How Does In Vitro Digestion Change the Amount of Phenolics in Morus alba L. Leaf? Analysis of Preparations and Infusions
Abstract
1. Introduction
2. Materials and Methods
2.1. Material
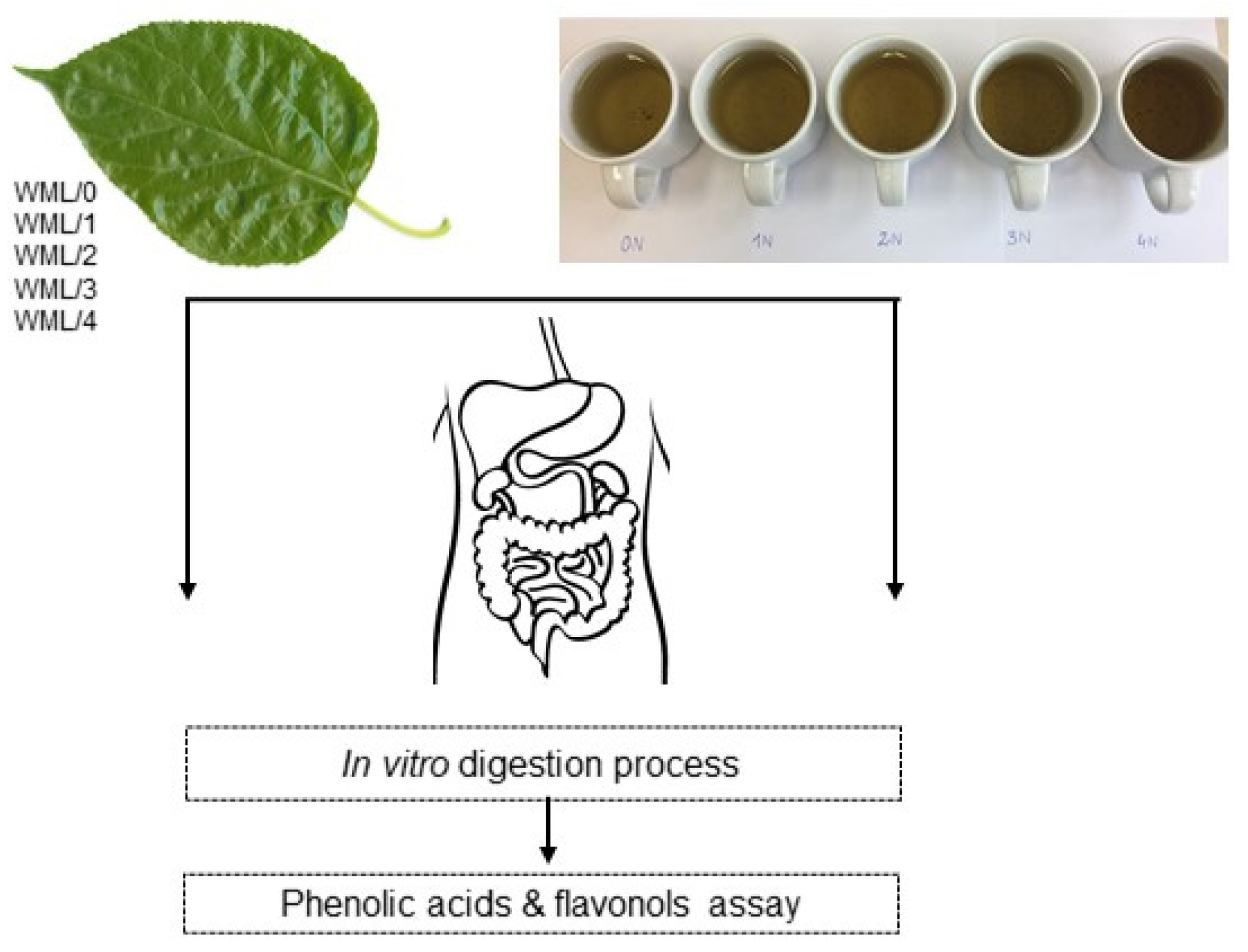
2.2. Conditioning Process of Leaves
- 1 h–conditioned for 1 h;
- 2 h–conditioned for 2 h;
- 3 h–conditioned for 3 h;
- 4 h–conditioned for 4 h.
2.3. Extraction Process
2.4. Infusions Preparation
- 0N–infusion from non-conditioned leaves;
- 1N–infusion from leaves conditioned for 1 h;
- 2N–infusion from leaves conditioned for 2 h;
- 3N–infusion from leaves conditioned for 3 h;
- 4N–infusion from leaves conditioned for 4 h.
