Zoopharmacology: A Way to Discover New Cancer Treatments
Abstract
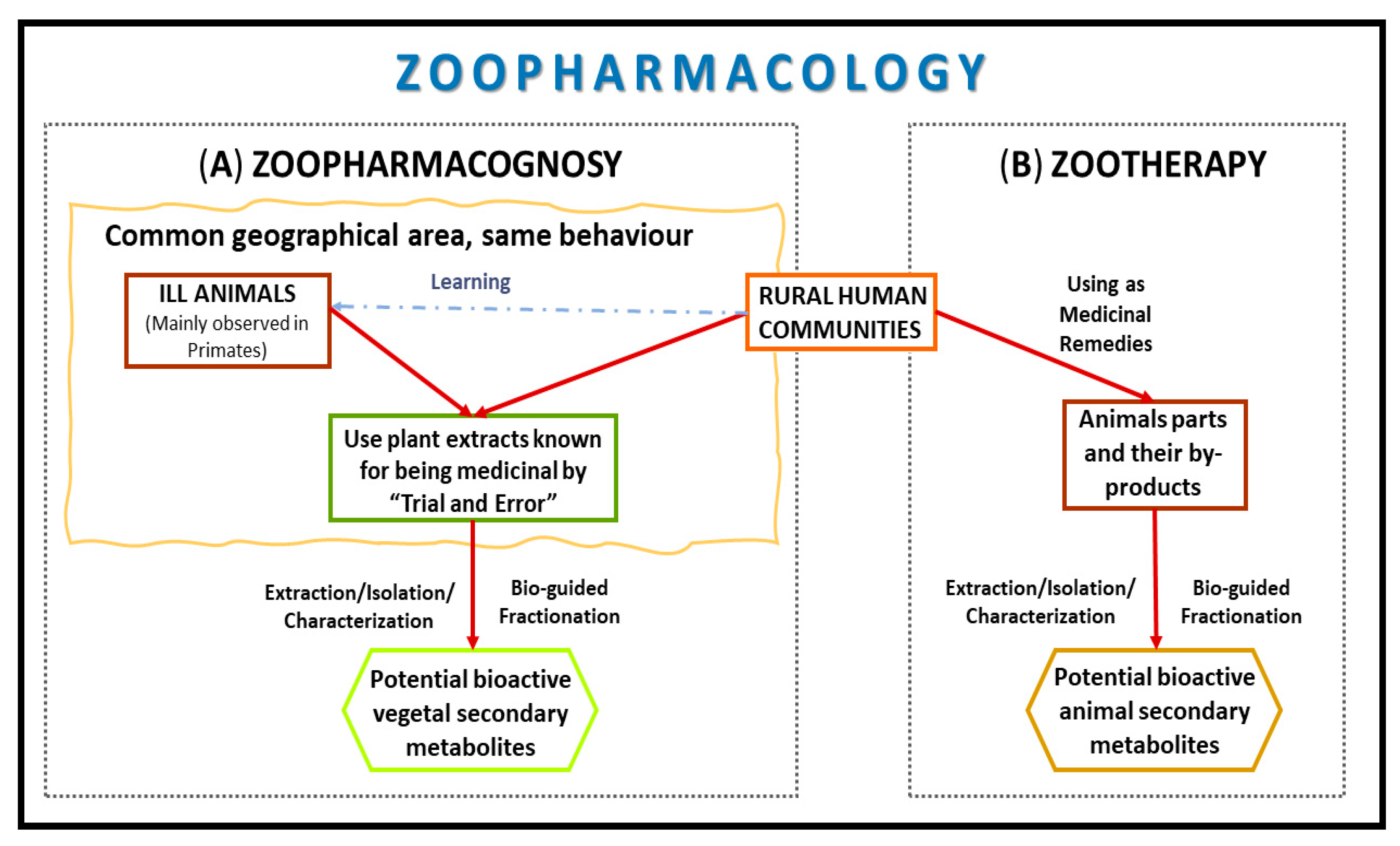
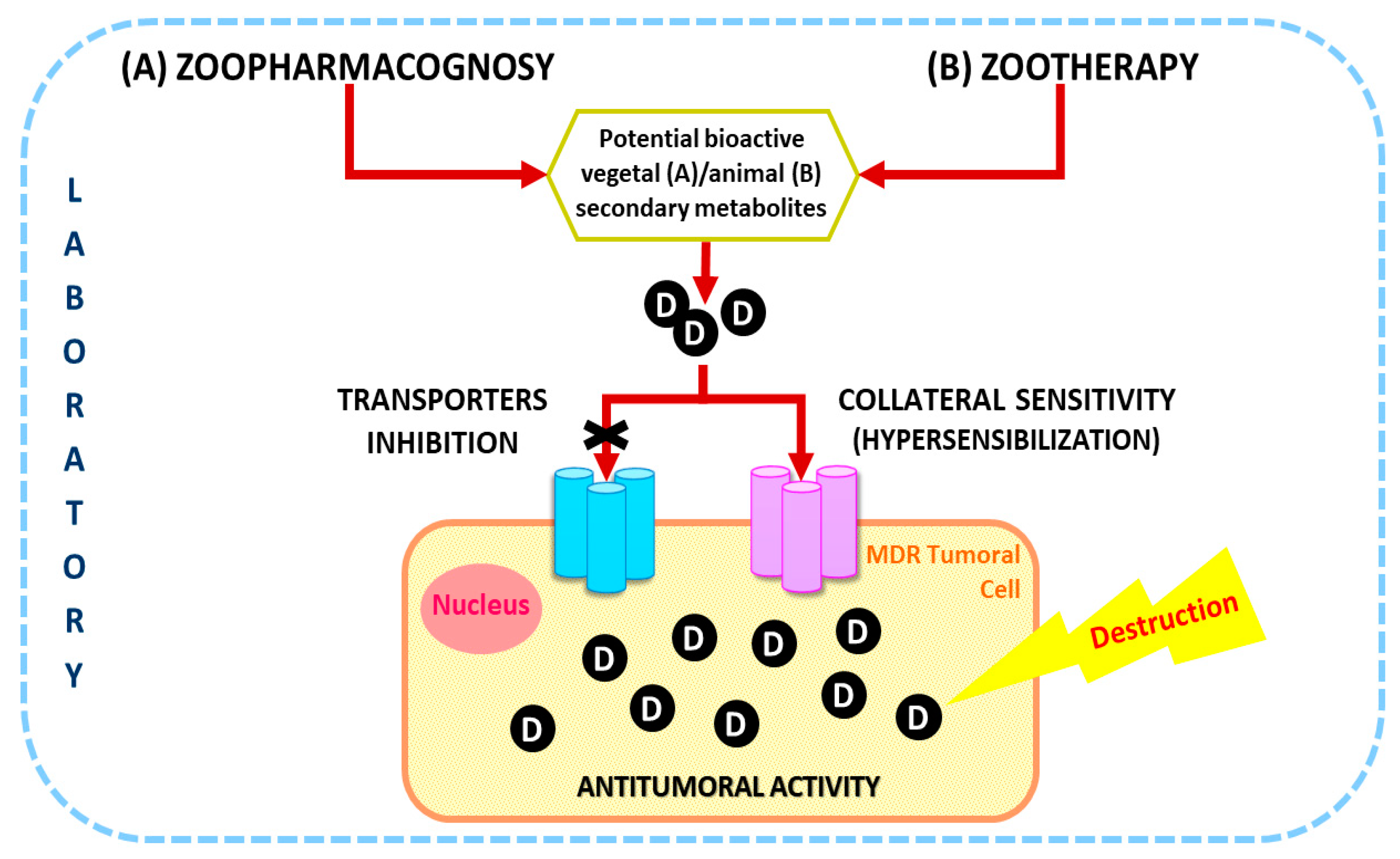
1. Introduction
2. Zoopharmacognosy
- The substance in question must be deliberately contacted.
- The substance must be detrimental, in this case, to tumoral cells.
- The detrimental effect on cancer cells must lead to increased host fitness.
- The substance must have a detrimental effect on the host in the absence of tumoral cells [18].
3. Zootherapy
| Group of Animals | Order | Family | Representative Species | Bioactive Compounds Found in Venoms | Ref. |
|---|---|---|---|---|---|
| Amphibians | Anura | Bufonidae | Rhinella schneideri, R. marina | Different types of steroids (bufadienolides), amines, proteins, peptides, alkaloids | [71,82,83,84] |
| Bufo gargizans Cantor, B. melasnosticus | |||||
| Leptodactylidae | Physalaemus nattereri | ||||
| Snakes | Squamata | Elapidae | Naja naja among others | MPP, disintegrins, L-amino acid oxidase, C-type lecitins, polypeptides, phospholipase A2, acetyl cholinesterases, serine proteases | [85,86] |
| Crotalidae | Crotalus spp., Bothrops spp. | ||||
| Viperidae | Macrovipera spp. | ||||
| Mammals | Eulipotyphla | Soricidae | Blarina brevicauda | Soridin | [92,93] |
| Marine (Jellyfish) | Semaeostomeae | Pelagiidae | Chrysaora quinquecirrha | SNV peptide | [59] |
