Polysaccharide-Based Formulations for Healing of Skin-Related Wound Infections: Lessons from Animal Models and Clinical Trials
Abstract
1. Introduction
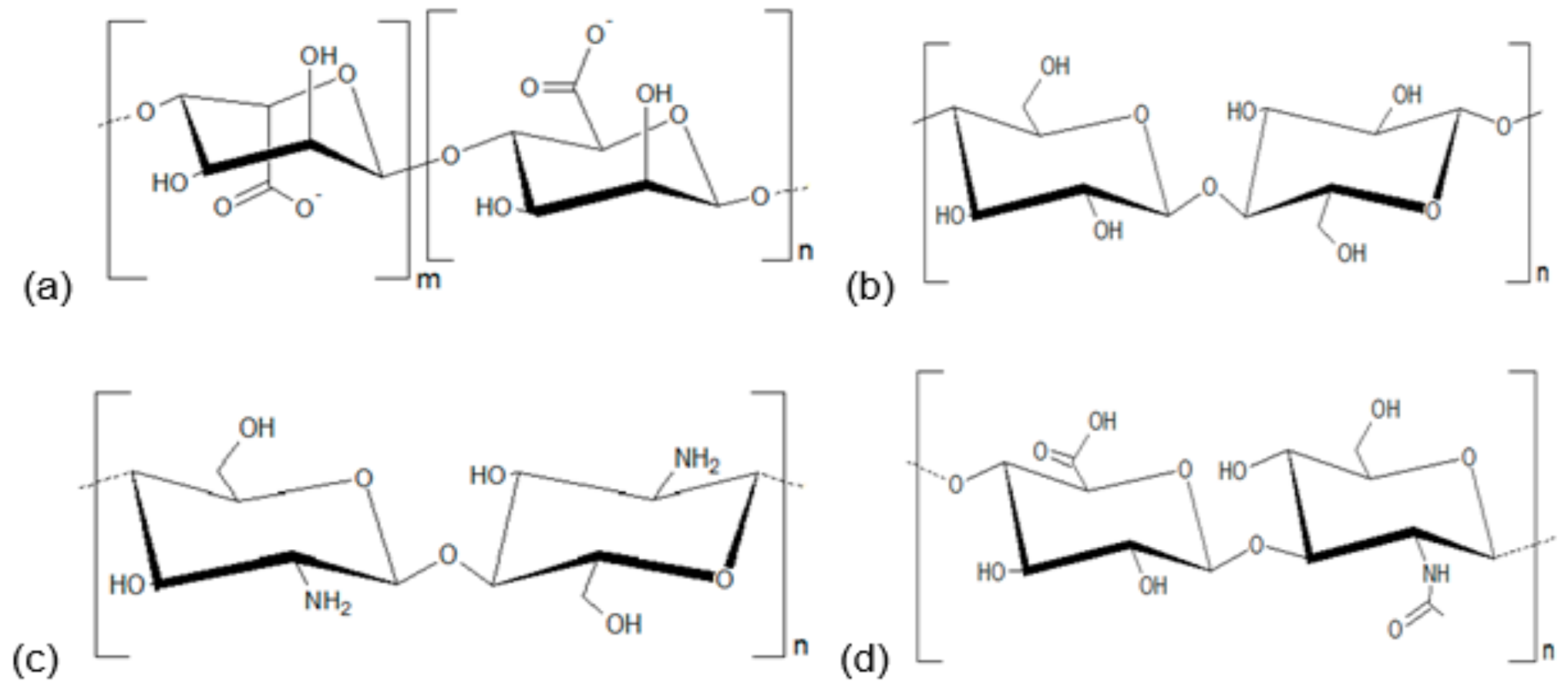
2. Polysaccharides and Development of Healing Agents
3. Alginate-Based Formulations
4. Cellulose-Based Formulations
5. Chitosan-Based Formulations
6. Hyaluronic Acid-Based Formulations
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Shaikh-Kader, A.; Houreld, N.N.; Rajendran, N.K.; Abrahamse, H. The link between advanced glycation end products and apoptosis in delayed wound healing. Cell Biochem. Funct. 2019, 37, 432–442. [Google Scholar] [CrossRef] [PubMed]
- Brazil, J.C.; Quiros, M.; Nusrat, A.; Parkos, C.A. Innate immune cell-epithelial crosstalk during wound repair. J. Clin. Investig. 2019, 129, 2983–2993. [Google Scholar] [CrossRef] [PubMed]
- Rahim, K.; Saleha, S.; Zhu, X.; Huo, L.; Basit, A.; Franco, O.L. Bacterial Contribution in Chronicity of Wounds. Microb. Ecol. 2017, 73, 710–721. [Google Scholar] [CrossRef] [PubMed]
- Omar, A.; Wright, J.B.; Schultz, G.; Burrell, R.; Nadworny, P. Microbial Biofilms and Chronic Wounds. Microorganisms 2017, 5, 9. [Google Scholar] [CrossRef]
- Cefalu, J.E.; Barrier, K.M.; Davis, A.H. Wound Infections in Critical Care. Crit. Care Nurs. Clin. N. Am. 2017, 29, 81–96. [Google Scholar] [CrossRef]
- Morton, L.M.; Phillips, T.J. Wound healing and treating wounds: Differential diagnosis and evaluation of chronic wounds. J. Am. Acad. Derm. 2016, 74, 589–605. [Google Scholar] [CrossRef]
- Chaudhary, N.A.; Munawar, M.D.; Khan, M.T.; Rehan, K.; Sadiq, A.; Tameez-Ud-Din, A.; Bhatti, H.W.; Rizvi, Z.A. Epidemiology, Bacteriological Profile, and Antibiotic Sensitivity Pattern of Burn Wounds in the Burn Unit of a Tertiary Care Hospital. Cureus 2019, 11, e4794. [Google Scholar] [CrossRef]
- Snyder, R.J.; Bohn, G.; Hanft, J.; Harkless, L.; Kim, P.; Lavery, L.; Schultz, G.; Wolcott, R. Wound Biofilm: Current Perspectives and Strategies on Biofilm Disruption and Treatments. Wounds 2017, 29, S1–S17. [Google Scholar]
- Guo, X.P.; Yang, Y.; Lu, D.P.; Niu, Z.S.; Feng, J.N.; Chen, Y.R.; Tou, F.Y.; Garner, E.; Xu, J.; Liu, M.; et al. Biofilms as a sink for antibiotic resistance genes (ARGs) in the Yangtze Estuary. Water Res. 2018, 129, 277–286. [Google Scholar] [CrossRef]
- Nesse, L.L.; Simm, R. Biofilm: A Hotspot for Emerging Bacterial Genotypes. Adv. Appl. Microbiol. 2018, 103, 223–246. [Google Scholar]
- Olsson, M.; Jarbrink, K.; Divakar, U.; Bajpai, R.; Upton, Z.; Schmidtchen, A.; Car, J. The humanistic and economic burden of chronic wounds: A systematic review. Wound Repair Regen. 2019, 27, 114–125. [Google Scholar] [CrossRef] [PubMed]
- Russo, A.; Concia, E.; Cristini, F.; De Rosa, F.G.; Esposito, S.; Menichetti, F.; Petrosillo, N.; Tumbarello, M.; Venditti, M.; Viale, P.; et al. Current and future trends in antibiotic therapy of acute bacterial skin and skin-structure infections. Clin. Microbiol. Infect. 2016, 22, S27–S36. [Google Scholar] [CrossRef]
- Boateng, J.S.; Matthews, K.H.; Stevens, H.N.; Eccleston, G.M. Wound healing dressings and drug delivery systems: A review. J. Pharm Sci 2008, 97, 2892–2923. [Google Scholar] [CrossRef] [PubMed]
- Chaganti, P.; Gordon, I.; Chao, J.H.; Zehtabchi, S. A systematic review of foam dressings for partial thickness burns. Am. J. Emerg Med. 2019, 37, 1184–1190. [Google Scholar] [CrossRef] [PubMed]
- Dwivedi, C.; Pandey, H.; Pandey, A.C.; Ramteke, P.W. Nanofibre Based Smart Pharmaceutical Scaffolds for Wound Repair and Regenerations. Curr. Pharm. Des. 2016, 22, 1460–1471. [Google Scholar] [CrossRef] [PubMed]
- Muzzarelli, R.A. Chitins and chitosans for the repair of wounded skin, nerve, cartilage and bone. Carbohydr. Polym. 2009, 76, 167–182. [Google Scholar] [CrossRef]
