Different Types of Non-Starch Polysaccharides Alter the Growth, Intestinal Flora and Serum Metabolite Profile of Grass Carp, Ctenopharyngodon idella
Abstract
1. Introduction
2. Material and Methods
2.1. Feed and Test Animal
2.2. Sampling Procedures
2.3. Analysis Methods
2.3.1. Chemical Analysis
2.3.2. Blood Biochemistry Parameters Measurement
2.3.3. Digestive Enzyme Activity Measurement
2.4. Gut Microbiota Analysis
2.5. Serum Metabolome Analysis
2.6. Statistical Analysis
3. Results
3.1. Growth Performance
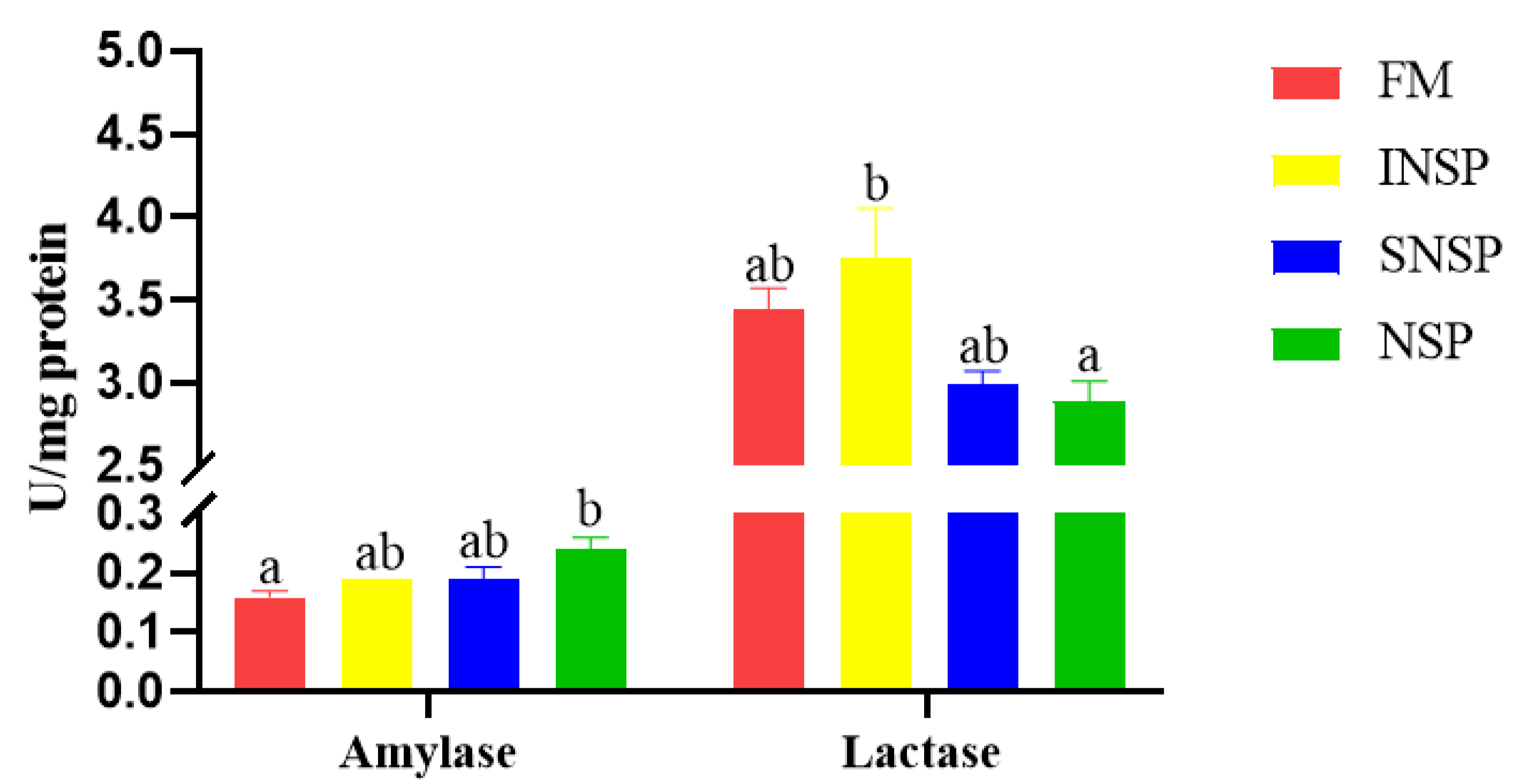
3.2. Digestive Enzyme Activity
3.3. Serum Lipoprotein Contents
3.4. Serum Protein Metabolism Indicators
3.5. Antioxidant Capacity Status
3.6. Whole-Body Composition and Dorsal Muscle Amino Profile
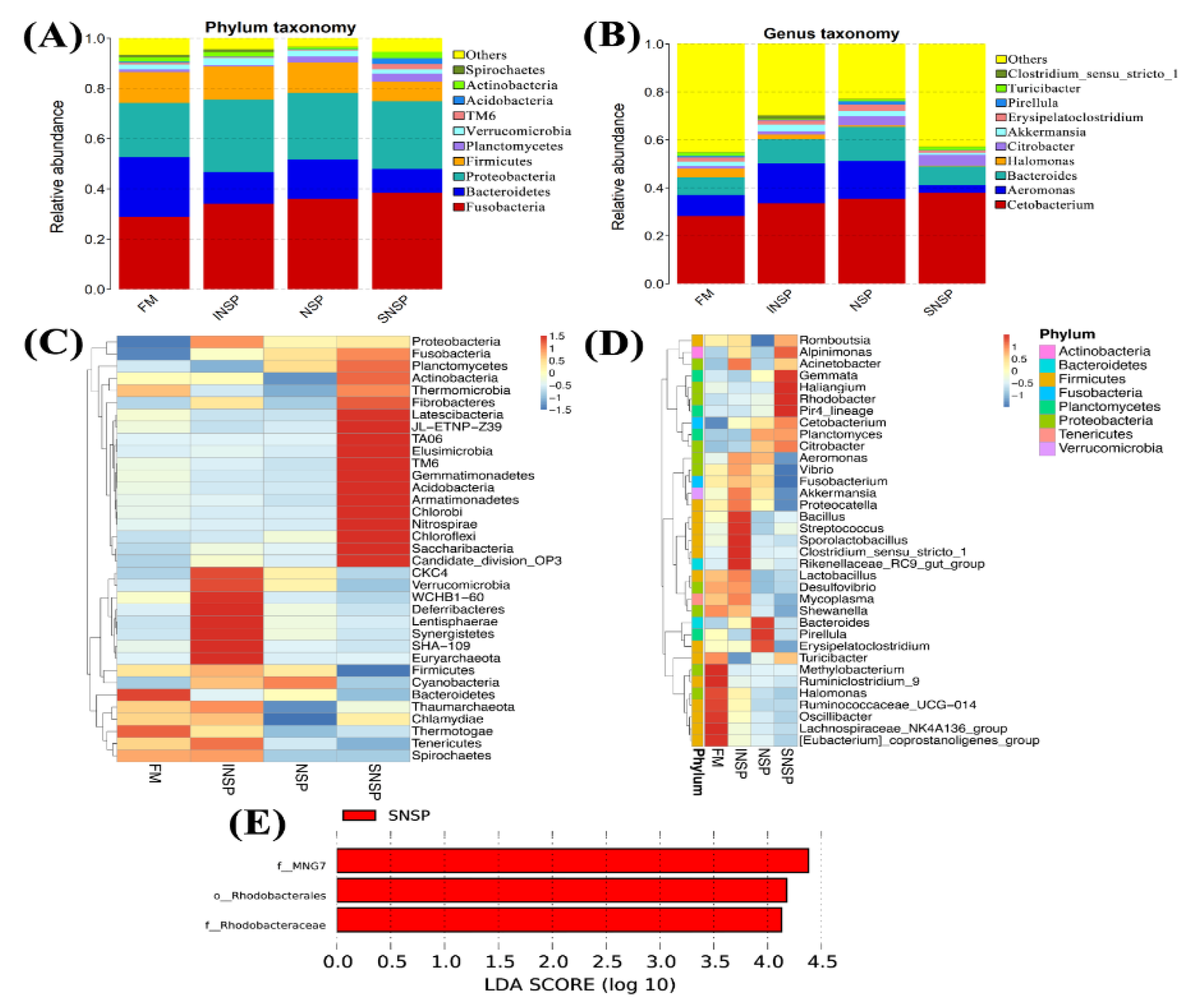
3.7. Gut Microbiota Structure
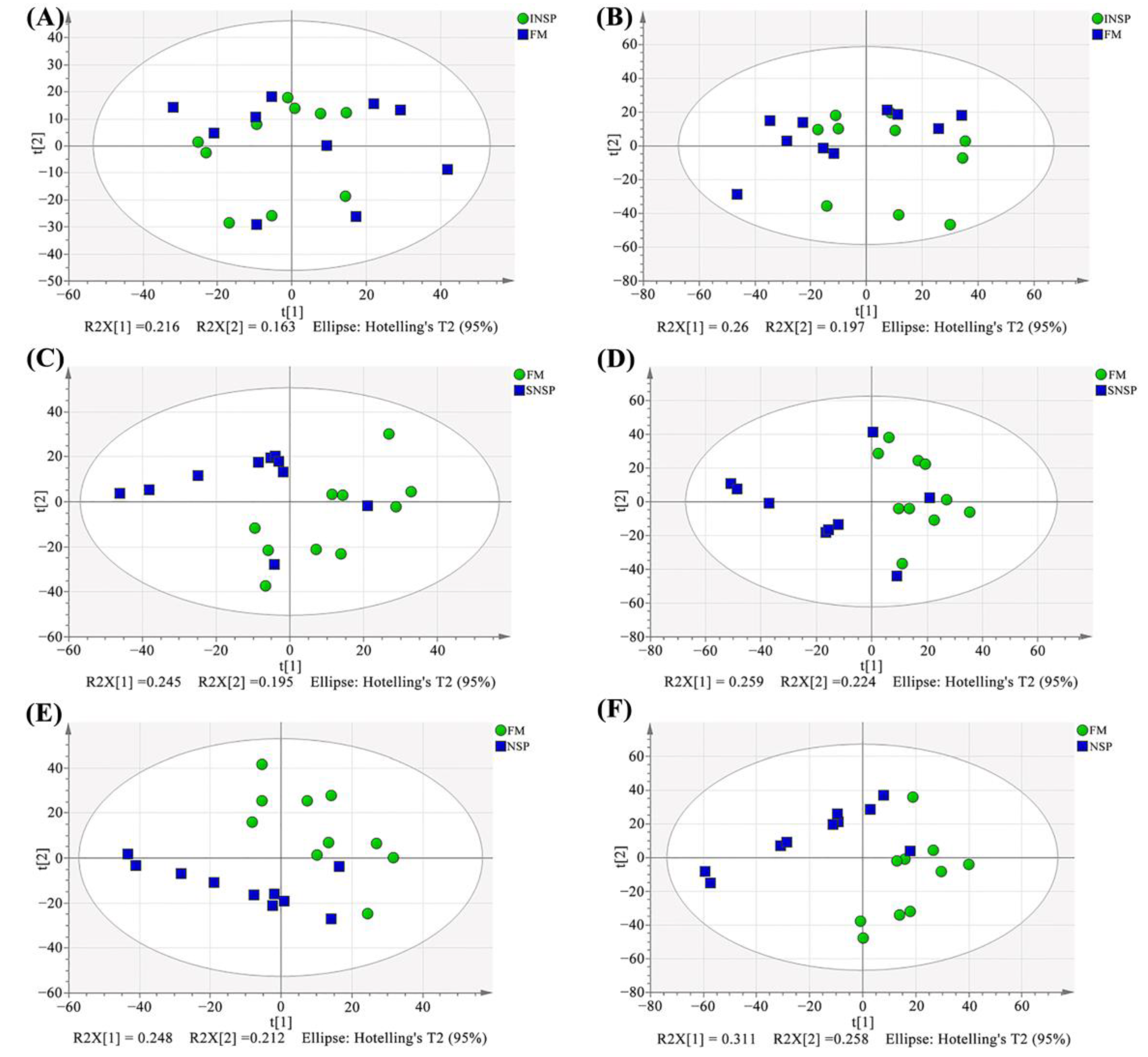
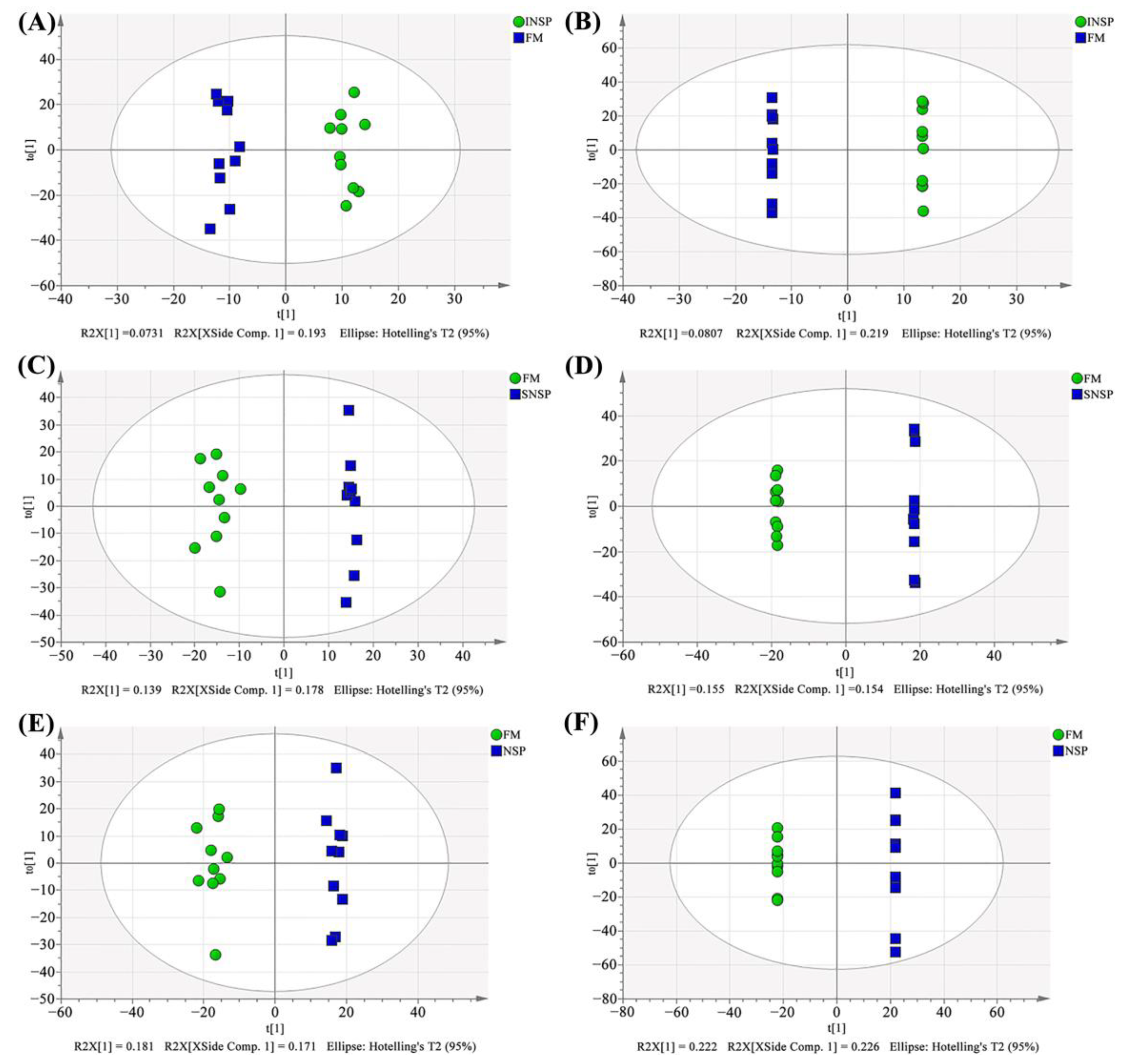
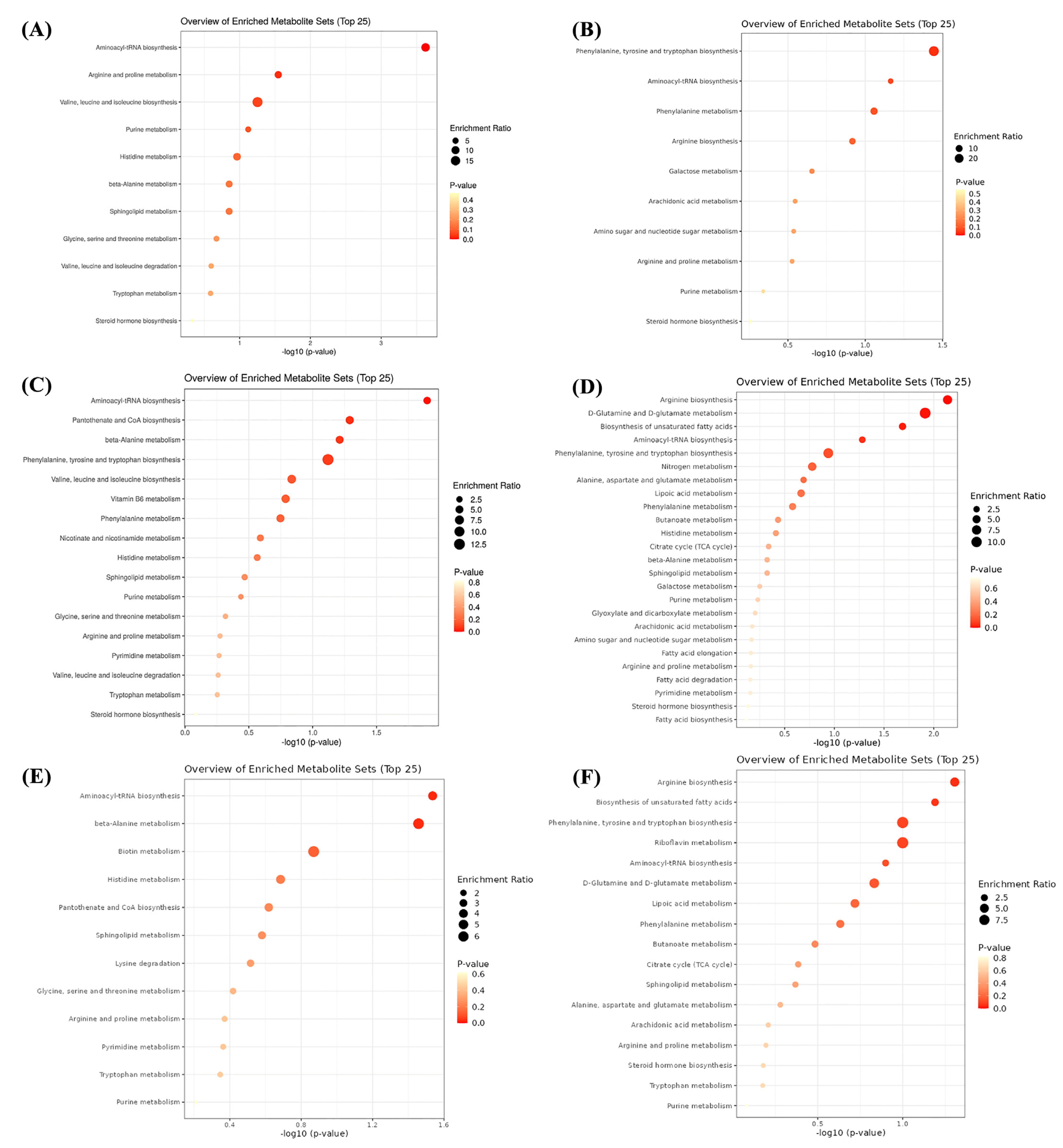
3.8. Serum Metabolome
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Deng, J.; Zhang, X.; Sun, Y.; Mi, H.; Zhang, L. Effects of different types of non-starch polysaccharides on growth, digestive enzyme activity, intestinal barrier function and antioxidant activity of rainbow trout (Oncorhynchus mykiss). Aqua. Rep. 2021, 21, 100864. [Google Scholar] [CrossRef]
- Ren, S.; Cai, C.; Cui, G.; Ni, Q.; Jiang, R.; Su, X.; Wang, Q.; Chen, W.; Zhang, J.; Wu, P.; et al. High dosages of pectin and cellulose cause different degrees of damage to the livers and intestines of Pelteobagrus fulvidraco. Aquaculture 2020, 514, 734445. [Google Scholar] [CrossRef]
- Kuz’mina, V.V. Influence of age on digestive enzyme activity in some freshwater teleosts. Aquaculture 1996, 148, 25–37. [Google Scholar] [CrossRef]
- Sinha, A.K.; Kumar, V.; Makkar, H.P.S.; De Boeck, G.; Becker, K. Non-starch polysaccharides and their role in fish nutrition—A review. Food Chem. 2011, 127, 1409–1426. [Google Scholar] [CrossRef]
- Cai, C.; Ren, S.; Cui, G.; Ni, Q.; Li, X.; Meng, Y.; Meng, Z.; Zhang, J.; Su, X.; Chen, H.; et al. Short-term stress due to dietary pectin induces cholestasis, and chronic stress induces hepatic steatosis and fibrosis in yellow catfish, Pelteobagrus fulvidraco. Aquaculture 2020, 516, 734607. [Google Scholar] [CrossRef]
- Liu, Y.; Deng, J.; Tan, B.; Xie, S.; Zhang, W. Effects of soluble and insoluble non-starch polysaccharides on growth performance, digestive enzyme activity, antioxidant capacity, and intestinal flora of juvenile genetic of improvement of farmed tilapia (Oreochromis niloticus). Front. Mar. Sci. 2022, 9, 872577. [Google Scholar] [CrossRef]
- Liu, Y.; Huang, H.; Fan, J.; Zhou, H.; Zhang, Y.; Cao, Y.; Jiang, W.; Zhang, W.; Deng, J.; Tan, B. Effects of dietary non-starch polysaccharides level on the growth, intestinal flora and intestinal health of juvenile largemouth bass Micropterus salmoides. Aquaculture 2022, 557, 738343. [Google Scholar] [CrossRef]
- Liu, Y.; Cao, Y.; Zhang, Y.; Fan, J.; Zhou, H.; Huang, H.; Jiang, W.; Zhang, W.; Deng, J.; Tan, B. Intestinal flora and immunity response to different viscous diets in juvenile largemouth bass, micropterus salmoides. Fish Shellfish Immunol. 2022, 127, 1012–1023. [Google Scholar] [CrossRef]
- Zhou, Q.; Mai, K.; Liu, Y.; Tan, B. Advances in animal and plant protein sources in place of fish meal. J. Fish. China 2005, 29, 404–410. [Google Scholar]
- Hossain, M.M.; Ali, M.L.; Khan, S.; Haque, M.M.; Shahjahan, M. Use of asian watergrass as feed of grass carp. Aquac. Rep. 2020, 18, 100434. [Google Scholar] [CrossRef]
- Huang, D.; Maulu, S.; Ren, M.; Liang, H.; Ge, X.; Ji, K.; Yu, H. Dietary lysine levels improved antioxidant capacity and immunity via the tor and p38 mapk signaling pathways in grass carp, ctenopharyngodon idellus fry. Front. Immunol. 2021, 12, 635015. [Google Scholar] [CrossRef] [PubMed]
