A Targeted and Tuneable DNA Damage Tool Using CRISPR/Cas9
Abstract
1. Introduction
2. Materials and Methods
2.1. Constructs
2.2. Protein Expression and Purification in Escherichia coli
2.3. Cell Culture
2.4. Drug Treatment
2.5. Electroporation
2.6. Immunofluorescence
2.7. Widefield Fluorescent Imaging
2.8. Confocal Imaging
2.9. Image Analysis
2.10. STochastic Optical Reconstruction Microscopy (STORM) Imaging
2.11. Clus-DoC
2.12. In Silico Design of Guide RNAs
3. Results
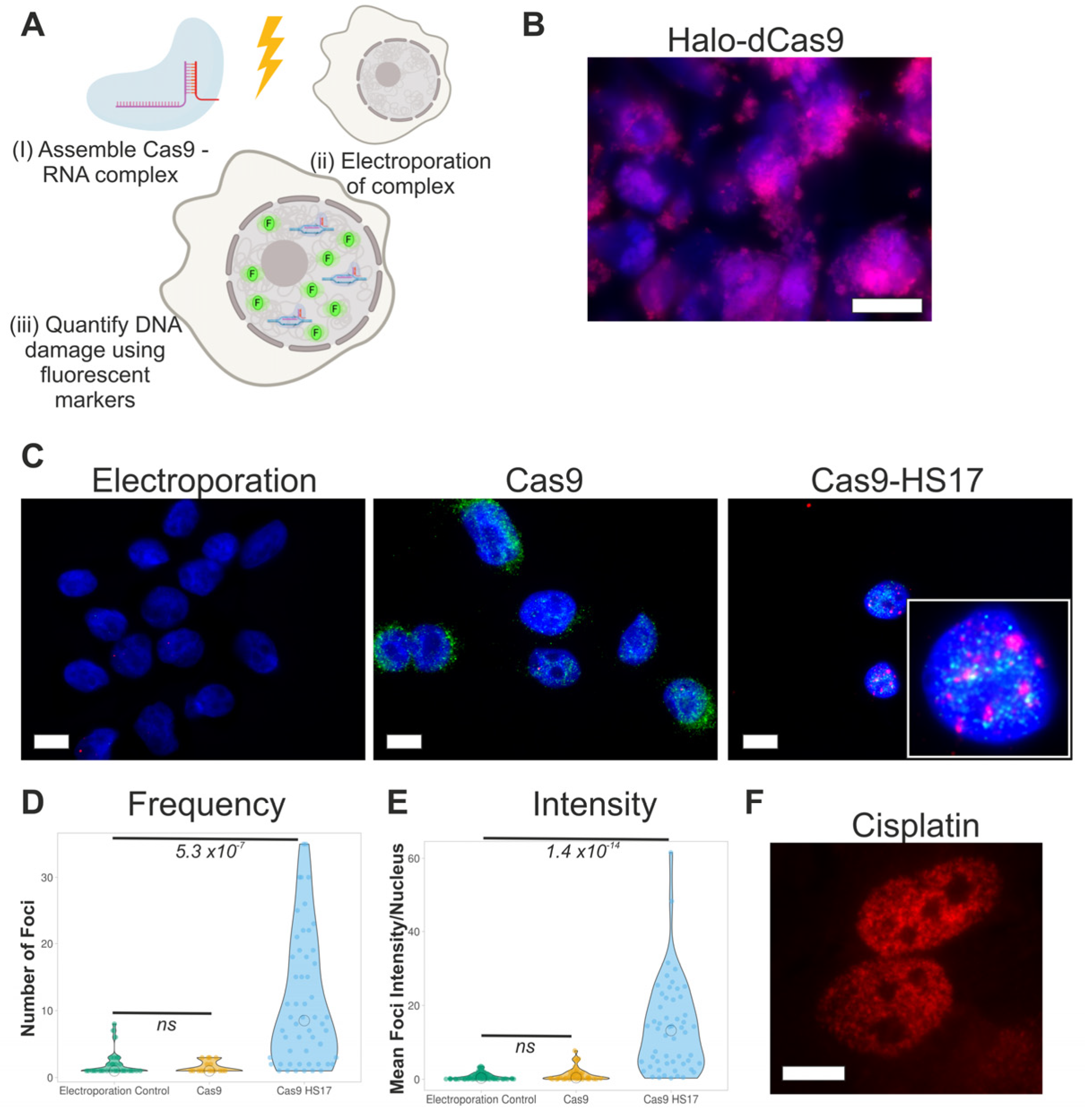
3.1. Proof-of-Concept Using Electroporation of Recombinant Cas9
3.2. Design of Promiscuous Guide RNAs
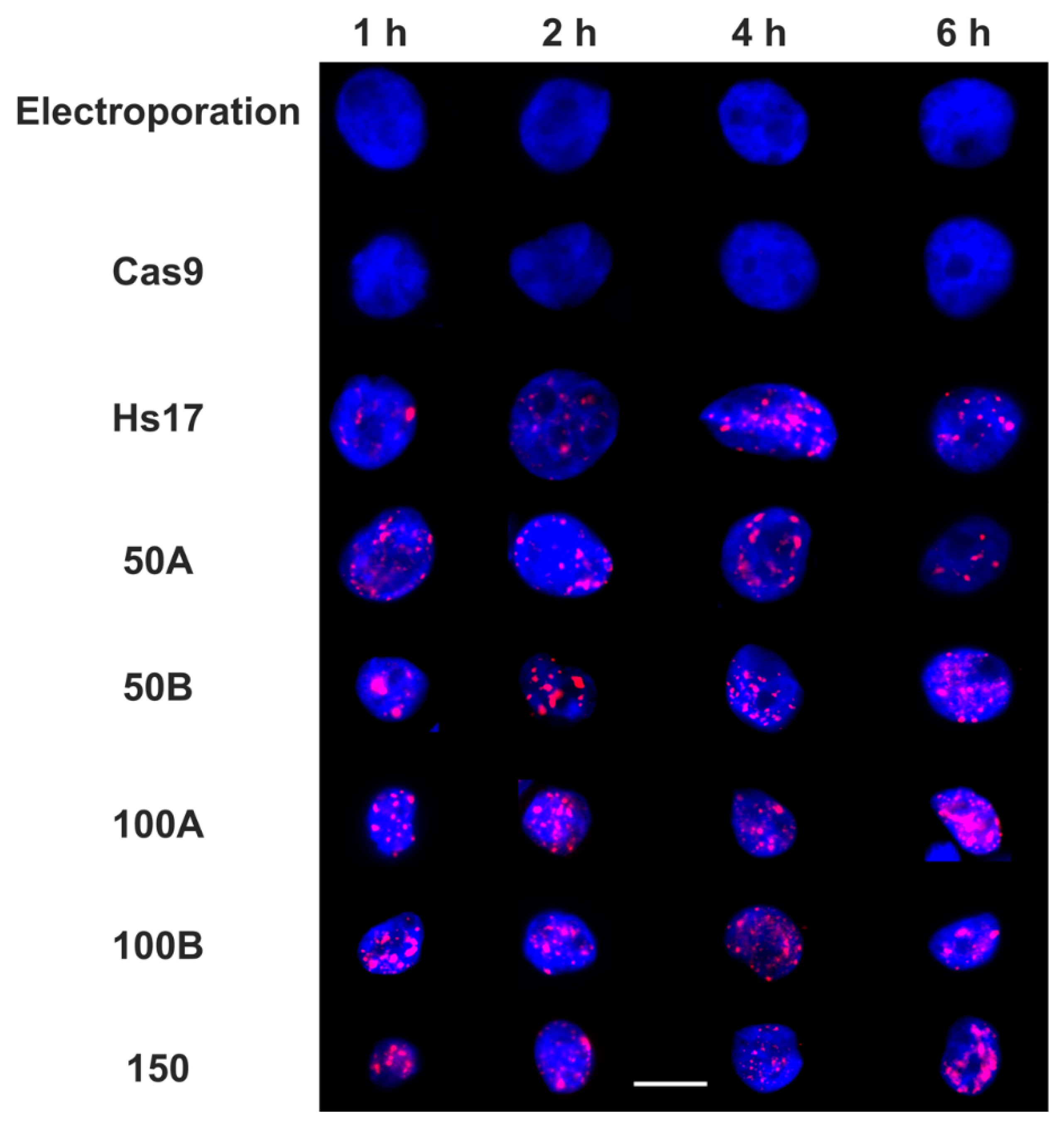
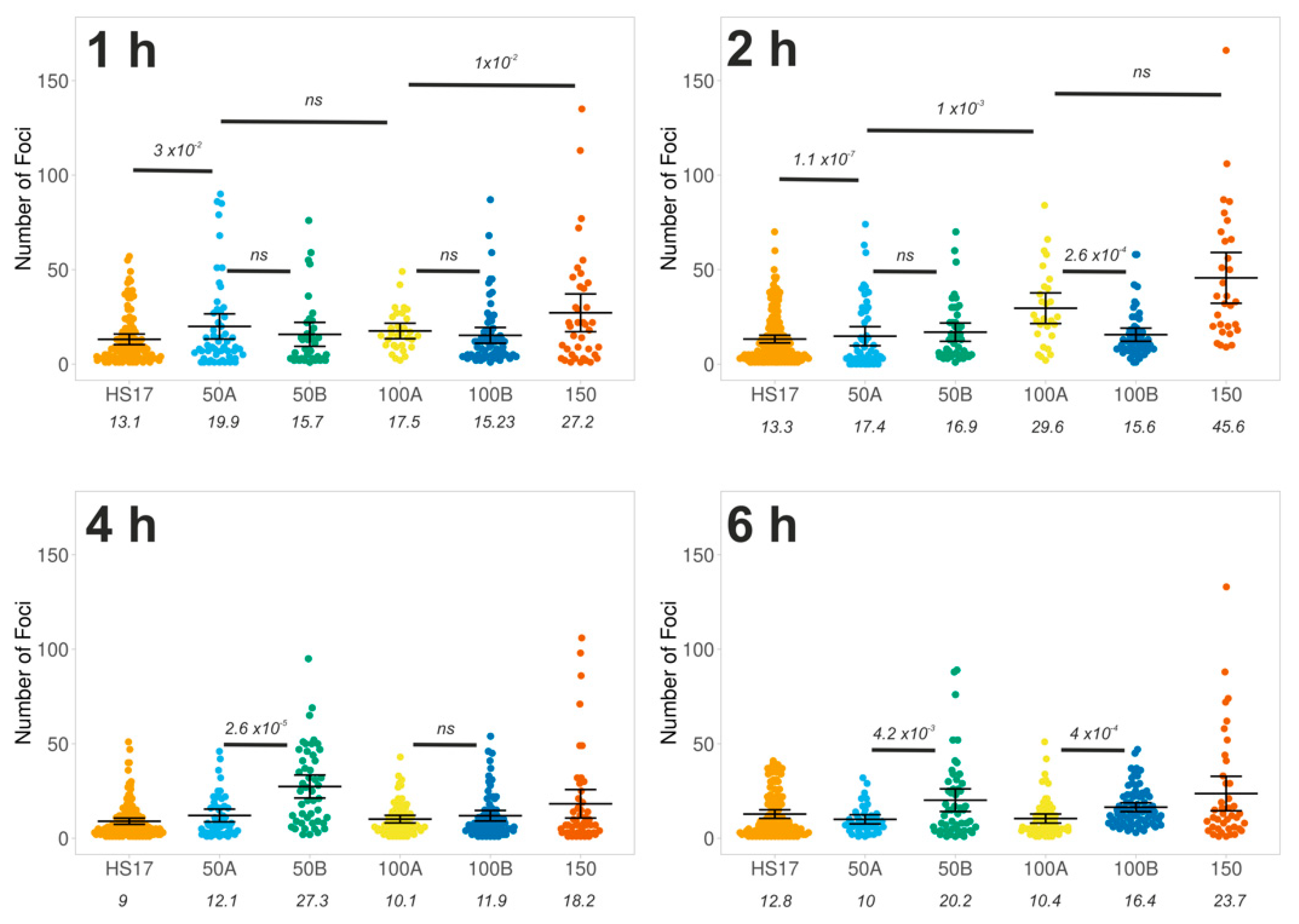
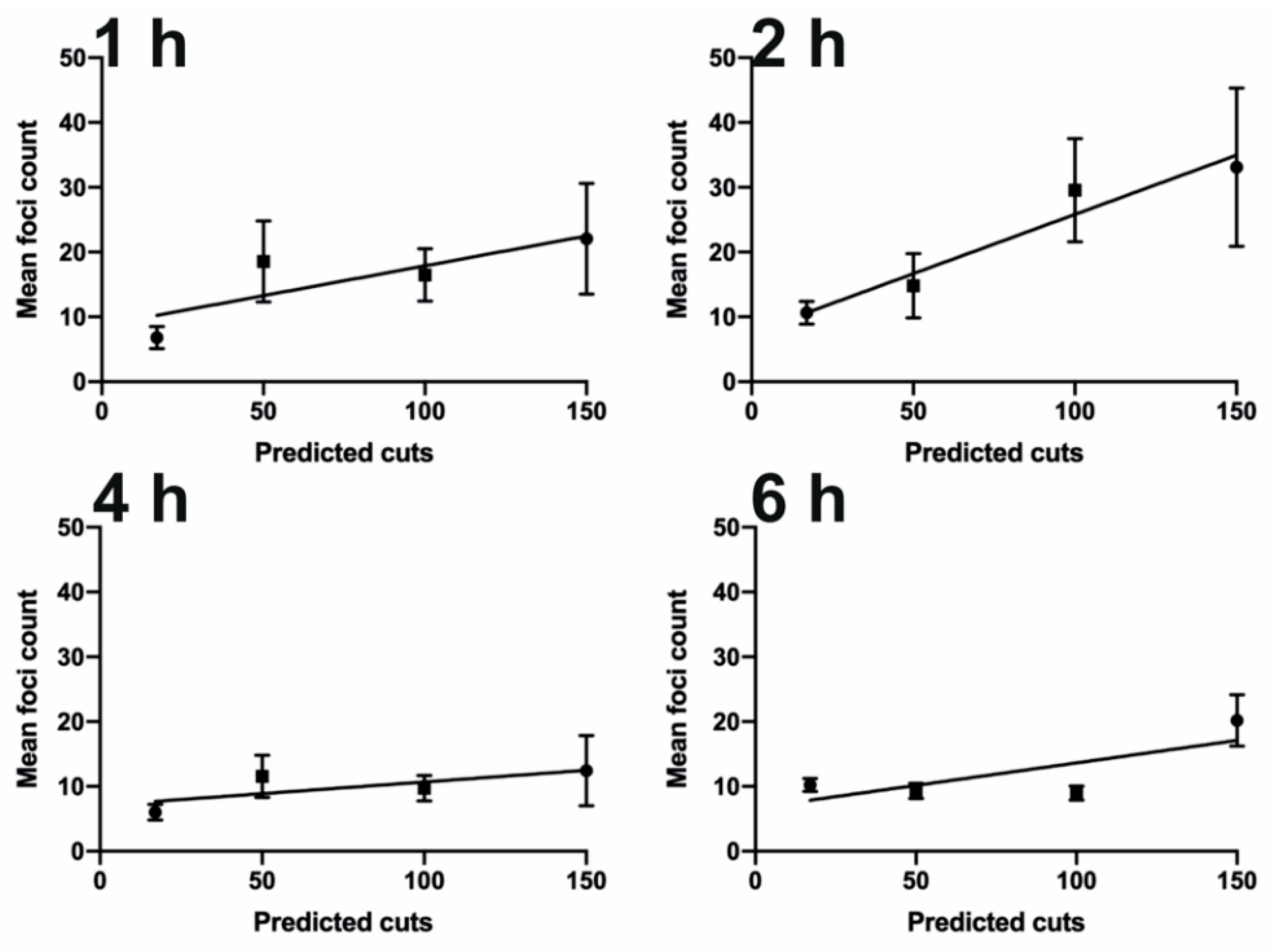
3.3. Tuning the Number of DNA Damage Sites Using Cas9 and Promiscuous Guides
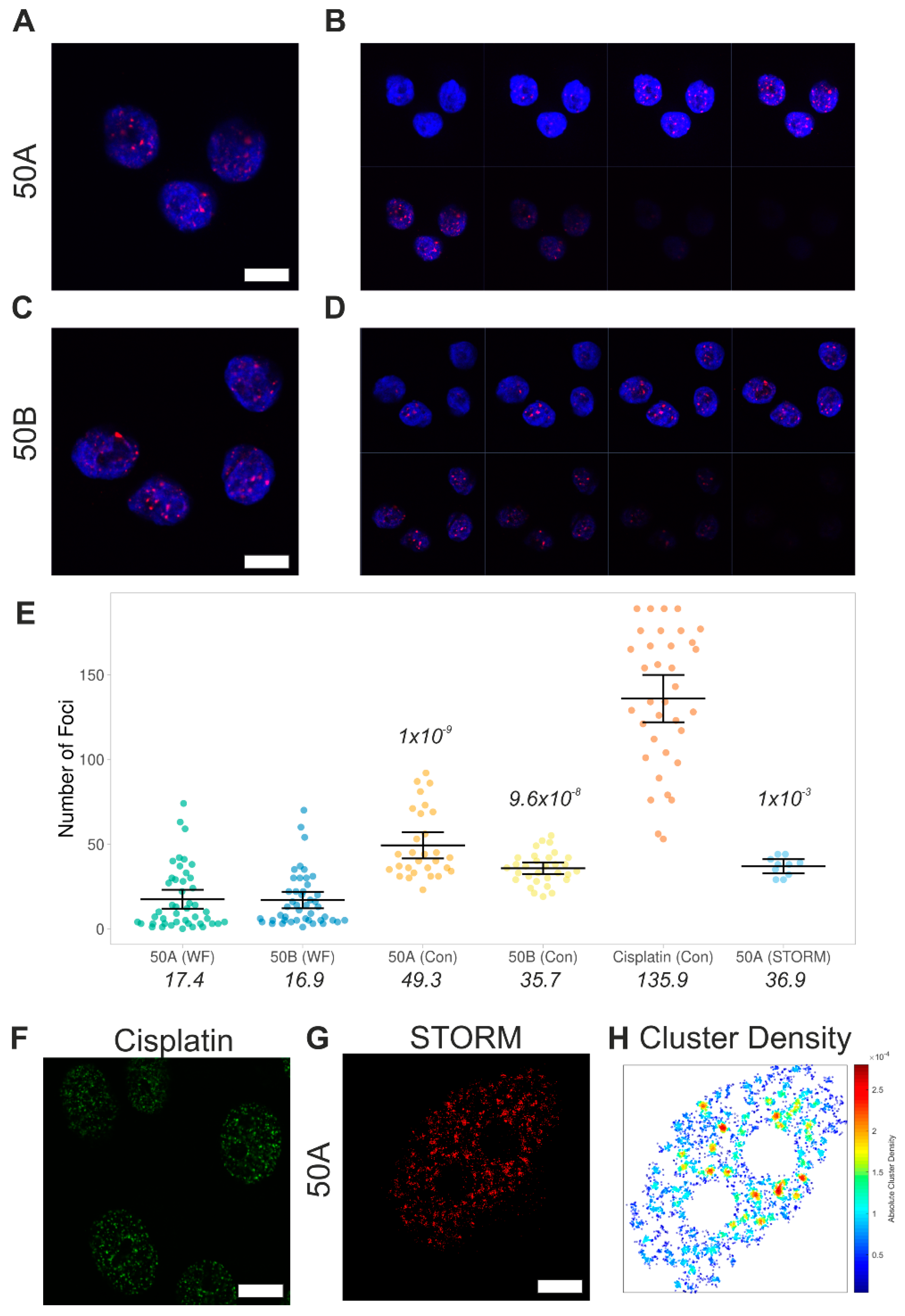
Single Particle Analysis of DNA Damage Foci
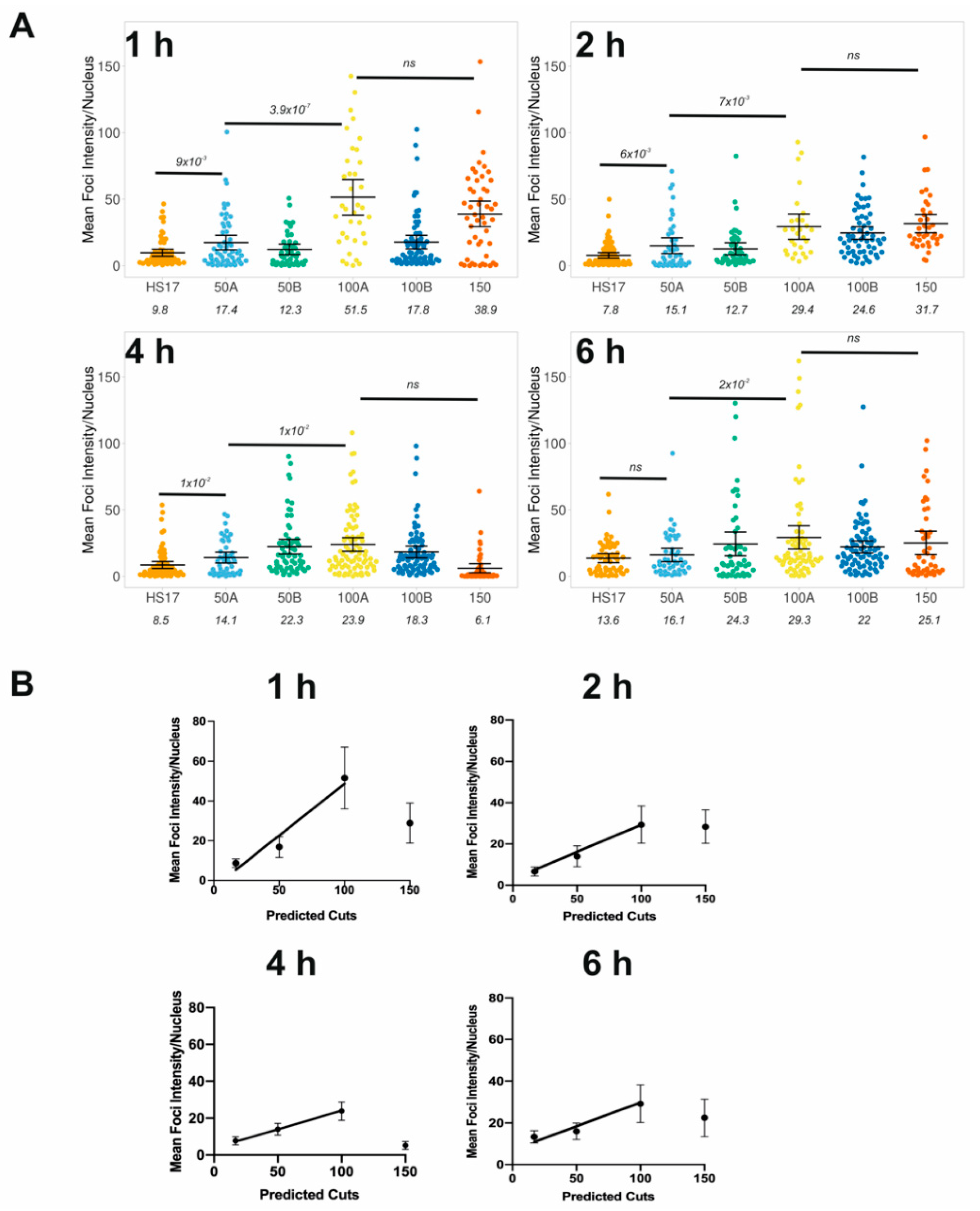
3.4. Tuning the Amount of DNA Damage Using Cas9 and Promiscuous Guides
Intensity-Based Quantification of DNA Damage