| Insects | Scorpiones | Buthidae | Leiurus quinquestriatus quinquestriatus | Chlorotoxin | [59,87] |
| Coleoptera | Meloidae | Epicauta hirticornis, Mylabris cichorii, M. phalerata | Cantharidin | [89,90] | |
| Araneae | Theraphosidae | Brachypelma albopilosum | Brachynin | ||
| Acanthoscurria gomesiana | Gomesin | ||||
| Lycosidae | Lycosa carolinensis | Lycosin-I | |||
| Hymenoptera | Apidae | Apis mellifera | Melittin | [94,95,96] | |
| Vespidae | Polybia paulista | Mastoparan | |||
| Lepidoptera | Saturniidae | Hyalophora cecropia | Cecropin | [96] |
4. Future Perspectives
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Weinberg, R.A. The Biology of Cancer, 2nd ed.; Garland Science: New York, NY, USA, 2014. [Google Scholar]
- Domínguez-Martin, E.M.; Díaz-Lanza, A.M.; Faustino, C.M.C. Anticancer hybrid combinations: Mechanisms of action, implications and future perspectives. Curr. Pharm. Des. 2018, 24, 4312–4333. [Google Scholar] [CrossRef]
- Garcia-Mayea, Y.; Mir, C.; Masson, F.; Paciucci, R.; LLeonart, M.E. Insights into new mechanisms and models of cancer stem cell multidrug resistance. Semin. Cancer Biol. 2020, 60, 166–180. [Google Scholar] [CrossRef] [PubMed]
- Stewart, B.W.; Wild, C.P. World Cancer Report 2014; International Agency for Research on Cancer: Lyon, France, 2014.