- Yuan, X.C.; Liang, X.F.; Li, A.X.; Cai, W.J. The feedback regulation of carbohydrates intake on food intake and appetite in grass carp (Ctenopharyngodon idella). Fish Physiol. Biochem. 2021, 47, 1395–1403. [Google Scholar] [CrossRef]
- Yang, G.; Yu, R.; Geng, S.; Xiong, L.; Yan, Q.; Kumar, V.; Wen, C.; Peng, M. Apple polyphenols modulates the antioxidant defense response and attenuates inflammatory response concurrent with hepatoprotective effect on grass carp (Ctenopharyngodon idellus) fed low fish meal diet. Aquaculture 2021, 534, 736284. [Google Scholar] [CrossRef]
- Yang, G.; Qiu, H.; Yu, R.; Xiong, L.; Yan, Q.; Wen, C.; Peng, M. Dietary supplementation of β-glucan, inulin and emodin modulates antioxidant response and suppresses intestinal inflammation of grass carp (Ctenopharyngodon idellus). Anim. Feed Sci. Technol. 2021, 272, 114789. [Google Scholar] [CrossRef]
- Dasgupta, M. Adaptation of the alimentary tract to feeding habits in the weed eating fish (grass carp) Ctenopharyngodon idella (Val.). J. Crop Weed 2009, 5, 201–205. [Google Scholar]
- Hao, Y.T.; Wu, S.G.; Jakovlić, I.; Zou, H.; Li, W.X.; Wang, G.T. Impacts of diet on hindgut microbiota and short-chain fatty acids in grass carp (Ctenopharyngodon idellus). Aquac. Res. 2017, 48, 5595–5605. [Google Scholar] [CrossRef]
- Li, X.-Q.; Xu, H.-B.; Sun, W.-T.; Xu, X.-Y.; Xu, Z.; Leng, X.-J. Grass carp fed a fishmeal-free extruded diet showed higher weight gain and nutrient utilization than those fed a pelleted diet at various feeding rates. Aquaculture 2018, 493, 283–288. [Google Scholar] [CrossRef]
- Cai, W.; Liang, X.-F.; Yuan, X.; Liu, L.; He, S.; Li, J.; Li, B.; Xue, M. Different strategies of grass carp (Ctenopharyngodon idella) responding to insufficient or excessive dietary carbohydrate. Aquaculture 2018, 497, 292–298. [Google Scholar] [CrossRef]
- Tian, L.; Liu, Y.; Yang, H.; Liang, G.; Niu, J. Effects of different dietary wheat starch levels on growth, feed efficiency and digestibility in grass carp (Ctenopharyngodon idella). Aquac. Int. 2011, 20, 283–293. [Google Scholar] [CrossRef]
- Tian, L.; Liu, Y.; Hung, S.; Deng, D.-F.; Yang, H.; Niu, J.; Liang, G. Effect of feeding strategy and carbohydrate source on carbohydrate utilization by grass carp (Ctenopharyngodon idella). Am. J. Agric. Biol. Sci. 2010, 5, 135–142. [Google Scholar] [CrossRef][Green Version]
- Fang, L.; Guo, X.; Liang, X.-F. First feeding of grass carp (Ctenopharyngodon idellus) with a high-carbohydrate diet: The effect on glucose metabolism in juveniles. Aquac. Rep. 2021, 21, 100830. [Google Scholar] [CrossRef]
- Takeuchi, T.; Hernández, M.; Watanabe, T. Nutritive value of gelatinized corn meal as a carbohydrate source to grass carp and hybrid tilapia Oreochromis niloticus × O. Aureus. Fish. Sci. 1994, 60, 573–577. [Google Scholar] [CrossRef][Green Version]
- Lin, S.M.; Zhou, X.M.; Zhou, Y.L.; Kuang, W.M.; Chen, Y.J.; Luo, L.; Dai, F.Y. Intestinal morphology, immunity and microbiota response to dietary fibers in largemouth bass, Micropterus salmoide. Fish Shellfish Immunol. 2020, 103, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Jian, S.Q.; Cao, H.; Wen, C.; Hu, B.; Peng, M.; Peng, L.; Yuan, J.; Liang, L. Changes in microbiota along the intestine of grass carp (Ctenopharyngodon idella): Community, interspecific interactions, and functions. Aquaculture 2019, 498, 151–161. [Google Scholar] [CrossRef]
- He, Y.; Chi, S.Y.; Tan, B.; Zhang, H.; Dong, X.H.; Yang, Q.; Liu, H.Y.; Zhang, S. Effect of yeast culture on intestinal microbiota of Litopenaeus vannamei. J. Guangdong Ocean Univ. 2017, 37, 21–27. [Google Scholar] [CrossRef]
- Beam, A.; Clinger, E.; Hao, L. Effect of diet and dietary components on the composition of the gut microbiota. Nutrients 2021, 13, 2795. [Google Scholar] [CrossRef]
- Zhu, B.P.; Zhou, J.; Wang, Z.; Hu, Y.; Cai, M.; Yang, L.; Dai, J.; Hu, Y. Interactions between intestinal morphology, digestion, inflammatory responses, and gut microbiota of juvenile channel catfish elicited by dietary enzymatic rice protein. Fish Shellfish Immunol. 2022, 127, 155–165. [Google Scholar] [CrossRef]