- Purna, S.K.; Babu, M. Collagen based dressings—A review. Burn.: J. Int. Soc. Burn Inj. 2000, 26, 54. [Google Scholar]
- Zhu, Y.; Zhang, J.; Song, J.; Yang, J.; Xu, T.; Pan, C.; Zhang, L. One-step synthesis of an antibacterial and pro-healing wound dressing that can treat wound infections. J. Mater. Chem. B. 2017, 5, 8451–8458. [Google Scholar] [CrossRef]
- Abdelrahman, T.; Newton, H. Wound dressings: Principles and practice. Surgery 2011, 29, 491–495. [Google Scholar] [CrossRef]
- Negut, I.; Grumezescu, V.; Grumezescu, A.M. Treatment Strategies for Infected Wounds. Molecules 2018, 23, 2392. [Google Scholar] [CrossRef]
- Davies, P.; McCarty, S.; Hamberg, K. Silver-containing foam dressings with Safetac: A review of the scientific and clinical data. J. Wound Care 2017, 26, S1–S32. [Google Scholar] [CrossRef] [PubMed]
- Mulani, M.S.; Kamble, E.E.; Kumkar, S.N.; Tawre, M.S.; Pardesi, K.R. Emerging Strategies to Combat ESKAPE Pathogens in the Era of Antimicrobial Resistance: A Review. Front. Microbiol. 2019, 10, 539. [Google Scholar] [CrossRef] [PubMed]
- Albuquerque, P.B.S.; Soares, P.A.G.; Aragao-Neto, A.C.; Albuquerque, G.S.; Silva, L.C.N.; Lima-Ribeiro, M.H.M.; Silva Neto, J.C.; Coelho, L.; Correia, M.T.S.; Teixeira, J.A.C.; et al. Healing activity evaluation of the galactomannan film obtained from Cassia grandis seeds with immobilized Cratylia mollis seed lectin. Int. J. Biol. Macromol. 2017, 102, 749–757. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, A.R., Jr.; Diniz, R.M.; Suarez, M.A.M.; Figueiredo, C.; Zagmignan, A.; Grisotto, M.A.G.; Fernandes, E.S.; da Silva, L.C.N. Use of Some Asteraceae Plants for the Treatment of Wounds: From Ethnopharmacological Studies to Scientific Evidences. Front. Pharm. 2018, 9, 784. [Google Scholar] [CrossRef]
- Ousey, K. A multipurpose dressing: A clinical review of the absorption, debridement and healing properties of Aquacel Foam. J. Wound Care 2019, 28, S1–S23. [Google Scholar] [CrossRef]
- Ramos-e-Silva, M.; Ribeiro de Castro, M.C. New dressings, including tissue-engineered living skin. Clin. Derm. 2002, 20, 715–723. [Google Scholar] [CrossRef]
- Vowden, K.; Vowden, P. Wound dressings: Principles and practice. Surgery 2017, 35, 489–494. [Google Scholar]
- Ahmed, E.M. Hydrogel: Preparation, characterization, and applications: A review. J. Adv. Res. 2015, 6, 105–121. [Google Scholar] [CrossRef]
- Caló, E.; Khutoryanskiy, V.V. Biomedical applications of hydrogels: A review of patents and commercial products. Eur. Polym. J. 2015, 65, 252–267. [Google Scholar] [CrossRef]
- Pott, F.S.; Meier, M.J.; Stocco, J.G.; Crozeta, K.; Ribas, J.D. The effectiveness of hydrocolloid dressings versus other dressings in the healing of pressure ulcers in adults and older adults: A systematic review and meta-analysis. Rev. Lat. Am. Enferm. 2014, 22, 511–520. [Google Scholar] [CrossRef]
- Dhivya, S.; Padma, V.V.; Santhini, E. Wound dressings—A review. BioMedicine 2015, 5, 22. [Google Scholar] [CrossRef] [PubMed]
- Babavalian, H.; Latifi, A.M.; Shokrgozar, M.A.; Bonakdar, S.; Mohammadi, S.; Moosazadeh Moghaddam, M. Analysis of Healing Effect of Alginate Sulfate Hydrogel Dressing Containing Antimicrobial Peptide on Wound Infection Caused by Methicillin-Resistant Staphylococcus aureus. Jundishapur. J. Microbiol. 2015, 8, e28320. [Google Scholar] [CrossRef] [PubMed]
- Wong, T.W.; Ramli, N.A. Carboxymethylcellulose film for bacterial wound infection control and healing. Carbohydr. Polym. 2014, 112, 367–375. [Google Scholar] [CrossRef] [PubMed]
