Wronska, A.K.; Ortiz-Merino, R.A.; Pronk, J.T.; Daran, J. Vitamin requirements and biosynthesis in Saccharomyces cerevisiae. Yeast 2020, 37, 283–304. [Google Scholar] [CrossRef]
- Levy, H.R. Glucose-6-Phosphate Dehydrogenases. Adv. Enzymol. Relat. Areas Mol. Biol. 1979, 48, 97–192. [Google Scholar]
- Lee, J.; Godon, C.; Lagniel, G.; Spector, D.; Garin, J.; Labarre, J.; Toledano, M.B. Yap1 and Skn7 Control Two Specialized Oxidative Stress Response Regulons in Yeast. J. Biol. Chem. 1999, 274, 16040–16046. [Google Scholar] [CrossRef]
- Minard, K.I.; McAlister-Henn, L. Sources of NADPH in Yeast Vary with Carbon Source. J. Biol. Chem. 2005, 280, 39890–39896. [Google Scholar] [CrossRef]
- Campbell, K.; Vowinckel, J.; Keller, M.A.; Ralser, M. Methionine Metabolism Alters Oxidative Stress Resistanceviathe Pentose Phosphate Pathway. Antioxid. Redox Signal. 2016, 24, 543–547. [Google Scholar] [CrossRef]
- Grabowska, D.; Chelstowska, A. The ALD6 Gene Product Is Indispensable for Providing NADPH in Yeast Cells Lacking Glucose-6-phosphate Dehydrogenase Activity. J. Biol. Chem. 2003, 278, 13984–13988. [Google Scholar] [CrossRef]
- Royt, P.W.; MacQuillan, A.M. The Pasteur effect and catabolite repression in an oxidative yeast, Kluyveromyces lactis. Antonie Leeuwenhoek 1979, 45, 241–252. [Google Scholar] [CrossRef] [PubMed]
- Epfeiffer, T.; Emorley, A. An evolutionary perspective on the Crabtree effect. Front. Mol. Biosci. 2014, 1, 17. [Google Scholar] [CrossRef]
- Strijbis, K.; Burg, J.V.D.; Visser, W.F.; Berg, M.V.D.; Distel, B. Alternative splicing directs dual localization of Candida albicans 6-phosphogluconate dehydrogenase to cytosol and peroxisomes. FEMS Yeast Res. 2011, 12, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Burgain, A.; Tebbji, F.; Khemiri, I.; Sellam, A. Metabolic Reprogramming in the Opportunistic Yeast Candida albicans in Response to Hypoxia. mSphere 2020, 5, e00913-19. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Ren, L.; Zeng, S.; Zhang, S.; Xu, D.; Zeng, X.; Li, F. Functional analysis of PGI1 and ZWF1 in thermotolerant yeast Kluyveromyces marxianus. Appl. Microbiol. Biotechnol. 2020, 104, 1–16. [Google Scholar] [CrossRef]
- Tsai, C.S.; Chen, Q. Regulation of D-glucose-6-phosphate dehydrogenase from Schizosaccharomyces pombe. Biochem. Cell Biol. 1998, 76, 645–648. [Google Scholar] [CrossRef]
- Jeffery, J.; Persson, B.; Wood, I.; Bergman, T.; Jeffery, R.; Jörnvall, H. Glucose-6-phosphate dehydrogenase. Structure-function relationships and the Pichia jadinii enzyme structure. Eur. J. Biochem. 1993, 212, 41–49. [Google Scholar] [CrossRef]
- Brown, J.; Sherlock, G.; Myers, C.L.; Burrows, N.M.; Deng, C.; Wu, H.I.; McCann, K.; Troyanskaya, O.G.; Brown, J.M. Global analysis of gene function in yeast by quantitative phenotypic profiling. Mol. Syst. Biol. 2006, 2, 2006-0001. [Google Scholar] [CrossRef]
- Byrne, K.P. The Yeast Gene Order Browser: Combining curated homology and syntenic context reveals gene fate in polyploid species. Genome Res. 2005, 15, 1456–1461. [Google Scholar] [CrossRef]
- Sinha, A.; Maitra, P.K. Induction of specific enzymes of the oxidative pentose phosphate pathway by glucono-lactone in Saccharomyces cerevisiae. J. Gen. Microbiol. 1992, 138, 1865–1873. [Google Scholar] [CrossRef] [PubMed]
- Berdis, A.J.; Cook, P.F. Overall kinetic mechanism of 6-phosphogluconate dehydrogenase from Candida utilis. Biochemistry 1993, 32, 2036–2040. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.S.; Chen, Q. Purification and kinetic characterization of 6-phosphogluconate dehydrogenase from Schizosaccharomyces pombe. Biochem. Cell Biol. 1998, 76, 637–644. [Google Scholar] [CrossRef] [PubMed]
- Miosga, T.; Zimmermann, F.K. Cloning and characterization of the first two genes of the non-oxidative part of the Saccharomyces cerevisiae pentose-phosphate pathway. Curr. Genet. 1996, 30, 404–409. [Google Scholar] [CrossRef] [PubMed]
