Abstract
Exercise can ameliorate cardiovascular dysfunctions in the diabetes condition, but its precise molecular mechanisms have not been entirely understood. The aim of the present study was to determine the impact of endurance training on expression of angiogenesis-related genes in cardiac tissue of diabetic rats. Thirty adults male Wistar rats were randomly divided into three groups (N = 10) including diabetic training (DT), sedentary diabetes (SD), and sedentary healthy (SH), in which diabetes was induced by a single dose of streptozotocin (50 mg/kg). Endurance training (ET) with moderate-intensity was performed on a motorized treadmill for six weeks. Training duration and treadmill speed were increased during five weeks, but they were kept constant at the final week, and slope was zero at all stages. Real-time polymerase chain reaction (RT-PCR) analysis was used to measure the expression of myocyte enhancer factor-2C (MEF2C), histone deacetylase-4 (HDAC4) and Calmodulin-dependent protein kinase II (CaMKII) in cardiac tissues of the rats. Our results demonstrated that six weeks of ET increased gene expression of MEF2C significantly (p < 0.05), and caused a significant reduction in HDAC4 and CaMKII gene expression in the DT rats compared to the SD rats (p < 0.05). We concluded that moderate-intensity ET could play a critical role in ameliorating cardiovascular dysfunction in a diabetes condition by regulating the expression of some angiogenesis-related genes in cardiac tissues.
1. Introduction
Diabetes Mellitus (DM), with an estimated worldwide prevalence of 285 million patients, is an increasingly prevalent metabolic disorder [1] that is characterized by persistent hyperglycemia caused by insulin resistance or a lack of insulin [2,3]. It has been documented that patients with DM are at a higher risk of central and peripheral cardiovascular diseases, because DM harms structure and function of micro vessels [4,5]. Among the others, abnormal angiogenesis and collateral vessel formation are the most prevalent cardiovascular manifestations seen in DM patients [6,7].
Angiogenesis, as a multilevel and complex process of a new capillary formation, is controlled by angiostatic and angiogenic factors [8,9]. In the recent years, researchers found new players in the angiogenesis process that are regulated by a complex network of transcriptional factors including myocyte enhancer factor-2C (MEF2C), histone deacetylase-4 (HDAC4), and Calmodulin-dependent protein kinase II (CaMKII) [10]. MEF2C is a direct transcriptional target of endothelial transcription factors that play an important role in angiogenesis and vasculogenesis during vascular development [11]. Previous studies have revealed that MEF2C was significantly down-regulated in cardiac tissue of diabetic rats [12,13]. HDACs control biological processes by de-acetylation of histone and regulating accessibility of transcription factors to the gene promoter [14]. Among all HDACs, HDAC4 plays an important role in mediating cardiovascular diseases [15]. This transcription factor is phosphorylated and activated by CaMKII, a central culprit in the development of heart failure and cardiac arrhythmia [16], and negatively interacts with MEF2C to control its repressive activity [17]. The activities of HDAC4 and CaMKII (representatives of histone acetylation) are increased in the diabetic condition [18,19].
More recently, researchers have introduced testosterone as another influencing factor on angiogenesis mainly in cardiovascular system of diabetic patients [20]. Some studies have demonstrated that androgens paucity are common in men with diabetes, and testosterone deficiency may modulate endothelial angiogenesis [21,22]. The other component of diabetics is insulin resistance, which is also associated with an increased risk of the premature development of coronary artery disease. Therefore, it seems that the combination of testosterone deficiency and impaired glucose tolerance increases the risk of cardiovascular disease in patients with DM [21,22]. The recent studies have demonstrated that physiological testosterone therapy could improve insulin resistance [23].
Sedentary lifestyle and poor diet are common in patients with DM that worsen the patient’s condition [24]. Lifestyle modifiers as well as medication are considered as helpful approaches to tackle this problem. As a lifestyle modifier, exercise training could play an inevitable role in glycemic control [25,26,27,28]. Endurance training (ET), as the most popular type of exercise training, is considered as the most effective for DM patients [29,30]. There are also no reports on ET side effects in DM patients, highlighting its safety for DM treatment [27,28,31,32]. In addition, it has been suggested that ET could lead to vascular modifications associated with capillary density and angiogenesis [3,33,34,35] and result in cardiac remolding. For example, Ardakanizade et al. [36] examined the effects of long-term and mid-term ET on angiogenesis and reported higher gene expression of vascular endothelial growth factor B (VEGF-B), MEF2C, and matrix metalloproteinase-2 (MMP-2), and lower gene expression of HDAC4 and ANGPT-1 in the long- than mid-term ET.
Although exercise training and DM result in cardiac remodeling [34], the effect of moderate-intensity ET on expression of MEF2C, HDAC4, and CaMKII has not been entirely understood in DM. Therefore, our study was aimed to indicate whether a moderate-intensity ET can change the gene expression of MEF2C, HDAC4, and CaMKII in the cardiac tissue of diabetic rats.