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Graphics
References
- Hustedt, N.; Durocher, D. The control of DNA repair by the cell cycle. Nat. Cell Biol. 2017, 19, 1–9. [Google Scholar] [CrossRef]
- Haber, J.E. DNA recombination: The replication connection. Trends Biochem. Sci. 1999, 24, 271–275. [Google Scholar] [CrossRef]
- Vilenchik, M.M.; Knudson, A.G. Endogenous DNA double-strand breaks: Production, fidelity of repair, and induction of cancer. Proc. Natl. Acad. Sci. USA 2003, 100, 12871–12876. [Google Scholar] [CrossRef]
- Jackson, S.P.; Bartek, J. The DNA-damage response in human biology and disease. Nat. Cell Biol. 2009, 461, 1071–1078. [Google Scholar] [CrossRef] [PubMed]
- Nowsheen, S.; Yang, E. The intersection between DNA damage response and cell death pathways. Exp. Oncol. 2012, 34, 243–254. [Google Scholar] [PubMed]
- Bryant, H.E.; Schultz, N.; Thomas, H.D.; Parker, K.M.; Flower, D.; Lopez, E.; Kyle, S.; Meuth, M.; Curtin, N.J.; Helleday, T. Specific killing of BRCA2-deficient tumours with inhibitors of poly(ADP-ribose) polymerase. Nat. Cell Biol. 2005, 434, 913–917. [Google Scholar] [CrossRef] [PubMed]