- Golbik, R.; Meshalkina, L.E.; Sandalova, T.; Tittmann, K.; Fiedler, E.; Neef, H.; König, S.; Kluger, R.; Kochetov, G.A.; Schneider, G.; et al. Effect of coenzyme modification on the structural and catalytic properties of wild-type transketolase and of the variant E418A from Saccharomyces cerevisiae. FEBS J. 2005, 272, 1326–1342. [Google Scholar] [CrossRef] [PubMed]
- Sundström, M.; Lindqvist, Y.; Schneider, G. Three-dimensional structure of apotransketolase flexible loops at the active site enable cofactor binding. FEBS Lett. 1992, 313, 229–231. [Google Scholar] [CrossRef]
- Ng, C.-H.; Tan, S.-X.; Perrone, G.G.; Thorpe, G.W.; Higgins, V.J.; Dawes, I.W. Adaptation to hydrogen peroxide in Saccharomyces cerevisiae: The role of NADPH-generating systems and the SKN7 transcription factor. Free. Radic. Biol. Med. 2008, 44, 1131–1145. [Google Scholar] [CrossRef] [PubMed]
- Matsushika, A.; Goshima, T.; Fujii, T.; Inoue, H.; Sawayama, S.; Yano, S. Characterization of non-oxidative transaldolase and transketolase enzymes in the pentose phosphate pathway with regard to xylose utilization by recombinant Saccharomyces cerevisiae. Enzym. Microb. Technol. 2012, 51, 16–25. [Google Scholar] [CrossRef]
- Villa-García, M.J.; Choi, M.S.; Hinz, F.I.; Gaspar, M.L.; Jesch, S.A.; Henry, S.A. Genome-wide screen for inositol auxotrophy in Saccharomyces cerevisiae implicates lipid metabolism in stress response signaling. Mol. Genet. Genom. 2011, 285, 125–149. [Google Scholar] [CrossRef]
- Sun, S.; Joris, L.; Tsolas, O. Purification and crystallization of transaldolase isozyme I and evidence for different genetic origin of isozymes I and III in Candida utilis. Arch. Biochem. Biophys. 1977, 178, 69–78. [Google Scholar] [CrossRef]
- Zhang, Q.; Bartels, R. Octulose: A forgotten metabolite? J. Exp. Bot. 2017, 68, 5689–5694. [Google Scholar] [CrossRef]
- Koendjbiharie, J.G.; Hon, S.; Pabst, M.; Hooftman, R.; Stevenson, D.M.; Cui, J.; Amador-Noguez, D.; Lynd, L.R.; Olson, D.G.; van Kranenburg, R. The pentose phosphate pathway of cellulolytic clostridia relies on 6-phosphofructokinase instead of transaldolase. J. Biol. Chem. 2020, 295, 1867–1878. [Google Scholar] [CrossRef]
- Rozova, O.N.; Khmelenina, V.N.; Trotsenko, Y.A. Characterization of recombinant PPi-dependent 6-phosphofructokinases from Methylosinus trichosporium OB3b and Methylobacterium nodulans ORS 2060. Biochemistry 2012, 77, 288–295. [Google Scholar] [CrossRef]
- Breitenbach-Schmitt, I.; Schmitt, H.D.; Heinisch, J.; Zimmermann, F.K. Genetic and physiological evidence for the existence of a second glycolytic pathway in yeast parallel to the phosphofructokinase-aldolase reaction sequence. Mol. Genet. Genom. 1984, 195, 536–540. [Google Scholar] [CrossRef]
- Lagunas, R.; Gancedo, J.M. Reduced Pyridine-Nucleotides Balance in Glucose-Growing Saccharomyces cerevisiae. Eur. J. Biochem. 1973, 37, 90–94. [Google Scholar] [CrossRef] [PubMed]
- Ciriacy, M.; Breitenbach, I. Physiological Effects of Seven Different Blocks in Glycolysis in Saccharomyces cerevisiae. J. Bacteriol. 1979, 139, 152–160. [Google Scholar] [CrossRef] [PubMed]
- Goffrini, P.; Wésolowski-Louvel, M.; Ferrero, I. A phosphoglucose isomerase gene is involved in the Rag phenotype of the yeast Kluyveromyces lactis. Mol. Genet. Genom. 1991, 228, 401–409. [Google Scholar] [CrossRef] [PubMed]
- Heinisch, J.; Kirchrath, L.; Liesen, T.; Vogelsang, K.; Hollenberg, C.P. Molecular genetics of phosphofructokinase in the yeast Kluyveromyces lactis. Mol. Microbiol. 1993, 8, 559–570. [Google Scholar] [CrossRef]
- Boles, E.; Lehnert, W.; Zimmermann, F.K. The role of the NAD-dependent glutamate dehydrogenase in restoring growth on glucose of a Saccharomyces cerevisiae phosphoglucose isomerase mutant. JBIC J. Biol. Inorg. Chem. 1993, 217, 469–477. [Google Scholar] [CrossRef]
