Stoichiometry Based Steady-State Hepatic Flux Analysis: Computational and Experimental Aspects
Abstract
:1. Introduction
2. Functions of the Liver
3. Experimental Methods for Liver Systems
3.1. Liver Cell Isolation and Culturing
3.2. Liver Perfusion System
3.3. Micro Fluidic Devices
4. Stoichiometric Models for Hepatic Networks
4.1. Hepatic Network Construction
4.2. Steady State Flux Analysis
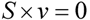
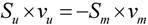
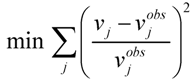 , where vj and vjobs are the predicted and observed external flux values, respectively). Another objective function that has also been used for hepatocytes is maximizing the summation of weighted fluxes (
, where vj and vjobs are the predicted and observed external flux values, respectively). Another objective function that has also been used for hepatocytes is maximizing the summation of weighted fluxes ( 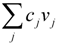 , where the coefficient cj is the weight of flux vj). Therefore, a weight can be attributed for each flux, which shows the importance of that flux. Yang et al. [66] and Nolan et al. [63] applied a bi-level optimization problem for the hepatic network where upper level objective function minimizes the relative error, whereas the lower level maximizes the summation of weighted fluxes. They solved the bi-level program by reformulating it into a single-level program using the Karush-Kuhn-Tucker or the primal-dual strategies. Furthermore, Uygun and co-workers [65] formulated a three-level optimization problem in order to determine a minimum set of fluxes that are of major importance to the liver cells while minimizing the relative error and maximizing the summation of weighted fluxes. They solved this mixed integer nonlinear programming problem (MINLP) in an iterative scheme until sufficient prediction accuracy had been obtained. By this data-mining procedure, they aimed to identify possible metabolic objectives for hepatocytes cultured in vitro.
, where the coefficient cj is the weight of flux vj). Therefore, a weight can be attributed for each flux, which shows the importance of that flux. Yang et al. [66] and Nolan et al. [63] applied a bi-level optimization problem for the hepatic network where upper level objective function minimizes the relative error, whereas the lower level maximizes the summation of weighted fluxes. They solved the bi-level program by reformulating it into a single-level program using the Karush-Kuhn-Tucker or the primal-dual strategies. Furthermore, Uygun and co-workers [65] formulated a three-level optimization problem in order to determine a minimum set of fluxes that are of major importance to the liver cells while minimizing the relative error and maximizing the summation of weighted fluxes. They solved this mixed integer nonlinear programming problem (MINLP) in an iterative scheme until sufficient prediction accuracy had been obtained. By this data-mining procedure, they aimed to identify possible metabolic objectives for hepatocytes cultured in vitro.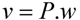 , where w denotes a vector involving the weight for each elementary mode; and P is the matrix of elementary modes). However, the decomposition of a steady state flux vector into pathways is not always unique for large networks because the number of pathways is not usually equal to the dimension of the null space of the stoichiometric matrix [77]. To overcome this problem, different objective functions have been proposed in the literature, such as maximization of the number of elementary modes, minimization of the elementary mode activity and the entropy maximization principle [78,79,80,81]. Orman et al. further introduced other optimization algorithms, including maximization of activity of short pathways and maximization of activity of liver-specific pathways, including urea and glucose production, to analyze the hepatic metabolism with the data sets obtained from perfused livers of fasted rats receiving burn injury [64].
, where w denotes a vector involving the weight for each elementary mode; and P is the matrix of elementary modes). However, the decomposition of a steady state flux vector into pathways is not always unique for large networks because the number of pathways is not usually equal to the dimension of the null space of the stoichiometric matrix [77]. To overcome this problem, different objective functions have been proposed in the literature, such as maximization of the number of elementary modes, minimization of the elementary mode activity and the entropy maximization principle [78,79,80,81]. Orman et al. further introduced other optimization algorithms, including maximization of activity of short pathways and maximization of activity of liver-specific pathways, including urea and glucose production, to analyze the hepatic metabolism with the data sets obtained from perfused livers of fasted rats receiving burn injury [64].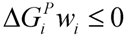 , where
, where 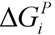 and wi are respectively Gibbs free energy and weight value of the pathway i [87]. This condition ensures that the pathway i can be active (wi ≥ 0) if its Gibbs free energy is less than zero (
and wi are respectively Gibbs free energy and weight value of the pathway i [87]. This condition ensures that the pathway i can be active (wi ≥ 0) if its Gibbs free energy is less than zero ( 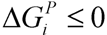 ), otherwise it is not active (wi = 0).