2.5. In vitro Simulation of Digestion Process
2.6. High-Performance Liquid Chromatography Assay–Phenolic Acids and Flavonols
2.7. Statistical Analysis
2.8. Reagents
3. Results
3.1. Simulated Digestion of White Mulberry Leaf Preparations
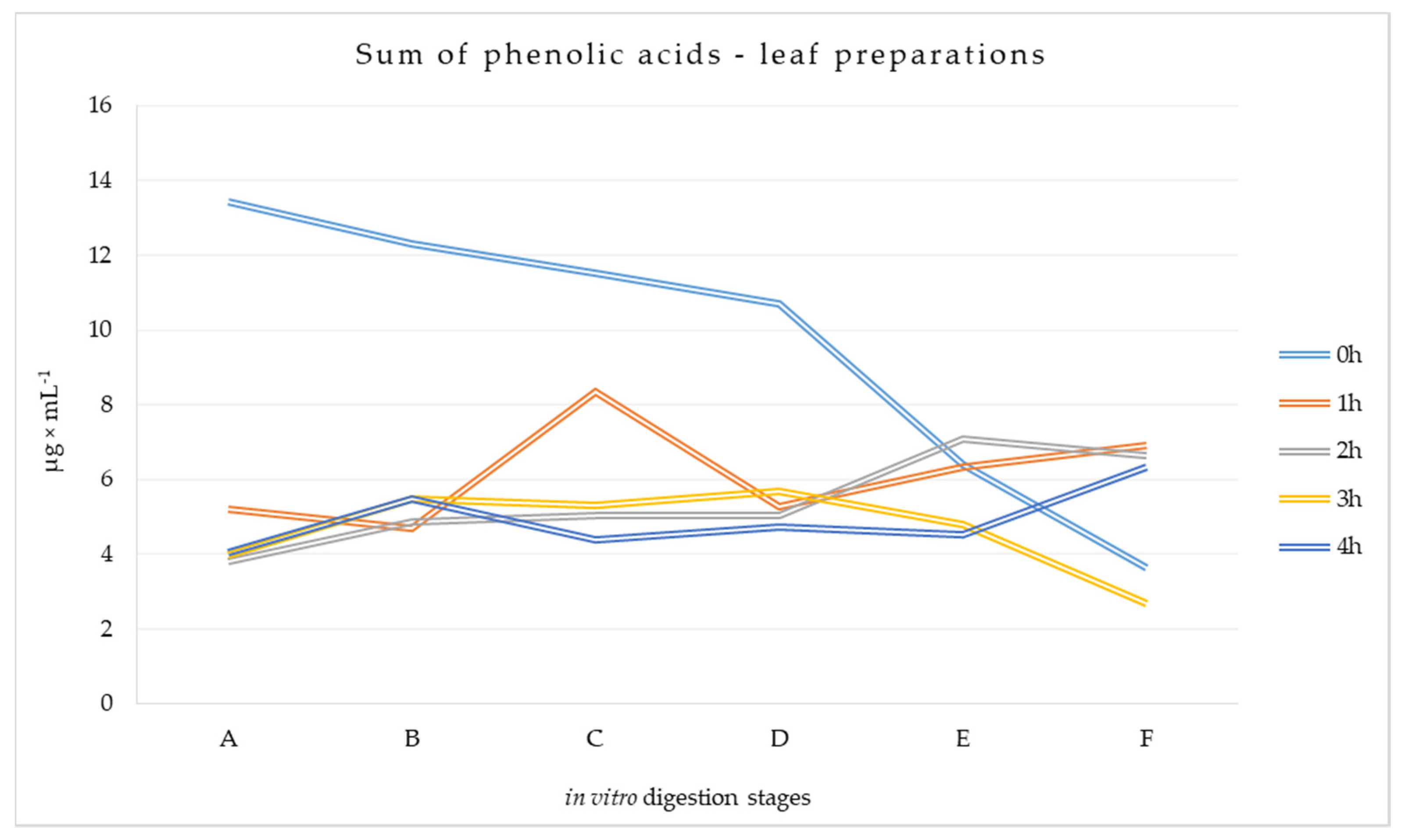
3.1.1. Phenolic Acid Content at Different Stages of Digestion of Leaf Preparations
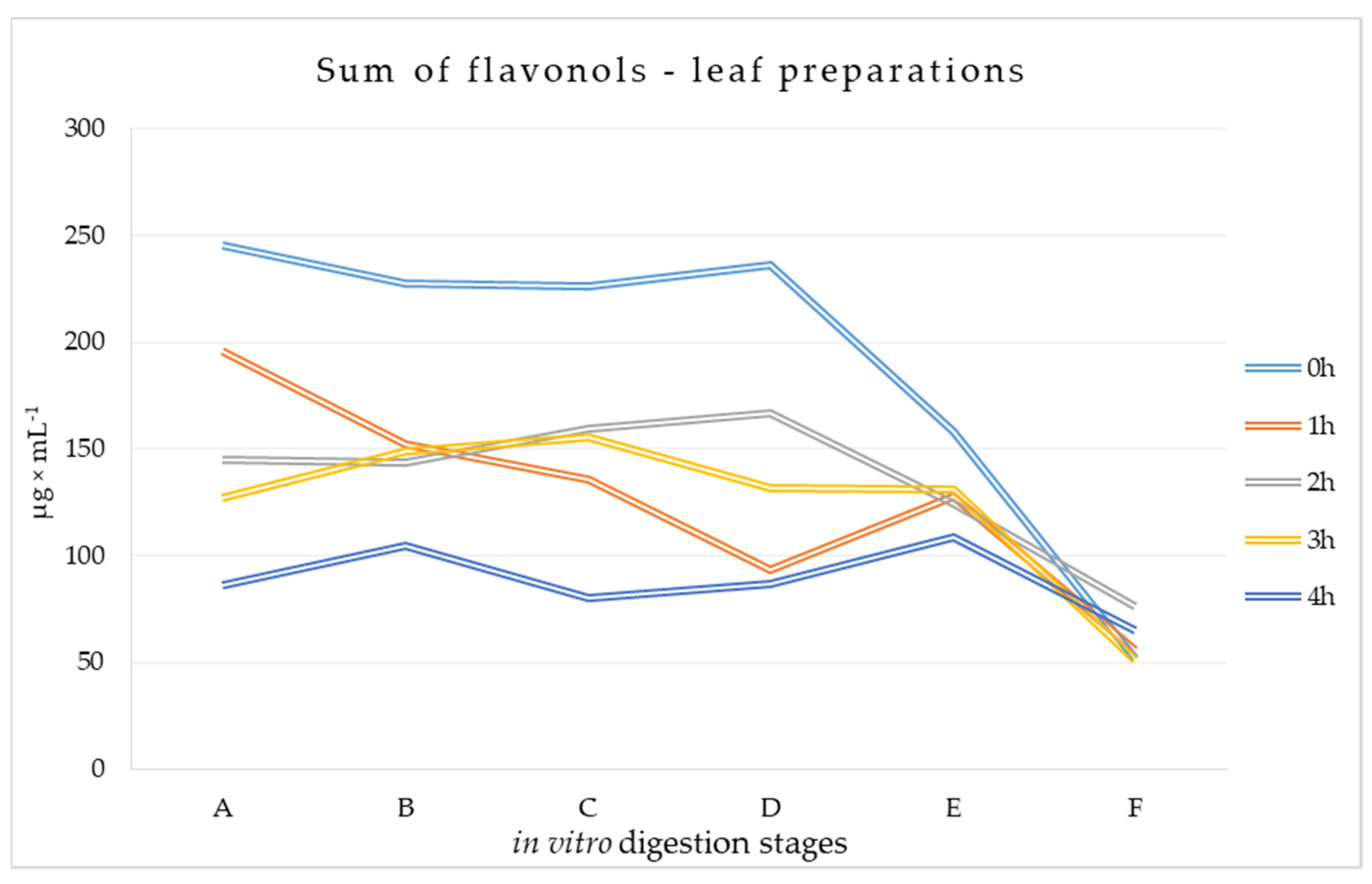
3.1.2. Flavonols Content at Different Stages of Digestion of Leaf Preparations
3.2. Simulated Digestion of White Mulberry Leaf Infusions
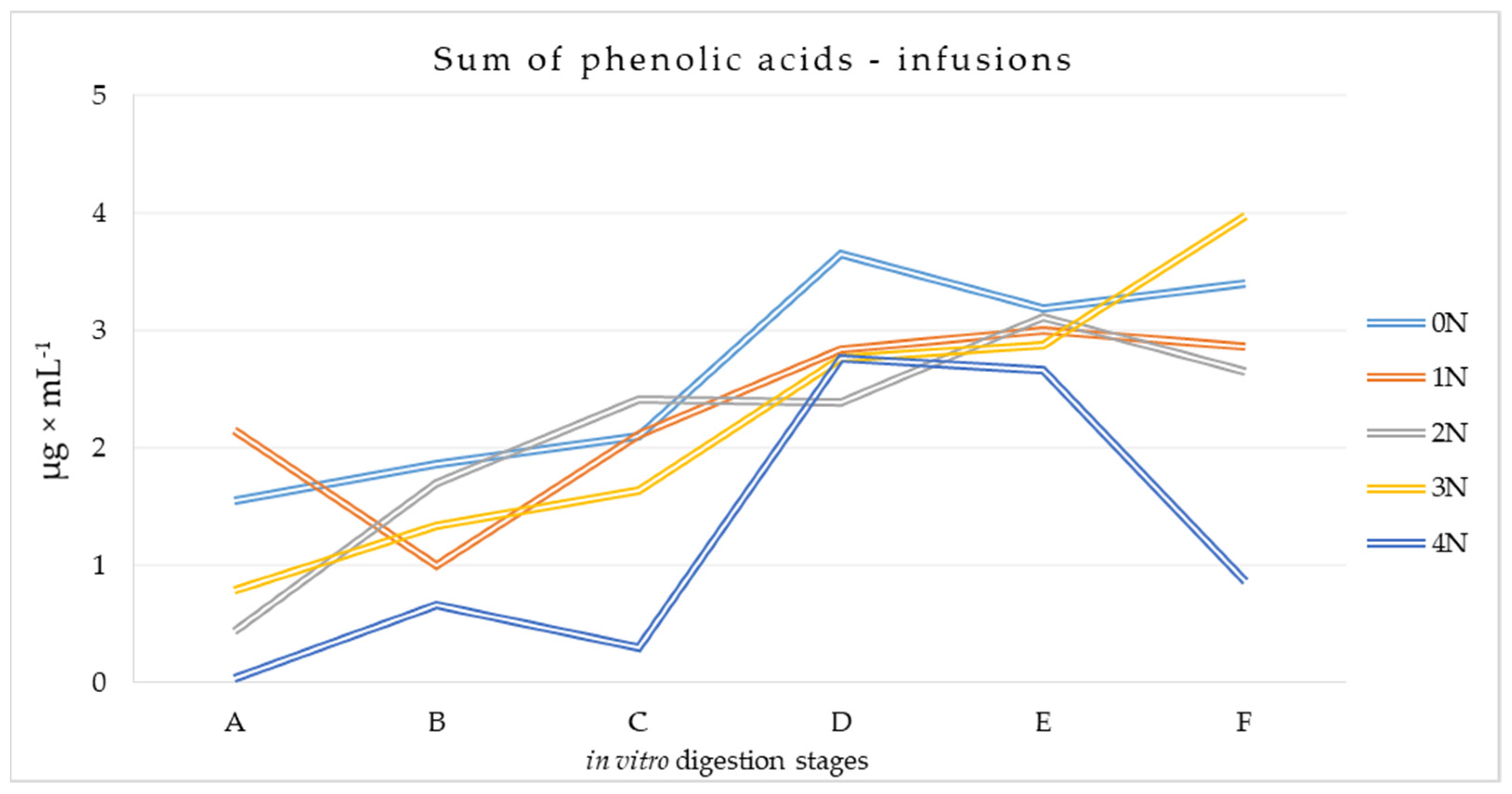
3.2.1. Phenolic Acids Content at Different Stages of the Digestion Process of Mulberry Infusions
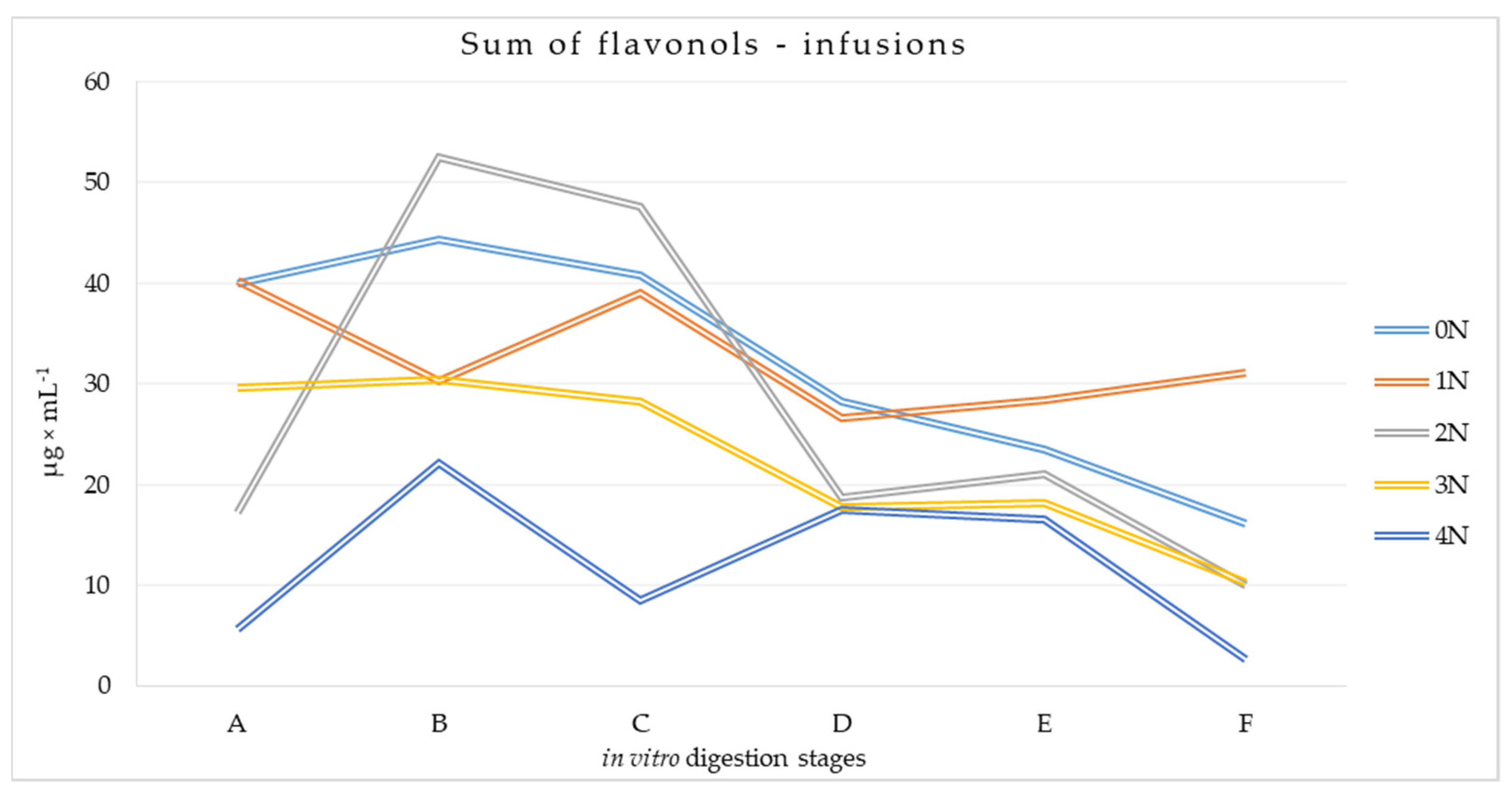
3.2.2. Flavonols Content at Different Stages of the Digestion Process of Mulberry Infusions
4. Discussion
5. Conclusions
Supplementary Materials
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bajpai, S.; Rao, A.V.B.; Muthukumaran, M.; Nagalakshmamma, K. History and Active Pharmacokinetic Principles of Mulberry: A Review. IOSR J. Pharm. 2012, 2, 13–16. [Google Scholar]
- Przeor, M. Some Common Medicinal Plants with Antidiabetic Activity, Known and Available in Europe (A Mini-Review). Pharmaceuticals 2022, 15, 3390. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.H.; Liu, F.; Xiong, L. Medicinal Parts of Mulberry (Leaf, Twig, Root Bark, and Fruit) and Compounds Thereof Are Excellent Traditional Chinese Medicines and Foods for Diabetes Mellitus. J. Funct. Foods 2023, 106, 105619. [Google Scholar] [CrossRef]
- Zou, Y.-X. The Roles of Fermentation Technologies in Mulberry Foods Processing: Application and Outlooks. Med. Chem. (Los. Angeles) 2015, 5, 4–5. [Google Scholar] [CrossRef]
- Sánchez-Salcedo, E.; Mena, P.; García-Viguera, C.; Hernández, F.; Martínez, J.J. (Poly)Phenolic Compounds and Antioxidant Activity of White (Morus alba) and Black (Morus nigra) Mulberry Leaves: Their Potential for New Products Rich in Phytochemicals. J. Funct. Foods 2015, 18, 1039–1046. [Google Scholar] [CrossRef]
- Kemsawasd, V.; Chaikham, P. Survival of Probiotics in Soyoghurt plus Mulberry (c.v. Chiang Mai 60) Leaf Extract during Refrigerated Storage and Their Ability to Tolerate Gastrointestinal Transit. LWT Food Sci. Technol. 2018, 93, 94–101. [Google Scholar] [CrossRef]