- Overkamp, K.M.; Bakker, B.M.; Steensma, H.Y.; Van Dijken, J.P.; Pronk, J.T. Two mechanisms for oxidation of cytosolic NADPH by Kluyveromyces lactis mitochondria. Yeast 2002, 19, 813–824. [Google Scholar] [CrossRef]
- Tarrío, N.; Prado, S.D.; Cerdán, M.E.; González-Siso, M.-I. The nuclear genes encoding the internal (KlNDI1) and external (KlNDE1) alternative NAD(P)H:ubiquinone oxidoreductases of mitochondria from Kluyveromyces lactis. Biochim. Biophys. Acta (BBA) Bioenerg. 2005, 1707, 199–210. [Google Scholar] [CrossRef]
- Gombert, A.K.; Dos Santos, M.M.; Christensen, B.; Nielsen, J. Network Identification and Flux Quantification in the Central Metabolism of Saccharomyces cerevisiae under Different Conditions of Glucose Repression. J. Bacteriol. 2001, 183, 1441–1451. [Google Scholar] [CrossRef]
- Zhang, J.; Martinez-Gomez, K.; Heinzle, E.; Wahl, S.A. Metabolic switches from quiescence to growth in synchronized Saccharomyces cerevisiae. Metabolomics 2019, 15, 1–13. [Google Scholar] [CrossRef]
- Kleijn, R.J.; Van Winden, W.A.; Van Gulik, W.M.; Heijnen, J.J. Revisiting the 13C-label distribution of the non-oxidative branch of the pentose phosphate pathway based upon kinetic and genetic evidence. FEBS J. 2005, 272, 4970–4982. [Google Scholar] [CrossRef] [PubMed]
- Blank, L.M.; Lehmbeck, F.; Sauer, U. Metabolic-flux and network analysis in fourteen hemiascomycetous yeasts. FEMS Yeast Res. 2005, 5, 545–558. [Google Scholar] [CrossRef]
- Krüger, A.; Grüning, N.-M.; Wamelink, M.M.; Kerick, M.; Kirpy, A.; Parkhomchuk, D.; Bluemlein, K.; Schweiger, M.-R.; Soldatov, A.; Lehrach, H.; et al. The Pentose Phosphate Pathway Is a Metabolic Redox Sensor and Regulates Transcription During the Antioxidant Response. Antioxid. Redox Signal. 2011, 15, 311–324. [Google Scholar] [CrossRef] [PubMed]
- Ralser, M.; Wamelink, M.M.; Kowald, A.; Gerisch, B.; Heeren, G.; Struys, E.; Klipp, E.; Jakobs, C.; Breitenbach, M.; Lehrach, H.; et al. Dynamic rerouting of the carbohydrate flux is key to counteracting oxidative stress. J. Biol. 2007, 6, 10. [Google Scholar] [CrossRef] [PubMed]
- Dick, T.P.; Ralser, M. Metabolic Remodeling in Times of Stress: Who Shoots Faster than His Shadow? Mol. Cell 2015, 59, 519–521. [Google Scholar] [CrossRef] [PubMed]
- Kuehne, A.; Emmert, H.; Soehle, J.; Winnefeld, M.; Fischer, F.; Wenck, H.; Gallinat, S.; Terstegen, L.; Lucius, R.; Hildebrand, J.; et al. Acute Activation of Oxidative Pentose Phosphate Pathway as First-Line Response to Oxidative Stress in Human Skin Cells. Mol. Cell 2015, 59, 359–371. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, A.; Lin, M. Regulation of glucose-6-phosphate dehydrogenase activity in red blood cells from hemolytic and non-hemolytic variant subjects. Blood 1973, 41, 877–891. [Google Scholar] [CrossRef] [PubMed]
- Røst, L.M.; Thorfinnsdottir, L.B.; Kumar, K.; Fuchino, K.; Langørgen, I.E.; Bartosova, Z.; Kristiansen, K.A.; Bruheim, P. Absolute Quantification of the Central Carbon Metabolome in Eight Commonly Applied Prokaryotic and Eukaryotic Model Systems. Metabolites 2020, 10, 74. [Google Scholar] [CrossRef]
- Bergman, A.; Vitay, D.; Hellgren, J.; Chen, Y.; Nielsen, J.; Siewers, V. Effects of overexpression of STB5 in Saccharomyces cerevisiae on fatty acid biosynthesis, physiology and transcriptome. FEMS Yeast Res. 2019, 19, 3. [Google Scholar] [CrossRef]
- Chechik, G.; Oh, E.; Rando, O.; Weissman, J.; Regev, A.; Koller, D. Activity motifs reveal principles of timing in transcriptional control of the yeast metabolic network. Nat. Biotechnol. 2008, 26, 1251–1259. [Google Scholar] [CrossRef]
- LaRochelle, M.; Drouin, S.; Robert, F.; Turcotte, B. Oxidative Stress-Activated Zinc Cluster Protein Stb5 Has Dual Activator/Repressor Functions Required for Pentose Phosphate Pathway Regulation and NADPH Production. Mol. Cell. Biol. 2006, 26, 6690–6701. [Google Scholar] [CrossRef] [PubMed]
