The Act of Controlling Adult Stem Cell Dynamics: Insights from Animal Models
Abstract
1. Introduction
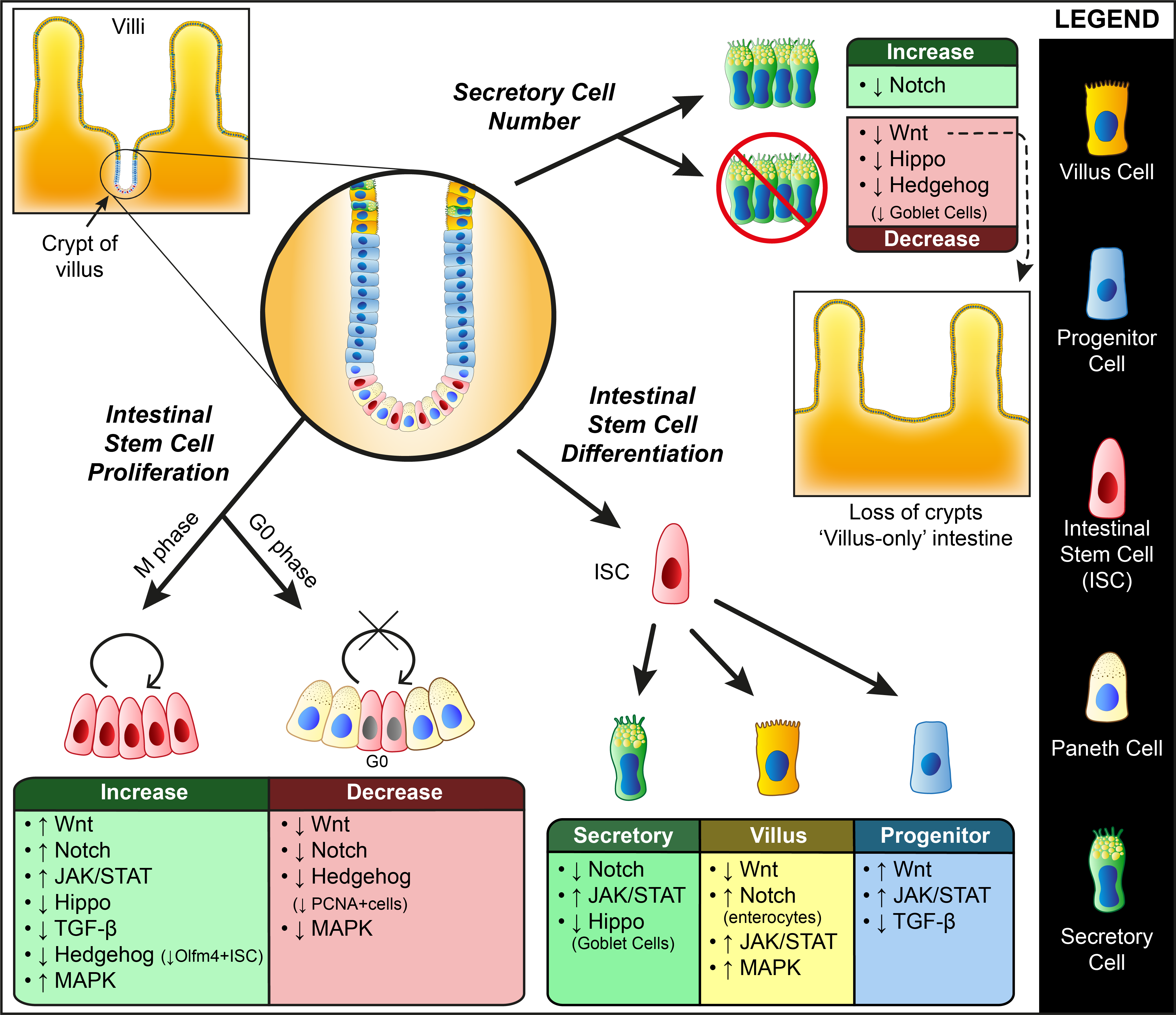
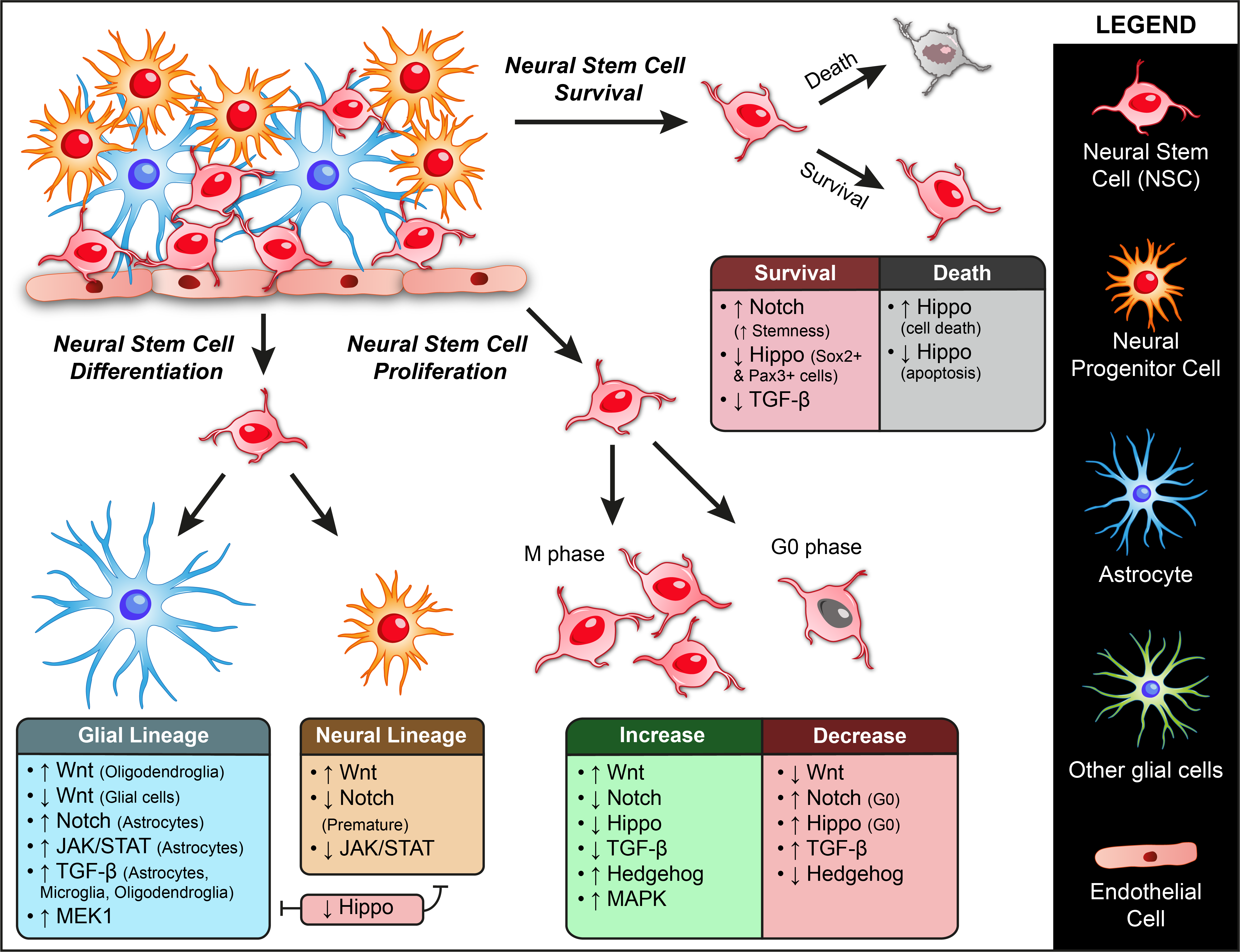
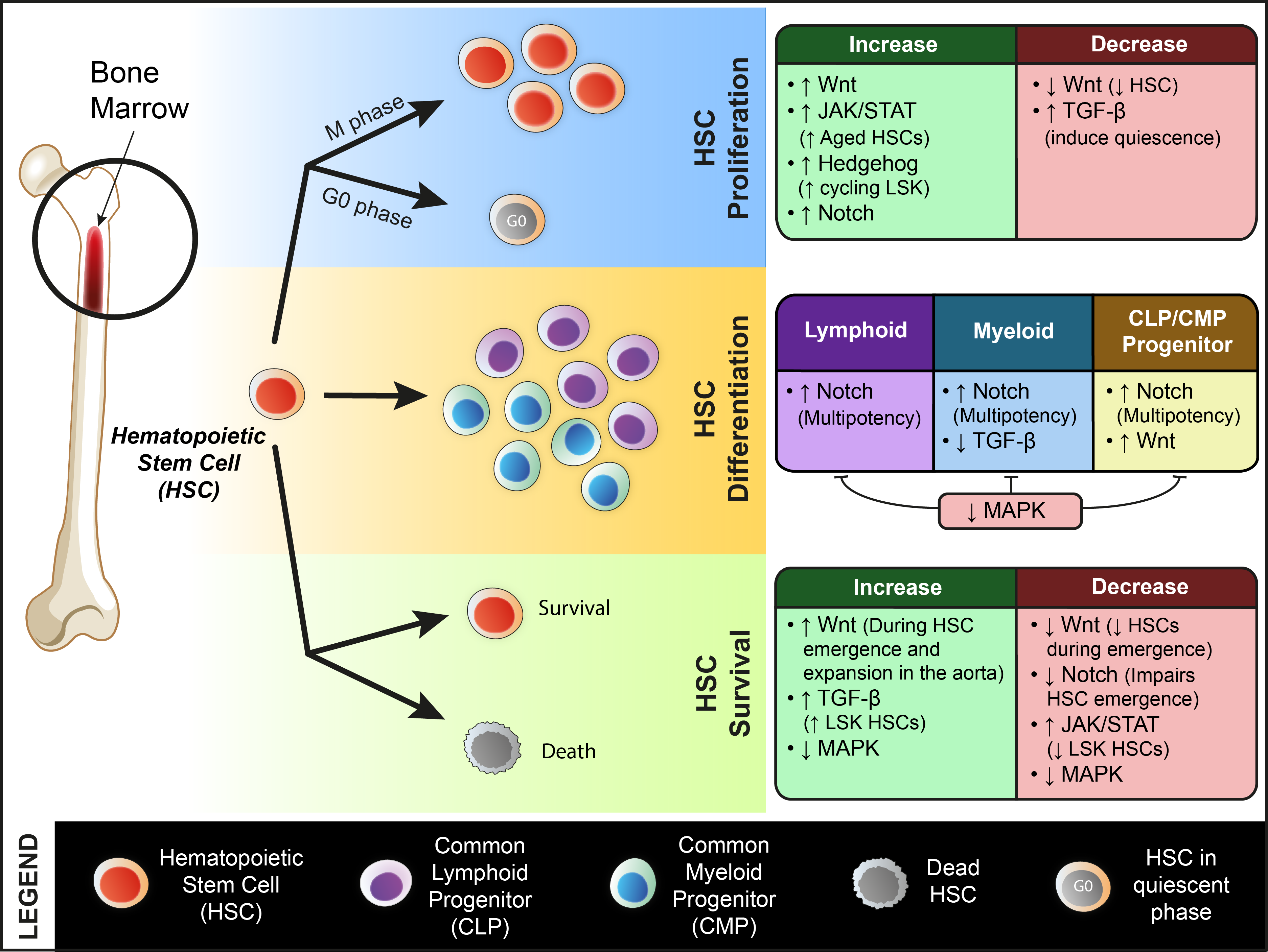
2. Genetic Factors Regulating ASC Dynamics
2.1. Wnt Signaling
2.2. Notch Signaling
2.3. JAK/STAT Pathway
2.4. Hippo Pathway
2.5. TGF-β Super-Family
2.6. Hedgehog Pathway
2.7. MAPK/ERK Signaling
3. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lowry, W.E.; Richter, L. Signaling in adult stem cells. Front. Biosci. 2007, 12, 3911–3927. [Google Scholar] [CrossRef] [PubMed]
- Logan, C.Y.; Nusse, R. The WNT Signaling Pathway in Development and Disease. Annu. Rev. Cell Dev. Biol. 2004, 20, 781–810. [Google Scholar] [CrossRef] [PubMed]
- Korinek, V.; Barker, N.; Moerer, P.; Van Donselaar, E.; Huls, G.; Peters, P.J.; Clevers, H. Depletion of epithelial stem-cell compartments in the small intestine of mice lacking Tcf-4. Nat. Genet. 1998, 19, 379–383. [Google Scholar] [CrossRef]
- Pinto, D.; Clevers, H. Wnt control of stem cells and differentiation in the intestinal epithelium. Exp. Cell Res. 2005, 306, 357–363. [Google Scholar] [CrossRef] [PubMed]
- Batlle, E.; Henderson, J.T.; Beghtel, H.; Born, M.M.V.D.; Sancho, E.; Huls, G.; Meeldijk, J.; Robertson, J.; van de Wetering, M.; Pawson, T.; et al. β-Catenin and TCF Mediate Cell Positioning in the Intestinal Epithelium by Controlling the Expression of EphB/EphrinB. Cell 2002, 111, 251–263. [Google Scholar] [CrossRef]
- Pinto, D.; Gregorieff, A.; Begthel, H.; Clevers, H. Canonical Wnt signals are essential for homeostasis of the intestinal epithelium. Genes Dev. 2003, 17, 1709–1713. [Google Scholar] [CrossRef]
- Sansom, O.J.; Reed, K.R.; Hayes, A.J.; Ireland, H.; Brinkmann, H.; Newton, I.P.; Batlle, E.; Simon-Assmann, P.; Clevers, H.; Nathke, I.S.; et al. Loss of Apc In Vivo immediately perturbs Wnt signaling, differentiation, and migration. Genes Dev. 2004, 18, 1385–1390. [Google Scholar] [CrossRef]
- Lin, G.; Xu, N.; Xi, R. Paracrine Wingless signalling controls self-renewal of Drosophila intestinal stem cells. Nat. Cell Biol. 2008, 455, 1119–1123. [Google Scholar] [CrossRef]
- Lee, W.-C.; Beebe, K.; Sudmeier, L.; Micchelli, C.A. Adenomatous polyposis coli regulates Drosophila intestinal stem cell proliferation. Development 2009, 136, 2255–2264. [Google Scholar] [CrossRef]
- Jiang, H.; Edgar, B.A. Intestinal stem cells in the adult Drosophila midgut. Exp. Cell Res. 2011, 317, 2780–2788. [Google Scholar] [CrossRef]
- Bengoa-Vergniory, N.; Kypta, R.M. Canonical and noncanonical Wnt signaling in neural stem/progenitor cells. Cell. Mol. Life Sci. 2015, 72, 4157–4172. [Google Scholar] [CrossRef]
- Kriska, J.; Honsa, P.; Dzamba, D.; Butenko, O.; Kolenicova, D.; Janeckova, L.; Nahacka, Z.; Andera, L.; Kozmik, Z.; Taketo, M.M.; et al. Manipulating Wnt signaling at different subcellular levels affects the fate of neonatal neural stem/progenitor cells. Brain Res. 2016, 1651, 73–87. [Google Scholar] [CrossRef]
- Wei, L.-C.; Ding, Y.-X.; Liu, Y.-H.; Duan, L.; Bai, Y.; Shi, M.; Chen, L.-W. Low-Dose Radiation Stimulates Wnt/β-Catenin Signaling, Neural Stem Cell Proliferation and Neurogenesis of the Mouse Hippocampus in vitro and In Vivo. Curr. Alzheimer Res. 2012, 9, 278–289. [Google Scholar] [CrossRef]
- Adachi, K.; Mirzadeh, Z.; Sakaguchi, M.; Yamashita, T.; Nikolcheva, T.; Gotoh, Y.; Peltz, G.; Gong, L.; Kawase, T.; Alvarez-Buylla, A.; et al. β-Catenin Signaling Promotes Proliferation of Progenitor Cells in the Adult Mouse Subventricular Zone. Stem Cells 2007, 25, 2827–2836. [Google Scholar] [CrossRef]
- Bowman, A.N.; Van Amerongen, R.; Palmer, T.D.; Nusse, R. Lineage tracing with Axin2 reveals distinct developmental and adult populations of Wnt/-catenin-responsive neural stem cells. Proc. Natl. Acad. Sci. USA 2013, 110, 7324–7329. [Google Scholar] [CrossRef]
- Piccin, D.; Morshead, C.M. Wnt Signaling Regulates Symmetry of Division of Neural Stem Cells in the Adult Brain and in Response to Injury. Stem Cells 2011, 29, 528–538. [Google Scholar] [CrossRef]
- Ortega, F.; Gascón, S.; Masserdotti, G.; Deshpande, A.; Simon, C.; Fischer, J.; Dimou, L.; Lie, D.C.; Schroeder, T.; Berninger, B. Oligodendrogliogenic and neurogenic adult subependymal zone neural stem cells constitute distinct lineages and exhibit differential responsiveness to Wnt signalling. Nat. Cell Biol. 2013, 15, 602–613. [Google Scholar] [CrossRef]
- Chen, B.-Y.; Wang, X.; Wang, Z.-Y.; Wang, Y.-Z.; Chen, L.-W.; Luo, Z.-J. Brain-derived neurotrophic factor stimulates proliferation and differentiation of neural stem cells, possibly by triggering the Wnt/β-catenin signaling pathway. J. Neurosci. Res. 2012, 91, 30–41. [Google Scholar] [CrossRef]
- Samarut, E.; Bekri, A.; Drapeau, P. Transcriptomic Analysis of Purified Embryonic Neural Stem Cells from Zebrafish Embryos Reveals Signaling Pathways Involved in Glycine-Dependent Neurogenesis. Front. Mol. Neurosci. 2016, 9, 22. [Google Scholar] [CrossRef]
- Shitasako, S.; Ito, Y.; Ito, R.; Ueda, Y.; Shimizu, Y.; Ohshima, T. Wnt and Shh signals regulate neural stem cell proliferation and differentiation in the optic tectum of adult zebrafish. Dev. Neurobiol. 2017, 77, 1206–1220. [Google Scholar] [CrossRef]
- Duncan, R.N.; Panahi, S.; Piotrowski, T.; Dorsky, R.I. Identification of Wnt Genes Expressed in Neural Progenitor Zones during Zebrafish Brain Development. PLoS ONE 2015, 10, e0145810. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.D.; Molinaro, A.M.; Pearson, B.J. The planarian TCF/LEF factor Smed-tcf1 is required for the regeneration of dorsal-lateral neuronal subtypes. Dev. Biol. 2018, 433, 374–383. [Google Scholar] [CrossRef] [PubMed]
- Reddien, P.W. The Cellular and Molecular Basis for Planarian Regeneration. Cell 2018, 175, 327–345. [Google Scholar] [CrossRef] [PubMed]
- Witchley, J.N.; Mayer, M.; Wagner, D.E.; Owen, J.H.; Reddien, P.W. Muscle Cells Provide Instructions for Planarian Regeneration. Cell Rep. 2013, 4, 633–641. [Google Scholar] [CrossRef]
- Petersen, C.P.; Reddien, P.W. Polarized notum Activation at Wounds Inhibits Wnt Function to Promote Planarian Head Regeneration. Science 2011, 332, 852–855. [Google Scholar] [CrossRef] [PubMed]
- Gurley, K.A.; Rink, J.C.; Alvarado, A.S. β-Catenin Defines Head Versus Tail Identity During Planarian Regeneration and Homeostasis. Science 2008, 319, 323–327. [Google Scholar] [CrossRef]
