Pyrroloquinoline-Quinone Is More Than an Antioxidant: A Vitamin-like Accessory Factor Important in Health and Disease Prevention
Abstract
1. Introduction
2. Chemistry and Biologic Mechanisms of Action
2.1. General Properties

2.2. PQQ as an Enzyme Cofactor
3. PQQ’s Role in Prokaryotes and Fungi
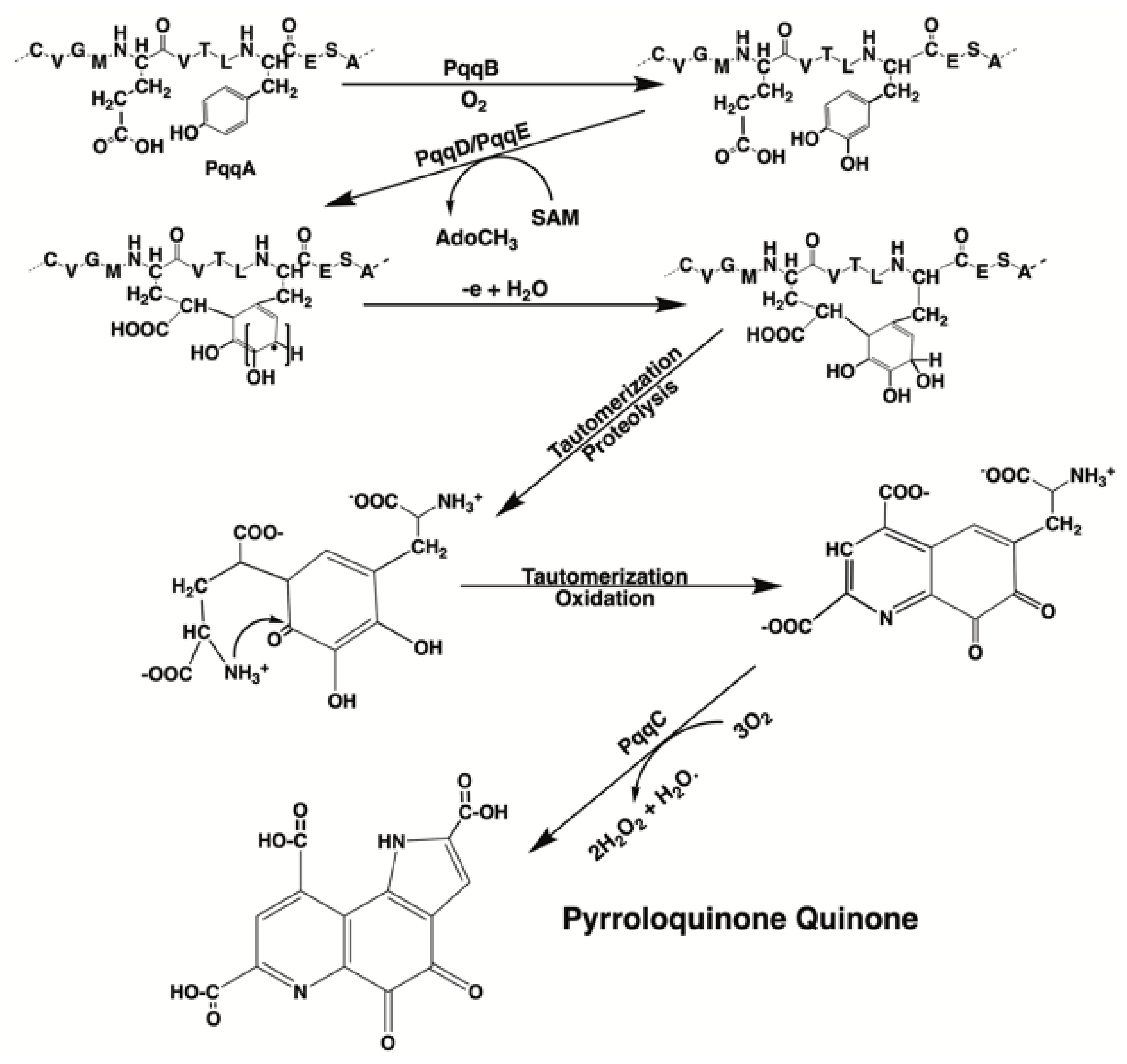
3.1. PQQ Synthesis
3.2. PQQ and Fungi
4. PQQ and Plant Growth
5. PQQ and Insects (Drosophila Melanogaster and Nematodes)
5.1. Drosophila Melanogaster
5.2. Nematodes
6. Nutritional Importance in Animal Models and Human Subjects
6.1. Murine and Rodents
6.2. Pigs
6.3. Chickens
6.4. Humans
7. PQQ and Its Derivatives in Diets and Supplements
8. Selected Clinical and Organ-Specific Dysfunctions Responsive to PQQ
8.1. Kidney and Liver
8.2. Intestinal Barrier Functions and the Microbiome
8.3. Cardiac and Skeletal Muscle Protection
8.4. PQQ and Neuroprotection
9. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hauge, J.G. Glucose dehydrogenase of Bacterium anitratum: An enzyme with a novel prosthetic group. J. Biol. Chem. 1964, 239, 3630–3639. [Google Scholar] [CrossRef]
- Frank, J.; Dijkstra, M.; Duine, J.A.; Balny, C.; Jzn, J.F. Kinetic and spectral studies on the redox forms of methanol dehydrogenase from Hyphomicrobium X. JBIC J. Biol. Inorg. Chem. 1988, 174, 331–338. [Google Scholar] [CrossRef]
- Duine, J.A.; Jzn, J.F.; Verwiel, P.E.J. Structure and activity of the prosthetic group of methanol dehydrogenase. JBIC J. Biol. Inorg. Chem. 1980, 108, 187–192. [Google Scholar] [CrossRef]
- Duine, J.A. Quinoproteins: Enzymes containing the quinonoid cofactor pyrroloquinoline quinone, topaquinone or tryptophan-tryptophan quinone. JBIC J. Biol. Inorg. Chem. 1991, 200, 271–284. [Google Scholar] [CrossRef]
- Anthony, C.; Ghosh, M. The structure and function of the PQQ-containing quinoprotein dehydrogenases. Prog. Biophys. Mol. Biol. 1998, 69, 1–21. [Google Scholar] [CrossRef]
- Salisbury, S.A.; Forrest, H.S.; Cruse, W.B.T.; Kennard, O. A novel coenzyme from bacterial primary alcohol dehydrogenases. Nat. Cell Biol. 1979, 280, 843–844. [Google Scholar] [CrossRef] [PubMed]
- Klinman, J.P.; Mu, D. Quinoenzymes in biology. Annu. Rev. Biochem. 1994, 63, 299–344. [Google Scholar] [CrossRef]
- Anthony, C. Pyrroloquinoline quinone (PQQ) and quinoprotein enzymes. Antioxid. Redox Signal. 2001, 3, 757–774. [Google Scholar] [CrossRef]
- Jonscher, K.R.; Rucker, R.B. Pyrroloquinoline quinone: Its profile. Effects on the liver and implications for health and disease prevention. In Dietary Interventions in Liver Disease; Watson, R.M., Preedy, V., Eds.; Academic Press: Cambridge, MA, USA, 2019; pp. 157–168. [Google Scholar]
- Zhao, L.; Gong, N.; Liu, M.; Pan, X.; Sang, S.; Sun, X.; Yu, Z.; Fang, Q.; Zhao, N.; Fei, G.; et al. Beneficial synergistic effects of microdose lithium with pyrroloquinoline quinone in an Alzheimer’s disease mouse model. Neurobiol. Aging 2014, 35, 2736–2745. [Google Scholar] [CrossRef]
- Nogueiras, R.; Habegger, K.M.; Chaudhary, N.; Finan, B.; Banks, A.S.; Dietrich, M.O.; Horvath, T.L.; Sinclair, D.A.; Pfluger, P.T.; Tschöop, M.H. Sirtuin 1 and sirtuin 3: Physiological modulators of metabolism. Physiol. Rev. 2012, 92, 1479–1514. [Google Scholar] [CrossRef] [PubMed]
- Stites, T.E.; Mitchell, A.E.; Rucker, R.B. Physiological importance of quinoenzymes and the O-quinone family of cofactors. J. Nutr. 2000, 130, 719–727. [Google Scholar] [CrossRef]
- McIntire, W.S. Newly discovered redox cofactors: Possible nutritional, medical, and pharmacological relevance to higher animals. Annu. Rev. Nutr. 1998, 18, 145–177. [Google Scholar] [CrossRef]
- Jayagobi, M.; Raghunathan, R.; Sainath, S.; Raghunathan, M. Synthesis and antibacterial property of pyrrolopyrano quinolinones and pyrroloquinolines. Eur. J. Med. Chem. 2011, 46, 2075–2082. [Google Scholar] [CrossRef]
- Várnai, A.; Umezawa, K.; Yoshida, M.; Eijsink, V.G.H. The Pyrroloquinoline-quinone-dependent pyranose dehydrogenase from Coprinopsis cinerea drives lytic polysaccharide monooxygenase action. Appl. Environ. Microbiol. 2018, 84, 00156–18. [Google Scholar] [CrossRef]
- Ouchi, A.; Ikemoto, K.; Nakano, M.; Nagaoka, S.I.; Mukai, K. Kinetic study of aroxyl radical scavenging and alpha-tocopheroxyl regeneration rates of pyrroloquinoline quinol (PQQH2, a reduced form of pyrroloquinoline quinone) in dimethyl sulfoxide solution: Finding of synergistic effect on the reaction rate due to the coexistence of alpha-tocopherol and PQQH2. J. Agric. Food Chem. 2013, 61, 11048–11060. [Google Scholar] [PubMed]
- Chan, S.I.; Chuankhayan, P.; Nareddy, P.K.R.; Tsai, I.-K.; Tsai, Y.-F.; Chen, K.H.-C.; Yu, S.S.-F.; Chen, C.-J. Mechanism of pyrroloquinoline quinone-dependent hydride transfer chemistry from spectroscopic and high-resolution X-ray structural studies of the methanol dehydrogenase from Methylococcus capsulatus (Bath). J. Am. Chem. Soc. 2021, 143, 3359–3372. [Google Scholar] [CrossRef]
- Akagawa, M.; Minematsu, K.; Shibata, T.; Kondo, T.; Ishii, T.; Uchida, K. Identification of lactate dehydrogenase as a mammalian pyrroloquinoline quinone (PQQ)-binding protein. Sci. Rep. 2016, 6, 26723. [Google Scholar] [CrossRef]
- Akagawa, M.; Nakano, M.; Ikemoto, K. Recent progress in studies on the health benefits of pyrroloquinoline quinone. Biosci. Biotechnol. Biochem. 2016, 80, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Tanno, M.; Sakamoto, J.; Miura, T.; Shimamoto, K.; Horio, Y. Nucleocytoplasmic Shuttling of the NAD+-dependent Histone Deacetylase SIRT1. J. Biol. Chem. 2007, 282, 6823–6832. [Google Scholar] [CrossRef]
