Gut Microbiota as the Link between Elevated BCAA Serum Levels and Insulin Resistance
Abstract
:1. Introduction
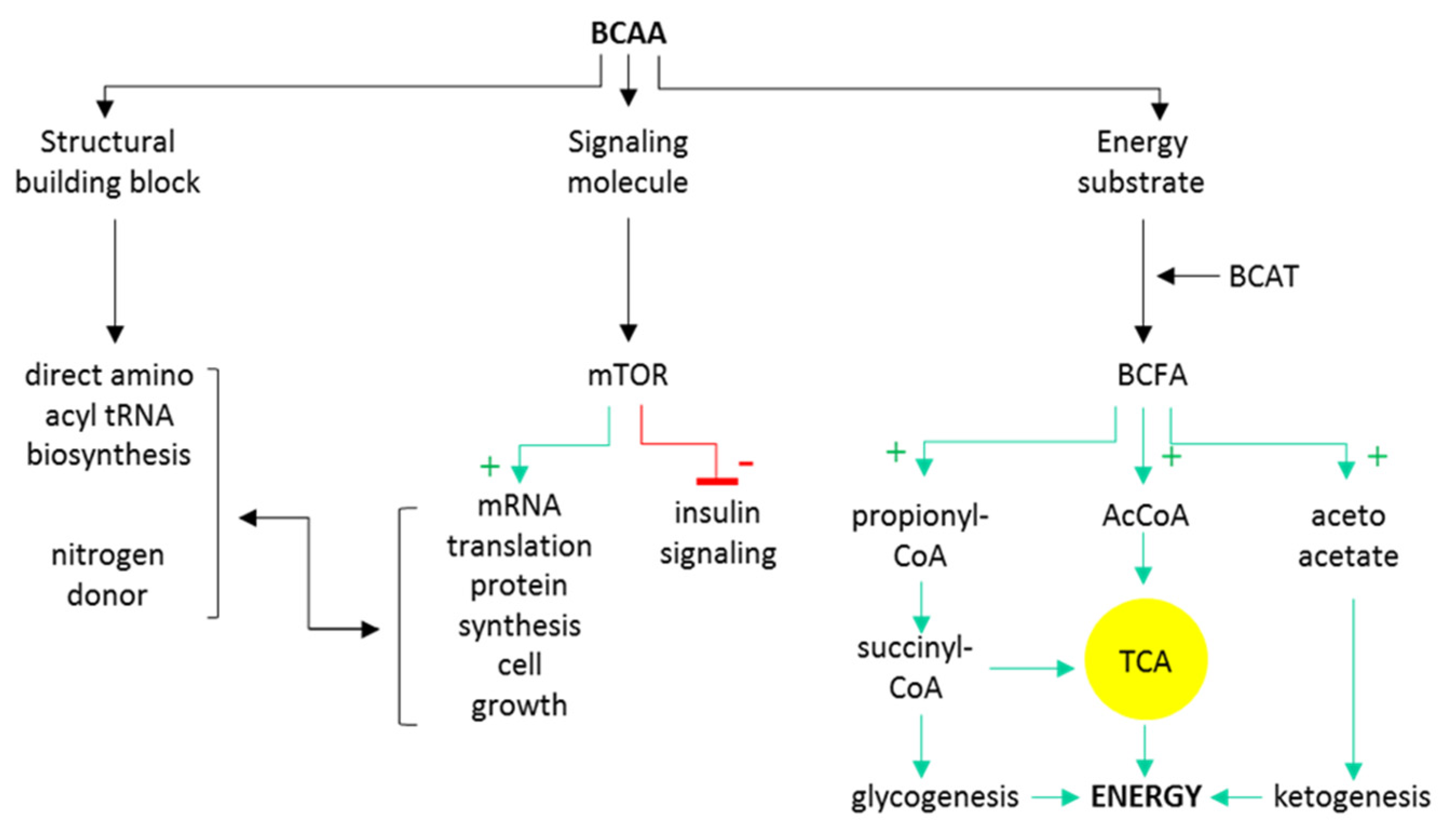
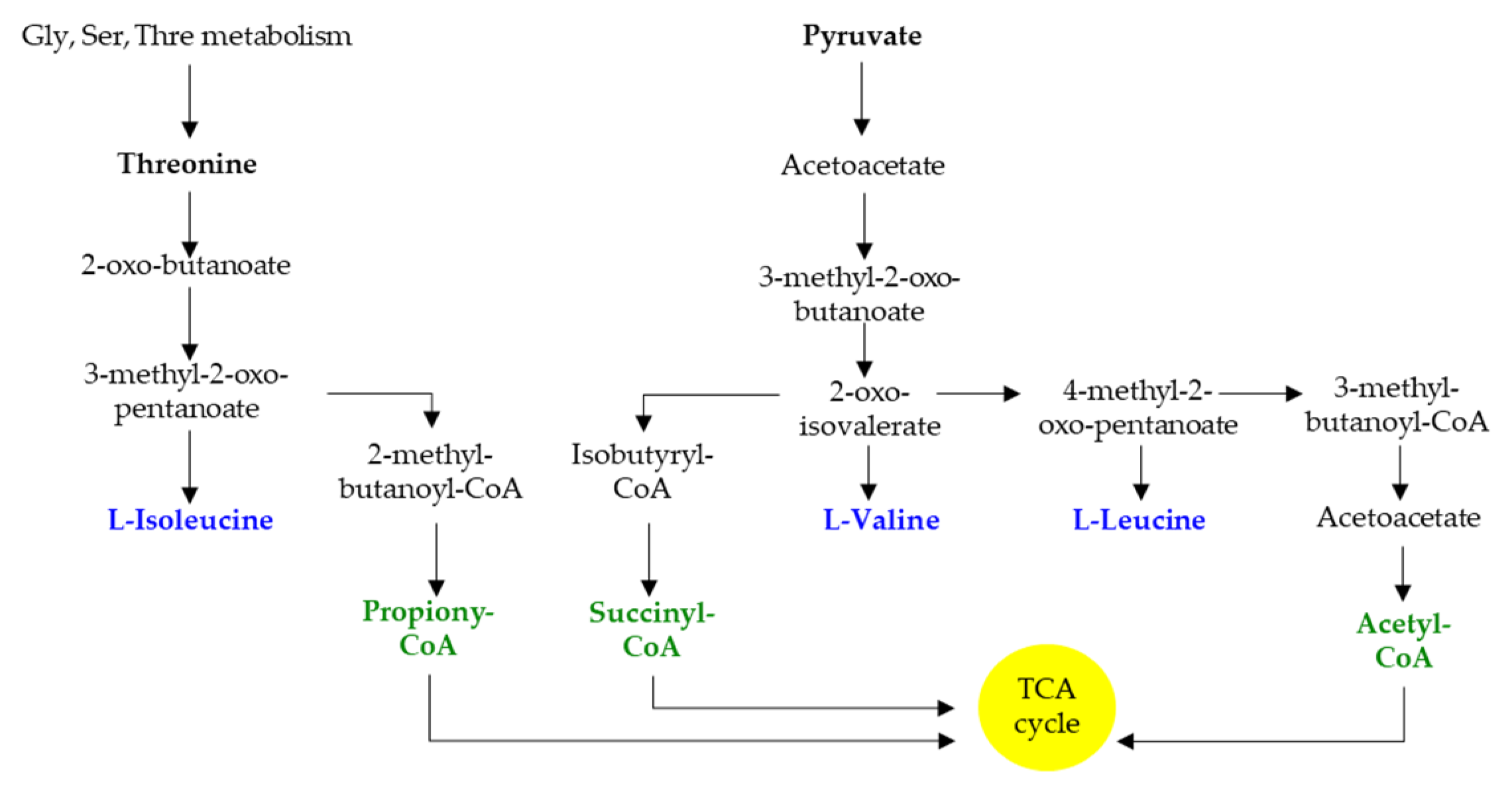
2. Branched-Chain Amino Acids Are Both Nutrients and Signaling Molecules
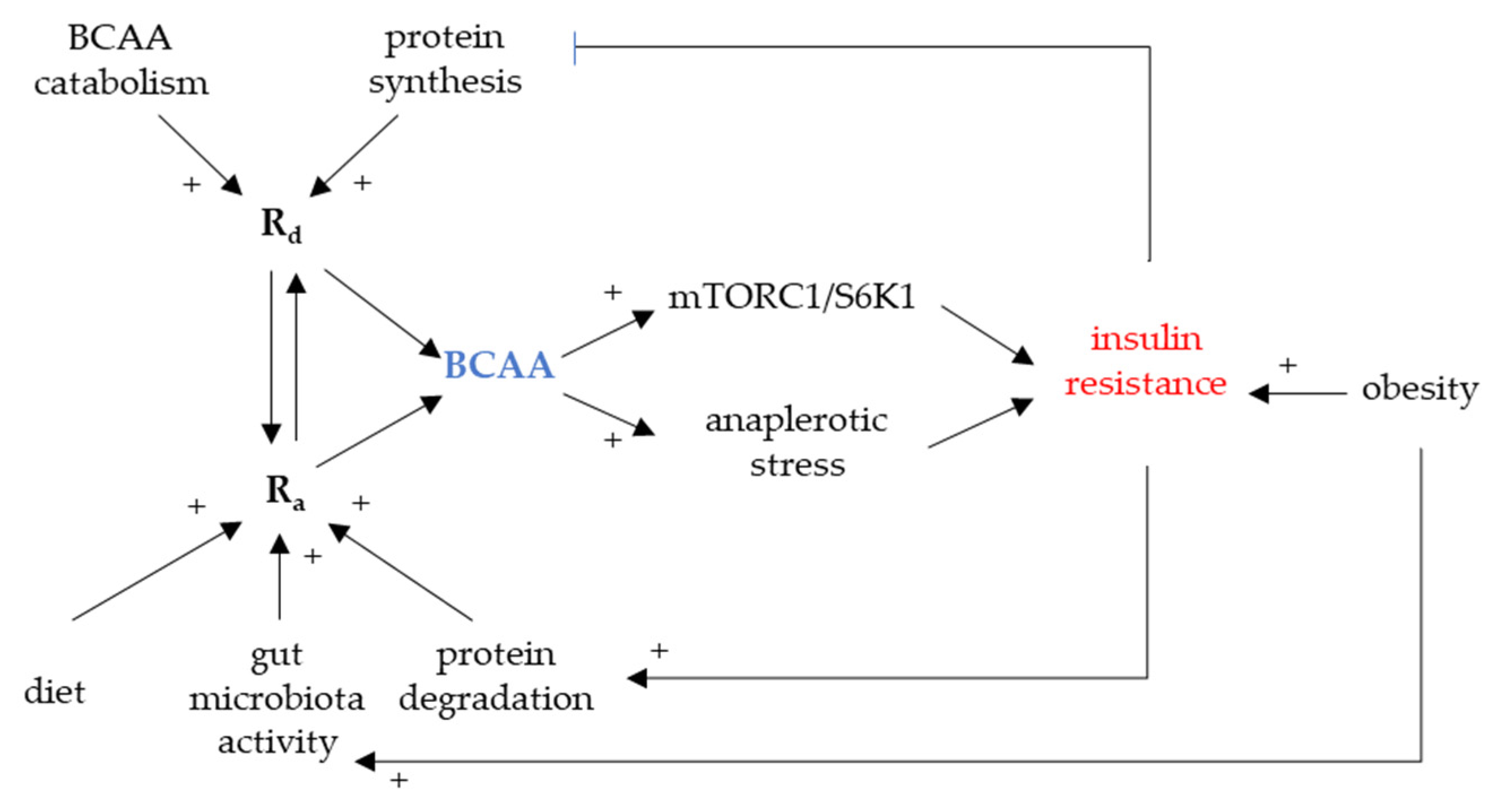
3. Circulating BCAAs Associated with Insulin Resistance
4. Gut Microbiota as the Source of Amino Acids for the Host
5. Findings Derived from Observational Studies
5.1. Human Studies
5.2. Animal Studies
6. Mechanistic Studies
6.1. The Effect of Bacterial Taxa Manipulation on the BCAA Metabolism
6.2. The Effect of Microbiota Modulation with Phytochemicals or Symbiotics on the BCAA Metabolism
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Noce, A.; Marrone, G.; Di Daniele, F.; Ottaviani, E.; Wilson Jones, G.; Bernini, R.; Romani, A.; Rovella, V. Impact of Gut Microbiota Composition on Onset and Progression of Chronic Non-Communicable Diseases. Nutrients 2019, 11, 1073. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cabello-Olmo, M.; Arana, M.; Urtasun, R.; Encio, I.J.; Barajas, M. Role of Postbiotics in Diabetes Mellitus: Current Knowledge and Future Perspectives. Foods 2021, 10, 1590. [Google Scholar] [CrossRef] [PubMed]
- Guilloteau, P.; Martin, L.; Eeckhaut, V.; Ducatelle, R.; Zabielski, R.; Van Immerseel, F. From the gut to the peripheral tissues: The multiple effects of butyrate. Nutr. Res. Rev. 2010, 23, 366–384. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cresci, G.A.; Glueck, B.; McMullen, M.R.; Xin, W.; Allende, D.; Nagy, L.E. Prophylactic tributyrin treatment mitigates chronic-binge ethanol-induced intestinal barrier and liver injury. J. Gastroenterol. Hepatol. 2017, 32, 1587–1597. [Google Scholar] [CrossRef] [PubMed]
- Arpaia, N.; Campbell, C.; Fan, X.; Dikiy, S.; van der Veeken, J.; de Roos, P.; Liu, H.; Cross, J.R.; Pfeffer, K.; Coffer, P.J.; et al. Metabolites produced by commensal bacteria promote peripheral regulatory T-cell generation. Nature 2013, 504, 451–455. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Mao, Z.; Ye, X.; Zuo, T. Human Gut Microbiome and Liver Diseases: From Correlation to Causation. Microorganisms 2021, 9, 1017. [Google Scholar] [CrossRef]
- Schattenberg, J.M.; Galle, P.R. Animal models of non-alcoholic steatohepatitis: Of mice and man. Dig. Dis. 2010, 28, 247–254. [Google Scholar] [CrossRef]
- Rao, R.K. Acetaldehyde-induced barrier disruption and paracellular permeability in Caco-2 cell monolayer. Methods Mol. Biol. 2008, 447, 171–183. [Google Scholar] [CrossRef] [PubMed]
- Newgard, C.B.; An, J.; Bain, J.R.; Muehlbauer, M.J.; Stevens, R.D.; Lien, L.F.; Haqq, A.M.; Shah, S.H.; Arlotto, M.; Slentz, C.A.; et al. A branched-chain amino acid-related metabolic signature that differentiates obese and lean humans and contributes to insulin resistance. Cell Metab. 2009, 9, 311–326. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Katagiri, R.; Goto, A.; Nakagawa, T.; Nishiumi, S.; Kobayashi, T.; Hidaka, A.; Budhathoki, S.; Yamaji, T.; Sawada, N.; Shimazu, T.; et al. Increased Levels of Branched-Chain Amino Acid Associated With Increased Risk of Pancreatic Cancer in a Prospective Case-Control Study of a Large Cohort. Gastroenterology 2018, 155, 1474–1482.e1. [Google Scholar] [CrossRef]
- Tobias, D.K.; Hazra, A.; Lawler, P.R.; Chandler, P.D.; Chasman, D.I.; Buring, J.E.; Lee, I.M.; Cheng, S.; Manson, J.E.; Mora, S. Circulating branched-chain amino acids and long-term risk of obesity-related cancers in women. Sci. Rep. 2020, 10, 16534. [Google Scholar] [CrossRef] [PubMed]