), otherwise it is not active (wi = 0).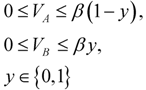
5. Applications and Major Outcomes of Stoichiometry-Based Hepatic Models
| Aim | Method | References |
|---|---|---|
| To investigate the metabolic objectives of cultured hepatocytes. | FBA | Uygun et al. [65] |
| To analyze the liver metabolism under fasted state | FBA | Orman et al. [7] |
| To analyze the liver metabolism under fasted state | FBA, MPA | Orman et al. [87] |
| To identify the metabolic changes associated with the cytotoxicity of saturated free fatty acids | MFA | Srivastava and Chan [88] |
| To design an optimum amino acid supplementation to increase the liver functions (computational) | FBA | Sharma et al. [70] |
| To design an optimum amino acid supplementation to increase the liver functions (computational and empirical) | FBA | Yang et al. [66,68,69] |
| To improve the hepatic functions with insulin, amino acid and plasma supplementation | MFA | Chan et al. [59,89] |
| To improve the perfused livers’ functions with red blood cells | FBA, MPA | Orman et al. [46] |
| To analyze the effects of burn injury on the flux distribution of liver metabolism | MFA | Lee et al. [5,6] |
| To determine hepatic flux changes caused by burn and septic shocks | MFA | Banta et al. [1] |
| To investigate the D-Galactosamine induced rat liver failure | MFA | Arai et al. [4] |
| To investigate D-Galactosamine induced rat liver failure (fluxes were measured at multiple time points) | MFA | Yokoyama et al. [3] |
| To investigate the effects of glucose and insulin on hepatic carcinoma cells | FBA | Iyer et al. [85] |
| To analyze the therapeutic effects of Dehydroepiandrosterone | MFA | Banta et al. [2] |
| To investigate the effects of hepatotoxic compounds on liver metabolism | MFA | Niklas et al. [90] |
| To analyze the effects of triadimefon on the primary rat hepatocytes | FBA | Iyer et al. [84] |
5.1. Characterization of Liver Functions and Objectives
5.2. Improving in Vitro Liver Functions
5.3. Characterization of Disease-Related Changes in the Liver
5.4. Proposing Treatment Techniques and Analyzing Effects of Drugs
6. Current Challenges
7. Conclusions
Acknowledgments
References
- Banta, S.; Vemula, M.; Yokoyama, T.; Jayaraman, A.; Berthiaume, F.; Yarmush, M.L. Contribution of gene expression to metabolic fluxes in hypermetabolic livers induced through burn injury and cecal ligation and puncture in rats. Biotechnol. Bioeng. 2007, 97, 118–137. [Google Scholar]
- Banta, S.; Yokoyama, T.; Berthiaume, F.; Yarmush, M.L. Effects of dehydroepiandrosterone administration on rat hepatic metabolism following thermal injury. J. Surg. Res. 2005, 127, 93–105. [Google Scholar]
- Yokoyama, T.; Banta, S.; Berthiaume, F.; Nagrath, D.; Tompkins, R.G.; Yarmush, M.L. Evolution of intrahepatic carbon, nitrogen, and energy metabolism in a D-galactosamine-induced rat liver failure model. Metab. Eng. 2005, 7, 88–103. [Google Scholar]
- Arai, K.; Lee, K.; Berthiaume, F.; Tompkins, R.G.; Yarmush, M.L. Intrahepatic amino acid and glucose metabolism in a D-galactosamine-induced rat liver failure model. Hepatology 2001, 34, 360–371. [Google Scholar]
- Lee, K.; Berthiaume, F.; Stephanopoulos, G.N.; Yarmush, D.M.; Yarmush, M.L. Metabolic flux analysis of postburn hepatic hypermetabolism. Metab. Eng. 2000, 2, 312–327. [Google Scholar]
