RETRACTED: Optimized Ellagic Acid–Ca Pectinate Floating Beads for Gastroprotection against Indomethacin-Induced Gastric Injury in Rats
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Experimental Design of EA–Ca Pectinate Beads
2.3. EA–Ca Pectinate Bead Preparation
2.4. Determination of EA–Ca Pectinate Buoyancy Time
2.5. Determination of EA Entrapped Percent
2.6. In Vitro Release Evaluation of EA–Ca Pectinate Beads
2.7. In Vitro Release Evaluation of EA–Ca Pectinate Beads
2.8. In Vivo Evaluation of Optimized EA–Ca Pectinate Beads
2.8.1. Animals
2.8.2. Animal Groups
2.8.3. Induction of Gastric Ulcers
2.8.4. Assessment of Gastric Mucosal Lesions
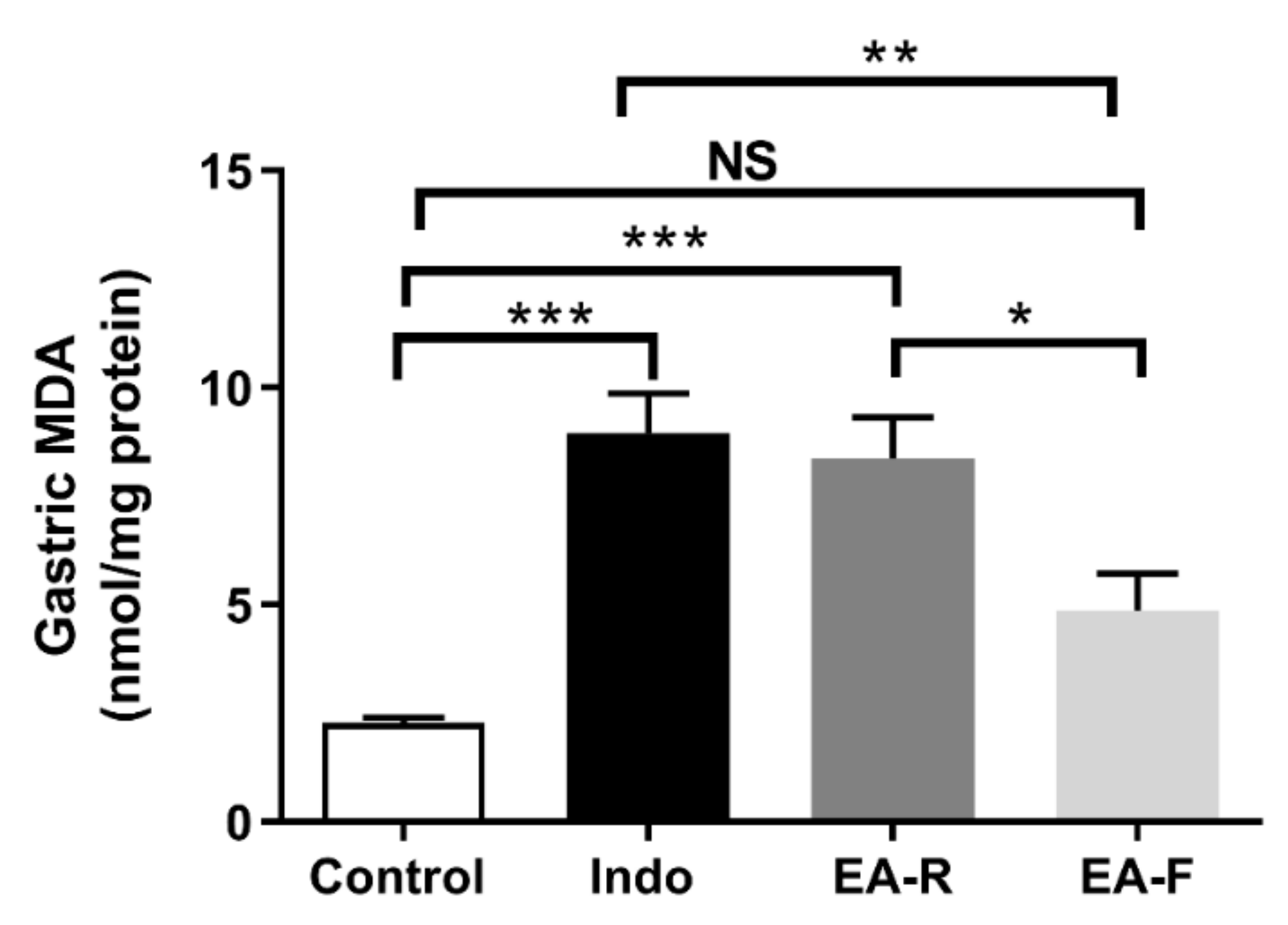
2.8.5. Determination of Gastric Mucosal Lipid Peroxidation
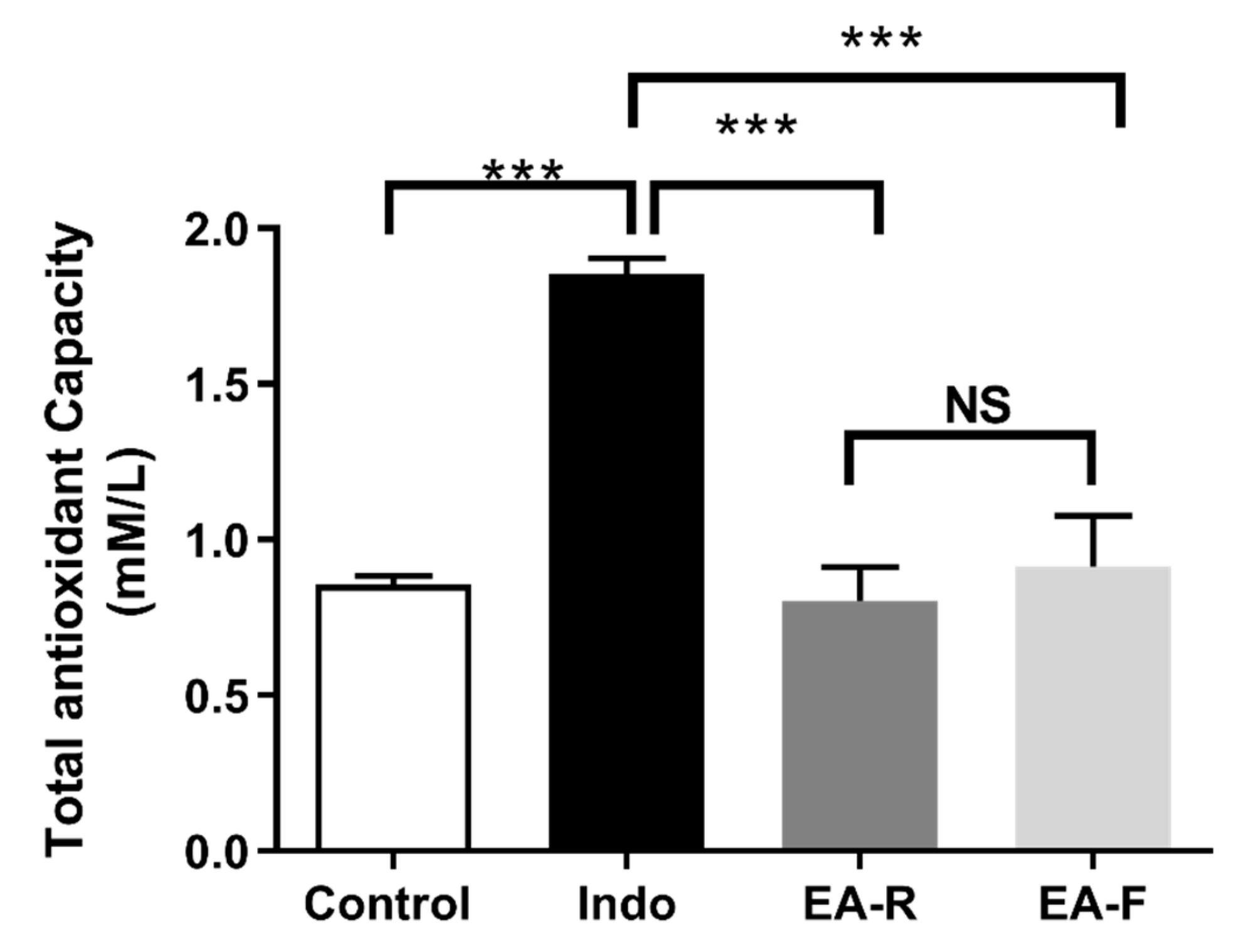
2.8.6. Determination of Serum Total Antioxidant Capacity
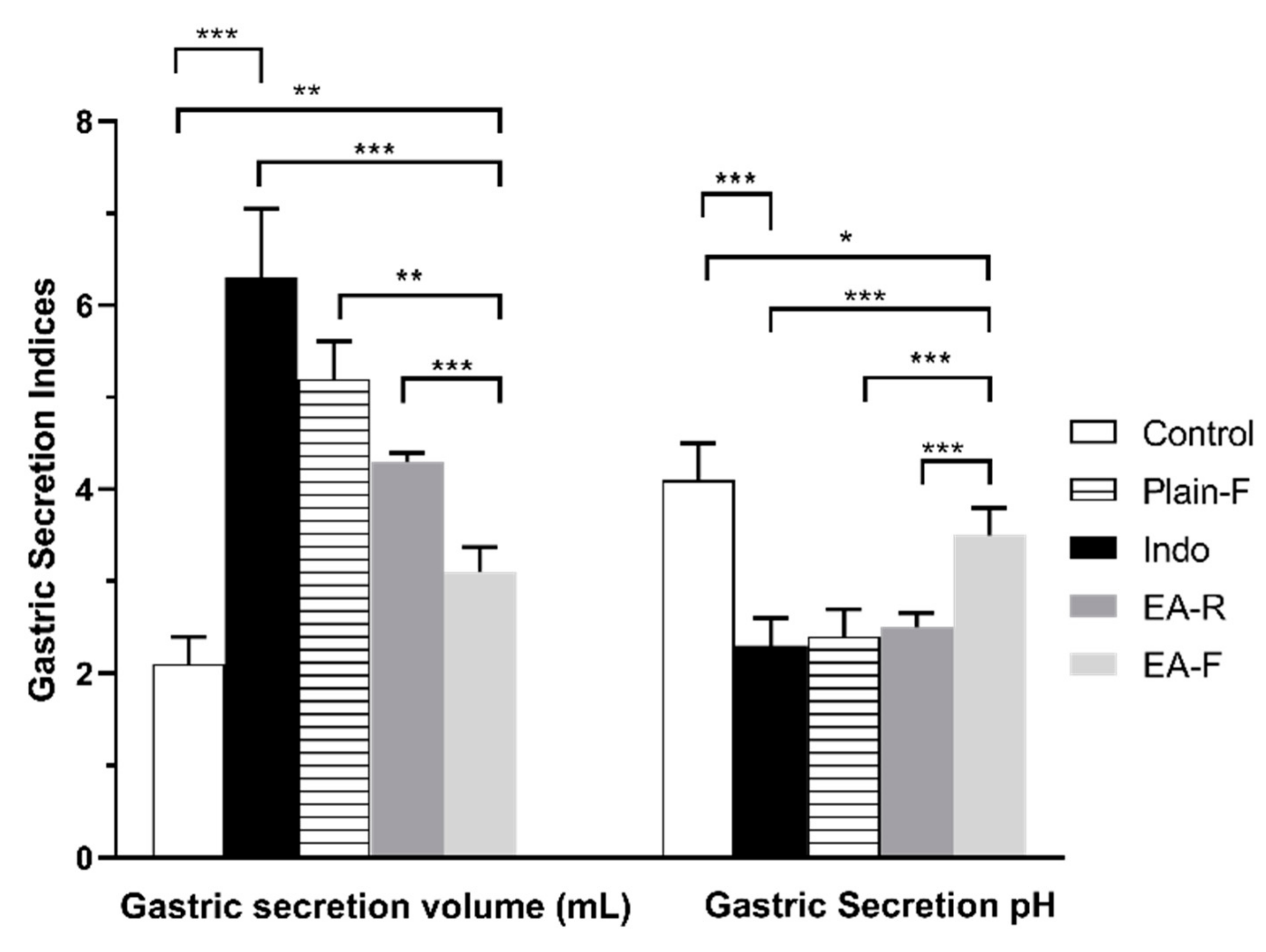
2.8.7. Determination of Gastric Secretion Parameters
2.8.8. Statistical Analysis
3. Results
3.1. Determination of EA–Ca Pectinate Buoyancy Time
3.2. Statistical Analysis of the Factorial Design
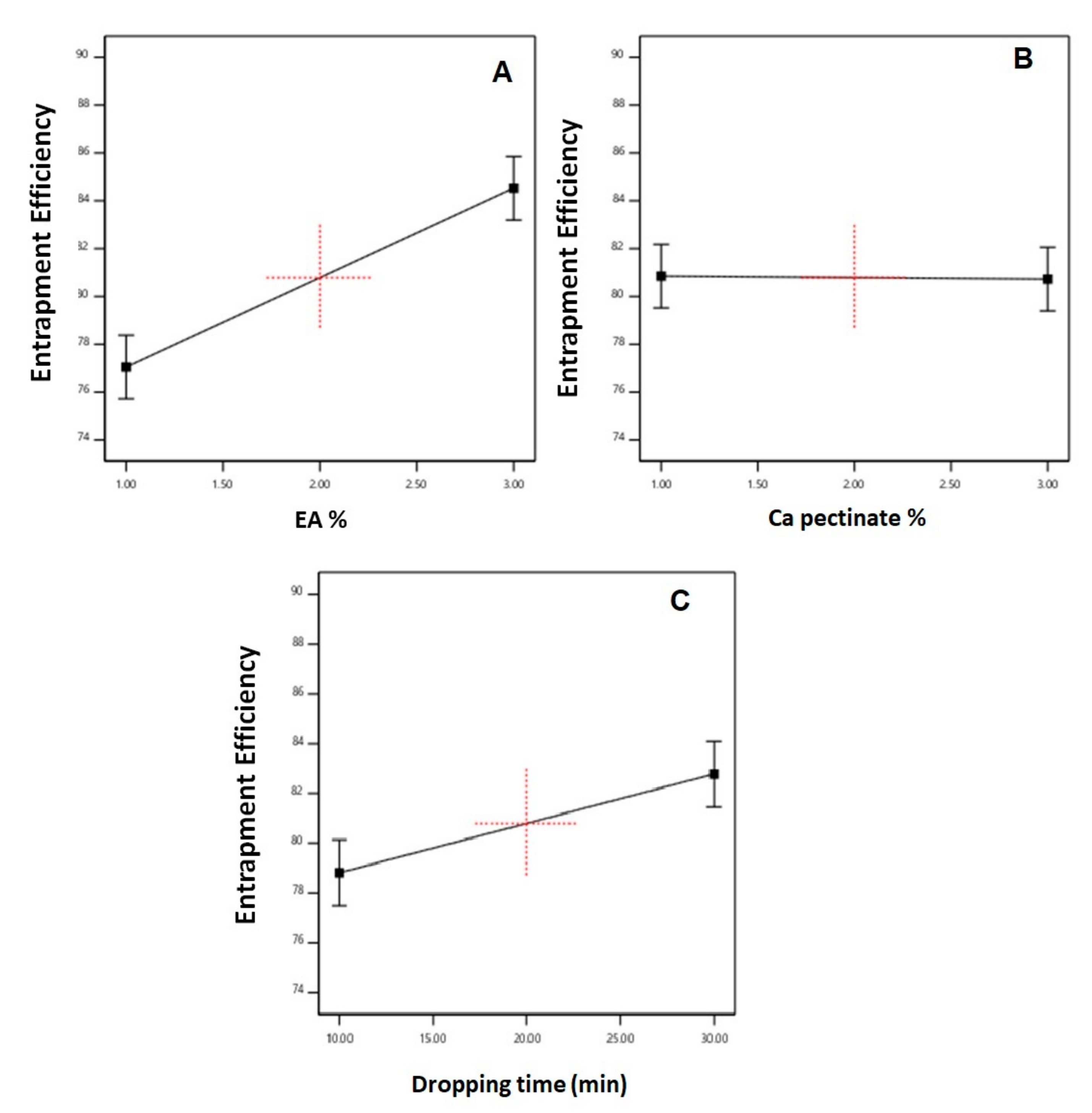
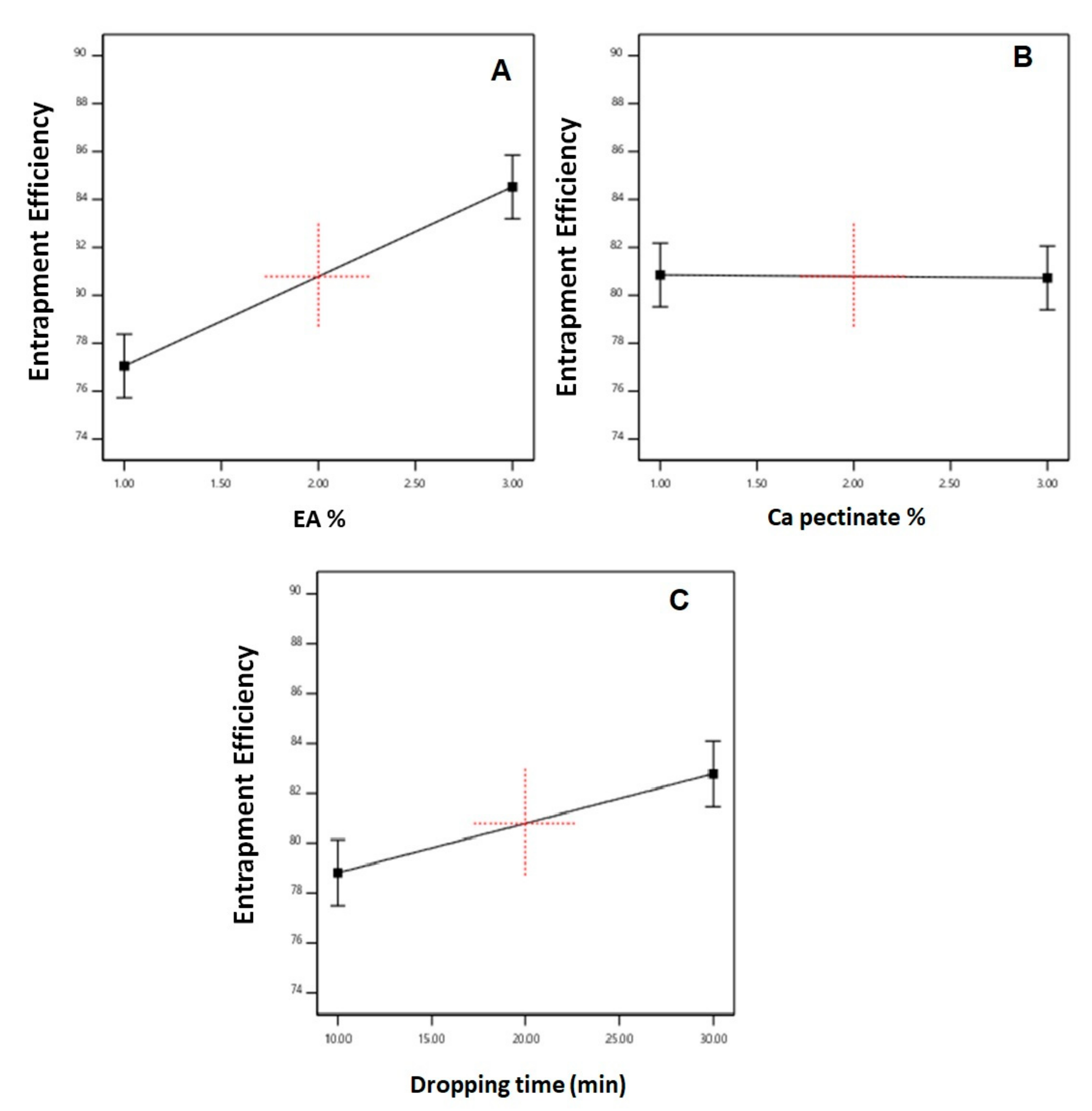
3.2.1. Effect of Variables on Entrapment Efficiency (Y1)
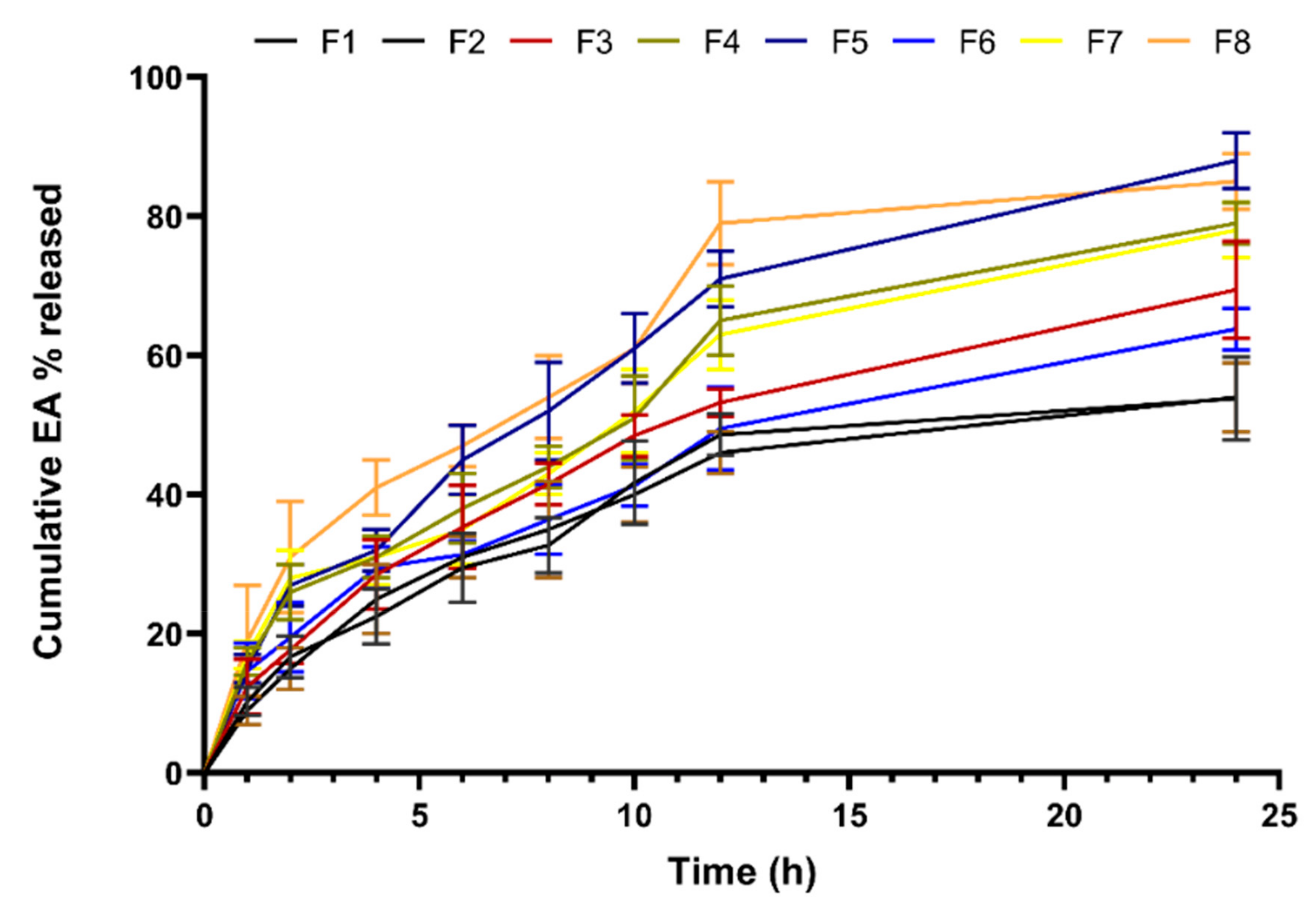
3.2.2. Effect of Variables on Cumulative EA% Released (Y2)
3.2.3. Selection of the Optimized EA–Ca Pectinate Beads
3.3. In Vivo Evaluation of Optimized EA Formulation
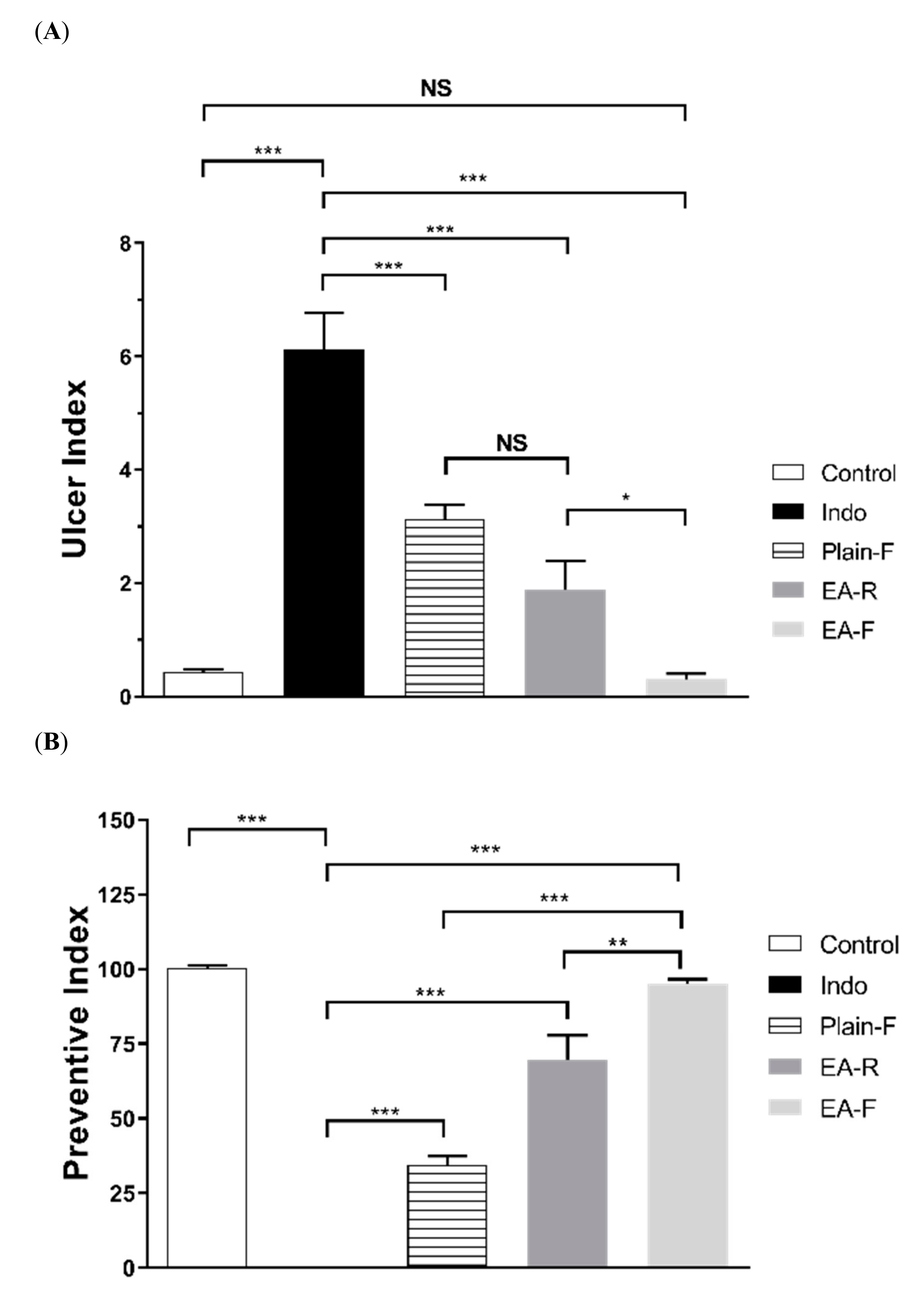
3.3.1. Effect of Pure EA and Optimized EA–Ca Pectinate Beads on Indomethacin-Induced Gastric Lesions
3.3.2. Effect of Pure EA and Optimized EA Formulation on Gastric pH
3.3.3. Effect of Pure EA and Optimized EA Formulation on Gastric Mucosal Oxidative Stress
3.3.4. Effect of Pure EA and Optimized EA Formulation on Serum Total Antioxidant Capacity
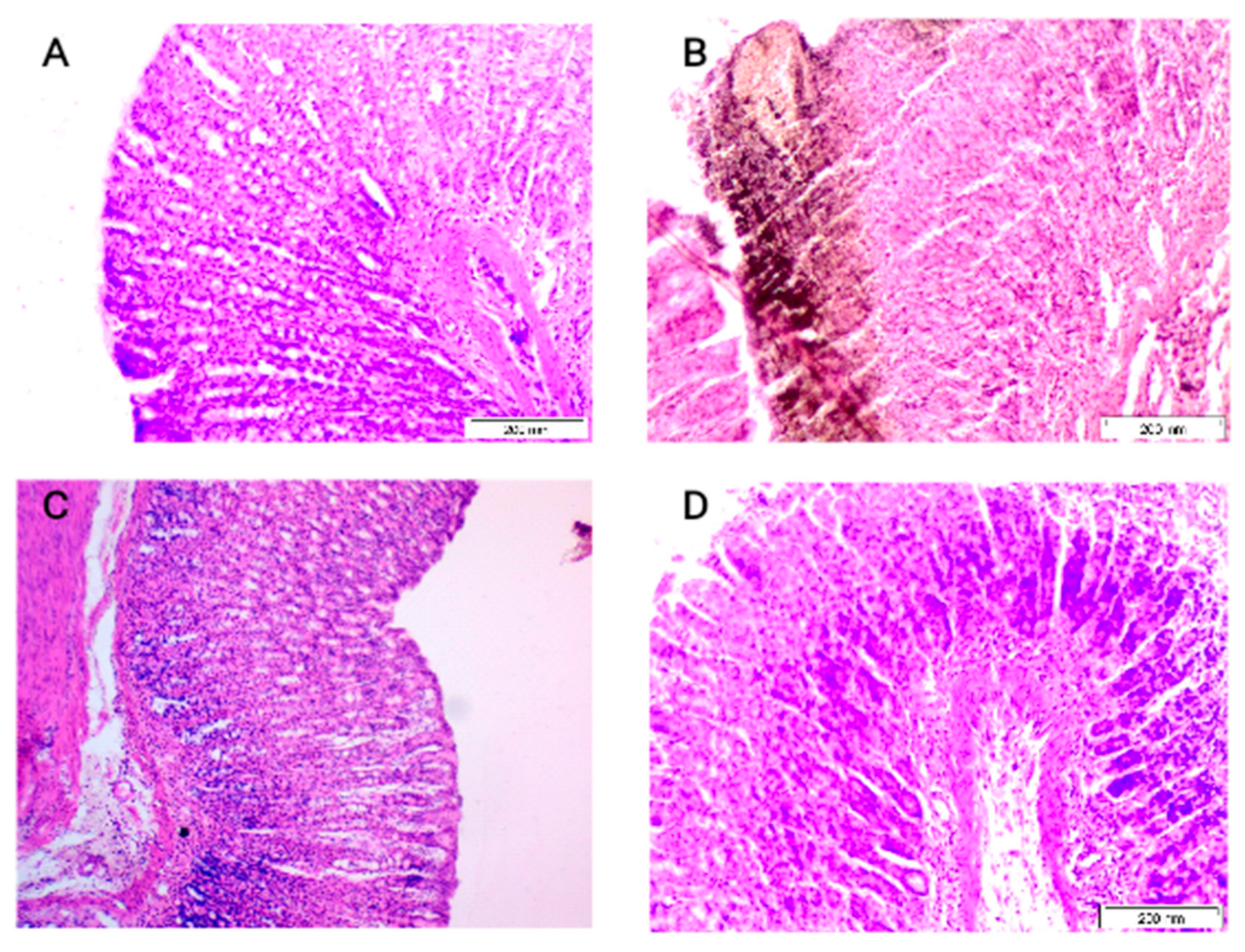
3.3.5. Effect of Pure EA and Optimized EA Formulation on the Histopathological Features of the Stomach
