Distinctive CD26 Expression on CD4 T-Cell Subsets
Abstract
:1. Introduction
2. Materials and Methods
2.1. Biological Samples
2.2. Ethics Statement
2.3. Flow Cytometry Analysis
2.4. Cell Culture and Polarization
2.5. Measurement of Soluble CD26 Protein
2.6. Statistical Analysis
3. Results
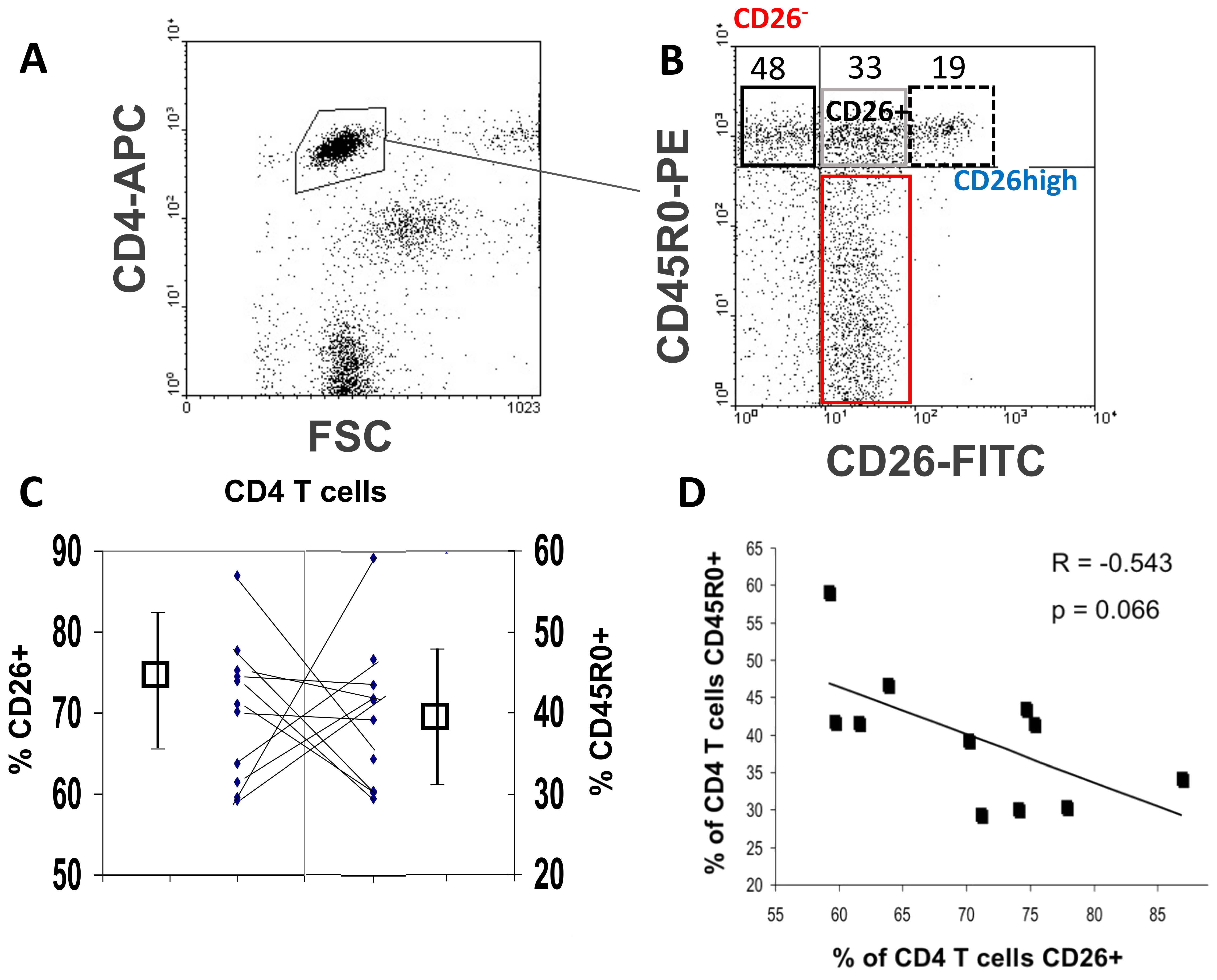
3.1. Relationship between the Cell Surface CD26 and CD45R0 Isoform in Human Peripheral Blood CD4 T Lymphocytes
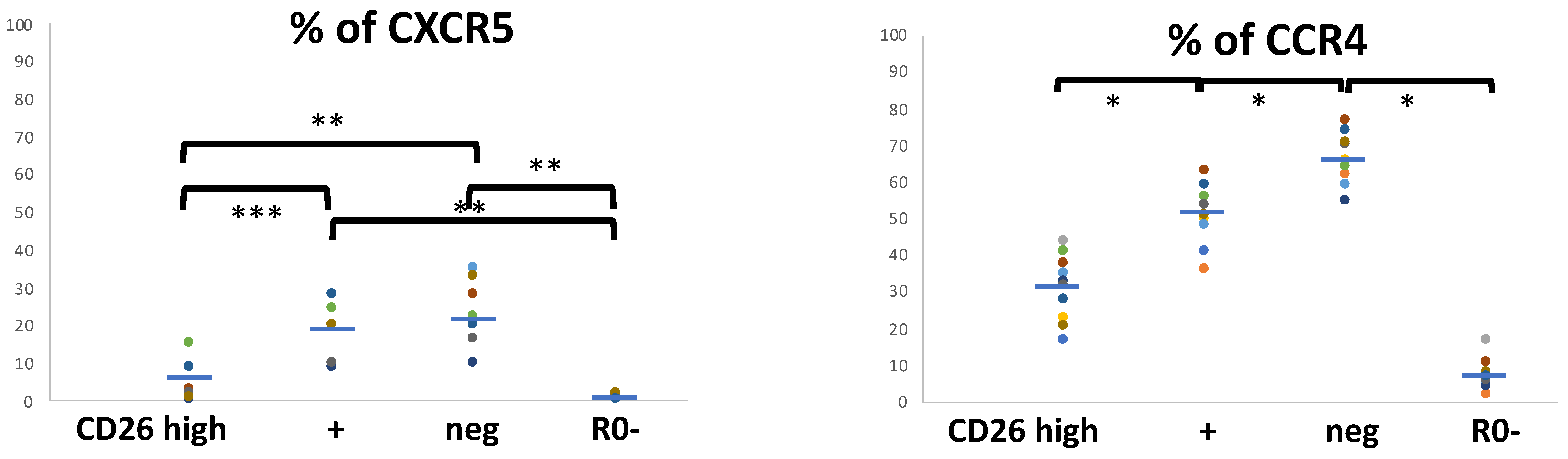
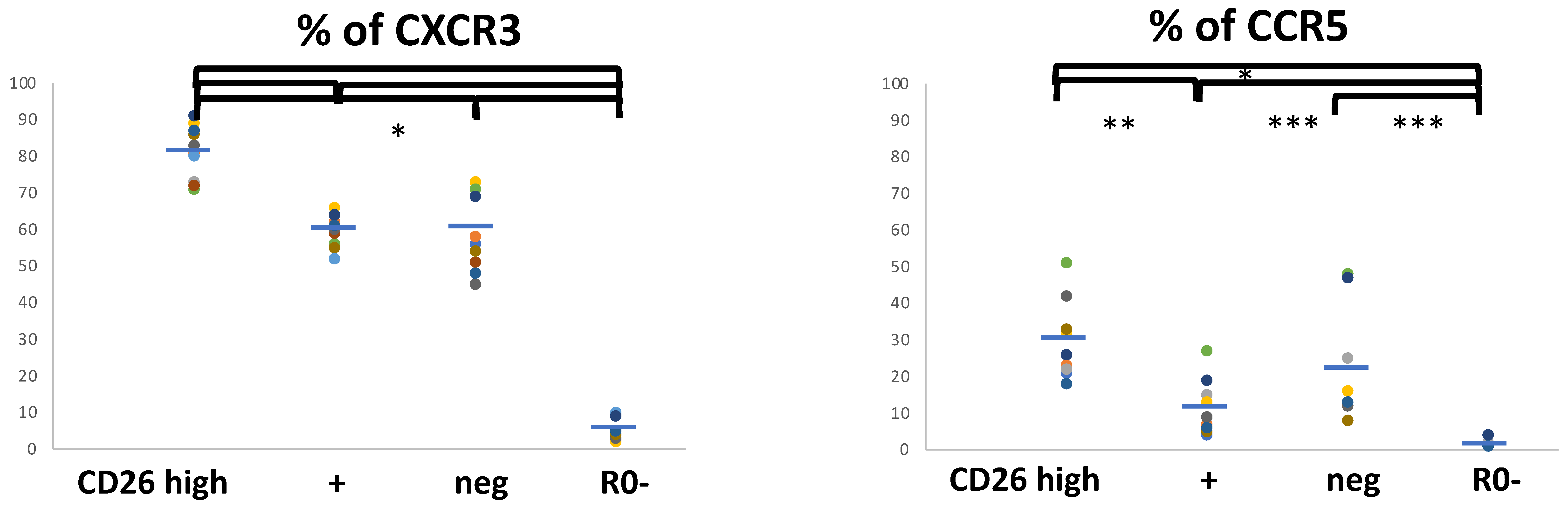
3.2. Relationship between the Cell-Surface CD26 and the Phenotype of Central (CM), Effector/Memory (EM), and Treg CD4 T Cell Subsets
3.3. Functional Programs in CM and EM Human T CD4 Cell Subsets in Relation to the Cell-Surface CD26
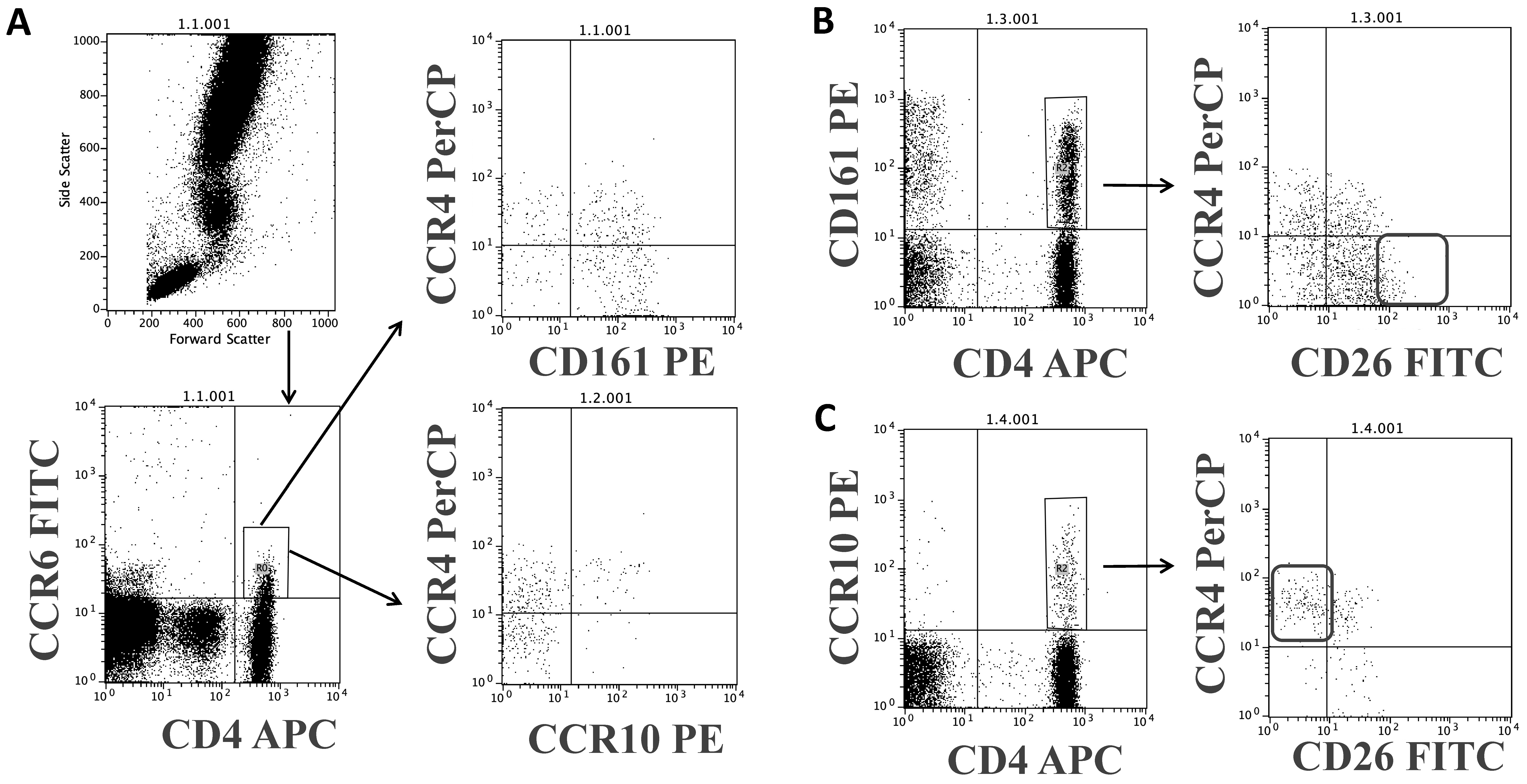
3.4. Relationship between CD26 and Th17 and Th22 CD4 Subsets
3.5. Cell-Surface CD26 in In Vitro Polarized CD4 T Lymphocytes
3.6. sCD26 in the Secretome of In Vitro Polarized CD4 T Lymphocytes
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bailey, S.R.; Nelson, M.H.; Majchrzak-Kuligowska, K.; Bowers, J.; Wyatt, M.M.; Smith, A.S.; Neal, L.R.; Shirai, K.; Carpenito, C.; June, C.H.; et al. Human CD26high T cells elicit tumor immunity against multiple malignancies via enhanced migration and persistence. Nat. Commun. 2017, 8, 1961. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nelson, M.H.; Knochelmann, H.M.; Bailey, S.R.; Huff, L.W.; Bowers, J.S.; Majchrzak-Kuligowska, K.; Wyatt, M.M.; Rubinstein, M.P.; Mehrotra, S.; Nishimura, M.I.; et al. Identification of human CD4+ T cell populations with distinct antitumor activity. Sci. Adv. 2020, 6, eaba7443. [Google Scholar] [CrossRef] [PubMed]
- Yawalkar, N.; Hunger, R.E.; Pichler, W.J.; Braathen, L.R.; Brand, C.U. Human afferent lymph from normal skin contains an increased number of mainly memory/effector CD4(+) T cells expressing activation, adhesion and co-stimulatory molecules. Eur. J. Immunol. 2000, 30, 491–497. [Google Scholar] [CrossRef]
- Krakauer, M.; Sorensen, P.S.; Sellebjerg, F. CD4+ memory T cells with high CD26 surface expression are enriched for Th1 markers and correlate with clinical severity of multiple sclerosis. J. Neuroimmunol. 2006, 181, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Cordero, O.J.; Salgado, F.J.; Viñuela, J.E.; Nogueira, M. Interleukin-12 enhances CD26 expression and dipeptidyl peptidase IV function on human activated lymphocytes. Immunobiology 1997, 197, 522–533. [Google Scholar] [CrossRef]
