Abstract
Alzheimer’s disease (AD) is a progressive neurodegenerative disorder associated with cognitive decline and characterized by amyloid-β plaques and neurofibrillary tau tangles. Although AD’s exact pathophysiology remains unclear, oxidative stress is known to play a role in the neurodegenerative process. Since no curative treatment exists, antioxidants represent a potential treatment for AD due to their ability to modulate oxidative stress. Therefore, this review aims to examine the impact of antioxidant supplementation and its potential mechanisms on cognitive function. The review primarily discusses research articles published between 2012 and 2022 reporting the results of clinical trials involving antioxidant supplementation on cognitive function in individuals with AD. Antioxidant supplementation included probiotics, selenium, melatonin, resveratrol, rosmarinic acid, carotenoids, curcumin, vitamin E, and coenzyme Q. While the studies included in this review did not provide much evidence for the beneficial role of antioxidant supplements on cognitive function in AD, the results varied from antioxidant to antioxidant and among trials examining the same antioxidant. Furthermore, many of the studies’ findings face several limitations, including short trial durations, small sample sizes, and a lack of diversity among study participants. As a result, more research is required to examine the impact of antioxidant supplementation on cognitive function in AD.
Keywords:
Alzheimer’s disease; antioxidants; coenzyme Q; curcumin; melatonin; probiotics; resveratrol; rosmarinic acid; selenium; vitamin E 1. Introduction
Globally, 697 individuals over the age of fifty suffer from dementia per 100,000 persons, with the prevalence of Alzheimer’s Disease (AD) being 324 per 10,000 [1]. In Europe, an estimated 7.1% of the population has AD, with the prevalence being higher in females than males and in individuals with an older age [2]. In 2020, more than 6 million adults in the United States had AD, with the prevalence forecast to exceed 13 million by 2060 [3]. During 2020, the healthcare costs associated with AD in the United States reached $305 billion, and the economic burden is expected to increase with the aging population [4]. AD affects an individual’s quality of life and that of those who care for them [4,5,6]. As a result, treating AD is critical to improving the health and well-being of individuals with AD and those who care for them.
AD is a multifactorial neurodegenerative disorder associated with a gradual decline in cognitive function and characterized by extracellular amyloid-β (Aβ) plaques and intracellular neurofibrillary tau tangles (NFT) [7,8]. Since years may pass before the onset of neurodegeneration leads to the development of cognitive decline, AD represents a continuum in which an affected individual progresses from normal cognitive function to mild cognitive impairment (MCI) and, finally, dementia [8,9,10]. Since a definitive diagnosis of AD cannot be made without an autopsy, establishing biomarkers for diagnosing AD, particularly in the early phases of the disease, is critical for early diagnosis and detection [7]. For example, several trials indicated that cerebral spinal fluid (CSF) Aβ42 levels decreased and tau levels increased with disease progression [11].
While Aβ plaques and NFT represent the histopathological hallmarks of AD, they may be the result of other neurodegenerative processes and not the initiating factor in AD [9]. The formation of Aβ plaques involves the cleavage of the single transmembrane protein, amyloid precursor protein (APP) [12]. In the non-amyloidogenic pathway, APP is cleaved by α-secretase to soluble APPα, while in the amyloidogenic pathway, APP is cleaved by β-secretase and γ-secretase to Aβ [12,13,14]. Mutations in presenilin-1 (PS1) and PS2, the catalytic subunits of γ-secretase, are associated with increased Aβ production [14]. NFTs are primarily composed of hyperphosphorylated tau, a microtubule-associated protein that can interact with other hyperphosphorylated tau proteins to form paired helical filaments [7,15]. Nevertheless, the exact role of Aβ and NFT in the pathophysiology of AD remains unclear.
Although the exact pathophysiology of AD is uncertain, mechanisms involved in neurodegeneration may include oxidative stress [7]. Oxidative stress occurs when the generation of reactive oxygen species (ROS) and reactive nitrogen species exceeds the body’s antioxidant defense systems [16,17,18,19]. Aging is associated with a decline in antioxidant enzymes and increased oxidative stress and inflammation, which may contribute to the development of chronic diseases [20]. Compared to other cells in the body, neurons possess more polyunsaturated fatty acids in their cell membranes, and their higher metabolic rate results in greater oxygen consumption [21]. As a result, the brain is particularly vulnerable to oxidative stress, which can damage lipids, proteins, and deoxyribonucleic acid (DNA) in cells [19,21,22]. Moreover, higher levels of serum malondialdehyde (MDA), a marker of lipid peroxidation, are observed in individuals with AD compared to healthy controls [23]. In addition to lipid, protein, and DNA damage, oxidative stress may contribute to the aggregation and misfolding of Aβ plaques and NFT [24]. There are several potential sources of ROS in AD. Research indicates that Aβ plaques, mitochondrial dysfunction, metal dyshomeostasis (iron, copper, and zinc), and inflammation may contribute to the generation of ROS [21,25,26,27,28,29]. For example, copper and iron can accumulate in Aβ plaques and contribute to the generation of ROS [25]. Additionally, neuroinflammation involves microglia activating nicotinamide adenine dinucleotide phosphate oxidase, which also generates ROS [27]. Overall, oxidative stress contributes to neuron damage and cell death [24]. Therefore, oxidative stress plays an important role in the neurodegeneration associated with AD.
Currently, there is no cure for AD, and treatment focuses on alleviating symptoms. Therapeutic drugs include cholinesterase inhibitors (donepezil, rivastigmine, and galantamine), N-methyl-D-aspartate receptor antagonists (memantine), and anti-Aβ monoclonal antibodies (aducanumab) [30,31,32,33]. During the pathophysiology of AD, cholinergic neuron death is thought to contribute to the decline in cognitive function [34]. Cholinesterase inhibitors function by preventing the enzyme acetylcholinesterase from breaking down the neurotransmitter acetylcholine [31,34,35]. As a result, more acetylcholine remains in the synaptic cleft, thus improving cholinergic neuron activity and cognitive function [34,36]. In AD, N-methyl-D-aspartate receptors are thought to be overactivated by the neurotransmitter glutamate, making it difficult for the brain to distinguish normal glutamate signaling [37]. N-methyl-D-aspartate receptor antagonists may operate by reducing the activation of the receptors by glutamate, thus improving cognitive function [37]. In addition to only alleviating AD symptoms, cholinesterase inhibitors and N-methyl-D-aspartate antagonists are associated with adverse side effects (decreased appetite, diarrhea, dizziness, insomnia, and nausea) and drug interactions [31,38]. Anti-Aβ monoclonal antibodies are thought to facilitate Aβ clearance from the CSF [32,33]. The Food and Drug Administration recently approved the anti-Aβ monoclonal antibody, aducanumab, for use in mild AD treatment [32,33]. Despite data indicating that aducanumab reduces Aβ plaques, current data on the impact of aducanumab on cognitive function is inconclusive, and the drug is associated with cerebral edema and intracerebral hemorrhages [32,33]. Given the limitations of the current treatments for AD, more research is needed to identify and develop strategies for modifying disease progression.
Since oxidative stress is involved in the pathophysiology of AD, antioxidants represent a potential treatment for AD. A previous review by Pritam et al. [39] discussed a wide range of antioxidants with therapeutic potential in AD but lacked information on the results of recent clinical trials. Therefore, this review aims to examine the impact of antioxidant supplementation and its potential mechanisms on cognitive function in individuals with AD. PubMed searches were used to identify articles published in English between 2012 and 2022, reporting the results of clinical trials that involved antioxidant supplementation in individuals with AD and included cognitive function as an outcome. The following review discusses the results of clinical trials involving supplementation with probiotics [40,41,42,43], selenium [44,45,46], melatonin [47], resveratrol [48,49,50,51], rosmarinic acid [52], carotenoids [53,54,55], curcumin [56], vitamin E [57,58,59,60], and coenzyme Q [58] (Table 1).

Table 1.
Summary of Clinical Trials on Antioxidant Supplementation in Individuals with AD.
2. Probiotics
According to the Food and Agriculture Organization of the United Nations and the World Health Organization, the term ‘probiotics’ refers to “live microorganisms that, when administered in adequate amounts, confer a health benefit on the host” [61,62]. The beneficial role of probiotics stems from the ability of bacteria to modify the composition and, thus, the activity of the gut microbiome (the microorganisms residing along the host’s gastrointestinal tract) [63,64]. The ability of the gut microbiome to communicate with the central nervous system via the gut-brain axis has led to an interest in using probiotics to treat neurological diseases [65,66,67]. Additionally, a systematic review and meta-analysis by Hung et al. [68] observed a significant reduction in the diversity of the gut microbiome of individuals with AD compared to healthy controls. Furthermore, pathogenic microorganisms may contribute to the pathophysiology of AD through the generation of lipopolysaccharides and bacterial amyloid fiber [69,70]. The beneficial effects of probiotics can depend on their strain, but research has linked some species of probiotic bacteria to antioxidant defense [71]. For example, administering Lactobacillus reuteri, Lactobacillus rhamnosus, and Bifidobacterium infantis alleviated oxidative stress and inflammation in rats treated with Aβ1–40 [72]. Therefore, probiotics with potential antioxidant functions are of interest for possibly treating AD. Four studies [40,41,42,43] examined the impact of probiotic supplementation among individuals with AD.
In Iran, Akbari et al. [40] examined the consumption of 200 mL/day of milk either with (n = 30) or without (n = 30) probiotic supplementation (2 × 109 CFU/g of Lactobacillus acidophilus, Lactobacillus casei, Lactobacillus fermentum, and Bifidobacterium bifidum) for 12 weeks in individuals with AD. After treatment, the probiotic group experienced a significant improvement in cognitive function compared to the control group, as indicated by an increase in Mini-Mental State Examination (MMSE) scores [40]. Additionally, the probiotic group experienced a significant improvement in high-sensitivity C-reactive protein (hs-CRP), the homeostatic model of assessment of insulin resistance (HOMA-IR), the homeostatic model of assessment for B-cell function (HOMA-B), MDA, the quantitative insulin sensitivity check index (QUICKI), triglycerides, and very low-density lipoprotein [40]. As a result, the study indicated that probiotic supplementation might improve cognitive function among individuals with AD by improving the lipid profile and decreasing insulin resistance and oxidative stress. However, no significant differences between the two groups were observed in total antioxidant capacity (TAC), glutathione, nitric oxide (NO), fasting blood glucose, low-density lipoprotein, high-density lipoprotein, total cholesterol, or body mass index after 12 weeks of treatment [40]. The study’s small sample size, short time frame, and lack of data on the host’s microbiome before and after supplementation limit the study’s results.
Furthermore, in Iran, Agahi et al. [41] examined the effects of probiotic supplementation on cognitive function using the Test Your Memory (TYM) test modified for an Iranian population. Men and women with AD were randomized to receive either a placebo (n = 30) or a probiotic (n = 30) for 12 weeks [41]. The probiotic group received two capsules every other day, each containing 3 × 109 CFU of bacteria. The first capsule contained Lactobacillus fermentum, Lactobacillus plantarum, and Bifidobacterium lactis, while the second capsule contained Lactobacillus acidophilus, Bifidobacterium bifidum, and Bifidobacterium longum [41]. At the end of 12 weeks, 23 participants remained in the control group and 25 in the probiotic group and were included in the final analysis [41]. After 12 weeks of treatment, statistical analysis indicated a significant difference between the two groups for markers of oxidative stress (TAC, MDA, and NO), but post hoc analysis made these differences negligible [41]. Additionally, no significant differences were observed between the two groups for cognitive function (TYM), markers of inflammation (tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), IL-10, or other markers of oxidative stress (total glutathione and 8-hydroxy-2′-deoxyguanosine) [41]. As a result, the study does not support the role of probiotics in improving cognitive function or markers of inflammation and oxidative stress in AD. Nevertheless, the authors [41] noted that the difference in the study results with those of Akbari et al. [40] could be accounted for by the different supplements and the higher percentage of participants with severe AD.
In Austria, Leblhuber et al. [42] examined the impacts of probiotic supplementation on cognitive function and inflammation. Twenty individuals with AD received 3 g of a probiotic supplement (Lactobacillus casei, Lactococcus lactis, Lactobacillus acidophilus, Bifidobacterium lactis, Lactobacillus paracasei, Lactobacillus plantarum, Bifidobacterium lactis, Bifidobacterium bifidum, and Lactobacillus salivarius) once daily for four weeks [42,73]. After the study, paired serum samples were available for 15 participants, and paired stool samples were available for 18 participants [42]. After four weeks of treatment, no significant changes were observed in cognitive function (MMSE, clock drawing test) [42]. Although the lack of change in cognitive function may indicate that the probiotic supplementation delayed cognitive decline, the study lacked a control group, and four weeks may not be sufficient for observing changes in cognitive function. While analysis of fecal ribonucleic acid (RNA) indicated that the concentration of Faecalibacterium prausnitzii increased after four weeks of treatment, no changes were observed for Clostridium cluster I or Akkermansia muciniphila RNA [42]. Additionally, a significant decline in fecal zonulin was observed, indicating a possible improvement in gut permeability [42]. Although no significant changes were observed in the ratio of serum kynurenine/tryptophan (an indicator of tryptophan breakdown), the levels of serum kynurenine increased, indicating a potential stimulation of the body’s immune system [42]. Despite the study being limited by its small sample size, short trial duration, and lack of a control group, Leblhuber et al.’s [42] data indicate that probiotic supplementation may modulate the composition of the gut microbiota and impact systemic inflammation.
In Brazil, Ton et al. [43] examined the impact of 90-day supplementation with kefir-fermented milk containing Acetobacter aceti, Acetobacter sp., Candida famata, Candida krusei, Enterococcus faecium, Lactobacillus delbrueckii delbrueckii, Lactobacillus fermentum, Lactobacillus fructivorans, Lactobacillus kefiranofaciens, and Leuconostoc spp. in individuals with AD taking 10 mg of donepezil. While 16 individuals with AD were initially enrolled in the study and received the intervention (2 mL/kg/day), only 13 completed the study and were included in the final analysis [43]. After 90 days of treatment, Ton et al. [43] observed a significant improvement in cognitive status (MMSE). In addition to examining the impact of probiotic consumption on cognitive function, the impact on measures of inflammation and oxidative stress using blood samples was examined [43]. Probiotic supplementation improved inflammation as indicated by a significant decline in TNF-α, IL-8, and IL12p70 (proinflammatory cytokines), but no change in IL-1b and IL-6 (proinflammatory cytokines) or IL-10 (anti-inflammatory cytokine) levels were observed [43]. Furthermore, serum oxidative stress decreased, as indicated by a decline in ROS (superoxide anion, hydrogen peroxide, and peroxynitrite/hydroxyl radical), a decline in advanced oxidative protein products, and an increase in NO [43]. While the study by Ton et al. [43] indicated that probiotic supplementation could potentially improve cognitive function through a reduction in inflammation and oxidative stress, the results of this study are limited by the small sample size and lack of a control group. Additionally, changes in serum inflammation and oxidative stress markers may not represent changes in brain tissue. The study also lacked data on the composition of the host’s microbiome before and after treatment. Nevertheless, the study supports the potential benefits of probiotic consumption in AD.