- Gültekin-Özgüven, M.; Karadaʇ, A.; Duman, Ş.; Özkal, B.; Özçelik, B. Fortification of Dark Chocolate with Spray Dried Black Mulberry (Morus nigra) Waste Extract Encapsulated in Chitosan-Coated Liposomes and Bioaccessability Studies. Food Chem. 2016, 201, 205–212. [Google Scholar] [CrossRef]
- Kobus-Cisowska, J.; Gramza-Michalowska, A.; Kmiecik, D.; Flaczyk, E.; Korczak, J. Mulberry Fruit as an Antioxidant Component in Muesli. Agric. Sci. 2013, 4, 130–135. [Google Scholar] [CrossRef]
- Przeor, M.; Flaczyk, E. Antioxidant Properties of Paratha Type Flat Bread Enriched with White Mulberry Leaf Extract. Indian J. Tradit. Knowl. 2016, 15, 237–244. [Google Scholar]
- Przeor, M.; Jokiel, M. Morus Alba L. Leaves (WML) Modulate Sweet (TAS1R) and Bitter (TAS2R) Taste in the Studies on—A New Perspective on the Utilization of White Mulberry Leaves in Food Production? Plant Foods Hum. Nutr. 2023. [Google Scholar] [CrossRef]
- Teng, Z.; Yuan, C.; Zhang, F.; Huan, M.; Cao, W.; Li, K.; Yang, J.; Cao, D.; Zhou, S.; Mei, Q. Intestinal Absorption and First-Pass Metabolism of Polyphenol Compounds in Rat and Their Transport Dynamics in Caco-2 Cells. PLoS ONE 2012, 7, e29647. [Google Scholar] [CrossRef] [PubMed]
- Soriano Sancho, R.A.; Pavan, V.; Pastore, G.M. Effect of in vitro Digestion on Bioactive Compounds and Antioxidant Activity of Common Bean Seed Coats. Food Res. Int. 2014, 76, 74–78. [Google Scholar] [CrossRef]
- Szczepaniak, O.; Jokiel, M.; Stuper-Szablewska, K.; Szymanowska, D.; Dziedziński, M.; Kobus-Cisowska, J. Can Cornelian Cherry Mask Bitter Taste of Probiotic Chocolate? Human TAS2R Receptors and a Sensory Study with Comprehensive Characterisation of New Functional Product. PLoS ONE 2021, 16, e0243871. [Google Scholar] [CrossRef] [PubMed]
- Hur, S.J.; Lim, B.O.; Decker, E.A.; McClements, D.J. In vitro Human Digestion Models for Food Applications. Food Chem. 2011, 125, 1–12. [Google Scholar] [CrossRef]
- Tian, W.; Hu, R.; Chen, G.; Zhang, Y.; Wang, W.; Li, Y. Potential Bioaccessibility of Phenolic Acids in Whole Wheat Products during in vitro Gastrointestinal Digestion and Probiotic Fermentation. Food Chem. 2021, 362, 130135. [Google Scholar] [CrossRef] [PubMed]
- Lucas-González, R.; Viuda-Martos, M.; Pérez-Alvarez, J.A.; Fernández-López, J. In vitro Digestion Models Suitable for Foods: Opportunities for New Fields of Application and Challenges. Food Res. Int. 2018, 107, 423–436. [Google Scholar] [CrossRef] [PubMed]