2. Materials and Methods
2.1. Animal Models and Ethical Statement
The present study was in accordance with the guidelines for the care and use of laboratory animals approved by the Ethics Committee on the use of animal of Ardabil University of Medical Sciences (IR.ARUMS.REC.1398.251). In this experimental study, 30 male Wistar rats (249 ± 8.3 g) were obtained from the Iran Pasteur Institute (Tehran, Iran). Animals were kept under controlled in Plexiglas cages with a stable temperature of 23 ± 5 °C and humidity of 35 ± 5% on a cycle of 12-h light/dark. All animals had free access to standard food and water throughout the study with no difference in accessibility. Rats were randomly divided into three groups: Diabetic training (DT), sedentary diabetic (SD), and sedentary healthy (SH). Familiarization to treadmill was conducted for two weeks at the speed of ten meters per mins for 10–15 min, five days a week in all groups. At the end of familiarization and following an overnight fast, diabetes was induced to DT and SD groups by single intraperitoneal injection of Streptozotocin (STZ) at a dosage of 50 mg/kg (Sigma, St. Louis MO, USA). STZ was prepared in a fresh citrate buffer (0.5 M with pH 4.5), as described previously [37]. The same volume of citrate solution was injected into the SH group to simulate the stress of injection. Serum glucose level was measured 72 h after STZ injection using a portable glucometer (Roche Diagnostics K.K., Tokyo, Japan). Serum glucose higher than 250 mg/dL was considered as the benchmark to identify the diabetic rats [38], which was met by all rats in DT and SD groups. To eliminate the effects of food consumption, all blood samples were taken after 12-h of fasting. While the DT group rats conducted six-week ET, the SD and SH groups did not participate in exercise training program during the experiment period.
2.2. ET Protocol
Animals in the ET group performed an exercise protocol five days a week, for six weeks as shown in Table 1. Before and after each exercise training session, three-min warm-up and cool-down were carried out, respectively, and the treadmill slope was zero at all stages. Treadmill speed and training duration were kept constant during the final week (sixth week) to conserve the adaptations that resulted from 6 weeks ET [39]. All training sessions were conducted between 08:00–12:00 AM.

Table 1.
Endurance training protocol in different weeks.
2.3. Biochemical Assays
Twenty-four hours after the last ET session, all animals were sacrificed by intraperitoneal injection of ketamine (75 mg/kg) and xylazine (5 mg/kg) following a 12-h fasting to measure testosterone concentration, and blood samples were taken from the animals’ heart and centrifuged for 15 min at 3000 rpm to obtain serum. Serums were kept at −20 °C until analysis. Testosterone concentration was measured using an ELISA kit (Monobind, Accubind, Costa Mesa, CA, USA) in a multiple ELISA reader (Bio Tek, Winooski, VT, USA) based on the recommended protocol by the manufacturer.
2.4. qRT-PCR Analysis
Cardiac tissues were removed, submerged in liquid nitrogen, and kept at −70 °C until further analysis. The extraction of RNAs was performed by RNXTM reagent according to the manufacturer’s procedure (Sina Clon Bioscience, Tehran, Iran). Concentration of RNA, and its purity were calculated by measuring the ratio of 260/280 nm optical density using Nanodrop spectrophotometry (Eppendorf, cologne, Germany), and values between 1.8–2 were defined as an acceptable purity. The cDNA synthesis was performed using qPCRTM Green Master Kit for SYBR Green I® (Yekta Tajhiz, Tehran, Iran) according to the instructions of the manufacturer. Real-time PCR was performed in Roche Light-Cycler detection system (Basel, Switzerland) with the following steps: Initial denaturation for 5 min at 95 °C and 45 cycles of denaturation for 15 s at 95 °C, annealing for 30 s at 60 °C, an extension for 20 s at 72 °C followed by melt curve analysis (50–99 °C) [40]. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as the reference gene to measure relative gene expression. The results were evaluated by using 2−ΔΔCt comparative method and Light Cycler SW1.1 software. The sequence of the primers is shown in Table 2.

Table 2.
The sequence of primers for quantitative real-time polymerase chain reaction (RT-PCR).
2.5. Statistical Analysis
Data were analyzed using Statistical Package for Social Sciences (SPSS) version 23. Shapiro–Wilk normality test and one-way ANOVA were used to determine the normal distribution of variables and to compare changes between three groups, respectively. Tukey was also used as a post hoc test. The significance level was set at p < 0.05.
3. Results
Blood glucose and body weight changes are shown in Figure 1. Blood glucose levels increased significantly in the second week compared to before exercise in DT and SD groups, and this increase continued until the sixth week (p < 0.001). In addition, blood glucose levels were significantly reduced in the DT group compared to the SD group at the sixth week (p < 0.001). There was no significant change in blood glucose levels during the experimental period in the SH group (Figure 1A). STZ-treated animals (SD and DT groups) showed a decrease in body weight compared to the SH group in the fourth and sixth weeks (p < 0.005). DT group showed higher weights than SD in the fourth and sixth weeks, but this difference was not significant (Figure 1B).
