DNA Double-Strand Breaks Induced in Human Cells by Twelve Metallic Species: Quantitative Inter-Comparisons and Influence of the ATM Protein
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cell Lines
2.2. Metals
2.3. Treatment with Zoledronate and Pravastatine (ZoPra)
2.4. Irradiations
2.5. Immunofluorescence
2.6. Statistical Analysis
3. Results
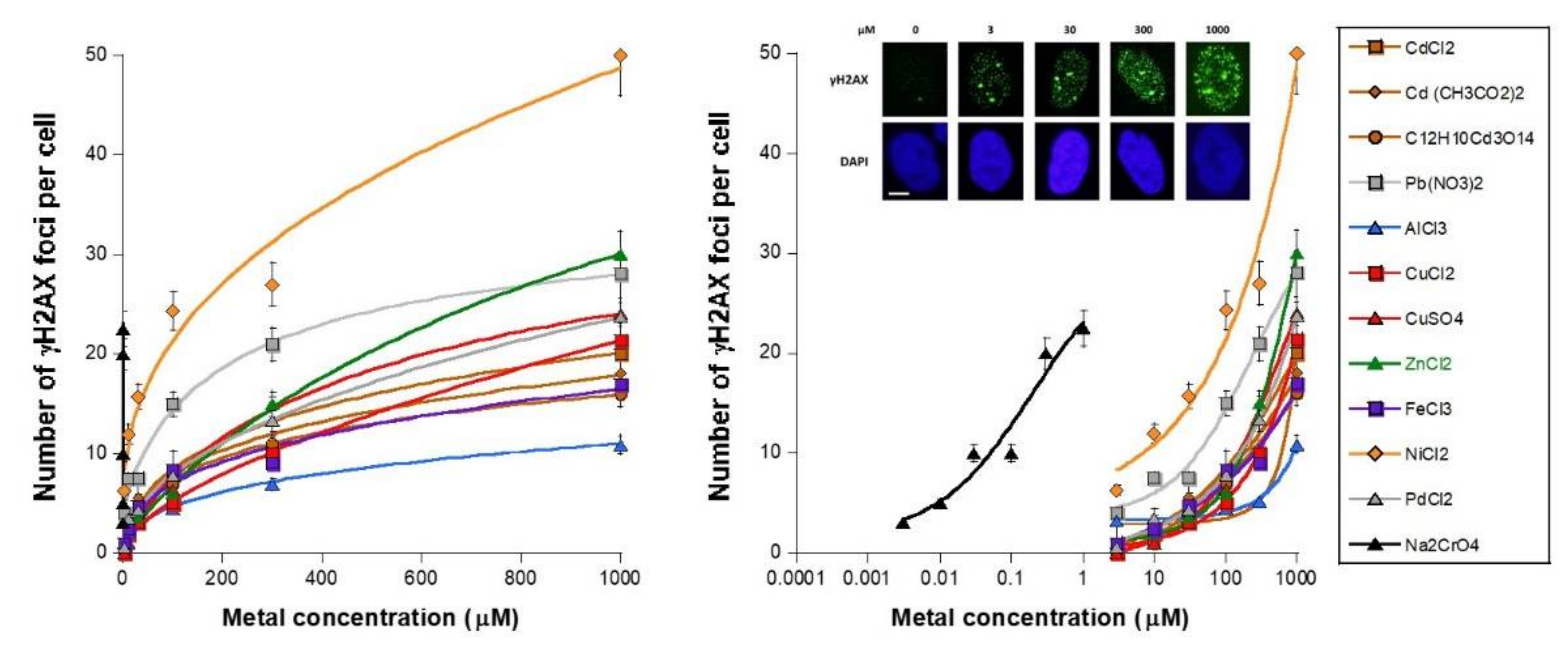
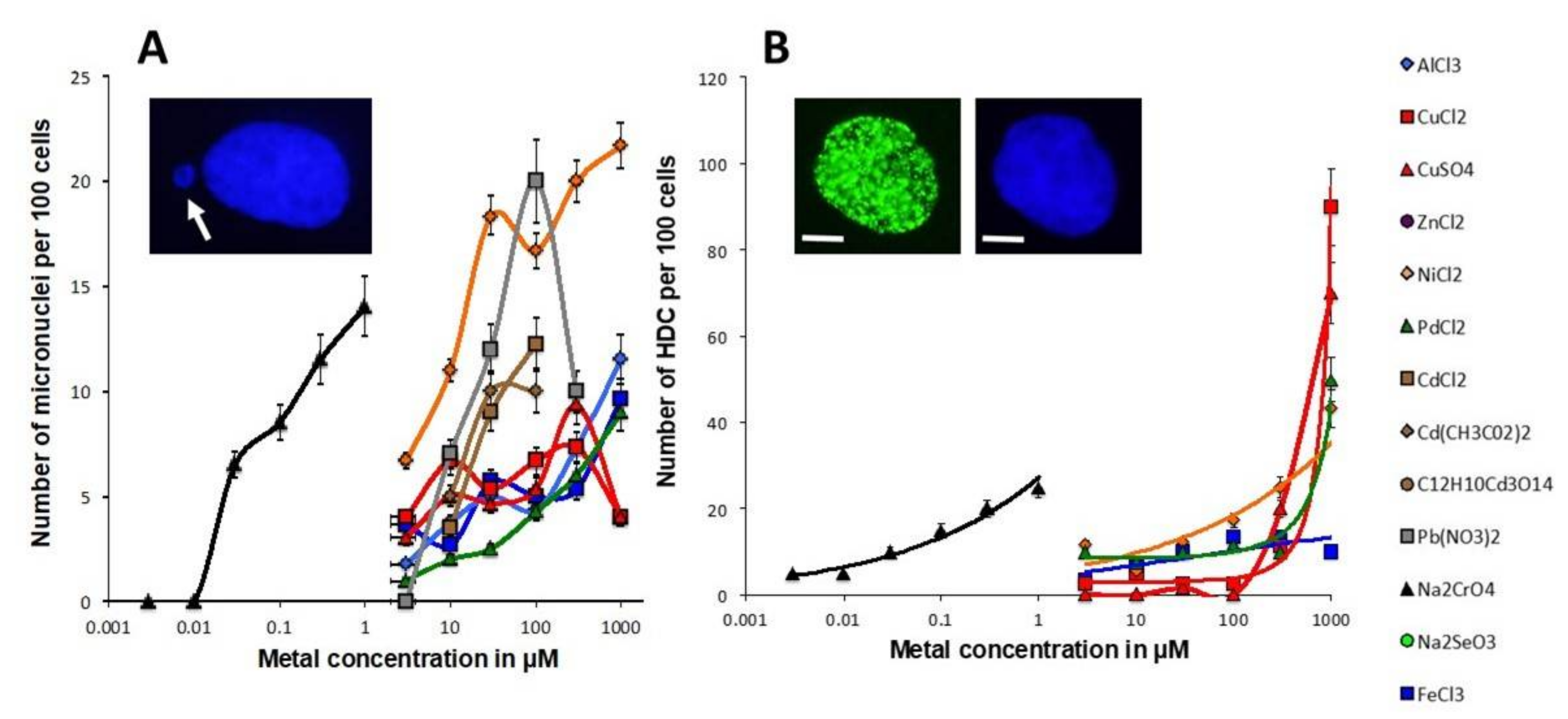
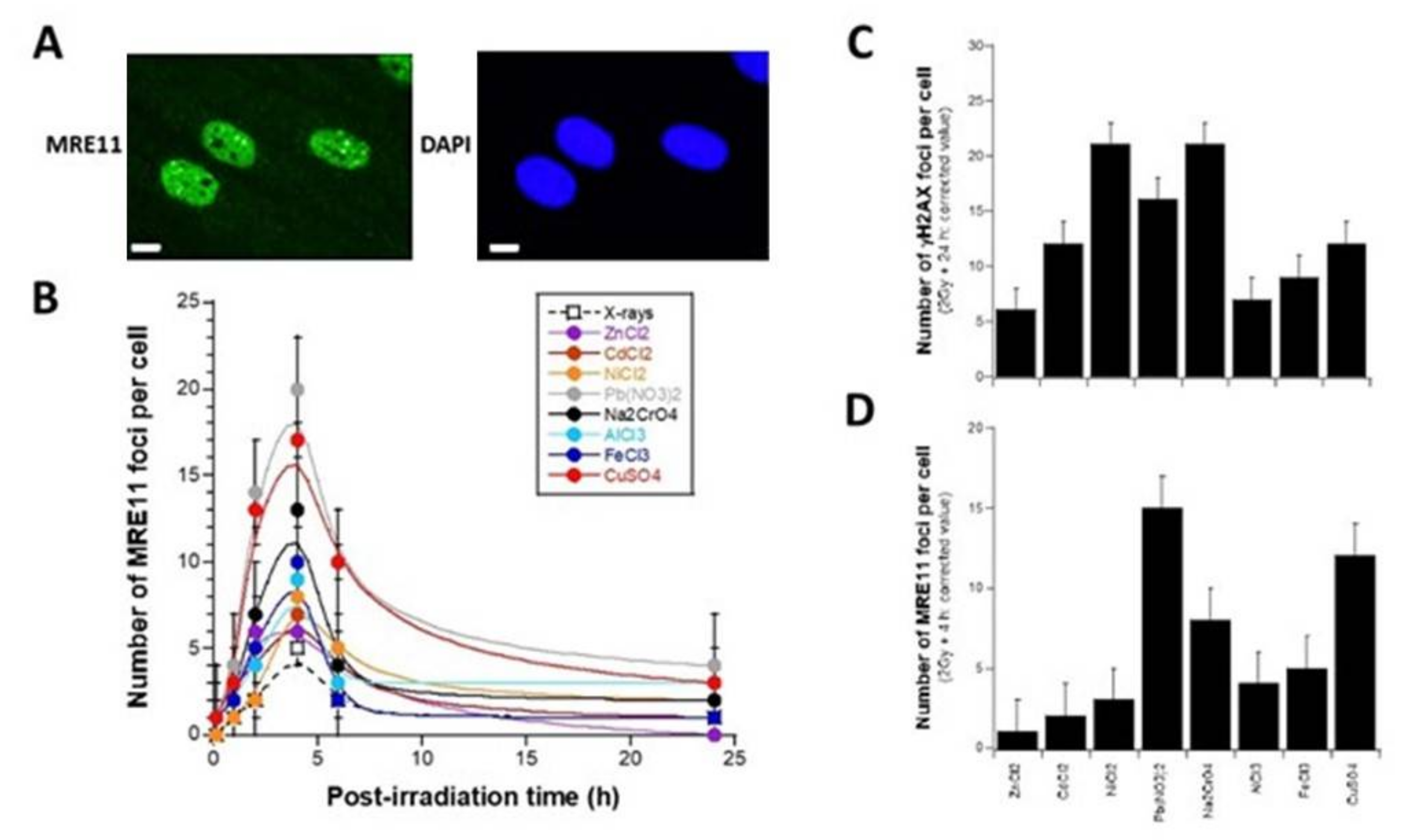