- Abd El-Hack, M.E.; de Oliveira, M.C.; Attia, Y.A.; Kamal, M.; Almohmadi, N.H.; Youssef, I.M.; Khalifa, N.E.; Moustafa, M.; Al-Shehri, M.; Taha, A.E. The Efficacy of Polyphenols as an Antioxidant Agent: An Updated Review. Int. J. Biol. Macromol. 2023, 250, 126525. [Google Scholar] [CrossRef]
- Shahwan, M.; Alhumaydhi, F.; Ashraf, G.M.; Hasan, P.M.Z.; Shamsi, A. Role of Polyphenols in Combating Type 2 Diabetes and Insulin Resistance. Int. J. Biol. Macromol. 2022, 206, 567–579. [Google Scholar] [CrossRef]
- Xie, F.; Yang, W.; Xing, M.; Zhang, H.; Ai, L. Natural Polyphenols-Gut Microbiota Interactions and Effects on Glycolipid Metabolism via Polyphenols-Gut-Brain Axis: A State-of-the-Art Review. Trends Food Sci. Technol. 2023, 140, 104171. [Google Scholar] [CrossRef]
- Przeor, M.; Flaczyk, E.; Beszterda, M.; Szymandera-Buszka, K.E.; Piechocka, J.; Kmiecik, D.; Szczepaniak, O.; Kobus-Cisowska, J.; Jarzębski, M.; Tylewicz, U. Air-Drying Temperature Changes the Content of the Phenolic Acids and Flavonols in White Mulberry (Morus Alba l.) Leaves. Cienc. Rural 2019, 49, e20190489. [Google Scholar] [CrossRef]
- Stuper-Szablewska, K.; Kurasiak-Popowska, D.; Nawracała, J.; Perkowski, J. Quantitative Profile of Phenolic Acids and Antioxidant Activity of Wheat Grain Exposed to Stress. Eur. Food Res. Technol. 2019, 245, 1595–1603. [Google Scholar] [CrossRef]
- Dżugan, M.; Miłek, M.; Grabek-Lejko, D.; Hęclik, J.; Jacek, B.; Litwińczuk, W. Antioxidant Activity, Polyphenolic Profiles and Antibacterial Properties of Leaf Extract of Various Paulownia spp. Clones. Agronomy 2021, 11, 2001. [Google Scholar] [CrossRef]
- Przeor, M.; Flaczyk, E.; Kmiecik, D.; Buchowski, M.S.; Staniek, H.; Tomczak-Graczyk, A.; Kobus-Cisowska, J.; Gramza-Michałowska, A.; Foksowicz-Flaczyk, J. Functional Properties and Antioxidant Activity of Morus alba L. Leaves Var. Zolwinska Wielkolistna (WML-P)—Tthe Effect of Controlled Conditioning Process. Antioxidants 2020, 9, 668. [Google Scholar] [CrossRef] [PubMed]
- Łochyńska, M. Wymagania i Agrotechnika Polskiej Odmiany Morwy Białej “Żółwińska Wielkolistna”. Zagadnienia Doradz. Rol. 2018, 4, 99–111. [Google Scholar]