- Guo, R.; Lan, Y.; Xue, W.; Cheng, B.; Zhang, Y.; Wang, C.; Ramakrishna, S. Collagen-cellulose nanocrystal scaffolds containing curcumin-loaded microspheres on infected full-thickness burns repair. J. Tissue Eng. Regen. Med. 2017, 11, 3544–3555. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, V.Q.; Ishihara, M.; Mori, Y.; Nakamura, S.; Kishimoto, S.; Fujita, M.; Hattori, H.; Kanatani, Y.; Ono, T.; Miyahira, Y.; et al. Preparation of size-controlled silver nanoparticles and chitosan-based composites and their anti-microbial activities. Biomed. Mater. Eng. 2013, 23, 473–483. [Google Scholar] [CrossRef]
- Cui, F.; Li, G.; Huang, J.; Zhang, J.; Lu, M.; Lu, W.; Huan, J.; Huang, Q. Development of chitosan-collagen hydrogel incorporated with lysostaphin (CCHL) burn dressing with anti-methicillin-resistant Staphylococcus aureus and promotion wound healing properties. Drug Deliv. 2011, 18, 173–180. [Google Scholar] [CrossRef]
- Ali, A.; Ahmed, S. A review on chitosan and its nanocomposites in drug delivery. Int. J. Biol. Macromol. 2018, 109, 273–286. [Google Scholar] [CrossRef]
- Tayeb, A.H.; Amini, E.; Ghasemi, S.; Tajvidi, M. Cellulose Nanomaterials-Binding Properties and Applications: A Review. Molecules 2018, 23, 2684. [Google Scholar] [CrossRef]
- Lee, K.Y.; Mooney, D.J. Alginate: Properties and biomedical applications. Prog. Polym. Sci. 2012, 37, 106–126. [Google Scholar] [CrossRef]
- Albuquerque, P.; Coelho, L.C.; Teixeira, J.A.; Carneiro-da-Cunha, M.G. Approaches in biotechnological applications of natural polymers. AIMS Mol. Sci. 2016, 3, 386–425. [Google Scholar] [CrossRef]
- Gim, S.; Zhu, Y.; Seeberger, P.H.; Delbianco, M. Carbohydrate-based nanomaterials for biomedical applications. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2019, 11, e1558. [Google Scholar] [CrossRef] [PubMed]
- Hamedi, H.; Moradi, S.; Hudson, S.M.; Tonelli, A.E. Chitosan based hydrogels and their applications for drug delivery in wound dressings: A review. Carbohydr. Polym. 2018, 199, 445–460. [Google Scholar] [CrossRef] [PubMed]
- Baldrick, P. The safety of chitosan as a pharmaceutical excipient. Regul. Toxicol. Pharm. 2010, 56, 290–299. [Google Scholar] [CrossRef] [PubMed]
- Mondal, M.; Hosain, M. Biodegradable surfactant from natural starch for the reduction of environmental pollution and safety for water living organism. Int. J. Innov. Res. Adv. Eng. 2014, 1, 424–433. [Google Scholar]
- Oladoja, N.A.; Unuabonah, E.I.; Amuda, O.S.; Kolawole, O.M. Polysaccharides as a Green and Sustainable Resources for Water and Wastewater Treatment; Springer: Berlin, Germany, 2017. [Google Scholar]
- Ahmad, S.; Ahmad, M.; Manzoor, K.; Purwar, R.; Ikram, S. A review on latest innovations in natural gums based hydrogels: Preparations & applications. Int. J. Biol. Macromol. 2019, 136, 870–890. [Google Scholar]
- Hamman, H.; Steenekamp, J.; Hamman, J. Use of Natural Gums and Mucilages as Pharmaceutical Excipients. Curr. Pharm. Des. 2015, 21, 4775–4797. [Google Scholar] [CrossRef]
- Ribeiro, A.J.; de Souza, F.R.L.; Bezerra, J.; Oliveira, C.; Nadvorny, D.; de La Roca Soares, M.F.; Nunes, L.C.C.; Silva-Filho, E.C.; Veiga, F.; Soares Sobrinho, J.L. Gums’ based delivery systems: Review on cashew gum and its derivatives. Carbohydr. Polym. 2016, 147, 188–200. [Google Scholar] [CrossRef]
- Salarbashi, D.; Tafaghodi, M. An update on physicochemical and functional properties of newly seed gums. Int. J. Biol. Macromol. 2018, 119, 1240–1247. [Google Scholar] [CrossRef]
- Rana, V.; Rai, P.; Tiwary, A.K.; Singh, R.S.; Kennedy, J.F.; Knill, C.J. Modified gums: Approaches and applications in drug delivery. Carbohydr. Polym. 2011, 83, 1031–1047. [Google Scholar] [CrossRef]
- Thombare, N.; Jha, U.; Mishra, S.; Siddiqui, M.Z. Guar gum as a promising starting material for diverse applications: A review. Int. J. Biol. Macromol. 2016, 88, 361–372. [Google Scholar] [CrossRef]