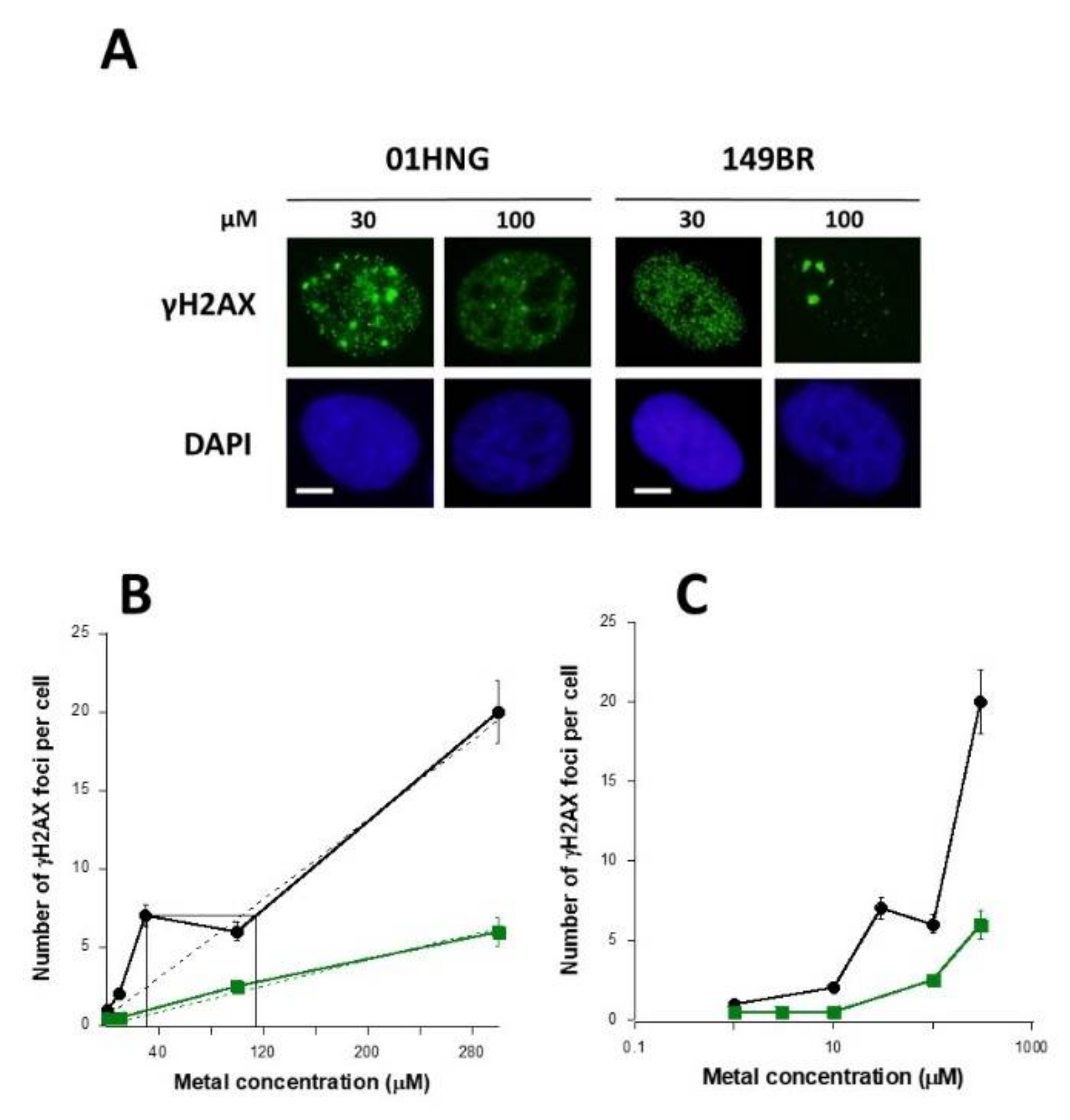
3.1. Unrepaired MI DSB Assessed with Different Endpoints
- − a metal concentration range in which the number of γH2AX foci is not significant or does not increase significantly with metal concentration;
- − a metal concentration range in which the number of γH2AX foci becomes significant and increases, generally linearly, with the metal concentration;
- − a metal concentration range in which the number of γH2AX foci reaches its maximum in a pseudo-plateau.