- Onyango, P.; Celic, I.; McCaffery, J.M.; Boeke, J.D.; Feinberg, A.P. SIRT3, a human SIR2 homologue, is an NAO-dependent deacetylase localized to mitochondria. Proc. Natl. Acad. Sci. USA 2002, 99, 13653–13658. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Kerins, M.J.; Tian, W.; Neupane, D.; Zhang, D.D.; Ooi, A. Differential and overlapping targets of the transcriptional regulators NRF1, NRF2, and NRF3 in human cells. J. Biol. Chem. 2019, 294, 18131–18149. [Google Scholar] [CrossRef]
- Tchaparian, E.; Marshal, L.; Cutler, G.; Bauerly, K.; Chowanadisai, W.; Satre, M.; Harris, C.; Rucker, R.B. Identification of transcriptional networks responding to pyrroloquinoline quinone dietary supplementation and their influence on thioredoxin expression, and the JAK/STAT and MAPK pathways. Biochem. J. 2010, 429, 515–526. [Google Scholar] [CrossRef] [PubMed]
- Arthur, J.S.C.; Ley, S.C. Mitogen-activated protein kinases in innate immunity. Nat. Rev. Immunol. 2013, 13, 679–692. [Google Scholar] [CrossRef]
- Dong, C.; Davis, R.J.; Flavell, R.A. MAP kinases in the immune response. Annu. Rev. Immunol. 2002, 20, 55–72. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.K.; Choi, E.-J. Compromised MAPK signaling in human diseases: An update. Arch. Toxicol. 2015, 89, 867–882. [Google Scholar] [CrossRef]
- Lyons, C.L.; Roche, H.M. Nutritional modulation of AMPK-impact upon metabolic-inflammation. Int. J. Mol. Sci. 2018, 19, 3092. [Google Scholar] [CrossRef] [PubMed]
- Peti, W.; Page, R. Molecular basis of MAP kinase regulation. Protein Sci. 2013, 22, 1698–1710. [Google Scholar] [CrossRef] [PubMed]
- Salminen, A.; Kauppinen, A.; Kaarniranta, K. AMPK activation inhibits the functions of myeloid-derived suppressor cells (MDSC): Impact on cancer and aging. J. Mol. Med. 2019, 97, 1049–1064. [Google Scholar] [CrossRef]
- Sun, Y.; Liu, W.Z.; Liu, T.; Feng, X.; Yang, N.; Zhou, H.F. Signaling pathway of MAP Kl ERK in cell proliferation, differentiation, migration, senescence and apoptosis. J. Recept. Signal Transduct. Res. 2015, 35, 600–604. [Google Scholar] [CrossRef]
- Villarino, A.V.; Kanno, Y.; O’Shea, J.J. Mechanisms and consequences of Jak–STAT signaling in the immune system. Nat. Immunol. 2017, 18, 374–384. [Google Scholar] [CrossRef]
- Xin, P.; Xu, X.; Deng, C.; Liu, S.; Wang, Y.; Zhou, X.; Ma, H.; Wei, D.; Sun, S. The role of JAK/STAT signaling pathway and its inhibitors in diseases. Int. Immunopharmacol. 2020, 80, 106210. [Google Scholar] [CrossRef]
- Shen, Y.-Q.; Bonnot, F.; Imsand, E.M.; RoseFigura, J.M.; Sjölander, K.; Klinman, J.P. Distribution and properties of the genes encoding the biosynthesis of the bacterial cofactor, pyrroloquinoline quinone. Biochemistry 2012, 51, 2265–2275. [Google Scholar] [CrossRef]
- Nguyen, T.M.; Naoki, K.; Kataoka, N.; Matsutani, M.; Ano, Y.; Adachi, O.; Matsushita, K.; Yakushi, T. Characterization of a cryptic, pyrroloquinoline quinone-dependent dehydrogenase of Gluconobacter sp. strain CHM43. Biosci. Biotechnol. Biochem. 2021, 85, 998–1004. [Google Scholar] [CrossRef]
- Yakushi, T.; Terada, Y.; Ozaki, S.; Kataoka, N.; Akakabe, Y.; Adachi, O.; Matsutani, M.; Matsushita, K. Aldopentoses as new substrates for the membrane-bound, pyrroloquinoline quinone-dependent glycerol (polyol) dehydrogenase of Gluconobacter sp. Appl. Microbiol. Biotechnol. 2018, 102, 3159–3171. [Google Scholar] [CrossRef]
- Zhu, W.; Klinman, J.P. Biogenesis of the peptide-derived redox cofactor pyrroloquinoline quinone. Curr. Opin. Chem. Biol. 2020, 59, 93–103. [Google Scholar] [CrossRef] [PubMed]
- Pereira, J.; Lupas, A.N. The VCBS superfamily forms a third supercluster of beta-propellers that includes tachylectin and integrins. Bioinformatics 2021, 36, 5618–5622. [Google Scholar] [CrossRef] [PubMed]
- Matsumura, H.; Umezawa, K.; Takeda, K.; Sugimoto, N.; Ishida, T.; Samejima, M.; Ohno, H.; Yoshida, M.; Igarashi, K.; Nakamura, N. Discovery of a eukaryotic pyrroloquinoline quinone-dependent oxidoreductase belonging to a new auxiliary activity family in the database of carbohydrate-active enzymes. PLoS ONE 2014, 9, e104851. [Google Scholar] [CrossRef] [PubMed]
- Takeda, K.; Ishida, T.; Yoshida, M.; Samejima, M.; Ohno, H.; Igarashi, K.; Nakamura, N. Crystal structure of the catalytic and cytochrome b domains in a eukaryotic pyrroloquinoline quinone-dependent dehydrogenase. Appl. Environ. Microbiol. 2019, 85, e01692–19. [Google Scholar] [CrossRef]
- Takeda, K.; Matsumura, H.; Ishida, T.; Samejima, M.; Ohno, H.; Yoshida, M.; Igarashi, K.; Nakamura, N. Characterization of a novel pqq-dependent quinohemoprotein pyranose dehydrogenase from Coprinopsis cinerea Classified into auxiliary activities family 12 in carbohydrate-active enzymes. PLoS ONE 2015, 10, e0115722. [Google Scholar] [CrossRef]
- Takeda, K.; Umezawa, K.; Várnai, A.; Eijsink, V.G.; Igarashi, K.; Yoshida, M.; Nakamura, N. Fungal PQQ-dependent dehydrogenases and their potential in biocatalysis. Curr. Opin. Chem. Biol. 2019, 49, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Ohsugi, M.; Noda, H.; Muro, K.; Ishiba, A.; Kondo, Y.; Nakao, S. Effects of the yeast extract components pyrroloquinoline quinone and aspartic acid on vitamin B12 production in Klebsiella pneumoniae IFO 13541. J. Nutr. Sci. Vitaminol. 1989, 35, 661–665. [Google Scholar] [CrossRef]
- Bharwad, K.; Rajkumar, S. Modulation of PQQ-dependent glucose dehydrogenase (mGDH and sGDH) activity by succinate in phosphate solubilizing plant growth promoting Acinetobacter sp. SK2. 3 Biotech 2020, 10, 5. [Google Scholar] [CrossRef]
- Choi, O.; Kim, J.; Kim, J.G.; Jeong, Y.; Moon, J.S.; Park, C.S.; Hwang, I. Pyrroloquinoline quinone is a plant growth promotion factor produced by Pseudomonas fluorescens B16. Plant Physiol. 2008, 146, 657–668. [Google Scholar] [CrossRef] [PubMed]