- dos Santos, A.; Cook, A.W.; Gough, R.E.; Schilling, M.; Olszok, A.N.; Brown, I.; Wang, L.; Aaron, J.; Martin-Fernandez, M.L.; Rehfeldt, F.; et al. DNA damage alters nuclear mechanics through chromatin reorganization. Nucleic Acids Res. 2021, 49, 340–353. [Google Scholar] [CrossRef] [PubMed]
- Berkovich, E.; Monnat, R.J., Jr.; Kastan, M.B. Roles of ATM and NBS1 in chromatin structure modulation and DNA double-strand break repair. Nat. Cell Biol. 2007, 9, 683–690. [Google Scholar] [CrossRef]
- Bellaiche, Y.; Mogila, V.; Perrimon, N. I-SceI endonuclease, a new tool for studying DNA double-strand break repair mechanisms in Drosophila. Genet. 1999, 152, 1037–1044. [Google Scholar]
- Iacovoni, J.S.; Caron, P.; Lassadi, I.; Nicolas, E.; Massip, L.; Trouche, D.; Legube, G. High-resolution profiling of γH2AX around DNA double strand breaks in the mammalian genome. EMBO J. 2010, 29, 1446–1457. [Google Scholar] [CrossRef]
- Gaj, T.; Gersbach, C.A.; Barbas, C.F. ZFN, TALEN, and CRISPR/Cas-based methods for genome engineering. Trends Biotechnol. 2013, 31, 397–405. [Google Scholar] [CrossRef] [PubMed]
- Jinek, M.; Chylinski, K.; Fonfara, I.; Hauer, M.; Doudna, J.A.; Charpentier, E. A Programmable Dual-RNA-Guided DNA Endonuclease in Adaptive Bacterial Immunity. Science 2012, 337, 816–821. [Google Scholar] [CrossRef]
- Maeder, M.L.; Thibodeau-Beganny, S.; Osiak, A.; Wright, D.A.; Anthony, R.M.; Eichtinger, M.; Jiang, T.; Foley, J.E.; Winfrey, R.J.; Townsend, J.A.; et al. Rapid “Open-Source” Engineering of Customized Zinc-Finger Nucleases for Highly Efficient Gene Modification. Mol. Cell 2008, 31, 294–301. [Google Scholar] [CrossRef]
- Miller, J.C.; Tan, S.; Qiao, G.; Barlow, K.A.; Wang, J.; Xiangdong, M.; Meng, X.; Paschon, D.E.; Leung, E.; Hinkley, S.J.; et al. A TALE nuclease architecture for efficient genome editing. Nat. Biotechnol. 2010, 29, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Mussolino, C.; Morbitzer, R.; Lütge, F.; Dannemann, N.; Lahaye, T.; Cathomen, T. A novel TALE nuclease scaffold enables high genome editing activity in combination with low toxicity. Nucleic Acids Res. 2011, 39, 9283–9293. [Google Scholar] [CrossRef] [PubMed]