- Ferreira, R.J.; dos Santos, D.J.; Ferreira, M.J. P-glycoprotein and membrane roles in multidrug resistance. Future Med. Chem. 2015, 7, 929–946. [Google Scholar] [CrossRef] [PubMed]
- Reis, M.A.; Matos, A.M.; Duarte, N.; Ahmed, O.B.; Ferreira, R.J.; Lage, H.; Ferreira, M. Epoxylathyrane Derivatives as MDR Selective Compounds for Disabling Multidrug Resistance in Cancer. Front. Pharmacol. 2020, 11, 599. [Google Scholar] [CrossRef]
- Chaachouay, H.; Ohneseit, P.; Toulany, M.; Kehlbach, R.; Multhoff, G.; Rodemann, H.P. Autophagy contributes to resistance of tumor cells to ionizing radiation. Radiother. Oncol. 2011, 99, 287–292. [Google Scholar] [CrossRef] [PubMed]
- Efferth, T.; Saeed, M.E.M.; Kadioglu, O.; Seo, E.J.; Shirooie, S.; Mbaveng, A.T.; Nabavi, S.M.; Kuete, V. Collateral sensitivity of natural products in drug-resistant cancer cells. Biotechnol. Adv. 2020, 38, 107342. [Google Scholar] [CrossRef]
- Cragg, G.M.; Newman, D.J. Natural products as sources of anticancer agents: Current approaches and perspectives. In Natural Products as Source of Molecules with Therapeutic Potential; Filho, V.C., Ed.; Springer International Publishing: Cham, Switzerland, 2018; pp. 309–331. [Google Scholar]
- Schmidt, B.; Ribnicky, D.M.; Poulev, A.; Logendra, S.; Cefalu, W.T.; Raskin, I. A natural history of botanical therapeutics. Metabolism 2008, 57, S3–S9. [Google Scholar] [CrossRef]
- Dhami, N. Trends in Pharmacognosy: A modern science of natural medicines. J. Herb. Med. 2013, 3, 123–131. [Google Scholar] [CrossRef]
- Heinrich, M. Ethnopharmacology: Quo vadis? Challenges for the future. Rev. Bras. Farm. 2014, 24, 99–102. [Google Scholar] [CrossRef][Green Version]
- Tasdemir, D.; MacIntosh, A.J.J.; Stergiou, P.; Kaiser, M.; Mansour, N.R.; Bickle, Q.; Huffman, M.A. Antiprotozoal and antihelminthic properties of plants ingested by wild Japanese macaques (Macaca fuscata yakui) in Yakushima Island. J. Ethnopharmacol. 2020, 247, 112270. [Google Scholar] [CrossRef]
- Huffman, M.A. Animal origins of herbal medicine. In Des Sources du Savoir aux Médicaments du Futur—From the Sources of Knowledge to the Medicines of the Future; Fleurentin, J., Pelt, J.M., Mazars, G., Eds.; Institut de Recherche pour le Développement (IRD): Marseille, France, 2002; pp. 31–42. [Google Scholar]
- Huffman, M.A. Animal self-medication and ethno-medicine: Exploration and exploitation of the medicinal properties of plants. Proc. Nutr. Soc. 2003, 62, 371–381. [Google Scholar] [CrossRef] [PubMed]
- Costa-Neto, E.M. Zoopharmacognosy, the self-medication behavior of animals. Interfaces Cient. Saúde Ambiente 2012, 1, 61–72. [Google Scholar] [CrossRef][Green Version]
- Mezcua-Martín, A.; Revuelta-Rueda, L.; Sánchez de Lollano-Prieto, J. The origins of zoopharmacognosy: How humans learned about self-medication from animals. Int. J. Appl. Res. 2019, 5, 73–79. [Google Scholar]
- Abbott, J. Self-medication in insects: Current evidence and future perspectives. Ecol. Entomol. 2014, 39, 273–280. [Google Scholar] [CrossRef]
- Huffman, M.A. Primate self-medication, passive prevention and active treatment—A brief review. IJMS 2016, 3, 1–10. [Google Scholar] [CrossRef]
- Lozano, G.A. Parasitic stress and self-medication in wild animals. Adv. Study Behav. 1998, 27, 291–317. [Google Scholar]
- Krief, S. Do animals use natural properties of plants to self-medicate? In Applied Equine Nutrition and Training; Wageningen Academic Publishers: Wageningen, The Netherlands, 2012; pp. 159–170. [Google Scholar]
- Villalba, J.J.; Miller, J.; Ungar, E.D.; Landau, S.Y.; Glendinning, J. Ruminant self-medication against gastrointestinal nematodes: Evidence, mechanism, and origins. Parasite 2014, 21, 31. [Google Scholar] [CrossRef] [PubMed]
- Lisonbee, L.D.; Villalba, J.J.; Provenza, F.D.; Hall, J.O. Tannins and self-medication: Implications for sustainable parasite control in herbivores. Behav. Process. 2009, 82, 184–189. [Google Scholar] [CrossRef] [PubMed]
- Johnson, W.; Tchounwou, P.B.; Yedjou, C.G. Therapeutic mechanisms of Vernonia amygdalina delile in the treatment of prostate cancer. Molecules 2017, 22, 1594. [Google Scholar] [CrossRef] [PubMed]
- Rohin, M.; Ridzwan, N.; Jumli, M.N.; Hadi, N.A.; Johari, S.; Latif, A. Cytotoxicity study and morphological changes of different extraction for Bismillah leaf (Vernonia amygdalina) in human glioblastoma multiforme cell line (U-87). Biomed Res. 2017, 28, 1472–1478. [Google Scholar]
