Pesticides and Male Fertility: A Dangerous Crosstalk
Abstract
1. Introduction
2. Routes of Exposure to Pesticides
3. Classes of Pesticides—A Brief Description
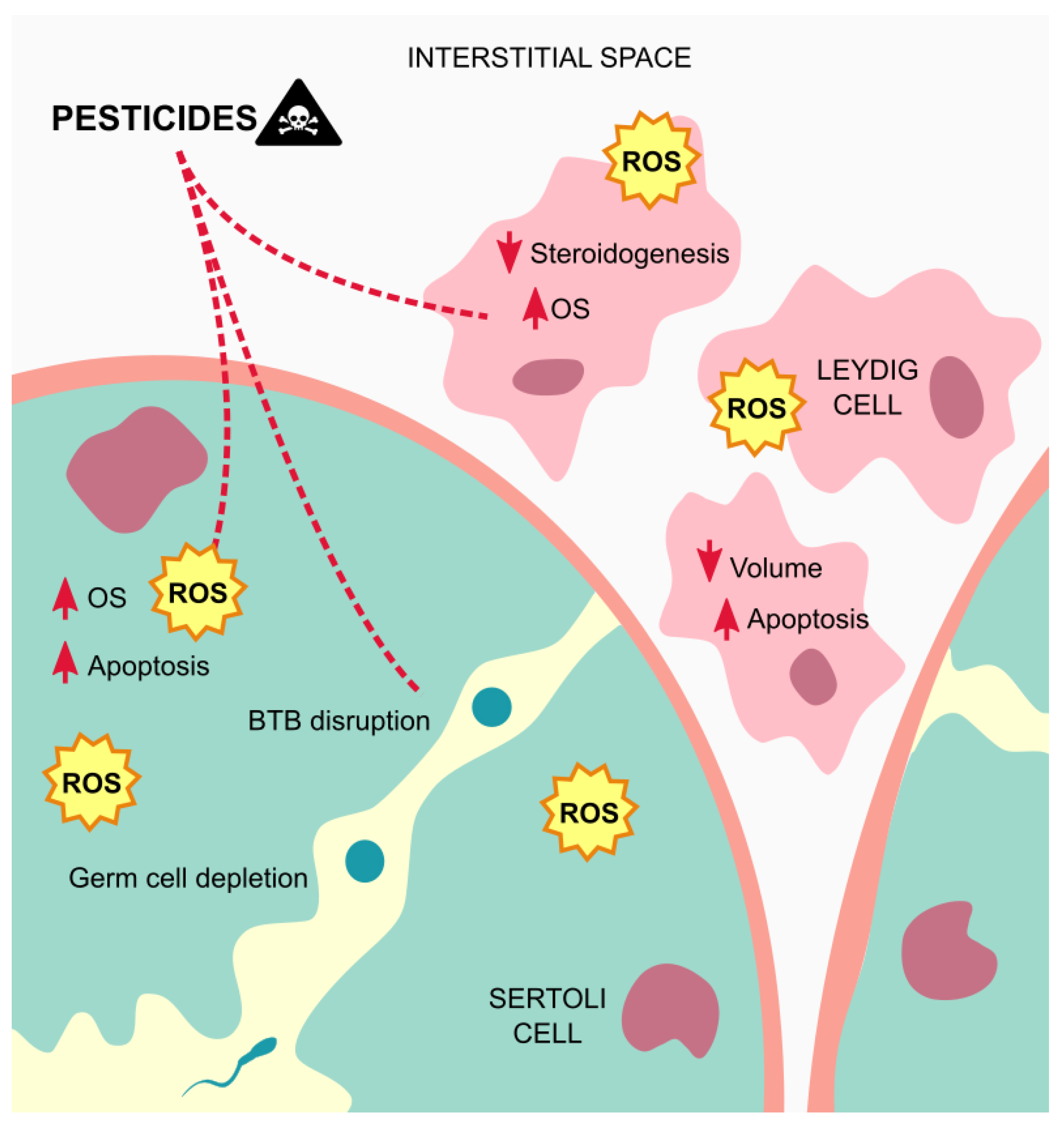
4. Effects and Mechanism of Action of Pesticides on Testicular Somatic Cells
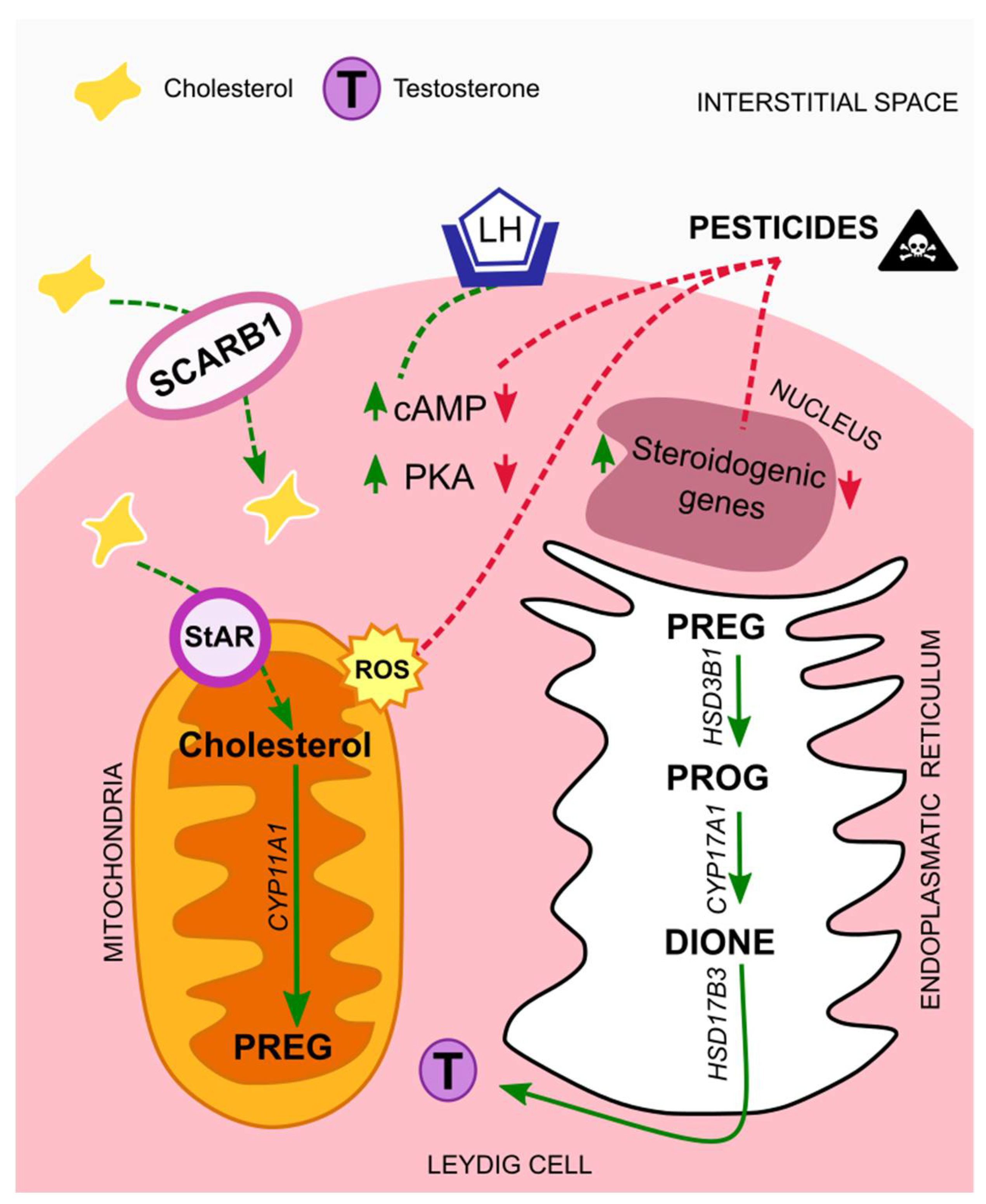
4.1. Leydig Cells
4.2. Peritubular Myoid Cells
4.3. Sertoli Cells
5. Effects and Mechanisms of Action of Pesticides on Testicular Tissue
6. Effects and Mechanisms of Action of Pesticides on Testicular Metabolism
7. How to Detect Pesticides and Their Impact on Cellular Metabolism for Male Fertility Assessment?
8. Materials and Methods
9. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Clementi, M.; Tiboni, G.M.; Causin, R.; La Rocca, C.; Maranghi, F.; Raffagnato, F.; Tenconi, R. Pesticides and fertility: An epidemiological study in northeast Italy and review of the literature. Reprod. Toxicol. 2008, 26, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Zegers-Hochschild, F.; Adamson, G.D.; de Mouzon, I.; Ishihara, O.; Mansour, R.; Nygren, K.; Sullivan, E.; Vanderpoel, S. International committee for monitoring assisted reproductive technology (ICMART) and the world health organization (WHO) revised glossary of ART terminology, 2009. Fertil. Steril. 2009, 92, 1520–1524. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Zhang, P.; Zhao, Y.; Zhang, H. Low dose carbendazim disrupts mouse spermatogenesis might be through estrogen receptor related histone and DNA methylation. Ecotoxicol. Environ. Saf. 2019, 176, 242–249. [Google Scholar] [CrossRef]
- Leaver, R.B. Male infertility: An overview of causes and treatment options. Br. J. Nurs. 2016, 25, S35–S40. [Google Scholar] [CrossRef]
- Babakhanzadeh, E.; Nazari, M.; Ghasemifar, S.; Khodadadian, A. Some of the factors involved in male infertility: A prospective review. Int. J. Gen. Med. 2020, 13, 29–41. [Google Scholar] [CrossRef]
- Murshidi, M.M.; Choy, J.T.; Eisenberg, M.L. Male Infertility and Somatic Health. Urol. Clin. N. Am. 2020, 47, 211–217. [Google Scholar] [CrossRef]
- Agarwal, A.; Baskaran, S.; Parekh, N.; Cho, C.L.; Henkel, R.; Vij, S.; Arafa, M.; Panner Selvam, M.K.; Shah, R. Male infertility. Lancet 2021, 397, 319–333. [Google Scholar] [CrossRef]
- Pesticidas. Available online: http://repositorio.insa.pt/bitstream/10400.18/772/1/Pesticidas%202011.pdf/ (accessed on 20 March 2020).
- Sharma, A.; Kumar, V.; Shahzad, B.; Tanveer, M.; Sidhu, G.P.S.; Handa, N.; Kohli, S.K.; Yadav, P.; Bali, A.S.; Parihar, R.D.; et al. Worldwide pesticide usage and its impacts on ecosystem. SN Appl. Sci. 2019, 1, 1446. [Google Scholar] [CrossRef]
- Mehrpour, O.; Karrari, P.; Zamani, N.; Tsatsakis, A.M.; Abdollahi, M. Occupational exposure to pesticides and consequences on male semen and fertility: A review. Toxicol. Lett. 2014, 230, 146–156. [Google Scholar] [CrossRef]