- Lee, K.; Berthiaume, F.; Stephanopoulos, G.N.; Yarmush, M.L. Profiling of dynamic changes in hypermetabolic livers. Biotechnol. Bioeng. 2003, 83, 400–415. [Google Scholar] [CrossRef]
- Orman, M.A.; Arai, K.; Yarmush, M.L.; Androulakis, I.P.; Berthiaume, F.; Ierapetritou, M.G. Metabolic flux determination in perfused livers by mass balance analysis: Effect of fasting. Biotechnol. Bioeng. 2010, 107, 825–835. [Google Scholar]
- Michalopoulos, G.K. Liver regeneration. J. Cell. Physiol. 2007, 213, 286–300. [Google Scholar] [CrossRef]
- Lavon, N.; Benvenisty, N. Study of hepatocyte differentiation using embryonic stem cells. J. Cell. Biochem. 2005, 96, 1193–1202. [Google Scholar] [CrossRef]
- Katz, J.; Tayek, J.A. Gluconeogenesis and the Cori cycle in 12-, 20-, and 40-h-fasted humans. Am. J. Physiol. Endocrinol. Metab. 1998, 275, E537–E542. [Google Scholar]
- Wang, S.; Raju, B.I.; Leyvi, E.; Weinstein, D.A.; Seip, R. Acoustic accessibility investigation for ultrasound mediated treatment of glycogen storage disease type ia patients. Ultrasound Med. Biol. 2011, 37, 1469–1477. [Google Scholar] [CrossRef]
- Heeneman, S.; Deutz, N.E. Effects of decreased glutamine supply on gut and liver metabolism in vivo in rats. Clin. Sci. (Lond) 1993, 85, 437–444. [Google Scholar]
- Gachon, F.; Firsov, D. The role of circadian timing system on drug metabolism and detoxification. Expert Opin. Drug Metab. Toxicol. 2011, 7, 147–158. [Google Scholar] [CrossRef]
- Guengerich, F.P. Cytochrome p450 and chemical toxicology. Chem. Res. Toxicol. 2008, 21, 70–83. [Google Scholar] [CrossRef]
- Fernandez-Sanchez, A.; Madrigal-Santillan, E.; Bautista, M.; Esquivel-Soto, J.; Morales-Gonzalez, A.; Esquivel-Chirino, C.; Durante-Montiel, I.; Sanchez-Rivera, G.; Valadez-Vega, C.; Morales-Gonzalez, J.A. Inflammation, oxidative stress, and obesity. Int. J. Mol. Sci. 2011, 12, 3117–3132. [Google Scholar]
- Michell, D.L.; Andrews, K.L.; Chin-Dusting, J.P. Endothelial dysfunction in hypertension: The role of arginase. Front. Biosci. (Schol Ed) 2011, 3, 946–960. [Google Scholar]
- Munder, M. Arginase: An emerging key player in the mammalian immune system. Br. J. Pharmacol. 2009, 158, 638–651. [Google Scholar]
- Jungermann, K.; Katz, N. Functional specialization of different hepatocyte populations. Physiol. Rev. 1989, 69, 708–764. [Google Scholar]
- Sasse, D.; Spornitz, U.M.; Maly, I.P. Liver architecture. Enzyme 1992, 46, 8–32. [Google Scholar]
- Jungermann, K. Zonation of metabolism and gene expression in liver. Histochem. Cell. Biol. 1995. [Google Scholar]
- Quistorff, B.; Katz, N.; Witters, L.A. Hepatocyte heterogeneity in the metabolism of fatty acids: Discrepancies on zonation of acetyl-CoA carboxylase. Enzyme 1992, 46, 59–71. [Google Scholar]
- Sasse, D. Dynamics of liver glycogen: the topochemistry of glycogen synthesis, glycogen content and glycogenolysis under the experimental conditions of glycogen accumulation and depletion. Histochemistry 1975, 45, 237–254. [Google Scholar] [CrossRef]
- Haussinger, D.; Lamers, W.H.; Moorman, A.F. Hepatocyte heterogeneity in the metabolism of amino acids and ammonia. Enzyme 1992, 46, 72–93. [Google Scholar]
- Jungermann, K.; Kietzmann, T. Zonation of parenchymal and nonparenchymal metabolism in liver. Annu. Rev. Nutr. 1996, 16, 179–203. [Google Scholar]
- Katz, J.; Kuwajima, M.; Foster, D.W.; Denis McGarry, J. The glucose paradox: New perspectives on hepatic carbohydrate metabolism. Trends Biochem. Sci. 1986, 11, 136–140. [Google Scholar]
