Genetic, Environmental and Lifestyle Determinants of Accelerated Telomere Attrition as Contributors to Risk and Severity of Multiple Sclerosis
Abstract
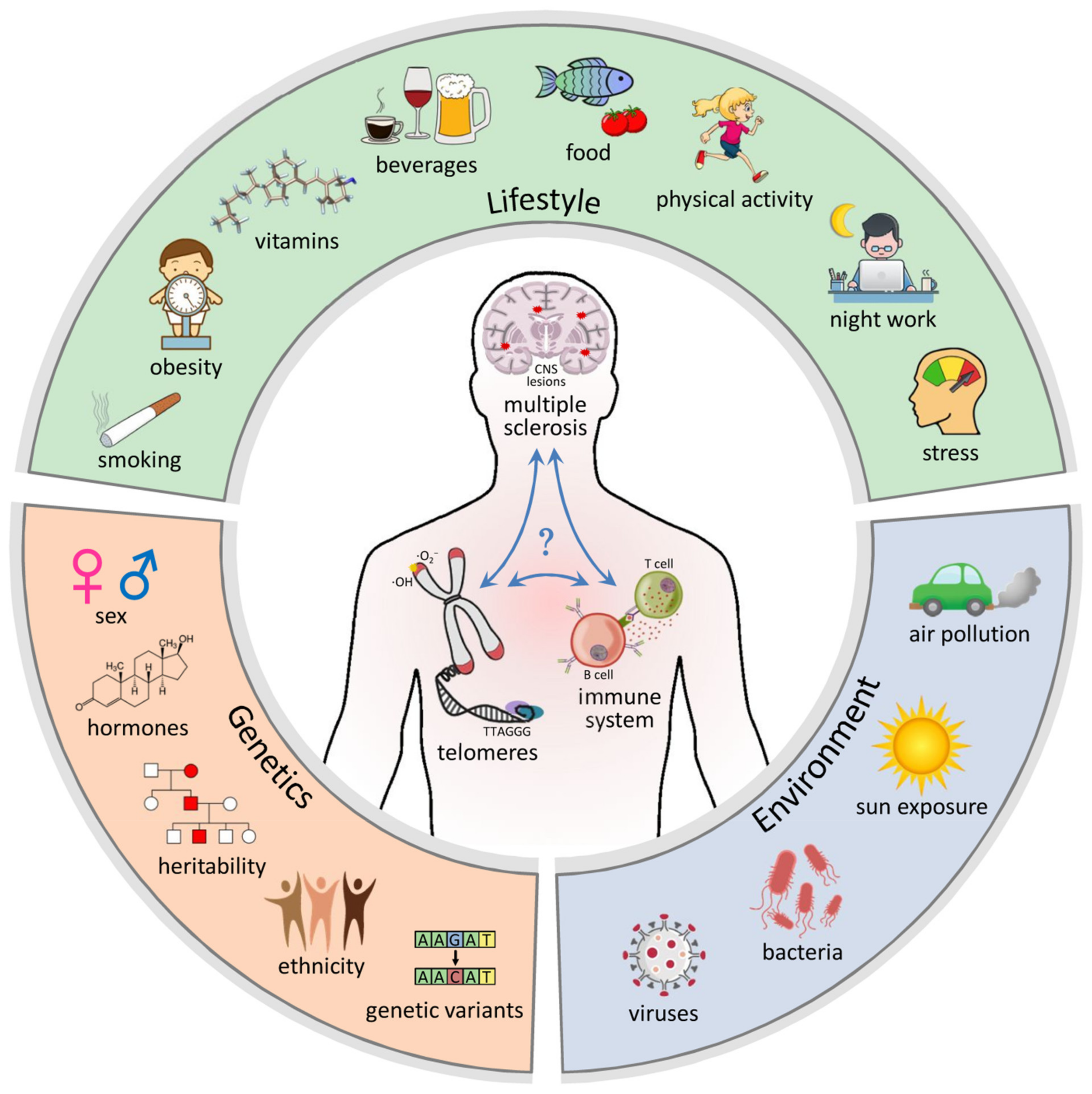
1. Telomere Biology
2. Multiple Sclerosis
3. Genetic Factors
3.1. Race/Ethnicity
3.2. Sex Differences
3.3. Familial Heritability
3.4. Genetic Variants
4. Environmental Factors
4.1. Sun Exposure
4.2. Viral Infections
4.3. Other Infections
4.4. Air Pollution
5. Lifestyle Factors
5.1. Dietary Habits
5.2. Vitamin Supply
5.3. Drinking Behavior
5.4. Cigarette Smoking
5.5. Physical Activity
5.6. Psychological Stress
5.7. Shift Work
6. Concluding Remarks
Author Contributions
Funding
Conflicts of Interest
Glossary
Abbreviations
| 8-OHdG | 8-hydroxy-desoxyguanosine |
| ALT | alternative lengthening of telomeres |
| BMI | body mass index |
| bp | base pair |
| CHI3L1 | chitinase 3-like protein 1 |
| CIS | clinically isolated syndrome |
| CMV | cytomegalovirus |
| CNS | central nervous system |
| CRP | C-reactive protein |
| EBNA1 | Epstein–Barr virus nuclear antigen 1 |
| EBV | Epstein–Barr virus |
| EDSS | Expanded Disability Status Scale |
| GWAS | genome-wide association study |
| HHV | human herpesvirus |
| HLA | human leukocyte antigen |
| H.p. | Helicobacter pylori |
| HR | hazard ratio |
| IgG | immunoglobulin G |
| IM | infectious mononucleosis |
| kb | kilobase |
| LD | linkage disequilibrium |
| LTL | leukocyte telomere length |
| MHC | major histocompatibility complex |
| MRI | magnetic resonance imaging |
| MS | multiple sclerosis |
| MSSS | Multiple Sclerosis Severity Score |
| OR | odds ratio |
| OS | oxidative stress |
| PA | physical activity |
| PCR | polymerase chain reaction |
| PM | particulate matter |
| PPMS | primary progressive multiple sclerosis |
| Q-FISH | quantitative fluorescence in situ hybridization |
| RNA | ribonucleic acid |
| RR | relative risk |
| RRMS | relapsing-remitting multiple sclerosis |
| S | single-copy gene signal |
| SAA | serum amyloid A |
| SASP | senescence-associated secretory phenotype |
| SD | standard deviation |
| SMD | standardized mean difference |
| SNP | single-nucleotide polymorphism |
| SPMS | secondary progressive multiple sclerosis |
| T | telomere signal |
| TERC | telomerase RNA component |
| TERT | telomerase reverse transcriptase |
| TeSLA | telomere shortest length assay |
| TL | telomere length |
| TRF | terminal restriction fragment |
| U-STELA | universal single telomere length analysis |
References
- Saretzki, G. Telomeres, Telomerase and Ageing. Subcell. Biochem. 2018, 90, 221–308. [Google Scholar] [CrossRef] [PubMed]
- Moyzis, R.K.; Buckingham, J.M.; Cram, L.S.; Dani, M.; Deaven, L.L.; Jones, M.D.; Meyne, J.; Ratliff, R.L.; Wu, J.R. A highly conserved repetitive DNA sequence, (TTAGGG)n, present at the telomeres of human chromosomes. Proc. Natl. Acad. Sci. USA 1988, 85, 6622–6626. [Google Scholar] [CrossRef]
- De Lange, T. Shelterin: The Protein Complex That Shapes and Safeguards Human Telomeres. Genes Dev. 2005, 19, 2100–2110. [Google Scholar] [CrossRef]
- Levy, M.Z.; Allsopp, R.C.; Futcher, A.; Greider, C.; Harley, C.B. Telomere end-replication problem and cell aging. J. Mol. Biol. 1992, 225, 951–960. [Google Scholar] [CrossRef]
- Hayflick, L.; Moorhead, P.S. The serial cultivation of human diploid cell strains. Exp. Cell Res. 1961, 25, 585–621. [Google Scholar] [CrossRef]
- Reichert, S.; Stier, A. Does oxidative stress shorten telomeres in vivo? A review. Biol. Lett. 2017, 13, 20170463. [Google Scholar] [CrossRef] [PubMed]
- Von Zglinicki, T. Oxidative stress shortens telomeres. Trends Biochem. Sci. 2002, 27, 339–344. [Google Scholar] [CrossRef]
- Visvikis-Siest, S. The future of telomere length in personalized medicine. Front. Biosci. 2018, 23, 1628–1654. [Google Scholar] [CrossRef]
- Liu, Z.; Zhang, J.; Yan, J.; Wang, Y.; Li, Y. Leucocyte Telomere Shortening in relation to Newly Diagnosed Type 2 Diabetic Patients with Depression. Oxidative Med. Cell. Longev. 2014, 2014, 1–8. [Google Scholar] [CrossRef]
- Wong, J.; De Vivo, I.; Lin, X.; Fang, S.C.; Christiani, D.C. The Relationship between Inflammatory Biomarkers and Telomere Length in an Occupational Prospective Cohort Study. PLoS ONE 2014, 9, e87348. [Google Scholar] [CrossRef]
- Criscuolo, F.; Sorci, G.; Behaim-Delarbre, M.; Zahn, S.; Faivre, B.; Bertile, F. Age-related response to an acute innate immune challenge in mice: Proteomics reveals a telomere maintenance-related cost. Proc. R. Soc. B Boil. Sci. 2018, 285, 20181877. [Google Scholar] [CrossRef]
- Henckel, E.; James, A.; Konradsen, J.; Nordlund, B.; Kjellberg, M.; Berggren-Broström, E.; Hedlin, G.; Degerman, S.; Bohlin, K. A Novel Association between YKL-40, a Marker of Structural Lung Disease, and Short Telomere Length in 10-Year-Old Children with Bronchopulmonary Dysplasia. Children 2021, 8, 80. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Schiffer, E.; Song, Z.; Wang, J.; Zürbig, P.; Thedieck, K.; Moes, S.; Bantel, H.; Saal, N.; Jantos, J.; et al. Proteins induced by telomere dysfunction and DNA damage represent biomarkers of human aging and disease. Proc. Natl. Acad. Sci. USA 2008, 105, 11299–11304. [Google Scholar] [CrossRef]
- Chakravarti, D.; Labella, K.A.; Depinho, R.A. Telomeres: History, health, and hallmarks of aging. Cell 2021, 184, 306–322. [Google Scholar] [CrossRef]
- Wright, W.E.; Piatyszek, M.A.; Rainey, W.E.; Byrd, W.; Shay, J.W. Telomerase activity in human germline and embryonic tissues and cells. Dev. Genet. 1996, 18, 173–179. [Google Scholar] [CrossRef]
- Kim, W.; Ludlow, A.T.; Min, J.; Robin, J.D.; Stadler, G.; Mender, I.; Lai, T.-P.; Zhang, N.; Wright, W.E.; Shay, J.W. Regulation of the Human Telomerase Gene TERT by Telomere Position Effect-Over Long Distances (TPE-OLD): Implications for Aging and Cancer. PLoS Biol. 2016, 14, e2000016. [Google Scholar] [CrossRef]
- Shay, J.W.; Wright, W.E. Telomeres and telomerase: Three decades of progress. Nat. Rev. Genet. 2019, 20, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Gaspar, T.B.; Sá, A.; Lopes, J.M.; Sobrinho-Simões, M.; Soares, P.; Vinagre, J. Telomere Maintenance Mechanisms in Cancer. Genes 2018, 9, 241. [Google Scholar] [CrossRef]
- Cawthon, R.M. Telomere measurement by quantitative PCR. Nucl. Acids Res. 2002, 30, e47. [Google Scholar] [CrossRef] [PubMed]
- Cawthon, R.M. Telomere length measurement by a novel monochrome multiplex quantitative PCR method. Nucl. Acids Res. 2009, 37, e21. [Google Scholar] [CrossRef] [PubMed]
- Lai, T.-P.; Zhang, N.; Noh, J.; Mender, I.; Tedone, E.; Huang, E.; Wright, W.E.; Danuser, G.; Shay, J.W. A method for measuring the distribution of the shortest telomeres in cells and tissues. Nat. Commun. 2017, 8, 1356. [Google Scholar] [CrossRef]
- Lai, T.-P.; Wright, W.E.; Shay, J.W. Comparison of telomere length measurement methods. Philos. Trans. R. Soc. B Biol. Sci. 2018, 373, 20160451. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Viswanathan, R.; Hande, M.P.; Loh, A.H.P.; Cheow, L.F. Massively parallel single-molecule telomere length measurement with digital real-time PCR. Sci. Adv. 2020, 6, eabb7944. [Google Scholar] [CrossRef]
- Grigorev, K.; Foox, J.; Bezdan, D.; Butler, D.; Luxton, J.J.; Reed, J.; McKenna, M.J.; Taylor, L.; George, K.A.; Meydan, C.; et al. Haplotype diversity and sequence heterogeneity of human telomeres. Genome Res. 2021, 31, 1269–1279. [Google Scholar] [CrossRef]
- Smith, L.; Luchini, C.; Demurtas, J.; Soysal, P.; Stubbs, B.; Hamer, M.; Nottegar, A.; Lawlor, R.T.; Sánchez, G.F.L.; Firth, J.; et al. Telomere length and health outcomes: An umbrella review of systematic reviews and meta-analyses of observational studies. Ageing Res. Rev. 2019, 51, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Anitha, A.; Thanseem, I.; Vasu, M.M.; Viswambharan, V.; Poovathinal, S.A. Telomeres in neurological disorders. Adv. Appl. Microbiol. 2019, 90, 81–132. [Google Scholar] [CrossRef]
- Barthel, F.P.; Wei, W.; Tang, M.; Martinez-Ledesma, E.; Hu, X.; Amin, S.B.; Akdemir, K.C.; Seth, S.; Song, X.; Wang, Q.; et al. Systematic analysis of telomere length and somatic alterations in 31 cancer types. Nat. Genet. 2017, 49, 349–357. [Google Scholar] [CrossRef]
- Guan, J.-Z.; Guan, W.-P.; Maeda, T.; Guoqing, X.; Guangzhi, W.; Makino, N. Patients with multiple sclerosis show increased oxidative stress markers and somatic telomere length shortening. Mol. Cell. Biochem. 2015, 400, 183–187. [Google Scholar] [CrossRef]
- Guan, J.Z.; Guan, W.-P.; Maeda, T. Vitamin E administration erases an enhanced oxidation in multiple sclerosis. Can. J. Physiol. Pharmacol. 2018, 96, 1–3. [Google Scholar] [CrossRef]
- Habib, R.; Ocklenburg, S.; Hoffjan, S.; Haghikia, A.; Epplen, J.T.; Arning, L. Association between shorter leukocyte telomeres and multiple sclerosis. J. Neuroimmunol. 2020, 341, 577187. [Google Scholar] [CrossRef] [PubMed]