- Choudhary, P.; Bhowmik, A.; Chakdar, H.; Khan, M.A.; Selvaraj, C.; Singh, S.K.; Murugan, K.; Kumar, S.; Saxena, A.K. Understanding the biological role of PqqB in Pseudomonas stutzeri using molecular dynamics simulation approach. J. Biomol. Struct. Dyn. 2020, 8, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Crespo, J.M.; Boiardi, J.L.; Luna, M.F. Mineral phosphate solubilization activity of Gluconacetobacter diazotrophicus under P-limitation and plant root environment. Agric. Sci. 2011, 2, 16–22. [Google Scholar] [CrossRef]
- Eotieno, N.; Lally, R.D.; Ekiwanuka, S.; Elloyd, A.; Eryan, D.; Germaine, K.J.; Dowling, D.N. Plant growth promotion induced by phosphate solubilizing endophytic Pseudomonas isolates. Front. Microbiol. 2015, 6, 745. [Google Scholar] [CrossRef]
- You, M.; Fang, S.; MacDonald, J.; Xu, J.; Yuan, Z.-C. Isolation and characterization of Burkholderia cenocepacia CR318, a phosphate solubilizing bacterium promoting corn growth. Microbiol. Res. 2019, 233, 126395. [Google Scholar] [CrossRef] [PubMed]
- Naveed, M.; Sohail, Y.; Khalid, N.; Ahmed, I.; Mumtaz, A.S. Evaluation of glucose dehydrogenase and pyrroloquinoline quinine (pqq) mutagenesis that renders functional inadequacies in host plants. J. Microbiol. Biotechnol. 2015, 25, 1349–1360. [Google Scholar] [CrossRef]
- Shrivastava, M.; Rajpurohit, Y.S.; Misra, H.S.; D’Souza, S.F. Survival of phosphate-solubilizing bacteria against DNA damaging agents. Can. J. Microbiol. 2010, 56, 822–830. [Google Scholar] [CrossRef][Green Version]
- Li, L.; Jiao, Z.; Hale, L.; Wu, W.; Guo, Y. Disruption of gene pqqA or pqqb reduces plant growth promotion activity and biocontrol of crown gall disease by Rahnella aquatilis HX2. PLoS ONE 2014, 9, e115010. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, K.; Twyman, R.M.; Vilcinskas, A. Insects as models to study the epigenetic basis of disease. Prog. Biophys. Mol. Biol. 2015, 118, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Ng, C.H.; Basil, A.H.; Hang, L.; Tan, R.; Goh, K.L.; O’Neill, S.; Zhang, X.; Yu, F.; Lim, K.L. Genetic or pharmacological activation of the Drosophila PGC-1alpha ortholog spargel rescues the disease phenotypes of genetic models of Parkinson’s disease. Neurobiol. Aging 2017, 55, 33–37. [Google Scholar] [CrossRef] [PubMed]
- Puschmann, A. Monogenic Parkinson’s disease and parkinsonism: Clinical phenotypes and frequencies of known mutations. Parkinsonism Relat. Disord. 2013, 19, 407–415. [Google Scholar] [CrossRef]
- Shin, S.C.; Kim, S.-H.; You, H.; Kim, B.; Kim, A.C.; Lee, K.-A.; Yoon, J.-H.; Ryu, J.-H.; Lee, W.-J. Drosophila microbiome modulates host developmental and metabolic homeostasis via insulin signaling. Science 2011, 334, 670–674. [Google Scholar] [CrossRef]
- Wu, J.; Huang, J.; Khanabdali, R.; Kalionis, B.; Xia, S.; Cai, W. Pyrroloquinoline quinone enhances the resistance to oxidative stress and extends lifespan upon DAF-16 and SKN-1 activities in C. elegans. Exp. Gerontol. 2016, 80, 43–50. [Google Scholar] [CrossRef]
- Sasakura, H.; Moribe, H.; Nakano, M.; Ikemoto, K.; Takeuchi, K.; Mori, I. Lifespan extension by peroxidase/dual oxidase-mediated ROS signaling through pyrroloquinoline quinone in C. elegans. J. Cell Sci. 2017, 130, 2631–2643. [Google Scholar] [CrossRef] [PubMed]
- Smidt, C.R.; Unkefer, C.J.; Houck, D.R.; Rucker, R.B. Intestinal absorption and tissue distribution of [14C]pyrroloquinoline quinone in mice. Exp. Biol. Med. 1991, 197, 27–31. [Google Scholar] [CrossRef]
- Smidt, C.R.; Bean-Knudsen, D.; Kirsch, D.G.; Rucker, R.B. Does the intestinal microflora synthesize pyrroloquinoline quinone? BioFactors 1991, 3, 53–59. [Google Scholar]
- Steinberg, F.; Stites, T.E.; Anderson, P.; Storms, D.; Chan, I.; Eghbali, S.; Rucker, R. pyrroloquinoline quinone improves growth and reproductive performance in mice fed chemically defined diets. Exp. Biol. Med. 2003, 228, 160–166. [Google Scholar] [CrossRef] [PubMed]
- Steinberg, F.M.; Gershwin, M.E.; Rucker, R.B. Dietary pyrroloquinoline quinone: Growth and immune response in BALB/c mice. J. Nutr. 1994, 124, 744–753. [Google Scholar] [CrossRef]
- Rucker, R.; Killgore, J.; Duich, L.; Romero-Chapman, N.; Smidt, C.; Tinker, D. Nutritional essentiality of pyrroloquinoline quinone. In PQQ and Quinoproteins: Proceedings of the First International Symposium on PQQ and Quinoprotein, Delft, The Netherlands; Jongejan, J.A., Duine, J.A., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1989; pp. 159–161. [Google Scholar]
- Yin, X.; Ming, D.; Bai, L.; Wu, F.; Liu, H.; Chen, Y.; Sun, L.; Wan, Y.; Thacker, P.A.; Wu, G.; et al. Effects of pyrroloquinoline quinone supplementation on growth performance and small intestine characteristics in weaned pigs. J. Anim. Sci. 2019, 97, 246–256. [Google Scholar] [CrossRef]
- Wang, C.; Zhang, B.; Zhang, H.; Yang, W.; Meng, Q.; Shi, B.; Shan, A. Effect of dietary pyrroloquinoline quinone disodium in sows on intestinal health of the offspring. Food Funct. 2020, 11, 7804–7816. [Google Scholar] [CrossRef]
- Zhang, H.; Li, J.; Cao, C.; Zhang, B.; Yang, W.; Shi, B.; Shan, A. Pyrroloquinoline quinone inhibits the production of inflammatory cytokines via the SIRTl/NF-κΒ signal pathway in weaned piglet jejunum. Food Funct. 2020, 11, 2137–2153. [Google Scholar] [CrossRef] [PubMed]
- Samuel, K.; Zhang, H.; Wang, J.; Wu, S.; Yue, H.; Sun, L.; Qi, G. Effects of dietary pyrroloquinoline quinone disodium on growth performance, carcass yield and antioxidant status of broiler chicks. Animal 2015, 9, 409–416. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Zhang, J.; Zhou, H.; Guo, Y.; Ma, Q.; Ji, C.; Zhao, L. Effects of dietary pyrroloquinoline quinone disodium supplementation on inflammatory responses, oxidative stress, and intestinal morphology in broiler chickens challenged with lipopolysaccharide. Poult. Sci. 2020, 99, 5389–5398. [Google Scholar] [CrossRef]