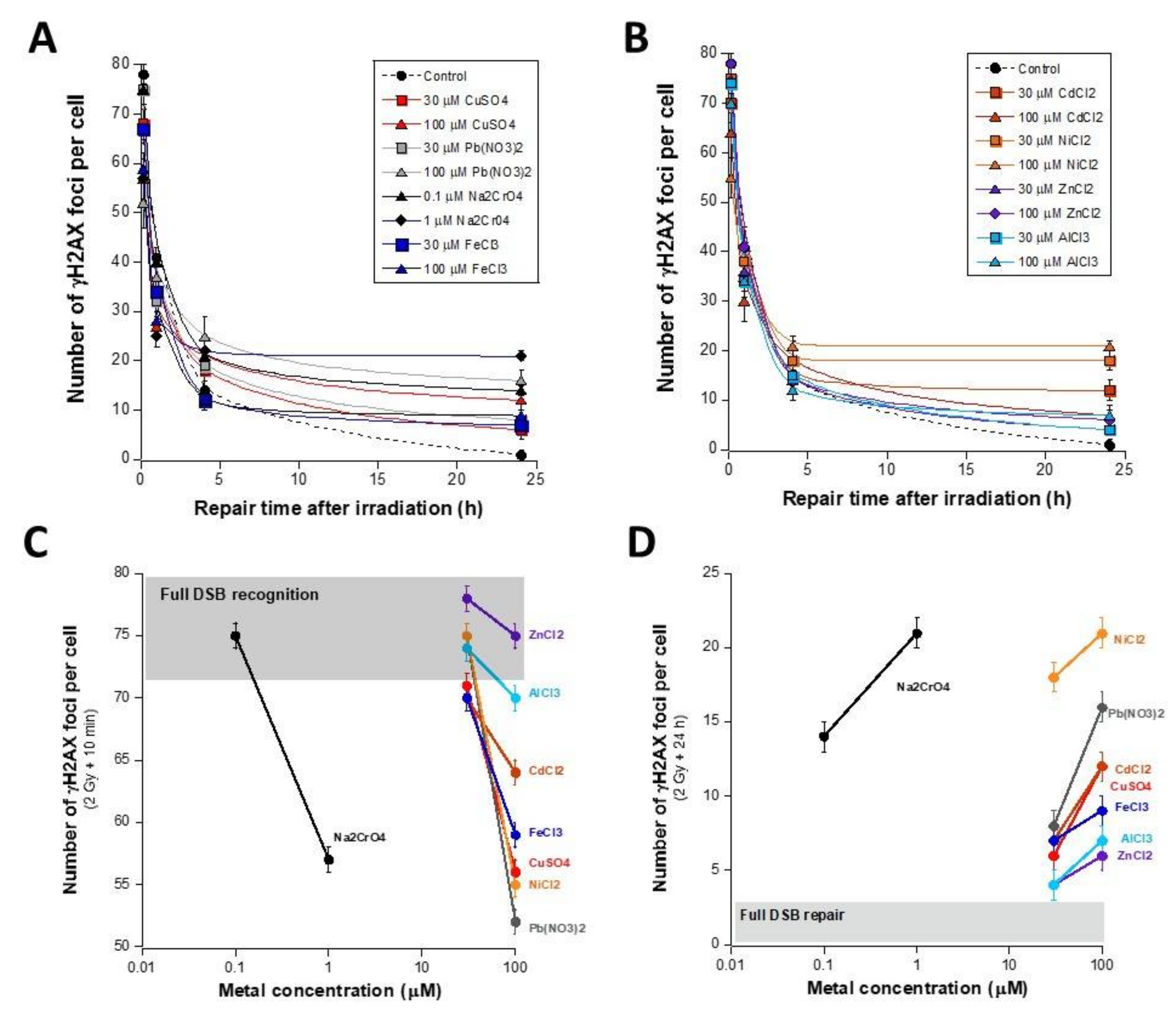
3.2. Influence of the Presence of Metal during the RI DSB Recognition and Repair Process
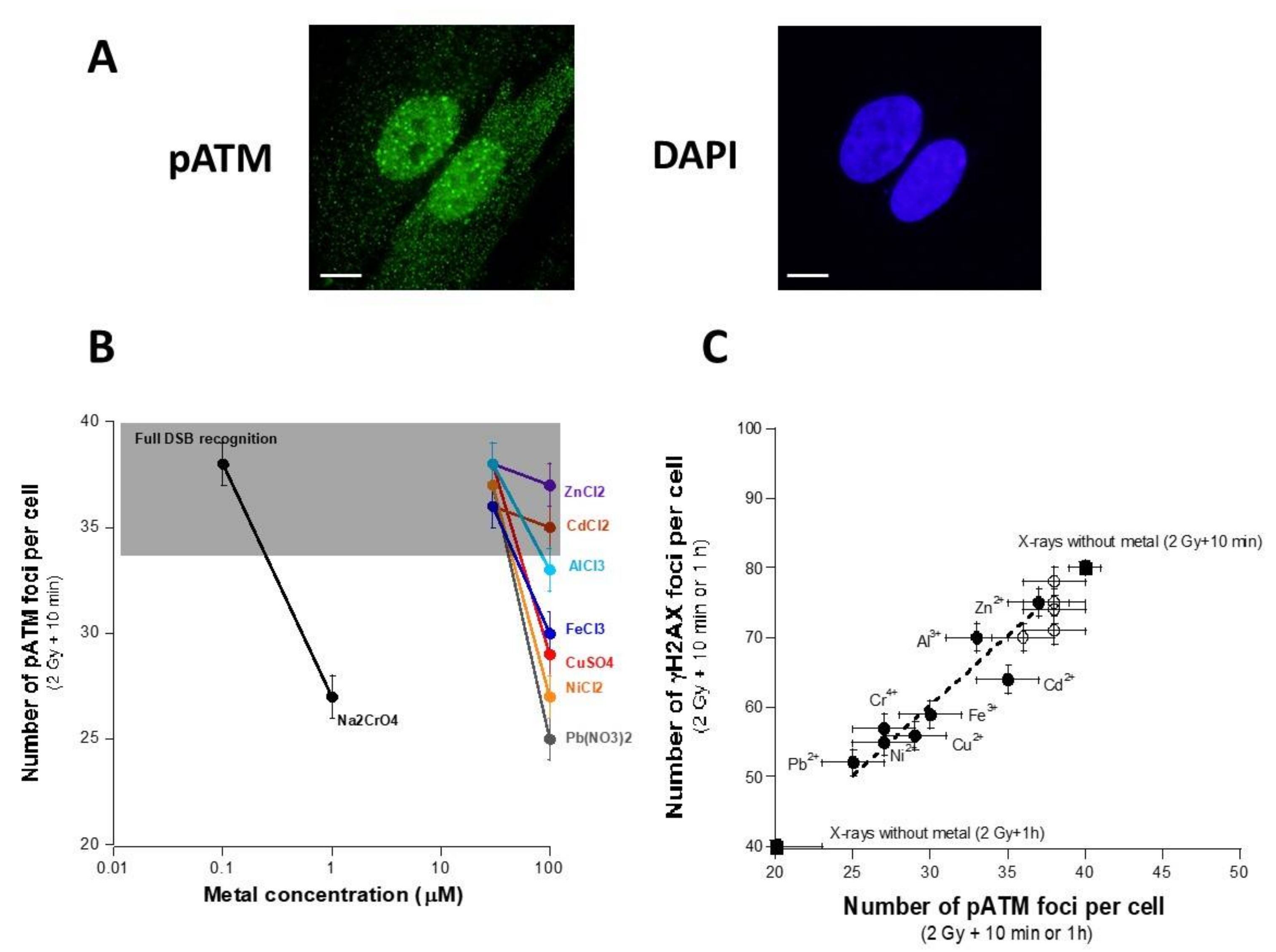
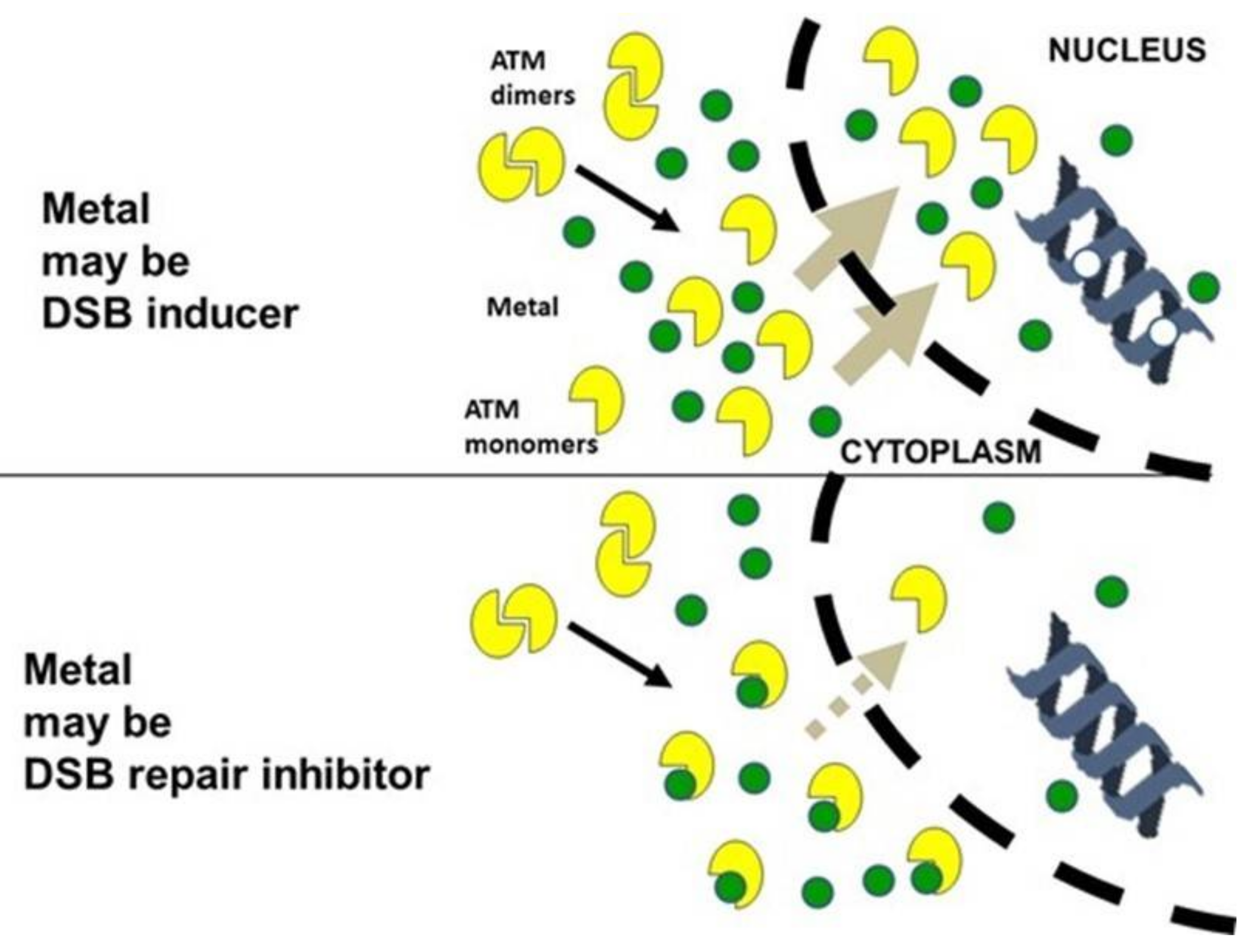
3.3. Influence of the Presence of Metal on the Nucleo-Shuttling of the ATM Protein
3.4. Influence of the Presence of Metal on the Activity of the MRE11 Nuclease
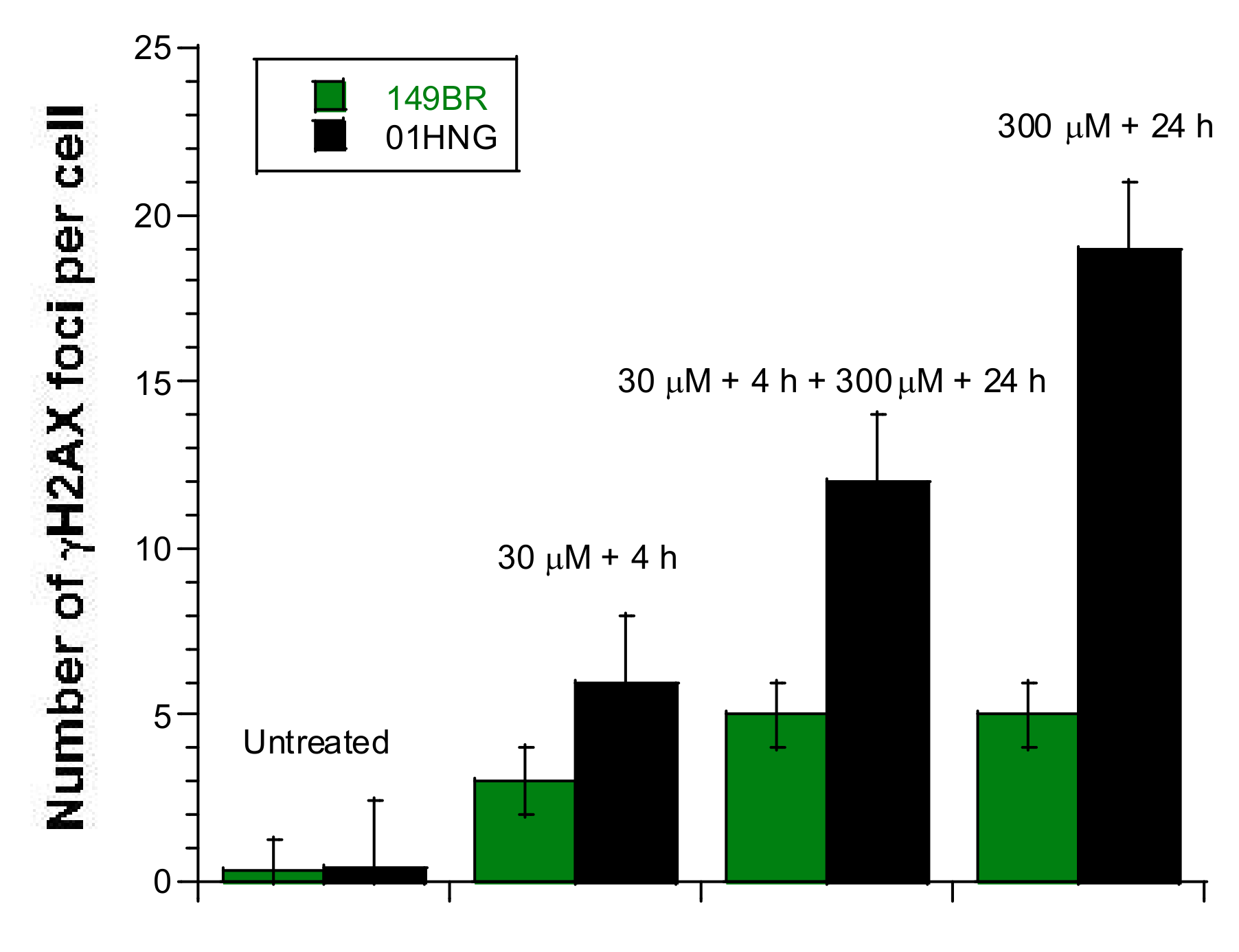
3.5. The Hypersensitivity Phenomenon Observed after Exposure to Low Metal Concentrations
3.6. The Adaptive Response Observed after Exposure to Low Metal Concentrations
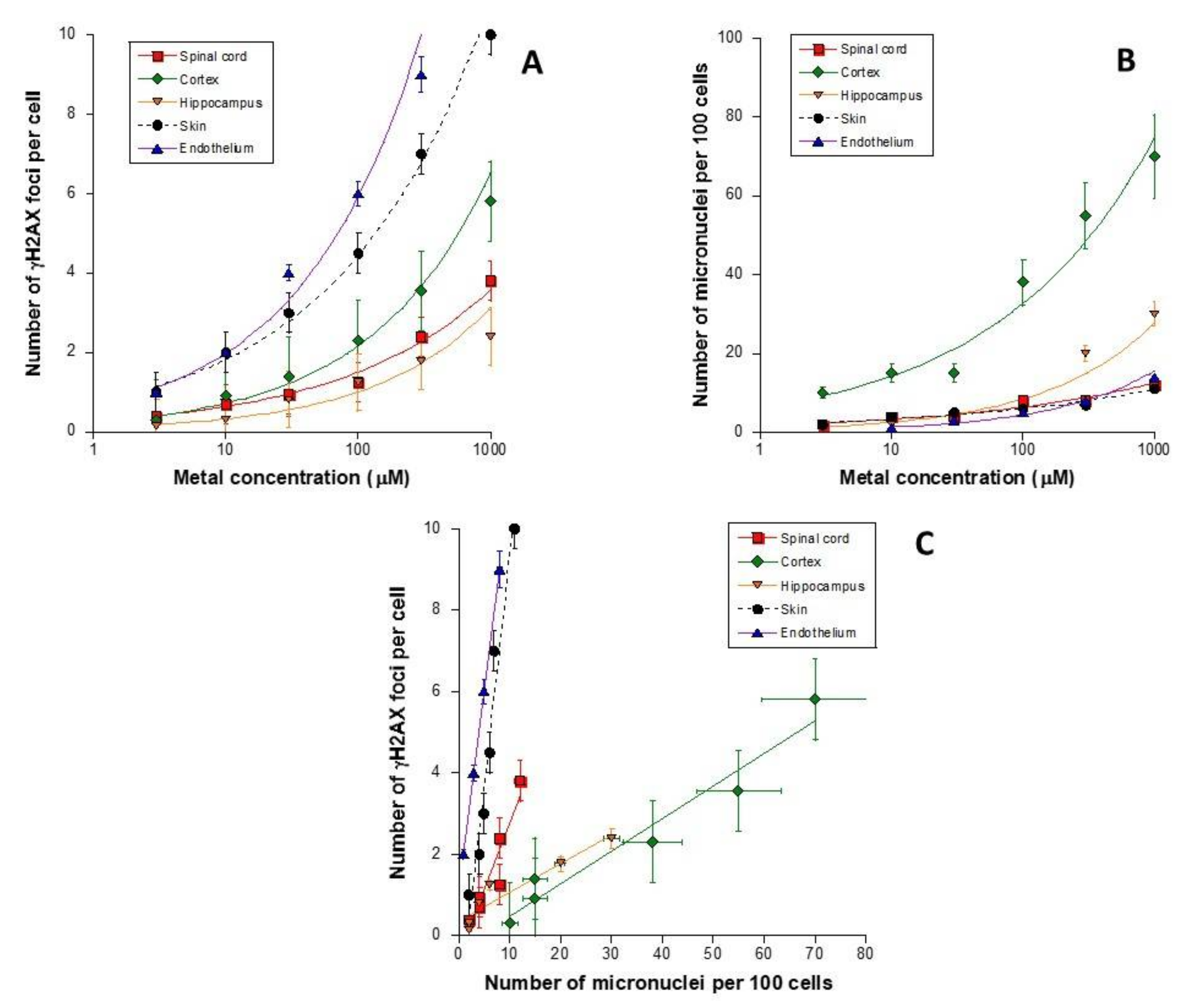