- Wu, Y.; Li, R.; Shen, G.; Huang, F.; Yang, Q.; Tan, B.; Chi, S.Y. Effects of dietary small peptides on growth, antioxidant capacity, nonspecific immunity and ingut microflora structure of Litopenaeus vannamei. J. Guangdong Ocean Univ. 2021, 41, 1–9. [Google Scholar]
- Gong, B.; Bao, F.; Wang, Y.; Liu, H.; Xiao, M.; He, J. Metabonomics study on the effect of traditional chinese medicines feed addition on growth performance and serum metabolic profile of juvenile chinese softshell turtle (Pelodiscus sinensis wiegmann). Aquac. Rep. 2021, 20, 100632. [Google Scholar] [CrossRef]
- Shi, H.-T.; Zhao, S.-Z.; Wang, K.-L.; Fan, M.-X.; Han, Y.-Q.; Wang, H.-L. Effects of dietary astragalus membranaceus supplementation on growth performance, and intestinal morphology, microbiota and metabolism in common carp (Cyprinus carpio). Aquac. Rep. 2022, 22, 100955. [Google Scholar] [CrossRef]
- Bar, N.; Korem, T.; Weissbrod, O.; Zeevi, D.; Rothschild, D.; Leviatan, S.; Kosower, N.; Lotan-Pompan, M.; Weinberger, A.; Le Roy, C.I.; et al. A reference map of potential determinants for the human serum metabolome. Nature 2020, 588, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Bertocci, F.; Mannino, G. Can agri-food waste be a sustainable alternative in aquaculture? A bibliometric and meta-analytic study on growth performance, innate immune system, and antioxidant defenses. Foods 2022, 11, 1861. [Google Scholar] [CrossRef] [PubMed]
- Deng, J.; Lin, B.; Zhang, X.; Guo, L.; Chen, L.; Li, G.; Wang, Q.; Yu, C.; Mi, H. Effects of dietary sodium humate on growth, antioxidant capacity, non-specific immune response, and resistance to aeromonas hydrophila in genetic improvement of farmed tilapia (GIFT, Oreochromis niloticus). Aquaculture 2020, 520, 734788. [Google Scholar] [CrossRef]
- AOAC. Official Methods of Analysis of AOAC International, 18th ed.; Association of Official Analytical Chemists: Gaithersburg, MD, USA, 2005. [Google Scholar]
- Friedewald, W.T.; Levy, R.I.; Fredrickson, D.S. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin. Chem. 1972, 18, 499–502. [Google Scholar] [CrossRef]
- Zhang, W.; Tan, B.; Pang, A.; Deng, J.; Yang, Q.; Zhang, H. Screening of potential biomarkers for soybean meal induced enteritis in pearl gentian grouper (Epinephelus fuscoguttatus ♀ × Epinephelus lanceolatus ♂). J. Guangdong Ocean Univ. 2022, 42, 1–12. [Google Scholar] [CrossRef]
- Li, J.; Wang, C.; Wang, L.; Zhao, Z.; Luo, L.; Xu, Q. Effects of glutamate supplementation in low phosphorus diets on intestinal digestive enzyme activities and intestinal morphology of juvenile songpu mirror carp (Cyprinus carpio L.). J. Guangdong Ocean Univ. 2019, 39, 20–26. [Google Scholar] [CrossRef]
- Mardones, O.; Oyarzún-Salazar, R.; Labbé, B.S.; Miguez, J.M.; Vargas-Chacoff, L.; Muñoz, J.L.P. Intestinal variation of serotonin, melatonin, and digestive enzymes activities along food passage time through git in salmo salar fed with supplemented diets with tryptophan and melatonin. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2022, 266, 111159. [Google Scholar] [CrossRef]
- Li, J.; Wang, C.; Zhang, Y.; Wu, D.; Fan, Z.; Wang, L. Effect of arginine supplementation in high starch diets on intestinal digestive enzyme activities and intestinal morphology of songpu mirror carp (Cyprinus carpio L.). J. Guangdong Ocean Univ. 2020, 41, 39–46. [Google Scholar] [CrossRef]
- Wang, A.; Yang, Q.; Tan, B.; Xiao, W.; Jia, J.; Dong, X.H.; Chi, S.Y.; Liu, H.Y.; Zhang, S. Effects of enzymolytic soybean meal on growth performance, serum biochemical indices, non-specific immunity and disease resistance of juvenile Litopenaeus vannamei. J. Guangdong Ocean Univ. 2018, 38, 14–21. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, Y.; Fan, J.; Zhou, H.; Huang, H.; Cao, Y.; Jiang, W.; Zhang, W.; Deng, J.; Tan, B. Effects of different viscous guar gums on growth, apparent nutrient digestibility, intestinal development and morphology in juvenile largemouth bass, Micropterus salmoides. Front. Physiol. 2022, 13, 927819. [Google Scholar] [CrossRef]