- Barnett, J.A. A history of research on yeasts 5: The fermentation pathway. Yeast 2003, 20, 509–543. [Google Scholar] [CrossRef] [PubMed]
- Nevoigt, E. Progress in Metabolic Engineering of Saccharomyces cerevisiae. Microbiol. Mol. Biol. Rev. 2008, 72, 379–412. [Google Scholar] [CrossRef] [PubMed]
- Scholey, D.; Burton, E.; Williams, P. The bio refinery; producing feed and fuel from grain. Food Chem. 2016, 197, 937–942. [Google Scholar] [CrossRef][Green Version]
- Jansen, M.L.A.; Bracher, J.M.; Papapetridis, I.; Verhoeven, M.D.; De Bruijn, H.; De Waal, P.P.; Van Maris, A.J.A.; Klaassen, P.; Pronk, J.T. Saccharomyces cerevisiae strains for second-generation ethanol production: From academic exploration to industrial implementation. FEMS Yeast Res. 2017, 17, fox044. [Google Scholar] [CrossRef]
- Cunha, J.T.; Soares, P.O.; Baptista, S.L.; Costa, C.E.; Domingues, L. Engineered Saccharomyces cerevisiae for lignocellulosic valorization: A review and perspectives on bioethanol production. Bioengineered 2020, 11, 883–903. [Google Scholar] [CrossRef] [PubMed]
- Amore, R.; Kötter, P.; Küster, C.; Ciriacy, M.; Hollenberg, C.P. Cloning and expression in Saccharomyces cerevisiae of the NAD(P)H-dependent xylose reductase-encoding gene (XYL1) from the xylose-assimilating yeast Pichia stipitis. Gene 1991, 109, 89–97. [Google Scholar] [CrossRef]
- Kötter, P.; Amore, R.; Hollenberg, C.P.; Ciriacy, M. Isolation and characterization of the Pichia stipitis xylitol dehydrogenase gene, XYL2, and construction of a xylose-utilizing Saccharomyces cerevisiae transformant. Curr. Genet. 1990, 18, 493–500. [Google Scholar] [CrossRef]
- Runquist, D.; Hahn-Hagerdal, B.; Bettiga, M.; Hahn-Hägerdal, B. Increased Ethanol Productivity in Xylose-Utilizing Saccharomyces cerevisiae via a Randomly Mutagenized Xylose Reductase. Appl. Environ. Microbiol. 2010, 76, 7796–7802. [Google Scholar] [CrossRef] [PubMed]
- Brat, D.; Boles, E.; Wiedemann, B. Functional Expression of a Bacterial Xylose Isomerase in Saccharomyces cerevisiae. Appl. Environ. Microbiol. 2009, 75, 2304–2311. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, Y.; Sahara, T.; Ohgiya, S.; Kamagata, Y.; Fujimori, K.E. Systematic optimization of gene expression of pentose phosphate pathway enhances ethanol production from a glucose/xylose mixed medium in a recombinant Saccharomyces cerevisiae. AMB Express 2018, 8, 1–11. [Google Scholar] [CrossRef]
- Kuyper, M.; Hartog, M.M.; Toirkens, M.J.; Almering, M.J.; Winkler, A.A.; Van Dijken, J.P.; Pronk, J.T. Metabolic engineering of a xylose-isomerase-expressing Saccharomyces cerevisiae strain for rapid anaerobic xylose fermentation. FEMS Yeast Res. 2005, 5, 399–409. [Google Scholar] [CrossRef] [PubMed]
- Qi, X.; Zha, J.; Liu, G.-G.; Zhang, W.; Li, B.-Z.; Yuan, Y.-J. Heterologous xylose isomerase pathway and evolutionary engineering improve xylose utilization in Saccharomyces cerevisiae. Front. Microbiol. 2015, 6, 1165. [Google Scholar] [CrossRef]
- Zhou, H.; Cheng, J.-S.; Wang, B.L.; Fink, G.R.; Stephanopoulos, G. Xylose isomerase overexpression along with engineering of the pentose phosphate pathway and evolutionary engineering enable rapid xylose utilization and ethanol production by Saccharomyces cerevisiae. Metab. Eng. 2012, 14, 611–622. [Google Scholar] [CrossRef]
- Tran, P.H.N.; Ko, J.K.; Gong, G.; Um, Y.; Lee, S.-M. Improved simultaneous co-fermentation of glucose and xylose by Saccharomyces cerevisiae for efficient lignocellulosic biorefinery. Biotechnol. Biofuels 2020, 13, 12–14. [Google Scholar] [CrossRef]
- Subtil, T.; Boles, E. Improving L-arabinose utilization of pentose fermenting Saccharomyces cerevisiae cells by heterologous expression of L-arabinose transporting sugar transporters. Biotechnol. Biofuels 2011, 4, 38. [Google Scholar] [CrossRef]