- Wong, F.C.; Woo, C.C.; Hsu, A.; Tan, B.K. The anti-cancer activities of Vernonia amygdalina extract in human breast cancer cell lines are mediated through caspase-dependent and p53-independent pathways. PLoS ONE 2013, 8, e78021. [Google Scholar] [CrossRef] [PubMed]
- Toyang, N.J.; Verpoorte, R. A review of the medicinal potentials of plants of the genus Vernonia (Asteraceae). J. Ethnopharmacol. 2013, 146, 681–723. [Google Scholar] [CrossRef] [PubMed]
- Tambour, M. La Zoopharmacognosie ou L’étude de L’automédication chez les Animaux: Voie D’avenir Pour de Nouveaux Médicaments. Ph.D. Thesis, Université de Lille, Lille, France, 2016. (In French). [Google Scholar]
- Omosa, L.K.; Midiwo, J.O.; Masila, V.M.; Gisacho, B.M.; Munayi, R.; Kamakama, F.; Chemutai, K.P.; Elhaboob, G.; Saeed, M.E.; Hamdoun, S.; et al. Cytotoxicity of 91 Kenyan indigenous medicinal plants towards human CCRF-CEM leukemia cells. J. Ethnopharmacol. 2016, 179, 177–196. [Google Scholar] [CrossRef] [PubMed]
- Singh, G.; Passsari, A.K.; Leo, V.V.; Mishra, V.K.; Subbarayan, S.; Singh, B.P.; Kumar, B.; Kumar, S.; Gupta, V.K.; Lalhlenmawia, H.; et al. Evaluation of phenolic content variability along with antioxidant, antimicrobial, and cytotoxic potential of selected traditional medicinal plants from india. Front. Plant Sci. 2016, 7, 407. [Google Scholar] [CrossRef] [PubMed]
- Desai, T.H.; Joshi, S.V. Anticancer activity of saponin isolated from Albizia lebbeck using various in vitro models. J. Ethnopharmacol. 2019, 231, 494–502. [Google Scholar] [CrossRef] [PubMed]
- Simo, L.M.; Noté, O.P.; Mbing, J.N.; Aouazou, S.A.; Guillaume, D.; Muller, C.D.; Pegnyemb, D.E.; Lobstein, A. New cytotoxic triterpenoid saponins from the roots of Albizia gummifera (J.F.Gmel.) C.A.Sm. Chem. Biodivers. 2017, 14. [Google Scholar] [CrossRef] [PubMed]
- Noté, O.P.; Kamto, E.L.D.; Toukea, D.D.; Aouazou, S.A.; Mbing, J.N.; Muller, C.D.; Guillaume, D.; Pegnyemb, D.E. Pro-apoptotic activity of new triterpenoid saponins from the roots of Albizia adianthifolia (Schumach.) W.Wight. Fitoterapia 2018, 129, 34–41. [Google Scholar]
- Noté, O.P.; Jihu, D.; Antheaume, C.; Zeniou, M.; Pegnyemb, D.E.; Guillaume, D.; Chneiwess, H.; Kilhoffer, M.C.; Lobstein, A. Triterpenoid saponins from Albizia lebbeck (L.) Benth and their inhibitory effect on the survival of high grade human brain tumor cells. Carbohydr. Res. 2015, 404, 26–33. [Google Scholar]
- Noté, O.P.; Simo, L.; Mbing, J.N.; Guillaume, D.; Aouazou, S.A.; Muller, C.D.; Pegnyemb, D.E.; Lobstein, A. Two new triterpenoid saponins from the roots of Albizia zygia (DC.) J.F. Macbr. Phytochem. Lett. 2016, 18, 128–135. [Google Scholar]
- Han, Q.; Qian, Y.; Wang, X.; Zhang, Q.; Cui, J.; Tu, P.; Liang, H. Cytotoxic oleanane triterpenoid saponins from Albizia julibrissin. Fitoterapia 2017, 121, 183–193. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Akihisa, T.; Kurita, M.; Kikuchi, T.; Zhu, W.F.; Ye, F.; Dong, Z.H.; Liu, W.Y.; Feng, F.; Xu, J. Melanogenesis-inhibitory and cytotoxic activities of triterpene glycoside constituents from the bark of Albizia procera. J. Nat. Prod. 2018, 81, 2612–2620. [Google Scholar] [CrossRef] [PubMed]
- Lacaille-Dubois, M.A.; Pegnyemb, D.E.; Note, O.P.; Mitaine-Offer, A.C. A review of acacic acid-type saponins from Leguminosae-Mimosoideae as potent cytotoxic and apoptosis inducing agents. Phytochem. Rev. 2011, 10, 565–584. [Google Scholar] [CrossRef]
- Fotso, G.W.; Kamga, J.; Ngameni, B.; Uesugi, S.; Ohno, M.; Kimura, K.I.; Momma, H.; Kwon, E.; Furuno, H.; Shiono, Y.; et al. Secondary metabolites with antiproliferative effects from Albizia glaberrima var glabrescens Oliv. (Mimosoideae). Nat. Prod. Res. 2017, 31, 1981–1987. [Google Scholar] [CrossRef] [PubMed]
- Kuete, V.; Tchinda, C.F.; Mambe, F.T.; Beng, V.P.; Efferth, T. Cytotoxicity of methanol extracts of 10 Cameroonian medicinal plants towards multi-factorial drug-resistant cancer cell lines. BMC Complement. Altern. Med. 2016, 16, 267. [Google Scholar] [CrossRef] [PubMed]
- Petroni, L.M.; Huffman, M.A.; Rodrigues, E. Medicinal plants in the diet of woolly spider monkeys (Brachyteles arachnoides, E. Geoffroy, 1806)—A bio-rational for the search of new medicines for human use? Rev. Bras. Farm. 2017, 27, 135–142. [Google Scholar] [CrossRef]