- Boonacker, E.P.; Wierenga, E.A.; Smits, H.H.; Van Noorden, C.J. CD26/DPPIV Signal Transduction Function, but Not Proteolytic Activity, Is Directly Related to Its Expression Level on Human Th1 and Th2 Cell Lines as Detected with Living Cell Cytochemistry. J. Histochem. Cytochem. 2002, 50, 1169–1177. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cordero, O.J.; Varela-Calviño, R.; López-González, T. CD26 Expression on T Helper Populations and sCD26 Serum Levels in Patients with Rheumatoid Arthritis. PLoS ONE 2015, 10, e0139535. [Google Scholar] [CrossRef]
- A Hafler, D.; A Fox, D.; Benjamin, D.; Weiner, H.L. Antigen reactive memory T cells are defined by Ta1. J. Immunol. 1986, 137. [Google Scholar]
- Morimoto, C.; Torimoto, Y.; Levinson, G.; E Rudd, C.; Schrieber, M.; Dang, N.H.; Letvin, N.L.; Schlossman, S.F. 1F7, a novel cell surface molecule, involved in helper function of CD4 cells. J. Immunol. 1989, 143, 3430–3439. [Google Scholar]
- Bengsch, B.; Seigel, B.; Flecken, T.; Wolanski, J.; Blum, H.E.; Thimme, R. Human Th17 Cells Express High Levels of Enzymatically Active Dipeptidylpeptidase IV (CD26). J. Immunol. 2012, 188, 5438–5447. [Google Scholar] [CrossRef] [Green Version]
- Dang, N.H.; Torimoto, Y.; Shimamura, K.; Tanaka, T.; Daley, J.F.; Schlossman, S.F.; Morimoto, C. 1F7 (CD26): A marker of thymic maturation involved in the differential regulation of the CD3 and CD2 pathways of human thymocyte activation. J. Immunol. 1991, 147, 2825–2832. [Google Scholar] [PubMed]
- Amlot, P.L.; Tahami, F.; Chinn, D.; Rawlings, E. Activation antigen expression on human T cells. I. Analysis by two-colour flow cytometry of umbilical cord blood, adult blood and lymphoid tissue. Clin. Exp. Immunol. 1996, 105. [Google Scholar] [CrossRef] [PubMed]
- Cordero, O.J.; Yang, C.P.; Bell, E.B. On the role of CD26 in CD4 memory T cells. Immunobiology 2007, 212, 85–94. [Google Scholar] [CrossRef]
- Ibegbu, C.C.; Xu, Y.X.; Fillos, D.; Radziewicz, H.; Grakoui, A.; Kourtis, A.P. Differential expression of CD26 on virus-specific CD8(+) T cells during active, latent and resolved infection. Immunology 2009, 126, 346–353. [Google Scholar] [CrossRef]