- Gojda, J.; Strakova, R.; Plihalova, A.; Tuma, P.; Potockova, J.; Polak, J.; Andel, M. Increased Incretin But Not Insulin Response after Oral versus Intravenous Branched Chain Amino Acids. Ann. Nutr. Metab. 2017, 70, 293–302. [Google Scholar] [CrossRef] [PubMed]
- Wahren, J.; Felig, P.; Hagenfeldt, L. Effect of protein ingestion on splanchnic and leg metabolism in normal man and in patients with diabetes mellitus. J. Clin. Investig. 1976, 57, 987–999. [Google Scholar] [CrossRef]
- Vellai, T. How the amino acid leucine activates the key cell-growth regulator mTOR. Nature 2021, 596, 192–194. [Google Scholar] [CrossRef] [PubMed]
- Pedroso, J.A.; Zampieri, T.T.; Donato, J., Jr. Reviewing the Effects of L-Leucine Supplementation in the Regulation of Food Intake, Energy Balance, and Glucose Homeostasis. Nutrients 2015, 7, 3914–3937. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Holecek, M. Branched-chain amino acids in health and disease: Metabolism, alterations in blood plasma, and as supplements. Nutr. Metab. 2018, 15, 33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cole, J.T. Metabolism of BCAAs. In Branched Chain Amino Acids in Clinical Nutrition; Rajendram, R., Preedy, V.R., Patel, V.B., Eds.; Humana Press: Totowa, NJ, USA, 2015; Volume 1, pp. 13–24. [Google Scholar]
- Tremblay, F.; Krebs, M.; Dombrowski, L.; Brehm, A.; Bernroider, E.; Roth, E.; Nowotny, P.; Waldhausl, W.; Marette, A.; Roden, M. Overactivation of S6 kinase 1 as a cause of human insulin resistance during increased amino acid availability. Diabetes 2005, 54, 2674–2684. [Google Scholar] [CrossRef] [Green Version]
- Nair, K.S.; Woolf, P.D.; Welle, S.L.; Matthews, D.E. Leucine, glucose, and energy metabolism after 3 days of fasting in healthy human subjects. Am. J. Clin. Nutr. 1987, 46, 557–562. [Google Scholar] [CrossRef] [PubMed]
- Kamaura, M.; Nishijima, K.; Takahashi, M.; Ando, T.; Mizushima, S.; Tochikubo, O. Lifestyle modification in metabolic syndrome and associated changes in plasma amino acid profiles. Circ. J. 2010, 74, 2434–2440. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wurtz, P.; Makinen, V.P.; Soininen, P.; Kangas, A.J.; Tukiainen, T.; Kettunen, J.; Savolainen, M.J.; Tammelin, T.; Viikari, J.S.; Ronnemaa, T.; et al. Metabolic signatures of insulin resistance in 7098 young adults. Diabetes 2012, 61, 1372–1380. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, T.J.; Larson, M.G.; Vasan, R.S.; Cheng, S.; Rhee, E.P.; McCabe, E.; Lewis, G.D.; Fox, C.S.; Jacques, P.F.; Fernandez, C.; et al. Metabolite profiles and the risk of developing diabetes. Nat. Med. 2011, 17, 448–453. [Google Scholar] [CrossRef]
- Lips, M.A.; Van Klinken, J.B.; van Harmelen, V.; Dharuri, H.K.; AC’t Hoen, P.A.; Laros, J.F.; van Ommen, G.J.; Janssen, I.M.; Van Ramshorst, B.; Van Wagensveld, B.A.; et al. Roux-en-Y gastric bypass surgery, but not calorie restriction, reduces plasma branched-chain amino acids in obese women independent of weight loss or the presence of type 2 diabetes. Diabetes Care 2014, 37, 3150–3156. [Google Scholar] [CrossRef] [Green Version]
- Yin, Q.; Brameld, J.M.; Parr, T.; Murton, A.J. Leucine and mTORc1 act independently to regulate 2-deoxyglucose uptake in L6 myotubes. Amino Acids 2020, 52, 477–486. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lynch, C.J.; Adams, S.H. Branched-chain amino acids in metabolic signalling and insulin resistance. Nat. Rev. Endocrinol. 2014, 10, 723–736. [Google Scholar] [CrossRef] [Green Version]
- Biswas, D.; Duffley, L.; Pulinilkunnil, T. Role of branched-chain amino acid-catabolizing enzymes in intertissue signaling, metabolic remodeling, and energy homeostasis. FASEB J. 2019, 33, 8711–8731. [Google Scholar] [CrossRef] [PubMed]