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Stack, W.A.; Hawkey, G.M.; Atherton, J.C.; Logan, R.F.A. Interactions between Helicobacter pylori and other risk factors for peptic ulcer bleeding. Aliment. Pharmacol. Ther. 2002, 16, 497–506. [Google Scholar] [CrossRef]
- Talley, N.J.; Locke, G.R.; Moayyedi, P.; West, J.; Ford, A.C.; Saito, Y.A. GI Epidemiology: Diseases and Clinical Methodology, 2nd ed.; Wiley Blackwell: Hoboken, NJ, USA, 2014; ISBN 9781118727072. [Google Scholar]
- Dunlap, J.; Patterson, S. PEPTIC ULCER DISEASE. Gastroenterol. Nurs. 2019, 42, 451–454. [Google Scholar] [CrossRef]
- Dumic, I.; Nordin, T.; Jecmenica, M.; Lalosevic, M.S.; Milosavljevic, T.; Alempijević, T. Gastrointestinal Tract Disorders in Older Age. Can. J. Gastroenterol. Hepatol. 2019, 2019, 6757524. [Google Scholar] [CrossRef] [PubMed]
- Goriacko, P.; Veltri, K.T. Adverse Drug Effects Involving the Gastrointestinal System (Pharmacist Perspective); Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2019; pp. 1–44. [Google Scholar]
- Rodríguez, L.A.G.; Lanas, A.; Soriano-Gabarró, M.; Soriano, L.C. Low-dose aspirin and risk of upper/lower gastrointestinal bleeding by bleed severity: A cohort study with nested case-control analysis using primary care electronic health records from the United Kingdom. Ann. Med. 2019, 51, 182–192. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Wu, H.-Q.; Cui, H.-L.; Li, Y.-Y.; Li, C.-Z. Gastroprotective and anti-ulcer effects of oxymatrine against several gastric ulcer models in rats: Possible roles of antioxidant, antiinflammatory, and prosurvival mechanisms. Phyther. Res. 2018, 32, 2047–2058. [Google Scholar] [CrossRef]
- Cheong, A.M.; Tan, Z.W.; Patrick, N.O.; Tan, C.; Lim, Y.M.; Nyam, K.-L. Improvement of gastroprotective and anti-ulcer effect of kenaf seed oil-in-water nanoemulsions in rats. Food Sci. Biotechnol. 2018, 27, 1175–1184. [Google Scholar] [CrossRef] [PubMed]
- Vaxman, I.; Dispenzieri, A.; Muchtar, E.; Gertz, M. New developments in diagnosis, risk assessment and management in systemic amyloidosis. Blood Rev. 2020, 40, 100636. [Google Scholar] [CrossRef] [PubMed]