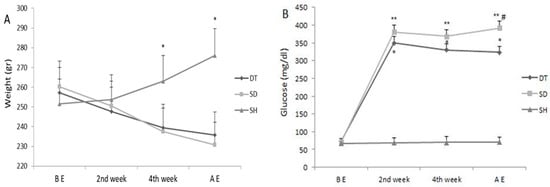
Figure 1.
(A) Weight change during and after ET. *; Significant difference between the SH group with DT and SD groups in 4th week and AE. (B) Changes in the serum levels of glucose during six weeks ET period. *; significant difference (p < 0.001) between DT and SH groups in 2nd week, 4th week, and AE with SH group.**; significant difference (p < 0.001) between SD and SH groups in 2nd week, 4th week, and AE with SH group #; significant difference (p < 0.001) between DT and SD groups AE. Abbreviations; ET: Endurance training, BE: Before Exercise, AE: After exercise, DT: Diabetic training, SD: Sedentary diabetic, SH: Sedentary healthy. Data are expressed as mean ± SEM.
As displayed in Figure 2, the SD group showed a significantly lower testosterone levels compared to the DT and SH groups (p < 0.001). In addition, the DT group showed significantly lower testosterone levels compared to the SH group (p < 0.001) (Figure 2).
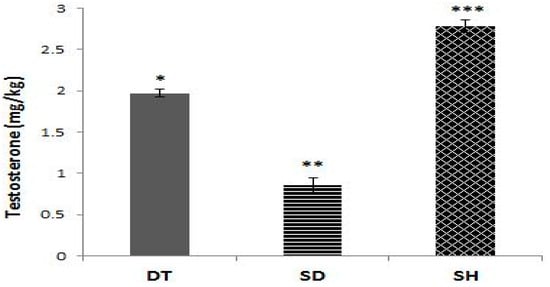
Figure 2.
Different serum levels of testosterone in the DT, SD, and SH groups after six weeks of ET. *; significant difference (p < 0.001) between DT and SD groups, **; significant difference (p < 0.001) between SD and SH groups, ***; significant difference (p < 0.001) between DT and SH groups. Abbreviations; DT: Diabetic training, SD: Sedentary diabetic, SH: Sedentary healthy, ET: Endurance training. Data are expressed as mean ± SEM.
The results of angiogenesis-related genes expression in cardiac tissues are shown in Figure 3A–C. MEF2C gene expression was higher in DT group compared to the SD group (p < 0.001), but it is lowered compared to the SH group significantly (p < 0.001) (Figure 3A). A significant difference in HDAC4 and CaMKII was observed in the DT group compared to the SD group (p < 0.001). Gene expression levels of HDAC4 and CaMKII were higher in both diabetic groups compared to the SH group (Figure 3B,C).
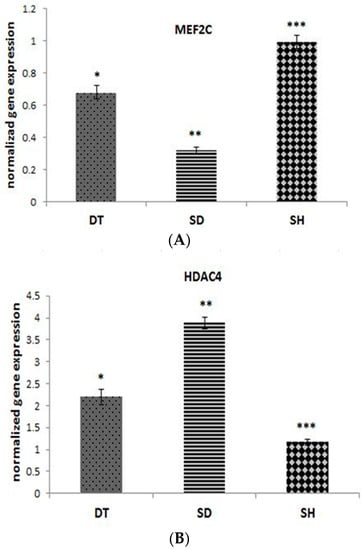
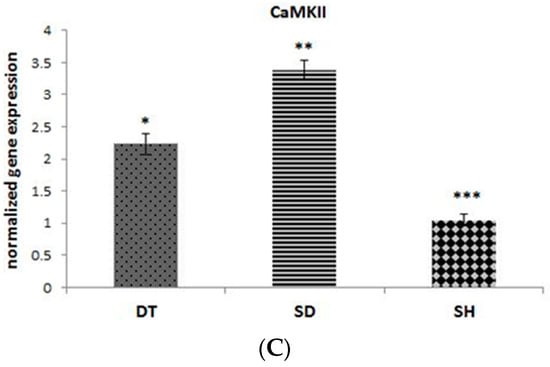
Figure 3.
The normalized gene expression of MEF2C (A), HDAC4 (B), and CaMKII (C) at the end of six weeks ET in DT, SD, and SH groups. *; significant difference p < 0.001 between DT and SD groups, **; significant difference (p < 0.001) between SD and SH groups, ***; significant difference (p < 0.001) between DT and SH groups. Abbreviations; ET: Endurance training, DT: Diabetic training, SD: Sedentary diabetic, SH: Sedentary healthy. Data are expressed as mean ± SEM.