- Newgard, C.B. Interplay between lipids and branched-chain amino acids in development of insulin resistance. Cell Metab. 2012, 15, 606–614. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, T.; Yao, W.; He, Q.; Shao, Y.; Zheng, R.; Huang, F. L-leucine stimulates glutamate dehydrogenase activity and glutamate synthesis by regulating mTORC1/SIRT4 pathway in pig liver. Anim. Nutr. 2018, 4, 329–337. [Google Scholar] [CrossRef] [PubMed]
- Binder, E.; Bermudez-Silva, F.J.; Andre, C.; Elie, M.; Romero-Zerbo, S.Y.; Leste-Lasserre, T.; Belluomo, I.; Duchampt, A.; Clark, S.; Aubert, A.; et al. Leucine supplementation protects from insulin resistance by regulating adiposity levels. PLoS ONE 2013, 8, e74705. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guo, K.; Yu, Y.H.; Hou, J.; Zhang, Y. Chronic leucine supplementation improves glycemic control in etiologically distinct mouse models of obesity and diabetes mellitus. Nutr. Metab. 2010, 7, 57. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hey, P.; Gow, P.; Testro, A.G.; Apostolov, R.; Chapman, B.; Sinclair, M. Nutraceuticals for the treatment of sarcopenia in chronic liver disease. Clin. Nutr. ESPEN 2021, 41, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Leenders, M.; van Loon, L.J. Leucine as a pharmaconutrient to prevent and treat sarcopenia and type 2 diabetes. Nutr. Rev. 2011, 69, 675–689. [Google Scholar] [CrossRef]
- Maykish, A.; Sikalidis, A.K. Utilization of Hydroxyl-Methyl Butyrate, Leucine, Glutamine and Arginine Supplementation in Nutritional Management of Sarcopenia-Implications and Clinical Considerations for Type 2 Diabetes Mellitus Risk Modulation. J. Pers. Med. 2020, 10, 19. [Google Scholar] [CrossRef] [Green Version]
- Sikalidis, A.K.; Maykish, A. The Gut Microbiome and Type 2 Diabetes Mellitus: Discussing a Complex Relationship. Biomedicines 2020, 8, 8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Soares, J.D.P.; Howell, S.L.; Teixeira, F.J.; Pimentel, G.D. Dietary Amino Acids and Immunonutrition Supplementation in Cancer-Induced Skeletal Muscle Mass Depletion: A Mini-Review. Curr. Pharm. Des. 2020, 26, 970–978. [Google Scholar] [CrossRef] [PubMed]
- Boulet, M.M.; Chevrier, G.; Grenier-Larouche, T.; Pelletier, M.; Nadeau, M.; Scarpa, J.; Prehn, C.; Marette, A.; Adamski, J.; Tchernof, A. Alterations of plasma metabolite profiles related to adipose tissue distribution and cardiometabolic risk. Am. J. Physiol. Endocrinol. Metab. 2015, 309, E736–E746. [Google Scholar] [CrossRef] [Green Version]
- Lu, J.; Xie, G.; Jia, W.; Jia, W. Insulin resistance and the metabolism of branched-chain amino acids. Front. Med. 2013, 7, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Green, C.R.; Wallace, M.; Divakaruni, A.S.; Phillips, S.A.; Murphy, A.N.; Ciaraldi, T.P.; Metallo, C.M. Branched-chain amino acid catabolism fuels adipocyte differentiation and lipogenesis. Nat. Chem. Biol. 2016, 12, 15–21. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Crown, S.B.; Marze, N.; Antoniewicz, M.R. Catabolism of Branched Chain Amino Acids Contributes Significantly to Synthesis of Odd-Chain and Even-Chain Fatty Acids in 3T3-L1 Adipocytes. PLoS ONE 2015, 10, e0145850. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Herman, M.A.; She, P.; Peroni, O.D.; Lynch, C.J.; Kahn, B.B. Adipose tissue branched chain amino acid (BCAA) metabolism modulates circulating BCAA levels. J. Biol. Chem. 2010, 285, 11348–11356. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matthews, D.E. Observations of branched-chain amino acid administration in humans. J. Nutr. 2005, 135, 1580S–1584S. [Google Scholar] [CrossRef] [Green Version]