- Goenka, M.K.; Majumder, S.; Sethy, P.K.; Chakraborty, M. Helicobacter pylori negative, non-steroidal anti-inflammatory drug-negative peptic ulcers in India. Indian J. Gastroenterol. 2011, 30, 33–37. [Google Scholar] [CrossRef]
- Ng, E.K.W.; Chung, S.C.S.; Sung, J.J.Y.; Lam, Y.H.; Lee, D.W.H.; Lau, J.Y.W.; Ling, T.K.W.; Lau, W.Y.; Li, A.K.C. High prevalence of Helicobacter pylori infection in duodenal ulcer perforations not caused by non-steroidal anti-inflammatory drugs. Br. J. Surg. 1996, 83, 1779–1781. [Google Scholar] [CrossRef]
- Voutilainen, M.; Mäntynen, T.; Fär, M. Impact of Non-steroidal Anti-inflammatory Drug and Aspirin Use on the Prevalence of Dyspepsia and Uncomplicated Peptic Ulcer Disease. Scand. J. Gastroenterol. 2001, 36, 817–821. [Google Scholar] [CrossRef]
- González-Sarrías, A.; Larrosa, M.; Tomás-Barberán, F.A.; Dolara, P.; Espín, J.C. NF-kB-dependent anti-inflammatory activity of urolithins, gut microbiota ellagic acid-derived metabolites, in human colonic fibroblasts. Br. J. Nutr. 2010, 104, 503–512. [Google Scholar] [CrossRef]
- Kaya, I.; Karapehlivan, M. The Investigation of Biochemical and Histological Effects of Ellagic Acid in Experimental Chronic Fluorosis Induced Mice. Int. J. Health Sci. Res. 2017, 7, 396–406. [Google Scholar]
- Masamune, A.; Satoh, M.; Kikuta, K.; Suzuki, N.; Satoh, K.; Shimosegawa, T. Ellagic acid blocks activation of pancreatic stellate cells. Biochem. Pharmacol. 2005, 70, 869–878. [Google Scholar] [CrossRef]
- Ahad, A.; Ganai, A.A.; Mujeeb, M.; Siddiqui, W.A. Ellagic acid, an NF-κB inhibitor, ameliorates renal function in experimental diabetic nephropathy. Chem. Biol. Interact. 2014, 219, 64–75. [Google Scholar] [CrossRef] [PubMed]
- Poynard, T.; Vernisse, B.; Agostini, H. Randomized, multicentre comparison of sodium alginate and cisapride in the symptomatic treatment of uncomplicated gastro-oesophageal reflux. Aliment. Pharmacol. Ther. 1998, 12, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Jain, A.; Jain, R.; Jain, S.; Khatik, R.; Kohli, D.V. Minicapsules encapsulating nanoparticles for targeting, apoptosis induction and treatment of colon cancer. Artif. Cells Nanomed. Biotechnol. 2019, 47, 1085–1093. [Google Scholar] [CrossRef]