- Gui, L.; Mai, H.; Chi, S.Y.; Zhou, W.; Li, Y.; Tan, B.; Dong, X.H.; Yang, Q.; Liu, H.Y.; Zhang, S. Effects of yeast culture on growth performance, hematological parameters, immunity and disease resistance of Litopenaeus vannamei. J. Guangdong Ocean Univ. 2019, 39, 30–37. [Google Scholar] [CrossRef]
- Wen, C.; Ma, S.; Tian, H.; Jiang, W.; Jia, X.; Zhang, W.; Jiang, G.; Li, X.; Chi, C.; He, C.; et al. Evaluation of the protein-sparing effects of carbohydrates in the diet of the crayfish, Procambarus clarkii. Aquaculture 2022, 556, 738275. [Google Scholar] [CrossRef]
- Hu, B.; Song, L.; Mao, S.; Xu, P. Effects of four chinese herbal preparations on growth performance and antioxidant activity in juvenile Micropterus salmoides. J. Guangdong Ocean Univ. 2019, 39, 101–107. [Google Scholar] [CrossRef]
- Kim, J.-H.; Kang, Y.J.; Kim, K.I.; Kim, S.K.; Kim, J.-H. Toxic effects of nitrogenous compounds (ammonia, nitrite, and nitrate) on acute toxicity and antioxidant responses of juvenile olive flounder, paralichthys olivaceus. Environ. Toxicol. Pharmacol. 2019, 67, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.-J.; Mou, M.-M.; Pu, D.-C.; Lin, S.-M.; Chen, Y.-J.; Luo, L. Effect of dietary starch level on growth, metabolism enzyme and oxidative status of juvenile largemouth bass, Micropterus salmoides. Aquaculture 2019, 498, 482–487. [Google Scholar] [CrossRef]
- Guo, H.; Tan, C.; You, L.; Shen, Y.; Lu, Z.; Zhu, C. Effects of nitrite stress on gene expression of antioxidant enzymes, heat shock protein and cathepsin b in hepatopancreas of Litopenaeus vannamei. J. Guangdong Ocean Univ. 2017, 37, 117–122. [Google Scholar] [CrossRef]
- Jia, Y.; Jing, Q.; Zhai, J.; Guan, C.; Huang, B. Alternations in oxidative stress, apoptosis, and innate-immune gene expression at mrna levels in subadult tiger puffer (Takifugu rubripes) under two different rearing systems. Fish Shellfish Immunol. 2019, 92, 756–764. [Google Scholar] [CrossRef]
- Luo, J.; Fu, W.; Yang, E.; Huang, J.; Xie, R.; Cheng, G. Effects of quercetin on growth performance, antioxidant capacity and intestinal microflora of hybrid grouper (Epinephelus fuscoguttatus ♀ × Epinephelus polyphekadion ♂). J. Guangdong Ocean Univ. 2022, 42, 13–22. [Google Scholar] [CrossRef]
- Li, Z.F.; Xie, J.; Yu, E.M.; Wang, G.J.; Zhang, X.K.; Yu, D.G.; Zhang, K.; Gong, W.B. Diet influences the accumulation of short-chain fatty acids associated with the gut microbiota in the grass carp (Ctenopharyngodon idellus) hindgut. Appl. Ecol. Environ. Res. 2019, 17, 13435–13453. [Google Scholar] [CrossRef]
- Li, S.; Heng, X.; Guo, L.; Lessing, D.J.; Chu, W. Scfas improve disease resistance via modulate gut microbiota, enhance immune response and increase antioxidative capacity in the host. Fish Shellfish Immunol. 2022, 120, 560–568. [Google Scholar] [CrossRef]
- Li, Y.; Lu, Z.; Zhan, F.; Yang, M.; Zhao, L.; Shi, F.; Li, J.; Lin, L.; Qin, Z. Nrf2 modulates host defense during antibacterial immunity response in grass carp (Ctenopharyngodon idellus). Aquaculture 2021, 536, 736474. [Google Scholar] [CrossRef]
- Pérez-Burillo, S.; Mehta, T.; Pastoriza, S.; Kramer, D.L.; Paliy, O.; Rufián-Henares, J.Á. Potential probiotic salami with dietary fiber modulates antioxidant capacity, short chain fatty acid production and gut microbiota community structure. LWT 2019, 105, 355–362. [Google Scholar] [CrossRef]
- Huang, D.; Liang, H.; Ren, M.; Ge, X.; Ji, K.; Yu, H.; Maulu, S. Effects of dietary lysine levels on growth performance, whole body composition and gene expression related to glycometabolism and lipid metabolism in grass carp, Ctenopharyngodon idellus fry. Aquaculture 2021, 530, 735806. [Google Scholar] [CrossRef]