While both the studies by Agahi et al. [41] and Leblhuber et al. [42] observed no impact of probiotic supplementation on cognitive function, the studies by Akbari et al. [40] and Ton et al. [43] observed that probiotic supplementation could improve MMSE scores and provided some insight into the potential mechanisms behind this improvement. While Akbari et al. [40] observed no change in serum NO levels, Ton et al. [43] observed a significant increase in NO levels. As mentioned above, the beneficial effects of probiotics may be specific to some bacterial species [71]. As a result, the different probiotic compositions could account for the differences between the two studies. While NO contributes to vasodilation, inhibits platelet aggregation, and functions as a neurotransmitter, it also plays a role in inflammation and can damage cells, particularly after reacting with superoxide anions to generate peroxynitrite anions [74]. Therefore, the impact of increased NO levels on AD in the study by Ton et al. [43] is unclear. Both studies observed a potential beneficial mechanism through the reduction of inflammation, with Ton et al. [43] reporting a decline in TNF-α, IL-8, and IL12p70 and Akbari et al. [40] noting a decline in hs-CRP. Additionally, both studies observed a potential beneficial mechanism through the reduction of oxidative stress, with Ton et al. [43] observing a decline in superoxide anion, hydrogen peroxide, and peroxynitrite/hydroxyl radical and Akbari et al. [40] documenting a decline in MDA. These results agree with preclinical research indicating that probiotics may improve cognitive function in rodent models of AD by reducing oxidative stress and inflammation. In rats who received an intrahippocampal injection of Aβ1–40 followed by probiotic supplementation with Lactobacillus reuteri, Lactobacillus rhamnosus, and Bifidobacterium infantis, Mehrabadi and Sadr [72] observed a significant improvement in cognitive function along with a decline in MDA, IL-1, and TNF-α. Furthermore, probiotics may improve cognitive function in AD by decreasing insulin resistance. Akbari et al. [40] observed a reduction in insulin resistance, which may have also contributed to improvements in cognitive function. Overall, two of the four studies support the beneficial potential of probiotics in improving cognitive function in AD.
3. Selenium
Selenium, an essential trace mineral, can be obtained from several dietary sources, including Brazil nuts, bread, broccoli, cereals, meat, fish, eggs, milk, and dairy [75]. In the form of selenomethionine and the amino acid selenocysteine, selenium is a component of several proteins in the body. These selenium-containing proteins are known as selenoproteins. Selenoproteins play a critical role in cognitive and thyroid function, along with antioxidant defense [75,76]. For example, glutathione peroxidase 4 (GPX4) is essential in regulating ferroptosis and reducing lipid peroxides [76]. A meta-analysis by Reddy et al. [77] indicated that serum selenium levels are significantly lower in individuals with AD, and the difference remained after controlling for age. Among individuals with AD, a direct correlation existed between serum selenium levels and GPX levels [77]. The prevention of AD by vitamin E and selenium trials observed no significant differences in AD risk among males over 60 supplemented with vitamin E (400 IU), selenium (200 μg), or both in combination or alone compared to a placebo for 5.4 years [78]. Conversely, a six-month clinical trial among individuals with MCI indicated that daily supplementation with Brazil nuts containing 288 μg of selenium improved erythrocyte GPX levels and verbal fluency [79]. As a result, selenium is a compound of interest in possibly treating and preventing AD due to its role in antioxidant defense. Two clinical trials were identified examining the impact of selenium supplementation on cognitive function in AD.
The first clinical trial examined the impact of two different doses of selenium. In Australia, researchers conducted a double-blind, placebo-controlled trial examining the impact of sodium selenate (Na2SeO4) supplementation among individuals with mild-to-moderate AD [44,45]. Participants were randomized to consume either supranutrional (10 mg sodium selenate; n = 20), nutritional (0.32 mg sodium selenate; n = 10), or a placebo (n = 10) three times a day for 24 weeks [44,45]. The study by Maplas et al. [44] focused on reporting the trial’s primary outcomes (safety and tolerability), while Cardoso et al. [45] detailed the results of an exploratory analysis. Four participants withdrew from the study due to a skin rash (supranutritional group), nail changes (supranutritional group), a diagnosis of normal pressure hydrocephalus (control group), and a change in medications (control group); the control group was composed of participants receiving either nutritional sodium selenate or the placebo [44]. Maplas et al. [44] observed drug-related adverse events (decreased appetite, dizziness, fatigue, headache, lethargy, myalgia, muscle spasms, nail disorder, and nausea) among 68% of study participants. Furthermore, Maplas et al. [44] reported only one severe adverse event (pre-syncope) in a member of the supranutritional group. With 16 members of the supranutritional group and 11 members of the control group experiencing drug-related adverse events, the authors concluded that supplementation with supranutritional selenate for 24 weeks was tolerable and safe [44]. Analysis of secondary variables revealed no significant changes in CSF phosphorylated tau, CSF total tau (t-tau), CSF Aβ1–42, MMSE, Alzheimer’s Disease Assessment Scale-cognitive (ADAS-Cog), controlled oral word association test (COWAT), category fluency test (CFT), one-card learning memory task (OCL), identification reaction time task (IDN), and detection reaction time task (DET) [44]. As a result, future studies with larger sample sizes and longer durations may reveal that sodium selenate can improve AD biomarkers and cognitive function.
The exploratory analysis by Cardoso et al. [45] expanded upon the trial’s results by examining the impact of sodium selenate supplementation on plasma and CSF selenium. While plasma and CSF selenium levels increased among participants receiving both nutritional and supranutritional doses, the increase in serum selenium only corresponded to a 3% increase in CSF levels [45]. Furthermore, serum and CSF selenium levels were highly variable within each experimental group [45]. As a result, Cardoso et al. [45] examined study data by regrouping participants as ‘responsive’ and ‘non-responsive’ to sodium selenate based on an increase in serum (responsive, n = 17; non-responsive, n = 18) and CSF (responsive, n = 12; non-responsive, n = 14) selenium three times that of baseline levels. There were no differences in the number of drug-related adverse events between the responsive and non-responsive groups [45]. Compared to CSF non-responsive individuals, participants in the CSF responsive group did not experience a significant decline in the MMSE. No significant differences emerged for ADAS-Cog, COWAT, CFT, OCL, IDN, and DET [45]. Nevertheless, the lack of significance in measures of cognitive function could still be accounted for by the study’s small sample size and short duration. Overall, Cardoso et al. [45] highlighted the importance of examining changes in serum and CSF levels of selenium in future studies.
The effects of selenium supplementation and other therapeutic compounds have been investigated. In Iran, Tamtaji et al. [46] examined the role of selenium (200 μg/day) with and without a probiotic (2 × 109 CFU/day of Lactobacillus acidophilus, Bifidobacterium bifidum, and Bifidobacterium longum) supplement among institutionalized individuals with AD. Participants were randomized to consume selenium and a probiotic (n = 30), selenium-only (n = 30), or a placebo (n = 30) for 12 weeks [46]. Following participant withdrawal due to personal reasons, the final analysis included 27 participants in the combination group, 26 in the selenium-only group, and 26 in the placebo group [46]. Similar to Malpas et al. [44], Tamtaji et al. [46] observed no significant difference in MMSE scores between the selenium-only and placebo groups. Malpas et al. [44] estimated that a sample size of 352 participants would be necessary to detect changes in MMSE scores using an 80% power level at a p-value less than 0.05. Since the trial by Tamtaji et al. [46] lasted for 12 weeks less than that of Malpas et al. [44], future clinical trials with more extended periods and a greater number of participants may reveal different results. Tamtaji et al. [46] also observed a significant improvement in the MMSE among participants supplemented with both selenium and probiotics. As a result, the combined actions of both selenium and probiotics could improve cognitive function among individuals with AD. Considering the trials by Akbari et al. [40] and Ton et al. [43] that observed a significant improvement in MMSE scores following the consumption of probiotics, future clinical trials should consider adding a probiotic-only group to assess whether the improvement in the selenium and probiotics group results solely from the addition of the probiotic. While the impact of selenium supplementation in individuals with AD remains unclear, future research may clarify the role of selenium supplementation in AD.
The mechanism behind selenium’s potential beneficial role in AD is currently unclear. One possible explanation is that a decline in the body’s selenium levels corresponds to a decrease in selenoproteins that protect against oxidative stress in the central nervous system [77]. For example, Sharma et al. [80] observed that the mice receiving a selenium-deficient diet had significantly lower selenium levels in the cortex and hippocampus than the mice receiving a selenium-adequate diet. Selenium deficiency also decreased GPX activity in the mouse cortex, hippocampus, and cerebellum and increased lipid peroxidation in the cortex [80]. Compared to selenium-adequate mice, the selenium-deficient mice displayed anxiety-like behavior (open field test) and decreased spatial memory (Morris water maze) [80]. As a result, increasing selenium levels through supplementation may reduce oxidative stress and improve cognitive function. Tamtaji et al. [46] observed that selenium supplementation increased serum glutathione levels and TAC. Additionally, after grouping participants as selenium-responsive and non-responsive, a significant improvement in MMSE scores emerged among individuals with AD supplemented with selenium [44,45]. As a result, inadequate selenium levels may contribute to cognitive decline in AD.
The mineral’s role in ferroptosis further supports the importance of adequate selenium. Ferroptosis is an iron-dependent form of programmed cell death in which excess lipid peroxidation, particularly of polyunsaturated fatty acids in the lipid bilayer, leads to loss of membrane integrity, organelle rupturing, and cell death [81]. GPX4, using reduced glutathione (GSH) as a cofactor, protects cells against ferroptosis by reducing lipid hydroperoxides to lipid alcohols [82,83]. Iron dyshomeostasis can participate in the Fenton reaction by increasing intracellular free iron, leading to an increase in oxidative stress and lipid peroxidation. Compared to mice receiving a selenium-adequate diet, those receiving a selenium-deficient diet showed increased iron levels and neuronal loss in their cortex and hippocampus [80]. Additionally, magnetic resonance imaging (MRI) indicated elevated iron levels in the cortex of individuals with AD compared to age-matched healthy controls [84]. As a result, inadequate selenium decreases GPX4 and GSH levels, decreasing the cell’s resistance to ferroptosis.
As seen in the study by Tamtaji et al., the beneficial role of selenium supplementation in AD [46] may be partially accounted for by the inclusion of probiotic supplementation. In this study [46], both the combination group and the selenium-only group experienced a significant improvement in markers of inflammation (hs-CRP), oxidative stress (glutathione), and insulin resistance (insulin, HOMA-IR, and QUICKI). Nevertheless, compared to both the placebo and selenium-only groups, the combination group experienced a significant improvement in cognitive function (MMSE), inflammation (hs-CRP), oxidative stress (TAC and glutathione), and insulin resistance (insulin, HOMA-IR, and QUICKI). Previously, the results by Akbari et al. [40] and Ton et al. [43] observed that probiotic supplementation could improve cognitive function (MMSE), inflammation (hs-CRP, TNF-α, IL-8, and IL12p70), oxidative stress (MDA and ROS), and insulin resistance (HOMA-IR and QUICKI). While these two studies support the potential ability of probiotics to contribute to the improvement in cognitive decline seen in the combination group, the type of probiotic bacteria may influence their mechanisms in human health. A study by Kim et al. [85] observed that the consumption of a probiotic containing Bifidobacterium bifidum and Bifidobacterium longum for 12 weeks compared to a placebo led to an improvement in cognitive function among community-dwelling older adults. Since the exact contribution of probiotics to cognitive function in AD is unclear [46], future studies should add a probiotic-only group to understand the beneficial changes in this study.
4. Melatonin
Melatonin (n-acetyl-5-methoxytryptamine) is a peptide hormone secreted by the pineal gland and is involved in regulating the circadian rhythm [86,87,88]. A systematic review by Nous et al. [89] indicated lower nighttime melatonin levels in the plasma and CSF of individuals with AD. Furthermore, the presence of sleep disorders in individuals with AD increases the risk of early institutionalization [90]. As a result, there is increased interest in using melatonin for the possible treatment of AD. Melatonin supplements include prolonged-release melatonin and fast-release melatonin. Compared to fast-release melatonin supplements, prolonged-release melatonin extended the time plasma melatonin levels remained above basal levels from three to four hours to five to seven hours [91,92]. In addition to increasing the risk of early institutionalization, disruptions of circadian rhythms may contribute to the generation of oxidative stress in AD [90]. As a result, melatonin’s antioxidant properties, particularly its free radical scavenging activity, could play a role in treating AD by reducing oxidative stress [88,93]. Rondanelle et al. [94] observed a significant improvement in MMSE scores among individuals with MCI supplemented with 5 mg melatonin in the presence of 720 mg docosahexaenoic acid (DHA), 286 mg eicosapentaenoic acid (EPA), 16 mg vitamin E, 160 mg soy phospholipids, and 95 mg tryptophan for 12 weeks compared to a placebo. Therefore, melatonin is a compound of interest in treating AD due to its antioxidant activity and ability to regulate circadian rhythms. One clinical trial was identified that examined the impact of melatonin supplementation on cognitive function in AD.
Wade et al. [47] examined the effect of prolonged-release melatonin among individuals with mild-to-moderate AD treated with acetylcholinesterase inhibitors. Participants were randomized to consume either a 2 mg melatonin supplement (n = 39) or a placebo (n = 34) daily for 24 weeks following a 2-week placebo run-in period and to precede a 2-week placebo run-out period [47]. All enrolled participants were included in the safety analysis [47]. No significant differences were reported between the melatonin (n = 8) and placebo (n = 2) groups for patient-reported drug-related adverse events [47]. Furthermore, none of the severe adverse events (melatonin group, n = 2; placebo group, n = 5) that occurred during the trial were study-related [47]. As a result, Wade et al. [47] concluded that supplementation with 2 mg of melatonin for 24 weeks in individuals with AD was safe and tolerable. After excluding participants who withdrew from the trial, 31 participants in the melatonin group and 29 in the placebo group were included in the final analysis [47]. Though the placebo group experienced no significant changes in overall sleep scores as measured by the Pittsburgh Sleep Quality Index (PSQI), the melatonin group experienced an improvement in overall sleep scores, as indicated by a significant decrease in PSQI scores [47]. These results agree with Cruz-Aguilar et al. [95,96], who observed that melatonin (5-mg, fast-release) supplementation had a beneficial impact on sleep among individuals with mild-to-moderate AD (n = 8) by reducing sleep latency. In contrast, an earlier clinical trial observed no significant differences in sleep measures using wrist actigraphy among individuals with AD supplemented with either 2.5 mg prolonged-release melatonin, 10 mg fast-release melatonin, or a placebo for eight weeks [97]. While neither the placebo nor melatonin group in the study by Wade et al. [47] experienced a significant change in ADAS-Cog scores, the melatonin group experienced significantly less decline in MMSE and an increase in Instrumental Activities of Daily Living (IADL) compared to the placebo group. After 24 weeks, neither the placebo nor the melatonin group experienced a significant change in the Neuropsychiatric Inventory (NPI) [47]. As a result, Wade et al. [47] concluded that prolonged-release melatonin might improve cognitive function in individuals with AD due to improved sleep efficacy. Nevertheless, further studies are needed to examine the relationship between melatonin and AD, including studies with larger sample sizes and longer durations.