- Liu, Q.S.; Deng, R.; Li, S.; Li, X.; Li, K.; Kebaituli, G.; Li, X.; Liu, R. Ellagic acid protects against neuron damage in ischemic stroke through regulating the ratio of Bcl-2/Bax expression. Appl. Physiol. Nutr. Metab. 2017, 42, 855–860. [Google Scholar] [CrossRef]
- Malik, A.; Afaq, S.; Assiri, A.; Shahid, M.; Akhtar, K. Influence of ellagic acid on prostate cancer cell proliferation: A caspase–dependent pathway. Asian Pac. J. Trop. Med. 2011, 4, 550–555. [Google Scholar] [CrossRef]
- Rosillo, M.A.; Sánchez-Hidalgo, M.; Cárdeno, A.; De La Lastra, A. Protective effect of ellagic acid, a natural polyphenolic compound, in a murine model of Crohn’s disease. Biochem. Pharmacol. 2011, 82, 737–745. [Google Scholar] [CrossRef]
- Ratnam, D.V.; Bhardwaj, V.; Kumar, M.N.V.R. Simultaneous analysis of ellagic acid and coenzyme Q10 by derivative spectroscopy and HPLC. Talanta 2006, 70, 387–391. [Google Scholar] [CrossRef]
- Kuo, M.-Y.; Ou, H.-C.; Lee, W.-J.; Kuo, W.-W.; Hwang, L.-L.; Song, T.-Y.; Huang, C.-Y.; Chiu, T.-H.; Tsai, K.-L.; Tsai, C.-S.; et al. Ellagic Acid Inhibits Oxidized Low-Density Lipoprotein (OxLDL)-Induced Metalloproteinase (MMP) Expression by Modulating the Protein Kinase C-α/Extracellular Signal-Regulated Kinase/Peroxisome Proliferator-Activated Receptor γ/Nuclear Factor-κB (PKC-α/ERK/PPAR-γ/NF-κB) Signaling Pathway in Endothelial Cells. J. Agric. Food Chem. 2011, 59, 5100–5108. [Google Scholar] [CrossRef]
- Ahmed, O.A.A.; Abourehab, M.A.; Khaled, K.A.; Sarhan, H.A. Evaluation of combined famotidine with quercetin for the treatment of peptic ulcer: In vivo animal study. Drug Des. Dev. Ther. 2015, 9, 2159–2169. [Google Scholar] [CrossRef] [PubMed]
- Uchiyama, M.; Mihara, M. Determination of malonaldehyde precursor in tissues by thiobarbituric acid test. Anal. Biochem. 1978, 86, 271–278. [Google Scholar] [CrossRef]
- Ponchel, G.; Irache, J.M. Specific and non-specific bioadhesive particulate systems for oral delivery to the gastrointestinal tract. Adv. Drug Deliv. Rev. 1998, 34, 191–219. [Google Scholar] [CrossRef]