3.7. Cell-Type Dependence in the Response to Metal Exposure
4. Discussion
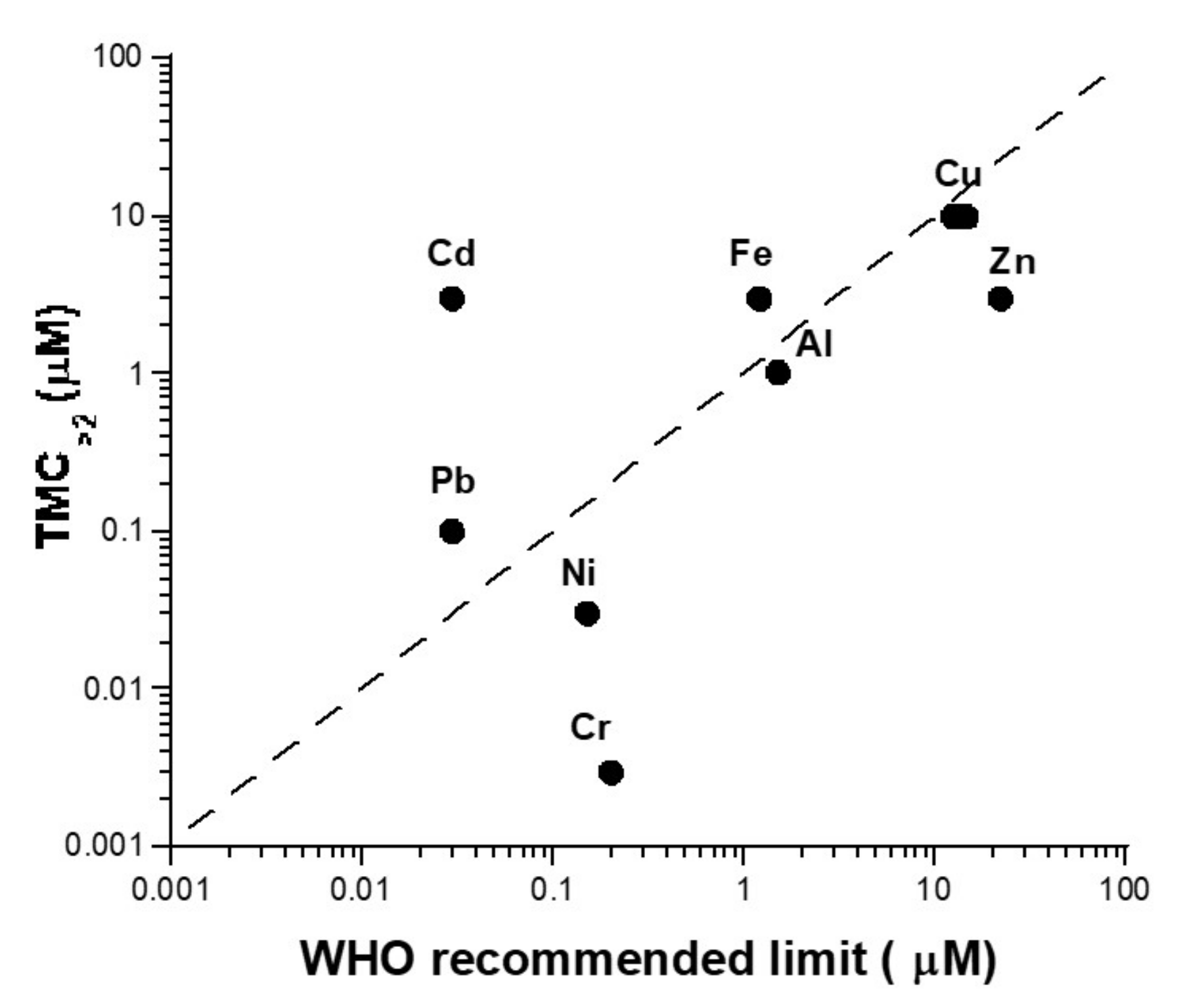
4.1. The Need for a Consensual Scale to Account for Toxicity and Carcinogenicity of Metals
4.2. Unrepaired DSB as a Unit to Account for Toxicity of Metals?
4.3. Misrepaired DSB as a Unit to Account for Carcinogenicity of Metals?
4.4. Toward a Unified Model for Understanding the Response to Metals?
5. Conclusions
6. Patents
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- IARC. Inorganic and organic lead compounds. IARC Monogr. Eval. Carcinog. Risks Hum. 2006, 87, 1. Available online: https://publications.iarc.fr/Book-And-Report-Series/Iarc-Monographs-On-The-Identification-Of-Carcinogenic-Hazards-To-Humans/Inorganic-And-Organic-Lead-Compounds-2006 (accessed on 3 October 2021).
- IARC. Beryllium, cadmium, mercury, and exposures in the glass manufacturing industry. Working Group views and expert opinions, Lyon, 9–16 February 1993. IARC Monogr. Eval. Carcinog. Risks Hum 1993, 58, 1–415. Available online: https://publications.iarc.fr/Book-And-Report-Series/Iarc-Monographs-On-The-Identification-Of-Carcinogenic-Hazards-To-Humans/Beryllium-Cadmium-Mercury-And-Exposures-In-The-Glass-Manufacturing-Industry-1993 (accessed on 3 October 2021).