- Hecker, M.; Fitzner, B.; Jäger, K.; Bühring, J.; Schwartz, M.; Hartmann, A.; Walter, M.; Zettl, U.K. Leukocyte Telomere Length in Patients with Multiple Sclerosis and Its Association with Clinical Phenotypes. Mol. Neurobiol. 2021, 58, 2886–2896. [Google Scholar] [CrossRef]
- Bühring, J.; Hecker, M.; Fitzner, B.; Zettl, U.K. Systematic Review of Studies on Telomere Length in Patients with Multiple Sclerosis. Aging Dis. 2021, 12, 1272–1286. [Google Scholar] [CrossRef]
- Codd, V.; Wang, Q.; Allara, E.; Musicha, C.; Kaptoge, S.; Stoma, S.; Jiang, T.; Hamby, S.E.; Braund, P.S.; Bountziouka, V.; et al. Polygenic basis and bio-medical consequences of telomere length variation. Nat. Genet. 2021, 53, 1425–1433. [Google Scholar] [CrossRef]
- Demanelis, K.; Jasmine, F.; Chen, L.S.; Chernoff, M.; Tong, L.; Delgado, D.; Zhang, C.; Shinkle, J.; Sabarinathan, M.; Lin, H.; et al. Determinants of telomere length across human tissues. Science 2020, 369, eaaz6876. [Google Scholar] [CrossRef] [PubMed]
- Filippi, M.; Bar-Or, A.; Piehl, F.; Preziosa, P.; Solari, A.; Vukusic, S.; Rocca, A.M. Multiple sclerosis. Nat. Rev. Dis. Prim. 2018, 4, 43. [Google Scholar] [CrossRef] [PubMed]
- Walton, C.; King, R.; Rechtman, L.; Kaye, W.; Leray, E.; Marrie, R.A.; Robertson, N.; La Rocca, N.; Uitdehaag, B.; Van Der Mei, I.; et al. Rising prevalence of multiple sclerosis worldwide: Insights from the Atlas of MS, third edition. Mult. Scler. J. 2020, 26, 1816–1821. [Google Scholar] [CrossRef] [PubMed]
- Olsson, T.; Barcellos, L.F.; Alfredsson, L. Interactions between genetic, lifestyle and environmental risk factors for multiple sclerosis. Nat. Rev. Neurol. 2017, 13, 25–36. [Google Scholar] [CrossRef]
- Zettl, U.K.; Stüve, O.; Patejdl, R. Immune-mediated CNS diseases: A review on nosological classification and clinical features. Autoimmun. Rev. 2012, 11, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Rommer, P.S.; Eichstädt, K.; Ellenberger, D.; Flachenecker, P.; Friede, T.; Haas, J.; Kleinschnitz, C.; Pöhlau, D.; Rienhoff, O.; Stahmann, A.; et al. Symptomatology and symptomatic treatment in multiple sclerosis: Results from a nationwide MS registry. Mult. Scler. J. 2019, 25, 1641–1652. [Google Scholar] [CrossRef]
- Orton, S.-M.; Herrera, B.M.; Yee, I.M.; Valdar, W.; Ramagopalan, S.V.; Sadovnick, A.D.; Ebers, G. Sex ratio of multiple sclerosis in Canada: A longitudinal study. Lancet Neurol. 2006, 5, 932–936. [Google Scholar] [CrossRef]
- Wallin, M.T.; Culpepper, W.J.; Campbell, J.D.; Nelson, L.M.; Langer-Gould, A.; Marrie, R.A.; Cutter, G.R.; Kaye, W.E.; Wagner, L.; Tremlett, H.; et al. The prevalence of MS in the United States. Neurology 2019, 92, e1029–e1040. [Google Scholar] [CrossRef] [PubMed]
- Kurtzke, J.F. Rating neurologic impairment in multiple sclerosis: An expanded disability status scale (EDSS). Neurology 1983, 33, 1444–1452. [Google Scholar] [CrossRef] [PubMed]
- Thompson, A.J.; Banwell, B.L.; Barkhof, F.; Carroll, W.M.; Coetzee, T.; Comi, G.; Correale, J.; Fazekas, F.; Filippi, M.; Freedman, M.S.; et al. Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol. 2018, 17, 162–173. [Google Scholar] [CrossRef]
- Rommer, P.S.; Zettl, U.K. Applying the 2017 McDonald diagnostic criteria for multiple sclerosis. Lancet Neurol. 2018, 17, 497–498. [Google Scholar] [CrossRef]
- Lublin, F.D.; Reingold, S.C.; Cohen, J.A.; Cutter, G.R.; Sørensen, P.S.; Thompson, A.J.; Wolinsky, J.S.; Balcer, L.J.; Banwell, B.; Barkhof, F.; et al. Defining the clinical course of multiple sclerosis: The 2013 revisions. Neurology 2014, 83, 278–286. [Google Scholar] [CrossRef]
- Goodin, D. The epidemiology of multiple sclerosis. Handb. Clin. Neurol. 2016, 138, 173–206. [Google Scholar] [CrossRef]
- Confavreux, C.; Vukusic, S. The clinical course of multiple sclerosis. Handb. Clin. Neurol. 2014, 122, 343–369. [Google Scholar] [CrossRef]
- Galea, I.; Ward-Abel, N.; Heesen, C. Relapse in multiple sclerosis. BMJ 2015, 350, h1765. [Google Scholar] [CrossRef]
- Blechinger, S.; Ehler, J.; Bsteh, G.; Winkelmann, A.; Leutmezer, F.; Meister, S.; Santer, A.; Hecker, M.; Berger, T.; Rommer, P.; et al. Therapeutic plasma exchange in steroid-refractory multiple sclerosis relapses. A retrospective two-center study. Ther. Adv. Neurol. Disord. 2021, 14, 756286420975642. [Google Scholar] [CrossRef] [PubMed]
- Rommer, P.S.; Milo, R.; Han, M.H.; Satyanarayan, S.; Sellner, J.; Hauer, L.; Illes, Z.; Warnke, C.; Laurent, S.; Weber, M.S.; et al. Immunological Aspects of Approved MS Therapeutics. Front. Immunol. 2019, 10, 1564. [Google Scholar] [CrossRef] [PubMed]
- Hauser, S.L.; Cree, B.A. Treatment of Multiple Sclerosis: A Review. Am. J. Med. 2020, 133, 1380–1390.e2. [Google Scholar] [CrossRef]
- Moiola, L.; Rommer, P.S.; Zettl, U.K. Prevention and management of adverse effects of disease modifying treatments in multiple sclerosis. Curr. Opin. Neurol. 2020, 33, 286–294.e2. [Google Scholar] [CrossRef] [PubMed]
- Rotstein, D.; Montalban, X. Reaching an evidence-based prognosis for personalized treatment of multiple sclerosis. Nat. Rev. Neurol. 2019, 15, 287–300. [Google Scholar] [CrossRef] [PubMed]
- Ellenberger, D.; Flachenecker, P.; Haas, J.; Hellwig, K.; Paul, F.; Stahmann, A.; Warnke, C.; Zettl, U.K.; Rommer, P.S. Is benign MS really benign? What a meaningful classification beyond the EDSS must take into consideration. Mult. Scler. Relat. Disord. 2020, 46, 102485. [Google Scholar] [CrossRef]
- Dendrou, C.A.; Fugger, L.; Friese, M.A. Immunopathology of multiple sclerosis. Nat. Rev. Immunol. 2015, 15, 545–558. [Google Scholar] [CrossRef] [PubMed]
- Cramer, S.; Simonsen, H.; Frederiksen, J.; Rostrup, E.; Larsson, H. Abnormal blood–brain barrier permeability in normal appearing white matter in multiple sclerosis investigated by MRI. NeuroImage Clin. 2014, 4, 182–189. [Google Scholar] [CrossRef]
- Kaskow, B.J.; Baecher-Allan, C. Effector T Cells in Multiple Sclerosis. Cold Spring Harb. Perspect. Med. 2018, 8, a029025. [Google Scholar] [CrossRef]
- Sospedra, M. B cells in multiple sclerosis. Curr. Opin. Neurol. 2018, 31, 256–262. [Google Scholar] [CrossRef]
- Duddy, M.; Niino, M.; Adatia, F.; Hebert, S.; Freedman, M.; Atkins, H.; Kim, H.J.; Bar-Or, A. Distinct Effector Cytokine Profiles of Memory and Naive Human B Cell Subsets and Implication in Multiple Sclerosis. J. Immunol. 2007, 178, 6092–6099. [Google Scholar] [CrossRef]
- Jelcic, I.; Al Nimer, F.; Wang, J.; Lentsch, V.; Planas, R.; Jelčić, I.; Madjovski, A.; Ruhrmann, S.; Faigle, W.; Frauenknecht, K.; et al. Memory B Cells Activate Brain-Homing, Autoreactive CD4+ T Cells in Multiple Sclerosis. Cell 2018, 175, 85–100.e23. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Patterson, K.R.; Bar-Or, A. Reassessing B cell contributions in multiple sclerosis. Nat. Immunol. 2018, 19, 696–707. [Google Scholar] [CrossRef] [PubMed]
- Fraussen, J.; Claes, N.; Van Wijmeersch, B.; van Horssen, J.; Stinissen, P.; Hupperts, R.; Somers, V. B cells of multiple sclerosis patients induce autoreactive proinflammatory T cell responses. Clin. Immunol. 2016, 173, 124–132. [Google Scholar] [CrossRef] [PubMed]
- Oost, W.; Talma, N.; Meilof, J.F.; Laman, J.D. Targeting senescence to delay progression of multiple sclerosis. J. Mol. Med. 2018, 96, 1153–1166. [Google Scholar] [CrossRef] [PubMed]
- Lassmann, H. Pathology and disease mechanisms in different stages of multiple sclerosis. J. Neurol. Sci. 2013, 333, 1–4. [Google Scholar] [CrossRef]
- Sanai, S.A.; Saini, V.; Benedict, R.H.; Zivadinov, R.; Teter, B.E.; Ramanathan, M.; Weinstock-Guttman, B. Aging and multiple sclerosis. Mult. Scler. J. 2016, 22, 717–725. [Google Scholar] [CrossRef] [PubMed]
- Leray, E.; Yaouanq, J.; Le Page, E.; Coustans, M.; Laplaud, D.; Oger, J.; Edan, G. Evidence for a two-stage disability progression in multiple sclerosis. Brain 2010, 133, 1900–1913. [Google Scholar] [CrossRef]
- Vaughn, C.B.; Jakimovski, D.; Kavak, K.S.; Ramanathan, M.; Benedict, R.H.B.; Zivadinov, R.; Weinstock-Guttman, B. Epidemiology and treatment of multiple sclerosis in elderly populations. Nat. Rev. Neurol. 2019, 15, 329–342. [Google Scholar] [CrossRef]
- Ibitoye, R.; Kemp, K.; Rice, C.; Hares, K.; Scolding, N.; Wilkins, A. Oxidative stress-related biomarkers in multiple sclerosis: A review. Biomark. Med. 2016, 10, 889–902. [Google Scholar] [CrossRef]
- Tasset, I.; Agüera, E.; Sanchez-Lopez, F.; Feijóo, M.; Giraldo, A.I.; Cruz, A.H.; Gascón, F.; Túnez, I. Peripheral oxidative stress in relapsing–remitting multiple sclerosis. Clin. Biochem. 2012, 45, 440–444. [Google Scholar] [CrossRef]
- Ljubisavljevic, S.; Stojanovic, I.; Bašić, J.; Pavlovic, D.A. The Validation Study of Neurofilament Heavy Chain and 8-hydroxy-2′-deoxyguanosine as Plasma Biomarkers of Clinical/Paraclinical Activity in First and Relapsing-Remitting Demyelination Acute Attacks. Neurotox. Res. 2016, 30, 530–538. [Google Scholar] [CrossRef] [PubMed]
- Miller, E.; Walczak, A.; Saluk-Bijak, J.; Ponczek, M.B.; Majsterek, I. Oxidative modification of patient’s plasma proteins and its role in pathogenesis of multiple sclerosis. Clin. Biochem. 2012, 45, 26–30. [Google Scholar] [CrossRef] [PubMed]