- Wideman, R.; Bowen, O.; Erf, G. Broiler pulmonary hypertensive responses during lipopolysaccharide-induced tolerance and cyclooxygenase inhibition. Poult. Sci. 2009, 88, 72–85. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Li, J.; Zhang, S.; Mi, Y.; Zhang, C. Attenuating effect of melatonin on lipopolysaccharide-induced chicken small intestine inflammation. Poult. Sci. 2018, 97, 2295–2302. [Google Scholar] [CrossRef]
- Harris, C.B.; Chowanadisai, W.; Mishchuk, D.O.; Satre, M.A.; Slupsky, C.M.; Rucker, R.B. Dietary pyrroloquinoline quinone (PQQ) alters indicators of inflammation and mitochondrial-related metabolism in human subjects. J. Nutr. Biochem. 2013, 24, 2076–2084. [Google Scholar] [CrossRef]
- Hwang, P.S.; Machek, S.B.; Cardaci, T.D.; Wilburn, D.; Kim, C.S.; Suezaki, E.S.; Willoughby, D.S. Effects of pyrroloquinoline quinone (PQQ) supplementation on aerobic exercise performance and indices of mitochondrial biogenesis in untrained men. J. Am. Coll. Nutr. 2019, 39, 547–556. [Google Scholar] [CrossRef] [PubMed]
- Wen, H.; He, Y.; Zhang, K.; Yang, X.; Hao, D.; Jiang, Y.; He, B. Mini-review: Functions and action mechanisms of PQQ in osteoporosis and neuro injury. Curr. Stem Cell Res. Ther. 2020, 15, 32–36. [Google Scholar] [CrossRef]
- Shiojima, Y.; Takahashi, M.; Takahashi, R.; Moriyama, H.; Bagchi, D.; Bagchi, M.; Akanuma, M. Effect of dietary pyrroloquinoline quinone disodium salt on cognitive function in healthy volunteers: A randomized, double-blind, placebo-controlled, parallel-group study. J. Am. Coll. Nutr. 2021, 20, 1–14. [Google Scholar] [CrossRef]
- Nakano, M.; Murayama, Y.; Hu, L.; Ikemoto, K.; Uetake, T.; Sakatani, K. Effects of antioxidant supplements (BioPQQ™) on cerebral blood flow and oxygen metabolism in the prefrontal cortex. Adv. Exp. Med. Biol. 2016, 923, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, A.; Jones, A.; Mercer, R.; Rucker, R. Characterization of pyrroloquinoline quinone amino acid derivatives by electrospray ionization mass spectrometry and detection in human milk. Anal. Biochem. 1999, 269, 317–325. [Google Scholar] [CrossRef] [PubMed]
- Kato, C.; Kawai, E.; Shimizu, N.; Mikekado, T.; Kimura, F.; Miyazawa, T.; Nakagawa, K. Determination of pyrroloquinoline quinone by enzymatic and LC-MS/MS methods to clarify its levels in foods. PLoS ONE 2018, 13, e0209700. [Google Scholar] [CrossRef]
- Stites, T.E.; Sih, T.R.; Rucker, R.B. Synthesis of [14C]pyrroloquinoline quinone (PQQ) in E. coli using genes for PQQ synthesis from K. pneumoniae. Biochim. Biophys. Acta (BBA)-Gen. Subj. 2000, 1524, 247–252. [Google Scholar] [CrossRef]
- Misset-Smits, M.; Oltshoorn, A.; Dewanti, A.; Duine, J. [11] Production, assay, and occurrence of pyrroloquinoline quinone. Methods Enzymol. 1997, 280, 89–98. [Google Scholar] [CrossRef]
- Stites, T.; Storms, D.; Bauerly, K.; Mah, J.; Harris, C.; Fascetti, A.; Rogers, Q.; Tchaparian, E.; Satre, M.; Rucker, R.B. Pyrroloquinoline quinone modulates mitochondrial quantity and function in mice. J. Nutr. 2006, 136, 390–396. [Google Scholar] [CrossRef] [PubMed]
- Wallace, T.C.; Giusti, M.M. Anthocyanins. Adv. Nutr. 2015, 6, 620–622. [Google Scholar] [CrossRef]
- Flückiger, R.; Apaz, M.; Gallop, P.M. [11] Redox-cycling detection of dialyzable pyrroloquinoline quinone and quinoproteins. Methods Enzymol. 1995, 258, 140–149. [Google Scholar] [CrossRef]
- Fluckiger, R.; Paz, M.; Mah, J.; Bishop, A.; Gallop, P. Characterization of the glycine-dependent redox-cycling activity in animal fluids and tissues using specific inhibitors and activators: Evidence for presence of PQQ. Biochem. Biophys. Res. Commun. 1993, 196, 61–68. [Google Scholar] [CrossRef]
- Rucker, R.; Chowanadisai, W.; Nakano, M. Potential physiological importance of pyrroloquinoline quinone. Altern. Med. Rev. J. Clin. Ther. 2009, 14, 268–277. [Google Scholar]
- Naito, Y.; Kumazawa, T.; Kino, I.; Suzuki, O. Effects of pyrroloquinoline quinone (PQQ) and PQQ-oxazole on DNA synthesis of cultured human fibroblasts. Life Sci. 1993, 52, 1909–1915. [Google Scholar] [CrossRef]
- Yamada, Y.; Nishii, K.; Kuwata, K.; Nakamichi, M.; Nakanishi, K.; Sugimoto, A.; Ikemoto, K. Effects of pyrroloquinoline quinone and imidazole pyrroloquinoline on biological activities and neural functions. Heliyon 2020, 6, e03240. [Google Scholar] [CrossRef] [PubMed]
- Tsuchida, T.; Yasuyama, T.; Higuchi, K.; Watanabe, A.; Urakami, T.; Akaike, T.; Sato, K.; Maeda, H. The protective effect of pyrroloquinoline quinone and its derivatives against carbon tetrachloride-induced liver injury of rats. J. Gastroenterol. Hepatol. 1993, 8, 342–347. [Google Scholar] [CrossRef]
- GRAS Notice for Pyrroloquinoline Quinone (PQQ) Disodium Salt; (GRN) No. 694; Office of Food Additive Safety (FHS-200) Center for Food Safety and Applied Nutrition Food and Drug Administration: Washinton, DC, USA, 2017.
- Liang, C.; Zhang, X.; Wang, W.; Song, Y.; Jia, X. A subchronic oral toxicity study on pyrroloquinoline quinone (PQQ) disodium salt in rats. Food Chem. Toxicol. 2015, 75, 146–150. [Google Scholar] [CrossRef]
- Nakano, M.; Suzuki, H.; Imamura, T.; Lau, A.; Lynch, B. Genotoxicity of pyrroloquinoline quinone (PQQ) disodium salt (BioPQQ™). Regul. Toxicol. Pharmacol. 2013, 67, 189–197. [Google Scholar] [CrossRef] [PubMed]
- Nakano, M.; Takahashi, H.; Koura, S.; Chung, C.; Tafazoli, S.; Roberts, A. Acute and subchronic toxicity studies of pyrroloquinoline quinone (PQQ) disodium salt (BioPQQ™) in rats. Regul. Toxicol. Pharmacol. 2014, 70, 107–121. [Google Scholar] [CrossRef]