While the exact mechanism of melatonin in AD remains unclear, melatonin may play a beneficial role in AD by improving circadian rhythms and reducing inflammation, oxidative stress, and the formation of Aβ plaque and NFT [88,90,98]. Both Wade et al. [47] and Cruz-Aguilar et al. [95,96] observed a significant improvement in sleep among individuals with mild-to-moderate AD following melatonin supplementation. As a result, the beneficial role of melatonin in AD may be due to its ability to restore disruptions in circadian rhythms. Although Wade et al. [47] did not examine melatonin’s impact on oxidative stress and inflammation biomarkers, several preclinical studies provided evidence that melatonin can reduce inflammation and oxidative stress related to AD. For example, melatonin ameliorated DNA fragmentation and apoptosis in rat hippocampal tissue cultures treated with okadaic acid, a pro-oxidant compound that can generate NFT in rat models [99]. In PC12 cells, agomelatine—an agonist of melatonin receptors MT1 and MT2—attenuated Aβ25–35-induced oxidative stress (MDA and ROS) and phosphorylated tau levels in addition to restoring cell viability [100]. Additionally, in rats injected with lipopolysaccharides to induce neuroinflammation, melatonin treatment significantly attenuated inflammation (TNF-α and IL-1β), oxidative stress (MDA and GSH), and acetylcholinesterase activity [101]. As a result, the beneficial impact of melatonin on cognitive function may be due to its ability to reduce oxidative stress and inflammation. Moreover, melatonin may improve cognitive function by reducing the formation of Aβ plaques and NFT. For example, Roy et al. [102] hypothesized that melatonin improved acetylcholine neurotransmission in AD by reducing the formation of Aβ plaques and NFT. The ability of melatonin to prevent Aβ plaques from forming could be related to melatonin contributing to the increased activity of α-secretase and decreased activity of β-secretase [98]. Nevertheless, the results from cell and animal studies do not always translate to humans, and therefore, more research is needed on the role of melatonin in AD and its potential mechanisms.
5. Resveratrol
Resveratrol (3,5,4′-trihydroxystillbene) is a polyphenol that can be obtained from various plant sources, such as blackberries, blueberries, bilberries, cranberries, grapes, and peanuts [103,104,105]. A 6-month pilot study indicated that consumption of 72 g of freeze-dried grape powder, compared to a placebo, protected individuals with mild cognitive decline from cerebral metabolic changes and correlated with improved attention and working memory [106]. Additionally, low-to-moderate consumption of red wine, another source of resveratrol, is associated with a lower risk of AD [107,108,109]. Previous preclinical research indicated that resveratrol might possess anti-inflammatory, anti-apoptotic, antioxidant, anticarcinogenic, and cardioprotective properties [110,111]. Furthermore, a previous systematic review [112] indicated that resveratrol might impact cognitive function in AD. As a result, resveratrol is an antioxidant of interest for potentially treating AD. Four articles were identified that examined the impact of resveratrol supplementation on cognitive function in AD.
In the United States, Turner et al. [48] conducted a phase 2 randomized control trial (RCT) among individuals with mild-to-moderate AD. Participants were randomized to receive either resveratrol (n = 62) or a placebo (n = 55) daily for 52 weeks, starting at a 500 mg dose and increasing it by 500 mg every 13 weeks [48]. While safety and tolerability examination revealed no significant differences between the two groups for neurological examination, physical examination, routine laboratory tests, and vital signs, 95% of the 119 participants experienced an adverse event [48]. There was no significant difference between the groups for the most common adverse events (nausea and diarrhea) [48]. Furthermore, the 36 serious adverse events (27 hospitalizations and three deaths) were determined not to be related to the study or drug [48]. As a result, Turner et al. [48] concluded that resveratrol supplementation was safe and tolerable among individuals with mild-to-moderate AD. After 52 weeks, the placebo group experienced a significantly greater decline in CSF and plasma Aβ40 compared to the resveratrol group, but no significant change was observed in CSF and plasma Aβ42, CSF tau, CSF phosphorylated tau 181 (p-tau), or plasma glucose and insulin metabolism [48]. A systematic review by Olsson et al. [113] observed no significant differences in the CSF Aβ40 for individuals with AD compared to those with MCI. Furthermore, MRIs of participant brain volume revealed a greater decrease in total brain volume among the treatment group [48]. Lastly, Turner et al. [48] observed no significant differences between the groups for ADAS-Cog, Clinical Dementia Rating (CDR)-sum of boxes, MMSE, or NPI but a significantly reduced decline in Alzheimer’s Disease Cooperative Study (ADCS) Activities of Daily Living (ADCS-ADL) among the treatment group. The study had several limitations, and the authors noted that the study might have been underpowered to detect changes in some variables [48].
To understand the mechanisms behind the beneficial role of resveratrol on cognitive function, Moussa et al. [49] examined biomarkers of inflammation at baseline and after 52 weeks in the CSF and plasma of a subset (resveratrol, n = 19; placebo, n = 19) of Turner et al.’s [48] participants. As reported in the previous study [48], the placebo group experienced a greater decline in ADCS-ADL scores compared to the resveratrol group [49]. Additionally, MMSE declined significantly in the placebo group but not in the resveratrol group [49]. While changes in plasma biomarkers of inflammation were observed, these changes lost significance after adjustment for multiple comparisons [49]. In contrast to Turner et al. [48], Moussa et al. [49] observed a significant decline in CSF Aβ40 in the resveratrol group instead of the placebo group after 52 weeks. While the placebo and resveratrol groups experienced a significant decrease in CSF Aβ42, the levels were significantly lower in the placebo group [49]. Additionally, matrix metalloprotease (MMP) 9 and CSF macrophage-derived chemokine levels were significantly higher in the resveratrol group compared to the placebo group at 52 weeks [49]. The significant decrease in CSF MMP9 levels indicated that resveratrol might improve cognitive function by reducing the blood-brain barrier’s permeability to inflammation [49].
In a study conducted in the United States, Zhu et al. [50] hypothesized that adding dextrose and malate to a resveratrol supplement would contribute electrons to the metabolism so it could regenerate reduced resveratrol in the brain. Individuals with mild-to-moderate AD taking donepezil were randomized to receive either the intervention (n = 17)—resveratrol (5 mg) in combination with dextrose (5 g) and malate (5 g)—or a placebo (n = 15) twice daily for a year [50]. Subsequently, one participant withdrew from the intervention group and two from the control group and were excluded from the final analysis [50]. Since none of the adverse events (control group, n = 4; intervention group, n = 3) were deemed to be related to the study drug, it was concluded that supplementation of 5 mg resveratrol with 5 g dextrose and 5 g malate was safe and tolerable among individuals with AD [50]. No significant differences were observed between the control and intervention groups for ADAS-Cog, ADCS-ADL, ADCS Clinician’s Global Impression of Change (ADCS-CGIC), MMSE, or NPI after one year of treatment [50]. Nevertheless, early discontinuation of recruitment resulted in the study being underpowered to detect a significant change in the primary outcome variable, ADAS-Cog [50]. Therefore, studies with a larger sample size on resveratrol supplementation in combination with dextrose and malate may yield different results.
In China, Fang et al. [51] examined the impact of resveratrol on cognitive function in hospitalized individuals with AD. Participants were randomized to receive either donepezil hydrochloride with (n = 45) or without (n = 45) 1 g or 2 g of resveratrol for two months [51]. Participants received a 5 mg dose of donepezil for the first month, which could be increased to 10 mg if needed for the last month of the trial [51]. After two months of treatment, the resveratrol group had significantly higher MMSE and lower ADAS-Cog scores, indicating better cognitive function [51]. The resveratrol group also had better measures of living ability, as indicated by the Functional Independence Measure (FIM) [51]. Additionally, the resveratrol group had significantly lower levels of biomarkers of inflammation (IL-6 and TNF-α) [51]. As a result, the study indicated that resveratrol supplementation could potentially improve cognitive function in individuals with AD by reducing inflammation. Nevertheless, the study faced several limitations. For example, the study did not include outcome data comparing individuals in the experimental group who received 1 g versus 2 g of resveratrol. In addition, the generalizability of the study’s results was limited by the small sample size and short duration.
While the mechanisms explaining the beneficial role of resveratrol in AD remain unclear, resveratrol may improve cognitive function by modulating inflammation and oxidative stress. For example, both Moussa et al. [49] and Fang et al. [51] observed changes in markers of inflammation. Moussa et al. [49] observed significantly lower CSF MMP9 levels in individuals treated with AD, indicating that resveratrol might reduce MMP9 levels and, thus, the permeability of the blood-brain barrier to inflammation. Fang et al. [51] observed a significant decline in serum biomarkers of inflammation (IL-6 and TNF-α). Several preclinical studies indicated that the antioxidant and anti-inflammatory properties of resveratrol might involve signaling pathways dependent on AMP-activated protein kinase (AMPK) [114,115] and SIRT1 [116]. For example, Chiang et al. [115] indicated that resveratrol reduced Aβ1–42-induced inflammation (IL-1β and TNF-α) and restored cell viability in human neural stem cells by activating AMPK. Kong et al. [117] observed that intraperitoneal resveratrol improved cognitive function and enhanced the gene expression of GPX while decreasing MDA tissue levels in the brains of a mouse model of AD.
6. Rosmarinic Acid
First isolated from rosemary (Rosmarinus officinalis), rosmarinic acid (labiatenic acid) is an ester of caffeic acid and 3,4-dihydroxyphenyllactic acid [118,119,120]. Other sources of rosmarinic acid include basil, lemon balm, mint, oregano, sage, and thyme [121,122,123]. Recent research indicated that rosmarinic acid possesses antioxidant, antibacterial, antiviral, anti-inflammatory, antihyperglycemic, hepatoprotective, anticancer, cardioprotective, and neuroprotective properties [123,124,125,126]. The antioxidant and anti-inflammatory properties of rosmarinic acid may benefit the central nervous system. For example, two clinical studies showed anxiety- and insomnia-related symptoms improved after supplementation with Melissa officinalis (lemon balm) [127] or Melissa officinalis and Nepeta menthoides [128]. Herrlinger et al. [129] observed an improvement in working memory among individuals with age-associated memory impairment after the daily consumption of a spearmint supplement for 90 days. As a result, rosmarinic acid is an antioxidant of interest in potentially treating and preventing AD, and one clinical trial was identified examining the impact of rosmarinic acid on cognitive function in AD.
In Japan, Noguchi-Shinohara et al. [52] conducted a clinical trial to examine the safety and efficacy of the daily consumption of Melissa officinalis extract (containing 500 mg of rosmarinic acid) in patients with mild AD. The first phase of the study included a double-blind trial and randomized participants to receive either Melissa officinalis (n = 12) or a placebo (n = 11) for 24 weeks [52]. The study’s second phase began as an open-label trial, with all participants allocated to the intervention group [52]. While no significant differences in vital signs, physical examination, or neurological examination were observed between the two groups, the placebo group experienced significantly more adverse events than the Melissa officinalis group [52]. Therefore, it was concluded that 500 mg of Melissa officinalis extract was safe and tolerable in individuals with AD. The study also examined secondary outcomes related to cognitive function and AD-related biomarkers. In the Melissa officinalis group, serum rosmarinic acid and its conjugate forms increased from baseline to week 24, but levels in the CSF were undetectable [52]. Noguchi-Shinohara et al. [52] observed a significant improvement in NPI scores among the Melissa officinalis group, particularly for the irritability and liability components of the NPI. Nevertheless, there were no significant changes in MMSE, ADAS-Cog, Disability for Dementia (DAD), or CDR scores [52]. Additionally, the researchers observed no significant changes in disease-related biomarkers (CSF Aβ1–42, CSF tau, and CSF p-tau) [52]. As a result, the study indicated that Melissa officinalis supplementation might improve agitation in individuals with AD but no other markers of cognitive function [52]. Nonetheless, the study’s small sample size and short duration limited the generalizability of the findings. As a result, the study may have been underpowered to detect changes in cognitive function in individuals with AD.
Several in vitro and in vivo studies have examined the potential mechanism of action of rosmarinic acid in AD treatments. Potential mechanisms for improving cognitive function in AD include metal chelation and reducing oxidative stress and inflammation. For example, excess copper can contribute to Aβ plaque formation, and copper-containing Aβ plaques can generate ROS [26]. In a cell culture study, Kola et al. [122] observed that rosmarinic acid protected NIH3T3 cells from copper toxicity by forming a 2:1 complex with cupric copper. Mirza et al. [124] observed a significant improvement in spatial memory in mice injected with the Aβ1–42 peptide treated with rosmarinic acid compared to those treated without rosmarinic acid. Furthermore, in a rat model of neuroinflammation, rosmarinic acid treatment attenuated hippocampal neuron loss and damage, short-term working memory decline, inflammation (TNF-α, IL-1β, and IL-6), and markers of oxidative stress (MDA) [121]. As a result, rosmarinic acid may improve cognitive function in AD by reducing oxidative stress and inflammation. Nevertheless, the results from cell and animal studies do not always translate to human beings, and Noguchi-Shinohara et al. [52] observed no changes in measures of cognitive function (MMSE, ADAS-Cog, DAD, or CDR) in individuals treated with rosmarinic acid.
7. Carotenoids
Carotenoids represent a large class of lipid-soluble pigment (yellow, orange, and red) molecules such as α- and β-carotene, cryptoxanthin, lutein, lycopene, and zeaxanthin [130,131]. Carotenoids can be classified based on their ability to be converted to vitamin A in the human body as provitamin A (α- and β-carotene and β-cryptoxanthin) and non-provitamin A (lutein, lycopene, and zeaxanthin) [130,131,132]. Carotenoids play a variety of roles in the human body. For example, lutein and zeaxanthin, which can be obtained through the diet from egg yolks, corn, kale, peas, and spinach, are associated with eye health and a reduced risk of age-related macular degeneration [133]. Additionally, carotenoids can function as antioxidants and may help prevent Aβ aggregation and toxicity [134,135]. An observational study observed a reduced risk of AD mortality in adults with either high serum lycopene or lutein and zeaxanthin but no relationship with serum α-carotene, β-carotene, or β-cryptoxanthin [136]. In Japan, a clinical trial indicated that supplementation with polyunsaturated fatty acids (290 mg EPA and 203 mg DHA), lycopene (84 mg), and Ginkgo biloba (240 mg) for three years improved cognitive function in community-dwelling older adults compared to controls [137]. As a result, carotenoids are an antioxidant of interest in treating AD. Three articles were identified that examined the impact of carotenoid supplementation on cognitive function in individuals with AD.
In Ireland, Nolan et al. [53] examined the role of carotenoid supplementation on cognitive function in individuals with mild-to-moderate AD and healthy controls. Participants were randomized to one of four groups: AD and carotenoid (n = 16); AD and placebo (n = 15); healthy control and carotenoid (n = 15); or healthy control and placebo (n = 16) [53]. The carotenoid supplement contained 10 mg of lutein, 2 mg of zeaxanthin, and 10 mg of meso-zeaxanthin and was consumed daily for six months, while the placebo consisted of sunflower oil [53]. Following six months of treatment, both groups that received the carotenoid supplement experienced a significant improvement in serum lutein, zeaxanthin, and meso-zeaxanthin levels [53]. No significant changes were observed for cognitive function (MMSE) in any of the four groups [53]. As a result, carotenoid supplementation did not improve cognitive function in individuals with AD.
A follow-up study by Nolan et al. [54] supplemented individuals with AD with either the carotenoid supplement (n = 12; 10 mg lutein, 2 mg zeaxanthin, and 10 mg meso-zeaxanthin) or the carotenoid supplement in combination with a fish oil supplement (n = 13; 430 mg DHA, 90 mg EPA) for 18 months. Compared to the carotenoid-only group, the combined intervention group experienced less functional decline based on medical observations and increased serum lutein and meso-zeaxanthin levels following treatment [54]. Therefore, Nolan et al. [55] conducted a 12-month RCT examining supplementation with carotenoids, fish oil, and vitamin E in individuals with mild-to-moderate AD. Participants were randomized to either the supplement group (n = 50; 10 mg lutein, 10 mg meso-zeaxanthin, 2 mg zeaxanthin, 500 mg DHA, 150 mg EPA, and 15 mg α-tocopherol) or a placebo (n = 27; sunflower oil) [55]. After participant withdrawal and loss to follow-up, 38 individuals remained in the intervention group and 19 in the placebo group and were included in the final analysis [55]. Following treatment, the intervention group experienced a significant increase in skin carotenoid scores [55]. After controlling for increased dietary carotenoid intake in the intervention group, serum nutrient levels (lutein, meso-zeaxanthin, zeaxanthin, DHA, EPA, and vitamin E) increased significantly compared to the placebo group [55]. Although the intervention group experienced less decline in caregiver-reported patient memory than the placebo group, no significant changes were observed for mean cognitive function as indicated by the MMSE and the Dementia Severity Rating Scale (DSRS) [55]. Upon categorizing participants by dementia severity (DSRS; mild, moderate, and severe), a significant improvement was observed in the intervention group, while the placebo group experienced a significant decline [55]. Lastly, no significant changes were observed for the clinical frailty score or patient- or caregiver-reported perceived quality of life [55]. While the study indicates that carotenoid supplementation combined with fish oil and vitamin E can potentially improve cognitive function in AD, the study was underpowered to detect changes in the MMSE [55]. Therefore, more research is required to examine the impact of carotenoid supplementation on cognitive function in AD.