- Bertin, G.; Averbeck, D. Cadmium: Cellular effects, modifications of biomolecules, modulation of DNA repair and genotoxic consequences (a review). Biochimie 2006, 88, 1549–1559. [Google Scholar] [CrossRef]
- Drago, D.; Bolognin, S.; Zatta, P. Role of metal ions in the abeta oligomerization in Alzheimer’s disease and in other neurological disorders. Curr. Alzheimer Res. 2008, 5, 500–507. [Google Scholar] [CrossRef]
- Boll, M.C.; Alcaraz-Zubeldia, M.; Montes, S.; Rios, C. Free copper, ferroxidase and SOD1 activities, lipid peroxidation and NO(x) content in the CSF. A different marker profile in four neurodegenerative diseases. Neurochem. Res. 2008, 33, 1717–1723. [Google Scholar] [CrossRef]
- Charlet, N.; Chapron, Y.; Faller, P.; Kirsch, R.; Stone, A.T.; Baveye, P.C. Neurodegenerative diseases and exposure to the environmental metals Mn, Pb, and Hg. Coord. Chem. Rev. 2012, 256, 2147–2163. [Google Scholar] [CrossRef]
- Smith, A.H.; Goycolea, M.; Haque, R.; Biggs, M.L. Marked increase in bladder and lung cancer mortality in a region of Northern Chile due to arsenic in drinking water. Am. J. Epidemiol. 1998, 147, 660–669. [Google Scholar] [CrossRef]
- Smith, A.H.; Hopenhayn-Rich, C.; Bates, M.N.; Goeden, H.M.; Hertz-Picciotto, I.; Duggan, H.M.; Wood, R.; Kosnett, M.J.; Smith, M.T. Cancer risks from arsenic in drinking water. Environ. Health Perspect. 1992, 97, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.H.; Marshall, G.; Yuan, Y.; Ferreccio, C.; Liaw, J.; von Ehrenstein, O.; Steinmaus, C.; Bates, M.N.; Selvin, S. Increased mortality from lung cancer and bronchiectasis in young adults after exposure to arsenic in utero and in early childhood. Environ. Health Perspect. 2006, 114, 1293–1296. [Google Scholar] [CrossRef] [Green Version]
- Wild, P.; Bourgkard, E.; Paris, C. Lung cancer and exposure to metals: The epidemiological evidence. Cancer Epidemiol. 2009, 472, 139–167. [Google Scholar]
- Gastaldo, J.; Viau, M.; Bencokova, Z.; Joubert, A.; Charvet, A.M.; Balosso, J.; Foray, N. Lead contamination results in late and slowly repairable DNA double-strand breaks and impacts upon the ATM-dependent signaling pathways. Toxicol. Lett. 2007, 173, 201–214. [Google Scholar] [CrossRef]
- Viau, M.; Gastaldo, J.; Bencokova, Z.; Joubert, A.; Foray, N. Cadmium inhibits non-homologous end-joining and over-activates the MRE11-dependent repair pathway. Mutat. Res. 2008, 654, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Joubert, A.; Zimmerman, K.M.; Bencokova, Z.; Gastaldo, J.; Rénier, W.; Chavaudra, N.; Favaudon, V.; Arlett, C.; Foray, N. DNA double-strand break repair defects in syndromes associated with acute radiation response: At least two different assays to predict intrinsic radiosensitivity? Int. J. Radiat. Biol. 2008, 84, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Foray, N.; Bourguignon, M.; Hamada, N. Individual response to ionizing radiation. Mutat. Res. Rev. 2016, 770, 369–386. [Google Scholar] [CrossRef]
- Morales, M.E.; Derbes, R.S.; Ade, C.M.; Ortego, J.C.; Stark, J.; Deininger, P.L.; Roy-Engel, A.M. Heavy Metal Exposure Influences Double Strand Break DNA Repair Outcomes. PLoS ONE 2016, 11, e0151367. [Google Scholar] [CrossRef]
- Rittich, B.; Spanova, A.; Falk, M.; Benes, M.J.; Hruby, M. Cleavage of double stranded plasmid DNA by lanthanide complexes. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2004, 800, 169–173. [Google Scholar] [CrossRef]
- Tenan, M.R.; Nicolle, A.; Moralli, D.; Verbouwe, E.; Jankowska, J.D.; Durin, M.A.; Green, C.M.; Mandriota, S.J.; Sappino, A.P. Aluminum Enters Mammalian Cells and Destabilizes Chromosome Structure and Number. Int. J. Mol. Sci. 2021, 22, 9515. [Google Scholar] [CrossRef]
- Zhang, S.; Hao, S.; Qiu, Z.; Wang, Y.; Zhao, Y.; Li, Y.; Gao, W.; Wu, Y.; Liu, C.; Xu, X.; et al. Cadmium disrupts the DNA damage response by destabilizing RNF168. Food Chem. Toxicol. 2019, 133, 110745. [Google Scholar] [CrossRef]
- Shah, A.J.; Lakkad, B.C.; Rao, M.V. Genotoxicity in lead treated human lymphocytes evaluated by micronucleus and comet assays. Indian J. Exp. Biol. 2016, 54, 502–508. [Google Scholar]
- DeLoughery, Z.; Luczak, M.W.; Ortega-Atienza, S.; Zhitkovich, A. DNA double-strand breaks by Cr(VI) are targeted to euchromatin and cause ATR-dependent phosphorylation of histone H2AX and its ubiquitination. Toxicol. Sci. Off. J. Soc. Toxicol. 2015, 143, 54–63. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pottier, G.; Viau, M.; Ricoul, M.; Shim, G.; Bellamy, M.; Cuceu, C.; Hempel, W.M.; Sabatier, L. Lead Exposure Induces Telomere Instability in Human Cells. PLoS ONE 2013, 8, e67501. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ayene, I.S.; Koch, C.J.; Krisch, R.E. DNA strand breakage by bivalent metal ions and ionizing radiation. Int. J. Radiat. Biol. 2007, 83, 195–210. [Google Scholar] [CrossRef] [PubMed]
- Rothkamm, K.; Lobrich, M. Evidence for a lack of DNA double-strand break repair in human cells exposed to very low x-ray doses. Proc. Natl. Acad. Sci. USA 2003, 100, 5057–5062. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Granzotto, A.; Benadjaoud, M.A.; Vogin, G.; Devic, C.; Ferlazzo, M.L.; Bodgi, L.; Pereira, S.; Sonzogni, L.; Forcheron, F.; Viau, M.; et al. Influence of Nucleoshuttling of the ATM Protein in the Healthy Tissues Response to Radiation Therapy: Toward a Molecular Classification of Human Radiosensitivity. Int. J. Radiat. Oncol. Biol. Phys. 2016, 94, 450–460. [Google Scholar] [CrossRef] [PubMed]
- Belkacemi, Y.; Colson-Durand, L.; Granzotto, A.; Husheng, S.; To, N.H.; Majdoul, S.; Guet, S.; Herve, M.L.; Fonteneau, G.; Diana, C.; et al. The Henri Mondor Procedure of Morbidity and Mortality Review Meetings: Prospective Registration of Clinical, Dosimetric, and Individual Radiosensitivity Data of Patients With Severe Radiation Toxicity. Int. J. Radiat. Oncol. Biol. Phys. 2016, 96, 629–636. [Google Scholar] [CrossRef]
- Pereira, S.; Bodgi, L.; Duclos, M.; Canet, A.; Ferlazzo, M.L.; Devic, C.; Granzotto, A.; Deneuve, S.; Vogin, G.; Foray, N. Fast and binary assay for predicting radiosensitivity based on the nucleoshuttling of ATM protein: Development, validation and performances. Int. J. Radiat. Oncol. Biol. Phys. 2018, 100, 353–360. [Google Scholar] [CrossRef]