- Richter, J.; Traver, D.; Willert, K. The role of Wnt signaling in hematopoietic stem cell development. Crit. Rev. Biochem. Mol. Biol. 2017, 52, 414–424. [Google Scholar] [CrossRef]
- Famili, F.; Perez, L.G.; Naber, B.A.; Noordermeer, J.N.; Fradkin, L.G.; Staal, F.J. The non-canonical Wnt receptor Ryk regulates hematopoietic stem cell repopulation in part by controlling proliferation and apoptosis. Cell Death Dis. 2016, 7, e2479. [Google Scholar] [CrossRef]
- Famili, F.; Brugman, M.H.; Taskesen, E.; Naber, B.E.; Fodde, R.; Staal, F.J. High Levels of Canonical Wnt Signaling Lead to Loss of Stemness and Increased Differentiation in Hematopoietic Stem Cells. Stem Cell Rep. 2016, 6, 652–659. [Google Scholar] [CrossRef]
- Baba, Y.; Garrett, K.P.; Kincade, P.W. Constitutively Active β-Catenin Confers Multilineage Differentiation Potential on Lymphoid and Myeloid Progenitors. Immunity 2005, 23, 599–609. [Google Scholar] [CrossRef]
- Kabiri, Z.; Numata, A.; Kawasaki, A.; Edison; Tenen, D.G.; Virshup, D.M. Wnts are dispensable for differentiation and self-renewal of adult murine hematopoietic stem cells. Blood 2015, 126, 1086–1094. [Google Scholar] [CrossRef]
- Zhang, C.; Patient, R.; Liu, F. Hematopoietic stem cell development and regulatory signaling in zebrafish. Biochim. Biophys. Acta (BBA) Gen. Subj. 2013, 1830, 2370–2374. [Google Scholar] [CrossRef]
- Clements, W.K.; Kim, A.D.; Ong, K.G.; Moore, J.C.; Lawson, N.D.; Traver, D. A somitic Wnt16/Notch pathway specifies haematopoietic stem cells. Nat. Cell Biol. 2011, 474, 220–224. [Google Scholar] [CrossRef]
- Grainger, S.; Richter, J.; Palazón, R.E.; Pouget, C.; Lonquich, B.; Wirth, S.; Grassme, K.S.; Herzog, W.; Swift, M.R.; Weinstein, B.M.; et al. Wnt9a Is Required for the Aortic Amplification of Nascent Hematopoietic Stem Cells. Cell Rep. 2016, 17, 1595–1606. [Google Scholar] [CrossRef]
- Huelsken, J.; Vogel, R.; Erdmann, B.; Cotsarelis, G.; Birchmeier, W. β-Catenin Controls Hair Follicle Morphogenesis and Stem Cell Differentiation in the Skin. Cell 2001, 105, 533–545. [Google Scholar] [CrossRef]
- Watt, F.; Collins, C. Role of -catenin in Epidermal Stem Cell Expansion, Lineage Selection, and Cancer. Cold Spring Harb. Symp. Quant. Biol. 2008, 73, 503–512. [Google Scholar] [CrossRef]
- Watt, F.M.; Jensen, K.B. Epidermal stem cell diversity and quiescence. EMBO Mol. Med. 2009, 1, 260–267. [Google Scholar] [CrossRef]
- Kandyba, E.; Leung, Y.; Chen, Y.-B.; Widelitz, R.; Chuong, C.-M.; Kobielak, K. Competitive balance of intrabulge BMP/Wnt signaling reveals a robust gene network ruling stem cell homeostasis and cyclic activation. Proc. Natl. Acad. Sci. USA 2013, 110, 1351–1356. [Google Scholar] [CrossRef]
- Lim, X.; Tan, S.H.; Koh, W.L.C.; Chau, R.M.W.; Yan, K.S.; Kuo, C.J.; Van Amerongen, R.; Klein, A.M.; Nusse, R. Interfollicular Epidermal Stem Cells Self-Renew via Autocrine Wnt Signaling. Science 2013, 342, 1226–1230. [Google Scholar] [CrossRef]
- Choi, Y.S.; Zhang, Y.; Xu, M.; Yang, Y.; Ito, M.; Peng, T.; Cui, Z.; Nagy, A.; Hadjantonakis, A.-K.; Lang, R.A.; et al. Distinct Functions for Wnt/β-Catenin in Hair Follicle Stem Cell Proliferation and Survival and Interfollicular Epidermal Homeostasis. Cell Stem Cell 2013, 13, 720–733. [Google Scholar] [CrossRef]
- Kretzschmar, K.; Cottle, D.L.; Schweiger, P.J.; Watt, F.M. The Androgen Receptor Antagonizes Wnt/β-Catenin Signaling in Epidermal Stem Cells. J. Investig. Dermatol. 2015, 135, 2753–2763. [Google Scholar] [CrossRef] [PubMed]
- Abnave, P.; Aboukhatwa, E.; Kosaka, N.; Thompson, J.; Hill, M.A.; Aboobaker, A.A. Epithelial-mesenchymal transition transcription factors control pluripotent adult stem cell migration In Vivo in planarians. Development 2017, 144, 3440–3453. [Google Scholar] [CrossRef] [PubMed]
- Pellegrinet, L.; Rodilla, V.; Liu, Z.; Chen, S.; Koch, U.; Espinosa, L.; Kaestner, K.H.; Kopan, R.; Lewis, J.; Radtke, F. Dll1 and Dll4-Mediated Notch Signaling Are Required for Homeostasis of Intestinal Stem Cells. Gastroenterology 2011, 140, 1230–1240.e7. [Google Scholar] [CrossRef] [PubMed]
- Tsai, Y.-H.; VanDussen, K.L.; Sawey, E.T.; Wade, A.W.; Kasper, C.; Rakshit, S.; Bhatt, R.G.; Stoeck, A.; Maillard, I.; Crawford, H.C.; et al. ADAM10 Regulates Notch Function in Intestinal Stem Cells of Mice. Gastroenterology 2014, 147, 822–834.e13. [Google Scholar] [CrossRef]
- Tian, H.; Biehs, B.; Chiu, C.; Siebel, C.W.; Wu, Y.; Costa, M.; de Sauvage, F.J.; Klein, O.D. Opposing Activities of Notch and Wnt Signaling Regulate Intestinal Stem Cells and Gut Homeostasis. Cell Rep. 2015, 11, 33–42. [Google Scholar] [CrossRef]
- Fre, S.; Hannezo, E.; Sale, S.; Huyghe, M.; Lafkas, D.; Kissel, H.; Louvi, A.; Greve, J.; Louvard, D.; Artavanis-Tsakonas, S. Notch Lineages and Activity in Intestinal Stem Cells Determined by a New Set of Knock-In Mice. PLoS ONE 2011, 6, e25785. [Google Scholar] [CrossRef]
- Carulli, A.J.; Keeley, T.M.; Demitrack, E.S.; Chung, J.; Maillard, I.; Samuelson, L.C. Notch receptor regulation of intestinal stem cell homeostasis and crypt regeneration. Dev. Biol. 2015, 402, 98–108. [Google Scholar] [CrossRef]
- Bohin, N.; Keeley, T.M.; Carulli, A.J.; Walker, E.M.; Carlson, E.A.; Gao, J.; Aifantis, I.; Siebel, C.W.; Rajala, M.W.; Myers, M.G.; et al. Rapid Crypt Cell Remodeling Regenerates the Intestinal Stem Cell Niche after Notch Inhibition. Stem Cell Rep. 2020, 15, 156–170. [Google Scholar] [CrossRef]
- Micchelli, C.A.; Perrimon, N. Evidence that stem cells reside in the adult Drosophila midgut epithelium. Nat. Cell Biol. 2005, 439, 475–479. [Google Scholar] [CrossRef]
- Ohlstein, B.; Spradling, A.C. The adult Drosophila posterior midgut is maintained by pluripotent stem cells. Nat. Cell Biol. 2005, 439, 470–474. [Google Scholar] [CrossRef]
- Ohlstein, B.; Spradling, A. Multipotent Drosophila Intestinal Stem Cells Specify Daughter Cell Fates by Differential Notch Signaling. Science 2007, 315, 988–992. [Google Scholar] [CrossRef]
- Imayoshi, I.; Kageyama, R. The Role of Notch Signaling in Adult Neurogenesis. Mol. Neurobiol. 2011, 44, 7–12. [Google Scholar] [CrossRef]
- Matsumoto, A.; Onoyama, I.; Sunabori, T.; Kageyama, R.; Okano, H.; Nakayama, K.I. Fbxw7-dependent Degradation of Notch Is Required for Control of “Stemness” and Neuronal-Glial Differentiation in Neural Stem Cells. J. Biol. Chem. 2011, 286, 13754–13764. [Google Scholar] [CrossRef]