- Mostafalou, S.; Abdollahi, M. Pesticides and human chronic diseases: Evidences, mechanisms, and perspectives. Toxicol. Appl. Pharm. 2013, 268, 157–177. [Google Scholar] [CrossRef]
- Zoeller, R.T.; Brown, T.R.; Doan, L.L.; Gore, A.C.; Skakkebaek, N.E.; Soto, A.M.; Woodruff, T.J.; Vom Saal, F.S. Endocrine-disrupting chemicals and public health protection: A statement of principles from The Endocrine Society. Endocrinology 2012, 153, 4097–4110. [Google Scholar] [CrossRef] [PubMed]
- Heindel, J.J. History of the obesogen field: Looking back to look forward. Front. Endocrinol. 2019, 10, 14. [Google Scholar] [CrossRef]
- Cardoso, A.M.; Alves, M.G.; Mathur, P.P.; Oliveira, P.F.; Cavaco, J.E.; Rato, L. Obesogens and male fertility. Obes. Rev. 2016, 18, 109–125. [Google Scholar] [CrossRef] [PubMed]
- Ren, X.M.; Kuo, Y.; Blumberg, B. Agrochemicals and obesity. Mol. Cell Endocrinol. 2020, 515, 110926. [Google Scholar] [CrossRef]
- Oliveira, P.F.; Sousa, M.; Silva, B.M.; Monteiro, M.P.; Alves, M.G. Obesity, energy balance and spermatogenesis. Reproduction 2017, 153, 173–185. [Google Scholar] [CrossRef] [PubMed]
- Skakkebaek, N.E.; Rajpert-De Meyts, E.; Main, K.M. Testicular dysgenesis syndrome: An increasingly common developmental disorder with environmental aspects. Hum. Reprod. 2001, 16, 972–978. [Google Scholar] [CrossRef]
- Durairajanayagam, D. Lifestyle causes of male infertility. Arab. J. Urol. 2018, 16, 10–20. [Google Scholar] [CrossRef]
- Comninos, A.N.; Jayasena, C.N.; Dhillo, W.S. The relationship between gut and adipose hormones, and reproduction. Hum. Reprod. Update 2014, 20, 153–174. [Google Scholar] [CrossRef]
- Mostafalou, S.; Abdollahi, M. Pesticides: An update of human exposure and toxicity. Arch. Toxicol. 2017, 91, 549–599. [Google Scholar] [CrossRef]
- Nassan, F.L.; Chavarro, J.E.; Tanrikut, C. Diet and men´s fertility: Does diet affect sperm quality? Fertil. Steril. 2018, 110, 570–577. [Google Scholar] [CrossRef]
- Marques-Pinto, A.; Carvalho, D. Human infertility: Are endocrine disruptors to blame? Endocr. Connect. 2013, 2, R15–R29. [Google Scholar] [CrossRef] [PubMed]
- Martin-Reina, J.; Duarte, J.A.; Cerrillos, L.; Bautista, J.D.; Moreno, I. Insecticide reproductive toxicity profile: Organophosphate, carbamate and pyrethroids. J. Toxins 2017, 4, 1–7. [Google Scholar]
- Morais, S.; Dias, E.; Pereira, M.L. Carbamates: Human exposure and health effects. In The Impact of Pesticides, 1st ed.; Jokanovic, M., Ed.; AcademyPublish.org.: London, UK, 2012; pp. 21–38. [Google Scholar]
- Leemans, M.; Couderg, S.; Demeneix, B.; Fini, J.B. Pesticides with potential thrydoid hormone-disrupting effects: A review of recent data. Front. Endocrinol. 2019, 10, 743. [Google Scholar] [CrossRef]