- Gojda, J.; Waldauf, P.; Hruskova, N.; Blahutova, B.; Krajcova, A.; Urban, T.; Tuma, P.; Rasova, K.; Duska, F. Lactate production without hypoxia in skeletal muscle during electrical cycling: Crossover study of femoral venous-arterial differences in healthy volunteers. PLoS ONE 2019, 14, e0200228. [Google Scholar] [CrossRef] [Green Version]
- Everman, S.; Meyer, C.; Tran, L.; Hoffman, N.; Carroll, C.C.; Dedmon, W.L.; Katsanos, C.S. Insulin does not stimulate muscle protein synthesis during increased plasma branched-chain amino acids alone but still decreases whole body proteolysis in humans. Am. J. Physiol. Endocrinol. Metab. 2016, 311, E671–E677. [Google Scholar] [CrossRef] [Green Version]
- Calbet, J.A.; MacLean, D.A. Plasma glucagon and insulin responses depend on the rate of appearance of amino acids after ingestion of different protein solutions in humans. J. Nutr. 2002, 132, 2174–2182. [Google Scholar] [CrossRef] [Green Version]
- Nilsson, M.; Stenberg, M.; Frid, A.H.; Holst, J.J.; Bjorck, I.M. Glycemia and insulinemia in healthy subjects after lactose-equivalent meals of milk and other food proteins: The role of plasma amino acids and incretins. Am. J. Clin. Nutr. 2004, 80, 1246–1253. [Google Scholar] [CrossRef]
- Pedersen, H.K.; Gudmundsdottir, V.; Nielsen, H.B.; Hyotylainen, T.; Nielsen, T.; Jensen, B.A.; Forslund, K.; Hildebrand, F.; Prifti, E.; Falony, G.; et al. Human gut microbes impact host serum metabolome and insulin sensitivity. Nature 2016, 535, 376–381. [Google Scholar] [CrossRef]
- Liu, R.; Hong, J.; Xu, X.; Feng, Q.; Zhang, D.; Gu, Y.; Shi, J.; Zhao, S.; Liu, W.; Wang, X.; et al. Gut microbiome and serum metabolome alterations in obesity and after weight-loss intervention. Nat. Med. 2017, 23, 859–868. [Google Scholar] [CrossRef] [PubMed]
- Zeng, S.L.; Li, S.Z.; Xiao, P.T.; Cai, Y.Y.; Chu, C.; Chen, B.Z.; Li, P.; Li, J.; Liu, E.H. Citrus polymethoxyflavones attenuate metabolic syndrome by regulating gut microbiome and amino acid metabolism. Sci. Adv. 2020, 6, eaax6208. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blachier, F.; Mariotti, F.; Huneau, J.F.; Tome, D. Effects of amino acid-derived luminal metabolites on the colonic epithelium and physiopathological consequences. Amino Acids 2007, 33, 547–562. [Google Scholar] [CrossRef]
- Davila, A.M.; Blachier, F.; Gotteland, M.; Andriamihaja, M.; Benetti, P.H.; Sanz, Y.; Tome, D. Intestinal luminal nitrogen metabolism: Role of the gut microbiota and consequences for the host. Pharmacol. Res. 2013, 68, 95–107. [Google Scholar] [CrossRef]
- Reitzer, L. Biosynthesis of Glutamate, Aspartate, Asparagine, L-Alanine, and D-Alanine. EcoSal Plus 2004, 1. [Google Scholar] [CrossRef] [PubMed]
- Torrallardona, D.; Harris, C.I.; Coates, M.E.; Fuller, M.F. Microbial amino acid synthesis and utilization in rats: Incorporation of 15N from 15NH4Cl into lysine in the tissues of germ-free and conventional rats. Br. J. Nutr. 1996, 76, 689–700. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Metges, C.C.; El-Khoury, A.E.; Henneman, L.; Petzke, K.J.; Grant, I.; Bedri, S.; Pereira, P.P.; Ajami, A.M.; Fuller, M.F.; Young, V.R. Availability of intestinal microbial lysine for whole body lysine homeostasis in human subjects. Am. J. Physiol. 1999, 277, E597–E607. [Google Scholar] [CrossRef] [PubMed]
- Metges, C.C.; Petzke, K.J.; Hennig, U. Gas chromatography/combustion/isotope ratio mass spectrometric comparison of N-acetyl- and N-pivaloyl amino acid esters to measure 15N isotopic abundances in physiological samples: A pilot study on amino acid synthesis in the upper gastro-intestinal tract of minipigs. J. Mass Spectrom. 1996, 31, 367–376. [Google Scholar] [CrossRef]
- Millward, D.J.; Forrester, T.; Ah-Sing, E.; Yeboah, N.; Gibson, N.; Badaloo, A.; Boyne, M.; Reade, M.; Persaud, C.; Jackson, A. The transfer of 15N from urea to lysine in the human infant. Br. J. Nutr. 2000, 83, 505–512. [Google Scholar] [CrossRef] [Green Version]