- Dubost, J.; Lamxay, V.; Krief, S.; Falshaw, M.; Manithip, C.; Deharo, E. From plant selection by elephants to human and veterinary pharmacopeia of mahouts in Laos. J. Ethnopharmacol. 2019, 244, 112157. [Google Scholar] [CrossRef] [PubMed]
- Krief, S.; Hladik, C.M.; Haxaire, C. Ethnomedicinal and bioactive properties of plants ingested by wild chimpanzees in Uganda. J. Ethnopharmacol. 2005, 101, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Omara, T.; Kiprop, A.K.; Ramkat, R.C.; Cherutoi, J.; Kagoya, S.; Moraa Nyangena, D.; Azeze Tebo, T.; Nteziyaremye, P.; Nyambura Karanja, L.; Jepchirchir, A.; et al. Medicinal plants used in traditional management of cancer in Uganda: A review of ethnobotanical surveys, phytochemistry, and anticancer studies. Evid. Based Complement. Altern. Med. 2020, 2020, 3529081. [Google Scholar] [CrossRef]
- Ochwang’i, D.O.; Kimwele, C.N.; Oduma, J.A.; Gathumbi, P.K.; Mbaria, J.M.; Kiama, S.G. Medicinal plants used in treatment and management of cancer in Kakamega County, Kenya. J. Ethnopharmacol. 2014, 151, 1040–1055. [Google Scholar] [CrossRef]
- Farombi, E.O.; Owoeye, O. Antioxidative and chemopreventive properties of Vernonia amygdalina and Garcinia biflavonoid. Int. J. Environ. Res. Public Health 2011, 8, 2533–2555. [Google Scholar] [CrossRef]
- Rodríguez, E.; Richard, W. Zoopharmacognosy: The use of medicinal plants by animals. In Phytochemical Potential of Tropical Plants, 1st ed.; Downum, K.R., Romeo, J.T., Stafford, H.A., Eds.; Springer: New York, NY, USA, 1993; pp. 89–105. [Google Scholar]
- Messner, E.J.; Wrangham, R.W. In vitro testing of the biological activity of Rubia cordifolia leaves on primate strongyloides species. Primates 1996, 37, 105–108. [Google Scholar] [CrossRef]
- Xu, K.; Wang, P.; Yuan, B.; Cheng, Y.; Li, Q.; Lei, H. Structural and bioactive studies of terpenes and cyclopeptides from the Genus Rubia. Chem. Cent. J. 2013, 7, 81. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wang, Z.; Li, M.Y.; Mi, C.; Wang, K.S.; Ma, J.; Jin, X. Mollugin has an anti-cancer therapeutic effect by inhibiting TNF-alpha-induced NF-kappaB activation. Int. J. Mol. Sci. 2017, 18, 1619. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Wang, H.; Zhu, J.; Xu, J.; Ding, K. Mollugin induces tumor cell apoptosis and autophagy via the PI3K/AKT/mTOR/p70S6K and ERK signaling pathways. Biochem. Biophys. Res. Commun. 2014, 450, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.M.; Park, H.S.; Jun, D.Y.; Woo, H.J.; Woo, M.H.; Yang, C.H.; Kim, Y.H. Mollugin induces apoptosis in human Jurkat T cells through endoplasmic reticulum stress-mediated activation of JNK and caspase-12 and subsequent activation of mitochondria-dependent caspase cascade regulated by Bcl-xL. Toxicol. Appl. Pharmacol. 2009, 241, 210–220. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.Q.; Zhao, S.M.; Wang, Z.; Zeng, G.Z.; Huang, M.B.; Tan, N.H. Rubicordins A-C, new cyclopeptides from Rubia cordifolia with cytotoxicity and inhibiting NF-kB signaling pathway. Tetrahedron 2015, 71, 9673–9678. [Google Scholar] [CrossRef]
- Lee, Y.M.; Auh, Q.S.; Lee, D.W.; Kim, J.Y.; Jung, H.J.; Lee, S.H.; Kim, E.C. Involvement of Nrf2-mediated upregulation of heme oxygenase-1 in mollugin-induced growth inhibition and apoptosis in human oral cancer cells. BioMed Res. Int. 2013, 2013, 210604. [Google Scholar] [CrossRef]
- Wang, J.; Li, L.; Wang, J.; Song, L.; Tan, N.; Wang, Z. Natural naphthohydroquinone dimer rubioncolin C exerts anti-tumor activity by inducing apoptotic and autophagic cell death and inhibiting the NF-kappaB and Akt/mTOR/P70S6K pathway in human cancer cells. Cells 2019, 8, 1593. [Google Scholar] [CrossRef]
- van Asseldonk, T.; Kleijer, G.; Lans, C. Ethnoveterinary herb use in the Netherlands-between ethnobotany and zoopharmacognosy. In Proceedings of the Conference: BASEL VET Preconference GA, Basel, Switzerland, 3 September 2017; pp. 1–6. [Google Scholar]
- Saad, B.; Said, O. Natural drugs in greco-arabic and Islamic medicine. In Greco-Arab and Islamic Herbal Medicine; Saad, B., Said, O., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2011; pp. 101–121. [Google Scholar]
- Alves, R.R.N.; Rosa, I.L. Why study the use of animal products in traditional medicines? J. Ethnobiol. Ethnomed. 2005, 1, 5. [Google Scholar] [CrossRef]