- Hill, N.R.; Fatoba, S.T.; Oke, J.L.; Hirst, J.; O’Callaghan, C.A.; Lasserson, D.; Hobbs, R. Global prevalence of chronic kidney—A systematic review and meta-analysis. PLoS ONE 2016, 11, e0158765. [Google Scholar] [CrossRef]
- Pendse, S.; Singh, A.K. Complications of chronic kidney disease: Anemia, mineral metabolism, and cardiovascular disease. Med. Clin. N. Am. 2005, 89, 549–561. [Google Scholar] [CrossRef] [PubMed]
- Choi, B.; Crouzet, C.; Lau, W.L.; Cribbs, D.H.; Fisher, M.J. Cerebral blood flow in chronic kidney disease. J. Stroke Cerebrovasc. Dis. 2021, 30, 105702. [Google Scholar] [CrossRef]
- Koren, M.J.; Blumen, H.M.; Ayers, E.I.; Verghese, J.; Abramowitz, M.K. Cognitive dysfunction and gait abnormalities in CKD. Clin. J. Am. Soc. Nephrol. 2021, 16, 694–704. [Google Scholar] [CrossRef]
- Lee, Y.J.; Yoon, E.; Park, S.; Kim, Y.W.; Kim, S.E.; Ko, J.; Park, J.H.; Park, K.M.; Kim, I.H.; Park, B.S. Alteration of brain connectivity in neurologically asymptomatic patients with chronic kidney disease. Medicine 2021, 100, e25633. [Google Scholar] [CrossRef]
- Miller, L.M.; Rifkin, D.; Lee, A.K.; Tamura, M.K.; Pajewski, N.M.; Weiner, D.E.; Al-Rousan, T.; Shlipak, M.; Ix, J.H. Association of urine biomarkers of kidney tubule injury and dysfunction with frailty index and cognitive function in persons with CKD in SPRINT. Am. J. Kidney Dis. 2021, 78, 530–540. [Google Scholar] [CrossRef]
- Irazabal, M.V.; Torres, V.E. Reactive oxygen species and redox signaling in chronic kidney disease. Cells 2020, 9, 1342. [Google Scholar] [CrossRef]
- Che, R.; Yuan, Y.; Huang, S.; Zhang, A. Mitochondrial dysfunction in the pathophysiology of renal diseases. Am. J. Physiol. Renal. Physiol. 2014, 306, F367–F378. [Google Scholar] [CrossRef]
- Daenen, K.; Andries, A.; Mekahli, D.; Van Schepdael, A.; Jouret, F.; Bammens, B. Oxidative stress in chronic kidney disease. Pediatr. Nephrol. 2019, 34, 975–991. [Google Scholar] [CrossRef]
- Nezu, M.; Suzuki, N.; Yamamoto, M. Targeting the KEAP1-NRF2 system to prevent kidney disease progression. Am. J. Nephrol. 2017, 45, 473–483. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.; Wang, P.; Qiao, Y.; Jiang, C.; Ge, Y.; Flickinger, B.; Malhotra, D.K.; Dworkin, L.D.; Liu, Z.; Gong, R. GSK3β-mediated Keap1-independent regulation of Nrf2 antioxidant response: A molecular rheostat of acute kidney injury to chronic kidney disease transition. Redox Biol. 2019, 26, 101275. [Google Scholar] [CrossRef]
- Surh, Y.-J.; Kundu, J.K.; Na, H.-K. Nrf2 as a master redox switch in turning on the cellular signaling involved in the induction of cytoprotective genes by some chemopreventive phytochemicals. Planta Medica 2008, 74, 1526–1539. [Google Scholar] [CrossRef] [PubMed]
- Yamawaki, K.; Kanda, H.; Shimazaki, R. Nrf2 activator for the treatment of kidney diseases. Toxicol. Appl. Pharmacol. 2018, 360, 30–37. [Google Scholar] [CrossRef] [PubMed]
- Choi, B.-H.; Kang, K.-S.; Kwak, M.-K. Effect of redox modulating NRF2 activators on chronic kidney disease. Molecules 2014, 19, 12727–12759. [Google Scholar] [CrossRef]
- Lin, X.; Yang, F.; Huang, J.; Jiang, S.; Tang, Y.; Li, J. Ameliorate effect of pyrroloquinoline quinone against cyclophosphamide-induced nephrotoxicity by activating the Nrf2 pathway and inhibiting the NLRP3 pathway. Life Sci. 2020, 256, 117901. [Google Scholar] [CrossRef]
- Wang, Z.; Han, N.; Zhao, K.; Li, Y.; Chi, Y.; Wang, B. Protective effects of pyrroloquinoline quinone against oxidative stress-induced cellular senescence and inflammation in human renal tubular epithelial cells via Keap1/Nrf2 signaling pathway. Int. Immunopharmacol. 2019, 72, 445–453. [Google Scholar] [CrossRef]
- Friedman, J.E.; Dobrinskikh, E.; Alfonso-Garcia, A.; Fast, A.; Janssen, R.C.; Soderborg, T.K.; Anderson, A.L.; Reisz, J.A.; D’Alessandro, A.; Frank, D.N.; et al. Pyrroloquinoline quinone prevents developmental programming of microbial dysbiosis and macrophage polarization to attenuate liver fibrosis in offspring of obese mice. Hepatol. Commun. 2018, 2, 313–328. [Google Scholar] [CrossRef] [PubMed]
- Devasani, K.; Kaul, R.; Majumdar, A. Supplementation of pyrroloquinoline quinone with atorvastatin augments mitochondrial biogenesis and attenuates low grade inflammation in obese rats. Eur. J. Pharmacol. 2020, 881, 173273. [Google Scholar] [CrossRef]
- Raghuvanshi, R.; Chaudhari, A.; Kumar, G.N. Amelioration of cadmium- and mercury-induced liver and kidney damage in rats by genetically engineered probiotic Escherichia coli Nissle 1917 producing pyrroloquinoline quinone with oral supplementation of citric acid. Nutrition 2016, 32, 1285–1294. [Google Scholar] [CrossRef] [PubMed]
- Assimakopoulos, S.F.; Triantos, C.; Maroulis, I.; Gogos, C. The role of the gut barrier function in health and disease. Gastroenterol. Res. 2018, 11, 261–263. [Google Scholar] [CrossRef]
- Massier, L.; Blüher, M.; Kovacs, P.; Chakaroun, R.M. Impaired intestinal barrier and tissue bacteria: Pathomechanisms for metabolic diseases. Front. Endocrinol. 2021, 12, 616506. [Google Scholar] [CrossRef] [PubMed]
- Pat, Y.; Ogulur, I. The epithelial barrier hypothesis: A 20-year journey. Allergy 2021. [Google Scholar] [CrossRef] [PubMed]
- Aldars-García, L.; Chaparro, M.; Gisbert, J. Systematic review: The gut microbiome and its potential clinical application in inflammatory bowel disease. Microorganisms 2021, 9, 977. [Google Scholar] [CrossRef]
- Axelrad, J.E.; Cadwell, K.H.; Colombel, J.-F.; Shah, S.C. The role of gastrointestinal pathogens in inflammatory bowel disease: A systematic review. Ther. Adv. Gastroenterol. 2021, 14, 17562848211004493. [Google Scholar] [CrossRef] [PubMed]