8. Curcumin
Curcumin (1,7-bis-(4-hydroxy-3-methoxyphenyl)-1, 6-heptadiene-3,5-dione), also known as diferuloylmethane, is a component of the spice turmeric, which is derived from the plant Curcuma longa [138,139,140,141,142]. Previous research indicates that the consumption of curry, which is rich in curcumin, is associated with greater cognitive performance among older adults [143,144]. Curcumin possesses anti-inflammatory, antioxidant, and metal-chelating properties [140,141,142,145]. Additionally, several preclinical trials indicate that curcumin can attenuate cognitive decline by interacting with Aβ peptides to prevent plaque formation [139]. In fact, curcumin’s ability to interact with Aβ has led to recent interest in using curcumin compounds and derivatives with imaging technology to potentially diagnose AD [146]. An 18-month randomized clinical trial of older dementia-free adults observed an improvement in memory in participants supplemented with curcumin (n = 21) compared to a placebo (n = 19) [147]. As a result, curcumin is an antioxidant of interest in treating AD. One clinical trial was identified that examined the impact of curcumin supplementation on cognitive function in AD.
In the United States, Ringman et al. [56] examined the role of curcumin supplementation in individuals with mild-to-moderate AD. Participants were randomized to receive either 2 g of curcumin (n = 12), 4 g of curcumin (n = 12), or a placebo daily for 24 weeks [56]. After a few weeks, the RCT was converted to an open-label trial, and the placebo group was randomized to either 2 g or 4 g of curcumin for 24 more weeks. Following participant withdrawal, nine individuals remained in the 2 g curcumin group, 10 in the 4 g curcumin group, and 11 in the placebo group [56]. While Ringman et al. [56] observed no significant differences between the three groups for measures of cognitive function (MMSE, ADAS-Cog, ADCS-ADL, NPI) after 24 weeks of treatment, the authors noted a non-significant trend towards worsening cognitive function (MMSE, ADAS-Cog) in the curcumin groups combined compared to the placebo group. Additionally, no significant differences were observed between the three groups for biomarkers of AD (plasma Aβ40 and Aβ42; CSF Aβ42, t-tau, and p-tau) and CSF isoprostanes [56]. As a result, the study indicated that curcumin supplementation did not improve cognitive function in individuals with AD. Nevertheless, the study had several limitations, including a small sample size. Additionally, research [145,148,149] indicates that curcumin has low bioavailability, and Ringman et al. [56] were unable to detect curcuminoids in the CSF, though a slight increase was observed in the plasma of treated individuals. While Small et al. [147] observed a beneficial effect of curcumin supplementation on memory in older adults, the study’s participants were dementia-free, and the study drug had been formulated to increase curcumin absorption in the gastrointestinal tract. Therefore, future research using different curcumin formulations and larger sample sizes may yield different results.
9. Vitamin E
Vitamin E is a fat-soluble vitamin consisting of eight different isoforms, including α-, β-, γ-, and δ-tocopherols and tocotrienols [150]. Dietary sources of vitamin E may vary in the amount of a particular isoform, with some plant-based sources being richer in one isoform over another [150,151]. For example, α-tocopherol—the most common isoform of vitamin E in the human body—is more abundant than other isoforms of vitamin E in hazelnuts, almond oil, and sunflower oil [150,151,152]. Vitamin E is an antioxidant that can function as a free radical scavenger to prevent the peroxidation of polyunsaturated fatty acids in the lipid bilayer of cells [153]. A meta-analysis by Lopes da Silva et al. [154] reported significantly lower plasma vitamin E levels in individuals with AD compared to controls. In 1997, a study published by Sano et al. [155] indicated that supplementation with 2000 IU of vitamin E reduced the progression to either death, institutionalization, loss of ADL, or severe dementia in individuals with moderately severe AD compared to a placebo. As a result, vitamin E is an antioxidant of interest in potentially treating AD. Four clinical trials were identified that examined the impact of vitamin E supplementation on cognitive function in individuals with AD.
In Germany, Arlt et al. [57] conducted an open-label trial to examine the impact of combined vitamin E and C supplementations in individuals with mild-to-moderate AD who take cholinesterase inhibitors. While the control group (n = 11) received standard care, the intervention group (n = 12) received vitamin E (400 IU) and vitamin C (1000 mg) daily for a year [57]. At baseline, the intervention group was composed of younger participants (mean age 67.7 ± 7.2 vs. 73.7 ± 5.3) with an earlier age at diagnosis (mean age 63.9 ± 7.6 vs. 71.1 ± 5.5) than the control group [57]. After one month of treatment, the intervention group experienced a significant increase in CSF α-tocopherol of 42% and a 14% increase in ascorbate, which remained relatively stable for the duration of the trial [57]. Additionally, Arlt et al. [57] observed a significant reduction in the rate of CSF oxidation after one year but not after one month. Despite these changes, no significant differences emerged between the two groups for cognitive function (MMSE, immediate and delayed word recall, word fluency, and the trail-making test) at baseline or after one year of treatment [57]. While the difference between the two groups for mean change in MMSE scores after one year was not statistically significant (intervention group, −2.08 ± 3.73; control group, −1.64 ± 2.26), the decline in MMSE scores experienced by the intervention group was statistically significant. Nevertheless, the study’s data is limited by its small sample size, open-label design, and absence of data on oxidative stress in the control group. Overall, the study does not support the beneficial role of vitamin E supplementation in combination with vitamin C in improving cognitive function in AD.
In the United States, Galasko et al. [58] examined the impact of an antioxidant supplement containing vitamin E (α-tocopherol) in comparison to supplementation with coenzyme Q alone or a placebo among individuals with mild-to-moderate AD. Participants were randomized to receive either an antioxidant supplement (800 IU vitamin E, 500 mg vitamin C, and 900 mg α-lipoic acid; n = 26), 400 mg coenzyme Q three times a day (n = 26), or a placebo (n = 26) for 16 weeks [58]. After excluding participants who withdrew from the study, 24 individuals remained in the antioxidant supplement group, 20 in the coenzyme Q group, and 18 in the placebo group and were included in the final analysis [58]. Compared to the coenzyme Q and placebo groups, the combined antioxidant group experienced a significant decline in cognitive function (MMSE) following treatment [58]. As a result, vitamin E supplementation combined with vitamin C and α-lipoic acid may negatively impact cognition [58]. Additionally, F2-isoprostane levels significantly decreased after treatment in the combined antioxidant group, indicating decreased oxidative stress in the central nervous system [58]. No significant differences emerged between the three groups for ADAS-ADL or CSF biomarkers (Aβ42, t-tau, or p-tau) [58]. Therefore, the study observed either no impact (coenzyme Q) or a negative impact (combined antioxidant group) of antioxidant supplementation on cognitive function in individuals with AD.
In the United States, Dysken et al. [59] examined the impact of vitamin E supplementation (α-tocopherol) with or without memantine on cognitive function in individuals with mild-to-moderate AD. Participants were randomized to one of four groups: 2000 IU vitamin E (n = 152), 20 mg memantine (n = 155), vitamin E and memantine (n = 154), or placebo (n = 152) daily [59]. Participants were scheduled for a follow-up every six months for four years, with the average participant being followed for 2.27 years [59]. After excluding participants without follow-up data, the study included 140 participants from the vitamin E group, 142 from the memantine group, 139 from the combination group, and 140 from the placebo group in the final analysis. Compared to the placebo group, participants receiving vitamin E alone experienced significantly less decline in ADCS-ADL scores. The average decline in ADCS-ADL was reduced by 3.15 units, indicating a clinically significant attenuation in the loss of functional ability. Although the placebo group experienced a greater decline in ADCS-ADL scores than the combined treatment group, the result was not statistically significant. Additionally, caregivers spent significantly less time (mean = 2.17 h) assisting participants (Caregiver Activity Survey) in the vitamin E group compared to the memantine group [59]. No other significant differences were detected for the MMSE, ADAS-Cog, NPI, or Dependence Scale. As a result, the trial indicated that 2000 IU of vitamin E without memantine might prevent an individual with AD’s ADL from declining and alleviate the time caregivers spend assisting individuals. In terms of safety, Dysken et al. [59] observed no significant differences between the vitamin E and placebo groups for adverse events, serious adverse events, or mortality. Conversely, significantly more serious adverse events were reported in the memantine and combination groups than in the placebo group [59]. While the study indicated that 2000 IU of vitamin E is safe for individuals with AD, the study’s generalizability is limited by the predominantly Caucasian (86%) male (97%) sample [59]. Additionally, larger sample sizes may be required to detect changes in other markers of cognitive function (MMSE and ADAS-Cog).
Furthermore, in the United States, Remington et al. [60] examined the impact of a nutraceutical supplement containing vitamin E in individuals with AD. Participants were randomized to receive either a supplement containing 400 μg folic acid, 30 IU α-tocopherol, 6 μg vitamin B12, 400 mg S-adenosyl methionine, 600 mg N-acetyl cysteine, and 500 mg acetyl-L-carnitine (n = 86) or a placebo (n = 57) twice daily for three to six months, followed by all participants being allocated to the intervention group for another six months [60]. After excluding participants lost to follow-up, 84 individuals remained in the intervention group and 57 in the placebo group [60]. Following three months of supplementation, participants in the intervention group had a significant improvement in cognitive function (Dementia Rating Scale) but no significant changes in NPI or ADCS-ADL scores [60]. Following the end of the open-label portion of the trial, both participants initially allocated to the intervention or placebo groups experienced either an improvement or maintenance of cognitive function [60]. Remington et al. [156] also reported that a cohort of 24 individuals with AD who consumed the neutraceutical supplement under open-label conditions for one year experienced no significant changes in the Dementia Rating Scale, NPI, or ADCS-ADL. The authors indicated that this lack of change suggested that the neutraceutical supplement prevented cognitive decline in participants [156]. As a result, the authors concluded that the nutraceutical supplement could improve cognitive function or prevent cognitive decline in individuals with AD [60,156]. Nevertheless, the study had several limitations, including small sample sizes and a lack of diversity among participants. Additionally, the inclusion of other nutrients besides vitamin E prevented researchers from drawing conclusions about the benefits of vitamin E supplements in individuals with AD.
While the mechanisms explaining the benefits of vitamin E in AD are unclear, vitamin E may improve cognitive function in individuals with AD by reducing inflammation and oxidative stress [153,157]. In a study by Giraldo et al. [158], vitamin E treatment attenuated p38 activation and tau phosphorylation in Aβ1–42-treated cortical neurons. Vitamin E supplementation also reduced p38 activation but not tau phosphorylation in the hippocampus of APP/PS1 mice [158]. Additionally, Ibrahim et al. [159] observed that vitamin E supplementation attenuated cognitive decline and Aβ accumulation in the cortex but not the hippocampus of APP/PS1 mice. Desrumaux et al. [160] examined the impact of vitamin E supplementation in phospholipid transfer protein (PLTP) knock-out mice with intracerebrovascular injection of Aβ25–35. PLTP is involved in the transfer of α-tocopherol throughout the body, and its deficiency results in increased circulating vitamin E and decreased vitamin E concentrations in the brain [160]. Vitamin E supplementation significantly restored vitamin E levels in the brains of PLTP knock-out mice and reduced oxidative stress (the cerebral oxidative stress index) and Aβ1–42 accumulation but did not affect inflammation (IL-6 and TNF-α) [160]. Nevertheless, the results from cell and animal studies do not always translate to human beings, and Dysken et al. [59] and Sano et al. [155] observed no changes in measures of cognitive function (MMSE) in individuals supplemented with vitamin E.
10. Coenzyme Q
Coenzyme Q10 (2,3-dimethoxy-5-methyl-6-multiprenyl-1,4-benzoquinone) is known as ubiquinone in its oxidized form and as ubiquinol in its reduced form [161,162,163]. Coenzyme Q10 can be synthesized by the human body, where it is involved in the electron transport chain of the mitochondria [163,164]. Additionally, it can be obtained from dietary sources, including fatty fish, spinach, and nuts, or from supplements [163]. Coenzyme Q10 is an antioxidant of interest in treating AD due to its free-radical scavenging capability and role in the regeneration of vitamins C and E [165]. Two systematic reviews and meta-analyses examining coenzyme Q10 supplementation and markers of oxidative stress observed that coenzyme Q10 significantly reduces MDA, increases superoxide dismutase and TAC, and does not impact catalase, GPX, or GSH [161,162]. While preclinical studies indicate that coenzyme Q10 is potentially therapeutic in dementia treatment, clinical studies on coenzyme Q10 supplementation published before 2012 have yielded mixed results [165]. Furthermore, a systematic review of three studies observed no significant difference between the coenzyme Q10 serum/plasma levels of individuals with AD and controls [165]. Nevertheless, coenzyme Q10 remains an antioxidant of interest in treating AD. One clinical trial was identified that examined the impact of coenzyme Q10 supplementation on cognitive function in AD.
As mentioned earlier, Galasko et al. [58] examined the impact of coenzyme Q (400 mg, three times a day) in comparison to an antioxidant supplement (800 IU vitamin E, 500 mg vitamin C, and 900 mg α-lipoic acid) or a placebo for 16 weeks in individuals with mild-to-moderate AD. While the combined supplementation increased the rate of cognitive decline (MMSE), coenzyme Q had no significant impact [58]. Coenzyme Q did not impact ADAS-ADL scores or CSF biomarkers (F2-isoprostane, Aβ42, t-tau, and p-tau) [58]. While the study is already limited by its small sample size, its data could also be impacted by the type of coenzyme Q supplement used. For example, Mantle et al. [164] noted that the bioavailability of coenzyme Q10 could vary based on the formulation of the supplement. In light of the promising role of coenzyme Q10 being beneficial for potentially treating neurodegenerative diseases and clinical studies on its role in AD yielding mixed results, more research is needed to determine the impact of coenzyme Q10 on cognitive function and oxidative stress in AD [163,165].