- Tejera-Alhambra, M.; Casrouge, A.; de Andrés, C. Low DPP4 expression and activity in multiple sclerosis. Clin. Immunol. 2014, 150, 170–183. [Google Scholar] [CrossRef]
- Ma, Y.; Visser, L.; Blokzijl, T.; Harms, G.; Atayar, Ç.; Poppema, S.; Berg, A.V.D. The CD4+CD26− T-cell population in classical Hodgkin’s lymphoma displays a distinctive regulatory T-cell profile. Lab. Investig. 2008, 88, 482–490. [Google Scholar] [CrossRef]
- Casrouge, A.; Sauer, A.; Da Silva, R.B.; Tejera-Alhambra, M.; Sánchez-Ramón, S.; B, I.C.A.R.; Cancrini, C.; Ingersoll, M.; Aiuti, A.; Albert, M. Lymphocytes are a major source of circulating soluble dipeptidyl peptidase 4. Clin. Exp. Immunol. 2018, 194, 166–179. [Google Scholar] [CrossRef] [Green Version]
- Cordero, J.; Salgado, F.J.; Nogueira, M. On the origin of serum CD26 and its altered concentration in cancer patients. Cancer Immunol. Immunother. 2009, 58, 1723–1747. [Google Scholar] [CrossRef] [PubMed]
- Klemann, C.; Wagner, L.; Stephan, M.; von Hörsten, S. Cut to the chase: A review of CD26/dipeptidyl peptidase-4's (DPP4) entanglement in the immune system. Clin. Exp. Immunol. 2016, 185, 1–21. [Google Scholar] [CrossRef] [Green Version]
- Nargis, T.; Chakrabarti, P. Significance of circulatory DPP4 activity in metabolic diseases. IUBMB Life 2018, 70, 112–119. [Google Scholar] [CrossRef] [Green Version]
- Morimoto, C.; Schlossman, S.F. The structure and function of CD26 in the T-cell immune response. Immunol. Rev. 1998, 161, 55–70. [Google Scholar] [CrossRef]
- Herlihy, S.E.; Brown, M.L.; Pilling, D.; Weeks, B.; Myers, L.K.; Gomer, R.H. Role of the Neutrophil Chemorepellent Soluble Dipeptidyl Peptidase IV in Decreasing Inflammation in a Murine Model of Arthritis. Arthritis Rheumatol. 2015, 67, 2634–2638. [Google Scholar] [CrossRef]
- White, M.J.; Chinea, L.E.; Pilling, D.; Gomer, R.H. Protease activated-receptor 2 is necessary for neutrophil chemorepulsion induced by trypsin, tryptase, or dipeptidyl peptidase IV. J. Leukoc. Biol. 2017, 103, 119–128. [Google Scholar] [CrossRef] [Green Version]
- Consalvo, K.; Rijal, R.; Tang, Y.; Kirolos, S.A.; Smith, M.; Gomer, R.H. Extracellular signaling in Dictyostelium. Int. J. Dev. Biol. 2019, 63, 395–405. [Google Scholar] [CrossRef] [Green Version]