- Ristori, G.; Laurenti, F.; Stacchini, P.; Gasperini, C.; Buttinelli, C.; Pozzilli, C.; Salvetti, M. Serum amyloid A protein is elevated in relapsing–remitting multiple sclerosis. J. Neuroimmunol. 1998, 88, 9–12. [Google Scholar] [CrossRef]
- Soilu-Hanninen, M.; Koskinen, J.O.; Laaksonen, M.; Hanninen, A.; Lilius, E.-M.; Waris, M. High sensitivity measurement of CRP and disease progression in multiple sclerosis. Neurology 2005, 65, 153–155. [Google Scholar] [CrossRef]
- Hinsinger, G.; Galéotti, N.; Nabholz, N.; Urbach, S.; Rigau, V.; Demattei, C.; Lehmann, S.; Camu, W.; Labauge, P.; Castelnovo, G.; et al. Chitinase 3-like proteins as diagnostic and prognostic biomarkers of multiple sclerosis. Mult. Scler. J. 2015, 21, 1251–1261. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Miralles, F.; Prefasi, D.; García-Merino, A.; Gascón-Giménez, F.; Medrano, N.; Castillo-Villalba, J.; Cubas, L.; Alcalá, C.; Gil-Perotín, S.; Gómez-Ballesteros, R.; et al. CSF chitinase 3-like-1 association with disability of primary progressive MS. Neurol.—Neuroimmunol. Neuroinflam. 2020, 7, e815. [Google Scholar] [CrossRef]
- Makhani, N.; Morrow, S.; Fisk, J.; Evans, C.; Beland, S.; Kulaga, S.; Kingwell, E.; Marriott, J.; Dykeman, J.; Jetté, N.; et al. MS incidence and prevalence in Africa, Asia, Australia and New Zealand: A systematic review. Mult. Scler. Relat. Disord. 2014, 3, 48–60. [Google Scholar] [CrossRef]
- Albor, C.; Du Sautoy, T.; Vanan, N.K.; Turner, B.P.; Boomla, K.; Schmierer, K. Ethnicity and prevalence of multiple sclerosis in east London. Mult. Scler. J. 2016, 23, 36–42. [Google Scholar] [CrossRef]
- Bhigjee, A.; Moodley, K.; Ramkissoon, K. Multiple sclerosis in KwaZulu Natal, South Africa: An epidemiological and clinical study. Mult. Scler. J. 2007, 13, 1095–1099. [Google Scholar] [CrossRef]
- Wallin, M.T.; Culpepper, W.J.; Coffman, P.; Pulaski, S.; Maloni, H.; Mahan, C.M.; Haselkorn, J.K.; Kurtzke, J.F.; Veterans Affairs Multiple Sclerosis Centres of Excellence Epidemiology Group. The Gulf War era multiple sclerosis cohort: Age and incidence rates by race, sex and service. Brain 2012, 135, 1778–1785. [Google Scholar] [CrossRef]
- Ventura, R.E.; Antezana, A.O.; Bacon, T.; Kister, I. Hispanic Americans and African Americans with multiple sclerosis have more severe disease course than Caucasian Americans. Mult. Scler. J. 2017, 23, 1554–1557. [Google Scholar] [CrossRef]
- Marrie, R.A.; Cutter, G.; Tyry, T.; Vollmer, T.; Campagnolo, D. Does multiple sclerosis-associated disability differ between races? Neurology 2006, 66, 1235–1240. [Google Scholar] [CrossRef]
- Hansen, M.E.; Hunt, S.C.; Stone, R.C.; Horvath, K.; Herbig, U.; Ranciaro, A.; Hirbo, J.; Beggs, W.; Reiner, A.P.; Wilson, J.G.; et al. Shorter telomere length in Europeans than in Africans due to polygenetic adaptation. Hum. Mol. Genet. 2016, 25, 2324–2330. [Google Scholar] [CrossRef]
- Hunt, S.C.; Chen, W.; Gardner, J.P.; Kimura, M.; Srinivasan, S.R.; Eckfeldt, J.H.; Berenson, G.S.; Aviv, A. Leukocyte telomeres are longer in AfricanAmericans than in whites: The National Heart, Lung, and Blood Institute Family Heart Study and the Bogalusa Heart Study. Aging Cell 2008, 7, 451–458. [Google Scholar] [CrossRef]
- Codd, V.; Denniff, M.; Swinfield, C.; Warner, S.C.; Papakonstantinou, M.; Sheth, S.; Nanus, D.E.; Budgeon, C.A.; Musicha, C.; Bountziouka, V.; et al. A major population resource of 474,074 participants in UK Biobank to in-vestigate determinants and biomedical consequences of leukocyte telomere length. MedRxiv 2021. [Google Scholar] [CrossRef]
- Okuda, K.; Bardeguez, A.; Gardner, J.P.; Rodriguez, P.; Ganesh, V.; Kimura, M.; Skurnick, J.; Awad, G.; Aviv, A. Telomere Length in the Newborn. Pediatr. Res. 2002, 52, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Voskuhl, R.R. The effect of sex on multiple sclerosis risk and disease progression. Mult. Scler. J. 2020, 26, 554–560. [Google Scholar] [CrossRef] [PubMed]
- Rommer, P.S.; Ellenberger, D.; Hellwig, K.; Haas, J.; Pöhlau, D.; Stahmann, A.; Zettl, U.K.; on behalf of the Scientific Advisory Group of the German MS-Register by the German MS Society. Relapsing and progressive MS: The sex-specific perspective. Ther. Adv. Neurol. Disord. 2020, 13, 175628642095649. [Google Scholar] [CrossRef] [PubMed]
- Kalincik, T.; Vivek, V.; Jokubaitis, V.; Lechner-Scott, J.; Trojano, M.; Izquierdo, G.; Lugaresi, A.; Grand’Maison, F.; Hupperts, R.; Oreja-Guevara, C.; et al. Sex as a determinant of relapse incidence and progressive course of multiple sclerosis. Brain 2013, 136, 3609–3617. [Google Scholar] [CrossRef] [PubMed]
- Miclea, A.; Salmen, A.; Zoehner, G.; Diem, L.; Kamm, C.P.; Chaloulos-Iakovidis, P.; Miclea, M.; Briner, M.; Kilidireas, K.; Stefanis, L.; et al. Age-dependent variation of female preponderance across different phenotypes of multiple sclerosis: A retrospective cross-sectional study. CNS Neurosci. Ther. 2019, 25, 527–531. [Google Scholar] [CrossRef]
- Shirani, A.; Zhao, Y.; Kingwell, E.; Rieckmann, P.; Tremlett, H. Temporal trends of disability progression in multiple sclerosis: Findings from British Columbia, Canada (1975–2009). Mult. Scler. J. 2011, 18, 442–450. [Google Scholar] [CrossRef] [PubMed]
- Ribbons, K.A.; McElduff, P.; Boz, C.; Trojano, M.; Izquierdo, G.; Duquette, P.; Girard, M.; Grand’Maison, F.; Hupperts, R.; Grammond, P.; et al. Male Sex Is Independently Associated with Faster Disability Accumulation in Relapse-Onset MS but Not in Primary Progressive MS. PLoS ONE 2015, 10, e0122686. [Google Scholar] [CrossRef]
- Schoonheim, M.M.; Popescu, V.; Lopes, F.C.R.; Wiebenga, O.T.; Vrenken, H.; Douw, L.; Polman, C.H.; Geurts, J.J.; Barkhof, F. Subcortical atrophy and cognition: Sex effects in multiple sclerosis. Neurology 2012, 79, 1754–1761. [Google Scholar] [CrossRef] [PubMed]
- Savettieri, G.; Messina, D.; Andreoli, V.; Bonavita, S.; Caltagirone, C.; Cittadella, R.; Farina, D.; Fazio, M.C.; Girlanda, P.; Le Pira, F.; et al. Gender-related effect of clinical and genetic variables on the cognitive impairment in multiple sclerosis. J. Neurol. 2004, 251, 1208–1214. [Google Scholar] [CrossRef]
- Barrett, E.L.B.; Richardson, D.S. Sex differences in telomeres and lifespan. Aging Cell 2011, 10, 913–921. [Google Scholar] [CrossRef] [PubMed]
- Gardner, M.; Bann, D.; Wiley, L.; Cooper, R.; Hardy, R.; Nitsch, D.; Martin-Ruiz, C.; Shiels, P.; Sayer, A.A.; Barbieri, M.; et al. Gender and telomere length: Systematic review and meta-analysis. Exp. Gerontol. 2014, 51, 15–27. [Google Scholar] [CrossRef]
- Stindl, R. Tying it all together: Telomeres, sexual size dimorphism and the gender gap in life expectancy. Med. Hypotheses 2004, 62, 151–154. [Google Scholar] [CrossRef]
- Gomes, N.M.V.; Ryder, O.A.; Houck, M.L.; Charter, S.J.; Walker, W.; Forsyth, N.R.; Austad, S.N.; Venditti, C.; Pagel, M.; Shay, J.W.; et al. Comparative biology of mammalian telomeres: Hypotheses on ancestral states and the roles of telomeres in longevity determination. Aging Cell 2011, 10, 761–768. [Google Scholar] [CrossRef]
- Ysrraelit, M.C.; Correale, J. Impact of sex hormones on immune function and multiple sclerosis development. Immunology 2019, 156, 9–22. [Google Scholar] [CrossRef]
- Turner, K.J.; Vasu, V.; Griffin, D.K. Telomere Biology and Human Phenotype. Cells 2019, 8, 73. [Google Scholar] [CrossRef]
- Chitnis, T. Role of puberty in multiple sclerosis risk and course. Clin. Immunol. 2013, 149, 192–200. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, B.M.; Noyce, A.J.; Bestwick, J.; Belete, D.; Giovannoni, G.; Dobson, R. Gene-Environment Interactions in Multiple Sclerosis. Neurol.—Neuroimmunol. Neuroinflam. 2021, 8, e1007. [Google Scholar] [CrossRef]
- Nguyen, A.-L.; Eastaugh, A.; van der Walt, A.; Jokubaitis, V.G. Pregnancy and multiple sclerosis: Clinical effects across the lifespan. Autoimmun. Rev. 2019, 18, 102360. [Google Scholar] [CrossRef] [PubMed]
- Confavreux, C.; Hutchinson, M.; Hours, M.M.; Cortinovis-Tourniaire, P.; Moreau, T. Rate of Pregnancy-Related Relapse in Multiple Sclerosis. N. Engl. J. Med. 1998, 339, 285–291. [Google Scholar] [CrossRef] [PubMed]
- Collongues, N.; Patte-Mensah, C.; De Seze, J.; Mensah-Nyagan, A.-G.; Derfuss, T. Testosterone and estrogen in multiple sclerosis: From pathophysiology to therapeutics. Exp. Rev. Neurother. 2018, 18, 515–522. [Google Scholar] [CrossRef] [PubMed]
- Voskuhl, R.R.; Wang, H.; Wu, T.C.J.; Sicotte, N.L.; Nakamura, K.; Kurth, F.; Itoh, N.; Bardens, J.; Bernard, J.T.; Corboy, J.R.; et al. Estriol combined with glatiramer acetate for women with relapsing-remitting multiple sclerosis: A randomised, placebo-controlled, phase 2 trial. Lancet Neurol. 2016, 15, 35–46. [Google Scholar] [CrossRef]
- Kyo, S.; Takakura, M.; Kanaya, T.; Zhuo, W.; Fujimoto, K.; Nishio, Y.; Orimo, A.; Inoue, M. Estrogen activates telomerase. Cancer Res. 1999, 59, 5917–5921. [Google Scholar]