- Nijland, J.G.; Driessen, A.J.M. Engineering of Pentose Transport in Saccharomyces cerevisiae for Biotechnological Applications. Front. Bioeng. Biotechnol. 2020, 7, 464. [Google Scholar] [CrossRef] [PubMed]
- Young, E.M.; Poucher, A.; Comer, A.; Bailey, A.; Alper, H. Functional Survey for Heterologous Sugar Transport Proteins, Using Saccharomyces cerevisiae as a Host. Appl. Environ. Microbiol. 2011, 77, 3311–3319. [Google Scholar] [CrossRef] [PubMed]
- Fletcher, E.; Gao, K.; Mercurio, K.; Ali, M.; Baetz, K. Yeast chemogenomic screen identifies distinct metabolic pathways required to tolerate exposure to phenolic fermentation inhibitors ferulic acid, 4-hydroxybenzoic acid and coniferyl aldehyde. Metab. Eng. 2019, 52, 98–109. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Chen, Y.; Nielsen, J. Harnessing xylose pathways for biofuels production. Curr. Opin. Biotechnol. 2019, 57, 56–65. [Google Scholar] [CrossRef]
- Marella, E.R.; Holkenbrink, C.; Siewers, V.; Borodina, I. Engineering microbial fatty acid metabolism for biofuels and biochemicals. Curr. Opin. Biotechnol. 2018, 50, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Pfleger, B.F.; Gossing, M.; Nielsen, J. Metabolic engineering strategies for microbial synthesis of oleochemicals. Metab. Eng. 2015, 29, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Misawa, N. Pathway engineering for functional isoprenoids. Curr. Opin. Biotechnol. 2011, 22, 627–633. [Google Scholar] [CrossRef]
- Paddon, C.J.; Keasling, J.D. Semi-synthetic artemisinin: A model for the use of synthetic biology in pharmaceutical development. Nat. Rev. Microbiol. 2014, 12, 355–367. [Google Scholar] [CrossRef]
- Kwak, S.; Yun, E.J.; Lane, S.; Oh, E.J.; Kim, K.H.; Jin, Y. Redirection of the Glycolytic Flux Enhances Isoprenoid Production in Saccharomyces cerevisiae. Biotechnol. J. 2019, 15, e1900173. [Google Scholar] [CrossRef]
- Reshamwala, S.M.S.; Lali, A.M. Exploiting the NADPH pool for xylitol production using recombinant Saccharomyces cerevisiae. Biotechnol. Prog. 2020, 36, e2972. [Google Scholar] [CrossRef]
- Park, S.-H.; Lee, K.; Jang, J.W.; Hahn, J.-S. Metabolic Engineering of Saccharomyces cerevisiae for Production of Shinorine, a Sunscreen Material, from Xylose. ACS Synth. Biol. 2019, 8, 346–357. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Cozad, M.A.; Holland, D.A.; Zhang, Y.; Luesch, H.; Ding, Y. Photosynthetic Production of Sunscreen Shinorine Using an Engineered Cyanobacterium. ACS Synth. Biol. 2018, 7, 664–671. [Google Scholar] [CrossRef]
- Curran, K.A.; Leavitt, J.M.; Karim, A.S.; Alper, H.S. Metabolic engineering of muconic acid production in Saccharomyces cerevisiae. Metab. Eng. 2013, 15, 55–66. [Google Scholar] [CrossRef] [PubMed]
- Hassing, E.-J.; de Groot, P.A.; Marquenie, V.R.; Pronk, J.T.; Daran, J.-M.G. Connecting central carbon and aromatic amino acid metabolisms to improve de novo 2-phenylethanol production in Saccharomyces cerevisiae. Metab. Eng. 2019, 56, 165–180. [Google Scholar] [CrossRef]
- Suástegui, M.; Ng, C.Y.; Chowdhury, A.; Sun, W.; Cao, M.; House, E.; Maranas, C.D.; Shao, Z. Multilevel engineering of the upstream module of aromatic amino acid biosynthesis in Saccharomyces cerevisiae for high production of polymer and drug precursors. Metab. Eng. 2017, 42, 134–144. [Google Scholar] [CrossRef]
- Nakagawa, Y.; Kasumi, T.; Ogihara, J.; Tamura, M.; Arai, T.; Tomishige, K. Erythritol: Another C4 Platform Chemical in Biomass Refinery. ACS Omega 2020, 5, 2520–2530. [Google Scholar] [CrossRef] [PubMed]
- Jeffries, T.W.; Van Vleet, J.R.H. Pichia stipitis genomics, transcriptomics, and gene clusters. FEMS Yeast Res. 2009, 9, 793–807. [Google Scholar] [CrossRef] [PubMed]
- Ruchala, J.; Sibirny, A. Pentose metabolism and conversion to biofuels and high-value chemicals in yeasts. FEMS Microbiol. Rev. 2020, 10, fuaa069. [Google Scholar] [CrossRef]