- Albuquerque, P.B.S.; Cerqueira, M.A.; Vicente, A.A.; Teixeira, J.A.; Carneiro-da-Cunha, M.G. Immobilization of bioactive compounds in Cassia grandis galactomannan-based films: Influence on physicochemical properties. Int. J. Biol. Macromol. 2017, 96, 727–735. [Google Scholar] [CrossRef] [PubMed]
- Aragao-Neto, A.C.; Soares, P.A.; Lima-Ribeiro, M.H.; Carvalho, E.J.; Correia, M.T.; Carneiro-da-Cunha, M.G. Combined therapy using low level laser and chitosan-policaju hydrogel for wound healing. Int. J. Biol. Macromol. 2017, 95, 268–272. [Google Scholar] [CrossRef] [PubMed]
- de Sousa, F.D.; Vasconselos, P.D.; da Silva, A.F.B.; Mota, E.F.; da Rocha Tome, A.; Mendes, F.; Gomes, A.M.M.; Abraham, D.J.; Shiwen, X.; Owen, J.S.; et al. Hydrogel and membrane scaffold formulations of Frutalin (breadfruit lectin) within a polysaccharide galactomannan matrix have potential for wound healing. Int. J. Biol. Macromol. 2019, 121, 429–442. [Google Scholar] [CrossRef] [PubMed]
- Knopf-Marques, H.; Pravda, M.; Wolfova, L.; Velebny, V.; Schaaf, P.; Vrana, N.E.; Lavalle, P. Hyaluronic Acid and Its Derivatives in Coating and Delivery Systems: Applications in Tissue Engineering, Regenerative Medicine and Immunomodulation. Adv. Healthc. Mater. 2016, 5, 2841–2855. [Google Scholar] [CrossRef]
- Ahsan, S.M.; Thomas, M.; Reddy, K.K.; Sooraparaju, S.G.; Asthana, A.; Bhatnagar, I. Chitosan as biomaterial in drug delivery and tissue engineering. Int. J. Biol. Macromol. 2018, 110, 97–109. [Google Scholar] [CrossRef]
- Rastogi, P.; Kandasubramanian, B. Review of alginate-based hydrogel bioprinting for application in tissue engineering. Biofabrication 2019, 11, 042001. [Google Scholar] [CrossRef]
- Portela, R.; Leal, C.R.; Almeida, P.L.; Sobral, R.G. Bacterial cellulose: A versatile biopolymer for wound dressing applications. Microb. Biotechnol. 2019, 12, 586–610. [Google Scholar] [CrossRef]
- Martinez-Martinez, M.; Rodriguez-Berna, G.; Gonzalez-Alvarez, I.; Hernandez, M.J.; Corma, A.; Bermejo, M.; Merino, V.; Gonzalez-Alvarez, M. Ionic Hydrogel Based on Chitosan Cross-Linked with 6-Phosphogluconic Trisodium Salt as a Drug Delivery System. Biomacromolecules 2018, 19, 1294–1304. [Google Scholar] [CrossRef]
- Meaume, S.; Vallet, D.; Morere, M.N.; Teot, L. Evaluation of a silver-releasing hydroalginate dressing in chronic wounds with signs of local infection. J. Wound Care 2005, 14, 411–419. [Google Scholar] [CrossRef]
- Woo, K.Y.; Coutts, P.M.; Sibbald, R.G. A randomized controlled trial to evaluate an antimicrobial dressing with silver alginate powder for the management of chronic wounds exhibiting signs of critical colonization. Adv. Skin Wound Care 2012, 25, 503–508. [Google Scholar] [CrossRef]
- Mirzaei, B.; Etemadian, S.; Goli, H.R.; Bahonar, S.; Gholami, S.A.; Karami, P.; Farhadi, M.; Tavakoli, R. Construction and analysis of alginate-based honey hydrogel as an ointment to heal of rat burn wound related infections. Int. J. Burns Trauma 2018, 8, 88–97. [Google Scholar] [PubMed]
- Bjorn, C.; Noppa, L.; Naslund Salomonsson, E.; Johansson, A.L.; Nilsson, E.; Mahlapuu, M.; Hakansson, J. Efficacy and safety profile of the novel antimicrobial peptide PXL150 in a mouse model of infected burn wounds. Int. J. Antimicrob. Agents 2015, 45, 519–524. [Google Scholar] [CrossRef] [PubMed]
- Burkatovskaya, M.; Tegos, G.P.; Swietlik, E.; Demidova, T.N.; Castano A, P.; Hamblin, M.R. Use of chitosan bandage to prevent fatal infections developing from highly contaminated wounds in mice. Biomaterials 2006, 27, 4157–4164. [Google Scholar] [CrossRef] [PubMed]
- Burkatovskaya, M.; Castano, A.P.; Demidova-Rice, T.N.; Tegos, G.P.; Hamblin, M.R. Effect of chitosan acetate bandage on wound healing in infected and noninfected wounds in mice. Wound Repair Regen. 2008, 16, 425–431. [Google Scholar] [CrossRef]