- Lin, J.; Kroenke, C.H.; Epel, E.; Kenna, H.A.; Wolkowitz, O.M.; Blackburn, E.; Rasgon, N.L. Greater endogenous estrogen exposure is associated with longer telomeres in postmenopausal women at risk for cognitive decline. Brain Res. 2011, 1379, 224–231. [Google Scholar] [CrossRef]
- Yeap, B.B.; Knuiman, M.; Divitini, M.L.; Hui, J.; Arscott, G.M.; Handelsman, D.J.; McLennan, S.V.; Twigg, S.M.; McQuillan, B.; Hung, J.; et al. Epidemiological and Mendelian Randomization Studies of Dihydrotestosterone and Estradiol and Leukocyte Telomere Length in Men. J. Clin. Endocrinol. Metab. 2016, 101, 1299–1306. [Google Scholar] [CrossRef]
- Pakpoor, J.; Goldacre, R.; Schmierer, K.; Giovannoni, G.; Goldacre, M.J. Testicular hypofunction and multiple sclerosis risk: A record-linkage study. Ann. Neurol. 2014, 76, 625–628. [Google Scholar] [CrossRef]
- Bove, R.; Musallam, A.; Healy, B.; Raghavan, K.; Glanz, B.; Bakshi, R.; Weiner, H.; De Jager, P.; Miller, K.; Chitnis, T. Low testosterone is associated with disability in men with multiple sclerosis. Mult. Scler. J. 2014, 20, 1584–1592. [Google Scholar] [CrossRef] [PubMed]
- Fang, C.; Huang, H.; Zhang, Q.; Wang, N.; Jing, X.; Guo, J.; Ferianc, M.; Xu, Z. Relation between sex hormones and leucocyte telomere length in men with idiopathic pulmonary fibrosis. Respirology 2020, 25, 1265–1273. [Google Scholar] [CrossRef] [PubMed]
- O’Gorman, C.; Lin, R.; Stankovich, J.; Broadley, S.A. Modelling Genetic Susceptibility to Multiple Sclerosis with Family Data. Neuroepidemiology 2012, 40, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Hensiek, A.E.; Seaman, S.R.; Barcellos, L.F.; Oturai, A.; Eraksoi, M.; Cocco, E.; Vecsei, L.; Stewart, G.; Dubois, B.; Bellman-Strobl, J.; et al. Familial effects on the clinical course of multiple sclerosis. Neurology 2007, 68, 376–383. [Google Scholar] [CrossRef] [PubMed]
- Delgado, D.A.; Zhang, C.; Gleason, K.; Demanelis, K.; Chen, L.S.; Gao, J.; Roy, S.; Shinkle, J.; Sabarinathan, M.; Argos, M.; et al. The contribution of parent-to-offspring transmission of telomeres to the heritability of telomere length in humans. Qual. Life Res. 2019, 138, 49–60. [Google Scholar] [CrossRef] [PubMed]
- Kalmbach, K.; Robinson, L.G.; Wang, F.; Liu, L.; Keefe, D. Telomere Length Reprogramming in Embryos and Stem Cells. BioMed Res. Int. 2014, 2014, 1–7. [Google Scholar] [CrossRef]
- Kim, J.-H.; Nam, C.M.; Lee, D.; Bang, H.; Ko, J.-H.; Lim, I.; Kim, G.J.; Koes, B.W.; Lee, D.-C. Heritability of telomere length across three generations of Korean families. Pediatr. Res. 2019, 87, 1060–1065. [Google Scholar] [CrossRef]
- Hjelmborg, J.B.; Dalgård, C.; Möller, S.; Steenstrup, T.; Kimura, M.; Christensen, K.; Kyvik, K.O.; Aviv, A. The heritability of leucocyte telomere length dynamics. J. Med. Genet. 2015, 52, 297–302. [Google Scholar] [CrossRef]
- Faul, J.D.; Mitchell, C.M.; Smith, J.A.; Zhao, W. Estimating Telomere Length Heritability in an Unrelated Sample of Adults: Is Heritability of Telomere Length Modified by Life Course Socioeconomic Status? Biodemography Soc. Biol. 2016, 62, 73–86. [Google Scholar] [CrossRef][Green Version]
- Lincoln, M.R.; Montpetit, A.; Cader, Z.; Saarela, J.; Dyment, D.A.; Tiislar, M.; Ferretti, V.; Tienari, P.J.; Sadovnick, A.D.; Peltonen, L.; et al. A predominant role for the HLA class II region in the association of the MHC region with multiple sclerosis. Nat. Genet. 2005, 37, 1108–1112. [Google Scholar] [CrossRef]
- The International Multiple Sclerosis Genetics Consortium. Class II HLA interactions modulate genetic risk for multiple sclerosis. Nat. Genet. 2015, 47, 1107–1113. [Google Scholar] [CrossRef] [PubMed]
- Hollenbach, J.A.; Oksenberg, J.R. The immunogenetics of multiple sclerosis: A comprehensive review. J. Autoimmun. 2015, 64, 13–25. [Google Scholar] [CrossRef]
- Yates, R.L.; Esiri, M.M.; Palace, J.; Mittal, A.; DeLuca, G.C. The influence of HLA-DRB1*15 on motor cortical pathology in multiple sclerosis. Neuropathol. Appl. Neurobiol. 2015, 41, 371–384. [Google Scholar] [CrossRef] [PubMed]
- Okuda, D.; Srinivasan, R.; Oksenberg, J.R.; Goodin, D.S.; Baranzini, S.E.; Beheshtian, A.; Waubant, E.; Zamvil, S.S.; Leppert, D.; Qualley, P.; et al. Genotype–Phenotype correlations in multiple sclerosis: HLA genes influence disease severity inferred by 1HMR spectroscopy and MRI measures. Brain 2009, 132, 250–259. [Google Scholar] [CrossRef] [PubMed]
- Baranzini, S.E.; Oksenberg, J.R. The Genetics of Multiple Sclerosis: From 0 to 200 in 50 Years. Trends Genet. 2017, 33, 960–970. [Google Scholar] [CrossRef]
- Patsopoulos, N.A.; Baranzini, S.E.; Santaniello, A.; Shoostari, P.; Cotsapas, C.; Wong, G. Multiple sclerosis genomic map implicates peripheral immune cells and microglia in susceptibility. Science 2019, 365, eaav7188. [Google Scholar] [CrossRef]
- Pan, G.; Simpson, S.; Van Der Mei, I.; Charlesworth, J.; Lucas, R.; Ponsonby, A.-L.; Zhou, Y.; Wu, F.; Taylor, B.V. Role of genetic susceptibility variants in predicting clinical course in multiple sclerosis: A cohort study. J. Neurol. Neurosurg. Psychiatry 2016, 87, 1204–1211. [Google Scholar] [CrossRef]
- Mamutse, G.; Woolmore, J.; Pye, E.; Partridge, J.; Boggild, M.; Young, C.; Fryer, A.; Hoban, P.R.; Rukin, N.; Alldersea, J.; et al. Vitamin D receptor gene polymorphism is associated with reduced disability in multiple sclerosis. Mult. Scler. J. 2008, 14, 1280–1283. [Google Scholar] [CrossRef] [PubMed]
- International Multiple Sclerosis Genetics Consortium. Genome-wide association study of severity in multiple sclerosis. Genes Immun. 2011, 12, 615–625. [Google Scholar] [CrossRef] [PubMed]
- George, M.F.; Briggs, F.B.; Shao, X.; Gianfrancesco, M.A.; Kockum, I.; Harbo, H.F.; Celius, E.G.; Bos, S.D.; Hedström, A.; Shen, L.; et al. Multiple sclerosis risk loci and disease severity in 7,125 individuals from 10 studies. Neurol. Genet. 2016, 2, e87. [Google Scholar] [CrossRef] [PubMed]
- Schönland, S.; Lopez, C.; Widmann, T.; Zimmer, J.; Bryl, E.; Goronzy, J.J.; Weyand, C.M. Premature telomeric loss in rheumatoid arthritis is genetically determined and involves both myeloid and lymphoid cell lineages. Proc. Natl. Acad. Sci. USA 2003, 100, 13471–13476. [Google Scholar] [CrossRef] [PubMed]
- Codd, V.; Nelson, C.; Albrecht, E.; Mangino, M.; Deelen, J.; Buxton, J.L.; Hottenga, J.J.; Fischer, K.; Esko, T.; Surakka, I.; et al. Identification of seven loci affecting mean telomere length and their association with disease. Nat. Genet. 2013, 45, 422–427. [Google Scholar] [CrossRef]
- Delgado, D.A.; Zhang, C.; Chen, L.S.; Gao, J.; Roy, S.; Shinkle, J.; Sabarinathan, M.; Argos, M.; Tong, L.; Ahmed, A.; et al. Genome-wide association study of telomere length among South Asians identifies a second RTEL1 association signal. J. Med. Genet. 2017, 55, 64–71. [Google Scholar] [CrossRef]
- Liu, H.; Yang, Y.; Ge, Y.; Liu, J.; Zhao, Y. TERC promotes cellular inflammatory response independent of telomerase. Nucl. Acids Res. 2019, 47, 8084–8095. [Google Scholar] [CrossRef]
- Garcia-Segura, L.M.; Veiga, S.; Sierra, A.; Melcangi, C.R.; Azcoitia, I. Aromatase: A neuroprotective enzyme. Prog. Neurobiol. 2003, 71, 31–41. [Google Scholar] [CrossRef]
- Hart, P.H.; Gorman, S. Exposure to UV Wavelengths in Sunlight Suppresses Immunity. To What Extent is UV-induced Vitamin D3 the Mediator Responsible? Clin. Biochem. Rev. 2013, 34, 3–13. [Google Scholar]
- Maglio, D.H.G.; Paz, M.L.; Leoni, J. Sunlight Effects on Immune System: Is There Something Else in addition to UV-Induced Immunosuppression? BioMed Res. Int. 2016, 2016, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Dobson, R.; Giovannoni, G.; Ramagopalan, S. The month of birth effect in multiple sclerosis: Systematic review, meta-analysis and effect of latitude. J. Neurol. Neurosurg. Psychiatry 2012, 84, 427–432. [Google Scholar] [CrossRef] [PubMed]
- Lucas, R.M.; Byrne, S.N.; Correale, J.; Ilschner, S.; Hart, P.H. Ultraviolet radiation, vitamin D and multiple sclerosis. Neurodegener. Dis. Manag. 2015, 5, 413–424. [Google Scholar] [CrossRef] [PubMed]
- Tremlett, H.; Zhu, F.; Ascherio, A.; Munger, K.L. Sun exposure over the life course and associations with multiple sclerosis. Neurology 2018, 90, e1191–e1199. [Google Scholar] [CrossRef]
- Orton, S.-M.; Wald, L.; Confavreux, C.; Vukusic, S.; Krohn, J.P.; Ramagopalan, S.V.; Herrera, B.M.; Sadovnick, A.D.; Ebers, G.C. Association of UV radiation with multiple sclerosis prevalence and sex ratio in France. Neurology 2011, 76, 425–431. [Google Scholar] [CrossRef] [PubMed]
- Ostkamp, P.; Salmen, A.; Pignolet, B.; Görlich, D.; Andlauer, T.F.M.; Schulte-Mecklenbeck, A.; Gonzalez-Escamilla, G.; Bucciarelli, F.; Gennero, I.; Breuer, J.; et al. Sunlight exposure exerts immunomodulatory effects to reduce multiple sclerosis severity. Proc. Natl. Acad. Sci. USA 2021, 118, e2018457118. [Google Scholar] [CrossRef] [PubMed]
- Rochette, P.; Brash, D.E. Human Telomeres Are Hypersensitive to UV-Induced DNA Damage and Refractory to Repair. PLoS Genet. 2010, 6, e1000926. [Google Scholar] [CrossRef]