4. Discussion
This is the first study that examined the effect of moderate-intensity ET on MEF2C, HDAC4, and CaMKII gene expression and testosterone levels in diabetic hearts. It has been shown that exercise training improves glucose control and could affect both the structure and function of the myocardium, which could improve cardiovascular health in DM patients [41]. The results of the current study show significant weight loss in diabetic groups due to frequent urination, dehydration, and skeletal muscle atrophy. Researchers showed that exposure to high levels of glucose resulted in expression of muscle atrophy–related genes like Atrogin1 and Murf1 [20]. It should be noted that weight loss was lower in the DT group, because exercise training can stimulate muscle hypertrophy and inhibit muscle atrophy [42]. In the SH group, the weight gain process occurred naturally due to sufficient calorie intake. In addition, our results showed that ET controlled serum levels of glucose, increased MEF2C, and decreased HDAC4 and CaMKII gene expression and rose serum testosterone levels in diabetic rats. A study by Grossmann et al. [42] showed that testosterone levels are lower in STZ-induced diabetic rats compared to non-diabetes. Lower testosterone was also documented in diabetic men compared to non-diabetics [43,44]. Changes in the serum levels of testosterone could improve insulin sensitivity and oxidative capacity, as well as trigger anti-inflammatory processes and capillarization [45]. It has also been suggested that the regulatory effects of exercise on glucose metabolism and angiogenesis genes expression are facilitated by increased testosterone levels [46], which is in line with our results. Testosterone could probably increase expression of its target genes (Spred-1 and PI3KR2), which stimulate proliferation of vascular cells that are required for vessel angiogenesis [40]. In fact, testosterone is the principal masculine gonadal androgen hormone that modulates angiogenesis and endothelial functions [40]. While testosterone deficiency is predominant in STZ-induced diabetic rats, it seems that exercise constitutes this deficiency, leading to increased angiogenesis genes expression.
To elucidate the effect of ET on the cardiac angiogenesis process, we studied gene expression of MEF2C, HDAC4, and CaMKII in the diabetic heart, and our results showed a higher gene expression of MEF2C and lower gene expression of HDAC4 and CaMKII in DT than another diabetic group. It seems that down-regulation and deacetylation of HDAC4 allowing MEF2C to activate angiogenesis process [47,48]. We believe that lower expression of HDAC4 in the DT group is accompanied by higher expression of the MEF2C. These results are in line with the previous research findings on the role of HDAC4 in the angiogenesis process [47,48]. On the other hand, ET induced an increase in the antioxidant potential, which could be another explanation for changes in MEF2C and HDAC4 gene expression because it has been reported that oxidative stress, as a novel phosphorylation-independent post-translational modification, regulates subcellular localization of MEF2C in cardiomyocytes [49]. It has been indicated that increased MEF2C gene expression can up-regulate vascular endothelial growth factor (VEGF)-B, which is a key regulator of angiogenesis [11]. Although the amount of VEGF-B has not been measured in the current study, due to financial limitations, based on the previous studies [36], we hypothesize that ET could increase the gene expression of VEGF-B. It is observed that HDAC4 down-regulation increases angiogenesis through stimulation of VEGF-B gene expression [50], and it has also been reported in the cerebral ischemia that higher expression of the HIF-VEGF signaling gene has be seen through the phosphorylation of the HDAC4 protein [51].
Another finding of the present study was down-regulation of CaMKII gene expression by ET, which is in accordance with other studies [52,53]. Stolen et al. [53] has shown that in diabetic rats with reduced cardiomyocyte contractile function, Ca2+ handling and chronically increased cardiac CaMKII aerobic interval exercise training resulted in reduced CaMKII levels. It has been demonstrated that CaMKII is capable of regulating the angiogenesis factors [19], and stimulates glucose uptake, sarcolemma ion fluxes, energy production, sarcoplasmic reticulum Ca2+ release/reuptake, and myocyte contraction/relaxation during acute activation [54]. Contrary to physiological condition, in a disease condition such as diabetes, CaMKII leads to mitochondrial dysfunction, cell fibrosis, remodeling of ion channel, inefficient substrate utilization, impaired intracellular Ca2+ handling, inflammation, and a contractile dysfunction leading to increased risk of arrhythmias [55]. CaMKII activity was up-regulated in the heart of diabetic rats in our study. CaMKII modulates transcription of HDAC4 at multiple levels [56], as we see higher HDAC4 gene expression in diabetic rats but both of them were controlled by ET. Therefore, ET-induced decrease in CaMKII gene expression may leads to decrease of HDAC4 as well. It seems that inhibition of CaMKII expression could possibly be a therapeutic strategy for DM by remodeling and promote angiogenesis in cardiac tissue. Beyond, it is possible that a decrease in CaMKII gene expression attenuates HDAC4 gene expression [56], but paradoxically lead to a greater increase in MEF2C. Thus, we provide support for the hypothesis that MEF2C regulation is under the control of HDAC4 and CaMKII during the regulatory adaptation to moderate-intensity ET in diabetic myocardium. However, more studies is needed to prove these results and shed light on the exact it’s signaling pathways.