- Haussinger, D.; Stehle, T. Hepatocyte heterogeneity in response to icosanoids. The perivenous scavenger cell hypothesis. Eur. J. Biochem. 1988, 175, 395–403. [Google Scholar] [CrossRef]
- Guzman, M.; Castro, J. Zonation of fatty acid metabolism in rat liver. Biochem. J. 1989, 264, 107–113. [Google Scholar]
- Seglen, P.O. Preparation of isolated rat liver cells. Methods Cell Biol. 1976, 13, 29–83. [Google Scholar]
- Jennison, M.W. Bacterial collagenase. J. Bacteriol. 1945, 50, 369–370. [Google Scholar]
- Behnia, K.; Bhatia, S.; Jastromb, N.; Balis, U.; Sullivan, S.; Yarmush, M.; Toner, M. Xenobiotic Metabolism by Cultured Primary Porcine Hepatocytes. Tissue Eng. 2000, 6, 467–479. [Google Scholar]
- Schwenk, M.; Burr, R.; Pfaff, E. Influence of viability on bromosulfophthalein uptake by isolated hepatocytes. Naunyn Schmiedebergs Arch. Pharmacol. 1976, 295, 99–102. [Google Scholar]
- Page, D.T.; Garvey, J.S. Isolation and characterization of hepatocytes and Kupffer cells. J. Immunol. Methods 1979, 27, 159–173. [Google Scholar]
- Wincek, T.J.; Hupka, A.L.; Sweat, F.W. Stimulation of adenylate cyclase from isolated hepatocytes and Kupffer cells. J. Biol. Chem. 1975, 250, 8863–8873. [Google Scholar]
- Elsdale, T.; Bard, J. Collagen substrata for studies on cell behavior. J. Cell Biol. 1972, 54, 626–637. [Google Scholar]
- Bissell, D.M. Primary hepatocyte culture: substratum requirements and production of matrix components. Fed. Proc. 1981, 40, 2469–2473. [Google Scholar]
- Kang, Y.H.; Berthiaume, F.O.; Nath, B.D.; Yarmush, M.L. Growth factors and nonparenchymal cell conditioned media induce mitogenic responses in stable long-term adult rat hepatocyte cultures. Exp. Cell Res. 2004, 293, 239–247. [Google Scholar]
- Sharma, N.S.; Nagrath, D.; Yarmush, M.L. Metabolic profiling based quantitative evaluation of hepatocellular metabolism in presence of adipocyte derived extracellular matrix. PLoS One 2011, 6, e20137. [Google Scholar]
- Borel Rinkes, I.H.M.; Toner, M.; Ezzell, R.M.; Tompkins, R.G.; Yarmush, M.L. Effects of dimethyl sulfoxide on cultured rat hepatocytes in sandwich configuration. Cryobiology 1992, 29, 443–453. [Google Scholar]
- Wilson, R.; Warner, N.; Ryan, K.; Selleck, L.; Colledge, D.; Rodgers, S.; Li, K.; Revill, P.; Locarnini, S. The hepatitis B e antigen suppresses IL-1beta-mediated NF-kappaB activation in hepatocytes. J. Viral. Hepat. 2011, 18, e499–e507. [Google Scholar]
- Berg, T.; Morland, J. Induction of tryptophan oxygenase by dexamethasone in isolated hepatocytes. Dependence on composition of medium and pH. Biochim. Biophys. Acta 1975, 392, 233–241. [Google Scholar] [CrossRef]
- Horiuti, Y.; Nakamura, T.; Ichihara, A. Role of serum in maintenance of functional hepatocytes in primary culture. J. Biochem. 1982, 92, 1985–1994. [Google Scholar]
- Yamaguchi, Y.; Yu, Y.M.; Zupke, C.; Yarmush, D.M.; Berthiaume, F.; Tompkins, R.G.; Yarmush, M.L. Effect of burn injury on glucose and nitrogen metabolism in the liver: Preliminary studies in a perfused liver system. Surgery 1997, 121, 295–303. [Google Scholar]
- Gores, G.J.; Kost, L.J.; LaRusso, N.F. The isolated perfused rat liver: conceptual and practical considerations. Hepatology 1986, 6, 511–517. [Google Scholar]
- Sahin, S.; Rowland, M. Development of an optimal method for the dual perfusion of the isolated rat liver. J. Pharmacol. Toxicol. Methods 1998, 39, 35–43. [Google Scholar]