- Neis, E.P.; Dejong, C.H.; Rensen, S.S. The role of microbial amino acid metabolism in host metabolism. Nutrients 2015, 7, 2930–2946. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Metges, C.C. Contribution of microbial amino acids to amino acid homeostasis of the host. J. Nutr. 2000, 130, 1857S–1864S. [Google Scholar] [CrossRef]
- Sridharan, G.V.; Choi, K.; Klemashevich, C.; Wu, C.; Prabakaran, D.; Pan, L.B.; Steinmeyer, S.; Mueller, C.; Yousofshahi, M.; Alaniz, R.C.; et al. Prediction and quantification of bioactive microbiota metabolites in the mouse gut. Nat. Commun. 2014, 5, 5492. [Google Scholar] [CrossRef] [Green Version]
- Smith, E.A.; Macfarlane, G.T. Enumeration of human colonic bacteria producing phenolic and indolic compounds: Effects of pH, carbohydrate availability and retention time on dissimilatory aromatic amino acid metabolism. J. Appl. Bacteriol. 1996, 81, 288–302. [Google Scholar] [CrossRef]
- Dai, Z.L.; Wu, G.; Zhu, W.Y. Amino acid metabolism in intestinal bacteria: Links between gut ecology and host health. Front. Biosci. 2011, 16, 1768–1786. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Allison, C.; Macfarlane, G.T. Influence of pH, nutrient availability, and growth rate on amine production by Bacteroides fragilis and Clostridium perfringens. Appl. Environ. Microbiol. 1989, 55, 2894–2898. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Davila, A.M.; Blachier, F.; Gotteland, M.; Andriamihaja, M.; Benetti, P.H.; Sanz, Y.; Tome, D. Re-print of “Intestinal luminal nitrogen metabolism: Role of the gut microbiota and consequences for the host”. Pharmacol. Res. 2013, 69, 114–126. [Google Scholar] [CrossRef]
- Aguirre, M.; Eck, A.; Koenen, M.E.; Savelkoul, P.H.; Budding, A.E.; Venema, K. Diet drives quick changes in the metabolic activity and composition of human gut microbiota in a validated in vitro gut model. Res. Microbiol. 2016, 167, 114–125. [Google Scholar] [CrossRef]
- Kaiser, J.C.; Heinrichs, D.E. Branching Out: Alterations in Bacterial Physiology and Virulence Due to Branched-Chain Amino Acid Deprivation. mBio 2018, 9, e01188-18. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.M.; Rock, C.O. Membrane lipid homeostasis in bacteria. Nat. Rev. Microbiol. 2008, 6, 222–233. [Google Scholar] [CrossRef]
- Scully, S.M.; Orlygsson, J. Branched-chain amino acid catabolism of Thermoanaerobacter pseudoethanolicus reveals potential route to branched-chain alcohol formation. Extremophiles 2020, 24, 121–133. [Google Scholar] [CrossRef]
- Sun, Y.; Meng, Q.; Zhang, Y.; Gao, H. Derepression of bkd by the FadR loss dictates elevated production of BCFAs and isoleucine starvation. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2020, 1865, 158577. [Google Scholar] [CrossRef]
- David, L.A.; Maurice, C.F.; Carmody, R.N.; Gootenberg, D.B.; Button, J.E.; Wolfe, B.E.; Ling, A.V.; Devlin, A.S.; Varma, Y.; Fischbach, M.A.; et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 2014, 505, 559–563. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shortt, C.; Hasselwander, O.; Meynier, A.; Nauta, A.; Fernandez, E.N.; Putz, P.; Rowland, I.; Swann, J.; Turk, J.; Vermeiren, J.; et al. Systematic review of the effects of the intestinal microbiota on selected nutrients and non-nutrients. Eur. J. Nutr. 2018, 57, 25–49. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ottosson, F.; Brunkwall, L.; Ericson, U.; Nilsson, P.M.; Almgren, P.; Fernandez, C.; Melander, O.; Orho-Melander, M. Connection Between BMI-Related Plasma Metabolite Profile and Gut Microbiota. J. Clin. Endocrinol. Metab. 