- Tong, Y.; Gao, H.; Qi, Q.; Liu, X.; Li, J.; Gao, J.; Li, P.; Wang, Y.; Du, L.; Wang, C. High fat diet, gut microbiome and gastrointestinal cancer. Theranostics 2021, 11, 5889–5910. [Google Scholar] [CrossRef] [PubMed]
- Boddy, S.L.; Giovannelli, I.; Sassani, M.; Cooper-Knock, J.; Snyder, M.P.; Segal, E.; Elinav, E.; Barker, L.A.; Shaw, P.J.; McDermott, C.J. The gut microbiome: A key player in the complexity of amyotrophic lateral sclerosis (ALS). BMC Med. 2021, 19, 13. [Google Scholar] [CrossRef] [PubMed]
- Lorente-Picón, M.; Laguna, A. New avenues for Parkinson’s Disease therapeutics: Disease-modifying strategies based on the gut microbiota. Biomolecules 2021, 11, 433. [Google Scholar] [CrossRef]
- Niesler, B.; Kuerten, S.; Demir, I.E.; Schäfer, K.-H. Disorders of the enteric nervous system—A holistic view. Nat. Rev. Gastroenterol. Hepatol. 2021, 18, 393–410. [Google Scholar] [CrossRef]
- Ojeda, J.; Ávila, A.; Vidal, P. Gut Microbiota interaction with the central nervous system throughout life. J. Clin. Med. 2021, 10, 1299. [Google Scholar] [CrossRef]
- Snethlage, C.M.F.; Nieuwdorp, M.; van Raalte, D.H.; Rampanelli, E.; Verchere, B.C.; Hanssen, N.M. Auto-immunity and the gut microbiome in type 1 diabetes: Lessons from rodent and human studies. Best Pract. Res. Clin. Endocrinol. Metab. 2021, 35, 101544. [Google Scholar] [CrossRef]
- Takewaki, D.; Yamamura, T. Gut microbiome research in multiple sclerosis. Neurosci. Res. 2021, 168, 28–31. [Google Scholar] [CrossRef]
- Akdis, C.A. Does the epithelial barrier hypothesis explain the increase in allergy, autoimmunity and other chronic conditions? Nat. Rev. Immunol. 2021, 1–13. [Google Scholar] [CrossRef]
- Li, R.; Meng, X.; Chen, B.; Zhao, L.; Zhang, X. Gut Microbiota in Lupus: A Butterfly Effect? Curr. Rheumatol. Rep. 2021, 23, 27. [Google Scholar] [CrossRef]
- Murdaca, G.; Greco, M.; Borro, M.; Gangemi, S. Hygiene hypothesis and autoimmune diseases: A narrative review of clinical evidences and mechanisms. Autoimmun. Rev. 2021, 20, 102845. [Google Scholar] [CrossRef] [PubMed]
- Olejniczak-Staruch, I.; Ciążyńska, M.; Sobolewska-Sztychny, D.; Narbutt, J.; Skibińska, M.; Lesiak, A. Alterations of the skin and gut microbiome in psoriasis and psoriatic arthritis. Int. J. Mol. Sci. 2021, 22, 3998. [Google Scholar] [CrossRef] [PubMed]
- Arora, A.; Behl, T.; Sehgal, A.; Singh, S.; Sharma, N.; Bhatia, S.; Sobarzo-Sanchez, E.; Bungau, S. Unravelling the involvement of gut microbiota in type 2 diabetes mellitus. Life Sci. 2021, 273, 119311. [Google Scholar] [CrossRef] [PubMed]
- Duttaroy, A.K. Role of gut microbiota and their metabolites on atherosclerosis, hypertension and human blood platelet function: A review. Nutrients 2021, 13, 144. [Google Scholar] [CrossRef]
- He, L. Alterations of gut microbiota by overnutrition impact gluconeogenic gene expression and insulin signaling. Int. J. Mol. Sci. 2021, 22, 2121. [Google Scholar] [CrossRef]
- Liébana-García, R.; Olivares, M.; Bullich-Vilarrubias, C.; López-Almela, I.; Romaní-Pérez, M.; Sanz, Y. The gut microbiota as a versatile immunomodulator in obesity and associated metabolic disorders. Best Pract. Res. Clin. Endocrinol. Metab. 2021, 35, 101542. [Google Scholar] [CrossRef]
- Massey, W.; Brown, J.M. The gut microbial endocrine organ in type 2 diabetes. Endocrinology 2021, 162, 235. [Google Scholar] [CrossRef]
- Petraroli, M.; Castellone, E.; Patianna, V.; Esposito, S. Gut microbiota and obesity in adults and children: The state of the art. Front. Pediatr. 2021, 9, 657020. [Google Scholar] [CrossRef]
- Régnier, M.; Van Hul, M.; Knauf, C.; Cani, P.D. Gut microbiome, endocrine control of gut barrier function and metabolic diseases. J. Endocrinol. 2021, 248, R67–R82. [Google Scholar] [CrossRef]
- Rovella, V.; Rodia, G.; Di Daniele, F.; Cardillo, C.; Campia, U.; Noce, A.; Candi, E.; Della-Morte, D.; Tesauro, M. Association of gut hormones and microbiota with vascular dysfunction in obesity. Nutrients 2021, 13, 613. [Google Scholar] [CrossRef]
- Salamone, D.; Rivellese, A.A.; Vetrani, C. The relationship between gut microbiota, short-chain fatty acids and type 2 diabetes mellitus: The possible role of dietary fibre. Acta Diabetol. 2021, 58, 1131–1138. [Google Scholar] [CrossRef] [PubMed]
- Van Son, J.; Koekkoek, L.; La Fleur, S.; Serlie, M.; Nieuwdorp, M. The role of the gut microbiota in the gut–brain axis in obesity: Mechanisms and future implications. Int. J. Mol. Sci. 2021, 22, 2993. [Google Scholar] [CrossRef]
- Zhang, B.; Yang, W.; Zhang, H.; He, S.; Meng, Q.; Chen, Z.; Shan, A. Effect of pyrroloquinoline quinone disodium in female rats during gestating and lactating on reproductive performance and the intestinal barrier functions in the progeny. Br. J. Nutr. 2019, 121, 818–830. [Google Scholar] [CrossRef]
- Huang, C.; Ming, D.; Wang, W.; Wang, Z.; Hu, Y.; Ma, X.; Wang, F. Pyrroloquinoline quinone alleviates jejunal mucosal barrier function damage and regulates colonic microbiota in piglets challenged with enterotoxigenic Escherichia coli. Front. Microbiol. 2020, 11, 1754. [Google Scholar] [CrossRef]
- Gasaly, N.; Hermoso, M.; Gotteland, M. Butyrate and the fine-tuning of colonic homeostasis: Implication for inflammatory bowel diseases. Int. J. Mol. Sci. 2021, 22, 3061. [Google Scholar] [CrossRef]
- Huang, X.; Oshima, T.; Tomita, T.; Fukui, H.; Miwa, H. Butyrate alleviates cytokine-induced barrier dysfunction by modifying claudin-2 levels. Biology 2021, 10, 205. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.K.; Pandey, S.K.; Kumar, G.N. Pyrroloquinoline quinone-secreting probiotic Escherichia coli Nissle 1917 ameliorates ethanol-induced oxidative damage and hyperlipidemia in rats. Alcohol. Clin. Exp. Res. 2014, 38, 2127–2137. [Google Scholar] [CrossRef]