- Treffers, L.W.; Hiemstra, I.H.; Kuijpers, T.W.; van den Berg, T.K.; Matlung, H.L. Neutrophils in cancer. Immunol. Rev. 2016, 273, 312–328. [Google Scholar] [CrossRef] [PubMed]
- Coffelt, S.; Wellenstein, M.D.; De Visser, S.B.C.M.D.W.K.E. Neutrophils in cancer: Neutral no more. Nat. Rev. Cancer 2016, 16, 431–446. [Google Scholar] [CrossRef] [Green Version]
- Arpinati, L.; Shaul, M.E.; Kaisar-Iluz, N.; Mali, S.; Mahroum, S.; Fridlender, Z.G. NETosis in cancer: A critical analysis of the impact of cancer on neutrophil extracellular trap (NET) release in lung cancer patients vs. mice. Cancer Immunol. Immunother. 2020, 69, 199–213. [Google Scholar] [CrossRef]
- Wronkowitz, N.; Görgens, S.W.; Romacho, T.; Villalobos, L.A.; Sánchez-Ferrer, C.F.; Peiró, C.; Sell, H.; Eckel, J. Soluble DPP4 induces inflammation and proliferation of human smooth muscle cells via protease-activated receptor 2. Biochim. Biophys. Acta -Mol. Basis Dis. 2014, 1842, 1613–1621. [Google Scholar] [CrossRef] [Green Version]
- Ohnuma, K.; Inoue, H.; Uchiyama, M. T-cell activation via CD26 and caveolin-1 in rheumatoid synovium. Mod. Rheumatol. 2006, 16, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Ohnuma, K.; Uchiyama, M.; Yamochi, T.; Nishibashi, K.; Hosono, O.; Takahashi, N.; Kina, S.; Tanaka, H.; Lin, X.; Dang, N.H.; et al. Caveolin-1 Triggers T-cell Activation via CD26 in Association with CARMA1. J. Biol. Chem. 2007, 282, 10117–10131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dismuke, W.M.; Klingeborn, M.; Stamer, W.D. Mechanism of Fibronectin Binding to Human Trabecular Meshwork Exosomes and Its Modulation by Dexamethasone. PLoS ONE 2016, 11, e0165326. [Google Scholar] [CrossRef]
- Schmidt, H.; Gelhaus, C.; Nebendahl, M.; Lettau, M.; Lucius, R.; Leippe, M.; Kabelitz, D.; Janssen, O. Effector granules in human T lymphocytes: The luminal proteome of secretory lysosomes from human T cells. Cell Commun. Signal. 2011, 9, 4. [Google Scholar] [CrossRef] [Green Version]
- Lettau, M.; Dietz, M.; Vollmers, S.; Armbrust, F.; Peters, C.; Dang, T.M.; Chitadze, G.; Kabelitz, D.; Janssen, O. Degranulation of human cytotoxic lymphocytes is a major source of proteolytically active soluble CD26/DPP4. Cell. Mol. Life Sci. 2019, 77, 751–764. [Google Scholar] [CrossRef]