5. Conclusions
Taken together, the results of this study indicated that six weeks of moderate-intensity ET allowed more effective control of glucose homeostasis, increased testosterone levels, and induced up-regulation of MEF2C and down-regulation of HDAC4 and CaMKII in cardiac tissue of diabetic rats. These results suggest improvements in managing the diabetic-induced cardiac dysfunction. However, future studies should cover our limitation by analyzing angiogenesis markers as well.
Author Contributions
Conceptualization, M.K., L.B., M.S.; methodology, M.K. and M.R.; software, validation and formal analysis, M.T. and M.R.; investigation and resources, K.K.; data curation, K.S.; writing—original draft preparation, M.K.; writing—review and editing, M.R. and K.K.; supervision and project administration, L.B. and M.S.; All authors have read and agreed to the published version of the manuscript.
Funding
This study is funded by the University of Mohaghegh Ardabili.
Institutional Review Board Statement
This study was approved by the Ethics Committee on the use of animal of Ardabil University of Medical Sciences (IR.ARUMS.REC.1398.251).
Informed Consent Statement
Not applicable.
Data Availability Statement
All data related to this study is available at https://uma.ac.ir/index.php?slc_lang=en.
Acknowledgments
This research is a result of Mojdeh Khajehlandi Ph.D. thesis conducted at University of Mohaghegh Ardabili, and thus we would like to thank this university for financial help. Also, we would like to thank Shahid Chamran University of Ahvaz for its cooperation in this research.
Conflicts of Interest
There is no conflict of interest.
References
- Shaw, J.; Sicree, R.; Zimmet, P. Global estimates of the prevalence of diabetes for 2010 and 2030. Diabetes Res. Clin. Prac. 2010, 87, 4–14. [Google Scholar] [CrossRef]
- Berlanga-Acosta, J.; Mendoza-Marí, Y.; Rodríguez-Rodríguez, N.; Herrera, D.G.D.B.; García-Ojalvo, A.; Fernández-Mayola, M.; Guillén-Nieto, G.; Valdés-Sosa, P.A. Burn injury insulin resistance and central nervous system complications: A review. Burn. Open 2020, 4, 41–52. [Google Scholar] [CrossRef]
- Zarich, S.W. Treating the diabetic patient: Appropriate care for glycemic control and cardiovascular disease risk factors. Rev. Cardiovasc. Med. 2003, 4, 19–28. [Google Scholar]
- Costa, P.Z.; Soares, R. Neovascularization in diabetes and its complications. Unraveling the angiogenic paradox. Life Sci. 2013, 92, 1037–1045. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, M.Z.; Yeasmin, F.; Rabi, D.M.; Ronksley, P.E.; Turin, T.C. Prognostic tools for cardiovascular disease in patients with type 2 diabetes: A systematic review and meta-analysis of C-statistics. J. Diabetes Complicat. 2019, 33, 98–111. [Google Scholar] [CrossRef] [PubMed]
- Hou, R.; Shen, M.; Wang, R.; Liu, H.; Gao, C.; Xu, J.; Tao, L.; Yin, Z.; Yin, T. Thioredoxin1 Inactivation Mediates the Impairment of Ischemia-Induced Angiogenesis and Further Injury in Diabetic Myocardium. J. Vasc. Res. 2020, 57, 76–85. [Google Scholar] [CrossRef]
- Abacı, A.; Oguzhan, A.; Kahraman, S.; Eryol, N.K.; Unal, S.; Arınc, H.; Ergin, A. Effect of Diabetes Mellitus on Formation of Coronary Collateral Vessels. Circulation 1999, 99, 2239–2242. [Google Scholar] [CrossRef] [PubMed]
- Carmeliet, P. Angiogenesis in health and disease. Nat. Med. 2003, 9, 653–660. [Google Scholar] [CrossRef]
- Jośko, J.; Mazurek, M. Transcription factors having impact on vascular endothelial growth factor (VEGF) gene expression in angiogenesis. Med Sci. Monit. 2004, 10, 89. [Google Scholar]
- Hamik, A.; Wang, B.; Jain, M.K. Transcriptional Regulators of Angiogenesis. Arter. Thromb. Vasc. Biol. 2006, 26, 1936–1947. [Google Scholar] [CrossRef] [PubMed]