- Kaufmann, A.; Greten, H.J.; Efferth, T. Animals and animal toxins in traditional and modern medicine. Br. J. Appl. Sci. Technol. 2014, 4, 578–593. [Google Scholar] [CrossRef]
- Borah, M.P.; Prasad, S.B. Ethnozoological study of animals based medicine used by traditional healers and indigenous inhabitants in the adjoining areas of Gibbon Wildlife Sanctuary, Assam, India. J. Ethnobiol. Ethnomed. 2017, 13, 39. [Google Scholar] [CrossRef] [PubMed]
- Dhakal, P.; Chettri, B.; Lepcha, S.; Acharya, B.K. Rich yet undocumented ethnozoological practices of socio-culturally diverse indigenous communities of Sikkim Himalaya, India. J. Ethnopharmacol. 2020, 249, 112386. [Google Scholar] [CrossRef] [PubMed]
- Das, K.S.; Choudhury, S.; Nonglait, K. Zootherapy among the ethnic groups of North Eastern Region of India-A Critical Review. J. Crit. Rev. 2017, 4, 1–9. [Google Scholar] [CrossRef]
- Alves, R.R.; Alves, H.N. The faunal drugstore: Animal-based remedies used in traditional medicines in Latin America. J. Ethnobiol. Ethnomed. 2011, 7, 9. [Google Scholar] [CrossRef] [PubMed]
- Solís, L.; Casas, A. Cuicatec ethnozoology: Traditional knowledge, use, and management of fauna by people of San Lorenzo Pápalo, Oaxaca, Mexico. J. Ethnobiol. Ethnomed. 2019, 15, 58. [Google Scholar] [CrossRef] [PubMed]
- Mahomoodally, F.; Samoisy, A.K.; Suroowan, S. Ethnozoological practices in Rodrigues Island of the Mascarene archipelago. J. Ethnopharmacol. 2019, 245, 112163. [Google Scholar] [CrossRef] [PubMed]
- Khusro, A.; Aarti, C.; Barbabosa-Pliego, A.; Rivas-Cáceres, R.R.; Cipriano-Salazar, M. Venom as therapeutic weapon to combat dreadful diseases of 21st century: A systematic review on cancer, TB, and HIV/AIDS. Microb. Pathog. 2018, 125, 96–107. [Google Scholar] [CrossRef]
- Jang, A.; Jo, C.; Kang, K.; Lee, M. Antimicrobial and human cancer cell cytotoxic effect of synthetic angiotensin-converting enzyme (ACE) inhibitory peptides. Food Chem. 2008, 107, 327–336. [Google Scholar] [CrossRef]
- Yu, Z.J.; Xu, Y.; Peng, W.; Liu, Y.J.; Zhang, J.M.; Li, J.S.; Sun, T.; Wang, P. Calculus bovis: A review on traditional usages, origin, chemistry, pharmacological activities and toxicology. J. Ethnopharmacol. 2020, 254, 112649. [Google Scholar] [CrossRef]
- Wang, L.; Dong, C.; Li, X.; Han, W.; Su, X. Anticancer potential of bioactive peptides from animal sources (Review). Oncol. Rep. 2017, 38, 637–651. [Google Scholar] [CrossRef]
- Sánchez-Pedraza, R.; Gamba-Rincón, M.R.; González-Rangel, A.L. Use of black vulture (Coragyps atratus) in complementary and alternative therapies for cancer in Colombia: A qualitative study. J. Ethnobiol. Ethnomed. 2012, 8, 20. [Google Scholar] [CrossRef] [PubMed]
- Jeyamogan, S.; Khan, N.A.; Siddiqui, R. Animals living in polluted environments are a potential source of anti-tumor molecule(s). Cancer Chemother. Pharmacol. 2017, 80, 919–924. [Google Scholar] [CrossRef] [PubMed]
- Song, W.; Shen, D.Y.; Kang, J.H.; Li, S.S.; Zhan, H.W.; Shi, Y.; Xiong, Y.X.; Liang, G.; Chen, Q.X. Apoptosis of human cholangiocarcinoma cells induced by ESC-3 from Crocodylus siamensis bile. World J. Gastroenterol. 2012, 18, 704–711. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Chen, D.; Xie, G.R. Effects of Gekko sulfated polysaccharide on the proliferation and differentiation of hepatic cancer cell line. Cell Biol. Int. 2006, 30, 659–664. [Google Scholar] [CrossRef] [PubMed]
- Ding, X.L.; Man, Y.N.; Hao, J.; Zhu, C.H.; Liu, C.; Yang, X.; Wu, X.Z. The antitumor effect of gekko sulfated glycopeptide by inhibiting bFGF-induced lymphangiogenesis. BioMed Res. Int. 2016, 2016, 7396392. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Mao, X.; Zhang, T.; Guo, X.; Ge, Y.; Ma, C.; Zhang, X. Separation and nanoencapsulation of antitumor peptides from Chinese three-striped box turtle (Cuora trifasciata). J. Microencapsul. 2016, 33, 344–354. [Google Scholar] [CrossRef]
- El Ouar, I.; Braicu, C.; Naimi, D.; Irimie, A.; Berindan-Neagoe, I. Effect of Helix aspersa extract on TNFα, NF-κB and some tumor suppressor genes in breast cancer cell line Hs578T. Pharmacol. Mag. 2017, 13, 281–285. [Google Scholar] [CrossRef]
- Ruiz-Torres, V.; Encinar, J.A.; Herranz-Lopez, M.; Perez-Sanchez, A.; Galiano, V.; Barrajon-Catalan, E.; Micol, V. An updated review on marine anticancer compounds: The use of virtual screening for the discovery of small-molecule cancer drugs. Molecules 2017, 22, 1037. [Google Scholar] [CrossRef]