- Gasser, B.; Prielhofer, R.; Marx, H.; Maurer, M.; Nocon, J.; Steiger, M.; Puxbaum, V.; Sauer, M.; Mattanovich, D. Pichia pastoris: Protein production host and model organism for biomedical research. Futur. Microbiol. 2013, 8, 191–208. [Google Scholar] [CrossRef] [PubMed]
- Nocon, J.; Steiger, M.; Mairinger, T.; Hohlweg, J.; Rußmayer, H.; Hann, S.; Gasser, B.; Mattanovich, D. Increasing pentose phosphate pathway flux enhances recombinant protein production in Pichia pastoris. Appl. Microbiol. Biotechnol. 2016, 100, 5955–5963. [Google Scholar] [CrossRef] [PubMed]
- Nurcholis, M.; Lertwattanasakul, N.; Rodrussamee, N.; Kosaka, T.; Murata, M.; Yamada, M. Integration of comprehensive data and biotechnological tools for industrial applications of Kluyveromyces marxianus. Appl. Microbiol. Biotechnol. 2019, 104, 475–488. [Google Scholar] [CrossRef]
- Ortiz-Merino, R.A.; Varela, J.A.; Coughlan, A.Y.; Hoshida, H.; Da Silveira, W.B.; Wilde, C.; Kuijpers, N.G.A.; Geertman, J.-M.; Wolfe, K.H.; Morrissey, J.P. Ploidy Variation in Kluyveromyces marxianus Separates Dairy and Non-dairy Isolates. Front. Genet. 2018, 9, 94. [Google Scholar] [CrossRef]
- Zhang, B.; Zhu, Y.; Zhang, J.; Wang, D.; Sun, L.; Hong, J. Engineered Kluyveromyces marxianus for pyruvate production at elevated temperature with simultaneous consumption of xylose and glucose. Bioresour. Technol. 2017, 224, 553–562. [Google Scholar] [CrossRef] [PubMed]
- Miller, K.K.; Alper, H.S. Yarrowia lipolytica: More than an oleaginous workhorse. Appl. Microbiol. Biotechnol. 2019, 103, 9251–9262. [Google Scholar] [CrossRef]
- Cappellini, M.D.; Fiorelli, G. Glucose-6-phosphate dehydrogenase deficiency. Lancet 2008, 371, 64–74. [Google Scholar] [CrossRef]
- Ge, T.; Yang, J.; Zhou, S.; Wang, Y.; Li, Y.; Tong, X. The Role of the Pentose Phosphate Pathway in Diabetes and Cancer. Front. Endocrinol. 2020, 11, 365. [Google Scholar] [CrossRef] [PubMed]
- Hayes, J.D.; Dinkova-Kostova, A.T.; Tew, K.D. Oxidative Stress in Cancer. Cancer Cell 2020, 38, 167–197. [Google Scholar] [CrossRef] [PubMed]
- Javed, M.; Ahmad, I.; Javed, H.; Naseem, S. d-ribose and pathogenesis of Alzheimer’s disease. Mol. Biol. Rep. 2020, 47, 2289–2299. [Google Scholar] [CrossRef]
- Tang, B.L. Neuroprotection by glucose-6-phosphate dehydrogenase and the pentose phosphate pathway. J. Cell. Biochem. 2019, 120, 14285–14295. [Google Scholar] [CrossRef]
- Wamelink, M.M.C.; Struys, E.A.; Jakobs, C. The biochemistry, metabolism and inherited defects of the pentose phosphate pathway: A review. J. Inherit. Metab. Dis. 2008, 31, 703–717. [Google Scholar] [CrossRef] [PubMed]
- Cha, Y.J.; Jung, W.H.; Koo, J.S. Differential Site-Based Expression of Pentose Phosphate Pathway-Related Proteins among Breast Cancer Metastases. Dis. Markers 2017, 2017, 1–10. [Google Scholar] [CrossRef]
- Kaur, P.; Wamelink, M.M.; Van Der Knaap, M.S.; Girisha, K.M.; Shukla, A. Confirmation of a Rare Genetic Leukoencephalopathy due to a Novel Bi-allelic Variant in RPIA. Eur. J. Med. Genet. 2019, 62, 103708. [Google Scholar] [CrossRef]
- Wamelink, M.M.C.; Grüning, N.-M.; Jansen, E.E.W.; Bluemlein, K.; Lehrach, H.; Jakobs, C.; Ralser, M. The difference between rare and exceptionally rare: Molecular characterization of ribose 5-phosphate isomerase deficiency. J. Mol. Med. 2010, 88, 931–939. [Google Scholar] [CrossRef]
- Boyle, L.; Wamelink, M.M.; Salomons, G.S.; Roos, B.; Pop, A.; Dauber, A.; Hwa, V.; Andrew, M.; Douglas, J.; Feingold, M.; et al. Mutations in TKT are the cause of a syndrome including short stature, developmental delay, and congenital heart defects. Am. J. Hum. Genet. 2016, 98, 1235–1242. [Google Scholar] [CrossRef] [PubMed]
- Peake, R.; Godber, I.; Maguire, D. The effect of magnesium administration on erythrocyte transketolase activity in alcoholic patients treated with thiamine. Scott. Med. J. 2013, 58, 139–142. [Google Scholar] [CrossRef] [PubMed]