- Ma, H.-M.; Liu, W.; Zhang, P.; Yuan, X.-Y. Human Skin Fibroblast Telomeres are Shortened after Ultraviolet Irradiation. J. Int. Med. Res. 2012, 40, 1871–1877. [Google Scholar] [CrossRef]
- Ikeda, H.; Aida, J.; Hatamochi, A.; Hamasaki, Y.; Izumiyama-Shimomura, N.; Nakamura, K.-I.; Ishikawa, N.; Poon, S.S.; Fujiwara, M.; Tomita, K.-I.; et al. Quantitative fluorescence in situ hybridization measurement of telomere length in skin with/without sun exposure or actinic keratosis. Hum. Pathol. 2014, 45, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Choi, S.; Roh, W.; Lee, J.; Kim, T.-G. Cellular Senescence and Inflammaging in the Skin Microenvironment. Int. J. Mol. Sci. 2021, 22, 3849. [Google Scholar] [CrossRef] [PubMed]
- Abrahamyan, S.; Eberspächer, B.; Hoshi, M.-M.; Aly, L.; Luessi, F.; Groppa, S.; Klotz, L.; Meuth, S.G.; Schroeder, C.; Grüter, T.; et al. Complete Epstein-Barr virus seropositivity in a large cohort of patients with early multiple sclerosis. J. Neurol. Neurosurg. Psychiatry 2020, 91, 681–686. [Google Scholar] [CrossRef]
- Levin, L.I.; Munger, K.M.; O’Reilly, E.J.; Falk, K.I.; Ascherio, A. Primary infection with the epstein-barr virus and risk of multiple sclerosis. Ann. Neurol. 2010, 67, 824–830. [Google Scholar] [CrossRef]
- Belbasis, L.; Bellou, V.; Evangelou, E.; Ioannidis, J.P.; Tzoulaki, I. Environmental risk factors and multiple sclerosis: An umbrella review of systematic reviews and meta-analyses. Lancet Neurol. 2015, 14, 263–273. [Google Scholar] [CrossRef]
- Langer-Gould, A.; Wu, J.; Lucas, R.; Smith, J.; Gonzales, E.; Amezcua, L.; Haraszti, S.; Chen, L.H.; Quach, H.; James, J.A.; et al. Epstein-Barr virus, cytomegalovirus, and multiple sclerosis susceptibility. Neurology 2017, 89, 1330–1337. [Google Scholar] [CrossRef]
- Farrell, R.A.; Antony, D.; Wall, G.R.; Clark, D.A.; Fisniku, L.; Swanton, J.; Khaleeli, Z.; Schmierer, K.; Miller, D.H.; Giovannoni, G. Humoral immune response to EBV in multiple sclerosis is associated with disease activity on MRI. Neurology 2009, 73, 32–38. [Google Scholar] [CrossRef]
- Lunemann, J.; Tintore, M.; Messmer, B.; Strowig, T.; Rovira, Á.; Perkal, H.; Caballero, E.; Münz, C.; Montalban, X.; Comabella, M. Elevated Epstein-Barr virus-encoded nuclear antigen-1 immune responses predict conversion to multiple sclerosis. Ann. Neurol. 2009, 67, 159–169. [Google Scholar] [CrossRef] [PubMed]
- Ingram, G.; Bugert, J.; Loveless, S.; Robertson, N.P. Anti-EBNA-1 IgG is not a reliable marker of multiple sclerosis clinical disease activity. Eur. J. Neurol. 2010, 17, 1386–1389. [Google Scholar] [CrossRef]
- Kamranvar, S.A.; Masucci, M.G. Regulation of Telomere Homeostasis during Epstein-Barr virus Infection and Immortalization. Viruses 2017, 9, 217. [Google Scholar] [CrossRef] [PubMed]
- Plunkett, F.J.; Soares, M.V.D.; Annels, N.; Hislop, A.; Ivory, K.; Lowdell, M.; Salmon, M.; Rickinson, A.; Akbar, A.N. The flow cytometric analysis of telomere length in antigen-specific CD8+ T cells during acute Epstein-Barr virus infection. Blood 2001, 97, 700–707. [Google Scholar] [CrossRef] [PubMed]
- Dowd, J.B.; Bosch, J.A.; Steptoe, A.; Jayabalasingham, B.; Lin, J.; Yolken, R.; Aiello, A.E. Persistent Herpesvirus Infections and Telomere Attrition Over 3 Years in the Whitehall II Cohort. J. Infect. Dis. 2017, 216, 565–572. [Google Scholar] [CrossRef]
- Engdahl, E.; Gustafsson, R.; Huang, J.; Biström, M.; Lima Bomfim, I.; Stridh, P.; Khademi, M.; Brenner, N.; Butt, J.; Michel, A.; et al. Increased Serological Response Against Human Herpesvirus 6A Is Associated with Risk for Multiple Sclerosis. Front. Immunol. 2019, 10, 2715. [Google Scholar] [CrossRef]
- Simpson, J.S.; Taylor, B.; Dwyer, D.E.; Taylor, J.; Blizzard, L.; Ponsonby, A.-L.; Pittas, F.; Dwyer, T.; van der Mei, I. Anti-HHV-6 IgG titer significantly predicts subsequent relapse risk in multiple sclerosis. Mult. Scler. J. 2012, 18, 799–806. [Google Scholar] [CrossRef]
- Arbuckle, J.H.; Medveczky, M.M.; Luka, J.; Hadley, S.H.; Luegmayr, A.; Ablashi, D.; Lund, T.C.; Tolar, J.; De Meirleir, K.; Montoya, J.G.; et al. The latent human herpesvirus-6A genome specifically integrates in telomeres of human chromosomes in vivo and in vitro. Proc. Natl. Acad. Sci. USA 2010, 107, 5563–5568. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Hidalgo-Bravo, A.; Zhang, E.; Cotton, V.E.; Mendez-Bermudez, A.; Wig, G.; Medina-Calzada, Z.; Neumann, R.; Jeffreys, A.J.; Winney, B.; et al. Human telomeres that carry an integrated copy of human herpesvirus 6 are often short and unstable, facilitating release of the viral genome from the chromosome. Nucl. Acids Res. 2013, 42, 315–327. [Google Scholar] [CrossRef]
- Zuhair, M.; Smit, G.S.A.; Wallis, G.; Jabbar, F.; Smith, C.; Devleesschauwer, B.; Griffiths, P. Estimation of the worldwide seroprevalence of cytomegalovirus: A systematic review and meta-analysis. Rev. Med Virol. 2019, 29, e2034. [Google Scholar] [CrossRef]
- Sundqvist, E.; Bergström, T.; Daialhosein, H.; Nyström, M.; Sundström, P.; Hillert, J.; Alfredsson, L.; Kockum, I.; Olsson, T. Cytomegalovirus seropositivity is negatively associated with multiple sclerosis. Mult. Scler. J. 2013, 20, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Horáková, D.; Zivadinov, R.; Weinstock-Guttman, B.; Havrdova, E.K.; Qu, J.; Tamaño-Blanco, M.; Badgett, D.; Tyblova, M.; Bergsland, N.; Hussein, S.; et al. Environmental Factors Associated with Disease Progression after the First Demyelinating Event: Results from the Multi-Center SET Study. PLoS ONE 2013, 8, e53996. [Google Scholar] [CrossRef] [PubMed]
- Zivadinov, R.; Nasuelli, D.; Tommasi, M.A.; Serafin, M.; Bratina, A.; Ukmar, M.; Pirko, I.; Johnson, A.J.; Furlan, C.; Pozzi-Mucelli, R.S.; et al. Positivity of cytomegalovirus antibodies predicts a better clinical and radiological outcome in multiple sclerosis patients. Neurol. Res. 2006, 28, 262–269. [Google Scholar] [CrossRef] [PubMed]
- Van de Berg, P.J.E.J.; Griffiths, S.J.; Yong, S.-L.; Macaulay, R.; Bemelman, F.J.; Jackson, S.; Henson, S.M.; Berge, I.J.M.T.; Akbar, A.N.; van Lier, R.A.W. Cytomegalovirus Infection Reduces Telomere Length of the Circulating T Cell Pool. J. Immunol. 2010, 184, 3417–3423. [Google Scholar] [CrossRef]
- Fletcher, J.; Vukmanovic-Stejic, M.; Dunne, P.; Birch, K.E.; Cook, J.E.; Jackson, S.E.; Salmon, M.; Rustin, M.H.; Akbar, A.N. Cytomegalovirus-Specific CD4+T Cells in Healthy Carriers Are Continuously Driven to Replicative Exhaustion. J. Immunol. 2005, 175, 8218–8225. [Google Scholar] [CrossRef]
- Ranjbar, R.; Karampoor, S.; Jalilian, F.A. The protective effect of Helicobacter Pylori infection on the susceptibility of multiple sclerosis. J. Neuroimmunol. 2019, 337, 577069. [Google Scholar] [CrossRef]
- Kountouras, J.; Papaefthymiou, A.; Gavalas, E.; Polyzos, S.A.; Boziki, M.; Kyriakou, P.; Katsinelos, P.; Zavos, C.; Liatsos, C.; Tzivras, D.; et al. Helicobacter pylori infection as a potential risk factor for multiple sclerosis. Med. Hypotheses 2020, 143, 110135. [Google Scholar] [CrossRef]
- Huang, J.; Xie, C.; Niu, Z.; He, L.; Li, J. The relation between Helicobacter pylori immunoglobulin G seropositivity and leukocyte telomere length in US adults from NHANES 1999–2000. Helicobacter 2020, 25, e12760. [Google Scholar] [CrossRef] [PubMed]
- Noppert, G.A.; Feinstein, L.; Dowd, J.B.; Stebbins, R.C.; Zang, E.; Needham, B.L.; Meier, H.C.S.; Simanek, A.; Aiello, A.E. Pathogen burden and leukocyte telomere length in the United States. Immun. Ageing 2020, 17, 36. [Google Scholar] [CrossRef] [PubMed]
- Farahmandfard, M.A.; Naghibzadeh-Tahami, A.; Khanjani, N. Ambient air pollution and multiple sclerosis: A systematic review. Rev. Environ. Health 2021. [Google Scholar] [CrossRef]
- Türk Börü, Ü.; Bölük, C.; Taşdemir, M.; Gezer, T.; Serim, V.A. Air pollution, a possible risk factor for multiple sclerosis. Acta Neurol. Scand. 2020, 141, 431–437. [Google Scholar] [CrossRef]
- Tateo, F.; Grassivaro, F.; Ermani, M.; Puthenparampil, M.; Gallo, P. PM2.5 levels strongly associate with multiple sclerosis prevalence in the Province of Padua, Veneto Region, North-East Italy. Mult. Scler. J. 2018, 25, 1719–1727. [Google Scholar] [CrossRef] [PubMed]
- Angelici, L.; Piola, M.; Cavalleri, T.; Randi, G.; Cortini, F.; Bergamaschi, R.; Baccarelli, A.A.; Bertazzi, P.A.; Pesatori, A.C.; Bollati, V. Effects of particulate matter exposure on multiple sclerosis hospital admission in Lombardy region, Italy. Environ. Res. 2016, 145, 68–73. [Google Scholar] [CrossRef]
- Zhao, B.; Vo, H.Q.; Johnston, F.H.; Negishi, K. Air pollution and telomere length: A systematic review of 12,058 subjects. Cardiovasc. Diagn. Ther. 2018, 8, 480–492. [Google Scholar] [CrossRef]
- Bijnens, E.M.; Zeegers, M.P.; Derom, C.; Martens, D.S.; Gielen, M.; Hageman, G.J.; Plusquin, M.; Thiery, E.; Vlietinck, R.; Nawrot, T.S. Telomere tracking from birth to adulthood and residential traffic exposure. BMC Med. 2017, 15, 205. [Google Scholar] [CrossRef] [PubMed]