- El-Gibaly, A.M.; Scheuer, C.; Menger, M.D.; Vollmar, B. Improvement of rat liver graft quality by pifithrin-alpha-mediated inhibition of hepatocyte necrapoptosis. Hepatology 2004, 39, 1553–1562. [Google Scholar] [CrossRef]
- Orman, M.A.; Ierapetritou, M.G.; Androulakis, I.P.; Berthiaume, F. Metabolic response of perfused livers to various oxygenation conditions. Biotechnol. Bioeng. 2011, 108, 2947–2957. [Google Scholar]
- Mik, E.G.; Johannes, T.; Zuurbier, C.J.; Heinen, A.; Houben-Weerts, J.H.; Balestra, G.M.; Stap, J.; Beek, J.F.; Ince, C. In vivo mitochondrial oxygen tension measured by a delayed fluorescence lifetime technique. Biophys. J. 2008, 95, 3977–3790. [Google Scholar]
- Rupenko, A.P.; Kruglik, O.V.; Morgulis, I.I. Functional activity of isolated perfused rat liver depends on medium composition. Bull. Exp. Biol. Med. 2008, 146, 107–110. [Google Scholar]
- Plauth, M.; Zimmermann, B.; Raible, A.; Vieillard-Baron, D.; Bauder-Gross, D.; Hartmann, F. Use of an artificial oxygen carrier in isolated rat liver perfusion: First demonstration of net glucose uptake at physiological portal glucose concentrations using a hemoglobin-free perfusate. Res. Exp. Med. (Berl) 1991, 191, 339–347. [Google Scholar]
- Fisher, R.L.; Gandolfi, A.J.; Sipes, I.G.; Brendel, K. Culture medium composition affects the relative toxicities of chlorobenzenes in rat liver slices and the isolated perfused liver. Drug Chem. Toxicol. 1993, 16, 321–339. [Google Scholar]
- Chen, Y.A.; King, A.D.; Shih, H.C.; Peng, C.C.; Wu, C.Y.; Liao, W.H.; Tung, Y.C. Generation of oxygen gradients in microfluidic devices for cell culture using spatially confined chemical reactions. Lab Chip 2011, 11, 3626–3633. [Google Scholar]
- Yamada, M.; Kano, K.; Tsuda, Y.; Kobayashi, J.; Yamato, M.; Seki, M.; Okano, T. Microfluidic devices for size-dependent separation of liver cells. Biomed. Microdevices 2007, 9, 637–645. [Google Scholar] [CrossRef]
- Anderson, K.; Cooper, J.M.; Haswell, S.J.; Marshall, D.; Yin, H.; Zhang, X. Microfluidic-based measurements of cytochrome P450 enzyme activity of primary mammalian hepatocytes. Analyst 2011, 135, 1282–1287. [Google Scholar]
- Nakao, Y.; Kimura, H.; Sakai, Y.; Fujii, T. Bile canaliculi formation by aligning rat primary hepatocytes in a microfluidic device. Biomicrofluidics 2011, 5, 22212. [Google Scholar] [CrossRef]
- Zhao, S.; Huang, Y.; Liu, Y.M. Microchip electrophoresis with chemiluminescence detection for assaying ascorbic acid and amino acids in single cells. J. Chromatogr. A 2009, 1216, 6746–6751. [Google Scholar]
- Kane, B.J.; Zinner, M.J.; Yarmush, M.L.; Toner, M. Liver-specific functional studies in a Microfluidic array of primary mammalian hepatocytes. Anal. Chem. 2006, 78, 4291–4298. [Google Scholar]
- Hofmann, U.; Maier, K.; Niebel, A.; Vacun, G.;Reuss, M.; Mauch, K. Identification of metabolic fluxes in hepatic cells from transient 13C-labeling experiments: Part I. Experimental observations. Biotechnol. Bioeng. 2008, 100, 344–354. [Google Scholar]
- Lee, P.J.; Hung, P.J.; Lee, L.P. An artificial liver sinusoid with a microfluidic endothelial-like barrier for primary hepatocyte culture. Biotechnol. Bioeng. 2007, 97, 1340–1346. [Google Scholar]
- Chan, C.; Berthiaume, F.; Lee, K.; Yarmush, M.L. Metabolic flux analysis of cultured hepatocytes exposed to plasma. Biotechnol. Bioeng. 2003, 81, 33–49. [Google Scholar]