- Schenk, G.; Duggleby, R.G.; Nixon, P.F. Properties and functions of the thiamin diphosphate dependent enzyme transketolase. Int. J. Biochem. Cell Biol. 1998, 30, 1297–1318. [Google Scholar] [CrossRef]
- Lafcı, N.G.; Colak, F.K.; Sahin, G.; Sakar, M.; Çetinkaya, S.; Savas-Erdeve, S. Hypergonadotrophic hypogonadism in a patient with transaldolase deficiency: Novel mutation in the pentose phosphate pathway. Hormones 2020, 1–5. [Google Scholar] [CrossRef]
- Oaks, Z.; Jimah, J.; Grossman, C.C.; Beckford, M.; Kelly, R.; Banerjee, S.; Niland, B.; Miklossy, G.; Kuloglu, Z.; Kansu, A.; et al. Transaldolase haploinsufficiency in subjects with acetaminophen-induced liver failure. J. Inherit. Metab. Dis. 2020, 43, 496–506. [Google Scholar] [CrossRef] [PubMed]
- Samland, A.K.; Sprenger, G.A. Transaldolase: From biochemistry to human disease. Int. J. Biochem. Cell Biol. 2009, 41, 1482–1494. [Google Scholar] [CrossRef]
- Williams, M.; Valayannopoulos, V.; Altassan, R.; Chung, W.K.; Heijboer, A.C.; Keng, W.T.; Lapatto, R.; McClean, P.; Mulder, M.F.; Tylki-Szymańska, A.; et al. Clinical, biochemical, and molecular overview of transaldolase deficiency and evaluation of the endocrine function: Update of 34 patients. J. Inherit. Metab. Dis. 2019, 42, 147–158. [Google Scholar] [CrossRef] [PubMed]
- Patra, K.C.; Hay, N. The pentose phosphate pathway and cancer. Trends Biochem. Sci. 2014, 39, 347–354. [Google Scholar] [CrossRef]
- Tu, D.; Gao, Y.; Yang, R.; Guan, T.; Hong, J.-S.; Gao, H.-M. The pentose phosphate pathway regulates chronic neuroinflammation and dopaminergic neurodegeneration. J. Neuroinflammation 2019, 16, 1–17. [Google Scholar] [CrossRef]
- Alamoudi, A.A. Why do cancer cells break from host circadian rhythm? Insights from unicellular organisms. BioEssays 2021, 43, 2000205. [Google Scholar] [CrossRef] [PubMed]
- Rey, G.; Valekunja, U.K.; Feeney, K.A.; Wulund, L.; Milev, N.B.; Stangherlin, A.; Ansel-Bollepalli, L.; Velagapudi, V.; O’Neill, J.S.; Reddy, A.B. The Pentose Phosphate Pathway Regulates the Circadian Clock. Cell Metab. 2016, 24, 462–473. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.-C.; Wu, Y.-H.; Liu, H.-Y.; Stern, A.; Chiu, D.T.-Y. What has passed is prolog: New cellular and physiological roles of G6PD. Free. Radic. Res. 2016, 50, 1047–1064. [Google Scholar] [CrossRef]
- Aziz, H.; Mohiuddin, S.S. Biochemistry, Hexose Monophosphate Pathway; StatPearls: Treasure Island, FL, USA, 2021. [Google Scholar]
- Tasdogan, A.; Faubert, B.; Ramesh, V.; Ubellacker, J.M.; Shen, B.; Solmonson, A.; Murphy, M.M.; Gu, Z.; Gu, W.; Martin, M.; et al. Metabolic heterogeneity confers differences in melanoma metastatic potential. Nature 2020, 577, 115–120. [Google Scholar] [CrossRef]
- Grabowska, D.; Jablonska-Skwiecinska, E.; Plochocka, D.; Chelstowska, A.; Lewandowska, I.; Witos, I.; Majewska, Z.; Rokicka-Milewska, R.; Burzynska, B. A novel mutation in the glucose-6-phosphate dehydrogenase gene in a subject with chronic nonspherocytic hemolytic anemia—Characterization of enzyme using yeast expression system and molecular modeling. Blood Cells Mol. Dis. 2004, 32, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Obiol-Pardo, C.; Rubio-Martinez, J. Homology modeling of human Transketolase: Description of critical sites useful for drug design and study of the cofactor binding mode. J. Mol. Graph. Model. 2009, 27, 723–734. [Google Scholar] [CrossRef]
- Raben, N.; Exelbert, R.; Spiegel, R.; Sherman, J.B.; Nakajima, H.; Plotz, P.; Heinisch, J. Functional expression of human mutant phosphofructokinase in yeast: Genetic defects in French Canadian and Swiss patients with phosphofructokinase deficiency. Am. J. Hum. Genet. 1995, 56, 131–141. [Google Scholar]
- Heinisch, J.J.; Buchwald, U.; Gottschlich, A.; Heppeler, N.; Rodicio, R.R. A tool kit for molecular genetics of Kluyveromyces lactis comprising a congenic strain series and a set of versatile vectors. FEMS Yeast Res. 2010, 10, 333–342. [Google Scholar] [CrossRef]