- Dai, T.; Tegos, G.P.; Burkatovskaya, M.; Castano, A.P.; Hamblin, M.R. Chitosan acetate bandage as a topical antimicrobial dressing for infected burns. Antimicrob. Agents Chemother. 2009, 53, 393–400. [Google Scholar] [CrossRef]
- Huang, L.; Dai, T.; Xuan, Y.; Tegos, G.P.; Hamblin, M.R. Synergistic combination of chitosan acetate with nanoparticle silver as a topical antimicrobial: Efficacy against bacterial burn infections. Antimicrob. Agents Chemother. 2011, 55, 3432–3438. [Google Scholar] [CrossRef]
- Peng, Y.; Song, C.; Yang, C.; Guo, Q.; Yao, M. Low molecular weight chitosan-coated silver nanoparticles are effective for the treatment of MRSA-infected wounds. Int. J. Nanomed. 2017, 12, 295–304. [Google Scholar] [CrossRef]
- Zhu, C.; Zhao, J.; Kempe, K.; Wilson, P.; Wang, J.; Velkov, T.; Li, J.; Davis, T.P.; Whittaker, M.R.; Haddleton, D.M. A Hydrogel-Based Localized Release of Colistin for Antimicrobial Treatment of Burn Wound Infection. Macromol. Biosci. 2017, 17. [Google Scholar] [CrossRef]
- Sun, L.; Jiang, W.; Zhang, H.; Guo, Y.; Chen, W.; Jin, Y.; Chen, H.; Du, K.; Dai, H.; Ji, J.; et al. Photosensitizer-Loaded Multifunctional Chitosan Nanoparticles for Simultaneous in Situ Imaging, Highly Efficient Bacterial Biofilm Eradication, and Tumor Ablation. ACS Appl. Mater. Interfaces 2019, 11, 2302–2316. [Google Scholar] [CrossRef]
- Yang, S.; Han, X.; Jia, Y.; Zhang, H.; Tang, T. Hydroxypropyltrimethyl Ammonium Chloride Chitosan Functionalized-PLGA Electrospun Fibrous Membranes as Antibacterial Wound Dressing: In Vitro and In Vivo Evaluation. Polymer 2017, 9, 697. [Google Scholar] [CrossRef]
- Tian, R.; Qiu, X.; Yuan, P.; Lei, K.; Wang, L.; Bai, Y.; Liu, S.; Chen, X. Fabrication of Self-Healing Hydrogels with On-Demand Antimicrobial Activity and Sustained Biomolecule Release for Infected Skin Regeneration. ACS Appl. Mater. Interfaces 2018, 10, 17018–17027. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Q.; Jiang, M.; Liu, Q.; Yan, S.; Feng, L.; Lan, Y.; Shan, G.; Xue, W.; Guo, R. Enhanced healing activity of burn wound infection by a dextran-HA hydrogel enriched with sanguinarine. Biomater. Sci. 2018, 6, 2472–2486. [Google Scholar] [CrossRef] [PubMed]
- Gazzabin, L.; Serantoni, S.; Palumbo, F.P.; Giordan, N. Hyaluronic acid and metallic silver treatment of chronic wounds: Healing rate and bacterial load control. J. Wound Care 2019, 28, 482–490. [Google Scholar] [CrossRef] [PubMed]
- Aderibigbe, B.A.; Buyana, B. Alginate in Wound Dressings. Pharmaceutics 2018, 10, 42. [Google Scholar] [CrossRef]
- Moghaddam, M.M.; Abolhassani, F.; Babavalian, H.; Mirnejad, R.; Azizi Barjini, K.; Amani, J. Comparison of in vitro antibacterial activities of two cationic peptides CM15 and CM11 against five pathogenic bacteria: Pseudomonas aeruginosa, Staphylococcus aureus, Vibrio cholerae, Acinetobacter baumannii, and Escherichia coli. Probiotics. Antimicrob. Proteins 2012, 4, 133–139. [Google Scholar] [CrossRef]
- Mohammadi Azad, Z.; Moravej, H.; Fasihi-Ramandi, M.; Masjedian, F.; Nazari, R.; Mirnejad, R.; Moosazadeh Moghaddam, M. In vitro synergistic effects of a short cationic peptide and clinically used antibiotics against drug-resistant isolates of Brucella melitensis. J. Med. Microbiol. 2017, 66, 919–926. [Google Scholar] [CrossRef]
- Moosazadeh Moghaddam, M.; Eftekhary, M.; Erfanimanesh, S.; Hashemi, A.; Fallah Omrani, V.; Farhadihosseinabadi, B.; Lasjerdi, Z.; Mossahebi-Mohammadi, M.; Pal Singh Chauhan, N.; Seifalian, A.M.; et al. Comparison of the antibacterial effects of a short cationic peptide and 1% silver bioactive glass against extensively drug-resistant bacteria, Pseudomonas aeruginosa and Acinetobacter baumannii, isolated from burn patients. Amino Acids 2018, 50, 1617–1628. [Google Scholar] [CrossRef]