11. Summary and Conclusions
In summary, this review article examined the results of clinical trials involving supplementation with antioxidants on cognitive function in individuals with AD (Figure 1). First, though Agahi et al. [41] and Leblhuber et al. [42] observed no relationship between probiotic supplementation and cognitive function, the studies by Akbari et al. [40] and Ton et al. [43] observed that probiotic supplementation could improve cognitive function in individuals with AD. The authors also provided evidence that probiotics may improve cognitive function by reducing oxidative stress and inflammation [40,43]. Second, while Maplas et al. [44] observed no benefits of selenium supplementation on cognitive function in individuals with AD, Cardoso et al. [45] observed that selenium supplementation prevented cognitive decline after grouping the study’s participants into selenium ‘responsive’ and ‘non-responsive’ groups based on changes in CSF and plasma selenium concentrations. Tamtaji et al. [46] also observed a significant improvement in cognitive function in individuals with AD supplemented with both selenium and probiotics but not selenium alone. Since the trial did not include a probiotic-only group, it is unclear whether the beneficial effects resulted from probiotic supplementation or the combination of the two supplements. Third, Wade et al. [47] observed that melatonin supplementation prevented cognitive decline in individuals with AD. Fourth, in contrast to Turner et al. [48] and Zhu et al. [50], Fang et al. [51] observed a beneficial impact of resveratrol supplementation on cognitive function in individuals with AD. Additionally, both Turner et al. [48] and Fang et al. [51] observed a beneficial impact of resveratrol supplementation on functional ability. Fifth, though Noguchi-Shinohara et al. [52] observed no significant changes in measures of cognitive function following supplementation with rosmarinic acid, the study’s participants experienced a significant improvement in measures of irritability and liability. Sixth, the studies by Nolan et al. [53,54,55] observed little benefit of carotenoid supplementation on cognitive function in individuals with AD, though Nolan et al. [55] observed beneficial changes in the AD severity category and caregiver-reported patient memory. Seventh, Ringman et al. [56] observed no relationship between curcumin supplementation and cognitive function, though the authors’ noted a non-significant trend toward worsening cognitive function. Eight, while Dysken et al. [59] observed no significant benefits of vitamin E on cognitive function, participants receiving vitamin E supplementation experienced significantly less decline in ADL than controls. In contrast, Remington et al. [60] observed a significant improvement in cognitive function but no difference in ADL in individuals with AD receiving a vitamin E supplement compared to controls. Nevertheless, the vitamin E supplement in the study by Remington et al. [60] included a lower dose of vitamin E and contained other nutraceutical compounds (folic acid, vitamin B12, S-adenosyl methionine, N-acetyl cysteine, and acetyl-L-carnitine). Arlt et al. [57] observed no relationship between vitamin E and C supplementation and cognitive function. Galasko et al. [58] also observed a significant decline in cognitive function in individuals receiving a vitamin E-containing supplement compared to either coenzyme Q or a placebo. As a result, the comparability of the four studies is limited. Overall, the ability of antioxidant supplements to improve cognitive function in individuals with AD remains unclear.
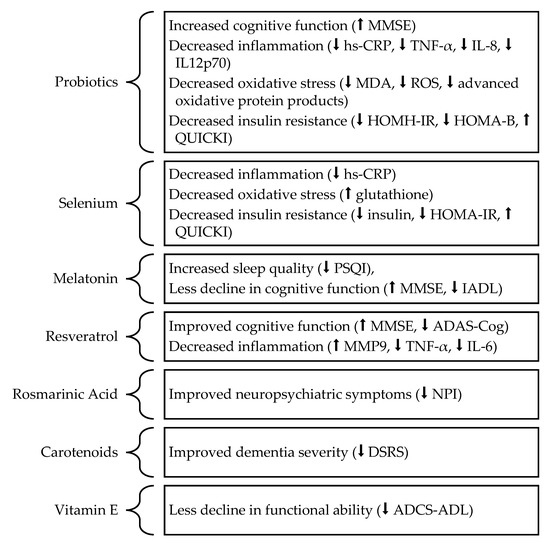
Figure 1.
The potential beneficial effects of different antioxidant supplements in individuals with AD. Upward arrows (⬆) indicate that the beneficial relationship results from the indicated value increasing, while downward arrows (⬇) indicate that the beneficial relationship results from the indicated value decreasing. Abbreviations: AD, Alzheimer’s disease; ADAS-Cog, Alzheimer’s Disease Assessment Scale-cognitive; ADCS, Alzheimer’s Disease Cooperative Study; ADCS-ADL, Alzheimer’s Disease Cooperative Study Activities of Daily Living; DSRS, Dementia Severity Rating Scale; HOMA-B, Homeostatic Model of Assessment for B-cell Function; HOMA-IR, Homeostatic Model of Assessment for Insulin Resistance; hs-CRP, high-sensitivity C-reactive protein; IADL, Instrumental Activities of Daily Living; IL, Interleukin; MDA, Malondialdehyde; MMP, Matrix Metalloprotease; MMSE, Mini-Mental State Examination; NPI, Neuropsychiatric Inventory; PSQI, Pittsburgh Sleep Quality Index; QUICKI, Quantitative Insulin Sensitivity Check Index; ROS, Reactive Oxygen Species; TNF-α, Tumor Necrosis Factor α.
As of January 2023, there are three ongoing clinical trials examining probiotic supplementation (NCT05521477, NCT04606420, and NCT05145881) in individuals with MCI or AD registered with clinicaltrials.gov [166,167,168]. Additionally, two ongoing clinical trials examining melatonin supplementation (NCT03954899 and NCT05543681) and another examining vitamin E supplementation (NCT05343611) and one examining coenzyme Q, probiotics, and curcumin supplementation (NCT04606420) in individuals with MCI or AD are registered [167,169,170,171]. There are no active clinical trials on selenium, resveratrol, rosmarinic acid, or carotenoid supplementation in AD.
In conclusion, this review article examined the results of clinical trials involving supplementation with antioxidants on cognitive function in individuals with AD. While the studies included in this review did not provide much evidence for the beneficial role of antioxidant supplements on cognitive function in individuals with AD, the results varied from antioxidant to antioxidant among trials examining the same antioxidant. Furthermore, the studies face several limitations, including short trial durations, small sample sizes, and a lack of diversity among study participants. As a result, more research is required to examine the impact of antioxidant supplementation on cognitive function in individuals with AD.
Author Contributions
E.K.: writing—original draft preparation, review, and editing. T.G.: funding acquisition, writing—review and editing. T.L.B.: writing—review and editing. J.R.B.: conceptualization, visualization, funding acquisition, writing—review and editing. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the Alabama Agricultural Experimental Station (AAES), the Hatch/Multistate Funding program, and the AAES Award for Interdisciplinary Research (AAES-AIR) that was awarded to T.G. and J.R.B.
Conflicts of Interest
The authors declare that they have no known competing interests regarding the publication of this article.
Abbreviations
Aβ, Amyloid Beta; AD, Alzheimer’s disease; ADAS-Cog, Alzheimer’s Disease Assessment Scale-cognitive; ADCS, Alzheimer’s Disease Cooperative Study; ADCS-ADL, Alzheimer’s Disease Cooperative Study Activities of Daily Living; ADCS-CGIC, Alzheimer’s Disease Cooperative Study Clinician’s Global Impression of Change; AMPK, AMP-Activated Protein Kinase; APP, Amyloid Precursor Protein; CDR, Clinical Dementia Rating; CFT, Category Fluency Test; CSF, Cerebral Spinal Fluid; COWAT, Controlled Oral Word Association Test; DAD, Disability Assessment for Dementia; DET, Detection Reaction Time Task; DHA, Docosahexaenoic Acid; DNA, Deoxyribonucleic Acid; DSRS, Dementia Severity Rating Scale; EPA, Eicosapentaenoic Acid; FIM, Functional Independence Measure; GPX, Glutathione Peroxidase; GSH, glutathione; HOMA-B, Homeostatic Model of Assessment for B-cell Function; HOMA-IR, Homeostatic Model of Assessment for Insulin Resistance; hs-CRP, high-sensitivity C-reactive protein; IADL, Instrumental Activities of Daily Living; IDN, Identification Reaction Time Task; IL, Interleukin; MCI, Mild Cognitive Impairment; MDA, Malondialdehyde; MMP, Matrix Metalloprotease; MMSE, Mini-Mental State Examination; MRI, Magnetic Resonance Imaging; n, sample size; NFT, Neurofibrillary Tau Tangles; NO, Nitric Oxide; NPI, Neuropsychiatric Inventory; OCL, One-Card Learning Memory Task; PLTP, Phospholipid Transfer Protein; PSQI, Pittsburgh Sleep Quality Index; PS1, Presenilin 1; p-tau, Phosphorylated Tau 181; QUICKI, Quantitative Insulin Sensitivity Check Index; RNA, Ribonucleic Acid; ROS, Reactive Oxygen Species; SIRT1, Sirtuin1; TAC, Total Antioxidant Capacity; TNF-α, Tumor Necrosis Factor α; t-tau, Total Tau; TYM, Test Your Memory.
References
- Cao, Q.; Tan, C.C.; Xu, W.; Hu, H.; Cao, X.P.; Dong, Q.; Tan, L.; Yu, J.T. The Prevalence of Dementia: A Systematic Review and Meta-Analysis. J. Alzheimer Dis. 2020, 73, 1157–1166. [Google Scholar] [CrossRef]
- Bacigalupo, I.; Mayer, F.; Lacorte, E.; Di Pucchio, A.; Marzolini, F.; Canevelli, M.; Di Fiandra, T.; Vanacore, N. A Systematic Review and Meta-Analysis on the Prevalence of Dementia in Europe: Estimates from the Highest-Quality Studies Adopting the DSM IV Diagnostic Criteria. J. Alzheimer Dis. 2018, 66, 1471–1481. [Google Scholar] [CrossRef] [PubMed]
- Rajan, K.B.; Weuve, J.; Barnes, L.L.; McAninch, E.A.; Wilson, R.S.; Evans, D.A. Population estimate of people with clinical Alzheimer’s disease and mild cognitive impairment in the United States (2020–2060). Alzheimer Dement. 2021, 17, 1966–1975. [Google Scholar] [CrossRef]
- Wong, W. Economic burden of Alzheimer disease and managed care considerations. Am. J. Manag. Care 2020, 26, S177–S183. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Wang, X.; He, R.; Liang, R.; Zhou, L. Measuring the Caregiver Burden of Caring for Community-Residing People with Alzheimer’s Disease. PLoS ONE 2015, 10, e0132168. [Google Scholar] [CrossRef]
- Vu, M.; Mangal, R.; Stead, T.; Lopez-Ortiz, C.; Ganti, L. Impact of Alzheimer’s Disease on Caregivers in the United States. Health Psychol. Res. 2022, 10, 37454. [Google Scholar] [CrossRef]
- Breijyeh, Z.; Karaman, R. Comprehensive Review on Alzheimer’s Disease: Causes and Treatment. Molecules 2020, 25, 5789. [Google Scholar] [CrossRef] [PubMed]
- Jack, C.R., Jr.; Bennett, D.A.; Blennow, K.; Carrillo, M.C.; Dunn, B.; Haeberlein, S.B.; Holtzman, D.M.; Jagust, W.; Jessen, F.; Karlawish, J.; et al. NIA-AA Research Framework: Toward a biological definition of Alzheimer’s disease. Alzheimer Dement. 2018, 14, 535–562. [Google Scholar] [CrossRef] [PubMed]
- Scheltens, P.; De Strooper, B.; Kivipelto, M.; Holstege, H.; Chételat, G.; Teunissen, C.E.; Cummings, J.; van der Flier, W.M. Alzheimer’s disease. Lancet 2021, 397, 1577–1590. [Google Scholar] [CrossRef]
- McKhann, G.M.; Knopman, D.S.; Chertkow, H.; Hyman, B.T.; Jack, C.R., Jr.; Kawas, C.H.; Klunk, W.E.; Koroshetz, W.J.; Manly, J.J.; Mayeux, R.; et al. The diagnosis of dementia due to Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimer Dement. 2011, 7, 263–269. [Google Scholar] [CrossRef]
- Hansson, O.; Lehmann, S.; Otto, M.; Zetterberg, H.; Lewczuk, P. Advantages and disadvantages of the use of the CSF Amyloid β (Aβ) 42/40 ratio in the diagnosis of Alzheimer’s Disease. Alzheimer Res. Ther. 2019, 11, 34. [Google Scholar] [CrossRef] [PubMed]
- Hampel, H.; Hardy, J.; Blennow, K.; Chen, C.; Perry, G.; Kim, S.H.; Villemagne, V.L.; Aisen, P.; Vendruscolo, M.; Iwatsubo, T.; et al. The Amyloid-β Pathway in Alzheimer’s Disease. Mol. Psychiatry 2021, 26, 5481–5503. [Google Scholar] [CrossRef]
- Ma, C.; Hong, F.; Yang, S. Amyloidosis in Alzheimer’s Disease: Pathogeny, Etiology, and Related Therapeutic Directions. Molecules 2022, 27, 1210. [Google Scholar] [CrossRef] [PubMed]
- Hur, J.Y. γ-Secretase in Alzheimer’s disease. Exp. Mol. Med. 2022, 54, 433–446. [Google Scholar] [CrossRef]
- Naseri, N.N.; Wang, H.; Guo, J.; Sharma, M.; Luo, W. The complexity of tau in Alzheimer’s disease. Neurosci. Lett. 2019, 705, 183–194. [Google Scholar] [CrossRef]
- Pisoschi, A.M.; Pop, A. The role of antioxidants in the chemistry of oxidative stress: A review. Eur. J. Med. Chem. 2015, 97, 55–74. [Google Scholar] [CrossRef]
- Nishimura, Y.; Kanda, Y.; Sone, H.; Aoyama, H. Oxidative Stress as a Common Key Event in Developmental Neurotoxicity. Oxid. Med. Cell. Longev. 2021, 2021, 6685204. [Google Scholar] [CrossRef]
- Sies, H. Oxidative stress: A concept in redox biology and medicine. Redox. Biol. 2015, 4, 180–183. [Google Scholar] [CrossRef] [PubMed]
- Teleanu, D.M.; Niculescu, A.G.; Lungu, I.I.; Radu, C.I.; Vladâcenco, O.; Roza, E.; Costăchescu, B.; Grumezescu, A.M.; Teleanu, R.I. An Overview of Oxidative Stress, Neuroinflammation, and Neurodegenerative Diseases. Int. J. Mol. Sci. 2022, 23, 5938. [Google Scholar] [CrossRef]
- Liguori, I.; Russo, G.; Curcio, F.; Bulli, G.; Aran, L.; Della-Morte, D.; Gargiulo, G.; Testa, G.; Cacciatore, F.; Bonaduce, D.; et al. Oxidative stress, aging, and diseases. Clin. Interv. Aging 2018, 13, 757–772. [Google Scholar] [CrossRef]
- Chen, Z.; Zhong, C. Oxidative stress in Alzheimer’s disease. Neurosci. Bull. 2014, 30, 271–281. [Google Scholar] [CrossRef]
- Cioffi, F.; Adam, R.H.I.; Bansal, R.; Broersen, K. A Review of Oxidative Stress Products and Related Genes in Early Alzheimer’s Disease. J. Alzheimer Dis. 2021, 83, 977–1001. [Google Scholar] [CrossRef] [PubMed]