- Chan, C.; Hwang, D.; Stephanopoulos, G.N.; Yarmush, M.L.; Stephanopoulos, G. Application of multivariate analysis to optimize function of cultured hepatocytes. Biotechnol. Prog. 2003, 19, 580–598. [Google Scholar] [CrossRef]
- Gille, C.; Bolling, C.; Hoppe, A.; Bulik, S.; Hoffmann, S.; Hubner, K.; Karlstadt, A.; Ganeshan, R.; Konig, M.; Rother, K.; et al. HepatoNet1: A comprehensive metabolic reconstruction of the human hepatocyte for the analysis of liver physiology. Mol. Syst. Biol. 2010, 6, 411. [Google Scholar]
- Jerby, L.; Shlomi, T.; Ruppin, E. Computational reconstruction of tissue-specific metabolic models: application to human liver metabolism. Mol. Syst. Biol. 2010, 6, 401. [Google Scholar]
- Nolan, R.P.; Fenley, A.P.; Lee, K. Identification of distributed metabolic objectives in the hypermetabolic liver by flux and energy balance analysis. Metab. Eng. 2006, 8, 30–45. [Google Scholar] [CrossRef]
- Orman, M.A.; Berthiaume, F.; Androulakis, I.P.; Ierapetritou, M.G. Pathway analysis of liver metabolism under stressed condition. J. Theor. Biol. 2011, 272, 131–140. [Google Scholar]
- Uygun, K.; Matthew, H.W.T.; Huang, Y. Investigation of metabolic objectives in cultured hepatocytes. Biotechnol. Bioeng. 2007, 97, 622–637. [Google Scholar] [CrossRef]
- Yang, H.; Roth, C.M.; Ierapetritou, M.G. Analysis of amino acid supplementation effects on hepatocyte cultures using flux balance analysis. OMICS 2011, 449–460. [Google Scholar]
- alik, P.; Akbay, A. Mass flux balance-based model and metabolic flux analysis for collagen synthesis in the fibrogenesis process of human liver. Med. Hypotheses 2000, 55, 5–14. [Google Scholar]
- Yang, H.; Roth, C.M.; Ierapetritou, M.G. A rational design approach for amino acid supplementation in hepatocyte culture. Biotechnol. Bioeng. 2009, 103, 1176–1191. [Google Scholar]
- Yang, H.; Ierapetritou, M.G.; Roth, C.M. Effects of amino acid transport limitations on cultured hepatocytes. Biophys. Chem. 2010, 152, 89–98. [Google Scholar]
- Sharma, N.S.; Ierapetritou, M.G.; Yarmush, M.L. Novel quantitative tools for engineering analysis of hepatocyte cultures in bioartificial liver systems. Biotechnol. Bioeng. 2005, 92, 321–335. [Google Scholar]
- Nagrath, D.; Avila-Elchiver, M.; Berthiaume, F.; Tilles, A.; Messac, A.; Yarmush, M. Integrated Energy and flux balance based multiobjective framework for large-scale metabolic networks. Ann. Biomed. Eng. 2007, 35, 863–885. [Google Scholar] [CrossRef]
- Nagrath, D.; Avila-Elchiver, M.; Berthiaume, F.; Tilles, A.W.; Messac, A.; Yarmush, M.L. Soft constraints-based multiobjective framework for flux balance analysis. Metab. Eng. 2010, 12, 429–445. [Google Scholar]
- Klamt, S.; Stelling, J. Two approaches for metabolic pathway analysis? Trends Biotechnol. 2003, 21, 64–69. [Google Scholar] [CrossRef]
- Trinh, C.T.; Wlaschin, A.; Srienc, F. Elementary mode analysis: A useful metabolic pathway analysis tool for characterizing cellular metabolism. Appl. Microbiol. Biotechnol. 2009, 81, 813–826. [Google Scholar]
- Klamt, S.; Saez-Rodriguez, J.; Gilles, E.D. Structural and functional analysis of cellular networks with CellNetAnalyzer. BMC Syst. Biol. 2007, 1, 2. [Google Scholar] [CrossRef]