- Gao, F.; Zhang, Y.-F.; Zhang, Z.-P.; Fu, L.-A.; Cao, X.-L.; Zhang, Y.-Z.; Guo, C.-J.; Yan, X.-C.; Yang, Q.-C.; Hu, Y.-Y.; et al. miR-342-5p Regulates Neural Stem Cell Proliferation and Differentiation Downstream to Notch Signaling in Mice. Stem Cell Rep. 2017, 8, 1032–1045. [Google Scholar] [CrossRef]
- Imayoshi, I.; Sakamoto, M.; Yamaguchi, M.; Mori, K.; Kageyama, R. Essential Roles of Notch Signaling in Maintenance of Neural Stem Cells in Developing and Adult Brains. J. Neurosci. 2010, 30, 3489–3498. [Google Scholar] [CrossRef]
- Ehm, O.; Göritz, C.; Covic, M.; Schäffner, I.; Schwarz, T.J.; Karaca, E.; Kempkes, B.; Kremmer, E.; Pfrieger, F.W.; Espinosa, L.; et al. RBPJ -Dependent Signaling Is Essential for Long-Term Maintenance of Neural Stem Cells in the Adult Hippocampus. J. Neurosci. 2010, 30, 13794–13807. [Google Scholar] [CrossRef]
- Li, J.; Han, Y.; Li, M.; Nie, C. Curcumin Promotes Proliferation of Adult Neural Stem Cells and the Birth of Neurons in Alzheimer’s Disease Mice via Notch Signaling Pathway. Cell. Reprogram. 2019, 21, 152–161. [Google Scholar] [CrossRef]
- Aguirre, A.; Rubio, M.E.; Gallo, V. Notch and EGFR pathway interaction regulates neural stem cell number and self-renewal. Nat. Cell Biol. 2010, 467, 323–327. [Google Scholar] [CrossRef]
- Lin, R.; Cai, J.; Kenyon, L.; Iozzo, R.; Rosenwasser, R.; Iacovitti, L. Systemic Factors Trigger Vasculature Cells to Drive Notch Signaling and Neurogenesis in Neural Stem Cells in the Adult Brain. Stem Cells 2019, 37, 395–406. [Google Scholar] [CrossRef]
- Chapouton, P.; Skupien, P.; Hesl, B.; Coolen, M.; Moore, J.C.; Madelaine, R.; Kremmer, E.; Faus-Kessler, T.; Blader, P.; Lawson, N.D.; et al. Notch Activity Levels Control the Balance between Quiescence and Recruitment of Adult Neural Stem Cells. J. Neurosci. 2010, 30, 7961–7974. [Google Scholar] [CrossRef]
- Berberoglu, M.A.; Dong, Z.; Li, G.; Zheng, J.; Martinez, L.D.C.G.T.; Peng, J.; Wagle, M.; Reichholf, B.; Petritsch, C.; Li, H.; et al. Heterogeneously expressed fezf2 patterns gradient Notch activity in balancing the quiescence, proliferation, and differentiation of adult neural stem cells. J. Neurosci. 2014, 34, 13911–13923. [Google Scholar] [CrossRef]
- Arya, R.; Sarkissian, T.; Tan, Y.; White, K. Neural stem cell progeny regulate stem cell death in a Notch and Hox dependent manner. Cell Death Differ. 2015, 22, 1378–1387. [Google Scholar] [CrossRef]
- Egger, B.; Gold, K.S.; Brand, A.H. Notch regulates the switch from symmetric to asymmetric neural stem cell division in the Drosophila optic lobe. Development 2010, 137, 2981–2987. [Google Scholar] [CrossRef]
- Pajcini, K.V.; Speck, N.A.; Pear, W.S. Notch signaling in mammalian hematopoietic stem cells. Leukemia 2011, 25, 1525–1532. [Google Scholar] [CrossRef]
- Calvi, L.M.; Adams, G.B.; Weibrecht, K.W.; Weber, J.; Olson, D.P.; Knight, M.C.; Martin, R.P.; Schipani, E.; Divieti, P.; Bringhurst, F.R.; et al. Osteoblastic cells regulate the haematopoietic stem cell niche. Nat. Cell Biol. 2003, 425, 841–846. [Google Scholar] [CrossRef]
- Tanigaki, K.; Han, H.; Yamamoto, N.; Tashiro, K.; Ikegawa, M.; Kuroda, K.; Suzuki, A.; Nakano, T.; Honjo, T. Notch–RBP-J signaling is involved in cell fate determination of marginal zone B cells. Nat. Immunol. 2002, 3, 443–450. [Google Scholar] [CrossRef]
- Han, H.; Tanigaki, K.; Yamamoto, N.; Kuroda, K.; Yoshimoto, M.; Nakahata, T.; Ikuta, K.; Honjo, T. Inducible gene knockout of transcription factor recombination signal binding protein-J reveals its essential role in T versus B lineage decision. Int. Immunol. 2002, 14, 637–645. [Google Scholar] [CrossRef]
- Lampreia, F.P.; Carmelo, J.G.; Anjos-Afonso, F. Notch Signaling in the Regulation of Hematopoietic Stem Cell. Curr. Stem Cell Rep. 2017, 3, 202–209. [Google Scholar] [CrossRef]
- Perlin, J.R.; Robertson, A.L.; Zon, L.I. Efforts to enhance blood stem cell engraftment: Recent insights from zebrafish hematopoiesis. J. Exp. Med. 2017, 214, 2817–2827. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Tu, H.; Kang, Y.; Xue, Y.; Ma, D.; Zhao, C.; Li, H.; Wang, L.; Liu, F. Primary cilia regulate hematopoietic stem and progenitor cell specification through Notch signaling in zebrafish. Nat. Commun. 2019, 10, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Konantz, M.; Alghisi, E.; Müller, J.S.; Lenard, A.; Esain, V.; Carroll, K.J.; Kanz, L.; E North, T.; Lengerke, C. Evi1 regulates Notch activation to induce zebrafish hematopoietic stem cell emergence. EMBO J. 2016, 35, 2315–2331. [Google Scholar] [CrossRef] [PubMed]
- Gerri, C.; Marass, M.; Rossi, A.; Stainier, D.Y.R. Hif-1α and Hif-2α regulate hemogenic endothelium and hematopoietic stem cell formation in zebrafish. Blood 2018, 131, 963–973. [Google Scholar] [CrossRef] [PubMed]
- Watt, F.M.; Estrach, S.; Ambler, C.A. Epidermal Notch signalling: Differentiation, cancer and adhesion. Curr. Opin. Cell Biol. 2008, 20, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Vauclair, S.; Nicolas, M.; Barrandon, Y.; Radtke, F. Notch1 is essential for postnatal hair follicle development and homeostasis. Dev. Biol. 2005, 284, 184–193. [Google Scholar] [CrossRef]
- Pan, Y.; Lin, M.-H.; Tian, X.; Cheng, H.-T.; Gridley, T.; Shen, J.; Kopan, R. γ-Secretase Functions through Notch Signaling to Maintain Skin Appendages but Is Not Required for Their Patterning or Initial Morphogenesis. Dev. Cell 2004, 7, 731–743. [Google Scholar] [CrossRef]
- Uyttendaele, H.; Panteleyev, A.A.; de Berker, D.; Tobin, D.T.; Christiano, A.M. Activation of Notch1 in the hair follicle leads to cell-fate switch and Mohawk alopecia. Differentiation 2004, 72, 396–409. [Google Scholar] [CrossRef]
- Yang, R.; Wang, J.; Chen, X.; Shi, Y.; Xie, J. Epidermal Stem Cells in Wound Healing and Regeneration. Stem Cells Int. 2020, 2020, 9148310. [Google Scholar] [CrossRef]
- Wang, P.; Shu, B.; Xu, Y.; Zhu, J.; Liu, J.; Zhou, Z.; Chen, L.; Zhao, J.; Liu, X.; Qi, S.; et al. Basic fibroblast growth factor reduces scar by inhibiting the differentiation of epidermal stem cells to myofibroblasts via the Notch1/Jagged1 pathway. Stem Cell Res. Ther. 2017, 8, 114. [Google Scholar] [CrossRef]
- Yang, R.-H.; Qi, S.-H.; Shu, B.; Ruan, S.-B.; Lin, Z.-P.; Lin, Y.; Shen, R.; Zhang, F.-G.; Chen, X.-D.; Xie, J.-L. Epidermal stem cells (ESCs) accelerate diabetic wound healing via the Notch signalling pathway. Biosci. Rep. 2016, 36, e00364. [Google Scholar] [CrossRef]