- Biasato, I.; Chemello, G.; Caimi, C.; Bellezza Oddon, S.; Capucchio, M.T.; Colombino, E.; Schiavone, A.; Ceccotti, C.; Terova, G.; Gasco, L. Taurine supplementation in plant-based diets for juvenile rainbow trout (Oncorhynchus mykiss): Effects on growth performance, whole body composition, and histomorphological features. Anim. Feed Sci. Technol. 2022, 289, 115314. [Google Scholar] [CrossRef]
- Knutsen, H.R.; Ottesen, O.H.; Palihawadana, A.M.; Sandaa, W.; Sørensen, M.; Hagen, Ø. Muscle growth and changes in chemical composition of spotted wolffish juveniles (anarhichas minor) fed diets with and without microalgae (Scenedesmus obliquus). Aquac. Rep. 2019, 13, 100175. [Google Scholar] [CrossRef]
- Oliveira, T.S.; Khan, K.U.; Boaratti, A.Z.; Rodrigues, A.T.; Reis, M.P.; Sakomura, N.K.; Fernandes, J.B.K. Evaluation of the optimum dietary essential amino acid pattern for adult pacu (Piaractus mesopotamicus). Aquaculture 2021, 540, 736686. [Google Scholar] [CrossRef]
- Yang, X.; Zhi, X.; Song, Z.; Wang, G.; Zhao, X.; Chi, S.; Tan, B. Flesh quality of hybrid grouper (Epinephelus fuscoguttatus ♀ × Epinephelus lanceolatus ♂) fed with hydrolyzed porcine mucosa-supplemented low fishmeal diet. Anim. Nutr. 2022, 8, 114–124. [Google Scholar] [CrossRef]
- Cheng, Y.; Zhao, J.L.; Song, L.Y.; Wu, H.Y.; Zhou, H.T. Effects of salinity and alkalinity on growth performance and muscle quality of nile tilapia Oreochromis niloticus. Fish Sci. 2020, 39, 341–350. [Google Scholar] [CrossRef]
- Yang, G.; Tao, Z.; Xiao, J.; Tu, G.; Kumar, V.; Wen, C. Characterization of the gastrointestinal microbiota in paddlefish (Polyodon spathula). Aquac. Rep. 2020, 17, 100402. [Google Scholar] [CrossRef]
- Chen, K.; Huang, W.; Liao, Y.; Yang, C.; Wang, Q. Community structure of culturable bacterial communities in the intestine of Pinctada fucata martensii and its aquaculture water. J. Guangdong Ocean Univ. 2021, 41, 47–54. [Google Scholar] [CrossRef]
- Huang, B.; Zhang, S.; Dong, X.; Chi, S.; Yang, Q.; Liu, H.; Tan, B.; Xie, S. Effects of fishmeal replacement by black soldier fly on growth performance, digestive enzyme activity, intestine morphology, intestinal flora and immune response of pearl gentian grouper (Epinephelus fuscoguttatus ♀ × Epinephelus lanceolatus ♂). Fish Shellfish Immunol. 2022, 120, 497–506. [Google Scholar] [CrossRef] [PubMed]
- Nayak, S.K. Role of gastrointestinal microbiota in fish. Aquac. Res. 2010, 41, 1553–1573. [Google Scholar] [CrossRef]
- Peng, M.; Xue, J.; Hu, Y.; Wen, C.; Hu, B.; Jian, S.; Liang, L.; Yang, G. Disturbance in the homeostasis of intestinal microbiota by a high-fat diet in the rice field eel (Monopterus albus). Aquaculture 2019, 502, 347–355. [Google Scholar] [CrossRef]
- Zhou, J.S.; Chen, H.J.; Shi, X.-c.; Li, X.X.; Chen, L.Q.; Du, Z.-Y.; Yu, H. Effect of dietary bile acids on growth, body composition, lipid metabolism and microbiota in grass carp (Ctenopharyngodon idella). Aquac. Nutr. 2017, 24, 802–813. [Google Scholar] [CrossRef]
- Meng, X.-L.; Cao, H.; Li, H.; Li, K.-K.; Yang, G.-K.; Zhang, Y.-M.; Chang, X.-L.; Zhang, X.-D.; Zhang, J.-X. Effect of dietary honeysuckle (Lonicera caerulea L.) supplementation on lipid metabolism, immunity and intestinal microbiota in grass carp (Ctenopharyngodon idellus). Aquac. Rep. 2022, 23, 101063. [Google Scholar] [CrossRef]
- Liu, S.; Yu, H.; Li, P.; Wang, C.; Liu, G.; Zhang, X.; Zhang, C.; Qi, M.; Ji, H. Dietary nano-selenium alleviated intestinal damage of juvenile grass carp (Ctenopharyngodon idella) induced by high-fat diet: Insight from intestinal morphology, tight junction, inflammation, anti-oxidization and intestinal microbiota. Anim. Nutr. 2022, 8, 235–248. [Google Scholar] [CrossRef]
- Ghanbari, M.; Kneifel, W.; Domig, K.J. A new view of the fish gut microbiome: Advances from next-generation sequencing. Aquaculture 2015, 448, 464–475. [Google Scholar] [CrossRef]