- Maiti, D.; Xu, Z.; Duh, E.J. Vascular endothelial growth factor induces MEF2C and MEF2-dependent activity in endothe-lial cells. Investig. Ophthalmol. Vis. Sci. 2008, 49, 3640–3648. [Google Scholar] [CrossRef] [PubMed]
- Razeghi, P.; Young, M.E.; Cockrill, T.C.; Frazier, O.H.; Taegtmeyer, H. Downregulation of myocardial myocyte enhancer factor 2C and myocyte enhancer factor 2C-regulated gene expression in diabetic patients with nonischemic heart failure. Circulation 2002, 106, 407–411. [Google Scholar] [CrossRef] [PubMed]
- Boudina, S.; Abel, E.D. Diabetic cardiomyopathy, causes and effects. Rev. Endocr. Metab. Disord. 2010, 11, 31–39. [Google Scholar] [CrossRef]
- Ruijter, A.J.D.; Gennip, A.H.V.; Caron, H.N.; Kemp, S.; Kuilenburg, A.B.V. Histone deacetylases (HDACs): Characterization of the classical HDAC family. Biochem. J. 2003, 370, 737–749. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Xiao, C.; Long, F.; Su, Z.; Jia, W.; Qin, M.; Huang, M.; Wu, W.; Suguro, R.; Liu, X.; et al. HDAC4 regulates vascular inflammation via activation of autophagy. Cardiovasc. Res. 2018, 114, 1016–1028. [Google Scholar] [CrossRef] [PubMed]
- Cuello, F.; Lorenz, K. Inhibition of cardiac CaMKII to cure heart failure: Step by step towards translation? Basic Res. Cardiol. 2016, 111, 1–4. [Google Scholar] [CrossRef]
- Backs, J.; Backs, T.; Bezprozvannaya, S.; McKinsey, T.A.; Olson, E.N. Histone Deacetylase 5 Acquires Calcium/Calmodulin-Dependent Kinase II Responsiveness by Oligomerization with Histone Deacetylase 4. Mol. Cell. Biol. 2008, 28, 3437–3445. [Google Scholar] [CrossRef]
- Chen, Y.; Du, J.; Zhao, Y.T.; Zhang, L.; Lv, G.; Zhuang, S.; Qin, G.; Zhao, T.C. Histone deacetylase (HDAC) inhibition improves myocardial function and prevents cardiac remodeling in diabetic mice. Cardiovasc. Diabetol. 2015, 14, 99. [Google Scholar] [CrossRef] [PubMed]
- Mollova, M.Y.; Katus, H.A.; Ebacks, J. Regulation of CaMKII signaling in cardiovascular disease. Front. Pharmacol. 2015, 6, 178. [Google Scholar] [CrossRef]
- Hirata, Y.; Nomura, K.; Senga, Y.; Okada, Y.; Kobayashi, K.; Okamoto, S.; Minokoshi, Y.; Imamura, M.; Takeda, S.; Hosooka, T.; et al. Hyperglycemia induces skeletal muscle atrophy via a WWP1/KLF15 axis. JCI Insight 2019, 4, 1–12. [Google Scholar] [CrossRef]
- Death, A.K.; McGrath, K.C.; Sader, M.A.; Nakhla, S.; Jessup, W.; Handelsman, D.J.; Celermajer, D.S. Dihydrotestosterone promotes vascular cell adhesion molecule-1 expression in male human endothelial cells via a nuclear factor-κB-dependent pathway. Endocrinology 2004, 145, 1889–1897. [Google Scholar] [CrossRef]
- Ng, M.K.; Quinn, C.M.; McCrohon, J.A.; Nakhla, S.; Jessup, W.; Handelsman, D.J.; Celermajer, D.S.; Death, A.K. Androgens up-regulate atherosclerosis-related genes in macrophages from males but not females: Molecular insights into gender differences in atherosclerosis. J. Am. Coll. Cardiol. 2003, 42, 1306–1313. [Google Scholar] [CrossRef]
- Kapoor, D.; Goodwin, E.; Channer, K.S.; Jones, T.H. Testosterone replacement therapy improves insulin resistance, glycaemic control, visceral adiposity and hypercholesterolemia in hypogonadal men with type 2 diabetes. Eur. J. Endocrinol. 2006, 154, 899–906. [Google Scholar] [CrossRef]
- Sami, W.; Ansari, T.; Butt, N.S.; Ab Hamid, M.R. Effect of diet on type 2 diabetes mellitus: A review. Int. J. Health Sci. 2017, 11, 65–71. [Google Scholar]
- Booth, F.W.; Roberts, C.K.; Thyfault, J.P.; Ruegsegger, G.N.; Toedebusch, R.G. Role of Inactivity in Chronic Diseases: Evolutionary Insight and Pathophysiological Mechanisms. Physiol. Rev. 2017, 97, 1351–1402. [Google Scholar] [CrossRef] [PubMed]
- Hambrecht, R.; Wolf, A.; Gielen, S.; Linke, A.; Hofer, J.; Erbs, S.; Schoene, N.; Schuler, G. Effect of Exercise on Coronary Endothelial Function in Patients with Coronary Artery Disease. N. Engl. J. Med. 2000, 342, 454–460. [Google Scholar] [CrossRef]