- Zamkowska, D.; Karwacka, A.; Jurewicz, J.; Radwan, M. Environmental exposure to non-persistent endocrine disrupting chemicals and semen quality: An overview of the current epidemiological evidence. Int. J. Occup. Med. Environ. Health 2018, 31, 377–414. [Google Scholar] [CrossRef]
- Gupta, R.C.; Mukherjee, I.R.M.; Doss, R.B.; Malik, J.K.; Milatovic, D. Chapter 35—Organophosphates and Carbamates. In Reproductive and Developmental Toxicology, 2nd ed.; Gupta, R.C., Ed.; Academic Press: Amsterdam, The Netherlands, 2017; pp. 609–631. [Google Scholar]
- Simon-Delso, N.; Amaral-Rogers, V.; Belzunces, L.P.; Bonmatin, J.M.; Chagnon, M.; Downs, C.; Furlan, L.; Gibbons, D.W.; Giorio, C.; Girolami, V.; et al. Systemic insecticides (neonicotinoids and fipronil): Trends, uses, mode of action and metabolites. Environ. Sci. Pollut. Res. 2015, 22, 5–34. [Google Scholar] [CrossRef] [PubMed]
- Zhou, R.; Wu, J.; Liu, B.; Jiang, Y.; Chen, W.; Li, J.; He, Q.; He, Z. The roles and mechanisms of Leydig cells and myoid cells in regulating spermatogenesis. Cell Mol. Life Sci. 2019, 76, 2681–2695. [Google Scholar] [CrossRef]
- Yawer, A.; Sychrová, E.; Labohá, P.; Raška, J.; Jambor, T.; Babica, P.; Sovadinová, I. Endocrine-disrupting chemicals rapidly affect intercellular signaling in Leydig cells. Toxicol. Appl. Pharm. 2020, 404, 115177. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chen, F.; Ye, L.; Zirkin, B.; Chen, H. Steroidogenesis in Leydig cells: Effects of aging and environmental factors. Reproduction 2017, 154, R111–R122. [Google Scholar] [CrossRef]
- Rato, L.; Alves, M.G.; Socorro, S.; Duarte, A.I.; Cavaco, J.E.; Oliveira, P.F. Metabolic regulation is important for spermatogenesis. Nat. Rev. Urol. 2012, 9, 330–338. [Google Scholar] [CrossRef]
- Van Cauwenbergh, O.; Di Serafino, A.; Tytgat, J.; Soubry, A. Transgenerational epigenetic effects from male exposure to endocrine-disrupting compounds: A systematic review on research in mammals. Clin. Epigenetics 2020, 12, 65. [Google Scholar] [CrossRef]
- Roy, P.; Phukan, P.K.; Changmai, D.; Boruah, S. Pesticides, insecticides and male infertility. Int. J. Reprod. Contracept. Obs. Gynecol. 2017, 6, 3387–3391. [Google Scholar] [CrossRef]
- Darbre, P.D. Endocrine disruptors and obesity. Curr. Obes. Rep. 2017, 6, 18–27. [Google Scholar] [CrossRef]
- Zirkin, B.R.; Papadopoulos, V. Leydig cells: Formation, function, and regulation. Biol. Reprod. 2018, 99, 101–111. [Google Scholar] [CrossRef]
- Landschulz, K.T.; Pathak, R.K.; Rigotti, A.; Krieger, M.; Hobbs, H.H. Regulation of scavenger receptor, class B, type I, a high density lipoprotein receptor, in liver and steroidogenic tissues of the rat. J. Clin. Investig. 1996, 98, 984–995. [Google Scholar] [CrossRef]
- Stocco, D.M. Clinical disorders associated with abnormal cholesterol transport: Mutations in the steroidogenic acute regulatory protein. Mol. Cell Endocrinol. 2002, 191, 19–25. [Google Scholar] [CrossRef]