- Olivera-Martinez, I.; Schurch, N.; Li, R.A.; Song, J.; Halley, P.A.; Das, R.M.; Burt, D.W.; Barton, G.J.; Storey, K.G. Major transcriptome re-organisation and abrupt changes in signalling, cell cycle and chromatin regulation at neural differentiation In Vivo. Development 2014, 141, 3266–3276. [Google Scholar] [CrossRef]
- Jiang, H.; Patel, P.H.; Kohlmaier, A.; Grenley, M.O.; McEwen, D.G.; Edgar, B.A. Cytokine/Jak/Stat Signaling Mediates Regeneration and Homeostasis in the Drosophila Midgut. Cell 2009, 137, 1343–1355. [Google Scholar] [CrossRef]
- Liu, W.; Singh, S.R.; Hou, S.X. JAK-STAT is restrained by Notch to control cell proliferation of the Drosophila intestinal stem cells. J. Cell. Biochem. 2010, 109, 992–999. [Google Scholar] [CrossRef]
- Lin, G.; Xu, N.; Xi, R. Paracrine Unpaired Signaling through the JAK/STAT Pathway Controls Self-renewal and Lineage Differentiation of Drosophila Intestinal Stem Cells. J. Mol. Cell Biol. 2009, 2, 37–49. [Google Scholar] [CrossRef]
- Xu, N.; Wang, S.Q.; Tan, D.; Gao, Y.; Lin, G.; Xi, R. EGFR, Wingless and JAK/STAT signaling cooperatively maintain Drosophila intestinal stem cells. Dev. Biol. 2011, 354, 31–43. [Google Scholar] [CrossRef]
- Gonneaud, A.; Turgeon, N.; Boisvert, F.-M.; Boudreau, F.; Asselin, C. JAK-STAT Pathway Inhibition Partially Restores Intestinal Homeostasis in Hdac1 and Hdac2-Intestinal Epithelial Cell-Deficient Mice. Cells 2021, 10, 224. [Google Scholar] [CrossRef]
- Wang, T.; Yuan, W.; Liu, Y.; Zhang, Y.; Wang, Z.; Zhou, X.; Ning, G.; Zhang, L.; Yao, L.; Feng, S.; et al. The role of the JAK-STAT pathway in neural stem cells, neural progenitor cells and reactive astrocytes after spinal cord injury. Biomed. Rep. 2015, 3, 141–146. [Google Scholar] [CrossRef]
- Stine, R.R.; Matunis, E.L. JAK-STAT signaling in stem cells. Adv. Exp. Med. Biol. 2013, 786, 247–267. [Google Scholar] [CrossRef]
- Tanaka, Y.; Yasugi, T.; Nagayama, M.; Sato, M.; Ei, S.-I. JAK/STAT guarantees robust neural stem cell differentiation by shutting off biological noise. Sci. Rep. 2018, 8, 1–9. [Google Scholar] [CrossRef]
- Du, L.; Wang, J. JAK/STAT pathway promotes Drosophila neuroblast proliferation via the direct CycE regulation. bioRxiv 2020. [Google Scholar] [CrossRef]
- Wang, W.; Li, Y.; Zhou, L.; Yue, H.; Luo, H. Role of JAK/STAT signaling in neuroepithelial stem cell maintenance and proliferation in the Drosophila optic lobe. Biochem. Biophys. Res. Commun. 2011, 410, 714–720. [Google Scholar] [CrossRef]
- Zyuz’Kov, G.N.; Miroshnichenko, L.A.; Polyakova, T.Y.; Stavrova, L.A.; Simanina, E.V.; Zhdanov, V.V. Specific Roles of JAKs and STAT3 in Functions of Neural Stem Cells and Committed Neuronal Progenitors during Ethanol-Induced Neurodegeneration. Bull. Exp. Biol. Med. 2020, 168, 356–360. [Google Scholar] [CrossRef]
- Schneider, L.; Pellegatta, S.; Favaro, R.; Pisati, F.; Roncaglia, P.; Testa, G.; Nicolis, S.K.; Finocchiaro, G.; di Fagagna, F.D. DNA Damage in Mammalian Neural Stem Cells Leads to Astrocytic Differentiation Mediated by BMP2 Signaling through JAK-STAT. Stem Cell Rep. 2013, 1, 123–138. [Google Scholar] [CrossRef]
- Zhang, Y.-L.; Zhou, Z.; Han, W.-W.; Zhang, L.-L.; Song, W.-S.; Huang, J.-H.; Liu, S. Oleanolic Acid Inhibiting the Differentiation of Neural Stem Cells into Astrocyte by Down-Regulating JAK/STAT Signaling Pathway. Am. J. Chin. Med. 2016, 44, 103–117. [Google Scholar] [CrossRef]
- Mao, S.; Li, X.; Wang, J.; Ding, X.; Zhang, C.; Li, L. miR-17-92 facilitates neuronal differentiation of transplanted neural stem / precursor cells under neuroinflammatory conditions. J. Neuroinflamm. 2016, 1–14. [Google Scholar] [CrossRef]
- Gregory, L.; Came, P.J.; Brown, S. Stem cell regulation by JAK/STAT signaling in Drosophila. Semin. Cell Dev. Biol. 2008, 19, 407–413. [Google Scholar] [CrossRef]
- Kirschner, K.; Chandra, T.; Kiselev, V.; Cruz, D.F.-S.; Macaulay, I.C.; Park, H.J.; Li, J.; Kent, D.G.; Kumar, R.; Pask, D.C.; et al. Proliferation Drives Aging-Related Functional Decline in a Subpopulation of the Hematopoietic Stem Cell Compartment. Cell Rep. 2017, 19, 1503–1511. [Google Scholar] [CrossRef]
- Li, J.; Spensberger, D.; Ahn, J.S.; Anand, S.; Beer, P.A.; Ghevaert, C.; Chen, E.; Forrai, A.; Scott, L.M.; Ferreira, R.; et al. JAK2 V617F impairs hematopoietic stem cell function in a conditional knock-in mouse model of JAK2 V617F–positive essential thrombocythemia. Blood 2010, 116, 1528–1538. [Google Scholar] [CrossRef]
- Woods, B.; Chen, W.; Chiu, S.; Marinaccio, C.; Fu, C.; Gu, L.; Bulic, M.; Yang, Q.; Zouak, A.; Jia, S.; et al. Activation of JAK/STAT Signaling in Megakaryocytes Sustains Myeloproliferation In Vivo. Clin. Cancer Res. 2019, 25, 5901–5912. [Google Scholar] [CrossRef]
- Ma, A.C.H.; Ward, A.C.; Liang, R.; Leung, A.Y.H. The role of jak2a in zebrafish hematopoiesis. Blood 2007, 110, 1824–1830. [Google Scholar] [CrossRef]
- Sano, S.; Itami, S.; Takeda, K.; Tarutani, M.; Yamaguchi, Y.; Miura, H.; Yoshikawa, K.; Akira, S.; Takeda, J. Keratinocyte-specific ablation of Stat3 exhibits impaired skin remodeling, but does not affect skin morphogenesis. EMBO J. 1999, 18, 4657–4668. [Google Scholar] [CrossRef]
- Harel, S.; Higgins, C.A.; Cerise, J.E.; Dai, Z.; Chen, J.C.; Clynes, R.; Christiano, A.M. Pharmacologic inhibition of JAK-STAT signaling promotes hair growth. Sci. Adv. 2015, 1, e1500973. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, J.; Fletcher, S.; Roth, E.; Wu, C.; Chun, A.; Horsley, V. Calcineurin/Nfatc1 signaling links skin stem cell quiescence to hormonal signaling during pregnancy and lactation. Genes Dev. 2014, 28, 983–994. [Google Scholar] [CrossRef] [PubMed]
- Ren, F.; Wang, B.; Yue, T.; Yun, E.-Y.; Ip, Y.T.; Jiang, J. Hippo signaling regulates Drosophila intestine stem cell proliferation through multiple pathways. Proc. Natl. Acad. Sci. USA 2010, 107, 21064–21069. [Google Scholar] [CrossRef] [PubMed]
- Camargo, F.D.; Gokhale, S.; Johnnidis, J.B.; Fu, D.; Bell, G.W.; Jaenisch, R.; Brummelkamp, T.R. YAP1 Increases Organ Size and Expands Undifferentiated Progenitor Cells. Curr. Biol. 2007, 17, 2054–2060. [Google Scholar] [CrossRef]
- Zhao, B.; Tumaneng, K.; Guan, K.-L. The Hippo pathway in organ size control, tissue regeneration and stem cell self-renewal. Nat. Cell Biol. 2011, 13, 877–883. [Google Scholar] [CrossRef]