- Clemente, D.B.; Vrijheid, M.; Martens, D.S.; Bustamante, M.; Chatzi, L.; Danileviciute, A.; De Castro, M.; Grazuleviciene, R.; Gutzkow, K.B.; Lepeule, J.; et al. Prenatal and Childhood Traffic-Related Air Pollution Exposure and Telomere Length in European Children: The HELIX Project. Environ. Health Perspect. 2019, 127, 87001. [Google Scholar] [CrossRef]
- Lee, H.A.; Lee, W.K.; Kong, K.-A.; Chang, N.; Ha, E.-H.; Hong, Y.S.; Park, H. The Effect of Eating Behavior on Being Overweight or Obese During Preadolescence. J. Prev. Med. Public Health 2011, 44, 226–233. [Google Scholar] [CrossRef] [PubMed]
- Hedström, A.K.; Olsson, T.; Alfredsson, L. High body mass index before age 20 is associated with increased risk for multiple sclerosis in both men and women. Mult. Scler. J. 2012, 18, 1334–1336. [Google Scholar] [CrossRef]
- Langer-Gould, A.; Brara, S.M.; Beaber, B.E.; Koebnick, C. Childhood obesity and risk of pediatric multiple sclerosis and clinically isolated syndrome. Neurology 2013, 80, 548–552. [Google Scholar] [CrossRef]
- Mokry, L.E.; Ross, S.; Timpson, N.J.; Sawcer, S.; Smith, G.D.; Richards, J.B. Obesity and Multiple Sclerosis: A Mendelian Randomization Study. PLoS Med. 2016, 13, e1002053. [Google Scholar] [CrossRef]
- Hedström, A.K.; Bomfim, I.L.; Barcellos, L.; Gianfrancesco, M.; Schaefer, C.; Kockum, I.; Olsson, T.; Alfredsson, L. Interaction between adolescent obesity and HLA risk genes in the etiology of multiple sclerosis. Neurology 2014, 82, 865–872. [Google Scholar] [CrossRef]
- Bove, R.; Musallam, A.; Xia, Z.; Baruch, N.; Messina, S.; Healy, B.; Chitnis, T. Longitudinal BMI trajectories in multiple sclerosis: Sex differences in association with disease severity. Mult. Scler. Relat. Disord. 2016, 8, 136–140. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.; Martin, H.; Firpo, M.A.; Demerath, E.W. Inverse association between adiposity and telomere length: The fels longitudinal study. Am. J. Hum. Biol. 2011, 23, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Buxton, J.L.; Walters, R.G.; Visvikis-Siest, S.; Meyre, D.; Froguel, P.; Blakemore, A.I.F. Childhood Obesity Is Associated with Shorter Leukocyte Telomere Length. J. Clin. Endocrinol. Metab. 2011, 96, 1500–1505. [Google Scholar] [CrossRef]
- Mundstock, E.; Sarria, E.E.; Zatti, H.; Mattos Louzada, F.; Kich Grun, L.; Herbert Jones, M.; Guma, F.T.; Mazzola In Memoriam, J.; Epifanio, M.; Stein, R.T.; et al. Effect of obesity on telomere length: Systematic review and meta-analysis. Obesity 2015, 23, 2165–2174. [Google Scholar] [CrossRef]
- Castro, A.; la Concha, L.M.-D.; Pantoja-Meléndez, C. Low-grade inflammation and its relation to obesity and chronic degenerative diseases. Rev. Méd. Hosp. General México 2017, 80, 101–105. [Google Scholar] [CrossRef]
- Evans, E.; Levasseur, V.; Cross, A.H.; Piccio, L. An overview of the current state of evidence for the role of specific diets in multiple sclerosis. Mult. Scler. Relat. Disord. 2019, 36, 101393. [Google Scholar] [CrossRef] [PubMed]
- Bäärnhielm, M.; Olsson, T.; Alfredsson, L. Fatty fish intake is associated with decreased occurrence of multiple sclerosis. Mult. Scler. J. 2013, 20, 726–732. [Google Scholar] [CrossRef]
- AlAmmar, W.A.; Albeesh, F.H.; Ibrahim, L.M.; Algindan, Y.Y.; Yamani, L.Z.; Khattab, R.Y. Effect of omega-3 fatty acids and fish oil supplementation on multiple sclerosis: A systematic review. Nutr. Neurosci. 2021, 24, 569–579. [Google Scholar] [CrossRef]
- Azary, S.; Schreiner, T.; Graves, J.; Waldman, A.; Belman, A.; Guttman, B.W.; Aaen, G.; Tillema, J.-M.; Mar, S.; Hart, J.; et al. Contribution of dietary intake to relapse rate in early paediatric multiple sclerosis. J. Neurol. Neurosurg. Psychiatry 2018, 89, 28–33. [Google Scholar] [CrossRef]
- Farez, M.F.; Fiol, M.P.; Gaitán, M.I.; Quintana, F.J.; Correale, J. Sodium intake is associated with increased disease activity in multiple sclerosis. J. Neurol. Neurosurg. Psychiatry 2015, 86, 26–31. [Google Scholar] [CrossRef]
- Waubant, E.; Lucas, R.; Mowry, E.; Graves, J.; Olsson, T.; Alfredsson, L.; Langer-Gould, A. Environmental and genetic risk factors for MS: An integrated review. Ann. Clin. Transl. Neurol. 2019, 6, 1905–1922. [Google Scholar] [CrossRef] [PubMed]
- Mische, L.J.; Mowry, E.M. The Evidence for Dietary Interventions and Nutritional Supplements as Treatment Options in Multiple Sclerosis: A Review. Curr. Treat. Options Neurol. 2018, 20, 8. [Google Scholar] [CrossRef]
- Shahi, S.K.; Freedman, S.N.; Mangalam, A.K. Gut microbiome in multiple sclerosis: The players involved and the roles they play. Gut Microbes 2017, 8, 607–615. [Google Scholar] [CrossRef] [PubMed]
- Caire-Juvera, G.; Navarro-Ibarra, M.J.; Hernández, J. Diet, physical activity and telomere length in adults. Nutr. Hosp. 2019, 36, 1403–1417. [Google Scholar] [CrossRef] [PubMed]
- Meshkani, S.E.; Kooshki, A.; Alahabadi, A.; Najafi, M.L.; Rad, A.; Riahimanesh, F.; Miri, M. Dietary pattern and telomere length in preschool children in a middle-income country. Matern. Child Nutr. 2021, 17, e13146. [Google Scholar] [CrossRef] [PubMed]
- Galiè, S.; Canudas, S.; Muralidharan, J.; García-Gavilán, J.; Bulló, M.; Salas-Salvadó, J. Impact of Nutrition on Telomere Health: Systematic Review of Observational Cohort Studies and Randomized Clinical Trials. Adv. Nutr. 2019, 11, 576–601. [Google Scholar] [CrossRef] [PubMed]
- Welendorf, C.; Nicoletti, C.F.; Pinhel, M.A.D.S.; Noronha, N.; de Paula, B.M.F.; Nonino, C.B. Obesity, weight loss, and influence on telomere length: New insights for personalized nutrition. Nutrition 2019, 66, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Munger, K.L.; Levin, L.I.; Hollis, B.W.; Howard, N.S.; Ascherio, A. Serum 25-Hydroxyvitamin D Levels and Risk of Multiple Sclerosis. JAMA 2006, 296, 2832–2838. [Google Scholar] [CrossRef] [PubMed]
- Munger, K.L.; Åivo, J.; Hongell, K.; Soilu-Hänninen, M.; Surcel, H.-M.; Ascherio, A. Vitamin D Status During Pregnancy and Risk of Multiple Sclerosis in Offspring of Women in the Finnish Maternity Cohort. JAMA Neurol. 2016, 73, 515–519. [Google Scholar] [CrossRef]
- Harroud, A.; Richards, J.B. Mendelian randomization in multiple sclerosis: A causal role for vitamin D and obesity? Mult. Scler. J. 2018, 24, 80–85. [Google Scholar] [CrossRef]
- Jacobs, B.M.; Noyce, A.J.; Giovannoni, G.; Dobson, R. BMI and low vitamin D are causal factors for multiple sclerosis. Neurol.—Neuroimmunol. Neuroinflam. 2020, 7, e662. [Google Scholar] [CrossRef] [PubMed]
- Hedström, A.K.; Olsson, T.; Kockum, I.; Hillert, J.; Alfredsson, L. Low fish consumption is associated with a small increased risk of MS. Neurol.—Neuroimmunol. Neuroinflam. 2020, 7, e717. [Google Scholar] [CrossRef]
- Martínez-Lapiscina, E.H.; Mahatanan, R.; Lee, C.-H.; Charoenpong, P.; Hong, J.-P. Associations of serum 25(OH) vitamin D levels with clinical and radiological outcomes in multiple sclerosis, a systematic review and meta-analysis. J. Neurol. Sci. 2020, 411, 116668. [Google Scholar] [CrossRef]
- Yeh, W.Z.; Gresle, M.; Jokubaitis, V.; Stankovich, J.; Van Der Walt, A.; Butzkueven, H. Immunoregulatory effects and therapeutic potential of vitamin D in multiple sclerosis. Br. J. Pharmacol. 2020, 177, 4113–4133. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.J.; Prescott, J.; Giovannucci, E.; Hankinson, S.E.; Rosner, B.; Han, J.; De Vivo, I. Plasma Vitamin D Biomarkers and Leukocyte Telomere Length. Am. J. Epidemiol. 2013, 177, 1411–1417. [Google Scholar] [CrossRef]
- Vetter, V.M.; Spira, D.; Banszerus, V.L.; DeMuth, I. Epigenetic Clock and Leukocyte Telomere Length Are Associated with Vitamin D Status but not with Functional Assessments and Frailty in the Berlin Aging Study II. Journals Gerontol. Ser. A Boil. Sci. Med. Sci. 2020, 75, 2056–2063. [Google Scholar] [CrossRef]
- Kim, J.-H.; Kim, G.J.; Lee, D.; Ko, J.-H.; Lim, I.; Bang, H.; Koes, B.W.; Seong, B.; Lee, D.-C. Higher maternal vitamin D concentrations are associated with longer leukocyte telomeres in newborns. Matern. Child Nutr. 2018, 14, e12475. [Google Scholar] [CrossRef]
- Borras, M.; Panizo, S.; Sarró, F.; Valdivielso, J.M.; Fernandez, E. Assessment of the Potential Role of Active Vitamin D Treatment in Telomere Length: A Case–Control Study in Hemodialysis Patients. Clin. Ther. 2012, 34, 849–856. [Google Scholar] [CrossRef]
- Dupuis, M.L.; Pagano, M.T.; Pierdominici, M.; Ortona, E. The role of vitamin D in autoimmune diseases: Could sex make the difference? Biol. Sex Differ. 2021, 12, 12. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; He, Z.-Y.; Liu, H.-N. Meta-analysis of the relationship between homocysteine, vitamin B12, folate, and multiple sclerosis. J. Clin. Neurosci. 2011, 18, 933–938. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.; Parks, C.; DeRoo, L.A.; Cawthon, R.M.; Sandler, D.P.; Chen, H. Multivitamin use and telomere length in women. Am. J. Clin. Nutr. 2009, 89, 1857–1863. [Google Scholar] [CrossRef] [PubMed]