- Pedersen, L.R.; Olsen, R.H.; Anholm, C.; Astrup, A.; Eugen-Olsen, J.; Fenger, M.; Simonsen, L.; Walzem, R.L.; Haugaard, S.B.; Prescott, E. Effects of 1 year of exercise training versus combined exercise training and weight loss on body compo-sition, low-grade inflammation and lipids in overweight patients with coronary artery disease: A randomized trial. Cardio-Vasc. Diabetol. 2019, 18, 127. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Tan, S.; Wang, Z.; Guo, Z.; Li, Q.; Wang, J. Aerobic exercise training at maximal fat oxidation intensity improves body composition, glycemic control, and physical capacity in older people with type 2 diabetes. J. Exerc. Sci. Fit. 2020, 18, 7–13. [Google Scholar] [CrossRef]
- Yakasai, A.M.; Maharaj, S.S.; Nuhu, J.M. Moderate intensity endurance exercise on responses of relative cardiovascular parameters of primary and secondary hypertensive patients: Protocol for a randomized controlled trials. Eur. J. Physiother. 2019, 21, 1–6. [Google Scholar] [CrossRef]
- Venables, M.C.; Jeukendrup, A.E. Endurance training and obesity: Effect on substrate metabolism and insulin sensitivity. Med. Sci. Sports Exerc. 2008, 40, 495–502. [Google Scholar] [CrossRef]
- Colberg, S.R.; Niederseer, D. Impact of Exercise on Cardiovascular Risk Factors: Diabetes Mellitus. In Textbook of Sports and Exercise Cardiology; Metzler, J.B., Ed.; Springer: New York, NY, USA, 2020; pp. 769–792. [Google Scholar]
- Crisafulli, A.; Pagliaro, P.; Roberto, S.; Cugusi, L.; Mercuro, G.; Lazou, A.; Beauloye, C.; Bertrand, L.; Hausenloy, D.J.; Aragno, M.; et al. Diabetic cardiomyopathy and ischemic heart disease: Prevention and therapy by exercise and condition-ing. Int. J. Mol. Sci. 2020, 21, 2896. [Google Scholar] [CrossRef]
- Després, J.-P. Physical Activity, Sedentary Behaviours, and Cardiovascular Health: When Will Cardiorespiratory Fitness Become a Vital Sign? Can. J. Cardiol. 2016, 32, 505–513. [Google Scholar] [CrossRef] [PubMed]
- Erekat, N.S.; Al-Jarrah, M.D.; Al Khatib, A.J. Treadmill Exercise Training Improves Vascular Endothelial Growth Factor Expression in the Cardiac Muscle of Type I Diabetic Rats. Cardiol. Res. 2014, 5, 23–29. [Google Scholar] [CrossRef][Green Version]
- Graham, K.; Yarar-Fisher, C.; Li, J.; McCully, K.M.; Rimmer, J.H.; Powell, D.; Bickel, C.S.; Fisher, G. Effects of High-Intensity Interval Training Versus Moderate-Intensity Training on Cardiometabolic Health Markers in Individuals With Spinal Cord Injury: A Pilot Study. Top. Spinal Cord Inj. Rehabil. 2019, 25, 248–259. [Google Scholar] [CrossRef]
- Ardakanizade, M. The effects of mid- and long-term endurance exercise on heart angiogenesis and oxidative stress. Iran. J. Basic. Med. Sci. 2018, 21, 800–805. [Google Scholar] [PubMed]
- Ne’Eman, Z.; Barash, V.; Rosenmann, E.; Shafrir, E. Localization of glycogen in the placenta of diabetic rats: A Light and electron microscopic study. Placenta 1987, 8, 201–208. [Google Scholar] [CrossRef]
- Sugimoto, K.; Rashid, I.B.; Shoji, M.; Suda, T.; Yasujima, M. Early changes in insulin receptor signaling and pain sensation in streptozotocin-induced diabetic neu-ropathy in rats. J. Pain 2008, 9, 237–245. [Google Scholar] [CrossRef]
- Chae, C.H.; Jung, S.L.; An, S.H.; Jung, C.K.; Nam, S.N.; Kim, H.T. Treadmill exercise suppresses muscle cell apoptosis by increasing nerve growth factor levels and stimulat-ing p-phosphatidylinositol 3-kinase activation in the soleus of diabetic rats. J. Physiol. Biochem. 2011, 67, 235–241. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2− ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Seguro, C.; Viana, R.; Lima, G.; Galvão, L.; Silva, L.; Jardim, T.; Jardim, P.; Gentil, P. Improvements in health parameters of a diabetic and hypertensive patient with only 40 min of exercise per week: A case study. Disabil. Rehabil. 2020, 42, 3119–3125. [Google Scholar] [CrossRef] [PubMed]