- Sulaeva, I.; Henniges, U.; Rosenau, T.; Potthast, A. Bacterial cellulose as a material for wound treatment: Properties and modifications. A review. Biotechnol. Adv. 2015, 33, 1547–1571. [Google Scholar] [CrossRef]
- Ramli, N.A.; Wong, T.W. Sodium carboxymethylcellulose scaffolds and their physicochemical effects on partial thickness wound healing. Int. J. Pharm. 2011, 403, 73–82. [Google Scholar] [CrossRef]
- Ogawa, A.; Nakayama, S.; Uehara, M.; Mori, Y.; Takahashi, M.; Aiba, T.; Kurosaki, Y. Pharmaceutical properties of a low-substituted hydroxypropyl cellulose (L-HPC) hydrogel as a novel external dressing. Int. J. Pharm. 2014, 477, 546–552. [Google Scholar] [CrossRef]
- Myhrman, E.; Hakansson, J.; Lindgren, K.; Bjorn, C.; Sjostrand, V.; Mahlapuu, M. The novel antimicrobial peptide PXL150 in the local treatment of skin and soft tissue infections. Appl. Microbiol. Biotechnol. 2013, 97, 3085–3096. [Google Scholar] [CrossRef] [PubMed]
- Hakansson, J.; Bjorn, C.; Lindgren, K.; Sjostrom, E.; Sjostrand, V.; Mahlapuu, M. Efficacy of the novel topical antimicrobial agent PXL150 in a mouse model of surgical site infections. Antimicrob. Agents Chemother. 2014, 58, 2982–2984. [Google Scholar] [CrossRef] [PubMed]
- Tejada, S.; Manayi, A.; Daglia, M.; Nabavi, S.F.; Sureda, A.; Hajheydari, Z.; Gortzi, O.; Pazoki-Toroudi, H.; Nabavi, S.M. Wound Healing Effects of Curcumin: A Short Review. Curr. Pharm. Biotechnol. 2016, 17, 1002–1007. [Google Scholar] [CrossRef] [PubMed]
- Shin, M.S.; Yu, J.S.; Lee, J.; Ji, Y.S.; Joung, H.J.; Han, Y.M.; Yoo, H.H.; Kang, K.S. A Hydroxypropyl Methylcellulose-Based Solid Dispersion of Curcumin with Enhanced Bioavailability and its Hepatoprotective Activity. Biomolecules 2019, 9, 281. [Google Scholar] [CrossRef]
- Freitas, M.A.; Pereira, A.H.; Pinto, J.G.; Casas, A.; Ferreira-Strixino, J. Bacterial viability after antimicrobial photodynamic therapy with curcumin on multiresistant Staphylococcus aureus. Future Microbiol. 2019, 14, 739–748. [Google Scholar] [CrossRef]
- Guran, M.; Sanliturk, G.; Kerkuklu, N.R.; Altundag, E.M.; Suha Yalcin, A. Combined effects of quercetin and curcumin on anti-inflammatory and antimicrobial parameters in vitro. Eur. J. Pharm. 2019, 859, 172486. [Google Scholar] [CrossRef]
- Paul, P.; Kolesinska, B.; Sujka, W. Chitosan and Its Derivatives—Biomaterials with Diverse Biological Activity for Manifold Applications. Mini Rev. Med. Chem. 2019, 19, 737–750. [Google Scholar] [CrossRef]
- Patrulea, V.; Ostafe, V.; Borchard, G.; Jordan, O. Chitosan as a starting material for wound healing applications. Eur. J. Pharm. Biopharm. 2015, 97, 417–426. [Google Scholar] [CrossRef]
- Penchev, H.; Paneva, D.; Manolova, N.; Rashkov, I. Electrospun hybrid nanofibers based on chitosan or N-carboxyethylchitosan and silver nanoparticles. Macromol. Biosci. 2009, 9, 884–894. [Google Scholar] [CrossRef]
- Penchev, H.; Paneva, D.; Manolova, N.; Rashkov, I. Hybrid nanofibrous yarns based on N-carboxyethylchitosan and silver nanoparticles with antibacterial activity prepared by self-bundling electrospinning. Carbohydr. Res. 2010, 345, 2374–2380. [Google Scholar] [CrossRef]
- Jayakumar, R.; Prabaharan, M.; Sudheesh Kumar, P.T.; Nair, S.V.; Tamura, H. Biomaterials based on chitin and chitosan in wound dressing applications. Biotechnol. Adv. 2011, 29, 322–337. [Google Scholar] [CrossRef] [PubMed]
- Shariatinia, Z. Pharmaceutical applications of chitosan. Adv. Colloid. Interface Sci. 2019, 263, 131–194. [Google Scholar] [CrossRef] [PubMed]
- Dai, T.; Tanaka, M.; Huang, Y.Y.; Hamblin, M.R. Chitosan preparations for wounds and burns: Antimicrobial and wound-healing effects. Expert. Rev. Anti-Infect. 2011, 9, 857–879. [Google Scholar] [CrossRef] [PubMed]