- Dong, Y.; Wang, Y.; Zhu, Q.; Li, X.; Huang, T.; Li, H.; Zhao, J.; Ge, R.S. Dimethoate blocks pubertal differentiation of Leydig cells in rats. Chemosphere 2020, 241, 125036. [Google Scholar] [CrossRef]
- Volle, D.H.; Duggavathi, R.; Magnier, B.C.; Houten, S.M.; Cummins, C.L.; Lobaccaro, J.M.; Verhoeven, G.; Schoonjans, K.; Auwerx, J. The small heterodimer partner is a gonadal gatekeeper of sexual maturation in male mice. Genes Dev. 2007, 21, 303–315. [Google Scholar] [CrossRef]
- Veja, A.; Martinot, E.; Baptissart, M.; De Haze, A.; Saru, J.P.; Baron, S.; Caira, F.; Schoonjans, K.; Lobaccaro, J.M.; Volle, D.H. Identification of the link between the hypothalamo-pituitary axis and the testicular orphan nuclear receptor NR0B2 in adult male mice. Endocrinology 2015, 156, 660–669. [Google Scholar] [CrossRef]
- Chen, R.; Cui, Y.; Zhang, X.; Zhang, Y.; Chen, M.; Zhou, T.; Lan, X.; Dong, W.; Pan, C. Chlorpyrifos Induction of Testicular-Cell Apoptosis through Generation of Reactive Oxygen Species and Phosphorylation of AMPK. J. Agric. Food Chem. 2018, 66, 12455–12470. [Google Scholar] [CrossRef] [PubMed]
- Eze, U.A.; Huntriss, J.D.; Routledge, M.N.; Gong, Y.Y. In vitro effects of single and binary mixtures of regulated mycotoxins and persistent organochloride pesticides on steroid hormone production in MA-10 Leydig cell line. Toxicol. Vitr. 2019, 60, 272–280. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wang, Y.; Fang, Y.; Ni, C.; Ma, L.; Zheng, W.; Bao, S.; Li, X.; Lian, Q.; Ge, R.S. Gestational exposure to ziram disrupts rat fetal Leydig cell development. Chemosphere 2018, 203, 393–401. [Google Scholar] [CrossRef] [PubMed]
- Kong, D.; Zhang, J.; Hou, X.; Zhang, S.; Tan, J.; Chen, Y.; Yang, W.; Zeng, J.; Han, Y.; Liu, X.; et al. Acetamiprid inhibits testosterone synthesis by affecting the mitochondrial function and cytoplasmic adenosine triphosphate production in rat Leydig cells. Biol. Reprod. 2017, 96, 254–265. [Google Scholar] [CrossRef] [PubMed]
- Silvestroni, L.; Rossi, F.; Magnanti, M.; Lubrano, C.; Santiemma, V.; Palleschi, S. A novel aspect of lindane testicular toxicity: In vitro effects on peritubular myoid cells. Reprod. Toxicol. 1999, 3, 431–441. [Google Scholar] [CrossRef]
- Rey, F.; González, M.; Zayas, M.A.; Stoker, C.; Durando, M.; Luque, E.H.; Muñoz-de-Toro, M. Prenatal exposure to pesticides disrupts testicular histoarchitecture and alters testosterone levels in male Caiman latirostris. Gen. Comp. Endocrinol. 2009, 162, 286–292. [Google Scholar] [CrossRef]
- Urióstegui-Acosta, M.; Tello-Mora, P.; Solís-Heredia, M.J.; Ortega-Olvera, J.M.; Piña-Guzmán, B.; Martín-Tapia, D.; González-Mariscal, L.; Quintanilla-Veja, B. Methyl parathion causes genetic damage in sperm and disrupts the permeability of the blood-testis barrier by an oxidant mechanism in mice. Toxicology 2020, 438, 152463. [Google Scholar] [CrossRef]