- Gustaw-Rothenberg, K.; Kowalczuk, K.; Stryjecka-Zimmer, M. Lipids’ peroxidation markers in Alzheimer’s disease and vascular dementia. Geriatr. Gerontol. Int. 2010, 10, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Kukreti, R.; Saso, L.; Kukreti, S. Oxidative Stress: A Key Modulator in Neurodegenerative Diseases. Molecules 2019, 24, 1583. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Zhao, B. Oxidative stress and the pathogenesis of Alzheimer’s disease. Oxid. Med. Cell. Longev. 2013, 2013, 316523. [Google Scholar] [CrossRef]
- Ejaz, H.W.; Wang, W.; Lang, M. Copper Toxicity Links to Pathogenesis of Alzheimer’s Disease and Therapeutics Approaches. Int. J. Mol. Sci. 2020, 21, 7660. [Google Scholar] [CrossRef]
- Ganguly, U.; Kaur, U.; Chakrabarti, S.S.; Sharma, P.; Agrawal, B.K.; Saso, L.; Chakrabarti, S. Oxidative Stress, Neuroinflammation, and NADPH Oxidase: Implications in the Pathogenesis and Treatment of Alzheimer’s Disease. Oxid. Med. Cell. Longev. 2021, 2021, 7086512. [Google Scholar] [CrossRef]
- Lei, P.; Ayton, S.; Bush, A.I. The essential elements of Alzheimer’s disease. J. Biol. Chem. 2021, 296, 100105. [Google Scholar] [CrossRef]
- Cheignon, C.; Tomas, M.; Bonnefont-Rousselot, D.; Faller, P.; Hureau, C.; Collin, F. Oxidative stress and the amyloid beta peptide in Alzheimer’s disease. Redox. Biol. 2018, 14, 450–464. [Google Scholar] [CrossRef]
- Dou, K.X.; Tan, M.S.; Tan, C.C.; Cao, X.P.; Hou, X.H.; Guo, Q.H.; Tan, L.; Mok, V.; Yu, J.T. Comparative safety and effectiveness of cholinesterase inhibitors and memantine for Alzheimer’s disease: A network meta-analysis of 41 randomized controlled trials. Alzheimer Res. Ther. 2018, 10, 126. [Google Scholar] [CrossRef]
- Grossberg, G.T.; Tong, G.; Burke, A.D.; Tariot, P.N. Present Algorithms and Future Treatments for Alzheimer’s Disease. J. Alzheimer Dis. 2019, 67, 1157–1171. [Google Scholar] [CrossRef] [PubMed]
- Shi, M.; Chu, F.; Zhu, F.; Zhu, J. Impact of Anti-amyloid-β Monoclonal Antibodies on the Pathology and Clinical Profile of Alzheimer’s Disease: A Focus on Aducanumab and Lecanemab. Front Aging Neurosci. 2022, 14, 870517. [Google Scholar] [CrossRef]
- Vaz, M.; Silva, V.; Monteiro, C.; Silvestre, S. Role of Aducanumab in the Treatment of Alzheimer’s Disease: Challenges and Opportunities. Clin. Interv. Aging 2022, 17, 797–810. [Google Scholar] [CrossRef] [PubMed]
- Hampel, H.; Mesulam, M.M.; Cuello, A.C.; Farlow, M.R.; Giacobini, E.; Grossberg, G.T.; Khachaturian, A.S.; Vergallo, A.; Cavedo, E.; Snyder, P.J.; et al. The cholinergic system in the pathophysiology and treatment of Alzheimer’s disease. Brain 2018, 141, 1917–1933. [Google Scholar] [CrossRef]
- Sharma, K. Cholinesterase inhibitors as Alzheimer’s therapeutics (Review). Mol. Med. Rep. 2019, 20, 1479–1487. [Google Scholar] [CrossRef]
- Moreira, N.; Lima, J.; Marchiori, M.F.; Carvalho, I.; Sakamoto-Hojo, E.T. Neuroprotective Effects of Cholinesterase Inhibitors: Current Scenario in Therapies for Alzheimer’s Disease and Future Perspectives. J. Alzheimer Dis. Rep. 2022, 6, 177–193. [Google Scholar] [CrossRef]
- Parsons, C.G.; Danysz, W.; Dekundy, A.; Pulte, I. Memantine and cholinesterase inhibitors: Complementary mechanisms in the treatment of Alzheimer’s disease. Neurotox. Res. 2013, 24, 358–369. [Google Scholar] [CrossRef] [PubMed]
- Arvanitakis, Z.; Shah, R.C.; Bennett, D.A. Diagnosis and Management of Dementia: Review. JAMA 2019, 322, 1589–1599. [Google Scholar] [CrossRef]
- Pritam, P.; Deka, R.; Bhardwaj, A.; Srivastava, R.; Kumar, D.; Jha, A.K.; Jha, N.K.; Villa, C.; Jha, S.K. Antioxidants in Alzheimer’s Disease: Current Therapeutic Significance and Future Prospects. Biology 2022, 11, 212. [Google Scholar] [CrossRef]
- Akbari, E.; Asemi, Z.; Daneshvar Kakhaki, R.; Bahmani, F.; Kouchaki, E.; Tamtaji, O.R.; Hamidi, G.A.; Salami, M. Effect of Probiotic Supplementation on Cognitive Function and Metabolic Status in Alzheimer’s Disease: A Randomized, Double-Blind and Controlled Trial. Front. Aging Neurosci. 2016, 8, 256. [Google Scholar] [CrossRef]
- Agahi, A.; Hamidi, G.A.; Daneshvar, R.; Hamdieh, M.; Soheili, M.; Alinaghipour, A.; Esmaeili Taba, S.M.; Salami, M. Does Severity of Alzheimer’s Disease Contribute to Its Responsiveness to Modifying Gut Microbiota? A Double Blind Clinical Trial. Front. Neurol. 2018, 9, 662. [Google Scholar] [CrossRef]
- Leblhuber, F.; Steiner, K.; Schuetz, B.; Fuchs, D.; Gostner, J.M. Probiotic Supplementation in Patients with Alzheimer’s Dementia—An Explorative Intervention Study. Curr. Alzheimer Res. 2018, 15, 1106–1113. [Google Scholar] [CrossRef] [PubMed]
- Ton, A.M.M.; Campagnaro, B.P.; Alves, G.A.; Aires, R.; Côco, L.Z.; Arpini, C.M.; Guerra, E.O.T.; Campos-Toimil, M.; Meyrelles, S.S.; Pereira, T.M.C.; et al. Oxidative Stress and Dementia in Alzheimer’s Patients: Effects of Synbiotic Supplementation. Oxid. Med. Cell. Longev. 2020, 2020, 2638703. [Google Scholar] [CrossRef]
- Malpas, C.B.; Vivash, L.; Genc, S.; Saling, M.M.; Desmond, P.; Steward, C.; Hicks, R.J.; Callahan, J.; Brodtmann, A.; Collins, S.; et al. A Phase IIa Randomized Control Trial of VEL015 (Sodium Selenate) in Mild-Moderate Alzheimer’s Disease. J. Alzheimer Dis. 2016, 54, 223–232. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, B.R.; Roberts, B.R.; Malpas, C.B.; Vivash, L.; Genc, S.; Saling, M.M.; Desmond, P.; Steward, C.; Hicks, R.J.; Callahan, J.; et al. Supranutritional Sodium Selenate Supplementation Delivers Selenium to the Central Nervous System: Results from a Randomized Controlled Pilot Trial in Alzheimer’s Disease. Neurotherapeutics 2019, 16, 192–202. [Google Scholar] [CrossRef] [PubMed]
- Tamtaji, O.R.; Heidari-Soureshjani, R.; Mirhosseini, N.; Kouchaki, E.; Bahmani, F.; Aghadavod, E.; Tajabadi-Ebrahimi, M.; Asemi, Z. Probiotic and selenium co-supplementation, and the effects on clinical, metabolic and genetic status in Alzheimer’s disease: A randomized, double-blind, controlled trial. Clin. Nutr. 2019, 38, 2569–2575. [Google Scholar] [CrossRef]
- Wade, A.G.; Farmer, M.; Harari, G.; Fund, N.; Laudon, M.; Nir, T.; Frydman-Marom, A.; Zisapel, N. Add-on prolonged-release melatonin for cognitive function and sleep in mild to moderate Alzheimer’s disease: A 6-month, randomized, placebo-controlled, multicenter trial. Clin. Interv. Aging 2014, 9, 947–961. [Google Scholar] [CrossRef]
- Turner, R.S.; Thomas, R.G.; Craft, S.; van Dyck, C.H.; Mintzer, J.; Reynolds, B.A.; Brewer, J.B.; Rissman, R.A.; Raman, R.; Aisen, P.S. A randomized, double-blind, placebo-controlled trial of resveratrol for Alzheimer disease. Neurology 2015, 85, 1383–1391. [Google Scholar] [CrossRef]
- Moussa, C.; Hebron, M.; Huang, X.; Ahn, J.; Rissman, R.A.; Aisen, P.S.; Turner, R.S. Resveratrol regulates neuro-inflammation and induces adaptive immunity in Alzheimer’s disease. J. Neuroinflamm. 2017, 14, 1. [Google Scholar] [CrossRef]
- Zhu, C.W.; Grossman, H.; Neugroschl, J.; Parker, S.; Burden, A.; Luo, X.; Sano, M. A randomized, double-blind, placebo-controlled trial of resveratrol with glucose and malate (RGM) to slow the progression of Alzheimer’s disease: A pilot study. Alzheimer Dement. 2018, 4, 609–616. [Google Scholar] [CrossRef]
- Fang, X.; Zhang, J.; Zhao, J.; Wang, L. Effect of Resveratrol Combined with Donepezil Hydrochloride on Inflammatory Factor Level and Cognitive Function Level of Patients with Alzheimer’s Disease. J. Healthc. Eng. 2022, 2022, 9148650. [Google Scholar] [CrossRef]
- Noguchi-Shinohara, M.; Ono, K.; Hamaguchi, T.; Nagai, T.; Kobayashi, S.; Komatsu, J.; Samuraki-Yokohama, M.; Iwasa, K.; Yokoyama, K.; Nakamura, H.; et al. Safety and efficacy of Melissa officinalis extract containing rosmarinic acid in the prevention of Alzheimer’s disease progression. Sci. Rep. 2020, 10, 18627. [Google Scholar] [CrossRef] [PubMed]
- Nolan, J.M.; Loskutova, E.; Howard, A.; Mulcahy, R.; Moran, R.; Stack, J.; Bolger, M.; Coen, R.F.; Dennison, J.; Akuffo, K.O.; et al. The impact of supplemental macular carotenoids in Alzheimer’s disease: A randomized clinical trial. J. Alzheimer Dis. 2015, 44, 1157–1169. [Google Scholar] [CrossRef]
- Nolan, J.M.; Mulcahy, R.; Power, R.; Moran, R.; Howard, A.N. Nutritional Intervention to Prevent Alzheimer’s Disease: Potential Benefits of Xanthophyll Carotenoids and Omega-3 Fatty Acids Combined. J. Alzheimer Dis. 2018, 64, 367–378. [Google Scholar] [CrossRef]
- Nolan, J.M.; Power, R.; Howard, A.N.; Bergin, P.; Roche, W.; Prado-Cabrero, A.; Pope, G.; Cooke, J.; Power, T.; Mulcahy, R. Supplementation With Carotenoids, Omega-3 Fatty Acids, and Vitamin E Has a Positive Effect on the Symptoms and Progression of Alzheimer’s Disease. J. Alzheimer Dis. 2022, 90, 233–249. [Google Scholar] [CrossRef] [PubMed]
- Ringman, J.M.; Frautschy, S.A.; Teng, E.; Begum, A.N.; Bardens, J.; Beigi, M.; Gylys, K.H.; Badmaev, V.; Heath, D.D.; Apostolova, L.G.; et al. Oral curcumin for Alzheimer’s disease: Tolerability and efficacy in a 24-week randomized, double blind, placebo-controlled study. Alzheimer Res. Ther. 2012, 4, 43. [Google Scholar] [CrossRef]
- Arlt, S.; Müller-Thomsen, T.; Beisiegel, U.; Kontush, A. Effect of one-year vitamin C- and E-supplementation on cerebrospinal fluid oxidation parameters and clinical course in Alzheimer’s disease. Neurochem. Res. 2012, 37, 2706–2714. [Google Scholar] [CrossRef] [PubMed]
- Galasko, D.R.; Peskind, E.; Clark, C.M.; Quinn, J.F.; Ringman, J.M.; Jicha, G.A.; Cotman, C.; Cottrell, B.; Montine, T.J.; Thomas, R.G.; et al. Antioxidants for Alzheimer disease: A randomized clinical trial with cerebrospinal fluid biomarker measures. Arch. Neurol. 2012, 69, 836–841. [Google Scholar] [CrossRef] [PubMed]
- Dysken, M.W.; Sano, M.; Asthana, S.; Vertrees, J.E.; Pallaki, M.; Llorente, M.; Love, S.; Schellenberg, G.D.; McCarten, J.R.; Malphurs, J.; et al. Effect of vitamin E and memantine on functional decline in Alzheimer disease: The TEAM-AD VA cooperative randomized trial. JAMA 2014, 311, 33–44. [Google Scholar] [CrossRef]
- Remington, R.; Bechtel, C.; Larsen, D.; Samar, A.; Doshanjh, L.; Fishman, P.; Luo, Y.; Smyers, K.; Page, R.; Morrell, C.; et al. A Phase II Randomized Clinical Trial of a Nutritional Formulation for Cognition and Mood in Alzheimer’s Disease. J. Alzheimer Dis. 2015, 45, 395–405. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations, World Health Organization. Probiotics in Food: Health and Nutritional Properties and Guidelines for Evaluation: Report of a Joint FAO/WHO Expert Consultation on Evaluation of Health and Nutritional Properties of Probiotics in Food including Powder Milk with Live Lactic Acid Bacteria, Cordoba, Argentina, 1–4 October 2001, [and] Report of a Joint FAO/WHO Working Group on Drafting Guidelines for the Evaluation of Probiotics in Food, London, Ontario, Canada, 30 April–1 May 2002; Food and Agriculture Organization of the United Nations, World Health Organization: Rome, Italy, 2006.
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef]
- Douglas, L.C.; Sanders, M.E. Probiotics and Prebiotics in Dietetics Practice. J. Am. Diet. Assoc. 2008, 108, 510–521. [Google Scholar] [CrossRef] [PubMed]
- Markowiak, P.; Śliżewska, K. Effects of Probiotics, Prebiotics, and Synbiotics on Human Health. Nutrients 2017, 9, 1021. [Google Scholar] [CrossRef] [PubMed]
- Westfall, S.; Lomis, N.; Kahouli, I.; Dia, S.Y.; Singh, S.P.; Prakash, S. Microbiome, probiotics and neurodegenerative diseases: Deciphering the gut brain axis. Cell. Mol. Life Sci. 2017, 74, 3769–3787. [Google Scholar] [CrossRef] [PubMed]
- Socała, K.; Doboszewska, U.; Szopa, A.; Serefko, A.; Włodarczyk, M.; Zielińska, A.; Poleszak, E.; Fichna, J.; Wlaź, P. The role of microbiota-gut-brain axis in neuropsychiatric and neurological disorders. Pharmacol. Res. 2021, 172, 105840. [Google Scholar] [CrossRef] [PubMed]
- Osadchiy, V.; Martin, C.R.; Mayer, E.A. The Gut-Brain Axis and the Microbiome: Mechanisms and Clinical Implications. Clin. Gastroenterol. Hepatol. 2019, 17, 322–332. [Google Scholar] [CrossRef] [PubMed]
- Hung, C.C.; Chang, C.C.; Huang, C.W.; Nouchi, R.; Cheng, C.H. Gut microbiota in patients with Alzheimer’s disease spectrum: A systematic review and meta-analysis. Aging 2022, 14, 477–496. [Google Scholar] [CrossRef] [PubMed]
- Naomi, R.; Embong, H.; Othman, F.; Ghazi, H.F.; Maruthey, N.; Bahari, H. Probiotics for Alzheimer’s Disease: A Systematic Review. Nutrients 2021, 14, 20. [Google Scholar] [CrossRef]
- Lekchand Dasriya, V.; Samtiya, M.; Dhewa, T.; Puniya, M.; Kumar, S.; Ranveer, S.; Chaudhary, V.; Vij, S.; Behare, P.; Singh, N.; et al. Etiology and management of Alzheimer’s disease: Potential role of gut microbiota modulation with probiotics supplementation. J. Food Biochem. 2022, 46, e14043. [Google Scholar] [CrossRef]
- Mishra, V.; Shah, C.; Mokashe, N.; Chavan, R.; Yadav, H.; Prajapati, J. Probiotics as potential antioxidants: A systematic review. J. Agric. Food Chem. 2015, 63, 3615–3626. [Google Scholar] [CrossRef]
- Mehrabadi, S.; Sadr, S.S. Assessment of Probiotics Mixture on Memory Function, Inflammation Markers, and Oxidative Stress in an Alzheimer’s Disease Model of Rats. Iran Biomed. J. 2020, 24, 220–228. [Google Scholar] [CrossRef]
- OMNi-BiOTiC® STRESS Repair—Stressed? Do Something about It! Available online: https://www.omni-biotic.com/en/products/omni-biotic-stress-repair/ (accessed on 3 March 2023).