- Klamt, S.; Stelling, J.; Ginkel, M.; Gilles, E.D. FluxAnalyzer: Exploring structure, pathways, and flux distributions in metabolic networks on interactive flux maps. Bioinformatics 2003, 19, 261–269. [Google Scholar]
- Schilling, C.H.; Letscher, D.; Palsson, B.O. Theory for the systemic definition of metabolic pathways and their use in interpreting metabolic function from? A pathway-oriented perspective. J. Theor. Biol. 2000, 203, 229–248. [Google Scholar]
- Nookaew, I.; Meechai, A.; Thammarongtham, C.; Laoteng, K.; Ruanglek, V.; Cheevadhanarak, S.; Nielsen, J.; Bhumiratana, S. Identification of flux regulation coefficients from elementary flux modes: A systems biology tool for analysis of metabolic networks. Biotechnol. Bioeng. 2007, 97, 1535–1549. [Google Scholar]
- Schwartz, J.M.; Kanehisa, M. A quadratic programming approach for decomposing steady-state metabolic flux distributions onto elementary modes. Bioinformatics 2005, 21, 204–205. [Google Scholar]
- Zhao, Q.Y.; Kurata, H. Maximum entropy decomposition of flux distribution at steady state to elementary modes. J. Biosci. Bioeng. 2009, 107, 84–89. [Google Scholar]
- Schwartz, J.M.; Kanehisa, M. Quantitative elementary mode analysis of metabolic pathways: The example of yeast glycolysis. BMC Bioinformatics 2006, 7, 186. [Google Scholar] [CrossRef]
- Jankowski, M.D.; Henry, C.S.; Broadbelt, L.J.; Hatzimanikatis, V. Group contribution method for thermodynamic analysis of complex metabolic networks. Biophys. J. 2008, 95, 1487–1499. [Google Scholar] [CrossRef]
- Henry, C.S.; Broadbelt, L.J.; Hatzimanikatis, V. Thermodynamics-based metabolic flux analysis. Biophys. J. 2007, 92, 1792–1805. [Google Scholar] [CrossRef]
- Iyer, V.V.; Ovacik, M.A.; Androulakis, I.P.; Roth, C.M.; Ierapetritou, M.G. Transcriptional and metabolic flux profiling of triadimefon effects on cultured hepatocytes. Toxicol. Appl. Pharmacol. 2010, 248, 165–177. [Google Scholar]
- Iyer, V.V.; Yang, H.; Ierapetritou, M.G.; Roth, C.M. Effects of glucose and insulin on HepG2-C3A cell metabolism. Biotechnol. Bioeng. 2010, 107, 347–356. [Google Scholar]
- Yoon, J.; Si, Y.; Nolan, R.; Lee, K. Modular decomposition of metabolic reaction networks based on flux analysis and pathway projection. Bioinformatics 2007, 23, 2433–2440. [Google Scholar]
- Orman, M.A.; Androulakis, I.P.; Berthiaume, F.; Ierapetritou, M.G. Metabolic network analysis of perfused livers under fed and fasted states: Incorporating thermodynamic and futile-cycle-associated regulatory constraints. J. Theor. Biol. 2012, 293, 101–110. [Google Scholar]
- Srivastava, S.; Chan, C. Application of metabolic flux analysis to identify the mechanisms of free fatty acid toxicity to human hepatoma cell line. Biotechnol. Bioeng. 2008, 99, 399–410. [Google Scholar] [CrossRef]
- Chan, C.; Berthiaume, F.; Lee, K.; Yarmush, M.L. Metabolic flux analysis of hepatocyte function in hormone- and amino acid-supplemented plasma. Metab. Eng. 2003, 5, 1–15. [Google Scholar]
- Niklas, J.; Noor, F.; Heinzle, E. Effects of drugs in subtoxic concentrations on the metabolic fluxes in human hepatoma cell line Hep G2. Toxicol. Appl. Pharmacol. 2009, 240, 327–336. [Google Scholar]
- Evsikov, A.; Dolan, M.; Genrich, M.; Patek, E.; Bult, C. MouseCyc: A curated biochemical pathways database for the laboratory mouse. Genome Biol. 2009, 10, R84. [Google Scholar] [CrossRef]
- Orman, M.A.; Ierapetritou, M.G.; Berthiaume, F.; Androulakis, I.P. The dynamics of the early inflammatory response in double-hit burn and sepsis animal models. Cytokine 2011, 56, 494–502. [Google Scholar] [CrossRef]