- Berg, J.V.D.; Manjón, A.G.; Kielbassa, K.; Feringa, F.M.; Freire, R.; Medema, R.H. A limited number of double-strand DNA breaks is sufficient to delay cell cycle progression. Nucleic Acids Res. 2018, 46, 10132–10144. [Google Scholar] [CrossRef]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: An open-source platform for biological-image analysis. Nat. Methods 2012, 9, 676–682. [Google Scholar] [CrossRef]
- Pageon, S.V.; Nicovich, P.R.; Mollazade, M.; Tabarin, T.; Gaus, K. Clus-DoC: A combined cluster detection and colocalization analysis for single-molecule localization microscopy data. Mol. Biol. Cell 2016, 27, 3627–3636. [Google Scholar] [CrossRef] [PubMed]
- Postma, M.; Goedhart, J. PlotsOfData—A web app for visualizing data together with their summaries. PLoS Biol. 2019, 17, e3000202. [Google Scholar] [CrossRef]
- McKenna, A.; Shendure, J. FlashFry: A fast and flexible tool for large-scale CRISPR target design. BMC Biol. 2018, 16, 1–6. [Google Scholar] [CrossRef] [PubMed]
- dos Santos, Á.; Fili, N.; Hari-Gupta, Y.; Gough, R.E.; Wang, L.; Martin-Fernandez, M.L.; Aaron, J.; Waite, E.; Chew, T.-L.; Toseland, C.P. Binding partners regulate unfolding of myosin VI to activate the molecular motor. bioRxiv 2020. [Google Scholar] [CrossRef]
- Hari-Gupta, Y.; Fili, N.; dos Santos, Á.; Cook, A.W.; Gough, R.E.; Reed, H.C.; Wang, L.; Aaron, J.; Venit, T.; Wait, E.; et al. Nuclear myosin VI regulates the spatial organization of mammalian transcription initiation. bioRxiv 2020. [Google Scholar] [CrossRef]
- Zhou, J.; Wang, J.; Shen, B.; Chen, L.; Su, Y.; Yang, J.; Zhang, W.; Tian, X.; Huang, X. Dual sgRNAs facilitate CRISPR/Cas9-mediated mouse genome targeting. FEBS J. 2014, 281, 1717–1725. [Google Scholar] [CrossRef] [PubMed]
- Biernacka, A.; Zhu, Y.; Skrzypczak, M.; Forey, R.; Pardo, B.; Grzelak, M.; Nde, J.; Mitra, A.; Kudlicki, A.; Crosetto, N.; et al. i-BLESS is an ultra-sensitive method for detection of DNA double-strand breaks. Commun. Biol. 2018, 1, 1–9. [Google Scholar] [CrossRef]