- Angevin, E.; Isambert, N.; Trillet-Lenoir, V.; You, B.; Alexandre, J.; Zalcman, G.; Vielh, P.; Farace, F.; Valleix, F.; Podoll, T.; et al. First-in-human phase 1 of YS110, a monoclonal antibody directed against CD26 in advanced CD26-expressing cancers. Br. J. Cancer 2017, 116, 1126–1134. [Google Scholar] [CrossRef] [Green Version]
- Nishida, H.; Hayashi, M.; Morimoto, C.; Sakamoto, M.; Yamada, T. CD26 is a potential therapeutic target by humanized monoclonal antibody for the treatment of multiple myeloma. Blood Cancer J. 2018, 8, 99. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gómez-Touriño, I.; Sánchez-Espinel, C.; Hernández-Fernández, A.; González, F.; Pena-González, E.; Rodríguez, J.; García-López, J.M.; Varela-Calvino, R. Galectin-1 synthesis in type 1 diabetes by different immune cell types: Reduced synthesis by monocytes and Th1 cells. Cell. Immunol. 2011, 271, 319–328. [Google Scholar] [CrossRef] [PubMed]
- Sallusto, F.; Geginat, J.; Lanzavecchia, A. Central memory and effector memory T cell subsets: Function, generation, and maintenance. Annu. Rev. Immunol. 2004, 22, 745–763. [Google Scholar] [CrossRef] [PubMed]
- Wan, Q.; Kozhaya, L.; ElHed, A.; Ramesh, R.; Carlson, T.J.; Djuretic, I.M.; Sundrud, M.S.; Unutmaz, D. Cytokine signals through PI-3 kinase pathway modulate Th17 cytokine production by CCR6+ human memory T cells. J. Exp. Med. 2011, 208, 1875–1887. [Google Scholar] [CrossRef] [Green Version]
- Eyerich, S.; Eyerich, K.; Pennino, D.; Carbone, T.; Nasorri, F.; Pallotta, S.; Cianfarani, F.; Odorisio, T.; Traidl-Hoffmann, C.; Behrendt, H.; et al. Th22 cells represent a distinct human T cell subset involved in epidermal immunity and remodeling. J. Clin. Investig. 2009, 119, 3573–3585. [Google Scholar] [CrossRef] [Green Version]
- Zhao, F.; Hoechst, B.; Gamrekelashvili, J.; Ormandy, L.A.; Voigtländer, T.; Wedemeyer, H.; Ylaya, K.; Wang, X.W.; Hewitt, S.M.; Manns, M.P.; et al. Human CCR4+CCR6+Th17 Cells Suppress Autologous CD8+ T Cell Responses. J. Immunol. 2012, 188, 6055–6062. [Google Scholar] [CrossRef] [Green Version]
- Truchetet, M.-E.; Brembilla, N.C.; Montanari, E.; Allanore, Y.; Chizzolini, C. Increased frequency of circulating Th22 in addition to Th17 and Th2 lymphocytes in systemic sclerosis: Association with interstitial lung disease. Arthritis Res. Ther. 2011, 13, R166. [Google Scholar] [CrossRef] [Green Version]