- Picón-Pagès, P.; Garcia-Buendia, J.; Muñoz, F.J. Functions and dysfunctions of nitric oxide in brain. Biochim. Biophys. Acta Mol. Basis Dis. 2019, 1865, 1949–1967. [Google Scholar] [CrossRef] [PubMed]
- Roman, M.; Jitaru, P.; Barbante, C. Selenium biochemistry and its role for human health. Metallomics 2014, 6, 25–54. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.H.; Song, G.L. Roles of Selenoproteins in Brain Function and the Potential Mechanism of Selenium in Alzheimer’s Disease. Front Neurosci. 2021, 15, 646518. [Google Scholar] [CrossRef] [PubMed]
- Reddy, V.S.; Bukke, S.; Dutt, N.; Rana, P.; Pandey, A.K. A systematic review and meta-analysis of the circulatory, erythrocellular and CSF selenium levels in Alzheimer’s disease: A metal meta-analysis (AMMA study-I). J. Trace Elem. Med. Biol. 2017, 42, 68–75. [Google Scholar] [CrossRef]
- Kryscio, R.J.; Abner, E.L.; Caban-Holt, A.; Lovell, M.; Goodman, P.; Darke, A.K.; Yee, M.; Crowley, J.; Schmitt, F.A. Association of Antioxidant Supplement Use and Dementia in the Prevention of Alzheimer’s Disease by Vitamin E and Selenium Trial (PREADViSE). JAMA Neurol. 2017, 74, 567–573. [Google Scholar] [CrossRef]
- Rita Cardoso, B.; Apolinário, D.; da Silva Bandeira, V.; Busse, A.L.; Magaldi, R.M.; Jacob-Filho, W.; Cozzolino, S.M. Effects of Brazil nut consumption on selenium status and cognitive performance in older adults with mild cognitive impairment: A randomized controlled pilot trial. Eur. J. Nutr. 2016, 55, 107–116. [Google Scholar] [CrossRef]
- Sharma, S.K.; Bansal, M.P.; Sandhir, R. Altered dietary selenium influences brain iron content and behavioural outcomes. Behav. Brain. Res. 2019, 372, 112011. [Google Scholar] [CrossRef]
- Jiang, X.; Stockwell, B.R.; Conrad, M. Ferroptosis: Mechanisms, biology and role in disease. Nat. Rev. Mol. Cell. Biol. 2021, 22, 266–282. [Google Scholar] [CrossRef]
- Forcina, G.C.; Dixon, S.J. GPX4 at the Crossroads of Lipid Homeostasis and Ferroptosis. Proteomics 2019, 19, 1800311. [Google Scholar] [CrossRef]
- Stockwell, B.R.; Friedmann Angeli, J.P.; Bayir, H.; Bush, A.I.; Conrad, M.; Dixon, S.J.; Fulda, S.; Gascón, S.; Hatzios, S.K.; Kagan, V.E.; et al. Ferroptosis: A Regulated Cell Death Nexus Linking Metabolism, Redox Biology, and Disease. Cell 2017, 171, 273–285. [Google Scholar] [CrossRef]
- Damulina, A.; Pirpamer, L.; Soellradl, M.; Sackl, M.; Tinauer, C.; Hofer, E.; Enzinger, C.; Gesierich, B.; Duering, M.; Ropele, S.; et al. Cross-sectional and Longitudinal Assessment of Brain Iron Level in Alzheimer Disease Using 3-T MRI. Radiology 2020, 296, 619–626. [Google Scholar] [CrossRef]
- Kim, C.S.; Cha, L.; Sim, M.; Jung, S.; Chun, W.Y.; Baik, H.W.; Shin, D.M. Probiotic Supplementation Improves Cognitive Function and Mood with Changes in Gut Microbiota in Community-Dwelling Older Adults: A Randomized, Double-Blind, Placebo-Controlled, Multicenter Trial. J. Gerontol. A Biol. Sci. Med. Sci. 2021, 76, 32–40. [Google Scholar] [CrossRef]
- Claustrat, B.; Leston, J. Melatonin: Physiological effects in humans. Neurochirurgie 2015, 61, 77–84. [Google Scholar] [CrossRef]
- Vasey, C.; McBride, J.; Penta, K. Circadian Rhythm Dysregulation and Restoration: The Role of Melatonin. Nutrients 2021, 13, 3480. [Google Scholar] [CrossRef] [PubMed]
- Shukla, M.; Govitrapong, P.; Boontem, P.; Reiter, R.J.; Satayavivad, J. Mechanisms of Melatonin in Alleviating Alzheimer’s Disease. Curr. Neuropharmacol. 2017, 15, 1010–1031. [Google Scholar] [CrossRef] [PubMed]
- Nous, A.; Engelborghs, S.; Smolders, I. Melatonin levels in the Alzheimer’s disease continuum: A systematic review. Alzheimer Res. Ther. 2021, 13, 52. [Google Scholar] [CrossRef] [PubMed]
- Homolak, J.; Mudrovčić, M.; Vukić, B.; Toljan, K. Circadian Rhythm and Alzheimer’s Disease. Med. Sci. 2018, 6, 52. [Google Scholar] [CrossRef]
- Aldhous, M.; Franey, C.; Wright, J.; Arendt, J. Plasma concentrations of melatonin in man following oral absorption of different preparations. Br. J. Clin. Pharmacol. 1985, 19, 517–521. [Google Scholar] [CrossRef]
- Zisapel, N. Melatonin and Sleep. Open Neuroendocrinol. J. 2010, 3, 85–95. [Google Scholar]
- Tan, D.X.; Manchester, L.C.; Esteban-Zubero, E.; Zhou, Z.; Reiter, R.J. Melatonin as a Potent and Inducible Endogenous Antioxidant: Synthesis and Metabolism. Molecules 2015, 20, 18886–18906. [Google Scholar] [CrossRef] [PubMed]
- Rondanelli, M.; Opizzi, A.; Faliva, M.; Mozzoni, M.; Antoniello, N.; Cazzola, R.; Savarè, R.; Cerutti, R.; Grossi, E.; Cestaro, B. Effects of a diet integration with an oily emulsion of DHA-phospholipids containing melatonin and tryptophan in elderly patients suffering from mild cognitive impairment. Nutr. Neurosci. 2012, 15, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Aguilar, M.A.; Ramírez-Salado, I.; Cruz-Ulloa, C.; Benítez-King, G.A. Melatonin effects on macro sleep architecture in Alzheimer ‘ s disease patients. Salud Ment. 2013, 36, 243–449. [Google Scholar]
- Cruz-Aguilar, M.A.; Ramírez-Salado, I.; Guevara, M.A.; Hernández-González, M.; Benitez-King, G. Melatonin Effects on EEG Activity During Sleep Onset in Mild-to-Moderate Alzheimer’s Disease: A Pilot Study. J. Alzheimer Dis. Rep. 2018, 2, 55–65. [Google Scholar] [CrossRef]
- Singer, C.; Tractenberg, R.E.; Kaye, J.; Schafer, K.; Gamst, A.; Grundman, M.; Thomas, R.; Thal, L.J. A multicenter, placebo-controlled trial of melatonin for sleep disturbance in Alzheimer’s disease. Sleep 2003, 26, 893–901. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, J.; Wan, J.; Liu, A.; Sun, J. Melatonin regulates Aβ production/clearance balance and Aβ neurotoxicity: A potential therapeutic molecule for Alzheimer’s disease. Biomed. Pharmacother. 2020, 132, 110887. [Google Scholar] [CrossRef]
- Solís-Chagoyán, H.; Domínguez-Alonso, A.; Valdés-Tovar, M.; Argueta, J.; Sánchez-Florentino, Z.A.; Calixto, E.; Benítez-King, G. Melatonin Rescues the Dendrite Collapse Induced by the Pro-Oxidant Toxin Okadaic Acid in Organotypic Cultures of Rat Hilar Hippocampus. Molecules 2020, 25, 5508. [Google Scholar] [CrossRef]
- Yao, K.; Zhao, Y.F.; Zu, H.B. Melatonin receptor stimulation by agomelatine prevents Aβ-induced tau phosphorylation and oxidative damage in PC12 cells. Drug Des. Devel. Ther. 2019, 13, 387–396. [Google Scholar] [CrossRef]
- Tyagi, E.; Agrawal, R.; Nath, C.; Shukla, R. Effect of melatonin on neuroinflammation and acetylcholinesterase activity induced by LPS in rat brain. Eur. J. Pharmacol. 2010, 640, 206–210. [Google Scholar] [CrossRef]
- Roy, J.; Tsui, K.C.; Ng, J.; Fung, M.L.; Lim, L.W. Regulation of Melatonin and Neurotransmission in Alzheimer’s Disease. Int. J. Mol. Sci. 2021, 22, 6841. [Google Scholar] [CrossRef]
- Arbo, B.D.; André-Miral, C.; Nasre-Nasser, R.G.; Schimith, L.E.; Santos, M.G.; Costa-Silva, D.; Muccillo-Baisch, A.L.; Hort, M.A. Resveratrol derivatives as potential treatments for Alzheimer’s and Parkinson’s disease. Front. Aging Neurosci. 2020, 12, 103. [Google Scholar] [CrossRef] [PubMed]
- Tian, B.; Liu, J. Resveratrol: A review of plant sources, synthesis, stability, modification and food application. J. Sci. Food Agric. 2020, 100, 1392–1404. [Google Scholar] [CrossRef] [PubMed]
- Grinan-Ferre, C.; Bellver-Sanchis, A.; Izquierdo, V.; Corpas, R.; Roig-Soriano, J.; Chillón, M.; Andres-Lacueva, C.; Somogyvári, M.; Sőti, C.; Sanfeliu, C. The pleiotropic neuroprotective effects of resveratrol in cognitive decline and Alzheimer’s disease pathology: From antioxidant to epigenetic therapy. Ageing Res. Rev. 2021, 67, 101271. [Google Scholar] [CrossRef]
- Lee, J.; Torosyan, N.; Silverman, D.H. Examining the impact of grape consumption on brain metabolism and cognitive function in patients with mild decline in cognition: A double-blinded placebo controlled pilot study. Exp. Gerontol. 2017, 87, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Caruana, M.; Cauchi, R.; Vassallo, N. Putative Role of Red Wine Polyphenols against Brain Pathology in Alzheimer’s and Parkinson’s Disease. Front. Nutr. 2016, 3, 31. [Google Scholar] [CrossRef]
- Fischer, K.; Melo van Lent, D.; Wolfsgruber, S.; Weinhold, L.; Kleineidam, L.; Bickel, H.; Scherer, M.; Eisele, M.; van den Bussche, H.; Wiese, B.; et al. Prospective Associations between Single Foods, Alzheimer’s Dementia and Memory Decline in the Elderly. Nutrients 2018, 10, 852. [Google Scholar] [CrossRef]
- Hu, N.; Yu, J.T.; Tan, L.; Wang, Y.L.; Sun, L.; Tan, L. Nutrition and the risk of Alzheimer’s disease. Biomed. Res. Int. 2013, 2013, 524820. [Google Scholar] [CrossRef] [PubMed]
- Drygalski, K.; Fereniec, E.; Koryciński, K.; Chomentowski, A.; Kiełczewska, A.; Odrzygóźdź, C.; Modzelewska, B. Resveratrol and Alzheimer’s disease. Mol. Pathophysiol. Clin. Trials Exp. Gerontol. 2018, 113, 36–47. [Google Scholar] [CrossRef]
- Ma, T.; Tan, M.-S.; Yu, J.-T.; Tan, L. Resveratrol as a Therapeutic Agent for Alzheimer’s Disease. BioMed Res. Int. 2014, 2014, 350516. [Google Scholar] [CrossRef]
- Tosatti, J.A.G.; Fontes, A.; Caramelli, P.; Gomes, K.B. Effects of Resveratrol Supplementation on the Cognitive Function of Patients with Alzheimer’s Disease: A Systematic Review of Randomized Controlled Trials. Drugs Aging 2022, 39, 285–295. [Google Scholar] [CrossRef]
- Olsson, B.; Lautner, R.; Andreasson, U.; Öhrfelt, A.; Portelius, E.; Bjerke, M.; Hölttä, M.; Rosén, C.; Olsson, C.; Strobel, G.; et al. CSF and blood biomarkers for the diagnosis of Alzheimer’s disease: A systematic review and meta-analysis. Lancet Neurol. 2016, 15, 673–684. [Google Scholar] [CrossRef] [PubMed]
- Dasgupta, B.; Milbrandt, J. Resveratrol stimulates AMP kinase activity in neurons. Proc. Natl. Acad. Sci. USA 2007, 104, 7217–7222. [Google Scholar] [CrossRef] [PubMed]
- Chiang, M.C.; Nicol, C.J.; Cheng, Y.C. Resveratrol activation of AMPK-dependent pathways is neuroprotective in human neural stem cells against amyloid-beta-induced inflammation and oxidative stress. Neurochem. Int. 2018, 115, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Nguyen, M.D.; Dobbin, M.M.; Fischer, A.; Sananbenesi, F.; Rodgers, J.T.; Delalle, I.; Baur, J.A.; Sui, G.; Armour, S.M.; et al. SIRT1 deacetylase protects against neurodegeneration in models for Alzheimer’s disease and amyotrophic lateral sclerosis. EMBO J. 2007, 26, 3169–3179. [Google Scholar] [CrossRef] [PubMed]
- Kong, D.; Yan, Y.; He, X.Y.; Yang, H.; Liang, B.; Wang, J.; He, Y.; Ding, Y.; Yu, H. Effects of Resveratrol on the Mechanisms of Antioxidants and Estrogen in Alzheimer’s Disease. BioMed Res. Int. 2019, 2019, 8983752. [Google Scholar] [CrossRef] [PubMed]
- Ngo, Y.L.; Lau, C.H.; Chua, L.S. Review on rosmarinic acid extraction, fractionation and its anti-diabetic potential. Food Chem. Toxicol. 2018, 121, 687–700. [Google Scholar] [CrossRef]
- Ghasemzadeh Rahbardar, M.; Hosseinzadeh, H. Effects of rosmarinic acid on nervous system disorders: An updated review. Naunyn Schmiedebergs Arch. Pharmacol. 2020, 393, 1779–1795. [Google Scholar] [CrossRef]
- Petersen, M.; Abdullah, Y.; Benner, J.; Eberle, D.; Gehlen, K.; Hücherig, S.; Janiak, V.; Kim, K.H.; Sander, M.; Weitzel, C.; et al. Evolution of rosmarinic acid biosynthesis. Phytochemistry 2009, 70, 1663–1679. [Google Scholar] [CrossRef]
- Hassanzadeh-Taheri, M.; Ahmadi-Zohan, A.; Mohammadifard, M.; Hosseini, M. Rosmarinic acid attenuates lipopolysaccharide-induced neuroinflammation and cognitive impairment in rats. J. Chem. Neuroanat. 2021, 117, 102008. [Google Scholar] [CrossRef]
- Kola, A.; Hecel, A.; Lamponi, S.; Valensin, D. Novel Perspective on Alzheimer’s Disease Treatment: Rosmarinic Acid Molecular Interplay with Copper(II) and Amyloid β. Life 2020, 10, 118. [Google Scholar] [CrossRef]
- Hitl, M.; Kladar, N.; Gavarić, N.; Božin, B. Rosmarinic Acid-Human Pharmacokinetics and Health Benefits. Planta Med. 2021, 87, 273–282. [Google Scholar] [CrossRef]