- Mandal, U.K.; Chatterjee, B.; Senjoti, F.G. Gastro-retentive drug delivery systems and their in vivo success: A recent update. Asian J. Pharm. Sci. 2016, 11, 575–584. [Google Scholar] [CrossRef]
- Arora, S.; Ali, J.; Ahuja, A.; Khar, R.K.; Baboota, S. Floating drug delivery systems: A review. AAPS PharmSciTech 2005, 6, E372–E390. [Google Scholar] [CrossRef] [PubMed]
- Ciriminna, R.; Fidalgo, A.; DeLisi, R.; Tamburino, A.; Carnaroglio, D.; Cravotto, G.; Ilharco, L.M.; Pagliaro, M. Controlling the Degree of Esterification of Citrus Pectin for Demanding Applications by Selection of the Source. ACS Omega 2017, 2, 7991–7995. [Google Scholar] [CrossRef]
- Sriamornsak, P.; Thirawong, N.; Puttipipatkhachorn, S. Emulsion gel beads of calcium pectinate capable of floating on the gastric fluid: Effect of some additives, hardening agent or coating on release behavior of metronidazole. Eur. J. Pharm. Sci. 2005, 24, 363–373. [Google Scholar] [CrossRef]
- Das, S.; Ng, K.-Y. Resveratrol-loaded calcium-pectinate beads: Effects of formulation parameters on drug release and bead characteristics. J. Pharm. Sci. 2010, 99, 840–860. [Google Scholar] [CrossRef]
- Ahmed, O.; Fahmy, U.; Bakhaidar, R.; El-Moselhy, M.A.; Okbazghi, S.Z.; Ahmed, A.-S.F.; Hammad, A.S.A.; Alhakamy, N. Omega-3 Self-Nanoemulsion Role in Gastroprotection against Indomethacin-Induced Gastric Injury in Rats. Pharmaceutics 2020, 12, 140. [Google Scholar] [CrossRef]
- Hosny, K.M.; Bahmdan, R.H.; Alhakamy, N.A.; AlFaleh, M.A.; Ahmed, O.A.; Elkomy, M.H. Physically Optimized Nano-Lipid Carriers Augment Raloxifene and Vitamin D Oral Bioavailability in Healthy Humans for Management of Osteoporosis. J. Pharm. Sci. 2020. [Google Scholar] [CrossRef] [PubMed]
- Roy, P.; Shahiwala, A. Statistical optimization of ranitidine HCl floating pulsatile delivery system for chronotherapy of nocturnal acid breakthrough. Eur. J. Pharm. Sci. 2009, 37, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Priyadarshi, R.; Sauraj; Kumar, B.; Deeba, F.; Kulshreshtha, A.; Negi, Y.S. Chitosan films incorporated with Apricot (Prunus armeniaca) kernel essential oil as active food packaging material. Food Hydrocoll. 2018, 85, 158–166. [Google Scholar] [CrossRef]