- Malissen, B.; Bongrand, P. Early T Cell Activation: Integrating Biochemical, Structural, and Biophysical Cues. Annu. Rev. Immunol. 2015, 33, 539–561. [Google Scholar] [CrossRef]
- Sharma, P.; Wong, E.B.; Napier, R.J.; Bishai, W.R.; Ndung'U, T.; Kasprowicz, V.O.; Lewinsohn, D.A.; Lewinsohn, D.; Gold, M.C. High expression of CD26 accurately identifies human bacteria-reactive MR1-restricted MAIT cells. Immunology 2015, 145, 443–453. [Google Scholar] [CrossRef] [PubMed]
- Sugimoto, C.; Konno, T.; Wakao, R.; Fujita, H.; Fujita, H.; Wakao, H. Mucosal-Associated Invariant T Cell Is a Potential Marker to Distinguish Fibromyalgia Syndrome from Arthritis. PLoS ONE 2015, 10, e0121124. [Google Scholar] [CrossRef] [PubMed]
- Whiteside, T.L. Regulatory T cell subsets in human cancer: Are they regulating for or against tumor progression? Cancer Immunol. Immunother 2014, 63, 67–72. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Booth, N.J.; McQuaid, A.J.; Sobande, T.; Kissane, S.; Agius, E.; Jackson, S.E.; Salmon, M.; Falciani, F.; Yong, K.; Rustin, M.H.; et al. Different Proliferative Potential and Migratory Characteristics of Human CD4+ Regulatory T Cells That Express either CD45RA or CD45RO. J. Immunol. 2010, 184, 4317–4326. [Google Scholar] [CrossRef] [Green Version]
- DeLeeuw, R.J.; Kost, S.E.; Kakal, J.; Nelson, B. The Prognostic Value of FoxP3+ Tumor-Infiltrating Lymphocytes in Cancer: A Critical Review of the Literature. Clin. Cancer Res. 2012, 18, 3022–3029. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mandapathil, M.; Szczepanski, M.; Harasymczuk, M.; Ren, J.; Cheng, D.; Jackson, E.K.; Gorelik, E.; Johnson, J.; Lang, S.; Whiteside, T.L. CD26 expression and adenosine deaminase activity in regulatory T cells (Treg) and CD4+T effector cells in patients with head and neck squamous cell carcinoma. OncoImmunology 2012, 1, 659–669. [Google Scholar] [CrossRef] [Green Version]
- Jacks, R.D.; Keller, T.J.; Nelson, A.; Nishimura, M.I.; White, P.; Iwashima, M. Cell intrinsic characteristics of human cord blood naïve CD4 T cells. Immunol. Lett. 2017, 193, 51–57. [Google Scholar] [CrossRef]