- Degnan, P.H.; Taga, M.E.; Goodman, A.L. Vitamin b12 as a modulator of gut microbial ecology. Cell Metab. 2014, 20, 769–778. [Google Scholar] [CrossRef]
- Tsuchiya, C.; Sakata, T.; Sugita, H. Novel ecological niche of cetobacterium somerae, an anaerobic bacterium in the intestinal tracts of freshwater fish. Lett. Appl. Microbiol. 2008, 46, 43–48. [Google Scholar] [CrossRef]
- Chang, X.; Shen, Y.; Yun, L.; Wang, X.; Feng, J.; Yang, G.; Meng, X.; Zhang, J.; Su, X. The antipsychotic drug olanzapine altered lipid metabolism in the common carp (Cyprinus carpio L.): Insight from the gut microbiota-scfas-liver axis. Sci. Total Envir. 2022, 856, 159054. [Google Scholar] [CrossRef]
- Ottaviani, D.; Parlani, C.; Citterio, B.; Masini, L.; Leoni, F.; Canonico, C.; Sabatini, L.; Bruscolini, F.; Pianetti, A. Putative virulence properties of aeromonas strains isolated from food, environmental and clinical sources in italy: A comparative study. Int. J. Food Microbiol. 2011, 144, 538–545. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Pan, J.; Chen, L.; Li, R.; Han, Y.; Di, Z.; Ling, B.; Ahmad, A.; Yang, N.; Fan, L.; et al. Prevalence, virulence-related genes and antimicrobial resistance of Aeromonas spp. From loach misgurnus anguillicaudatus with skin ulcer and healthy controls in southern china. Aquaculture 2022, 552, 738040. [Google Scholar] [CrossRef]
- Torregrosa-Crespo, J.; Martínez-Espinosa, R.M.; Esclapez, J.; Bautista, V.; Pire, C.; Camacho, M.; Richardson, D.J.; Bonete, M.J. Anaerobic Metabolism in Haloferax Genus: Denitrification as Case of Study; Academic Press: Cambridge, MA, USA, 2016; pp. 41–85. [Google Scholar]
- Zumla, A. Mandell, douglas, and bennett’s principles and practice of infectious diseases. Lancet Infect. Dis. 2010, 10, 303–304. [Google Scholar] [CrossRef]
- Liu, Y.; Zhou, H.; Fan, J.; Huang, H.; Deng, J.; Tan, B. Assessing effects of guar gum viscosity on the growth, intestinal flora, and intestinal health of Micropterus salmoides. In. J. Biol. Macrol. 2022, 222, 1037–1047. [Google Scholar] [CrossRef] [PubMed]
- Garrett, W.S.; Onderdonk, A.B. Bacteroides, Prevotella, Porphyromonas, and Fusobacterium Species (and Other Medically Important Anaerobic Gram-Negative Bacilli); W.B. Saunders: Philadelphia, PA, USA, 2015; pp. 2773–2780. [Google Scholar]
- Xu, W.; Kenéz, Á.; Mann, S.; Overton, T.R.; Wakshlag, J.J.; Nydam, D.V.; Feng, T.; Yepes, F.L. Effects of dietary branched-chain amino acid supplementation on serum and milk metabolome profiles in dairy cows during early lactation. J. Dairy Sci. 2022, 105, 8497–8508. [Google Scholar] [CrossRef]
- Ibba, M.; Soll, D. Aminoacyl-trna synthesis. Annu. Rev. Biochem. 2000, 69, 617–650. [Google Scholar] [CrossRef]
- Tan, B.; Yin, Y.; Liu, Z.; Li, X.; Xu, H.; Kong, X.; Huang, R.; Tang, W.; Shinzato, I.; Smith, S.B.; et al. Dietary l-arginine supplementation increases muscle gain and reduces body fat mass in growing-finishing pigs. Amino Acids 2009, 37, 169–175. [Google Scholar] [CrossRef]
- Hoseini, S.M.; Ahmad Khan, M.; Yousefi, M.; Costas, B. Roles of arginine in fish nutrition and health: Insights for future researches. Rev. Aquac. 2020, 12, 2091–2108. [Google Scholar] [CrossRef]
- Wang, Q.; Xu, Z.; Ai, Q. Arginine metabolism and its functions in growth, nutrient utilization, and immunonutrition of fish. Anim. Nutr. 2021, 7, 716–727. [Google Scholar] [CrossRef]
- Wu, G.; Meininger, C.J. Nitric oxide and vascular insulin resistance. Biofactors 2009, 35, 21–27. [Google Scholar] [CrossRef]
- Li, P.; Wu, G. Roles of dietary glycine, proline, and hydroxyproline in collagen synthesis and animal growth. Amino Acids 2018, 50, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Mai, K.; Trushenski, J.; Wu, G. New developments in fish amino acid nutrition: Towards functional and environmentally oriented aquafeeds. Amino Acids 2009, 37, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zheng, S.; Ma, X.; Cheng, K.; Wu, G. Effects of dietary starch and lipid levels on the protein retention and growth of largemouth bass (Micropterus salmoides). Amino Acids 2020, 52, 999–1016. [Google Scholar] [CrossRef] [PubMed]