- Zhou, D.; Zhang, Y.; Wu, H.; Barry, E.; Yin, Y.; Lawrence, E.; Dawson, D.; Willis, J.E.; Markowitz, S.D.; Camargo, F.D.; et al. Mst1 and Mst2 protein kinases restrain intestinal stem cell proliferation and colonic tumorigenesis by inhibition of Yes-associated protein (Yap) overabundance. Proc. Natl. Acad. Sci. USA 2011, 108, E1312–E1320. [Google Scholar] [CrossRef]
- Imajo, M.; Ebisuya, M.; Nishida, E. Dual role of YAP and TAZ in renewal of the intestinal epithelium. Nat. Cell Biol. 2015, 17, 7–19. [Google Scholar] [CrossRef]
- Cai, J.; Zhang, N.; Zheng, Y.; De Wilde, R.F.; Maitra, A.; Pan, D. The Hippo signaling pathway restricts the oncogenic potential of an intestinal regeneration program. Genes Dev. 2010, 24, 2383–2388. [Google Scholar] [CrossRef]
- Barry, E.R.; Morikawa, T.; Butler, B.L.; Shrestha, K.; De La Rosa, R.; Yan, K.S.; Fuchs, C.S.; Magness, S.T.; Smits, R.; Ogino, S.; et al. Restriction of intestinal stem cell expansion and the regenerative response by YAP. Nature 2013, 493, 106–110. [Google Scholar] [CrossRef]
- Poon, C.L.; Mitchell, K.A.; Kondo, S.; Cheng, L.Y.; Harvey, K.F. The Hippo Pathway Regulates Neuroblasts and Brain Size in Drosophila melanogaster. Curr. Biol. 2016, 26, 1034–1042. [Google Scholar] [CrossRef]
- Ding, R.; Weynans, K.; Bossing, T.; Barros, C.S.; Berger, C. The Hippo signalling pathway maintains quiescence in Drosophila neural stem cells. Nat. Commun. 2016, 7, 10510. [Google Scholar] [CrossRef]
- Gil-Ranedo, J.; Gonzaga, E.; Jaworek, K.J.; Berger, C.; Bossing, T.; Barros, C.S. STRIPAK Members Orchestrate Hippo and Insulin Receptor Signaling to Promote Neural Stem Cell Reactivation. Cell Rep. 2019, 27, 2921–2933.e5. [Google Scholar] [CrossRef]
- Ding, R.; Berger, C. Hippo pathway regulates neural stem cell quiescence. Cell Cycle 2016, 15, 1525–1526. [Google Scholar] [CrossRef] [PubMed]
- Gee, S.T.; Milgram, S.L.; Kramer, K.L.; Conlon, F.L.; Moody, S.A. Yes-Associated Protein 65 (YAP) Expands Neural Progenitors and Regulates Pax3 Expression in the Neural Plate Border Zone. PLoS ONE 2011, 6, e20309. [Google Scholar] [CrossRef]
- Cao, X.; Pfaff, S.L.; Gage, F.H. YAP regulates neural progenitor cell number via the TEA domain transcription factor. Genes Dev. 2008, 22, 3320–3334. [Google Scholar] [CrossRef]
- Lavado, A.; Park, J.Y.; Paré, J.; Finkelstein, D.; Pan, H.; Xu, B.; Fan, Y.; Kumar, R.P.; Neale, G.; Kwak, Y.D.; et al. The Hippo Pathway Prevents YAP/TAZ-Driven Hypertranscription and Controls Neural Progenitor Number. Dev. Cell 2018, 47, 576–591.e8. [Google Scholar] [CrossRef]
- Han, D.; Lee, S.M.; Kwon, M.; Noh, H.; Lee, J.H.; Yoon, Y.; Cho, J.Y.; Yoon, K. Yes-Associated Protein Enhances Fibroblast Growth Factor 2-Dependent Neural Stem Cell Proliferation by Induction of Fibroblast Growth Factor Receptor Expression. Stem Cells Dev. 2020, 29, 1240–1246. [Google Scholar] [CrossRef]
- Jansson, L.; Larsson, J. Normal Hematopoietic Stem Cell Function in Mice with Enforced Expression of the Hippo Signaling Effector YAP1. PLoS ONE 2012, 7, e32013. [Google Scholar] [CrossRef]
- Beverdam, A.; Claxton, C.; Zhang, X.; James, G.; Harvey, K.F.; Key, B. Yap Controls Stem/Progenitor Cell Proliferation in the Mouse Postnatal Epidermis. J. Investig. Dermatol. 2013, 133, 1497–1505. [Google Scholar] [CrossRef]
- Schlegelmilch, K.; Mohseni, M.; Kirak, O.; Pruszak, J.; Rodriguez, J.R.; Zhou, D.; Kreger, B.T.; Vasioukhin, V.; Avruch, J.; Brummelkamp, T.R.; et al. Yap1 Acts Downstream of α-Catenin to Control Epidermal Proliferation. Cell 2011, 144, 782–795. [Google Scholar] [CrossRef]
- Zhang, H.; Pasolli, H.A.; Fuchs, E. Yes-associated protein (YAP) transcriptional coactivator functions in balancing growth and differentiation in skin. Proc. Natl. Acad. Sci. USA 2011, 108, 2270–2275. [Google Scholar] [CrossRef] [PubMed]
- Oren, M.; Aylon, Y. The Hippo Signaling Pathway and Cancer. EMBO Rep. 2014, 15, 642–656. [Google Scholar] [CrossRef]
- Akladios, B.; Reinoso, V.M.; Cain, J.E.; Wang, T.; Lambie, D.L.; Watkins, D.N.; Beverdam, A. Positive regulatory interactions between YAP and Hedgehog signalling in skin homeostasis and BCC development in mouse skin In Vivo. PLoS ONE 2017, 12, e0183178. [Google Scholar] [CrossRef] [PubMed]
- Totaro, A.; Castellan, M.; Battilana, G.; Zanconato, F.; Azzolin, L.; Giulitti, S.; Cordenonsi, M.; Piccolo, S. YAP/TAZ link cell mechanics to Notch signalling to control epidermal stem cell fate. Nat. Commun. 2017, 8, 15206. [Google Scholar] [CrossRef]
- Mendoza-Reinoso, V.; Beverdam, A. Epidermal YAP activity drives canonical WNT16/β-catenin signaling to promote keratinocyte proliferation in vitro and in the murine skin. Stem Cell Res. 2018, 29, 15–23. [Google Scholar] [CrossRef]
- De Sousa, N.; Rodríguez-Esteban, G.; Rojo-Laguna, J.I.; Saló, E.; Adell, T. Hippo signaling controls cell cycle and restricts cell plasticity in planarians. PLoS Biol. 2018, 16, e2002399. [Google Scholar] [CrossRef]
- He, X.C.; Zhang, J.; Tong, W.-G.; Tawfik, O.; Ross, J.; Scoville, D.H.; Tian, Q.; Zeng, X.; Wiedemann, L.M.; Mishina, Y.; et al. BMP signaling inhibits intestinal stem cell self-renewal through suppression of Wnt–β-catenin signaling. Nat. Genet. 2004, 36, 1117–1121. [Google Scholar] [CrossRef]
- Qi, Z.; Li, Y.; Zhao, B.; Xu, C.; Liu, Y.; Li, H.; Zhang, B.; Wang, X.; Yang, X.; Xie, W.; et al. BMP restricts stemness of intestinal Lgr5+ stem cells by directly suppressing their signature genes. Nat. Commun. 2017, 8, 13824. [Google Scholar] [CrossRef]
- Tian, A.; Jiang, J. Dual role of BMP signaling in the regulation of Drosophila intestinal stem cell self-renewal. Fly 2017, 11, 297–302. [Google Scholar] [CrossRef]
- Tian, A.; Jiang, J. Intestinal epithelium-derived BMP controls stem cell self-renewal in Drosophila adult midgut. eLife 2014, 3, e01857. [Google Scholar] [CrossRef]
- Ayyaz, A.; Li, H.; Jasper, H. Haemocytes control stem cell activity in the Drosophila intestine. Nat. Cell Biol. 2015, 17, 736–748. [Google Scholar] [CrossRef]
- Cai, X.T.; Li, H.; Safyan, A.; Gawlik, J.; Pyrowolakis, G.; Jasper, H. AWD regulates timed activation of BMP signaling in intestinal stem cells to maintain tissue homeostasis. Nat. Commun. 2019, 10, 1–16. [Google Scholar] [CrossRef]
- Aigner, L.; Bogdahn, U. TGF-beta in neural stem cells and in tumors of the central nervous system. Cell Tissue Res. 2008, 331, 225–241. [Google Scholar] [CrossRef]
- Falk, S.; Wurdak, H.; Ittner, L.M.; Ille, F.; Sumara, G.; Schmid, M.-T.; Draganova, K.; Lang, K.S.; Paratore, C.; Leveen, P.; et al. Brain Area-Specific Effect of TGF-β Signaling on Wnt-Dependent Neural Stem Cell Expansion. Cell Stem Cell 2008, 2, 472–483. [Google Scholar] [CrossRef]