| Enzyme | Yeast | Gene (Systematic Name)/Accession Number 1 | Structure/ Identity 2 | Cofactors | Specific Activity (mU/mg) 3 | References |
|---|---|---|---|---|---|---|
| Glucose-6-phosphate dehydrogenase (G6PD) EC 1.1.1.49 | S. cerevisiae | ZWF1 (YNL241C) NC_001146.8 | 2(4) × 59 kDa | NADP+ | 100–180 | [26,27,28,29,30] |
| K. lactis | KlZWF1 (KLLA0D19855g) | 2(4) × 59 kDa 69% | NADP+ | 135–430 | [26,27,31,32] | |
| 6-Phosphoglucono-lactonase (6PGL) EC 3.1.1.31 | S. cerevisiae | SOL4 (YGR248W) NC_001139.9 SOL3 (YHR163W) NC_001140.6 | (?) × 28 kDa (?) × 28 kDa | - | n.d. | [33,34] |
| K. lactis | KlSOL4 (KLLA0A05390g) | (?) × 28 kDa 45%/53% | n.d. | |||
| 6-phosphogluconate dehydrogenase (6PGD) EC 1.1.1.44 | S. cerevisiae | GND1 (YHR183W) NC_001140.6 GND2 (YGR256W) NC_001139.9 | 2 × 52 kDa | NADP+ | 48c | [28,30,35,36] |
| K. lactis | KlGND1 (KLLA0A09339g) | (?) × 54 kDa 85%/81% | NADP+ | n.d. | ||
| Ribulosephosphate epimerase (RPE) EC 5.1.3.1 | S. cerevisiae | RPE1 (YJL121C) NC_001142.9 | (?) × 26 kDa | - | 1900–2200 | [28,37] |
| K. lactis | KlRPE1 (KLLA0E15071g) | (?) × 26 kDa 69% | ||||
| Ribosephosphate ketol isomerase (RKI) EC 5.3.1.6 | S. cerevisiae | RKI1 (YOR095C) NC_001147.6 | 4 × 28 kDa | - | 91 | [37,38] |
| K. lactis | KlRKI1 (KLLA0C13541g) | (?) × 30 kDa 65% | n.d. | |||
| Transketolase (TKL) EC 2.2.1.1 | S. cerevisiae | TKL1 (YPR074C) NC_001148.4 TKL2 (YBR117C) NC_001134.8 | 2 × 74 kDa | TPP Mg2+ | 80–100 | [27,39,40,41,42,43] |
| K. lactis | KlTKL1 (KLLA0B09152g) | (?) × 74 kDa 77%/70% | 230–260 | [27] | ||
| Transaldolase (TAL) EC 2.2.1.2 | S. cerevisiae | TAL1 (YLR354C) NM_001182243.1 | 2 × 38 kDa | - | 45–73 | [28,37,41,44] |
| K. lactis | KlTAL1 (KLLA0A02607g) | (?) × 36 kDa 75% | 298 | [44] | ||
| Sedoheptulose-1,7-bisphosphatase (SHB17) EC 3.1.3.37 | S. cerevisiae | SHB17 (YKR043C) NC_001143.9 | 2 × 31 kDa | Mg2+ | n.d. | [25] |
| K. lactis | KlSHB17 (KLLA0E14961g) | (?) × 31 kDa 71% |
| Enzyme | Disease | References |
|---|---|---|
| Glucose-6-phosphate Dehydrogenase (G6PD) | hemolytic anemia, diabetes, lung, liver, colorectal, prostate, and cervical cancer, leukemia, heart defects, Parkinson’s disease, Alzheimer’s disease | [45,135,136,137,138,139,140] |
| 6-Phosphoglucono-Lactonase (6PGL) | metastases in bones originating from breast cancer | [141] |
| 6-Phosphogluconate Dehydrogenase (6PGD) | lung and brain cancer | [136,137] |
| Ribosephosphate Isomerase (RPI) | pancreatic cancer and leukoencephalopathy | [136,142,143] |
| Ribulosephosphate Epimerase (RPE) | pancreatic cancer | [136] |
| Transketolase (TKL) | lung, liver and pancreatic cancer, Wernicke-Korsakoff syndrome, heart defects | [136,144,145,146] |
| Transaldolase (TAL) | lung cancer, liver cirrhosis, anemia, thrombocytopenia, heart defects, renal malfunction and neonatal edema multiple sclerosis, rheumatoid arthritis | [136,140,147,148,149,150] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bertels, L.-K.; Fernández Murillo, L.; Heinisch, J.J. The Pentose Phosphate Pathway in Yeasts–More Than a Poor Cousin of Glycolysis. Biomolecules 2021, 11, 725. https://doi.org/10.3390/biom11050725
Bertels L-K, Fernández Murillo L, Heinisch JJ. The Pentose Phosphate Pathway in Yeasts–More Than a Poor Cousin of Glycolysis. Biomolecules. 2021; 11(5):725. https://doi.org/10.3390/biom11050725
Chicago/Turabian StyleBertels, Laura-Katharina, Lucía Fernández Murillo, and Jürgen J. Heinisch. 2021. "The Pentose Phosphate Pathway in Yeasts–More Than a Poor Cousin of Glycolysis" Biomolecules 11, no. 5: 725. https://doi.org/10.3390/biom11050725
APA StyleBertels, L.-K., Fernández Murillo, L., & Heinisch, J. J. (2021). The Pentose Phosphate Pathway in Yeasts–More Than a Poor Cousin of Glycolysis. Biomolecules, 11(5), 725. https://doi.org/10.3390/biom11050725