- Jia, S.; Li, X.; Zheng, S.; Wu, G. Amino acids are major energy substrates for tissues of hybrid striped bass and zebrafish. Amino Acids 2017, 49, 2053–2063. [Google Scholar] [CrossRef] [PubMed]
- Parthasarathy, A.; Cross, P.J.; Dobson, R.C.J.; Adams, L.E.; Savka, M.A.; Hudson, A.O. A three-ring circus: Metabolism of the three proteogenic aromatic amino acids and their role in the health of plants and animals. Front. Mol. Biosci. 2018, 5, 29. [Google Scholar] [CrossRef]
- Hopkinson, G. A neurochemical theory of appetite and weight changes in depressive states. Acta Psychiatr. Scand. 1981, 64, 217–225. [Google Scholar] [CrossRef]
| Group | FM | INSP | SNSP | NSP |
|---|---|---|---|---|
| Ingredients% | ||||
| Fish meal 1 | 33.00 | 33.00 | 33.00 | 33.00 |
| Wheat meal 1 | 15.00 | 15.00 | 15.00 | 15.00 |
| Wheat bran 1 | 25.00 | 25.00 | 25.00 | 25.00 |
| α-Starch | 22.38 | 17.98 | 13.14 | 8.74 |
| INSP | 0.00 | 4.40 | 0.00 | 4.40 |
| SNSP 2 | 0.00 | 0.00 | 9.24 | 9.24 |
| Soy oil 1 | 1.00 | 1.00 | 1.00 | 1.00 |
| Soy lectin 1 | 0.50 | 0.50 | 0.50 | 0.50 |
| Ca(H2PO4)2 | 1.20 | 1.20 | 1.20 | 1.20 |
| NaCl | 0.20 | 0.20 | 0.20 | 0.20 |
| Choline chloride 1 | 0.30 | 0.30 | 0.30 | 0.30 |
| Vitamin C 3 | 0.02 | 0.02 | 0.02 | 0.02 |
| Mineral premix | 1.00 | 1.00 | 1.00 | 1.00 |
| Vitamin premix | 0.40 | 0.40 | 0.40 | 0.40 |
| Nutrient level% | ||||
| Dry matter | 94.19 | 94.22 | 94.02 | 94.26 |
| Crude protein | 29.66 | 30.02 | 29.76 | 30.53 |
| Crude lipid | 4.76 | 4.81 | 4.97 | 4.78 |
| Ash | 9.90 | 9.76 | 9.68 | 9.52 |
| Gross energy (MJ/kg) | 18.59 | 18.47 | 19.16 | 19.16 |
| Group | FM | INSP | SNSP | NSP |
|---|---|---|---|---|
| IBW (g) | 0.79 ± 0.01 | 0.80 ± 0.01 | 0.79 ± 0.01 | 0.80 ± 0.01 |
| FBW (g) | 24.56 ± 0.46 ab | 27.79 ± 0.77 b | 23.59 ± 1.37 a | 28.01 ± 0.35 b |
| SR (%) | 99.19 ± 0.83 | 99.19 ± 0.81 | 99.15 ± 0.85 | 100.0 ± 0.01 |
| FI (g/kg MBW/day) | 12.05 ± 0.22 a | 12.84 ± 0.28 ab | 13.66 ± 0.24 b | 13.34 ± 0.27 b |
| WGR (%) | 29.96 ± 0.56 ab | 33.92 ± 1.00 b | 28.76 ± 1.73 a | 34.23 ± 0.44 b |
| DGC (%/day) | 3.30 ± 0.03 ab | 3.50 ± 0.05 b | 3.24 ± 0.09 a | 3.52 ± 0.02 b |
| FCR | 1.01 ± 0.01 a | 1.04 ± 0.03 ab | 1.17 ± 0.04 b | 1.08 ± 0.03 ab |
| PER | 3.32 ± 0.05 b | 3.21 ± 0.09 ab | 2.88 ± 0.10 a | 3.04 ± 0.07 ab |
| Group | FM | INSP | SNSP | NSP |
|---|---|---|---|---|
| Triglyceride (mmol/L) | 2.94 ± 0.13 | 3.11 ± 0.04 | 2.87 ± 0.08 | 2.86 ± 0.07 |
| Total cholesterol (mmol/L) | 6.16 ± 0.18 ab | 6.48 ± 0.17 b | 6.05 ± 0.17 ab | 5.68 ± 0.09 a |
| HDL-C (mmol/L) | 4.47 ± 0.19 b | 4.10 ± 0.12 ab | 3.83 ± 0.19 ab | 3.60 ± 0.20 a |
| LDL-C (mmol/L) | 1.10 ± 0.06 a | 1.75 ± 0.07 b | 1.65 ± 0.09 b | 1.50 ± 0.12 b |
| LDL-C/HDL-C | 0.25 ± 0.02 a | 0.43 ± 0.02 b | 0.44 ± 0.04 b | 0.43 ± 0.06 b |
| Free cholesterol (mmol/L) | 0.66 ± 0.13 a | 1.36 ± 0.23 b | 1.33 ± 0.18 b | 0.79 ± 0.08 ab |
| Cholesterol ester (mmol/L) | 5.49 ± 0.28 | 5.12 ± 0.31 | 4.71 ± 0.26 | 4.89 ± 0.09 |
| Free/total cholesterol | 0.11 ± 0.02 a | 0.21 ± 0.04 b | 0.22 ± 0.04 b | 0.14 ± 0.01 ab |
| Group | FM | INSP | SNSP | NSP |
|---|---|---|---|---|
| Protein (g/L) | 28.12 ± 0.95 bc | 25.82 ± 0.46 ab | 23.42 ± 0.63 a | 30.43 ± 0.99 c |
| TAA (mol/L) | 0.11 ± 0.01 b | 0.07 ± 0.01 a | 0.12 ± 0.01 b | 0.08 ± 0.01 a |
| BUN (mmol/L) | 6.92 ± 0.17 a | 6.24 ± 0.22 a | 8.95 ± 0.44 b | 6.98 ± 0.35 a |
| AST (U/L) | 12.87 ± 0.47 b | 3.92 ± 0.22 a | 16.99 ± 1.20 c | 11.65 ± 0.22 b |
| ALT (U/L) | 4.86 ± 0.06 ab | 4.75 ± 0.03 a | 5.00 ± 0.07 ab | 5.05 ± 0.07 b |
| GGT (U/L) | 0.62 ± 0.15 | 0.62 ± 0.15 | 1.09 ± 0.16 | 0.62 ± 0.15 |
| Group | FM | INSP | SNSP | NSP |
|---|---|---|---|---|