- Oike, H.; Nagai, K.; Fukushima, T.; Ishida, N.; Kobori, M. Feeding cues and injected nutrients induce acute expression of multiple clock genes in the mouse liver. PLoS One 2011, 6, e23709. [Google Scholar]
- Lotze, M.T.; Frana, L.W.; Sharrow, S.O.; Robb, R.J.; Rosenberg, S.A. In vivo administration of purified human interleukin 2. I. Half-life and immunologic effects of the Jurkat cell line-derived interleukin 2. J. Immunol 1985, 134, 157–166. [Google Scholar]
- Kontermann, R.E. Strategies for extended serum half-life of protein therapeutics. Curr. Opin. Biotechnol. 2011, 22, 868–876. [Google Scholar]
- Reis, E.S.; Lange, T.; Kohl, G.; Herrmann, A.; Tschulakow, A.V.; Naujoks, J.; Born, J.; Kohl, J. Sleep and circadian rhythm regulate circulating complement factors and immunoregulatory properties of C5a. Brain Behav. Immun. 2011, 25, 1416–1426. [Google Scholar]
- Bouwens, L.; De Bleser, P.; Vanderkerken, K.; Geerts, B.; Wisse, E. Liver cell heterogeneity: Functions of non-parenchymal cells. Enzyme 1992, 46, 155–168. [Google Scholar]
- Allen, J.W.; Khetani, S.R.; Bhatia, S.N. In vitro zonation and toxicity in a hepatocyte bioreactor. Toxicol. Sci. 2005, 84, 110–119. [Google Scholar]
- Ying, M.; Pey, A.L.; Aarsaether, N.; Martinez, A. Phenylalanine hydroxylase expression in primary rat hepatocytes is modulated by oxygen concentration. Mol. Genet. Metab. 2010, 101, 279–281. [Google Scholar] [CrossRef]
- Ohtake, Y.; Kobayashi, T.; Maruko, A.; Oh-Ishi, N.; Yamamoto, F.; Katoh, S.; Ohkubo, Y. Norepinephrine modulates the zonally different hepatocyte proliferation through the regulation of transglutaminase activity. Am. J. Physiol. Gastrointest. Liver Physiol. 2011, 299, G106–14. [Google Scholar]
- Previs, S.F.; Fernandez, C.A.; Yang, D.; Soloviev, M.V.; David, F.; Brunengraber, H. Limitations of the mass isotopomer distribution analysis of glucose to study gluconeogenesis. Substrate cycling between glycerol and triose phosphates in liver. J. Biol. Chem. 1995, 270, 19806–19815. [Google Scholar]
- Nakao, Y.; Kimura, H.; Sakai, Y.; Fujii, T. Bile canaliculi formation by aligning rat primary hepatocytes in a microfluidic device. Biomicrofluidics 2011, 5, 22212. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Orman, M.A.; Mattick, J.; Androulakis, I.P.; Berthiaume, F.; Ierapetritou, M.G. Stoichiometry Based Steady-State Hepatic Flux Analysis: Computational and Experimental Aspects. Metabolites 2012, 2, 268-291. https://doi.org/10.3390/metabo2010268
Orman MA, Mattick J, Androulakis IP, Berthiaume F, Ierapetritou MG. Stoichiometry Based Steady-State Hepatic Flux Analysis: Computational and Experimental Aspects. Metabolites. 2012; 2(1):268-291. https://doi.org/10.3390/metabo2010268
Chicago/Turabian StyleOrman, Mehmet A., John Mattick, Ioannis P. Androulakis, Francois Berthiaume, and Marianthi G. Ierapetritou. 2012. "Stoichiometry Based Steady-State Hepatic Flux Analysis: Computational and Experimental Aspects" Metabolites 2, no. 1: 268-291. https://doi.org/10.3390/metabo2010268
APA StyleOrman, M. A., Mattick, J., Androulakis, I. P., Berthiaume, F., & Ierapetritou, M. G. (2012). Stoichiometry Based Steady-State Hepatic Flux Analysis: Computational and Experimental Aspects. Metabolites, 2(1), 268-291. https://doi.org/10.3390/metabo2010268