- Fathi, N.; Almasi, H.; PirouziFard, M.K. Effect of ultraviolet radiation on morphological and physicochemical properties of sesame protein isolate based edible films. Food Hydrocoll. 2018, 85, 136–143. [Google Scholar] [CrossRef]
- Verkempinck, S.; Kyomugasho, C.; Salvia-Trujillo, L.; Denis, S.; Bourgeois, M.; Van Loey, A.; Hendrickx, M.; Grauwet, T. Emulsion stabilizing properties of citrus pectin and its interactions with conventional emulsifiers in oil-in-water emulsions. Food Hydrocoll. 2018, 85, 144–157. [Google Scholar] [CrossRef]
- Beserra, A.M.S.E.S.; Calegari, P.I.; Souza, M.D.C.; Dos Santos, R.A.N.; Lima, J.C.D.S.; Silva, R.M.; Balogun, S.O.; Martins, D.T.D.O. Gastroprotective and Ulcer-Healing Mechanisms of Ellagic Acid in Experimental Rats. J. Agric. Food Chem. 2011, 59, 6957–6965. [Google Scholar] [CrossRef] [PubMed]
- Murakami, S.; Isobe, Y.; Kijima, H.; Nagai, H.; Muramatu, M.; Otomo, S. Inhibition of Gastric H+, K (+)-ATPase and Acid Secretion by Ellagic Acid. Planta Med. 1991, 57, 305–308. [Google Scholar] [CrossRef]
- Iino, T.; Nakahara, K.; Miki, W.; Kiso, Y.; Ogawa, Y.; Kato, S.; Takeuchi, K. Less Damaging Effect of Whisky in Rat Stomachs in Comparison with Pure Ethanol. Digestion 2001, 64, 214–221. [Google Scholar] [CrossRef]
- Iino, T.; Tashima, K.; Umeda, M.; Ogawa, Y.; Takeeda, M.; Takata, K.; Takeuchi, K. Effect of ellagic acid on gastric damage induced in ischemic rat stomachs following ammonia or reperfusion. Life Sci. 2002, 70, 1139–1150. [Google Scholar] [CrossRef]
- Chung, J.-G.; Wu, L.-T.; Chang, S.H.; Lo, H.H.; Hsieh, S.E.; Li, Y.C.; Hung, C.F. Inhibitory Actions of Berberine on Growth and Arylamine N-Acetyltransferase Activity in Strains of Helicobacter Pylori from Peptic Ulcer Patients. Int. J. Toxicol. 1999, 18, 35–40. [Google Scholar] [CrossRef]