- Newman, D.J. From natural products to drugs. Phys. Sci. Rev. 2018, 4, 20180111. [Google Scholar] [CrossRef]
- Abraham, I.; El Sayed, K.; Chen, Z.S.; Guo, H. Current status on marine products with reversal effect on cancer multidrug resistance. Mar. Drugs 2012, 10, 2312–2321. [Google Scholar] [CrossRef]
- Lopez, D.; Martinez-Luis, S. Marine natural products with P-glycoprotein inhibitor properties. Mar. Drugs 2014, 12, 525–546. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.C.; Yang, H.; Zhang, L.L.; Zhang, Q.; Chen, B.; Wang, Y. Biotoxins for cancer therapy. Asian Pac. J. Cancer Prev. 2014, 15, 4753–4758. [Google Scholar] [CrossRef] [PubMed]
- Abdelfatah, S.; Lu, X.; Schmeda-Hirschmann, G.; Efferth, T. Cytotoxicity and antimitotic activity of Rhinella schneideri and Rhinella marina venoms. J. Ethnopharmacol. 2019, 242, 112049. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.L.; Hou, J.J.; Wang, X.; Yu, Y.; Li, H.J.; Li, Z.W.; Feng, Z.J.; Qu, H.; Wu, W.Y.; Guo, D.A. Venenum bufonis: An overview of its traditional use, natural product chemistry, pharmacology, pharmacokinetics and toxicology. J. Ethnopharmacol. 2019, 237, 215–235. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Zhang, Q.; Zou, G.; Gao, G.; Yue, Q. Arenobufagin, isolated from toad venom, inhibited epithelial-to-mesenchymal transition and suppressed migration and invasion of lung cancer cells via targeting IKKβ/NFκβ signal cascade. J. Ethnopharmacol. 2020, 250, 112492. [Google Scholar] [CrossRef] [PubMed]
- Calderon, L.A.; Sobrinho, J.C.; Zaqueo, K.D.; De Moura, A.A.; Grabner, A.N.; Mazzi, M.V.; Marcussi, S.; Nomizo, A.; Fernandes, C.F.C.; Zuliani, J.P.; et al. Antitumoral activity of snake venom proteins: New trends in cancer therapy. BioMed Res. Int. 2014, 2014, 203639. [Google Scholar] [CrossRef]
- Urra, F.A.; Araya-Maturana, R. Targeting metastasis with snake toxins: Molecular mechanisms. Toxins (Basel) 2017, 9, 390. [Google Scholar] [CrossRef]
- Cohen-Inbar, O.; Zaaroor, M. Glioblastoma multiforme targeted therapy: The Chlorotoxin story. J. Clin. Neurosci. 2016, 33, 52–58. [Google Scholar] [CrossRef]
- Ding, J.; Chua, P.J.; Bay, B.H.; Gopalakrishnakone, P. Scorpion venoms as a potential source of novel cancer therapeutic compounds. Exp. Biol. Med. (Maywood) 2014, 239, 387–393. [Google Scholar] [CrossRef]
- Verma, A.K.; Prasad, S.B. Antitumor effect of blister beetles: An ethno-medicinal practice in Karbi community and its experimental evaluation against a murine malignant tumor model. J. Ethnopharmacol. 2013, 148, 869–879. [Google Scholar] [CrossRef]
- Deng, Y.; Zhang, W.; Li, N.; Lei, X.; Gong, X.; Zhang, D.; Wang, L.; Ye, W. Cantharidin derivatives from the medicinal insect Mylabris phalerata. Tetrahedron 2017, 73, 5932–5939. [Google Scholar] [CrossRef]
- Akef, H.M. Anticancer, antimicrobial, and analgesic activities of spider venoms. Toxicol. Res. 2018, 7, 381–395. [Google Scholar] [CrossRef] [PubMed]
- Ma, R.; Mahadevappa, R.; Kwok, H.F. Venom-based peptide therapy: Insights into anti-cancer mechanism. Oncotarget 2017, 8, 100908–100930. [Google Scholar] [CrossRef] [PubMed]
- Balamurugan, E.; Reddy, B.V.; Menon, V.P. Antitumor and antioxidant role of Chrysaora quinquecirrha (sea nettle) nematocyst venom peptide against Ehrlich ascites carcinoma in Swiss Albino mice. Mol. Cell. Biochem. 2010, 338, 69–76. [Google Scholar] [CrossRef]
- Lyu, C.; Fang, F.; Li, B. Anti-tumor effects of melittin and its potential applications in clinic. Curr. Protein Pept. Sci. 2018, 20, 240–250. [Google Scholar] [CrossRef] [PubMed]
- Huerta-Reyes, M.; Anselme, C.; Cherqui, A.; Decocq, G. Exploration through the venoms from hymenoptera as potential therapeutic agents in cancer therapy. Int. J. Pharmacol. 2017, 13, 507–515. [Google Scholar] [CrossRef]
- El-Tantawy, N.L. Helminthes and insects: Maladies or therapies. Parasitol. Res. 2015, 114, 359–377. [Google Scholar] [CrossRef]
- Olgac, A.; Orhan, I.E.; Banoglu, E. The potential role of in silico approaches to identify novel bioactive molecules from natural resources. Future Med. Chem. 2017, 9, 1665–1686. [Google Scholar] [CrossRef]
- Rifaioglu, A.S.; Atas, H.; Martin, M.J.; Cetin-Atalay, R.; Atalay, V.; Dogan, T. Recent applications of deep learning and machine intelligence on in silico drug discovery: Methods, tools and databases. Brief Bioinform. 2019, 20, 1878–1912. [Google Scholar] [CrossRef]