- Pineda, J.R.; Daynac, M.; Chicheportiche, A.; Cebrian-Silla, A.; Felice, K.S.; Garcia-Verdugo, J.M.; Boussin, F.D.; Mouthon, M. Vascular-derived TGF-β increases in the stem cell niche and perturbs neurogenesis during aging and following irradiation in the adult mouse brain. EMBO Mol. Med. 2013, 5, 548–562. [Google Scholar] [CrossRef]
- Kandasamy, M.; Lehner, B.; Kraus, S.; Sander, P.R.; Marschallinger, J.; Rivera, F.J.; Trümbach, D.; Ueberham, U.; Reitsamer, H.A.; Strauss, O.; et al. TGF-beta signalling in the adult neurogenic niche promotes stem cell quiescence as well as generation of new neurons. J. Cell. Mol. Med. 2014, 18, 1444–1459. [Google Scholar] [CrossRef]
- Arnold, C.P.; Benham-Pyle, B.W.; Lange, J.J.; Wood, C.J.; Alvarado, A.S. Wnt and TGFβ coordinate growth and patterning to regulate size-dependent behaviour. Nat. Cell Biol. 2019, 572, 655–659. [Google Scholar] [CrossRef]
- Zhang, J.; Niu, C.; Ye, L.; Huang, H.; He, X.; Tong, W.-G.; Ross, J.; Haug, J.S.; Johnson, T.; Feng, J.Q.; et al. Identification of the haematopoietic stem cell niche and control of the niche size. Nat. Cell Biol. 2003, 425, 836–841. [Google Scholar] [CrossRef]
- Wilkinson, R.N.; Pouget, C.; Gering, M.; Russell, A.J.; Davies, S.G.; Kimelman, D.; Patient, R. Hedgehog and Bmp Polarize Hematopoietic Stem Cell Emergence in the Zebrafish Dorsal Aorta. Dev. Cell 2009, 16, 909–916. [Google Scholar] [CrossRef]
- Blank, U.; Karlsson, S. TGF-β signaling in the control of hematopoietic stem cells. Blood 2015, 125, 3542–3550. [Google Scholar] [CrossRef]
- Langer, J.C.; Henckaerts, E.; Orenstein, J.; Snoeck, H.-W. Quantitative Trait Analysis Reveals Transforming Growth Factor-β2 as a Positive Regulator of Early Hematopoietic Progenitor and Stem Cell Function. J. Exp. Med. 2004, 199, 5–14. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, S.; Ema, H.; Karlsson, G.; Yamaguchi, T.; Miyoshi, H.; Shioda, S.; Taketo, M.M.; Karlsson, S.; Iwama, A.; Nakauchi, H. Nonmyelinating Schwann Cells Maintain Hematopoietic Stem Cell Hibernation in the Bone Marrow Niche. Cell 2011, 147, 1146–1158. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; He, X.C.; Tong, W.-G.; Johnson, T.; Wiedemann, L.M.; Mishina, Y.; Feng, J.Q.; Li, L. Bone Morphogenetic Protein Signaling Inhibits Hair Follicle Anagen Induction by Restricting Epithelial Stem/Progenitor Cell Activation and Expansion. Stem Cells 2006, 24, 2826–2839. [Google Scholar] [CrossRef] [PubMed]
- Kobielak, K.; Stokes, N.; De La Cruz, J.; Polak, L.; Fuchs, E. Loss of a quiescent niche but not follicle stem cells in the absence of bone morphogenetic protein signaling. Proc. Natl. Acad. Sci. USA 2007, 104, 10063–10068. [Google Scholar] [CrossRef]
- Sharov, A.A.; Sharova, T.Y.; Mardaryev, A.N.; Di Vignano, A.T.; Atoyan, R.; Weiner, L.; Yang, S.; Brissette, J.L.; Dotto, G.P.; Botchkarev, V.A. Bone morphogenetic protein signaling regulates the size of hair follicles and modulates the expression of cell cycle-associated genes. Proc. Natl. Acad. Sci. USA 2006, 103, 18166–18171. [Google Scholar] [CrossRef]
- Plikus, M.V.; Mayer, J.A.; De La Cruz, D.; Baker, R.E.; Maini, P.K.; Maxson, R.; Chuong, C.-M. Cyclic dermal BMP signalling regulates stem cell activation during hair regeneration. Nat. Cell Biol. 2008, 451, 340–344. [Google Scholar] [CrossRef]
- Utani, A. Laminin alpha3 chain-derived peptide promotes keratinocyte migration and wound closure: Clustering of syndecan-4 and integrin beta1. Seikagaku. J. Jpn. Biochem. Soc. 2010, 82, 327–331. [Google Scholar]
- Oshimori, N.; Fuchs, E. Paracrine TGF-β Signaling Counterbalances BMP-Mediated Repression in Hair Follicle Stem Cell Activation. Cell Stem Cell 2012, 10, 63–75. [Google Scholar] [CrossRef]
- Liu, N.; Yin, Y.; Wang, H.; Zhou, Z.; Sheng, X.; Fu, H.; Guo, R.; Wang, H.; Yang, J.; Gong, P.; et al. Telomere dysfunction impairs epidermal stem cell specification and differentiation by disrupting BMP/pSmad/P63 signaling. PLoS Genet. 2019, 15, e1008368. [Google Scholar] [CrossRef]
- Wurtzel, O.; Oderberg, I.M.; Reddien, P.W. Planarian epidermal stem cells respond to positional cues to promote cell type diversity. Dev. Cell 2017, 40, 491–504.e5. [Google Scholar] [CrossRef]
- Van den Brink, G.R.; Bleuming, S.A.; Hardwick, J.C.H.; Schepman, B.L.; Offerhaus, G.J.; Keller, J.J.; Nielsen, C.; Gaffield, W.; Van Deventer, S.J.H.; Roberts, D.J.; et al. Indian Hedgehog is an antagonist of Wnt signaling in colonic epithelial cell differentiation. Nat. Genet. 2004, 36, 277–282. [Google Scholar] [CrossRef]
- Kosinski, C.; Stange, D.E.; Xu, C.; Chan, A.S.; Ho, C.; Yuen, S.T.; Mifflin, R.C.; Powell, D.W.; Clevers, H.; Leung, S.Y.; et al. Indian Hedgehog Regulates Intestinal Stem Cell Fate Through Epithelial−Mesenchymal Interactions During Development. Gastroenterology 2010, 139, 893–903. [Google Scholar] [CrossRef]
- Wang, L.C.; Nassir, F.; Liu, Z.-Y.; Ling, L.; Kuo, F.; Crowell, T.; Olson, D.; Davidson, N.O.; Burkly, L.C. Disruption of hedgehog signaling reveals a novel role in intestinal morphogenesis and intestinal-specific lipid metabolism in mice. Gastroenterology 2002, 122, 469–482. [Google Scholar] [CrossRef]
- Gagné-Sansfaçon, J.; Allaire, J.M.; Jones, C.; Boudreau, F.; Perreault, N. Loss of Sonic Hedgehog Leads to Alterations in Intestinal Secretory Cell Maturation and Autophagy. PLoS ONE 2014, 9, e98751. [Google Scholar] [CrossRef]
- Ramalho-Santos, M.; A Melton, D.; McMahon, A.P. Hedgehog signals regulate multiple aspects of gastrointestinal development. Development 2000, 127, 2763–2772. [Google Scholar]
- Jiang, H.; Tian, A.; Jiang, J. Intestinal stem cell response to injury: Lessons from Drosophila. Cell. Mol. Life Sci. 2016, 73, 3337–3349. [Google Scholar] [CrossRef]
- Tian, A.; Shi, Q.; Jiang, A.; Li, S.; Wang, B.; Jiang, J. Injury-stimulated Hedgehog signaling promotes regenerative proliferation of Drosophila intestinal stem cells. J. Cell Biol. 2015, 208, 807–819. [Google Scholar] [CrossRef]
- Forsthoefel, D.J.; I Cejda, N.; Khan, U.W.; A Newmark, P. Cell-type diversity and regionalized gene expression in the planarian intestine. eLife 2020, 9, 1–38. [Google Scholar] [CrossRef]
- Ahn, S.; Joyner, A.L. In Vivo analysis of quiescent adult neural stem cells responding to Sonic hedgehog. Nat. Cell Biol. 2005, 437, 894–897. [Google Scholar] [CrossRef]