| SOD (U/mL) | 44.58 ± 2.31 | 40.70 ± 2.49 | 40.24 ± 1.95 | 45.23 ± 1.39 |
| CAT (U/mL) | 6.35 ± 0.46 a | 5.90 ± 0.24 a | 7.19 ± 0.31 ab | 8.73 ± 0.81 b |
| POD (U/mL) | 10.81 ± 0.82 a | 22.96 ± 1.55 b | 18.96 ± 1.07 b | 24.00 ± 1.36 b |
| TAC (U/mL) | 2.77 ± 0.17 a | 3.48 ± 0.16 ab | 3.50 ± 0.16 ab | 3.74 ± 0.20 b |
| Group | Initial Fish | FM | INSP | SNSP | NSP |
|---|---|---|---|---|---|
| Crude protein | 11.60 | 15.54 ± 0.12 ab | 15.52 ± 0.16 ab | 14.72 ± 0.12 a | 15.69 ± 0.34 b |
| Crude lipid | 8.27 | 8.57 ± 0.10 ab | 8.54 ± 0.23 ab | 8.35 ± 0.09 a | 9.32 ± 0.25 b |
| Ash | 1.79 | 2.90 ± 0.02 bc | 2.77 ± 0.02 b | 2.99 ± 0.07 c | 2.56 ± 0.04 a |
| Group | Initial Fish | FM | INSP | SNSP | NSP |
|---|---|---|---|---|---|
| Arg | 3.42 | 3.59 ± 0.03 | 3.74 ± 0.04 | 3.64 ± 0.04 | 3.73 ± 0.05 |
| His | 1.06 | 1.15 ± 0.01 | 1.16 ± 0.04 | 1.25 ± 0.02 | 1.17 ± 0.01 |
| Ile | 2.03 | 1.88 ± 0.02 a | 2.01 ± 0.02 c | 1.89 ± 0.03 ab | 1.97 ± 0.01 bc |
| Leu | 3.41 | 3.29 ± 0.04 a | 3.52 ± 0.02 c | 3.32 ± 0.04 ab | 3.45 ± 0.02 bc |
| Lys | 3.89 | 3.85 ± 0.02 a | 4.12 ± 0.03 c | 3.92 ± 0.05 ab | 4.09 ± 0.05 bc |
| Met | 1.21 | 1.14 ± 0.01 a | 1.21 ± 0.02 b | 1.16 ± 0.02 ab | 1.19 ± 0.01 ab |
| Phe | 1.56 | 1.55 ± 0.03 a | 1.64 ± 0.02 b | 1.56 ± 0.02 ab | 1.60 ± 0.01 ab |
| Thr | 1.92 | 1.86 ± 0.02 a | 1.96 ± 0.03 ab | 1.91 ± 0.02 ab | 1.99 ± 0.04 b |
| Val | 2.20 | 2.09 ± 0.02 a | 2.22 ± 0.03 b | 2.11 ± 0.03 a | 2.19 ± 0.01 ab |
| ∑EAA | 20.70 | 20.41 ± 0.17 a | 21.59 ± 0.16 c | 20.75 ± 0.22 ab | 21.39 ± 0.14 bc |
| Glu | 6.46 | 7.03 ± 0.09 a | 7.23 ± 0.09 ab | 7.15 ± 0.15 ab | 7.57 ± 0.07 b |
| Gly | 1.72 | 1.85 ± 0.01 | 1.93 ± 0.07 | 1.87 ± 0.04 | 1.95 ± 0.02 |
| Ala | 3.17 | 3.24 ± 0.01 | 3.36 ± 0.05 | 3.28 ± 0.06 | 3.43 ± 0.03 |
| Tyr | 1.43 | 1.18 ± 0.01 | 1.28 ± 0.01 | 1.25 ± 0.06 | 1.24 ± 0.01 |
| Asp | 4.63 | 5.20 ± 0.07 a | 5.32 ± 0.05 b | 5.27 ± 0.10 a | 5.63 ± 0.06 b |
| Ser | 1.48 | 1.50 ± 0.03 | 1.55 ± 0.02 | 1.56 ± 0.02 | 1.60 ± 0.03 |
| Pro | 2.37 | 2.39 ± 0.03 | 2.51 ± 0.05 | 2.42 ± 0.03 | 2.48 ± 0.02 |
| Cys | 0.34 | 0.37 ± 0.01 | 0.37 ± 0.02 | 0.36 ± 0.01 | 0.35 ± 0.01 |
| ∑NEAA | 21.60 | 22.78 ± 0.24 a | 23.55 ± 0.21 ab | 23.17 ± 0.44 ab | 24.26 ± 0.21 b |
| ∑TAA | 42.30 | 42.89 ± 0.40 a | 44.83 ± 0.37 ab | 43.62 ± 0.66 ab | 45.35 ± 0.35 b |
| Group | FM | INSP | SNSP | NSP |
|---|---|---|---|---|
| Shannon | 4.43 ± 0.93 | 4.43 ± 1.05 | 5.09 ± 1.46 | 4.05 ± 0.41 |
| Simpson | 0.79 ± 0.07 | 0.79 ± 0.09 | 0.75 ± 0.18 | 0.81 ± 0.03 |
| Chao1 | 1414.40 ± 276.79 | 1234.13 ± 278.92 | 1719.01 ± 514.53 | 1001.48 ± 205.58 |
| ACE | 1486.38 ± 280.07 | 1269.27 ± 279.67 | 1784.94 ± 532.06 | 1041.58 ± 205.97 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Y.; Fu, X.; Zhou, H.; Fan, J.; Huang, H.; Deng, J.; Tan, B. Different Types of Non-Starch Polysaccharides Alter the Growth, Intestinal Flora and Serum Metabolite Profile of Grass Carp, Ctenopharyngodon idella. Metabolites 2022, 12, 1003. https://doi.org/10.3390/metabo12101003
Liu Y, Fu X, Zhou H, Fan J, Huang H, Deng J, Tan B. Different Types of Non-Starch Polysaccharides Alter the Growth, Intestinal Flora and Serum Metabolite Profile of Grass Carp, Ctenopharyngodon idella. Metabolites. 2022; 12(10):1003. https://doi.org/10.3390/metabo12101003
Chicago/Turabian StyleLiu, Yu, Xinlangji Fu, Hang Zhou, Jiongting Fan, Huajing Huang, Junming Deng, and Beiping Tan. 2022. "Different Types of Non-Starch Polysaccharides Alter the Growth, Intestinal Flora and Serum Metabolite Profile of Grass Carp, Ctenopharyngodon idella" Metabolites 12, no. 10: 1003. https://doi.org/10.3390/metabo12101003
APA StyleLiu, Y., Fu, X., Zhou, H., Fan, J., Huang, H., Deng, J., & Tan, B. (2022). Different Types of Non-Starch Polysaccharides Alter the Growth, Intestinal Flora and Serum Metabolite Profile of Grass Carp, Ctenopharyngodon idella. Metabolites, 12(10), 1003. https://doi.org/10.3390/metabo12101003