- Brzozowski, T.; Konturek, P.C.; Sliwowski, Z.; Kwiecień, S.; Drozdowicz, D.; Pawlik, M.; Mach, K.; Konturek, S.J.; Pawlik, W.W. Interaction of nonsteroidal anti-inflammatory drugs (NSAID) with Helicobacter pylori in the stomach of humans and experimental animals. J. Physiol. Pharmacol. 2006, 57, 67–79. [Google Scholar]
- Woodward, M.C.; Huff, N.K.; Garza, F.; Keowen, M.L.; Kearney, M.T.; Andrews, F.M. Effect of pectin, lecithin, and antacid feed supplements (Egusin®) on gastric ulcer scores, gastric fluid pH and blood gas values in horses. BMC Veter. Res. 2014, 10, S4. [Google Scholar] [CrossRef] [PubMed]
- Murray, M.J.; Grady, T.C. The effect of a pectin-lecithin complex on prevention of gastric mucosal lesions induced by feed deprivation in ponies. Equine Veter. J. 2002, 34, 195–198. [Google Scholar] [CrossRef] [PubMed]
- Kirchner, T.; Aparicio, B.; Argentieri, D.; Lau, C.; Ritchie, D. Effects of tepoxalin, a dual inhibitor of cyclooxygenase/5-lipoxygenase, on events associated with NSAID-induced gastrointestinal inflammation. Prostaglandins, Leukot. Essent. Fat. Acids 1997, 56, 417–423. [Google Scholar] [CrossRef]
- Rainsford, K.D. Gastric ulcerogenicity of non-steroidal anti-inflammatory drugs in mice with mucosa sensitized by cholinomimetic treatment. J. Pharm. Pharmacol. 1987, 39, 669–670. [Google Scholar] [CrossRef]


| Independent Variables | Levels | |
|---|---|---|
| (−1) | (+1) | |
| X1: EA% | 1.00 | 3.00 |
| X2: Ca pectinate % | 1.00 | 3.00 |
| X3: dropping time (min) | 10.00 | 30.00 |
| Responses | Desirability Constraints | |
| Y1: % EE (%) | Maximize | |
| Y2: cumulative % EA released | Maximize | |
| Experimental Run | Independent Variables | EE% ± SD | Cum. % EA Released ± SD | ||
|---|---|---|---|---|---|
| EA% | Ca Pectinate % | Dropping Time (Min) | |||
| F-1 | 3.00 | 1.00 | 30.00 | 88.1 ± 2.31 | 53.8 ± 1.12 |
| F-2 | 3.00 | 1.00 | 10.00 | 82.8 ± 1.89 | 54.6 ± 1.34 |
| F-3 | 3.00 | 3.00 | 30.00 | 85.6 ± 3.21 | 69.4 ± 2.76 |
| F-4 | 1.00 | 1.00 | 10.00 | 74.7 ± 1.45 | 79.8 ± 1.99 |
| F-5 | 1.00 | 3.00 | 30.00 | 79.6 ± 2.38 | 88.6 ± 3.22 |