- Mirza, F.J.; Amber, S.; Sumera; Hassan, D.; Ahmed, T.; Zahid, S. Rosmarinic acid and ursolic acid alleviate deficits in cognition, synaptic regulation and adult hippocampal neurogenesis in an Aβ(1-42)-induced mouse model of Alzheimer’s disease. Phytomedicine 2021, 83, 153490. [Google Scholar] [CrossRef]
- Vasileva, L.V.; Savova, M.S.; Tews, D.; Wabitsch, M.; Georgiev, M.I. Rosmarinic acid attenuates obesity and obesity-related inflammation in human adipocytes. Food Chem. Toxicol. 2021, 149, 112002. [Google Scholar] [CrossRef] [PubMed]
- Dahchour, A. Anxiolytic and antidepressive potentials of rosmarinic acid: A review with a focus on antioxidant and anti-inflammatory effects. Pharmacol. Res. 2022, 184, 106421. [Google Scholar] [CrossRef] [PubMed]
- Cases, J.; Ibarra, A.; Feuillère, N.; Roller, M.; Sukkar, S.G. Pilot trial of Melissa officinalis L. leaf extract in the treatment of volunteers suffering from mild-to-moderate anxiety disorders and sleep disturbances. Mediterr. J. Nutr. Metab. 2011, 4, 211–218. [Google Scholar] [CrossRef]
- Ranjbar, M.; Firoozabadi, A.; Salehi, A.; Ghorbanifar, Z.; Zarshenas, M.M.; Sadeghniiat-Haghighi, K.; Rezaeizadeh, H. Effects of Herbal combination (Melissa officinalis L. and Nepeta menthoides Boiss. & Buhse) on insomnia severity, anxiety and depression in insomniacs: Randomized placebo controlled trial. Integr. Med. Res. 2018, 7, 328–332. [Google Scholar] [CrossRef] [PubMed]
- Herrlinger, K.A.; Nieman, K.M.; Sanoshy, K.D.; Fonseca, B.A.; Lasrado, J.A.; Schild, A.L.; Maki, K.C.; Wesnes, K.A.; Ceddia, M.A. Spearmint Extract Improves Working Memory in Men and Women with Age-Associated Memory Impairment. J. Altern. Complement. Med. 2018, 24, 37–47. [Google Scholar] [CrossRef]
- Milani, A.; Basirnejad, M.; Shahbazi, S.; Bolhassani, A. Carotenoids: Biochemistry, pharmacology and treatment. Br. J. Pharmacol. 2017, 174, 1290–1324. [Google Scholar] [CrossRef]
- Carazo, A.; Macáková, K.; Matoušová, K.; Krčmová, L.K.; Protti, M.; Mladěnka, P. Vitamin A Update: Forms, Sources, Kinetics, Detection, Function, Deficiency, Therapeutic Use and Toxicity. Nutrients 2021, 13, 1703. [Google Scholar] [CrossRef]
- Toti, E.; Chen, C.O.; Palmery, M.; Villaño Valencia, D.; Peluso, I. Non-Provitamin A and Provitamin A Carotenoids as Immunomodulators: Recommended Dietary Allowance, Therapeutic Index, or Personalized Nutrition? Oxid. Med. Cell. Longev. 2018, 2018, 4637861. [Google Scholar] [CrossRef]
- Abdel-Aal el, S.M.; Akhtar, H.; Zaheer, K.; Ali, R. Dietary sources of lutein and zeaxanthin carotenoids and their role in eye health. Nutrients 2013, 5, 1169–1185. [Google Scholar] [CrossRef] [PubMed]
- Kabir, M.T.; Rahman, M.H.; Shah, M.; Jamiruddin, M.R.; Basak, D.; Al-Harrasi, A.; Bhatia, S.; Ashraf, G.M.; Najda, A.; El-Kott, A.F.; et al. Therapeutic promise of carotenoids as antioxidants and anti-inflammatory agents in neurodegenerative disorders. Biomed. Pharmacother. 2022, 146, 112610. [Google Scholar] [CrossRef]
- Bhatti, A.B.; Usman, M.; Ali, F.; Satti, S.A. Vitamin Supplementation as an Adjuvant Treatment for Alzheimer’s Disease. J. Clin. Diagn. Res. 2016, 10, OE07–OE11. [Google Scholar] [CrossRef] [PubMed]
- Min, J.Y.; Min, K.B. Serum lycopene, lutein and zeaxanthin, and the risk of Alzheimer’s disease mortality in older adults. Dement. Geriatr. Cogn. Disord. 2014, 37, 246–256. [Google Scholar] [CrossRef] [PubMed]
- Yasuno, F.; Tanimukai, S.; Sasaki, M.; Ikejima, C.; Yamashita, F.; Kodama, C.; Mizukami, K.; Asada, T. Combination of antioxidant supplements improved cognitive function in the elderly. J. Alzheimer Dis. 2012, 32, 895–903. [Google Scholar] [CrossRef]
- Mecocci, P.; Polidori, M.C. Antioxidant clinical trials in mild cognitive impairment and Alzheimer’s disease. Biochim. Biophys. Acta 2012, 1822, 631–638. [Google Scholar] [CrossRef]
- Reddy, P.H.; Manczak, M.; Yin, X.; Grady, M.C.; Mitchell, A.; Tonk, S.; Kuruva, C.S.; Bhatti, J.S.; Kandimalla, R.; Vijayan, M.; et al. Protective Effects of Indian Spice Curcumin Against Amyloid-β in Alzheimer’s Disease. J. Alzheimer Dis. 2018, 61, 843–866. [Google Scholar] [CrossRef]
- Priyadarsini, K.I. The Chemistry of Curcumin: From Extraction to Therapeutic Agent. Molecules 2014, 19, 20091–20112. [Google Scholar] [CrossRef]
- Kim, Y.; Clifton, P. Curcumin, Cardiometabolic Health and Dementia. Int. J. Environ. Res. Public Health 2018, 15, 2093. [Google Scholar] [CrossRef]
- Polidori, M.C.; Nelles, G. Antioxidant clinical trials in mild cognitive impairment and Alzheimer’s disease—Challenges and perspectives. Curr. Pharm. Des. 2014, 20, 3083–3092. [Google Scholar] [CrossRef]
- Ng, T.P.; Chiam, P.C.; Lee, T.; Chua, H.C.; Lim, L.; Kua, E.H. Curry consumption and cognitive function in the elderly. Am. J. Epidemiol. 2006, 164, 898–906. [Google Scholar] [CrossRef]
- Ng, T.P.; Nyunt, M.S.Z.; Gao, Q.; Gwee, X.; Chua, D.Q.L.; Yap, K.B. Curcumin-Rich Curry Consumption and Neurocognitive Function from 4.5-Year Follow-Up of Community-Dwelling Older Adults (Singapore Longitudinal Ageing Study). Nutrients 2022, 14, 1189. [Google Scholar] [CrossRef] [PubMed]
- Prasad, S.; DuBourdieu, D.; Srivastava, A.; Kumar, P.; Lall, R. Metal-Curcumin Complexes in Therapeutics: An Approach to Enhance Pharmacological Effects of Curcumin. Int. J. Mol. Sci. 2021, 22, 7094. [Google Scholar] [CrossRef]
- Chen, M.; Du, Z.Y.; Zheng, X.; Li, D.L.; Zhou, R.P.; Zhang, K. Use of curcumin in diagnosis, prevention, and treatment of Alzheimer’s disease. Neural Regen. Res. 2018, 13, 742–752. [Google Scholar] [CrossRef]
- Small, G.W.; Siddarth, P.; Li, Z.; Miller, K.J.; Ercoli, L.; Emerson, N.D.; Martinez, J.; Wong, K.P.; Liu, J.; Merrill, D.A.; et al. Memory and Brain Amyloid and Tau Effects of a Bioavailable Form of Curcumin in Non-Demented Adults: A Double-Blind, Placebo-Controlled 18-Month Trial. Am. J. Geriatr. Psychiatry 2018, 26, 266–277. [Google Scholar] [CrossRef]
- Voulgaropoulou, S.D.; van Amelsvoort, T.; Prickaerts, J.; Vingerhoets, C. The effect of curcumin on cognition in Alzheimer’s disease and healthy aging: A systematic review of pre-clinical and clinical studies. Brain Res. 2019, 1725, 146476. [Google Scholar] [CrossRef]
- Dei Cas, M.; Ghidoni, R. Dietary Curcumin: Correlation between Bioavailability and Health Potential. Nutrients 2019, 11, 2147. [Google Scholar] [CrossRef]
- Jiang, Q. Natural forms of vitamin E: Metabolism, antioxidant, and anti-inflammatory activities and their role in disease prevention and therapy. Free. Radic. Biol. Med. 2014, 72, 76–90. [Google Scholar] [CrossRef] [PubMed]
- Zaaboul, F.; Liu, Y. Vitamin E in foodstuff: Nutritional, analytical, and food technology aspects. Compr. Rev. Food Sci. Food Saf. 2022, 21, 964–998. [Google Scholar] [CrossRef] [PubMed]
- Engin, K.N. Alpha-tocopherol: Looking beyond an antioxidant. Mol. Vis. 2009, 15, 855–860. [Google Scholar]
- Browne, D.; McGuinness, B.; Woodside, J.V.; McKay, G.J. Vitamin E and Alzheimer’s disease: What do we know so far? Clin. Interv. Aging 2019, 14, 1303–1317. [Google Scholar] [CrossRef] [PubMed]
- Lopes da Silva, S.; Vellas, B.; Elemans, S.; Luchsinger, J.; Kamphuis, P.; Yaffe, K.; Sijben, J.; Groenendijk, M.; Stijnen, T. Plasma nutrient status of patients with Alzheimer’s disease: Systematic review and meta-analysis. Alzheimer Dement. 2014, 10, 485–502. [Google Scholar] [CrossRef]
- Sano, M.; Ernesto, C.; Thomas, R.G.; Klauber, M.R.; Schafer, K.; Grundman, M.; Woodbury, P.; Growdon, J.; Cotman, C.W.; Pfeiffer, E.; et al. A controlled trial of selegiline, alpha-tocopherol, or both as treatment for Alzheimer’s disease. The Alzheimer’s Disease Cooperative Study. N. Engl. J. Med. 1997, 336, 1216–1222. [Google Scholar] [CrossRef]
- Remington, R.; Bechtel, C.; Larsen, D.; Samar, A.; Page, R.; Morrell, C.; Shea, T.B. Maintenance of Cognitive Performance and Mood for Individuals with Alzheimer’s Disease Following Consumption of a Nutraceutical Formulation: A One-Year, Open-Label Study. J. Alzheimer Dis. 2016, 51, 991–995. [Google Scholar] [CrossRef]
- Boccardi, V.; Baroni, M.; Mangialasche, F.; Mecocci, P. Vitamin E family: Role in the pathogenesis and treatment of Alzheimer’s disease. Alzheimer Dement. Transl. Res. Clin. Interv. 2016, 2, 182–191. [Google Scholar] [CrossRef] [PubMed]
- Giraldo, E.; Lloret, A.; Fuchsberger, T.; Viña, J. Aβ and tau toxicities in Alzheimer’s are linked via oxidative stress-induced p38 activation: Protective role of vitamin E. Redox Biol. 2014, 2, 873–877. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, N.F.; Yanagisawa, D.; Durani, L.W.; Hamezah, H.S.; Damanhuri, H.A.; Wan Ngah, W.Z.; Tsuji, M.; Kiuchi, Y.; Ono, K.; Tooyama, I. Tocotrienol-Rich Fraction Modulates Amyloid Pathology and Improves Cognitive Function in AβPP/PS1 Mice. J. Alzheimer Dis. 2017, 55, 597–612. [Google Scholar] [CrossRef]
- Desrumaux, C.; Pisoni, A.; Meunier, J.; Deckert, V.; Athias, A.; Perrier, V.; Villard, V.; Lagrost, L.; Verdier, J.M.; Maurice, T. Increased amyloid-β peptide-induced memory deficits in phospholipid transfer protein (PLTP) gene knockout mice. Neuropsychopharmacology 2013, 38, 817–825. [Google Scholar] [CrossRef]
- Sedaghat, A.; Samadi, M.; Shirvani, H.; Sepandi, M.; Tahmasebi, W. Coenzyme Q10 Supplementation and Oxidative Stress Parameters: An Updated Systematic Review and Meta-analysis of Randomized Controlled Clinical Trials. Asian J. Sports Med. 2022, 13, e131308. [Google Scholar] [CrossRef]
- Akbari, A.; Mobini, G.R.; Agah, S.; Morvaridzadeh, M.; Omidi, A.; Potter, E.; Fazelian, S.; Ardehali, S.H.; Daneshzad, E.; Dehghani, S. Coenzyme Q10 supplementation and oxidative stress parameters: A systematic review and meta-analysis of clinical trials. Eur. J. Clin. Pharmacol. 2020, 76, 1483–1499. [Google Scholar] [CrossRef]
- Testai, L.; Martelli, A.; Flori, L.; Cicero, A.F.G.; Colletti, A. Coenzyme Q(10): Clinical Applications beyond Cardiovascular Diseases. Nutrients 2021, 13, 1697. [Google Scholar] [CrossRef] [PubMed]
- Mantle, D.; Heaton, R.A.; Hargreaves, I.P. Coenzyme Q10, Ageing and the Nervous System: An Overview. Antioxidants 2021, 11, 2. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Jiménez, F.J.; Alonso-Navarro, H.; García-Martín, E.; Agúndez, J.A.G. Coenzyme Q10 and Dementia: A Systematic Review. Antioxidants 2023, 12, 533. [Google Scholar] [CrossRef] [PubMed]
- University Hospital, Geneva. Dynamic Response to Probiotics in Context of Alzheimer Disease: A Proof-of-Concept Study. Available online: https://ClinicalTrials.gov/show/NCT05521477 (accessed on 10 January 2023).
- Preventive Medicine Research Institute; University of California, San Francisco; Harvard Medical School; University of California, San Diago; The Cleveland Clinic; Renown Health. Can Lifestyle Changes Reverse Early-Stage Alzheimer’s Disease. Available online: https://ClinicalTrials.gov/show/NCT04606420 (accessed on 10 January 2023).
- Hsieh-Hsun, H. Effect of Probiotics in Alzheimer’s Disease. Available online: https://ClinicalTrials.gov/show/NCT05145881 (accessed on 10 January 2023).
- Natalie, D. Disease Modifying Potential of 5mg of Melatonin on Cognition and Brain Health in Aging. Available online: https://ClinicalTrials.gov/show/NCT03954899 (accessed on 10 January 2023).
- India Globalization Capital Inc. IGC-AD1 Trial on Agitation in Dementia Due to Alzheimer’s. Available online: https://ClinicalTrials.gov/show/NCT05543681 (accessed on 10 January 2023).
- Venturelli, M.; University of Perugia; INCLIVA; Molde University College; University of Liverpool; Molecular Horizon S.r.l.; Nestlé Italiana, S.p.A. Chocolate and Physical Exercise to Reduce Malnutrition in Pre-dementia Aged People. Available online: https://ClinicalTrials.gov/show/NCT05343611 (accessed on 10 January 2023).
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).