- Corina, A.; Rangel-Zuñiga, O.A.; Jiménez-Lucena, R.; Alcalá-Díaz, J.F.; Quintana-Navarro, G.; Yubero-Serrano, E.M.; López-Moreno, J.; Delgado-Lista, J.; Tinahones, F.; Ordovás, J.M.; et al. Low Intake of Vitamin E Accelerates Cellular Aging in Patients with Established Cardiovascular Disease: The CORDIOPREV Study. J. Gerontol. Ser. A Boil. Sci. Med. Sci. 2019, 74, 770–777. [Google Scholar] [CrossRef]
- Hedström, A.K.; Hillert, J.; Olsson, T.; Alfredsson, L. Alcohol as a Modifiable Lifestyle Factor Affecting Multiple Sclerosis Risk. JAMA Neurol. 2014, 71, 300–305. [Google Scholar] [CrossRef] [PubMed]
- Pakpoor, J.; Goldacre, R.; Disanto, G.; Giovannoni, G.; Goldacre, M.J. Alcohol Misuse Disorders and Multiple Sclerosis Risk. JAMA Neurol. 2014, 71, 1188–1189. [Google Scholar] [CrossRef] [PubMed]
- Hedström, A.K.; Mowry, E.; Gianfrancesco, M.; Shao, X.; Schaefer, C.A.; Shen, L.; Olsson, T.; Barcellos, L.F.; Alfredsson, L. High consumption of coffee is associated with decreased multiple sclerosis risk; results from two independent studies. J. Neurol. Neurosurg. Psychiatry 2016, 87, 454–460. [Google Scholar] [CrossRef] [PubMed]
- D’Hooghe, M.B.; Haentjens, P.; Nagels, G.; De Keyser, J. Alcohol, coffee, fish, smoking and disease progression in multiple sclerosis. Eur. J. Neurol. 2011, 19, 616–624. [Google Scholar] [CrossRef] [PubMed]
- Diaz-Cruz, C.; Chua, A.S.; Malik, M.T.; Kaplan, T.; Glanz, B.I.; Egorova, S.; Guttmann, C.R.; Bakshi, R.; Weiner, H.L.; Healy, B.C.; et al. The effect of alcohol and red wine consumption on clinical and MRI outcomes in multiple sclerosis. Mult. Scler. Relat. Disord. 2017, 17, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Navarro-Mateu, F.; Husky, M.; Cayuela-Fuentes, P.; Álvarez, F.; Roca-Vega, A.; Rubio-Aparicio, M.; Chirlaque, M.D.; Cayuela, M.L.; Martínez, S.; Sánchez-Meca, J. The association of telomere length with substance use disorders: A systematic review and meta-analysis of observational studies. Addiction 2021, 116, 1954–1972. [Google Scholar] [CrossRef]
- Liu, J.J.; Crous-Bou, M.; Giovannucci, E.; De Vivo, I. Coffee Consumption Is Positively Associated with Longer Leukocyte Telomere Length in the Nurses’ Health Study. J. Nutr. 2016, 146, 1373–1378. [Google Scholar] [CrossRef]
- Degelman, M.L.; Herman, K.M. Smoking and multiple sclerosis: A systematic review and meta-analysis using the Bradford Hill criteria for causation. Mult. Scler. Relat. Disord. 2017, 17, 207–216. [Google Scholar] [CrossRef] [PubMed]
- Hedström, A.K.; Bomfim, I.L.; Barcellos, L.F.; Briggs, F.; Schaefer, C.; Kockum, I.; Olsson, T.; Alfredsson, L. Interaction between passive smoking and two HLA genes with regard to multiple sclerosis risk. Int. J. Epidemiol. 2014, 43, 1791–1798. [Google Scholar] [CrossRef]
- Paz-Ballesteros, W.C.; Monterrubio-Flores, E.A.; Flores-Rivera, J.D.J.; Corona-Vázquez, T.; Hernández-Girón, C. Cigarette Smoking, Alcohol Consumption and Overweight in Multiple Sclerosis: Disability Progression. Arch. Med. Res. 2017, 48, 113–120. [Google Scholar] [CrossRef]
- Zivadinov, R.; Weinstock-Guttman, B.; Hashmi, K.; Abdelrahman, N.; Stosic, M.; Dwyer, M.; Hussein, S.; Durfee, J.; Ramanathan, M. Smoking is associated with increased lesion volumes and brain atrophy in multiple sclerosis. Neurology 2009, 73, 504–510. [Google Scholar] [CrossRef]
- Ramanujam, R.; Hedström, A.-K.; Manouchehrinia, A.; Alfredsson, L.; Olsson, T.; Bottai, M.; Hillert, J. Effect of Smoking Cessation on Multiple Sclerosis Prognosis. JAMA Neurol. 2015, 72, 1117–1123. [Google Scholar] [CrossRef] [PubMed]
- Astuti, Y.; Wardhana, A.; Watkins, J.; Wulaningsih, W. Cigarette smoking and telomere length: A systematic review of 84 studies and meta-analysis. Environ. Res. 2017, 158, 480–489. [Google Scholar] [CrossRef]
- Valdes, A.; Andrew, T.; Gardner, J.; Kimura, M.; Oelsner, E.; Cherkas, L.; Aviv, A.; Spector, T. Obesity, cigarette smoking, and telomere length in women. Lancet 2005, 366, 662–664. [Google Scholar] [CrossRef]
- Vyas, C.M.; Ogata, S.; Reynolds, C.F.; Mischoulon, D.; Chang, G.; Cook, N.R.; Manson, J.E.; Crous-Bou, M.; De Vivo, I.; Okereke, O.I. Telomere length and its relationships with lifestyle and behavioural factors: Variations by sex and race/ethnicity. Age Ageing 2021, 50, 838–846. [Google Scholar] [CrossRef] [PubMed]
- Wesnes, K.; Myhr, K.-M.; Riise, T.; Cortese, M.; Pugliatti, M.; Boström, I.; Landtblom, A.-M.; Wolfson, C.; Bjornevik, K. Physical activity is associated with a decreased multiple sclerosis risk: The EnvIMS study. Mult. Scler. J. 2017, 24, 150–157. [Google Scholar] [CrossRef]
- Dorans, K.S.; Massa, J.; Chitnis, T.; Ascherio, A.; Munger, K.L. Physical activity and the incidence of multiple sclerosis. Neurology 2016, 87, 1770–1776. [Google Scholar] [CrossRef] [PubMed]
- Cortese, M.; Riise, T.; Bjornevik, K.; Myhr, K.-M.; The Multiple Sclerosis Conscript Service Database Study Group. Body size and physical exercise, and the risk of multiple sclerosis. Mult. Scler. J. 2018, 24, 270–278. [Google Scholar] [CrossRef]
- Rooney, S.; Riemenschneider, M.; Dalgas, U.; Jørgensen, M.-L.K.; Michelsen, A.-S.; Brønd, J.C.; Hvid, L.G. Physical activity is associated with neuromuscular and physical function in patients with multiple sclerosis independent of disease severity. Disabil. Rehabil. 2021, 43, 632–639. [Google Scholar] [CrossRef]
- Kjølhede, T.; Siemonsen, S.; Wenzel, D.; Stellmann, J.-P.; Ringgaard, S.; Pedersen, B.G.; Stenager, E.; Petersen, T.; Vissing, K.; Heesen, C.; et al. Can resistance training impact MRI outcomes in relapsing-remitting multiple sclerosis? Mult. Scler. J. 2017, 24, 1356–1365. [Google Scholar] [CrossRef]
- Dalgas, U.; Langeskov-Christensen, M.; Stenager, E.; Riemenschneider, M.; Hvid, L.G. Exercise as Medicine in Multiple Sclerosis-Time for a Paradigm Shift: Preventive, Symptomatic, and Disease-Modifying Aspects and Perspectives. Curr. Neurol. Neurosci. Rep. 2019, 19, 88. [Google Scholar] [CrossRef] [PubMed]
- Shin, Y.-A. How Does Obesity and Physical Activity Affect Aging?: Focused on Telomere as a Biomarker of Aging. J. Obes. Metab. Syndr. 2019, 28, 92–104. [Google Scholar] [CrossRef]
- Borghini, A.; Giardini, G.; Tonacci, A.; Mastorci, F.; Mercuri, A.; Mrakic-Sposta, S.; Moretti, S.; Andreassi, M.G.; Pratali, L. Chronic and acute effects of endurance training on telomere length. Mutagenesis 2015, 30, 711–716. [Google Scholar] [CrossRef]
- Grant, I.; Brown, G.W.; Harris, T.; McDonald, W.I.; Patterson, T.; Trimble, M.R. Severely threatening events and marked life difficulties preceding onset or exacerbation of multiple sclerosis. J. Neurol. Neurosurg. Psychiatry 1989, 52, 8–13. [Google Scholar] [CrossRef]
- Jiang, X.; Olsson, T.; Hillert, J.; Kockum, I.; Alfredsson, L. Stressful life events are associated with the risk of multiple sclerosis. Eur. J. Neurol. 2020, 27, 2539–2548. [Google Scholar] [CrossRef] [PubMed]
- Riise, T.; Mohr, D.C.; Munger, K.L.; Rich-Edwards, J.W.; Kawachi, I.; Ascherio, A. Stress and the risk of multiple sclerosis. Neurology 2011, 76, 1866–1871. [Google Scholar] [CrossRef]
- Spitzer, C.; Bouchain, M.; Winkler, L.Y.; Wingenfeld, K.; Gold, S.M.; Grabe, H.J.; Barnow, S.; Otte, C.; Heesen, C. Childhood Trauma in Multiple Sclerosis. Psychosom. Med. 2012, 74, 312–318. [Google Scholar] [CrossRef]
- Starkweather, A.R.; Alhaeeri, A.A.; Montpetit, A.; Brumelle, J.; Filler, K.; Montpetit, M.; Mohanraj, L.; Lyon, D.E.; Jackson-Cook, C.K. An Integrative Review of Factors Associated with Telomere Length and Implications for Biobehavioral Research. Nurs. Res. 2014, 63, 36–50. [Google Scholar] [CrossRef]
- Kemp, B.R.; Ferraro, K.F. Are Biological Consequences of Childhood Exposures Detectable in Telomere Length Decades Later? J. Gerontol. Ser. A Boil. Sci. Med. Sci. 2021, 76, 7–14. [Google Scholar] [CrossRef]
- Schutte, N.S.; Malouff, J.M. The Relationship Between Perceived Stress and Telomere Length: A Meta-analysis. Stress Health 2016, 32, 313–319. [Google Scholar] [CrossRef]
- Epel, E.S. Psychological and metabolic stress: A recipe for accelerated cellular aging? Hormones 2009, 8, 7–22. [Google Scholar] [CrossRef]
- Hedström, A.K.; Åkerstedt, T.; Hillert, J.; Olsson, T.; Alfredsson, L. Shift work at young age is associated with increased risk for multiple sclerosis. Ann. Neurol. 2011, 70, 733–741. [Google Scholar] [CrossRef] [PubMed]
- Hedstrom, A.; Åkerstedt, T.; Olsson, T.; Alfredsson, L. Shift work influences multiple sclerosis risk. Mult. Scler. J. 2015, 21, 1195–1199. [Google Scholar] [CrossRef]
- Gustavsen, S.; Søndergaard, H.; Oturai, D.; Laursen, B.; Laursen, J.; Magyari, M.; Ullum, H.; Larsen, M.; Sellebjerg, F. Shift work at young age is associated with increased risk of multiple sclerosis in a Danish population. Mult. Scler. Relat. Disord. 2016, 9, 104–109. [Google Scholar] [CrossRef]
- Damasceno, A.; Moraes, A.S.; Farias, A.; Damasceno, B.P.; dos Santos, L.M.B.; Cendes, F. Disruption of melatonin circadian rhythm production is related to multiple sclerosis severity: A preliminary study. J. Neurol. Sci. 2015, 353, 166–168. [Google Scholar] [CrossRef] [PubMed]