- Vogin, G.; Bastogne, T.; Bodgi, L.; Gillet-Daubin, J.; Canet, A.; Pereira, S.; Foray, N. The Phosphorylated ATM Immunofluorescence Assay: A High-performance Radiosensitivity Assay to Predict Postradiation Therapy Overreactions. Int. J. Radiat. Oncol. Biol. Phys. 2018, 101, 690–693. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maalouf, M.; Granzotto, A.; Devic, C.; Bodgi, L.; Ferlazzo, M.; Peaucelle, C.; Bajard, M.; Giraud, J.Y.; Balosso, J.; Herault, J.; et al. Influence of Linear Energy Transfer on the Nucleo-shuttling of the ATM Protein: A Novel Biological Interpretation Relevant for Particles and Radiation. Int. J. Radiat. Oncol. Biol. Phys. 2019, 103, 709–718. [Google Scholar] [CrossRef]
- Berthel, E.; Ferlazzo, M.; Devic, C.; Bourguignon, M.; Foray, N. What does the History of Research on the Repair of DNA Double-Strand Breaks Tell Us?—A Comprehensive Review of Human Radiosensitivity. Int. J. Mol. Sci. 2019, 20, 5339. [Google Scholar] [CrossRef] [Green Version]
- Berthel, E.; Foray, N.; Ferlazzo, M.L. The Nucleoshuttling of the ATM Protein: A Unified Model to Describe the Individual Response to High- and Low-Dose of Radiation? Cancers 2019, 11, 905. [Google Scholar] [CrossRef] [Green Version]
- Bodgi, L.; Foray, N. The nucleo-shuttling of the ATM protein as a basis for a novel theory of radiation response: Resolution of the linear-quadratic model. Int. J. Radiat. Biol. 2016, 92, 117–131. [Google Scholar] [CrossRef]
- Foray, N.; Fertil, B.; Alsbeih, M.G.; Badie, C.; Chavaudra, N.; Iliakis, G.; Malaise, E.P. Dose-rate effect on radiation-induced DNA double-strand breaks in the human fibroblast HF19 cell line. Int. J. Radiat. Biol. 1996, 69, 241–249. [Google Scholar] [CrossRef]
- Foray, N.; Priestley, A.; Alsbeih, G.; Badie, C.; Capulas, E.P.; Arlett, C.F.; Malaise, E.P. Hypersensitivity of ataxia telangiectasia fibroblasts to ionizing radiation is associated with a repair deficiency of DNA double-strand breaks. Int. J. Radiat. Biol. 1997, 72, 271–283. [Google Scholar] [PubMed]
- Gastaldo, J.; Bencokova, Z.; Massart, C.; Joubert, A.; Balosso, J.; Charvet, A.M.; Foray, N. Specific molecular and cellular events induced by irradiated X-ray photoactivatable drugs raise the problem of co-toxicities: Particular consequences for anti-cancer synchrotron therapy. J. Synchrotron Radiat. 2011, 18, 456–463. [Google Scholar] [CrossRef] [PubMed]
- Varela, I.; Pereira, S.; Ugalde, A.P.; Navarro, C.L.; Suarez, M.F.; Cau, P.; Cadinanos, J.; Osorio, F.G.; Foray, N.; Cobo, J.; et al. Combined treatment with statins and aminobisphosphonates extends longevity in a mouse model of human premature aging. Nat. Med. 2008, 14, 767–772. [Google Scholar] [CrossRef]
- Foray, N.; Marot, D.; Gabriel, A.; Randrianarison, V.; Carr, A.M.; Perricaudet, M.; Ashworth, A.; Jeggo, P. A subset of ATM- and ATR-dependent phosphorylation events requires the BRCA1 protein. EMBO J. 2003, 22, 2860–2871. [Google Scholar] [CrossRef] [Green Version]
- Ferlazzo, M.; Berthel, E.; Granzotto, A.; Devic, C.; Sonzogni, L.; Bachelet, J.T.; Pereira, S.; Bourguignon, M.; Sarasin, A.; Mezzina, M.; et al. Some mutations in the xeroderma pigmentosum D gene may lead to moderate but significant radiosensitivity associated with a delayed radiation-induced ATM nuclear localization. Int. J. Radiat. Biol. 2019, 96, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Colin, C.; Devic, C.; Noël, A.; Rabilloud, M.; Zabot, M.-T.; Pinet-Isaac, S.; Giraud, S.; Riche, B.; Valette, P.-J.; Rodriguez-Lafrasse, C.; et al. DNA double-strand breaks induced by mammographic screening procedures in human mammary epithelial cells. Int. J. Radiat. Biol. 2011, 87, 1103–1112. [Google Scholar] [CrossRef]
- Bodgi, L.; Granzotto, A.; Devic, C.; Vogin, G.; Lesne, A.; Bottollier-Depois, J.F.; Victor, J.M.; Maalouf, M.; Fares, G.; Foray, N. A single formula to describe radiation-induced protein relocalization: Towards a mathematical definition of individual radiosensitivity. Int. J. Theor. Biol. 2013, 333, 135–145. [Google Scholar] [CrossRef]
- Grote, S.J.; Joshi, G.P.; Revell, S.H.; Shaw, C.A. Observations of radiation-induced chromosome fragment loss in live mammalian cells in culture, and its effect on colony-forming ability. Int. J. Radiat. Biol. Relat. Stud. Phys. Chem. Med. 1981, 39, 395–408. [Google Scholar] [CrossRef]
- Nascarella, M.A.; Stoffolano Jr, J.G.; Stanek 3rd, E.J.; Kostecki, P.T.; Calabrese, E.J. Hormesis and stage specific toxicity induced by cadmium in an insect model, the queen blowfly, Phormia regina Meig. Environ. Pollut. 2003, 124, 257–262. [Google Scholar] [CrossRef] [Green Version]
- Bleavins, K.; Perone, P.; Naik, M.; Rehman, M.; Aslam, M.N.; Dame, M.K.; Meshinchi, S.; Bhagavathula, N.; Varani, J. Stimulation of fibroblast proliferation by insoluble gadolinium salts. Biol. Trace Elem. Res. 2012, 145, 257–267. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Philips, N.; Hwang, H.; Chauhan, S.; Leonardi, D.; Gonzalez, S. Stimulation of cell proliferation and expression of matrixmetalloproteinase-1 and interluekin-8 genes in dermal fibroblasts by copper. Connect Tissue Res. 2010, 51, 224–229. [Google Scholar] [CrossRef] [PubMed]
- Beyersmann, D.; Haase, H. Functions of zinc in signaling, proliferation and differentiation of mammalian cells. Biometals 2001, 14, 331–341. [Google Scholar] [CrossRef]
- Wagner, S.; Hussain, M.Z.; Hunt, T.K.; Bacic, B.; Becker, H.D. Stimulation of fibroblast proliferation by lactate-mediated oxidants. Wound Repair Regen. 2004, 12, 368–373. [Google Scholar] [CrossRef]
- Ferlazzo, M.L.; Bach-Tobdji, M.K.E.; Djerad, A.; Sonzogni, L.; Burlet, S.F.; Devic, C.; Granzotto, A.; Bodgi, L.; Djeffal-Kerrar, A.; Foray, N. Radiobiological characterization of tuberous sclerosis: A delay in the nucleo-shuttling of ATM may be responsible for radiosensitivity. Mol. Neurobiol. 2017, 55, 4973–4983. [Google Scholar] [CrossRef]
- Ferlazzo, M.L.; Sonzogni, L.; Granzotto, A.; Bodgi, L.; Lartin, O.; Devic, C.; Vogin, G.; Pereira, S.; Foray, N. Mutations of the Huntington’s Disease Protein Impact on the ATM-Dependent Signaling and Repair Pathways of the Radiation-Induced DNA Double-Strand Breaks: Corrective Effect of Statins and Bisphosphonates. Mol. Neurobiol. 2014, 49, 1200–1211. [Google Scholar] [CrossRef] [PubMed]
- Varela, I.; Cadinanos, J.; Pendas, A.M.; Gutierrez-Fernandez, A.; Folgueras, A.R.; Sanchez, L.M.; Zhou, Z.; Rodriguez, F.J.; Stewart, C.L.; Vega, J.A.; et al. Accelerated ageing in mice deficient in Zmpste24 protease is linked to p53 signalling activation. Nature 2005, 437, 564–568. [Google Scholar] [CrossRef] [PubMed]
- Renier, W.; Joubert, A.; Bencokova, Z.; Gastaldo, J.; Massart, C.; Foray, N. Consequences of the bleed-through phenomenon in immunofluorescence of proteins forming radiation-induced nuclear foci. Int. J. Radiat. Biol. 2007, 83, 543–549. [Google Scholar] [CrossRef] [PubMed]