- Pisani, C.; Voisin, S.; Arafah, K.; Durand, P.; Perrard, M.H.; Guichaoua, M.R.; Bulet, P.; Prat, O. Ex vivo assessment of testicular toxicity induced by carbendazim and iprodione, alone or in a mixture. ALTEX 2016, 33, 393–413. [Google Scholar] [CrossRef]
- Carvalho, R.K.; Rodrigues, T.C.; Júnior, W.D.; Mota, G.M.P.; Andersen, M.L.; Mazaro, E.; Costa, R. Short- and long-term exposure to methamidophos impairs spermatogenesis in mice. Reprod. Biol. 2020, 20, 357–364. [Google Scholar] [CrossRef] [PubMed]
- Rastogi, D.; Narayan, R.; Saxena, D.K.; Chowdhuri, D.K. Endosulfan induced cell death in Sertoli-germ cells of male Wistar rat follows intrinsic mode of cell death. Chemosphere 2014, 94, 104–115. [Google Scholar] [CrossRef]
- Hung, J.H.; Chen, C.Y.; Omar, H.A.; Huang, K.Y.; Tsao, C.C.; Chiu, C.C.; Chen, Y.L.; Chen, P.H.; Teng, Y.N. Reactive oxygen species mediate Terbufos-induced apoptosis in mouse testicular cell lines via the modulation of cell cycle and pro-apoptotic proteins. Environ. Toxicol. 2016, 31, 1888–1898. [Google Scholar] [CrossRef]
- Ham, J.; You, S.; Lim, W.; Song, G. Bifenthrin impairs the functions of Leydig and Sertoli cells in mice via mitochondrion-endoplasmic reticulum dysregulation. Environ. Pollut. 2020, 266, 115174. [Google Scholar] [CrossRef]
- Yan, M.; Shi, Y.; Wang, Y.; Wang, C.; Zhou, J.; Quan, C.; Liu, C.; Yang, K. Effects of p,p′-DDE on the mRNA and protein expressions of vimentin, N-cadherin and FSHR in rats testes: An in vivo and in vitro study. Environ. Toxicol. Pharm. 2013, 35, 486–494. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Wang, H.X.; Shen, J.Y.; Zhang, R.; Hong, J.W.; Li, Z.; Chen, G.; Li, M.X.; Ding, Z.; Li, J.; et al. The anti-androgenic effects of cypermethrin mediated by non-classical testosterone pathway activation of mitogen-activated protein kinase cascade in mouse Sertoli cells. Ecotoxicol. Environ. Saf. 2019, 177, 58–65. [Google Scholar] [CrossRef]
- de Barros, A.L.; Bae, J.H.; Borges, C.S.; Rosa, J.L.; Cavariani, M.M.; Silva, P.V.; Pinheiro, P.F.F.; Anselmo-Franci, J.A.; Arena, A.C. Perinatal exposure to insecticide fipronil: Effects on the reproductive system in male rats. Reprod. Fertil. Dev. 2017, 29, 1130–1143. [Google Scholar] [CrossRef] [PubMed]
- Alves, M.G.; Rato, L.; Carvalho, R.A.; Moreira, P.I.; Socorro, S.; Oliveira, P.F. Hormonal control of Sertoli cell metabolism regulates spermatogenesis. Cell Mol. Life Sci. 2013, 70, 777–793. [Google Scholar] [CrossRef]
- Alves-Lopes, J.P.; Söder, O.; Stukenborg, J.B. Testicular organoid generation by a novel in vitro three-layer gradient system. Biomaterials 2017, 130, 76–89. [Google Scholar] [CrossRef]