- Lee, W.J.; Choi, S.W. Quantitative Changes of Polyphenolic Compounds in Mulberry (Morus alba L.) Leaves in Relation to Varieties, Harvest Period, and Heat Processing. Prev. Nutr. food Sci. 2012, 17, 280–285. [Google Scholar] [CrossRef] [PubMed]
- Enkhmaa, B.; Shiwaku, K.; Katsube, T.; Kitajima, K.; Anuurad, E.; Yamasaki, M.; Yamane, Y. Mulberry (Morus alba L.) Leaves and Their Major Flavonol Quercetin 3-(6-Malonylglucoside) Attenuate Atherosclerotic Lesion Development in LDL Receptor-Deficient Mice. J. Nutr. 2005, 135, 729–734. [Google Scholar] [CrossRef]
- Katsube, T.; Tsurunaga, Y.; Sugiyama, M.; Furuno, T.; Yamasaki, Y. Effect of Air-Drying Temperature on Antioxidant Capacity and Stability of Polyphenolic Compounds in Mulberry (Morus alba L.) Leaves. Food Chem. 2009, 113, 964–969. [Google Scholar] [CrossRef]
- Hunyadi, A.; Martins, A.; Hsieh, T.-J.; Seres, A.; Zupkó, I. Chlorogenic Acid and Rutin Play a Major Role in the in vivo Anti-Diabetic Activity of Morus Alba Leaf Extract on Type II Diabetic Rats. PLoS ONE 2012, 7, e50619. [Google Scholar] [CrossRef]
- Kim, Y.; Goodner, K.L.; Park, J.D.; Choi, J.; Talcott, S.T. Changes in Antioxidant Phytochemicals and Volatile Composition of Camellia Sinensis by Oxidation during Tea Fermentation. Food Chem. 2011, 129, 1331–1342. [Google Scholar] [CrossRef]
- Thabti, I.; Elfalleh, W.; Hannachi, H.; Ferchichi, A.; Campos, M.D.G. Identification and Quantification of Phenolic Acids and Flavonol Glycosides in Tunisian Morus Species by HPLC-DAD and HPLC-MS. J. Funct. Foods 2012, 4, 367–374. [Google Scholar] [CrossRef]
- Gupta, J.; Gupta, A. Isolation and Identification of Flavonoid Rutin from Rauwolfia Serpentina. Int. J. Chem. Stud. 2015, 3, 113–115. [Google Scholar]
- Kamiloglu, S.; Capanoglu, E.; Bilen, F.D.; Gonzales, G.B.; Grootaert, C.; Van de Wiele, T.; Van Camp, J. Bioaccessibility of Polyphenols from Plant-Processing Byproducts of Black Carrot (Daucus carota L.). J. Agric. Food Chem. 2016, 64, 2450–2458. [Google Scholar] [CrossRef] [PubMed]
- Siracusa, V.; Blanco, I.; Romani, S.; Tylewicz, U.; Dalla Rosa, M. Gas Permeability and Thermal Behavior of Polypropylene Films Used for Packaging Minimally Processed Fresh-Cut Potatoes: A Case Study. J. Food Sci. 2012. [Google Scholar] [CrossRef] [PubMed]