- Zhu, Y.; Biernacka, A.; Pardo, B.; Dojer, N.; Forey, R.; Skrzypczak, M.; Fongang, B.; Nde, J.; Yousefi, R.; Pasero, P.; et al. qDSB-Seq is a general method for genome-wide quantification of DNA double-strand breaks using sequencing. Nat. Commun. 2019, 10, 2313. [Google Scholar] [CrossRef] [PubMed]
- Kaya-Okur, H.S.; Janssens, D.H.; Henikoff, J.G.; Ahmad, K.; Henikoff, S. Efficient low-cost chromatin profiling with CUT&Tag. Nat. Protoc. 2020, 15, 1–20. [Google Scholar] [CrossRef]
- Skene, P.J.; Henikoff, S. An efficient targeted nuclease strategy for high-resolution mapping of DNA binding sites. eLife 2017, 6, e21856. [Google Scholar] [CrossRef]
| Name | crRNA Sequence (5′-3′) | FlashFry Hits | Ensembl Hits |
|---|---|---|---|
| 50A | ACCCCTGGCAGCTGCGGTTCAGG | 54 | 53 |
| 50B | TATAATAAGCAAATTGCAATGGG | 52 | 50 |
| 100A | GGGGCTTCCAGGTCACAGGTAGG | 111 | 100 |
| 100B | ACTTTAAGTTTTAGGGTACATGG | 100 | 100 |