2018, 103, 1491–1501. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Org, E.; Blum, Y.; Kasela, S.; Mehrabian, M.; Kuusisto, J.; Kangas, A.J.; Soininen, P.; Wang, Z.; Ala-Korpela, M.; Hazen, S.L.; et al. Relationships between gut microbiota, plasma metabolites, and metabolic syndrome traits in the METSIM cohort. Genome Biol. 2017, 18, 70. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Wan, Y.; Yin, K.; Wei, Y.; Wang, B.; Yu, X.; Ni, Y.; Zheng, J.; Huang, T.; Song, M.; et al. Lower Circulating Branched-Chain Amino Acid Concentrations Among Vegetarians are Associated with Changes in Gut Microbial Composition and Function. Mol. Nutr. Food Res. 2019, 63, e1900612. [Google Scholar] [CrossRef]
- Mesnage, R.; Grundler, F.; Schwiertz, A.; Le Maho, Y.; Wilhelmi de Toledo, F. Changes in human gut microbiota composition are linked to the energy metabolic switch during 10 d of Buchinger fasting. J. Nutr. Sci. 2019, 8, e36. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, L.; Yue, Y.; Shi, M.; Tian, M.; Ji, J.; Liao, X.; Hu, X.; Chen, F. Dietary Luffa cylindrica (L.) Roem promotes branched-chain amino acid catabolism in the circulation system via gut microbiota in diet-induced obese mice. Food Chem. 2020, 320, 126648. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Nie, Q.; Hu, J.; Huang, X.; Yin, J.; Nie, S. Multiomics Approach to Explore the Amelioration Mechanisms of Glucomannans on the Metabolic Disorder of Type 2 Diabetic Rats. J. Agric. Food Chem. 2021, 69, 2632–2645. [Google Scholar] [CrossRef] [PubMed]
- Ridaura, V.K.; Faith, J.J.; Rey, F.E.; Cheng, J.; Duncan, A.E.; Kau, A.L.; Griffin, N.W.; Lombard, V.; Henrissat, B.; Bain, J.R.; et al. Gut microbiota from twins discordant for obesity modulate metabolism in mice. Science 2013, 341, 1241214. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yue, S.J.; Liu, J.; Wang, A.T.; Meng, X.T.; Yang, Z.R.; Peng, C.; Guan, H.S.; Wang, C.Y.; Yan, D. Berberine alleviates insulin resistance by reducing peripheral branched-chain amino acids. Am. J. Physiol. Endocrinol. Metab. 2019, 316, E73–E85. [Google Scholar] [CrossRef]
- Wu, W.; Zhang, L.; Xia, B.; Tang, S.; Liu, L.; Xie, J.; Zhang, H. Bioregional Alterations in Gut Microbiome Contribute to the Plasma Metabolomic Changes in Pigs Fed with Inulin. Microorganisms 2020, 8, 111. [Google Scholar] [CrossRef] [Green Version]
- Sun, Y.; He, Z.; Li, J.; Gong, S.; Yuan, S.; Li, T.; Ning, N.; Xing, L.; Zhang, L.; Chen, F.; et al. Gentamicin Induced Microbiome Adaptations Associate With Increased BCAA Levels and Enhance Severity of Influenza Infection. Front. Immunol. 2020, 11, 608895. [Google Scholar] [CrossRef]

| Organism | Condition | Effect on Physiology | Microbiome Composition | Microbiome Functionality | Metabolome | |
|---|---|---|---|---|---|---|
| Pedersen H. et al. [38] | human | IR/MetSy | increased Prevotella. copri, B. vulgatus; decreased Butyrivibrio crosstus, Eubacterium siraeum | upregulated BCAA biosynthesis (correlates with P. copri and B. vulgatus); downregulated inward BCAA transport (correlates with B. crosstus and E. siraeum) | increased s-BCAA | |
| T2D | increased P. copri, B. vulgatus; decreased Butyrivibrio crosstus, Eubacterium siraeum | Upregulated BCAA biosynthesis; downregulated inward BCAA transport. | increased s-BCAA | |||
| Ottosson F. et al. [56] | Human (Malmo Offspring Study) | obesity | increased Dorea, Blautia, Ruminococcus, positively correlate with PC-BMI decreased SHA-98, negatively correlate with PC-BMI | increased PC-BMI metabolites: glutamate, BCAA | ||
| Org E. et al. [57] | Human (METSIM study) | MetSy | Blautia positively correlated with s-BCAAs, high BMI and HOMA-IR Christensenellaceae negatively correlated with s-BCAAs | |||