- Sebastian, R.; Raghavan, S.C. Exposure to Endosulfan can result in male infertility due to testicular atrophy and reduced sperm count. Cell Death Discov. 2015, 1, 15020. [Google Scholar] [CrossRef]
- Erthal, R.P.; Staurengo-Ferrari, L.; Fattori, V.; Luiz, K.G.; Cunha, F.Q.; Pescim, R.R.; Cecchini, R.; Verri, W.A., Jr.; Guarnier, F.A.; Alves Fernandes, G.S. Exposure to low doses of malathion during juvenile and peripubertal periods impairs testicular and sperm parameters in rats: Role of oxidative stress and testosterone. Reprod. Toxicol. 2020, 96, 17–26. [Google Scholar] [CrossRef]
- Elsharkawy, E.E.; Yahia, D.; El-Nisr, N.A. Chlorpyrifos induced testicular damage in rats: Ameliorative effect of glutathione antioxidant. Environ. Toxicol. 2014, 29, 1011–1019. [Google Scholar] [CrossRef] [PubMed]
- Saleh, H.; Nassar, A.M.K.; Noreldin, A.E.; Samak, D.; Elshony, N.; Wasef, L.; Elewa, Y.H.A.; Hassan, S.M.A.; Saati, A.A.; Hetta, H.F.; et al. Chemo-Protective Potential of Cerium Oxide Nanoparticles against Fipronil-Induced Oxidative Stress, Apoptosis, Inflammation and Reproductive Dysfunction in Male White Albino Rats. Molecules 2020, 25, 3479. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, A.A.; Abdellatief, S.A.; Khater, S.I.; Ali, H.; Al-Gabri, N.A. Fenpropathrin induces testicular damage, apoptosis, and genomic DNA damage in adult rats: Protective role of camel milk. Ecotoxicol. Environ. Saf. 2019, 181, 548–558. [Google Scholar] [CrossRef]
- Mahajan, L.; Verma, P.K.; Raina, R.; Sood, S. Potentiating effect of imidacloprid on arsenic-induced testicular toxicity in Wistar rats. BMC Pharm. Toxicol. 2018, 19, 48. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi-Sardoo, M.; Mandegary, A.; Nabiuni, M.; Nematollahi-Mahani, S.N.; Amirheidari, B. Mancozeb induces testicular dysfunction through oxidative stress and apoptosis: Protective role of N-acetylcysteine antioxidant. Toxicol. Ind. Health 2018, 34, 798–811. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Sun, J.; Fan, Y.; Su, J.; Xie, J.; Wu, Y.; Liu, X.; Wang, C. Exposure to Pb and Cd alters MCT4/CD147 expression and MCT4/CD147-dependent lactate transport in mice Sertoli cells cultured in vitro. Toxicol. Vitr. 2019, 56, 30–40. [Google Scholar] [CrossRef]
- Crisóstomo, L.; Alves, M.G.; Calamita, G.; Sousa, M.; Oliveira, P.F. Glycerol and testicular activity: The good, the bad and the ugly. Mol. Hum. Reprod. 2017, 23, 725–737. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, A.M.; Alves, M.G.; Sousa, A.C.; Jarak, I.; Carvalho, R.A.; Oliveira, P.F.; Cavaco, J.E.; Rato, L. The effects of the obesogen tributyltin on the metabolism of Sertoli cells cultured ex vivo. Arch. Toxicol. 2018, 92, 601–610. [Google Scholar] [CrossRef] [PubMed]
- Sengupta, P.; Banerjee, R. Environmental toxins: Alarming impacts of pesticides on male fertility. Hum. Exp. Toxicol. 2013, 33, 1017–1039. [Google Scholar] [CrossRef] [PubMed]