- Pineda-Vadillo, C.; Nau, F.; Dubiard, C.; Cheynier, V.; Meudec, E.; Sanz-Buenhombre, M.; Guadarrama, A.; Tóth, T.; Csavajda, E.; Hingyi, H.; et al. In vitro Digestion of Dairy and Egg Products Enriched with Grape Extracts: Effect of the Food Matrix on Polyphenol Bioaccessibility and Antioxidant Activity. Food Res. Int. 2016, 88, 284–292. [Google Scholar] [CrossRef]
- Parkar, S.G.; Trower, T.M.; Stevenson, D.E. Fecal Microbial Metabolism of Polyphenols and Its Effects on Human Gut Microbiota. Anaerobe 2013, 23, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Sadeghi Ekbatan, S.; Sleno, L.; Sabally, K.; Khairallah, J.; Azadi, B.; Rodes, L.; Prakash, S.; Donnelly, D.J.; Kubow, S. Biotransformation of Polyphenols in a Dynamic Multistage Gastrointestinal Model. Food Chem. 2016, 204, 453–462. [Google Scholar] [CrossRef] [PubMed]
- Stalmach, A.; Mullen, W.; Barron, D.; Uchida, K.; Yokota, T.; Cavin, C.; Steiling, H.; Williamson, G.; Crozier, A. Metabolite Profiling of Hydroxycinnamate Derivatives in Plasma and Urine after the Ingestion of Coffee by Humans: Identification of Biomarkers of Coffee Consumption. Drug Metab. Dispos. 2009, 37, 1749–1758. [Google Scholar] [CrossRef]
- Tagliazucchi, D.; Verzelloni, E.; Bertolini, D.; Conte, A. In vitro Bio-Accessibility and Antioxidant Activity of Grape Polyphenols. Food Chem. 2010, 120, 599–606. [Google Scholar] [CrossRef]
- Rechner, A.R.; Smith, M.A.; Kuhnle, G.; Gibson, G.R.; Debnam, E.S.; Srai, S.K.S.; Moore, K.P.; Rice-Evans, C.A. Colonic Metabolism of Dietary Polyphenols: Influence of Structure on Microbial Fermentation Products. Free Radic. Biol. Med. 2004, 36, 212–225. [Google Scholar] [CrossRef]
- Celep, E.; Charehsaz, M.; Akyüz, S.; Acar, E.T.; Yesilada, E. Effect of in vitro Gastrointestinal Digestion on the Bioavailability of Phenolic Components and the Antioxidant Potentials of Some Turkish Fruit Wines. Food Res. Int. 2015, 78, 209–215. [Google Scholar] [CrossRef]
- Ozkan, G.; Kamiloglu, S.; Ozdal, T.; Boyacioglu, D.; Capanoglu, E. Potential Use of Turkish Medicinal Plants in the Treatment of Various Diseases. Molecules 2016, 21, 257. [Google Scholar] [CrossRef] [PubMed]
- Duda-Chodak, A.; Tarko, T.; Satora, P.; Sroka, P. Interaction of Dietary Compounds, Especially Polyphenols, with the Intestinal Microbiota: A Review. Eur. J. Nutr. 2015, 54, 325–341. [Google Scholar] [CrossRef] [PubMed]