| Wang F. et al. [58] | human | VG VEG OMNI | VG and VEG vs OMNI: lower intake of energy, fat, chol.; higher intake of carbohydrates and fiber | VG and VEG vs OMNI: higher Prevotella (including P. copri) lower Alistipes, Bacteroides (NOT B. thetaiotaomicron or B. ovatus), Bilophila, Collinsella, Parabacteroides | VG and VEG vs OMNI: upregulated BCAAs degradation. | VG and VEG vs OMNI: lower s-BCAAs |
| Mesnage R. et al. [59] | human | 10-day fasting (1046 kJ/day) | switch from carbohydrate to fatty acid oxidation, ketosis | decreased Lachnospiraceae, Ruminococcaceae; increased E.coli, B. fragilis, B. thetaiotaomicron, Bilophila waldsworthia | increased s-BCAA; negative correlation: BCAA/Lachnospiraceae positive correlation: BCAA/B.fragilis, B. thetaiotaomicron, Bilophila |
| Organism | Condition | Effect on Physiology | Microbiome Composition | Microbiome Functionality | Metabolome | |
|---|---|---|---|---|---|---|
| Zeng et al. [40] | mice | HFD | weight gain; compromised glucose homeostasis and serum lipid profile; stimulated mTOR/p70S6K/SREB pathway | increased Firmicutes to Bacteroidetes ratio, Ruminococcus; decreased S24-7, Ruminococcaceae, Lachnospiraceae, Bacteroides, Oscillospira, Rikenellaceae | upregulated BCAA biosynthesis, decreased BCAA degradation | serum: shift in 73 metabolites feces: shift in 91 metabolites both including elevated BCAA |
| Zhang et al. [60] | mice | HFD | obesity-associated insulin resistance | increased Enterorhabdus, Acetatifactor, Butyricoccus, Sterptococcus, Eubacterium_xylanophilum_group, Escherichia-Shigella; decreased Alloprevotella, Parasutterella, Parabacteroides, Eubacterium_coprostanoligenes_group, Christensenellaceae_R-7_group. | upregulated BCAA synthesis; no change in BCAA degradation | increased s-BCAA |
| Chen et al. [61] | rat | diabetes model (HFD + STZ) | fasting hyperglycemia; decreased tissue BCAA metabolism | Increased Proteobacteria, Lachnospiraceae_uncultured and Lachnoospiraceae_NK4A136 decreased Firmicutes to Bacteroidetes ratio, Muribaculaceae | Upregulated AA and BCAA biosynthesis; no change in BCAA degradation | altered AA metabolism, increased s-BCAA; positive correlation between Bacteroides pectinophilus group, Bacteroides, Klebsiella, Prevotellaceae Ga6A1 group, Prevotellaceae NK3B31 group, Prevotellaceae UCG-001, Ruminiclostridium, Ruminiclostridium 1, 5 and 9, Staphylococcus, Streptococcus and BCAA |
| Model | Treatment | Effect on Physiology | Microbiome Composition | Microbiome Functionality | Metabolome | |
|---|---|---|---|---|---|---|
| Ridaura et al. [62] | humanized mice | fecal Tx from obese twin | reproduction of obese phenotype | upregulated AA-metabolism related pathways (Phe, Lys, Leu, Ile, Val, Arg, Cys, Tyr) | increased s-BCAA, Met, Ser, Gly, Phe, Ala and Tyr | |
| Pedersen H. et al. [38] | mice | HFD, P. copri/sham gavage | P. copri aggravated glucose tolerance, reduced insulin sensitivity | elevated P. copri; no other changes in microbiota composition | upregulated BCAA biosynthesis | increased s-BCAA |
| Zeng S. et al. [40] | mice | HFD + B. ovatus/sham gavage | decreased fat accumulation, ameliorated lipid profile and liver function tests | increased B. ovatus | decreased f- and s-BCAA | |
| Liu R. et al. [39] | mice | HFD + B. thetaiota-omicron/sham gavage | lower adiposity; improved inflammatory status | increased B. thetaiotaomicrone, no substantial change in the whole microbiome | decreased circulating AA (glutamate, Phe, Leu, Val) |
| Model | Treatment | Effect on Physiology | Microbiome Composition | Microbiome Functionality | Metabolome | |
|---|---|---|---|---|---|---|