- Alves, M.G.; Neuhaus-Oliveira, A.; Moreira, P.I.; Socorro, S.; Oliveira, P.F. Exposure to 2,4-dichlorophenoxyacetic acid alters glucose metabolism in immature rat Sertoli cells. Reprod. Toxicol. 2013, 38, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Harada, Y.; Tanaka, N.; Ichikawa, M.; Kamijo, Y.; Sugiyama, E.; Gonzalez, F.J.; Aoyama, T. PPARα-dependent cholesterol/testosterone disruption in Leydig cells mediates 2,4-dichlorophenoxyacetic acid-induced testicular toxicity in mice. Arch. Toxicol. 2016, 90, 3061–3071. [Google Scholar] [CrossRef]
- Michalik, L.; Auwerx, J.; Berger, J.P.; Chatterjee, V.K.; Glass, C.K.; Gonzalez, F.J.; Grimaldi, P.A.; Kadowaki, T.; Lazar, M.A.; O’Rahilly, S.; et al. International Union of Pharmacology. LXI. Peroxisome proliferator-activated receptors. Pharm. Rev. 2006, 58, 726–741. [Google Scholar] [CrossRef]
- Krey, G.; Braissant, O.; L’Horset, F.; Kalkhoven, E.; Perroud, M.; Parker, M.G.; Wahli, W. Fatty acids, eicosanoids, and hypolipidemic agents identified as ligands of peroxisome proliferator-activated receptors by coactivator-dependent receptor ligand assay. Mol. Endocrinol. 1997, 11, 779–791. [Google Scholar] [CrossRef]
- Regueira, M.; Riera, M.F.; Galardo, M.N.; Pellizzari, E.H.; Cigorraga, S.B.; Meroni, S.B. Activation of PPAR α and PPAR β/δ regulates Sertoli cell metabolism. Mol. Cell Endocrinol. 2014, 382, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Nittoli, V.; Colella, M.; Porciello, A.; Reale, C.; Roberto, L.; Russo, F.; Russo, N.A.; Porreca, I.; De Felice, M.; Mallardo, M.; et al. Multi Species Analyses Reveal Testicular T3 Metabolism and Signalling as a Target of Environmental Pesticides. Cells 2021, 10, 2187. [Google Scholar] [CrossRef] [PubMed]
- Clauson, S.L.; Sylvia, J.M.; Arcury, T.A.; Summers, P.; Spencer, K.M. Detection of Pesticides and Metabolites Using Surface-Enhanced Raman Spectroscopy (SERS): Acephate. Appl. Spectrosc. 2015, 69, 785–793. [Google Scholar] [CrossRef] [PubMed]
- Dudek, C.A.; Reuse, C.; Fuchs, R.; Hendriks, J.; Starck, V.; Hiller, K. MIAMI--a tool for non-targeted detection of metabolic flux changes for mode of action identification. Bioinformatics 2020, 36, 3925–3926. [Google Scholar] [CrossRef] [PubMed]
- Hiller, K.; Wegner, A.; Weindl, D.; Cordes, T.; Metallo, C.M.; Kelleher, J.K.; Stephanopoulos, G. NTFD—A stand-alone application for the non-targeted detection of stable isotope-labeled compounds in GC/MS data. Bioinformatics 2013, 29, 1226–1228. [Google Scholar] [CrossRef] [PubMed][Green Version]

| Classes of Pesticides | Properties | Mechanisms of Action | Examples |
|---|---|---|---|
| Organochlorine pesticides (OCPs) [25] | Chlorinated hydrocarbon compounds | Alter ion exchange dynamics in axons, in the peripheral and central nervous systems, leading to decreased action potentials | Dichlorodiphenyltrichloroethane (DDT), Hexachlorobenzene (HCB) |