- Yamada, M.; Elias, M.D.; Matsushita, K.; Migita, C.T.; Adachi, O. Escherichia coli PQQ-containing quinoprotein glucose dehydrogenase: Its structure comparison with other quinoproteins. Biochim. Biophys. Acta (BBA)-Proteins Proteom. 2003, 1647, 185–192. [Google Scholar] [CrossRef]
- Canani, R.B.; Di Costanzo, M.; Leone, L.; Pedata, M.; Meli, R.; Calignano, A. Potential beneficial effects of butyrate in intestinal and extraintestinal diseases. World J. Gastroenterol. 2011, 17, 1519–28. [Google Scholar] [CrossRef]
- Gao, Z.; Yin, J.; Zhang, J.; Ward, R.E.; Martin, R.J.; Lefevre, M.; Cefalu, W.T.; Ye, J. Butyrate improves insulin sensitivity and increases energy expenditure in mice. Diabetes 2009, 58, 1509–1517. [Google Scholar] [CrossRef]
- Sun, J.; Wu, Q.; Sun, H.; Qiao, Y. Inhibition of histone deacetylase by butyrate protects rat liver from chemic reperfusion injury. Int. J. Mol. Sci. 2014, 15, 21069–21079. [Google Scholar] [CrossRef] [PubMed]
- Jonscher, K.R.; Stewart, M.S.; Alfonso-Garcia, A.; De Felice, B.C.; Wang, X.X.; Luo, Y.; Levi, M.; Heerwagen, M.J.R.; Janssen, R.C.; de la Houssaye, B.A.; et al. Early PQQ supplementation has persistent long-term protective effects on developmental programming of hepatic lipotoxicity and inflammation in obese mice. FASEB J. 2017, 31, 1434–1448. [Google Scholar] [CrossRef] [PubMed]
- Guirro, M.; Costa, A.; Gual-Grau, A.; Herrero, P.; Torrell, H.; Canela, N.; Arola, L. Effects from diet-induced gut microbiota dysbiosis and obesity can be ameliorated by fecal microbiota transplantation: A multiomics approach. PLoS ONE 2019, 14, e0218143. [Google Scholar] [CrossRef]
- Bauerly, K.; Harris, C.; Chowanadisai, W.; Graham, J.; Havel, P.J.; Tchaparian, E.; Satre, M.; Karliner, J.S.; Rucker, R.B. Altering pyrroloquinoline quinone nutritional status modulates mitochondrial, lipid, and energy metabolism in rats. PLoS ONE 2011, 6, e21779. [Google Scholar] [CrossRef]
- Tao, R.; Karliner, J.S.; Simonis, U.; Zheng, J.; Zhang, J.; Honbo, N.; Alano, C.C. Pyrroloquinoline quinone preserves mitochondrial function and prevents oxidative injury in adult rat cardiac myocytes. Biochem. Biophys. Res. Commun. 2007, 363, 257–262. [Google Scholar] [CrossRef][Green Version]
- Zhu, B.Q.; Simonis, U.; Cecchini, G.; Zhou, H.Z.; Li, L.; Teerlink, J.R.; Karliner, J.S. Comparison of pyrroloquinoline quinone and/or metoprolol on myocardial infarct size and mitochondrial damage in a rat model of ischemialreperfusion injury. J. Cardiovasc. Pharmacol. Ther. 2006, 11, 119–128. [Google Scholar] [CrossRef]
- Zhu, B.Q.; Zhou, H.Z.; Teerlink, J.R.; Karliner, J.S. Pyrroloquinoline quinone (PQQ) decreases myocardial infarct size and improves cardiac function in rat models of ischemia and ischemial reperfusion. Cardiovasc. Drugs Ther. 2004, 18, 421–431. [Google Scholar] [CrossRef]
- Liu, L.; Zhang, Y.; Liu, T.; Ke, C.; Huang, J.; Fu, Y.; Lin, Z.; Chen, F.; Wu, X.; Chen, Q. Pyrroloquinoline quinone protects against exercise-induced fatigue and oxidative damage via improving mitochondrial function in mice. FASEB J. 2021, 35, e21394. [Google Scholar] [CrossRef]
- Chrétien, D.; Bénit, P.; Ha, H.H.; Keipert, S.; El-Khoury, R.; Chang, Y.T.; Jastroch, M.; Jacobs, H.T.; Rustin, P.; Rak, M. Mitochondria are physiologically maintained at close to 50 degrees C. PLoS Biol. 2018, 16, e2003992. [Google Scholar] [CrossRef] [PubMed]
- Wilkening, A.; Rüb, C.; Sylvester, M.; Voos, W. Analysis of heat-induced protein aggregation in human mitochondria. J. Biol. Chem. 2018, 293, 11537–11552. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Zhou, J.; Shen, M.; Xu, H.; Yu, S.; Cheng, Q.; Ding, F. Pyrroloquinoline quinone inhibits rotenone-induced microglia inflammation by enhancing autophagy. Molecules 2020, 25, 4359. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.I.; Chen, S.; Yu, S.; Qin, J.; Zhang, J.; Cheng, Q.; Ke, K.; Ding, F. Neuroprotective effects of pyrroloquinoline quinone against rotenone injury in primary cultured midbrain neurons and in a rat model of Parkinson’s disease. Neuropharmacology 2016, 108, 238–251. [Google Scholar] [CrossRef] [PubMed]
- Qin, J.; Wu, M.; Yu, S.; Gao, X.; Zhang, J.; Dong, X.; Ji, J.; Zhang, Y.; Zhou, L.; Zhang, Q.; et al. Pyrroloquinoline quinone-conferred neuroprotection in rotenone models of Parkinson’s disease. Toxicol. Lett. 2015, 238, 70–82. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Chen, S.; Shen, M.; He, Q.; Zhang, Y.; Shi, Y.; Ding, F.; Zhang, Q. Mitochondrial regulation by pyrroloquinoline quinone prevents rotenone-induced neurotoxicity in Parkinson’s disease models. Neurosci. Lett. 2018, 687, 104–110. [Google Scholar] [CrossRef] [PubMed]
- Chowanadisai, W.; Bauerly, K.A.; Tchaparian, E.; Wong, A.; Cortopassi, G.A.; Rucker, R.B. Pyrroloquinoline quinone stimulates mitochondrial biogenesis through cAMP response element-binding protein phosphorylation and increased PGC-1α expression. J. Biol. Chem. 2010, 285, 142–152. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Q.; Chen, J.; Guo, H.; Lu, J.-L.; Zhou, J.; Guo, X.-Y.; Shi, Y.; Zhang, Y.; Yu, S.; Zhang, Q.; et al. Pyrroloquinoline quinone promotes mitochondrial biogenesis in rotenone-induced Parkinson’s disease model via AMPK activation. Acta Pharmacol. Sin. 2021, 42, 665–678. [Google Scholar] [CrossRef]
- Betarbet, R.; Sherer, T.B.; MacKenzie, G.; Garcia-Osuna, M.; Panov, A.V.; Greenamyre, J.T. Chronic systemic pesticide exposure reproduces features of Parkinson’s disease. Nat. Neurosci. 2000, 3, 1301–1306. [Google Scholar] [CrossRef]
- Kim, J.; Harada, R.; Kobayashi, M.; Kobayashi, N.; Sode, K. The inhibitory effect of pyrroloquinoline quinone on the amyloid formation and cytotoxicity of truncated alpha-synuclein. Mol. Neurodegener. 2010, 5, 20. [Google Scholar] [CrossRef]