- Ledda, C.; Loreto, C.; Rapisarda, V. Telomere Length as a Biomarker of Biological Aging in Shift Workers. Appl. Sci. 2020, 10, 2764. [Google Scholar] [CrossRef]
- Carugno, M.; Maggioni, C.; Crespi, E.; Bonzini, M.; Cuocina, S.; Dioni, L.; Tarantini, L.; Consonni, D.; Ferrari, L.; Pesatori, A.C. Night Shift Work, DNA Methylation and Telomere Length: An Investigation on Hospital Female Nurses. Int. J. Environ. Res. Public Health 2019, 16, 2292. [Google Scholar] [CrossRef]
- Samulin Erdem, J.; Notø, H.Ø.; Skare, Ø.; Lie, J.S.; Petersen-Øverleir, M.; Reszka, E.; Pepłońska, B.; Zienolddiny, S. Mechanisms of breast cancer risk in shift workers: Association of telomere shortening with the duration and intensity of night work. Cancer Med. 2017, 6, 1988–1997. [Google Scholar] [CrossRef]
- Helby, J.; Nordestgaard, B.G.; Benfield, T.; Bojesen, S.E. Shorter leukocyte telomere length is associated with higher risk of infections: A prospective study of 75,309 individuals from the general population. Haematology 2017, 102, 1457–1465. [Google Scholar] [CrossRef]
- Wang, S.; Madu, C.O.; Lu, Y. Telomere and Its Role in Diseases. Oncomedicine 2019, 4, 1–9. [Google Scholar] [CrossRef]
- Redondo, J.; Sarkar, P.; Kemp, K.; Virgo, P.F.; Pawade, J.; Norton, A.; Emery, D.C.; Guttridge, M.G.; Marks, D.I.; Wilkins, A.; et al. Reduced cellularity of bone marrow in multiple sclerosis with decreased MSC expansion potential and premature ageing in vitro. Mult. Scler. J. 2017, 24, 919–931. [Google Scholar] [CrossRef]
- Krysko, K.M.; Henry, R.G.; Cree, B.A.C.; Lin, J.; Caillier, S.; Santaniello, A.; Zhao, C.; Gomez, R.; Bevan, C.; Smith, D.L.; et al. Telomere Length Is Associated with Disability Progression in Multiple Sclerosis. Ann. Neurol. 2019, 86, 671–682. [Google Scholar] [CrossRef]
- Aubert, G.; Baerlocher, G.M.; Vulto, I.; Poon, S.S.; Lansdorp, P.M. Collapse of Telomere Homeostasis in Hematopoietic Cells Caused by Heterozygous Mutations in Telomerase Genes. PLoS Genet. 2012, 8, e1002696. [Google Scholar] [CrossRef]
- Hedström, A.K.; Hössjer, O.; Katsoulis, M.; Kockum, I.; Olsson, T.; Alfredsson, L. Organic solvents and MS susceptibility. Neurology 2018, 91, e455–e462. [Google Scholar] [CrossRef]
- Gerhardsson, L.; Hou, L.; Pettersson, K. Work-related exposure to organic solvents and the risk for multiple sclerosis—A systematic review. Int. Arch. Occup. Environ. Health 2021, 94, 221–229. [Google Scholar] [CrossRef] [PubMed]
- Liang, G.; Schernhammer, E.; Qi, L.; Gao, X.; De Vivo, I.; Han, J. Associations between Rotating Night Shifts, Sleep Duration, and Telomere Length in Women. PLoS ONE 2011, 6, e23462. [Google Scholar] [CrossRef] [PubMed]
- Jackowska, M.; Hamer, M.; Carvalho, L.A.; Erusalimsky, J.D.; Butcher, L.; Steptoe, A. Short Sleep Duration Is Associated with Shorter Telomere Length in Healthy Men: Findings from the Whitehall II Cohort Study. PLoS ONE 2012, 7, e47292. [Google Scholar] [CrossRef]
- James, S.; McLanahan, S.; Brooks-Gunn, J.; Mitchell, C.; Schneper, L.; Wagner, B.; Notterman, D.A. Sleep Duration and Telomere Length in Children. J. Pediatr. 2017, 187, 247–252.e1. [Google Scholar] [CrossRef]
- Dreyfus, D.H. Anti-Viral Therapy, Epstein–Barr Virus, Autoimmunity, and Chaos (The Butterfly Effect). Infect. Autoimmun. 2015, 301–317. [Google Scholar] [CrossRef]
- Hedström, A.K.; Brenner, N.; Butt, J.; Hillert, J.; Waterboer, T.; Olsson, T.; Alfredsson, L. Overweight/obesity in young adulthood interacts with aspects of EBV infection in MS etiology. Neurol.—Neuroimmunol. Neuroinflam. 2021, 8, e912. [Google Scholar] [CrossRef] [PubMed]
- Wijnands, J.M.A.; Kingwell, E.; Zhu, F.; Zhao, Y.; Högg, T.; Stadnyk, K.; Ekuma, O.; Lu, X.; Evans, C.; Fisk, J.D.; et al. Health-care use before a first demyelinating event suggestive of a multiple sclerosis prodrome: A matched cohort study. Lancet Neurol. 2017, 16, 445–451. [Google Scholar] [CrossRef]
- Bovis, F.; Signori, A.; Carmisciano, L.; Maietta, I.; Steinerman, J.R.; Li, T.; Tansy, A.P.; Sormani, M.P. Expanded disability status scale progression assessment heterogeneity in multiple sclerosis according to geographical areas. Ann. Neurol. 2018, 84, 621–625. [Google Scholar] [CrossRef]
- King, K.; Kozlitina, J.; Rosenberg, R.N.; Peshock, R.; McColl, R.W.; Garcia, C.K. Effect of Leukocyte Telomere Length on Total and Regional Brain Volumes in a Large Population-Based Cohort. JAMA Neurol. 2014, 71, 1247–1254. [Google Scholar] [CrossRef]
- Pathak, G.A.; Wendt, F.R.; Levey, D.F.; Mecca, A.P.; van Dyck, C.H.; Gelernter, J.; Polimanti, R. Pleiotropic effects of telomere length loci with brain morphology and brain tissue expression. Hum. Mol. Genet. 2021, 30, 1360–1370. [Google Scholar] [CrossRef] [PubMed]
- Guo, M.H.; Sama, P.; LaBarre, B.A.; Lokhande, H.; Balibalos, J.; Chu, C.; Du, X.; Kheradpour, P.; Kim, C.C.; Oniskey, T.; et al. Dissection of multiple sclerosis genetics identifies B and CD4+ T cells as driver cell subsets. BioRxiv 2021. [Google Scholar] [CrossRef]
- Spittau, B. Aging Microglia—Phenotypes, Functions and Implications for Age-Related Neurodegenerative Diseases. Front. Aging Neurosci. 2017, 9, 194. [Google Scholar] [CrossRef]
- Baker, D.J.; Petersen, R.C. Cellular senescence in brain aging and neurodegenerative diseases: Evidence and perspectives. J. Clin. Investig. 2018, 128, 1208–1216. [Google Scholar] [CrossRef]
- Hug, A.; Korporal, M.; Schröder, I.; Haas, J.; Glatz, K.; Storch-Hagenlocher, B.; Wildemann, B. Thymic export function and T cell homeostasis in patients with relapsing remitting multiple sclerosis. J. Immunol. 2003, 171, 432–437. [Google Scholar] [CrossRef] [PubMed]
- Thewissen, M.; Linsen, L.; Geusens, P.; Raus, J.; Stinissen, P. Impaired activation-induced telomerase activity in PBMC of early but not chronic rheumatoid arthritis patients. Immunol. Lett. 2005, 100, 205–210. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Adamczyk, B.; Adamczyk-Sowa, M. New Insights into the Role of Oxidative Stress Mechanisms in the Pathophysiology and Treatment of Multiple Sclerosis. Oxid. Med. Cell. Longev. 2016, 2016, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Jäger, K.; Walter, M. Therapeutic Targeting of Telomerase. Genes 2016, 7, 39. [Google Scholar] [CrossRef] [PubMed]
- Di Micco, R.; Krizhanovsky, V.; Baker, D.; di Fagagna, F.D. Cellular senescence in ageing: From mechanisms to therapeutic opportunities. Nat. Rev. Mol. Cell Biol. 2021, 22, 75–95. [Google Scholar] [CrossRef]
| MS Risk | MS Severity | TL | ||||
|---|---|---|---|---|---|---|
| Factor | Effect | Evidence | Effect | Evidence | Effect | Evidence |
| Genetics | ||||||
| Ancestry (European) | ➚ | *** | ➘ | * | ➘ | *** |
| Sex (female) | ➚ | *** | • | *** | ➚ | *** |
| Estrogen in women | ➚ | * | ➘ | ** | ➚ | * |
| Testosterone in men | ➘ | * | ➘ | * | ➚ | * |
| Familial heritability | • | *** | − | − | • | *** |
| HLA alleles | • | *** | • | * | • | * |
| SNPs | • | ** | − | − | • | ** |
| Environment | ||||||
| Sun exposure | ➘ | ** | ➘ | * | • | * |
| Epstein–Barr virus | ➚ | *** | ➚ | ** | • | * |
| HHV-6A | ➚ | * | ➚ | * | ➘ | ** |
| CMV | ➘ | * | • | * | ➘ | ** |
| Bacterial infections | • | * | − | − | ➘ | * |
| Air pollution | ➚ | * | ➚ | * | ➘ | ** |
| Lifestyle | ||||||
| Obesity | ➚ | *** | • | * | ➘ | *** |
| Mediterranean diet | ➘ | * | ➘ | * | ➚ | ** |
| Vitamin D sufficiency | ➘ | ** | ➘ | * | ➚ | ** |
| Other vitamins | ➘ | * | − | − | ➚ | * |
| Alcohol | • | * | ➘ | * | ➘ | * |
| Coffee | ➘ | * | − | − | ➚ | * |
| Smoking | ➚ | *** | ➚ | *** | ➘ | *** |
| Physical activity | ➘ | ** | ➘ | ** | • | ** |
| Psychological stress | ➚ | * | ➚ | * | ➘ | ** |
| Night work | ➚ | ** | − | − | ➘ | * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hecker, M.; Bühring, J.; Fitzner, B.; Rommer, P.S.; Zettl, U.K. Genetic, Environmental and Lifestyle Determinants of Accelerated Telomere Attrition as Contributors to Risk and Severity of Multiple Sclerosis. Biomolecules 2021, 11, 1510. https://doi.org/10.3390/biom11101510
Hecker M, Bühring J, Fitzner B, Rommer PS, Zettl UK. Genetic, Environmental and Lifestyle Determinants of Accelerated Telomere Attrition as Contributors to Risk and Severity of Multiple Sclerosis. Biomolecules. 2021; 11(10):1510. https://doi.org/10.3390/biom11101510
Chicago/Turabian StyleHecker, Michael, Jan Bühring, Brit Fitzner, Paulus Stefan Rommer, and Uwe Klaus Zettl. 2021. "Genetic, Environmental and Lifestyle Determinants of Accelerated Telomere Attrition as Contributors to Risk and Severity of Multiple Sclerosis" Biomolecules 11, no. 10: 1510. https://doi.org/10.3390/biom11101510
APA StyleHecker, M., Bühring, J., Fitzner, B., Rommer, P. S., & Zettl, U. K. (2021). Genetic, Environmental and Lifestyle Determinants of Accelerated Telomere Attrition as Contributors to Risk and Severity of Multiple Sclerosis. Biomolecules, 11(10), 1510. https://doi.org/10.3390/biom11101510