- Palma, V.; Lim, D.A.; Dahmane, N.; Sánchez, P.; Brionne, T.C.; Herzberg, C.D.; Gitton, Y.; Carleton, A.; Álvarez-Buylla, A.; I Altaba, A.R. Sonic hedgehog controls stem cell behavior in the postnatal and adult brain. Development 2005, 132, 335–344. [Google Scholar] [CrossRef]
- Lai, K.; Kaspar, B.K.; Gage, F.H.; Schaffer, D.V. Sonic hedgehog regulates adult neural progenitor proliferation In Vitro and In Vivo. Nat. Neurosci. 2002, 6, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Machold, R.; Hayashi, S.; Rutlin, M.; Muzumdar, M.D.; Nery, S.; Corbin, J.G.; Gritli-Linde, A.; Dellovade, T.; Porter, J.A.; Rubin, L.L.; et al. Sonic Hedgehog Is Required for Progenitor Cell Maintenance in Telencephalic Stem Cell Niches. Neuron 2003, 39, 937–950. [Google Scholar] [CrossRef]
- Daynac, M.; Tirou, L.; Faure, H.; Mouthon, M.-A.; Gauthier, L.R.; Hahn, H.; Boussin, F.D.; Ruat, M. Hedgehog Controls Quiescence and Activation of Neural Stem Cells in the Adult Ventricular-Subventricular Zone. Stem Cell Rep. 2016, 7, 735–748. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liu, G.; Guo, T.; Liang, X.G.; Du, H.; Yang, L.; Bhaduri, A.; Li, X.; Xu, Z.; Zhang, Z.; et al. Cortical Neural Stem Cell Lineage Progression Is Regulated by Extrinsic Signaling Molecule Sonic Hedgehog. Cell Rep. 2020, 30, 4490–4504.e4. [Google Scholar] [CrossRef]
- Peeters, M.; Ottersbach, K.; Bollerot, K.; Orelio, C.; De Bruijn, M.; Wijgerde, M.; Dzierzak, E. Ventral embryonic tissues and Hedgehog proteins induce early AGM hematopoietic stem cell development. Development 2009, 136, 2613–2621. [Google Scholar] [CrossRef]
- Trowbridge, J.J.; Scott, M.P.; Bhatia, M. Hedgehog modulates cell cycle regulators in stem cells to control hematopoietic regeneration. Proc. Natl. Acad. Sci. USA 2006, 103, 14134–14139. [Google Scholar] [CrossRef]
- Gao, J.; Graves, S.; Koch, U.; Liu, S.; Jankovic, V.; Buonamici, S.; El Andaloussi, A.; Nimer, S.D.; Kee, B.L.; Taichman, R.; et al. Hedgehog Signaling Is Dispensable for Adult Hematopoietic Stem Cell Function. Cell Stem Cell 2009, 4, 548–558. [Google Scholar] [CrossRef]
- Hofmann, I.; Stover, E.H.; Cullen, D.E.; Mao, J.; Morgan, K.J.; Lee, B.H.; Kharas, M.G.; Miller, P.G.; Cornejo, M.G.; Okabe, R.; et al. Hedgehog Signaling Is Dispensable for Adult Murine Hematopoietic Stem Cell Function and Hematopoiesis. Cell Stem Cell 2009, 4, 559–567. [Google Scholar] [CrossRef]
- Çankirili, N.K.; Altundag, O.; Çelebi-Saltik, B. Skin Stem Cells, Their Niche and Tissue Engineering Approach for Skin Regeneration. Adv. Exp. Med. Biol. 2019, 1212, 107–126. [Google Scholar] [CrossRef]
- Abe, Y.; Tanaka, N. Roles of the Hedgehog Signaling Pathway in Epidermal and Hair Follicle Development, Homeostasis, and Cancer. J. Dev. Biol. 2017, 5, 12. [Google Scholar] [CrossRef]
- Paladini, R.D.; Saleh, J.; Qian, C.; Xu, G.-X.; Rubin, L.L. Modulation of Hair Growth with Small Molecule Agonists of the Hedgehog Signaling Pathway. J. Investig. Dermatol. 2005, 125, 638–646. [Google Scholar] [CrossRef]
- Pedro, M.P.; Lund, K.; Iglesias-Bartolome, R. The landscape of GPCR signaling in the regulation of epidermal stem cell fate and skin homeostasis. Stem Cells 2020, 38, 1520–1531. [Google Scholar] [CrossRef]
- Mill, P.; Mo, R.; Fu, H.; Grachtchouk, M.; Kim, P.C.; Dlugosz, A.A.; Hui, C.-C. Sonic hedgehog-dependent activation of Gli2 is essential for embryonic hair follicle development. Genes Dev. 2003, 17, 282–294. [Google Scholar] [CrossRef]
- Epstein, E.H. Basal cell carcinomas: Attack of the hedgehog. Nat. Rev. Cancer 2008, 8, 743–754. [Google Scholar] [CrossRef]
- Adolphe, C.; Narang, M.; Ellis, T.; Wicking, C.; Kaur, P.; Wainwright, B. An In Vivo comparative study of sonic, desert and Indian hedgehog reveals that hedgehog pathway activity regulates epidermal stem cell homeostasis. Development 2004, 131, 5009–5019. [Google Scholar] [CrossRef]
- Cui, S.; Chang, P.-Y. Current understanding concerning intestinal stem cells. World J. Gastroenterol. 2016, 22, 7099–7110. [Google Scholar] [CrossRef]
- Jiang, H.; Grenley, M.O.; Bravo, M.-J.; Blumhagen, R.Z.; Edgar, B.A. EGFR/Ras/MAPK Signaling Mediates Adult Midgut Epithelial Homeostasis and Regeneration in Drosophila. Cell Stem Cell 2011, 8, 84–95. [Google Scholar] [CrossRef]
- Biteau, B.; Jasper, H. EGF signaling regulates the proliferation of intestinal stem cells in Drosophila. Development 2011, 138, 1045–1055. [Google Scholar] [CrossRef]
- Kabiri, Z.; Greicius, G.; Zaribafzadeh, H.; Hemmerich, A.; Counter, C.M.; Virshup, D.M. Wnt signaling suppresses MAPK-driven proliferation of intestinal stem cells. J. Clin. Investig. 2018, 128, 3806–3812. [Google Scholar] [CrossRef]
- Park, J.-S.; Kim, Y.-S.; Yoo, M.-A. The role of p38b MAPK in age-related modulation of intestinal stem cell proliferation and differentiation in Drosophila. Aging 2009, 1, 637–651. [Google Scholar] [CrossRef]
- Barberán, S.; Fraguas, S.; Cebrià, F. The EGFR signaling pathway controls gut progenitor differentiation during planarian regeneration and homeostasis. Development 2016, 143, 2089–2102. [Google Scholar] [CrossRef] [PubMed]
- Barberán, S.; Cebrià, F. The role of the EGFR signaling pathway in stem cell differentiation during planarian regeneration and homeostasis. Semin. Cell Dev. Biol. 2019, 87, 45–57. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.-R.; Lee, J.; Kim, H.-J. Differential effects of MEK inhibitors on rat neural stem cell differentiation: Repressive roles of MEK2 in neurogenesis and induction of astrocytogenesis by PD98059. Pharmacol. Res. 2019, 149, 104466. [Google Scholar] [CrossRef] [PubMed]
- Jiang, P.; Zhu, T.; Xia, Z.; Gao, F.; Gu, W.; Chen, X.; Yuan, T.; Yu, H. Inhibition of MAPK/ERK signaling blocks hippocampal neurogenesis and impairs cognitive performance in prenatally infected neonatal rats. Eur. Arch. Psychiatry Clin. Neurosci. 2015, 265, 497–509. [Google Scholar] [CrossRef]
- Kim, K.-T.; Kim, H.-J.; Cho, D.-C.; Bae, J.-S.; Park, S.-W. Substance P stimulates proliferation of spinal neural stem cells in spinal cord injury via the mitogen-activated protein kinase signaling pathway. Spine J. 2015, 15, 2055–2065. [Google Scholar] [CrossRef]
- Liu, W.; Wu, W.; Lin, G.; Cheng, J.; Zeng, Y.; Shi, Y. Physical exercise promotes proliferation and differentiation of endogenous neural stem cells via ERK in rats with cerebral infarction. Mol. Med. Rep. 2018, 18, 1455–1464. [Google Scholar] [CrossRef]