- Bouayed, J.; Hoffmann, L.; Bohn, T. Total Phenolics, Flavonoids, Anthocyanins and Antioxidant Activity Following Simulated Gastro-Intestinal Digestion and Dialysis of Apple Varieties: Bioaccessibility and Potential Uptake. Food Chem. 2011, 128, 14–21. [Google Scholar] [CrossRef] [PubMed]
| Digestion Stage | Actor | Action Time | pH |
|---|---|---|---|
| Stomach | Pepsin | 2 h | 2.0 |
| Duodenum | Pancreatic extract + bile acids | 0.5 h | 6.0 |
| Small intestine | Intestine microflora | 2 h | 7.4 |
| Large intestine | 18 h | 8.0 |
| Phenolic Acids | Conditioning Process | Digestion Stage | ||||
|---|---|---|---|---|---|---|
| Spearman Rank Order | Gamma | τ Kendall | Spearman Rank Order | Gamma | τ Kendall | |
| GAL | 0.111 | 0.065 | 0.058 | 0.201 | 0.173 * | 0.159 * |
| PRO | 0.612 * | 0.539 * | 0.485 * | −0.559 * | −0.469 * | −0.431 * |
| HYD | 0.548 * | 0.481 * | 0.433 * | 0.280 * | 0.220 * | 0.202 * |
| VAN | 0.276 * | 0.205 * | 0.184 * | 0.489 * | 0.417 * | 0.383 * |
| CHL | 0.217 * | 0.154 | 0.138 | −0.582 * | −0.492 * | −0.452 * |
| CAF | 0.249 * | 0.198 * | 0.178 * | 0.167 | 0.129 | 0.119 |
| SYR | 0.132 | 0.101 | 0.089 | −0.840 * | −0.760 * | −0.701 * |
| COU | 0.152 | 0.138 | 0.124 | −0.072 | −0.054 | −0.050 |
| FER | 0.022 | −0.033 | −0.030 | −0.136 | −0.121 | −0.111 |
| SIN | 0.328 * | 0.240 * | 0.216 * | −0.391 * | −0.321 * | −0.295 * |
| sum | −0.601 * | −0.112 | ||||
| Flavonol | Conditioning Process | Digestion Stage | ||||
|---|---|---|---|---|---|---|
| Spearman Rank Order | Gamma | τ Kendall | Spearman Rank Order | Gamma | τ Kendall | |
| RUT | 0.022 | −0.034 | −0.031 | −0.678 * | −0.575 * | −0.528 * |
| AST | 0.273 * | 0.244 * | 0.219 * | −0.046 | −0.051 | −0.047 |
| QUE | 0.462 * | 0.388 * | 0.349 * | 0.211 * | 0.163 * | 0.149 * |
| KEM | −0.022 | −0.023 | −0.021 | −0.135 | −0.102 | −0.094 |
| ISR | −0.002 | 0.017 | 0.015 | −0.047 * | −0.374 * | −0.344 |
| Pearson | ||||||
| ISQ | 0.350 * p = 0.001 | −0.433 * p = 0.000 | ||||
| MAL | 0.200 p = 0.059 | −0.424 * p = 0.000 | ||||
| MYR | 0.058 p = 0.590 | −0.305 * p = 0.003 | ||||
| Sum | −0.541 * | −0.540 * | ||||
| Phenolic Acids | Conditioning Process | Digestion Stage | ||||
|---|---|---|---|---|---|---|
| Spearman Rank Order | Gamma | τ Kendall | Spearman Rank Order | Gamma | τ Kendall | |
| GAL | −0.201 | −0.162 * | −0.145 * | 0.767 * | 0.661 * | 0.607 * |
| PRO | −0.647 * | −0.565 * | −0.504 * | −0.186 | −0.142 | −0.129 |