- Chen, B.; Harrison, R.F.; Papadatos, G.; Willett, P.; Wood, D.J.; Lewell, X.Q.; Greenidge, P.; Stiefl, N. Evaluation of machine-learning methods for ligand-based virtual screening. J. Comput. Aided Mol. Des. 2007, 21, 53–62. [Google Scholar] [CrossRef]
- Duran-Frigola, M.; Pauls, E.; Guitart-Pla; Bertoni, M.; Alcalde, V.; Amat, D.; Juan-Blanco, T.; Aloy, P. Extending the small-molecule similarity principle to all levels of biology with the chemical checker. Nat. Biotechnol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Acevedo, C.H.; Scotti, L.; Scotti, M.T. In silico studies designed to select sesquiterpene lactones with potential antichagasic activity from an in-house Asteraceae database. ChemMedChem 2018, 13, 634–645. [Google Scholar] [CrossRef] [PubMed]
- Chagas-Paula, D.A.; Oliveira, T.B.; Zhang, T.; Edrada-Ebel, R.; Da Costa, F.B. Prediction of anti-inflammatory plants and discovery of their biomarkers by machine learning algorithms and metabolomic studies. Planta Med. 2015, 81, 450–458. [Google Scholar] [CrossRef] [PubMed]
- Blondeau, S.; Do, Q.T.; Scior, T.; Bernard, P.; Morin-Allory, L. Reverse pharmacognosy: Another way to harness the generosity of nature. Curr. Pharm. Des. 2010, 16, 1682–1696. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.; Yang, Y.; Xiong, W.; Yao, Y.; Wu, H.; Zhang, M. Network pharmacology-based research on the active component and mechanism of the antihepatoma effect of Rubia cordifolia L. J. Cell. Biochem. 2019, 120, 12461–12472. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.J.; Zhao, H.Y.; Wei, X.L.; Guo, F.F.; Wei, J.Y.; Wang, H.J.; Yang, J.; Yang, Z.G.; Si, N.; Bian, B.L. The lipid homeostasis regulation study of arenobufagin in zebrafish HepG2 xenograft model and HepG2 cells using integrated lipidomics-proteomics approach. J. Ethnopharmacol. 2020. [Google Scholar] [CrossRef]
- Sen, T.; Samanta, S.K. Medicinal plants, human health and biodiversity: A broad review. Adv. Biochem. Eng. Biotechnol. 2015, 147, 59–110. [Google Scholar]
| Animal Specie | Product | Molecule(s) | Type of Cancer | Studied Cell Line | Ref. |
|---|---|---|---|---|---|
| Bos primigenius taurus | Meat peptides | GFHI, DFHING, FHG, GLSDGEWQ | Breast | MCF-7 | [67] |
| Stomach adenocarcinoma | AGS | ||||
| Bos Taurus domesticus Gmalin | Dried gallstones | Not specified | Sarcoma | S180 | [68] |
| Breast | 4T1 | ||||
| Hepatoma | HepG2 | ||||
| Capra aegagrus hircus | Spleen, liver extracts | ACPB-3 | Gastric | BCG-823 | [69] |
| GCSC | |||||
| Colorectal | HCT-116 | ||||
| Coragyps atratus | Blood extracts | - | Hematopoietic system | - | [70] |
| Crocodylus siamensis | Bile acid extracts | ESC-3 | Cholangiocarcinoma | SK-ChA-1 | [72] |
| Mz-ChA-1 | |||||
| QBC939 | |||||
| Bile components | Human papillomavirus-related endocervical adenocarcinoma | SMM 7721 | [71] | ||
| Cholangiocarcinoma resistant to 5-Fluorouracil | QBC939/5-FU | ||||
| White blood cell extract | - | Cervical Cancer (Anti-angiogenic properties) | HeLa | ||
| Gekko swinhonis Güenter | Whole animal aqueous extracts | Gepsine | Hepatocarcinoma | Bel-702 | [73,74] |
| Colon carcinoma | HT-29 | ||||
| Anti-angiogenic properties | hLECs | ||||
| Cuora trifasciata | Enzymatic hydrolysates extracts | Fraction T1 (peptides T1-1 RGVKGPR, T1-2 KLGPKGPR), Fraction T2 SSPGPPVH | Liver | HepG2 | [75] |
| Breast | MCF-7 | ||||
| Helix aspersa | Snails aqueous extracts | Not specified | Breast | H5578T | [76] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Domínguez-Martín, E.M.; Tavares, J.; Ríjo, P.; Díaz-Lanza, A.M. Zoopharmacology: A Way to Discover New Cancer Treatments. Biomolecules 2020, 10, 817. https://doi.org/10.3390/biom10060817
Domínguez-Martín EM, Tavares J, Ríjo P, Díaz-Lanza AM. Zoopharmacology: A Way to Discover New Cancer Treatments. Biomolecules. 2020; 10(6):817. https://doi.org/10.3390/biom10060817
Chicago/Turabian StyleDomínguez-Martín, Eva María, Joana Tavares, Patrícia Ríjo, and Ana María Díaz-Lanza. 2020. "Zoopharmacology: A Way to Discover New Cancer Treatments" Biomolecules 10, no. 6: 817. https://doi.org/10.3390/biom10060817
APA StyleDomínguez-Martín, E. M., Tavares, J., Ríjo, P., & Díaz-Lanza, A. M. (2020). Zoopharmacology: A Way to Discover New Cancer Treatments. Biomolecules, 10(6), 817. https://doi.org/10.3390/biom10060817