| Organophosphates (OPs) [26] | Esters of phosphoric acid | Induce irreversible inhibition of acetylcholinesterase enzyme (AChE), causing accumulation of acetylcholine (ACh) in muscarinic and nicotinic cholinergic synapses, consequently, overstimulating ACh receptors in the nervous system and neuromuscular junctions | Malathion, Parathion, Diazinon, Dichlorvos, Chlorpyrifos, Tribufos (DEF) |
| Carbamates [27] | Esters of N-methyl carbamic acid | Reversibly inhibit the AChE, which catalyzes the hydrolysis of ACh, leading to its increase in nerve synapses and neuromuscular junctions, triggering increased stimulation of these nerve endings | Carbendazim, Carbaryl, Aminocarb, Thiodicarb, Carbofuran, Mancozeb |
| Pyrethroids [25] | Synthetic pesticides based on the chemistry of natural pyrethrins | Prevent the closure of voltage-gated sodium channels in axonal membranes, blocking normal nerve impulses, thereby paralyzing and, eventually, killing the organism | Cypermethrin, Bifenthrin, Fenvalerate, Permethrin |
| Phenylpyrazoles [28] | Chemical structure characterized by a central pyrazole ring with a phenyl group attached to one of the nitrogen atoms of the pyrazole | Block non-competitive gamma-aminobutyric acid (GABA)-gated chloride channels, creating excessive neuronal stimulation and death | Fipronil, Pyriprole |
| Neonicotinoids [28] | Structurally similar to the natural insecticide nicotine | Enhanced selectivity and potency to bind to nicotinic ACh receptors (nAChRs), leading to a large influx of cations into the postsynaptic membrane of nerve cells in the central nervous system, triggering excessive excitatory neurotransmission, which results in paralysis and death | Imidacloprid, Acetamiprid, Chlothianidin |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moreira, S.; Pereira, S.C.; Seco-Rovira, V.; Oliveira, P.F.; Alves, M.G.; Pereira, M.d.L. Pesticides and Male Fertility: A Dangerous Crosstalk. Metabolites 2021, 11, 799. https://doi.org/10.3390/metabo11120799
Moreira S, Pereira SC, Seco-Rovira V, Oliveira PF, Alves MG, Pereira MdL. Pesticides and Male Fertility: A Dangerous Crosstalk. Metabolites. 2021; 11(12):799. https://doi.org/10.3390/metabo11120799
Chicago/Turabian StyleMoreira, Sílvia, Sara C. Pereira, Vicente Seco-Rovira, Pedro F. Oliveira, Marco G. Alves, and Maria de Lourdes Pereira. 2021. "Pesticides and Male Fertility: A Dangerous Crosstalk" Metabolites 11, no. 12: 799. https://doi.org/10.3390/metabo11120799
APA StyleMoreira, S., Pereira, S. C., Seco-Rovira, V., Oliveira, P. F., Alves, M. G., & Pereira, M. d. L. (2021). Pesticides and Male Fertility: A Dangerous Crosstalk. Metabolites, 11(12), 799. https://doi.org/10.3390/metabo11120799