- El-Nachef, L.; Al-Choboq, J.; Restier-Verlet, J.; Granzotto, A.; Berthel, E.; Sonzogni, L.; Ferlazzo, M.L.; Bouchet, A.; Leblond, P.; Combemale, P.; et al. Human Radiosensitivity and Radiosusceptibility: What Are the Differences? Int. J. Mol. Sci. 2021, 22, 7158. [Google Scholar] [CrossRef]
- Joiner, M.C.; Marples, B.; Lambin, P.; Short, S.C.; Turesson, I. Low-dose hypersensitivity: Current status and possible mechanisms. Int. J. Radiat. Oncol. Biol. Phys. 2001, 49, 379–389. [Google Scholar] [CrossRef]
- Calabrese, E.J. Hormesis is central to toxicology, pharmacology and risk assessment. Hum. Exp. Toxicol. 2008, 29, 249–261. [Google Scholar] [CrossRef]
- Calabrese, E.J. Hormesis: A fundamental concept in biology. Microb. Cell 2014, 1, 145–149. [Google Scholar] [CrossRef] [Green Version]
- Devic, C.; Ferlazzo, M.L.; Foray, N. Influence of Individual Radiosensitivity on the Adaptive Response Phenomenon: Toward a Mechanistic Explanation Based on the Nucleo-Shuttling of ATM Protein. Dose-Response Publ. Int. Hormesis Soc. 2018, 16, 1–11. [Google Scholar] [CrossRef]
- Granzotto, A.; Bencokova, Z.; Vogin, G.; Devic, C.; Joubert, A.; Balosso, J.; Foray, N. DNA double-strand breaks repair and signaling of human gliomas and normal brain cells in response to radiation: Po-tential impact of the ATM- and BRCA1-dependent pathways. In Brain Tumors/Book 3; Abujamra, A.L., Ed.; Intechweb: Rijeka, Croatia, 2011. [Google Scholar]
- Hartwig, A. Current aspects in metal genotoxicity. Biometals 1995, 8, 3–11. [Google Scholar] [CrossRef]
- Lay, P.A.; Levina, A. Activation of Molecular Oxygen during the Reactions of Chromium(VI/V/IV) with Biological Reductants: Implications for Chromium-Induced Genotoxicities. J. Am. Chem. Soc. 1998, 120, 6704–6714. [Google Scholar] [CrossRef]
- Levina, A.; Codd, R.; Dillon, C.T.; Lay, P.A. Chromium in Biology. In Toxicology and Nutritional Aspects Progress in Inorganic Chemistry; Karlin, K.D., Ed.; Wiley Publishers: New York, NY, USA, 2003; Volume 51, pp. 145–250. [Google Scholar]
- Averbeck, D.; Candeias, S.; Chandna, S.; Foray, N.; Friedl, A.A.; Haghdoost, S.; Jeggo, P.A.; Lumniczky, K.; Paris, F.; Quintens, R.; et al. Establishing mechanisms affecting the individual response to ionizing radiation. Int. J. Radiat. Biol. 2020, 96, 297–323. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Misono, M.; Ochini, E.; Saito, Y.; Yoneda, Y. A new dual parameter scale for the strength of Lewis acids and bases with the evaluation of their softness. J. Inorg. Nucl. Chem. 1967, 29, 2685–2691. [Google Scholar] [CrossRef]
- Pearson, R.G. Hard and Soft acids and bases, Part I. J. Chem. Educ. 1968, 45, 581–587. [Google Scholar] [CrossRef]
- WHO. Guidelines for Drinking-Water Quality; WHO: Geneva, Switzerland, 2004; Available online: https://www.who.int/water_sanitation_health/dwq/GDWQ2004web.pdf (accessed on 3 October 2021).
- Paull, T.T. Mechanisms of ATM Activation. Annu. Rev. Biochem. 2015, 84, 711–738. [Google Scholar] [CrossRef] [PubMed]
- Pagacova, E.; Stefancikova, L.; Schmidt-Kaler, F.; Hildenbrand, G.; Vicar, T.; Depes, D.; Lee, J.H.; Bestvater, F.; Lacombe, S.; Porcel, E.; et al. Challenges and Contradictions of Metal Nano-Particle Applications for Radio-Sensitivity Enhancement in Cancer Therapy. Int. J. Mol. Sci. 2019, 20, 588. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Metal Species | TMC>2 $$$$(µM) | Sigmoidal* Data Fit Parameters | ||||
|---|---|---|---|---|---|---|
| m1 | m2 | m3 | m4 | r | ||
| Na2CrO4 | 0.003 | 30 | 1.71 | 1.90 × 10−1 | 0.67 | 0.97 |
| NiCl2 | 0.03 | 3380 | 4.49 | 2.77 × 107 | 0.42 | 0.98 |
| Pb(NO3)2 | 0.1 | 35 | 3.69 | 2.31 × 102 | 0.80 | 0.99 |
| AlCl3 | 1 | 36 | 0.29 | 5.75 × 103 | 0.48 | 0.99 |
| FeCl3 | 3 | 735 | 0 | 6.53 × 107 | 0.33 | 0.98 |
| ZnCl2 | 3 | 72 | 1.00 | 1.50 × 103 | 0.89 | 0.99 |
| CdCl2 | 3 | 58 | 0 | 3.54 × 103 | 0.46 | 0.99 |
| Cd(CH3CO2)2 | 3 | 603 | 0 | 2.29 × 108 | 0.26 | 0.99 |
| C12H10Cd3O14 | 10 | 80 | 0 | 5.25 × 104 | 0.28 | 0.99 |
| PdCl2 | 10 | 1045 | 0 | 2.94 × 106 | 0.47 | 0.99 |
| CuSO4 | 10 | 43 | 0 | 7.07 × 102 | 0.75 | 0.99 |
| CuCl2 | 10 | 1501 | 0.13 | 8.84 × 105 | 0.62 | 0.99 |
| Metallic Species | Misono Softness Parameter | TMC > 2$$$$ (µM) | TMC > 8$$$$ (µM) | γH2AX Foci at 24 h 2 | MRE11 Foci at 4 h 3 | WHO Limits in Tap Water (µM) |
|---|---|---|---|---|---|---|
| Na2CrO4 | na 1 | 0.003 | 0.06 | 21 (1 µM) | 8 (1 µM) | 0.2 |
| NiCl2 | 2.82 | 0.03 | 9 | 21 | 3 | 0.15 |
| Pb(NO3)2 | 3.58 | 0.1 | 30 | 16 | 15 | 0.03 |
| AlCl3 | 1.6 | 1 | 1000 | 7 | 4 | 1.5 |
| FeCl3 | 3.09 | 3 | 300 | 9 | 5 | 1.2 |
| ZnCl2 | 2.34 | 3 | 700 | 6 | 1 | 22 |
| CdCl2 | 3.04 | 3 | 100 | 12 | 2 | 0.03 |
| Cd(CH3CO2)2 | 3.04 | 3 | 100 | na 1 | na 1 | na 1 |
| C12H10Cd3O14 | 3.04 | 10 | 100 | na 1 | na 1 | na 1 |
| PdCl2 | na 1 | 10 | 100 | na 1 | na 1 | na 1 |
| CuCl2 | 2.89 | 10 | 800 | 14.9 | ||
| CuSO4 | 2.89 | 10 | 100 | 12 | 12 | 12.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Viau, M.; Sonzogni, L.; Ferlazzo, M.L.; Berthel, E.; Pereira, S.; Bodgi, L.; Granzotto, A.; Devic, C.; Fervers, B.; Charlet, L.; et al. DNA Double-Strand Breaks Induced in Human Cells by Twelve Metallic Species: Quantitative Inter-Comparisons and Influence of the ATM Protein. Biomolecules 2021, 11, 1462. https://doi.org/10.3390/biom11101462
Viau M, Sonzogni L, Ferlazzo ML, Berthel E, Pereira S, Bodgi L, Granzotto A, Devic C, Fervers B, Charlet L, et al. DNA Double-Strand Breaks Induced in Human Cells by Twelve Metallic Species: Quantitative Inter-Comparisons and Influence of the ATM Protein. Biomolecules. 2021; 11(10):1462. https://doi.org/10.3390/biom11101462
Chicago/Turabian StyleViau, Muriel, Laurène Sonzogni, Mélanie L. Ferlazzo, Elise Berthel, Sandrine Pereira, Larry Bodgi, Adeline Granzotto, Clément Devic, Béatrice Fervers, Laurent Charlet, and et al. 2021. "DNA Double-Strand Breaks Induced in Human Cells by Twelve Metallic Species: Quantitative Inter-Comparisons and Influence of the ATM Protein" Biomolecules 11, no. 10: 1462. https://doi.org/10.3390/biom11101462
APA StyleViau, M., Sonzogni, L., Ferlazzo, M. L., Berthel, E., Pereira, S., Bodgi, L., Granzotto, A., Devic, C., Fervers, B., Charlet, L., & Foray, N. (2021). DNA Double-Strand Breaks Induced in Human Cells by Twelve Metallic Species: Quantitative Inter-Comparisons and Influence of the ATM Protein. Biomolecules, 11(10), 1462. https://doi.org/10.3390/biom11101462