| 150 | GTGCCAGAAATCTGGCCACCAGG | 158 | 154 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Emmanouilidis, I.; Fili, N.; Cook, A.W.; Hari-Gupta, Y.; dos Santos, Á.; Wang, L.; Martin-Fernandez, M.L.; Ellis, P.J.I.; Toseland, C.P. A Targeted and Tuneable DNA Damage Tool Using CRISPR/Cas9. Biomolecules 2021, 11, 288. https://doi.org/10.3390/biom11020288
Emmanouilidis I, Fili N, Cook AW, Hari-Gupta Y, dos Santos Á, Wang L, Martin-Fernandez ML, Ellis PJI, Toseland CP. A Targeted and Tuneable DNA Damage Tool Using CRISPR/Cas9. Biomolecules. 2021; 11(2):288. https://doi.org/10.3390/biom11020288
Chicago/Turabian StyleEmmanouilidis, Ioannis, Natalia Fili, Alexander W. Cook, Yukti Hari-Gupta, Ália dos Santos, Lin Wang, Marisa L. Martin-Fernandez, Peter J. I. Ellis, and Christopher P. Toseland. 2021. "A Targeted and Tuneable DNA Damage Tool Using CRISPR/Cas9" Biomolecules 11, no. 2: 288. https://doi.org/10.3390/biom11020288
APA StyleEmmanouilidis, I., Fili, N., Cook, A. W., Hari-Gupta, Y., dos Santos, Á., Wang, L., Martin-Fernandez, M. L., Ellis, P. J. I., & Toseland, C. P. (2021). A Targeted and Tuneable DNA Damage Tool Using CRISPR/Cas9. Biomolecules, 11(2), 288. https://doi.org/10.3390/biom11020288