- Tocharus, C.; Puriboriboon, Y.; Junmanee, T.; Ekthuwapranee, K.; Govitrapong, P. Melatonin enhances adult rat hippocampal progenitor cell proliferation via ERK signaling pathway through melatonin receptor. Neuroscience 2014, 275, 314–321. [Google Scholar] [CrossRef]
- Chen, X.; Wu, H.; Chen, H.; Wang, Q.; Xie, X.-J.; Shen, J. Astragaloside VI Promotes Neural Stem Cell Proliferation and Enhances Neurological Function Recovery in Transient Cerebral Ischemic Injury via Activating EGFR/MAPK Signaling Cascades. Mol. Neurobiol. 2018, 56, 3053–3067. [Google Scholar] [CrossRef]
- Lao, C.L.; Lu, C.-S.; Chen, J.-C. Dopamine D3receptor activation promotes neural stem/progenitor cell proliferation through AKT and ERK1/2 pathways and expands type-B and -C cells in adult subventricular zone. Glia 2013, 61, 475–489. [Google Scholar] [CrossRef]
- Shivaraj, M.C.; Marcy, G.; Low, G.; Ryu, J.R.; Zhao, X.; Rosales, F.J.; Goh, E.L.K. Taurine Induces Proliferation of Neural Stem Cells and Synapse Development in the Developing Mouse Brain. PLoS ONE 2012, 7, e42935. [Google Scholar] [CrossRef]
- Hamanoue, M.; Morioka, K.; Ohsawa, I.; Ohsawa, K.; Kobayashi, M.; Tsuburaya, K.; Akasaka, Y.; Mikami, T.; Ogata, T.; Takamatsu, K. Cell-permeable p38 MAP kinase promotes migration of adult neural stem/progenitor cells. Sci. Rep. 2016, 6, 24279. [Google Scholar] [CrossRef]
- Moors, M.; E Cline, J.; Abel, J.; Fritsche, E. ERK-dependent and -independent pathways trigger human neural progenitor cell migration. Toxicol. Appl. Pharmacol. 2007, 221, 57–67. [Google Scholar] [CrossRef]
- Li, X.; Newbern, J.M.; Wu, Y.; Morgan-Smith, M.; Zhong, J.; Charron, J.; Snider, W.D. MEK Is a Key Regulator of Gliogenesis in the Developing Brain. Neuron 2012, 75, 1035–1050. [Google Scholar] [CrossRef]
- Bajetto, A.; Barbero, S.; Bonavia, R.; Piccioli, P.; Pirani, P.; Florio, T.; Schettini, G. Stromal cell-derived factor-1α induces astrocyte proliferation through the activation of extracellular signal-regulated kinases 1/2 pathway. J. Neurochem. 2001, 77, 1226–1236. [Google Scholar] [CrossRef]
- Fraguas, S.; Barberán, S.; Iglesias, M.; Rodríguez-Esteban, G.; Cebrià, F. egr-4, a target of EGFR signaling, is required for the formation of the brain primordia and head regeneration in planarians. Development 2014, 141, 1835–1847. [Google Scholar] [CrossRef]
- Tasaki, J.; Shibata, N.; Nishimura, O.; Itomi, K.; Tabata, Y.; Son, F.; Suzuki, N.; Araki, R.; Abe, M.; Agata, K.; et al. ERK signaling controls blastema cell differentiation during planarian regeneration. Development 2011, 138, 2417–2427. [Google Scholar] [CrossRef]
- Lei, K.; Vu, H.T.-K.; Mohan, R.D.; McKinney, S.A.; Seidel, C.W.; Alexander, R.; Gotting, K.; Workman, J.L.; Alvarado, A.S. Egf Signaling Directs Neoblast Repopulation by Regulating Asymmetric Cell Division in Planarians. Dev. Cell 2016, 38, 413–429. [Google Scholar] [CrossRef]
- Cebrià, F.; Kobayashi, C.; Umesono, Y.; Nakazawa, M.; Mineta, K.; Ikeo, K.; Gojobori, T.; Itoh, M.; Taira, M.; Alvarado, A.S.; et al. FGFR-related gene nou-darake restricts brain tissues to the head region of planarians. Nature 2002, 419, 620–624. [Google Scholar] [CrossRef]
- Platanias, L.C. Map kinase signaling pathways and hematologic malignancies. Blood 2003, 101, 4667–4679. [Google Scholar] [CrossRef]
- Chung, E.; Kondo, M. Role of Ras/Raf/MEK/ERK signaling in physiological hematopoiesis and leukemia development. Immunol. Res. 2010, 49, 248–268. [Google Scholar] [CrossRef]
- Wandzioch, E.; Edling, C.E.; Palmer, R.H.; Carlsson, L.; Hallberg, B. Activation of the MAP kinase pathway by c-Kit is PI-3 kinase dependent in hematopoietic progenitor/stem cell lines. Blood 2004, 104, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Chan, G.; Gu, S.; Neel, B.G. Erk1 and Erk2 are required for maintenance of hematopoietic stem cells and adult hematopoiesis. Blood 2013, 121, 3594–3598. [Google Scholar] [CrossRef] [PubMed]
- Ito, K.; Hirao, A.; Arai, F.; Takubo, K.; Matsuoka, S.; Miyamoto, K.; Ohmura, M.; Naka, K.; Hosokawa, K.; Ikeda, Y.; et al. Reactive oxygen species act through p38 MAPK to limit the lifespan of hematopoietic stem cells. Nat. Med. 2006, 12, 446–451. [Google Scholar] [CrossRef] [PubMed]
- Khavari, T.A.; Rinn, J.L. Ras/Erk MAPK Signaling in Epidermal Homeostasis and Neoplasia. Cell Cycle 2007, 6, 2928–2931. [Google Scholar] [CrossRef] [PubMed]
- Doma, E.; Rupp, C.; Baccarini, M. EGFR-Ras-Raf Signaling in Epidermal Stem Cells: Roles in Hair Follicle Development, Regeneration, Tissue Remodeling and Epidermal Cancers. Int. J. Mol. Sci. 2013, 14, 19361–19384. [Google Scholar] [CrossRef]
- Scholl, F.A.; Dumesic, P.A.; Khavari, P.A. Mek1 Alters Epidermal Growth and Differentiation. Cancer Res. 2004, 64, 6035–6040. [Google Scholar] [CrossRef]
- Tarutani, M.; Cai, T.; Dajee, M.; A Khavari, P. Inducible activation of Ras and Raf in adult epidermis. Cancer Res. 2003, 63, 319–323. [Google Scholar]
- Hiratsuka, T.; Bordeu, I.; Pruessner, G.; Watt, F.M. Regulation of ERK basal and pulsatile activity control proliferation and exit from the stem cell compartment in mammalian epidermis. Proc. Natl. Acad. Sci. USA 2020, 117, 17796–17807. [Google Scholar] [CrossRef]
- Chou, Y.-T.; Lai, F.-J.; Chang, N.-S.; Hsu, L.-J. Wwox Deficiency Causes Downregulation of Prosurvival ERK Signaling and Abnormal Homeostatic Responses in Mouse Skin. Front. Cell Dev. Biol. 2020, 8, 558432. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krishnan, M.; Kumar, S.; Kangale, L.J.; Ghigo, E.; Abnave, P. The Act of Controlling Adult Stem Cell Dynamics: Insights from Animal Models. Biomolecules 2021, 11, 667. https://doi.org/10.3390/biom11050667
Krishnan M, Kumar S, Kangale LJ, Ghigo E, Abnave P. The Act of Controlling Adult Stem Cell Dynamics: Insights from Animal Models. Biomolecules. 2021; 11(5):667. https://doi.org/10.3390/biom11050667
Chicago/Turabian StyleKrishnan, Meera, Sahil Kumar, Luis Johnson Kangale, Eric Ghigo, and Prasad Abnave. 2021. "The Act of Controlling Adult Stem Cell Dynamics: Insights from Animal Models" Biomolecules 11, no. 5: 667. https://doi.org/10.3390/biom11050667
APA StyleKrishnan, M., Kumar, S., Kangale, L. J., Ghigo, E., & Abnave, P. (2021). The Act of Controlling Adult Stem Cell Dynamics: Insights from Animal Models. Biomolecules, 11(5), 667. https://doi.org/10.3390/biom11050667