- Salgado, F.J.; Lojo, J.; Alonso-Lebrero, J.L. A role for interleukin-12 in the regulation of T cell plasma membrane com-partmentation. J. Biol. Chem. 2003, 278, 24849–24857. [Google Scholar] [CrossRef] [Green Version]
- Mattern, T.; Reich, C.; Duchrow, M.; Ansorge, S.; Ulmer, A.J.; Flad, H.D. Antibody-induced modulation of CD26 surface expression. Immunology 1995, 84, 595–600. [Google Scholar]
- Salgado, F.J.; Vela, E.; Martín, M.; Franco, R.; Nogueira, M.; Cordero, O.J. Mechanisms of CD26/dipeptidyl peptidase IV cy-tokine-dependent regulation on human activated lymphocytes. Cytokine 2000, 12, 1136–1141. [Google Scholar] [CrossRef]
- Yeganeh, F.; Mousavi, S.M.J.; Hosseinzadeh-Sarband, S.; Ahmadzadeh, A.; Bahrami-Motlagh, H.; Hoseini, M.H.M.; Sattari, M.; Sohrabi, M.R.; Pouriran, R.; Dehghan, P. Association of CD26/dipeptidyl peptidase IV mRNA level in peripheral blood mononuclear cells with disease activity and bone erosion in rheumatoid arthritis. Clin. Rheumatol. 2018, 37, 3183–3190. [Google Scholar] [CrossRef] [PubMed]
- Bohm, S.K.; Gum, J.R., Jr.; Erickson, R.H.; Hicks, J.W.; Kim, Y.S. Human dipeptidyl peptidase IV gene promoter: Tissue-specific regulation from a TATA-less GC-rich sequence characteristic of a housekeeping gene promoter. Biochem. J. 1995, 311, 835–843. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bauvois, B.; Djavaheri-Mergny, M.; Rouillard, D.; Dumont, J.; Wietzerbin, J. Regulation of CD26/DPPIV gene expression by interferons and retinoic acid in tumor B cells. Oncogene 2000, 19, 265–272. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cordero, O.J.; Varela-Calvino, R.; López-González, T.; Grujic, M.; Juranic, Z.; Mouriño, C.; Hernández, R.; Rodríguez-López, M.; De La Iglesia, B.A.; Pego-Reigosa, J.M. Anti-CD26 autoantibodies are involved in rheumatoid arthritis and show potential clinical interest. Clin. Biochem. 2017, 50, 903–910. [Google Scholar] [CrossRef]
- Yazbeck, R.; Jaenisch, S.E.; Abbott, C.A. Potential disease biomarkers: Dipeptidyl peptidase 4 and fibroblast activation protein. Protoplasma 2017, 255, 375–386. [Google Scholar] [CrossRef]
- Cordero, O.J.; Imbernon, M.; Chiara, L.D. Potential of soluble CD26 as a serum marker for colorectal cancer detection. World J. Clin. Oncol. 2011, 2, 245–261. [Google Scholar] [CrossRef] [PubMed]