- Percival, S.L.; Thomas, J.; Linton, S.; Okel, T.; Corum, L.; Slone, W. The antimicrobial efficacy of silver on antibiotic-resistant bacteria isolated from burn wounds. Int. Wound J. 2012, 9, 488–493. [Google Scholar] [CrossRef] [PubMed]
- Ishihara, M.; Nguyen, V.Q.; Mori, Y.; Nakamura, S.; Hattori, H. Adsorption of Silver Nanoparticles onto Different Surface Structures of Chitin/Chitosan and Correlations with Antimicrobial Activities. Int. J. Mol. Sci. 2015, 16, 13973–13988. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.C.; Lall, R.; Srivastava, A.; Sinha, A. Hyaluronic Acid: Molecular Mechanisms and Therapeutic Trajectory. Front. Vet. Sci. 2019, 6, 192. [Google Scholar] [CrossRef]
- Litwiniuk, M.; Krejner, A.; Speyrer, M.S.; Gauto, A.R.; Grzela, T. Hyaluronic Acid in Inflammation and Tissue Regeneration. Wounds 2016, 28, 78–88. [Google Scholar]
- Neuman, M.G.; Nanau, R.M.; Oruna-Sanchez, L.; Coto, G. Hyaluronic acid and wound healing. J. Pharm Pharm. Sci. 2015, 18, 53–60. [Google Scholar] [CrossRef]
- Liu, M.; Liu, T.; Zhang, X.; Jian, Z.; Xia, H.; Yang, J.; Hu, X.; Xing, M.; Luo, G.; Wu, J. Fabrication of KR-12 peptide-containing hyaluronic acid immobilized fibrous eggshell membrane effectively kills multi-drug-resistant bacteria, promotes angiogenesis and accelerates re-epithelialization. Int. J. Nanomed. 2019, 14, 3345–3360. [Google Scholar] [CrossRef]
- Su, Z.; Ma, H.; Wu, Z.; Zeng, H.; Li, Z.; Wang, Y.; Liu, G.; Xu, B.; Lin, Y.; Zhang, P.; et al. Enhancement of skin wound healing with decellularized scaffolds loaded with hyaluronic acid and epidermal growth factor. Mater. Sci. Eng. C. Mater. Biol. Appl. 2014, 44, 440–448. [Google Scholar] [CrossRef]
- Salwowska, N.M.; Bebenek, K.A.; Zadlo, D.A.; Wcislo-Dziadecka, D.L. Physiochemical properties and application of hyaluronic acid: A systematic review. J. Cosmet. Derm. 2016, 15, 520–526. [Google Scholar] [CrossRef] [PubMed]
- Keen, M.A. Hyaluronic Acid in Dermatology. Skinmed 2017, 15, 441–448. [Google Scholar] [PubMed]
- Park, J.H.; Park, E.J.; Yi, H.S. Wound Healing and Anti-inflammatory Effects of Topical Hyaluronic Acid Injection in Surgical-Site Infection Caused by Staphylococcus aureus. Int. J. Low Extrem. Wounds 2017, 16, 202–207. [Google Scholar] [CrossRef] [PubMed]
- Hamoud, R.; Reichling, J.; Wink, M. Synergistic antimicrobial activity of combinations of sanguinarine and EDTA with vancomycin against multidrug resistant bacteria. Drug Metab. Lett. 2014, 8, 119–128. [Google Scholar] [CrossRef]
- Godowski, K.C. Antimicrobial action of sanguinarine. J. Clin. Dent. 1989, 1, 96–101. [Google Scholar] [PubMed]
- Croaker, A.; King, G.J.; Pyne, J.H.; Anoopkumar-Dukie, S.; Liu, L. Sanguinaria canadensis: Traditional Medicine, Phytochemical Composition, Biological Activities and Current Uses. Int. J. Mol. Sci. 2016, 17, 1414. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Li, H.; Yao, H.; Mu, Q.; Zhao, G.; Li, Y.; Hu, H.; Niu, X. Pharmacokinetic and anti-inflammatory effects of sanguinarine solid lipid nanoparticles. Inflammation 2014, 37, 632–638. [Google Scholar] [CrossRef]
- Abdou, A.G.; Maraee, A.H.; Al-Bara, A.M.; Diab, W.M. Immunohistochemical expression of TGF-beta1 in keloids and hypertrophic scars. Am. J. Derm. 2011, 33, 84–91. [Google Scholar] [CrossRef]
- Liu, Y.; Li, Y.; Li, N.; Teng, W.; Wang, M.; Zhang, Y.; Xiao, Z. TGF-beta1 promotes scar fibroblasts proliferation and transdifferentiation via up-regulating MicroRNA-21. Sci. Rep. 2016, 6, 32231. [Google Scholar] [CrossRef]
- Lichtman, M.K.; Otero-Vinas, M.; Falanga, V. Transforming growth factor beta (TGF-beta) isoforms in wound healing and fibrosis. Wound Repair Regen. 2016, 24, 215–222. [Google Scholar] [CrossRef]
| Fundamental Characteristics | Secondary Characteristics |
|---|---|
|
|
| Formulation Type | Indications | Advantages | Disadvantages | References |
| Foams |
|
|