- Grossmann, M.; Thomas, M.C.; Panagiotopoulos, S.; Sharpe, K.; MacIsaac, R.J.; Clarke, S.; Zajac, J.D.; Jerums, G. Low Testosterone Levels Are Common and Associated with Insulin Resistance in Men with Diabetes. J. Clin. Endocrinol. Metab. 2008, 93, 1834–1840. [Google Scholar] [CrossRef]
- Khaneshi, F.; Nasrolahi, O.; Azizi, S.; Nejati, V. Sesame effects on testicular damage in streptozotocin-induced diabetes rats. Avicenna J. Phytomedicine 2013, 3, 347–355. [Google Scholar]
- Zumoff, B.; Strain, G.W.; Miller, L.K.; Rosner, W.; Senie, R.; Seres, D.S.; Rosenfeld, R.S. Plasma Free and Non-Sex-Hormone-Binding-Globulin Bound Testosterone Are Decreased in Obese Men in Proportion to Their Degree of Obesity. J. Clin. Endocrinol. Metab. 1990, 71, 929–931. [Google Scholar] [CrossRef] [PubMed]
- Akerstrom, T.; Laub, L.; Vedel, K.; Brand, C.L.; Pedersen, B.K.; Lindqvist, A.K.; Wojtaszewski, J.F.P.; Hellsten, Y. Increased skeletal muscle capillarization enhances insulin sensitivity. Am. J. Physiol. Metab. 2014, 307, E1105–E1116. [Google Scholar] [CrossRef] [PubMed]
- Rao, P.M.; Kelly, D.M.; Jones, T.H. Testosterone and insulin resistance in the metabolic syndrome and T2DM in men. Nat. Rev. Endocrinol. 2013, 9, 479–493. [Google Scholar] [CrossRef]
- Sacilotto, N.; Chouliaras, K.M.; Nikitenko, L.L.; Lu, Y.W.; Fritzsche, M.; Wallace, M.D.; Nornes, S.; García-Moreno, F.; Payne, S.; Bridges, E.; et al. MEF2 transcription factors are key regulators of sprouting angiogenesis. Genes Dev. 2016, 30, 2297–2309. [Google Scholar] [CrossRef] [PubMed]
- Tao, Z.; Shi, A.; Zhao, J. Epidemiological Perspectives of Diabetes. Cell Biophys. 2015, 73, 181–185. [Google Scholar] [CrossRef] [PubMed]
- Ago, T.; Liu, T.; Zhai, P.; Chen, W.; Li, H.; Molkentin, J.D.; Vatner, S.F.; Sadoshima, J. A Redox-Dependent Pathway for Regulating Class II HDACs and Cardiac Hypertrophy. Cell 2008, 133, 978–993. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Wei, L.; Chen, Q.; Terek, R.M. HDAC4 Represses Vascular Endothelial Growth Factor Expression in Chondrosarcoma by Modulating RUNX2 Activity. J. Biol. Chem. 2009, 284, 21881–21890. [Google Scholar] [CrossRef]
- Liu, J.; Zhou, X.; Li, Q.; Zhou, S.-M.; Hu, B.; Hu, G.-W.; Niu, X.; Guo, S.-C.; Wang, Y.; Deng, Z.-F. Role of Phosphorylated HDAC4 in Stroke-Induced Angiogenesis. BioMed Res. Int. 2017, 2017, 2957538 . [Google Scholar] [CrossRef] [PubMed]
- Kaurstad, G.; Alves, M.N.; Kemi, O.J.; Rolim, N.; Høydal, M.A.; Wisløff, H.; Stølen, T.O.; Wisløff, U. Chronic CaMKII inhibition blunts the cardiac contractile response to exercise training. Graefe’s Arch. Clin. Exp. Ophthalmol. 2012, 112, 579–588. [Google Scholar] [CrossRef] [PubMed]
- Stølen, T.O.; Høydal, M.A.; Kemi, O.J.; Catalucci, D.; Ceci, M.; Aasum, E.; Larsen, T.; Rolim, N.; Condorelli, G.; Smith, G.L.; et al. Interval training normalizes cardiomyocyte function, diastolic Ca2+ control, and SR Ca2+ release syn-chronicity in a mouse model of diabetic cardiomyopathy. Circ. Res. 2009, 105, 527–536. [Google Scholar] [CrossRef] [PubMed]
- Turtle, C. Altered Contractile Mechanics and Ca2+ Handling Contribute to Cardiomyopathy Pathogenesis; Oxford University: Oxford, UK, 2015. [Google Scholar]
- Hegyi, B.; Bers, D.M.; Bossuyt, J. CaMKII signaling in heart diseases: Emerging role in diabetic cardiomyopathy. J. Mol. Cell. Cardiol. 2019, 127, 246–259. [Google Scholar] [CrossRef] [PubMed]
- Awad, S.; Kunhi, M.; Little, G.H.; Bai, Y.; An, W.; Bers, D.; Kedes, L.; Poizat, C. Nuclear CaMKII enhances histone H3 phosphorylation and remodels chromatin during cardiac hypertro-phy. Nucleic Acids Res. 2013, 41, 7656–7672. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).