| HYD | −0.147 | −0.089 | −0.079 | 0.247 * | 0.208 * | 0.186 * |
| VAN | −0.117 | −0.095 | −0.083 | 0.127 | 0.106 | 0.093 |
| CHL | −0.597 * | −0.496 * | −0.447 * | −0.449 * | −0.377 * | −0.346 * |
| CAF | −0.813 * | −0.760 * | −0.684 * | 0.189 | 0.156 * | 0.143 * |
| SYR | −0.458 * | −0.579 * | −0.375 * | −0.355 * | −0.462 * | −0.302 * |
| COU | −0.594 * | −0.503 * | −0.459 * | −0.192 | −0.161 * | −0.147 * |
| FER | −0.818 * | −0.732 * | −0.666 * | −0.296 * | −0.241 * | −0.221 * |
| SIN | −0.499 * | −1.000 * | −0.437 * | −0.264 * | −0.521 * | −0.221 * |
| sum | −0.402 * | 0.699 * | ||||
| Flavonol | Conditioning Process | Digestion Stage | ||||
|---|---|---|---|---|---|---|
| Spearman Rank Order | Gamma | τ Kendall | Spearman Rank Order | Gamma | τ Kendall | |
| RUT | −0.639 * | −0.550 * | −0.495 * | −0.463 * | −0.391 * | −0.359 * |
| KEM | −0.622 * | −0.521 * | −0.468 * | −0.366 * | −0.305 * | −0.281 * |
| ISQ | −0.435 * | −0.353 * | −0.318 * | −0.572 * | −0.477 * | −0.438 * |
| MAL | −0.551 * | −0.452 * | −0.406 * | −0.402 * | −0.340 * | −0.313 * |
| AST | −0.609 * | −0.537 * | −0.453 * | −0.399 * | −0.348 * | −0.319 * |
| MYR | −0.589 * | −0.470 * | −0.423 * | −0.319 * | −0.276 * | −0.253 * |
| QUE | −0.412 * | −0.333 * | −0.299 * | −0.458 * | −0.371 * | −0.341 * |
| sum | −0.567 * | −0.454 * | ||||
| Test | Digested Preparations/Digested Infusions |
|---|---|
| flavonols | 0.661 * |
| phenolic acids | 0.094 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Przeor, M. How Does In Vitro Digestion Change the Amount of Phenolics in Morus alba L. Leaf? Analysis of Preparations and Infusions. Metabolites 2024, 14, 31. https://doi.org/10.3390/metabo14010031
Przeor M. How Does In Vitro Digestion Change the Amount of Phenolics in Morus alba L. Leaf? Analysis of Preparations and Infusions. Metabolites. 2024; 14(1):31. https://doi.org/10.3390/metabo14010031
Chicago/Turabian StylePrzeor, Monika. 2024. "How Does In Vitro Digestion Change the Amount of Phenolics in Morus alba L. Leaf? Analysis of Preparations and Infusions" Metabolites 14, no. 1: 31. https://doi.org/10.3390/metabo14010031
APA StylePrzeor, M. (2024). How Does In Vitro Digestion Change the Amount of Phenolics in Morus alba L. Leaf? Analysis of Preparations and Infusions. Metabolites, 14(1), 31. https://doi.org/10.3390/metabo14010031