| Zeng S. et al. [40] | Mice HFD | PMFE | PMFE protected against MetSy in HFD mice PMFE inhibited mTOR/P70S6K/SREBP pathway | increased Bacteroides, S24-7, Ruminococcaceae, Oscillospira, Lachnospiraceae; decreased Paraprevotella, Streptococcus | upregulated BCAA degradation | prevention of HFD-induced increase in s-BCAA and f-BCAA |
| fermentation in vitro | fresh feces (HFD mice) + PMFE added in vitro | increased B. ovatus, B. thetaiotaomicron, B. vulgatus, B. caccae, B. stercoris, B. uniformis decreased B. fragilis, B. finegoldii, B. coprophilus | ||||
| Zhang L. et al. [60] | Mice HFD | Luffa cylindrica | alleviation of obesity-associated insulin resistance | HFD induced significant shift in microbiota composition, luffa induced decrease in Enterorhabdus, Butyricoccus, Eubacterium_xylanophilum_group | downregulated BCAA synthesis; no change in BCAA degradation | decreased s-BCAA |
| Luffa cylindrica + ATB | no effect on obesity-related parameters; partial alleviation of glucose intolerance | microbiota depletion | no effect on s-BCAA compared with HFD-fed mice | |||
| Yue S. et al. [63] | Mice HFD | berberine | alleviation of HFD-induced obesity and glucose intolerance | restoration of HFD-induced shift in Firmicutes to Bacteroidetes ratio increased Akkermansia, selective reductions in Clostridiales, Streptococcaceae, Clostridiaceae, Prevotellaceae, decreased Streptococcus, Prevotella | downregulated BCAA biosynthesis; upregulated BCAA degradation | prevented HFD-induced increase in s-BCAA (valine p < 0.05, leucine p = 0.17, isoleucine p = 0.06); downregulated bacterial taxa correlate positively with s-BCAA |
| Chen H. et al. [61] | rat HFD + streptozotocin | gluco-mannans | hypoglycemic, hypolipidemic, kidney-protective effect | decreased Clostridium spp., Bacteroides spp., Prevotella spp., Klebsiella spp., Escherichia coli, Streptococcus spp., Staphylococcus aureus | downregulated BCAA biosynthesis; no change in BCAA degradation | decreased s-BCAA |
| Wu W. et al. [64] | growing pig | inulin | lower s-cholesterol and s-glucose | increased abundance of 10 genera (including Prevotella and Succinivibrio), decreased abundance of six genera | upregulated BCAA degradation | decreased s-BCAA positive correlations between Prevotella/Val and Leu Succinivibrio/ValClostridium_sensu_stricto_1/Ile and Leu |
| Crovesy L. et al. [65] | obese women | low-energy diet | decreased glycerol; increased Arg, Glu and 2-oxoisovalerate | |||
| low-energy diet + SG | dtto + increased Pyr, Ala; decreased citrate, Ile and total BCAA; positive correlations∆ Verrucomicrobia/∆ Ile∆ Firmicutes/∆ Ile and ∆ total BCAA | |||||
| Sun Y. et al. [66] | mice influenza virus infection | gentamicin | reduced survival; decreased number of suppressor CD11b + Ly6G + cells, increased CD8 + cells in lung, enhanced inflammation | increased Bacteroidetes decreased Proteobacteria | increased s-BCAA |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gojda, J.; Cahova, M. Gut Microbiota as the Link between Elevated BCAA Serum Levels and Insulin Resistance. Biomolecules 2021, 11, 1414. https://doi.org/10.3390/biom11101414
Gojda J, Cahova M. Gut Microbiota as the Link between Elevated BCAA Serum Levels and Insulin Resistance. Biomolecules. 2021; 11(10):1414. https://doi.org/10.3390/biom11101414
Chicago/Turabian StyleGojda, Jan, and Monika Cahova. 2021. "Gut Microbiota as the Link between Elevated BCAA Serum Levels and Insulin Resistance" Biomolecules 11, no. 10: 1414. https://doi.org/10.3390/biom11101414
APA StyleGojda, J., & Cahova, M. (2021). Gut Microbiota as the Link between Elevated BCAA Serum Levels and Insulin Resistance. Biomolecules, 11(10), 1414. https://doi.org/10.3390/biom11101414