| F-6 | 3.00 | 3.00 | 10.00 | 81.6 ± 3.11 | 63.8 ± 2.39 |
| F-7 | 1.00 | 1.00 | 30.00 | 77.8 ± 1.98 | 78.8 ± 1.76 |
| F-8 | 1.00 | 3.00 | 10.00 | 76.1 ± 2.26 | 85.7 ± 2.67 |
| Responses | R2 | Adjusted R2 | Predicted R2 | Adequate Precision | Significant Factors |
|---|---|---|---|---|---|
| Y1: EE% | 0.9514 | 0.9149 | 0.8056 | 12.09 | X1, X3 |
| Y2: Cum. EA% released | 0.9801 | 0.9652 | 0.9204 | 19.42 | X1, X2 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alhakamy, N.A.; Badr-Eldin, S.M.; Ahmed, O.A.A.; Halwani, A.A.; Aldawsari, H.M.; El-Moselhy, M.A.; Anter, A.; Sharkawi, S.S.; Sultan, M.H.; Madkhali, O.A.A.; et al. RETRACTED: Optimized Ellagic Acid–Ca Pectinate Floating Beads for Gastroprotection against Indomethacin-Induced Gastric Injury in Rats. Biomolecules 2020, 10, 1006. https://doi.org/10.3390/biom10071006
Alhakamy NA, Badr-Eldin SM, Ahmed OAA, Halwani AA, Aldawsari HM, El-Moselhy MA, Anter A, Sharkawi SS, Sultan MH, Madkhali OAA, et al. RETRACTED: Optimized Ellagic Acid–Ca Pectinate Floating Beads for Gastroprotection against Indomethacin-Induced Gastric Injury in Rats. Biomolecules. 2020; 10(7):1006. https://doi.org/10.3390/biom10071006
Chicago/Turabian StyleAlhakamy, Nabil A., Shaimaa M. Badr-Eldin, Osama A. A. Ahmed, Abdulrahman A. Halwani, Hibah M. Aldawsari, Mohamed A. El-Moselhy, Aliaa Anter, Sara S. Sharkawi, Muhammad H. Sultan, Osama A. A. Madkhali, and et al. 2020. "RETRACTED: Optimized Ellagic Acid–Ca Pectinate Floating Beads for Gastroprotection against Indomethacin-Induced Gastric Injury in Rats" Biomolecules 10, no. 7: 1006. https://doi.org/10.3390/biom10071006
APA StyleAlhakamy, N. A., Badr-Eldin, S. M., Ahmed, O. A. A., Halwani, A. A., Aldawsari, H. M., El-Moselhy, M. A., Anter, A., Sharkawi, S. S., Sultan, M. H., Madkhali, O. A. A., Bakhrebah, M. A., Alomary, M. N., Abdulaal, W. H., & A. Fahmy, U. (2020). RETRACTED: Optimized Ellagic Acid–Ca Pectinate Floating Beads for Gastroprotection against Indomethacin-Induced Gastric Injury in Rats. Biomolecules, 10(7), 1006. https://doi.org/10.3390/biom10071006