- Jensen, F.E.; Gardner, G.J.; Williams, A.P.; Gallop, P.M.; Aizenman, E.; Rosenberg, P.A. The putative essential nutrient pyrroloquinoline quinone is neuroprotective in a rodent model of hypoxiclischemic brain injury. Neuroscience 1994, 62, 399–406. [Google Scholar] [CrossRef]
- Zhang, L.; Liu, J.; Cheng, C.; Yuan, Y.; Yu, B.; Shen, A.; Yan, M. The neuroprotective effect of pyrroloquinoline quinone on traumatic brain injury. J. Neurotrauma 2012, 29, 851–864. [Google Scholar] [CrossRef]
- Zhang, Y.; Feustel, P.; Kimelberg, H.K. Neuroprotection by pyrroloquinoline quinone (PQQ) in reversible middle cerebral artery occlusion in the adult rat. Brain Res. 2006, 1094, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Ralto, K.M.; Rhee, E.P.; Parikh, S.M. NAD+ homeostasis in renal health and disease. Nat. Rev. Nephrol. 2020, 16, 99–111. [Google Scholar] [CrossRef] [PubMed]


| Attribute | Description |
|---|---|
| PQQ is required for essential physiological functions | PQQ is one of only a few nutritionally important biofactors for which a nutritional deficiency can be defined in multiple species of eukaryotes [9,60,61,66,83]. PQQ has been tentatively identified as a component of interstellar dust [83]. Thus, PQQ may have been present throughout early biological conception and evolution. PQQ is also a plant growth factor [43,44,45,46,47,48,49,50,51], i.e., for animals and humans, there has been constant exposure to PQQ [83]. |
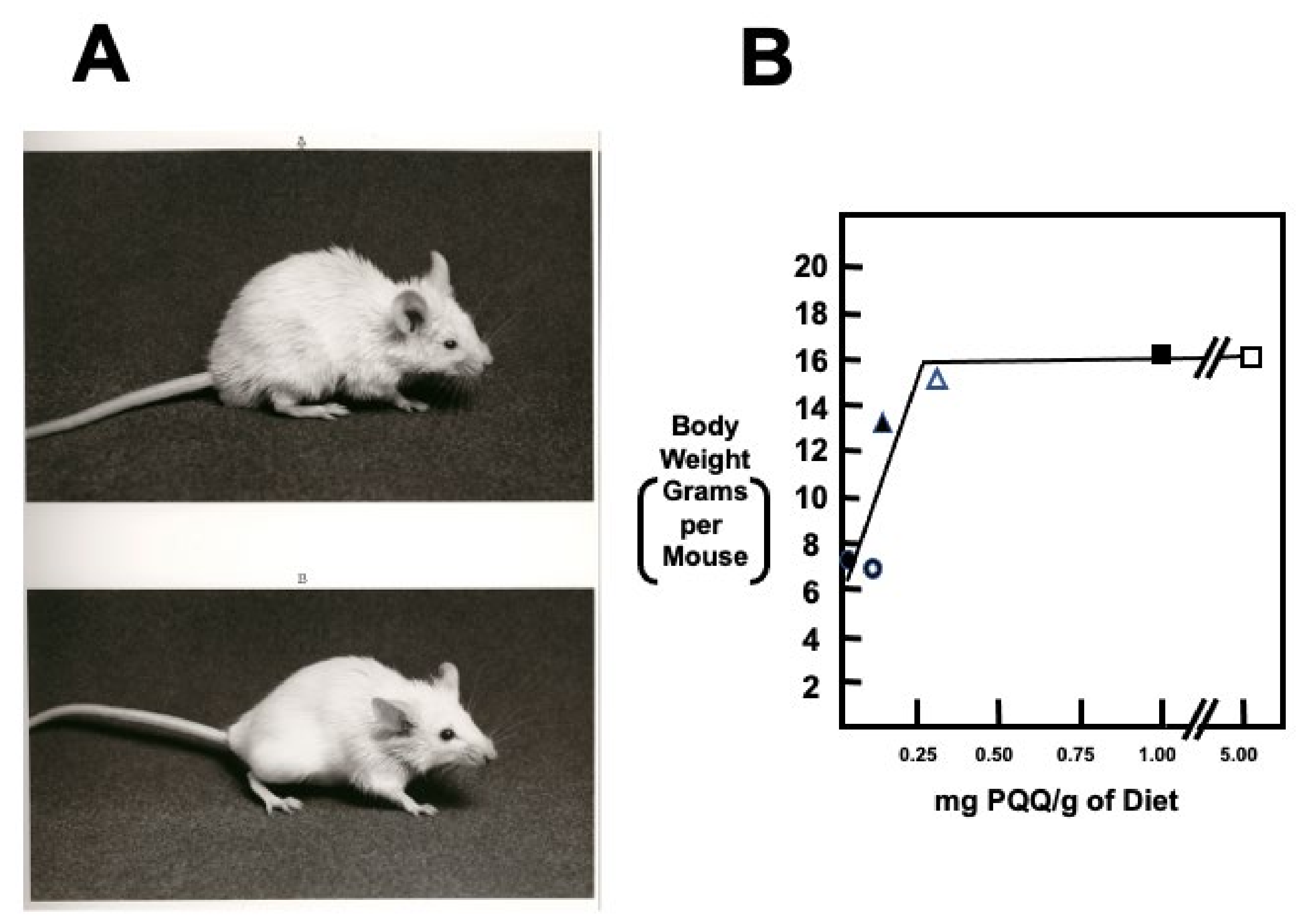
| The apparent need for PQQ is nutritionally attainable without the need for supplementation | PQQ effects are elicited in the nM to μM range of exposure, in contrast to the mM concentrations needed for other biofactors [9,60,61]. PQQ and its derivatives, such as imidazopyrroloquinoline (IPQ) compounds, are found in the milk of mammals in concentrations like vitamins, such as folate, riboflavin and, biotin [12,75]. In rodents, PQQ appears essential for reproduction [60,61,136]. |
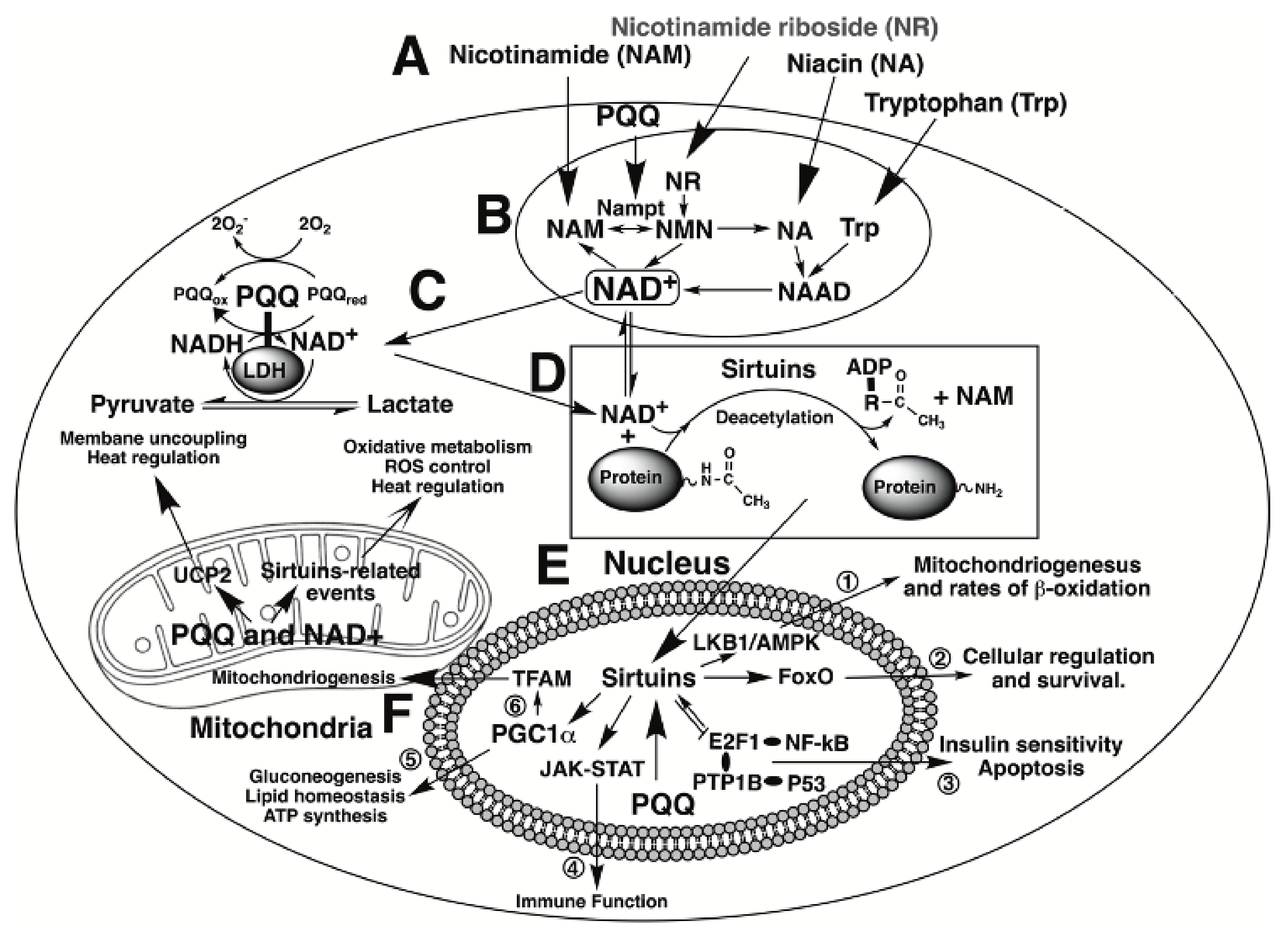
| PQQ aids in sustaining cellular NAD+ levels and mitochondriogenesis | PQQ acts as an accessory factor for lactate and potentially other dehydrogenases in NADH oxidation to NAD+ [18,19]. Accordingly, the effects of PQQ exposure mimic those of cellular NAD+ (e.g., sustaining mitochondriogenesis [11,165]. |
| PQQ stimulates sirtuin activity | Sirtuins modulate the activity of factors involved in DNA repair and mitochondriogenesis [23]. PQQ stimulates the expression of proteins in the sirtuin family of enzymes and, by sustaining NAD+ levels, optimizes NAD+, a sirtuin cofactor and co-substrate [11,165]. |
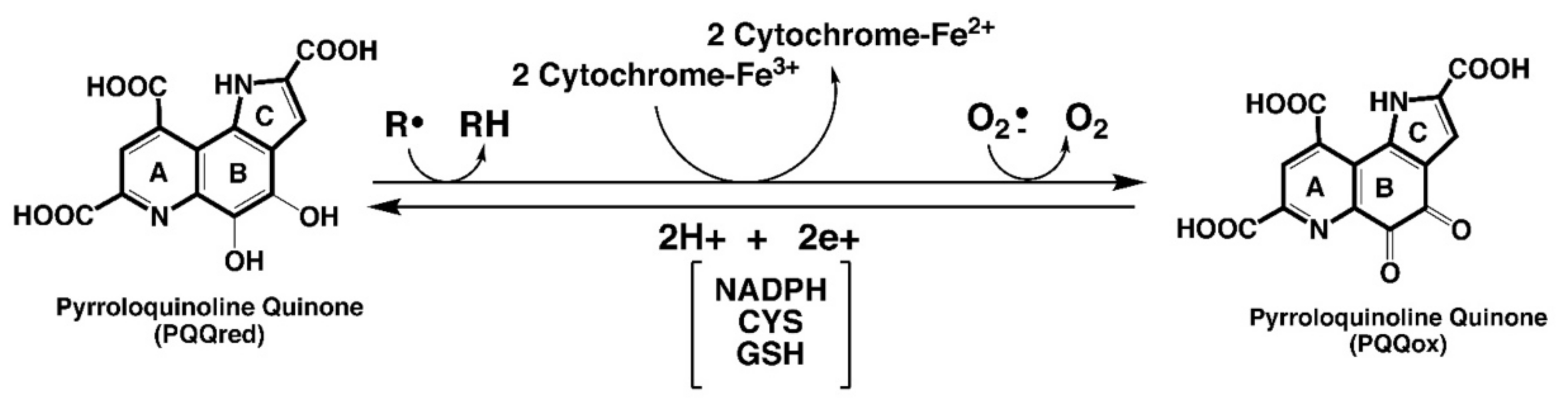
| Relationships involving PQQ are important to the aging process, immunity, and ROS defense | PQQ exposure increases longevity in models used in the study of aging [53], improves immune responsiveness to cytokines [9,70,145], and acts as a potent redox cycling agent that helps maximize ROS defense [9,12,13,16,107,145]. |
| PQQ is associated with the attenuation of clinically relevant dysfunctions such as ischemia, neurogenic losses, inflammation, and lipotoxicity. | PQQ is neuroprotective and appears to improve memory [74,155,162,163,164]. PQQ protects NMDA mediated receptors in neurological injury. PQQ can reverse hepatic steatosis and has potential as a therapeutic for nonalcoholic fatty liver disease (NAFLD) or nonalcoholic steatohepatitis (NASH) [107,145]. PQQ also influences serum lipid metabolism in ways relevant to protection from heart disease [147]. Moreover, in models of ischemia and reperfusion injury, the cardioprotective effectiveness of PQQ is equivalent to β1-selective adrenoceptor antagonists, such as metoprolol [149]. PQQ alleviates jejunal mucosal inflammatory injury in animal models by inhibiting events associated with NF-κB-related pathways and improving the imbalance of colonic microbiota caused by various agents [9,107,145]. As noted by Naveed et al. [49], inflammation of the gastrointestinal tract has a strong association with ROS genesis. PQQ, as a ROS scavenger, acts as a protectant. PQQ also acts to reduce C-reactive protein and IL-6 levels [61]. |
| PQQ is safe. | The no observed effect level (NOAEL) for PQQ has been determined to be 400 mg/kg bw/day in a sub chronic toxicity study in rats. By applying a safety margin of 100, it can be concluded that doses up to 4 mg/kg BW/day or 240 mg/person/day would be safe in adult humans weighing 60 kg [87,88,89,90]. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jonscher, K.R.; Chowanadisai, W.; Rucker, R.B. Pyrroloquinoline-Quinone Is More Than an Antioxidant: A Vitamin-like Accessory Factor Important in Health and Disease Prevention. Biomolecules 2021, 11, 1441. https://doi.org/10.3390/biom11101441
Jonscher KR, Chowanadisai W, Rucker RB. Pyrroloquinoline-Quinone Is More Than an Antioxidant: A Vitamin-like Accessory Factor Important in Health and Disease Prevention. Biomolecules. 2021; 11(10):1441. https://doi.org/10.3390/biom11101441
Chicago/Turabian StyleJonscher, Karen R., Winyoo Chowanadisai, and Robert B. Rucker. 2021. "Pyrroloquinoline-Quinone Is More Than an Antioxidant: A Vitamin-like Accessory Factor Important in Health and Disease Prevention" Biomolecules 11, no. 10: 1441. https://doi.org/10.3390/biom11101441
APA StyleJonscher, K. R., Chowanadisai, W., & Rucker, R. B. (2021). Pyrroloquinoline-Quinone Is More Than an Antioxidant: A Vitamin-like Accessory Factor Important in Health and Disease Prevention. Biomolecules, 11(10), 1441. https://doi.org/10.3390/biom11101441