- Cho, E.-H.; Kim, S.-W. Soluble Dipeptidyl Peptidase-4 Levels Are Associated with Decreased Renal Function in Patients with Type 2 Diabetes Mellitus. Diabetes Metab. J. 2019, 43, 97–104. [Google Scholar] [CrossRef]
- Beckenkamp, A.; Davies, S.; Willig, J.B.; Buffon, A. DPPIV/CD26: A tumor suppressor or a marker of malignancy? Tumour. Biol. 2016, 7, 7059–7073. [Google Scholar] [CrossRef]
- Liu, Y.; Li, Y.; Gong, Y.; Yu, N.; Zhang, Y.; You, R.; Qu, C.; Lu, G.; Huang, Y.; Gao, Y.; et al. CD26 expression is down-regulated on CD8+ T cells in patients with Hashimoto's thyroiditis. Int. Immunopharmacol. 2017, 54, 280–285. [Google Scholar] [CrossRef] [PubMed]
- Manocha, E.; Bugatti, A.; Belleri, M.; Zani, A.; Marsico, S.; Caccuri, F.; Presta, M.; Caruso, A. Avian Reovirus P17 Suppresses Angiogenesis by Promoting DPP4 Secretion. Cells 2021, 10, 259. [Google Scholar] [CrossRef]

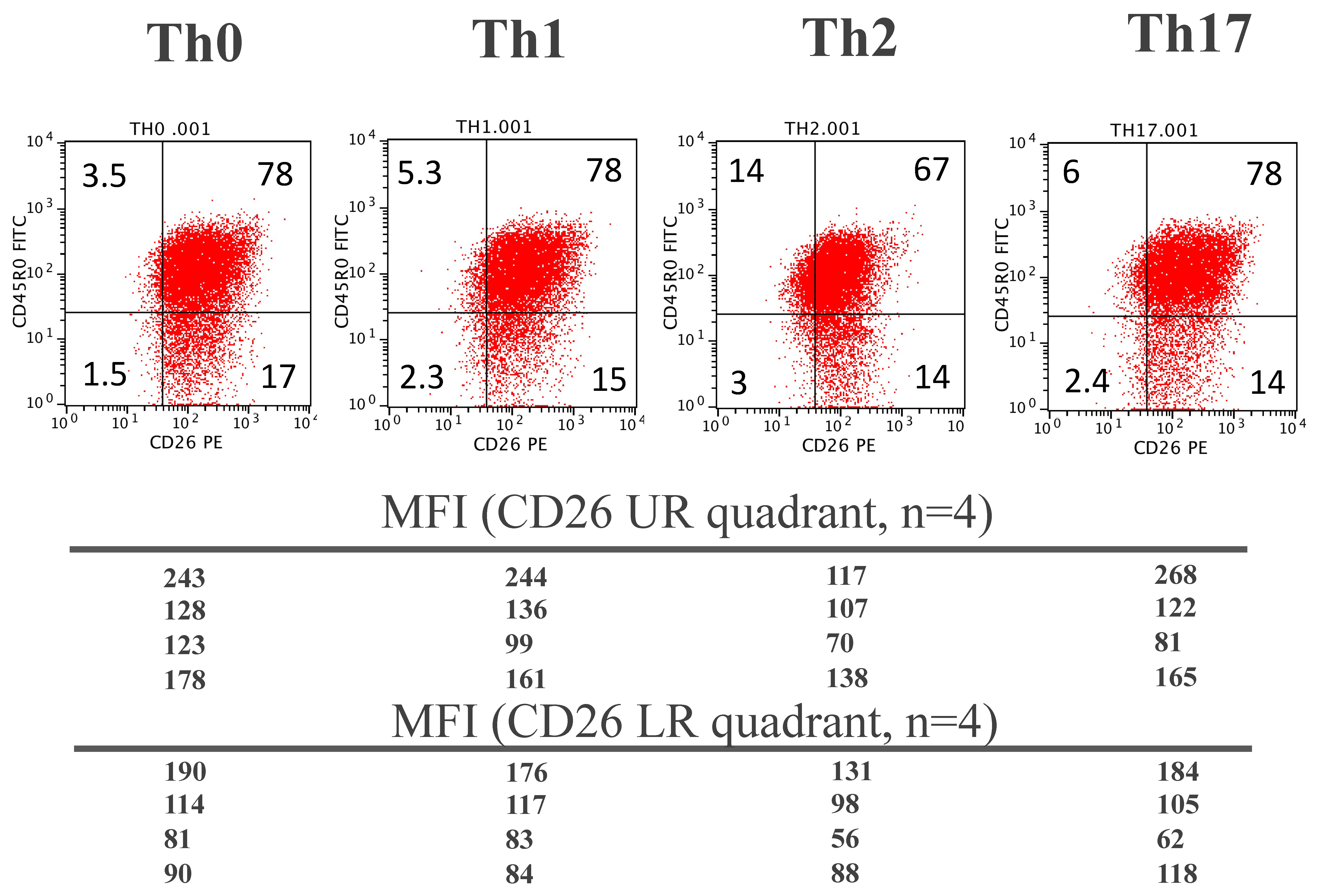
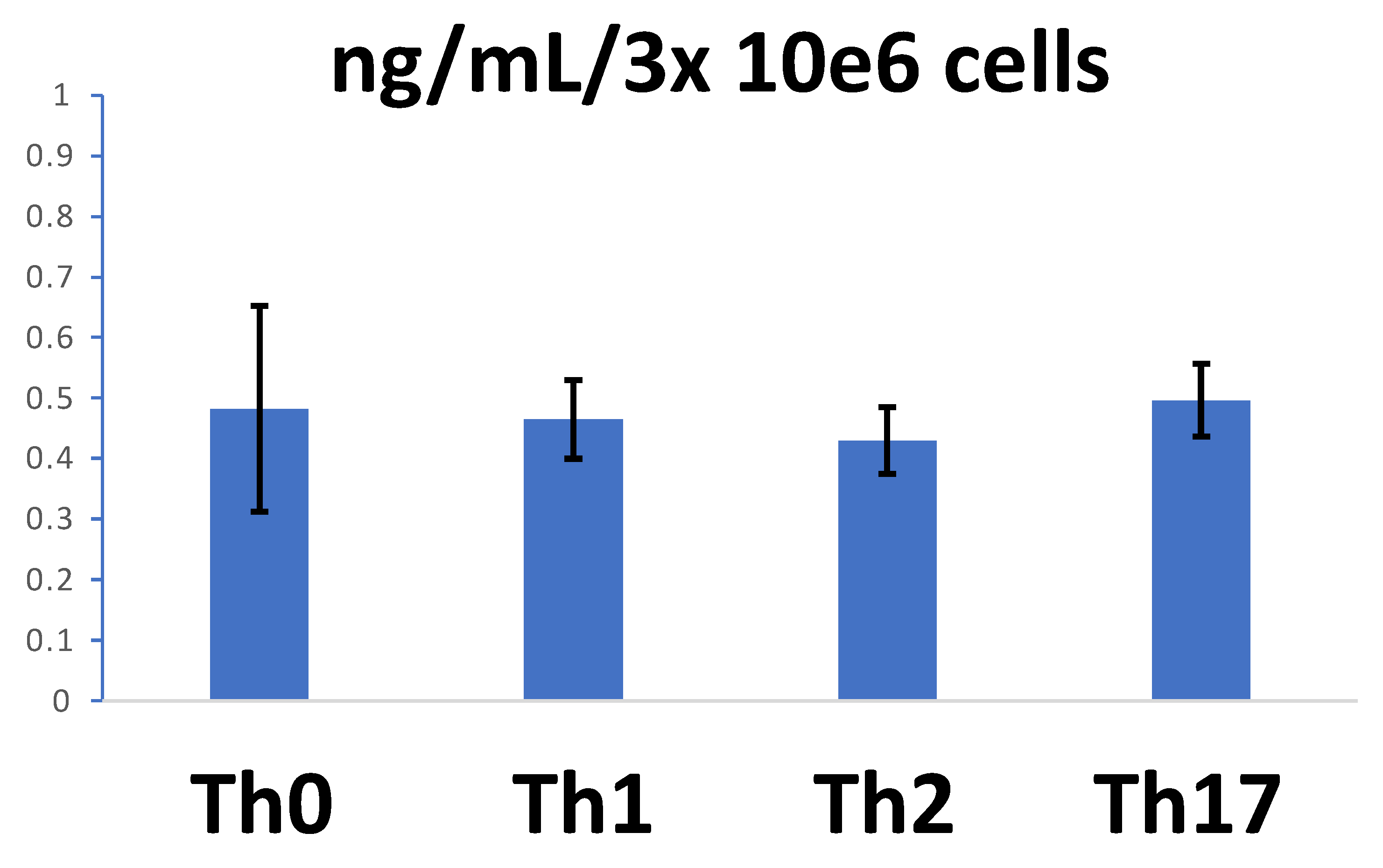
| Polarization Condition | |||
|---|---|---|---|
| Th1 | Th2 | Th17 | |
| −9 | −11 | −18 | |
| Soluble CD26 (sCD26) | −13 | −15 | −17 |
| −4 | −14 | 10 | |
| −20 | −22 | - | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cordero, O.J.; Rafael-Vidal, C.; Varela-Calviño, R.; Calviño-Sampedro, C.; Malvar-Fernández, B.; García, S.; Viñuela, J.E.; Pego-Reigosa, J.M. Distinctive CD26 Expression on CD4 T-Cell Subsets. Biomolecules 2021, 11, 1446. https://doi.org/10.3390/biom11101446
Cordero OJ, Rafael-Vidal C, Varela-Calviño R, Calviño-Sampedro C, Malvar-Fernández B, García S, Viñuela JE, Pego-Reigosa JM. Distinctive CD26 Expression on CD4 T-Cell Subsets. Biomolecules. 2021; 11(10):1446. https://doi.org/10.3390/biom11101446
Chicago/Turabian StyleCordero, Oscar J., Carlos Rafael-Vidal, Rubén Varela-Calviño, Cristina Calviño-Sampedro, Beatriz Malvar-Fernández, Samuel García, Juan E. Viñuela, and José M. Pego-Reigosa. 2021. "Distinctive CD26 Expression on CD4 T-Cell Subsets" Biomolecules 11, no. 10: 1446. https://doi.org/10.3390/biom11101446
APA StyleCordero, O. J., Rafael-Vidal, C., Varela-Calviño, R., Calviño-Sampedro, C., Malvar-Fernández, B., García, S., Viñuela, J. E., & Pego-Reigosa, J. M. (2021). Distinctive CD26 Expression on CD4 T-Cell Subsets. Biomolecules, 11(10), 1446. https://doi.org/10.3390/biom11101446