| [13,19,25,26,27] |
| Hydrogels |
|
|
| [19,27,28,29] |
| Hydrocolloids |
|
|
| [19,27,30] |
| Films |
|
|
| [19,20,27,31] |
| Membranes |
|
|
| [19,27,31] |
| Polysaccharide | Co-Polymer | Formulation Type | Incorporated Agent | Infection Model | Bacteria | Ref. |
|---|---|---|---|---|---|---|
| Alginate | - | Alginate dressing | Silver | Human | - | [60] |
| Alginate dressing | Silver | Human | - | [61] | ||
| - | Alginate dressing | CM11 peptide | Animal (mice) | MRSA | [32] | |
| - | Hydrogel | Honey | Animal (rats) | A. baumannii; K. pneumoniae; P. aeruginosa; S. aureus | [62] | |
| Cellulose (Sodium carboxymethyl cellulose) | - | Film | - | Animal (rats) | P. aeruginosa; S. aureus | [33] |
| Cellulose (Hydroxypropyl cellulose) | - | Gel | PXL150 peptide | Animal (mice) | P. aeruginosa | [63] |
| Cellulose | Collagen | Scaffolds | Curcumin (loaded in gelatin microspheres) | Animal (rats) | E. coli; P. aeruginosa; S. aureus | [34] |
| Chitosan acetate | - | Dressing | - | Animal (rats) | P. mirabilis; P. aeruginosa; S. aureus | [64,65,66] |
| - | Dressing | Silver nanoparticles | Animal (mice) | A. baumannii; MRSA; P. mirabilis; P. aeruginosa | [67] | |
| Chitosan | Dressing | Silver nanoparticles | Animal (mice) | MRSA | [68] | |
| Collagen | Hydrogel | Lysostaphin | Animal (rabbits) | MRSA | [36,69] | |
| Chitosan (glycol chitosan) | Aldehyde-modified poly(ethylene glycol) derivative | Hydrogel | Colistin | Animal (mice) | P. aeruginosa | [36,69] |
| Chitosan (Carboxymethyl chitosan) | - | - Nanoparticles | - | Animal (rabbits) | P. aeruginosa; S. aureus | [70] |
| Chitosan (Hydroxypropyltrimethyl ammonium chloride chitosan) | Poly(lactic-co-glycolic acid) | Nanofibrous Membranes | - | Animal (mice) | S. aureus | [71] |
| Hyaluronic acid | - | Hydrogel | EDTA−Fe3+; PDGF-BB growth factor | Animal (mice) | E. coli; S. aureus | [72] |
| Dextran | Hydrogel | Sanguinarine (loaded in gelatin microspheres) | Animal (mice) | E. coli; MRSA | [73] | |
| - | Topical spray | Metallic silver | Human | - | [74] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ribeiro, D.M.L.; Carvalho Júnior, A.R.; Vale de Macedo, G.H.R.; Chagas, V.L.; Silva, L.d.S.; Cutrim, B.d.S.; Santos, D.M.; Soares, B.L.L.; Zagmignan, A.; de Miranda, R.d.C.M.; et al. Polysaccharide-Based Formulations for Healing of Skin-Related Wound Infections: Lessons from Animal Models and Clinical Trials. Biomolecules 2020, 10, 63. https://doi.org/10.3390/biom10010063
Ribeiro DML, Carvalho Júnior AR, Vale de Macedo GHR, Chagas VL, Silva LdS, Cutrim BdS, Santos DM, Soares BLL, Zagmignan A, de Miranda RdCM, et al. Polysaccharide-Based Formulations for Healing of Skin-Related Wound Infections: Lessons from Animal Models and Clinical Trials. Biomolecules. 2020; 10(1):63. https://doi.org/10.3390/biom10010063
Chicago/Turabian StyleRibeiro, Diogo Marcelo Lima, Alexsander Rodrigues Carvalho Júnior, Gustavo Henrique Rodrigues Vale de Macedo, Vitor Lopes Chagas, Lucas dos Santos Silva, Brenda da Silva Cutrim, Deivid Martins Santos, Bruno Luis Lima Soares, Adrielle Zagmignan, Rita de Cássia Mendonça de Miranda, and et al. 2020. "Polysaccharide-Based Formulations for Healing of Skin-Related Wound Infections: Lessons from Animal Models and Clinical Trials" Biomolecules 10, no. 1: 63. https://doi.org/10.3390/biom10010063
APA StyleRibeiro, D. M. L., Carvalho Júnior, A. R., Vale de Macedo, G. H. R., Chagas, V. L., Silva, L. d. S., Cutrim, B. d. S., Santos, D. M., Soares, B. L. L., Zagmignan, A., de Miranda, R. d. C. M., de Albuquerque, P. B. S., & Nascimento da Silva, L. C. (2020). Polysaccharide-Based Formulations for Healing of Skin-Related Wound Infections: Lessons from Animal Models and Clinical Trials. Biomolecules, 10(1), 63. https://doi.org/10.3390/biom10010063








